Published online Jun 21, 2016. doi: 10.3748/wjg.v22.i23.5415
Peer-review started: January 8, 2016
First decision: March 7, 2016
Revised: April 8, 2016
Accepted: May 4, 2016
Article in press: May 4, 2016
Published online: June 21, 2016
Processing time: 157 Days and 18.9 Hours
AIM: To evaluate the effect of oral Escherichia coli (E. coli) Nissle application on the outcome of intestinal-borne dermatoses.
METHODS: In a randomized, controlled, non-blinded prospective clinical trial 82 patients with intestinal-borne facial dermatoses characterized by an erythematous papular-pustular rash were screened. At the initiation visit 37 patients entered the experimental arm and 20 patients constituted the control arm. All 57 patients were treated with a vegetarian diet and conventional topical therapy of the dermatoses with ointments containing tetracycline, steroids and retinoids. In the experimental arm patients received a one month therapy with oral E. coli Nissle at a maintenance dose of 2 capsules daily. The experimental group was compared to a non-treatment group only receiving the diet and topical therapy. The primary outcome parameter was improvement of the dermatoses, secondary parameters included life quality and adverse events. In addition the immunological reaction profile (IgA, interleucin-8 and interferon-α) was determined. Furthermore the changes of stool consistency and the microbiota composition over the time of intervention were recorded.
RESULTS: Eighty-nine percent of the patients with acne, papular-pustular rosacea and seborrhoic dermatitis responded to E. coli Nissle therapy with significant amelioration or complete recovery in contrast to 56% in the control arm (P < 0.01). Accordingly, in the E. coli Nissle treated patients life quality improved significantly (P < 0.01), and adverse events were not recorded. The clinical improvement was associated with a significant increase of IgA levels to normal values in serum as well as suppression of the proinflammatory cytokine IL-8 (P < 0.01 for both parameters). In the E. coli Nissle treated group a shift towards a protective microbiota with predominance of bifidobacteria and lactobacteria (> 107 CFU/g stool) was observed in 79% and 63% of the patients, respectively (P < 0.01), compared to no change in the control group without E. coli Nissle. Moreover, the detection rate of a pathogenic flora dropped from 73% to 14 % of the patients in the experimental arm (P < 0.01) with no significant change in the control arm (accounting 80% before and 70% after the observation period, P > 0.05). Accordingly, stool consistency, color and smell normalized in the E. coli Nissle treated patients.
CONCLUSION: E. coli Nissle protects the mucus barrier by overgrowth of a favorable gut microbiota with less immunoreactive potential which finally leads to clinical improvement of intestinal borne dermatoses.
Core tip: The occurrence of facial dermatoses with erythematous papular-pustular exanthemas is often linked to intestinal inflammation. However, the underlying mechanism remains unclear, and innovative treatment options are missing. Here we show that patients with these dermatoses carry a more aggressive microbiota associated with suppressed serum IgA levels, but increase of the proinflammatory cytokines interleukin-8 and interferon-α. Clinical manifestation, microbiota and inflammatory parameters are significantly improved by application of Escherichia coli Nissle. It indicates the usefulness of this probiotic therapy in a neglected patient population in desperate need for effective help.
- Citation: Manzhalii E, Hornuss D, Stremmel W. Intestinal-borne dermatoses significantly improved by oral application of Escherichia coli Nissle 1917. World J Gastroenterol 2016; 22(23): 5415-5421
- URL: https://www.wjgnet.com/1007-9327/full/v22/i23/5415.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i23.5415
Gastrointestinal diseases are often associated with facial dermatoses including acne, rosacea or seborrhoic dermatitis which impair the quality of life of these patients[1-3]. Common feature of these manifestations is an erythematous papular-pustular rash. The digestive system reveals in these cases often infections or an altered microbiota[1,4-8]. Some bacterial genera or species, e.g., bacteroides, firmicutes or bifidobacteria, are predominant in comparison to others like Escherichia coli (E. coli), lactobacilli and enterococci[4,5,9-13]. Staphylococci, proteus and candida belong to the transient microflora[14]. The composition of the intestinal microbiota is mainly determined by dietary patterns[12]. The function of the microbiota has recently been the focus of scientific interest because it is not only responsible for maintaining a physiological immune response, but also for metabolic processes connected with insulin resistance, obesity and manifestation of fatty liver disease[1,2,8,15-17]. It has also been suggested that an intestinal micro-ecologic imbalance may cause dermatoses induced by an overstimulated immune system[17-19]. This could have therapeutic implications by changing the microbiota towards less aggressive bacterial colonization. One example is the oral application of the E. coli strain Nissle 1917 (EcN). By means of special adhesive organelles (by the type F-1A, F-1C and curly fimbriae), the strain has an ability to attach to the mucus membrane of the large intestine and to arrange as microcolonies, forming of a biofilm[20]. Due to the presence of flagella, the bacteria are also mobile, which gives them the advantage of colonizing the colon[21,22]. Therefore, these bacteria were shown to strengthen the mucosal barrier also by interacting with immune modulatory and anti-inflammatory mechanisms[23,24]. E. coli Nissle inhibits the growth of Gram-negative anaerobic bacteria by its secretion of antimicrobial substances (microcins) and by siderophores which capture iron and, thus, prevent the growth of certain pathological bacterial strain[16,25]. A postulated overstimulation of the immune system in intestinal disease-related dermatoses by a pathologic microbiota could be identified by elevation of cytokines and chemokines in the circulation[18,26]. Central players are interleukin-8 (IL-8) and α-interferon, which attract mononuclear cells to the site of inflammation to destroy pathogens by activation of the immune system[3]. Before a pathological microbiota invades the organism it has to pass the mucosal barrier. There are several lines of defense which have to be broken[3,26]. The mucus is the first hurdle which has to be taken. Within the mucus there is IgA which is known to inactivate invading bacteria. Since it is secreted from systemic sources, patients with IgA deficiency are prone to intestinal-borne infections[27].
Accordingly, in this study we evaluate the role of IL-8, interferon (INF)-α and IgA as players in the pathogenesis of intestinal disease related dermatoses and the effect of oral administration of E. coli Nissle in these conditions.
In the randomized, controlled, non-blinded, prospective clinical trial 82 patients met the criterion of papular-pustular exanthema with facial manifestation. They were instructed to participate in a clinical trial, informed about the nature of the study and randomized by a closed envelope drawing to the experimental (EA) or control (CA) arm population. Between the evaluation and initiation visit (up to 4 wk interval), 4 and 21 of the participants in the EA and CA groups, respectively, were lost for the study population. The high loss of patients in the control arm was due to the information of the patients that they did not participate in active treatment protocol with E. coli Nissle. Thus, finally 37 patients entered the EA and 20 patients constituted the CA group. All included patients underwent physical examinations including the consultation of a dermatologist to verify the diagnosis of the skin dermatoses.
For basic treatment of chronic dermatoses, a diet with predominance of vegetable products was prescribed for all patients. The patients of the control arm (CA) only received standard topical therapy prescribed by a dermatologist, consisting of ointments containing tetracycline, steroids and retinoids (Kremgen® and Lokoid®). The patients of the experimental arm (EA) received a combination treatment which included the standard topical therapy of the dermatoses in combination with oral administration of E. coli Nissle 1917 (Mutaflor®): 1 capsule daily for 4 d, then 2 capsules daily for the following month. One capsule of the E. coli Nissle 1917 contained 2.5 - 25 × 109 live bacteria (CFU). The capsules are resistant to gastric juice and do not disintegrate before they reach the terminal small intestine. The patients were informed about the need to store the medication at a cool place. Follow-up examinations of the dermatoses were performed after a month. The therapeutic effect was estimated according to the dynamics of improvement of the dermatological manifestations. Another criterion was the subjective evaluation of the patients in regard to tolerability and adverse events. Life quality was measured by a scale of 4 index points including: good, acceptable, impaired and not acceptable. For testing of the immunological response in blood, a white blood cell (WBC) differentiation was performed, and the concentration of IL-8 and INF-α was determined by an immunoassay. IgA was quantified by an immunoassay method. The stool of the patients was evaluated in regard to its consistency, color, smell, mucus content and WBC.
Furthermore the stool neutral fat, fatty acid, starch content and presence of muscle fibers were determined[14]. Quantification of bacterial strains was performed by standard techniques[14]. The trial was approved by the local ethical committee.
All study participants were informed about the study nature and signed a written consent form. The study protocol was approved by the regional committee for research ethics.
Statistical analysis performed by using SPSS-20 software. All data in this study were expressed as mean ± SD or percent. The Kolmogorov-Smirnov normality test was used for data distribution analysis. All the values had parametric distribution. Analysis of Variance was applied for multiple comparisons and if the results were significant, a post-hoc Turkey’s test was performed. The comparison of the connected values namely the data from the same patient before and after treatment was done using the Student’s t-test for paired samples. The differences between groups were considered significant at P < 0.05.
Out of 123 patients with dermatoses primarily evaluated, 82 patients revealed a papular-pustular exanthema with facial manifestation. Of these, 57 patients agreed to participate in the study and were finally included in the trial with an age range from 18 to 42 years, a disease duration range from 1 to 10 years, and a gender distribution of 35 women and 22 men. The experimental arm (EA) consisted of 37 and the control arm (CA) of 20 patients (Table 1).
| Characteristic | Experimental | Control |
| arm (n = 37) | arm (n = 20) | |
| Sex (women) | 63% | 61% |
| Sex (male) | 37% | 39% |
| Age (yr) | 29 ± 3.1 | 28 ± 2.5 |
| Smoker | 34% | 36% |
| Oral contraception (women) | 4% | 5% |
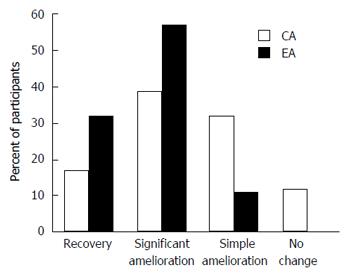
All patients revealed the predominant feature of erythema in conjunction with papular-pustular elements. Twenty-two percent out of the patients were diagnosed with acne, 36% with papular-pustular rosacea and 57% with seborrhoic dermatitis. In 10% of the cases the entire facial skin was involved. Concerning the primary end point of the trial, the improvement of the dermatologic features was significantly greater in the EA compared to the CA group (P < 0.01). After one month, in the EA group 32% showed recovery and 57% significant amelioration (11% simple amelioration), whereas in the CA group only 17% revealed recovery and 39% significant amelioration (32% simple amelioration and 12% no change) (Figure 1).
The resolution of clinical manifestations of the inflammatory process of the facial skin occurred in the reversed order of their development. Initially, edema and swelling decreased, later papular rush and erythema faded as well as the formation of new papulae and pustulae discontinued. This was followed by disappearance of crusts in the area of the lesions, and nodular eruptions gradually flattened (Figure 2 as an example).
All patients in both groups tolerated the treatment very well, and adverse events were not recorded. All patients in the EA showed an increase of life quality by 1.7 ± 0.6 index points (< 0.01) revealing an acceptable or good condition in all patients of the EA group in contrast to the CA group with an overall unchanged impaired life quality (P > 0.05). Accordingly, E. coli Nissle showed high therapeutic efficacy in addition to good tolerability and absence of serious adverse reactions reported by the patients.
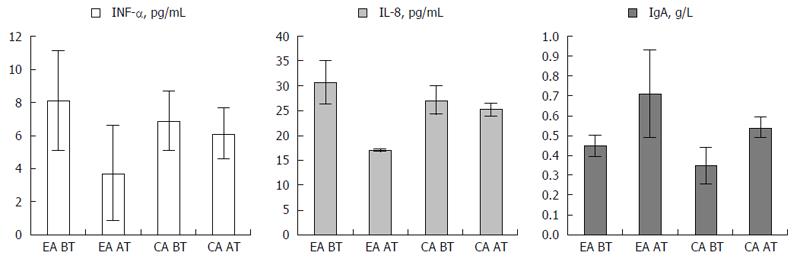
Elevated INF-α values showed a trend towards reduction in the EA patient population after therapy, but did not reach statistical significance. Low serum IgA levels were initially recorded in the EA and CA group. After treatment, the IgA level was normalized only in the EA arm (Table 2 and Figure 3). The same is true for the elevation of IL-8 cytokine levels which were normalized after treatment in the EA patient group. This is probably due to the immunomodulatory properties of E. coli Nissle 1917 which decreases the level of newly activated T-lymphocytes migration into the focus of inflammation. Accordingly, also the lymphocytosis disappeared in 78% of the EA group, whereas it improved in the CA group only in 42 % of the patients (Table 2 and Figure 3).
| INF-α, pg/mL | IL-8, pg/mL | IgA, g/L | ||||
| Arms of the patients | Before | After | Before | After | Before | After |
| treatment | treatment | treatment | treatment | treatment | treatment | |
| Experimental | 8.07 ± 2.97 | 3.73 ± 2.88df | 30.8 ± 4.42 | 17.1 ± 0.65df | 0.45 ± 0.05b | 0.71±0.22df |
| arm (n = 37) | ||||||
| Control | 6.88 ± 1.77 | 6.12 ± 1.53 | 27.0 ± 2.72 | 25.2 ± 1.23b | 0.35 ± 0.9 | 0.54 ±0.05d |
| arm (n = 20) | ||||||
In regard to stool appearance, 82% of the patients initially had loose stools of grey color, sticky consistency with strong smell and mucus in large amounts. After treatment, 71% of participants in the EA had a formed stool of typical color and smell, and only small amounts of mucus.
Before treatment, bacteriological stool culture showed a decrease in the number of bifidobacteria and lactobacteria in both patient groups but an increase in potential pathogenic bacteria, i.e., staphylococci, yeasts, bacteroides, proteus, citrobacter and klebsiellae (Table 3).
| Experimental | Control | |||
| arm (n = 37) | arm (n = 20) | |||
| Microflora | Before | After | Before | After |
| characteristics | treatment | treatment | treatment | treatment |
| Bifidobacteria | ||||
| > 107 CFU/g | 5 (14) | 29 (79)b | 3 (15) | 3 (15) |
| Normal | ||||
| < 107 CFU/g | 26 (70) | 7 (19)b | 15 (75) | 14 (70) |
| Below normal | ||||
| absent | 6 (16) | 1 (2)b | 2 (10) | 3 (15) |
| Lactobacteria | ||||
| > 107 CFU/g | 3 (8) | 23 (63)b | 3 (15) | 2 (10) |
| Normal | ||||
| < 107 CFU/g | 27 (73) | 13 (35)b | 15 (75) | 15 (75) |
| Below normal | ||||
| absent | 7 (19) | 1 ( 2)b | 2 (10) | 3 (15) |
| Pathogenic | ||||
| microflora | 27 (73) | 5 (14)b | 16 (80) | 14 (70) |
After therapy with E. coli Nissle (EA), an increase of bifidobacteria and lactobacteria in stool cultures was noted (P < 0.01 for both species). There was a significant decrease recorded in the number of staphylococci, yeasts, bacteroides, proteus, citrobacter, klebsieallae in 59% of the EA-patients as compared to no change in the CA-group (P < 0.01) (Table 3).
The mechanism by which intestinal diseases induce related dermatoses is obscure. Here we show that intestinal-borne dermatoses are accompanied by a shift towards a more aggressive intestinal microbiota spectrum. Due to their potential to invade the mucosal barrier, they activate the immune system with elevation of IL-8 and interferon-α. This could be the reason for induction of dermatoses because they attract mononuclear cells to preformed lesions of the skin, leading to inflammation. The dermatoses are significantly improved after oral application of E. coli Nissle. Whether the low IgA levels initially recorded in the patient population are the consequence or origin of invasion of pathogenic bacteria remains to be determined. However, the consumption of IgA within the mucus seems more likely because it returns to normal values after the treatment course.
In addition to the observed changes of the microbiota distribution, the biofilm of E. coli Nissle per se may also have an impact on stool consistency. It may be due to an effect on motility as well as the functionality of the mucus barrier. The production of short-chain fatty acids increases the nutritional state of the mucus and thus its capability to absorb water[14,20]. This improves the motility as well as the absorption of water and sodium. All of this helps to form a more consolidated stool. More importantly, because the mucosal barrier is strengthened, pathogens cannot easily penetrate and, thus, the E. coli Nissle application prohibits systemic activation of the immune system eventually inducing intestinal-borne dermatoses.
In conclusion, we report that E. coli Nissle is very effective to treat intestinal-borne chronic dermatoses. It shows good tolerability and no adverse events. The mode of action relates to change of the intestinal microbiota towards less aggressive bacteria. This in turn ameliorates the immune response characterized by a normalization of IgA and IL-8. Thus, it represents a treatment option for patients with intestinal borne dermatosis.
Intestinal-borne dermatoses have unknown etiology and there is a medical need for their therapy. The recent observation that the gut microbiota has impact on the immune system guided us to the question whether this induces dermatoses and whether microbiota modulation may be of therapeutic use.
The present study focuses on etiology and therapy of intestinal dermatoses as a neglected field in gastroenterology, although a large number of patients suffer from this entity. It covers the areas of microbiota and the associated systemic immune response.
It is an innovative approach to link the gut microbiota to the pathogenesis of intestinal-borne dermatoses. The fact that modulation of the microbiota by application of Escherichia coli (E. coli) Nissle 1917 improves the dermatoses was unexpected and opens a new avenue of therapy for these patients in need.
The study provides a rationale for the therapy of intestinal-borne dermatoses. Indeed it is shown that E. coli Nissle 1917 improves these dermatoses by suppressing the intestinal microbiota-triggered immune response. It will open avenues of new therapies also with other microbiota-modulating regimens. The study may also stimulate basic research to unravel the interaction of the gut microbiota and the immune system.
The paper deals with different intestinal-borne dermatoses, the adaptive immune response mechanism, the composition of the gut microbiota in regard to protective and aggressive bacterial colonization and the biological activity of E. coli Nissle 1917.
The manuscript is very interesting for the readers.
P- Reviewer: Casadesus D S- Editor: Qi Y L- Editor: A E- Editor: Wang CH
| 1. | Fölster-Holst R. Probiotics in the treatment and prevention of atopic dermatitis. Ann Nutr Metab. 2010;57 Suppl:16-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 2. | Dahten A, Koch C, Ernst D, Schnöller C, Hartmann S, Worm M. Systemic PPARgamma ligation inhibits allergic immune response in the skin. J Invest Dermatol. 2008;128:2211-2218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 3. | Okada H, Kuhn C, Feillet H, Bach JF. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: an update. Clin Exp Immunol. 2010;160:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 715] [Cited by in RCA: 754] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 4. | Penders J, Thijs C, van den Brandt PA, Kummeling I, Snijders B, Stelma F, Adams H, van Ree R, Stobberingh EE. Gut microbiota composition and development of atopic manifestations in infancy: the KOALA Birth Cohort Study. Gut. 2007;56:661-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 539] [Cited by in RCA: 570] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 5. | Sekirov I, Russell SL, Antunes LC, Finlay BB. Gut microbiota in health and disease. Physiol Rev. 2010;90:859-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2501] [Cited by in RCA: 2743] [Article Influence: 182.9] [Reference Citation Analysis (1)] |
| 6. | Fujimura KE, Slusher NA, Cabana MD, Lynch SV. Role of the gut microbiota in defining human health. Expert Rev Anti Infect Ther. 2010;8:435-454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 312] [Cited by in RCA: 288] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 7. | Falkow S, Small P, Isberg R, Hayes SF, Corwin D. A molecular strategy for the study of bacterial invasion. Rev Infect Dis. 1987;9 Suppl 5:S450-S455. [PubMed] |
| 8. | Abraham C, Medzhitov R. Interactions between the host innate immune system and microbes in inflammatory bowel disease. Gastroenterology. 2011;140:1729-1737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 423] [Cited by in RCA: 405] [Article Influence: 28.9] [Reference Citation Analysis (1)] |
| 9. | Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, Gill SR, Nelson KE, Relman DA. Diversity of the human intestinal microbial flora. Science. 2005;308:1635-1638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5700] [Cited by in RCA: 5584] [Article Influence: 279.2] [Reference Citation Analysis (2)] |
| 10. | Bäckhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI. Host-bacterial mutualism in the human intestine. Science. 2005;307:1915-1920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3394] [Cited by in RCA: 3540] [Article Influence: 177.0] [Reference Citation Analysis (5)] |
| 11. | Robles Alonso V, Guarner F. Linking the gut microbiota to human health. Br J Nutr. 2013;109 Suppl 2:S21-S26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 207] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 12. | Wu GD, Chen J, Hoffmann C, Bittinger K, Chen YY, Keilbaugh SA, Bewtra M, Knights D, Walters WA, Knight R. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334:105-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4098] [Cited by in RCA: 4530] [Article Influence: 323.6] [Reference Citation Analysis (1)] |
| 13. | Rajilić-Stojanović M, de Vos WM. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol Rev. 2014;38:996-1047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 765] [Cited by in RCA: 780] [Article Influence: 70.9] [Reference Citation Analysis (0)] |
| 14. | Sonnenborn U, Schulze J. The non-pathogenicEscherichia colistrain Nissle 1917 - features of a versatile probiotic. Microb Ecol Health Dis. 2009;21:122-158. [RCA] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 144] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 15. | Wehkamp J, Harder J, Wehkamp K, Wehkamp-von Meissner B, Schlee M, Enders C, Sonnenborn U, Nuding S, Bengmark S, Fellermann K. NF-kappaB- and AP-1-mediated induction of human beta defensin-2 in intestinal epithelial cells by Escherichia coli Nissle 1917: a novel effect of a probiotic bacterium. Infect Immun. 2004;72:5750-5758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 342] [Cited by in RCA: 343] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 16. | Cichon C, Enders C, Sonnenborn U. DNA-microarray-based comparison of cellular responses in polarized T84 epithelial cells triggered by probiotics: E. coli Nissle 1917 (EcN) and Lactobacillus acidophilus PZ1041. Gastroenterology. 2004;126:A578-579. |
| 17. | Eyerich K, Böckelmann R, Pommer AJ, Foerster S, Hofmeister H, Huss-Marp J, Cavani A, Behrendt H, Ring J, Gollnick H. Comparative in situ topoproteome analysis reveals differences in patch test-induced eczema: cytotoxicity-dominated nickel versus pleiotrope pollen reaction. Exp Dermatol. 2010;19:511-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 18. | Schreiber S, Heinig T, Thiele HG, Raedler A. Immunoregulatory role of interleukin 10 in patients with inflammatory bowel disease. Gastroenterology. 1995;108:1434-1444. [PubMed] |
| 19. | Chen WX, Ren LH, Shi RH. Enteric microbiota leads to new therapeutic strategies for ulcerative colitis. World J Gastroenterol. 2014;20:15657-15663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 43] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 20. | Hancock V, Dahl M, Klemm P. Probiotic Escherichia coli strain Nissle 1917 outcompetes intestinal pathogens during biofilm formation. J Med Microbiol. 2010;59:392-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 92] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 21. | Altenhoefer A, Oswald S, Sonnenborn U, Enders C, Schulze J, Hacker J, Oelschlaeger TA. The probiotic Escherichia coli strain Nissle 1917 interferes with invasion of human intestinal epithelial cells by different enteroinvasive bacterial pathogens. FEMS Immunol Med Microbiol. 2004;40:223-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 158] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 22. | Jacobi CA, Malfertheiner P. Escherichia coli Nissle 1917 (Mutaflor): new insights into an old probiotic bacterium. Dig Dis. 2011;29:600-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 23. | Yoon SS, Sun J. Probiotics, nuclear receptor signaling, and anti-inflammatory pathways. Gastroenterol Res Pract. 2011;2011:971938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 24. | Zyrek AA, Cichon C, Helms S, Enders C, Sonnenborn U, Schmidt MA. Molecular mechanisms underlying the probiotic effects of Escherichia coli Nissle 1917 involve ZO-2 and PKCzeta redistribution resulting in tight junction and epithelial barrier repair. Cell Microbiol. 2007;9:804-816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 310] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 25. | Kamada N, Maeda K, Inoue N, Hisamatsu T, Okamoto S, Hong KS, Yamada T, Watanabe N, Tsuchimoto K, Ogata H. Nonpathogenic Escherichia coli strain Nissle 1917 inhibits signal transduction in intestinal epithelial cells. Infect Immun. 2008;76:214-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 49] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 26. | Adam E, Delbrassine L, Bouillot C, Reynders V, Mailleux AC, Muraille E, Jacquet A. Probiotic Escherichia coli Nissle 1917 activates DC and prevents house dust mite allergy through a TLR4-dependent pathway. Eur J Immunol. 2010;40:1995-2005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Schultz M, Strauch UG, Linde HJ, Watzl S, Obermeier F, Göttl C, Dunger N, Grunwald N, Schölmerich J, Rath HC. Preventive effects of Escherichia coli strain Nissle 1917 on acute and chronic intestinal inflammation in two different murine models of colitis. Clin Diagn Lab Immunol. 2004;11:372-378. [PubMed] |