Published online Aug 28, 2011. doi: 10.4329/wjr.v3.i8.205
Revised: July 12, 2011
Accepted: July 19, 2011
Published online: August 28, 2011
AIM: To assess the feasibility of performing acoustic radiation force impulse (ARFI) elastography in patients with ascites and its predictive value for the cirrhotic or non-cirrhotic etiology of ascites. METHODS: Our study included 153 patients with ascites, mean age 58.8 ± 13.1 years. One hundred and fifteen (75.2%) patients had ascites in the context of cirrhosis, 29 (18.9%) had non-cirrhotic ascites (diagnosed by clinical, ultrasound, endoscopic and/or laparoscopic criteria) and in 9 (5.9%) cases we could not establish the etiology of ascites. We performed 10 ARFI measurements and the median value was calculated and expressed in meters/second (m/s). Among the 29 patients with non-cirrhotic ascites were included: 20 laparoscopically demonstrated peritoneal carcinomatosis with histological confirmation, 7 acute pancreatitis with ascites which later resolved, and one case each of lymphatic ascites and ascites in the context of a liver abscess. In 11 of the 20 patients with peritoneal carcinomatosis, the liver structure was homogenous in the ultrasound examination and in 9 patients the ultrasound exam revealed liver metastases. RESULTS: We could not obtain valid ARFI measurements in 5 patients (3.2%). The mean liver stiffness measurements by means of ARFI were statistically significantly higher in patients with cirrhotic ascites than in those with non-cirrhotic ascites: 3.04 ± 0.70 vs 1.45 ± 0.59 m/s (P < 0.001). For a cut-off value of 1.8 m/s for predicting cirrhosis (and ascites in the context of cirrhosis), as obtained in a previous study, ARFI had 98.1% sensitivity, 86.2% specificity, 96.4% positive predictive value, 92.5% negative predictive value and 95.6% accuracy for predicting cirrhotic ascites. For a cut-off value of 1.9 m/s the accuracy was 94.9% and for a 2 m/s cut-off value it was 92.8%. CONCLUSION: ARFI elastography is feasible in most patients with ascites and has a very good predictive value for the cirrhotic or non-cirrhotic etiology of ascites.
- Citation: Bota S, Sporea I, Şirli R, Popescu A, Dănilă M, Şendroiu M. Value of acoustic radiation force impulse elastography for the assessment of ascites syndrome. World J Radiol 2011; 3(8): 205-209
- URL: https://www.wjgnet.com/1949-8470/full/v3/i8/205.htm
- DOI: https://dx.doi.org/10.4329/wjr.v3.i8.205
Ascites is a fluid accumulation in the peritoneal cavity. Mild ascites is hard to notice by clinical exam, but severe ascites leads to abdominal distension. Patients with ascites generally will complain of progressive abdominal heaviness and pressure, as well as shortness of breath due to mechanical impingement on the diaphragm. The International Club of Ascites classified ascites as follows: grade 1 (mild, visible only on ultrasound); grade 2 (detectable with flank bulging and shifting dullness); and grade 3 (directly visible, confirmed with fluid thrill)[1].
The peritoneal fluid can be transudate or exudate. The serum-ascites albumin gradient (SAAG) is probably the best method to differentiate transudate and exudate ascites[2]. A high SAAG (> 1.1 g/dL) is suggestive of a portal hypertensive cause of ascites. A low gradient (< 1.1 g/dL) indicates that the cause of ascites was not portal hypertension.
The causes of ascites with high SAAG (transudate) are: liver cirrhosis, heart failure, hepatic venous occlusion (Budd-Chiari syndrome or veno-occlusive disease), constrictive pericarditis, Kwashiorkor syndrome[3]. Causes of ascites with low SAAG (exudate) are: cancer (primary peritoneal carcinomatosis and metastasis), tuberculosis, pancreatitis, serositis, nephrotic syndrome or protein losing enteropathy[3]. Other causes of ascites are: Meigs syndrome, vasculitis, hypothyroidism, peritoneum mesothelioma[3].
Acoustic radiation force impulse (ARFI) elastography is a new method used for the assessment of liver fibrosis, thyroid gland nodules, breast nodules, liver and kidney tumors[4-10]. The principle of ultrasound elastography is that compression of the examined tissue induces a smaller strain in hard tissues than in soft ones. The ultrasound probe automatically produces an acoustic “push” pulse that generates shear-waves which propagate into the tissue. Their speed, measured in meters/second (m/s), is displayed on the screen. The propagation speed increases with fibrosis severity. Using image-based localization and a proprietary implementation of ARFI technology, shear wave speed may be quantified in a precise anatomical region, focused on a region of interest, with a predefined size provided by the system. Measurement value and depth are also reported and the results of the elasticity are in m/s[11-13].
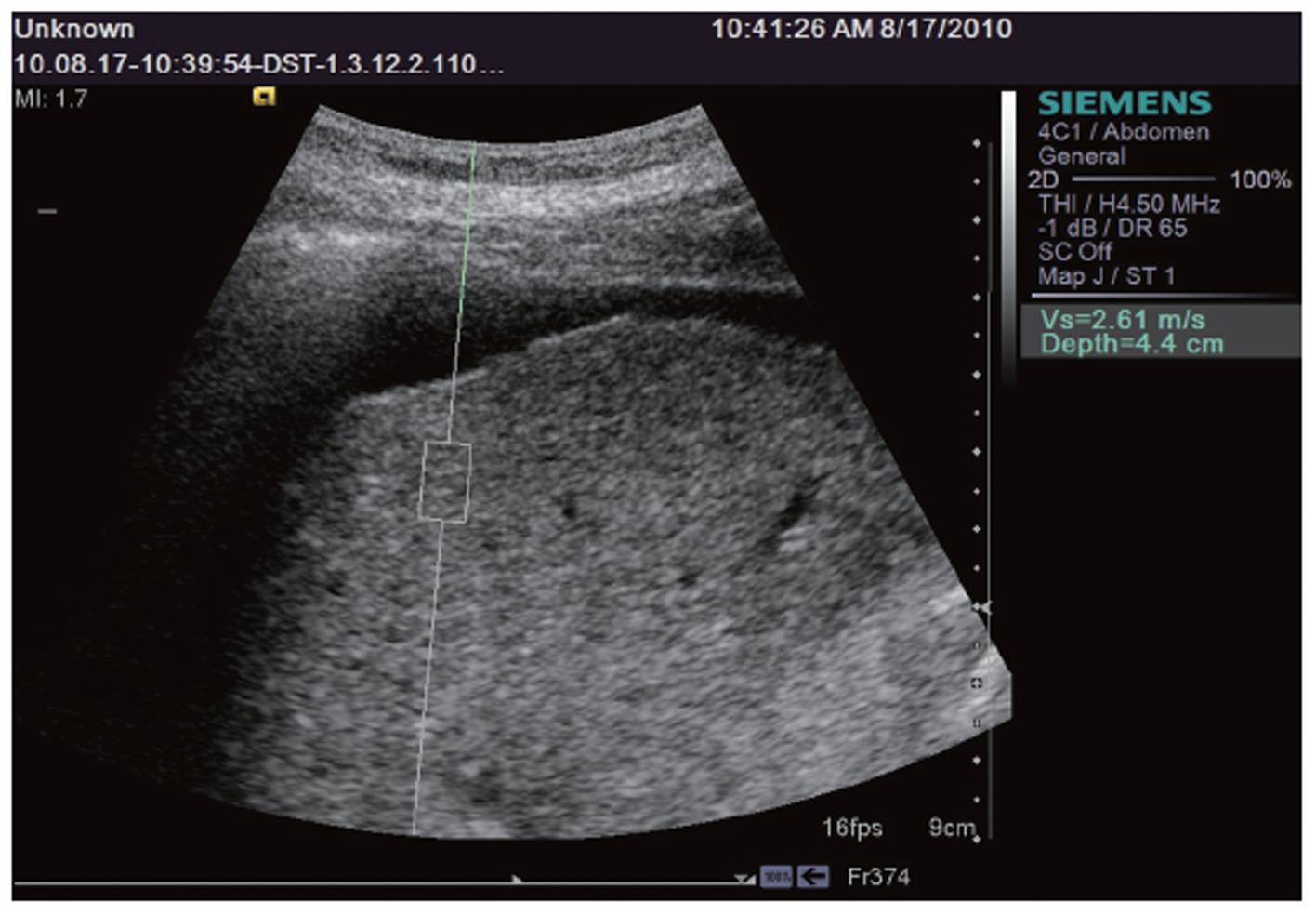
We included in our study 153 consecutive patients, mean age 58.8 ± 13.1 years, 61 women and 92 men. In each patient we measured the liver stiffness (LS) by means of ARFI in the right liver lobe, 1 cm below the liver capsule, by intercostal approach, with the patient laying in left lateral decubitus (Figure 1). ARFI measurements were performed with a Siemens Acuson S2000™ ultrasound system. In each patient, 10 valid ARFI measurements were performed and the median values were calculated, the results being expressed in meters/second (m/s). The investigator performed ARFI measurements blinded to the patient’s clinical and biochemical data.
Our study included 115 patients (75.2%) with ascites caused by liver cirrhosis (diagnosed by clinical, ultrasound, endoscopic and/or laparoscopic criteria), 29 patients (18.9%) with non-cirrhotic ascites (20 laparoscopically demonstrated peritoneal carcinomatosis with histological confirmation, 7 acute pancreatitis with ascites which later resolved and one case each of lymphatic ascites and ascites in the context of a liver abscess) and 9 patients (5.9%) in whom we could not establish the etiology of ascites (the patients refused the laparoscopy). All the patients signed the informed consent and the study was approved ethically by the Emergency County Hospital Timisoara.
Data obtained from our patients were collected in a Microsoft Excel file, the statistical analysis being performed using the MedCalc program and WINK Statistical Data Analysis Research Software. ARFI measurements were numeric variables, so the mean values and standard deviation were calculated. The 2-way ANOVA test and t test were used to compare mean ARFI values of LS.
The characteristics of the patients are presented in Table 1. In 11 of the 20 patients with peritoneal carcinomatosis, the liver structure was homogenous in the ultrasound examination and in 9 patients the ultrasound exam revealed liver metastases.
| Parameter | All patients | Cirrhotic patients | Non-cirrhotic patients |
| Mean age (yr) | 58.8 ± 13.1 | 58.6 ± 12.1 | 57.4 ± 15.3 |
| Sex, n (%) | |||
| Female | 61 (39.8) | 50 (43.5) | 7 (24.1) |
| Male | 92 (60.2) | 65 (56.5) | 22 (75.9) |
| Classification of ascites, n (%) | |||
| Grade 1 | 70 (45.7) | 53 (46.1) | 14 (48.3) |
| Grade 2 | 50 (32.7) | 36 (31.3) | 10 (34.4) |
| Grade 3 | 33 (21.5) | 26 (22.6) | 5 (17.3) |
| Mean ARFI values (range, m/s) | 1.60 ± 0.66 (0.83-4.87) | 3.04 ± 0.70 (1.62-4.87) | 1.45 ± 0.59 (0.83-3.94) |
| Mean AST (U/L) (normal value = 5-34 U/L) | 83.5 ± 72.8 | 95.1 ± 76.2 | 47.2 ± 43.4 |
| Mean ALT (U/L) (normal value = 10-35 U/L) | 47.7 ± 37.1 | 49.3 ± 33.5 | 47.1 ± 50 |
| Mean total bilirubin (mg/dL) (normal value = 0.2-1 mg/dL) | 3.25 ± 3.78 | 3.29 ± 2.80 | 3.41 ± 6.55 |
| Mean total protein (g/dL) (normal value = 5.6-8 g/dL) | 6.2 ± 1.2 | 6.5 ± 1.1 | 6 ± 1.2 |
| Mean albumin (g/dL) (normal value = 3.5-5 g/dL) | 3.02 ± 0.67 | 2.95 ± 0.66 | 3.38 ± 0.68 |
| Mean prothrombin index time (%) (normal value > 75%) | 70.1 ± 14.4 | 65.7 ± 16 | 86.3 ± 15.9 |
| Mean cholinesterase value (U/L) (normal value = 4500-8000 U/L) | 3252.9 ± 1743.1 | 2810.2 ± 1414.3 | 5220.3 ± 1977.2 |
Of the 115 cirrhotic patients, 85 (73.9%) had esophageal varices, 23 (20%) had a history of variceal bleeding, 21 (18.2%) had hepatocellular carcinoma.
In 5 of the patients, we could not obtain valid measurements (3.2%) and in the other cases ARFI measurements ranged between 0.83 and 4.87 m/s. Our statistical analysis was made on 139 patients with valid ARFI measurements and proven cause of ascites.
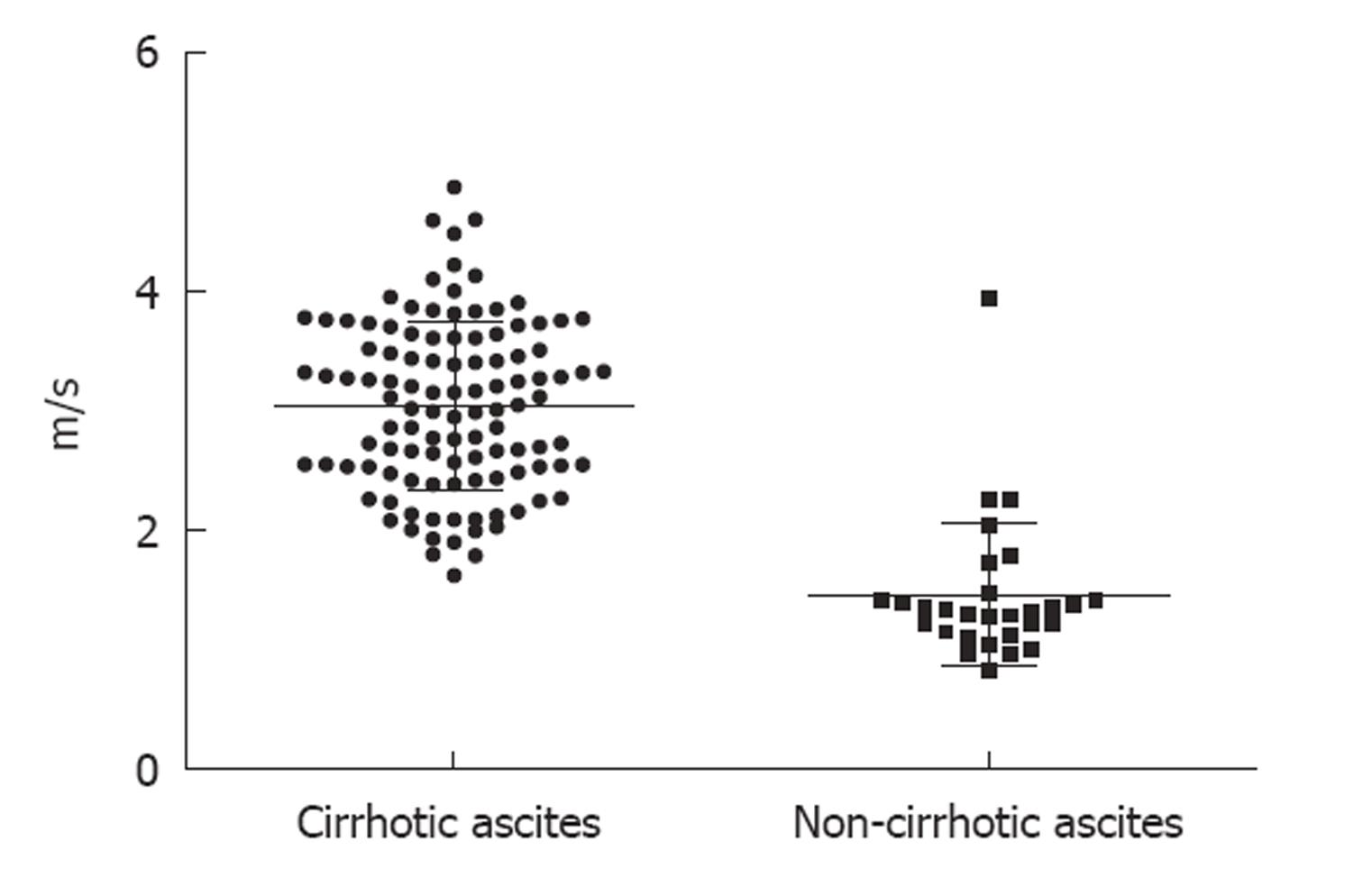
The mean LS measurements by means of ARFI were statistically significantly higher in patients with cirrhotic ascites than in those with non-cirrhotic ascites: 3.04 ± 0.70 m/s vs 1.45 ± 0.59 m/s (P < 0.001) (Figure 2).
For a cut-off value of 1.8 m/s for predicting cirrhosis (and ascites in the context of liver cirrhosis), which is a figure resulting from our previous studies[8,9], ARFI had very good predictive value for the cirrhotic etiology of the ascites (Table 2). If the cut-off value was set at 1.9 and 2 m/s, the predictive value was also very good (Table 2).
| Cut-off | Se | Sp | PPV | NPV | Accuracy |
| 1.8 m/s | 98.1 | 86.2 | 96.4 | 92.5 | 95.6 |
| 1.9 m/s | 97.2 | 86.2 | 96.2 | 89.2 | 94.9 |
| 2 m/s | 94.5 | 86.2 | 96.2 | 80.6 | 92.8 |
Standard ultrasonography can be a useful tool for the etiological diagnosis of ascites syndrome. For instance, the surface of the liver is irregular in liver cirrhosis and regular in most other causes of ascites. In cirrhosis we can also find an enlarged spleen, a large portal vein, a thickened wall of the gallbladder with bowel loops freely floating within the ascitic fluid. If the peritoneal fluid is an exudate we can find small amorphous echoes within the fluid (“debris”), loculated fluid, matted bowel loops, hepatic metastases[14,15]. However, the differentiation between benign and malignant ascites by means of ultrasound is frequently difficult or impossible.
ARFI elastography is a new method used for the assessment of LS. Unlike transient elastography (TE) (FibroScan, EchoSens-Paris, France), this new technique can additionally be performed in patients with ascites.
In our present study, we found statistically significant differences between LS measurements by means of ARFI in patients with cirrhotic ascites vs those with non-cirrhotic ascites (3.04 ± 0.70 m/s vs 1.45 ± 0.59 m/s) (P < 0.001). Although the patient groups were unbalanced, our results were confident enough to discriminate between the cirrhotic or non-cirrhotic etiology of ascites. For an LS cut-off value of 1.8 m/s, ARFI elastography had 95.6% accuracy for predicting the cirrhotic etiology of ascites (2 patients with cirrhotic ascites and 4 patients with non-cirrhotic ascites were incorrectly classified), for a cut-off value of 1.9 m/s, the accuracy was 94.9% (3 patients with cirrhotic ascites and 4 patients with non-cirrhotic ascites were incorrectly classified), and for a cut-off value of 2 m/s, it was 92.8% (6 patients with cirrhotic ascites and 4 patients with non-cirrhotic ascites were incorrectly classified). Thus, from our data it follows that the best ARFI cut-off for prediction of cirrhosis (and ascites in the context of the liver cirrhosis) is 1.8 m/s.
Of the 4 patients with non-cirrhotic ascites incorrectly classified for a 1.8 m/s cutoff value, in 2 cases ARFI was difficult to determine (10 valid ARFI measurements and more than 7 invalid measurements) and in one case after we performed contrast enhanced ultrasonography we discovered multiple metastases (so ARFI was performed in the liver masses).
Regarding the 2 patients with cirrhotic ascites incorrectly classified for a 1.8 m/s cut-off value, in one case ARFI was difficult to determine (10 valid ARFI measurements and more than 7 invalid measurements).
Several studies have demonstrated the value of ARFI elastography for the prediction of liver cirrhosis and for the study of the liver in healthy subjects.
In a previous study performed by our group[9], in which we included 114 subjects (38 healthy volunteers, 76 patients with chronic hepatopathies in whom liver biopsy (LB) was performed, and 23 with clinical, ultrasonographic and/or endoscopic signs of cirrhosis) investigated by means of ARFI, a direct, strong, linear correlation (Spearman rho = 0.675) was found between ARFI measurements and fibrosis (P < 0.0001). The predictive value of ARFI for the presence of cirrhosis was also excellent; for a cut-off value of 1.8 m/s (for measurements made 1-2 cm under the capsule) the area under the receiver operating characteristic curve (AUROC) was 0.92.
In another study published in 2010 by our group[8], which included 71 patients with chronic viral hepatopathies, all with LB, the cut-off value of ARFI measurement for the diagnosis of cirrhosis was 1.8 m/s [AUROC 0.868, with 100% sensitivity (Se), 77% specificity (Sp), 58% positive predictive value (PPV) and 100% negative predictive value (NPV)].
In a study performed by Lupşor et al[7], 112 patients with chronic HCV hepatitis were evaluated by means of LB (the stage of fibrosis was assessed according to the Metavir scoring system), ARFI and TE (FibroScan). For a cut-off value of > 2 m/s for the prediction of cirrhosis, ARFI had 80% Se, 95.4% Sp, 90.3% PPV and 90% NPV (AUROC = 0.93). Also, in the study of Fierbinteanu-Braticevici et al[16] which evaluated 79 patients with chronic HCV hepatitis who underwent LB and ARFI, this new elastographic method had very good sensitivity and specificity for prediction of cirrhosis (for a cut-off value > 1.94 m/s ARFI had 100% Se, 98% Sp, 95% PPV and 100% NPV).
A study published by Goertz et al[10] included 57 patients with chronic viral hepatitis B and C who underwent ARFI and consecutive LB. The results were compared to the histological stage of fibrosis (F). ARFI values were significantly correlated with the histological stage of fibrosis (P < 0.001). The AUROC curve for the accuracy of ARFI imaging was 87% for prediction of cirrhosis (F = 4).
A study published by Haque et al[17] included 22 patients with chronic hepatopathies, in whom LB and ARFI were performed in the same session. ARFI values were compared with both Batts-Ludwing score (F0 to F4) and modified Ishak score (F0 to F4). For the prediction of liver cirrhosis, the AUROC was 0.85 for both Ishak and Batts-Ludwing scores.
In a study published by Piscaglia et al[18], these authors included 133 patients with chronic liver disease evaluated by means of ARFI elastography and TE. In 70 patients they performed LB. The diagnostic accuracy for cirrhosis was first assessed in the 90 patients submitted to TE with > 13 kPa (47% of patients) as diagnostic for cirrhosis values. The best cut-off for cirrhosis with ARFI was then tested in the 70 patients with biopsy (cirrhosis in 38% of patients). The AUROC of ARFI for the diagnosis of cirrhosis (reference TE) was 0.941 with 1.75 m/s as the best cut-off (Se 93.0%; Sp 85.1%). ARFI demonstrated good performance also in patients with bioptic diagnosis of cirrhosis (AUROC 0.908, Se 81.5%, Sp 88.4%).
In all the studies presented above, the cirrhotic patients did not have ascites.
Regarding the evaluation of LS in healthy subjects, Gallotti et al[19] measured the stiffness of various abdominal organs by ARFI elastography in 35 healthy subjects, obtaining for the liver a mean value of 1.59 m/s. Goertz et al[10] obtained a mean LS value of 1.09 m/s in a group of subjects without liver pathology. Popescu et al[20] evaluated by means of ARFI elastography 82 subjects without known liver pathology. The mean value of ARFI measurements in normal individuals was 1.15 ± 0.21 m/s.
The results of our study support the conclusion that in the investigation of an ascites syndrome, the first investigation that should be performed after abdominal ultrasound should be ARFI elastography. Regarding the limitations of this study, ARFI is not a very good method for the evaluation of LS in cases of multiple metastases, in cases when performing ARFI in liver masses (when the liver structure is homogeneous in standard ultrasonography) or in cases with a success rate < 60% (success rate = the ratio of the number of successful acquisitions over the total number of acquisitions).
ARFI elastography is feasible in most patients with ascites (96.8%) and it has a very good predictive value for the cirrhotic or non-cirrhotic etiology of ascites.
Ascites syndrome is a quite common discovery in abdominal ultrasonography and several methods are used to establish the cause of ascites.
Acoustic radiation force impulse (ARFI) elastography is a new method, based on ultrasound, used for the evaluation of liver stiffness. The advantage of this elastographic method compared to transient elastography, is that it can be used in the presence of ascites. In our study, ARFI was determinable in 96.8% of the patients with ascites. ARFI had a very good accuracy (95.6% for a 1.8 m/s cut-off value) for the prediction of cirrhotic or non-cirrhotic etiology of the ascites.
This study supports the conclusion that in the investigation of an ascites syndrome, the first investigation that should be performed after abdominal ultrasound should be ARFI elastography.
This is an interesting cross-sectional study evaluating liver stiffness by ARFI in subjects with ascites. The authors noticed higher values in subjects with cirrhosis, which is biological plausible. Additionally authors presented some cut-off points which could be used for the diagnosis of cirrhosis in subjects with ascites. This study is clearly presented and is well written; however, some revisions are needed to improve the manuscript.
Peer reviewers: Yasunori Minami, MD, PhD, Assistant Professor, Division of Gastroenterology and Hepatology, Department of Internal Medicine, Kinki University School of Medicine, 377-2 Ohno-higashi, Osaka-sayama, Osaka, 589-8511, Japan; Wellington P Martins, PhD, Departamento de Ginecologia e, Obstetrícia da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo, Avenida dos Bandeirantes 3900, 8º andar, Ribeirão Preto, São Paulo 14049-900, Brazil; Mirko D’Onofrio, MD, Assistant Professor, Department of Radiology, GB Rossi University Hospital, University of Verona, Piazzale LA Scuro 10, Verona, 37134, Italy
S- Editor Cheng JX L- Editor Logan S E- Editor Zheng XM
| 1. | Ashby MA, Fleming BG, Brooksbank M, Rounsefell B, Runciman WB, Jackson K, Muirden N, Smith M. Description of a mechanistic approach to pain management in advanced cancer. Preliminary report. Pain. 1992;51:153-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 611] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 2. | Runyon BA, Montano AA, Akriviadis EA, Antillon MR, Irving MA, McHutchison JG. The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med. 1992;117:215-220. [PubMed] |
| 3. | Warrell DA. Oxford textbook of medicine. 3rd ed. Oxford: Oxford University Press 2003; . |
| 4. | Friedrich-Rust M, Ong MF, Herrmann E, Dries V, Samaras P, Zeuzem S, Sarrazin C. Real-time elastography for noninvasive assessment of liver fibrosis in chronic viral hepatitis. AJR Am J Roentgenol. 2007;188:758-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 238] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 5. | Havre RF, Elde E, Gilja OH, Odegaard S, Eide GE, Matre K, Nesje LB. Freehand real-time elastography: impact of scanning parameters on image quality and in vitro intra- and interobserver validations. Ultrasound Med Biol. 2008;34:1638-1650. [PubMed] [DOI] [Full Text] |
| 6. | Friedrich-Rust M, Wunder K, Kriener S, Sotoudeh F, Richter S, Bojunga J, Herrmann E, Poynard T, Dietrich CF, Vermehren J. Liver fibrosis in viral hepatitis: noninvasive assessment with acoustic radiation force impulse imaging versus transient elastography. Radiology. 2009;252:595-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Lupsor M, Badea R, Stefanescu H, Sparchez Z, Branda H, Serban A, Maniu A. Performance of a new elastographic method (ARFI technology) compared to unidimensional transient elastography in the noninvasive assessment of chronic hepatitis C. Preliminary results. J Gastrointestin Liver Dis. 2009;18:303-310. [PubMed] |
| 8. | Sporea I, Sirli R, Popescu A, Danilă M. Acoustic Radiation Force Impulse (ARFI)--a new modality for the evaluation of liver fibrosis. Med Ultrason. 2010;12:26-31. [PubMed] |
| 9. | Sporea I, Sirli RL, Deleanu A, Popescu A, Focsa M, Danila M, Tudora A. Acoustic radiation force impulse elastography as compared to transient elastography and liver biopsy in patients with chronic hepatopathies. Ultraschall Med. 2011;32 Suppl 1:S46-S52. [PubMed] |
| 10. | Goertz RS, Zopf Y, Jugl V, Heide R, Janson C, Strobel D, Bernatik T, Haendl T. Measurement of liver elasticity with acoustic radiation force impulse (ARFI) technology: an alternative noninvasive method for staging liver fibrosis in viral hepatitis. Ultraschall Med. 2010;31:151-155. [PubMed] |
| 11. | Zhai L, Palmeri ML, Bouchard RR, Nightingale RW, Nightingale KR. An integrated indenter-ARFI imaging system for tissue stiffness quantification. Ultrason Imaging. 2008;30:95-111. [PubMed] |
| 12. | Nightingale K, Soo MS, Nightingale R, Trahey G. Acoustic radiation force impulse imaging: in vivo demonstration of clinical feasibility. Ultrasound Med Biol. 2002;28:227-235. [PubMed] |
| 13. | Mauldin FW, Zhu HT, Behler RH, Nichols TC, Gallippi CM. Robust principal component analysis and clustering methods for automated classification of tissue response to ARFI excitation. Ultrasound Med Biol. 2008;34:309-325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Edell SL, Gefter WB. Ultrasonic differentiation of types of ascitic fluid. AJR Am J Roentgenol. 1979;133:111-114. [PubMed] |
| 15. | Yeh HC, Wolf BS. Ultrasonography in ascites. Radiology. 1977;124:783-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 16. | Fierbinteanu-Braticevici C, Andronescu D, Usvat R, Cretoiu D, Baicus C, Marinoschi G. Acoustic radiation force imaging sonoelastography for noninvasive staging of liver fibrosis. World J Gastroenterol. 2009;15:5525-5532. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 133] [Cited by in RCA: 143] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 17. | Haque M, Robinson C, Owen D, Yoshida EM, Harris A. Comparison of acoustic radiation force impulse imaging (ARFI) to liver biopsy histologic scores in the evaluation of chronic liver disease: A pilot study. Ann Hepatol. 2010;9:289-293. [PubMed] |
| 18. | Piscaglia F, Salvatore V, Di Donato R, D'Onofrio M, Gualandi S, Gallotti A, Peri E, Borghi A, Conti F, Fattovich G. Accuracy of VirtualTouch Acoustic Radiation Force Impulse (ARFI) imaging for the diagnosis of cirrhosis during liver ultrasonography. Ultraschall Med. 2011;32:167-175. [PubMed] |
| 19. | Gallotti A, D'Onofrio M, Pozzi Mucelli R. Acoustic Radiation Force Impulse (ARFI) technique in ultrasound with Virtual Touch tissue quantification of the upper abdomen. Radiol Med. 2010;115:889-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 131] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 20. | Popescu A, Sporea I, Sirli R, Bota S, Focşa M, Dănilă M, Nicoliţă D, Martie A, Sendroiu M, Juchiş A. The mean values of liver stiffness assessed by Acoustic Radiation Force Impulse elastography in normal subjects. Med Ultrason. 2011;13:33-37. [PubMed] |