Published online Jun 7, 2018. doi: 10.3748/wjg.v24.i21.2236
Peer-review started: April 4, 2018
First decision: April 19, 2018
Revised: April 27, 2018
Accepted: May 18, 2018
Article in press: May 18, 2018
Published online: June 7, 2018
Processing time: 61 Days and 7.5 Hours
Gastric cancer (GC), with its high incidence and mortality rates, is a highly fatal cancer that is common in East Asia particularly in China. Its recurrence and metastasis are the main causes of its poor prognosis. Circulating tumor cells (CTCs) or other blood biomarkers that are released into the circulating blood stream by tumors are thought to play a crucial role in the recurrence and metastasis of gastric cancer. Therefore, the detection of CTCs and other blood biomarkers has an important clinical significance; in fact, they can help predict the prognosis, assess the staging, monitor the therapeutic effects and determine the drug susceptibility. Recent research has identified many blood biomarkers in GC, such as various serum proteins, autoantibodies against tumor associated antigens, and cell-free DNAs. The analysis of CTCs and circulating cell-free tumor DNA (ctDNA) in the peripheral blood of patients with gastric cancer is called as liquid biopsy. These blood biomarkers provide the disease status for individuals and have clinical meaning. In this review, we focus on the recent scientific advances regarding CTCs and other blood biomarkers, and discuss their origins and clinical meaning.
Core tip: As liquid biopsy, the detection of circulating tumor cells (CTCs) and other blood biomarkers have their certain clinical significance. In this review, we focus on the recent scientific advances of CTCs and some other blood biomarkers, and discuss their origin and clinical usefulness.
- Citation: Li TT, Liu H, Yu J, Shi GY, Zhao LY, Li GX. Prognostic and predictive blood biomarkers in gastric cancer and the potential application of circulating tumor cells. World J Gastroenterol 2018; 24(21): 2236-2246
- URL: https://www.wjgnet.com/1007-9327/full/v24/i21/2236.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i21.2236
Gastric cancer (GC) ranks as the fifth most common malignant tumor and the third leading cause of cancer deaths, with more than 951000 new cases and 723000 deaths estimated per year (GLOBOCAN 2012)[1]. Despite the development of diagnostic techniques, surgical techniques and perioperative management in recent years, the prognostic outcomes for GC remain poor. Because early stage GC tends to be asymptomatic and because mass screening is not popular, most patients in China are diagnosed at an advanced stage[2]. The prognosis of peritoneal metastasis from gastric cancer is very poor. In addition, the median survival is 4-12 mo, and the 5-year actuarial survival rate of patients with peritoneal metastasis is less than 5%[3,4]. Therefore, finding useful diagnostic and monitoring tools for gastric cancer patients should be considered as the most important clinical objectives.
A “liquid biopsy” for gastric cancer patients is used to detect physiological indicators or parameters in the serum; the procedure is less invasive than an endoscopic or surgical biopsy, and it allows practitioners to detect the disease earlier and visualize the dynamics and development of gastric tumors, as well as treatment efficiency and chemotherapy resistance. Carcinoembryonic antigen (CEA), cancer antigen 19-9 (CA19-9) and cancer antigen 72-4 (CA72-4) are regarded as clinically popular gastrointestinal tumor biomarkers. However, their positivity rates are less than 40% in GC patients, and the sensitivity and specificity of these blood biomarkers are not sufficient[5,6]. Indeed, if a blood biomarker is to be used in a population-based screening program, it should be reliable in repeated applications and easily measurable in blood serum or plasma by common laboratory equipment. Moreover, it should be present in the bloodstream before the onset of manifestations and clinical symptoms, be able to distinguish between cancer and inflammation and have high positive predictive value for malignant tumors. Therefore, there is an urgent need to identify more precise and effective blood biomarkers to provide optimal management for GC patients; these blood biomarkers should be able to provide early detection, clinical staging, therapy response monitoring, and prognosis for GC.
Cells can be released into the blood stream from the original tumor and/or corresponding distant metastatic sites. These circulating tumor cells (CTCs) could be collected and detected through respective technologies according to their physical and biologic features. CTCs from cancer patients may be considered as a type of real-time “liquid biopsy” that could provide real-time information about the cancer status. CTCs have already been accepted by the FDA as a prognostic biomarker for monitoring patients with breast, prostate and colorectal cancer[7]. Currently, the concept of “liquid biopsy” has also been accepted for the clinical application of evaluating ctDNAs that apoptotic and necrotic cancer cells discharge into the blood circulation[8]. As we know, there are numerous genetic and epigenetic aberrations that could activate oncogenes and promote tumor progression. Therefore, we have developed sensitive molecular assays for the detection of ctDNAs in the blood plasma to find tumor-specific aberrations. Moreover, several autoantibodies against specific tumor associated antigens (TAAs) that are expressed by cancer cells and can be detected in the blood plasma more than five years prior to diagnosis have already been identified[9]. Therefore, CTC, ctDNAs and autoantibodies could become potential blood biomarkers for gastric cancer[10].
In this article, we focus on the clinical applications of CTC, ctDNAs and autoantibodies after a brief introduction of the biology and detection technologies, and we explore the future prospects of blood biomarkers in gastric cancer patients.
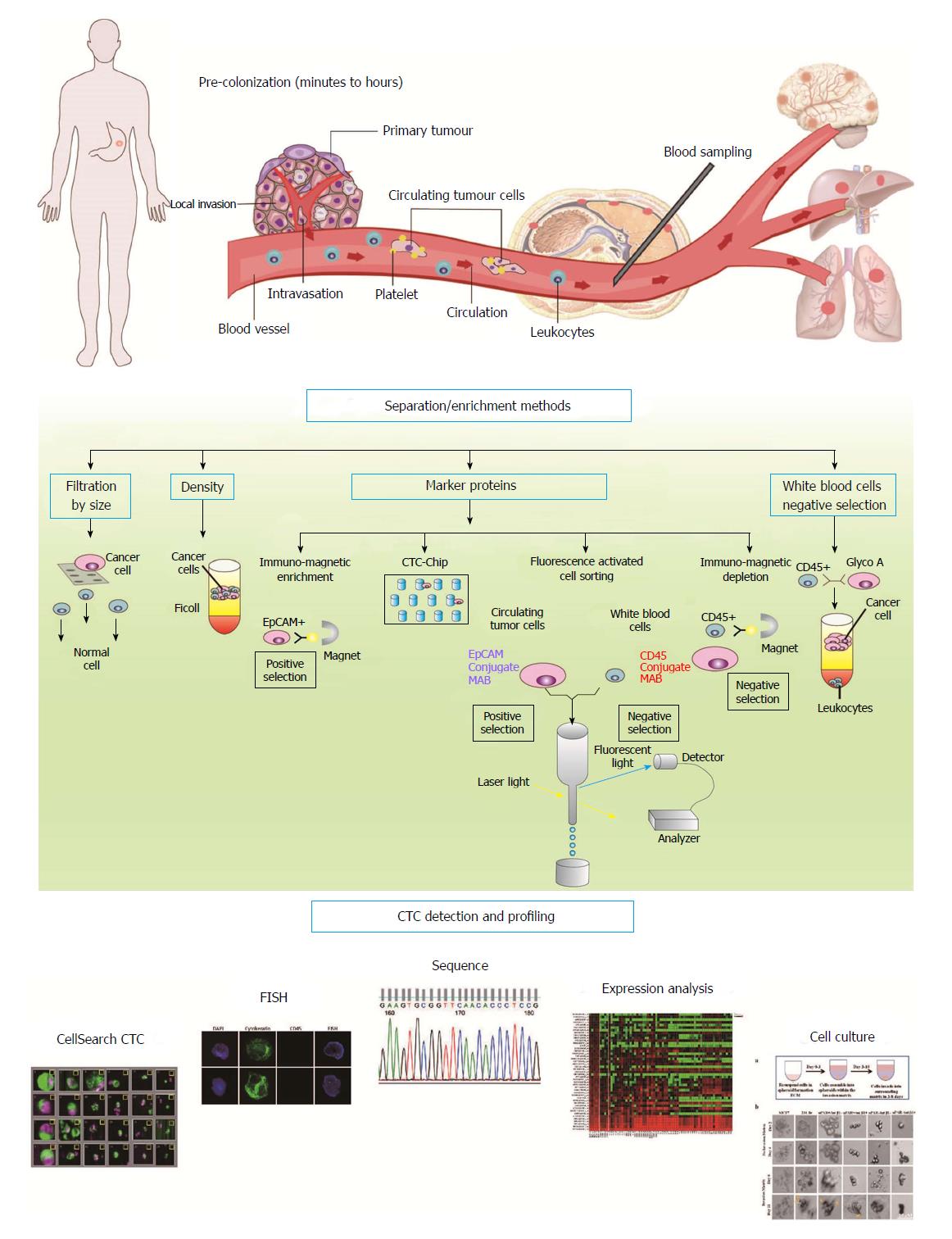
The cancer cells that are released from the original tumor or corresponding distant metastatic sites into the circulating blood are called CTCs. However, these epithelial tumor cells cannot stay in the harsh conditions of the bloodstream, and it is possible that CTCs are selected through these harsh conditions[11]. This proposal is consistent with the phenomenon that there are many apoptotic or fragmented CTCs in the peripheral blood stream of cancer patients[12]. The treacherous journey through the vasculature is necessary for the spread of cancer cells to additional sites. CTCs are closely associated with activated platelets and macrophages[13]. Moreover, the transference of metastatic cancer cells into the circulating blood often relies on various chemokines, such as CCR4, CCR7, CCR9, and CXCR4, which guide the cancer cells across the blood vessels[14]. Even a few months or years after primary tumor removal, CTCs can be detected in the peripheral bloodstream of cancer patients, which indicates that cancer cells can be released into the circulation from other metastatic sites[15,16]. However, how these CTCs give rise to tumor metastasis and progression remains unclear. Future comparative genomic analyses of primary carcinoma and metastatic specimens along with CTCs from the same patient might provide more insight (Figure 1).
Currently, CTCs are often detected by epithelial markers such as epithelial cell adhesion molecule (EpCAM) and cytokeratins (CKs), which are not expressed on the surface of blood cells and distinguish CTCs from the masses of blood cells[17]. Epithelial cancer cells can make an epithelial-to-mesenchymal transition (EMT) that leads to decreased epithelial marker expression and enhanced plasticity and migration and invasion capacity. The CTCs that undergo EMT could be resistant to anoikis, which are necessary for the survival and dissemination of CTCs[17]. It has been previously indicated that EMT might particularly affect the stemness of tumor cells[18]. CTCs that undergo EMT might escape detection by EpCAM-based collection methods, such as the CellSearch system. Our previous study explored mesenchymal markers (Vimentin and Twist) to identify the mesenchymal phenotypes of CTCs in the bloodstream and their relevance to therapy responses[19].
The evolution of various technologies to enrich and detect CTCs has been considerable, even resulting in the detection and verification of new CTC markers[17]. It is vital that we pay close attention to the biological characteristics of tumor cells dissemination and potential stem cell like properties that are affected by EMT, particularly in the field of CTCs[18]. Therefore, many companies have optimized their devices to select and detect CTCs that have undergone EMT[17].
After an enrichment step, we could greatly increase the concentration of CTCs and enable the easy detection of even a single tumor cell. Then, CTCs can be detected by different techniques. In theory, CTCs could be positively or negatively chosen based on physical features (e.g., size, density, deforming character, and electric charges) and biologic features (e.g., the expression of protein markers). The enrichment of positively or negatively chosen CTCs could also be achieved based on particular combinations of physical and biologic features in a device. Then, the CTCs could be detected through immunologic, molecular, and/or functional assays. Recently, increasing numbers of research teams have attempted functional tests using cultures and xenografts of CTCs[20,21]. In vitro and in vivo CTCs models can be applied to detect individualized drug susceptibility. However, the ability to establish CTCs cultures and xenografts of CTCs should be improved to design personalized medicine. Currently, hundreds or thousands of CTCs are required to construct cancer cell cultures or xenografts, which limits this approach to individual therapy (Figure 1).
The new technical developments that we focus on are based on new discoveries in CTC biology. A lack of knowledge has hindered the development of the application of CTCs for clinical diagnosis. However, new significant perspectives regarding the biological meaning of CTCs and various revolutionary techniques have been reported[22]. We believe that equipment for the combined collection, detection, and characterization of CTCs will soon be applied clinically.
Recurrence and metastasis not only predict clinical outcomes but also affect the quality of life of GC patients. They are the most critical factors in the treatment of GC. It was originally thought that incomplete surgical resection resulted in recurrence and metastasis after the operative treatment of GC; therefore, extensive radical resection was applied. However, this procedure was not successful, indicating that there are other possible reasons for recurrence and metastasis. Some researchers found that tumor cells could be released into the bloodstream at the early stage of solid tumors (e.g., breast, colon, lung, and gastric cancer)[7]. Therefore, CTCs may also play a vital role in monitoring the dissemination of gastric cancer and guiding the treatment of GC patients with recurrence and metastasis.
As summarized in Table 1, many studies have reported the clinical value of CTCs as prognostic indicators by different detection methods, including the CellSearch system, RT-PCR/qRT-PCR, and FISH. Uenosono et al[23] detected CTCs using the CellSearch system in 251 gastric cancer patients and found that the overall survival (OS) was obviously lower in patients with CTCs than in patients without CTCs (P < 0.0001). Subgroup analysis revealed that the relapse-free survival and OS were significantly lower in patients with CTCs than in patients without CTCs in the resection group (P < 0.0001). In a prospective study, Matsusaka et al[24] also assessed the correlation between CTCs detected by the CellSearch system and chemotherapy and clinical outcomes. They found that GC patients with at least 4 CTCs at 2 and 4 wk after the onset of chemotherapy had an obviously shorter overall survival and progression-free survival than the patients with less than 4 CTCs. However, the CTCs levels at baseline (i.e., before chemotherapy) had no positive correlation with the clinical outcomes. These findings may indicate that the treatment response of CTCs is correlated with clinical outcomes. The number of studies using RT-PCR/qRT-PCR methods is relatively small. However, Mimori et al[25] detected a candidate marker, the membrane type 1 matrix metalloproteinase (MT1-MMP) mRNA level, in more than 800 GC patients. This marker was chosen based on the results of a cDNA microarray analysis, and its correlation with prognosis was subsequently validated using qRT-PCR. As a consequence, the MT1-MMP mRNA level in the peripheral blood may be an independent prognostic indicator of recurrence and metastasis in GC patients (P = 0.0018).
| Characteristic and number of patients | Detection method | Statistic value | Ref. | ||||
| 17 | RT-PCR | CA19 mRNA | OS | P = 0.014 | CK19 (+) vs (-) | Yeh et al[44], 1998 | |
| I-IV | 57 | RT-PCR | CEA mRNA | Liver metastasis recurrence | P = 0.03 | CEA (+) vs (-) | Miyazono et al[45], 2001 |
| I-IV | 106 | RT-PCR | CEA mRNA | Recurrence/metastasis | P = 0.02 | CEA (+) vs (-) | Sumikura et al[46], 2003 |
| I-IV | 46 | qRT-PCR | CK20 mRNA | 2-yr-survival | P < 0.05 | CK20 (+) vs (-) | Friederichs et al[47], 2005 |
| I-IV | 41 | RT-PCR | CK20 mRNA | OS | P = 0.0363 | CK20 (+) vs (-) | Illert et al[48], 2005 |
| I-III | 46 | RT-PCR | CEA mRNA | Recurrence | P ≤ 0.00022 | CEA after sugery (+) vs (-) | Seo et al[49], 2005 |
| I-IV | 52 | RT-PCR | C-Met mRNA | OS | P = 0.0178 | C-Met (+) vs (-) | Uen et al[50], 2006 |
| MUC1 mRNA | OS | P = 0.0352 | MUC1 (+) vs (-) | ||||
| I-IV | 42 | qRT-PCR | CEA mRNA | Recurrence/metastasis | P = 0.032 | CEA (+) vs (-) | Wu et al[51], 2006 |
| I-IV | 64 | MAH | hTERT/CK19/CEA/MUC1 | Recurrence/metastasis | P = 0.009 | All marker (+) vs the others | Wu et al[52], 2006 |
| I-IV | 57 | RT-PCR | CK20 mRNA | 5-yr survival | P > 0.05 | CK20 (+) vs (-) | Pituch-Noworolska et al[53], 2007 |
| Metastatic | 27 | CellSearch System | EpCAM CK8/18/19 | OS | P = 0.039 | CTC ≥ 2 vs < 2 | Hiraiwa et al[54], 2008 |
| I-IV | 69 | RT-PCR | CK19 mRNA | OS | P = 0.0347 | CK19 (+) vs (-) | Koga et al[55], 2008 |
| CK20 mRNA | OS | P = 0.049 | CK20 (+) vs (-) | ||||
| I-IV | 810 | RT-PCR | MT1-MMP | Recurrence/metastasis | P = 0.0018 | MT1-MMP (+) vs (-) | Mimori et al[25], 2008 |
| I-IV | 55 | RT-PCR, ELISA | Survivin mRNA | RFS | P = 0.026 | Survivin (+) vs (-) | Yie et al[56], 2008 |
| I-IV | 70 | qRT-PCR | Survivin mRNA | OS | P = 0.036 | Survivin high vs low | Bertazza et al[57], 2009 |
| Advanced | 51 (2 wk after chemotherapy) 48 (4 wk after chemotherapy) | CellSearch system | EpCAM CK8/18/19 | PFS ,OS (2 wk after chemotherapy) PFS ,OS (4 wk after chemotherapy) | P < 0.001 | CTC ≥ 4 vs < 4 | Matsusaka et al[24], 2010 |
| I-IV | 123 | qRT-PCR | CEA mRNA | Recurrence | P = 0.001 | CEA (+) vs (-) | Qiu et al[58], 2010 |
| DFS | P = 0.001 | ||||||
| I-IV | 30 | qRT-PCR | CK18 mRNA | RFS | P < 0.001 | CK18 (+) vs (-) | Saad et al[59], 2010 |
| OS | P = 0.001 | ||||||
| I-IV | 95 | qRT-PCR | B7-H3 mRNA | OS | P = 0.046 | B7-H3 high vs low | Arigami et al[60], 2011 |
| I-IV | 98 | RT-PCR, ELISA | Survivin mRNA | DFS | P < 0.001 | Survivin (+) vs (-) | Cao et al[61], 2011 |
| I-IV | 52 | qRT-PCR | miR-200c | OS | P = 0.016 | miR-200c high vs low | Valladares-Ayerbes et al[62], 2012 |
| RFS | P = 0.044 | ||||||
| I-IV | 75 | Immunofluorescence | GFP | OS | P =0.0021 | CTC ≥ 5 vs < 5 | Ito et al[63], 2012 |
| I-IV | 251 | CellSearch system | EpCAM CK8/18/19 | OS | P < 0.001 | CTC (+) vs (-) | Uenosono et al[23], 2013 |
| RFS | P < 0.001 | ||||||
| I-IV | 22 | CellSearch system | EpCAM CK8/18/19 | OS | P = 0.23 | CTC ≥ 2 vs < 2 | Sclafani et al[64], 2014 |
| PFS | P = 0.91 | ||||||
| I-IV | 62 | qRT-PCR | KRT19/MUC1/EPCAM/CEACAM5/BIRC5 mRNA | OS | P = 0.003 | All marker (+) vs the others | Kubisch et al[65], 2015 |
| PFS | P < 0.001 | ||||||
| I-IV | 36 | Flow cytometry | CD133 ABCG2 | OS | P = 0.034 | CD133 (+) vs (-) | Xia et al[66], 2015 |
| I-IV | 136 | CellSearch system | EpCAM CK8/18/19 | PFS | P = 0.016 | CTC (+) vs (-) | Okabe et al[67], 2015 |
| I-IV | 100 | Cell Search system | EpCAM CK8/18/19 | OS | P = 0.004 | CTC ≥ 5 vs < 5 | Lee et al[68], 2015 |
| PFS | P = 0.004 | ||||||
| I-IV | 24 | FACS-ICC | EpCAM | OS | P = 0.014 | CTC ≥ 2 vs < 2 | Meulendijks et al[69], 2016 |
| PFS | P = 0.007 | ||||||
| I-IV | 136 | CellSearch system | EpCAM CK8/18/19 | OS | P < 0.001 | CTC ≥ 3 vs < 3 | Li et al[70], 2016 |
| PFS | P = 0.001 | ||||||
| I-IV | 65 | Immunofluorescence | OBP-401 | OS | P = 0.183 | OBP-401 (+) vs (-) | Ito et al[71], 2016 |
| RFS | P = 0.034 | ||||||
| I-IV | 106 | CellSearch system | EpCAM CK8/18/19 | OS | P = 0.003 | CTC ≥ 2 vs < 2 | Peront et al[72], 2017 |
| RFS | P = 0.0002 | ||||||
| I-IV | 43 | IsoFlux platform | EpCAM | OS | P = 0.0013 | CTC ≥ 17 vs < 17 | Brungs et al[73], 2018 |
Taken together, these studies indicate that CTCs result in GC recurrence and metastasis and may act as vital therapeutic targets for the treatment of GC recurrence and metastasis after radical resection.
Tumor DNA can be released into the blood stream from the primary tumors, circulating tumor cells, or metastases of cancer patients. The majority of circulating cell-free tumor DNAs (ctDNAs) come from apoptotic or necrotic cancer cells that release fragmented DNA into the circulating blood. Dying nonmalignant host cells can also release cell-free DNAs (cfDNAs) into the circulating blood. These normal cfDNAs can dilute the ctDNAs concentrations in cancer patients, particularly in circumstances when tissue-damaging procedures, including surgery, chemotherapy, or radiotherapy, were carried out. Even though the length of DNA fragments might provide some information about the derivation of cfDNAs[26,27], we should further explore the biological features of the ctDNAs in the circulating blood. Several studies have indicated that ctDNAs can even be absorbed by host cells, and this uptake can affect the biology of these host cells[28,29]. Thus, ctDNAs may be indicated as a new target for anti-tumor treatment in order to dilute this type of oncogenic DNA, an idea proposed decades ago[30]. Several clinical applications of ctDNAs have been used for gastric cancer. ctDNAs is not only a tool for the early detection of cancer but also a prognostic or predictive factor (Table 2).
| Candidate biomarkers | Sample size | Sample type | Method/technology | Diagnostic value/outcome | Ref. |
| Total cell-free DNA level b-actin | GC = 53, HC = 21 | Plasma | qPCR | AUC = 0.75, P < 0.0001 | Sai et al[31], 2007 |
| DNA methylation markers RPRM (Reprimo) | GC = 43, HC = 31 | GC tissues and plasma | MSP | 95.3% GC, 9.7% HC, P < 0.00001; Strong correlation between methyl status in tissues and plasma | Bernal et al[74], 2008 |
| Gene amplification MYC gene copy number (MYC/GAPDH ratio) | GC = 57, HC = 39 | Tissues and plasma | qPCR | AUC = 0.816; Strong positive correlation between MYC levels in GC tissues and plasma (r = 0.342; P = 0.009) | Park et al[75], 2009 |
| RUNX3 | GC (preoperative) = 65, GC (postoperative) = 43, HC = 50 | Tissues and serum | qMSP | AUC = 0.8651, Sn = 95.5%, Sp = 62.5%; Decrease after surgical resection | Sakakura et al[76], 2009 |
| KCNA4 + CYP26B1 | GC = 46, GPL = 46, HC = 30 | Serum | Discovery: Methylation microarray in tissues; Testing: MSP | AUC = 0.917, Sn = 91.3%, Sp = 92.1% | Zheng et al[77], 2011 |
| SLC19A3 | Discovery: GC = 45, HC = 60; Validation: GC = 20, HC = 20 | Plasma | MSRED-qPCR | Increased in GC, P < 0.0001 | Ng et al[78], 2011 |
| Alu DNA sequences | GC = 54, HC = 59 | Plasma | Alu81-qPCR | AUC = 0.784, Sn = 75%, Sp = 63% | Park et al[32], 2012 |
| FAM5C + MYLK | GC = 58, GPL = 46, HC = 30 | Serum | Discovery: MeDIP in cell lines; Testing: MSP | AUC = 0.838, Sn = 77.6%, Sp = 90% for GC vs HC; Sn = 30.4% for GPL vs HC; Decrease after surgical resection | Chen et al[79], 2012 |
| XAF1 | GC = 202, HC = 88 | Tumor tissues and serum | qMSP | AUC = 0.909, P < 0.0001; 83.9% concordance between tissues and serum | Ling et al[33], 2013 |
| Total cfDNA level | Early GC = 16; advanced GC = 14; HC = 34 | Plasma | Measurement of cfDNA concentration | AUC = 0.991, Sn = 96.67%, Sp = 94.11% for GC vs HC | Kim et al[80], 2014 |
| HER2 + MYC | GC = 81; gastritis = 63; HC = 32 | Plasma and tissues | FISH and qPCR | AUC = 0.850, Sn = 69%, Sp = 92% | Park et al[34], 2014 |
| HER2 gene copy number (HER2/RPPH1 ratio) | Discovery: GC = 52 (pre and post-operative treatment), HC = 40;Validation: GC = 25 plasma | Plasma and tissues | qPCR | AUC = 0.746, Sn = 53.9%, Sp = 96.7%; Positive correlation between GC tissues and plasma (r = 0.424; P = 0.00721); Decrease in post-treatment plasma in HER2 + GC cases; Sn = 66.7%, Sp = 100% | Shoda et al[35], 2015 |
| TP53 | GC = 6 | Plasma | Parallel sequencing | ctDNATP53 mutation in three out of six patients (50%) | Hamakawa et al[81], 2015 |
| AKT1, AKT3, PIK3CA, PTEN, ARID1A, TP53 and BRAF | GC = 277 | Plasma and tissues | MassARRAY system | 32 out of 94 patients (34%) with a tissue mutation had a corresponding mutation in plasma | Fang et al[82], 2016 |
| HER2 | GC = 70 | Plasma and tissues | dual-color ISH assay | ctDNA had a high concordance of HER2 amplification with tumor tissues(91.4%, Kappa index = 0.784, P < 0.001) | Gao et al[83], 2017 |
| HER2 | GC = 60; HC = 30 | Plasma and tissues | digital droplet PCR | The preoperative plasma HER2 ratio correlated with the tumor HER2 status (P < 0.001); Sn = 73.3%, Sp = 93.3% | Shoda et al[36], 2017 |
Among previous studies of ctDNAs in GC patients, we found that some studies focused on the concentration of ctDNAs. In these studies, the housekeeping gene, beta-actin[31], and a non-coding DNA sequence, ALU[32], were assessed. By comparison, the most widely used method for detecting ctDNAs is the measurement of methylated DNA in the plasma or serum; this measurement is often performed with methylation specific-PCR (MSP) or quantitative methylation specific-PCR (qMSP) assays. With the advances in technology and verification of more sensitive and specific genes, evidence has accumulated in this field. Comprehensive analyses using methylation CpG island microarray have indicated the possibility of more meaningful genes for measuring methylated DNA. Furthermore, Ling et al[33] have shown the effective application of methylated XAF1 DNA. This DNA could be used as a diagnostic or prognostic biomarker with high specificity and sensitivity. In addition to mutation analyses, gene amplification appears to provide relevant blood biomarkers. Park et al[34] found that the combination of plasma HER2 and MYC concentrations to diagnose GC had a sensitivity and specificity of 69% and 92%, respectively. To determine the effect of sequencing methods upon the overall diagnostic accuracy, Shoda et al[35] compared qPCR and digital droplet PCR (ddPCR)[36] for detecting the HER2 amplification ratio in 60 patients with GC. A correlation between the plasma and tissue HER2 amplification ratios was observed by ddPCR (ρ = 0.424, 95%CI: 0.125-0.652, P = 0.00721).
Cancer-associated autoantibodies
IgG Autoantibodies against specific tumor associated antigens (TAAs) can be detected in the blood more than five years prior to a clinical diagnosis of cancer, thus indicating their important role in the prognosis of early-stage cancer[37,38]. Additionally, autoantibodies have other promising biomarker qualities: they can be detected in every type of tumor that has ever been tested[39,40] and they are very stable and have antigen specificity. Assessing the autoantibody response against TAAs with multiplex immunoassays is supposed to be viable, and this method might make them clinically applicable.
To the best of our knowledge, ten studies have reported the clinical diagnosis values of diverse GC associated autoantibodies or their combinations (Table 3). In these studies, the recognized biomarkers can distinguish GC patients from healthy controls with comparatively excellent specificity (87%-100%), but discrepant sensitivity (19.3%-98.9%). There are three studies that described the AUC: Zhou et al[41] reported that autoantibodies against seven TAAs could distinguish GC patients from healthy subjects with an AUC of 0.73. Zayakin et al[42] showed that 45 autoantibodies could distinguish GC patients from healthy subjects with an AUC of 0.79, while Meistere et al[43] reported an AUC of 0.60. These ten studies of autoantibodies in GC vary greatly regarding the number of autoantibodies measured (ranging from 2 to 102), the techniques used to detect the autoantibodies, the definition of suitable control groups, and the methods used to normalize the data and define cut-off values. Taken together, these factors may greatly hinder the clinical application of the reported biomarkers.
| Biomarker signature description | Technology | Study design | Sample size (GC/controls) | Diagnostic value | Ref. |
| 2 TAAs-p62 and Koc | ELISA | GC vs HC | 135/82 | Sn = 19.3%, Sp = 97.6%, P < 0.01 | Zhang et al[84], 2001 |
| 3TAAs-IQGAP3, KRT23 and REG3A | PARSE assay | GC vs HC (age and sex matched) | 48/46 | Sn = 22.9%, Sp = 100%, P < 0.001 | Xu et al[85], 2012 |
| 3 TAAs-p16, p53 and c-myc | ELISA | GC vs HC | 74/82 | Sn = 21.6%, Sp = 97.6%; P < 0.001 | Looi et al[86], 2006 |
| 6 TAAs-p53, Hsp70, HCC-22-5, PrxVI, KM-HN-1 and p90 | ELISA | GC vs HC, training set | 100/79 | Sn = 49.0%, Sp = 92.4%, P < 0.01 | Hoshion et al[87], 2017 |
| GC vs HC, validation set | 248/74 | Sn = 52.0%, Sp = 90.5%, P < 0.01 | |||
| 7 TAAs - p53, C-myc, p16, IMP1, Koc, p62 and Survivin | ELISA | Cardia GC vs HC | 88/140 | AUC = 0.73, Sn = 64%, Sp = 87%, P < 0.001 | Zhou et al[41], 2015 |
| 7 TAAs - C-myc, Cyclin B1, IMP1, Koc, P53, p62 and Survivin | ELISA, fixed cut-off | GC vs HC | 91/346 | Sn = 52.7%, Sp = 89.9%, P < 0.01 | Zhang et al[88], 2003 |
| ELISA, individual cut-off | GC vs HC | 91/346 | Sn = 98.9%, Sp = 93.1%, P < 0.001 | Koziol et al[89], 2003 | |
| 45 T7 phage-displayed TAA clones (including NY-ESO-1, DDX53, MAGE antigens etc.) | T7 phage displayed TAA microarray | GC vs HC (age and sex matched) | T:100/100 | AUC = 0.79, Sn = 59%, Sp = 90%, P < 0.001 | Zayakin et al[42], 2013 |
| V:235/213 | AUC = 0.64, Sn = 58.7%, Sp = 55%, P < 0.001 | ||||
| GC vs gastritis | 235/100 | AUC = 0.76, Sn = 58.7%, Sp = 81.5%, P < 0.001 | |||
| GC vs gastric ulcer | 235/54 | ||||
| 64 TAAs (including MAGEA4, CTAG1, TP53, ERBB2_C and SDCCAG8 antigens etc.) | Bead-based multiplex serology | GC vs HC | T:155/224 | Sn = 0-12%, Sp = 98%; P > 0.05 | Werner et al[90], 2016 |
| GC vs HC | V:146/97 | Sn = 32%, Sp = 87%; P < 0.001 | |||
| 102 TAAs (including CTAG1B/CTAG2, DDX53, IGF2BP2, TP53 and MAGEA3 antigens etc.) | A recombinant antigen microarray | GC vs HC | 829/929 | AUC = 0.60, Sn = 21%, Sp = 91%, P < 0.001 | Meistere et al[43], 2017 |
In general, measuring autoantibodies against TAAs has been reported to have excellent specificity but general sensitivity, which would hamper its use in clinical medicine. The biological mechanisms underlying the limitations of autoantibody sensitivity are currently unknown. Additionally, the heterogeneity of TAAs among cancer patients is very high, and one cancer-specific autoantibody usually has a low probability of detection and is thus unlikely to have statistical significance. Therefore, recently published studies are likely to be statistically inefficient. However, diagnostic biomarker panels result in the low repeatability of initial results and reduce the diagnostic value of autoantibodies, but this issue could be remedied by analyzing combinations with good statistical significance.
In general, the field of CTCs, ctDNAs and autoantibodies is stimulating discovery regarding the tumor recurrence and metastasis, but it is still in the early stages. The transformation of these blood biomarkers into conventional clinical indicators is hampered by the absence of consistency among different technical methods. The CellSearch system is the first standardized semi-automatic technique approved by the FDA to enrich and detect CTCs in patients with breast, prostate or colorectal cancer. Many studies have shown that the results of CTCs detection with the CellSearch system could serve as a clinical prognostic and therapeutic effectiveness indicator for these cancers. Recently, a few studies have shown that detection of CTCs in GC patients using the CellSearch system could be used for staging, predicting patients’ overall survival and evaluating the treatment effectiveness. However, large-scale clinical studies are needed to further validate the important role of CTCs and to explore an applicable cut-off value for the CTCs score in GC patients.
Although various methods and techniques have been recommended for ultimately establishing an applicable, sensitive and real-time monitoring system using circulating blood, few methods can currently be applied in clinical practice. Large-scale clinical trials and further exploration of the biology and significance of blood biomarkers might solve the associated problems and improve their application as blood biomarkers. Therefore, the exploration of revolutionary blood biomarkers, such as CTCs, ctDNAs and autoantibodies, could provide many advantages for gastric cancer patients and improve their clinical outcomes in the future.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Aoyagi K, Surlin VM S- Editor: Gong ZM L- Editor: A E- Editor: Huang Y
| 1. | Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359-E386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20108] [Cited by in RCA: 20495] [Article Influence: 2049.5] [Reference Citation Analysis (20)] |
| 2. | Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11444] [Cited by in RCA: 13201] [Article Influence: 1466.8] [Reference Citation Analysis (3)] |
| 3. | Sarela AI, Miner TJ, Karpeh MS, Coit DG, Jaques DP, Brennan MF. Clinical outcomes with laparoscopic stage M1, unresected gastric adenocarcinoma. Ann Surg. 2006;243:189-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 75] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 4. | Lasithiotakis K, Antoniou SA, Antoniou GA, Kaklamanos I, Zoras O. Gastrectomy for stage IV gastric cancer. a systematic review and meta-analysis. Anticancer Res. 2014;34:2079-2085. [PubMed] |
| 5. | Li Y, Yang Y, Lu M, Shen L. Predictive value of serum CEA, CA19-9 and CA72.4 in early diagnosis of recurrence after radical resection of gastric cancer. Hepatogastroenterology. 2011;58:2166-2170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 6. | Ishigami S, Natsugoe S, Hokita S, Che X, Tokuda K, Nakajo A, Iwashige H, Tokushige M, Watanabe T, Takao S. Clinical importance of preoperative carcinoembryonic antigen and carbohydrate antigen 19-9 levels in gastric cancer. J Clin Gastroenterol. 2001;32:41-44. [PubMed] |
| 7. | Pantel K, Alix-Panabières C. Circulating tumour cells in cancer patients: challenges and perspectives. Trends Mol Med. 2010;16:398-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 576] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 8. | Diaz LA Jr, Bardelli A. Liquid biopsies: genotyping circulating tumor DNA. J Clin Oncol. 2014;32:579-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1413] [Cited by in RCA: 1682] [Article Influence: 152.9] [Reference Citation Analysis (0)] |
| 9. | Turk MJ, Wolchok JD, Guevara-Patino JA, Goldberg SM, Houghton AN. Multiple pathways to tumor immunity and concomitant autoimmunity. Immunol Rev. 2002;188:122-135. [PubMed] |
| 10. | Haber DA, Velculescu VE. Blood-based analyses of cancer: circulating tumor cells and circulating tumor DNA. Cancer Discov. 2014;4:650-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 462] [Cited by in RCA: 556] [Article Influence: 50.5] [Reference Citation Analysis (0)] |
| 11. | Kang Y, Pantel K. Tumor cell dissemination: emerging biological insights from animal models and cancer patients. Cancer Cell. 2013;23:573-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 321] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 12. | Larson CJ, Moreno JG, Pienta KJ, Gross S, Repollet M, O’hara SM, Russell T, Terstappen LW. Apoptosis of circulating tumor cells in prostate cancer patients. Cytometry A. 2004;62:46-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 115] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 13. | Smith HA, Kang Y. The metastasis-promoting roles of tumor-associated immune cells. J Mol Med (Berl). 2013;91:411-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 283] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 14. | Bonecchi R, Galliera E, Borroni EM, Corsi MM, Locati M, Mantovani A. Chemokines and chemokine receptors: an overview. Front Biosci (Landmark Ed). 2009;14:540-551. [PubMed] |
| 15. | Meng S, Tripathy D, Frenkel EP, Shete S, Naftalis EZ, Huth JF, Beitsch PD, Leitch M, Hoover S, Euhus D. Circulating tumor cells in patients with breast cancer dormancy. Clin Cancer Res. 2004;10:8152-8162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 666] [Cited by in RCA: 728] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 16. | Müller V, Stahmann N, Riethdorf S, Rau T, Zabel T, Goetz A, Jänicke F, Pantel K. Circulating tumor cells in breast cancer: correlation to bone marrow micrometastases, heterogeneous response to systemic therapy and low proliferative activity. Clin Cancer Res. 2005;11:3678-3685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 303] [Cited by in RCA: 282] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 17. | Alix-Panabières C, Pantel K. Challenges in circulating tumour cell research. Nat Rev Cancer. 2014;14:623-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 917] [Cited by in RCA: 1011] [Article Influence: 91.9] [Reference Citation Analysis (0)] |
| 18. | Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133:704-715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6972] [Cited by in RCA: 6793] [Article Influence: 399.6] [Reference Citation Analysis (0)] |
| 19. | Li TT, Liu H, Li FP, Hu YF, Mou TY, Lin T, Yu J, Zheng L, Li GX. Evaluation of epithelial-mesenchymal transitioned circulating tumor cells in patients with resectable gastric cancer: Relevance to therapy response. World J Gastroenterol. 2015;21:13259-13267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 20. | Pantel K, Alix-Panabières C. Functional Studies on Viable Circulating Tumor Cells. Clin Chem. 2016;62:328-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 21. | Alix-Panabières C, Pantel K. Liquid biopsy in cancer patients: advances in capturing viable CTCs for functional studies using the EPISPOT assay. Expert Rev Mol Diagn. 2015;15:1411-1417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 22. | Pantel K, Speicher MR. The biology of circulating tumor cells. Oncogene. 2016;35:1216-1224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 374] [Article Influence: 41.6] [Reference Citation Analysis (0)] |
| 23. | Uenosono Y, Arigami T, Kozono T, Yanagita S, Hagihara T, Haraguchi N, Matsushita D, Hirata M, Arima H, Funasako Y. Clinical significance of circulating tumor cells in peripheral blood from patients with gastric cancer. Cancer. 2013;119:3984-3991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 24. | Matsusaka S, Chìn K, Ogura M, Suenaga M, Shinozaki E, Mishima Y, Terui Y, Mizunuma N, Hatake K. Circulating tumor cells as a surrogate marker for determining response to chemotherapy in patients with advanced gastric cancer. Cancer Sci. 2010;101:1067-1071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 93] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 25. | Mimori K, Fukagawa T, Kosaka Y, Ishikawa K, Iwatsuki M, Yokobori T, Hirasaki S, Takatsuno Y, Sakashita H, Ishii H. A large-scale study of MT1-MMP as a marker for isolated tumor cells in peripheral blood and bone marrow in gastric cancer cases. Ann Surg Oncol. 2008;15:2934-2942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 26. | Heitzer E, Auer M, Hoffmann EM, Pichler M, Gasch C, Ulz P, Lax S, Waldispuehl-Geigl J, Mauermann O, Mohan S. Establishment of tumor-specific copy number alterations from plasma DNA of patients with cancer. Int J Cancer. 2013;133:346-356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 134] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 27. | Lo YM, Zhang J, Leung TN, Lau TK, Chang AM, Hjelm NM. Rapid clearance of fetal DNA from maternal plasma. Am J Hum Genet. 1999;64:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 812] [Cited by in RCA: 810] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 28. | Schwarzenbach H, Hoon DS, Pantel K. Cell-free nucleic acids as biomarkers in cancer patients. Nat Rev Cancer. 2011;11:426-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1954] [Cited by in RCA: 2140] [Article Influence: 152.9] [Reference Citation Analysis (0)] |
| 29. | Trejo-Becerril C, Pérez-Cárdenas E, Taja-Chayeb L, Anker P, Herrera-Goepfert R, Medina-Velázquez LA, Hidalgo-Miranda A, Pérez-Montiel D, Chávez-Blanco A, Cruz-Velázquez J. Cancer progression mediated by horizontal gene transfer in an in vivo model. PLoS One. 2012;7:e52754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 99] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 30. | DE LAMIRANDE G. Action of deoxyribonuclease and ribonuclease on the growth of Ehrlich ascites carcinoma in mice. Nature. 1961;192:52-54. [PubMed] |
| 31. | Sai S, Ichikawa D, Tomita H, Ikoma D, Tani N, Ikoma H, Kikuchi S, Fujiwara H, Ueda Y, Otsuji E. Quantification of plasma cell-free DNA in patients with gastric cancer. Anticancer Res. 2007;27:2747-2751. [PubMed] |
| 32. | Park JL, Kim HJ, Choi BY, Lee HC, Jang HR, Song KS, Noh SM, Kim SY, Han DS, Kim YS. Quantitative analysis of cell-free DNA in the plasma of gastric cancer patients. Oncol Lett. 2012;3:921-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 33. | Ling ZQ, Lv P, Lu XX, Yu JL, Han J, Ying LS, Zhu X, Zhu WY, Fang XH, Wang S. Circulating Methylated XAF1 DNA Indicates Poor Prognosis for Gastric Cancer. PLoS One. 2013;8:e67195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 34. | Park KU, Lee HE, Nam SK, Nam KH, Park DJ, Kim HH, Kim WH, Lee HS. The quantification of HER2 and MYC gene fragments in cell-free plasma as putative biomarkers for gastric cancer diagnosis. Clin Chem Lab Med. 2014;52:1033-1040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Shoda K, Masuda K, Ichikawa D, Arita T, Miyakami Y, Watanabe M, Konishi H, Imoto I, Otsuji E. HER2 amplification detected in the circulating DNA of patients with gastric cancer: a retrospective pilot study. Gastric Cancer. 2015;18:698-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 36. | Shoda K, Ichikawa D, Fujita Y, Masuda K, Hiramoto H, Hamada J, Arita T, Konishi H, Komatsu S, Shiozaki A. Monitoring the HER2 copy number status in circulating tumor DNA by droplet digital PCR in patients with gastric cancer. Gastric Cancer. 2017;20:126-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 104] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 37. | Chapman C, Murray A, Chakrabarti J, Thorpe A, Woolston C, Sahin U, Barnes A, Robertson J. Autoantibodies in breast cancer: their use as an aid to early diagnosis. Ann Oncol. 2007;18:868-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 200] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 38. | Zhong L, Coe SP, Stromberg AJ, Khattar NH, Jett JR, Hirschowitz EA. Profiling tumor-associated antibodies for early detection of non-small cell lung cancer. J Thorac Oncol. 2006;1:513-519. [PubMed] |
| 39. | Preuss KD, Zwick C, Bormann C, Neumann F, Pfreundschuh M. Analysis of the B-cell repertoire against antigens expressed by human neoplasms. Immunol Rev. 2002;188:43-50. [PubMed] |
| 40. | Scanlan MJ. Identification of human tumor antigens by serological analysis of recombinant cDNA expression libraries (SEREX). Curr Protoc Immunol. 2005;20:Unit 20.7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 41. | Zhou SL, Ku JW, Fan ZM, Yue WB, Du F, Zhou YF, Liu YL, Li Y, Tang S, Hu YL. Detection of autoantibodies to a panel of tumor-associated antigens for the diagnosis values of gastric cardia adenocarcinoma. Dis Esophagus. 2015;28:371-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 42. | Zayakin P, Ancāns G, Siliņa K, Meistere I, Kalniņa Z, Andrejeva D, Endzeliņš E, Ivanova L, Pismennaja A, Ruskule A. Tumor-associated autoantibody signature for the early detection of gastric cancer. Int J Cancer. 2013;132:137-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 43. | Meistere I, Werner S, Zayakin P, Siliņa K, Rulle U, Pismennaja A, Šantare D, Kikuste I, Isajevs S, Leja M. The Prevalence of Cancer-Associated Autoantibodies in Patients with Gastric Cancer and Progressive Grades of Premalignant Lesions. Cancer Epidemiol Biomarkers Prev. 2017;26:1564-1574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 44. | Yeh KH, Chen YC, Yeh SH, Chen CP, Lin JT, Cheng AL. Detection of circulating cancer cells by nested reverse transcription-polymerase chain reaction of cytokeratin-19 (K19)--possible clinical significance in advanced gastric cancer. Anticancer Res. 1998;18:1283-1286. [PubMed] |
| 45. | Miyazono F, Natsugoe S, Takao S, Tokuda K, Kijima F, Aridome K, Hokita S, Baba M, Eizuru Y, Aikou T. Surgical maneuvers enhance molecular detection of circulating tumor cells during gastric cancer surgery. Ann Surg. 2001;233:189-194. [PubMed] |
| 46. | Sumikura S, Ishigami S, Natsugoe S, Miyazono F, Tokuda K, Nakajo A, Okumura H, Matsumoto M, Hokita S, Aikou T. Disseminated cancer cells in the blood and expression of sialylated antigen in gastric cancer. Cancer Lett. 2003;200:77-83. [PubMed] |
| 47. | Friederichs J, Gertler R, Rosenberg R, Nahrig J, Führer K, Holzmann B, Dittler HJ, Dahm M, Thorban S, Nekarda H. Prognostic impact of CK-20-positive cells in peripheral venous blood of patients with gastrointestinal carcinoma. World J Surg. 2005;29:422-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 48. | Illert B, Fein M, Otto C, Cording F, Stehle D, Thiede A, Timmermann W. Disseminated tumor cells in the blood of patients with gastric cancer are an independent predictive marker of poor prognosis. Scand J Gastroenterol. 2005;40:843-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 49. | Seo JH, Choi CW, Kim BS, Shin SW, Kim YH, Kim JS, Lee SW, Choi JH, Park YT, Mok YJ. Follow-up study of peripheral blood carcinoembryonic antigen mRNA using reverse transcription-polymerase chain reaction as an early marker of clinical recurrence in patients with curatively resected gastric cancer. Am J Clin Oncol. 2005;28:24-29. [PubMed] |
| 50. | Uen YH, Lin SR, Wu CH, Hsieh JS, Lu CY, Yu FJ, Huang TJ, Wang JY. Clinical significance of MUC1 and c-Met RT-PCR detection of circulating tumor cells in patients with gastric carcinoma. Clin Chim Acta. 2006;367:55-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 51. | Wu CH, Lin SR, Hsieh JS, Chen FM, Lu CY, Yu FJ, Cheng TL, Huang TJ, Huang SY, Wang JY. Molecular detection of disseminated tumor cells in the peripheral blood of patients with gastric cancer: evaluation of their prognostic significance. Dis Markers. 2006;22:103-109. [PubMed] |
| 52. | Wu CH, Lin SR, Yu FJ, Wu DC, Pan YS, Hsieh JS, Huang SY, Wang JY. Development of a high-throughput membrane-array method for molecular diagnosis of circulating tumor cells in patients with gastric cancers. Int J Cancer. 2006;119:373-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 53. | Pituch-Noworolska A, Kolodziejczyk P, Kulig J, Drabik G, Szczepanik A, Czupryna A, Popiela T, Zembala M. Circulating tumour cells and survival of patients with gastric cancer. Anticancer Res. 2007;27:635-640. [PubMed] |
| 54. | Hiraiwa K, Takeuchi H, Hasegawa H, Saikawa Y, Suda K, Ando T, Kumagai K, Irino T, Yoshikawa T, Matsuda S. Clinical significance of circulating tumor cells in blood from patients with gastrointestinal cancers. Ann Surg Oncol. 2008;15:3092-3100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 150] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 55. | Koga T, Tokunaga E, Sumiyoshi Y, Oki E, Oda S, Takahashi I, Kakeji Y, Baba H, Maehara Y. Detection of circulating gastric cancer cells in peripheral blood using real time quantitative RT-PCR. Hepatogastroenterology. 2008;55:1131-1135. [PubMed] |
| 56. | Yie SM, Lou B, Ye SR, Cao M, He X, Li P, Hu K, Rao L, Wu SM, Xiao HB. Detection of survivin-expressing circulating cancer cells (CCCs) in peripheral blood of patients with gastric and colorectal cancer reveals high risks of relapse. Ann Surg Oncol. 2008;15:3073-3082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 57. | Bertazza L, Mocellin S, Marchet A, Pilati P, Gabrieli J, Scalerta R, Nitti D. Survivin gene levels in the peripheral blood of patients with gastric cancer independently predict survival. J Transl Med. 2009;7:111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 58. | Qiu MZ, Li ZH, Zhou ZW, Li YH, Wang ZQ, Wang FH, Huang P, Aziz F, Wang DY, Xu RH. Detection of carcinoembryonic antigen messenger RNA in blood using quantitative real-time reverse transcriptase-polymerase chain reaction to predict recurrence of gastric adenocarcinoma. J Transl Med. 2010;8:107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 59. | Saad AA, Awed NM, Abd Elkerim NN, El-Shennawy D, Alfons MA, Elserafy ME, Darwish YW, Barakat EM, Ezz-Elarab SS. Prognostic significance of E-cadherin expression and peripheral blood micrometastasis in gastric carcinoma patients. Ann Surg Oncol. 2010;17:3059-3067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 60. | Arigami T, Uenosono Y, Hirata M, Yanagita S, Ishigami S, Natsugoe S. B7-H3 expression in gastric cancer: a novel molecular blood marker for detecting circulating tumor cells. Cancer Sci. 2011;102:1019-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 61. | Cao W, Yang W, Li H, Lou G, Jiang J, Geng M, Xi W, Ren R, Qu Q, Jin X. Using detection of survivin-expressing circulating tumor cells in peripheral blood to predict tumor recurrence following curative resection of gastric cancer. J Surg Oncol. 2011;103:110-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 62. | Valladares-Ayerbes M, Reboredo M, Medina-Villaamil V, Iglesias-Díaz P, Lorenzo-Patiño MJ, Haz M, Santamarina I, Blanco M, Fernández-Tajes J, Quindós M. Circulating miR-200c as a diagnostic and prognostic biomarker for gastric cancer. J Transl Med. 2012;10:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 123] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 63. | Ito H, Inoue H, Sando N, Kimura S, Gohda K, Sato J, Murakami K, Ito S, Odaka N, Satodate H. Prognostic impact of detecting viable circulating tumour cells in gastric cancer patients using a telomerase-specific viral agent: a prospective study. BMC Cancer. 2012;12:346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 64. | Sclafani F, Smyth E, Cunningham D, Chau I, Turner A, Watkins D. A pilot study assessing the incidence and clinical significance of circulating tumor cells in esophagogastric cancers. Clin Colorectal Cancer. 2014;13:94-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 65. | Kubisch I, de Albuquerque A, Schuppan D, Kaul S, Schaich M, Stölzel U. Prognostic Role of a Multimarker Analysis of Circulating Tumor Cells in Advanced Gastric and Gastroesophageal Adenocarcinomas. Oncology. 2015;89:294-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 66. | Xia P, Song CL, Liu JF, Wang D, Xu XY. Prognostic value of circulating CD133(+) cells in patients with gastric cancer. Cell Prolif. 2015;48:311-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 67. | Okabe H, Tsunoda S, Hosogi H, Hisamori S, Tanaka E, Tanaka S, Sakai Y. Circulating Tumor Cells as an Independent Predictor of Survival in Advanced Gastric Cancer. Ann Surg Oncol. 2015;22:3954-3961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 68. | Lee SJ, Lee J, Kim ST, Park SH, Park JO, Park YS, Lim HY, Kang WK. Circulating tumor cells are predictive of poor response to chemotherapy in metastatic gastric cancer. Int J Biol Markers. 2015;30:e382-e386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 69. | Meulendijks D, de Groot JW, Los M, Boers JE, Beerepoot LV, Polee MB, Beeker A, Portielje JE, Goey SH, de Jong RS. Bevacizumab combined with docetaxel, oxaliplatin, and capecitabine, followed by maintenance with capecitabine and bevacizumab, as first-line treatment of patients with advanced HER2-negative gastric cancer: A multicenter phase 2 study. Cancer. 2016;122:1434-1443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 70. | Li Y, Gong J, Zhang Q, Lu Z, Gao J, Li Y, Cao Y, Shen L. Dynamic monitoring of circulating tumour cells to evaluate therapeutic efficacy in advanced gastric cancer. Br J Cancer. 2016;114:138-145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 78] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 71. | Ito H, Sato J, Tsujino Y, Yamaguchi N, Kimura S, Gohda K, Murakami K, Onimaru M, Ohmori T, Ishikawa F. Long-term prognostic impact of circulating tumour cells in gastric cancer patients. World J Gastroenterol. 2016;22:10232-10241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 72. | Pernot S, Badoual C, Terme M, Castan F, Cazes A, Bouche O, Bennouna J, Francois E, Ghiringhelli F, De La Fouchardiere C. Dynamic evaluation of circulating tumour cells in patients with advanced gastric and oesogastric junction adenocarcinoma: Prognostic value and early assessment of therapeutic effects. Eur J Cancer. 2017;79:15-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 73. | Brungs D, Lynch D, Luk AW, Minaei E, Ranson M, Aghmesheh M, Vine KL, Carolan M, Jaber M, de Souza P. Cryopreservation for delayed circulating tumor cell isolation is a valid strategy for prognostic association of circulating tumor cells in gastroesophageal cancer. World J Gastroenterol. 2018;24:810-818. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 74. | Bernal C, Aguayo F, Villarroel C, Vargas M, Díaz I, Ossandon FJ, Santibáñez E, Palma M, Aravena E, Barrientos C. Reprimo as a potential biomarker for early detection in gastric cancer. Clin Cancer Res. 2008;14:6264-6269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 96] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 75. | Park KU, Lee HE, Park DJ, Jung EJ, Song J, Kim HH, Choe G, Kim WH, Lee HS. MYC quantitation in cell-free plasma DNA by real-time PCR for gastric cancer diagnosis. Clin Chem Lab Med. 2009;47:530-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 76. | Sakakura C, Hamada T, Miyagawa K, Nishio M, Miyashita A, Nagata H, Ida H, Yazumi S, Otsuji E, Chiba T. Quantitative analysis of tumor-derived methylated RUNX3 sequences in the serum of gastric cancer patients. Anticancer Res. 2009;29:2619-2625. [PubMed] |
| 77. | Zheng Y, Chen L, Li J, Yu B, Su L, Chen X, Yu Y, Yan M, Liu B, Zhu Z. Hypermethylated DNA as potential biomarkers for gastric cancer diagnosis. Clin Biochem. 2011;44:1405-1411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 78. | Ng EK, Leung CP, Shin VY, Wong CL, Ma ES, Jin HC, Chu KM, Kwong A. Quantitative analysis and diagnostic significance of methylated SLC19A3 DNA in the plasma of breast and gastric cancer patients. PLoS One. 2011;6:e22233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 79. | Chen L, Su L, Li J, Zheng Y, Yu B, Yu Y, Yan M, Gu Q, Zhu Z, Liu B. Hypermethylated FAM5C and MYLK in serum as diagnosis and pre-warning markers for gastric cancer. Dis Markers. 2012;32:195-202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 80. | Kim K, Shin DG, Park MK, Baik SH, Kim TH, Kim S, Lee S. Circulating cell-free DNA as a promising biomarker in patients with gastric cancer: diagnostic validity and significant reduction of cfDNA after surgical resection. Ann Surg Treat Res. 2014;86:136-142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 114] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 81. | Hamakawa T, Kukita Y, Kurokawa Y, Miyazaki Y, Takahashi T, Yamasaki M, Miyata H, Nakajima K, Taniguchi K, Takiguchi S. Monitoring gastric cancer progression with circulating tumour DNA. Br J Cancer. 2015;112:352-356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 82. | Fang WL, Lan YT, Huang KH, Liu CA, Hung YP, Lin CH, Jhang FY, Chang SC, Chen MH, Chao Y. Clinical significance of circulating plasma DNA in gastric cancer. Int J Cancer. 2016;138:2974-2983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 65] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 83. | Gao J, Wang H, Zang W, Li B, Rao G, Li L, Yu Y, Li Z, Dong B, Lu Z. Circulating tumor DNA functions as an alternative for tissue to overcome tumor heterogeneity in advanced gastric cancer. Cancer Sci. 2017;108:1881-1887. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 84. | Zhang JY, Chan EK, Peng XX, Lu M, Wang X, Mueller F, Tan EM. Autoimmune responses to mRNA binding proteins p62 and Koc in diverse malignancies. Clin Immunol. 2001;100:149-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 85. | Xu QW, Zhao W, Wang Y, Sartor MA, Han DM, Deng J, Ponnala R, Yang JY, Zhang QY, Liao GQ. An integrated genome-wide approach to discover tumor-specific antigens as potential immunologic and clinical targets in cancer. Cancer Res. 2012;72:6351-6361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 86. | Looi K, Megliorino R, Shi FD, Peng XX, Chen Y, Zhang JY. Humoral immune response to p16, a cyclin-dependent kinase inhibitor in human malignancies. Oncol Rep. 2006;16:1105-1110. [PubMed] |
| 87. | Hoshino I, Nagata M, Takiguchi N, Nabeya Y, Ikeda A, Yokoi S, Kuwajima A, Tagawa M, Matsushita K, Satoshi Y. Panel of autoantibodies against multiple tumor-associated antigens for detecting gastric cancer. Cancer Sci. 2017;108:308-315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 88. | Zhang JY, Casiano CA, Peng XX, Koziol JA, Chan EK, Tan EM. Enhancement of antibody detection in cancer using panel of recombinant tumor-associated antigens. Cancer Epidemiol Biomarkers Prev. 2003;12:136-143. [PubMed] |
| 89. | Koziol JA, Zhang JY, Casiano CA, Peng XX, Shi FD, Feng AC, Chan EK, Tan EM. Recursive partitioning as an approach to selection of immune markers for tumor diagnosis. Clin Cancer Res. 2003;9:5120-5126. [PubMed] |
| 90. | Werner S, Chen H, Butt J, Michel A, Knebel P, Holleczek B, Zörnig I, Eichmüller SB, Jäger D, Pawlita M. Evaluation of the diagnostic value of 64 simultaneously measured autoantibodies for early detection of gastric cancer. Sci Rep. 2016;6:25467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |