Published online Jul 15, 2017. doi: 10.4239/wjd.v8.i7.365
Peer-review started: October 28, 2016
First decision: January 20, 2017
Revised: May 13, 2017
Accepted: May 13, 2017
Article in press: June 8, 2017
Published online: July 15, 2017
Processing time: 252 Days and 12.4 Hours
To determine the predictive role of body mass index (BMI) and waist circumference (WC) for diabetes and prediabetes risk in future in total sample as well as in men and women separately.
In a population based cohort study, 1765 with mean ± SD age: 42.32 ± 6.18 healthy participants were followed up from 2003 till 2013 (n = 960). Anthropometric and biochemical measures of participants were evaluated regularly during the follow up period. BMI and WC measures at baseline and diabetes and prediabetes status of participants at 2013 were determined. Multivariable logistic regression analysis was used for determining the risk of diabetes and prediabetes considering important potential confounding variables. Receiver operating characteristic curve analysis was conducted to determine the best cut of values of BMI and WC for diabetes and prediabetes.
At 2013, among participants who had complete data, 45 and 307 people were diabetic and prediabetic, respectively. In final fully adjusted model, BMI value was a significant predictor of diabetes (RR = 1.39, 95%CI: 1.06-1.82 and AUC = 0.68, 95%CI: 0.59-0.75; P < 0.001) however not a significant risk factor for prediabetes. Also, WC was a significant predictor for diabetes (RR = 1.2, 95%CI: 1.05-1.38 and AUC = 0.67, 95%CI: 0.6-0.75) but not significant risk factor for prediabetes. Similar results were observed in both genders.
General and abdominal obesity are significant risk factors for diabetes in future.
Core tip: The predictive powers of body mass index (BMI) and waist circumference (WC) were similar in predicting the incidence risk of diabetes in either gender. The cut-off points for predicting diabetes in men and women were different. Defined cut-off points based on maximum sensitivity plus specificity values suggested that in men, BMI of 26.2 kg/m2 and WC of 89.7 cm, and in women, BMI of 28.6 kg/m2 and WC of 84.3 cm would predict Isfahanian population at high risk for developing diabetes.
- Citation: Haghighatdoost F, Amini M, Feizi A, Iraj B. Are body mass index and waist circumference significant predictors of diabetes and prediabetes risk: Results from a population based cohort study. World J Diabetes 2017; 8(7): 365-373
- URL: https://www.wjgnet.com/1948-9358/full/v8/i7/365.htm
- DOI: https://dx.doi.org/10.4239/wjd.v8.i7.365
The increased prevalence of obesity in the world[1] is a major concern as it is strongly related to multiple metabolic disorders[2]. General obesity measured by body mass index (BMI) is a known risk factor for diabetes[2]. Although BMI is often advocated as a simple measure to determine disease risk, it has several limitations. First, lean mass and fat mass could not be differentiated for a given BMI across age, sex and race[3]. Second, fat distribution could not be distinguished by BMI[4,5], whilst it has been generally accepted that visceral adiposity plays more important role in developing insulin resistance and diabetes rather than overall adiposity[6-8]. Therefore, waist circumference (WC) was developed as an abdominal adiposity measure which considers fat distribution.
Although in most populations WC is a stronger predictor for diabetes compared with BMI[7,8], no significant differences were observed between WC and BMI in Japanese[9] and Iranians[10] to predict disease risk. In addition, available risk thresholds predominantly come from European populations which might not be applicable to the Asian population due to differences in genetics and obesity pattern. Therefore, it is essential to identify the best anthropometric index and effective risk thresholds for adiposity measures to develop appropriate preventive strategies in each population.
Based on International Diabetes Federation (IDF)’s recommendation, WC cut-off values for clinical practice should be determined in different ethnicities[11]. Although IDF has suggested WC greater than 90 and 80 cm, respectively for Asian men and women, as cut-off point for abdominal obesity, there is no consensus for WC cut-off point in Iranians. Studies in this regard have suggested that 89-95 cm for men and 85-97 cm for women may be optimal cut-off points for abdominal obesity[12-15]. Nevertheless, to the best of our knowledge, there is only one longitudinal study among Iranians which has determined cut-off point of WC for detecting cardiovascular disease risk[15], and other cut-off points come from cross-sectional surveys[12-14,16]. These values might be limited because of the design of study.
The present study aimed to prospectively determine the effective anthropometric measures to predict the risk of type 2 diabetes and prediabetes also estimate the optimal cut-off point of WC and BMI by following up non-diabetic participants at baseline examination. Estimated cut-off points by this study will contribute to detect individuals at higher risk of developing type 2 diabetes as well as prediabetes in the Iranian population.
Subjects in the present study were the participants in the Isfahan Diabetes Prevention Study (IDPS), an ongoing cohort study in central Iran. The aim of this study is evaluating the role of diet and physical activity in the prevention or delay the developing of diabetes in first-degree relatives (FDRs) of patients with type 2 diabetes. This study was run between 2003 and 2013. One thousand, seven hundred and sixty-five healthy participants including 446 (25.3%) males and 1371 (74.6%) females were selected from a consecutive sample who attended in the clinics of Isfahan Endocrine and Metabolism Research Center. Data from 960 people including 255 (25.5%) male and 705 (73.4%) female at 2013 were subjected to statistical analysis. Health status and potential risk factors for diabetes were assessed using a questionnaire. To update demographic, anthropometric, and lifestyle information as well as diagnosis new diabetic cases, follow-up tests were run according to a medical care standard in diabetes[17]. Accordingly, participants with impaired 2-h OGTT at baseline were annually tested, and individuals with normal 75 g 2-h oral glucose test tolerance (2-h OGTT) were tested at least at 3-year intervals. More details regarding the participants and methodology of IDPS have been described elsewhere[18]. Informed written consent was obtained from all study participants and the Ethical Committee of Isfahan University of Medical Sciences approved the protocol of study.
Anthropometric indices were measured by well-trained examiners at baseline while participants were minimally clothed and without footwear. Weight was measured using a balanced scale and recorded to the nearest 0.1 kg. Height was determined using a wall-fixed tape measure while participants were in a normal standing position and recorded to the nearest 0.5 cm. WC and hip circumference were determined using a metal tape measure without imposing any pressure to body surface and were recorded to the nearest 0.5 cm. The location for measuring WC was considered as the narrowest level between the lowest rib and iliac crest, whilst for hip circumference was conserved as the largest level[19]. BMI was calculated as body weight in kilogram divided by height in Square meter.
Biochemical tests including lipid profile, fasting plasma glucose (FPG) and OGTT were carried out for all participants. To determine lipid profile and FPG, a blood sample was drawn from all participants after 10-12 h overnight fasting. Postprandial plasma glucose was measured using venous blood sample at 30, 60, and 120 min after oral glucose administration. Plasma glucose and lipid profile concentrations were determined using enzymatic colorimetric method (ParsAzmoon, Tehran, Iran) adapted to a Selectra-2 auto-analyzer (Vital Scientific, Spankeren, Netherlands). Serum concentration of low-density lipoprotein cholesterol (LDL-C) was calculated by Friedwald equation in individuals with serum TG levels < 400 mg/dL[20]. HbA1c concentrations were measured in whole blood samples via the pink reagent kit on a DS5 analyzer. Both intra- and inter-assay coefficients of variability (CVs) were < 2.2% for all markers.
The criteria for the diagnosis of diabetes and impaired glucose tolerance test were based on the American diabetes association (ADA) definition. Accordingly, cut-off point for impaired fasting glucose was considered as 100 mg/dL[17]. Diabetes was defined as FPG ≥ 126 mg/dL, or HbA1C ≥ 6.5% or 2-h OGTT ≥ 200 mg/dL.
Blood pressure was measured using a Mercury sphygmomanometer while subjects were in seated position two times with at least 30 s interval between measurements. The mean of two measurements was recorded as the subject’s blood pressure. Hypertriglyceridemia was defined as serum TG ≥ 150 mg/dL, high LDL-C as LDL-C ≥ 130 mg/dL, hypercholesterolemia asTC ≥ 200 mg/dL and low HDL-C as HDL-C < 50 mg/dL in female and < 40 mg/dL in male. According to the JNC and WHO Guideline criteria, hypertension was defined as systolic blood pressure (SBP) ≥ 130 mmHg, diastolic blood pressure (DBP) ≥ 85 mmHg and/or antihypertensive medications[21].
Continuous and categorical data were presented as mean ± SD. Normality of quantitative data was evaluated using Kolmogorov-Smirnov test and Q-Q plot. Positive skewed data was subjected to logarithmic transformation. χ2 test was used for evaluating the association between categorical data. Between groups comparisons of quantitative data were conducted using Analysis of variance (ANOVA) or nonparametric Kruskal-Wallis tests. To determine the association between BMI and WC values at baseline (2003) as an independent variable and type 2diabetes and prediabetes at 2013, we used binary logistic regression analysis in different models. In these analyses, after obtaining relative risk (RR) and 95% confidence interval (95%CI) in crude model, adjustment was made for age and sex, smoking, positive family history in the first model. Additional adjustment was made for physical activity and energy intake in the second model. In third model adjustment additionally was done for FBS and HbA1c. Finally, adjustment was made for all mentioned variables and lipid profile indices (including TG, LDL, HDL and cholesterol) and blood pressure.
The predictive values of BMI and WC values for type 2 diabetes and prediabetes were evaluated using receiver operating characteristic curve (ROC) analysis and area under the curve (AUC) and its 95%CI. The optimal sensitivity and specificity for different cut off values of BMI and WC were calculated using Youden index. Statistical analyses were performed using statistical package for social science (SPSS version 15, SPSS, Inc., IL, United States).
General characteristics of participants at baseline are presented in Table 1. Individuals who affected by diabetes after 10 years follow up had greater BMI, WC, hip circumference, waist to hip ratio, fasting blood sugar, glycemic response, total cholesterol, triglyceride and systolic and diastolic blood pressures at baseline. Abdominal obesity at baseline was more prevalent among those who developed (affected by) diabetes.
| Normal (n = 599)1 | Pre-diabetes (n = 307)1 | Diabetes (n = 45)1 | P value | |
| Whole population | ||||
| Age (yr) | 42.06 ± 6.17 | 42.72 ± 6.20 | 43.20 ± 6.19 | 0.197 |
| Energy intake (kcal/d) | 1844.32 ± 553.85 | 1793.43 ± 571.48 | 1908.95 ± 598.94 | 0.646 |
| Weight (kg) | 72.09 ± 12.10 | 73.51 ± 12.49 | 78.22 ± 11.96 | 0.943 |
| Height (cm) | 160.21 ± 8.31 | 160.09 ± 8.62 | 159.81 ± 8.58 | 0.197 |
| Body mass index (kg/m2) | 28.09 ± 4.17 | 28.66 ± 4.18 | 30.63 ± 4.15 | < 0.0001 |
| Waist circumference (cm) | 87.00 ± 9.58 | 88.72 ± 9.75 | 92.86 ± 9.08 | < 0.0001 |
| Hip circumference (cm) | 106.21 ± 8.54 | 107.35 ± 8.77 | 110.35 ± 9.27 | 0.003 |
| Waist to hip ratio | 0.82 ± 0.07 | 0.83 ± 0.07 | 0.84 ± 0.05 | 0.078 |
| Fasting blood sugar | 87.04 ± 7.99 | 89.30 ± 7.03 | 91.0 ± 6.14 | < 0.0001 |
| Blood sugar after 30 min (mg/dL) | 127.41 ± 25.19 | 136.43 ± 26.48 | 141.49 ± 25.48 | < 0.0001 |
| Blood sugar after 60 min (mg/dL) | 123.02 ± 32.06 | 136.19 ± 31.17 | 151.81 ± 36.61 | < 0.0001 |
| Blood sugar after 120 min (mg/dL) | 98.11 ± 21.15 | 104.81 ± 21.08 | 110.94 ± 18.80 | < 0.0001 |
| HbA1c (%) | 4.94 ± 0.78 | 5.08 ± 0.75 | 5.17 ± 0.78 | 0.014 |
| Triglyceride (mg/dL) | 150.15 ± 77.29 | 156.50 ± 83.83 | 200.82 ± 130.48 | < 0.0001 |
| Total cholesterol (mg/dL) | 190.30 ± 38.16 | 192.47 ± 35.97 | 205.51 ± 50.56 | 0.034 |
| LDL-C (mg/dL) | 116.34 ± 33.45 | 117.04 ± 32.26 | 123.52 ± 52.88 | 0.436 |
| HDL-C (mg/dL) | 45.04 ± 11.75 | 44.78 ± 10.31 | 45.12 ± 11.55 | 0.944 |
| Systolic blood pressure (mmHg) | 110.23 ± 10.49 | 110.73 ± 10.66 | 110.76 ± 10.64 | < 0.0001 |
| Diastolic blood pressure (mmHg) | 70.37 ± 10.11 | 70.64 ± 10.20 | 70.65 ± 10.26 | 0.002 |
| Abdominal obese (WC > 90 cm) (%) | 32.9 | 41.8 | 57.4 | < 0.0001 |
| Men | ||||
| Age (yr) | 43.04 ± 6.73 | 42.48 ± 6.20 | 43.85 ± 7.16 | 0.71 |
| Energy intake (kcal/d) | 2384.54 ± 558.37 | 2176.44 ± 648.34 | 2536.88 ± 651.42 | 0.343 |
| Weight (kg) | 77.98 ± 12.95 | 79.43 ± 13.44 | 81.38 ± 6.13 | 0.53 |
| Height (cm) | 170.78 ± 6.47 | 170.56 ± 6.30 | 169.55 ± 5.22 | 0.802 |
| Body mass index (kg/m2) | 26.66 ± 3.53 | 27.22 ± 3.82 | 28.32 ± 1.82 | 0.201 |
| Waist circumference (cm) | 92.63 ± 9.43 | 93.14 ± 9.86 | 97.65 ± 7.64 | 0.19 |
| Hip circumference (cm) | 102.76 ± 6.68 | 103.91 ± 7.76 | 108.15 ± 7.19 | 0.025 |
| Waist to hip ratio | 0.90 ± 0.06 | 0.89 ± 0.05 | 0.90 ± 0.03 | 0.754 |
| Fasting blood sugar | 87.55 ± 8.25 | 90.34 ± 6.91 | 92.54 ± 4.11 | 0.005 |
| Blood sugar after 30 min (mg/dL) | 133.20 ± 28.96 | 140.95 ± 28.86 | 144.42 ± 31.05 | 0.091 |
| Blood sugar after 60 min (mg/dL) | 128.50 ± 35.76 | 136.26 ± 35.35 | 147.69 ± 44.80 | 0.081 |
| Blood sugar after 120 min (mg/dL) | 91.18 ± 24.14 | 92.40 ± 21.22 | 110.08 ± 20.20 | 0.019 |
| HbA1c (%) | 4.94 ± 0.66 | 5.09 ± 0.89 | 5.15 ± 1.01 | 0.293 |
| Triglyceride (mg/dL) | 178.85 ± 97.60 | 177.54 ± 92.37 | 219.23 ± 101.25 | 0.328 |
| Total cholesterol (mg/dL) | 189.49 ± 383.49 | 189.45 ± 31.95 | 204.31 ± 51.08 | 0.312 |
| LDL-C (mg/dL) | 114.74 ± 30.58 | 113.56 ± 31.07 | 118.60 ± 43.79 | 0.87 |
| HDL-C (mg/dL) | 40.92 ± 12.01 | 41.49 ± 9.21 | 45.92 ± 12.16 | 0.301 |
| Systolic blood pressure (mmHg) | 110.46 ± 10.51 | 110.87 ± 10.75 | 120.69 ± 10.48 | 0.011 |
| Diastolic blood pressure (mmHg) | 70.48 ± 10.13 | 70.83 ± 10.30 | 80.08 ± 10.08 | 0.04 |
| Abdominal obese (WC > 90 cm) (%) | 59.1 | 63.5 | 92.3 | 0.058 |
| Women | ||||
| Age (yr) | 41.70 ± 5.92 | 42.81 ± 6.21 | 42.94 ± 5.86 | 0.059 |
| Energy intake (kcal/d) | 1738.47 ± 488.91 | 1674.05 ± 491.68 | 1715.73 ± 447.60 | 0.643 |
| Weight (kg) | 70.05 ± 11.11 | 71.17 ± 11.30 | 77.06 ± 13.36 | 0.002 |
| Height (cm) | 156.52 ± 5.12 | 156.96 ± 5.25 | 156.27 ± 6.59 | 0.43 |
| Body mass index (kg/m2) | 28.58 ± 4.26 | 29.23 ± 4.19 | 31.47 ± 4.45 | < 0.0001 |
| Waist circumference (cm) | 85.10 ± 8.88 | 87.00 ± 9.16 | 91.03 ± 9.02 | < 0.0001 |
| Hip circumference (cm) | 107.37 ± 8.79 | 108.68 ± 8.79 | 111.21 ± 9.94 | 0.02 |
| Waist to hip ratio | 0.79 ± 0.05 | 0.80 ± 0.05 | 0.82 ± 0.03 | 0.018 |
| Fasting blood sugar | 86.87 ± 7.91 | 88.89 ± 7.05 | 90.41 ± 6.71 | 0.001 |
| Blood sugar after 30 min (mg/dL) | 125.21 ± 23.32 | 134.55 ± 25.26 | 140.42 ± 23.59 | < 0.0001 |
| Blood sugar after 60 min (mg/dL) | 120.96 ± 30.41 | 136.16 ± 29.46 | 153.38 ± 33.60 | < 0.0001 |
| Blood sugar after 120 min (mg/dL) | 100.58 ± 19.45 | 109.63 ± 18.99 | 111.26 ± 18.55 | < 0.0001 |
| HbA1c (%) | 4.94 ± 0.82 | 5.08 ± 0.68 | 5.18 ± 0.69 | 0.05 |
| Triglyceride (mg/dL) | 140.11 ± 66.06 | 148.32 ± 79.00 | 193.34 ± 141.41 | < 0.0001 |
| Total cholesterol (mg/dL) | 190.74 ± 39.60 | 193.63 ± 37.41 | 206.00 ± 51.16 | 0.092 |
| LDL-C (mg/dL) | 117.04 ± 34.27 | 118.31 ± 32.67 | 125.64 ± 56.94 | 0.44 |
| HDL-C (mg/dL) | 46.48 ± 11.34 | 45.99 ± 10.45 | 44.77 ± 11.48 | 0.658 |
| Systolic blood pressure (mmHg) | 110.15 ± 10.48 | 110.67 ± 10.63 | 110.40 ± 10.58 | < 0.0001 |
| Diastolic blood pressure (mmHg) | 70.33 ± 10.10 | 70.56 ± 10.15 | 70.48 ± 10.31 | 0.044 |
| Abdominal obese (WC > 90 cm) (%) | 24.1 | 33.3 | 44.1 | 0.004 |
There were 45 incident cases of physician-diagnosed diabetic patients during follow up from 2003 to 2013. Overall, there was a positive link between BMI and WC in crude and all adjusted models (Table 2). After controlling for various confounders and mediators, relative risk for diabetes increased by 38% for 1 s.d. increase in BMI (95%CI: 1.05-1.82, P = 0.019). One s.d. increase in WC was associated with 20% higher risk for developing diabetes (95%CI: 1.04-1.38, P = 0.010), after controlling for potential confounders and mediators. In men, 1 s.d. increase either in BMI or in WC could not significantly affect the risk of prediabetes and diabetes; however, in women, 1 s.d. increase in both BMI and WC were associated with higher risk of diabetes, but not prediabetes. In the full adjusted model, 1 s.d increase in BMI and WC increased risk of diabetes by 51% (95%CI: 1.06-2.14) and 22% (95%CI: 1.03-1.45) in women, respectively.
| Total | Men | Women | ||||
| Pre-diabetes | Diabetes | Pre-diabetes | Diabetes | Pre-diabetes | Diabetes | |
| BMI | ||||||
| Crude | 1.03 (1.00, 1.07) | 1.14 (1.07, 1.21) | 1.04 (0.97, 1.13) | 1.14 (0.97, 1.34) | 1.04 (1.00, 108) | 1.15 (1.07, 1.23) |
| Model 1 | 1.04 (1.00, 1.07) | 1.14 (1.06, 1.22) | 1.14 (0.97, 1.35) | 1.04 (0.97, 1.12) | 1.03 (0.99, 1.07) | 1.14 (1.06, 1.23) |
| Model 2 | 1.06 (0.98, 1.15) | 1.24 (1.06, 1.46) | 1.06 (0.80, 1.40) | 1.30 (0.75, 2.26) | 1.07 (0.98, 1.17) | 1.25 (1.02, 1.53) |
| Model 3 | 1.05 (0.96, 1.14) | 1.36 (1.05, 1.77) | 1.00 (0.75, 1.35) | 1.44 (0.42, 4.88) | 1.07 (0.97, 1.17) | 1.44 (1.05, 1.98) |
| Model 4 | 1.04 (0.96, 1.14) | 1.38 (1.05, 1.82) | 1.00 (0.75, 1.35) | Inestimable | 1.07 (0.97, 1.18) | 1.51 (1.06, 2.14) |
| WC | ||||||
| Crude | 1.02 (1.00, 1.03) | 1.06 (1.03, 1.09) | 1.01 (0.98, 1.03) | 1.06 (1.00, 1.13) | 1.02 (1.01, 1.04) | 1.07 (1.03, 1.11) |
| Model 1 | 1.02 (1.00, 1.03) | 1.07 (1.03, 1.10) | 1.01 (0.98, 1.03) | 1.06 (1.00, 1.13) | 1.02 (1.00, 1.04) | 1.07 (1.03, 1.11) |
| Model 2 | 1.04 (1.00, 1.08) | 1.16 (1.06, 1.27) | 1.00 (0.90, 1.12) | 1.08 (0.87, 1.33) | 1.05 (1.01, 1.09) | 1.20 (1.07, 1.35) |
| Model 3 | 1.03 (0.99, 1.07) | 1.20 (1.04, 1.38) | 0.97 (0.85, 1.09) | 1.58 (0.37, 6.76) | 1.04 (1.00, 1.09) | 1.21 (1.03, 1.42) |
| Model 4 | 1.03 (0.99, 1.07) | 1.20 (1.04, 1.38) | 0.97 (0.86, 1.09) | Inestimable | 1.04 (1.00, 1.09) | 1.22 (1.03, 1.45) |
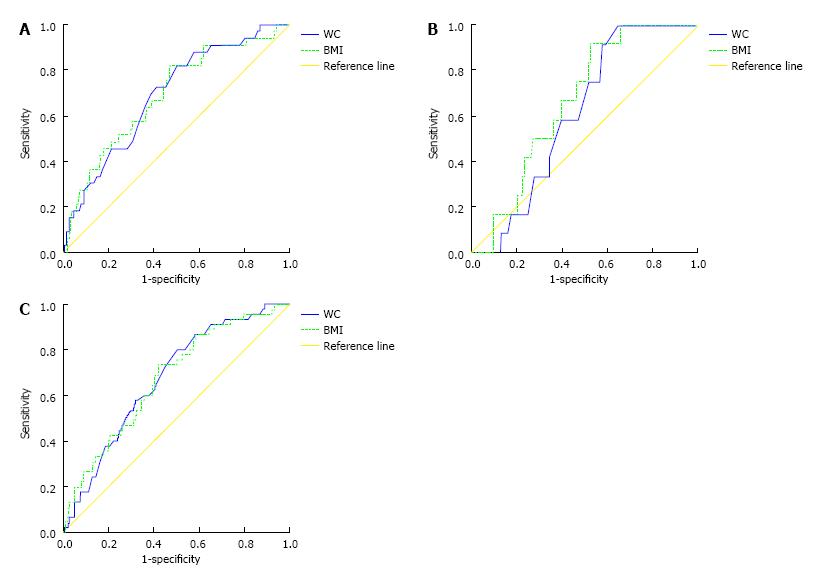
The AUCs (and 95%CIs) of BMI and WC in the prediction of pre-diabetes and diabetes are shown in Table 3. AUCs for both measures were larger for diabetes rather than pre-diabetes. As can be seen the significant predictive roles were detected for both BMI and WC on predicting diabetes while positive but not significant for prediabetes. Nevertheless, AUC of WC did not differ substantially from AUC of BMI either for pre-diabetes or for diabetes in the whole population. When analyses were run for men and women separately, similar results were obtained. Figure 1 supports the similar predictive powers of BMI and WC in predicting the incidence risk of diabetes in the whole population and either gender.
| Total | Men | Women | ||||
| Pre-diabetes | Diabetes | Pre-diabetes | Diabetes | Pre-diabetes | Diabetes | |
| Body mass index | 0.541 (0.502, 0.581) | 0.673 (0.596, 0.749) | 0.538 (0.460, 0.617) | 0.664 (0.551, 0.778) | 0.544 (0.498, 0.590) | 0.691 (0.598, 0.784) |
| Waist circumference | 0.552 (0.513, 0.592) | 0.674 (0.602, 0.746) | 0.508 (0.432, 0.585) | 0.613 (0.505, 0.721) | 0.564 (0.518, 0.611) | 0.691 (0.604, 0.778) |
Table 4 indicates the optimal cutoff points for general obesity and abdominal adiposity to predict incidence of pre-diabetes and diabetes. Defined cut-off points based on maximum sensitivity plus specificity values suggested that in men, BMI of 26.2 kg/m2 and WC of 89.7 cm, and in women, BMI of 28.6 kg/m2 and WC of 84.3 cm would predict the incident risk of diabetes. In the whole population, BMI of 28.5 kg/m2 and WC of 86.25 and 86.75 cm had the highest maximum sensitivity plus specificity. The optimal cutoff points for BMI to predict pre-diabetes in the whole population, men and women were 28.3, 29.6, 28.3 kg/m2, respectively. Corresponding values for WC were 86.0, 89.7, 88.2 cm, respectively.
| Whole population | Men | Women | ||||
| BMI | WC | BMI | WC | BMI | WC | |
| Diabetes | ||||||
| Cutoff point 1 | 28.5 | 86.25 | 26.2 | 89.7 | 28.6 | 84.3 |
| Sensitivity | 0.733 | 0.787 | 0.917 | 1 | 0.818 | 0.818 |
| Specificity | 0.579 | 0.495 | 0.477 | 0.356 | 0.53 | 0.5 |
| Cutoff point 2 | 29 | 86.75 | 27 | 90 | 29 | 85 |
| Sensitivity | 0.6 | 0.766 | 0.75 | 0.917 | 0.667 | 0.758 |
| Specificity | 0.614 | 0.514 | 0.53 | 0.423 | 0.566 | 0.532 |
| Pre-diabetes | ||||||
| Cutoff point 1 | 28.3 | 86 | 29.6 | 89.7 | 28.3 | 88.2 |
| Sensitivity | 0.528 | 0.787 | 0.294 | 0.706 | 0.583 | 0.45 |
| Specificity | 0.559 | 0.495 | 0.819 | 0.356 | 0.516 | 0.686 |
| Cutoff point 2 | 29 | 87 | 30 | 90 | 29 | 89 |
| Sensitivity | 0.427 | 0.723 | 0.235 | 0.635 | 0.468 | 0.376 |
| Specificity | 0.614 | 0.453 | 0.826 | 0.409 | 0.564 | 0.718 |
In this prospective study, BMI was strongly associated with diabetes incidence in the whole population and women. WC was moderately related to diabetes incident in the whole population and women. These associations remained significant after controlling either for confounding variables or mediators. The associations of BMI and WC with incidence of diabetes in men were not significant, and in overall both BMI and WC were weakly correlated with pre-diabetes incidence.
Adjustment for mediators increased the risk of diabetes incidence which might be attributed to adiposity alone or adipocytokines or other unmeasured risk factors such as dietary intake, lifestyle, inflammatory factors and family history[22,23]. Effects of these variables on anthropometric measures have been well-established[24,25]. In the DECODA (Diabetes Epidemiology: Collaborative Analysis of Diagnostic criteria in Asia) study, BMI and WC were not differently associated with the incidence risk of diabetes in men, but in women, WC was stronger anthropometric predictor of diabetes than BMI[7]. A meta-analysis of the Asian cohorts suggested that BMI and WC were similarly related to incident of diabetes[26]; however, in European, WC is stronger predictor for developing diabetes than BMI[8]. Using prospective analyses, similar associations were found for BMI and WC to predict the progression of diabetes in an Iranian population[10]. Nevertheless, in the current analyses, we observed that BMI was a stronger predictor for the incident diabetes in women and the whole population. The discrepancies regarding BMI and WC relation with diabetes in the current analyses might reveal that the length of follow-up duration might be a relevant determinant of estimating incident risk. Regardless of the contradictory results on which of anthropometric measures is better, all studies indicated that both BMI and WC are directly associated with the incidence risk of diabetes. For pre-diabetes, in the whole population, cut points were similar to cut points of diabetes. In men, in spite of similar cut points for WC to predict pre-diabetes vs diabetes, there was considerable difference for BMI while greater BMI value was identified as the best cut point. In contrast, in women, BMI cut point for predicting pre-diabetes was similar to the one for diabetes, but WC cut point was considerably higher. This finding confirms that WC in women and BMI in men are better index for predicting pre-diabetes as well as for diabetes.
To determine appropriate cutoff points for anthropometric measures, some variables such as age and sex need to be taken into account. In men and older age adults, higher cut-off points are more suitable. However, in the current study, we determined cut-off points only based on sex since majority of participated subjects at baseline were younger than 60 years which is defined as the age of elderly[27].
In this study, there was no difference in the overall predictive discrimination (as determined by AUC) of BMI and WC in either gender, that is in line with other studies[28,29]. Yoon et al[29] indicated that BMI and WC have similar predictive power for insulin resistance and diabetes among Korean adults. Another population-based cross-sectional study on Iranian men and women aged 20-80 years found no difference in the predictive power of BMI and WC for diabetes[28]. Nevertheless, Johnston Alperet et al[30] revealed that central obesity measures (WC and waist to height ratio) are better than BMI for the diagnosis of uninvestigated diabetes mellitus in three major Asian ethnic groups (Chinese, Malays, and Asian-Indians).
Available evidences to determine suitable cut-off points for WC and BMI have been obtained from cross-sectional studies[12,13,16]. To the best of our knowledge, there is only one prospective cohort study to predict appropriate cut-off points for diabetes among Iranians[10]. Difference in study design may lead to inconsistency regarding the determined clinically relevant cut-off points in different studies. Moreover, the follow-up duration and the sample size of study may influence these cut points in studies with similar design. Our study suggested that in the whole population, the BMI cut points of 28.5 kg/m2 and 86 cm for WC yielded the maximum sensitivity plus specificity for predicting the presence of diabetes. Corresponding values in men were 26.2 kg/m2 and 89.7 cm, and in women were 28.6 kg/m2 and 84.3 cm, respectively. Generally, women had higher values of BMI but lower values of WC cut points; and this means that in women, central obesity performed better than BMI to predict diabetes risk, whilst in men BMI perform better. In this analysis, ROC analysis was run to identify cut points. It should be taken into account that ROC method is dependent on the distribution of anthropometric measures in the study population. On the other hand, increasing mean values of anthropometrics by corresponding higher distributions would automatically increase derived cut points by the ROC analysis[31]. Therefore, higher cut points of WC in men and BMI in women could be explained by the higher mean values of WC and BMI in men and women, respectively. Moreover, ROC method is equally weighted for sensitivity and specificity[32]. This might lead to low sensitivity for anthropometric measures to predict the incidence risk of diabetes in clinical practice. Furthermore, defined cut points in our study could not be optimal points in clinic, since sensitivity vs specificity need to be weighed against other factors such as seriousness of the complaint, the applied test for evaluation (whether it is invasive or feasible) and how often the test must be done[33]. Furthermore, due to high prevalence of diabetes in Iran[25], it is relevant to identify a sensible proportion of the population at risk. Our defined cut-off points’ sensitivity are higher than 80% that means only 20% diabetic subjects would be missed by these cut points. However, for pre-diabetes sensitivity is very low.
This study has several limitations that should be taken into account. The main limitation is few numbers of cases with diabetes that decrease the statistical power of analyses. Furthermore, our study population was not a representative sample of Iranians and therefore more studies are needed to confirm whether our findings are generalizable to other Iranian populations. In addition, a recent research has shown that non-alcoholic fatty liver disease (NAFLD) might be a new criterion for metabolic syndrome[34]. Regarding the high prevalence of NAFLD among Iranians[35] and due to its close relation with insulin resistance, further studies are needed to determine the suitable cut points for BMI and WC for predicting the NAFLD incidence among Iranians. Nevertheless, this study has some strength. Using measured anthropometric variables, not self-reported values, in a large sample of men and women with very reliable data are the main strengths of this study. Furthermore, confounding effects of various confounders and mediators were taken into account in data analyses. Finally, based on our prospective study design an association between fat accumulation and diabetes mellitus could be concluded.
In conclusion, we observed that the predictive powers of BMI and WC were similar in predicting the incidence risk of diabetes in either gender. The cut-off points for predicting diabetes in men and women were different. Defined cut-off points based on maximum sensitivity plus specificity values suggested that in men, BMI of 26.2 kg/m2 and WC of 89.7 cm, and in women, BMI of 28.6 kg/m2 and WC of 84.3 cm would predict Isfahanian population at high risk for developing diabetes.
The increased prevalence of obesity in the world is a major concern as it is strongly related to multiple metabolic disorders, among them diabetes. Therefore, it is essential to identify the best anthropometric index and effective risk thresholds for adiposity measures to develop appropriate preventive strategies in each population.
Current study aimed to prospectively determine the effective anthropometric measures to predict the risk of type 2 diabetes and prediabetes also estimate the optimal cut-off point of body mass index (BMI) and waist circumference (WC) by following up non-diabetic participants at baseline examination.
To the best of our knowledge, there is only one longitudinal study among Iranians which has determined cut-off point of WC for detecting cardiovascular disease risk and no study on BMI threshold, and other cut-off points come from cross-sectional surveys, on the other hand no study available on determining the best cut of values for prediabetes. These values are strengthening by the authors longitudinal study design.
Estimated cut-off points by the study will contribute to detect individuals at higher risk of developing type 2 diabetes as well as prediabetes.
BMI and WC’s best cut of values for predicting diabetes and prediabetes.
The article is an important epidemiological study in which the authors studied a cohort of 1765 healthy participants followed up from 2003 till 2013. The study is well conducted, with appropriate statistics methodology and the results, previously known, confirm the relationship between anthropometric data and diabetes mellitus.
Manuscript source: Invited manuscript
Specialty type: Endocrinology and metabolism
Country of origin: Iran
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Gómez-Sáez JM, Tarantino G, Xie ZJ S- Editor: Kong JX L- Editor: A E- Editor: Lu YJ
| 1. | World Health Organization. Obesity and overweight. Fact sheet 311. [accessed 2016 Aug 13]. Available from: http://www.who.int/mediacentre/factsheets/fs311/en/. |
| 2. | Guh DP, Zhang W, Bansback N, Amarsi Z, Birmingham CL, Anis AH. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health. 2009;9:88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2406] [Cited by in RCA: 2538] [Article Influence: 158.6] [Reference Citation Analysis (0)] |
| 3. | Jackson AS, Stanforth PR, Gagnon J, Rankinen T, Leon AS, Rao DC, Skinner JS, Bouchard C, Wilmore JH. The effect of sex, age and race on estimating percentage body fat from body mass index: The Heritage Family Study. Int J Obes Relat Metab Disord. 2002;26:789-796. [PubMed] |
| 4. | Nevill AM, Stewart AD, Olds T, Holder R. Relationship between adiposity and body size reveals limitations of BMI. Am J Phys Anthropol. 2006;129:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 251] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 5. | Heymsfield SB, Scherzer R, Pietrobelli A, Lewis CE, Grunfeld C. Body mass index as a phenotypic expression of adiposity: quantitative contribution of muscularity in a population-based sample. Int J Obes (Lond). 2009;33:1363-1373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 115] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 6. | Cornier MA, Després JP, Davis N, Grossniklaus DA, Klein S, Lamarche B, Lopez-Jimenez F, Rao G, St-Onge MP, Towfighi A. Assessing adiposity: a scientific statement from the American Heart Association. Circulation. 2011;124:1996-2019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 535] [Cited by in RCA: 652] [Article Influence: 46.6] [Reference Citation Analysis (0)] |
| 7. | Nyamdorj R, Qiao Q, Lam TH, Tuomilehto J, Ho SY, Pitkäniemi J, Nakagami T, Mohan V, Janus ED, Ferreira SR. BMI compared with central obesity indicators in relation to diabetes and hypertension in Asians. Obesity (Silver Spring). 2008;16:1622-1635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 165] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 8. | Langenberg C, Sharp SJ, Schulze MB, Rolandsson O, Overvad K, Forouhi NG, Spranger J, Drogan D, Huerta JM, Arriola L. Long-term risk of incident type 2 diabetes and measures of overall and regional obesity: the EPIC-InterAct case-cohort study. PLoS Med. 2012;9:e1001230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 137] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 9. | Oda E, Kawai R. Comparison among body mass index (BMI), waist circumference (WC), and percent body fat (%BF) as anthropometric markers for the clustering of metabolic risk factors in Japanese. Intern Med. 2010;49:1477-1482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Janghorbani M, Amini M. Comparison of body mass index with abdominal obesity indicators and waist-to-stature ratio for prediction of type 2 diabetes: The Isfahan diabetes prevention study. Obes Res Clin Pract. 2010;4:e1-e82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Alberti KG, Zimmet P, Shaw J. Metabolic syndrome--a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet Med. 2006;23:469-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3852] [Cited by in RCA: 4248] [Article Influence: 223.6] [Reference Citation Analysis (0)] |
| 12. | Delavari A, Forouzanfar MH, Alikhani S, Sharifian A, Kelishadi R. First nationwide study of the prevalence of the metabolic syndrome and optimal cutoff points of waist circumference in the Middle East: the national survey of risk factors for noncommunicable diseases of Iran. Diabetes Care. 2009;32:1092-1097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 285] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 13. | Esteghamati A, Ashraf H, Rashidi A, Meysamie A. Waist circumference cut-off points for the diagnosis of metabolic syndrome in Iranian adults. Diabetes Res Clin Pract. 2008;82:104-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 14. | Gozashti MH, Najmeasadat F, Mohadeseh S, Najafipour H. Determination of most suitable cut off point of waist circumference for diagnosis of metabolic syndrome in Kerman. Diabetes Metab Syndr. 2014;8:8-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 15. | Hadaegh F, Zabetian A, Sarbakhsh P, Khalili D, James WP, Azizi F. Appropriate cutoff values of anthropometric variables to predict cardiovascular outcomes: 7.6 years follow-up in an Iranian population. Int J Obes (Lond). 2009;33:1437-1445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 99] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 16. | Shabnam AA, Homa K, Reza MT, Bagher L, Hossein FM, Hamidreza A. Cut-off points of waist circumference and body mass index for detecting diabetes, hypercholesterolemia and hypertension according to National Non-Communicable Disease Risk Factors Surveillance in Iran. Arch Med Sci. 2012;8:614-621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | American Diabetes Association. Executive summary: Standards of medical care in diabetes--2014. Diabetes Care. 2014;37 Suppl 1:S5-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 154] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 18. | Amini M, Janghorbani M. Diabetes and impaired glucose regulation in first-degree relatives of patients with type 2 diabetes in isfahan, iran: prevalence and risk factors. Rev Diabet Stud. 2007;4:169-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 50] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 19. | World Health Organization. Regional Office for Europe, & WHO Consultation on the Epidemiology of Obesity 1987. (1989). Measuring obesity-classification and description of anthropometric data: Report on a WHO Consultation on the Epidemiology of Obesity Warsaw, 21-23, October 1987. Copenhagen: Copenhagen: WHO 1989; . |
| 20. | Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499-502. [PubMed] |
| 22. | Sakurai M, Nakamura K, Miura K, Takamura T, Yoshita K, Sasaki S, Nagasawa SY, Morikawa Y, Ishizaki M, Kido T. Family history of diabetes, lifestyle factors, and the 7-year incident risk of type 2 diabetes mellitus in middle-aged Japanese men and women. J Diabetes Investig. 2013;4:261-268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 23. | Ley SH, Ardisson Korat AV, Sun Q, Tobias DK, Zhang C, Qi L, Willett WC, Manson JE, Hu FB. Contribution of the Nurses’ Health Studies to Uncovering Risk Factors for Type 2 Diabetes: Diet, Lifestyle, Biomarkers, and Genetics. Am J Public Health. 2016;106:1624-1630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 24. | Marangoni F, Brignoli O, Cricelli C, Poli A. Lifestyle and specific dietary habits in the Italian population: focus on sugar intake and association with anthropometric parameters-the LIZ (Liquidi e Zuccheri nella popolazione Italiana) study. Eur J Nutr. 2017;56:1685-1691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 25. | Maracy MR, Feizi A, Bagherynejad M. The prevalence and correlated determinants of hypertension and type 2 diabetes: A large community-based study in Isfahan, Iran. Pak J Med Sci. 2012;28:247-252. |
| 26. | Vazquez G, Duval S, Jacobs DR, Silventoinen K. Comparison of body mass index, waist circumference, and waist/hip ratio in predicting incident diabetes: a meta-analysis. Epidemiol Rev. 2007;29:115-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 579] [Cited by in RCA: 646] [Article Influence: 35.9] [Reference Citation Analysis (0)] |
| 27. | World Health Organization. Proposed working definition of an older person in Africa for the MDS Project. [accessed 2016 Sep 13]. Available from: http://www.who.int/healthinfo/survey/ageingdefnolder/en/. |
| 28. | Hajian-Tilaki K, Heidari B. Is waist circumference a better predictor of diabetes than body mass index or waist-to-height ratio in Iranian adults? Int J Prev Med. 2015;6:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 29. | Yoon YS, Choi HS, Kim JK, Kim YI, Oh SW. Differences in the associations of anthropometric measures with insulin resistance and type 2 diabetes mellitus between Korean and US populations: Comparisons of representative nationwide sample data. Obes Res Clin Pract. 2015;10:642-651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 30. | Johnston Alperet D, Lim WY, MokKwee Heng D, Ma S, van Dam RM. Optimal anthropometric measures and thresholds to identify undiagnosed type 2 diabetes in three major Asian ethnic groups. Obesity (Silver Spring). 2016;24:2185-2193. [RCA] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 31. | Razak F, Anand SS, Shannon H, Vuksan V, Davis B, Jacobs R, Teo KK, McQueen M, Yusuf S. Defining obesity cut points in a multiethnic population. Circulation. 2007;115:2111-2118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 369] [Cited by in RCA: 377] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 32. | Perkins NJ, Schisterman EF. The inconsistency of “optimal” cutpoints obtained using two criteria based on the receiver operating characteristic curve. Am J Epidemiol. 2006;163:670-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1048] [Cited by in RCA: 1287] [Article Influence: 67.7] [Reference Citation Analysis (0)] |
| 33. | Glümer C, Carstensen B, Sandbaek A, Lauritzen T, Jørgensen T, Borch-Johnsen K. A Danish diabetes risk score for targeted screening: the Inter99 study. Diabetes Care. 2004;27:727-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 234] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 34. | Tarantino G, Finelli C. What about non-alcoholic fatty liver disease as a new criterion to define metabolic syndrome? World J Gastroenterol. 2013;19:3375-3384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 111] [Cited by in RCA: 134] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 35. | Zolfaghari H, Askari G, Siassi F, Feizi A, Sotoudeh G. Intake of Nutrients, Fiber, and Sugar in Patients with Nonalcoholic Fatty Liver Disease in Comparison to Healthy Individuals. Int J Prev Med. 2016;7:98. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (0)] |