Published online Feb 15, 2019. doi: 10.4239/wjd.v10.i2.96
Peer-review started: November 15, 2018
First decision: November 29, 2018
Revised: January 29, 2019
Accepted: February 11, 2019
Article in press: February 12, 2019
Published online: February 15, 2019
Processing time: 95 Days and 10.6 Hours
Royal jelly (RJ) has been observed to have therapeutic properties in diabetic individuals, including the reduction of high blood sugar. This systematic review synthesized existing evidence to investigate the effectiveness of RJ supplementation in managing measures of blood glucose.
To determine the effectiveness of RJ supplementation on glycemic responses in healthy and non-insulin dependent diabetic adults, as well as animal models of diabetes.
This was a systematic review employing the PRISMA strategy. Peer-reviewed, published articles were extracted from several databases using key words related to target population, intervention and outcome and hand-selected for inclusion. Included articles proceeded to data extraction phase, where information on target parameters and effectiveness of treatment was summarized. Following this, the risk of bias for each included study was evaluated. Then, the long-term and immediate effectiveness of RJ supplementation in glycemic control were assessed using the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) tool, which rates the quality of evidence.
Of 168 articles extracted from database searching, eighteen were included for analysis in this systematic review. Across the studies, studied populations, intervention styles and outcome measures were largely heterogeneous. Despite this, the results in studies indicate a general trend of positive effect of RJ in glycemic regulation in vitro and in vivo. Additionally, some dose-dependent glycemic effects were observed, along with some large effect sizes. The risk of bias for human and animal studies is generally low-unclear risk, although lack of blinding is a serious concern in both categories. Overall, as per the GRADE tool, the quality of evidence is low, and very low for long-term and immediate effectiveness of RJ, respectively. A major limitation affecting evidence quality is the heterogeneity among included studies. Fasting blood glucose and glucose clearance appear to be most affected by RJ supplementation.
Quality of evidence suggesting that RJ is an effective modulator of glycemic regulation is low for long-term effects of RJ, and very low for immediate effects.
Core tip: Royal jelly (RJ) is a promising natural treatment to improve high blood glucose. Insulin sensitivity, fasting blood glucose levels, and rate of glucose clearance are among the glycemic parameters investigated in the current systematic review that are shown to approach normal levels due to regular RJ intake.
- Citation: Omer K, Gelkopf MJ, Newton G. Effectiveness of royal jelly supplementation in glycemic regulation: A systematic review. World J Diabetes 2019; 10(2): 96-113
- URL: https://www.wjgnet.com/1948-9358/full/v10/i2/96.htm
- DOI: https://dx.doi.org/10.4239/wjd.v10.i2.96
The incidence of type 2 diabetes (T2D) has drastically increased over the past thirty years, making it among the most taxing diseases for health agencies worldwide[1-3]. As of 2017, projections for global prevalence of diabetes in 2030 have already been surpassed[4,5]. By 2040, the prevalence of T2D is expected to reach 642 million worldwide[5]. The epidemic is believed to stem from rapid changes in society since the 1980s that promote long periods of inactivity, energy and nutrient overconsumption; however, further epigenetic and genetic interactions continue to be explored[1,2]. T2D is heavily associated with cardiovascular disease and obesity, which largely accounts for the morbidity and mortality in patients with the disease[6]. Thus, to minimize risk of complications in patients, food intake and concentrations of blood glucose must be tightly managed[6].
Numerous natural health products, including royal jelly (RJ), have been explored as potential hypoglycemic agents[7,8]. RJ is a yellow, milky substance secreted by worker honey bees (Apis mellifera) through cephalic exocrine glands, such as the mandibular and hypopharyngeal glands[9]. RJ functions to nourish larvae for the first three days after being reared, although larvae destined to be reproductive queen bees are fed the material throughout their entire larval and adult life[9]. 10-hydroxy-2-decenoic acid (10H2DA), a fatty acid unique to RJ, is a major compound of interest in blood glucose management for its observed hypoglycemic effects[7]. Takikawa et al[7] found that 10H2DA significantly increases non-insulin dependent phosphorylation of AMP kinase (AMPK) in skeletal muscle, increasing translocation of glucose transporter type 4 (GLUT4) to cell surface and consequently, glucose transport into the cell. An in vivo study examining life-extending effects of RJ found that 10H2DA induces upregulation of molecules involved in caloric restriction, reducing energy intake[10]. 10H2DA has demonstrated interactions with estrogen receptors leading to alterations in gene expression, potentially including those involved in glucose regulation[11]. The glucose modulating role of 10H2DA is the most well-investigated mechanism by which RJ might benefit patients with T2D, although other RJ components, such as sebacic acid, may also be important.
In addition to in vitro studies, RJ administration has demonstrated therapeutic potential in human and rodent diabetic models. In a randomized controlled trial, Khoshpey et al[12] found that daily ingestion of capsules containing 3000 mg RJ for eight weeks significantly decreased fasting blood glucose (FBG) in diabetic individuals compared to a placebo group. However, the effects of RJ administration on glycemic control outcomes are inconsistent across studies, possibly due to considerable variation in studied population and intervention. For example, while Khoshpey et al[12] found no significant change in carbohydrate (CHO) intake in the RJ-treated group, Pourmoradian et al[13] found a significant decrease in CHO intake in diabetic individuals in response to daily ingestion of 1000 mg lyophilized RJ for eight weeks. Rodent studies have shown a more pronounced effect: Ghanbari et al[14] found that addition of 100 mg/kg RJ in drinking solution improved circulating insulin and FBG in diabetic mice to levels similar to the healthy control group. Zamami et al[15] observed a similar magnitude of effect in insulin-resistant rats following administration of 300 mg/kg enzymatically treated RJ. It is difficult to apply the results of rodent studies into a human context due to variation in physiologic processes, particularly absorption and distribution of nutrients such as 10H2DA[16]. Presently, there is a lack of synthesis and analysis of these human and animal studies investigating the therapeutic effects of RJ.
This systematic review will investigate the effectiveness of RJ as a therapeutic agent in individuals with T2D. Specifically, we will assess animal, human and in vitro studies examining the effects of administration of RJ and its constituents on various outcomes that relate to glycemic control, such as plasma glucose levels, plasma lipid levels and hemoglobin A1c (HbA1c) levels in healthy and diabetic individuals. Included studies will investigate outcomes following both acute and long-term administration of RJ on glycemic control. The synthesis and evaluation of existing trials provides individuals and health care professionals with a resource to make informed decisions regarding T2D therapy. To our knowledge, no systematic review investigating RJ as a treatment for diabetes exists.
The guidelines of the 2009 PRISMA model strategy were followed throughout this review[17].
We conducted a systematic search of peer-reviewed articles relating to the impact of RJ on glycemic outcomes. A set of keywords were developed by Kamel Omer and Genevieve Newton to yield trials with study variables that are appropriate for the research question. These were subsequently searched in five databases: Cochrane Library, CINAHL Plus, PubMed (via NCBI), Web of Science, and ProQuest. Operator commands were used to yield studies containing at least one keyword for each variable within the title or abstract. A review protocol does not exist for this systematic review.
Following extraction and compilation of articles from the database results, duplicate studies were electronically removed. Bibliographies of studies were manually scanned to capture relevant studies. Titles and abstracts were manually screened and articles not meeting inclusion criteria were removed. Full text screening was completed for the remaining articles, where studies not meeting the inclusion criteria were removed. This was conducted in duplicate by Kamel Omer and Maxwell J Gelkopf; final decisions were settled by discussion. The resultant articles proceeded to the quality appraisal stage.
(1) Population: Healthy or diabetic human adults or animal models; (2) Intervention: Oral administration of RJ or its constituents; (3) Outcomes assessed: Direct measures of glycemic control or measures pertinent to glycemic control; (4) In vitro studies: Effects of RJ (or constituents) administration were investigated on an outcome directly related to glycemic control; (5) Methodology includes control group for comparison with treatment; and (6) Available in English.
Data on study design, subjects, treatment, and relevant outcomes and results were abstracted for each included study, where applicable, qualitative and quantitative details were added to each of these study variables. For subjects, the extracted number of participants was those that completed the study. Data summaries of each included study were then classified into one of three different tables, depending on study population and intervention style. The classifications are: (1) Acute administration (examining immediate RJ effects) of RJ in human trials; (2) Long-term administration (examining long-term RJ effects) of RJ in human trials; and (3) Long-term administration of RJ in animal trials and in vitro trials.
For each included human and animal study, the Cochrane Collaboration’s risk of bias assessment tool was used to determine the risk of bias[18]. The tool covers five pre-specified areas of bias: Selection bias (allocation concealment and random sequence generation), performance bias (blinding of participants and researchers), detection bias (blinding of outcome assessment), attrition bias (incomplete outcome data), and reporting bias (selective reporting)[18]. The tool also includes an “other bias” category for other sources of bias that do not fall within these prespecified areas[18]. For each area of bias, the tool provides criteria to rate the study as low, high, or unclear risk of bias. A high risk of bias indicates a high possibility of bias that is likely to impact the study results, while a low risk suggests negligible risk[18]. An unclear risk of bias indicates that insufficient information is provided to determine if results are impacted by bias, but some doubt is raised[18].
Two authors, Kamel Omer and Maxwell J Gelkopf, assessed all included articles to reach a consensus on the risk of bias for each study. Higgins et al[18] provides a detailed guide on the ranking procedure, which was applied by reviewers throughout the risk of bias assessment. Although the tool is designed for human trials, O’Connor and Sargeant[19] developed a modified version for use in risk of bias assessment of animal trials. This modified version was used by the reviewers to guide ranking of risk of bias for the included animal studies. Justification was noted for each judgement, including paraphrases and direct quotes from the article when available.
Following determination of the risk of bias for each study, the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) tool was used to rate the overall quality evidence across all included studies for a single outcome[20]. In this review, GRADE was used to assess the quality of evidence for RJ’s capacity to manage blood glucose levels following long-term supplementation of RJ as well as acute effects of RJ administration. The GRADE tool encompasses five domains to determine overall quality: risk of bias, indirectness to research question, imprecision of results, inconsistency between studies and publication bias[20].
To represent the GRADE rating, a summary of findings table was created. Ranking begins at high quality due to the majority of trials being randomized controlled trial but are gradually degraded if serious concern exists in any of the five domains[20]. Ranking can also be upgraded if there is no plausible confounding bias, or if magnitude of effect is large or dose-dependent[20]. The overall ranking is determined by number and magnitude of all downgrades and upgrades[20]. Each of the four possible rankings directly correspond to a certain overall quality of evidence (very low, low, moderate, high)[19]. Ryan and Hill[21] provide criteria for what constitutes a concern in these domains, which was used as a guide for reviewers when determining the GRADE ranking. To factor animal studies in, a modified version of GRADE for animal studies developed by Wei et al[22] was used. Kamel Omer and Genevieve Newton reached a consensus on the GRADE score based on criteria and justification was provided for each decision on the summary of findings table.
To determine effect size and the precision of the quantitative data of the study results, appropriate measures were manually calculated. Standardized mean difference (SMD, also known as Cohen’s d) was calculated as described by Faraone[23] using a pooled standard deviation and sample means of the treatment and placebo group to estimate magnitude of treatment effect. A large effect size constitutes a SMD of > 0.8, while a small effect size is considered to have a SMD of < 0.2; all values in between are medium effect sizes[23]. A negative value indicates that the treatment reduces the parameter being investigated[23]. The SMD was calculated for values taken at the endpoint of the study. For studies with multiple treatment groups, the SMD was calculated for the group with highest dosage. Where numerical values were unavailable, values were interpolated from provided graphs. If values were reported as medians, these were used in place of the mean throughout the effect size calculation. As part of the GRADE evaluation, 95%CIs of SMDs for long-term, hypoglycemic outcomes were calculated to assess imprecision of the effect sizes. For simplicity of evaluation and comparison, SMDs were reported in this review for statistically significant outcomes only.
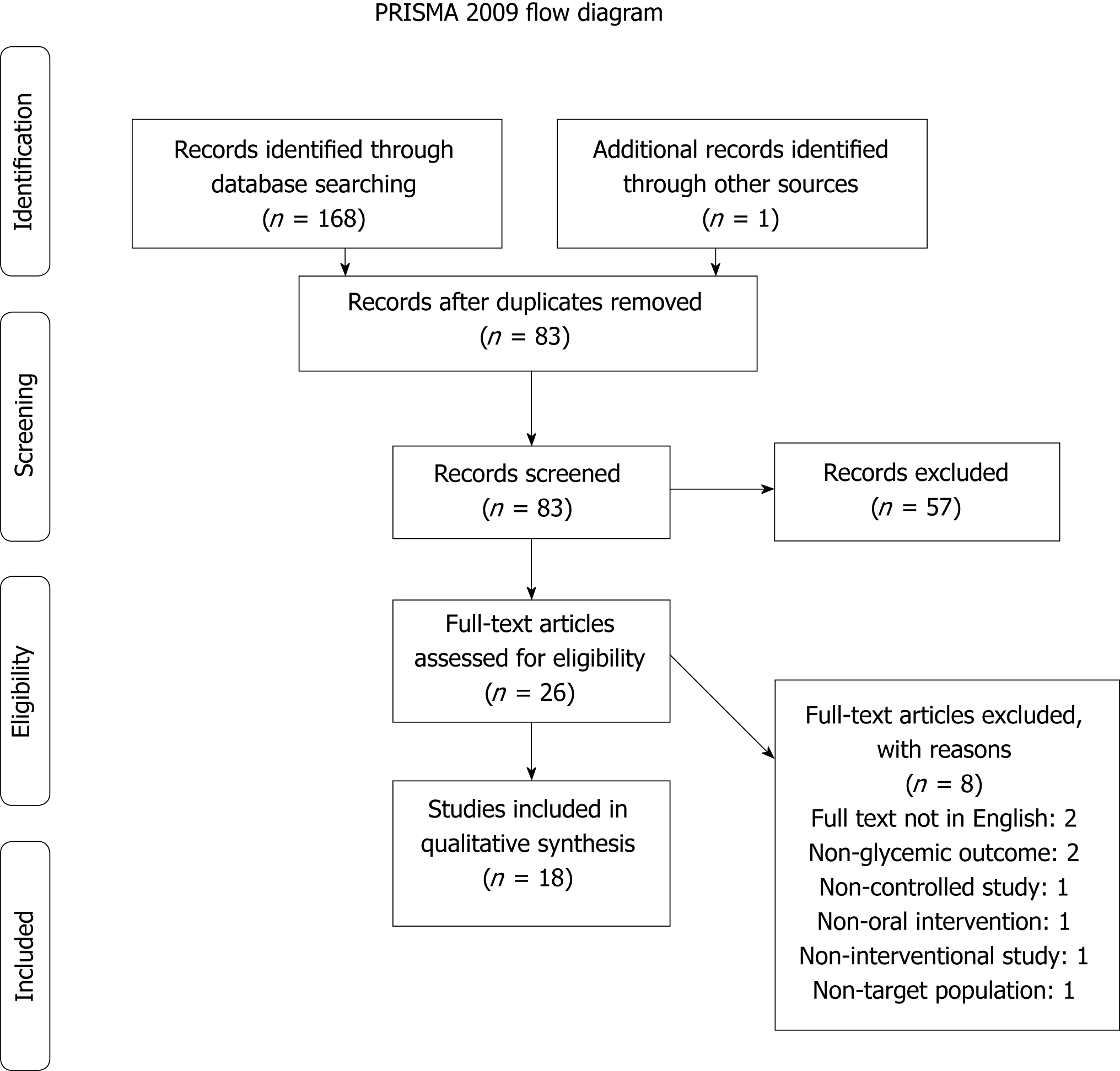
The pre-specified search strategy was conducted on March 21st, 2018, yielding 168 results. Grey literature databases were searched, but no pertinent articles were found. One study was captured from scanning the bibliographies of collected studies. After removal of duplicates, the total number of unique studies was determined to be 83. These studies went on to the title and abstract screening. Fifty-seven studies were removed due to not meeting the inclusion criteria at this stage. The remaining 26 underwent a full-text screening to determine eligibility. Of these 26, eight were excluded: two because they were not written in English, two for assessing outcomes not related to glycemic control, one because it lacked an oral intervention in treatment groups, one because the study design did not have a control group, one because it lacked an intervention, and one because it did not target the desired population for this review. The resultant 18 articles were included in the systematic literature review for quality of evidence appraisal (Figure 1).
Following the systematic search, study characteristics, including intervention style, length, participants and results were manually extracted and summarized into the Tables 1, 2 and 3[24-36].
| Ref. | Study design | Subjects | Treatment | Outcome measures | Effectiveness |
| Iaconelli et al[24] | Crossover study | N = 10 + 10 healthy individuals and individuals with type 2 diabetes | Each subject went through three studies on different days: 0 g, 12 g, or 23 g of sebacic acid substituted fats in a meal | Glucose clearance: Postprandial | Significantly improved glucose clearance in diabetic subjects only in dose-dependent manner (d = -1.70) |
| Insulin secretion/clearance rate | Significantly improved GLUT4 expression (d = 0.81) and glucose uptake in L6 cells (d = 0.67) | ||||
| GLUT4 expression in L6 myotube cells | Insulin secretion/clearance decreases significantly in similar fashion between healthy and diabetic patients. Dose-response relationship. For diabetics, d = -1.12 | ||||
| Mobasseri et al[25] | Randomized controlled trial | N = 20 + 20 adults with type 2 diabetes aged 30-65 in control and treatment groups | 15 g of royal jelly ingested orally after overnight fasting | Hyperglycemia: Fasting blood glucose, glucose clearance after royal jelly consumption | No significant difference in outcome measures between two groups |
| Hyperinsulinemia: Serum c-peptide and insulin | |||||
| Münstedt et al[26] | Controlled trial | N= 10 + 10 healthy males, split into experimental and control groups | 20 g of fresh royal jelly ingested orally | Glucose clearance: Plasma samples during OGTT | Significantly increased rate of glucose clearance (insufficient information for effect size calculation) |
| Insulin resistance: Serum insulin and c-peptide | No significant change in serum insulin profile | ||||
| Münstedt et al[27] | Randomized controlled trial | N = 15 healthy male adults aged 20-34, unspecified distribution between treatment and control groups | 0.55 g lyophilized royal jelly in enteric-coated capsule ingested orally | Hyperglycemia: Glucose clearance (OGTT) | Improved glucose clearance and decreased plasma insulin, unspecified statistical significance (insufficient information for effect size calculation) |
| Insulin resistance: Serum insulin and c-peptide |
| Ref. | Study design | Subjects | Treatment | Outcome measures | Effectiveness |
| Khoshpey et al[12] | Randomized double-blind controlled trial | N = 11 females + 12 males aged 20-65 with type 2 diabetes in control group (placebo) | 3000 mg royal jelly oral capsules once per day for 8 wk. Control received placebo | Macronutrient intake | No significant change in macronutrient intake |
| N = 13 females + 10 males aged 20-65 with type 2 diabetes in treatment group | Hyperglycemia: Fasting blood glucose | Fasting blood glucose significantly reduced in comparison to control group (d = -0.87) | |||
| Mobasseri et al[28] | Randomized controlled trial | N = 25 + 25 females with type 2 diabetes aged 30-65 in control and treatment groups | 200 mg royal jelly powder prepared in gel form and served with breakfast for 8 wk. Control group received placebo | Plasma triglyceride | Significantly decreased plasma triglyceride in comparison to control (d = -0.476) |
| Morita et al[29] | Randomized double-blind controlled trial | N = 30 healthy adults 42-83 yr of age in control (placebo) | 3000 mg royal jelly in 100 mL liquid daily for 6 mo. Control received placebo identical in appearance | Body weight: BMI | Significantly improved fasting blood glucose (d = -0.9596) |
| N = 31 healthy adults 42-83 yr of age in treatment group | Insulin resistance: HOMA-IR | No significant changes in other outcomes of interest | |||
| Hyperglycemia: HbA1c, fasting blood glucose | |||||
| Plasma triglyceride | |||||
| Pourmoradian et al[13] | Human double-blinded randomized clinical trial | N = 23 females aged 30-65 with type 2 diabetes in treatment group | 1000 mg lyophilized royal jelly in soft gel form served after breakfast for 8 wk. Control group received placebo soft gel | Body weight: weight scale before and after study period | Significantly decreased body weight within same group, before and after intervention (d = -0.3808) |
| N = 22 females aged 30-65 with type 2 diabetes in control group | Macronutrient intake: 24-h recall food questionnaire for 3 d before and after study period | Significantly decreased energy intake within same group, before and after intervention (d = -9.52) | |||
| Pourmoradian et al[30] | Human double-blinded randomized controlled trial | N = 21 females aged 30-65 with type 2 diabetes in treatment group | 1000 mg lyophilized royal jelly in soft gel form served after breakfast for 8 wk. Control group received placebo soft gel | Plasma insulin | Significantly decreased plasma insulin and HbA1c and insignificantly decreased fasting blood glucose compared to baseline within same group, before and after intervention. |
| N = 20 females aged 30-65 with type 2 diabetes in control group | HbA1c | d = 0.016 (HbA1c) | |||
| Hyperglycemia: Fasting blood glucose | d = -0.0785 (plasma insulin) | ||||
| Shidfar et al[31] | Human double-blinded randomized controlled trial | N = 23 + 23 adults 25-65 yr old with type 2 diabetes in experimental and control (placebo) groups | 1000 mg royal jelly in soft gelatin capsules 3 times daily for 8 wk. Control group received placebo identical in appearance to treatment | Fasting blood sugar | Significantly decreased fasting blood levels to more normal range (d = -0.3725) |
| Macronutrient intake: 24-h recall diet questionnaire | Did not significantly alter macronutrient intake | ||||
| Insulin resistance: HOMA-IR | Significantly decreased HOMA-IR: improved insulin sensitivity (d = -0.79) |
| Ref. | Study design | Subjects | Treatment | Outcome measures | Effectiveness |
| Ghanbari et al[14] | Randomized controlled trial | N = 8 healthy male Wistar rats aged 10-12 wk (control) | 100 mg/kg BW royal jelly dissolved in 1 mL of water daily for 6 wk | Hyperinsulinemia: ELISA test on plasma sample | Treatment significantly improved insulin levels (d = 1.67) and hyperglycemic fasting blood glucose (d = -2.72) levels to levels similar to healthy control group |
| N = 8 diabetic male Wistar rats aged 10-12 wk | Hyperglycemia: Fasting plasma glucose | ||||
| N = 8 healthy male Wistar rats aged 10-12 wk receiving treatment | |||||
| N = 8 diabetic male Wistar rats aged 10-12 wk receiving treatment | |||||
| Fujii et al[32] | Controlled trial | N = 80 male streptozotocin-diabetic rats aged 5 wk equally split into three experimental groups and one control group | Each experimental group had one of 1, 10, and 100 mg/kg body weight royal jelly administered orally by force for 4 wk. Control group received purified water | Hyperglycemia: Blood glucose (unknown whether fasting) | Royal jelly administration overall slightly decreased blood glucose levels in non-dose dependent manner (no information on statistical significance) |
| Body weight | No significant change in body weight between groups | ||||
| Membrez et al[33] | Randomized controlled trial | N = 15 male db/db mice aged 6-8 wk in control group | 1 g/kg body weight of sebacic acid was added to chow food in one experimental group, and 10 g/kg body weight SA to second experimental group’s chow for 6 wk | Hyperglycemia: OGTT and fasting (plasma samples) | In more heavily supplemented group: Hyperglycemia significantly improved (d = -1.86) and improved glucose clearance (d = -3.20), HbA1c significantly decreased (d = -1.89), ketone bodies significantly increased (d = 1.16), dose response relationship observed, gluconeogenic and lipogenic enzyme expression significantly decreased (insufficient information for SMD estimation), food intake was significantly decreased (d = -1.82). |
| N = 30 male db/db mice aged 6-8 wk equally split in two experimental groups | HbA1c: Plasma samples | ||||
| Liver gene expression: RNA extracted from liver samples | |||||
| Food intake: Chow consumed | |||||
| Takikawa et al[7] | In vitro | L6 myotubes grown in cell culture and collected from healthy male mice 7 wk of age | Cell cultured myotubes treated with 10H2DA | Glucose clearance: GLUT4 translocation to plasma membrane | Significantly improved GLUT4 translocation to plasma membrane in skeletal muscle cells compared to non-treated myotube cells (d = 0.4698) |
| Mice fed 1.6 mmol/kg 10H2DA | |||||
| Yoneshiro et al[34] | Controlled trial | N = 8 3-wk old healthy male mice (control) | High fat diet with 5% lyophilized royal jelly powder for 17 wk | Body weight gain | Body weight gain due to white adipose tissue significantly reduced compared to HFD group (d = -2.82) |
| N = 11 3-wk old healthy male mice fed HFD | Hyperlipidemia: Plasma sample | Significantly decreased levels of NEFA compared to HFD (d = -1.6072) | |||
| N = 11 3-wk old healthy male mice fed high fat diet with treatment | Hyperglycemia: Plasma sample | Significantly improved hyperglycemia compared to HFD group (d = -2.04) | |||
| Insulin resistance: HOMA-IR | HOMA-IR significantly decreased compared to HFD group, not significantly different from control group (d = -1.23) | ||||
| Zamami et al[15] | Controlled trial | N = 6 6-wk old healthy male Wistar rats (control, received water) | Two experimental groups: One fed 100 mg/kg and the other 300 mg/kg of dilute enzymatically treated royal jelly supplementation daily for 8 wk | Insulin resistance: HOMA-IR | High fructose diet induced insulin resistance in rats |
| N = 5 6-wk old healthy male Wistar rats as vehicle-treated group (received high fructose consumption) | Food intake | Plasma insulin levels and HOMA-IR similar between healthy control group and fructose drinking rats supplemented with 300 mg/kg royal jelly. Dose dependent relationship observed d = -0.7063 (effect size of 300 mg/kg royal jelly on fructose drinking rats) | |||
| N = 6 + 6 6-wk old healthy male Wistar rats (received high fructose consumption) in two treatment groups | Body weight | No significant difference in body weight and FBG between groups | |||
| Plasma triglycerides | Plasma triglycerides significantly decreased compared to control dose-dependently (d = -1.62) | ||||
| Watadani et al[35] | Controlled trial | N = 7 female KK-Ay mice 5 wk of age in control group | 3 mg/kg 10H2DA for 4 wk | Hyperglycemia: Plasma glucose samples collected in intervals after OGTT | Significantly improved glucose clearance (d = -1.33) and fasting blood glucose (d = -1.23) |
| N = 8 female KK-Ay mice 5 wk of age in treatment group | Body weight: Adiposity index of abdominal, mesenteric and retroperitoneal fat tissue | Body weight did not differ between groups | |||
| Insulin resistance: HOMA-IR | Significantly improved insulin sensitivity (d = -4.44) | ||||
| Glucose regulatory proteins: AMPK, G6Pase, Pck1 levels, GLUT4, GS/GSK in tissue homogenates | Significantly increased levels of G6Pase (d = 1.22) and Pck1(d = 0.77) mRNA in liver cells. Significantly increased levels of pAMPK in muscle (d = 3.13), but no change in liver. Insignificant increase in GLUT4 in muscle cells. No change in GS/GSK levels between groups | ||||
| Yoshida et al[36] | Controlled trial | 16 female KK-Ay mice split into control and experimental groups | 10 mg/kg royal jelly in 1/15M phosphate buffer 5 d/wk for 4 wk | Significantly improved rates of glucose clearance (d = -1.25) | |
| Insignificantly decreased body weight | |||||
| Significantly increased pAMPK levels in liver (d = 2.39) and skeletal muscle (d = 1.73). Significantly decreased G6Pase mRNA levels in liver (d = -1.65), but no change in Pck mRNA levels. Insignificantly increased GLUT4 levels in skeletal muscle | |||||
| Significantly decreased plasma NEFA (d = -1.42). No change in plasma TG | |||||
| No significant change in plasma insulin |
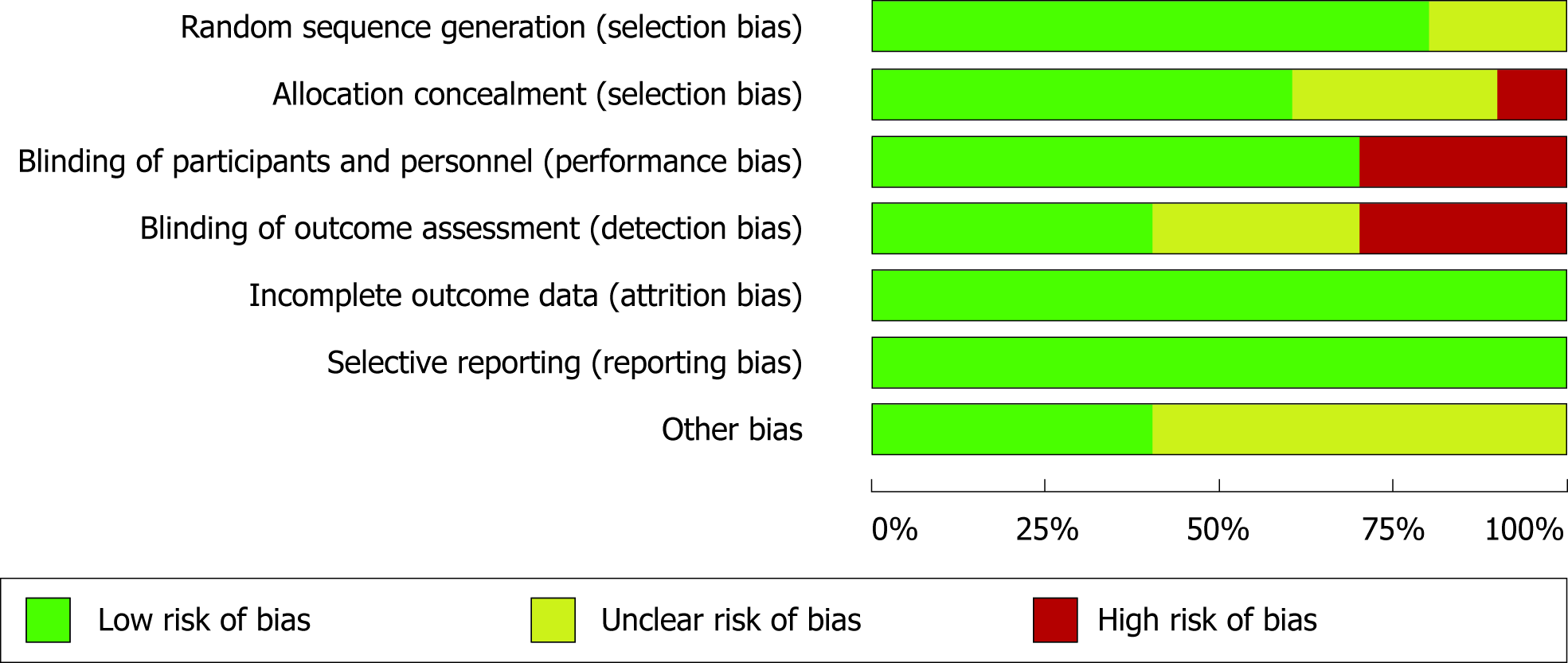
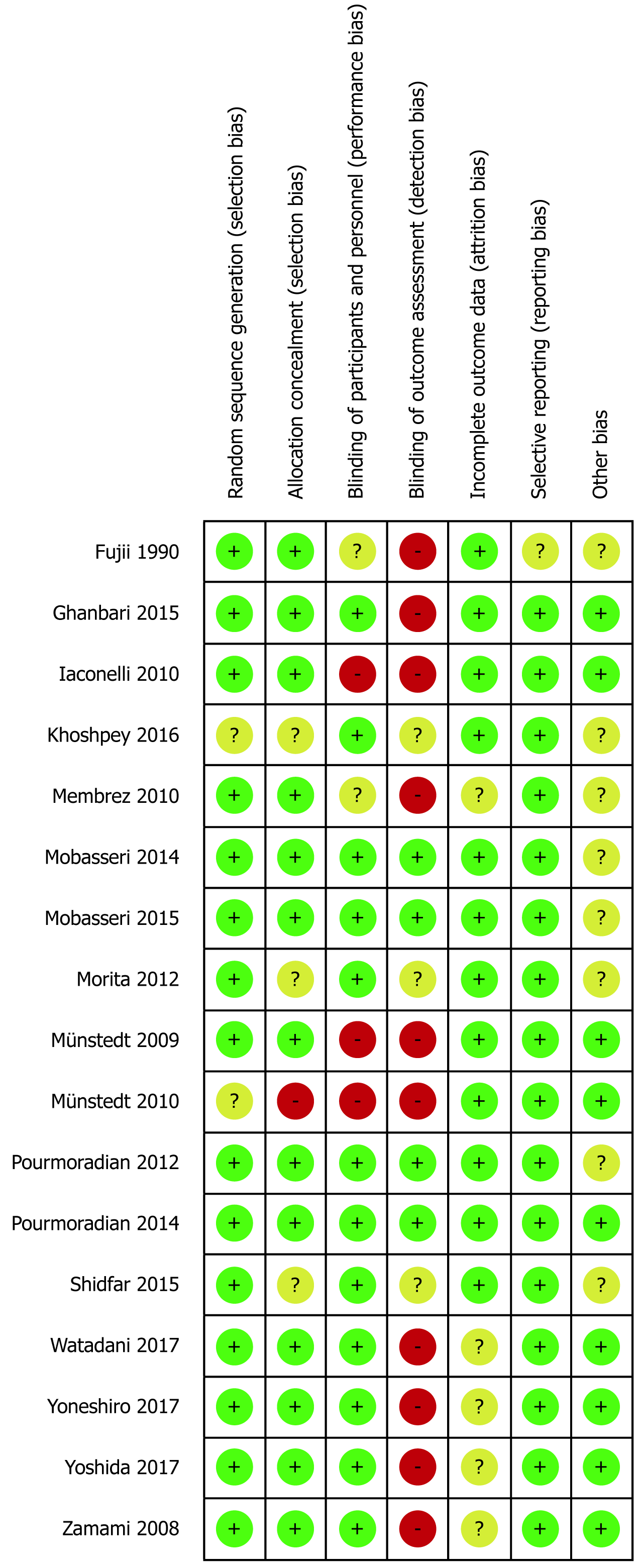
For human trials, the pre-specified areas of bias are generally low risk. The biggest source of concern for bias stems from the lack of transparency of measures taken to prevent a given area of bias. This was particularly evident in the allocation concealment category, as evidenced by the high proportion of human trials at high or unclear risk in that category (Figure 2). Moreover, although many studies claimed that participants and personnel were blinded, description of the actual blinding methodology was rarely provided, suggesting that performance and detection bias are considerable concerns among included human studies. The next biggest source of concern for bias is apparent in the “other bias” section-this is largely due to confounding bias arising from the recruitment of participants from a single source (e.g., common hospital). Overall, of the human trials, domains of bias at high risk are relatively few.
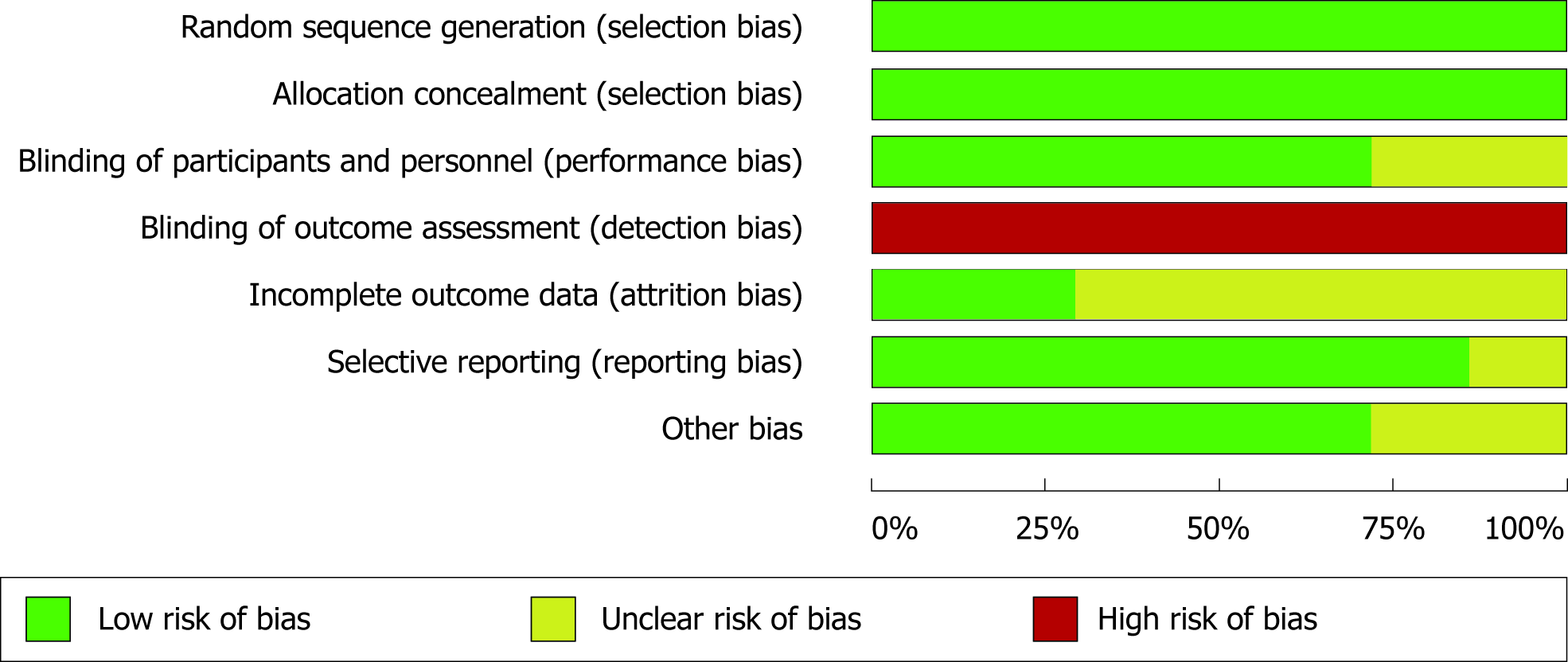
Across the animal studies, the risk of bias is a notably bigger concern. One hundred percent of the studies are considered high risk of detection bias (i.e., group allocations known to outcome assessors), potentially due to blinding being uncommon in animal studies (Figure 3). Attrition bias is another serious concern across the animal studies; most included animal studies excluded some individual subjects from analysis without any explanation. Performance and selection bias, however, are well accounted for in the included animal studies, with nearly 100% of both domains at low risk. Like human trials, reporting bias is not a serious concern in the animal trials (Figure 4).
The quality of evidence was evaluated as per the GRADE criteria with results shown in Tables 4 and 5. This evaluation integrated study results with risk of bias across all studies included in this review.
| GRADE criteria | Rating | Support for judgement | Overall quality of evidence |
| Outcome: Long term glycemic control (n = 14 studies) | |||
| RoB (assessed on Cochrane RoB Collaboration Tool) | No | Only one study had low RoB for all categories. Most studies had at least one item at high or unclear RoB | High |
| Serious (-1)1 | |||
| Very serious (-2) | |||
| Inconsistency | No | Generally, positive clinical effects demonstrated but some studies indicate null effects. There are also large variations in magnitude of effect. Heterogeneity is notable between the studies (in population, intervention and outcome assessment) | Moderate |
| Serious (-1) 1 | |||
| Very serious (-2) | |||
| Indirectness | No1 | Evidence synthesized from studies addresses review question with respect to population, interventions and outcome | Low1 |
| Serious (-1) | |||
| Very serious (-2) | |||
| Imprecision | No | All studies have groups with small sample sizes (≤ 20), with no indication that they meet required sample sizes to detect difference in outcome; observable but statistically insignificant measures in many studies suggest sample sizes were too small to detect difference. 95% confidence intervals of effect size estimates mostly suggest an appreciable benefit for treatment, but there are several that suggest possibility of no meaningful effect | Very low |
| Serious (-1) | |||
| Very serious (-2) 1 | |||
| Publication bias | Undetected1 | There is chance of publication bias considering the review is entirely “small-scale” trials; this area of research is not well-established and there is potential for publication bias, but none was overtly detected | |
| Strongly suspected (-1) | |||
| Other | Large effect (+11 or +2) | Standardized mean difference of studies (effect size) indicates large magnitude of effect of treatment. Dose-response relationships observed | |
| Dose response (+11 or +2) | |||
| No plausible confounding (+1 or +2) | |||
| GRADE criteria | Rating | Support for judgement | Overall quality of evidence |
| Outcomes: Acute glycemic control outcomes (n = 4 studies) | |||
| RoB (assessed on Cochrane RoB Collaboration Tool) | No | Majority of studies had overall high RoB, likely affecting the study results | High |
| Serious (-1) | |||
| Very serious (-2)1 | |||
| Inconsistency | No | Outcome effects are somewhat consistent, and studied population are similar enough to not be considered detrimental to evidence quality. Intervention, however, was heterogeneous across all relevant studies | Moderate |
| Serious (-1) 1 | |||
| Very serious (-2) | |||
| Indirectness | No1 | Research question is addressed by majority of the animal studies | Low |
| Serious (-1) | |||
| Very serious (-2) | |||
| Imprecision | No | All studies have groups with small sample sizes (≤ 20), with no indication that they meet required sample sizes to detect difference in outcome. For those with calculable effect sizes, the confidence intervals suggest potential for no appreciable benefit | Very low1 |
| Serious (-1) | |||
| Very serious (-2) 1 | |||
| Publication bias | Undetected1 | There is chance of publication bias considering the review is entirely “small-scale” trials; this area of research is not well-established and there is potential for publication bias, but none was overtly detected | |
| Strongly suspected (-1) | |||
| Other | Large effect (+1 or +2) | Some dose response relationships observed, however not enough studies to confirm this relationship. Insufficient effect size estimates to determine if effect is large or not | |
| Dose response (+1 or +2) | |||
| No plausible confounding (+1 or +2) | |||
Direct measures of glycemic control (FBG, glucose clearance rate, insulin levels) appear to be appreciably impacted by RJ administration. Most of the included studies which examined FBG and rate of glucose clearance observed significant change from baseline due to oral supplementation of RJ. Of these results, the majority had large effect size estimates, substantiating the role of the intervention in the observations. Most of the studies investigating insulin levels also reported a beneficial effect. Furthermore, in studies with multiple experimental groups, a dose-response relationship was observed in plasma insulin levels and rate of glucose clearance, but not FBG levels. Abnormal regulation of these parameters give rise to other secondary conditions associated with T2D, such as high levels of HbA1c.
The beneficial effect of RJ supplementation was demonstrated by improved indirect measures of glycemic control. These indirect measures are precursors (e.g., high circulating fat) or indicators (e.g., HbA1c) of hyperglycemia. The effectiveness of RJ administration in improving these parameters was not as apparent, as there was no clear trend in outcome responses to RJ treatment. However, when significant changes were detected in the experimental group, these results mostly had large effect magnitude estimates. The inconsistency between observed effects may be due to heterogeneity among the included studies.
The wide range of supplementation forms included in the evidence may explain some of the observed inconsistency between results. For example, many studies used lyophilized RJ, which is known to be chemically different and less bioactive compared to fresh RJ[27,37]. Numerous clinical trials used enteric coated capsules to deliver RJ, which is known to alter pharmacodynamic properties of compounds[38]. Some animal and human studies added the supplementation to meals, which may affect effectiveness due to food-drug interactions[39]. When similar populations were studied, some outcomes showed reduced, negated or contradictory effects in response to RJ as compared to 10H2DA and vice versa, such as between Yoshida et al[36] and Watadani et al[35]. This circumstance indicates the possibility of RJ constituents having interactions amplifying or diminishing effectiveness of certain glycemic outcomes.
Due to the different investigation types and subjects across the included studies, it is important to contextualize the effectiveness for the different populations. The largest improvements to blood glucose were observed in diabetic models, both human and animals. Healthy (human) models had some improvements in blood-glucose parameters; however, for most normal blood-glucose parameters, there is a limit on the possible difference from baseline. Only one in vitro was included and was used in this review primarily to elucidate mechanisms and support the in vivo results. Thus, the effectiveness of RJ as a glycemic regulator was determined through the lens of diabetic human patients, with the other populations used to support the findings.
Mostly displayed in the rodent trials, there was a marked difference on effectiveness of RJ administration between studies that investigated the effects of RJ between genders, with greater efficacy seen in males. The divergent effects may potentially be due to the estrogenic activity of compounds derived from RJ, particularly 10H2DA and a sterol, 24-methylenecholesterol[11]. These have weak affinity for estrogen receptors that induce changes in gene expression[11]. Because these compounds would compete with endogenous estrogen for receptor binding, RJ may not be effective in individuals with elevated estrogen levels (i.e., premenopausal females). Correspondingly, estrogen perfusion has previously been linked to improved hyperglycemic symptoms in postmenopausal females and males, which is consistent with the evidence in this review[40,41].
Although very few participants in the included study reported undesirable effects of RJ consumption, potential adverse effects in humans are an important factor in their feasibility as a glycemic regulator. One case report found an association between haemorrhagic colitis and daily RJ intake, which had never been documented previously[42]. Bronchospasm and anaphylaxis have also been noted in individual cases[43,44]. The aforementioned cases are possibly due to allergic reactions to RJ proteins[42,43,44]. Harmful RJ-drug interactions should be considered; consuming RJ while taking warfarin has been associated with hematuria[45].
The present systematic review suggests that RJ has a positive effect on both direct and indirect measures of glycemic control in diabetic and healthy individuals. This general trend was observed in both animal and adult human trials but was more pronounced in the former. In healthy individuals, supplementation of RJ may reduce risk of developing hyperglycemia and insulin resistance. With the evidence presented, RJ is likely more effective as a long-term dietary supplement rather than for acute treatment of hyperglycemia. Effective clinical doses appear to be as low as 1000 mg of fresh RJ daily for diabetic humans, but true values may vary between individuals and supplementation form.
In a normal state, intracellular protein-protein interactions arising from insulin binding to its surface receptor are critical to blood glucose regulation[46,47]. One major result of the signaling cascade induced by insulin on various tissue types is the translocation of the GLUT4 glucose transporter to the cell surface, which works to import glucose into the cell[46]. The insulin-dependent pathway also modifies gene expression and protein activity such as those involved in glycogen breakdown[46]. Insulin receptor substrate (IRS) proteins are key intermediates in the pathway[47]. With elevated levels of circulating fatty acids as in diabetic individuals, phosphorylation of IRS proteins is inhibited via activation of protein kinase C (PKC)[47,48]. As a result of decreased sensitivity to insulin, the cellular responses involved in regulating blood glucose are impaired, leading to hyperglycemia[48].
Intracellular AMPK, which induces similar glycemic responses to insulin in a non-insulin dependent pathway, has been observed to be activated by 10H2DA, the fatty acid derived from RJ that is thought to underlie its glycemic effects[7,49]. Takikawa et al[7] found AMPK to be activated by 10H2DA via activation of CaMKKβ (calcium/calmodulin-dependent protein kinase kinase beta). In skeletal muscle, pathways mediated by AMPK have been observed to increase GLUT4 gene expression and translocation[7,50]. In addition to this, AMPK-mediated regulation of GLUT4 has been observed to improve insulin-stimulated GLUT4 regulation, thus potentially improving response (sensitivity) of insulin receptors bound to ligand[51]. Moreover, activated AMPK in adipocytes and skeletal muscle are also responsible for enhancing enzymes involved in fatty acid oxidation, thus potentially leading to a decrease in body weight and circulating fatty acids[36]. Finally, in Yoshida et al[36], increased levels of AMPK following oral administration in mice appeared to stunt activity of glucose-6-phosphatase in hepatocytes, thus suggesting that the kinase can also regulate gluconeogenesis (and therefore glucose export) independently of insulin. By activating an enzyme that works to increase cellular energy levels in the cell, 10H2DA from RJ may mediate the desired hypoglycemic effects in diabetic subjects.
In the hypothalamus, AMPK plays an important regulatory role in food intake[52]. When activated, a signaling cascade that leads to increased energy intake is initiated[53]. Downstream of this cascade, mammalian target of rapamycin (mTOR) signaling is known to play a direct and important role[52]. However, in the presence of 10H2DA, mTOR activity has been observed to be decreased in vitro, resulting in decreased energy intake in hypothalamic cells[10,53]. The beneficial effect of RJ on food intake and body weight is observed in Pourmoradian et al[13], where both parameters were significantly decreased in diabetic female subjects. Following a decrease in macronutrient intake, there is a decrease in fatty acid synthesis and circulating lipids, leading to a decrease in activation of PKC. Thus, RJ’s possible action to decrease body weight and food intake is a mechanism that adds to its beneficial effects on glycemic regulation.
As previously mentioned, rodent studies showed a difference between effectiveness of RJ as administered to males compared to females. Fatty acids and sterols in RJ putatively have weak affinity for estrogen receptors, which when activated principally affect gene expression[11,54]. In addition to affecting transcriptional and translational activity that may regulate glycemic activity, estrogen receptors are able to induce activation of AMPK intracellularly[54]. Moreover, activated estrogen receptors in skeletal muscle have been shown to amplify GLUT4 translocation[54]. In the hypothalamus, estrogen suppresses energy intake through mechanisms not fully understood[54]. Thus, in accord with the evidence collected in this systematic review, activation of estrogen receptors in the body work to increase energy expenditure and decrease energy intake[54].
Generally, the principal limitation within the included evidence is the wide range of intervention methodology between the included studies. Although this heterogeneity provides data on the various ways to supplement RJ orally (e.g., prandial), there may not be sufficient evidence for one particular intervention method, including duration and dosage of supplementation, to be adopted by health care providers or researchers. As previously described, this variation in intervention methodology also likely contributes to the inconsistency across study results. Moreover, outcomes associated with different interventions at times contradict each other-Watadani et al[35] observed significantly increased expression of G6Pase in liver cells following 10H2DA administration, while Yoshida et al[36] observed significantly decreased expression of G6Pase in liver cells following RJ administration. The source of RJ also differs between studies, which is a potential confounder of the evidence, as the chemical composition of RJ is known to differ between time of year, honeybee age, and geographic location[55].Thus, the inconsistency across intervention methods and resultant outcomes is a notable limitation of the overall evidence.
Across human studies, limitations exist largely due to the study populations. Although most of the clinical trials included a balance of male and females, few studies examined exclusively females, and none examined males exclusively. Considering RJ’s potential estrogenic activity, a comparison of effects on male and female populations might have provided clearer evidence on the effectiveness of RJ as treatment for diabetes. Notably, the animal studies, which examined exclusively either male or female rodents, displayed differential responses to similar treatment between sexes. Similarly, the age ranges of included subjects in the included trials were relatively wide; inclusion criteria based on tighter ranges (e.g., younger adults, menopausal women) might have determined if certain groups are more affected by the treatment than others, once again potentially due to RJ’s activation of estrogen receptors. Moreover, the exclusion and inclusion criteria of the clinical trials affect the generalizability of RJ as treatment for diabetes: For example, many studies excluded individuals who had not taken glucose-lowering medications, or those who had diabetes for a certain length of time. Information on quality of participant dietary patterns and management of diabetes were rarely provided in the included studies but have a large impact on the effect of a supplement such as RJ. These factors are all important when considering the external validity of the synthesized evidence for clinical application.
The main limitation associated with animal studies is the physiological variability between animals and humans. Rodent studies, which comprise a considerable portion of the evidence, are good models of T2D in humans, but lack key pathologies in the disease, including pathologies found in pancreatic islets, the secretion site of insulin[8]. Moreover, when investigating the effects of an oral agent such as RJ, bioavailability is critical: absorption, distribution and metabolism of nutrients such as fatty acids found in RJ may vary between rodents and humans[16]. Our preliminary research indicates that bioavailability and metabolism of RJ in human models has not yet been established, so the enhanced response of RJ observed in animal trials may not be applicable to human patients.
Another limitation stems from the similarity between aspects of some studies. A considerable number of the included studies share the same authors, such as Pourmoradian et al[13], Pourmoradian et al[30], Mobasseri et al[28], and Mobasseri et al[25]. As a result, there is noteworthy overlap in the methodology of these studies, although different outcomes are assessed in each. In maintaining very similar study population and methodology, there is the potential for similar undetected bias to affect the results of these studies. Because it presents the risk of the overall evidence misrepresenting RJ’s true effect, overlapping methodology between studies is an important factor to consider when interpreting the evidence.
The evidence synthesized in this review relied completely on the included studies, which may be unrepresentative of RJ’s true effect on the probed outcomes. The exclusion of non-English studies may have removed a considerable number of studies; this is a concern particularly for this topic because almost all the included studies have non-English speaking origins. Also, despite having known insulin-like properties and thus potentially a role in glycemic regulation, no studies on RJ proteins were included in this review[56]. Lastly, while this review focused on RJ as a dietary supplement, other forms of administration (e.g., topical) may improve effectiveness or bioavailability.
In conclusion, RJ supplementation presents promising potential for treatment of glycemic T2D symptoms. The evidence synthesized in this review complements existing research that demonstrates other therapeutic effects of RJ administration in T2D symptoms, such as oxidative stress, impaired wound-healing and inflammation[32,57,58]. Future studies should examine the pharmacodynamic properties of RJ, particularly with respect to dosage forms, effectiveness and bioavailability in different populations to further elucidate the effectiveness of RJ as a therapeutic agent of hyperglycemia.
Existing evidence suggests that royal jelly (RJ) is a promising therapeutic option in hyperglycemic cases. Few studies have specifically examined the clinical viability of RJ as treatment, and no study has critically analyzed the existing evidence. Knowledge of the factors that influence effectiveness of RJ intake provides an alternative treatment for hyperglycemia, which is often associated with diabetes.
This systematic review demonstrated that the intervention style (e.g., length of supplementation, ingestion form) as well as pre-existing patient characteristics may be important factors in its effectiveness, and future research should further investigate these factors to inform patients and health care providers.
This review sought to examine whether there is support for RJ as a glycemic regulator in models of type 2 diabetes as well as healthy individuals. Our analysis found that the existing evidence suggests that RJ is a promising therapeutic option in hyperglycemic cases, with effective doses as low as 1000 mg of fresh RJ daily for diabetic patients.
This was a systematic review employing the PRISMA strategy. Five databases were searched using keywords pertinent to the research objectives. Two reviewers conducted full-text screening to select included articles that met eligibility criteria. Relevant information (i.e., intervention style, results, participant characteristics) was extracted from the included articles. Risk of bias was assessed by two reviewers. GRADE, a novel tool developed by Cochrane used to assess overall quality of evidence, was also determined by two reviewers.
Effective doses of RJ may be as low as 1000 mg of fresh RJ for a diabetic patient. Overall, the quality of evidence for RJ as a treatment is low for long-term effectiveness, and very low for acute effects of RJ consumption.
Synthesis and analysis of existing studies shows that RJ may be viable as part of a treatment plan in lowering blood sugar. Due to the heterogeneity in studied population and intervention, RJ may have more pronounced effects in certain dosage forms (e.g., fresh RJ) and in certain populations (e.g., postmenopausal females). This information may be useful for individuals and health care practitioners wishing to explore hyperglycemia treatment options.
Future clinical trials should consider the potential effects of intervention form and length, as well as the effect of participant characteristics to clarify which patient populations or conditions would benefit most from RJ supplementation.
Manuscript source: Unsolicited manuscript
Specialty type: Endocrinology and metabolism
Country of origin: Canada
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Koch TR S- Editor: Ji FF L- Editor: A E- Editor: Song H
| 1. | Chen L, Magliano DJ, Zimmet PZ. The worldwide epidemiology of type 2 diabetes mellitus--present and future perspectives. Nat Rev Endocrinol. 2011;8:228-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1333] [Cited by in RCA: 1445] [Article Influence: 103.2] [Reference Citation Analysis (0)] |
| 2. | Zimmet P, Alberti KG, Shaw J. Global and societal implications of the diabetes epidemic. Nature. 2001;414:782-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3804] [Cited by in RCA: 3689] [Article Influence: 153.7] [Reference Citation Analysis (0)] |
| 3. | Vijan S. In the clinic. Type 2 diabetes. Ann Intern Med. 2010;152:ITC31-15; quiz ITC316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 80] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 4. | Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9344] [Cited by in RCA: 8965] [Article Influence: 426.9] [Reference Citation Analysis (1)] |
| 5. | Reusch JE, Manson JE. Management of Type 2 Diabetes in 2017: Getting to Goal. JAMA. 2017;317:1015-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 107] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 6. | Khunti K, Bodicoat DH, Davies MJ. Type 2 diabetes: lifetime risk of advancing from prediabetes. Lancet Diabetes Endocrinol. 2016;4:5-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 7. | Takikawa M, Kumagai A, Hirata H, Soga M, Yamashita Y, Ueda M, Ashida H, Tsuda T. 10-Hydroxy-2-decenoic acid, a unique medium-chain fatty acid, activates 5'-AMP-activated protein kinase in L6 myotubes and mice. Mol Nutr Food Res. 2013;57:1794-1802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 8. | Tundis R, Loizzo MR, Menichini F. Natural products as alpha-amylase and alpha-glucosidase inhibitors and their hypoglycaemic potential in the treatment of diabetes: an update. Mini Rev Med Chem. 2010;10:315-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 399] [Cited by in RCA: 475] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 9. | Chen C, Chen SY. Changes in protein components and storage stability of royal jelly under various conditions. Food Chem. 1995;54:195-200. [RCA] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 45] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Honda Y, Fujita Y, Maruyama H, Araki Y, Ichihara K, Sato A, Kojima T, Tanaka M, Nozawa Y, Ito M, Honda S. Lifespan-extending effects of royal jelly and its related substances on the nematode Caenorhabditis elegans. PLoS One. 2011;6:e23527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 11. | Suzuki KM, Isohama Y, Maruyama H, Yamada Y, Narita Y, Ohta S, Araki Y, Miyata T, Mishima S. Estrogenic activities of Fatty acids and a sterol isolated from royal jelly. Evid Based Complement Alternat Med. 2008;5:295-302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 12. | Khoshpey B, Djazayeri S, Amiri F, Malek M, Hosseini AF, Hosseini S, Shidfar S, Shidfar F. Effect of Royal Jelly Intake on Serum Glucose, Apolipoprotein A-I (ApoA-I), Apolipoprotein B (ApoB) and ApoB/ApoA-I Ratios in Patients with Type 2 Diabetes: A Randomized, Double-Blind Clinical Trial Study. Can J Diabetes. 2016;40:324-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 13. | Pourmoradian S, Mahdavi R, Mobasseri M, Faramarzi E, Mobasseri M. Effects of royal jelly supplementation on body weight and dietary intake in type 2 diabetic females. Health Promot Perspect. 2012;2:231-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 14. | Ghanbari E, Nejati V, Khazaei M. Improvement in Serum Biochemical Alterations and Oxidative Stress of Liver and Pancreas following Use of Royal Jelly in Streptozotocin-Induced Diabetic Rats. Cell J. 2016;18:362-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 15. | Zamami Y, Takatori S, Goda M, Koyama T, Iwatani Y, Jin X, Takai-Doi S, Kawasaki H. Royal jelly ameliorates insulin resistance in fructose-drinking rats. Biol Pharm Bull. 2008;31:2103-2107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 16. | Baker DH. Animal models in nutrition research. J Nutr. 2008;138:391-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 72] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 17. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52948] [Cited by in RCA: 47199] [Article Influence: 2949.9] [Reference Citation Analysis (0)] |
| 18. | Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA; Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18487] [Cited by in RCA: 24863] [Article Influence: 1775.9] [Reference Citation Analysis (3)] |
| 19. | O'Connor AM, Sargeant JM. Critical appraisal of studies using laboratory animal models. ILAR J. 2014;55:405-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 20. | Schünemann HJ, Oxman AD, Brozek J, Glasziou P, Jaeschke R, Vist GE, Williams JW, Kunz R, Craig J, Montori VM, Bossuyt P, Guyatt GH; GRADE Working Group. Grading quality of evidence and strength of recommendations for diagnostic tests and strategies. BMJ. 2008;336:1106-1110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 970] [Cited by in RCA: 1168] [Article Influence: 68.7] [Reference Citation Analysis (0)] |
| 21. | Ryan R, Hill S; Cochrane consumers and communication group. How to GRADE the quality of the evidence. Cochrane consumers and communication group. 2016; Available from: URL: https://cc.cochrane.org/sites/cc.cochrane.org/files/public/uploads/how_to_grade.pdf. |
| 22. | Wei D, Tang K, Wang Q, Estill J, Yao L, Wang X, Chen Y, Yang K. The use of GRADE approach in systematic reviews of animal studies. J Evid Based Med. 2016;9:98-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 23. | Faraone SV. Interpreting estimates of treatment effects: implications for managed care. P T. 2008;33:700-711. [PubMed] |
| 24. | Iaconelli A, Gastaldelli A, Chiellini C, Gniuli D, Favuzzi A, Binnert C, Macé K, Mingrone G. Effect of oral sebacic Acid on postprandial glycemia, insulinemia, and glucose rate of appearance in type 2 diabetes. Diabetes Care. 2010;33:2327-2332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | Mobasseri M, Ghiyasvand S, Ostadrahimi A, Ghojazadeh M, Noshad H, Pourmoradian S. Effect of Fresh Royal Jelly Ingestion on Glycemic Response in Patients With Type 2 Diabetes. Iran Red Crescent Med J. 2015;17:e20074. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 26. | Münstedt K, Bargello M, Hauenschild A. Royal jelly reduces the serum glucose levels in healthy subjects. J Med Food. 2009;12:1170-1172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 27. | Münstedt K, Böhme M, Hrgovic I, Hauenschild A. An approach to the application of Royal Jelly: Encapsulation of lyophilized Royal Jelly and its effect on glucose metabolism in humans. J Api Product Api Med Sci. 2010;2:29-30. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 28. | Mobasseri M, Pourmoradian S, Mahdavi R, Faramarzi E. Effects of royal jelly supplementation on lipid profile and high-sensitivity c-reactive protein levels in type-2 diabetic women: A pilot study. Curr Top Nutraceutical Res. 2014;12:101-105. |
| 29. | Morita H, Ikeda T, Kajita K, Fujioka K, Mori I, Okada H, Uno Y, Ishizuka T. Effect of royal jelly ingestion for six months on healthy volunteers. Nutr J. 2012;11:77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 30. | Pourmoradian S, Mahdavi R, Mobasseri M, Faramarzi E, Mobasseri M. Effects of royal jelly supplementation on glycemic control and oxidative stress factors in type 2 diabetic female: a randomized clinical trial. Chin J Integr Med. 2014;20:347-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 31. | Shidfar F, Jazayeri S, Mousavi SN, Malek M, Hosseini AF, Khoshpey B. Does Supplementation with Royal Jelly Improve Oxidative Stress and Insulin Resistance in Type 2 Diabetic Patients? Iran J Public Health. 2015;44:797-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 32. | Fujii A, Kobayashi S, Kuboyama N, Furukawa Y, Kaneko Y, Ishihama S, Yamamoto H, Tamura T. Augmentation of wound healing by royal jelly (RJ) in streptozotocin-diabetic rats. Jpn J Pharmacol. 1990;53:331-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 94] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 33. | Membrez M, Chou CJ, Raymond F, Mansourian R, Moser M, Monnard I, Ammon-Zufferey C, Mace K, Mingrone G, Binnert C. Six weeks' sebacic acid supplementation improves fasting plasma glucose, HbA1c and glucose tolerance in db/db mice. Diabetes Obes Metab. 2010;12:1120-1126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 34. | Yoneshiro T, Kaede R, Nagaya K, Aoyama J, Saito M, Okamatsu-Ogura Y, Kimura K, Terao A. Royal jelly ameliorates diet-induced obesity and glucose intolerance by promoting brown adipose tissue thermogenesis in mice. Obes Res Clin Pract. 2018;12:127-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 35. | Watadani R, Kotoh J, Sasaki D, Someya A, Matsumoto K, Maeda A. 10-Hydroxy-2-decenoic acid, a natural product, improves hyperglycemia and insulin resistance in obese/diabetic KK-Ay mice, but does not prevent obesity. J Vet Med Sci. 2017;79:1596-1602. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 36. | Yoshida M, Hayashi K, Watadani R, Okano Y, Tanimura K, Kotoh J, Sasaki D, Matsumoto K, Maeda A. Royal jelly improves hyperglycemia in obese/diabetic KK-Ay mice. J Vet Med Sci. 2017;79:299-307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 37. | Messia MC, Caboni MF, Marconi E. Storage stability assessment of freeze-dried royal jelly by furosine determination. J Agric Food Chem. 2005;53:4440-4443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 38. | Maroni A, Moutaharrik S, Zema L, Gazzaniga A. Enteric coatings for colonic drug delivery: state of the art. Expert Opin Drug Deliv. 2017;14:1027-1029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 39. | Bushra R, Aslam N, Khan AY. Food-drug interactions. Oman Med J. 2011;26:77-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 140] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 40. | Louet JF, LeMay C, Mauvais-Jarvis F. Antidiabetic actions of estrogen: insight from human and genetic mouse models. Curr Atheroscler Rep. 2004;6:180-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 213] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 41. | Ahmed MA, Hassanein KM. Effects of estrogen on hyperglycemia and liver dysfunction in diabetic male rats. Int J Physiol Pathophysiol Pharmacol. 2012;4:156-166. [PubMed] |
| 42. | Yonei Y, Shibagaki K, Tsukada N, Nagasu N, Inagaki Y, Miyamoto K, Suzuki O, Kiryu Y. Case report: haemorrhagic colitis associated with royal jelly intake. J Gastroenterol Hepatol. 1997;12:495-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 43. | Laporte JR, Ibáãnez L, Vendrell L, Ballarín E. Bronchospasm induced by royal jelly. Allergy. 1996;51:440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 44. | Katayama M, Aoki M, Kawana S. Case of anaphylaxis caused by ingestion of royal jelly. J Dermatol. 2008;35:222-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 45. | Lee NJ, Fermo JD. Warfarin and royal jelly interaction. Pharmacotherapy. 2006;26:583-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 46. | Virkamäki A, Ueki K, Kahn CR. Protein-protein interaction in insulin signaling and the molecular mechanisms of insulin resistance. J Clin Invest. 1999;103:931-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 598] [Cited by in RCA: 571] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 47. | Morino K, Petersen KF, Shulman GI. Molecular mechanisms of insulin resistance in humans and their potential links with mitochondrial dysfunction. Diabetes. 2006;55 Suppl 2:S9-S15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 686] [Cited by in RCA: 614] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 48. | Boden G, Shulman GI. Free fatty acids in obesity and type 2 diabetes: defining their role in the development of insulin resistance and beta-cell dysfunction. Eur J Clin Invest. 2002;32 Suppl 3:14-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 890] [Cited by in RCA: 903] [Article Influence: 39.3] [Reference Citation Analysis (0)] |
| 49. | Mihaylova MM, Shaw RJ. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol. 2011;13:1016-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1847] [Cited by in RCA: 2321] [Article Influence: 165.8] [Reference Citation Analysis (0)] |
| 50. | Ojuka EO, Jones TE, Nolte LA, Chen M, Wamhoff BR, Sturek M, Holloszy JO. Regulation of GLUT4 biogenesis in muscle: evidence for involvement of AMPK and Ca(2+). Am J Physiol Endocrinol Metab. 2002;282:E1008-E1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 155] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 51. | Habegger KM, Hoffman NJ, Ridenour CM, Brozinick JT, Elmendorf JS. AMPK enhances insulin-stimulated GLUT4 regulation via lowering membrane cholesterol. Endocrinology. 2012;153:2130-2141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 101] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 52. | Minokoshi Y, Shiuchi T, Lee S, Suzuki A, Okamoto S. Role of hypothalamic AMP-kinase in food intake regulation. Nutrition. 2008;24:786-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 53. | Anderson KA, Ribar TJ, Lin F, Noeldner PK, Green MF, Muehlbauer MJ, Witters LA, Kemp BE, Means AR. Hypothalamic CaMKK2 contributes to the regulation of energy balance. Cell Metab. 2008;7:377-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 296] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 54. | Mauvais-Jarvis F, Clegg DJ, Hevener AL. The role of estrogens in control of energy balance and glucose homeostasis. Endocr Rev. 2013;34:309-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 732] [Cited by in RCA: 918] [Article Influence: 76.5] [Reference Citation Analysis (0)] |
| 55. | Zheng HQ, Hu FL, Dietemann V. Changes in composition of royal jelly harvested at different times: consequences for quality standards. Apidologie. 2011;42:39-47. [DOI] [Full Text] |
| 56. | Kramer KJ, Childs CN, Spiers RD, Jacobs RM. Purification of insulin-like peptides from insect haemolymph and royal jelly. Insect Biochem. 1982;12:91-98. [RCA] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 29] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 57. | El-Nekeety AA, El-Kholy W, Abbas NF, Ebaid A, Amra HA, Abdel-Wahhab MA. Efficacy of royal jelly against the oxidative stress of fumonisin in rats. Toxicon. 2007;50:256-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 72] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 58. | Kohno K, Okamoto I, Sano O, Arai N, Iwaki K, Ikeda M, Kurimoto M. Royal jelly inhibits the production of proinflammatory cytokines by activated macrophages. Biosci Biotechnol Biochem. 2004;68:138-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 160] [Article Influence: 7.6] [Reference Citation Analysis (0)] |