Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.110407
Revised: June 23, 2025
Accepted: July 23, 2025
Published online: August 28, 2025
Processing time: 83 Days and 14.8 Hours
Primary ciliary dyskinesia (PCD) is a rare condition characterised by dysmotile, immotile, or absent cilia. As a result of the impairment in respiratory mucociliary clearance, patients with PCD typically develop neonatal respiratory distress, nasal congestion, otitis media and recurrent respiratory infections leading to bron
To evaluate the efficacy of ultra-low dose CT (ULDCT) chest in identifying pul
Paediatric patients with PCD who presented for routine clinical outpatient follow-up within the study period, were eligible for inclusion in the study. ULDCT and CR were performed on these patients and the results compared. Comparison metrics included radiation dose, subjective and objective image quality and disease severity.
Six paediatric patients (mean age 9 years) underwent clinically indicated ULDCT chest examinations and CR for surveillance of their PCD. The mean effective dose was 0.08 ± 0.02 mSv, a dose that approximates that of a frontal and lateral chest radiograph. The average Brody II score across the entire cohort was 12.92, with excellent inter-rater reliability and intra-class correlation coefficient (ICC) of 0.98. The average Chrispin-Norman score on CR was 1 with excellent inter-rater reliability and ICC of 0.92.
ULDCT demonstrates superior diagnostic capabilities, minimal radiation dose penalty, and high interobserver reliability in comparison to CR. Thus, we advocate for ULDCT to be the preferred modality for surveillance imaging in paediatric PCD.
Core Tip: Ultra-low dose computed tomography (ULDCT) is an exciting technique developed over the last decade that allows acquisition of diagnostic quality images at remarkably low radiation doses. This technique is especially important in patients requiring regular repeated imaging such as children with primary ciliary dyskinesia (PCD). Patients with PCD frequently suffer from recurrent respiratory tract infections and develop structural lung changes that require regular imaging to monitor. While chest radiography is traditionally used, subtle findings may be missed due to limitations of this modality. Our study offers an alternative; ULDCT which is superior in diagnostic capability with a minimal radiation dose penalty.
- Citation: Waldron MG, O'Regan PW, Lane M, Shet SS, Kakish E, Moloney F, Moore N, Murphy MJ, Beagan L, Plant BJ, Mullane D, Ni Chroinin M, Ryan DJ, O'Regan K, Power SP, Maher MM. Ultra-low dose computed tomography chest vs chest radiography in paediatric primary ciliary dyskinesia: A prospective study. World J Radiol 2025; 17(8): 110407
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/110407.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.110407
Primary ciliary dyskinesia (PCD) is a rare, primarily autosomal recessive, inherited condition characterised by dysmotile, immotile, or absent cilia[1]. As a result of the impairment in respiratory mucociliary clearance, patients with PCD typically develop neonatal respiratory distress, nasal congestion, otitis media and recurrent respiratory infections leading to bronchiectasis[2]. A laterality defect, male infertility and congenital heart disease are also commonly associated with PCD[3].
Correct diagnosis of PCD can present challenges as its clinical manifestations mimic that of other diseases such as cystic fibrosis (CF), primary immunodeficiencies and asthma[4]. A knowledge of the disease manifestations and robust diagnostics are important to avoid delayed or incorrect diagnosis[5]. There is no specific single gold-standard test to diagnose PCD, and it requires multiple investigations such as genetic testing, mucosal biopsies, nasal nitric oxide levels and electron microscopy[6,7]. Management of PCD draws from the treatment strategies employed for CF and non-CF bronchiectasis and includes respiratory physiotherapy, comprehensive treatment of acute infections, lifestyle mana
Structural lung changes such as mucus plugging, bronchial wall thickening, atelectasis, and bronchiectasis have been shown by chest computed tomography (CT) to develop in infancy and early childhood in patients with PCD[9-11]. Chest CT has increased sensitivity over pulmonary function tests in diagnosing early lung disease in PCD[12]. A cornerstone of PCD management is regular surveillance and prompt treatment of any pulmonary exacerbation to minimise or prevent the establishment of permanent lung damage. In an inherently young PCD patient cohort that will receive early and regular diagnostic and surveillance chest CTs, it is prudent to minimise exposure to ionising radiation[13]. There has been recent development and subsequent refinement of radiation-reducing CT techniques that have allowed significant radiation dose reductions, with chest CT doses in the range of chest radiography[14-16]. The continued assurance of diagnostic integrity of these reduced-dose protocols is clearly of paramount importance.
This study is designed to prospectively evaluate ultra-low dose CT (ULDCT) chest in identifying pulmonary changes within a paediatric PCD cohort in comparison to chest radiography.
This prospective study received approval from the local institutional review board (Clinical Research Ethics Committee Reference Number: ECM 4 (n) 12/11/2019 & ECM 3 (ll) 28/06/2022). Written informed consent was obtained from each patient or their guardian prior to participation.
Paediatric patients with PCD between the ages of 4 and 16 years, who presented for routine clinical outpatient follow-up between July 1, 2022 and July 1, 2023, were eligible for inclusion in the study. The patients were scheduled to undergo a clinically indicated surveillance ULDCT of the chest. Those who did not have a recent chest radiograph had one performed in addition to the CT chest.
ULDCT Chest was performed on a single Aquilion Prime SP CT Scanner (Canon Medical Systems, Otawara, Tochigi, Japan). Only one lateral scout performed at 80Kv and 10mA. The ultra-low dose helical CT scan was performed with the following acquisition parameters; 120 Kv, 10 mA, 0.35 second rotation time, small focus, standard pitch factor of 0.813, standard helical pitch of 65 and thickness of 0.5 × 80. Images were reconstructed in 2 mm and 1 mm soft tissue and lung windows.
All images were acquired on inspiration to include lung apex to diaphragm and no additional expiration views were obtained. No conscious sedation or general anaesthesia were utilised. Spirometry assisted CT was not performed. Patients received coaching from a respiratory physiotherapist with satisfactory outcome in breath holding.
Disease severity: For the evaluation of PCD severity using chest CT, we utilized the Brody II CT scoring system[17]. The Brody II score comprises five components: Bronchiectasis, mucus plugging, peri-bronchial thickening, parenchymal abnormalities, and air trapping. Each component is evaluated based on the extent of involvement in central and peripheral lung areas and specific severity criteria. Bronchiectasis is scored from 1-12, considering the extent in both lung areas and bronchiectasis size, with multipliers ranging from 0.5 to 3. Mucus plugging is scored from 0-6, again based on the extent in both lung areas. Peribronchial thickening, scored from 0-9 and considers the extent and severity (mild, moderate, severe) in both lung areas. The parenchyma score, ranging from 0-9, is based on the extent of dense parenchymal opacity, ground glass opacity, and cysts or bullae. Lastly, air trapping is scored from 0-4.5, reflecting the extent of lung involvement and the size of affected areas. Our approach did not assess air trapping as to minimise radiation exposure, expiratory CT examinations were not acquired. This scoring system provides a detailed and standardized assessment of disease severity in PCD patients, enabling accurate comparisons within our study cohort.
Subjective image quality: Subjective image quality was assessed on a Picture Archiving and Communication System (Impax 6.5.3; Agfa Health- care, Mortsel, Belgium) on a monitor with a resolution of 3 megapixels.
Diagnostic acceptability on both soft tissue and lung windows, image noise on both soft tissue and lung windows, depiction of bronchovascular structures centrally and within 2 cm of the pleural surface and streak artefact were assessed by consensus by 2 radiologists utilising a previously validated subjective scoring system[14,15].
Briefly, images were graded on a scale of 1-5 with 1 = unacceptable, 2 = minimally acceptable, 3 = acceptable, 4 = highly acceptable and 5 = excellent. This assessment was carried out on six different levels of each CT scan; at the level of the apices, aortopulmonary window, carina, largest cardiac diameter, largest thoracic diameter, and upper abdomen (on soft tissue window assessment). Again, images were anonymised and randomly assessed to minimise bias.
Objective image quality: Objective image quality analysis was conducted on a dedicated workstation (Advantage Workstation VolumeShare 2, Version 4.4, GE Medical Systems) using previously validated quantitative signal to noise measurements by placing homogenous region of interests (ROIs) (diameter 10 mm) in predefined homogenous anatomic locations[14]. These ROIs were placed on the thoracic aorta and right paraspinal musculature at the level of the aortic arch, carina, and largest cardiac diameter. Each measurement was taken on three consecutive slices of the CT series and a mean value calculated to ensure accuracy. The mean attenuation value in Hounsfield units (HU) within the ROI and the standard deviation represented the signal and noise level, respectively. The signal-to-noise ratio (SNR) within each ROI was calculated by dividing mean HU by standard deviation.
A subgroup of 2 of the patients in this study had previously undergone modified low dose CT chest examinations prior to this study and these examinations [mean age 22 months and mean dose-length product (DLP) 10.5 mGycm] will serve as a baseline comparison/internal control group for image quality assessment.
Chest radiograph image assessment: The severity of pulmonary changes in chest radiographs was quantified with the Modified Chrispin-Norman score, with a possible range of 0 to 38[18,19]. Readers conducting the chest radiograph assessment were blinded to the ULDCT chest result. This scoring system evaluates five radiological parameters: Overinflation, bronchial line shadows, ring shadows, mottled shadows, and large soft shadows. Each parameter consists of specific elements that are assessed within different lung zones (right upper, left upper, right lower, and left lower). For overinflation, elements such as diaphragmatic depression, chest wall shape, and lung field appearance are scored. Each element within the parameters is rated on a scale from 0 to 2, where 0 signifies 'Not Present', 1 denotes 'Present, not marked', and 2 indicates 'Marked' presence of the feature.
Radiation dose assessment: The radiation dose delivered to the patient in each CT was calculated in terms of volumetric CT dose index (CTDIvol), DLP, size-specific dose estimate (SSDE) as per American Association of Physicists in Medicine, and effective dose (ED) was calculated by multiplying the DLP by an age specific multiplier for CT chest[20,21].
Clinical assessment: Comprehensive clinical data were collected for each participant to facilitate a thorough analysis of disease characteristics and treatment outcomes. Demographic information included gender and date of birth. Pulmonary function was assessed using spirometry measurements: Forced expiratory volume in one second (FEV1) and forced vital capacity (FVC), recorded both in litres (L) and as a percentage of the predicted values for the patient's age, gender, and height. The frequency of pulmonary exacerbations, particularly those necessitating intravenous antibiotic treatment, was recorded to evaluate disease severity and progression. Nutritional status was assessed using body mass index (BMI), Weight in kilograms, and weight centile, aligning with age-specific growth charts. Additionally, airway microbial composition was analysed to identify predominant bacterial colonisations or infections. This holistic approach to data collection aimed to capture a broad spectrum of clinical variables that influence the course and management of PCD.
Statistical analyses were carried out using statistical package for the social sciences (SPSS) version 29 (IBM SPSS Inc., Chicago, Il). Data were exported from Microsoft Office Excel (Microsoft Corporation, CA, United States) into SPSS for statistical analysis. For continuous variables, normality testing was first conducted using a Shapiro-Wilk test. Data are presented as mean ± SD unless otherwise specified.
Inter-rater agreement for ordinal-scaled Brody II ratings was estimated with Krippendorff alpha using the ordinal difference function, and calculated 95%CIs using percentile bootstrap intervals with 10000 bootstrap samples to account for clustering. Inter-rater agreement of the total Brody II scores was estimated using the intraclass correlation coefficient (ICC) with 95%CI. ICC values below 0.5 indicate poor reliability, between 0.5 and 0.75 moderate reliability, between 0.75 and 0.9 good reliability, and above 0.9 excellent reliability.
CT and radiograph image quality metrics were reported using descriptive statistics.
6 paediatric patients (67% male) underwent clinically indicated ULDCT chest examinations, with an additional CR if not already recently performed, for surveillance of their PCD. The patient ages were 4, 6, 6, 7, 11 and 16.
Mean FEV1 was 98.6 ± 7.5% predicted and 2.1 ± 1.1 L. The mean FVC was 99 ± 2.9% predicted and 2.0 ± 0.6 L.
Patients experienced a mean 4 ± 3.8 pulmonary exacerbations requiring intravenous antibiotics per year. Mean BMI was 19.8 ± 4.6 kg/m2 with a mean weight of 34.8 ± 14.8 kg and mean percentile for weight of 66 ± 34 percentile. The predominant airway microbial identified was pseudomonas aeruginosa (50%).
The mean ULDCT chest CTDIvol was 0.11 ± 0 mGy. The mean DLP was 2.11 ± 0.35 mGycm. The mean SSDE was 0.18 ± 0.02 mGy. The mean ED was 0.08 ± 0.02 mSv. In comparison, the mean ED from CR was 0.03 mSv ± 0.03 mSv.
CT disease severity: The average Brody II CT score across the entire cohort was 12.92, with excellent inter-rater reliability and ICC of 0.98 (95%CI: 0.87, 0.99). Inter-rater reliability of each component of the Brody II score in terms of Krippendorff alpha was good for mucus plugging (0.8) and parenchymal opacity (0.79) and poor for bronchiectasis (0.34) and pe
| Brody II criteria | Inter-rater reliability |
| Bronchiectasis | 0.34 (-0.46–0.87) |
| Peribronchial thickening | 0.34 (-0.33–0.92) |
| Mucus plugging | 0.8 (0.52–0.99) |
| Parenchymal opacity | 0.79 (0.67–0.91) |
| Air trapping | Not assessed |
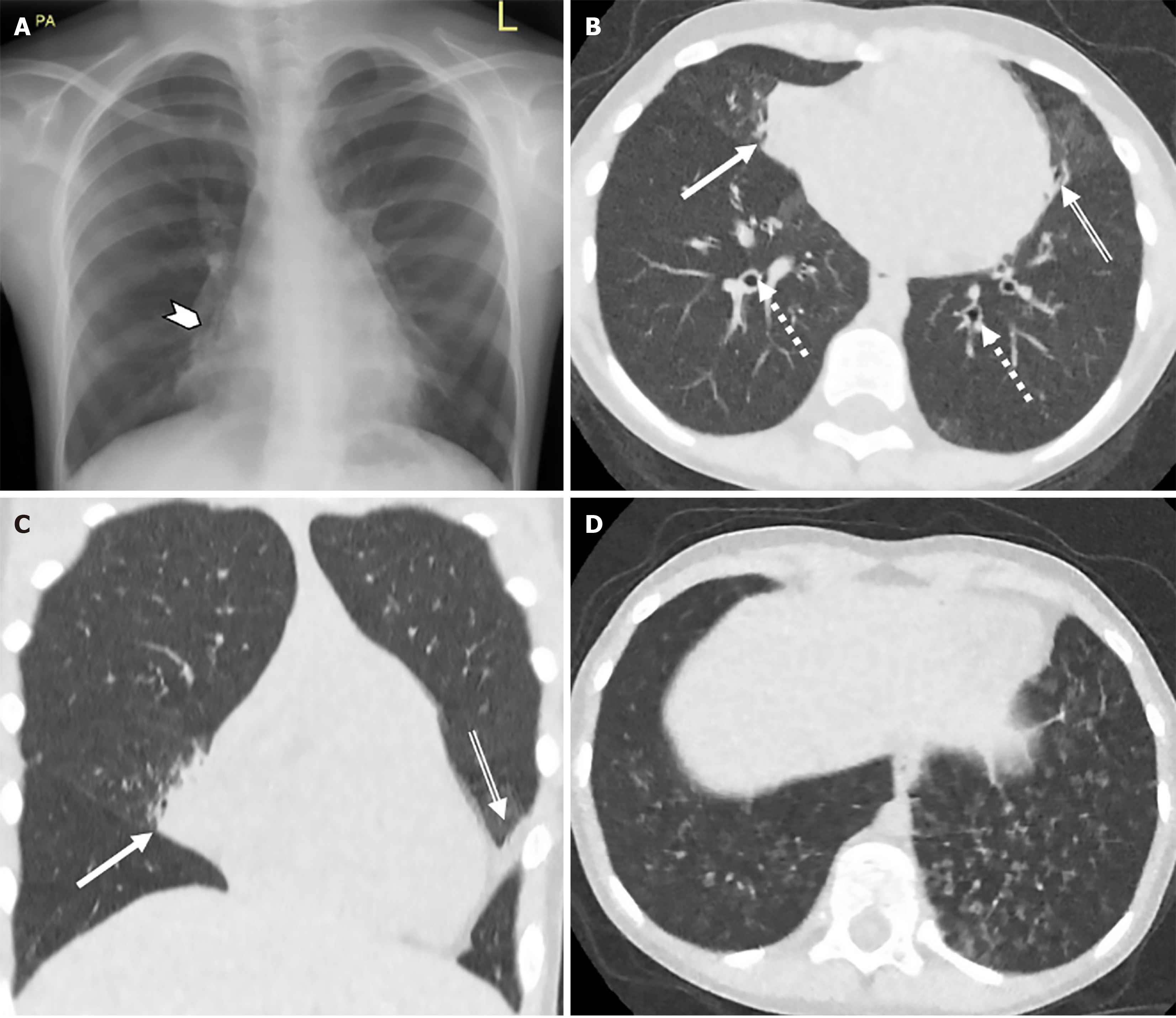
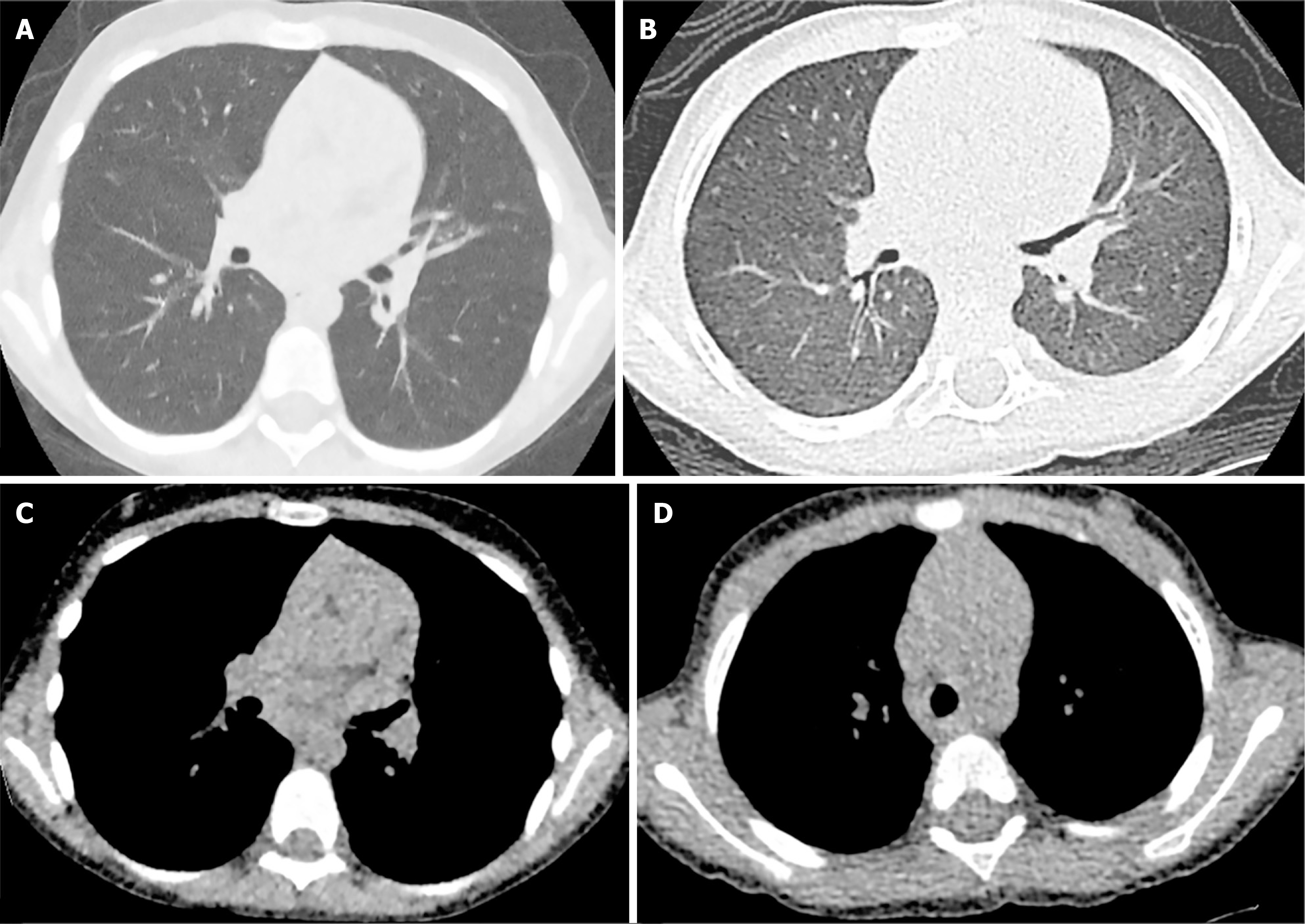
Figure 1 demonstrates representative ULDCT chest images of a 7-year-old female patient with PCD and the protocol’s ability to identify associated structural lung pathology. Figure 2 demonstrates comparative images of ULDCT chest and a modified low dose CT chest in the same patient obtained 7 years apart. The assessment of lung structure is acceptable in our ultra-low dose protocol with greater noise being evident in the mediastinum on soft tissue windows.
CT subjective image quality: The mean of both readers subjective quality scores was utilised for image quality asse
Subjective image quality was acceptable to highly acceptable on all categories. There were comparable quality scores in the small 2-patient subgroup who underwent prior modified low dose CT imaging (Table 2).
| Mean subjective image quality score | Image noise lung windows | Image noise soft tissue windows | Central bronchovascular structure depiction | Peripheral (within 2 cm of parietal pleura) bronchovascular structure depiction | Streak artifact | Overall diagnostic acceptability on lung windows | Overall diagnostic acceptability on soft tissue windows |
| Overall | 4.1 ± 0.49 | 3.2 ± 0.06 | 3.9 ± 0.91 | 3.5 ± 1.12 | 3.3 ± 0.15 | 3.9 ± 1.07 | 3.3 ± 0.11 |
| 2-patient subgroup | 3.5 ± 0.71 | 2.8 ± 0.05 | 3.9 ± 0.14 | 3.7 ± 0.21 | 3.4 ± 0.77 | 3.8 ± 0.28 | 3.0 ± 0.29 |
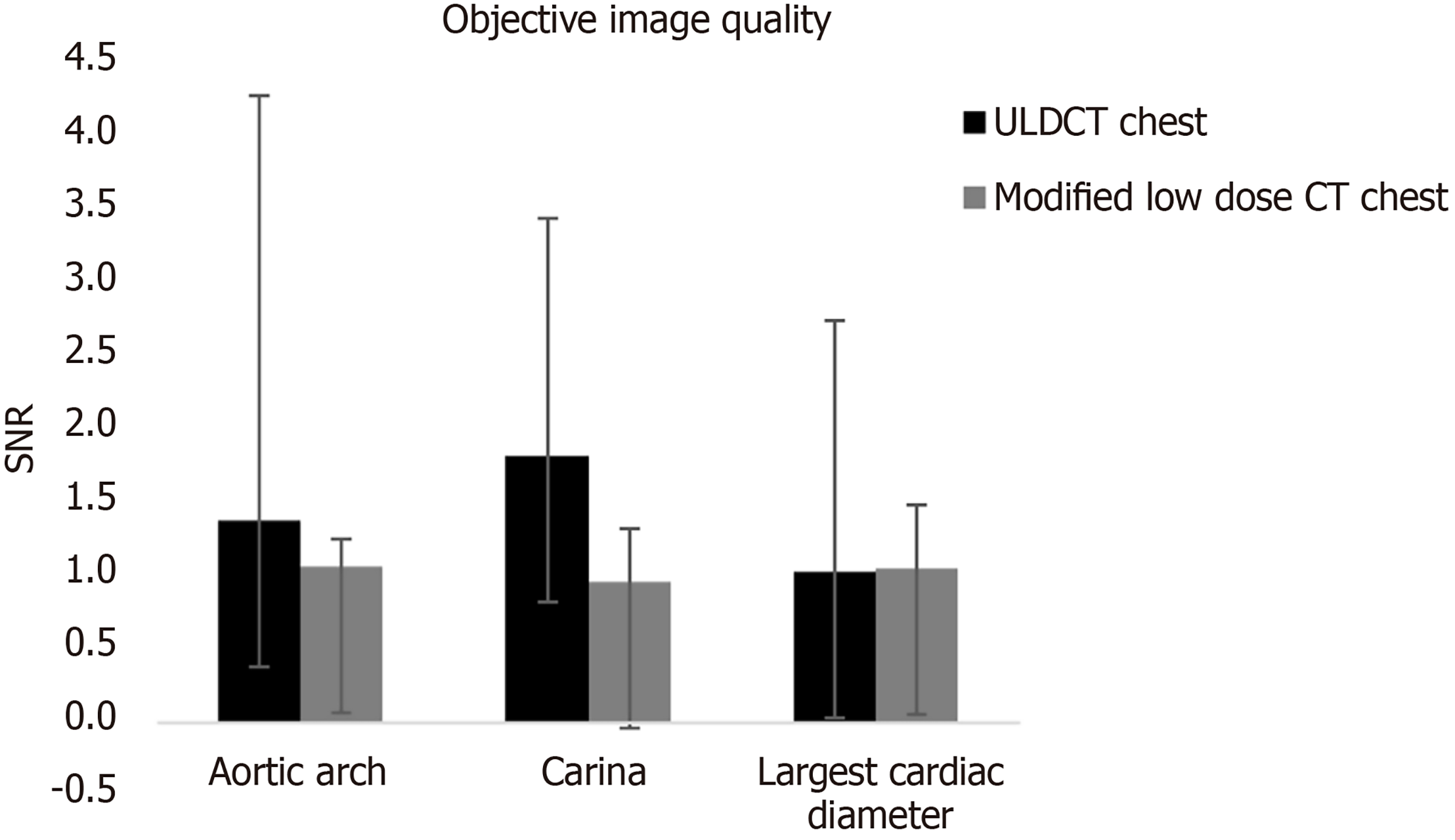
CT objective image quality: The mean SNR at the level of the arch, carina, and largest cardiac diameter for the ULDCT chest examinations were 1.38 ± 2.89, 1.82 ± 1.61 and 1.03 ± 1.7 respectively. For the 2-patient internal control subgroup that previously underwent modified low dose CT chest examinations prior to this study, the mean SNR at the level of the arch, carina, and largest cardiac diameter were 1.06 ± 0.19, 0.96 ± 0.36 and 1.05 ± 0.43 respectively (Figure 3).
Chest radiograph disease severity: The average Chrispin-Norman score on chest radiograph was 1 with excellent inter-rater reliability and ICC of 0.92 (95%CI: 0.41, 0.99).
This study illustrates the potential of ULDCT chest in paediatric patients with PCD to produce diagnostic-quality images capable of identifying key PCD-related pulmonary features, including bronchiectasis, peri-bronchial thickening, and airspace opacities, some of which are seen in Figures 1 and 2. These images facilitate relatively consistent and re
Our patient cohort demonstrated expected clinical characteristics of a PCD patient cohort including satisfactory PFTs, an expected average number of pulmonary exacerbations requiring hospital care annually and typical airway microbials. The spectrum of disease in PCD is like that of CF and utilising a CF specific scoring system to assess disease burden is a reasonable approach. Magnetic resonance imaging (MRI) based lung clearance index has been proposed as a possible reliable disease monitoring metric in PCD[22]. This, however, has the associated practical limitations of MRI in a paediatric population in terms of sedation, cost, expertise and limited access to appropriate MRI infrastructure.
It has been proposed that CF specific CT scoring systems do not capture the entirety of the PCD disease process[23]. Rademacher et al[24] were one of the first to describe a PCD specific CT scoring system (PCD-CT) in adults in 2021 and further work was carried out by Chowdhary et al[25] having proposed the Specific PCD Evaluation by CT (SPEC) score in a cohort of 30 patients that correlated well with PFTs. The PCD-CT scoring system was developed by evaluating several imaging markers of lung disease including mucus plugging, tree-in-bud pattern, atelectasis, peripheral and central consolidations, peripheral and central ground glass opacities, interlobular septal thickening, mosaic attenuation, cavities, emphysema and fibrosis. Using multivariate logistic regression, 5 variables: Absence of fibrosis and emphysema, predominance of bronchiectasis in the middle/Lower lobe and atelectasis, tree-in-bud pattern, and atelectasis or history of resection of a middle/Lower lobe were selected for the final scoring system. In contrast, the SPEC scoring system has several overlapping elements with the Brody II scoring system such as bronchiectasis severity and extent, extent of bronchial wall thickening, presence of atelectasis and presence of air trapping. In addition, each lobe is evaluated separately just like the Brody II scoring system. However, the SPEC system also assesses bronchoceles, the extent of tree-in-bud pattern of mucus plugging and the presence of interlobar septal thickening. The additional elements aim to cater specifically to the PCD pattern of disease. While the initial results for both scoring systems show promise with strong correlation to pulmonary function tests, they remain less well established than the Brody II score. Additionally, neither of these scoring systems have been compared directly with the Brody II score to evaluate any differences in performance[24,25]. Therefore, our study utilised the Brody II CF scoring system, which has previously been well established in PCD cohorts, to minimise unknown variables in the assessment of our novel ULDCT chest protocol[9,10].
As seen in Table 1, inter-rater reliability for both overall Brody II score on CT and Chrispin Norman score on CR was excellent. On more detailed analysis of the specific elements of the Brody II score, there was poor inter-rater reliability when assessing bronchiectasis and peri-bronchial thickening. A likely cause for this is the relatively mild disease burden of this young patient group and the inherent subjective nature of scoring systems.
Quantitative analysis of airway dilation and airway to artery ratios have been demonstrated in 13 paediatric patients with PCD in comparison with healthy controls, highlighting the potential benefit of novel software developments in the routine assessment of PCD patients[26]. Interestingly, in this study, airway thickening was not a predominant feature.
Objective and subjective image quality is grossly comparable to the small internal control group who underwent prior modified low dose CT chest examination prior to this study (Table 2, Figure 3). The ability to conduct CT examinations at such low radiation doses (mean ED 0.08 mSv), in combination with the added diagnostic value compared to CR, suggests substantial implications for PCD imaging guidelines.
This study highlights the potential significant radiation dose saving available with the application of relatively straightforward imaging protocol adjustments, without compromising diagnostic integrity. ULDCT chest will establish and maintain a central role in paediatric PCD diagnostics going forward. However, ULDCT may be further improved by a revolutionary new CT technology: The photon counting CT (PCCT). This technology makes use of photon counting detectors in place of the standard energy integrating detectors and shows great improvement in image resolution while reducing radiation dose. Indeed, in the paediatric chest, PCCT offers improved visualisation of peripheral airways, the bronchial wall and the bronchial segments, all of which may be useful in monitoring progression of PCD[27,28]. Furthermore, application of low dose image acquisition protocols to CT scanners with photon counting detectors shows greater reductions in radiation dose levels with reduced image noise when compared with standard energy integrating detectors[29]. However, although promising for future use in PCD diagnostics and follow-up, this technology is unfortunately limited by its scarce availability and high cost[30].
The strength of this study lies in its prospective design and the blinded image assessment of two consultant radio
ULDCT offers a viable alternative to conventional imaging for patients with PCD providing diagnostic quality images at a dose equivalent to a frontal and lateral CR. These low-dose images can be reliably assessed with the Brody II scoring system and offer superior diagnostic capability compared to CR. This study reinforces the limitations of CR and further supports the integration of ULDCT into routine care with potential for further enhancement through new technologies such as the photon counting CT. Although this study has limitations such as a small sample size and a single centre cohort, the prospective design and blinded scoring strengthen the findings. Future research may be directed towards validation of ULDCT in larger multicentre cohorts and assessment of newer technologies such as the photon counting CT in a PCD specific cohort.
| 1. | Knowles MR, Zariwala M, Leigh M. Primary Ciliary Dyskinesia. Clin Chest Med. 2016;37:449-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 147] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 2. | Leigh MW, Ferkol TW, Davis SD, Lee HS, Rosenfeld M, Dell SD, Sagel SD, Milla C, Olivier KN, Sullivan KM, Zariwala MA, Pittman JE, Shapiro AJ, Carson JL, Krischer J, Hazucha MJ, Knowles MR. Clinical Features and Associated Likelihood of Primary Ciliary Dyskinesia in Children and Adolescents. Ann Am Thorac Soc. 2016;13:1305-1313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 147] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 3. | Horani A, Ferkol TW. Understanding Primary Ciliary Dyskinesia and Other Ciliopathies. J Pediatr. 2021;230:15-22.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 87] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 4. | Shapiro AJ, Davis SD, Polineni D, Manion M, Rosenfeld M, Dell SD, Chilvers MA, Ferkol TW, Zariwala MA, Sagel SD, Josephson M, Morgan L, Yilmaz O, Olivier KN, Milla C, Pittman JE, Daniels MLA, Jones MH, Janahi IA, Ware SM, Daniel SJ, Cooper ML, Nogee LM, Anton B, Eastvold T, Ehrne L, Guadagno E, Knowles MR, Leigh MW, Lavergne V; American Thoracic Society Assembly on Pediatrics. Diagnosis of Primary Ciliary Dyskinesia. An Official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2018;197:e24-e39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 323] [Article Influence: 53.8] [Reference Citation Analysis (0)] |
| 5. | Shapiro AJ, Zariwala MA, Ferkol T, Davis SD, Sagel SD, Dell SD, Rosenfeld M, Olivier KN, Milla C, Daniel SJ, Kimple AJ, Manion M, Knowles MR, Leigh MW; Genetic Disorders of Mucociliary Clearance Consortium. Diagnosis, monitoring, and treatment of primary ciliary dyskinesia: PCD foundation consensus recommendations based on state of the art review. Pediatr Pulmonol. 2016;51:115-132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 280] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 6. | Lucas JS, Alanin MC, Collins S, Harris A, Johansen HK, Nielsen KG, Papon JF, Robinson P, Walker WT. Clinical care of children with primary ciliary dyskinesia. Expert Rev Respir Med. 2017;11:779-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Noone PG, Leigh MW, Sannuti A, Minnix SL, Carson JL, Hazucha M, Zariwala MA, Knowles MR. Primary ciliary dyskinesia: diagnostic and phenotypic features. Am J Respir Crit Care Med. 2004;169:459-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 523] [Cited by in RCA: 502] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 8. | Lucas JS, Carroll M. Primary ciliary dyskinesia and cystic fibrosis: different diseases require different treatment. Chest. 2014;145:674-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 9. | Santamaria F, Montella S, Tiddens HAWM, Guidi G, Casotti V, Maglione M, de Jong PA. Structural and functional lung disease in primary ciliary dyskinesia. Chest. 2008;134:351-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 75] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 10. | Jain K, Padley SP, Goldstraw EJ, Kidd SJ, Hogg C, Biggart E, Bush A. Primary ciliary dyskinesia in the paediatric population: range and severity of radiological findings in a cohort of patients receiving tertiary care. Clin Radiol. 2007;62:986-993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 53] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 11. | Kennedy MP, Noone PG, Leigh MW, Zariwala MA, Minnix SL, Knowles MR, Molina PL. High-resolution CT of patients with primary ciliary dyskinesia. AJR Am J Roentgenol. 2007;188:1232-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 111] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 12. | Maglione M, Bush A, Montella S, Mollica C, Manna A, Esposito A, Santamaria F. Progression of lung disease in primary ciliary dyskinesia: is spirometry less accurate than CT? Pediatr Pulmonol. 2012;47:498-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 13. | Joyce S, Carey BW, Moore N, Mullane D, Moore M, McEntee MF, Plant BJ, Maher MM, O'Connor OJ. Computed tomography in cystic fibrosis lung disease: a focus on radiation exposure. Pediatr Radiol. 2021;51:544-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 14. | O'Connor OJ, Vandeleur M, McGarrigle AM, Moore N, McWilliams SR, McSweeney SE, O'Neill M, Ni Chroinin M, Maher MM. Development of low-dose protocols for thin-section CT assessment of cystic fibrosis in pediatric patients. Radiology. 2010;257:820-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 67] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Moloney F, Kavanagh RG, Ronan NJ, Grey TM, Joyce S, Ryan DJ, Moore N, O'Connor OJ, Plant BJ, Maher MM. Ultra-low-dose thoracic CT with model-based iterative reconstruction (MBIR) in cystic fibrosis patients undergoing treatment with cystic fibrosis transmembrane conductance regulators (CFTR). Clin Radiol. 2021;76:393.e9-393.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 16. | Sheahan KP, O'Mahony AT, Morrissy D, Ibrahim H, Crowley C, Waldron MG, Sokol-Randell D, McMahon A, Maher MM, O'Connor OJ, Plant BJ. Replacing Plain Radiograph with ultra-low dose CT thorax in cystic fibrosis (CF) in the era of CFTR modulation and its impact on cumulative effective dose. J Cyst Fibros. 2023;22:715-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 17. | Brody AS, Klein JS, Molina PL, Quan J, Bean JA, Wilmott RW. High-resolution computed tomography in young patients with cystic fibrosis: distribution of abnormalities and correlation with pulmonary function tests. J Pediatr. 2004;145:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 258] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 18. | Chrispin AR, Norman AP. The systematic evaluation of the chest radiograph in cystic fibrosis. Pediatr Radiol. 1974;2:101-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 216] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 19. | de Jong PA, Achterberg JA, Kessels OA, van Ginneken B, Hogeweg L, Beek FJ, Terheggen-Lagro SW. Modified Chrispin-Norman chest radiography score for cystic fibrosis: observer agreement and correlation with lung function. Eur Radiol. 2011;21:722-729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 20. | Romanyukha A, Folio L, Lamart S, Simon SL, Lee C. BODY SIZE-SPECIFIC EFFECTIVE DOSE CONVERSION COEFFICIENTS FOR CT SCANS. Radiat Prot Dosimetry. 2016;172:428-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 21. | Brady SL, Kaufman RA. Investigation of American Association of Physicists in Medicine Report 204 size-specific dose estimates for pediatric CT implementation. Radiology. 2012;265:832-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 105] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 22. | Boon M, Vermeulen FL, Gysemans W, Proesmans M, Jorissen M, De Boeck K. Lung structure-function correlation in patients with primary ciliary dyskinesia. Thorax. 2015;70:339-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 23. | Tadd K, Morgan L, Rosenow T, Schultz A, Susanto C, Murray C, Robinson P. CF derived scoring systems do not fully describe the range of structural changes seen on CT scans in PCD. Pediatr Pulmonol. 2019;54:471-477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Rademacher J, Dettmer S, Fuge J, Vogel-Claussen J, Shin HO, Shah A, Pedro PI, Wilson R, Welte T, Wacker F, Loebinger MR, Ringshausen FC. The Primary Ciliary Dyskinesia Computed Tomography Score in Adults with Bronchiectasis: A Derivation und Validation Study. Respiration. 2021;100:499-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 25. | Chowdhary T, Bracken J, Morgan L, Schultz A, Robinson P. The SPEC score-A quantifiable CT scoring system for primary ciliary dyskinesia. Pediatr Pulmonol. 2024;59:72-80. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 26. | Ferraro V, Andrinopoulou ER, Sijbring AMM, Haarman EG, Tiddens HAWM, Pijnenburg MWH. Airway-artery quantitative assessment on chest computed tomography in paediatric primary ciliary dyskinesia. ERJ Open Res. 2020;6:00210-02019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 27. | Siegel MJ, Bugenhagen SM, Sanchez A, Kim S, Abadia A, Ramirez-Giraldo JC. Comparison of Radiation Dose and Image Quality of Pediatric High-Resolution Chest CT Between Photon-Counting Detector CT and Energy-Integrated Detector CT: A Matched Study. AJR Am J Roentgenol. 2023;221:363-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 28. | Aliukonyte I, Caudri D, Booij R, van Straten M, Dijkshoorn ML, Budde RPJ, Oei EHG, Saba L, Tiddens HAWM, Ciet P. Unlocking the potential of photon counting detector CT for paediatric imaging: a pictorial essay. BJR Open. 2024;6:tzae015. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 29. | Rajagopal JR, Farhadi F, Solomon J, Sahbaee P, Saboury B, Pritchard WF, Jones EC, Samei E. Comparison of Low Dose Performance of Photon-Counting and Energy Integrating CT. Acad Radiol. 2021;28:1754-1760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |