Published online Apr 26, 2020. doi: 10.4252/wjsc.v12.i4.277
Peer-review started: December 1, 2019
First decision: January 13, 2020
Revised: March 19, 2020
Accepted: March 26, 2020
Article in press: March 26, 2020
Published online: April 26, 2020
Processing time: 147 Days and 10.9 Hours
Premature ovarian insufficiency (POI) and premature ovarian failure (POF) have become one of the major problems threatening women of childbearing age. Studies have shown that stem cells transplanted from bone marrow, umbilical cord, peripheral blood and amniotic fluid can migrate and proliferate to the ovary, promote ovarian function repair, increase the number of follicles and granulosa cells at all levels of ovary, improve endocrine function, and can differentiate into oocytes in specific ovarian environment to restore fertility to some extent.
To study the ability of human umbilical cord mesenchymal stem cells (hUCMSCs) to repair ovarian injury after chemotherapy.
A total of 110 female BALB/c mice (aged 7-8 wk old) with body masses of 16.0-20.0 g were selected. The mice were fed until 12 wk of age, and cyclophosphamide was administered by intraperitoneal injection for 14 consecutive days to induce premature ovarian failure in mice. Seventy-five mice with estrous cycle disorder were screened and randomly divided into 3 groups according to their body weight: model group, positive control group and hUCMSC group, and each group had 25 mice. Another 25 mice were used as negative controls. The mice in the hUCMSC group were injected with hUCMSCs in the tail vein, and the mice in the positive control group were given an oestradiol valerate solution and a medroxyprogesterone acetate solution in the tail vein. On the 1st, 15th, 30th, 45th, and 60th days after intravenous administration, vaginal smears were made to monitor the estrous cycles of the mice. The ovaries were weighed, and pathological sections were made to observe the morphology of the follicles; blood samples were collected to monitor the concentration of sex hormones (oestradiol and follicle-stimulating hormone).
The estrous cycles of the model group mice were disrupted throughout the experiment. Mice in the hUCMSC group and the positive control group resumed normal estrous cycles. The ovarian weight of the model group mice continued to decline. The ovarian weight of the hUCMSC group mice and the positive control group mice decreased first and then gradually increased, and the ovarian weight of the hUCMSC group mice was heavier than that of the positive control group mice. The difference was statistically significant (P < 0.05). Compared with the negative control group, the model group experienced a decrease in oestradiol and an increase in follicle-stimulating hormone, and the difference was statistically significant (P < 0.05). Compared with the model group, the hUCMSC and positive control groups experienced a slight increase in oestradiol and a decrease in follicle-stimulating hormone; the difference was statistically significant (P < 0.05). The pathological examination revealed that the mouse ovaries from the model group were atrophied, the volume was reduced, the cortical and medullary structures were disordered, the number of follicles at all stages was significantly reduced, the number of atretic follicles increased, the number of primordial follicles and corpus luteum significantly decreased, and the corpus luteum had an irregular shape. Compared with those of the model group, the lesions of the hUCMSC and positive control groups significantly improved.
hUCMSCs can repair ovarian tissue damaged by chemotherapy to a certain extent, can improve the degree of apoptosis in ovarian tissue, and can improve the endocrine function of mouse ovaries.
Core tip: Human umbilical cord mesenchymal stem cells (hUCMSCs) can repair ovarian tissue damaged by chemotherapy to a certain extent, can improve the degree of apoptosis in ovarian tissue, and can improve the endocrine function of mouse ovaries. The effect of hUCMSCs on chemotherapy-induced premature ovarian failure mice was observed by closely monitoring the changes in mouse ovarian structure and endocrine function to further verify whether hUCMSCs can be used in the treatment of chemotherapy-induced premature ovarian failure.
- Citation: Shen J, Cao D, Sun JL. Ability of human umbilical cord mesenchymal stem cells to repair chemotherapy-induced premature ovarian failure. World J Stem Cells 2020; 12(4): 277-287
- URL: https://www.wjgnet.com/1948-0210/full/v12/i4/277.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v12.i4.277
Premature ovarian insufficiency (POI) and premature ovarian failure (POF) have become one of the major problems threatening women of childbearing age. These disorders can not only cause female reproductive endocrine dysfunction[1,2], but also seriously affect the physical and mental health and the quality of life of women of reproductive age[3]. At present, the treatment widely used for premature ovarian failure is mainly hormone replacement therapy (HRT). However, HRT mainly improves clinical symptoms, and it is impossible to fundamentally treat or repair damaged ovarian tissue[4], and long-term application of HRT can easily lead to side effects such as cardiovascular and cerebrovascular diseases, so HRT has not yet achieved satisfactory therapeutic effects. In recent years, with the deepening of research on mesenchymal stem cells (MSCs), MSC therapy has been considered as a treatment for repairing damaged tissues and reconstructing normal tissue function. Some of the research results have been applied in clinical trials, such as malignant tumours, autoimmune diseases, and cardiovascular diseases[5,6]. Related experimental studies have shown that MSCs can be colonized in ovarian tissue and can migrate towards the damaged tissue, thereby partially repairing the drug-induced ovarian damage caused by chemotherapy[7,8]. Therefore, the treatment of premature ovarian failure has also made some progress. According to animal experiments, human umbilical cord mesenchymal stem cells (hUCMSCs) have many advantages, such as easy collection, low immunogenicity, no tumourigenicity and no ethical restrictions[9-11], thus bringing hope to improve ovarian function and restore fertility in patients with POI/POF. In this study, an animal model of chemotherapy-induced premature ovarian failure was constructed. The effect of hUCMSCs on chemotherapy-induced premature ovarian failure mice was observed by closely monitoring the changes in mouse ovarian structure and endocrine function to further verify whether hUCMSCs can be used in the treatment of chemotherapy-induced premature ovarian failure.
hUCMSCs were provided by Shenyang Cell Therapy Engineering Technology Research and Development Center Co., Ltd.
A total of 110 SPL-level BALB/c female mice, aged 7-8 wk old with a body mass of 16.0-20.0 g, were purchased from Liaoning Changsheng Biotechnology Co., Ltd., Animal Quality Certificate No. 211002300048121, Animal License No. SCXK (Liao) 2015-0001. The mice were housed in the SPF-class animal room of the Animal Experimental Center at the Heping Campus of Northern War Zone General Hospital. The mice were grouped in 5 mice per cage, were free to eat and drink and were exposed to a day/night discontinuous illumination (12 h:12 h). The food, litter, water and cages used for feeding were strictly disinfected and sterilized.
Cyclophosphamide for injection (Jiangsu Disheng); oestradiol valerate tablets (Guangzhou Xianling); medroxyprogesterone acetate tablets (Zhejiang Xianju Pharma); sodium chloride injection (Huaren Pharmaceutical); mouse oestrogen 2 (E2) kit (Shanghai Guang Rui Biological Technology Co., Ltd., LOT 201905, RET ER2619); mouse follicle-stimulating hormone (FSH) kit (Shanghai Guang Rui Biological Technology Co., Ltd., LOT 201905, RET ER2936).
OLYMPUS Optical Microscope (OLYMPUS, Japan); Microscopic Image Analysis Software (Image-Pro, USA); rotary slicer, automatic dewatering machine, biological tissue embedding machine, automatic dyeing machine, spreading film drying machine (LEICA, Germany); Z236K Centrifuge (HERMLE); Nu-437-400E Biosafety Cabinet (Tianmei Technology Co., Ltd., China); BS224S Electronic Analytical Balance (Sartorius, Germany).
hUCMSCs were provided by Shenyang Cell Therapy Engineering Technology Research and Development Center Co., Ltd., and the samples in the public library are selected using strict and accurate detection methods. For the hUCMSCs used in this experiment, from the original sample collection, cell separation, stem cell (line) establishment, to the final cell preparation and release, all processes are in line with the "Good Manufacturing Practices" requirements, including personnel, materials, instruments and equipment, and environment management and control.
After raising SPF-class BALB/c mice to 12 wk of age, the mice were subjected to vaginal smear observation for 7 consecutive days. We observed under an optical microscope and determined the animal's estrous cycle based on cell classification. A total of 110 mice with a normal estrous cycle were randomly divided into a negative control group (25 mice) and a model group (85 mice). A model of chemotherapy-induced POF was established for the mice in the model group by intraperitoneal injection of cyclophosphamide for a first-time loading of 50 mg/kg, followed by a continuous intraperitoneal injection of 8 mg/(kg·d) for 14 d[12]. A vaginal smear was prepared and observed every day from day 4 to day 10 after administration of cyclophosphamide. Seventy-five mice with estrous cycle disorder were screened and randomly divided into the following three groups according to their body weight with 25 mice in each group: model group, hUCMSC group and positive control group. In the negative control group and the model group, the mice were injected with sodium chloride in the tail vein, 0.2 mL each; the hUCMSC group was injected with hUCMSCs at a concentration of 1 × 106 cells/mL in the tail vein, and the injection volume was 0.2 mL each. The injection was performed once a week for 4 wk. The positive control group was injected with 0.012 mg/mL oestradiol valerate solution in the tail vein in a volume of 10 mL/kg body weight. On day 4, the mice were given a tail vein injection of 0.096 mg/mL solution of medroxyprogesterone acetate in a volume of 10 mL/kg body weight (this is considered as a course of treatment). Subsequently, the two drugs were discontinued for 1 d, and then the next course of treatment was repeated for 4 wk.
Mouse vaginal smear and estrous cycle: The mice were observed once a day at 8:00 am, and the cotton swabs were taken lightly from the vaginal secretions to make a smear. After air drying, an appropriate amount of methylene blue dye solution was added dropwise. After 1 to 2 min, the mixture was rinsed with water, dried naturally and placed under a microscope.
Ovarian weighing and pathological section observation of follicular morphology: Five mice were sacrificed on the 1st, 15th, 30th, 45th and 60th days after intravenous administration, ovarian specimens were taken, and the ovaries were weighed. Paraffin-embedded sections were routinely prepared and stained with haematoxylin-eosin, and the ovarian structure was observed under a light microscope.
Determination of sex hormone (E2 and FSH) concentration: On the 1st, 15th, 30th, 45th and 60th day after intravenous administration, blood was collected from the eyelids, with heparin as an anticoagulant. After centrifugation for 15 min, the upper serum was taken and stored in a refrigerator at -80 °C. The E2 and FSH concentrations were determined at the elective stage.
Statistical processing was performed using SPSS 17.0 software. For those who conform to the normal distribution, the measurement data are expressed as the mean ± SD using one-way analysis of variance; data that do not conform to the normal distribution are statistically analysed using nonparametric tests. P < 0.05 indicates that the difference was statistically significant.
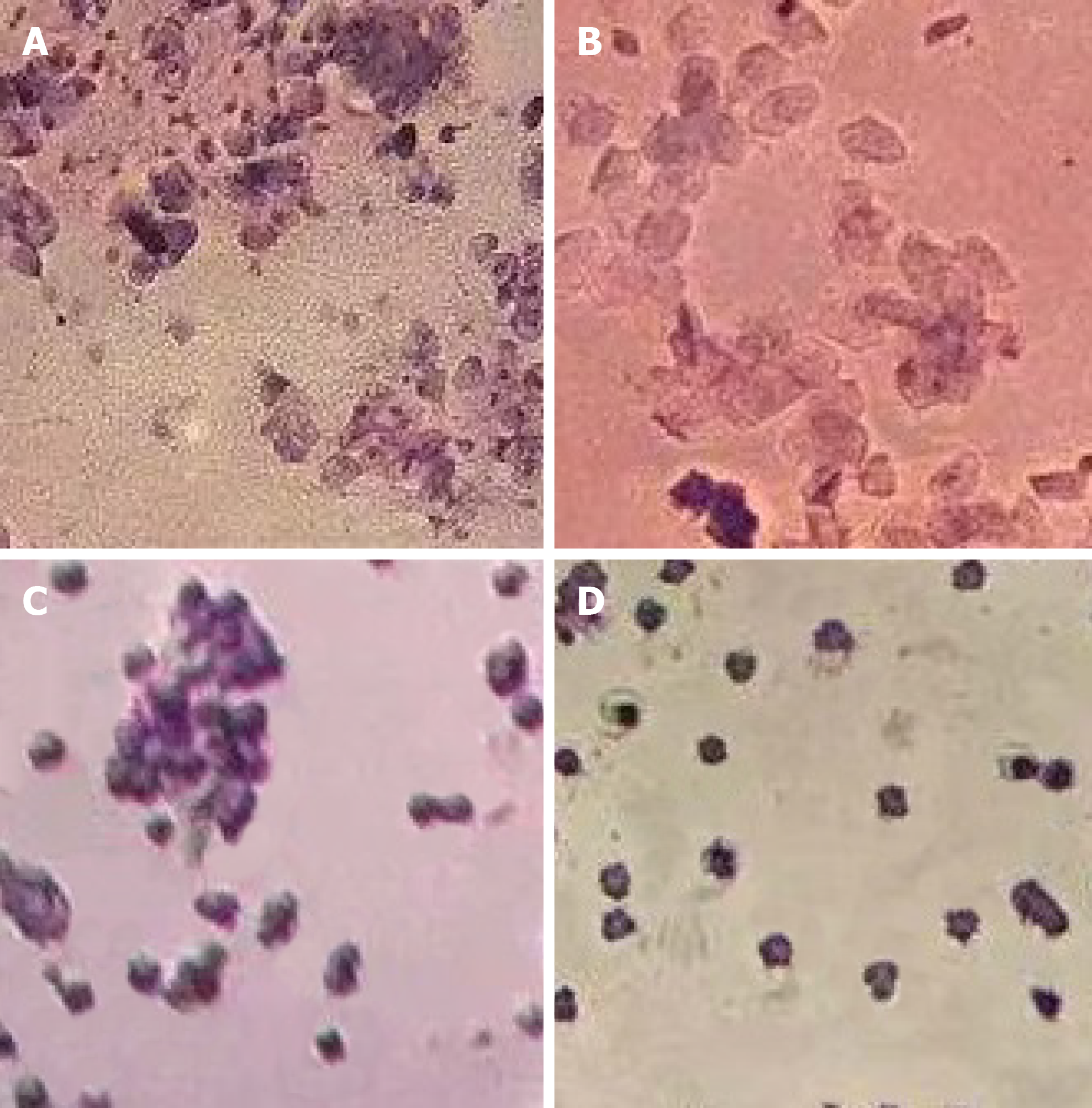
The estrous cycle of normal mice is approximately 3 to 5 d and includes the proestrus, oestrus, late oestrus and anestrous stages (Figure 1). The estrous cycle can be identified by changes in the cells in the vagina: The early stage of oestrus is characterized by small, round, squamous epithelial cells (nucleated epithelial cells); oestrus is characterized by irregular (keratinized) squamous epithelial cells; the late stage of oestrus is characterized by white blood cells and keratinocytes; and the anestrous stage is characterized by the appearance of white blood cells and round epithelial cells. In the experiment, the oestrus cycle of the normal control group did not change significantly and was the normal estrous cycle; the estrous cycle of the model group mice continued to be disordered. Within 1 to 15 d after cell transplantation, the estrous cycle of the hUCMSC group and the positive control group remained unresolved, and they were all in a disordered state. After 16 to 30 d, the hUCMSC group and the positive control group began to gradually return to the normal estrous cycle. Although the number of mice in the hUCMSC group that returned to the normal estrous cycle was greater than that in the positive control group, the difference was not statistically significant, as shown in Table 1.
| Groups | 1-15 d | 16-30 d | 31-45 d | 46-60 d |
| Negative control group | 20 | 15 | 10 | 5 |
| Model group | 0 | 0 | 0 | 0 |
| hUCMSC group | 0 | 7 | 4 | 3 |
| Positive control group | 0 | 6 | 4 | 4 |
Compared with that of the negative control group, the ovarian weights of the model, hUCMSC and positive control groups were significantly decreased on the first day and the 15th day after transplantation, and the difference was statistically significant (P < 0.05). There was no significant difference between the 3 groups (P > 0.05). On the 30th, 45th and 60th days after transplantation, the ovarian weight of the model group continued to decrease, and the ovarian weight of the hUCMSC group and the positive control group continued to increase. The difference was statistically significant (P < 0.05). There was no significant difference between the hUCMSC group and the positive control group (P > 0.05, Table 2).
| Items | After transplantation | ||||
| 1 d | 15 d | 30 d | 45 d | 60 d | |
| Negative control group | 36.67 ± 0.68 | 36.97 ± 0.32 | 37.45 ± 0.35 | 34.86 ± 0.43 | 38.13 ± 0.35 |
| Model group | 23.19 ± 0.98 | 20.23 ± 0.49 | 18.30 ± 0.28 | 18.02 ± 0.41 | 17.25 ± 0.28 |
| hUCMSC group | 21.19 ± 0.65 | 19.54 ± 0.27 | 21.77 ± 0.48 | 23.11 ± 0.38 | 24.28 ± 0.48 |
| Positive control group | 22.50 ± 0.62 | 19.30 ± 0.37 | 24.53 ± 0.93 | 27.64 ± 0.34 | 28.06 ± 0.93 |
| P value | P < 0.05 | ||||
Before the model group was established, there was no significant difference in the basic concentrations of the sex hormones E2 and FSH between the groups (P > 0.05). After the model group was established, the E2 concentration of the model group, the hUCMSC group and the positive control group decreased, and the FSH concentration increased, while the negative control group E2 and FSH concentrations fluctuated within the normal range. The difference between the 3 groups (model group, hUCMSC group, and the positive control group) and the negative control group was statistically significant (P < 0.05) and continued until the end of the experiment. On the 1st day after transplantation, the E2 concentration in the model group, hUCMSC group and positive control group was significantly different from that in the negative control group (P < 0.05). There was no significant difference between the 3 groups (P > 0.05). On the 15th day after transplantation, the E2 and FSH concentrations in the hUCMSC group and the positive control group were significantly different from those in the model group (P < 0.05). There was no significant difference between the hUCMSC group and the positive control group (P > 0.05). On the 30th, 45th, and 60th days after transplantation, the E2 and FSH concentrations in the hUCMSC group and the positive control group were significantly different from those in the model group (P < 0.05). There was no significant difference between the hUCMSC group and the positive control group (P < 0.05, Table 3 and Table 4).
| Items | Before transplantation | After transplantation | ||||
| 1 d | 15 d | 30 d | 45 d | 60 d | ||
| Negative control group | 56.94 ± 4.43 | 51.42 ± 2.35 | 53.99 ± 2.73 | 53.43 ± 3.26 | 52.04 ± 1.65 | 55.43 ± 1.57 |
| Model group | 55.23 ± 4.35 (P = 0.147) | 35.41 ± 2.25 | 28.32 ± 1.51 | 24.86 ± 0.90 | 22.16 ± 1.25 | 18.99 ± 1.40 |
| hUCMSC group | 55.83 ± 3.35 (P = 0.325) | 37.84 ± 1.69 | 38.33 ± 1.79 | 35.04 ± 2.31 | 35.12 ± 1.47 | 34.10 ± 1.42 |
| Positive control group | 55.03 ± 3.43 (P = 0.357) | 37.34 ± 1.86 | 37.80 ± 1.83 | 36.86 ± 2.09 | 36.86 ± 1.11 | 37.08 ± 1.47 |
| P value | P > 0. 05 | |||||
| Items | Before transplantation | After transplantation | ||||
| 1 d | 15 d | 30 d | 45 d | 60 d | ||
| Negative control group | 8.61 ± 0.03 | 9.25 ± 0.35 | 8.64 ± 0.56 | 9.27 ± 0.36 | 8.93 ± 0.26 | 9.11 ± 0.10 |
| Model group | 8.76 ± 0.17 (P = 0.19) | 18.33 ± 0.79 | 20.36 ± 0.74 | 22.38 ± 1.05 | 24.26 ± 1.15 | 25.19 ± 0.96 |
| hUCMSC group | 8.70 ± 0.38 (P = 0.93) | 19.44 ± 1.23 | 14.07 ± 0.53 | 15.52 ± 0.57 | 18.22 ± 1.14 | 18.55 ± 0.66 |
| Positive control group | 8.66 ± 0.45 (P = 0.10) | 18.5 ± 0.31 | 12.50 ± 0.63 | 12.39 ± 0.03 | 13.50 ± 1.04 | 14.28 ± 0.64 |
| P value | P > 0. 05 | |||||
In the negative control group, the ovary volume was larger, and the ovarian cortex medulla was clear. It was found that the follicles at all levels were active, arranged regularly, and developed normally. The corpus luteum was well developed, and the luteal cells were abundant. The model group had obvious ovarian atrophy, decreased volumes, and disordered cortical and medullary structures. The number of follicles at all levels was significantly reduced, and the number of atresia follicles increased. Only a small number of primordial follicles, ovarian oocytes and zona pellucida were abnormal. The corpus luteum number was significantly reduced, and the volume was reduced. The shape was not regular. The ovarian volume of the positive control group did not decrease significantly. The medullary structure of the ovarian cortex was slightly better than that of the model control group. The number of follicles at each level decreased, and the atresia follicles remained larger. The corpus luteum number decreased, the volume decreased, and the morphology was irregular. The ovarian volume of the hUCMSC group was slightly lower than that of the negative control group and the cortical medulla was structurally disordered but superior to that of the model group. The number of follicles at each level was reduced, the number of atresia follicles was significantly reduced, and the number of corpus luteum was slightly reduced, but the morphology was not regular (Figure 2).
With the development of modern science and technology, cancer treatment methods are increasing, and chemotherapy and radiotherapy treatments are widely used. Although the therapeutic effect of the disease is improved and the lifetime limit of patients is prolonged, the adverse consequences of chemotherapy radiotherapy for women of childbearing age also make patients troubled. According to some surveys, approximately 5% of women of childbearing age in the world are diagnosed with cancer[13-16]. After active treatment, they will face serious complications, such as ovarian insufficiency and even POF. Coupled with the fast pace of life and high work pressure, the incidence of POI has become younger, affecting more than 10% of women[17]. POI is a clinical syndrome in which women's ovarian activity declines before the age of 40, with menstrual disorders (such as menopause or thin menstruation), ovarian atrophy, low levels of oestrogen in the body, and gonadotropin up to the level of menopause[18,19]; if progression continues, ovarian failure before the age of 40 can lead to POF. POI and POF are caused by a variety of reasons, and their aetiology and pathogenesis are more complicated. The pathogenesis of this is not fully understood. Commonly known causes include genetic factors (number or structural abnormalities of X chromosomes), iatrogenic factors (such as radiotherapy and chemotherapy, surgery and other tumour treatments), infection factors, immune factors, etc.[20,21].
Stem cells are a class of early undifferentiated cells with self-renewal ability and multi-directional differentiation potential, which not only have tissue regeneration and repair functions but also have low immunogenicity, immunomodulatory effects and no toxicity[22-25]. At the same time, after entering the human body, the stem cells have the characteristics of automatic migration and homing to the tissues of the damaged organs, thereby having the function of participating in repairing the damaged tissue structure[26]. hUCMSCs are MSCs, which are mainly isolated from the Wharton's jelly of the umbilical cord after the foetus is born. hUCMSCs have many advantages, such as convenient material extraction, a wide range of sources, isolation and culture in vitro, stable biological properties, no tumourigenicity and low immunogenicity[27]. There is no ethical problem in culturing MSCs with umbilical cord, and it does not cause extra pain to patients, and it is not easy to cause disease spread among people[27-30]. Due to the above characteristics, hUCMSCs have gradually become a new research hotspot of MSCs[31-36].
In patients with POF, ovarian failure and a decreased ovary reserve causes the amount of E2 production to be reduced in sinusoid follicles, so the negative feedback effect on the gonadal axis is weakened, resulting in increased levels of FSH and LH hormones secreted by the pituitary. This experiment examined the changes in serum E2 and FSH in each group of mice to verify whether hUCMSCs can improve the endocrine function of the ovary. The results showed that compared with the negative control group, the E2 of the model group decreased, and the FSH increased, and the difference was statistically significant (P < 0.05 or 0.01), indicating that the cyclophosphamide-induced OPF model was established. Compared with the model group, the hUCMSC group and the positive control group had an increase in E2 to a certain extent, and FSH decreased; the difference was statistically significant (P < 0.05 or 0.01), indicating that both hUCMSCs and HRT can improve the ovary and that the OPI can be restored to a certain extent. In the pathological examination of mouse ovarian tissue, the model group had obvious ovarian atrophy, decreased volume, a disordered cortical and medullary structure, the number of follicles at all levels was significantly reduced, and the number of atresia follicles increased; only a small amount of primordial follicles, the number of corpus luteum decreased significantly and the shape was not regular. The lesions of the hUCMSC group and the positive control group were significantly improved compared with those of the model group. The oestrus cycle of the model group mice continued to be disordered during the experiment. The hUCMSC group and the positive control group mice resumed the normal estrous cycle. The total number of follicles and the number of follicles at all stages in the mouse ovaries were lower in the model group than in the normal group, and the ovarian weight continued to decrease. The total number of follicles and the number of follicles at all levels in the hUCMSC group and the positive control group were higher than those in the model group, and the ovarian weight continued to increase; the difference was statistically significant (P < 0.05). The results indicated that hUCMSCs can repair damaged ovarian tissue to a certain extent, improve the degree of apoptosis in ovarian tissue, and improve the endocrine function of mouse ovaries.
In summary, hUCMSCs have a certain repair effect on damaged ovarian tissue after cyclophosphamide-induced chemotherapy damage and regulate endocrine secretion of hormones, thereby further slowing ovarian failure. It can provide new treatments for chemotherapy patients and provide a corresponding experimental basis for more clinical experimental research. However, the specific molecular mechanism requires further study.
Premature ovarian insufficiency (POI) and premature ovarian failure (POF) have become one of the major problems threatening women of childbearing age. Studies have shown that stem cells transplanted from bone marrow, umbilical cord, peripheral blood and amniotic fluid can migrate and proliferate to the ovary, promote ovarian function repair, increase the number of follicles and granulosa cells at all levels of ovary, improve endocrine function, and can differentiate into oocytes in specific ovarian environment to restore fertility to some extent.
According to animal experiments, human umbilical cord mesenchymal stem cells (hUCMSCs) have many advantages, such as easy collection, low immunogenicity, no tumourigenicity and no ethical restrictions, it bringing hope to improve ovarian function and restore fertility in patients with POI/POF.
In this study, the authors aimed to study the ability of hUCMSCs to repair ovarian injury after chemotherapy.
A total of 110 female BALB/c mice were fed until 12 wk of age, and cyclophosphamide was administered by intraperitoneal injection for 14 consecutive days to induce premature ovarian failure in mice. The mice in the hUCMSC group were injected with hUCMSCs in the tail vein, and the mice in the positive control group were given an oestradiol valerate solution and a medroxyprogesterone acetate solution in the tail vein.
Mice in the hUCMSC group and the positive control group resumed normal estrous cycles. The ovarian weight of the model group mice continued to decline. The ovarian weight of the hUCMSC group mice and the positive control group mice decreased first and then gradually increased, and the ovarian weight of the hUCMSC group mice was heavier than that of the positive control group mice. The model group experienced a decrease in oestradiol and an increase in follicle-stimulating hormone. The hUCMSC and positive control groups experienced a slight increase in oestradiol and a decrease in follicle-stimulating hormone. The pathological examination revealed that the mouse ovaries from the model group were atrophied, the volume was reduced, the cortical and medullary structures were disordered, the number of follicles at all stages was significantly reduced, the number of atretic follicles increased, the number of primordial follicles and corpus luteum significantly decreased, and the corpus luteum had an irregular shape. The lesions of the hUCMSC and positive control groups significantly improved.
hUCMSCs can repair ovarian tissue damaged by chemotherapy to a certain extent. And it can improve the degree of apoptosis in ovarian tissue, and improve the endocrine function of mouse ovaries.
The specific molecular mechanism requires further study.
Manuscript source: Unsolicited manuscript
Specialty type: Cell and tissue engineering
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Lynch HT, Tanabe S, van der Laan LJW S-Editor: Wang JL L-Editor: A E-Editor: Wu YXJ
| 1. | Chen LH, Gao FX, Sun AJ. [Research progress in the evaluation of ovarian reserve function]. Shengzhi Yixue Zazhi. 2014;3:330-333. [DOI] [Full Text] |
| 2. | Wilkosz P, Greggains GD, Tanbo TG, Fedorcsak P. Female reproductive decline is determined by remaining ovarian reserve and age. PLoS One. 2014;9:e108343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 3. | Sheikhansari G, Aghebati-Maleki L, Nouri M, Jadidi-Niaragh F, Yousefi M. Current approaches for the treatment of premature ovarian failure with stem cell therapy. Biomed Pharmacother. 2018;102:254-262. [PubMed] |
| 4. | Kramer A, Yaoliwasi A, Yuemaier M, Tuerhong M, Kadeer N, Yilike X. [The effect of chronic stress on the levels of E2, FSH, LH and β - EP in serum of rats with premature ovarian failure]. Zhonghua Zhongyiyao Xuekan. 2019;37:631-634, 778-779. [DOI] [Full Text] |
| 5. | Ullah I, Subbarao RB, Rho GJ. Human mesenchymal stem cells - current trends and future prospective. Biosci Rep. 2015;35:e00191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 715] [Cited by in RCA: 912] [Article Influence: 91.2] [Reference Citation Analysis (0)] |
| 6. | Arutyunyan I, Elchaninov A, Makarov A, Fatkhudinov T. Umbilical Cord as Prospective Source for Mesenchymal Stem Cell-Based Therapy. Stem Cells Int. 2016;2016:6901286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 174] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 7. | Wang Z, Huang KR, Wang YL, Wang W, Bai HY, Yang XY. [Localization of transplanted menstrual-derived stem cells in premature ovarian failure model of mice]. Xi’an Jiaotong Daxue Xuebao (Yixue Ban). 2017;38:803-808. [DOI] [Full Text] |
| 8. | Fu X, He Y, Xie C, Liu W. Bone marrow mesenchymal stem cell transplantation improves ovarian function and structure in rats with chemotherapy-induced ovarian damage. Cytotherapy. 2008;10:353-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 144] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 9. | Fazeli Z, Abedindo A, Omrani MD, Ghaderian SMH. Mesenchymal Stem Cells (MSCs) Therapy for Recovery of Fertility: a Systematic Review. Stem Cell Rev Rep. 2018;14:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 77] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 10. | Paladino FV, Peixoto-Cruz JS, Santacruz-Perez C, Goldberg AC. Comparison between isolation protocols highlights intrinsic variability of human umbilical cord mesenchymal cells. Cell Tissue Bank. 2016;17:123-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 11. | Buravkova LB, Andreeva ER, Gogvadze V, Zhivotovsky B. Mesenchymal stem cells and hypoxia: where are we? Mitochondrion. 2014;19 Pt A:105-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 104] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 12. | Fu X, He Y, Wang X, Peng D, Chen X, Li X, Wang Q. Overexpression of miR-21 in stem cells improves ovarian structure and function in rats with chemotherapy-induced ovarian damage by targeting PDCD4 and PTEN to inhibit granulosa cell apoptosis. Stem Cell Res Ther. 2017;8:187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 151] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 13. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9172] [Cited by in RCA: 9958] [Article Influence: 995.8] [Reference Citation Analysis (0)] |
| 14. | Mohri J, Katada C, Ueda M, Sugawara M, Yamashita K, Moriya H, Komori S, Hayakawa K, Koizumi W, Atsuda K. Predisposing Factors for Chemotherapy-induced Nephrotoxicity in Patients with Advanced Esophageal Cancer Who Received Combination Chemotherapy with Docetaxel, Cisplatin, and 5-fluorouracil. J Transl Int Med. 2018;6:32-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Schizas D, Kapsampelis P, Mylonas KS MD. Adenosquamous Carcinoma of the Esophagus: A Literature Review. J Transl Int Med. 2018;6:70-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 16. | Michael JS, Lee BS, Zhang M, Yu JS. Nanotechnology for Treatment of Glioblastoma Multiforme. J Transl Int Med. 2018;6:128-133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 17. | Thakur M, Feldman G, Puscheck EE. Primary ovarian insufficiency in classic galactosemia: current understanding and future research opportunities. J Assist Reprod Genet. 2018;35:3-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 18. | Podfigurna-Stopa A, Czyzyk A, Grymowicz M, Smolarczyk R, Katulski K, Czajkowski K, Meczekalski B. Premature ovarian insufficiency: the context of long-term effects. J Endocrinol Invest. 2016;39:983-990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 138] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 19. | Barasoain M, Barrenetxea G, Huerta I, Télez M, Criado B, Arrieta I. Study of the Genetic Etiology of Primary Ovarian Insufficiency: FMR1 Gene. Genes (Basel). 2016;7:123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 20. | De Vos M, Devroy P, Fauser BC. Primary ovarian insufficiency. Lancet. 2010;376:911-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 502] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 21. | Fenton AJ. Premature ovarian insufficiency: Pathogenesis and management. J Midlife Health. 2015;6:147-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 22. | Niemeyer P, Kornacker M, Mehlhorn A, Seckinger A, Vohrer J, Schmal H, Kasten P, Eckstein V, Südkamp NP, Krause U. Comparison of immunological properties of bone marrow stromal cells and adipose tissue-derived stem cells before and after osteogenic differentiation in vitro. Tissue Eng. 2007;13:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 136] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 23. | Le Blanc K, Ringdén O. Immunomodulation by mesenchymal stem cells and clinical experience. J Intern Med. 2007;262:509-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 512] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 24. | Lazarov O, Marr RA. Neurogenesis and Alzheimer's disease: at the crossroads. Exp Neurol. 2010;223:267-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 236] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 25. | Volarevic V, Gazdic M, Simovic Markovic B, Jovicic N, Djonov V, Arsenijevic N. Mesenchymal stem cell-derived factors: Immuno-modulatory effects and therapeutic potential. Biofactors. 2017;43:633-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 26. | Kern S, Eichler H, Stoeve J, Klüter H, Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells. 2006;24:1294-1301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2248] [Cited by in RCA: 2347] [Article Influence: 123.5] [Reference Citation Analysis (0)] |
| 27. | Zhang YY, Yin HQ, Ni F, Jiang H. [Transplatation of bone marrow mesenchymal stem cells for reconstruction of the ovarian function in the premature ovarian insufficiency mice]. Anhui Nongye Daxue Xuebao. 2017;44:44-49. [DOI] [Full Text] |
| 28. | Zhao D, Zhao PW, Wu HB, Cai XY, Sun LP, Tao SY, Niu JZ, Lu D, Qi MY. [Effect of Erxian Decoction on proliferation and cycle of ovarian granulosa cells in cisplatin induced premature ovarian failure model]. Huanqiu Zhongyiyao. 2017;10:131-136. |
| 29. | Zhang LJ, Tao SY, Zhao PW, Mao XL. [Experimental study of cisplatin in the establishment of rat model of premature ovarian failure]. Shijie Zhongyiyao. 2015;10:1563-1565, 1569. [DOI] [Full Text] |
| 30. | Prasad S, Gupta T, Divya A. Correlation of the Day 3 FSH/LH Ratio and LH Concentration in Predicting IVF Outcome. J Reprod Infertil. 2013;14:23-28. [PubMed] |
| 31. | Fan C, Wang D, Zhang Q, Zhou J. Migration capacity of human umbilical cord mesenchymal stem cells towards glioma in vivo. Neural Regen Res. 2013;8:2093-2102. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 32. | Li YK, Wang H, Jiang CG, Huang H, Liu J, Wang YX, Xiao FJ, Wu ZZ, Xu ZJ. [Therapeutic effects and the underlying mechanism of umbilical cord-derived mesenchymal stem cells for bleomycin induced lung injury in rats]. Zhonghua Jie He He Hu Xi Za Zhi. 2013;36:808-813. [PubMed] |
| 33. | Elfayomy AK, Almasry SM, El-Tarhouny SA, Eldomiaty MA. Human umbilical cord blood-mesenchymal stem cells transplantation renovates the ovarian surface epithelium in a rat model of premature ovarian failure: Possible direct and indirect effects. Tissue Cell. 2016;48:370-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 69] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 34. | Liew A, O'Brien T, Egan L. Mesenchymal stromal cell therapy for Crohn's disease. Dig Dis. 2014;32 Suppl 1:50-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Conconi MT, Burra P, Di Liddo R, Calore C, Turetta M, Bellini S, Bo P, Nussdorfer GG, Parnigotto PP. CD105(+) cells from Wharton's jelly show in vitro and in vivo myogenic differentiative potential. Int J Mol Med. 2006;18:1089-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 36. | Liu S, Jia Y, Yuan M, Guo W, Huang J, Zhao B, Peng J, Xu W, Lu S, Guo Q. Repair of Osteochondral Defects Using Human Umbilical Cord Wharton's Jelly-Derived Mesenchymal Stem Cells in a Rabbit Model. Biomed Res Int. 2017;2017:8760383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |