Published online Mar 21, 2020. doi: 10.3748/wjg.v26.i11.1172
Peer-review started: November 15, 2019
First decision: February 14, 2019
Revised: March 5, 2020
Accepted: March 9, 2020
Article in press: March 9, 2020
Published online: March 21, 2020
Processing time: 126 Days and 11.7 Hours
Minimally invasive surgery for gastric cancer (GC) has gained widespread use as a safe curative procedure especially for early GC.
To determine risk factors for postoperative complications after minimally invasive gastrectomy for GC.
Between January 2009 and June 2019, 1716 consecutive patients were referred to our division for primary GC. Among them, 1401 patients who were diagnosed with both clinical and pathological Stage III or lower GC and underwent robotic gastrectomy (RG) or laparoscopic gastrectomy (LG) were enrolled. Retrospective chart review and multivariate analysis were performed for identifying risk factors for postoperative morbidity.
Morbidity following minimally invasive gastrectomy was observed in 7.5% of the patients. Multivariate analyses demonstrated that non-robotic minimally invasive surgery, male gender, and an operative time of ≥ 360 min were significant independent risk factors for morbidity. Therefore, morbidity was compared between RG and LG. Accordingly, propensity-matched cohort analysis revealed that the RG group had significantly fewer intra-abdominal infectious complications than the LG group (2.5% vs 5.9%, respectively; P = 0.038), while no significant differences were noted for other local or systemic complications. Multivariate analyses of the propensity-matched cohort revealed that non-robotic minimally invasive surgery [odds ratio = 2.463 (1.070–5.682); P = 0.034] was a significant independent risk factor for intra-abdominal infectious complications.
The findings showed that robotic surgery might improve short-term outcomes following minimally invasive radical gastrectomy by reducing intra-abdominal infectious complications.
Core tip: This study aimed to determine risk factors for postoperative complications after minimally invasive gastrectomy for gastric cancer. Accordingly, multivariate analysis identified non-robotic minimally invasive surgery as an independent risk factor for postoperative complications. Propensity score matching analysis showed that the robotic gastrectomy group had a significantly lower incidence of intra-abdominal infectious complications compared to the laparoscopic gastrectomy group. Additionally, multivariate analyses in the propensity score-matched cohort showed that non-robotic minimally invasive surgery was a significant independent risk factor for intra-abdominal infectious complications.
- Citation: Shibasaki S, Suda K, Nakauchi M, Nakamura K, Kikuchi K, Inaba K, Uyama I. Non-robotic minimally invasive gastrectomy as an independent risk factor for postoperative intra-abdominal infectious complications: A single-center, retrospective and propensity score-matched analysis. World J Gastroenterol 2020; 26(11): 1172-1184
- URL: https://www.wjgnet.com/1007-9327/full/v26/i11/1172.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i11.1172
Gastric cancer (GC) is the fifth most common malignancy and the third leading cause of cancer-related death worldwide[1]. Surgical resection with or without perioperative chemotherapy has remained the only curative treatment option, with regional lymphadenectomy being recommended as part of radical gastrectomy[2-4]. Recently, laparoscopic gastrectomy (LG) has gained widespread use as it is a minimally invasive and safe curative procedure for GC especially for early GC[5-7]. Since we demonstrated the comparability of the laparoscopic D2 gastrectomy over the open D2 gastrectomy in the short- and long-term outcomes[8,9], minimally invasive surgery (MIS) has been the first choice as the standard radical procedure for GC in our institute[10].
However, several recent studies using the nationwide web-based database of Japan have revealed that LG promoted higher postoperative local complications compared with open gastrectomy (OG)[11-13]. Two main reasons may explain such findings. First, LG requires more experience, at least 40–60 surgical procedures, to achieve optimal proficiency compared with OG[14-17]. Second, LG has several technical limitations, including limited range of motion with straight forceps and hand tremors, which need to be addressed to further improve surgical outcomes following minimally invasive gastrectomy. Accordingly, two possible measures may help overcome such limitations. First is the Endoscopic Surgical Skill Qualification System (ESSQS), which was launched in 2004 by the Japanese Society for Endoscopic Surgery to develop a tool for the reliable and reproducible evaluation of trainees’ surgical techniques[18]. In this system, two judges assess non-edited videotapes in a double-blinded fashion using strict criteria. Accordingly, surgeons determined to be qualified by this system experienced less frequent complications following laparoscopic distal gastrectomy (DG) compared with those who failed[18]. The second measure involves robotic surgery, which facilitates precise dissection in a confined surgical field with impressive dexterity[19-21]. In fact, a number of previous studies have shown that robotic gastrectomy (RG) resulted in significantly lower postoperative complication rates compared to LG[20,22,23].
Considering the aforementioned discussion, the present study aimed to determine risk factors for postoperative complications after MIS for GC, focusing on the impact of robotics and surgeon qualification by the ESSQS.
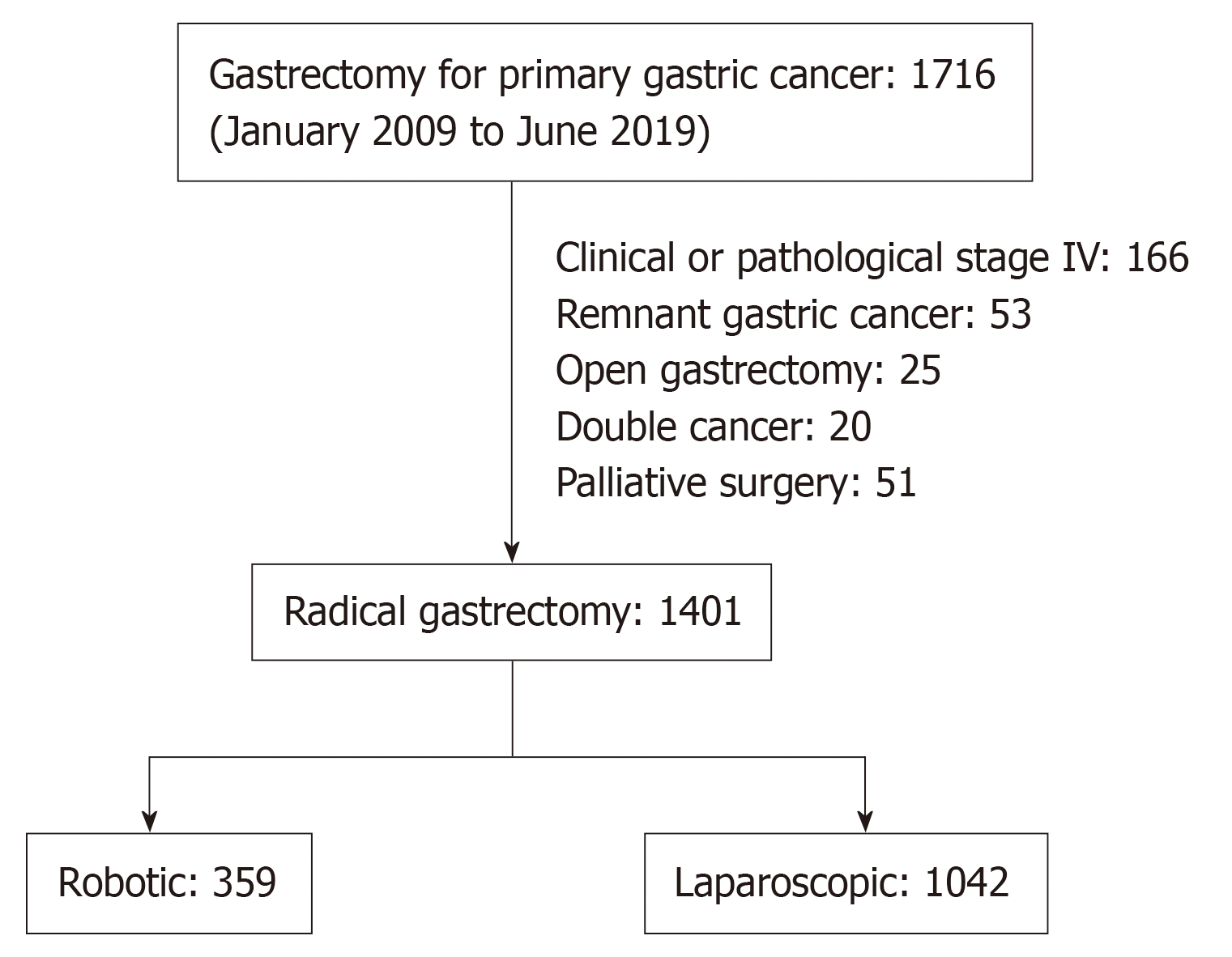
Between January 2009 and June 2019, 1716 consecutive patients were referred to our division for primary GC eligible for surgical treatment. The present study ultimately enrolled 1401 patients (robotic, n = 359 and laparoscopic, n = 1042) with both clinical and pathological Stage III or lower GC after excluding 315 patients who had clinical or pathological stage IV GC (n = 166), remnant GC (n = 53), OG (n = 25), double cancer (n = 20), and palliative or limited lymphadenectomy (n = 51) due to insufficient physical function. The patient selection process is summarized in Figure 1. This study included not only symptomatic patients but also those who were diagnosed as a result of the mass cancer screening programs, which have been executed nationwide and have contributed to earlier detection of GC. In the present study, the stage of the cancer was described according to the 15th edition of the Japanese Classification of Gastric Carcinoma[24]. Cancer staging was performed based on the findings of contrast-enhanced computed tomography, gastrography, endoscopic study, and endosonography before the beginning of any treatment and, when applicable, after the completion of chemotherapy, as we previously described[20]. Tumor invasion depth was measured ultrasonographically[25,26]. The gastric wall was assessed based on the standard five-layer sonographic structure. On the endosonographic image, the mucosal layer is visualized as a combination of the first and second hypoechoic layers, and the submucosal layer corresponds to the third hyperechoic layer. The layer of the muscularis propria is visualized as the fourth hypoechoic layer, and the fifth hyperechoic layer is the serosa, including the subserosa. Initial endoscopic diagnosis regarding invasion depth was confirmed based on the agreement by expert endoscopists at the medical conference prior to therapy. The indication of endoscopic treatment and radical gastrectomy including the extent of systematic lymph node dissection was determined based on the 2014 Japanese Gastric Cancer Treatment Guidelines[3]. The microscopic tumor-negative status in the cut end was routinely confirmed by intraoperative frozen section diagnosis as previously reported[27,28], and margins of resection (R0 or R1 resection) was pathologically diagnosed by permanent section diagnosis. In a considerable number of the enrolled patients, Helicobacter pylori was examined and systemically eradicated before surgery at each hospital or clinic at which GC of those patients was diagnosed. Details regarding indications for physical function assessment, surgical procedures, perioperative radical gastrectomy management, extent of gastric resection and lymph node dissection, type of anastomosis, and postoperative chemotherapy in addition to oncologic follow-up have been reported previously[8-10,20,21,29,30]. This study was approved by the Institutional Review Board of Fujita Health University.
Patients were completely involved in the decision-making process, and informed consent was obtained from all patients. However, during the study period, decision making on patient procedures was dependent on circumstances surrounding the national medical insurance coverage. Accordingly, RG had not been included in the national medical insurance coverage in Japan between January 2009 and March 2018, during which patients needed to be charged 2200000 JPY upon perioperative admission to undergo RG[20]. All patients were equally offered robotic surgery without considering their backgrounds, including physical and oncological status. Hence, 211 patients who agreed to uninsured da Vinci Surgical System (DVSS) application underwent RG, whereas the remaining 946 patients who refused uninsured DVSS application underwent LG with health insurance coverage. Meanwhile, between October 2014 and January 2017, we organized a multi-institutional, single-arm prospective clinical study approved for Advanced Medical Technology (“Senshiniryo”) B[23]. Accordingly, 94 patients with cStage I/II GC who were enrolled in our institution’s Senshiniryo B trial were also included in the present analysis. Since its approval for national medical insurance coverage based on the outcomes of the Senshiniryo B trial in April 2018, RG has been more favorably indicated for patients diagnosed with advanced GC who required total gastrectomy (TG) or proximal gastrectomy (PG) and desired to undergo RG at our institution. After April 2018, 52 patients underwent RG, whereas 96 underwent LG.
All LG procedures were performed or guided by the ESSQS-qualified surgeons. Meanwhile, RG was performed by surgeons certified to operate a DVSS console, qualified by the ESSQS, and certified by the Japanese Society of Gastroenterological Surgery. All procedures related to LG and RG were supervised by an expert gastric surgeon (I.U.) who had performed more than 1500 LG and 400 RG procedures.
All patients were observed for 30 d following surgery. The primary endpoint of this single-center retrospective analysis was morbidity. Secondary endpoints comprised clinicopathological characteristics and short-term surgical outcomes, including operative time, surgeon console time, estimated blood loss, number of dissected lymph nodes, complication rates, rates for intra-abdominal infectious complications (including postoperative pancreatic fistulas, leakage, and intra-abdominal abscesses), mortality rate, and length of postoperative hospitalization. All postoperative complications Grade IIIa or above based on the Clavien–Dindo (CD) classification were recorded[31] and classified according to the Japan Clinical Oncology Group Postoperative Complications Criteria based on the CD classification ver. 2.0[32]. Total operative time was defined as the duration from the start of abdominal incision until complete wound closure, while surgeon console time was defined as the duration of DVSS operation during surgery. Blood loss was estimated by weighing suctioned blood and gauze pieces that had absorbed blood.
Diagnosis and grading of pancreatic fistula were determined according to CD classification[31] as mentioned above. Our perioperative management for postoperative pancreatic fistula was conducted as follows[20,33]: Although pancreatic fistula is defined as output via an operatively placed drain (or a subsequently placed percutaneous drain) of any measurable volume of drain fluid on or after postoperative day 3, with an amylase level at least over 3 times as high as the upper normal range of the serum level, it was comprehensively diagnosed according to not only drain amylase levels, but also changes in the properties of the drain and the clinical, laboratory, and imaging findings including computed tomographic scans. Patients with high drain amylase level and no abnormal physical and laboratory findings were observed without any treatment (CD Grade I). The abdominal drainage tube was removed basically after the drain amylase level was sufficiently recovered. Patients with high drain amylase level accompanied by abnormal findings such as fever, abdominal pain, and high inflammatory markers, were intensively treated with antibiotics, octreotide acetate, and parenteral nutrition while the drainage tube position was urgently confirmed using computed tomographic scans and radiographic contrast study (CD Grade II). When the drainage tube position was not appropriate, an additional or alternative drainage tube was placed into the fluid cavity using percutaneous computed tomography or ultrasonography-guided technique (CD Grade IIIa), and irrigation and drainage with saline was performed. Parenteral nutrition was gradually switched to enteral nutrition without delay, once pancreatic fistula had been confined to a certain space and inflammatory response had settled.
Propensity score-matched (PSM) analysis was used to limit confounders and overcome possible patient selection bias. Propensity scores for all patients were calculated using a logistic regression model based on the following variables: Age, gender, body mass index (BMI), American Society of Anesthesiologist (ASA) classification, presence of neoadjuvant chemotherapy, history of laparotomy, cT, cN, cStage, pT, pN, pStage, type of gastrectomy, extent of lymph node dissection, and splenectomy. Consequently, rigorous adjustment for significant differences in the baseline characteristics of PSM patients was performed using nearest neighbor matching without replacement and a caliper width of 0.2 logit of the standard deviation. An absolute standardized difference (SD) was used to measure covariate balance, in which an absolute standardized mean difference above 0.1 indicated a meaningful imbalance[11,12].
All analyses were conducted using IBM SPSS Statistics 23 (IBM Corporation, Armonk, NY, United States). Between-group comparisons were performed using the χ2 test or Mann–Whitney U test. Univariate χ2 test and multivariate logistic regression analysis were used to determine risk factors for the occurrence of postoperative complications. Data were expressed as median (range) or odds ratio (OR) (95% confidence interval) unless otherwise specified. P < 0.05 (two-tailed) was considered statistically significant.
Patient characteristics and surgical outcomes of MIS for GC are summarized in Table 1. Accordingly, 939 (67%) and 856 (61%) patients had cStage I and pStage I disease, respectively, while 120 (8.6%) received preoperative chemotherapy. A total of 359 and 1042 patients underwent RG and LG, while 993 (70.9%), 89 (6.4%), and 319 (22.8%) underwent DG, PG, and TG, respectively. Moreover, 767 and 634 patients underwent D1+ and D2 dissection, respectively. The rates for conversion to open procedure, reoperation within 30 d, in-hospital mortality within 30 d, and morbidity within 30 d after operation were 0.1%, 1.1%, 0.3% and 7.5%, respectively (Table 1). All patients completed successfully R0 resection.
| Clinicopathological characteristics | Surgical outcomes | ||
| Age (yr) | 69 (24–93) | No. of operators (certified surgeon) | 33 (19) |
| Gender (M:F) | 973:428 | Qualified:non-qualified surgeons | 925:476 |
| Body mass index (kg/m2) | 22.3 (1 4.3–37.3) | Procedure (RG:LG) | 359:1042 |
| ASA grade (1:2:3) | 500:733:168 | Type of resection (DG:PG:TG) | 993:89:319 |
| History of laparotomy, n (%) | 263 (18.8) | Extent of lymphadenectomy (D1+:D2) | 767:634 |
| Tumor size (mm) | 30 (0–180) | Splenectomy, n (%) | 40 (2.9) |
| cT1 (1:2:3:4a) | 751:264:224:162 | Total operative time (min) | 348 (147–942) |
| cN1 (−:+) | 1093:308 | Estimated blood loss (mL) | 30 (0–2150) |
| cStage1 (I:IIA:IIB:III) | 939:76:154:232 | No. of dissected LNs | 35 (6–114) |
| pT1 (1:2:3:4a) | 797:164:178:262 | Conversion to open procedure, n (%) | 1 (0.1) |
| pN1 (0:1:2:3) | 949:174:137:141 | Reoperation rate, n (%) | 15 (1.1) |
| pStage1 (I:II:III) | 856:280:265 | In-hospital mortality, n (%) | 4 (0.3) |
| No. of metastatic LNs | 0 (0–63) | Morbidity, n (%) | 105 (7.5) |
| Use of preoperative chemotherapy, n (%) | 120 (8.6) | Hospital stay following surgery (d) | 13 (2–195) |
Univariate analysis identified seven significant factors for postoperative CD grade IIIa or more complications, including non-robotic MIS, male gender, cStage II or higher, type of gastrectomy (PG and TG), splenectomy, operative time ≥ 360 min, and estimated blood loss ≥ 50 mL. Multivariate analysis determined that non-robotic MIS [OR = 2.591 (1.418–4.717); P = 0.002], male gender [OR = 1.969 (1.142–3.390); P = 0.015], and operative time ≥ 360 min [OR = 1.800 (1.098–2.952); P = 0.020] were significant independent risk factors for morbidity (Table 2).
| Factors | Univariate analysis, OR (95%CI) | P value | Multivariate analysis, OR (95%CI) | P value |
| Non-robotic minimally invasive surgery | 2.438 (1.381–4.304) | 0.002 | 2.591 (1.418–4.717) | 0.002 |
| Age ≥ 70 yr | 1.020 (0.706–1.474) | 0.920 | ||
| Male | 2.277 (1.372–3.779) | 0.001 | 1.969 (1.142–3.390) | 0.015 |
| Body mass index ≥ 23 kg/m2 | 1.138 (0.763–1.698) | 0.538 | ||
| ASA score 2 or higher | 1.069 (0.703–1.625) | 0.832 | ||
| cT21 or higher | 1.296 (0.870–1.930) | 0.222 | ||
| cN1 positive | 1.183 (0.745–1.879) | 0.540 | ||
| cStage II1 or higher | 1.649 (1.102–2.467) | 0.017 | 1.247 (0.809–1.922) | 0.318 |
| Proximal or total gastrectomy | 1.847 (1.230–2.772) | 0.004 | 1.208 (0.753–1.937) | 0.433 |
| D2 lymph node dissection | 1.204 (0.809–1.792) | 0.415 | ||
| Splenectomy | 2.734 (1.179–6.339) | 0.026 | 1.360 (0.542–3.408) | 0.512 |
| History of laparotomy | 1.083 (0.658–1.783) | 0.795 | ||
| Operative time ≥ 360 min | 2.449 (1.613–3.718) | < 0.001 | 1.800 (1.098–2.952) | 0.020 |
| Estimated blood loss ≥ 50 mL | 2.039 (1.367–3.042) | < 0.001 | 1.368 (0.873–2.143) | 0.209 |
| Tumor size ≥ 30 mm | 1.119 (0.703–1.782) | 0.721 | ||
| pT21 or higher | 1.378 (0.926–2.052) | 0.125 | ||
| pN1 positive | 1.155 (0.761–1.753) | 0.516 | ||
| pStage II 1 or higher | 1.471 (0.987–2.192) | 0.061 | ||
| Use of neoadjuvant chemotherapy | 1.274 (0.662–2.452) | 0.467 | ||
| Non-qualified surgeons | 1.148 (0.785–1.679) | 0.521 |
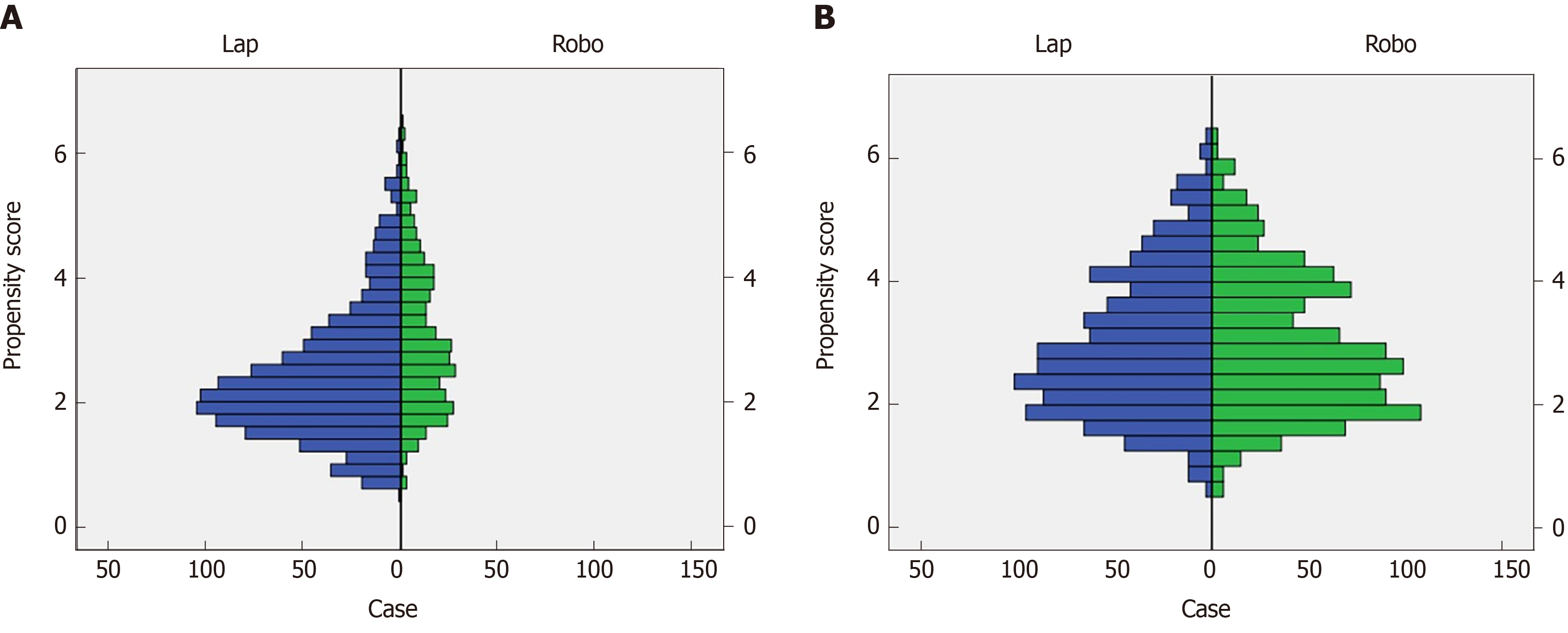
Our analysis subsequently focused on the comparison between RG and LG. Patient characteristics according to type of procedure are summarized in Table 3. Although no differences in BMI, history of laparotomy, tumor size, cT, cN, cStage, pT, pN, pStage, and number of metastatic lymph nodes were observed between the RG and LG group, significant differences were found in age [RG 67 (30–89) vs LG 70 (24–93); P < 0.001], gender (M:F, RG 233:126 vs LG 740:302; P = 0.033), ASA classification (1:2:3, RG 160:168:31 vs LG 340:565:137; P < 0.001), preoperative chemotherapy (RG 5.3% vs LG 9.7%; P = 0.010), type of resection (DG:PG:TG, RG 250:42:67 vs LG 743/47/252; P < 0.001), and extent of lymphadenectomy (D1+:D2, RG 178:181 vs LG 589:453; P = 0.023). Factors having an SD over 0.1 included age, gender, BMI, ASA classification, tumor size, use of preoperative chemotherapy, type of resection, extent of lymphade-nectomy, and splenectomy (Table 3). To compensate for such differences, PSM analysis was used. The average and standard deviation of the propensity score was 0.256 and 0.111, respectively, thus yielding a caliper width of 0.02 for this study. After propensity score matching, 354 patients were included in each group (Figure 2). Propensity score distributions for each case before and after matching are presented in Figure 2. After matching, the SD for age, gender, BMI, ASA classification, presence of neoadjuvant chemotherapy, history of laparotomy, tumor size, cT, cN, cStage, pT, pN, pStage, type of resection, extent of lymph node dissection, and splenectomy decreased to < 0.10, indicating that a sufficient balance was achieved (Table 3).
| Entire cohort (n = 1401) | P value | SD | Propensity-score matched cohort (n = 708) | P value | SD | |||
| RG (n = 359) | LG (n = 1042) | RG (n = 354) | LG (n = 354) | |||||
| Age (yr) | 67 (30–89) | 70 (24–93) | < 0.001 | 0.24 | 67 (30–89) | 66 (24–90) | 0.315 | 0.10 |
| Gender (M:F) | 233:126 | 740:302 | 0.033 | 0.12 | 230:124 | 230:124 | 1.000 | 0 |
| Body mass index (kg/m2) | 22.9 (14.3–32.0) | 22.2 (14.5–37.3) | 0.021 | 0.12 | 22.8 (14.3–32.0) | 22.4 (14.9–37.3) | 0.752 | 0.03 |
| ASA grade (1:2:3) | 160:168:31 | 340:565:137 | < 0.001 | 0.25 | 155:168:31 | 149:174:31 | 0.905 | 0.03 |
| History of laparotomy, n (%) | 66 (18.4) | 197 (18.9) | 0.876 | 0.01 | 65 (18.4) | 59 (16.7) | 0.621 | 0.04 |
| Tumor size (mm) | 30 (0–170) | 30 (0–180) | 0.013 | 0.14 | 30 (0–170) | 30 (0–180) | 0.208 | 0.09 |
| cT1 (1:2:3:4a) | 198:75:56:30 | 553:189:168:132 | 0.131 | 0.04 | 195:74:55:30 | 204:70:48:32 | 0.834 | 0.05 |
| cN1 (−:+) | 287:72 | 806:236 | 0.337 | 0.06 | 284:70 | 289:65 | 0.702 | 0.04 |
| cStage1 (I:II:III) | 249:62:48 | 690:168:184 | 0.183 | 0.07 | 246:61:47 | 252:59:43 | 0.962 | 0.04 |
| pT1 (1:2:3:4a) | 210:36:46:67 | 587:128:132:195 | 0.711 | 0.05 | 208:36:46:64 | 213:36:39:66 | 0.882 | 0.03 |
| pN1 (0:1:2:3) | 249:47:24:39 | 700:127:113:102 | 0.145 | 0.05 | 246:46:24:38 | 244:48:18:44 | 0.72 | 0.01 |
| pStage1 (I:II:III) | 221:80:58 | 635:200:207 | 0.200 | 0.01 | 219:78:57 | 224:71:59 | 0.823 | 0.03 |
| No. of metastatic LNs | 0 (0–43) | 0 (0–63) | 0.385 | 0.03 | 0 (0–43) | 0 (0–37) | 0.819 | 0.02 |
| Use of preoperative chemotherapy, n (%) | 19 (5.3) | 101 (9.7) | 0.011 | 0.17 | 19 (5.4) | 18 (5.1) | > 0.999 | 0.01 |
| Type of resection (DG:PG:TG) | 250:42:67 | 743:47:252 | < 0.001 | 0.13 | 248:39:67 | 252:33:69 | 0.778 | 0.01 |
| Extent of lymphadenectomy (D1+:D2) | 178:181 | 589:453 | 0.023 | 0.14 | 175:179 | 175:179 | > 0.999 | 0 |
| Splenectomy, n (%) | 6 (1.7) | 34 (3.3) | 0.142 | 0.11 | 6 (1.7) | 7 (2.0) | > 0.999 | 0.02 |
Surgical outcomes and short-term postoperative courses of the entire cohort and the PSM cohort are summarized in Table 4. Accordingly, 8 and 33 operating surgeons performed RG and LG, respectively. Moreover, 100% of the RG cases and only 56.5% (572/1042) of the LG cases (P < 0.001) were handled by qualified surgeons. The RG group had a significantly shorter duration of hospitalization following surgery compared to the LG group [RG 12 (2–195) d vs LG 13 (3–177) d; P < 0.001], despite having a slightly greater total operative time [RG 360 (174–942) min vs LG 342 (147–937) min; P < 0.001) and estimated blood loss [RG 36 (0–935) mL vs LG 29 (0–2150) mL; P < 0.001]. No significant differences were observed in the number of dissected lymph nodes, conversion to open procedure, and reoperation rate. In-hospital mortality was sufficiently low (RG 0.6% vs LG 0.3%; P = 0.578) throughout this series. After propensity score matching, results similar to those for the entire cohort were obtained (Table 4).
| Entire cohort (n = 1401) | P value | Propensity-score matched cohort (n = 708) | P value | |||
| RG (n = 359) | LG (n = 1042) | RG (n = 354) | LG (n = 354) | |||
| No. of operators (qualified surgeon) | 8 (8) | 33 (14) | NA | 8 (8) | 33 (14) | NA |
| Qualified:non-qualified surgeons | 359:0 | 572:475 | < 0.001 | 354:0 | 186:149 | < 0.001 |
| Total operative time (min) | 360 (174–942) | 342 (147–937) | < 0.001 | 360 (174–942) | 347 (149–937) | 0.001 |
| Console time (min) | 306 (136–860) | NA | NA | 307 (136–860) | NA | NA |
| Estimated blood loss (mL) | 36 (0–935) | 29 (0–2150) | 0.007 | 37 (0–935) | 28 (0–2150) | 0.005 |
| No. of dissected LNs | 37 (7–100) | 35 (6–114) | 0.058 | 37 (7–100) | 36 (6–108) | 0.807 |
| Conversion to open procedure, n (%) | 0 (0) | 1 (0.1) | > 0.999 | 0 | 0 | > 0.999 |
| Reoperation rate, n (%) | 4 (1.1) | 11 (1.1) | > 0.999 | 4 (1.1) | 6 (1.7) | 0.750 |
| Hospital stay following surgery (d) | 12 (2–195) | 13 (3–177) | < 0.001 | 12 (2–195) | 13 (3–131) | 0.001 |
| In-hospital mortality, n (%) | 2 (0.6) | 2 (0.3) | 0.578 | 2 (0.6) | 1 (0.3) | > 0.999 |
Postoperative complications are summarized in Table 5. Briefly, the RG group had a significantly better morbidity rate than the LG group (RG 3.6% vs LG 8.8%; P = 0.002). Robotic surgery promoted better attenuation of intra-abdominal infectious complications compared to non-robotic surgery (RG 2.5% vs LG 6.3%; P = 0.005), while no significant differences in other local (RG 0.8% vs LG 1.3%; P = 0.632) or systemic (RG 0.3% vs LG 1.6%; P = 0.091) complication rates were observed. After PSM analysis, results remained almost same (Table 5), with the RG group showing a significantly better morbidity rate than the LG group (RG 3.7% vs LG 7.6%; P = 0.033). Robotic surgery promoted better attenuation of intra-abdominal infectious complications compared to non-robotic surgery (RG 2.5% vs LG 5.9%; P = 0.038), while no significant differences in other local (RG 0.6% vs LG 1.1%; P = 0.682) or systemic (RG 0.3% vs LG 1.1%; P = 0.369) complication rates were observed.
| Entire cohort (n = 1401) | P value | Propensity-score matched cohort (n = 708) | P value | |||
| RG (n = 359) | LG (n = 1042) | RG (n = 354) | LG (n = 354) | |||
| Morbidity | 13 (3.6) | 92 (8.8) | 0.002 | 13 (3.7) | 27 (7.6) | 0.033 |
| Intra-abdominal infection | 9 (2.5) | 66 (6.3) | 0.005 | 9 (2.5) | 21 (5.9) | 0.038 |
| Anastomotic leakage | 6 (1.7) | 22 (2.1) | 0.670 | 6 (1.7) | 7 (2.0) | > 0.999 |
| Pancreatic fistula | 3 (0.8) | 28 (2.7) | 0.058 | 3 (0.8) | 9 (2.5) | 0.143 |
| Intraperitoneal abscess | 0 (0) | 16 (1.5) | 0.017 | 0 (0) | 5 (1.4) | 0.062 |
| Other local complications | 3 (0.8) | 14 (1.3) | 0.632 | 2 (0.6) | 4 (1.1) | 0.682 |
| Intra-abdominal bleeding | 2 (0.6) | 4 (0.4) | > 0.999 | 1 (0.3) | 2 (0.6) | > 0.999 |
| Bowel obstruction | 0 (0) | 7 (0.6) | 0.150 | 0 (0) | 0 (0) | > 0.999 |
| Anastomotic stenosis | 1 (0.3) | 3 (0.3) | > 0.999 | 1 (0.3) | 2 (0.6) | > 0.999 |
| Systemic complications | 1 (0.3) | 17 (1.6) | 0.091 | 1 (0.3) | 4 (1.1) | 0.369 |
| Pneumonia | 0 (0) | 9 (0.9) | 0.256 | 0 (0) | 2 (0.6) | 0.499 |
| Pulmonary embolism | 1 (0.3) | 2 (0.2) | 0.726 | 1 (0.3) | 0 (0) | > 0.999 |
| Renal dysfunction | 0 (0) | 1 (0.1) | 0.573 | 0 (0) | 0 (0) | > 0.999 |
| Cardiovascular disease | 0 (0) | 5 (0.5) | 0.337 | 0 (0) | 2 (0.6) | 0.499 |
Univariate analysis identified several significant risk factors for intra-abdominal infectious complications, including non-robotic MIS, male gender, PG or TG, operative time ≥ 360 min, estimated blood loss ≥ 50 mL, and non-qualified surgeons (Table 6). Multivariate analysis clearly demonstrated that non-robotic MIS [OR 2.463 (1.070–5.682); P = 0.034], male gender [OR 3.937 (1.157–13.333); P = 0.028], and operative time ≥ 360 min [OR 2.779 (1.003–7.701); P = 0.049] were significant independent risk factors for intra-abdominal infectious complications.
| Factors | Univariate analysis, OR (95%CI) | P value | Multivariate analysis, OR (95%CI) | P value |
| Non-robotic minimally invasive surgery | 2.333 (1.084–5.024) | 0.038 | 2.463 (1.070–5.682) | 0.034 |
| Age ≥ 70 yr | 1.141 (0.546–2.388) | 0.849 | ||
| Male | 4.852 (1.487–15.834) | 0.003 | 3.937 (1.157–13.333) | 0.028 |
| Body mass index ≥ 23 kg/m2 | 1.064 (0.511–2.214) | > 0.999 | ||
| ASA score 2 or higher | 1.135 (0.538–2.393) | 0.851 | ||
| cStage II1 or more | 1.394 (0.651–2.982) | 0.416 | ||
| Proximal or total gastrectomy | 2.513 (1.205–5.240) | 0.014 | 1.694 (0.722–3.974) | 0.226 |
| D2 dissection | 1.023 (0.508–2.061) | > 0.999 | ||
| Splenectomy | 1.579 (0.467–5.339) | 0.668 | ||
| Operative time ≥ 360 min | 4.863 (1.963–12.048) | < 0.001 | 2.779 (1.003–7.701) | 0.049 |
| Estimated blood loss ≥ 50 mL | 3.596 (1.682–7.687) | 0.001 | 2.204 (0.967–5.023) | 0.060 |
| Tumor size ≥ 30 mm | 1.057 (0.508–2.201) | > 0.999 | ||
| pStage II1 or more | 1.120 (0.531–2.363) | 0.848 | ||
| Use of neoadjuvant chemotherapy | 1.599 (0.224–11.419) | 0.729 | ||
| Non-qualified surgeons | 2.047 (1.018–4.118) | 0.045 | 1.852 (0.810–4.237) | 0.145 |
The present study sought to identify risk factors for complications after MIS for GC. Accordingly, multivariate analysis revealed that non-robotic MIS was among the independent risk factors for complications. To determine whether a cause–effect relationship existed between non-RG and morbidity, short-term outcomes between RG and LG were compared using PSM analysis. Subsequent results showed that the RG group had a significantly lower incidence of intra-abdominal infectious complications compared to LG group and was more likely to be handled by an ESSQS-qualified surgeon. However, multivariate analysis of the PSM cohort showed that non-robotic MIS, but not the lack of ESSQS surgeon qualification, was a significant independent risk factor for intra-abdominal infectious complications. These findings clearly suggest that robotic surgery is at least more effective in reducing morbidity after MIS for GC than ESSQS qualification. The results presented herein support our previous evidence suggesting that the use of a robotic system significantly reduced postoperative complications[18]. In addition, the present study yielded three major findings.
First, the current study observed a 3.6% and 2.5% incidence rate for CD grade IIIa or higher morbidity and intra-abdominal infectious complications following RG, respectively. This finding was comparable to results from other prospective trials in Japan (2.5%–5.0% and 0.6%–3.3%, respectively)[23,34,35] or in other countries (reported as a range from 1.0% to 8.9%)[36]. In particular, RG seemed to have greater beneficial effects against pancreatic fistulas and intraperitoneal abscesses rather than anastomotic leakage compared to LG, although no significant difference was observed. This may be partly attributed to the meticulosity and high-definition magnified three-dimensional image of the robotic systems, which could be more effective in pancreas-protective radical lymph node dissection rather than intracorporeal alimentary tract reconstruction[30,37]. Actually, according to Table 5, there is a trend towards decrease in intraperitoneal abscess as well as pancreatic fistula in the RG group. Since intraperitoneal abscess could be induced by subclinical pancreatic fistula, the following speculation has taken place considering the results of our previous study in which RG significantly reduced pancreatic fistula: Robotic articulating forceps in combination with the magnified vivid three dimensional image enable operating surgeons to conduct radical lymph node dissection with little touch on the pancreas, leading to reduction in postoperative intra-abdominal infectious complications including clinical and subclinical pancreatic fistula. In addition, the “double bipolar” method characterized by simultaneous use of Maryland bipolar forceps (bipolar forced coagulation, 420172, Intuitive) with the right hand and Fenestrated bipolar forceps (bipolar soft coagulation, 420205, Intuitive) with the left hand might also facilitate pancreas-protective dissection in RG[20,23]. However, there has been little evidence that minimally invasive gastrectomy is contributed to the reduction in postoperative pancreatic fistula, as shown in previous meta-analyses based on retrospective studies[38,39]. Therefore, further studies including multi-center randomized controlled trial are desired to establish solid evidence on RG.
Second, multivariate analysis showed that surgeon non-qualification was not an independent risk factor for morbidity. Two possible reasons may explain such a result. First is that a qualified surgeon could have guided the non-qualified surgeon performing the surgery. Second is that the qualified surgeons are able to perform high-quality surgeries even on technically demanding cases. In fact, our results showed that qualified surgeons were more likely to be in charge of more difficult procedures, including PG, TG, and D2 dissection, and there were no significant differences in morbidity rate of LG between the qualified and non-qualified surgeons (data not shown). Therefore, we still believe that ESSQS has played an important role in securing the safety and quality of MIS for GC.
Third, our findings showed that RG increased total operative time, a result consistent with those presented in many previous reports or meta-analyses[36]. In contrast, total operative time ≥ 360 min was identified as an independent risk factor for postoperative intra-abdominal infectious complications. This reduction in complications despite prolonged operative time with RG suggest its potential efficacy for addressing or overcoming certain factors that may induce complications in association with prolonged operative time. Considering that more technically-demanding procedures, such as PG/TG or D2 dissection, as well as complicated patient backgrounds, such as more advanced diseases, higher BMI, and use of preoperative chemotherapy, would likely extended operative time, they would constitute good indications for RG.
The present study has several limitations that need consideration. First, this study employed a single-center, retrospective, and non-randomized design. Moreover, financial resources necessary for RG had been changed from each patient’s own expense, Senshiniryo B, to the national insurance coverage. Therefore, considering possible data biases, overall results should be interpreted cautiously. Since October 2018, all patients who underwent RG using the national medical insurance must be prospectively registered to the web-based registry of the National Clinical Database[40]. Thus, large real-world data from this prospective registry would reveal actual outcomes of RG, including intraoperative and postoperative adverse effects and long-term oncologic outcomes. Second, this study has concerns regarding operator bias given that almost half of the LG cases were performed by non-qualified surgeons, while all RG procedures were performed by qualified surgeons. Accordingly, it remains largely unclear whether the protective effects of RG on morbidity observed herein could be extrapolated to RG conducted by a non-qualified surgeon. To address this issue, future studies comparing RG and LG performed by experts and non-qualified surgeons would be necessary. Third, the advantages of RG on oncological outcomes remained inconclusive give that long-term surveillance is still underway. However, we had previously reported that RG had long-term oncological outcomes comparable to those for LG[41]. In addition, some reports demonstrated that intra-abdominal infectious complications after gastrectomy had a negative impact on long-term oncological outcomes[42,43]. Further investigations are nonetheless warranted to determine whether RG’s effect in reducing intra-abdominal infectious complications can lead to improvement in oncological outcomes after RG in the present cohort. Fourth, robotic and technological advances may influence surgical outcomes. During this study period, three DVSS systems, i.e., S, Si, and Xi, had been used for RG. Although no differences in complication rates had been observe between these three systems (data not shown), further investigation on how differences in the version of the robotic system affect surgical outcomes is imperative. In addition, multivariate analysis involving the PSM cohort identified the male gender as an independent risk factor for intra-abdominal infectious complications. However, factors primarily affected have remained unclear. Hence, further investigation regarding differences in clinical and anatomical characteristics between males and females is necessary.
In conclusion, the present study shown that robotic surgery might improve short-term outcomes following minimally invasive radical gastrectomy by reducing intra-abdominal infectious complications.
Minimally invasive surgery for gastric cancer (GC) has gained widespread use as a safe curative procedure especially for early GC. However, several recent studies using the nationwide web-based database of Japan have revealed that laparoscopic gastrectomy (LG) promoted higher postoperative local complications compared with open gastrectomy.
We launched robotic gastrectomy (RG) for GC in 2009. Our previous studies have consistently suggested that use of the surgical robot in LG might reduce postoperative complications, although use of the surgical robot was determined in a non-randomized manner. Actually, only experienced surgeons have performed RG, and RG was used for patients who hoped for uninsured use of the robot between 2009 and 2017.
This study aimed to determine risk factors for postoperative complications after minimally invasive gastrectomy for GC using our prospectively maintained database between January 2009 and June 2019.
This study enrolled 1401 patients who underwent radical robotic gastrectomy (RG) or LG for clinical and pathological Stage III or lower GC. Retrospective chart review and multivariate analysis were performed for identifying risk factors for postoperative morbidity.
Multivariate analyses demonstrated that non-robotic minimally invasive surgery, male gender, and an operative time of ≥ 360 min were significant independent risk factors for morbidity. Therefore, morbidity was compared between RG and LG using propensity score matched analysis. As a result, RG induced significantly fewer intra-abdominal infectious complications than the LG (2.5% vs 5.9%, respectively; P = 0.038). Multivariate analyses of the propensity-matched cohort revealed that non-robotic minimally invasive surgery [OR = 2.463 (1.070–5.682); P = 0.034] was a significant independent risk factor for intra-abdominal infectious complications.
RG might improve short-term outcomes following minimally invasive radical gastrectomy by reducing intra-abdominal infectious complications.
We will conduct an RCT on this topic in the near future. Impact of RG on long-term outcomes should also be examined at least in this cohort.
Biostatistics statement: The biostatistics was supervised by Satista, Co., Ltd.. (URL: http://www.satista.jp/medical/, E-mail: medical@satista.jp).
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Japan
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Fusaroli P, Li Y, Petrucciani N, Wang DR, Guerra F S-Editor: Wang YQ L-Editor: A E-Editor: Ma YJ
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55842] [Article Influence: 7977.4] [Reference Citation Analysis (132)] |
| 2. | Sano T, Sasako M, Yamamoto S, Nashimoto A, Kurita A, Hiratsuka M, Tsujinaka T, Kinoshita T, Arai K, Yamamura Y, Okajima K. Gastric cancer surgery: morbidity and mortality results from a prospective randomized controlled trial comparing D2 and extended para-aortic lymphadenectomy--Japan Clinical Oncology Group study 9501. J Clin Oncol. 2004;22:2767-2773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 493] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 3. | Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer. 2017;20:1-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1575] [Cited by in RCA: 1915] [Article Influence: 239.4] [Reference Citation Analysis (1)] |
| 4. | Sasako M. Progress in the treatment of gastric cancer in Japan over the last 50 years. Ann Gastroenterol Surg. 2020;4:21-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 5. | Kim HH, Hyung WJ, Cho GS, Kim MC, Han SU, Kim W, Ryu SW, Lee HJ, Song KY. Morbidity and mortality of laparoscopic gastrectomy versus open gastrectomy for gastric cancer: an interim report--a phase III multicenter, prospective, randomized Trial (KLASS Trial). Ann Surg. 2010;251:417-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 562] [Cited by in RCA: 624] [Article Influence: 41.6] [Reference Citation Analysis (0)] |
| 6. | Katai H, Sasako M, Fukuda H, Nakamura K, Hiki N, Saka M, Yamaue H, Yoshikawa T, Kojima K; JCOG Gastric Cancer Surgical Study Group. Safety and feasibility of laparoscopy-assisted distal gastrectomy with suprapancreatic nodal dissection for clinical stage I gastric cancer: a multicenter phase II trial (JCOG 0703). Gastric Cancer. 2010;13:238-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 261] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 7. | Katai H, Mizusawa J, Katayama H, Takagi M, Yoshikawa T, Fukagawa T, Terashima M, Misawa K, Teshima S, Koeda K, Nunobe S, Fukushima N, Yasuda T, Asao Y, Fujiwara Y, Sasako M. Short-term surgical outcomes from a phase III study of laparoscopy-assisted versus open distal gastrectomy with nodal dissection for clinical stage IA/IB gastric cancer: Japan Clinical Oncology Group Study JCOG0912. Gastric Cancer. 2017;20:699-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 285] [Article Influence: 35.6] [Reference Citation Analysis (1)] |
| 8. | Shinohara T, Satoh S, Kanaya S, Ishida Y, Taniguchi K, Isogaki J, Inaba K, Yanaga K, Uyama I. Laparoscopic versus open D2 gastrectomy for advanced gastric cancer: a retrospective cohort study. Surg Endosc. 2013;27:286-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 158] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 9. | Nakauchi M, Suda K, Kadoya S, Inaba K, Ishida Y, Uyama I. Technical aspects and short- and long-term outcomes of totally laparoscopic total gastrectomy for advanced gastric cancer: a single-institution retrospective study. Surg Endosc. 2016;30:4632-4639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 10. | Uyama I, Suda K, Satoh S. Laparoscopic surgery for advanced gastric cancer: current status and future perspectives. J Gastric Cancer. 2013;13:19-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 11. | Yoshida K, Honda M, Kumamaru H, Kodera Y, Kakeji Y, Hiki N, Etoh T, Miyata H, Yamashita Y, Seto Y, Kitano S, Konno H. Surgical outcomes of laparoscopic distal gastrectomy compared to open distal gastrectomy: A retrospective cohort study based on a nationwide registry database in Japan. Ann Gastroenterol Surg. 2018;2:55-64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 12. | Hiki N, Honda M, Etoh T, Yoshida K, Kodera Y, Kakeji Y, Kumamaru H, Miyata H, Yamashita Y, Inomata M, Konno H, Seto Y, Kitano S. Higher incidence of pancreatic fistula in laparoscopic gastrectomy. Real-world evidence from a nationwide prospective cohort study. Gastric Cancer. 2018;21:162-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 13. | Kodera Y, Yoshida K, Kumamaru H, Kakeji Y, Hiki N, Etoh T, Honda M, Miyata H, Yamashita Y, Seto Y, Kitano S, Konno H. Introducing laparoscopic total gastrectomy for gastric cancer in general practice: a retrospective cohort study based on a nationwide registry database in Japan. Gastric Cancer. 2019;22:202-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 83] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 14. | Kim MC, Jung GJ, Kim HH. Learning curve of laparoscopy-assisted distal gastrectomy with systemic lymphadenectomy for early gastric cancer. World J Gastroenterol. 2005;11:7508-7511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 140] [Cited by in RCA: 153] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 15. | Jin SH, Kim DY, Kim H, Jeong IH, Kim MW, Cho YK, Han SU. Multidimensional learning curve in laparoscopy-assisted gastrectomy for early gastric cancer. Surg Endosc. 2007;21:28-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 122] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 16. | Kunisaki C, Makino H, Yamamoto N, Sato T, Oshima T, Nagano Y, Fujii S, Akiyama H, Otsuka Y, Ono HA, Kosaka T, Takagawa R, Shimada H. Learning curve for laparoscopy-assisted distal gastrectomy with regional lymph node dissection for early gastric cancer. Surg Laparosc Endosc Percutan Tech. 2008;18:236-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 89] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 17. | Hu WG, Ma JJ, Zang L, Xue P, Xu H, Wang ML, Lu AG, Li JW, Feng B, Zheng MH. Learning curve and long-term outcomes of laparoscopy-assisted distal gastrectomy for gastric cancer. J Laparoendosc Adv Surg Tech A. 2014;24:487-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 18. | Mori T, Kimura T, Kitajima M. Skill accreditation system for laparoscopic gastroenterologic surgeons in Japan. Minim Invasive Ther Allied Technol. 2010;19:18-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 149] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 19. | Suda K, Ishida Y, Kawamura Y, Inaba K, Kanaya S, Teramukai S, Satoh S, Uyama I. Robot-assisted thoracoscopic lymphadenectomy along the left recurrent laryngeal nerve for esophageal squamous cell carcinoma in the prone position: technical report and short-term outcomes. World J Surg. 2012;36:1608-1616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 110] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 20. | Suda K, Man-I M, Ishida Y, Kawamura Y, Satoh S, Uyama I. Potential advantages of robotic radical gastrectomy for gastric adenocarcinoma in comparison with conventional laparoscopic approach: a single institutional retrospective comparative cohort study. Surg Endosc. 2015;29:673-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 170] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 21. | Uyama I, Kanaya S, Ishida Y, Inaba K, Suda K, Satoh S. Novel integrated robotic approach for suprapancreatic D2 nodal dissection for treating gastric cancer: technique and initial experience. World J Surg. 2012;36:331-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 136] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 22. | Wang WJ, Li HT, Yu JP, Su L, Guo CA, Chen P, Yan L, Li K, Ma YW, Wang L, Hu W, Li YM, Liu HB. Severity and incidence of complications assessed by the Clavien-Dindo classification following robotic and laparoscopic gastrectomy for advanced gastric cancer: a retrospective and propensity score-matched study. Surg Endosc. 2019;33:3341-3354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 23. | Uyama I, Suda K, Nakauchi M, Kinoshita T, Noshiro H, Takiguchi S, Ehara K, Obama K, Kuwabara S, Okabe H, Terashima M. Clinical advantages of robotic gastrectomy for clinical stage I/II gastric cancer: a multi-institutional prospective single-arm study. Gastric Cancer. 2019;22:377-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 158] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 24. | Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2390] [Cited by in RCA: 2873] [Article Influence: 205.2] [Reference Citation Analysis (0)] |
| 25. | Yanai H, Matsumoto Y, Harada T, Nishiaki M, Tokiyama H, Shigemitsu T, Tada M, Okita K. Endoscopic ultrasonography and endoscopy for staging depth of invasion in early gastric cancer: a pilot study. Gastrointest Endosc. 1997;46:212-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 111] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 26. | Fusaroli P, Kypraios D, Eloubeidi MA, Caletti G. Levels of evidence in endoscopic ultrasonography: a systematic review. Dig Dis Sci. 2012;57:602-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 27. | Nakauchi M, Suda K, Nakamura K, Shibasaki S, Kikuchi K, Nakamura T, Kadoya S, Ishida Y, Inaba K, Taniguchi K, Uyama I. Laparoscopic subtotal gastrectomy for advanced gastric cancer: technical aspects and surgical, nutritional and oncological outcomes. Surg Endosc. 2017;31:4631-4640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 28. | Catena F, Di Battista M, Ansaloni L, Pantaleo M, Fusaroli P, Di Scioscio V, Santini D, Nannini M, Saponara M, Ponti G, Persiani R, Delrio P, Coccolini F, Di Saverio S, Biasco G, Lazzareschi D, Pinna A; GISTologist Study Group. Microscopic margins of resection influence primary gastrointestinal stromal tumor survival. Onkologie. 2012;35:645-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 29. | Shibasaki S, Suda K, Nakauchi M, Nakamura T, Kadoya S, Kikuchi K, Inaba K, Uyama I. Outermost layer-oriented medial approach for infrapyloric nodal dissection in laparoscopic distal gastrectomy. Surg Endosc. 2018;32:2137-2148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 30. | Nakamura K, Suda K, Suzuki A, Nakauchi M, Shibasaki S, Kikuchi K, Nakamura T, Kadoya S, Inaba K, Uyama I. Intracorporeal Isosceles Right Triangle-shaped Anastomosis in Totally Laparoscopic Distal Gastrectomy. Surg Laparosc Endosc Percutan Tech. 2018;28:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 31. | Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18532] [Cited by in RCA: 24849] [Article Influence: 1183.3] [Reference Citation Analysis (0)] |
| 32. | Katayama H, Kurokawa Y, Nakamura K, Ito H, Kanemitsu Y, Masuda N, Tsubosa Y, Satoh T, Yokomizo A, Fukuda H, Sasako M. Extended Clavien-Dindo classification of surgical complications: Japan Clinical Oncology Group postoperative complications criteria. Surg Today. 2016;46:668-685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 403] [Cited by in RCA: 619] [Article Influence: 61.9] [Reference Citation Analysis (0)] |
| 33. | Suda K, Nakauchi M, Inaba K, Ishida Y, Uyama I. Revising robotic surgery for stomach, potential benefits revised II: prevention of pancreatic fistula. Transl Gastrointest Cancer. 2015;4:461-467. [DOI] [Full Text] |
| 34. | Okabe H, Obama K, Tsunoda S, Matsuo K, Tanaka E, Hisamori S, Sakai Y. Feasibility of robotic radical gastrectomy using a monopolar device for gastric cancer. Surg Today. 2019;49:820-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 35. | Tokunaga M, Makuuchi R, Miki Y, Tanizawa Y, Bando E, Kawamura T, Terashima M. Late phase II study of robot-assisted gastrectomy with nodal dissection for clinical stage I gastric cancer. Surg Endosc. 2016;30:3362-3367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 36. | Shibasaki S, Suda K, Obama K, Yoshida M, Uyama I. Should robotic gastrectomy become a standard surgical treatment option for gastric cancer? Surg Today. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 37. | Inaba K, Satoh S, Ishida Y, Taniguchi K, Isogaki J, Kanaya S, Uyama I. Overlap method: novel intracorporeal esophagojejunostomy after laparoscopic total gastrectomy. J Am Coll Surg. 2010;211:e25-e29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 197] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 38. | Guerra F, Giuliani G, Iacobone M, Bianchi PP, Coratti A. Pancreas-related complications following gastrectomy: systematic review and meta-analysis of open versus minimally invasive surgery. Surg Endosc. 2017;31:4346-4356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 39. | Guerra F, Giuliani G, Formisano G, Bianchi PP, Patriti A, Coratti A. Pancreatic Complications After Conventional Laparoscopic Radical Gastrectomy Versus Robotic Radical Gastrectomy: Systematic Review and Meta-Analysis. J Laparoendosc Adv Surg Tech A. 2018;28:1207-1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 40. | Gotoh M, Miyata H, Hashimoto H, Wakabayashi G, Konno H, Miyakawa S, Sugihara K, Mori M, Satomi S, Kokudo N, Iwanaka T. National Clinical Database feedback implementation for quality improvement of cancer treatment in Japan: from good to great through transparency. Surg Today. 2016;46:38-47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 101] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 41. | Nakauchi M, Suda K, Susumu S, Kadoya S, Inaba K, Ishida Y, Uyama I. Comparison of the long-term outcomes of robotic radical gastrectomy for gastric cancer and conventional laparoscopic approach: a single institutional retrospective cohort study. Surg Endosc. 2016;30:5444-5452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 69] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 42. | Tokunaga M, Tanizawa Y, Bando E, Kawamura T, Terashima M. Poor survival rate in patients with postoperative intra-abdominal infectious complications following curative gastrectomy for gastric cancer. Ann Surg Oncol. 2013;20:1575-1583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 258] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 43. | Aurello P, Cinquepalmi M, Petrucciani N, Moschetta G, Antolino L, Felli F, Giulitti D, Nigri G, D'Angelo F, Valabrega S, Ramacciato G. Impact of Anastomotic Leakage on Overall and Disease-free Survival After Surgery for Gastric Carcinoma: A Systematic Review. Anticancer Res. 2020;40:619-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |