Published online May 7, 2016. doi: 10.3748/wjg.v22.i17.4380
Peer-review started: December 7, 2015
First decision: January 28, 2016
Revised: February 2, 2016
Accepted: March 2, 2016
Article in press: March 2, 2016
Published online: May 7, 2016
Processing time: 145 Days and 8 Hours
AIM: To identify prognostic factors and to correlate APC mutations with clinical features, including extracolic manifestations.
METHODS: One hundred thirty-five patients who underwent surgical procedures for familial adenomatous polyposis (FAP) were included. FAP was diagnosed when the number of adenomatous polyps was > 100. Data related to patient, extracoloic manifestations, cancer characteristics, operative procedure, follow up and surveillance were collected. APC mutation testing was performed in the 30 most recent patients. DNA was extracted from peripheral blood and polymerase chain reaction products using 31 primer pairs on APC gene were sequenced. A retrospective study was performed to investigate a causal relationship between prognosis and feature of patient.
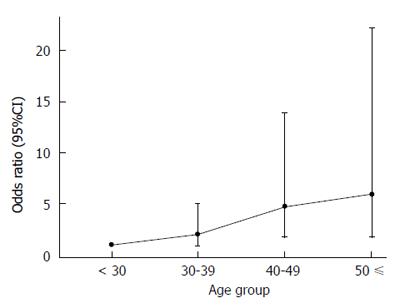
RESULTS: The mean age of the 51 patients with colorectal cancer (CRC) was older than that of those without CRC (30.5 vs 36.9, P = 0.002). Older individuals were more likely to have colon cancer at the time of FAP diagnosis [odds ratio, 4.75 (95%CI: 1.71-13.89) and 5.91(1.76-22.12) for 40-49 years and age > 50 vs age < 30). The number of confirmed deaths was 13 and the median age at death was 40 years (range, 27 to 85 years). Ten of the deaths (76.9%) were from CRC. Another cause of two cases of death were desmoid tumors (15.4%). Development of cancer on remnant rectal or ileal mucosa after surgery was not observed. The APC mutation testing revealed 23 pathogenic mutations and one likely pathogenic mutation, among which were four novel mutations. The correlation between mutational status and clinical manifestations was investigated. Mutations that could prodict poor prognosis were at codon 1309 which located on mutation cluster region, codon 1465 and codon 1507.
CONCLUSION: Identification of APC mutations should aid in the diagnosis and counseling of family members in terms of early diagnosis and management of FAP.
Core tip: Diagnostic delay to make adequate management harder, resulting in advanced colorectal cancer and accompanying morbidities, was not uncommon in patients with familial adenomatous polyposis. This study investigated prognostic factors and the correlation between APC mutations and clinical features, including extracolic manifestations. The present study revealed that early diagnosis and management of high-risk patients is essential and suggests the necessary APC mutations testing in the diagnosis and counseling of patients by informing on disease prognosis.
- Citation: Jung SM, Yoon YS, Lim SB, Yu CS, Kim JC. Clinicopathological features of familial adenomatous polyposis in Korean patients. World J Gastroenterol 2016; 22(17): 4380-4388
- URL: https://www.wjgnet.com/1007-9327/full/v22/i17/4380.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i17.4380
Familial adenomatous polyposis (FAP) is an autosomal dominantly inherited cancer syndrome caused by germ-line mutation of the adenomatous polyposis coli gene (APC) and characterized by the development of 100 to 1000 colorectal adenomatous polyps in its classic form[1]. Subjects with FAP have a risk of almost 100% of developing colorectal cancer by 40-50 years of age[2]. The extracolic phenotype is characterized by the development of adenomatous polyps in the upper gastrointerstinal tract, gastric fundic polyps, desmoid tumors, osteomas, fibrous bone dysplasia, skin fibromas, pineal blastomas, brain cysts, papillary thyroid cancers (PTCs) and congenital retinal pigment epithelial hypertrophy (CHRPE). Colorectal cancer is the most common cause of death in FAP patients and desmoid tumors are the second[3]. To prevent the development of colon cancer, prophylactic removal of the entire colorectal mucosa is recommended for FAP patients.
The FAP registry was performed to improve the prognosis of FAP through early diagnosis and surgery before the development of colorectal cancer[4]. In addition, a recent molecular genetic test for APC mutations makes it possible to detect asymptomatic or not developing polyposis patients. APC mutations can be detected in 80%-90% of patients who have a valid indication for the test[5]. Since most families are managed through hospital-based registries and not by the nation wide registry, there are few investigations of integrative and continuous follow-up to provide treatment guidelines for Korean families. Patients’ indifference and diagnostic delay appear to make adequate management harder, resulting in advanced colorectal cancers and accompanying morbidities.
The purpose of this study was to describe the clinicopathological features of the disease and the expression of extracolic manifestations, and to evaluate whether APC mutations have prognostic value in Korean patients. Finally, we propose management options for high-risk patients to improve outcome.
FAP was defined by the presence of more than 100 colorectal adenomatous polyps, according to intra-operative findings and pathologic reports. In this retrospective study, 135 consecutive patients who underwent surgical procedures for FAP at the Asan Medical Center (Seoul, Korea) between August 1991 and July 2014 were included with median follow-up periods of 54 mo. Patients with fewer than 100 adenomatous polyps and patients with hamartomatous polyposis were excluded. Demographics and length of follow-up were documented for each patient.
The extent of disease and developing cancer was assessed by colonoscopy, abdominopelvic computed tomography (CT) or magnetic resonance imaging (MRI). Positron emission tomography-computed tomography (PET-CT) was performed if advanced colorectal cancer was suspected. Investigation of extra-colonic manifestations was done when FAP was diagnosed or in the immediate postoperative period. Tests included physical examination for skin lesions and palpable masses, X-ray imaging for bone lesions, dental and ophthalmic examination and gastroduodenal endoscopy.
Patients were subjected to one of the following procedures: total proctocolectomy with ileal pouch anal anastomosis (TPC/IPAA) with hand sewn or stapled anastomosis, TPC with end ileostomy, and total colectomy with ileorectal anastomosis (TC/IRA). The patients who underwent TPC/IPAA with mucosectomy with hand sewn anastomosis or TPC with end ileostomy did not have any remnant rectal mucosa. By contrast, the TPC/IPAA with stapled anastomosis or TC/IRA procedures left remnant rectal mucosa.
Annual colonic and gastric endoscopic surveillance is recommended after surgery. Colonoscopy was performed using a standard adult colonoscope or a gastroscope. Any lesion suspected to be an adenoma during endoscopic surveillance was biopsied. If the polyps could not be controlled by endoscopic procedures, surgical procedures were performed. Surveillance endoscopies and associated pathologic reports were reviewed including the incidence, timing and histology of the adenoma and cancer development. In patients with colon cancer, a follow-up assessment was performed every 3 to 6 mo for the first 2 years and annually thereafter.
In our institution, APC germ-line mutation testing has been performed since 2008 in the clinical setting to diagnosis FAP and counsel patients and their family members. The 30 most recent patients among 135 FAP patients underwent mutation tests. Genomic DNA was extracted from peripheral blood using a Qiagen DNA extraction kit (Qiagen, Hilden, Germany). Thirty-one primer pairs were used, including primer pairs covering exons 1-14 and 17 primer pairs covering exon 15 of the APC gene. Polymerase chain reaction (PCR) was performed using Accu-Power PCR premix (Bioneer, Daejeon, Korea). Each PCR product was sequenced using a 3130xl or 3730 genetic analyzer (Applied Biosystems, Foster City, CA). Sequences were aligned using the Sequencher 4.9 software (Gene Codes Corporation, Ann Arbor, MI).
Continuous variables were described as mean ± SD or median with ranges and categorical variables were expressed as numbers and percentages. Differences in characteristics of patients with or without colorectal cancer were calculated using t-tests for continuous variables and χ2 tests or Fisher’s exact tests for categorical variables. Survival curves were generated by the Kaplan-Meier method, and univariate survival distributions were compared with the use of the log-rank test.
All statistical analyses were carried out using SPSS version 20.0 (IBM Corp., Armonk, NY, United States) and R version 3.1.2 (The Comprehensive R Archive Network: http://cran.r-project.org). The level of significance was set at P < 0.05.
The characteristics of the 135 patients are shown in Table 1. There was no significant difference in age at diagnosis between men and women (P = 0.613). The mean age of patients with cancer was significantly greater than that of patients without cancer (P = 0.002) and the mean age of the patients with cancer at an early stage (carcinoma in situ and carcinoma restricted to the submucosal layer), 32.7 years, was between the mean age of the patients with advanced cancer (38.7 years) and that of the patients without cancer. Thirty six of the 39 patients who had a family history of FAP also had a family history of colorectal cancer except three patients who only have a family history of FAP. The rates of familial history of FAP or CRC were significantly higher in patients without CRC than in patients with CRC. Only 12 of the 135 patients had been diagnosed with FAP prior to surgery and the median follow-up period was 5 years. TPC with end ileostomy was performed in patients with rectal cancer in 8 cases and total colectomy was performed in patients who presented a few rectal polyps (less than 5 and endoscopically removable) in 10 cases. There was no significant difference in operative procedure between patients with CRC and patients without CRC. Eighty-eight (65.2%) patients underwent a colonoscopy because of gastrointestinal symptoms at the time of diagnosis and 47 patients (34.8%) underwent a colonoscopy because of positive stool occult blood test or health medical examination without specific symptom (Table 2).
| Total(n = 135) | with CRC(n = 51) | without CRC (n = 84) | P value1 | |
| Age distribution2 | 31 (15-78) | 34 (15-78) | 28 (15-64) | 0.002 |
| < 20 | 11 (8.2) | 5 (9.8) | 6 (7.1) | |
| 20-39 | 86 (63.7) | 23 (45.1) | 63 (75.0) | |
| 40-59 | 34 (25.2) | 21 (41.2) | 13 (15.5) | |
| ≥ 60 | 4 (3.0) | 2 (4.0) | 2 (2.4) | |
| Sex ratio (M:F) | 1:0.78 | 1:0.59 | 1:0.91 | 0.239 |
| CRC FHx | 72 (53.3) | 20 (39.2) | 52 (61.9) | 0.010 |
| FAP FHx | 39 (28.9) | 7 (13.7) | 32 (38.1) | 0.003 |
| Malignancy FHx | 86 (63.7) | 28 (54.9) | 59 (70.2) | 0.071 |
| Follow-up (mo)2 | 54.2 (1-271) | 55.5 (1-271) | 53.49 (1-212) | 0.855 |
| Alive at follow-up | 122 (90.4) | 40 (78.4) | 82 (97.6) | < 0.001 |
| No. of polyps2 | 435 | 427 | 440 | 0.866 |
| OP procedures | 0.846 | |||
| Handsewn TPC/IPAA | 84 (62.2) | 29 (56.8) | 55 (65.5) | |
| Stapled TPC/ IPAA | 33 (24.4) | 10 (19.6) | 23 (27.4) | |
| TPC/end ileostomy | 8 (5.9) | 8 (15.7) | 0 | |
| TC | 10 (7.4) | 4 (7.8) | 6 (7.1) | |
| Mortality | 13 (9.6) | 11 (21.6) | 2 (2.9) | < 0.001 |
| Cancer specific | 10 (7.4) | 10 (19.6) | 0 | |
| Other reason | 3 (2.2) | 1 (2.0) | 2 (2.4) |
| n (%) | ||
| Chief complaint | Hemotochezia | 34 (25.2) |
| Diarrhea | 23 (17.0) | |
| Abdominal pain | 14 (10.4) | |
| Constipation | 8 (5.9) | |
| Abdominal mass | 2 (1.5) | |
| None | 47 (34.8) | |
| Others | 7 (5.2) | |
| Extracolic manifestation | Fundic polyps | 57 (42.2) |
| UGI adenoma | 46 (34.1) | |
| CHRPE | 29/100 (29.0) | |
| Desmoid tumor | 20 (14.8) | |
| Dental anomaly | 11/96 (11.5) | |
| Osteoma | 9/84 (10.7) | |
| Epidermoid cyst | 7 (5.2) | |
| Papillary thyroid cancer | 6 (4.4) | |
| Ampulla of vater adenoma | 5 (3.7) | |
| Adrenal adenoma | 4 (3.0) | |
| Gastric cancer | 4 (3.0) |
Extra-colonic manifestations are described in Table 2. Gastric polyps were found in 63 (46.7%) patients. The histological types of gastric polyps were fundic gland polyp in 57 patients (90.5%) and tubular adenoma biopsied from the antrum of the stomach in 16 patients (25.4%). Ten patients had both fundic gland polyps and tubular adenomas. Four cases of early gastric cancer were diagnosed and the median age was 41 (range, 34-51 years). Two of them were treated by endoscopic submucosal excision and two underwent distal gastrectomy. Among six patients with papillary thyroid cancer (PTC), two were diagnosed with PTC and underwent total thyroidectomy before receiving a diagnosis of FAP. Five patients (3.7%) underwent ampullectomy for polyps in the ampulla of vater in follow-up periods, and high-grade dysplasia was detected in one patient. Desmoid tumors were diagnosed at surgery in four patients and developed in 16 other cases during follow-up; the median age at diagnosis of desmoid tumors was 31 (range, 20-47 years). Desmoid tumors were mostly intra-abdominal in 19 cases, particularly mesenteric with one in the abdominal wall. The postoperative risk for desmoid tumors was not different between men and women (P = 0.625).
Clinicopathologic characteristics are shown in Table 3. The incidence of synchronous lesions and the rates of early colorectal cancer were relatively high. Odds ratio for having colon cancer at the time of FAP diagnosis for 40s and over 50s years were 4.75 (95%CI: 1.71-13.89) and 5.91 (95%CI: 1.76-22.12) when compared with under the age of 30 (Figure 1). Five cases of colorectal cancer were diagnosed in patients aged less than 20 years. Four of them had carcinoma in situ and one showed invasion to proper muscle of rectal wall without lymph node metastasis. Among 60 of de novo patients , who did not have a family history of colorectal cancer or FAP, 31 presented colorectal cancer and it is significantly higher than that of patients with family history of FAP or CRC (51.7% vs 27.8%, P = 0.013), but there was no significant difference in mean age of development of CRC between the two groups (P = 0.265). Patients with gastrointestinal symptoms were more frequently diagnosed with cancer than patients without symptoms and the difference was statistically significant (P = 0.042). Cancer patients without symptoms at the time of diagnosis had a higher incidence of early stage cancer than those with symptoms at diagnosis (P = 0.041).
| n (%) | |
| Tumor location | |
| Colon | 32 (62.7) |
| Rectum | 19 (37.3) |
| Synchronous lesion | 24 (47.0) |
| Definitive stoma | 8 (15.7) |
| Neoadjuvant treatment | 2 (3.9) |
| Tumor grade | |
| WD-MD | 46 (88.2) |
| PD | 5 (11.8) |
| Final pathological staging | |
| Tumor size (mm)1 | 41 (2.44) |
| LVI | 12 (23.5) |
| PNI | 8 (15.9) |
| pT0-1 | 15 (29.4) |
| pT2 | 6 (11.8) |
| pT3 | 25 (49.0) |
| pT4 | 5 (9.8) |
| Lymph node invasion | 25 (49.0) |
| Distance metastasis | 7 (13.7) |
| Staging | |
| 0-1 | 18 (35.3) |
| 2 | 6 (11.8) |
| 3 | 20 (39.2) |
| 4 | 7 (13.7) |
| Local relapse | 0 |
| Systemic relapse | 13 (25.5) |
The patients who have remnant rectum after rectal excision (68.1%) presented a significantly higher incidence of adenomatous polyps in remnant rectal mucosa than other patients underwent TPC and mucosectomy (P < 0.001). Development of cancer on remnant rectal mucosa was not observed, but one patient who underwent TPC/IPAA with stapled anastomosis was diagnosed with villotubular adenoma with high-grade dysplasia and underwent trans-anal excision. There was no significant difference in the incidence of adenomatous polyps in pouch or ileum regardless of operative procedure (P = 0.465) and malignant change or dysplasia was not observed on any ileal adenomas.
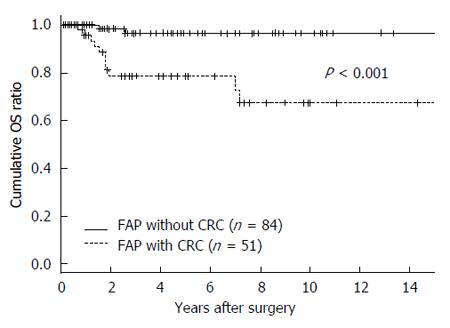
Confirmed deaths were observed in 13 patients with a median age at death of 40 years (range, 27-85 years). Deaths from colorectal cancer accounted for 10 cases (76.9%). In these 10 patients, five cases had distant metastasis at the time of diagnosis. Comparison of postoperative survival between patients with colorectal cancer and patients without cancer is shown in Figure 2. In patients without CRC, only two deaths (15.4%) were observed in patients with desmoids tumor causing major complication and there was no death caused by desmoid tumors in the CRC group.
The germ-line APC mutations that were detected in these patients are listed in Table 4 with clinical details. Two of them was a sister relationship, so 29 unrelated patients were included. Testing revealed 23 pathogenic mutations and one likely pathogenic mutation. Five patients were negative for APC mutations. Of the 24 pathogenic or likely pathogenic mutations, frameshift, nonsense and splice-site mutations represented 54.2%, 33.3% and 12.5%, respectively. Sixteen of 24 mutations were localized within exon 15. Just one mutation in codon 1309 was located within the mutation cluster region (MCR, mutations from codon 1250 to 1464). The mutations on codons 1061, 1309 and 1465 were frequently observed in two cases, respectively. Four novel mutations were identified based on the Human Gene Mutation Database (http://www.hgmd.cf.ac.uk/ac): c.1378G>T, c.3709C>T, c.4519-4520insTGAGCTCA and c.1958+3A>T. A patient with a mutation on codon 461 (c.1378G>T) presented with a desmoid tumor and a upper gastrointerstinal (UGI) adenoma. A man with a novel mutation of codon 1507 (c.4519-4520insTGAGCTCA) was diagnosed with advanced synchronous colorectal cancer and had a family history of colorectal cancer; his father died of colon cancer with liver metastasis at 35 years of age. His older brother also died of colon cancer which was not indicated for surgery at 19 years of age. His younger brother was diagnosed with colon cancer and underwent surgery at age 19, but died of multiple liver metastases at 20 years of age. He presented with CHRPE, dental anomaly and adrenal adenoma.
| Exons (introns, I) | Nucleotide change | AA change | Mutationtype | Age ofdiagnosis | Patient characteristics | ||||
| CRC | No. of polyps | CHRPE | PTC | DT | |||||
| 7 | 778C>T | p.Gln260X | Nonsense | 49 | No | 400 | |||
| 10 | 1378G>T1 | p.Glu460X | Nonsense | 21 | No | 1300 | O | ||
| 10 | 1381G>T1 | p.Glu461X | Nonsense | 23 | No | 400 | O | ||
| 11 | 1495C>T | p.Arg499X | Nonsense | 28 | Yes | 500 | |||
| 13 | 1690C>T | p.Arg564X | Nonsense | 54 | Yes | 400 | |||
| 15 | 2797-2800delAACA | p.Asn933LeufsX21 | Frameshift | 29 | No | 300 | O | ||
| 15 | 2805C>G | p.Tyr935X | Nonsense | 32 | Yes | 600 | O | O | |
| 15 | 3183-3187delACAAA | p.Lys1061fsX | Frameshift | 28 | No | 400 | O | ||
| 15 | 3183-3187delACAAA | p.Lys1061fsX | Frameshift | 12 | No | 800 | |||
| 15 | 3505-3509delGAGAA | p.Glu1169ThrfsX8 | Frameshift | 38 | No | 700 | O | ||
| 15 | 3523C>T | p.Gln1175X | Nonsense | 22 | No | 1400 | O | ||
| 20 | No | 450 | O | ||||||
| 15 | 3578delA | p.Gln1193ArgfsX72 | Frameshift | 34 | No | 100 | |||
| 15 | 3595-3596delAA | p.Lys1199GlufsX8 | Frameshift | 28 | No | 100 | O | O | |
| 15 | 3631-3632delAT | p.Met1211fsX5 | Frameshift | 31 | No | 200 | |||
| 15 | 3709C>T | p.Gln1237X | Nonsense | 20 | No | 100 | O | ||
| 15 | 3925-3928delGAAA2 | p.Glu1309ArgfsX11 | Frameshift | 30 | Yes | 200 | O | O | O |
| 15 | 3927-3931delAAAGA2 | p.Glu1309AspfsX4 | Frameshift | 17 | Yes | 2500 | |||
| 15 | 4393-4394delAG | p.Ser1465TrpfsX3 | Frameshift | 31 | No | 130 | O | ||
| 15 | 4393-4394delAG | p.Ser1465TrpfsX3 | Frameshift | 31 | No | 360 | O | ||
| 15 | 4519-4520insTGAGCTCA1 | p.Ser1507MetfsX6 | Frameshift | 26 | Yes | 1000 | O | ||
| 15 | 4782-4785delAGCC | p.Ala1595ArgfsX54 | Frameshift | 41 | Yes | 100 | |||
| (I 4) | 532-2A>C | Splice-site | 39 | Yes | 100 | ||||
| (I 4) | 532-2A>C | Splice-site | 38 | No | 100 | ||||
| (I 14) | 1958+3A>T1 | Splice-site | 19 | No | 740 | ||||
While CRC is the inescapable fate of untreated FAP patients, few studies provide information on the age distribution at the time of diagnosis of CRC in FAP since most cases are diagnosed in a premalignant stage. In this study, CRC occurred in more than one-third of FAP patients. The prevalence of CRC in de novo patients was significantly higher than that of patients with a family history of FAP or CRC, but there was no significant difference in the mean age of development of CRC between the two groups. The mean age at diagnosis of FAP was significantly different between patients with CRC and those without CRC. In the present study, the prevalence of CRC in patients with a family history was more frequent than that of a previous study for call up patients who diagnosed FAP through family screening[6] since most patients underwent a colonoscopy after developing intestinal symptoms despite having a family history of CRC or FAP. This means that development of CRC in FAP patients depends more on the age at diagnosis than on family history and that we have the opportunity to reduce the frequency of CRC, especially in patients with a family history. We found that diagnosis of colorectal cancer exceeded 10% at the age of 26 and 50% at the age of 43 and that this age distribution was similar to that described in a previous Western report[7]. We also found that there were no cancer-specific deaths in early cancer cases (carcinoma in situ and cancer restricted to the submucosa). Even though some cases of CRC before 20 years of age were observed in this study, all were diagnosed earlier than stage I and survived without recurrence. To improve survival, a reduction in the incidence of CRC at diagnosis should be achieved. These results suggest that a diagnosis of FAP should be obtained before the early twenties.
The standard prophylactic procedure for the majority of patients with FAP is TPC with IPAA. In selected patients, rectum-preserving surgery might be indicated. We performed TC with IRA in six patients who presented with a few rectal polyps. All of them developed adenomas on remnant rectal mucosa but did not develop any carcinomas. The cumulative risk of rectal cancer after IRA was reported to be about one-fourth of patients after 15-25 years despite surveillance[8]. Therefore continuing endoscopic surveillance is necessary for these patients. The incidence of developing adenomatous polyps after TPC in the anorectal transient zone was reported as 10%-15%[9]. There are several reports of adenomatous polyps and cancers arising from the ileal pouch mucosa as opposed to the anastomotic site[10]. We found nine cases of adenomatous polyps arising from the ileal pouch mucosa but did not observe cancer or dysplasia. Development of cancer on remnant rectal mucosa was also not observed, but one patient among 18 cases of adenomatous polyps arising from remnant mucosa was diagnosed with villotubular adenoma with high-grade dysplasia and underwent trans-anal excision. The incidence of tubular adenoma in remnant mucosa and ileal pouch increased over time, so periodical follow-up was necessary.
In the present study, gastric polyps were a common manifestation of FAP. The histological types of gastric polyps were mostly fundic gland polyps and secondary tubular adenomas biopsied from the antrum of the stomach. This is consistent with previous reports documenting that fundic gland polyps are the most common gastric polyps and that adenoma is the second most prevalent gastric lesion in individuals with FAP[11]. We observed that three patients had asymptomatic gastric carcinoma at their first gastroduodenal endoscopy while one developed carcinoma during the surveillance study 18 years after surgery and one developed TA with high-grade dysplasia (HGD) 10 years after surgery, which was treated with endoscopic submucosal dissection. All of these cancers and HGD developed in the antrum. The overall incidence of 3% for gastric carcinoma in this study is similar to the high prevalence of gastric cancer reported in previous study[12]. Nonetheless, the prevalence of gastric cancer in FAP was not reported to be higher than that in the general population in Western countries and the most common gastric cancer site in Western reports was the fundus of the stomach (fundic gland polyps)[11,13]. The high incidence of gastric cancer and the different sites may indicate inter-ethnic differences in the presentation of FAP. In contrast to Western reports of a relatively high incidence of duodenal cancer, we did not observe any malignant neoplasms on the duodenum[14]; however, it is important to consider the possibility of malignancy on the duodenum, since duodenal adenomatous polyps occurred in one-third of patients and since one case of HGD was detected on the ampulla of vater in our study.
We found that desmoid tumor was the only cause of death associated with FAP except caused by CRC in this study. The incidence of 14.8% and location of intra-abdominal site, particularly small bowel mesentery, in the present study is consistent with previous studies[15]. In particular, mutation on codons 1445-1580 is associated with postoperative development of desmoid tumors and recommended to postpone elective colectomy[16], but this correlation does not always appear to be consistent. In our study, four of 20 desmoid tumors developed without any history of surgical trauma. Desmoid tumors unrelated to surgical trauma are a relatively poor prognostic factor[17]. In two of four synchronous desmoid tumors, the same APC mutation (codon 1465; c.4393-4394delAG), reported as a desmoid tumor-associated mutation[18], was detected in unrelated patients. The mutation at codon 1465 located in exon 15 may implicate progression, so patients having this mutation and desmoid tumors should be considered for permanent stoma at the time of primary surgery. Other mutations associated with desmoid tumors in our study were at codons 461 (c.1381G>T) and 1309 (c.3925-3928delGAAA). The incidence of desmoid tumors increased over time, so periodic follow-up is necessary.
Another feature of FAP is the variation in clinical course between patients, so prediction of the severity of the disease is important in the interest of effective cancer prevention. Correlation between mutation and the age at onset of intestinal symptoms and development of CRC was reported[19], but the manifestation may be variable even in patients with identical germ-line mutations. Tumors with mutations localized in exon 15 between codons 1250 and 1464 (mutation cluster region or MCR) have generally a worse prognosis with early onset of the disease and may be candidates for IPAA due to the high incidence of secondary protectomy after primary colectomy[19,20]. In the present study, two kinds of mutations at codon 1309 within the MCR were detected: c.3925-3928delGAAA and c.3927-3931delAAAGA. Deletion of 5 base pairs at codon 1309 (c.3927-3931delAAAGA), the most common mutation, was detected in a patient who presented with CRC at age 17. This finding corresponds with previous results showing that mutation at codon 1309 (c.3927-3931delAAAGA) is associated with early development of intestinal symptoms and death from colorectal cancer[19]. The incidence of this mutation was 4% in our study and 29% in a previous study[16] and its frequency varied. No other MCR mutation was observed in the present study (Table 4). These findings implicate the possibility of ethnic differences in APC mutations. We also observed one interesting novel mutation at codon 1507 within exon 15, c.4519-4520insTGAGCTCA, which was not located in the MCR but presented in a patient with early onset and poor prognosis of CRC. This information may help support surgical decisions of method and timing. In the present study, two-thirds of APC mutations were localized to exon 15. Two of the most common mutations were also identified in this study: at codon 1061 (c.3183-3187delACAAA) and codon 1465 (c.4393-4394delAG). Mutation at codon 1061 has not been reported in Korean patients even though it is one of the most frequently reported mutations.
Extracolic manifestations showed correlations with APC mutations. The expression of CHRPE is associated with a clearly distinct region of mutations located between codons 311 and 1445 of APC[20,21]. All mutations detected with CHPRE in this study were located on reported distinct region but one mutation was not located within this region (codon1507; c.4519-4520insTGAGCTCA). It is necessary to observe carefully the clinical manifestation of this novel mutation. PTC affects 1%-2% of patients with FAP. PTC is associated with a mutation located between codons 140 and 1309 of APC, and a strong association with CHRPE also exists[21]. In the present study, three mutations on codons 935, 1199 and 1309 were detected and all of them presented CHRPE, which is consistent with previous reports. The mutation detection rate in our study is consistent with previous reports[5]. None of the cases can be explained by large allelic deletions, promoter deletions, deep intronic base changes and chromothripsis in germline[22], reduced or absent expression from one allele of APC or bi-allelic germ-line mutation of MYH[23].
A major limitation of this study was the relatively small number of patients and the fact that the study was based on one medical center, so the result may not be generalizable. A multicenter or nationwide registry investigation is needed. Secondly, the retrospective nature of the study and use of medical records for data collection prevented knowledge about affected family members because of unclear medical records and low rates of hospital utilization in the previous two generations in Korea. A prospective cohort study using the FAP registry is required. Finally, the APC mutation test has been used only recently in suspected patients in the clinical setting since gene analysis only became available in 2008 at our institute. Mutation testing should be conducted in patients who do not know their family history of FAP to identify affected members.
In conclusion, we reported clinicopathologic features and prognosis of FAP in Korean patients. The data suggest the necessity for early diagnosis and management of high-risk patients. APC mutations can inform on the severity of the disease and on prognosis and identify family members having the same mutation. The identification of APC gene mutations and the understanding of genotype-phenotype correlations should aid in the diagnosis and counseling of patients.
Familial adenomatous polyposis (FAP) have a risk of almost 100% of developing colorectal cancer by 40-50 years of age. Colorectal cancer is the most common cause of death in FAP patients and desmoid tumors are the second. A recent molecular genetic test for APC mutations makes it possible to detect asymptomatic or not developing polyposis patients. Studies on clinicopathological features of the disease and clinical availability of APC mutations testing were performed in order to improve the early detection and management. Diagnostic delay makes adequate management harder in patients with FAP. This study investigated prognostic factors and the correlation between APC mutations and clinical features.
Diagnostic delay makes adequate management harder in patients with FAP. This study investigated prognostic factors and the correlation between APC mutations and clinical features.
The present study revealed that early diagnosis and management of high-risk patients is essential and suggests the necessary APC mutations testing in the diagnosis and counseling of patients by informing on disease prognosis.
The type of APC mutation was one indicator that could be used to evaluate the poor prognosis of patients with FAP. This finding suggested that APC mutation testing can inform on the severity of the disease and on prognosis and identify family members having the same mutation.
APC gene located on chromosome 5q21-22 and found to be mutated in FAP patients. This gene encodes a tumor suppressor protein (APC protein, 2843 amino acids). APC protein acts as an antagonist of the Wnt signaling pathway and also play a role in process of cell migration and adhesion, transcriptional activation, and apoptosis.
It’s a very nice and significant work. Congrats and thanks to authors for meaningful success. Congrats for authors for writing so much nice topic.
P- Reviewer: Lee SC, Wani IA S- Editor: Yu J L- Editor: A E- Editor: Wang CH
| 1. | Kinzler KW, Nilbert MC, Su LK, Vogelstein B, Bryan TM, Levy DB, Smith KJ, Preisinger AC, Hedge P, McKechnie D. Identification of FAP locus genes from chromosome 5q21. Science. 1991;253:661-665. [PubMed] |
| 2. | Vasen HF, Möslein G, Alonso A, Aretz S, Bernstein I, Bertario L, Blanco I, Bülow S, Burn J, Capella G. Guidelines for the clinical management of familial adenomatous polyposis (FAP). Gut. 2008;57:704-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 564] [Cited by in RCA: 470] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 3. | Arvanitis ML, Jagelman DG, Fazio VW, Lavery IC, McGannon E. Mortality in patients with familial adenomatous polyposis. Dis Colon Rectum. 1990;33:639-642. [PubMed] |
| 4. | Barrow P, Khan M, Lalloo F, Evans DG, Hill J. Systematic review of the impact of registration and screening on colorectal cancer incidence and mortality in familial adenomatous polyposis and Lynch syndrome. Br J Surg. 2013;100:1719-1731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 91] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 5. | Giardiello FM, Brensinger JD, Petersen GM, Luce MC, Hylind LM, Bacon JA, Booker SV, Parker RD, Hamilton SR. The use and interpretation of commercial APC gene testing for familial adenomatous polyposis. N Engl J Med. 1997;336:823-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 277] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 6. | Järvinen HJ. Epidemiology of familial adenomatous polyposis in Finland: impact of family screening on the colorectal cancer rate and survival. Gut. 1992;33:357-360. [PubMed] |
| 7. | Bertario L, Russo A, Sala P, Eboli M, Radice P, Presciuttini S, Andreola S, Rodriguez-Bigas MA, Pizzetti P, Spinelli P. Survival of patients with hereditary colorectal cancer: comparison of HNPCC and colorectal cancer in FAP patients with sporadic colorectal cancer. Int J Cancer. 1999;80:183-187. [PubMed] |
| 8. | Nugent KP, Phillips RK. Rectal cancer risk in older patients with familial adenomatous polyposis and an ileorectal anastomosis: a cause for concern. Br J Surg. 1992;79:1204-1206. [PubMed] |
| 9. | von Roon AC, Will OC, Man RF, Neale KF, Phillips RK, Nicholls RJ, Clark SK, Tekkis PP. Mucosectomy with handsewn anastomosis reduces the risk of adenoma formation in the anorectal segment after restorative proctocolectomy for familial adenomatous polyposis. Ann Surg. 2011;253:314-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Tonelli F, Ficari F, Bargellini T, Valanzano R. Ileal pouch adenomas and carcinomas after restorative proctocolectomy for familial adenomatous polyposis. Dis Colon Rectum. 2012;55:322-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Garrean S, Hering J, Saied A, Jani J, Espat NJ. Gastric adenocarcinoma arising from fundic gland polyps in a patient with familial adenomatous polyposis syndrome. Am Surg. 2008;74:79-83. [PubMed] |
| 12. | Park JG, Park KJ, Ahn YO, Song IS, Choi KW, Moon HY, Choo SY, Kim JP. Risk of gastric cancer among Korean familial adenomatous polyposis patients. Report of three cases. Dis Colon Rectum. 1992;35:996-998. [PubMed] |
| 13. | Ngamruengphong S, Boardman LA, Heigh RI, Krishna M, Roberts ME, Riegert-Johnson DL. Gastric adenomas in familial adenomatous polyposis are common, but subtle, and have a benign course. Hered Cancer Clin Pract. 2014;12:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 14. | Bülow S, Björk J, Christensen IJ, Fausa O, Järvinen H, Moesgaard F, Vasen HF. Duodenal adenomatosis in familial adenomatous polyposis. Gut. 2004;53:381-386. [PubMed] |
| 15. | Nugent KP, Spigelman AD, Phillips RK. Life expectancy after colectomy and ileorectal anastomosis for familial adenomatous polyposis. Dis Colon Rectum. 1993;36:1059-1062. [PubMed] |
| 16. | Friedl W, Caspari R, Sengteller M, Uhlhaas S, Lamberti C, Jungck M, Kadmon M, Wolf M, Fahnenstich J, Gebert J. Can APC mutation analysis contribute to therapeutic decisions in familial adenomatous polyposis? Experience from 680 FAP families. Gut. 2001;48:515-521. [PubMed] |
| 17. | Jung WB, Kim CW, Kim JC. Clinical characteristics and adequate treatment of familial adenomatous polyposis combined with desmoid tumors. Cancer Res Treat. 2014;46:366-373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Martin-Denavit T, Duthel S, Giraud S, Olschwang S, Saurin JC, Plauchu H. Phenotype variability of two FAP families with an identical APC germline mutation at codon 1465: a potential modifier effect? Clin Genet. 2001;60:125-131. [PubMed] |
| 19. | Nieuwenhuis MH, Bülow S, Björk J, Järvinen HJ, Bülow C, Bisgaard ML, Vasen HF. Genotype predicting phenotype in familial adenomatous polyposis: a practical application to the choice of surgery. Dis Colon Rectum. 2009;52:1259-1263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 20. | Nieuwenhuis MH, Vasen HF. Correlations between mutation site in APC and phenotype of familial adenomatous polyposis (FAP): a review of the literature. Crit Rev Oncol Hematol. 2007;61:153-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 244] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 21. | Groen EJ, Roos A, Muntinghe FL, Enting RH, de Vries J, Kleibeuker JH, Witjes MJ, Links TP, van Beek AP. Extra-intestinal manifestations of familial adenomatous polyposis. Ann Surg Oncol. 2008;15:2439-2450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 171] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 22. | Kloosterman WP, Guryev V, van Roosmalen M, Duran KJ, de Bruijn E, Bakker SC, Letteboer T, van Nesselrooij B, Hochstenbach R, Poot M. Chromothripsis as a mechanism driving complex de novo structural rearrangements in the germline. Hum Mol Genet. 2011;20:1916-1924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 248] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 23. | Kim JC, Ka IH, Lee YM, Koo KH, Kim HC, Yu CS, Jang SJ, Kim YS, Lee HI, Lee KH. MYH, OGG1, MTH1, and APC alterations involved in the colorectal tumorigenesis of Korean patients with multiple adenomas. Virchows Arch. 2007;450:311-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |