Published online Jun 28, 2017. doi: 10.3748/wjg.v23.i24.4330
Peer-review started: February 10, 2017
First decision: March 16, 2017
Revised: April 13, 2017
Accepted: May 19, 2017
Article in press: May 19, 2017
Published online: June 28, 2017
Processing time: 148 Days and 6.2 Hours
The prognosis for patients who are diagnosed with advanced stage hepatocellular carcinoma (HCC) is poor because there are few treatment options. Recent research has focused on the identification of novel molecular entities that can be targeted to inhibit oncogenic signals that are involved in the carcinogenesis, proliferation and progression of HCC. Among all of the pathways that are involved in the development of HCC, Hedgehog (HH) signalling has demonstrated a substantial role in hepatocarcinogenesis and HCC progression. HH plays a physiological role in embryogenesis, through the induction of the differentiation of hepatocytes from endodermal progenitors. The re-activation of the HH pathway in chronic damaged liver is a mechanism of fibrotic degeneration and is implicated in various stages of HCC development. HH activation sustains the sub-population of immature liver epithelial cells that are involved in the pathogenesis of cirrhosis and HCC, and HH itself is a mediator of the alcohol-derived malignant transformation of liver cells. High levels of expression of HH protein markers in liver tumour tissues are correlated with aggressive histological and biological features and a poor clinical outcome. In vitro and in vivo inhibition models of the HH pathway confirm that HH is essential in maintaining tumour growth, metastasis and a mesenchymal phenotype.
Core tip: The hedgehog (HH) pathway is involved in the embryonic development of liver, and its reactivation plays a substantial role in sustaining cancer cell growth and progression in hepatocellular carcinoma (HCC). In hepatocarcinogenesis, HH signalling is required for differentiation, proliferation and polarity of liver embryonic cells. High levels of expression of HH components in HCC tissues correlate with mesenchymal properties and maintain the proliferation of cancer stem cells, which is a dynamic source of malignant cells in HCC progression. Current data on HH inhibition in preclinical models further confirm the role of HH and deserve future investigations in a clinical setting.
- Citation: Della Corte CM, Viscardi G, Papaccio F, Esposito G, Martini G, Ciardiello D, Martinelli E, Ciardiello F, Morgillo F. Implication of the Hedgehog pathway in hepatocellular carcinoma. World J Gastroenterol 2017; 23(24): 4330-4340
- URL: https://www.wjgnet.com/1007-9327/full/v23/i24/4330.htm
- DOI: https://dx.doi.org/10.3748/wjg.v23.i24.4330
Liver tumours are the third highest cause of cancer-related deaths and the sixth most frequent type of cancer worldwide[1]. In the past few years, the incidence of hepatocellular carcinoma (HCC) has been on the rise, especially among males, being correlated with chronic liver disease[2]. In particular, the majority of patients who develop HCC are infected with viral hepatitis B (HBV) or viral hepatitis C (HCV)[3,4]. Even though vaccination programmes are commonly used in Western populations, the incidence of HCC remains high because of other risk factors, such as alcoholic and non-alcoholic fatty liver disease and obesity, which are increasingly out of control in Occidental countries[5].
Screening procedures with periodic liver ultrasonography (every 6 mo), concomitant with the determination of serum alpha-fetoprotein (AFP), are recommended only for cirrhotic patients and HBV- and HCV- serum positive patients[6].
Treatment options with curative intent, including surgery, local treatment (such as percutaneous thermal ablation, intra-arterial chemotherapy or embolization or radiotherapy) and liver transplantation, are reserved only for select patients with localized tumour extension and/or with good performance status and liver function[7,8].
Unfortunately, most patients are diagnosed at an advanced stage; the five-year overall survival for unresectable HCC is less than 10% because all medical treatments that were given to these patients exhibited a very poor efficacy[2]. Actually, according to international oncologic guidelines, sorafenib is the only standard systemic therapy for patients with advanced HCC and adequate liver function[2]. Sorafenib is a multi-kinase inhibitor with potent inhibitory activity against RAF kinases, including mutant V600E B-RAF, and against various serine/threonine kinases, such as the pro-angiogenic receptor tyrosine kinases vascular endothelial growth factor receptor (VEGFR), platelet derived growth factor receptor (PDGFR) and fibroblast growth factor receptor (FGFR1), as well as tyrosine kinases such as KIT, Fms-related tyrosine kinase 3 (FLT-3) and RET[9]. More recent clinical data on new treatment options included regorafenib[10] and immunotherapy[11]. Treatment with regorafenib produced a survival benefit in HCC patients on sorafenib treatment (median survival 10.6 mo for regorafenib vs 7.8 mo for the placebo group, with a hazard ratio of 0.63, one-sided P < 0.0001)[10]. At the previous ASCO congress, efficacy data from the CheckMate 040 trial were presented for Nivolumab, an anti-PD1 (Programmed cell Death 1) agent, in advanced HCC patients (70% progressed to sorafenib and 30% were sorafenib naïve). Nivolumab monotherapy provided early, stable and durable responses, irrespective of the hepatitis C or B viral infection status, and of PD-L1 (Programmed cell Death Ligand 1) expression on the tumours[11]. The overall response rate was 19% in the overall population, and 21.7% in the first-line setting, with an overall 9 month survival rate of 77%[11].
Recent research has focused on identifying potential new molecular targets that can be “drugable” in order to inhibit the oncogenic signals that drive tumour carcinogenesis, growth and metastasis[12].
In HCC carcinogenesis and evolution, biological and genetic alterations lead to the activation of oncogenes (V-Myc avian myelocytomatosis viral oncogene homologue, myc, kirsten rat sarcoma viral oncogene homologue, k-ras, raf murine sarcoma viral oncogene homologue B, BRAF), of growth factor-induced signalling (transforming growth factor-α, TGF-α, TGF-β, insulin-like growth factor-2 receptor, IGF-2, epidermal growth factor receptor, EGFR, PDGFR, VEGFR), and/or re-activation of pathways that are implicated in development and embryogenesis (Wingless-Type MMTV integration site family, WNT, and Hedgehog, HH) or disabling of tumour suppressor genes (retinoblastoma, RB, phosphatase and tensin homologues, PTEN, Runt-related transcription factor 3, RUNX3, or p53)[13,14].
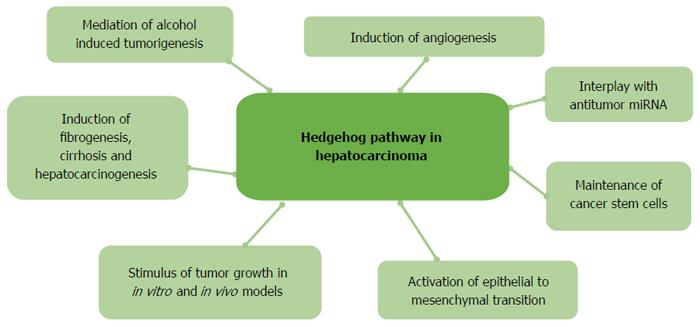
In this review we will focus on current data regarding the role of HH pathway reactivation in the development and progression of HCC. The main role of HH in embryonic hepatogenesis involves the induction of the differentiation of hepatocytes from endodermal progenitors[15,16]. In hepatocarcinoma cells, the re-induction of expression of HH proteins sustains cancer cell growth and reduces apoptotic stimuli[17] at various stages of HCC development (Figure 1).
Carcinogenesis is a multistep process that is characterized by changes at the cellular, genetic and epigenetic levels, and by abnormal cell division that leads to the formation of a malignant mass. There are common mechanisms for carcinogenesis in different human organs and others that are organ-specific[18].
The principal cause of liver carcinogenesis is an external stimulus: viral infection or alcoholic damage[5,19-22]. HBV DNA and HCV core proteins are oncogenic, as they induce the formation of pro-proliferative transcription factors and interplay with RAS, WNT or p53 signalling[19-21]. In fatty liver disease, especially in alcohol related damage, active adipocyte pathways induce chronic inflammation of the microenvironment and liver cell regeneration and proliferation[5,22].
An important mechanism of oncogenesis and tumour evolution in hepatocarcinoma is the activation of the oncogenes MYC and RAS[23-26]. MYC gene encodes for a protein that is involved in nucleic acid metabolism and in response to growth factors. MYC amplification has been detected in 12% of single nodular HCC, in 38% of multiple nodular HCC (P < 0.01), and with a higher frequency in metastatic and recurrent HCC patients than in primary tumours (45% vs 29%, and 60% vs 38%, respectively), suggesting that MYC alteration can occur in early stages of carcinogenesis rather than in later stages, leading to metastatic processes[26]. The RAS gene transcript is the RAS protein, a small GTPase protein that represents the main regulator of the mitogen activated protein kinase (MAPK) and of the phosphatidylinositol 3-kinase (PI3K)/AKT pathways that represent the principal signalling pathways for cell survival[23,24,27].
Among receptors that are involved in HCC carcinogenesis, the MET receptor and its ligand hepatocyte growth factor have been demonstrated to correlate with a poor prognosis in HCC[28,29], and the selective MET-inhibitor, tivantinib, has shown promising activity in a phase II trial in patients progressing on sorafenib and whose tumours produced high expression levels of MET[30].
In the evolution and metastasis of HCC, abnormal angiogenesis and epithelial-mesenchymal transition (EMT) are fundamental. VEGFR, FGFR and PDGFR are the main factors in the uncontrolled neo-angiogenesis of HCC[31,32]. Sorafenib was originally designed to inhibit VEGFR, PDGFR and KIT; the efficacy of sorafenib confirms the essential role of angiogenesis in the progression of HCC[10]. TGF-β is a key mediator of EMT and functions as an autocrine or paracrine growth factor for the regulation of inflammatory cells, T-cells and cancer-associated fibroblasts in the microenvironment[33,34].
A peculiar and interesting aspect of the early phase of hepatocarcinogenesis is the renewal of the developmental pathways of WNT and HH that conduct embryonic growth and the differentiation of normal liver cells in humans[35-37]. WNT/β-catenin signalling can be abnormally re-activated in HCC by β-catenin mutations, by nuclear translocations, or by a reduction of the adenomatous polyposis coli gene-related protein, which is a negative regulator of β-catenin[35,36]. A β-catenin signal induces metastatic behaviour and stem-like features in cancer cells[35,36].
Although there are data indicating that TGF-β and WNT regulate growth and repair responses in adult livers, the possibility that HH was involved in these processes was not considered until recently[17,37].
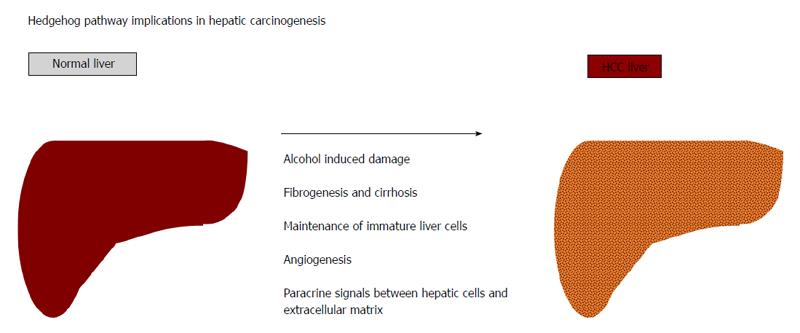
HH pathway activation is required for the differentiation, proliferation and polarity of liver embryonic cells and plays a role in hepatocarcinogenesis[16,37,38] (Figure 2). It is known that complete silencing of HH signalling in embryos cause a lethal block in the development of nervous and cardiovascular systems and the liver[38,39]. Partial silencing of HH during liver development is responsible for hepatic defects, along with cardiac, neurological and muscular malformations[38,39]. In human embryonic development, HH ligands are highly expressed in cells of the ventral endoderm that differentiate in hepatic progenitors, and the principal mediator of HH activity is foxa2, a specific transcription factor for hepatic cells[40]. Inhibition of HH in vitro reduced the proliferation of human foetal hepatoblasts[41], thus confirming that HH is essential in the differentiation process for hepatic and pancreatic cells from multipotent endodermal progenitor cells. HH activation progressively decreases during liver epithelial cell maturation[37,41]. HH signalling has no function in the adult liver; indeed, normal hepatocytes display undetectable levels of HH signal proteins with no clear HH pathway activity. The mesenchyme of healthy livers and liver sinusoidal cells, including endothelial and hepatic stellate cells, continues to produce Hhip, a soluble antagonist of HH ligands, which retain soluble HH ligands from the activation of the HH pathway in epithelial cells[42]. Interestingly, after partial hepatectomy, the hepatic expression of HH ligands re-increases significantly, concomitant with a downregulation of Hhip, and this increment represents a strong stimulus for liver regeneration[43,44]. Transient activation of the HH pathway is useful for liver regeneration after an acute injury. An alteration of the equilibrium between liver epithelial cells and neighbouring stromal cells is due to persistent re-induction of paracrine HH signalling that sustains the sub-population of immature liver epithelial cells that are involved in the pathogenesis of liver fibrosis, cirrhosis and HCC[44].
The HH pathway is activated by ligands. In humans, three ligands have been identified: Sonic hedgehog (SHH), Indian hedgehog (IHH) and Desert hedgehog, of which SHH is the most studied[45]. In target cells, SHH binds to its receptor Patched-1 (PTCH1). In the absence of SHH, PTCH1 inhibits Smoothened (SMO), a downstream protein in the HH pathway. Thus, the interaction between SHH and PTCH1 reduces SMO and the subsequent activation of the GLI transcription factors Gli1 and Gli2, which are known activators, as well as Gli3, a repressor[45]. The effect of this molecular cascade has only partially been studied, and the role of HH is currently the object of research in various types of human cancers (breast, pancreas, lung, colorectal, gastric and renal cancers and hepatocarcinoma)[16,17,46-51].
Different research groups have demonstrated that mature hepatocytes lack HH activity[37]. The difference in terms of HH protein marker expression between tumour and normal liver tissues has been investigated to assess the clinical impact of HH activation in HCC patients[52-54] (Table 1).
| Ref. | SMO | SHH | PTCH | Gli1 | Clinical correlation |
| Sicklick et al[52], 2006 | 42.90% (3-fold higher than normal tissues) | 50% (3- to 4-fold higher than normal tissues) | Tumour size | ||
| 21.4% (8- to 9-fold higher than normal tissues) | |||||
| Huang et al[53], 2006 | 59% positive tumours | 56% positive tumours | 72% positive tumours | Tumour size | |
| Che et al[59], 2012 | 32.61% positive tumours | 60.87% positive tumours | 50% positive tumours | 54.35% positive tumours | Gli1 with disease-free survival (DFS) and overall survival (OS) |
| Dugum et al[54], 2016 | 100% tumoural and surrounding tissue | 100% tumoural and surrounding tissue, with higher expression in tumour tissue | 100% tumoural and surrounding tissue | Tendency to correlation with HCC recurrence after liver transplantation |
The first prospective study was performed in 2006 by Sicklick et al[52] These authors examined levels of SHH, Gli1 and SMO in tumours and in adjacent normal tissues and confirmed the activation of the HH pathway in cancer cells with overexpression of SMO compared to respective non-tumour tissues. Moreover, elevated SMO levels were significantly associated with the size of the HCC nodules. The ratio of SMO to PTCH1 expression was higher in larger (> 5 cm diameter) than in smaller (< 5 cm diameter) HCC lesions (r 1⁄4 0.57, P < 0.04)[52].
Similar results have been presented by Huang et al[53] who published an interesting analysis of HH markers in HCC tumour tissues and adjacent normal liver tissues. In a case study of 115 patients, the expression of SHH and its target genes were quantified by in situ hybridization and polymerase chain reaction (PCR). Expression levels of SHH, Gli1 and PTCH1 in tumour cells were significantly higher than in adjacent normal cells in most cases. HCC cancer cells were positive for SHH in 59% of liver tumours, for Gli1 in 72%, and for PTCH1 in 56%; the majority of HH positive cases were tumours larger than 3 cm, thus underlying the role of this pathway in maintaining tumour growth and progression[53]. One single case exhibited positive Gli1 and PTCH1 expression both in tumour and in surrounding tissues; deeper analysis also detected positive HH proteins levels in altered pre-cancerous tissues (small cell dysplasia or dysplastic nodules) and in microscopic HCC, thus reinforcing the substantial role of HH in early stages of carcinogenesis. Another important finding that emerged from this study was the correlation between HH activation and β-catenin positivity, suggesting an interplay between the two developmental pathways (WNT and HH) that are involved in hepatocarcinogenesis[55].
Furthermore, HH activation appears to be linked to the ingestion of alcohol. A recent study by Chan et al[56] analysed a model of alcohol-derived hepatocarcinogenesis and described a correlation between increased alcohol ingestion and HH signalling activation, with subsequent malignant changes in liver cells. Alcohol exposure induces the over-activation of HH signalling through the elevation of HH ligands and HH target genes in animal models and human tissues. The proliferation of progenitor cells was accompanied by the expression of EMT markers and by an interaction between stroma and hepatic cells. The HH pathway can induce glycolytic activity in myofibroblasts of liver stroma, the so-called “reverse Warburg” effect[57], and the products of glycolytic stromal cells stimulate the proliferation of malignant liver cells[58].
Further evidence for a role of the HH pathway in the carcinogenesis of HCC came from the analysis of HH activation in cirrhotic patients[52]. Among these patients, levels of PTCH1 were variable, due to the presence of myofibroblastic hepatic stellate cells in cirrhotic non neoplastic tissue that are able to produce PTCH1[52]. The concomitant detection of SMO mutations and HH activation, demonstrated by high Gli1 levels, suggest that early genetic alteration in HH proteins can promote carcinogenesis and growth of HCC from HH signals[52].
To define the relationship between the expression of HH pathway components and clinical prognosis, a sample-expanded and prolonged follow-up study was performed on 46 surgically treated HCC patients[59]. All HH components were present in more than 50% of tumour samples, and in particular, Gli1 expression was significantly correlated with disease-free survival (DFS) and overall survival (OS) (P < 0.029 and P < 0.025, respectively). The authors concluded that Gli1 expression could become a strong prognostic biomarker for HCC progression. Since Gli1 is a key effector in the HH pathway, and HH components were associated with clinical malignant features (tumour size, capsular invasion and vascular invasion), we speculate that HH signal activation has a clear impact on the clinical progression and development of HCC in patients[59].
In 2016, Dugum et al[54] assessed HH protein marker expression in HCC and surrounding normal tissue in patients with HCC recurrence after liver transplantation (LT). These data have high clinical relevance, because a complete panel of prognostic biomarkers is required to identify patients who can be selected for LT. SHH, PTCH1 and Gli1 levels were positive in all tumour samples and in cirrhotic tissue, confirming previous data. In particular, PTCH1 expression was higher in HCC than in non-tumour tissue. Although no statistically significant correlation was observed between HH markers and recurrence after LT, the interval time between LT and recurrence indicated a trend towards this correlation. However, the small number of patients limited the value of this study[54].
In addition to the analysis of tumour tissues from HCC patients, in order to clarify the mechanisms of tumourigenesis and progression that are induced by the HH signalling pathway, Che et al[59] studied the effect of the SMO antagonist cyclopamine on proliferation and apoptosis in HCC cell lines. Cyclopamine is a natural plant corn lily extract[60] with the ability to downregulate HH signals through direct negative interaction with the SMO receptor. In vitro cyclopamine treatment affected cell viability, with a concomitant reduction of mRNA and protein levels of Gli1, demonstrating that HCC cell proliferation was dependent on HH signalling[59]. Treatment with cyclopamine also induced cell apoptosis through Gli1-dependent downregulation of Bcl-2 and cleavage of caspase 9 and caspase 3.
Similarly, Huang et al[53] tested a panel of HCC cells for the expression of HH components, and selected HH positive HCC cells were treated with cyclopamine or anti-SHH-specific antibodies. A cyclopamine-induced reduction of Gli1 mRNA with no substantial change in SHH levels was demonstrated. Each type of HH inhibitor affected the cell proliferation and vitality of HCC cells, supporting the ideas that Gli1 is the best marker of HH activation and that inhibition of Hh can be sufficient to reduce tumour growth in this model[53]. These results encourage the evaluation of the inhibition of the HH pathway as a promising new therapeutic strategy for treating HCC.
Moreover, the combination of HH inhibitor with radiotherapy enhances the radiosensitivity of HCC cells and orthotopic tumours[61], and the inhibition of HH induces autophagy and apoptosis in HCC cells[62], thus suggesting other various potential mechanisms of HH-mediated effects.
Based on these translational evidence, various in vivo preclinical models have been created to study the contribution of HH signalling to the pathogenesis of HCC.
Philips et al[63] focused on the role of the HH pathway in hepatocarcinogenesis and in the development of the pre-cancerous lesion represented by liver fibrosis. To demonstrate this hypothesis, they treated a model of murine HCC represented by Mdr2-deficient mice with the SMO inhibitor, GDC-0449. Mdr2 is the murine homologue protein of human MDR3, and its deletion eliminates P-glycoprotein, which transports phosphatidylcholine (PC) across the canalicular membrane. The absence of functional Mdr2 induces portal inflammation and fibrosis, with ductular proliferation and development of HCC within 60 wk[64]. Mdr2-deficient mice were the optimal model to study the evolution of HCC in vivo in fibrotic livers. Mice showed progressively increasing levels of SHH, IHH, and Gli2, concomitant, in early phases, with an elevation of liver enzymes. Treatment in vivo with GDC-0449 decreased the levels of hepatic TGF-β and PDGF-β, of alpha-smooth muscle actin in myofibroblastic hepatic stellate cells, concomitant with a reduction of Gli1 and Gli2, demonstrating that inhibition of HH improved liver fibrosis. Moreover, the expression of markers of liver progenitor cell populations decreased after HH inhibition, suggesting that HH signalling is essential for the regeneration of liver cell carcinogenesis, and for maintenance of progenitor cell populations in advanced stages of disease as a source of malignant cancer cells[63]. The correlation between HH and PDGF-β in liver fibrosis implied that the fibrogenic repair response favoured HH activation itself. Since stem-like progenitor cells express CD44, and the receptor for osteopontin and osteopontin itself is controlled by the HH pathway[65], the authors also investigated the effect of SMO inhibition on osteopontin levels. Of interest, osteopontin expression was significantly reduced after treatment. It is likely that osteopontin is a key point of regulation for cancer stem cell survival by the HH pathway[63]. Progenitor cells produce fibrogenic factors such as TGF-β and PDGF-β, which activate Gli2 in a non-canonical (SMO-independent) manner. HH is implicated as a principal component of all of these negative stimuli during chronic liver injury that lead to fibrosis and/or cancer[63].
A prospective in vivo animal study confirmed the potency of HH inhibition for reverting HCC growth[66]. Mice xenografted with mouse hepatoma Mistheton Lectin-1 cells were randomly assigned to receive a placebo or cyclopamine at a dose of 10 or 30 mg/kg per day for 10 d and were monitored for tumour growth. Tumour size, SHH and Gli1 mRNA levels were decreased by cyclopamine treatment, especially with the higher dose, in a statistically significant manner (P = 0.047 for tumour size)[66].
Moreover, Pinter et al[67] demonstrated that inhibition of HH with the SMO inhibitor GDC-0449 reduces angiogenesis in an orthotopic HCC model by decreasing tumour expression of VEGF, with a subsequent reduction of micro vessel formation and tumour size.
EMT is a complex dynamic process that determines changes in cellular features from an epithelial to a mesenchymal phenotype[68]. Recent data support the importance of cancer stem cells in inducing EMT because they represent a subpopulation that exhibits a metastatic profile[68]. Among the heterogeneity of cells that compose a tumour mass, cancer stem cells have been recognized as normal stem cells that are able to generate various types of cells that resemble different clones of tumour evolution and exhibit high invasive potential and drugs resistance[68]. EMT activation is triggered by cell signalling that represses E-cadherin expression in favour of proteins of the mesenchymal phenotype. In HCC cell lines, SLUG was demonstrated as a principal inducer of EMT and stem-like cancer cell growth, together with high levels of vimentin and over-expression of N-cadherin[69,70]. Consistent with the phenotype, cancer stem cells display a sphere-forming ability in vitro, confirming their invasive behaviour. Of interest, CD133+ cells isolated from a mouse Hepa16-derived tumour revealed over-expression of HH pathway components[71]. However, cancer stem cells constantly change dynamically in HCC. In particular, cancer stem cells that are characterized by an EMT phenotype, such as expression of vimentin, SNAIL, TGF-β and HH components, are located in invasive margins of HCC nodules. The location of these components serve as an indicator of their role in the regulation of HCC progression. Their potential role as therapeutic targets in HCC remains unknown[72,73].
Interestingly, a promising novel target that has been investigated in preclinical studies in HCC cell models is the Bromodomain-containing protein 4 (BRD4), which plays an important role in HCC cancerogenesis by the induction of various oncogenes, including HH signalling-related genes[74-76]. Wang et al[75] demonstrated that BRD4 is able to promote HCC cell migration and invasion in vitro and that BRD4 activity is mediated by HH signalling through the metalloproteases MMP - 2 and MMP - 9. The activation of metalloprotease by the HH pathway through focal adhesion kinase/AKT signalling has been confirmed also by other studies on liver cancer[76]. The inhibition of BRD4 by silencing-RNA reduces MMP-2 and MMP-9 levels, blocking cell progression and invasive abilities. All of these metastatic features can be totally restored by stimulating HCC cells with the recombinant ligand SHH, thus confirming that HH is an essential pathway in HCC metastasis[75].
Metalloprotease activation is not the only known downstream effector of HH pathway-induced progression in HCC. Another potential “targetable” protein is the kinesin family member 20A that is downstream of Gli2. Gli2 controls the cell cycle in normal and cancerous cells[77]. Similarly, kinesin family proteins are involved in the regulation of the cell cycle and mitosis[78]. Shi et al[77] correlated for the first time Gli2 and KIF20A in HCC cells and tissue samples from patients. They found that aberrant activation of the GLI2-KIF20A axis is crucial for the growth of hepatocarcinoma and that their interplay is mediated by activating Forkhead Box M1 (FoxM1)-MMB complex-mediated transcription. Gli2 or KIF20A knockdown affects the growth of HCC cells in vitro and in vivo. Analysis of HCC tissues reveals that GLI2, FoxM1 and KIF20A are highly increased in tumour cells and are correlated with HCC recurrence and overall survival of patients. Co-expression of these three markers could be considered a negative predictor for poor prognosis in HCC patients[77].
MicroRNAs (miRNAs) are endogenous non-coding RNAs and are characterized by a length of B22 nucleotides. miRNAs supress the expression of target genes at a post-transcriptional or translational level. In particular, a miRNA seed sequence (6-8 end nucleotides) interacts with a complementary sequence in messenger RNA of target genes. In humans their role is known in normal biological processes including proliferation and differentiation, and in tumours, including HCC[79,80].
MicroRNAs can regulate EMT in HCC; in particular, miR-338-3p expression is significantly low in HCC tissues with poor differentiation in advanced stage HCC patients[81].
MiR- 338-3p reduces HCC cell invasiveness in vitro by specifically inhibiting SMO, the receptor of HH signalling, and subsequently reducing MMP-9 activity[82,83]. In HCC cell lines, miR-338-3p expression levels correlate with the transcriptional activity of E-cadherin and with inhibited N-cadherin[83]. Transfection of the miR-338-3p mimic in HCC cells induces a regression of Gli1 protein and mRNA levels concomitant with a decrease of SNAIL activity. In contrast, SMO siRNA inhibits the effect of miR-338-3p, confirming that SMO is the mediator of miR-338-3p in HCC cells and that inhibition of miR-338-3p is a mechanism of activation of EMT and HH signalling in HCC development. miR-338-3p and N-cadherin expression levels were assessed in a cohort of 163 HCC tissue specimens by real time PCR, in which HCC tumours were classified as low and high expressers and correlated a low miR-338-3p with a poor clinical DFS. Similarly, high expression of N-cadherin and low expression of miR-338-3p correlated with HCC metastasis[79,83]. Moreover, real time PCR on HCC tissues for HH components revealed that high levels of miR-338-3p occur together with inhibition of HH signals, and with low expression of SMO, Gli1 and vimentin, thus confirming the complex link between miRNAs, HH and EMT.
In addition, cooperation of HH and miR-378 family members (miR-378a-3p, miR-378b and miR-378d) was demonstrated also in hepatic fibrogenesis. In most chronic liver diseases, fibrogenesis is a base for malignant transformation. The mechanism by which fibrogenesis is controlled by miR-378 is by the inhibition of GLI3 in activated hepatic stellate cells and chronically damaged liver[80]. Moreover, SMO is an inhibitor of the transcription of miR-378a-3p by nuclear factor-kB. In human tissues, miR-378a-3p levels are inversely correlated with Gli3. In experimental animal models, the introduction of a mimic miR-378a-3p can revert liver fibrotic damage and HH signalling[80].
In light of these results, miR-378a-3p is considered to be a useful biomarker for hepatic pathogenesis and a potentially new therapeutic agent for treatment of liver tumours.
Among all of the pathways that are involved in the development of HCC, HH signalling has a substantial role in hepatocarcinogenesis and HCC progression. The role of HH can have potential therapeutic implications, because chemical inhibitors of the HH pathway are now approved for use in other human tumours, such as in basocellular carcinoma[84]. All efforts that target the molecular components of the pathways that are activated in the development of HCC have not demonstrated a clinical benefit, and sorafenib remains the only available therapeutic option for advanced HCC patients. Numerous preclinical data are available that suggest a role of HH in hepatocarcinogenesis and EMT in HCC[38,75-77]. In HCC, reactivation of the HH pathway is responsible for the maintenance of cancer stem cells that harbour a mesenchymal phenotype, and HH inhibition in preclinical models has a clear anti-tumour effect[76,77]. Moreover, the correlation between the expression of HH proteins in HCC tumour tissues and a poor clinical outcome demonstrates that HH activation is implicated in HCC invasiveness and progression[52-54].
Thus, the HH pathway plays a crucial role in the process of carcinogenesis and progression of hepatocarcinoma; however, the prognostic value of the HH pathway protein marker expression in tissues from HCC patients requires further investigation. In the future, targeting the HH pathway in HCC could be an interesting subject for preclinical and clinical studies.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Italy
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Meng ZQ, Şendur MAN, Zhao HT S- Editor: Ma YJ L- Editor: A E- Editor: Wang CH
| 1. | White DL, Thrift AP, Kanwal F, Davila J, El-Serag HB. Incidence of Hepatocellular Carcinoma in All 50 United States, From 2000 Through 2012. Gastroenterology. 2017;152:812-820.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 331] [Article Influence: 41.4] [Reference Citation Analysis (1)] |
| 2. | Verslype C, Rosmorduc O, Rougier P; ESMO Guidelines Working Group. Hepatocellular carcinoma: ESMO-ESDO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23 Suppl 7:vii41-vii48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 282] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 3. | Chen JD, Yang HI, Iloeje UH, You SL, Lu SN, Wang LY, Su J, Sun CA, Liaw YF, Chen CJ; Risk Evaluation of Viral Load Elevation and Associated Liver Disease/Cancer in HBV (REVEAL-HBV) Study Group. Carriers of inactive hepatitis B virus are still at risk for hepatocellular carcinoma and liver-related death. Gastroenterology. 2010;138:1747-1754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 254] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 4. | Mahale P, Torres HA, Kramer JR, Hwang LY, Li R, Brown EL, Engels EA. Hepatitis C virus infection and the risk of cancer among elderly US adults: A registry-based case-control study. Cancer. 2017;123:1202-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 5. | Marrero JA, Fontana RJ, Fu S, Conjeevaram HS, Su GL, Lok AS. Alcohol, tobacco and obesity are synergistic risk factors for hepatocellular carcinoma. J Hepatol. 2005;42:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 350] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 6. | Dulku G, Dhillon R, Goodwin M, Cheng W, Kontorinis N, Mendelson R. The role of imaging in the surveillance and diagnosis of hepatocellular cancer. J Med Imaging Radiat Oncol. 2017;61:171-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 7. | Poon RT, Fan ST, Tsang FH, Wong J. Locoregional therapies for hepatocellular carcinoma: a critical review from the surgeon’s perspective. Ann Surg. 2002;235:466-486. [PubMed] |
| 8. | Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37:429-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2207] [Cited by in RCA: 2270] [Article Influence: 103.2] [Reference Citation Analysis (0)] |
| 9. | Josephs DH, Ross PJ. Sorafenib in hepatocellular carcinoma. Br J Hosp Med (Lond). 2010;71:451-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 10. | Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389:56-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2160] [Cited by in RCA: 2707] [Article Influence: 338.4] [Reference Citation Analysis (0)] |
| 11. | Melero I, Sangro B, Yau TC. Nivolumab dose escalation and expansion in patients with advanced hepatocellular carcinoma: The CheckMate 040 study. Presented January 20, 2017. : Gastrointestinal Cancers Symposium ASCO 2017; Abstract 226. |
| 12. | Fornaro L, Vivaldi C, Lorenzoni G, Masi G, Bargellini I. Moving beyond sorafenib alone in advanced hepatocellular carcinoma: is hepatic arterial infusion chemotherapy the best option? Ann Oncol. 2017;28:667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 13. | Kgatle MM, Setshedi M, Hairwadzi HN. Hepatoepigenetic Alterations in Viral and Nonviral-Induced Hepatocellular Carcinoma. Biomed Res Int. 2016;2016:3956485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Yoo JE, Kim YJ, Rhee H, Kim H, Ahn EY, Choi JS, Roncalli M, Park YN. Progressive Enrichment of Stemness Features and Tumor Stromal Alterations in Multistep Hepatocarcinogenesis. PLoS One. 2017;12:e0170465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Ramalho-Santos M, Melton DA, McMahon AP. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development. 2000;127:2763-2772. [PubMed] |
| 16. | Parkin CA, Ingham PW. The adventures of Sonic Hedgehog in development and repair. I. Hedgehog signaling in gastrointestinal development and disease. Am J Physiol Gastrointest Liver Physiol. 2008;294:G363-G367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Zheng X, Zeng W, Gai X, Xu Q, Li C, Liang Z, Tuo H, Liu Q. Role of the Hedgehog pathway in hepatocellular carcinoma (review). Oncol Rep. 2013;30:2020-2026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 18. | Vassilev A, DePamphilis ML. Links between DNA Replication, Stem Cells and Cancer. Genes (Basel). 2017;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 19. | Minami M, Daimon Y, Mori K, Takashima H, Nakajima T, Itoh Y, Okanoue T. Hepatitis B virus-related insertional mutagenesis in chronic hepatitis B patients as an early drastic genetic change leading to hepatocarcinogenesis. Oncogene. 2005;24:4340-4348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 62] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 20. | Yamanaka T, Kodama T, Doi T. Subcellular localization of HCV core protein regulates its ability for p53 activation and p21 suppression. Biochem Biophys Res Commun. 2002;294:528-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 21. | Fukutomi T, Zhou Y, Kawai S, Eguchi H, Wands JR, Li J. Hepatitis C virus core protein stimulates hepatocyte growth: correlation with upregulation of wnt-1 expression. Hepatology. 2005;41:1096-1105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 115] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 22. | McKillop IH, Schrum LW. Role of alcohol in liver carcinogenesis. Semin Liver Dis. 2009;29:222-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 83] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 23. | Zakaria MK, Khan I, Mani P, Chattopadhyay P, Sarkar DP, Sinha S. Combination of hepatocyte specific delivery and transformation dependent expression of shRNA inducing transcriptional gene silencing of c-Myc promoter in hepatocellular carcinoma cells. BMC Cancer. 2014;14:582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Kolch W. Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem J. 2000;351 Pt 2:289-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 25. | Huynh H, Nguyen TT, Chow KH, Tan PH, Soo KC, Tran E. Over-expression of the mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK in hepatocellular carcinoma: its role in tumor progression and apoptosis. BMC Gastroenterol. 2003;3:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 207] [Cited by in RCA: 219] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 26. | Wang Y, Wu MC, Sham JS, Zhang W, Wu WQ, Guan XY. Prognostic significance of c-myc and AIB1 amplification in hepatocellular carcinoma. A broad survey using high-throughput tissue microarray. Cancer. 2002;95:2346-2352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 167] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 27. | Chung TW, Lee YC, Kim CH. Hepatitis B viral HBx induces matrix metalloproteinase-9 gene expression through activation of ERK and PI-3K/AKT pathways: involvement of invasive potential. FASEB J. 2004;18:1123-1125. [PubMed] |
| 28. | Boix L, Rosa JL, Ventura F, Castells A, Bruix J, Rodés J, Bartrons R. c-met mRNA overexpression in human hepatocellular carcinoma. Hepatology. 1994;19:88-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 74] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 29. | Ueki T, Fujimoto J, Suzuki T, Yamamoto H, Okamoto E. Expression of hepatocyte growth factor and its receptor, the c-met proto-oncogene, in hepatocellular carcinoma. Hepatology. 1997;25:619-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 103] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 30. | Santoro A, Rimassa L, Borbath I, Daniele B, Salvagni S, Van Laethem JL, Van Vlierberghe H, Trojan J, Kolligs FT, Weiss A. Tivantinib for second-line treatment of advanced hepatocellular carcinoma: a randomised, placebo-controlled phase 2 study. Lancet Oncol. 2013;14:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 463] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 31. | Semela D, Dufour JF. Angiogenesis and hepatocellular carcinoma. J Hepatol. 2004;41:864-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 280] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 32. | El-Assal ON, Yamanoi A, Soda Y, Yamaguchi M, Igarashi M, Yamamoto A, Nabika T, Nagasue N. Clinical significance of microvessel density and vascular endothelial growth factor expression in hepatocellular carcinoma and surrounding liver: possible involvement of vascular endothelial growth factor in the angiogenesis of cirrhotic liver. Hepatology. 1998;27:1554-1562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 242] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 33. | Lu WQ, Qiu JL, Huang ZL, Liu HY. Enhanced circulating transforming growth factor beta 1 is causally associated with an increased risk of hepatocellular carcinoma: a mendelian randomization meta-analysis. Oncotarget. 2016;7:84695-84704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 34. | Mitra A, Yan J, Xia X, Zhou S, Chen J, Mishra L, Li S. IL6-mediated inflammatory loop reprograms normal to epithelial-mesenchymal transition(+) metastatic cancer stem cells in preneoplastic liver of transforming growth factor beta-deficient β2-spectrin(+/-) mice. Hepatology. 2017;65:1222-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 35. | Zhang Y, Wei W, Cheng N, Wang K, Li B, Jiang X, Sun S. Hepatitis C virus-induced up-regulation of microRNA-155 promotes hepatocarcinogenesis by activating Wnt signaling. Hepatology. 2012;56:1631-1640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 256] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 36. | Srisuttee R, Koh SS, Kim SJ, Malilas W, Boonying W, Cho IR, Jhun BH, Ito M, Horio Y, Seto E. Hepatitis B virus X (HBX) protein upregulates β-catenin in a human hepatic cell line by sequestering SIRT1 deacetylase. Oncol Rep. 2012;28:276-282. [PubMed] |
| 37. | McMahon AP, Ingham PW, Tabin CJ. Developmental roles and clinical significance of hedgehog signaling. Curr Top Dev Biol. 2003;53:1-114. [PubMed] |
| 38. | Omenetti A, Diehl AM. The adventures of sonic hedgehog in development and repair. II. Sonic hedgehog and liver development, inflammation, and cancer. Am J Physiol Gastrointest Liver Physiol. 2008;294:G595-G598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 39. | Ingham PW, McMahon AP. Hedgehog signaling in animal development: paradigms and principles. Genes Dev. 2001;15:3059-3087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2300] [Cited by in RCA: 2336] [Article Influence: 97.3] [Reference Citation Analysis (0)] |
| 40. | Lee CS, Friedman JR, Fulmer JT, Kaestner KH. The initiation of liver development is dependent on Foxa transcription factors. Nature. 2005;435:944-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 452] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 41. | Sicklick JK, Li YX, Melhem A, Schmelzer E, Zdanowicz M, Huang J, Caballero M, Fair JH, Ludlow JW, McClelland RE. Hedgehog signaling maintains resident hepatic progenitors throughout life. Am J Physiol Gastrointest Liver Physiol. 2006;290:G859-G870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 158] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 42. | Choi SS, Omenetti A, Witek RP, Moylan CA, Syn WK, Jung Y, Yang L, Sudan DL, Sicklick JK, Michelotti GA. Hedgehog pathway activation and epithelial-to-mesenchymal transitions during myofibroblastic transformation of rat hepatic cells in culture and cirrhosis. Am J Physiol Gastrointest Liver Physiol. 2009;297:G1093-G1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 197] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 43. | Ochoa B, Syn WK, Delgado I, Karaca GF, Jung Y, Wang J, Zubiaga AM, Fresnedo O, Omenetti A, Zdanowicz M. Hedgehog signaling is critical for normal liver regeneration after partial hepatectomy in mice. Hepatology. 2010;51:1712-1723. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 144] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 44. | Schmelzer E, Wauthier E, Reid LM. The phenotypes of pluripotent human hepatic progenitors. Stem Cells. 2006;24:1852-1858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 257] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 45. | Nybakken K, Perrimon N. Hedgehog signal transduction: recent findings. Curr Opin Genet Dev. 2002;12:503-511. [PubMed] |
| 46. | Habib JG, O’Shaughnessy JA. The hedgehog pathway in triple-negative breast cancer. Cancer Med. 2016;5:2989-3006. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 100] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 47. | Della Corte CM, Bellevicine C, Vicidomini G, Vitagliano D, Malapelle U, Accardo M, Fabozzi A, Fiorelli A, Fasano M, Papaccio F. SMO Gene Amplification and Activation of the Hedgehog Pathway as Novel Mechanisms of Resistance to Anti-Epidermal Growth Factor Receptor Drugs in Human Lung Cancer. Clin Cancer Res. 2015;21:4686-4697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 95] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 48. | Klieser E, Swierczynski S, Mayr C, Jäger T, Schmidt J, Neureiter D, Kiesslich T, Illig R. Differential role of Hedgehog signaling in human pancreatic (patho-) physiology: An up to date review. World J Gastrointest Pathophysiol. 2016;7:199-210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 13] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 49. | Papadopoulos V, Tsapakidis K, Riobo Del Galdo NA, Papandreou CN, Del Galdo F, Anthoney A, Sakellaridis N, Dimas K, Kamposioras K. The Prognostic Significance of the Hedgehog Signaling Pathway in Colorectal Cancer. Clin Colorectal Cancer. 2016;15:116-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 50. | Lu L, Wu M, Zhao F, Fu W, Li W, Li X, Liu T. Prognostic and clinicopathological value of Gli-1 expression in gastric cancer: A meta-analysis. Oncotarget. 2016;7:69087-69096. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 51. | Shigemura K, Fujisawa M. Hedgehog signaling and urological cancers. Curr Drug Targets. 2015;16:258-271. [PubMed] |
| 52. | Sicklick JK, Li YX, Jayaraman A, Kannangai R, Qi Y, Vivekanandan P, Ludlow JW, Owzar K, Chen W, Torbenson MS. Dysregulation of the Hedgehog pathway in human hepatocarcinogenesis. Carcinogenesis. 2006;27:748-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 215] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 53. | Huang S, He J, Zhang X, Bian Y, Yang L, Xie G, Zhang K, Tang W, Stelter AA, Wang Q. Activation of the hedgehog pathway in human hepatocellular carcinomas. Carcinogenesis. 2006;27:1334-1340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 184] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 54. | Dugum M, Hanouneh I, McIntyre T, Pai R, Aucejo F, Eghtesad B, Zein N. Sonic hedgehog signaling in hepatocellular carcinoma: A pilot study. Mol Clin Oncol. 2016;4:369-374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 55. | Giakoustidis A, Giakoustidis D, Mudan S, Sklavos A, Williams R. Molecular signalling in hepatocellular carcinoma: Role of and crosstalk among WNT/ß-catenin, Sonic Hedgehog, Notch and Dickkopf-1. Can J Gastroenterol Hepatol. 2015;29:209-217. [PubMed] |
| 56. | Chan IS, Guy CD, Machado MV, Wank A, Kadiyala V, Michelotti G, Choi S, Swiderska-Syn M, Karaca G, Pereira TA. Alcohol activates the hedgehog pathway and induces related procarcinogenic processes in the alcohol-preferring rat model of hepatocarcinogenesis. Alcohol Clin Exp Res. 2014;38:787-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 57. | Pavlides S, Whitaker-Menezes D, Castello-Cros R, Flomenberg N, Witkiewicz AK, Frank PG, Casimiro MC, Wang C, Fortina P, Addya S. The reverse Warburg effect: aerobic glycolysis in cancer associated fibroblasts and the tumor stroma. Cell Cycle. 2009;8:3984-4001. [PubMed] |
| 58. | Chan IS, Guy CD, Chen Y, Lu J, Swiderska-Syn M, Michelotti GA, Karaca G, Xie G, Krüger L, Syn WK. Paracrine Hedgehog signaling drives metabolic changes in hepatocellular carcinoma. Cancer Res. 2012;72:6344-6350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 59. | Che L, Yuan YH, Jia J, Ren J. Activation of sonic hedgehog signaling pathway is an independent potential prognosis predictor in human hepatocellular carcinoma patients. Chin J Cancer Res. 2012;24:323-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 60. | Chen JK, Taipale J, Cooper MK, Beachy PA. Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev. 2002;16:2743-2748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1185] [Cited by in RCA: 1170] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 61. | Tsai CL, Hsu FM, Tzen KY, Liu WL, Cheng AL, Cheng JC. Sonic Hedgehog inhibition as a strategy to augment radiosensitivity of hepatocellular carcinoma. J Gastroenterol Hepatol. 2015;30:1317-1324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 62. | Wang Y, Han C, Lu L, Magliato S, Wu T. Hedgehog signaling pathway regulates autophagy in human hepatocellular carcinoma cells. Hepatology. 2013;58:995-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 132] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 63. | Philips GM, Chan IS, Swiderska M, Schroder VT, Guy C, Karaca GF, Moylan C, Venkatraman T, Feuerlein S, Syn WK. Hedgehog signaling antagonist promotes regression of both liver fibrosis and hepatocellular carcinoma in a murine model of primary liver cancer. PLoS One. 2011;6:e23943. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 141] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 64. | Van Nieuwkerk CM, Elferink RP, Groen AK, Ottenhoff R, Tytgat GN, Dingemans KP, Van Den Bergh Weerman MA, Offerhaus GJ. Effects of Ursodeoxycholate and cholate feeding on liver disease in FVB mice with a disrupted mdr2 P-glycoprotein gene. Gastroenterology. 1996;111:165-171. [PubMed] |
| 65. | Syn WK, Choi SS, Liaskou E, Karaca GF, Agboola KM, Oo YH, Mi Z, Pereira TA, Zdanowicz M, Malladi P. Osteopontin is induced by hedgehog pathway activation and promotes fibrosis progression in nonalcoholic steatohepatitis. Hepatology. 2011;53:106-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 229] [Cited by in RCA: 216] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 66. | Jeng KS, Sheen IS, Jeng WJ, Yu MC, Tsai HH, Chang FY, Su JC. Blockade of the sonic hedgehog pathway effectively inhibits the growth of hepatoma in mice: An in vivo study. Oncol Lett. 2012;4:1158-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 67. | Pinter M, Sieghart W, Schmid M, Dauser B, Prager G, Dienes HP, Trauner M, Peck-Radosavljevic M. Hedgehog inhibition reduces angiogenesis by downregulation of tumoral VEGF-A expression in hepatocellular carcinoma. United European Gastroenterol J. 2013;1:265-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 68. | Jordan CT, Guzman ML, Noble M. Cancer stem cells. N Engl J Med. 2006;355:1253-1261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1172] [Cited by in RCA: 1189] [Article Influence: 62.6] [Reference Citation Analysis (0)] |
| 69. | Sun Y, Song GD, Sun N, Chen JQ, Yang SS. Slug overexpression induces stemness and promotes hepatocellular carcinoma cell invasion and metastasis. Oncol Lett. 2014;7:1936-1940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 70. | Mitra A, Satelli A, Xia X, Cutrera J, Mishra L, Li S. Cell-surface Vimentin: A mislocalized protein for isolating csVimentin(+) CD133(-) novel stem-like hepatocellular carcinoma cells expressing EMT markers. Int J Cancer. 2015;137:491-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 71. | Jeng KS, Sheen IS, Jeng WJ, Yu MC, Hsiau HI, Chang FY, Tsai HH. Activation of the sonic hedgehog signaling pathway occurs in the CD133 positive cells of mouse liver cancer Hepa 1-6 cells. Onco Targets Ther. 2013;6:1047-1055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 72. | Fan QM, Jing YY, Yu GF, Kou XR, Ye F, Gao L, Li R, Zhao QD, Yang Y, Lu ZH. Tumor-associated macrophages promote cancer stem cell-like properties via transforming growth factor-beta1-induced epithelial-mesenchymal transition in hepatocellular carcinoma. Cancer Lett. 2014;352:160-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 350] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 73. | Plaks V, Kong N, Werb Z. The cancer stem cell niche: how essential is the niche in regulating stemness of tumor cells? Cell Stem Cell. 2015;16:225-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 883] [Cited by in RCA: 1178] [Article Influence: 130.9] [Reference Citation Analysis (0)] |
| 74. | Tang Y, Gholamin S, Schubert S, Willardson MI, Lee A, Bandopadhayay P, Bergthold G, Masoud S, Nguyen B, Vue N. Epigenetic targeting of Hedgehog pathway transcriptional output through BET bromodomain inhibition. Nat Med. 2014;20:732-740. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 250] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 75. | Wang YH, Sui XM, Sui YN, Zhu QW, Yan K, Wang LS, Wang F, Zhou JH. BRD4 induces cell migration and invasion in HCC cells through MMP-2 and MMP-9 activation mediated by the Sonic hedgehog signaling pathway. Oncol Lett. 2015;10:2227-2232. [PubMed] |
| 76. | Chen JS, Huang XH, Wang Q, Huang JQ, Zhang LJ, Chen XL, Lei J, Cheng ZX. Sonic hedgehog signaling pathway induces cell migration and invasion through focal adhesion kinase/AKT signaling-mediated activation of matrix metalloproteinase (MMP)-2 and MMP-9 in liver cancer. Carcinogenesis. 2013;34:10-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 137] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 77. | Shi C, Huang D, Lu N, Chen D, Zhang M, Yan Y, Deng L, Lu Q, Lu H, Luo S. Aberrantly activated Gli2-KIF20A axis is crucial for growth of hepatocellular carcinoma and predicts poor prognosis. Oncotarget. 2016;7:26206-26219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 65] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 78. | Vale RD, Reese TS, Sheetz MP. Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell. 1985;42:39-50. [PubMed] |
| 79. | Chen JS, Liang LL, Xu HX, Chen F, Shen SL, Chen W, Chen LZ, Su Q, Zhang LJ, Bi J. miR-338-3p inhibits epithelial-mesenchymal transition and metastasis in hepatocellular carcinoma cells. Oncotarget. 2016; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 80. | Hyun J, Wang S, Kim J, Rao KM, Park SY, Chung I, Ha CS, Kim SW, Yun YH, Jung Y. MicroRNA-378 limits activation of hepatic stellate cells and liver fibrosis by suppressing Gli3 expression. Nat Commun. 2016;7:10993. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 185] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 81. | Zhang LY, Liu M, Li X, Tang H. miR-490-3p modulates cell growth and epithelial to mesenchymal transition of hepatocellular carcinoma cells by targeting endoplasmic reticulum-Golgi intermediate compartment protein 3 (ERGIC3). J Biol Chem. 2013;288:4035-4047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 129] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 82. | Huang XH, Chen JS, Wang Q, Chen XL, Wen L, Chen LZ, Bi J, Zhang LJ, Su Q, Zeng WT. miR-338-3p suppresses invasion of liver cancer cell by targeting smoothened. J Pathol. 2011;225:463-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 101] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 83. | Huang JY, Zhang K, Chen DQ, Chen J, Feng B, Song H, Chen Y, Zhu Z, Lu L, De W. MicroRNA-451: epithelial-mesenchymal transition inhibitor and prognostic biomarker of hepatocelluar carcinoma. Oncotarget. 2015;6:18613-18630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 69] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 84. | Becker LR, Aakhus AE, Reich HC, Lee PK. A Novel Alternate Dosing of Vismodegib for Treatment of Patients With Advanced Basal Cell Carcinomas. JAMA Dermatol. 2017;153:321-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |