Published online Apr 21, 2021. doi: 10.3748/wjg.v27.i15.1616
Peer-review started: January 28, 2021
First decision: February 24, 2021
Revised: March 5, 2021
Accepted: March 29, 2021
Article in press: March 29, 2021
Published online: April 21, 2021
Processing time: 75 Days and 15.2 Hours
Pancreatic ductal adenocarcinoma (PDAC) is the fourth leading cause of death among cancers, it is characterized by poor prognosis and strong chemoresistance. In the PDAC microenvironment, stromal cells release different extracellular components, including CXCL12. The CXCL12 is a chemokine promoting the communication between tumour and stromal cells. Six different splicing isoforms of CXCL12 are known (α, β, γ, δ, ε, θ) but their role in PDAC has not yet been characterized.
To investigate the specific role of α, β, and γ CXCL12 isoforms in PDAC onset.
We used hTERT-HPNE E6/E7/KRasG12D (Human Pancreatic Nestin-Expressing) cell line as a pancreatic pre-tumour model and exposed it to the α, β, and γ CXCL12 isoforms. The altered expression profiles were assessed by microarray analyses and confirmed by Real-Time polymerase chain reaction. The functional enrichment analyses have been performed by Enrichr tool to highlight Gene Ontology enriched terms. In addition, wound healing assays have been carried out to assess the phenotypic changes, in terms of migration ability, induced by the α, β, and γ CXCL12 isoforms.
Microarray analysis of hTERT-HPNE cells treated with the three different CXCL12 isoforms highlighted that the expression of only a few genes was altered. Moreover, the α and β isoforms showed an alteration in expression of different genes, whereas γ isoform affected the expression of genes also common with α and β isoforms. The β isoform altered the expression of genes mainly involved in cell cycle regulation. In addition, all isoforms affected the expression of genes associated to cell migration, adhesion and cytoskeleton. In vitro cell migration assay confirmed that CXCL12 enhanced the migration ability of hTERT-HPNE cells. Among the CXCL12 splicing isoforms, the γ isoform showed higher induction of migration than α and β isoforms.
Our data suggests an involvement and different roles of CXCL12 isoforms in PDAC onset. However, more investigations are needed to confirm these preliminary observations.
Core Tip: In the microenvironment of pancreatic ductal adenocarcinoma (PDAC), stromal cells release different extracellular components, including CXCL12, in order to communicate with cancer cells. Here, we investigated the specific role of α, β, and γ CXCL12 splicing isoforms in PDAC onset, by using a pre-tumour model. Microarray analysis suggested a role of CXCL12 in cell migration, and wound healing assays confirmed this hypothesis. In particular, γ isoform showed the highest promotion of migration. Our results shed light on the molecular basis of PDAC onset and progression.
- Citation: Cecati M, Giulietti M, Righetti A, Sabanovic B, Piva F. Effects of CXCL12 isoforms in a pancreatic pre-tumour cellular model: Microarray analysis. World J Gastroenterol 2021; 27(15): 1616-1629
- URL: https://www.wjgnet.com/1007-9327/full/v27/i15/1616.htm
- DOI: https://dx.doi.org/10.3748/wjg.v27.i15.1616
Pancreatic ductal adenocarcinoma (PDAC) is one of the most aggressive gastrointestinal tumours with a 5-year survival rate of 7%[1]. Due to the lack of early symptoms and specific diagnostic markers of early-stage disease, PDAC is often diagnosed at an advanced stage. At the time of diagnosis, the 90% of patients already presents advanced tumour progression and distant metastasis, therefore the surgical treatment is no longer applicable. This tumour also shows a high resistance rate to anti-tumour drugs, so chemio- and radio-therapy are not effective[2,3]. Progressive accumulation of genetic mutations, causing activation of different signalling pathways, drives tumour growth and development[4]. The earliest genetic event in the progression of the normal ductal epithelial cells to premalignant pancreatic intraepithelial neoplasia (PanIN) is the mutation of the K-Ras oncogene[5]. The activation of K-Ras protein triggers various downstream effector proteins which promote proliferation, metabolic reprogramming, anti-apoptosis, evasion of the immune response and remodelling of the microenvironment[6]. Recently, increasing interest has been focused on the PDAC microenvironment. The PDAC microenvironment is composed of a large portion of stroma surrounding cancer cells and contains cells such as cancer-associated fibroblasts (CAFs), T cells, stellate cells, macrophages, regulatory T cells, endothelial cells and others[7-9]. To maintain a favourable microenvironment for cancer cell survival, stromal cells secrete extracellular components, such as extracellular matrix (ECM), matrix metallo-proteinases, growth factors, transformation growth factor-β and cytokines.
One of the above-mentioned molecules is the chemokine CXCL12 (C-X-C motif chemokine ligand 12), a low molecular weight protein (about 12 kDa), belonging to the CXC chemokine family. In PDAC, CXCL12 is a key messenger in the intercellular communication between tumour and stromal cells[10]. In fact, CXCL12 promotes tumour proliferation, epithelial to mesenchymal transition, metastases, angiogenesis and immunosurveillance[10,11]. In PDAC setting, it acts through highly expressed receptors namely CXCR4 and CXCR7[11]. Although six different CXCL12 splicing variants have been described (α, β, γ, δ, ε, θ), only some properties of α, β and γ isoforms are known[12]. The sequence of the isoforms differ significantly one from another, thus also the function of the isoforms which are not yet characterized could be different or even contrary[12,13]. In breast cancer the role of the isoforms α, β, γ, and to a lesser extent δ, was studied[14,15], whereas in PDAC no information is available about the specific role played by each isoform of CXCL12. Moreover, often the papers focusing on CXCL12 role in PDAC actually report the results without stating which isoform in particular is under investigation[12].
The hTERT-HPNE E6/E7/KRasG12D (Human Pancreatic Nestin-Expressing) is a pancreatic pre-tumour cell line. These cells derive from normal pancreas duct epithelial cells and harbour the classical KRas (G12D) mutation, furthermore it bears inactivated p53 and Rb tumour suppressor genes. This status mimics the cells that have acquired the cancer predisposing mutations, but are not yet fully transformed. The phenotype of this cell line is similar to the third phase of the premalignant lesions, called PanIN-3[5,6,16].
Here, we investigated the transcriptomic alterations induced by treatments with the α, β and γ CXCL12 isoforms (the only ones commercially available) in a pancreatic pre-cancerous model in order to study PDAC onset. To this aim, we treated hTERT-HPNE cells with different CXCL12 isoforms, and assessed the gene expression profiles of these cells by microarray analyses. Since microarray analyses indicated several deregulated genes involved in cell migration, we also performed wound healing assays.
The human pancreatic pre-tumour cell line hTERT-HPNE E6/E7/K-RasG12D (ATCC® CRL-4038™) was purchased from American Type Culture Collection. These cells are adherent and have an epithelial-like morphology. Furthermore, they lack fundamental features of malignant pancreatic cancer cells, such as the anchorage independent growth in soft agar and the ability to engraft as tumours in athymic mice. This cell line was developed from human pancreatic duct cells by transduction of human telomerase gene (hTERT) in order to escape from cellular senescence. Further immortalization steps included the introduction of the human papillomavirus 16 E6 and E7 proteins, which are able to impair the function of key tumour suppressors p53 and Rb. In these cells, the KRAS G12D mutation is sufficient to induce formation of PanIN lesions, without inducing a fully malignant phenotype. Cells were maintained in high glucose Dulbecco’s Modified Eagle’s Medium (Lonza, Milan, Italy) supplemented with 10% fetal bovine serum, 1% L-Glutamine and 1% Penicillin/ Streptomycin (EuroClone, Milan, Italy). Cells were maintained at 37 °C in a humidified atmosphere of 5% CO2 in air.
A total of 2 × 106 hTERT-HPNE cells were seeded in p100 dishes for cell treatments. Once cells reached the right confluence, they were treated for 24 h with 100 ng/mL of each of the following isoforms: Human recombinant CXCL12-α (PHC1346, Thermo Fisher Scientific, Milan, Italy), human recombinant CXCL12-β (2716-SD, R&D Systems) and human recombinant CXCL12-γ (6448-SD-025, R&D Systems, Minneapolis, MN, United States). Cells cultured similarly for 24 h have been used as control. All experiments were carried out in biological triplicates.
Total RNA was isolated from harvested cells (12 samples in total) using the RNeasy Protect Cell Mini Kit (Qiagen, Milan, Italy) according to the manufacturer’s instructions. The RNA concentration in isolated samples was determined by ultraviolet absorption at 260 nm. The quality of total RNA was first assessed using an Agilent Bioanalyzer 2100 (Agilent Technologies, Palo Alto, CA, United States). Biotin-labelled cDNA targets were synthesized starting from 150 ng of total RNA. Double stranded cDNA synthesis and related cRNA was performed with GeneChip® WT Plus Kit (Affymetrix, Thermo Fisher Scientific, Milan, Italy). Same kit was used to synthesize the sense strand cDNA before fragmenting and labelling. All steps of the labelling protocol were performed as suggested by Affymetrix, starting from 5.5 μg of ssDNA.
Hybridization was performed using the GeneChip® Hybridization, Wash and Stain Kit (Affymetrix, Thermo Fisher Scientific, Milan, Italy). In particular, fragmented and labelled sscDNA were diluted in hybridization buffer at a concentration of 23 ng/μL for a 2.3 μg total and denatured at 99 °C for 5 min, incubated at 45 °C for 5 min and centrifuged at maximum speed for 1 min prior to introduction into the GeneChip® cartridge. A single GeneChip® Human Clariom S was then hybridized with each biotin-labelled sense target (12 samples in total). Hybridizations were performed for 16 h at 45 °C in a rotisserie oven (60 rpm). GeneChip® cartridges were washed and stained with GeneChip® Hybridization, Wash and Stain Kit in the Affymetrix Fluidics Station. GeneChip arrays were scanned using an Affymetrix GeneChip® Scanner 3000 7G using default parameters. Affymetrix GeneChip® Command Console software was used to acquire GeneChip® images and generate .DAT and .CEL files, which were used for subsequent analysis.
Analyses of the raw expression data (probe-level .CEL files) were carried out within R/Bioconductor environment (version 3.6 and 3.9, respectively). In particular, we applied the background correction and quantile normalization by employing the Robust Multi-array Average method available in the oligo R package[17]. Then, ComBat function from sva R package[18] was used to remove potential batch effects. Principal component analysis was used for the data quality control, before and after batch effect correction. For differential expression analysis, genefilter and limma R packages have been used[19]. In particular, for the identification of differentially expressed genes (DEGs), we considered the Benjamini-Hochberg corrected P value cut-off (FDR), set at 0.05, and a log2 Fold Change |log2FC| > 0.7.
Enrichr tool (http://amp.pharm.mssm.edu/Enrichr/)[20] was used to perform functional enrichment analysis of the DEGs, i.e., the identification of the most over-represented (enriched) Gene Ontology (GO) terms. In this tool, we selected the panels GO Biological Process, GO Molecular Function and GO Cellular Component. Only statistically significant results are reported (P < 0.05).
To confirm the microarray results, we assessed the expression of some DEGs by Real-Time polymerase chain reaction (PCR). We selected six genes (CXCL8, RRP12, ADH1B, UBA7, FEM1C, and CASP1) based on the commonly used criteria, that is their fold-changes in relative expression and P values[21]. For this purpose, we isolated RNA from hTERT-HPNE cells as described previously for microarray analysis. Then, RNA was reverse transcribed with HyperScript First Strand Synthesis Kit (GeneAll Biotechnology, Korea) using random primers to obtain complementary DNA (cDNA). The selected genes were run in duplicate by Real-Time PCR, using SYBR Green chemistry. The primer sequences are reported in Supplementary Table 1. All samples were tested in triplicate using GAPDH (glyceraldehyde-3-phosphate dehydrogenase) as the reference gene for data normalization to correct for variations in RNA quality and quantity. Threshold cycle (CT) values of genes of interest were normalized against CT values of GAPDH, and a relative fold change in expression with respect to a reference sample was calculated by the 2-ΔΔCt method.
To evaluate the cell migration capacity, hTERT-HPNE cells were seeded into a 24-well plate (2 × 104 cells/well) and allowed to attach and grow until reaching the 90%-100% confluence. Cell monolayers were scratched with a sterile 1000 μL pipette tip to make a wound. Then, wounded cell monolayers were washed with phosphate-buffered saline 1× to remove cell debris, and incubated for 24 h in DMEM with 100 ng/mL of each CXCL12 isoform. Cells were monitored under the Eclipse Ti2E microscope (Nikon, Tokyo, Japan) equipped with a camera (Hamamatsu Photonics, Japan) and photographed at 0 h, 12 h and 24 h. Each experiment was performed in triplicate and the data were presented as mean ± SD.
For Real-Time PCR and migration assays, differences in gene expression levels between the treated and untreated cells or difference in cell migration ability were determined using the t-test. P values less than 0.05 were considered statistically significant. All statistical analyses were performed by using the Stat6 Software for Windows (Stat6 Software, San Diego, CA, United States).
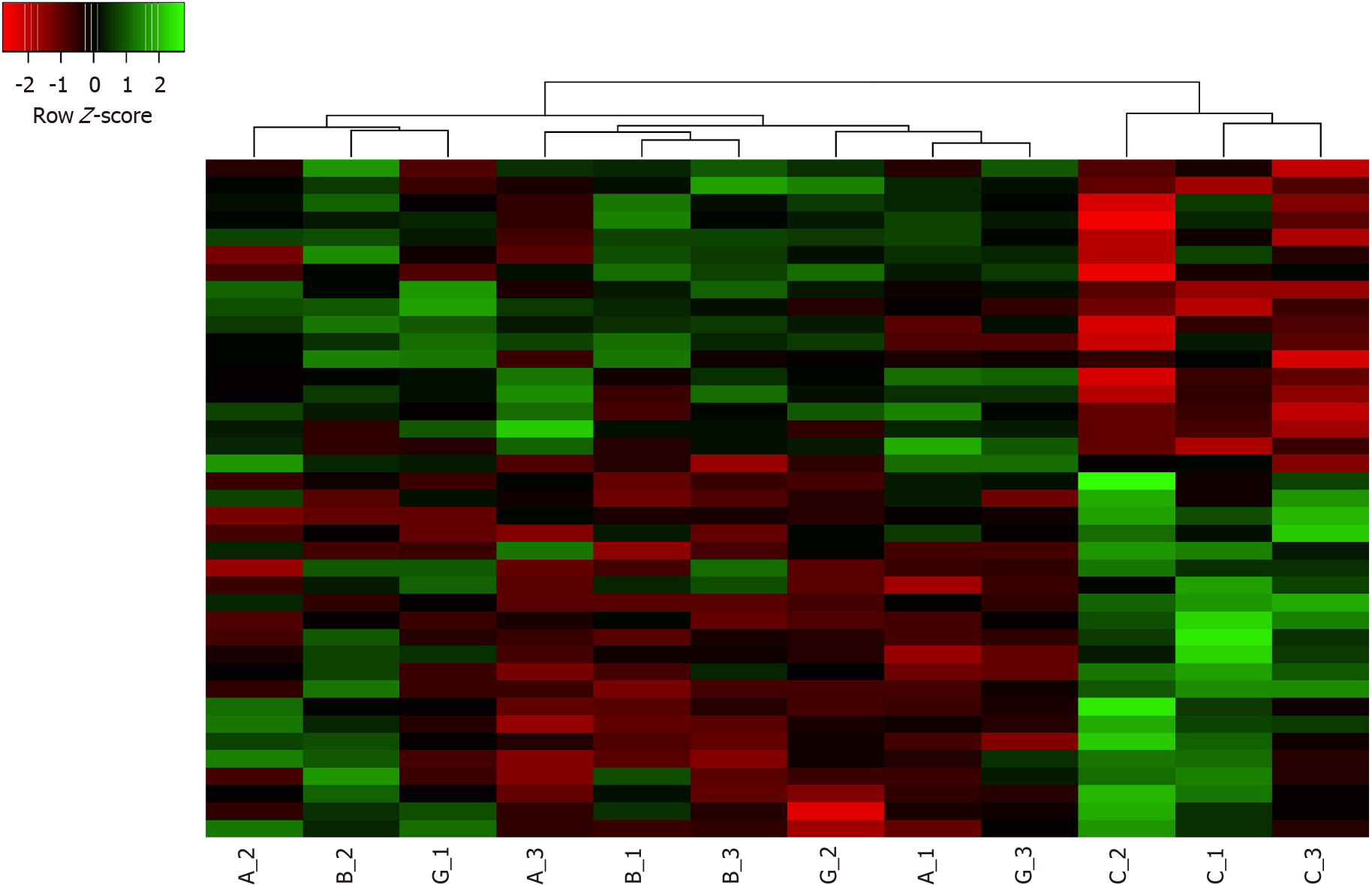
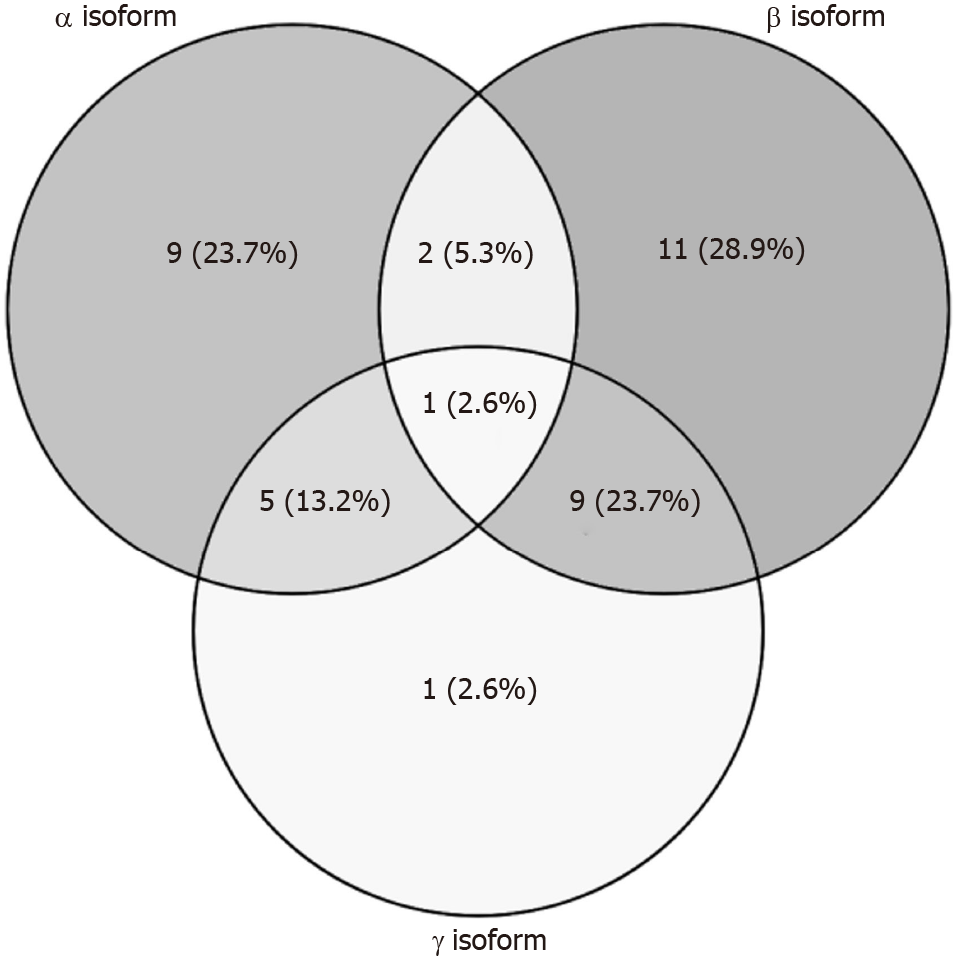
Gene expression alterations in pancreatic hTERT-HPNE cells treated with α, β or γ CXCL12 isoforms, compared to the untreated cells, have been assessed by microarray analyses on Affymetrix Human Clariom S Array. After data processing (i.e., quality control, normalization, batch effect removal), only a few genes resulted to be differentially expressed between treated and untreated cells. Indeed, pairwise comparisons between each treatment and the controls highlighted 17, 24 and 16 DEGs for α, β and γ isoforms, respectively (Table 1). This observation suggests that treatments with CXCL12 did not dramatically alter the gene expression of hTERT-HPNE cells and that, among tested isoforms, the β isoform may induce the highest effect on the transcriptome. Among DEGs regulated by α isoform, 7 were overexpressed and 10 were underexpressed; regarding β isoform, 12 genes were upregulated and 12 were downregulated; finally, for γ isoform, 2 genes were overexpressed and 14 were underexpressed. By hierarchical clustering, the expression levels of these DEGs allow to easily highlight control samples, but homogeneous clusters of samples treated with each CXCL12 isoform are not so clearly distinguishable (Figure 1). This result suggests that, although CXCL12 treatments alter gene expression in hTERT-HPNE cells, the specific effect of each isoform is quite difficult to identify. Therefore, we compared DEGs among different treatments, revealing common and exclusive genes (Figure 2). In particular, 10, 11 and 1 (i.e., LGR5) DEGs were exclusive for α, β and γ isoforms, respectively, suggesting that the effects on the transcriptome of γ isoform is intermediate between those induced by α and β isoforms. Indeed, α and β isoforms induced expression alteration in many different genes, whereas γ isoform affected the expression of genes also common with α and β isoforms (Figure 2).
| Probe name | Log2FC | P value | Adjusted P value | Gene name |
| CXCL12 α isoform vs control | ||||
| TC1100012755.hg.1 | -1.203589 | 3.12 × 10-8 | 0.000052 | KIRREL3 |
| TC1700010680.hg.1 | -1.036293 | 1.56 × 10-6 | 0.001479 | KRT14 |
| TC0200015894.hg.1 | -1.009543 | 1.37 × 10-6 | 0.001358 | DOCK10 |
| TC1700007330.hg.1 | -0.937268 | 9.64 × 10-6 | 0.007801 | KSR1 |
| TC0200007399.hg.1 | -0.920275 | 1.26 × 10-5 | 0.009220 | PLEKHH2 |
| TC1000011549.hg.1 | -0.908378 | 2.40 × 10-5 | 0.016397 | RRP12 |
| TC1100009990.hg.1 | -0.841133 | 5.45 × 10-5 | 0.032188 | OR6A2 |
| TC1000011923.hg.1 | -0.827117 | 7.17 × 10-5 | 0.038532 | GFRA1 |
| TC0300013528.hg.1 | -0.824444 | 7.59 × 10-5 | 0.038532 | GMNC |
| TC1800009242.hg.1 | -0.818366 | 1.03 × 10-4 | 0.049741 | SERPINB2 |
| TC0300007393.hg.1 | 0.807450 | 1.12 × 10-4 | 0.049980 | UBA7 |
| TSUnmapped00000150.hg.1 | 0.812168 | 1.05 × 10-4 | 0.049741 | OBP2B |
| TC0400008134.hg.1 | 0.841986 | 5.91 × 10-5 | 0.033939 | MMRN1 |
| TC1300010008.hg.1 | 0.854761 | 4.73 × 10-5 | 0.028684 | GAS6-AS1 |
| TC0100008815.hg.1 | 0.864724 | 1.12 × 10-4 | 0.049980 | IFI44L |
| TC1600010467.hg.1 | 0.873123 | 2.89 × 10-5 | 0.019144 | CNGB1 |
| TC0400011618.hg.1 | 0.958292 | 5.08 × 10-6 | 0.004269 | NEUROG2 |
| CXCL12 β isoform vs control | ||||
| TC0400007836.hg.1 | -1.317647 | 3.40 × 10-9 | 0.000008 | CXCL8 |
| TC0400011408.hg.1 | -1.007967 | 2.72 × 10-6 | 0.002057 | ADH1B |
| TC2100006784.hg.1 | -1.000559 | 9.12 × 10-7 | 0.000839 | JAM2 |
| TSUnmapped00000478.hg.1 | -0.944922 | 2.60 × 10-6 | 0.002052 | ZNF502 |
| TC0400006661.hg.1 | -0.799480 | 1.01 × 10-4 | 0.033702 | STK32B |
| TC0200014802.hg.1 | -0.797931 | 7.39 × 10-5 | 0.028291 | SCN3A |
| TC1000011549.hg.1 | -0.796631 | 8.77 × 10-5 | 0.030275 | RRP12 |
| TC0200015894.hg.1 | -0.792474 | 9.80 × 10-5 | 0.033299 | DOCK10 |
| TC0X00010399.hg.1 | -0.779084 | 1.41 × 10-4 | 0.042386 | BEX1 |
| TC0600014250.hg.1 | -0.776865 | 1.08 × 10-4 | 0.034696 | ZBED9 |
| TC0300007220.hg.1 | -0.767719 | 1.30 × 10-4 | 0.040552 | ZNF502 |
| TC0400009879.hg.1 | -0.764228 | 1.70 × 10-4 | 0.049472 | CYTL1 |
| TC0600013231.hg.1 | 0.768484 | 1.61 × 10-4 | 0.047319 | SGK1 |
| TC1300007248.hg.1 | 0.803510 | 8.35 × 10-5 | 0.029556 | CKAP2 |
| TC1100013221.hg.1 | 0.818187 | 4.63 × 10-5 | 0.019311 | CASP1 |
| TC0500012030.hg.1 | 0.819484 | 4.61 × 10-5 | 0.019311 | GDF9 |
| TC0300007393.hg.1 | 0.838000 | 3.14 × 10-5 | 0.014151 | UBA7 |
| TC0300012670.hg.1 | 0.853540 | 2.67 × 10-5 | 0.012705 | ATR |
| TC0100013700.hg.1 | 0.868308 | 2.28 × 10-5 | 0.011426 | CLSPN |
| TC0600007138.hg.1 | 0.884415 | 1.49 × 10-5 | 0.008040 | E2F3 |
| TC0600010241.hg.1 | 0.895254 | 9.02 × 10-6 | 0.005389 | KIF25 |
| TC0100008101.hg.1 | 0.951395 | 3.50 × 10-6 | 0.002479 | KIF2C |
| TC0500010150.hg.1 | 1.068352 | 1.53 × 10-7 | 0.000178 | DNAH5 |
| TC0500011752.hg.1 | 1.070992 | 1.39 × 10-7 | 0.000171 | FEM1C |
| CXCL12 γ isoform vs control | ||||
| TC1700010680.hg.1 | -1.080970 | 5.93 × 10-8 | 0.000105 | KRT14 |
| TC0400011408.hg.1 | -0.986416 | 1.69 × 10-5 | 0.011437 | ADH1B |
| TC0200015894.hg.1 | -0.932303 | 1.47 × 10-6 | 0.001696 | DOCK10 |
| TC0400007836.hg.1 | -0.928065 | 6.86 × 10-6 | 0.005275 | CXCL8 |
| TC0400006661.hg.1 | -0.899192 | 5.58 × 10-6 | 0.004432 | STK32B |
| TC2100006784.hg.1 | -0.897234 | 3.99 × 10-6 | 0.003542 | JAM2 |
| TC1000011549.hg.1 | -0.840455 | 1.83 × 10-5 | 0.012077 | GFRA1 |
| TC1100012755.hg.1 | -0.836071 | 2.44 × 10-5 | 0.015179 | KIRREL3 |
| TC1200008176.hg.1 | -0.818598 | 6.33 × 10-5 | 0.032429 | LGR5 |
| TC1700007330.hg.1 | -0.815796 | 2.93 × 10-5 | 0.017802 | KSR1 |
| TSUnmapped00000478.hg.1 | -0.787902 | 4.44 × 10-5 | 0.025573 | ZNF502 |
| TC1100009990.hg.1 | -0.785292 | 4.90 × 10-5 | 0.027540 | OR6A2 |
| TC0600014250.hg.1 | -0.774330 | 6.12 × 10-5 | 0.032045 | ZBED9 |
| TC0X00010399.hg.1 | -0.769031 | 1.00 × 10-4 | 0.047072 | BEX1 |
| TC0500011752.hg.1 | 0.893861 | 4.79 × 10-6 | 0.004094 | FEM1C |
| TC1100013221.hg.1 | 0.922373 | 2.14 × 10-6 | 0.002246 | CASP1 |
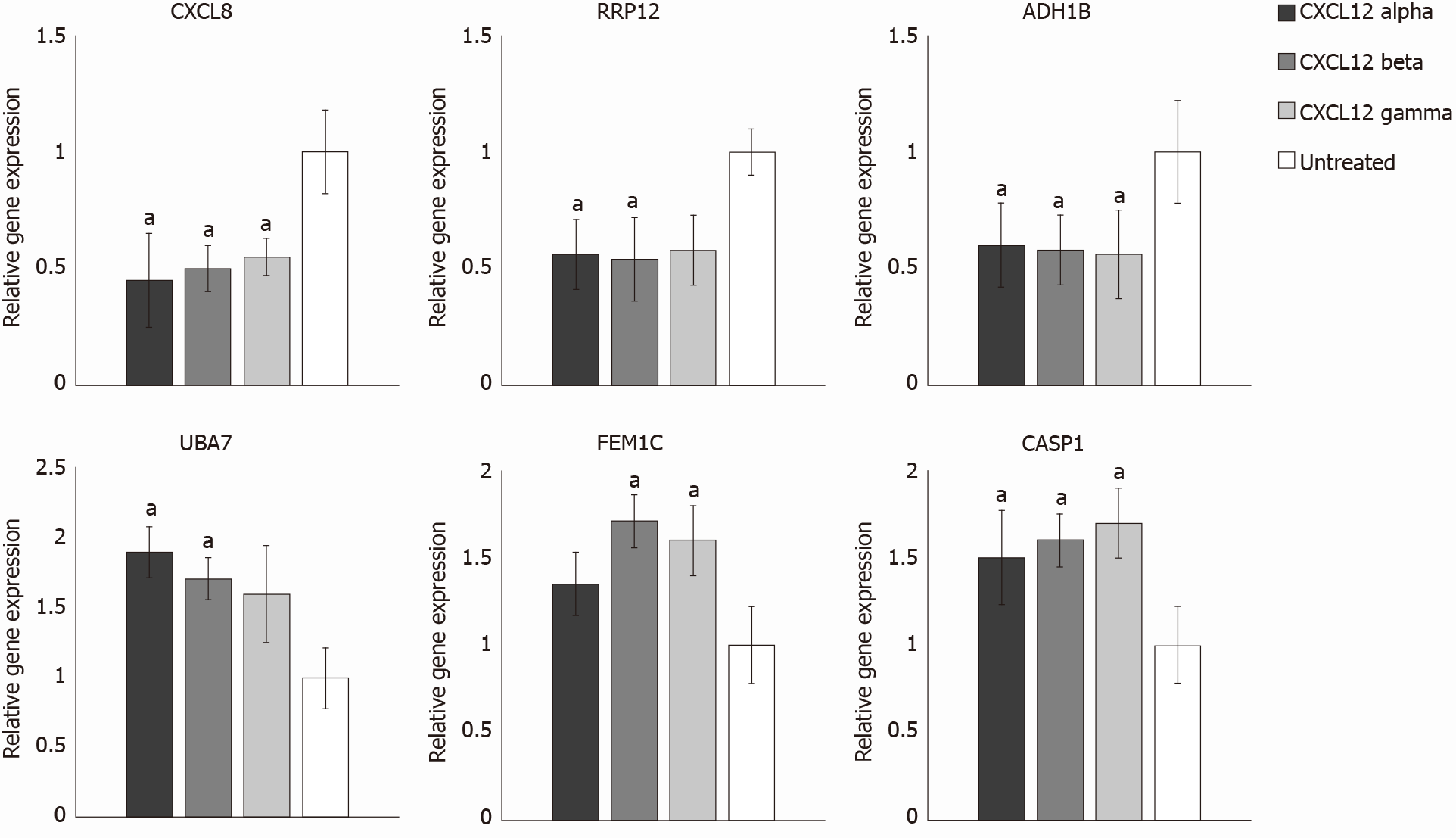
Real-Time PCR analysis was carried out on treated and control samples in order to validate microarray data. Among significant DEGs identified by microarray analysis, we chose 3 up-regulated (UBA7, FEM1C, CASP1) and 3 down-regulated (CXCL8, RRP12 and ADH1B) genes for Real-Time PCR validation. As reported in the Figure 3, the selected genes showed no discrepancies in their expression profiles, for each CXCL12 isoform, between microarray and Real-Time PCR. Indeed, for all isoforms, CXCL8, RRP12 and ADH1B genes resulted to be downregulated also in Real-Time PCR results. In particular, CXCL8, RRP12 and ADH1B genes showed an average fold change reduction of 2.01, 1.78 and 1.72, respectively. Regarding UBA7, FEM1C and CASP1 genes, Real-Time PCR confirmed that they were upregulated, on average, of 1.74, 1.55 and 1.60 times, respectively, after all CXCL12 isoform treatments.
The three lists of DEGs (i.e., for each CXCL12 isoform) have been submitted to Enrichr tool in order to provide an interpretation of the biological processes associated with these genes. This gene enrichment analysis has been performed considering GO terms, regarding GO Biological Process, GO Molecular Function and GO Cellular Component (Supplementary Table 2-4). Our analyses showed that β isoform seems to alter, more than α and γ isoforms the expression of genes involved in cell cycle regulation, DNA replication, G2/M checkpoints, p53 signalling pathway, regulation of apoptosis and senescence. In addition, enrichment analyses highlighted that, for all CXCL12 isoforms, most enriched GO terms involved cell migration and adhesion. Indeed, we often found terms such as "intermediate filament", "cytoskeleton", "microtubule", "kinesin complex", "motor activity", "tubulin binding", " positive chemotaxis", "hemidesmosome assembly", "cell-substrate junction assembly", "microtubule polymerization or depolymerisation", "regulation of cell motility", "regulation of actin filament depolymerisation" (Supplementary Tables 2-4). Since these terms seem to be uniformly distributed among the three CXCL12 isoforms, we decided to carry out in vitro assays to evaluate better the specific effect of each CXCL12 isoform on cell migration.
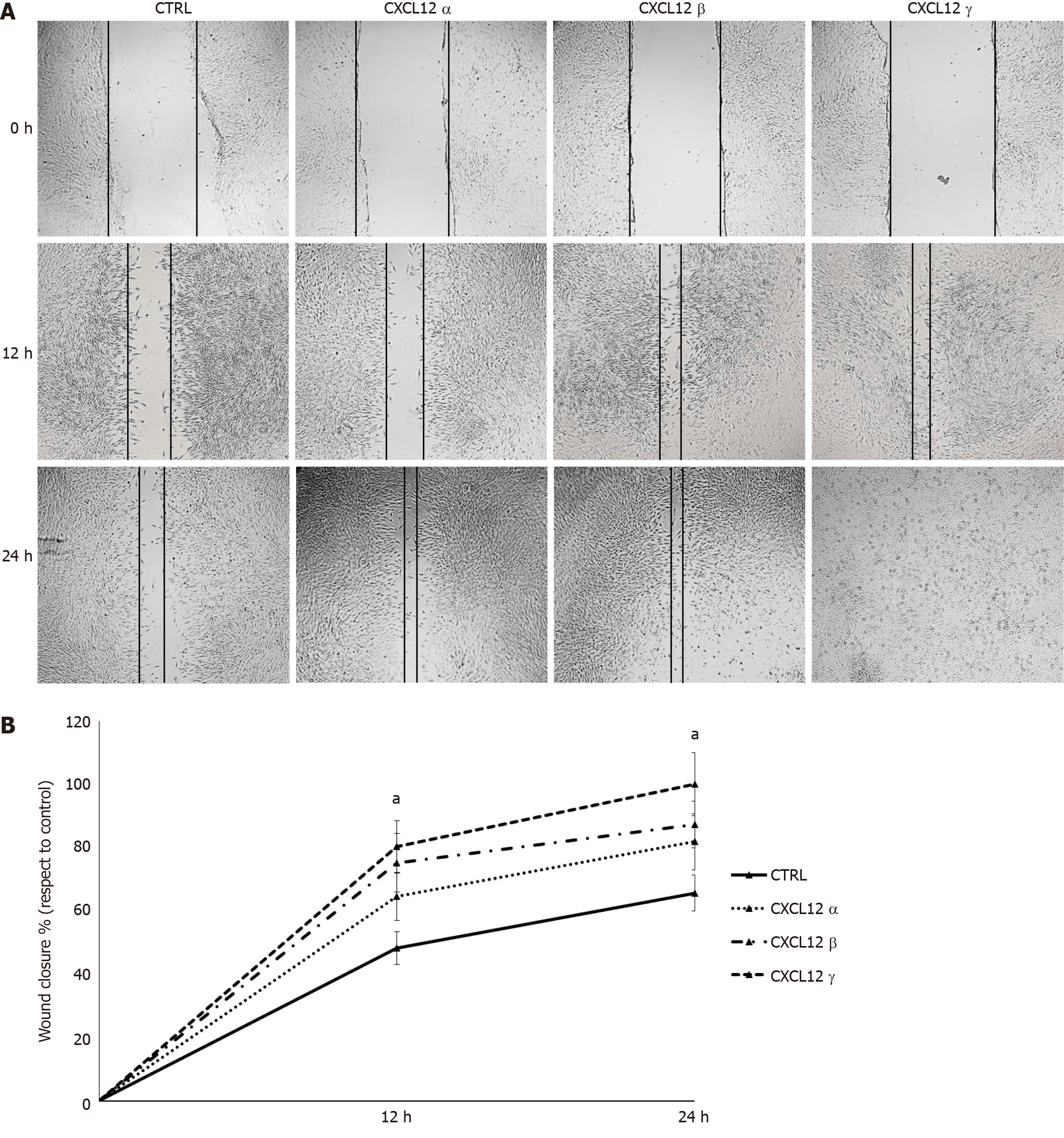
To assess the biological influence of each CXCL12 isoform on cell migration, hTERT-HPNE cells have been subjected to monolayer wound healing assay. Compared with untreated control, the migration ability of treated hTERT-HPNE cells significantly (P < 0.05) increased at 12 h and 24 h time-points after CXCL12 isoforms administration. In particular, the γ isoform increased (37% at 24 h) the hTERT-HPNE migration more than α (16% at 24 h) and β (22% at 24 h) CXCL12 isoforms (Figure 4). Our results confirmed the functional enrichment analyses on DEGs identified by microarray experiments.
The strong interaction between stroma and tumour cells is a typical characteristic of PDAC microenvironment. CAFs, representing the 50% of PDAC stroma, are involved in malignant progression, by releasing several chemokines such as CXCL12[12]. In cancer cells presenting the CXCL12 receptor (CXCR4), numerous signalling pathways are activated, which promote cell growth, proliferation, migration, invasion, metastasis and drug resistance[12]. In this study, we investigated the potential role of three different CXCL12 splicing isoforms in a pancreatic pre-tumour model. In particular, we used immortalized, epithelial-like pancreatic duct cells bearing a mutation in the KRAS gene (G12D), which is known to be present in 90% of low-grade PanIN-1 lesions. The G12D mutation triggers conformational changes that result in KRAS protein activation, which, in turn, can constitutively stimulate several effector pathways involved in tumour development. However, in pancreas, it leads only to a pre-malignant phenotype; indeed, further mutations, amplifications or inactivation of other genes are necessary for the tumour formation[5,6,16]. Therefore, this model has a great value as control in functional in vitro assays.
Our microarray data identified some DEGs in hTERT-HPNE cells upon treatment with different CXCL12 isoforms. The DEGs of each isoform were associated with cell migration, adhesion, and cytoskeleton. In vitro wound healing assays confirmed these gene expression results for all isoforms. Our data also showed that the treatment with CXCL12 γ isoform on hTERT-HPNE cells caused enhanced wound healing repair than α and β isoforms.
Similar previous results are not available, since the different CXCL12 isoforms have been usually evaluated in terms of their chemoattractant abilities, that is in in vitro chemotaxis assays with a CXCL12 gradient. In a study of Yu et al[22], β isoform was more efficient than the others, while in some other studies the isoform with the highest attractant ability was α isoform[23,24]. Furthermore, there are in other studies which determined that γ isoform induced greater migration than other isoforms[25,26]. In another in vitro chemotaxis assay, the use of different concentrations allowed to realize that α isoform induced greatest migration at low concentrations, whereas γ isoform at high concentrations[27]. It was also observed that the chemotactic responses of cells toward CXCL12 can be drawn as a bell-shaped curve with very low effects at both high and low concentrations of CXCL12 isoforms, maybe due the CXCR7 scavenging which alters the CXCL12 gradient to facilitate migration[25]. On the contrary, γ isoform drives chemotaxis to a much greater extent than α and β in in vivo studies. Indeed, γ isoform binds to the ECM components, with an extremely greater affinity than α isoform, allowing its local increase and so its activation by oligomerization[26,27].
Among genes with significantly altered expression upon treatments with the different CXCL12 isoforms, there are many genes already known to be involved in cell migration. Interestingly, this is the first study highlighting the possible genes targeted by different CXCL12 isoforms. For example, all CXCL12 isoforms were able to reduce the expression of DOCK10 gene, involved in several cellular processes, including the cellular migration. In particular, it regulates amoeboid motility[28] and it is able to induce filopodia and membrane ruffles[29]. In many tumour types, the overexpression of the receptor GFRA1 Leads to enhanced cancer cell proliferation and migration[30]. The GFRA1 downregulation caused by α and γ CXCL12 isoforms could highlight their potential anti-tumoral role. Both β and γ isoform affect the expression of the alcohol dehydrogenase gene (ADH1B), known to take part in several pathways promoting ovarian cancer cell infiltration[31]. Since CXCL12 β and γ isoform downregulate ADH1B gene, it suggests their possible anti-tumoral effect. Also JAM2 expression is downregulated by CXCL12 β and γ isoforms and, interestingly, JAM2 is known to affect cell invasion and migration abilities in pancreatic cancer cells[32]. GAS6-AS1 (GAS6 antisense RNA 1), overexpressed only upon treatments with the α isoform, promotes cancer cell growth, migration, and invasion ability in breast[33] and gastric cancer[34]. The CXCL12 β treatment induced overexpression of GDF9, CKAP2 and KIF2C. While GDF9 overexpression is correlated with a loss of the invasiveness, growth, and migration in human kidney cancer[35], CKAP2 overexpression increased cell proliferation, migration and invasion in HeLa cells[36]. Similarly to CKAP2, KIF2C mediates the cell migration in gastric cancer[37] and hepatocellular carcinoma[38]. Moreover, in KRas mutated cells, the knock down of KIF2C reduced the cell migration[39]. Our results showed that only CXCL12 γ isoform reduced the expression of LGR5. It is able to alter actin cytoskeleton, and in turn, reduce cell migration[40]. However, other studies demonstrated the central role of LGR5 in the tumour physiopathology, although its utility as a marker of cancer stem cells is still controversial[41].
Besides cell migration, CXCL12 treatments affected the expression of genes involved in cell cycle regulation, genome integrity, stroma remodelling, and inflammation. For example, only the CXCL12 β treatment induced overexpression of E2F3 and ATR genes. The upregulation of E2F3 gene, involved in regulation of cell cycle, promotes proliferation and progression of pancreatic cancer cells[42]. The DNA damage sensing by the ATR kinases plays critical role in the resistance to chemotherapeutic drugs. Indeed, the inhibition of ATR increased pancreatic cancer cells’ sensitivity to gemcitabine and radiation in vitro[43,44]. The SERPINB2 gene expression is downregulated only upon treatments with α isoform. Interestingly, since SERPINB2 mediates the remodelling of PDAC stroma, leading to the suppression of tumour growth and local invasion[45], α isoform may have a pro-tumoral activity in our hTERT-HPNE cell model. The expression of the pro-tumour KSR1 (Kinase suppressor of Ras-1) gene in our KRAS mutated pancreatic cells was downregulated upon treatments with CXCL12 α and γ isoforms. Interestingly, deletion of KSR1 prevented cell signalling, leading to the block of the transformation induced by mutated KRAS[46]. The inhibition of KSR1 gene determined a significant growth reduction of pancreatic tumours in vivo, as observed after injection of Panc-1 cells, a cellular model of KRAS-dependent PDAC, in nude mice[47]. Finally, CXCL12 β and γ isoforms downregulated the expression of the major metastasis-promoting inflammatory chemokine CXCL8 (also known as interleukin-8, IL-8). Indeed, KRAS mutation induces overexpression of CXCL8, which is necessary for cancer growth, vascularization and stromal remodelling[48]. Unlike CXCL12 β and γ isoforms, tumor necrosis factor-α, leukemia inhibitory factor, IL-1β, IL-6, and interferon-β induced the expression of CXCL8 in PDAC cells[49]. On the contrary, Matsuo et al[50] demonstrated that CXCL8 secretion by pancreatic cancer cell lines was significantly induced by CXCL12. However, unlike our experiments, these authors performed assays in a tumour model and without information about the involved CXCL12 isoform.
In conclusion, we showed that treatments with different CXCL12 isoforms prompt cell migration to different extents, probably due to different genes identified by our microarray analysis. Our results may facilitate the elucidation of the role of some CXCL12 isoforms in pancreatic cancer onset, and its underlying molecular mechanisms. In addition, the identified genes may represent novel candidates for diagnostic biomarkers and therapeutic targets for pancreatic cancer.
Pancreatic ductal adenocarcinoma (PDAC) is a highly lethal cancer type, since it is usually diagnosed late, it has a very poor prognosis and strong chemoresistance. In the tumour microenvironment, cancer cells and other cell types co-exist and communicate by exchanging several molecules, including the chemokine CXCL12.
CXCL12 pre-mRNA can be alternatively spliced into different isoforms (α, β, γ, δ, ε, θ). However, their specific roles in PDAC have not yet been fully described.
Here, we aim to evaluate the specific roles of the main CXCL12 isoforms (α, β, and γ) in PDAC onset.
We administered α, β, and γ CXCL12 isoforms to a pre-tumour model of PDAC, i.e., the hTERT-HPNE E6/E7/KRasG12D cells. Then, we performed microarray analysis and Real-Time polymerase chain reaction validation in order to evaluate the global gene expression alterations. We also carried out wound healing assays in order to evaluate the effect of α, β, and γ CXCL12 isoforms on the cell migration ability.
The transcriptomic analyses showed that the expression of only few genes was affected by the treatment with the three isoforms. In particular, α and β isoforms affect different genes, whereas γ isoform altered the expression of genes already affected by the other isoforms. Since many genes affected by all isoforms are involved in cell migration and cytoskeleton remodelling, we performed cell migration assays, which confirmed the role of CXCL12 in migration, mainly caused by the γ isoform.
Our results suggest that α, β and γ CXCL12 isoforms can trigger different responses in a pancreatic pre-tumour model. The γ isoform induced the highest level of cell migration.
Although our data shed light on the molecular basis of PDAC onset and progression, further studies are necessary for a deeper characterization of CXCL12 isoforms.
We thank the Microarray Unit of Cogentech S.R.L. Benefit Corporation (Milan, Italy) for their assistance in the experimental microarray analyses.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Reshkin SJ S-Editor: Gao CC L-Editor: A P-Editor: Liu JH
| 1. | Luchini C, Capelli P, Scarpa A. Pancreatic Ductal Adenocarcinoma and Its Variants. Surg Pathol Clin. 2016;9:547-560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 2. | Sleightholm RL, Neilsen BK, Li J, Steele MM, Singh RK, Hollingsworth MA, Oupicky D. Emerging roles of the CXCL12/CXCR4 axis in pancreatic cancer progression and therapy. Pharmacol Ther. 2017;179:158-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 128] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 3. | Ding Y, Du Y. Clinicopathological significance and prognostic role of chemokine receptor CXCR4 expression in pancreatic ductal adenocarcinoma, a meta-analysis and literature review. Int J Surg. 2019;65:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 4. | Adamska A, Domenichini A, Falasca M. Pancreatic Ductal Adenocarcinoma: Current and Evolving Therapies. Int J Mol Sci. 2017;18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 427] [Article Influence: 53.4] [Reference Citation Analysis (0)] |
| 5. | Deramaudt T, Rustgi AK. Mutant KRAS in the initiation of pancreatic cancer. Biochim Biophys Acta. 2005;1756:97-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 68] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 6. | Feldmann G, Beaty R, Hruban RH, Maitra A. Molecular genetics of pancreatic intraepithelial neoplasia. J Hepatobiliary Pancreat Surg. 2007;14:224-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 201] [Cited by in RCA: 180] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 7. | Pillarisetty VG. The pancreatic cancer microenvironment: an immunologic battleground. Oncoimmunology. 2014;3:e950171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 8. | Carr RM, Fernandez-Zapico ME. Pancreatic cancer microenvironment, to target or not to target? EMBO Mol Med. 2016;8:80-82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Ren B, Cui M, Yang G, Wang H, Feng M, You L, Zhao Y. Tumor microenvironment participates in metastasis of pancreatic cancer. Mol Cancer. 2018;17:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 182] [Cited by in RCA: 415] [Article Influence: 59.3] [Reference Citation Analysis (0)] |
| 10. | Pan B, Liao Q, Niu Z, Zhou L, Zhao Y. Cancer-associated fibroblasts in pancreatic adenocarcinoma. Future Oncol. 2015;11:2603-2610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 11. | Qian D, Lu Z, Xu Q, Wu P, Tian L, Zhao L, Cai B, Yin J, Wu Y, Staveley-O'Carroll KF, Jiang K, Miao Y, Li G. Galectin-1-driven upregulation of SDF-1 in pancreatic stellate cells promotes pancreatic cancer metastasis. Cancer Lett. 2017;397:43-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 58] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 12. | Righetti A, Giulietti M, Šabanović B, Occhipinti G, Principato G, Piva F. CXCL12 and Its Isoforms: Different Roles in Pancreatic Cancer? J Oncol. 2019;2019:9681698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 13. | Roy I, Zimmerman NP, Mackinnon AC, Tsai S, Evans DB, Dwinell MB. CXCL12 chemokine expression suppresses human pancreatic cancer growth and metastasis. PLoS One. 2014;9:e90400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 14. | Zhao S, Chang SL, Linderman JJ, Feng FY, Luker GD. A Comprehensive Analysis of CXCL12 Isoforms in Breast Cancer1,2. Transl Oncol. 2014;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 15. | Ray P, Stacer AC, Fenner J, Cavnar SP, Meguiar K, Brown M, Luker KE, Luker GD. CXCL12-γ in primary tumors drives breast cancer metastasis. Oncogene. 2015;34:2043-2051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 16. | Furukawa T, Sunamura M, Horii A. Molecular mechanisms of pancreatic carcinogenesis. Cancer Sci. 2006;97:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 17. | Carvalho BS, Irizarry RA. A framework for oligonucleotide microarray preprocessing. Bioinformatics. 2010;26:2363-2367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1295] [Cited by in RCA: 1158] [Article Influence: 77.2] [Reference Citation Analysis (0)] |
| 18. | Leek JT, Johnson WE, Parker HS, Jaffe AE, Storey JD. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics. 2012;28:882-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2446] [Cited by in RCA: 3980] [Article Influence: 306.2] [Reference Citation Analysis (0)] |
| 19. | Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015;43:e47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16184] [Cited by in RCA: 25589] [Article Influence: 2558.9] [Reference Citation Analysis (0)] |
| 20. | Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, Koplev S, Jenkins SL, Jagodnik KM, Lachmann A, McDermott MG, Monteiro CD, Gundersen GW, Ma'ayan A. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016;44:W90-W97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4582] [Cited by in RCA: 6887] [Article Influence: 765.2] [Reference Citation Analysis (0)] |
| 21. | Morey JS, Ryan JC, Van Dolah FM. Microarray validation: factors influencing correlation between oligonucleotide microarrays and real-time PCR. Biol Proced Online. 2006;8:175-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 391] [Cited by in RCA: 408] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 22. | Yu L, Cecil J, Peng SB, Schrementi J, Kovacevic S, Paul D, Su EW, Wang J. Identification and expression of novel isoforms of human stromal cell-derived factor 1. Gene. 2006;374:174-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 161] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 23. | Altenburg JD, Broxmeyer HE, Jin Q, Cooper S, Basu S, Alkhatib G. A naturally occurring splice variant of CXCL12/stromal cell-derived factor 1 is a potent human immunodeficiency virus type 1 inhibitor with weak chemotaxis and cell survival activities. J Virol. 2007;81:8140-8148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 51] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Altenburg JD, Jin Q, Alkhatib B, Alkhatib G. The potent anti-HIV activity of CXCL12gamma correlates with efficient CXCR4 binding and internalization. J Virol. 2010;84:2563-2572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Cavnar SP, Ray P, Moudgil P, Chang SL, Luker KE, Linderman JJ, Takayama S, Luker GD. Microfluidic source-sink model reveals effects of biophysically distinct CXCL12 isoforms in breast cancer chemotaxis. Integr Biol (Camb). 2014;6:564-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 26. | Chang SL, Cavnar SP, Takayama S, Luker GD, Linderman JJ. Cell, isoform, and environment factors shape gradients and modulate chemotaxis. PLoS One. 2015;10:e0123450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 27. | Rueda P, Balabanian K, Lagane B, Staropoli I, Chow K, Levoye A, Laguri C, Sadir R, Delaunay T, Izquierdo E, Pablos JL, Lendinez E, Caruz A, Franco D, Baleux F, Lortat-Jacob H, Arenzana-Seisdedos F. The CXCL12gamma chemokine displays unprecedented structural and functional properties that make it a paradigm of chemoattractant proteins. PLoS One. 2008;3:e2543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 59] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 28. | Gadea G, Sanz-Moreno V, Self A, Godi A, Marshall CJ. DOCK10-mediated Cdc42 activation is necessary for amoeboid invasion of melanoma cells. Curr Biol. 2008;18:1456-1465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 157] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 29. | Jaudon F, Raynaud F, Wehrlé R, Bellanger JM, Doulazmi M, Vodjdani G, Gasman S, Fagni L, Dusart I, Debant A, Schmidt S. The RhoGEF DOCK10 is essential for dendritic spine morphogenesis. Mol Biol Cell. 2015;26:2112-2127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 30. | Ma WR, Xu P, Liu ZJ, Zhou J, Gu LK, Zhang J, Deng DJ. Impact of GFRA1 gene reactivation by DNA demethylation on prognosis of patients with metastatic colon cancer. World J Gastroenterol. 2020;26:184-198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Gharpure KM, Lara OD, Wen Y, Pradeep S, LaFargue C, Ivan C, Rupaimoole R, Hu W, Mangala LS, Wu SY, Nagaraja AS, Baggerly K, Sood AK. ADH1B promotes mesothelial clearance and ovarian cancer infiltration. Oncotarget. 2018;9:25115-25126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | Zhang W, He R, Chen S, Zhang L, Cao G, Yang W, Li J. The JAM-B/c-src/MMP9 pathway is associated with progression and regulates the invasion of pancreatic cancer. J Cancer. 2020;11:3246-3255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 33. | Li S, Jia H, Zhang Z, Wu D. LncRNA GAS6-AS1 facilitates the progression of breast cancer by targeting the miR-324-3p/SETD1A axis to activate the PI3K/AKT pathway. Eur J Cell Biol. 2020;99:151124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 34. | Zhang P, Dong Q, Zhu H, Li S, Shi L, Chen X. Long non-coding antisense RNA GAS6-AS1 supports gastric cancer progression via increasing GAS6 expression. Gene. 2019;696:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 35. | Du P, Ye L, Li H, Ruge F, Yang Y, Jiang WG. Loss of expression of growth differentiation factor-9 (GDF9) in human kidney cancer and regulation of growth and migration of kidney cancer cells by GDF9. Anticancer Res. 2012;32:4375-4383. [PubMed] |
| 36. | Guo QS, Song Y, Hua KQ, Gao SJ. Involvement of FAK-ERK2 signaling pathway in CKAP2-induced proliferation and motility in cervical carcinoma cell lines. Sci Rep. 2017;7:2117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 37. | Nakamura Y, Tanaka F, Haraguchi N, Mimori K, Matsumoto T, Inoue H, Yanaga K, Mori M. Clinicopathological and biological significance of mitotic centromere-associated kinesin overexpression in human gastric cancer. Br J Cancer. 2007;97:543-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 76] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 38. | Wei S, Dai M, Zhang C, Teng K, Wang F, Li H, Sun W, Feng Z, Kang T, Guan X, Xu R, Cai M, Xie D. KIF2C: a novel link between Wnt/β-catenin and mTORC1 signaling in the pathogenesis of hepatocellular carcinoma. Protein Cell. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 39. | Zaganjor E, Osborne JK, Weil LM, Diaz-Martinez LA, Gonzales JX, Singel SM, Larsen JE, Girard L, Minna JD, Cobb MH. Ras regulates kinesin 13 family members to control cell migration pathways in transformed human bronchial epithelial cells. Oncogene. 2014;33:5457-5466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 40. | Carmon KS, Gong X, Yi J, Wu L, Thomas A, Moore CM, Masuho I, Timson DJ, Martemyanov KA, Liu QJ. LGR5 receptor promotes cell-cell adhesion in stem cells and colon cancer cells via the IQGAP1-Rac1 pathway. J Biol Chem. 2017;292:14989-15001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 41. | Andrikou K, Santoni M, Piva F, Bittoni A, Lanese A, Pellei C, Conti A, Loretelli C, Mandolesi A, Giulietti M, Scarpelli M, Principato G, Falconi M, Cascinu S. Lgr5 expression, cancer stem cells and pancreatic cancer: results from biological and computational analyses. Future Oncol. 2015;11:1037-1045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 42. | Sun FB, Lin Y, Li SJ, Gao J, Han B, Zhang CS. MiR-210 knockdown promotes the development of pancreatic cancer via upregulating E2F3 expression. Eur Rev Med Pharmacol Sci. 2018;22:8640-8648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 43. | Prevo R, Fokas E, Reaper PM, Charlton PA, Pollard JR, McKenna WG, Muschel RJ, Brunner TB. The novel ATR inhibitor VE-821 increases sensitivity of pancreatic cancer cells to radiation and chemotherapy. Cancer Biol Ther. 2012;13:1072-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 187] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 44. | Liu S, Ge Y, Wang T, Edwards H, Ren Q, Jiang Y, Quan C, Wang G. Inhibition of ATR potentiates the cytotoxic effect of gemcitabine on pancreatic cancer cells through enhancement of DNA damage and abrogation of ribonucleotide reductase induction by gemcitabine. Oncol Rep. 2017;37:3377-3386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 45. | Harris NLE, Vennin C, Conway JRW, Vine KL, Pinese M, Cowley MJ, Shearer RF, Lucas MC, Herrmann D, Allam AH, Pajic M, Morton JP; Australian Pancreatic Cancer Genome Initiative; Biankin AV, Ranson M, Timpson P, Saunders DN. SerpinB2 regulates stromal remodelling and local invasion in pancreatic cancer. Oncogene. 2017;36:4288-4298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 46. | Kortum RL, Lewis RE. The molecular scaffold KSR1 regulates the proliferative and oncogenic potential of cells. Mol Cell Biol. 2004;24:4407-4416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 123] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 47. | Xing HR, Cordon-Cardo C, Deng X, Tong W, Campodonico L, Fuks Z, Kolesnick R. Pharmacologic inactivation of kinase suppressor of ras-1 abrogates Ras-mediated pancreatic cancer. Nat Med. 2003;9:1266-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 48. | Sparmann A, Bar-Sagi D. Ras-induced interleukin-8 expression plays a critical role in tumor growth and angiogenesis. Cancer Cell. 2004;6:447-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 621] [Cited by in RCA: 653] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 49. | Kamohara H, Takahashi M, Ishiko T, Ogawa M, Baba H. Induction of interleukin-8 (CXCL-8) by tumor necrosis factor-alpha and leukemia inhibitory factor in pancreatic carcinoma cells: Impact of CXCL-8 as an autocrine growth factor. Int J Oncol. 2007;31:627-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 50. | Matsuo Y, Ochi N, Sawai H, Yasuda A, Takahashi H, Funahashi H, Takeyama H, Tong Z, Guha S. CXCL8/IL-8 and CXCL12/SDF-1alpha co-operatively promote invasiveness and angiogenesis in pancreatic cancer. Int J Cancer. 2009;124:853-861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 227] [Cited by in RCA: 234] [Article Influence: 14.6] [Reference Citation Analysis (0)] |