Published online Sep 26, 2021. doi: 10.4330/wjc.v13.i9.456
Peer-review started: March 19, 2021
First decision: May 13, 2021
Revised: May 26, 2021
Accepted: July 23, 2021
Article in press: July 23, 2021
Published online: September 26, 2021
Processing time: 177 Days and 0.8 Hours
Coronary artery vasospasm (CAVS) plays an important role in acute chest pain syndrome caused by transient and partial or complete occlusion of the coronary arteries. Pathophysiology of the disease remains incompletely understood, with autonomic and endothelial dysfunction thought to play an important role. Due to the dynamic nature of the disease, its exact prevalence is not entirely clear but is found to be more prevalent in East Asian and female population. Cigarette smoking remains a prominent risk factor, although CAVS does not follow traditional coronary artery disease risk factors. Many triggers continue to be identified, with recent findings identifying chemotherapeutics, allergens, and inflammatory mediators as playing some role in the exacerbation of CAVS. Provocative testing with direct visualization is currently the gold-standard for diagnosis, but non-invasive tests, including the use of biomarkers, are being increasingly studied to aid in the diagnosis. Treatment of the CAVS is an area of active research. Apart from risk factor modification, calcium channel blockers are currently the first line treatment, with nitrates playing an important adjunct role. High-risk patients with life-threatening complications should be considered for implantable cardioverter defibrillator (ICD), although timing criteria for escalated therapy require further investigation. The role of pharmaceuticals targeting oxidative stress remains incompletely understood.
Core Tip: Coronary artery vasospasm (CAVS) represents a spectrum of transient coronary arterial occlusion which can lead to serious complications, including sudden cardiac death. CAVS, often underdiagnosed and undertreated, should be considered in symptomatic patients with nonobstructive coronary arteries. Recent studies have expanded upon the etiology, epidemiology, and treatment options for CAVS.
- Citation: Jewulski J, Khanal S, Dahal K. Coronary vasospasm: A narrative review. World J Cardiol 2021; 13(9): 456-463
- URL: https://www.wjgnet.com/1949-8462/full/v13/i9/456.htm
- DOI: https://dx.doi.org/10.4330/wjc.v13.i9.456
Coronary artery vasospasm (CAVS) was first described as a “variant” of typical angina pectoris by Dr. Myron Prinzmetal in 1959, hence launching the study of a Prinzmetal (or “variant”) angina, which was subsequently termed vasospastic angina (VSA)[1]. Since then, the paradigm surrounding VSA has continued to expand. Symptoms develop upon transient partial or complete occlusion of coronary vessels resulting in a spectrum of clinical manifestations, ranging from stable angina to acute coronary syndrome (ACS), and in some cases sudden cardiac death (SCD)[1-6]. The disease process has been identified in patients both with and without coronary artery disease (CAD), affecting both epicardial and microvascular coronary arteries, and having both focal and diffuse involvement[2,4,7]. Certainly, traditional risk factors associated with CAD cannot be relied upon as predictors of CAVS, with the exception of cigarette smoking[1]. Although a prevailing precedent exists regarding the pathophysiology, diagnostics, and treatment of VSA, previous reviews have identified areas which require further study, especially in cases of refractory disease[8].
A number of mechanisms have been proposed regarding the mechanism of CAVS, including impairment of parasympathetic activity, coronary vascular and microvascular endothelial dysfunction, enhanced smooth muscle vasoconstriction, chronic inflammation, and oxidative stressors[4,5,7]. Recent studies have highlighted rarer causes of CAVS resulting from excessive adrenergic stimulation in cases of catecholamine-induced cardiomyopathy in pheochromocytomas and paragangliomas[9].
Approximately 5% to 30% of patients presenting with angina demonstrate normal or non-obstructive coronary arteries when worked up with coronary angiography, despite presenting with symptoms suggestive of acute coronary syndrome (ACS)[10-12]. Nearly half of these presentations may be attributed to CAVS[5,13], with an increased incidence amongst East Asians, especially Korean and Japanese populations[3,6,13,14]. This increased prevalence (in addition to environmental factors) has been attributed to genetic deficiency of variant aldehyde dehydrogenase 2 (ALDH2) genotype amongst these populations15. This correlates with increased toxic aldehyde accumulation, which may from the bases of future targeted treatment strategies in these populations[15]. Further studies demonstrate a need for exploring CAVS amongst Japanese adolescents less than 20 years old, whose clinical status matched severity of adults with refractory CAVS[16].
An increased incidence is additionally seen in women, with up to 70% of women showing ACS symptoms without CAD demonstrating findings suggestive of CAVS[17-19]. The incidence is particularly high for younger women under the age of 50 years, as well as non-white women[20,21]. For this reason, significant consideration should be given to a CAVS workup in patients from these demographics, especially given the tendency for worse outcomes in women compared to men[21]. Women with myocardial ischemia and no obstructive coronary artery disease (MINOCA) should undergo coronary reactivity testing (CRT), which reliably identifies the presence of CAVS[17].
Consideration of CAVS requires high clinical suspicion, and should also be considered in patients with rest angina or patients with anginal symptoms despite unremarkable coronary angiography[6,18]. Patients often develop symptoms between midnight and early morning, at times awoken from sleep by symptoms[15].
A number of triggers have been implicated in literature, with the most impactful trigger being cigarette smoking[2,18]. Other triggers include psychological stress, cold exposure, hyperventilation, alcohol consumption, stimulants (e.g., cocaine )[5,22]. Given increasing legalization within the United States, marijuana consumption has been considered as a contributor to CAVS exacerbation in a number of case reports[23].
Chemotherapies are other important causes of CAVS. Studies suggest that fluoropyrimidines (including 5-fluorouracil and capecitabine) induce vascular endothelial damage which can cause ischemia secondary to coronary artery vasospasm[24]. In such cases, chemotherapy should be halted and standard therapy for CAVS initiated, which should include calcium channel blockers (CCBs) and nitrates[24]. Further research is required surrounding cardioprotective agents such as coenzyme complex, glucagon-like peptide-1 (GLP-1) analogues, degradation inhibitors[24], and uridine triacetate[25]. Other chemotherapies should be considered if feasible, otherwise a CAD workup should be considered to stratify the patient’s risk for further fluoropyrimidine treatment[26].
Kounis syndrome (KS) is described as the occurrence of ACS in the setting of a mast-cell and platelet mediated hypersensitivity, anaphylactic, anaphylactoid, or allergic reaction[27,28]. One pathophysiology of KS includes CAVS, induced by inflammatory mediators[29]. Common triggers include antibiotics (especially beta-lactams[29]) and insect bites, which together account for half of reported cases[27]. Recent studies have expanded upon potential triggers, including supplements from traditional Chinese medicine (TCM)[27] and injection of cefuroxime[29].
Infectious myocarditis has been implicated in the development of CAVS in patients with otherwise non-obstructive coronary arteries. The underlying mechanism is believed to involve direct inflammatory and infectious interference with endothelial function of the coronary arteries[30]. Well documented etiologies include parasitic infections with Treponema cruzi (Chagas disease) and viral infections with parvovirus B19, amongst other causes[30].
Studies indicate that CAVS is an underdiagnosed and underreported disease[3,6,14]. Correct diagnosis can guide appropriate treatment, which can not only improve patient’s quality of life, but also reduce aforementioned serious risks associated with CAVS, including life-threatening arrhythmias and SCD[14]. Current gold standard diagnosis of CAVS utilizes pharmacological provocative testing with high-dose boluses of acetylcholine, ergonovine, or methylergonovine via intracoronary injection[2,3,5,13,14]. The response is then visualized as a coronary vasospasm with transient > 90% occlusion of coronary arteries[19], during coronary angiography, or via abnormalities of ventricular wall motion on echocardiogram[2,3]. A standard cardiac workup should initially include standard 12-lead electrocardiogram (ECG) during an attack, ambulatory cardiac monitoring, or exercise stress testing to exclude underlying CAD[18].
The Coronary Vasomotion Disorder International Study Group (COVADIS) developed diagnostic criteria for CAVS, which include: (1) Nitrate responsiveness to angina during spontaneous episodes with at least one of rest angina, marked diurnal variation in exercise tolerance, hyperventilation precipitating episodes, or calcium channel blockers (but not β-blockers) suppressing episodes; (2) Transient ischemic changes during spontaneous episodes including at least two contiguous leads with ST segment elevations ≥ 0.1 mV, ST segment depressions ≥ 0.1 mV, or new negative U waves; and (3) Coronary artery spasm visualized either spontaneously or during provocative testing[31]. Definitive CAVS is diagnosed if the first and either second or third criteria are fulfilled, which can guide further testing[31].
Although provocative testing is favored in the diagnosis of CAVS and is generally considered safe, the procedure does carry a small chance of serious complications. Recent studies have explored the use of biomarkers via blood test as a possible component in the workup for CAVS. One study explored a number of biomarkers including: (1) Inflammatory markers including C-reactive proteins (CRP), cytokines, lipoprotein (a), and cystatin-C as precipitating factors; (2) Vasoconstrictors including rho-kinase, serotonin (5-hydroxytryptamine), and endothelin-1 (ET-1); and (3) Oxidative stressors including thioredoxin and nitrotyrosine[32].
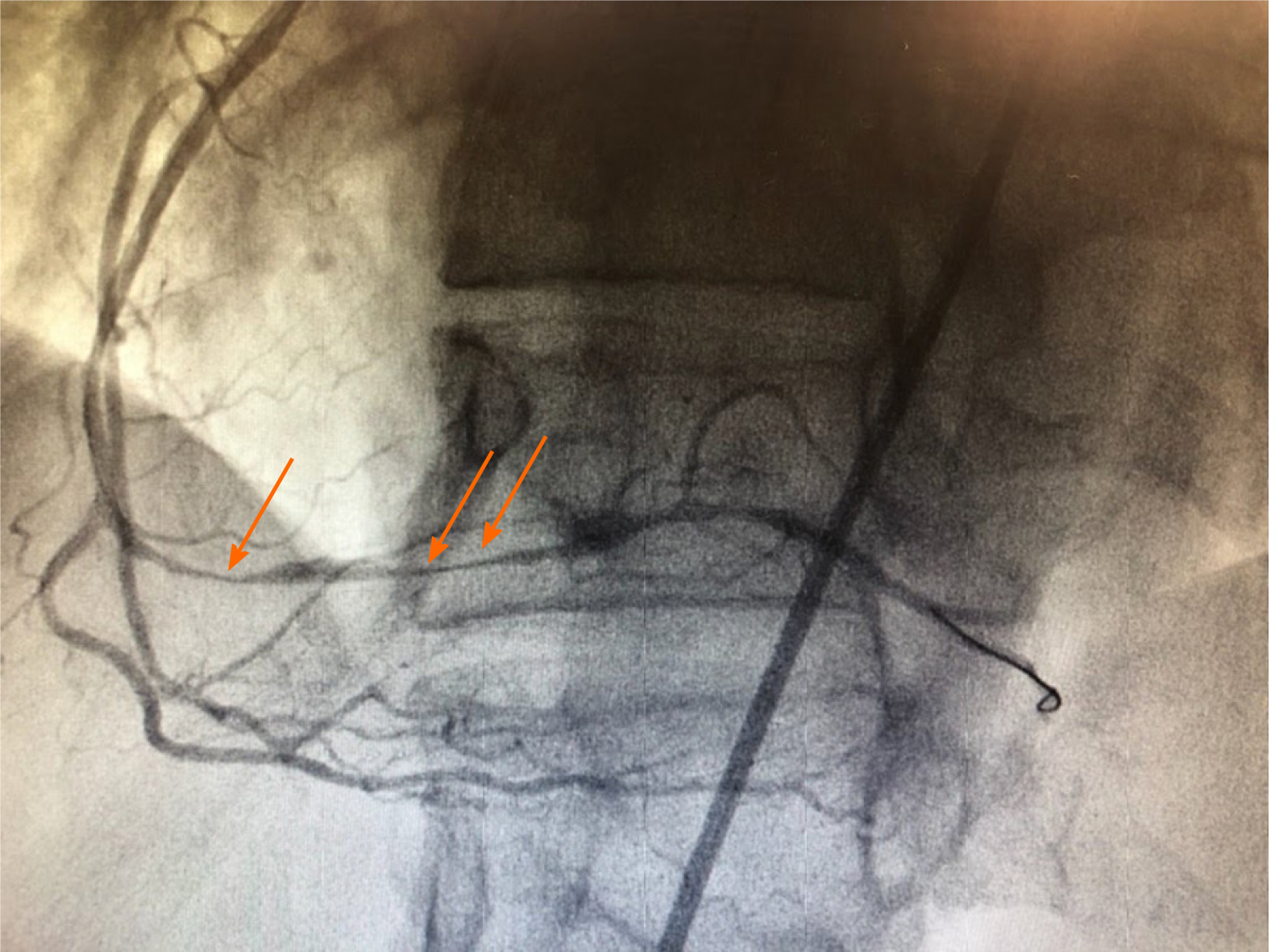
Previous studies have stipulated that increased testing and sensitivity of provocative testing can enhance detection of CAVS worldwide (especially Western countries), but this must be weighed against the risks associated with such testing[14,32]. Notably, rapid administration of intracoronary nitrate following provocative testing has resulted in no reported procedure-related deaths[13]. Furthermore, recent studies have demonstrated more efficacy and safety with local administration of nitroglycerin to spasming areas through a perforated balloon rather than proximal administration of nitroglycerin, which may further reduce minor risks associated with provocative testing[33]. The impact of local nitroglycerin administration on a spasming area of coronary artery can be visualized in Figures 1 and 2.
Initial approach to patients with CAVS should include lifestyle modifications, with the most prominent impact coming from smoking cessation[2,18]. Once this initial approach has been exhausted and symptoms remain refractory, a number of pharmacological options are available to supplement lifestyle notifications, noted in Table 1. First line pharmacologic treatment should include CCBs, with non-dihydropyridine being preferred but with similar efficacy to dihydropyridine CCBs[2,5]. Patients with persistent symptoms may benefit from the addition of long-acting nitrates, which reduce the frequency of anginal symptoms[2]. Additionally, sublingual nitrates may be useful for relieving acute episodes of angina[5]. Statins and angiotensin converting enzyme (ACE) inhibitors have shown efficacy in preventing CAVS episodes, and should be considered in all patients presenting with CAVS[2,5,19]. One study analyzed the use of sarpogrelate (a serotonin receptor antagonist) in addition to high-dose statins for CAVS, but did not find significant improvement of outcomes[34]. The use of magnesium and antioxidants (such as vitamin C and E) have demonstrated efficacy in many patients[5].
| Drug class | Common drugs | Usual drug dosage | Indication |
| CCB | Amlodipine; Diltiazem; Nifedipine-ER | 10 mg qD; 240 mg qD; 30-120 mg qD | First line for CAVS[40-42] |
| Long-acting nitrate | Isosorbide mononitrate | 60–240 mg qD (maintenance) | Symptomatic improvement in combination with first line therapy[2] |
| Short-acting nitrate | Sublingual nitroglycerin | 0.3 mg | Acute attack[43] |
| Statin | Lovastatin; Fluvastatin | 80 mg qD; 20-80 mg qD | All patients experiencing CAVS[15,44] |
| ACE inhibitor/ARB | Candesartan; Losartan | 8-16 mg qD; 25–50 mg qD | All patients experiencing CAVS[45] |
| BB | Bisoprolol; Nebivolol | 1.25–5 mg qD; 5–10 mg qD | DES-VSA[37,38] |
| Rho kinase inhibitors | Fasudil | 240 mg qD | Refractory CAVS[46] |
| Antioxidants | Vitamin E | 400 mg qD | Adjunct therapy[47] |
| Magnesium | Magnesium Chloride | 20 mEq | Replenishing deficiency[5] |
| Potassium channel activator | Nicorandil | 10–20 mg BID | If nitrates are ineffective[12] |
Patients with refractory disease who do not respond to lifestyle changes and first line pharmacotherapy may consider alpha 1-adrenergic receptor antagonists, rho-kinase inhibitors, or nicorandil[2,5,19]. Patients with significant atherosclerosis may benefit from percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG), which has shown benefit in patients with concomitant CAVS[5].
Patients who have experienced adverse outcomes of CAVS including life-threatening ventricular arrythmias or SCD may benefit from an implantable cardioverter defibrillator (ICD) in addition to medical therapy[2]. Notably, other reversible causes of SCD should be considered before escalating therapy. Although CAVS generally has a favorable prognosis, patients experiencing these adverse outcomes have worse outcomes[35]. Predictors of SCD in CAVS patients include advanced age, hypertension, hyperlipidemia, family history of sudden cardiac death, multivessel spasm, and left anterior descending (LAD) artery spasm, and should be considered for escalated therapy[35]. Recent studies have also explored the use sympathectomy, which reduced major adverse cardiac events in patients with refractory CAVS when compared to conventional treatment[36].
Beta blockers (BBs), both selective and nonselective forms (especially propranolol), should generally be avoided in patients with CAVS[2,5]. Blockage of beta-2 receptors, which prevents smooth muscle relaxation, can exacerbate anginal symptoms. The use of nebivolol, a BB with nitric oxide-releasing effects, has been compared to diltiazem for CAVS – both reduced the effects of CAVS, but diltiazem had greater reduction in symptoms at 12 wk[37]. A notable recent exception to this is drug-eluting stent-induced vasospastic angina (DES-VSA), which showed lower 2-year major cardiovascular events (MACE) with BBs compared to CCBs[38]. The use of aspirin (especially at high doses) in CAVS remains controversial, and there has been no clear demonstrated benefit of its use in patients with any degree of CAVS[2,5].
Although an understanding of CAVS continues to expand, there remains an ongoing need to better stratify the pathology. The effect of pharmacologic therapy, especially when considering generally avoided medications like BBs, should be tested in larger studies to ensure efficacy[38]. Studies have also emphasized the need to explore the use of fractional flow reserve (FFR) in the evaluation of moderate stenosis in the setting of MINOCA, nearly half of whom may have underlying CAVS[13,39]. Heart-type fatty acid-binding protein (h-FABP), myocardial performance (Tei) index, and genetic testing have been identified as potentially useful methods for tracking the development of CAVS in patients taking fluoropyrimidines[24,25]. Further studies should explore the timing of ICD implantation in high-risk patients with CAVS who would benefit from such therapy[3,35]. Future studies should consider populations in Western countries, where CAVS is generally underdiagnosed and undertreated[32].
CAVS is an important but underrecognized disease that can result in significant clinical symptoms and/or life-threatening complications. Its prevalence is decreasing due to decreasing prevalence of smoking, increasing use of the medications like calcium channel blockers, statins, and increasing use of non-invasive testing for CAD. It is important to consider CAVS in the differential of anginal chest pain in patients with non-obstructive CAD and treat it appropriately.
Manuscript source: Invited manuscript
Specialty type: Cardiac and cardiovascular systems
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ueda H S-Editor: Ma YJ L-Editor: A P-Editor: Li JH
| 1. | Swarup S, Patibandla S, Grossman SA. Coronary Artery Vasospasm. 2021 Aug 1. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021. [PubMed] |
| 2. | Picard F, Sayah N, Spagnoli V, Adjedj J, Varenne O. Vasospastic angina: A literature review of current evidence. Arch Cardiovasc Dis. 2019;112:44-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 85] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 3. | Song JK. Coronary Artery Vasospasm. Korean Circ J. 2018;48:767-777. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 4. | Cenko E, Bergami M, Varotti E, Bugiardini R. Vasospastic Angina and its Relationship with the Coronary Microcirculation. Curr Pharm Des. 2018;24:2906-2910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Matta A, Bouisset F, Lhermusier T, Campelo-Parada F, Elbaz M, Carrié D, Roncalli J. Coronary Artery Spasm: New Insights. J Interv Cardiol. 2020;2020:5894586. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 6. | Benamer H, Saighi Bouaouina M, Masri A, Sarkis G, El Beze N, Millien V. [Vasospastic angina: An under-diagnosed pathology]. Ann Cardiol Angeiol (Paris). 2019;68:341-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Ford TJ, Rocchiccioli P, Good R, McEntegart M, Eteiba H, Watkins S, Shaukat A, Lindsay M, Robertson K, Hood S, Yii E, Sidik N, Harvey A, Montezano AC, Beattie E, Haddow L, Oldroyd KG, Touyz RM, Berry C. Systemic microvascular dysfunction in microvascular and vasospastic angina. Eur Heart J. 2018;39:4086-4097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 135] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 8. | Teragawa H, Oshita C, Ueda T. Coronary spasm: It's common, but it's still unsolved. World J Cardiol. 2018;10:201-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 9. | Santos JRU, Brofferio A, Viana B, Pacak K. Catecholamine-Induced Cardiomyopathy in Pheochromocytoma: How to Manage a Rare Complication in a Rare Disease? Horm Metab Res. 2019;51:458-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 10. | van de Wiele C, Rimbu A, Belhocine T, de Spiegeleer B, Sathekge M, Maes A. Reversible myocardial perfusion defects in patients not suffering from obstructive epicardial coronary artery disease as assessed by coronary angiography. Q J Nucl Med Mol Imaging. 2018;62:325-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 11. | Leopoulou M, Mistakidi VC, Oikonomou E, Latsios G, Papaioannou S, Deftereos S, Siasos G, Antonopoulos A, Charalambous G, Tousoulis D. Acute Coronary Syndrome with Non-ruptured Plaques (NONRUPLA): Novel Ideas and Perspectives. Curr Atheroscler Rep. 2020;22:21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | Sidik NP, McEntegart M, Roditi G, Ford TJ, McDermott M, Morrow A, Byrne J, Adams J, Hargreaves A, Oldroyd KG, Stobo D, Wu O, Messow CM, McConnachie A, Berry C. Rationale and design of the British Heart Foundation (BHF) Coronary Microvascular Function and CT Coronary Angiogram (CorCTCA) study. Am Heart J. 2020;221:48-59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 13. | Tamis-Holland JE, Jneid H, Reynolds HR, Agewall S, Brilakis ES, Brown TM, Lerman A, Cushman M, Kumbhani DJ, Arslanian-Engoren C, Bolger AF, Beltrame JF; American Heart Association Interventional Cardiovascular Care Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; Council on Epidemiology and Prevention; and Council on Quality of Care and Outcomes Research. Contemporary Diagnosis and Management of Patients With Myocardial Infarction in the Absence of Obstructive Coronary Artery Disease: A Scientific Statement From the American Heart Association. Circulation. 2019;139:e891-e908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 559] [Article Influence: 93.2] [Reference Citation Analysis (0)] |
| 14. | Benamer H, Millien V. [Coronary spasm a diagnostic and therapeutic challenge]. Presse Med. 2018;47:798-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Yasue H, Mizuno Y, Harada E, Itoh T, Nakagawa H, Nakayama M, Ogawa H, Tayama S, Honda T, Hokimoto S, Ohshima S, Hokamura Y, Kugiyama K, Horie M, Yoshimura M, Harada M, Uemura S, Saito Y; SCAST (Statin and Coronary Artery Spasm Trial) Investigators. Effects of a 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitor, fluvastatin, on coronary spasm after withdrawal of calcium-channel blockers. J Am Coll Cardiol. 2008;51:1742-1748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 124] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 16. | Sueda S. Young Vasospastic Angina Patients Less Than 20 Years Old. Circ J. 2019;83:1925-1928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | AlBadri A, Mavromatis K, Bairey Merz CN. The role of coronary reactivity testing in women with no obstructive coronary artery disease. Curr Opin Cardiol. 2019;34:656-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 18. | Beijk MA, Vlastra WV, Delewi R, van de Hoef TP, Boekholdt SM, Sjauw KD, Piek JJ. Myocardial infarction with non-obstructive coronary arteries: a focus on vasospastic angina. Neth Heart J. 2019;27:237-245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 19. | Konst RE, Meeder JG, Wittekoek ME, Maas AHEM, Appelman Y, Piek JJ, van de Hoef TP, Damman P, Elias-Smale SE. Ischaemia with no obstructive coronary arteries. Neth Heart J. 2020;28:66-72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 20. | Henning RJ. Recognition and treatment of ischemic heart diseases in women. Future Cardiol. 2019;15:197-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 21. | Sluchinski SL, Pituskin E, Bainey KR, Norris CM. A Review of the Evidence for Treatment of Myocardial Infarction With Nonobstructive Coronary Arteries. CJC Open. 2020;2:395-401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Richards JR, Le JK. Cocaine Toxicity. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan. [PubMed] |
| 23. | Patel RS, Kamil SH, Bachu R, Adikey A, Ravat V, Kaur M, Tankersley WE, Goyal H. Marijuana use and acute myocardial infarction: A systematic review of published cases in the literature. Trends Cardiovasc Med. 2020;30:298-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 24. | Chong JH, Ghosh AK. Coronary Artery Vasospasm Induced by 5-fluorouracil: Proposed Mechanisms, Existing Management Options and Future Directions. Interv Cardiol. 2019;14:89-94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 25. | Depetris I, Marino D, Bonzano A, Cagnazzo C, Filippi R, Aglietta M, Leone F. Fluoropyrimidine-induced cardiotoxicity. Crit Rev Oncol Hematol. 2018;124:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 26. | Sara JD, Kaur J, Khodadadi R, Rehman M, Lobo R, Chakrabarti S, Herrmann J, Lerman A, Grothey A. 5-fluorouracil and cardiotoxicity: a review. Ther Adv Med Oncol. 2018;10:1758835918780140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 266] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 27. | Li J, Zheng J, Zhou Y, Liu X, Peng W. Acute coronary syndrome secondary to allergic coronary vasospasm (Kounis Syndrome): a case series, follow-up and literature review. BMC Cardiovasc Disord. 2018;18:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 28. | Sciatti E, Vizzardi E, Cani DS, Castiello A, Bonadei I, Savoldi D, Metra M, D'Aloia A. Kounis syndrome, a disease to know: Case report and review of the literature. Monaldi Arch Chest Dis. 2018;88:898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Mitsis A, Christodoulou E, Georgiou P. Coronary spasm secondary to cefuroxime injection, complicated with cardiogenic shock - a manifestation of Kounis syndrome: case report and literature review. Eur Heart J Acute Cardiovasc Care. 2018;7:624-630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Woudstra L, Juffermans LJM, van Rossum AC, Niessen HWM, Krijnen PAJ. Infectious myocarditis: the role of the cardiac vasculature. Heart Fail Rev. 2018;23:583-595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 31. | Beltrame JF, Crea F, Kaski JC, Ogawa H, Ong P, Sechtem U, Shimokawa H, Bairey Merz CN; Coronary Vasomotion Disorders International Study Group (COVADIS). International standardization of diagnostic criteria for vasospastic angina. Eur Heart J. 2017;38:2565-2568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 183] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 32. | Li L, Jin YP, Xia SD, Feng C. The Biochemical Markers Associated with the Occurrence of Coronary Spasm. Biomed Res Int. 2019;2019:4834202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 33. | Balaban Y, Kaya A, Satilmisoglu MH, Balaban MB. Intracoronary focal nitroglycerin injection through drilled balloon is very effective in the resolution of coronary spasm vs into proximal coronary artery: A prospective randomized comparison study. J Interv Cardiol. 2018;31:765-774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 34. | Kim SR, Choi KH, Song YB, Lee JM, Park TK, Yang JH, Hahn JY, Choi JH, Choi SH, Gwon HC. Effect of sarpogrelate and high-dose statin on the reduction of coronary spasm in vasospastic angina: A two by two factorial, pilot randomized study. Clin Cardiol. 2019;42:899-907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 35. | Kundu A, Vaze A, Sardar P, Nagy A, Aronow WS, Botkin NF. Variant Angina and Aborted Sudden Cardiac Death. Curr Cardiol Rep. 2018;20:26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 36. | Lin Y, Liu H, Yu D, Wu M, Liu Q, Liang X, Pang X, Chen K, Luo L, Dong S. Sympathectomy vs conventional treatment for refractory coronary artery spasm. Coron Artery Dis. 2019;30:418-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 37. | Kook H, Hong SJ, Yang KS, Lee S, Kim JS, Park CG. Comparison of nebivolol vs diltiazem in improving coronary artery spasm and quality of life in patients with hypertension and vasospastic angina: A prospective, randomized, double-blind pilot study. PLoS One. 2020;15:e0239039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 38. | Sawano M, Katsuki T, Kitai T, Tamita K, Obunai K, Ikegami Y, Yamane T, Ueda I, Endo A, Maekawa Y, Kawamura A, Fukuda K, Kohsaka S. Beta blockers vs calcium channel blockers for provocation of vasospastic angina after drug-eluting stent implantation: a multicentre prospective randomised trial. Open Heart. 2020;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 39. | Cruz Rodriguez JB, Kar S. Management of Angina Post Percutaneous Coronary Intervention. Curr Cardiol Rep. 2020;22:7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 40. | Ford TJ, Stanley B, Sidik N, Good R, Rocchiccioli P, McEntegart M, Watkins S, Eteiba H, Shaukat A, Lindsay M, Robertson K, Hood S, McGeoch R, McDade R, Yii E, McCartney P, Corcoran D, Collison D, Rush C, Sattar N, McConnachie A, Touyz RM, Oldroyd KG, Berry C. 1-Year Outcomes of Angina Management Guided by Invasive Coronary Function Testing (CorMicA). JACC Cardiovasc Interv. 2020;13:33-45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 177] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 41. | Higuma T, Oikawa K, Kato T, Mori Y, Kudo T, Yamamoto T, Hoshi Y, Kameda K, Suto N, Fujita N, Inokubo Y, Konta A, Osanai T, Okumura K. Comparison of the effects of long-acting nifedipine CR and diltiazem R in patients with vasospastic angina: Aomori coronary spastic angina study. J Cardiol. 2010;56:354-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 42. | Chahine RA, Feldman RL, Giles TD, Nicod P, Raizner AE, Weiss RJ, Vanov SK. Randomized placebo-controlled trial of amlodipine in vasospastic angina. Amlodipine Study 160 Group. J Am Coll Cardiol. 1993;21:1365-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 89] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 43. | Schroeder JS, Rosenthal S, Ginsburg R, Lamb I. Medical therapy of Prinzmetal's variant angina. Chest. 1980;78:231-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 44. | Treasure CB, Klein JL, Weintraub WS, Talley JD, Stillabower ME, Kosinski AS, Zhang J, Boccuzzi SJ, Cedarholm JC, Alexander RW. Beneficial effects of cholesterol-lowering therapy on the coronary endothelium in patients with coronary artery disease. N Engl J Med. 1995;332:481-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 919] [Cited by in RCA: 861] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 45. | Hirai N, Kawano H, Yasue H, Shimomura H, Miyamoto S, Soejima H, Kajiwara I, Sakamoto T, Yoshimura M, Nakamura H, Yodoi J, Ogawa H. Attenuation of nitrate tolerance and oxidative stress by an angiotensin II receptor blocker in patients with coronary spastic angina. Circulation. 2003;108:1446-1450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 42] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 46. | Vicari RM, Chaitman B, Keefe D, Smith WB, Chrysant SG, Tonkon MJ, Bittar N, Weiss RJ, Morales-Ballejo H, Thadani U; Fasudil Study Group. Efficacy and safety of fasudil in patients with stable angina: a double-blind, placebo-controlled, phase 2 trial. J Am Coll Cardiol. 2005;46:1803-1811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 158] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 47. | Miyamoto S, Kawano H, Takazoe K, Soejima H, Sakamoto T, Hokamaki J, Yoshimura M, Nakamura H, Yodoi J, Ogawa H. Vitamin E improves fibrinolytic activity in patients with coronary spastic angina. Thromb Res. 2004;113:345-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |