Published online Nov 15, 2022. doi: 10.4239/wjd.v13.i11.962
Peer-review started: September 14, 2022
First decision: October 5, 2022
Revised: October 22, 2022
Accepted: October 31, 2022
Article in press: October 31, 2022
Published online: November 15, 2022
Processing time: 57 Days and 12.7 Hours
The adverse consequences resulting from diabetes are often presented as severe complications. Diabetic wounds are one of the most commonly occurring complications in diabetes, and the control and treatment of this is costly. Due to a series of pathophysiological mechanisms, diabetic wounds remain in the inflammatory phase for a prolonged period of time, and face difficulty in entering the proliferative phase, thus leading to chronic non-healing wounds. The current consensus on the treatment of diabetic wounds is through multidisciplinary comprehensive management, however, standard wound treatment methods are still limited and therefore, more effective methods are required. In recent years, defensins have been found to play diverse roles in a variety of diseases; however, the molecular mechanisms underlying these activities are still largely unknown. Defensins can be constitutively or inductively produced in the skin, therefore, their local distribution is affected by the microenvironment of these diabetic wounds. Current evidence suggests that defensins are involved in the diabetic wound pathogenesis, and can potentially promote the early completion of each stage, thus making research on defensins a promising area for developing novel treatments for diabetic wounds. In this review, we describe the complex function of human defensins in the development of diabetic wounds, and suggest potential thera-peutic benefits.
Core Tip: Although previous studies have suggested that defensins have a function in the promotion of wound healing, their mechanism is still unclear. In this review, we discuss the potential role of various defensins in refractory diabetic wounds and their properties, including immunoregulation, promotion of re-epithelialization, collagen deposition, vascular regeneration, and neurological recovery, as well as antimicrobial activity.
- Citation: Tan ZX, Tao R, Li SC, Shen BZ, Meng LX, Zhu ZY. Role of defensins in diabetic wound healing. World J Diabetes 2022; 13(11): 962-971
- URL: https://www.wjgnet.com/1948-9358/full/v13/i11/962.htm
- DOI: https://dx.doi.org/10.4239/wjd.v13.i11.962
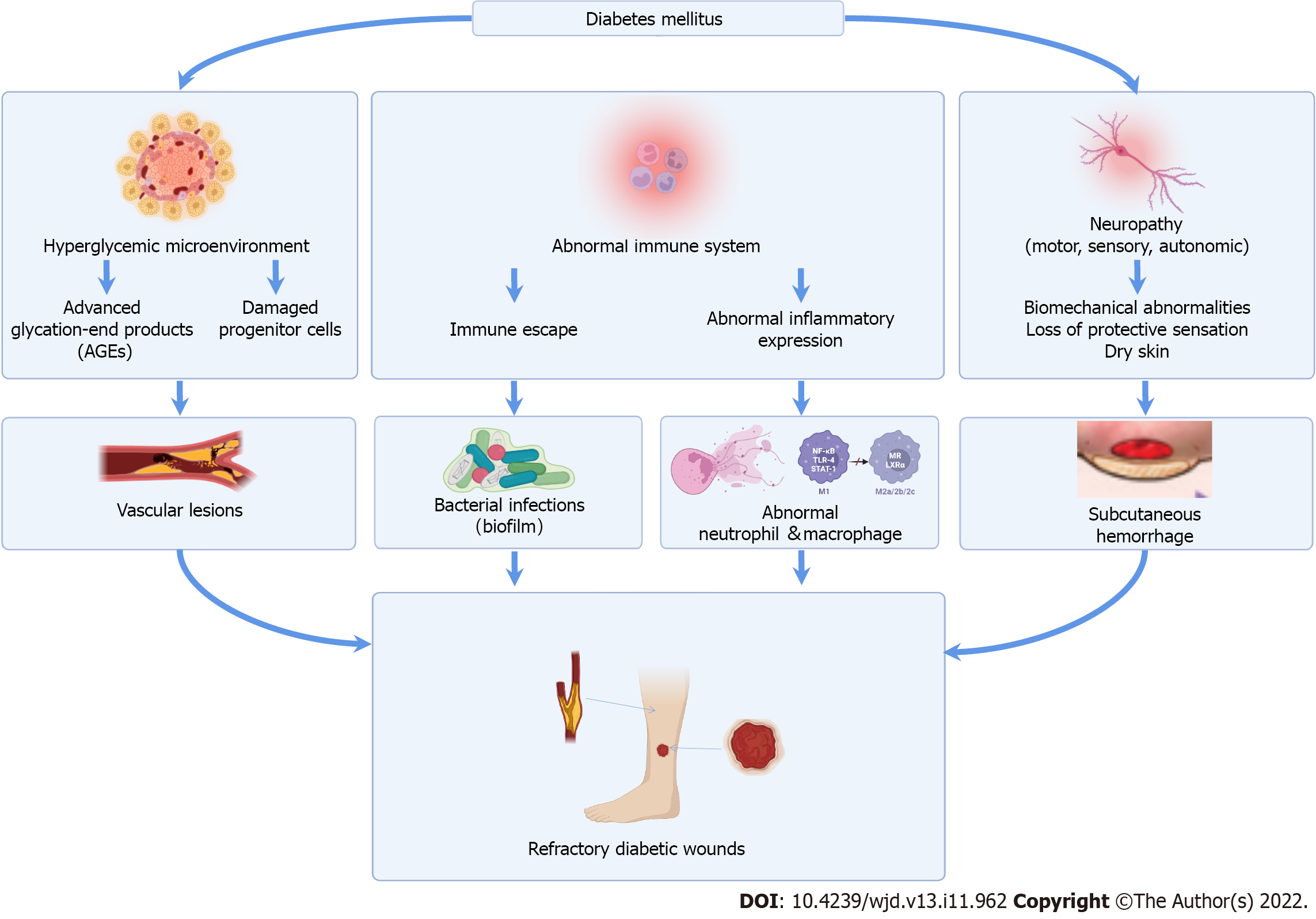
Diabetes was estimated to affect over 536.6 million people worldwide in 2021, and an increase in prevalence occurs at a faster rate among middle-income countries[1]. Diabetes mellitus gradually causes a series of complications, such as neuropathy, retinopathy, nephropathy, cardiovascular diseases, and diabetic wounds, as it develops. Due to lesions in the small blood vessels and peripheral nerves that are particularly prominent in the feet, diabetic wounds are usually presented as diabetic foot ulcers (DFUs), and are characterized by a delayed tissue growth and increased susceptibility to infection. Patients with DFUs have an increased risk of lower limb amputation, which has poor short-term prognosis associated with high mortality[2]. Statistically, the five-year mortality and direct costs of care for patients with DFUs have been comparable to that of cancer[3]. The large patient population and medical expenditure urgently requires an effective treatment method.
The mechanisms for diabetic wound development involve multifactorial etiologies, including a hyperglycemic microenvironment, abnormal host immune resistance, and neuropathy (Figure 1)[4]. These mechanisms influence each other, instead of occurring independently, causing irreversible diabetic complications. The local damage to vessels and nerves, reduced growth factors expression, and lower collagen accumulation contribute to repeated outbreaks and the protracted course of diabetic wounds, leading to further infections. Although current treatments, including glycemic control, anti-infective treatment, and advanced dressing application, promote wound healing by regulating the local microenvironment, they also have disadvantages, such as protracted treatment periods, high costs, and occasionally inefficient results[5]. Hence, studying methods that promote diabetic wound healing is ongoing.
With concern about antibiotic resistance growing more prominent, antimicrobial peptides (AMPs) have garnered attention as a new method of antibacterial therapy, including development of different formulation strategies for effective delivery to wounds, including AMPs loaded in nanoparticles, hydrogels, creams, gels, etc. As a representative AMP, the defensins properties are gradually being researched (Table 1)[6-23]. Human defensins are divided into α-defensins and β-defensins[23]. Human α-defensins mainly occur in neutrophils (human neutrophil peptide1-4, HNP1-4) or small intestinal Paneth cells (human defensin5-6, HD5-6). More extensively, 31 human β-defensins (HBDs) have been described, and HBD1-4 is most widely-studied[24]. Reportedly, the direct primary role of defensins is controlling microbial infections by killing bacteria and modulating the immune system. Moreover, defensins play different roles in different environments within the body, such as infected wounds, malignancy, atherosclerosis, pulmonary fibrosis, etc. In this review, we focused on investigating their mechanism of action in wound healing, especially chronic diabetic wounds.
| Defensin | Main cellular source | Action | ||
| α-defensin | HNP1 | Neutrophils; monocytes; macrophages; natural killer cells | Increase the healing rate of MRSA-infected wounds[6]; promote hemostasis[7]; r/affect the cardiovascular system[8]; inhibit thrombus formation[9] | Anti-infection and immunoregulation[23] |
| HNP2-3 | Anti-tumor activity[10] | |||
| HNP4 | Neutrophils | Characterize benign and malignant salivary gland tumors[11] | ||
| HD5-6 | Intestinal Paneth cells | Reverse dyslipidemia and improve glucoregulatory capacity[12]; anti-tumor ability[13]; amyloid inhibitor[14] | ||
| β-defensin | HBD1 | Epithelial cells; monocytes; macrophages | Anti-tumor activity[15]; potentiate osteoclastogenesis[16] | Induce the secretion of angiogenin[54]; anti-infection and immunoregulation[23] |
| HBD2 | Accelerate wound healing[17]; Oncolytic activity[18]; reduce alcoholic liver injury[19] | |||
| HBD3 | Accelerate wound healing[20]; induce IL-8 release and apoptosis in airway smooth muscle cells[21] | |||
| HBD4 | Epithelial cells | Stimulate/suppress cancer cell proliferation and viability[22] | ||
Although little is known about defensins involvement in diabetic wounds, existing studies indicate that they play potential roles in complex pathophysiological changes of diabetic wounds[25,26]. This review summarizes and analyzes known experimental data about the role of defensins in diabetic wound healing, particularly for inflammation, cell proliferation and migration, regeneration of blood vessels and nerves, and antibacterial activities. Research articles on the role of defensins in diabetic wounds, published between inception and September 10, 2022, were collected from various search engines, such as PubMed, Google Scholar, Web of Science, and Science Direct using the following keywords: AMPs, defensins, host defense peptides, diabetic, refractory, and chronic wounds, wound healing, etc. Identified studies and relevant citations within these studies were reviewed.
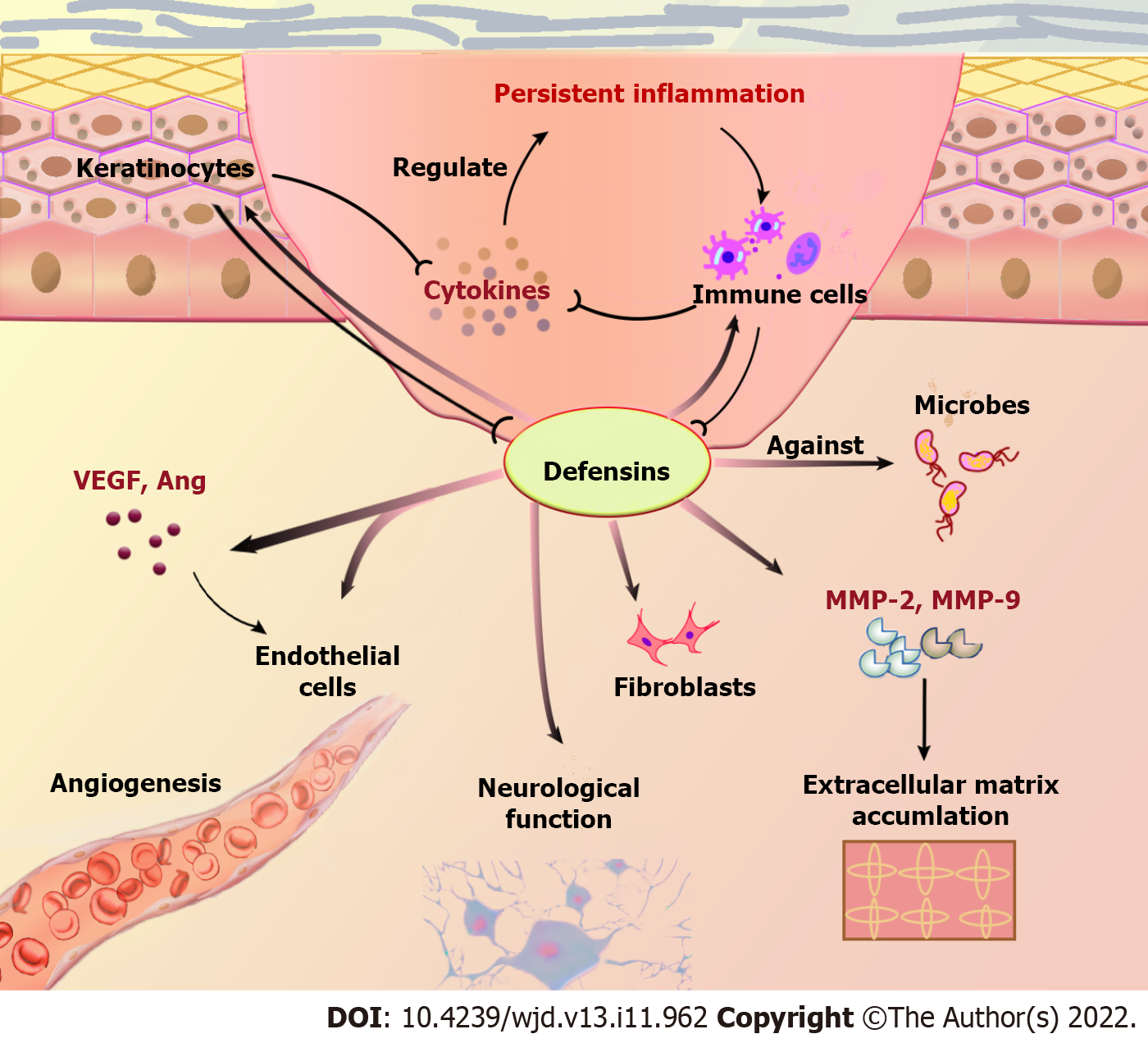
The response to tissue injury involves multiple cellular and extracellular events, including inflammation, re-epithelialization, and angiogenesis, followed by fibroplasia with collagen synthesis, and tissue remodeling. Defensins may be a multifactorial modulator in the management of this process, which interferes in diabetic wounds (Table 2, Figure 2).
| Stage | Defensin | Activation |
| Inflammation | HNP1-2, HBD1-3[28] | Recruitment of leukocytes |
| HNP1-4[29] | Secretion of inflammatory cytokines like IL-8 | |
| HBD2-4[32] | Activation of the p38 and ERK1/2 MAPK pathways | |
| HNP1, HBD2[35] | Activation of the p42/44 MAPK pathways | |
| HBD2, HBD3[36] | Down-regulate the TIR, TRAF-6, NF-κB of TLR signaling pathways | |
| HBD3[37] | Induce M2 macrophage differentiation | |
| Re-epithelialization | HNP1[43], HBD2-3[42] | Induce keratinocyte migration and proliferation |
| HBD1[44] | Protect keratinocytes from apoptosis | |
| Collagen synthesis | HNP1[45] | Enhance extracellular matrix deposition |
| HBD3[20,37] | Increase the expression of MMP-2, and down-regulate the expression of MMP-9 | |
| Fibroplasia | HNP1[45], HBD2[46] | Promote the proliferation and activation of fibroblasts |
| Angiogenesis | HNP1[51], HBD2[52], HBD3[53] | Induce VEGF |
| HBD1-4[54] | Induce angiogenin | |
| HNPs[55] | Inhibit adhesion and migration of endothelial cell | |
| Nerve reconstruction | HNP1[40] | Promote the recovery of neurological function |
| HBD3[57] | Modulate the expression of nerve elongation factors | |
| Antimicrobial activity | HNP1-4, HBD1-4[61] | Exhibit a broad range of antimicrobial properties |
| HBD2[60] | Reduce biofilm formation | |
| HNP1-3[62] | Neutralize bacterial toxins | |
| HNP1, HBD1, HBD3[63] | Show synergy of action with antibiotics |
The first phase of wound healing is the inflammatory phase, characterized by platelet aggregation and leukocytes migration, including neutrophils and macrophages that secrete defensins and consequently clear the wound area[27]. Studies suggest defensins promote recruitment and accumulation of leukocytes at inflammatory sites, and simultaneously release a series of chemokines[28,29]. In diabetic wounds, the number of neutrophils increases abnormally and macrophage polarization is suppressed, leading to an excessive inflammatory expression[30,31]. As defensins are released in response to inflammation from neutrophils and macrophages, which act as a signal to instigate recruitment of immune cells, a positive-feedback loop is created. HBDs can reportedly participate in degranulation of mast cells and induce secretion of proinflammatory factors by keratinocytes via the p38 and ERK1/2 MAPK pathways activation[32,33]. Through the same action sites, HNPs produce vasoactive by-products in endothelial cells via ROS-dependent mechanisms, and stimulate the increased expression of IL-6 and IL-8 by activating p42/44 MAPK pathways[34,35].
In contrast, studies investigating associations between defensins and inflammatory mediators exhibited controversial results. HBDs have demonstrated an immunosuppressive effect by down-regulating the TIR, TRAF-6, and NF-κB of TLR signaling pathways[36]. Moreover, HBDs contribute to their anti-inflammatory ability by inducing M2 macrophage differentiation[37]. Previous experiments established that HBDs can be beneficial in inflammatory diseases, such as periodontitis, considering its anti-inflammatory properties[38]. A study on HNPs from dying neutrophils exhibited an immunosuppressive effect of the α-defensins that inhibited macrophage stimulation[39]. The HNP1 “bipolar effect” represents the reduction of inflammatory responses with a physiological dose, enhanced expression of inflammatory factors with a high dose, and significant reductions in cell viability and interleukin-10 expression with increased concentration levels[40]. Overall, defensins perform different functions under different conditions, including concentration levels[40]. Defensins are often used as disease-related markers as dysregulation of their levels is caused by immune system disorders and effectors produced themselves or through associated cells[41]. However, the causal relationship and sequence of cascades remain unclear. Several studies emphasized the relationship between delayed wound healing and uncontrolled inflammatory responses, and defensins as efficient adjustors playing a regulatory role in the process.
Failure to re-epithelialize is one of the most significant indicators for chronic wounds. Re-epithelialization is achieved through keratinocyte migration, proliferation, and differentiation. The HNP1, HBD2, HBD3, and HBD4 can reportedly induce proliferation and migration of keratinocytes, which can consequently secrete HBDs, thereby promoting reconstruction of the cellular barrier to accelerate wound healing[42,43]. Subsequent studies reported that HBD3 enhances phosphorylation of the FGFR1/JAK2/STAT3 pathways to promote keratinocyte proliferation and migration[20]. HBD1 potentially acts as a relevant transcription factor by protecting keratinocytes from apoptosis during epithelial reorganization[44]. In other words, defensins have properties that promote wound epithelialization by affecting keratinocyte activity, and thus facilitating early wound closure.
Furthermore, defensins seemingly play an important role in fibroblasts and collagen matrix accumulation, which is essential for dermal reconstitution. HNP1 can reportedly promote proliferation and activation of fibroblasts more effectively than HBD2 at the same concentration, and the increased collagen gene expression can only be observed by its stimulation[45]. A study also proved that HBDs indirectly stimulate fibroblast migration by activating protein kinase C[46]. High levels of pro-inflammatory cytokines and inflammatory chemokines in diabetic wounds lead to an increased production of matrix metalloproteinases (MMPs), especially MMP-2 and MMP-9, thereby inhibiting extracellular matrix formation and dermis reconstruction[47,48]. Studies have suggested that the use of an inhibitor for MMP-2 and MMP-9 accelerates wound healing in diabetic mice by maintaining the balance between systematic inflammation and cytokine biosynthesis[49]. HBD3 may potentially reverse the pathological condition as they have shown an inhibitory effect on MMP-9, which may result from cytotoxicity for dendritic cells in high concentrations[50]. Instead, HBD3 reportedly increases the expression of MMP-2, which is essential for angiogenesis and prolonged matrix remodeling[20]. To explain these contradictory findings, further clarification and a comprehensive analysis on the mechanism of wound healing is necessary, as well as verification through specific experiments.
Angiogenesis is a vital physiological process in wound healing and largely regulated by growth factors, specifically vascular endothelial growth factor (VEGF) and angiogenin. HNP1, HBD2, and HBD3 was proven to bind to cell surface receptor proteins, thus promoting VEGF expression and improvement of vascularization[51-53]. The novel role of HBDs in angiogenesis was also identified, revealing that HBD1-4 increases secretion of angiogenin dose-dependently[54]. However, the opposing actions can be described as a consequence of on-site recruitment of distinct subpopulations from circulation. HNPs can reportedly inhibit adhesion and migration of endothelial cells, and block VEGF-induced endothelial cell proliferation and capillary formation upon inflammatory stimulation[55]. These studies have shed a light on the mechanistic complexity of HNPs angiogenesis.
Neuropathy caused by diabetes is the influencing factor for subcutaneous hemorrhage underneath the callus formation, ultimately leading to skin breakdown[56]. Studies have proven that HNP1 administration can promote recovery of neurological function following sciatic nerve injury[40]. Additionally, HBD3 modulates the expression of nerve elongation factors that are involved in epidermal hyperinnervation and hypersensitivity to warm sensations[57]. As a result, application of defensins can help prevent delayed treatment due to peripheral neuropathy and difficulty in mastering wound conditions in patients with diabetic wounds.
Healing of refractory diabetic wounds is often associated with susceptibility to bacterial infections and formation of biofilms[58]. As a class of small cationic molecule peptides with broad-spectrum antimicrobial activity, defensins are produced to eliminate invading pathogens during the initial stages of wound formation[59]. While the important role of the pore-formation mechanism has been recognized in many studies, other mechanisms, such as disruption of cell wall synthesis, metabolic activity, ATP and nucleic acid synthesis, and amino acid uptake, have also been proposed in recent years[60]. HNPs and HBDs both exhibit a strong tendency to eliminate various pathogens, including Staphylococcus aureus and Escherichia coli, which often invades chronic wounds[61]. Specifically, HBD2 exhibits biofilm inhibitory activity by inducing structural changes that interfere with the biofilm precursor’s transport into the extracellular space[60]. Additionally, HNPs were proven to protect leukocytes from neutralization by gram-positive pathogenic bacterial toxins[62]. Furthermore, they can potentially avoid the emergence of resistance when implemented with other antimicrobial therapies[63]. Defensins are not only more effective against drug-resistant bacteria, as compared to antibiotics, but can also preserve the resident bacteria, despite the lack of target specificity as an intractable problem preventing their use as a therapeutic drug[64].
Although there is ambiguity regarding its exact role, refractory healing of diabetic wounds is speculated result from interactions between multiple pathophysiological changes in the microenvironment of hyperglycemic and persistent inflammation. This affects immune cell function and composition of defensins at the wound site. In human skin, HBD1 is constitutively expressed in epithelial cells, while inducible HNP1-4 by neutrophils and HBD2-3 by keratinocytes mainly[65]. It was obtained through a biopsy that HBD2-4 were overexpressed in the border area of DFUs[25]. Studies generally agree that inadequate HBD expression is associated with poor wound healing, and many methods that promote diabetic wound healing are seemingly carried out by promoting defensins expression[66,67]. In diabetic wounds, higher HNP1, HNP3, and HNP4 expressions are more common in the central part than in the marginal areas, thus causing a significant increase in IL-8 expression under the influence of advanced glycation end products (AGEs)[29].
Defensins affect the expression and secretion of cytokines, cell proliferation, migration, and apoptosis, and are also involved in all stages of wound healing. Contrary to its proven activity in fighting pathogens and promoting tissue reconstruction, the role of defensins in inflammation and vascularization remains unclear. This discrepancy could be due to pro-inflammatory and anti-inflammatory properties being attributed to HBDs at lower concentrations[28,29], compared to antibacterial effects at higher concentrations exhibited in different experiments[48]. Thus, effects of defensins may vary depending on concentration. Furthermore, HNP1 and HBD3 exhibit increased cytotoxic effects with the increased concentration, which can also be related to a greater hydrophobicity[43,68]. Therefore, changing the local distribution or structure of defensins can have beneficial effects and prevent toxic side effects.
Studying every type of defensins within a single experiment is difficult. Additionally, certain defensins can exhibit different or contradictory effects within the same environment due to differences in experimental complexity and aims of the experiment. These factors create a huge obstacle in horizontal comparison among similar experiments. Cytotoxicity caused by defensins is difficult to assess, which indicates that topical application may be more appropriate than the systemic administration. Considering the unstable biochemical properties of defensins, topical application alone may be insufficient. To overcome this limitation, researchers are studying biological dressings as an alternative; however, formulation of an ideal material has not yet been achieved. However, animal studies on defensins exhibit improved healing outcomes, and display stable effects through application of new materials or genetic engineering methods[17,20,69]. These findings present defensins as a promising therapeutic approach owing to modern techniques, such as development of new materials to efficiently load active factors and novel protein sequences to highlight their beneficial effects.
Defensins regulates chronic inflammation, tissue regeneration, angiogenesis, and nerve recovery, as well as antimicrobial properties; therefore, they are a promising treatment for diabetic wounds. There is an urgent need to find the appropriate dosing regimens and develop new biological dressing alternatives to incorporate active factors. Hence, further preclinical investigations are necessary to understand extensive molecular mechanisms of defensins in the treatment of diabetic wounds, and consequently determine suitable therapeutic strategies.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Engineering, biomedical
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Dziegielewska-Gesiak S, Poland; Salim A, Pakistan S-Editor: Chen YL L-Editor: A P-Editor: Chen YL
| 1. | Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3033] [Cited by in RCA: 4809] [Article Influence: 1603.0] [Reference Citation Analysis (36)] |
| 2. | Rathnayake A, Saboo A, Malabu UH, Falhammar H. Lower extremity amputations and long-term outcomes in diabetic foot ulcers: A systematic review. World J Diabetes. 2020;11:391-399. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (4)] |
| 3. | Armstrong DG, Swerdlow MA, Armstrong AA, Conte MS, Padula WV, Bus SA. Five year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J Foot Ankle Res. 2020;13:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 499] [Article Influence: 99.8] [Reference Citation Analysis (2)] |
| 4. | Pouget C, Dunyach-Remy C, Pantel A, Schuldiner S, Sotto A, Lavigne JP. Biofilms in Diabetic Foot Ulcers: Significance and Clinical Relevance. Microorganisms. 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 5. | Holl J, Kowalewski C, Zimek Z, Fiedor P, Kaminski A, Oldak T, Moniuszko M, Eljaszewicz A. Chronic Diabetic Wounds and Their Treatment with Skin Substitutes. Cells. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 171] [Article Influence: 42.8] [Reference Citation Analysis (0)] |
| 6. | Bolatchiev A, Baturin V, Bazikov I, Maltsev A, Kunitsina E. Effect of antimicrobial peptides HNP-1 and hBD-1 on Staphylococcus aureus strains in vitro and in vivo. Fundam Clin Pharmacol. 2020;34:102-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 7. | Kim KC, Lee W, Lee J, Cha HJ, Hwang BH. Newly Identified HNP-F from Human Neutrophil Peptide-1 Promotes Hemostasis. Biotechnol J. 2019;14:e1800606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Higazi M, Abdeen S, Abu-Fanne R, Heyman SN, Masarwy A, Bdeir K, Maraga E, Cines DB, Higazi AA. Opposing effects of HNP1 (α-defensin-1) on plasma cholesterol and atherogenesis. PLoS One. 2020;15:e0231582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 9. | McDaniel JK, Abdelgawwad MS, Hargett A, Renfrow MB, Bdeir K, Cao W, Cines DB, Zheng XL. Human neutrophil peptide-1 inhibits thrombus formation under arterial flow via its terminal free cysteine thiols. J Thromb Haemost. 2019;17:596-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Ferdowsi S, Pourfathollah AA, Amiri F, Rafiee MH, Aghaei A. Evaluation of anticancer activity of α-defensins purified from neutrophils trapped in leukoreduction filters. Life Sci. 2019;224:249-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Winter J, Pantelis A, Kraus D, Reckenbeil J, Reich R, Jepsen S, Fischer HP, Allam JP, Novak N, Wenghoefer M. Human α-defensin (DEFA) gene expression helps to characterise benign and malignant salivary gland tumours. BMC Cancer. 2012;12:465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Larsen IS, Fritzen AM, Carl CS, Agerholm M, Damgaard MTF, Holm JB, Marette A, Nordkild P, Kiens B, Kristiansen K, Wehkamp J, Jensen BAH. Human Paneth cell α-defensin-5 treatment reverses dyslipidemia and improves glucoregulatory capacity in diet-induced obese mice. Am J Physiol Endocrinol Metab. 2019;317:E42-E52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 13. | Panjeta A, Preet S. Anticancer potential of human intestinal defensin 5 against 1, 2-dimethylhydrazine dihydrochloride induced colon cancer: A therapeutic approach. Peptides. 2020;126:170263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 14. | Tang Y, Zhang D, Gong X, Zheng J. Repurposing of intestinal defensins as multi-target, dual-function amyloid inhibitors via cross-seeding. Chem Sci. 2022;13:7143-7156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 15. | Sun CQ, Arnold RS, Hsieh CL, Dorin JR, Lian F, Li Z, Petros JA. Discovery and mechanisms of host defense to oncogenesis: targeting the β-defensin-1 peptide as a natural tumor inhibitor. Cancer Biol Ther. 2019;20:774-786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 16. | Makeudom A, Supanchart C, Montreekachon P, Khongkhunthian S, Sastraruji T, Krisanaprakornkit J, Krisanaprakornkit S. The antimicrobial peptide, human β-defensin-1, potentiates in vitro osteoclastogenesis via activation of the p44/42 mitogen-activated protein kinases. Peptides. 2017;95:33-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Mi B, Liu J, Liu Y, Hu L, Panayi AC, Zhou W, Liu G. The Designer Antimicrobial Peptide A-hBD-2 Facilitates Skin Wound Healing by Stimulating Keratinocyte Migration and Proliferation. Cell Physiol Biochem. 2018;51:647-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 18. | Bindra GK, Williams SA, Lay FT, Baxter AA, Poon IKH, Hulett MD, Phan TK. Human β-Defensin 2 (HBD-2) Displays Oncolytic Activity but Does Not Affect Tumour Cell Migration. Biomolecules. 2022;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 19. | Warner JB, Larsen IS, Hardesty JE, Song YL, Warner DR, McClain CJ, Sun R, Deng Z, Jensen BAH, Kirpich IA. Human Beta Defensin 2 Ameliorated Alcohol-Associated Liver Disease in Mice. Front Physiol. 2021;12:812882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Takahashi M, Umehara Y, Yue H, Trujillo-Paez JV, Peng G, Nguyen HLT, Ikutama R, Okumura K, Ogawa H, Ikeda S, Niyonsaba F. The Antimicrobial Peptide Human β-Defensin-3 Accelerates Wound Healing by Promoting Angiogenesis, Cell Migration, and Proliferation Through the FGFR/JAK2/STAT3 Signaling Pathway. Front Immunol. 2021;12:712781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 68] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 21. | Wang W, Qu X, Dang X, Shang D, Yang L, Li Y, Xu D, Martin JG, Hamid Q, Liu J, Chang Y. Human β-defensin-3 induces IL-8 release and apoptosis in airway smooth muscle cells. Clin Exp Allergy. 2017;47:1138-1149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Gerashchenko OL, Zhuravel EV, Skachkova OV, Khranovska NN, Filonenko VV, Pogrebnoy PV, Soldatkina MA. Biologic activities of recombinant human-beta-defensin-4 toward cultured human cancer cells. Exp Oncol. 2013;35:76-82. [PubMed] |
| 23. | Fruitwala S, El-Naccache DW, Chang TL. Multifaceted immune functions of human defensins and underlying mechanisms. Semin Cell Dev Biol. 2019;88:163-172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 110] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 24. | Park MS, Kim JI, Lee I, Park S, Bae JY, Park MS. Towards the Application of Human Defensins as Antivirals. Biomol Ther (Seoul). 2018;26:242-254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 25. | Rivas-Santiago B, Trujillo V, Montoya A, Gonzalez-Curiel I, Castañeda-Delgado J, Cardenas A, Rincon K, Hernandez ML, Hernández-Pando R. Expression of antimicrobial peptides in diabetic foot ulcer. J Dermatol Sci. 2012;65:19-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 26. | Sanapalli BKR, Yele V, Kalidhindi RSR, Singh SK, Gulati M, Karri VVSR. Human beta defensins may be a multifactorial modulator in the management of diabetic wound. Wound Repair Regen. 2020;28:416-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Petkovic M, Mouritzen MV, Mojsoska B, Jenssen H. Immunomodulatory Properties of Host Defence Peptides in Skin Wound Healing. Biomolecules. 2021;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 28. | Prasad SV, Fiedoruk K, Daniluk T, Piktel E, Bucki R. Expression and Function of Host Defense Peptides at Inflammation Sites. Int J Mol Sci. 2019;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 29. | Meng L, Guo X, Yang X, Liu H, Yu M, Wu Y, Zhu Z. Human α defensins promote the expression of the inflammatory cytokine interleukin-8 under high-glucose conditions: Novel insights into the poor healing of diabetic foot ulcers. J Biochem Mol Toxicol. 2019;33:e22351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 30. | Wong SL, Demers M, Martinod K, Gallant M, Wang Y, Goldfine AB, Kahn CR, Wagner DD. Diabetes primes neutrophils to undergo NETosis, which impairs wound healing. Nat Med. 2015;21:815-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 570] [Cited by in RCA: 873] [Article Influence: 87.3] [Reference Citation Analysis (0)] |
| 31. | Louiselle AE, Niemiec SM, Zgheib C, Liechty KW. Macrophage polarization and diabetic wound healing. Transl Res. 2021;236:109-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 369] [Article Influence: 92.3] [Reference Citation Analysis (0)] |
| 32. | Niyonsaba F, Ushio H, Nagaoka I, Okumura K, Ogawa H. The human beta-defensins (-1, -2, -3, -4) and cathelicidin LL-37 induce IL-18 secretion through p38 and ERK MAPK activation in primary human keratinocytes. J Immunol. 2005;175:1776-1784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 192] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 33. | Chen X, Niyonsaba F, Ushio H, Hara M, Yokoi H, Matsumoto K, Saito H, Nagaoka I, Ikeda S, Okumura K, Ogawa H. Antimicrobial peptides human beta-defensin (hBD)-3 and hBD-4 activate mast cells and increase skin vascular permeability. Eur J Immunol. 2007;37:434-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 131] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 34. | Syeda F, Tullis E, Slutsky AS, Zhang H. Human neutrophil peptides upregulate expression of COX-2 and endothelin-1 by inducing oxidative stress. Am J Physiol Heart Circ Physiol. 2008;294:H2769-H2774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 35. | Li J, Zhu HY, Beuerman RW. Stimulation of specific cytokines in human conjunctival epithelial cells by defensins HNP1, HBD2, and HBD3. Invest Ophthalmol Vis Sci. 2009;50:644-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 36. | Semple F, Dorin JR. β-Defensins: multifunctional modulators of infection, inflammation and more? J Innate Immun. 2012;4:337-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 266] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 37. | Cui D, Lyu J, Li H, Lei L, Bian T, Li L, Yan F. Human β-defensin 3 inhibits periodontitis development by suppressing inflammatory responses in macrophages. Mol Immunol. 2017;91:65-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 38. | Li L, Jiang H, Chen R, Zhou J, Xiao Y, Zhang Y, Yan F. Human β-defensin 3 gene modification promotes the osteogenic differentiation of human periodontal ligament cells and bone repair in periodontitis. Int J Oral Sci. 2020;12:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 39. | Miles K, Clarke DJ, Lu W, Sibinska Z, Beaumont PE, Davidson DJ, Barr TA, Campopiano DJ, Gray M. Dying and necrotic neutrophils are anti-inflammatory secondary to the release of alpha-defensins. J Immunol. 2009;183:2122-2132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 122] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 40. | Yuan YS, Niu SP, Yu F, Zhang YJ, Han N, Lu H, Yin XF, Xu HL, Kou YH. Intraoperative single administration of neutrophil peptide 1 accelerates the early functional recovery of peripheral nerves after crush injury. Neural Regen Res. 2020;15:2108-2115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 41. | Donnarumma G, Paoletti I, Fusco A, Perfetto B, Buommino E, de Gregorio V, Baroni A. β-Defensins: Work in Progress. In: Advances in Microbiology, Infectious Diseases and Public Health: Volume 2. Donelli G (editor). Cham: Springer International Publishing; 2016. pp. 59-76.. |
| 42. | Niyonsaba F, Ushio H, Nakano N, Ng W, Sayama K, Hashimoto K, Nagaoka I, Okumura K, Ogawa H. Antimicrobial peptides human beta-defensins stimulate epidermal keratinocyte migration, proliferation and production of proinflammatory cytokines and chemokines. J Invest Dermatol. 2007;127:594-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 376] [Cited by in RCA: 405] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 43. | Gursoy UK, Könönen E, Luukkonen N, Uitto VJ. Human neutrophil defensins and their effect on epithelial cells. J Periodontol. 2013;84:126-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 44. | Prado-Montes de Oca E. Human beta-defensin 1: a restless warrior against allergies, infections and cancer. Int J Biochem Cell Biol. 2010;42:800-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 76] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 45. | Oono T, Shirafuji Y, Huh WK, Akiyama H, Iwatsuki K. Effects of human neutrophil peptide-1 on the expression of interstitial collagenase and type I collagen in human dermal fibroblasts. Arch Dermatol Res. 2002;294:185-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 46. | van Kilsdonk JWJ, Jansen PAM, van den Bogaard EH, Bos C, Bergers M, Zeeuwen PLJM, Schalkwijk J. The Effects of Human Beta-Defensins on Skin Cells in vitro. Dermatology. 2017;233:155-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 47. | Rousselle P, Braye F, Dayan G. Re-epithelialization of adult skin wounds: Cellular mechanisms and therapeutic strategies. Adv Drug Deliv Rev. 2019;146:344-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 344] [Article Influence: 57.3] [Reference Citation Analysis (0)] |
| 48. | Dai J, Shen J, Chai Y, Chen H. IL-1β Impaired Diabetic Wound Healing by Regulating MMP-2 and MMP-9 through the p38 Pathway. Mediators Inflamm. 2021;2021:6645766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 49. | Gao M, Nguyen TT, Suckow MA, Wolter WR, Gooyit M, Mobashery S, Chang M. Acceleration of diabetic wound healing using a novel protease-anti-protease combination therapy. Proc Natl Acad Sci U S A. 2015;112:15226-15231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 129] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 50. | Raina M, Bates AM, Fischer CL, Progulske-Fox A, Abbasi T, Vali S, Brogden KA. Human beta defensin 3 alters matrix metalloproteinase production in human dendritic cells exposed to Porphyromonas gingivalis hemagglutinin B. J Periodontol. 2018;89:361-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 51. | Baroni A, Donnarumma G, Paoletti I, Longanesi-Cattani I, Bifulco K, Tufano MA, Carriero MV. Antimicrobial human beta-defensin-2 stimulates migration, proliferation and tube formation of human umbilical vein endothelial cells. Peptides. 2009;30:267-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 91] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 52. | Petrov V, Funderburg N, Weinberg A, Sieg S. Human β defensin-3 induces chemokines from monocytes and macrophages: diminished activity in cells from HIV-infected persons. Immunology. 2013;140:413-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 53. | Amenomori M, Mukae H, Ishimatsu Y, Sakamoto N, Kakugawa T, Hara A, Hara S, Fujita H, Ishimoto H, Hayashi T, Kohno S. Differential effects of human neutrophil peptide-1 on growth factor and interleukin-8 production by human lung fibroblasts and epithelial cells. Exp Lung Res. 2010;36:411-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 54. | Umehara Y, Takahashi M, Yue H, Trujillo-Paez JV, Peng G, Nguyen HLT, Okumura K, Ogawa H, Niyonsaba F. The Antimicrobial Peptides Human β-Defensins Induce the Secretion of Angiogenin in Human Dermal Fibroblasts. Int J Mol Sci. 2022;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 55. | Seignez C, Phillipson M. The multitasking neutrophils and their involvement in angiogenesis. Curr Opin Hematol. 2017;24:3-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 56. | Armstrong DG, Boulton AJM, Bus SA. Diabetic Foot Ulcers and Their Recurrence. N Engl J Med. 2017;376:2367-2375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1953] [Cited by in RCA: 2333] [Article Influence: 291.6] [Reference Citation Analysis (2)] |
| 57. | Umehara Y, Kamata Y, Tominaga M, Niyonsaba F, Ogawa H, Takamori K. Antimicrobial peptides human LL-37 and β-defensin-3 modulate the expression of nerve elongation factors in human epidermal keratinocytes. J Dermatol Sci. 2017;88:365-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 58. | Afonso AC, Oliveira D, Saavedra MJ, Borges A, Simões M. Biofilms in Diabetic Foot Ulcers: Impact, Risk Factors and Control Strategies. Int J Mol Sci. 2021;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 67] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 59. | Gao X, Ding J, Liao C, Xu J, Liu X, Lu W. Defensins: The natural peptide antibiotic. Adv Drug Deliv Rev. 2021;179:114008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 89] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 60. | Parducho KR, Beadell B, Ybarra TK, Bush M, Escalera E, Trejos AT, Chieng A, Mendez M, Anderson C, Park H, Wang Y, Lu W, Porter E. The Antimicrobial Peptide Human Beta-Defensin 2 Inhibits Biofilm Production of Pseudomonas aeruginosa Without Compromising Metabolic Activity. Front Immunol. 2020;11:805. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 61. | Pachón-Ibáñez ME, Smani Y, Pachón J, Sánchez-Céspedes J. Perspectives for clinical use of engineered human host defense antimicrobial peptides. FEMS Microbiol Rev. 2017;41:323-342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 108] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 62. | Cardot-Martin E, Casalegno JS, Badiou C, Dauwalder O, Keller D, Prévost G, Rieg S, Kern WV, Cuerq C, Etienne J, Vandenesch F, Lina G, Dumitrescu O. α-Defensins partially protect human neutrophils against Panton-Valentine leukocidin produced by Staphylococcus aureus. Lett Appl Microbiol. 2015;61:158-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 63. | Bolatchiev A. Antibacterial activity of human defensins against Staphylococcus aureus and Escherichia coli. PeerJ. 2020;8:e10455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 64. | Pasupuleti M, Schmidtchen A, Malmsten M. Antimicrobial peptides: key components of the innate immune system. Crit Rev Biotechnol. 2012;32:143-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 571] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 65. | Steinstraesser L, Koehler T, Jacobsen F, Daigeler A, Goertz O, Langer S, Kesting M, Steinau H, Eriksson E, Hirsch T. Host defense peptides in wound healing. Mol Med. 2008;14:528-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 120] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 66. | Hozzein WN, Badr G, Badr BM, Allam A, Ghamdi AA, Al-Wadaan MA, Al-Waili NS. Bee venom improves diabetic wound healing by protecting functional macrophages from apoptosis and enhancing Nrf2, Ang-1 and Tie-2 signaling. Mol Immunol. 2018;103:322-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 67. | Badr G. Camel whey protein enhances diabetic wound healing in a streptozotocin-induced diabetic mouse model: the critical role of β-Defensin-1, -2 and -3. Lipids Health Dis. 2013;12:46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 68. | Liu S, Zhou L, Li J, Suresh A, Verma C, Foo YH, Yap EP, Tan DT, Beuerman RW. Linear analogues of human beta-defensin 3: concepts for design of antimicrobial peptides with reduced cytotoxicity to mammalian cells. Chembiochem. 2008;9:964-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 66] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 69. | Luo G, Sun Y, Zhang J, Xu Z, Lu W, Wang H, Zhang Y, Li H, Mao Z, Ye S, Cheng B, Fang X. Nanodefensin-encased hydrogel with dual bactericidal and pro-regenerative functions for advanced wound therapy. Theranostics. 2021;11:3642-3660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |