Published online Nov 7, 2015. doi: 10.3748/wjg.v21.i41.11654
Peer-review started: May 22, 2015
First decision: June 23, 2015
Revised: July 16, 2015
Accepted: September 22, 2015
Article in press: September 22, 2015
Published online: November 7, 2015
Processing time: 166 Days and 12.7 Hours
Helicobacter pylori (H. pylori) plays a role in the pathogenesis of gastric cancer. The outcome of the infection depends on environmental factors and bacterial and host characteristics. Gastric carcinogenesis is a multistep process that is reversible in the early phase of mucosal damage, but the exact point of no return has not been identified. Therefore, two main therapeutic strategies could reduce gastric cancer incidence: (1) eradication of the already present infection; and (2) immunization (prior to or during the course of the infection). The success of a gastric cancer prevention strategy depends on timing because the prevention strategy must be introduced before the point of no return in gastric carcinogenesis. Although the exact point of no return has not been identified, infection should be eradicated before severe atrophy of the gastric mucosa develops. Eradication therapy rates remain suboptimal due to increasing H. pylori resistance to antibiotics and patient noncompliance. Vaccination against H. pylori would reduce the cost of eradication therapies and lower gastric cancer incidence. A vaccine against H. pylori is still a research challenge. An effective vaccine should have an adequate route of delivery, appropriate bacterial antigens and effective and safe adjuvants. Future research should focus on the development of rescue eradication therapy protocols until an efficacious vaccine against the bacterium becomes available.
Core tip: Two main therapeutic strategies could reduce the incidence of Helicobacter pylori (H. pylori)-related gastric cancer: eradication of the infection or vaccination. Success of a gastric cancer prevention strategy depends on the eradication of the infection or on vaccination before irreversible mucosal changes (severe atrophy, intestinal metaplasia or dysplasia) have occurred. Eradication therapy results are suboptimal due to increased antibiotic resistance in H. pylori and patient noncompliance. To improve the rates of eradication, rescue regimens have been developed. Concomitant and sequential protocols seem equally effective rescue strategies. An effective vaccine is not available at present, in spite of enormous effort by different researchers.
-
Citation: Sokic-Milutinovic A, Alempijevic T, Milosavljevic T. Role of
Helicobacter pylori infection in gastric carcinogenesis: Current knowledge and future directions. World J Gastroenterol 2015; 21(41): 11654-11672 - URL: https://www.wjgnet.com/1007-9327/full/v21/i41/11654.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i41.11654
Helicobacter pylori (H. pylori) is a spiral, gram-negative, microaerophilic bacterium that plays an undisputed role in the pathogenesis of gastric and duodenal ulcers, low grade B cell gastric lymphoma (MALT lymphoma) and gastric cancer[1]. In 1982, Warren and Marshall cultivated the bacterium. Their discovery changed the therapeutic algorithm for both peptic ulcer disease and gastric MALT lymphoma. The role of H. pylori in gastric carcinogenesis was clarified in 1991 when large epidemiological studies[1,2] reported a higher incidence of gastric cancer in H. pylori-infected individuals, which confirmed previously published reports[3-5]. Scientific evidence accumulated, and in 1994, H. pylori was named as a human carcinogen by the International Agency for Research on Cancer. The role of the infection in gastric cancer development was further supported by a study by Wang et al[6] that included 2722 early gastric cancer patients and 13976 controls. This study demonstrated a higher H. pylori prevalence in patients with early gastric cancer than in the control group (87% vs 61%, respectively).
Gastric cancer is common; it is the third most common of all cancers among males and the fifth most common among females[7]. The survival rate of advanced gastric cancer patients is very low (< 20%). The incidence of gastric cancer is declining in developed countries but rising in developing countries, and the overall burden of the disease is constantly increasing[7,8].
Distinct patterns of H. pylori gastritis are related to different outcomes of the infection. Chronic corpus-predominant and multifocal atrophic gastritis lead to increased risk of gastric cancer formation, while antrum-predominant gastritis leads to the formation of duodenal ulcer[9-11].
The outcome of H. pylori infection depends on the characteristics of the bacterium in addition to the characteristics of the host and environmental factors.
H. pylori infection is, in majority of cases, acquired during childhood. The bacterium has to overcome the gastric acid barrier and enter the mucus layer to complete the process of colonization[12] and to subsequently induce damage to the gastric mucosa. Furthermore, the persistence of the infection is also important, and it reflects the ability of the bacterium to adapt to its environment and to start multiplication[13].
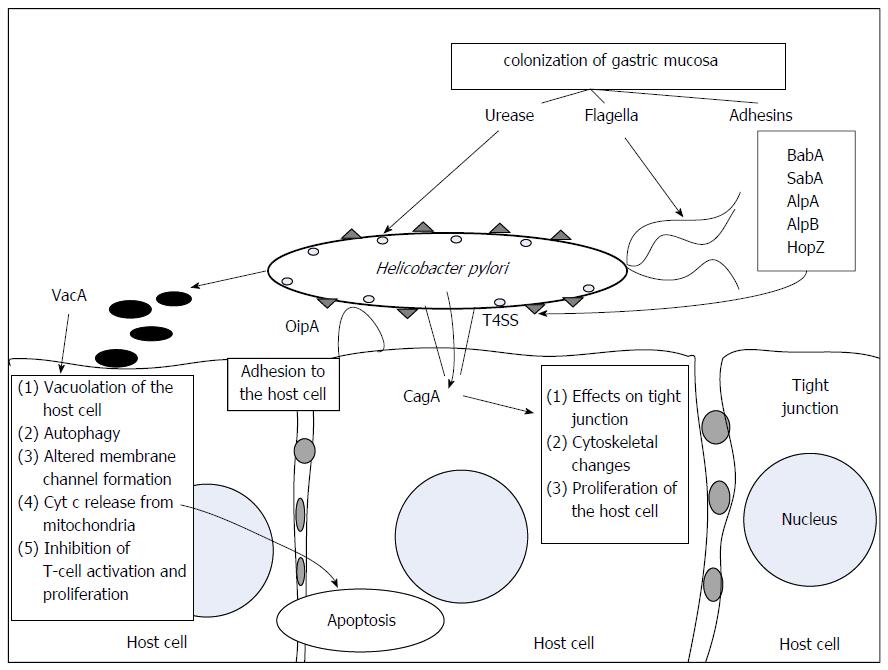
To colonize the gastric mucosa the bacterium uses urease activity, motility and adhesion mechanisms[14].
Urease activity is essential for colonization of the gastric mucosa because in the absence of urea, the bacterium can only survive in a pH range of 4.0-8.0, while in an environment containing urea, it remains viable at a low pH of 2.5. Urease catalyzes the hydrolysis of urea into ammonia and CO2, leading to the increased pH of the bacterial microenvironment. H. pylori urease has a high affinity for urea, which enables the bacteria to utilize the limited amounts of urea that are present in the human stomach[15].
H. pylori flagella-mediated motility is necessary for both colonization of the gastric mucosa and for the persistence of the infection[14]. Expression of two flagellar proteins, FlaA and FlaB, is required for full bacterial motility[16].
Adhesion of H. pylori to epithelial cells enables the bacterium to alter host cell function. Adhesion is mediated through outer membrane proteins that act as adhesins, including BabA, SabA, AlpA, AlpB and HopZ.
The interaction between the bacterium and the gastric mucus occurs through contacts between the bacterial outer membrane protein BabA[17,18] and the Lewisb blood group antigen. BabA is a highly variable protein that is encoded by two genes, babA1 and babA2. The protein encoded by babA2 is functionally active. A major adhesin of H. pylori is SabA (sialic-acid binding adhesin), which interacts with sialylated structures on mucins[14]. The proportion of sialylated structures increases in the gastric mucosa during the course of chronic H. pylori infection. SabA also binds to sialylated receptors on neutrophils and induces activation of the neutrophils. AlpA and AlpB are expressed in all bacterial strains and enable binding to host laminin[14,18]. HopZ also plays role in the colonization process[14,19].
H. pylori uses the thioredoxin system[14,20] to induce partial breaks and changes in the polymeric structure of mucus gel. H. pylori infection and non-specific mechanisms of inflammation simultaneously reduce the protective capabilities of gastric mucin. One-fifth of the presenting bacteria completely adhere to the gastric surface epithelium, while the remaining bacteria reside in the surface mucus layer[21]. The helical shape of the bacterium facilitates its penetration of gastric mucus[22].
H. pylori virulence depends on the above described and other factors that are responsible for damage to the gastric mucosa (Figure 1). Epidemiological studies have identified six distinct H. pylori strains in different geographic regions that are related to different incidences of gastric cancer[23]. These strains are termed hpEastAsia, hpAsia2, hpEurope, hpAfrica1, hpAfrica2 and hpNEAfrica. H. pylori produces various virulence factors and has the ability to modulate its reaction to the host immune response and to thereby adapt to individual host conditions[8].
H. pylori is a highly heterogeneous bacterium[24,25]. Virulence factors that contribute to gastric cancer development include the cytotoxin-associated gene (cagA) A and CagA protein (CagA), CagL, vacuolating cytotoxin (VacA) and outer inflammatory protein (OipA), while the possible role of the duodenal ulcer-promoting gene (dupA) remains unclear[8].
Two distinct types of H. pylori are the CagA-producing (cagA-positive) strains and the CagA-nonproducing (cagA-negative) strains. In animal models, gastric cancer develops only in animals infected with cagA-positive H. pylori strains or when CagA protein is artificially introduced into the host[26,27]. Because in humans, only some individuals infected with cagA-positive strains develop gastric cancer, further investigations have focused on cagA gene polymorphisms. The number of repeat sequences in the 3’ region of the cagA gene differs between H. pylori strains[28]. Each repeat region of the CagA protein contains EPIYA motifs, a term used to describe a specific sequence of amino acids (Glu-Pro-Ile-Tyr-Ala). There are two EPIYA motifs in the first repeat region (EPIYA-A and EPIYA-B) and two in the second (EPIYA-C or EPIYA-D) repeat region[24]. In Western-type H. pylori, CagA proteins have EPIYA ABC, ABCC or ABCCC repeat regions, while in East Asian-type H. pylori, CagA proteins have EPIYA ABD repeats[8,24].
The CagA protein consists of a C-terminal region that contains the EPIYA motifs and an N-terminal region[8]. After adhesion of the bacterium to the host cell, CagA is injected into the host cell via the cag pathogenicity island (cagPAI)-encoded type IV secretion system (T4SS), and electrostatic interactions with phosphatidylserine keep CagA linked to the inner leaflet of the cell membrane[8,29]. In the cytoplasm of the host cell, CagA is phosphorylated at its EPIYA motifs[30]. CagA alters host cell signaling in both a phosphorylation-dependent and a phosphorylation-independent manner[8]. The induction of heme oxygenase 1 reduced CagA phosphorylation in gastric epithelial cells in vitro, while in vivo H.pylori diminishes heme oxygenase 1 gene expression[8,31].
Carcinogenic and virulent H. pylori strains express CagL, a highly conserved protein component of T4SS that is involved in bacterial attachment to the host cell and also in the induction of host inflammatory responses and carcinogenesis[29]. CagL induces hypergastrinemia, which is a risk factor for the development of gastric adenocarcinoma[8,30]. Contact between CagL and α5β1 integrin induces IL-8 secretion from the host cell[32].
VacA induces vacuolization and apoptosis (as a consequence of cytochrome c release from mitochondria) of the host cell. It is also responsible for altered membrane-channel formation and the induction of autophagy and altered host immune responses[30,33,34], mainly through the inhibition of T cell activation and proliferation[35]. The vacA gene is functional in all H. pylori strains and differences in its vacuolating activity have been associated with its gene structure, which varies in the signal (s1 and s2), middle (m1 and m2) and intermediate (i1 and i2) regions[36]. The risk of developing different gastrointestinal pathology is attributable to different combinations of s, m and i region subtype.
The s1/m1 strains induce the highest level of cytotoxicity, the s1/m2 strains induce the lowest, and the s2/m2 strains have no cytotoxic activity (s2/m1 strains are rare)[37]. The risk of developing either gastric cancer or peptic ulcer disease is increased in individuals infected with s1 or m1 H. pylori strains compared with individuals infected with s2 or m2 strains[8,24]. In East Asia, most H. pylori strains are s1 type, and in these patients, the presence of the m1 region is related to an increased risk for gastric cancer[24].
The intermediate region of vacA is localized between the s and m regions. Type i1 is found in all s1/m1 strains, while all s2/m2 strains are type i2. Strains that are type s1/m2 can be either type i1 or i2. Strains with the i1 region are more pathogenic[8]. The type of the i-region has a better predictive value than s region type in some, but not all populations[38].
The deletion (d) region is localized between the i and the m regions[39]. The d region can be type d1 or d2. In patients infected with Western strains, the presence of the d1 region is a risk factor for gastric mucosal atrophy. In patients infected with the East Asia type of Helicobacter, all strains are classified as s1/i1/d1[24].
OipA is a protein that was identified in 2000[24] and is involved in H. pylori adhesion to the host cell, induction of the host pro-inflammatory response and the subsequent increase in mucosal interleukin-8 (IL8) levels[40]. Results from animal studies suggest a role for OipA in gastric carcinogenesis[8,26].
CagA, VacA and OipA synthesis is linked. Therefore, almost all H. pylori strains produce either all or none of these proteins. East Asian H. pylori strains are highly pathogenic and CagA, VacA and OipA-producing. According to the available data, the presence of these three genes is related to gastric cancer and peptic ulcer disease pathogenesis[24].
Duodenal ulcer-promoting gene (dupA) plays a role in T4SS formation and is localized in the plasticity zone. Lu et al[41] proposed dupA as an H. pylori virulence factor that is involved in duodenal ulcer pathogenesis and that confers a protective effect against gastric carcinogenesis[8], but other studies have failed to demonstrate a relationship between this gene and any distinct gastroduodenal pathology, and they have therefore not supported this hypothesis[23,42,43]. These results could be explained by polymorphism of the dupA gene[30].
Multilocus sequence typing of housekeeping genes revealed that there are six distinct H. pylori strains (hpEurope, hpEastAsia, hpAsia2, hpAfrica1, hpAfrica2 and hpNEAfrica). These strains are associated with different geographic regions and with the incidence of gastric cancer.
It is probable that housekeeping gene differences are merely markers for virulence factors that affect disease outcome[8,23]. Initially, four main clusters were identified by Falush et al[44]. HpEurope isolates were found in Europe and in countries colonized by Europeans, while the majority of isolates from East Asia were the hpEastAsia strain. HpAfrica1 is widely spread, while hpAfrica2 is found exclusively in South Africa. Two clusters were later identified: hpAsia (South and Southeast Asia) and hpNEAfrica (the predominant isolate in Northeast Africa)[8,25].
The geographical distribution of HpEastAsia isolates is concordant with the high incidence of gastric cancer in these areas. On the other hand, in low gastric cancer incidence areas, such as Africa and South Asia, most strains are hpNEAfrica, hpAfrica1, hpAfrica2 or hpAsia2. This is a plausible explanation for both African and Asian enigma. The high incidence of H. pylori infection is related to the high incidence of gastric cancer in East Asia, while a low incidence of gastric cancer is observed in populations with a high prevalence of H. pylori infection in Africa (the African enigma) and South Asia (the Asian enigma)[8,23].
After infection with H. pylori, inflammation and mucosal damage occur in the non-acid secreting gastric antrum. Over time, mucosal damage progresses into the gastric corpus. The atrophic border can be recognized endoscopically, and the damage progresses more rapidly along the lesser curve than the greater curve, as previously reported[44,45]. Chronic inflammation that is related to H. pylori affects cell differentiation and promotes metaplasia[46-48]. As the damage spreads into the corpus, pyloric metaplasia is observed near the atrophic border. Pyloric metaplasia exhibits similar immunohistochemical characteristics to spasmolytic polypeptide/trefoil factor family 2-expressing metaplasia (SPEM), which has been described in animal models of gastric carcinogenesis[49,50] and is probably an important step in gastric cancer formation.
Gastric adenocarcinoma can originate from both proximal (cardia) and distal (non-cardia) parts of stomach. Proximal and distal gastric cancers have different epidemiological and clinical characteristics[8]. Risk factors for proximal gastric cancer include increased body weight, gastro-esophageal reflux disease and Barrett’s esophagus[8,51], while distal gastric cancer risk is increased by the presence of H. pylori infection[9-11], family history of gastric cancer, low socioeconomic status, smoking and a diet rich in salty and smoked food with low consumption of fruits and vegetables[52].
According to the Lauren classification, gastric cancer is divided into intestinal and diffuse histological subtypes. The presence of H. pylori infection and corpus-predominant gastritis with intestinal metaplasia leads to intestinal-type gastric cancer, whereas diffuse gastric cancer arises from non-atrophic pangastritis[9-11].
Long-lasting precancerous processes result in intestinal-type gastric adenocarcinoma. In 1938, pathologists proposed that the presence of gastric intestinal metaplasia is related to gastric cancer[8]. This model, now known as the Correa cascade, was reintroduced and proposed by Correa et al[53] in 1975. The authors updated their model in 1988 and 1992. In the Correa cascade, consecutive histological changes in the gastric mucosa occur, leading to gastric cancer through the following steps: normal gastric mucosa, superficial (non-atrophic) gastritis, multifocal atrophic gastritis, complete (small intestine type) intestinal metaplasia followed by intestinal metaplasia of the incomplete (colonic) type, low-grade dysplasia, high-grade dysplasia and invasive adenocarcinoma[9-11,53]. It is now believed that intestinal metaplasia arises from SPEM and that SPEM may also provide the cells of origin for gastric cancer[50,54]. Intestinal metaplasia is considered by some authors as a surrogate marker for the presence and extent of gastric mucosal atrophy[55-57]. The concept of multifocal atrophic gastritis represents areas of intestinal metaplasia in SPEM-type atrophy damage[58,59]. Nevertheless, the critically important point of no return, up to which gastric cancer prevention is possible and histological changes are reversible, remains unidentified[8].
Apart from the bacterial strain, the characteristics of the host play a role in gastric carcinogenesis. Different host gene polymorphisms have been described, mainly as single nucleotide polymorphisms. These polymorphisms influence host inflammatory immune responses and affect host cell proliferation and mucosal protection. They also exert an effect on the metabolism of carcinogens and antioxidants[8].
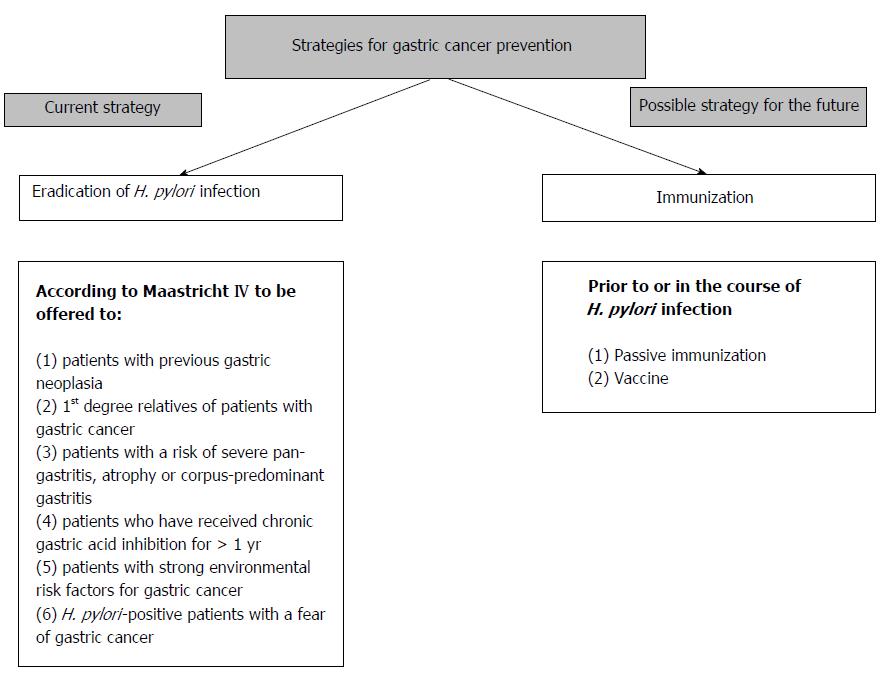
There are two main therapeutic strategies that could reduce the incidence of gastric cancer: eradication of an ongoing infection or immunization prior to or during the course of the infection (Figure 2). The success of a gastric cancer prevention strategy depends on its timing because prevention strategies should be introduced before the point of no return in gastric carcinogenesis. Although the exact point of no return has not been identified, infection should be eradicated before severe atrophy of the gastric mucosa develops.
Effects of eradication therapy for H. pylori infection on either invasive gastric cancer or premalignant histological lesions of the gastric mucosa have been reported in five randomized control trials (RCT)[60-65].
Gastric cancer incidence was evaluated in a RCT conducted by Wong et al[60] that lasted for 7.5 years. Healthy individuals were randomized to receive either eradication therapy or placebo in a region in China with a high gastric cancer incidence. The study results did not demonstrate a decrease in overall gastric cancer incidence, but the results did suggest a protective role for H. pylori eradication in subjects without precancerous lesions[8].
The low number of gastric cancer cases[60,63] and the study design (it did not aim to assess gastric cancer incidence as a primary outcome) of other previously published studies[61,62] has led to a lack of scientific evidence for the possible effects of H. pylori eradication on cancer occurrence. Evidence in favor of a protective effect of H. pylori eradication on gastric cancer formation came from a study by Ma et al[64]. They published the results of a RCT after 15 years of follow-up with 2258 H. pylori seropositive adults. In this study, participants were selected from the general population and randomly assigned to one of three intervention groups (H. pylori eradication, garlic supplements, or supplemental vitamins) or a control group. This study demonstrated reduced gastric cancer incidence in the group of patients treated with eradication therapy for H. pylori.
The benefits of mass eradication in populations with high incidence of H. pylori was assessed by Lee et al[66]. Individuals with positive H. pylori urea breath test underwent endoscopic screening and received eradication therapy. The success rate of the eradication therapy was 78.7%, and it led to a decrease in gastric atrophy incidence. However, no significant change in intestinal metaplasia was observed. The incidence of gastric cancer decreased by 25% during the study.
An important study from Uemura et al[67] assessed the effect of H. pylori eradication on metachronous gastric cancer development in patients who had a previous endoscopic resection of an early gastric carcinoma. H. pylori-positive patients who underwent endoscopic resection were randomized to receive either H. pylori-treatment or no treatment. After four years of follow up, metachronous cancer was not diagnosed in any of the H. pylori-treated patients compared to 9% in the group that received no treatment. These findings were confirmed in a larger study by Fukase et al[65], who demonstrated that eradication therapy in patients with previous endoscopic resection of early gastric cancer reduced the risk of metachronous gastric carcinoma by 65%. Current Japanese guidelines reflect an acceptance of the results of this study and suggest H. pylori eradication therapy after endoscopic resection of early gastric cancer[68].
Recently published data are available from a retrospective study performed in South Korea[69] that analyzed the relationship between the risk of metachronous gastric cancer in patients who underwent endoscopic resection of early gastric cancer and H. pylori eradication therapy. This study confirmed the results of Fukase et al[65] and demonstrated that successful H. pylori eradication may reduce the occurrence of metachronous gastric cancer.
Recent trial reports[64-66,69] have also provided evidence of a protective effect of H. pylori eradication in gastric cancer.
According to the Maastricht IV consensus, eradication of H. pylori reduces the risk of gastric cancer development. Patients should be treated during the initial phase of the infection, before preneoplastic changes in the gastric mucosa occur. Authors of the Maastricht IV consensus identified individuals with an increased risk for gastric cancer. They suggest that to decrease gastric cancer risk, eradication therapy should be offered to first-degree relatives of family members with a diagnosis of gastric cancer, patients with previous gastric neoplasia who have already been treated by endoscopic or subtotal gastric resection, patients with a risk of severe pan-gastritis, corpus-predominant gastritis or severe atrophy, patients who have received chronic gastric acid inhibition for more than 1 year, patients with strong environmental risk factors for gastric cancer (heavy smoking or high exposure to dust, coal, quartz, cement and/or work in quarries), and H. pylori-positive patients with a fear of gastric cancer[70]. Two other documents, the American College of Gastroenterology guidelines[71] and the Asia Pacific Consensus document[72], have also recommend H. pylori eradication in patients with endoscopic resection of early gastric cancer.
Population screening and eradication of H. pylori was a matter of scientific debate until recently. Currently, the Maastricht IV consensus encourages[70], while the Asia Pacific consensus strongly recommends, population screening and treatment for H. pylori in high-risk regions as a chemo-prophylactic measure for gastric cancer[71]. The rationale behind this is the cost effectiveness of eradication therapy when compared to the cost of treatment of advanced gastric cancer.
H. pylori eradication therapy should stop the progression of mucosal damage and reduce gastric cancer risk[73]. Eradication of the infection stops the inflammatory process and promotes healing of gastritis and a resolution of inflammation. H. pylori leads to gastric cancer through the Correa cascade; therefore, when severe atrophic damage and intestinal metaplasia occur, eradication cannot reverse mucosal changes.
After eradication therapy, individuals with non-atrophic gastritis have a negligible risk of developing gastric cancer, while individuals with atrophic gastritis have an increased risk. This risk is overall lower in eradicated patients when compared to untreated patients with the same pattern of gastritis. In untreated patients, the risk for gastric cancer increases yearly as the atrophy progresses[73], as demonstrated by Ohata et al[74] In a large, longitudinal cohort study on 4655 healthy asymptomatic subjects followed for 7.7 years, the authors aimed to determine the association between H. pylori infection and the progression of chronic atrophic gastritis (CAG) with gastric cancer. The authors identified 45 gastric cancer cases, none of which were H. pylori negative and CAG negative, during the study period. Development of CAG increased the risk of gastric cancer. Recently published data[75] from the same group confirmed that in subjects with a serologically diagnosed healthy stomach (H. pylori-negative/pepsinogen within normal range and therefore CAG-negative), the cancer incidence rate was low (16/100000 person-years). On the other hand, in the individuals with an H. pylori infection, they observed progression of chronic gastritis and increased gastric cancer risk. In patients with no atrophy and active inflammation, gastric cancer risk was estimated at 250/100000 person-years, which is comparable to the risk in subjects with CAG. Patients with active inflammation were at risk of diffuse gastric cancer. These results revealed that gastric cancer develops in some patients as a result of the Correa cascade, while in others it can result from a direct carcinogenic pathway based on active inflammation.
Eradication therapy, up to some point, prevents gastric cancer. Data on the possible reversion of atrophic changes in the gastric mucosa is conflicting. A longitudinal cohort study conducted by Yanaoka et al[76] with a mean follow up of 9.3 years demonstrated a significant reduction in cancer incidence after eradication in H. pylori positive patients with mild atrophic gastritis. The incidence in patients with persistent infection was 111/100000 person-years compared to 69/100000 among patients in whom the infection was eradiated. As expected, cancer incidence rates did not vary significantly (237 vs 223) among the patients with severe atrophy. The authors concluded that cancer development after eradication depends on the presence of extensive CAG before eradication and that H. pylori eradication is beneficial in subjects with mild CAG. A study by Sakakibara[77], who followed a small group of 8 patients who were surgically treated for gastric cancer for 9 years, suggested a prompt improvement in the atrophy score (reversion of atrophic changes) in the remaining gastric mucosa following eradication therapy. The authors concluded that H. pylori eradication improved possible precancerous lesions in the gastric remnant.
Watari et al[78], in a prospective study, followed 96 patients for 4 years who exhibited chronic gastritis with or without intestinal metaplasia or gastric intestinal metaplasia with dysplasia and failed to demonstrate a change in intestinal metaplasia score. Nevertheless, the authors reported a change in the intestinal metaplasia phenotype: they followed the expression of several biomarkers related to carcinogenesis and demonstrated regression in TC22-4. TC22 is a neoplastic marker that is expressed exclusively by transformed epithelial cells. Based on this finding, they suggested that the change of phenotype may be an important factor in the reduction of cancer incidence after eradication of H. pylori. H. pylori eradication prior to development of intestinal metaplasia was beneficial for patients with corpus gastritis. However, eradication in high risk patients (i.e., patients with atrophy with intestinal metaplasia, especially of the incomplete type or with a history of endoscopic treatment for gastric cancer) was not beneficial[79]. Identification of the point of no return for the development of malignancy is an important, but still unanswered, scientific goal.
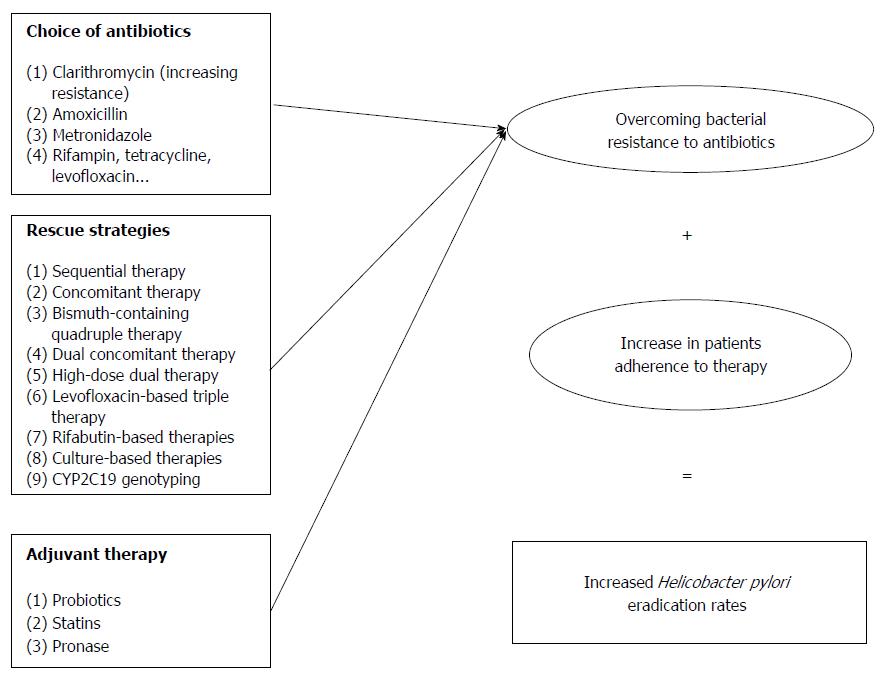
There are two main underlying causes of suboptimal results of eradication therapy: H. pylori resistance and patient noncompliance (Figure 3).
In the first years after the discovery of H. pylori and its role in the pathogenesis of major gastric diseases, eradication therapy seemed to be a safe and efficient strategy that would resolve and eradicate peptic ulcer disease, MALT lymphoma and majority of gastric cancer cases.
In 1993, an eradication protocol consisting of two antibiotics (clarithromycin and amoxicillin or metronidazole) and a proton pump inhibitor was proposed[80,81] and confirmed as efficacious in large studies[82,83].
Eradication therapy is effective when the mucosal concentration of the antibiotic is above the minimal bactericidal concentration (MBC) at the site of the infection for a sufficient time, which enables the eradication of all present bacteria[84]. Macrolides (clarithromycin), beta-lactams (amoxicillin), tetracycline, metronidazole, rifampin (rifabutin) and fluoroquinolones (levofloxacin) were identified as antibiotics with the best potential for eradication of the infection. For these drugs, therapeutic doses result in a mucosal concentration above the MBC.
Selected antibiotics need to be absorbed and released in the gastric mucosa over long periods of time. The in vitro efficacy of aminoglycosides and bismuth salts is compromised in vivo by a poor rate of absorption. In addition, some drugs from the above mentioned groups have displayed disappointing clinical effects, i.e., doxycycline (tetracycline) and ciprofloxacin (fluoroquinolone). There are also other factors that are described in detail in a review by Megraud[85] that should be considered when assessing antibiotic efficacy. These factors include the acidity of the stomach and bacterial resistance. The majority of antibiotics are not active at low pH and are only active in dividing bacteria, making the use of proton pump inhibitor (PPI) mandatory.
Clarithromycin is the basis for H. pylori treatment because it has MBC, good mucosal diffusion and is not affected by gastric acidity. The addition of a second antibiotic provides a high and permanent eradication rate. The second antibiotic of choice is either amoxicillin or metronidazole. Triple therapy was a standard treatment until recent years, in which we are facing increased H. pylori resistance to clarithromycin that is attributable to the selection of bacteria with point mutations that have occurred during replication[86,87].
The global clarithromycin resistance rate in Europe increased from 9% in 1998[88] to 17.6% in 2008[89]. It has been suggested that clarithromycin resistance is the major cause of eradication treatment failure. Data on clarithromycin resistance was obtained in a study that included 18 European countries. Primary H. pylori antibiotic resistance rates were, for adults, 17.5% for clarithromycin, 14.1% for levofloxacin and 34.9% for metronidazole. Higher resistance rates for clarithromycin and levofloxacin are observed in Western/Central and Southern Europe (> 20%) than in Northern European countries (< 10%). There are also data from individual countries showing a wide range of resistance rates to clarithromycin. Resistance rates range from 0% in India to 49.2% in Spain[90,91]. There is also cross-resistance for all of the other macrolides[85].
Resistance to rifampin, amoxicillin and tetracycline is rare[85]. Resistance to rifampin is observed in patients previously treated for tuberculosis, while resistance to tetracycline has been reported in some[92,93], but not all studies from Korea and Brazil[94,95]. Resistance to metronidazole is not frequently observed and depends on the other drugs used and the length of treatment[85].
The first Maastricht conference proposed a triple treatment including PPI-clarithromycin and amoxicillin or metronidazole[96] that currently has an eradication rate of 70% (the aimed-for eradication rate for any protocol should be over 80%)[97]. Possible explanations for this decrease in efficacy of the standard triple therapy include low compliance, high gastric acidity, high bacterial load, the type of strain and an increase in H. pylori resistance to clarithromycin. Maastricht IV therefore suggests that PPI-clarithromycin-containing triple therapy without prior susceptibility testing should not be prescribed in regions with a clarithromycin resistance rate of more than 15%-20%[70].
Because no new drug has been developed for this indication, a number of studies have focused on the use of different combinations of known antibiotics.
Possible scenarios proposed to overcome the problem of low eradication rates include the administration of sequential or concomitant therapy. Sequential therapy (ST) consists of a PPI and amoxicillin administered for the first five days followed by a PPI and 2 other antibiotics for the following 5 d. This sequential administration weakens the bacterial cell wall in the initial phase and helps to increase eradication rates, even in clarithromycin-resistant strains. Concomitant therapy (CT) regimen consists of all of the medication administered in ST, but given simultaneously. The efficacy of both ST[97,98] and CT[99,100] therapies has been supported in different studies. A recent meta-analysis provided data on the efficacy of concomitant vs sequential therapies[101]. The analysis was based on 7 RCTs that included 2412 individuals. The sequential regimen was successful in 83.8% of patients, while concomitant therapy eradicated the infection in 86.1% of patients. The adverse events and adherence to medications were not different between the two regimens.
The idea of a bismuth-containing quadruple therapy was revisited and improved through the single pill concept. Namely, a formulation containing bismuth salts, tetracycline and metronidazole in the same pill was developed[102-104].
Hsu et al[105] proposed hybrid (dual concomitant) therapy consisting of dual therapy (PPI and amoxicillin for 7 d) followed by a concomitant quadruple therapy (PPI, amoxicillin, clarithromycin and metronidazole for another 7 d). The eradication rate for this treatment was over 97%.
High-dose dual therapy consists of administration of PPI and amoxicillin three times a day for 2 wk and was initially designed for areas with high resistance to clarithromycin. It provides eradication in 78.4% of patients[106,107] and does so with fewer side effects and better compliance. The authors therefore suggested that larger studies are needed[108].
Levofloxacin-based triple therapy consists of PPI, levofloxacin, and amoxicillin for 10 d, and the eradication rate of levofloxacin-based triple therapy ranges from 74% to 96%[109-111]. This regimen is not recommended as the first line treatment because augmented use of quinolones for respiratory and urogenital infections increased H. pylori resistance to these drugs[109]. The resistance to levofloxacin is a consequence of a point mutation in a special region, the so-called quinolone resistance determining region. There is also cross-resistance in all fluoroquinolones[91]. Levofloxacin-based therapy is considered to be an efficient alternative regimen in populations with 15%-20% clarithromycin resistance and quinolone resistance less than 10% and is a second line treatment, according to the Maastricht IV consensus[70]. Selection of quinolone therapy should be based on the results of antibiotics susceptibility tests or geographic resistance patterns due to the rapid increase in the number of resistant strains[91].
Rifabutin-based therapies were introduced as rescue therapies based on the results of in vitro studies[112]. A triple regimen includes amoxicillin, PPI and rifabutin, but the optimal duration of treatment is not defined and ranges from 7 to 14 d[113-115]. Myelotoxicity is a rare but significant complication that limits its widespread use[113]. The potential for mycobacterial resistance also limits the use of this regimen, leaving it as valid option only as a rescue treatment.
Culture-based therapies are recommended after the failure of second-line treatments. An antimicrobial susceptibility test is recommended[69], and treatments adjusted to the results achieve more than 90% eradication rate after second-line therapy failure[116]. The test is invasive, expensive and has low sensitivity (less than 60%)[117]. Mixed infections with susceptible and resistant H. pylori strains also limit the efficacy of this therapeutic approach[118].
The relevance of CYP2C19 genotyping as a rescue strategy for improvement of eradication rates is based on the fact that the CYP2C19 polymorphism affects the H. pylori eradication rate, especially by omeprazole treatment[119]. This strategy is probably plausible in Asia, where poor metabolizers account for more than 15% of all patients[72,120].
Adjuvant therapy in H. pylori eradication is likely to be of interest to researchers aiming to increase eradication rates, but the majority of data dwells on the role of probiotics. There is, however, some data suggesting a role for statins and pronase.
Probiotics are considered safe. Thus, they are proposed as an adjuvant therapy to increase eradication rates and to decrease the side effects of therapeutic regimens, especially antibiotic-associated diarrhea. A meta-analysis published by Zhang et al[121] analyzed data from 45 RCTs and revealed that the addition of probiotics to a standard therapy was associated with an increased eradication rate (82.31% in probiotic group vs 72.08% in control group) and a lower incidence of adverse events. The mechanism of this action is probably related to the ability of probiotics to induce anti-inflammatory and anti-oxidative mechanisms that regulate the intestinal microbiota. Today, urease is considered a single possible target for probiotic action[122,123]. According to Ruggiero, it is more likely that probiotics exert indirect and non-specific, rather than direct and specific, anti-H. pylori activity[124].
HMG-CoA reductase inhibitors have many pleiotropic effects. Therefore, Nseir et al[125] tested, in a small RCT, the hypothesis that the addition of simvastatin could improve eradication rates. According to this study, a better eradication rate (91% vs 72%) was observed in the group whose treatment included statin. Further studies are needed in this field.
Pronase is proteolytic enzyme that causes the degradation of gastric mucus, and its addition to standard eradication therapy was investigated in 1995 and 2002[126,127]. A single RCT by Gotoh used pronase with an eradication therapy (lansoprazole once daily, 500 mg of amoxicillin, 250 mg of metronidazole and 18000 tyrosine units of pronase thrice daily for 2 wk) and demonstrated an increased eradication rate in the group treated with pronase (ITT: 94% vs 76.5%, P = 0.0041)[126]. More validation is needed because the therapeutic regimens used in these studies are not currently standard eradication therapy protocols.
Patient adherence to treatment regimens is important for the successful eradication of H. pylori. According to the available data, successful eradication was observed in 96% of patients who took more than 60% of the prescribed medication[128]. Adherence to sequential therapy varies from 81%-98%, while adherence to concomitant therapy has been reported as 78.7%-100% in different studies[129-135]. The lowest adherence is observed in Spain[131], and the highest is observed in Taiwan[135], as seen in Table 1.
| Ref. | Country | Adherence to treatment |
| Wu et al[130] | Taiwan | 98.2% concomitant therapy |
| 95.7% sequential therapy | ||
| Greenberg et al[131] | Latin America (Chile, Colombia, Costa Rica, Honduras, Nicaragua, and Mexico ) | 92.2% overall |
| 93.8% concomitant therapy | ||
| 93% sequential therapy | ||
| 89.9% standard therapy | ||
| McNicholl et al[132] | Spain | 83% concomitant therapy |
| 82% sequential therapy | ||
| Lim et al[133] | South Korea | 96.2% concomitant therapy |
| 95.3% sequential therapy | ||
| Huang et al[134] | Taiwan | 94% concomitant therapy |
| 95.3% sequential therapy | ||
| Hsu et al[135] | Taiwan | 100% concomitant therapy |
| 98% sequential therapy | ||
| 99% standard therapy |
Passive immunization is effective in the prevention and treatment of various infectious diseases[136-138], making it a plausible strategy for the treatment of H. pylori as well. Data from animal studies has supported this concept, together with a previously reported protective effect from breastfeeding[139-141]. It is probable that specific antibodies inhibit the adherence of H. pylori. Clinical studies have reported conflicting data. Some studies have suggested that treatment with bovine antibodies could eradicate or decrease H. pylori colonization density[142,143], while others have failed to demonstrate this effect[144,145]. The first RCT to evaluate the efficacy and safety of specific anti-H. pylori polyclonal bovine IgA antibodies to reduce the intragastric bacterial load and gastritis activity in humans was performed by den Hoed[146]; the authors concluded that the antibody-based oral immunotherapy appears to be safe but ineffective because it did not significantly reduce H. pylori colonization density.
Active immunization against H. pylori infection would reduce the cost and potential complications of eradication therapy and is expected to lower gastric cancer incidence. The development of a vaccine against H. pylori is complicated by the fact that the bacterium is noninvasive and remains strictly luminal without crossing the epithelium. An effective vaccine therefore must induce the appropriate Th memory cells that can be recruited to the mucosal surfaces. An effective vaccine against H. pylori should consist of appropriate bacterial antigens, an effective and safe adjuvant, and the route of delivery should be adequate. Different protocols have been tested using different antigens, adjuvants and application routes.
In studies on animal models using classical immunization protocols, H. pylori lysates or H. pylori proteins were used, and plausible candidate antigens were identified (i.e., urease, catalase, VacA, CagA, NapA, HpaA, AlpA and BabA). Better protection resulted from combinations of antigens[147,148]. Recently, new bacterial antigens have been proposed as candidates for vaccine development, including 20 kD outer membrane lipoprotein Lpp20[149], AhpC (alkyl hydroperoxide reductase)[150] and antioxidant proteins (e.g., superoxide dismutase and catalase)[151].
Some studies that have used attenuated Salmonella strains, which express H. pylori ureA and ureB antigens, as delivery systems have demonstrated significant protection, both with intranasal and oral administration[152,153]. Recently designed Salmonella vector approaches use outer inflammatory protein A[154] for oral therapeutic immunization in addition to CagA, VacA and UreB in the vector[155]. This OipA-Salmonella based approach seems to be effective at both inducing OipA-specific antibodies and reducing H. pylori colonization. Overall, the Salmonella-based approach seems to be successful in animal models.
A polio virus-based vaccination using urease B had both prophylactic and therapeutic efficacy[156].
A multi-epitope approach is the basis of several new vaccine candidates and is described in detail in several studies. Li et al[157] used B and T cell epitopes that were generated by software prediction aimed at inducing both humoral and cellular immune response. Epivac uses proteins consisting of predicted T cell epitopes from HpaA-, UreB- and CagA[158]-inducing serum but unfortunately no mucosal immunity. The induction of mucosal immunity could be an effective H. pylori vaccination protocol. Promising results were obtained in a study using chimeric flagellin consisting of the hypervariable domain of H. pylori FlaA and the C- and N-terminal segments of Escherichia coli (E. coli) flagellin that was designed by Mori[159]. The chimeric flagellin was designed to maintain H. pylori specificity and gain TLR5 activity.
The vaccine in animal models was administered using different routes such as intranasal, oral, intramuscular, subcutaneous, rectal and intraperitoneal. Different adjuvants have been tested in animal models, i.e., cholera toxin (CT) or heat-labile enterotoxin (LT), but the clinical use of these strong mucosal adjuvants is limited in humans because of their toxicity. A possible solution to this problem is to detoxify the adjuvant while maintaining its stimulatory effect[160]. However, these adjuvants have not been used in human clinical trials against H. pylori[161].
In humans, majority of clinical studies have used recombinant urease as an antigen. In humans, clinical trials have tested the ability of experimental vaccines to eradicate existing H. pylori infection or to prevent the colonization of the gastric mucosa after introduction of the bacterium in an experimental challenge[162].
In H. pylori-infected asymptomatic individuals, oral immunization was well tolerated but did not lead to a specific immune response[163], while adding LT induced an immune response[164] and reduced H. pylori colonization[165]. Diarrhea occurred as a consequence of LT toxicity, but limiting the amount of LT was not effective, in that it resolved the side effects of LT but also reduced the immune response. Rectal administration of urease and LT induced a weak immune response[166].
Urease-expressing Salmonella-based delivery vectors did not prove to be effective in humans, which is in opposition to findings in animal models[167,168], because immune reactions were undetectable. Initial data on the use of the urease-expressing Salmonella vaccine strain Ty21a was disappointing because the immunologic response in H. pylori-negative volunteers was weak. T cell memory was observed in very few subjects, and no urease-specific antibodies were detected[168]. However, further investigation revealed that administration of multiple doses of a Salmonella-Ty21a based recombinant vaccine or the use of another recombinant strain that expressed the HP0231 H. pylori antigen resulted in the development of an immune response specific to H. pylori infection[169] and a decrease in the number of bacteria in gastric biopsies, both in vaccinated subjects and in the control group. Attenuated vaccines are well tolerated and might be the correct direction for further research.
There is evidence that efficacy can be improved via multivalent subunit vaccines[170]. A multivalent subunit vaccine consisting of CagA, VacA and NapA[171] was administered intramuscularly to H. pylori-negative volunteers, in whom it induced both humoral and cellular immune responses without side effects. Unfortunately, protection from H. pylori in a clinical setting did not differ between the placebo and the vaccine groups[172].
Recently, CagL protein was proposed as promising candidate for use in a subunit vaccine[173].
The use of strong mucosal adjuvants that have been tested in animal models is limited in humans because of their toxicity (i.e., CT and LT).
In the field of mucosal adjuvants, important results have been published in recent years. Nedrud et al[174] demonstrated that the intranasal administration of a mucosal adjuvant, CTA1-DD (a derivative of the cholera toxin), is safe, effective and protected against a live H. pylori challenge in mice. The use of heat shock proteins (Hsp) as the mucosal adjuvant in a H. pylori vaccine has also been reported[175]. This vaccine was administered via respiratory route and induced systemic and mucosal antibodies; protective immunity was generated with a milder post-immunization inflammation. Nevertheless, sterilizing immunity was not achieved[175]. Promising results were obtained when a nontoxic double mutant of an E. coli toxin (R192G/L211A) (dm2T) was used as the mucosal adjuvant[176]. Altman et al[177] suggested that carbohydrate-based vaccines against H. pylori should use dextran-based conjugates, and Zhang et al[178] suggested recombinant Lactococcus lactis as a vector because the bacterium is already used in dairy products.
A report from Shirai et al[179] indicated that salivary antibody formation is a critical factor for successful vaccination against H. pylori. This was supported by the findings of Ng et al[180], who confirmed increased levels of salivary IgA without an increase in mucin production or cytokine level. Intranasal application in animal models seems to be promising and effective[174]. A multivalent subunit vaccine consisting of CagA, VacA and NapA[172] was administered intramuscularly. The sublingual[176] route appears to be promising, while an oral route of administration has repeatedly been tested with variable effects that depend on the antigen and the adjuvant or vector used[163-165,169,178]. Rectal administration was tested once and the results were somewhat disappointing[166].
To develop an effective vaccine for H. pylori, a better understanding of protective immune responses from data on animal models and a better understanding of host responses is needed. The identification of the underlying mechanisms that prevent a host from clearing the infection could help in the development of an effective vaccine.
The T helper cell response is an important part of the protective immune response because data on animal models suggest that mice lacking antibody molecules, including mucosal immunoglobulin IgA, are well-protected by vaccinations[181-183], while mice with deficient cellular immunity are not protected[182,184]. These data suggest that the T helper cell response is crucial for the induction of protective immunity, even in the absence of other forms of adaptive immunity[161]. This is based on the fact that Th1 or Th17 cell-induced increases in inflammation lead to the development of protective immunity. Therefore, a successful vaccine should induce a strong Th1 or Th17 response. According to Zawahir, possible multiple effector mechanisms that can eradicate H. pylori are not adequately defined. Therefore, vaccine efficacy could be improved through the enhancement of Th1 or Th17 responses to H. pylori[161]. It seems that the host immune response is age dependent, because a weaker Th1 response is observed in children[185].
In isolated cells, the removal of CD25+ T cells before H. pylori stimulation increases the IFNγ response[186]. The results obtained in an animal model revealed that T regulatory cells suppress the active host immune response to H. pylori infection[186-190]. Because the majority of infected individuals do not have an H. pylori-associated disease, the host might not recognize the bacterium as dangerous and may suppress the immune response. Another possible target for improvement of vaccine efficacy is therefore overcoming the host predisposition to reducing immune responses to bacteria. A vaccine should increase the host cellular immune response, as supported by findings in animal models where bacterial load was reduced using IL-17[191] and IL-12[192].
Recent publications by Muhsen et al[193,194] demonstrated higher seroconversion rates to a typhoid vaccine in H. pylori-infected subjects, and the authors also demonstrated that gastric H. pylori-associated inflammation promoted seroconversion. Based on these findings, the concept that active H. pylori infection could be beneficial for the efficacy of other vaccines becomes attractive, and further research is needed in this area.
Timely eradication of H. pylori infection is, at present, the single available evidence-based strategy for reducing H. pylori-related gastric cancer risk, incidence, and subsequent morbidity and mortality. It is mandatory to eradicate the infection before irreversible mucosal damage occurs. Eradication should precede the development of severe atrophic changes.
Future possible strategies include the development of an effective vaccine. It is rather disappointing that although some of the experimentally tested vaccines have shown promising results, there is limited funding and financial support from the pharmaceutical industry. At the moment, another possible strategy is the development of new antimicrobial agents that would be effective for eradication therapy, but these are long-lasting and time-consuming studies. Therefore, at present, it is advisable to insist on patient compliance during eradication therapy because this is the area where improvement is possible.
P- Reviewer: Engin AB, Mach TH S- Editor: Ma YJ L- Editor: A E- Editor: Wang CH
| 1. | Talley NJ, Zinsmeister AR, Weaver A, DiMagno EP, Carpenter HA, Perez-Perez GI, Blaser MJ. Gastric adenocarcinoma and Helicobacter pylori infection. J Natl Cancer Inst. 1991;83:1734-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 245] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 2. | Parsonnet J, Friedman GD, Vandersteen DP, Chang Y, Vogelman JH, Orentreich N, Sibley RK. Helicobacter pylori infection and the risk of gastric carcinoma. N Engl J Med. 1991;325:1127-1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2805] [Cited by in RCA: 2739] [Article Influence: 80.6] [Reference Citation Analysis (0)] |
| 3. | Scott N, Lansdown M, Diament R, Rathbone B, Murday V, Wyatt JI, McMahon M, Dixon MF, Quirke P. Helicobacter gastritis and intestinal metaplasia in a gastric cancer family. Lancet. 1990;335:728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 4. | Marwick C. Helicobacter: new name, new hypothesis involving type of gastric cancer. JAMA. 1990;264:2724, 2727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 5. | Caruso ML, Fucci L. Histological identification of Helicobacter pylori in early and advanced gastric cancer. J Clin Gastroenterol. 1990;12:601-602. [PubMed] |
| 6. | Wang C, Yuan Y, Hunt RH. The association between Helicobacter pylori infection and early gastric cancer: a meta-analysis. Am J Gastroenterol. 2007;102:1789-1798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 101] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 7. | Mbulaiteye SM, Hisada M, El-Omar EM. Helicobacter Pylori associated global gastric cancer burden. Front Biosci (Landmark Ed). 2009;14:1490-1504. [PubMed] |
| 8. | Sokic-Milutinovic A, Popovic D, Alempijevic T, Dragasevic S, Lukic S, Pavlovic-Markovic A. Helicobacter pylori Infection and Gastric Cancer — Is Eradication Enough to Prevent Gastric Cancer. Trends in Helicobacter pylori infection. Rijeka: In Tech 2014; 155-173. [DOI] [Full Text] |
| 9. | Correa P. A human model of gastric carcinogenesis. Cancer Res. 1988;48:3554-3560. [PubMed] |
| 10. | Correa P. Human gastric carcinogenesis: a multistep and multifactorial process--First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992;52:6735-6740. [PubMed] |
| 11. | Correa P, Piazuelo MB. The gastric precancerous cascade. J Dig Dis. 2012;13:2-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | De Luca A, Iaquinto G. Helicobacter pylori and gastric diseases: a dangerous association. Cancer Lett. 2004;213:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 53] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Manente L, Perna A, Buommino E, Altucci L, Lucariello A, Citro G, Baldi A, Iaquinto G, Tufano MA, De Luca A. The Helicobacter pylori’s protein VacA has direct effects on the regulation of cell cycle and apoptosis in gastric epithelial cells. J Cell Physiol. 2008;214:582-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Dunne C, Dolan B, Clyne M. Factors that mediate colonization of the human stomach by Helicobacter pylori. World J Gastroenterol. 2014;20:5610-5624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 79] [Cited by in RCA: 86] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 15. | Turbett GR, Høj PB, Horne R, Mee BJ. Purification and characterization of the urease enzymes of Helicobacter species from humans and animals. Infect Immun. 1992;60:5259-5266. [PubMed] |
| 16. | Josenhans C, Labigne A, Suerbaum S. Comparative ultrastructural and functional studies of Helicobacter pylori and Helicobacter mustelae flagellin mutants: both flagellin subunits, FlaA and FlaB, are necessary for full motility in Helicobacter species. J Bacteriol. 1995;177:3010-3020. [PubMed] |
| 17. | Ilver D, Arnqvist A, Ogren J, Frick IM, Kersulyte D, Incecik ET, Berg DE, Covacci A, Engstrand L, Borén T. Helicobacter pylori adhesin binding fucosylated histo-blood group antigens revealed by retagging. Science. 1998;279:373-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 872] [Cited by in RCA: 842] [Article Influence: 31.2] [Reference Citation Analysis (1)] |
| 18. | Senkovich OA, Yin J, Ekshyyan V, Conant C, Traylor J, Adegboyega P, McGee DJ, Rhoads RE, Slepenkov S, Testerman TL. Helicobacter pylori AlpA and AlpB bind host laminin and influence gastric inflammation in gerbils. Infect Immun. 2011;79:3106-3116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 87] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 19. | Peck B, Ortkamp M, Diehl KD, Hundt E, Knapp B. Conservation, localization and expression of HopZ, a protein involved in adhesion of Helicobacter pylori. Nucleic Acids Res. 1999;27:3325-3333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 132] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 20. | Newton JL, Jordan N, Oliver L, Strugala V, Pearson J, James OF, Allen A. Helicobacter pylori in vivo causes structural changes in the adherent gastric mucus layer but barrier thickness is not compromised. Gut. 1998;43:470-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 21. | Hessey SJ, Spencer J, Wyatt JI, Sobala G, Rathbone BJ, Axon AT, Dixon MF. Bacterial adhesion and disease activity in Helicobacter associated chronic gastritis. Gut. 1990;31:134-138. [PubMed] |
| 22. | Sycuro LK, Wyckoff TJ, Biboy J, Born P, Pincus Z, Vollmer W, Salama NR. Multiple peptidoglycan modification networks modulate Helicobacter pylori’s cell shape, motility, and colonization potential. PLoS Pathog. 2012;8:e1002603. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 116] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 23. | Yamaoka Y, Kato M, Asaka M. Geographic differences in gastric cancer incidence can be explained by differences between Helicobacter pylori strains. Intern Med. 2008;47:1077-1083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 179] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 24. | Yamaoka Y. Mechanisms of disease: Helicobacter pylori virulence factors. Nat Rev Gastroenterol Hepatol. 2010;7:629-641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 500] [Cited by in RCA: 461] [Article Influence: 30.7] [Reference Citation Analysis (1)] |
| 25. | Linz B, Balloux F, Moodley Y, Manica A, Liu H, Roumagnac P, Falush D, Stamer C, Prugnolle F, van der Merwe SW. An African origin for the intimate association between humans and Helicobacter pylori. Nature. 2007;445:915-918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 720] [Cited by in RCA: 637] [Article Influence: 35.4] [Reference Citation Analysis (3)] |
| 26. | Franco AT, Johnston E, Krishna U, Yamaoka Y, Israel DA, Nagy TA, Wroblewski LE, Piazuelo MB, Correa P, Peek RM. Regulation of gastric carcinogenesis by Helicobacter pylori virulence factors. Cancer Res. 2008;68:379-387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 239] [Cited by in RCA: 234] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 27. | Ohnishi N, Yuasa H, Tanaka S, Sawa H, Miura M, Matsui A, Higashi H, Musashi M, Iwabuchi K, Suzuki M. Transgenic expression of Helicobacter pylori CagA induces gastrointestinal and hematopoietic neoplasms in mouse. Proc Natl Acad Sci USA. 2008;105:1003-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 429] [Cited by in RCA: 465] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 28. | Yamaoka Y, Osato MS, Sepulveda AR, Gutierrez O, Figura N, Kim JG, Kodama T, Kashima K, Graham DY. Molecular epidemiology of Helicobacter pylori: separation of H. pylori from East Asian and non-Asian countries. Epidemiol Infect. 2000;124:91-96. [PubMed] |
| 29. | Hayashi T, Senda M, Morohashi H, Higashi H, Horio M, Kashiba Y, Nagase L, Sasaya D, Shimizu T, Venugopalan N. Tertiary structure-function analysis reveals the pathogenic signaling potentiation mechanism of Helicobacter pylori oncogenic effector CagA. Cell Host Microbe. 2012;12:20-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 117] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 30. | Cid TP, Fernández MC, Benito Martínez S, Jones NL. Pathogenesis of Helicobacter pylori infection. Helicobacter. 2013;18 Suppl 1:12-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 31. | Gobert AP, Verriere T, de Sablet T, Peek RM, Chaturvedi R, Wilson KT. Haem oxygenase-1 inhibits phosphorylation of the Helicobacter pylori oncoprotein CagA in gastric epithelial cells. Cell Microbiol. 2013;15:145-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 32. | Wiedemann T, Hofbaur S, Tegtmeyer N, Huber S, Sewald N, Wessler S, Backert S, Rieder G. Helicobacter pylori CagL dependent induction of gastrin expression via a novel αvβ5-integrin-integrin linked kinase signalling complex. Gut. 2012;61:986-996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 89] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 33. | Atherton JC. The pathogenesis of Helicobacter pylori-induced gastro-duodenal diseases. Annu Rev Pathol. 2006;1:63-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 394] [Cited by in RCA: 410] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 34. | Cover TL, Blanke SR. Helicobacter pylori VacA, a paradigm for toxin multifunctionality. Nat Rev Microbiol. 2005;3:320-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 372] [Cited by in RCA: 411] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 35. | Gebert B, Fischer W, Weiss E, Hoffmann R, Haas R. Helicobacter pylori vacuolating cytotoxin inhibits T lymphocyte activation. Science. 2003;301:1099-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 409] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 36. | Cover TL, Blaser MJ. Purification and characterization of the vacuolating toxin from Helicobacter pylori. J Biol Chem. 1992;267:10570-10575. [PubMed] |
| 37. | Atherton JC, Cao P, Peek RM, Tummuru MK, Blaser MJ, Cover TL. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J Biol Chem. 1995;270:17771-17777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1078] [Cited by in RCA: 1108] [Article Influence: 36.9] [Reference Citation Analysis (0)] |
| 38. | Ogiwara H, Graham DY, Yamaoka Y. vacA i-region subtyping. Gastroenterology. 2008;134:1267; author reply 1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 39. | Ogiwara H, Sugimoto M, Ohno T, Vilaichone RK, Mahachai V, Graham DY, Yamaoka Y. Role of deletion located between the intermediate and middle regions of the Helicobacter pylori vacA gene in cases of gastroduodenal diseases. J Clin Microbiol. 2009;47:3493-3500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 67] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 40. | Yamaoka Y, Kita M, Kodama T, Imamura S, Ohno T, Sawai N, Ishimaru A, Imanishi J, Graham DY. Helicobacter pylori infection in mice: Role of outer membrane proteins in colonization and inflammation. Gastroenterology. 2002;123:1992-2004. [PubMed] |
| 41. | Lu H, Hsu PI, Graham DY, Yamaoka Y. Duodenal ulcer promoting gene of Helicobacter pylori. Gastroenterology. 2005;128:833-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 223] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 42. | Douraghi M, Mohammadi M, Oghalaie A, Abdirad A, Mohagheghi MA, Hosseini ME, Zeraati H, Ghasemi A, Esmaieli M, Mohajerani N. dupA as a risk determinant in Helicobacter pylori infection. J Med Microbiol. 2008;57:554-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 43. | Nguyen LT, Uchida T, Tsukamoto Y, Kuroda A, Okimoto T, Kodama M, Murakami K, Fujioka T, Moriyama M. Helicobacter pylori dupA gene is not associated with clinical outcomes in the Japanese population. Clin Microbiol Infect. 2010;16:1264-1269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 44. | Falush D, Wirth T, Linz B, Pritchard JK, Stephens M, Kidd M, Blaser MJ, Graham DY, Vacher S, Perez-Perez GI. Traces of human migrations in Helicobacter pylori populations. Science. 2003;299:1582-1585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 708] [Article Influence: 32.2] [Reference Citation Analysis (1)] |
| 45. | Satoh K, Kimura K, Taniguchi Y, Yoshida Y, Kihira K, Takimoto T, Kawata H, Saifuku K, Ido K, Takemoto T. Distribution of inflammation and atrophy in the stomach of Helicobacter pylori-positive and -negative patients with chronic gastritis. Am J Gastroenterol. 1996;91:963-969. [PubMed] |
| 46. | Shiotani A, Iishi H, Uedo N, Ishiguro S, Tatsuta M, Nakae Y, Kumamoto M, Merchant JL. Evidence that loss of sonic hedgehog is an indicator of Helicobater pylori-induced atrophic gastritis progressing to gastric cancer. Am J Gastroenterol. 2005;100:581-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 47. | Shiotani A, Uedo N, Iishi H, Tatsuta M, Ishiguro S, Nakae Y, Kamada T, Haruma K, Merchant JL. Re-expression of sonic hedgehog and reduction of CDX2 after Helicobacter pylori eradication prior to incomplete intestinal metaplasia. Int J Cancer. 2007;121:1182-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 48. | Silberg DG, Sullivan J, Kang E, Swain GP, Moffett J, Sund NJ, Sackett SD, Kaestner KH. Cdx2 ectopic expression induces gastric intestinal metaplasia in transgenic mice. Gastroenterology. 2002;122:689-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 355] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 49. | Nomura S, Baxter T, Yamaguchi H, Leys C, Vartapetian AB, Fox JG, Lee JR, Wang TC, Goldenring JR. Spasmolytic polypeptide expressing metaplasia to preneoplasia in H. felis-infected mice. Gastroenterology. 2004;127:582-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 118] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 50. | Nozaki K, Ogawa M, Williams JA, Lafleur BJ, Ng V, Drapkin RI, Mills JC, Konieczny SF, Nomura S, Goldenring JR. A molecular signature of gastric metaplasia arising in response to acute parietal cell loss. Gastroenterology. 2008;134:511-522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 147] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 51. | Conteduca V, Sansonno D, Ingravallo G, Marangi S, Russi S, Lauletta G, Dammacco F. Barrett’s esophagus and esophageal cancer: an overview. Int J Oncol. 2012;41:414-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 52. | Fuccio L, Eusebi LH, Bazzoli F. Gastric cancer, Helicobacter pylori infection and other risk factors. World J Gastrointest Oncol. 2010;2:342-347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 53] [Cited by in RCA: 54] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 53. | Correa P, Haenszel W, Cuello C, Tannenbaum S, Archer M. A model for gastric cancer epidemiology. Lancet. 1975;2:58-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 725] [Cited by in RCA: 734] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 54. | Weis VG, Goldenring JR. Current understanding of SPEM and its standing in the preneoplastic process. Gastric Cancer. 2009;12:189-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 107] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 55. | Houghton J, Wang TC. Helicobacter pylori and gastric cancer: a new paradigm for inflammation-associated epithelial cancers. Gastroenterology. 2005;128:1567-1578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 186] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 56. | Shiotani A, Iishi H, Uedo N, Kumamoto M, Nakae Y, Ishiguro S, Tatsuta M, Graham DY. Histologic and serum risk markers for noncardia early gastric cancer. Int J Cancer. 2005;115:463-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 57] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 57. | Varon C, Dubus P, Mazurier F, Asencio C, Chambonnier L, Ferrand J, Giese A, Senant-Dugot N, Carlotti M, Mégraud F. Helicobacter pylori infection recruits bone marrow-derived cells that participate in gastric preneoplasia in mice. Gastroenterology. 2012;142:281-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 104] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 58. | Weis VG, Sousa JF, LaFleur BJ, Nam KT, Weis JA, Finke PE, Ameen NA, Fox JG, Goldenring JR. Heterogeneity in mouse spasmolytic polypeptide-expressing metaplasia lineages identifies markers of metaplastic progression. Gut. 2013;62:1270-1279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 92] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 59. | Nam KT, O’Neal RL, Coffey RJ, Finke PE, Barker N, Goldenring JR. Spasmolytic polypeptide-expressing metaplasia (SPEM) in the gastric oxyntic mucosa does not arise from Lgr5-expressing cells. Gut. 2012;61:1678-1685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 60. | Wong BC, Lam SK, Wong WM, Chen JS, Zheng TT, Feng RE, Lai KC, Hu WH, Yuen ST, Leung SY. Helicobacter pylori eradication to prevent gastric cancer in a high-risk region of China: a randomized controlled trial. JAMA. 2004;291:187-194. [PubMed] |
| 61. | Leung WK, Lin SR, Ching JY, To KF, Ng EK, Chan FK, Lau JY, Sung JJ. Factors predicting progression of gastric intestinal metaplasia: results of a randomised trial on Helicobacter pylori eradication. Gut. 2004;53:1244-1249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 318] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 62. | Correa P, Fontham ET, Bravo JC, Bravo LE, Ruiz B, Zarama G, Realpe JL, Malcom GT, Li D, Johnson WD. Chemoprevention of gastric dysplasia: randomized trial of antioxidant supplements and anti-helicobacter pylori therapy. J Natl Cancer Inst. 2000;92:1881-1888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 497] [Cited by in RCA: 483] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 63. | Mera R, Fontham ET, Bravo LE, Bravo JC, Piazuelo MB, Camargo MC, Correa P. Long term follow up of patients treated for Helicobacter pylori infection. Gut. 2005;54:1536-1540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 254] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 64. | Ma JL, Zhang L, Brown LM, Li JY, Shen L, Pan KF, Liu WD, Hu Y, Han ZX, Crystal-Mansour S. Fifteen-year effects of Helicobacter pylori, garlic, and vitamin treatments on gastric cancer incidence and mortality. J Natl Cancer Inst. 2012;104:488-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 353] [Article Influence: 27.2] [Reference Citation Analysis (1)] |
| 65. | Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, Terao S, Amagai K, Hayashi S, Asaka M. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 876] [Cited by in RCA: 936] [Article Influence: 55.1] [Reference Citation Analysis (0)] |
| 66. | Lee YC, Chen TH, Chiu HM, Shun CT, Chiang H, Liu TY, Wu MS, Lin JT. The benefit of mass eradication of Helicobacter pylori infection: a community-based study of gastric cancer prevention. Gut. 2013;62:676-682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 270] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 67. | Uemura N, Mukai T, Okamoto S, Yamaguchi S, Mashiba H, Taniyama K, Sasaki N, Haruma K, Sumii K, Kajiyama G. Effect of Helicobacter pylori eradication on subsequent development of cancer after endoscopic resection of early gastric cancer. Cancer Epidemiol Biomarkers Prev. 1997;6:639-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 65] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 68. | Asaka M, Kato M, Takahashi S, Fukuda Y, Sugiyama T, Ota H, Uemura N, Murakami K, Satoh K, Sugano K. Guidelines for the management of Helicobacter pylori infection in Japan: 2009 revised edition. Helicobacter. 2010;15:1-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 69. | Bae SE, Jung HY, Kang J, Park YS, Baek S, Jung JH, Choi JY, Kim MY, Ahn JY, Choi KS. Effect of Helicobacter pylori eradication on metachronous recurrence after endoscopic resection of gastric neoplasm. Am J Gastroenterol. 2014;109:60-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 100] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 70. | Malfertheiner P, Megraud F, O’Morain CA, Atherton J, Axon AT, Bazzoli F, Gensini GF, Gisbert JP, Graham DY, Rokkas T. Management of Helicobacter pylori infection--the Maastricht IV/ Florence Consensus Report. Gut. 2012;61:646-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1719] [Cited by in RCA: 1591] [Article Influence: 122.4] [Reference Citation Analysis (5)] |
| 71. | Chey WD, Wong BC; Practice Parameters Committee of the American College of Gastroenterology. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol. 2007;102:1808-1825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 72. | Fock KM, Katelaris P, Sugano K, Ang TL, Hunt R, Talley NJ, Lam SK, Xiao SD, Tan HJ, Wu CY. Second Asia-Pacific Consensus Guidelines for Helicobacter pylori infection. J Gastroenterol Hepatol. 2009;24:1587-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 73. | Graham DY, Shiotani A. The time to eradicate gastric cancer is now. Gut. 2005;54:735-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 92] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 74. | Ohata H, Kitauchi S, Yoshimura N, Mugitani K, Iwane M, Nakamura H, Yoshikawa A, Yanaoka K, Arii K, Tamai H. Progression of chronic atrophic gastritis associated with Helicobacter pylori infection increases risk of gastric cancer. Int J Cancer. 2004;109:138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 354] [Cited by in RCA: 373] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 75. | Yoshida T, Kato J, Inoue I, Yoshimura N, Deguchi H, Mukoubayashi C, Oka M, Watanabe M, Enomoto S, Niwa T. Cancer development based on chronic active gastritis and resulting gastric atrophy as assessed by serum levels of pepsinogen and Helicobacter pylori antibody titer. Int J Cancer. 2014;134:1445-1457. [PubMed] |
| 76. | Yanaoka K, Oka M, Ohata H, Yoshimura N, Deguchi H, Mukoubayashi C, Enomoto S, Inoue I, Iguchi M, Maekita T. Eradication of Helicobacter pylori prevents cancer development in subjects with mild gastric atrophy identified by serum pepsinogen levels. Int J Cancer. 2009;125:2697-2703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 110] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 77. | Sakakibara M, Ando T, Ishiguro K, Maeda O, Watanabe O, Hirayama Y, Morise K, Maeda K, Matsushita M, Furukawa K. Usefulness of Helicobacter pylori eradication for precancerous lesions of the gastric remnant. J Gastroenterol Hepatol. 2014;29 Suppl 4:60-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 78. | Watari J, Das KK, Amenta PS, Tanabe H, Tanaka A, Geng X, Lin JJ, Kohgo Y, Das KM. Effect of eradication of Helicobacter pylori on the histology and cellular phenotype of gastric intestinal metaplasia. Clin Gastroenterol Hepatol. 2008;6:409-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 79. | Shiotani A, Uedo N, Iishi H, Murao T, Kanzaki T, Kimura Y, Kamada T, Kusunoki H, Inoue K, Haruma K. H. pylori eradication did not improve dysregulation of specific oncogenic miRNAs in intestinal metaplastic glands. J Gastroenterol. 2012;47:988-998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 80. | Bazzoli F, Zagari R, Fossi S. Efficacy and tolerability of a short-term low-dose triple therapy for eradication of Helicobacter pylori. Am J Gastroenterol. 1993;104:40A. |
| 81. | Lamouliatte HC, Cayla R, Megraud F, Zerbib F, Stablo M, Bouchard S. Amoxicillin-clarithromycin-omeprazole: the best therapy for Helicobacter pylori infection. Acta Gastroenterol Belg. 1993;56:A14. |
| 82. | Lind T, Veldhuyzen van Zanten S, Unge P, Spiller R, Bayerdörffer E, O’Morain C, Bardhan KD, Bradette M, Chiba N, Wrangstadh M. Eradication of Helicobacter pylori using one-week triple therapies combining omeprazole with two antimicrobials: the MACH I Study. Helicobacter. 1996;1:138-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 83. | Lind T, Mégraud F, Unge P, Bayerdörffer E, O’morain C, Spiller R, Veldhuyzen Van Zanten S, Bardhan KD, Hellblom M, Wrangstadh M. The MACH2 study: role of omeprazole in eradication of Helicobacter pylori with 1-week triple therapies. Gastroenterology. 1999;116:248-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 277] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 84. | Mégraud F. Current recommendations for Helicobacter pylori therapies in a world of evolving resistance. Gut Microbes. 2013;4:541-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 85. | Mégraud F. H pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut. 2004;53:1374-1384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 607] [Cited by in RCA: 681] [Article Influence: 32.4] [Reference Citation Analysis (2)] |
| 86. | Mégraud F, Lehours P. Helicobacter pylori detection and antimicrobial susceptibility testing. Clin Microbiol Rev. 2007;20:280-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 488] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 87. | Versalovic J, Shortridge D, Kibler K, Griffy MV, Beyer J, Flamm RK, Tanaka SK, Graham DY, Go MF. Mutations in 23S rRNA are associated with clarithromycin resistance in Helicobacter pylori. Antimicrob Agents Chemother. 1996;40:477-480. [PubMed] |
| 88. | Glupczynski Y, Mégraud F, Lopez-Brea M, Andersen LP. European multicentre survey of in vitro antimicrobial resistance in Helicobacter pylori. Eur J Clin Microbiol Infect Dis. 2001;20:820-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 194] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 89. | Megraud F, Coenen S, Versporten A, Kist M, Lopez-Brea M, Hirschl AM, Andersen LP, Goossens H, Glupczynski Y. Helicobacter pylori resistance to antibiotics in Europe and its relationship to antibiotic consumption. Gut. 2013;62:34-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 632] [Article Influence: 52.7] [Reference Citation Analysis (3)] |
| 90. | De Francesco V, Giorgio F, Hassan C, Manes G, Vannella L, Panella C, Ierardi E, Zullo A. Worldwide H. pylori antibiotic resistance: a systematic review. J Gastrointestin Liver Dis. 2010;19:409-414. [PubMed] |
| 91. | Bang CS, Baik GH. Attempts to enhance the eradication rate of Helicobacter pylori infection. World J Gastroenterol. 2014;20:5252-5262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 92. | Lee JW, Kim N, Kim JM, Nam RH, Chang H, Kim JY, Shin CM, Park YS, Lee DH, Jung HC. Prevalence of primary and secondary antimicrobial resistance of Helicobacter pylori in Korea from 2003 through 2012. Helicobacter. 2013;18:206-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 177] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 93. | Ribeiro ML, Gerrits MM, Benvengo YH, Berning M, Godoy AP, Kuipers EJ, Mendonça S, van Vliet AH, Pedrazzoli J, Kusters JG. Detection of high-level tetracycline resistance in clinical isolates of Helicobacter pylori using PCR-RFLP. FEMS Immunol Med Microbiol. 2004;40:57-61. [PubMed] |
| 94. | Chung JW, Lee GH, Jeong JY, Lee SM, Jung JH, Choi KD, Song HJ, Jung HY, Kim JH. Resistance of Helicobacter pylori strains to antibiotics in Korea with a focus on fluoroquinolone resistance. J Gastroenterol Hepatol. 2012;27:493-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 95. | Suzuki RB, Almeida CM, Sperança MA. Absence of Helicobacter pylori high tetracycline resistant 16S rDNA AGA926-928TTC genotype in gastric biopsy specimens from dyspeptic patients of a city in the interior of São Paulo, Brazil. BMC Gastroenterol. 2012;12:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 96. | Malfertheiner P, Mégraud F, O’Morain C, Bell D, Bianchi Porro G, Deltenre M, Forman D, Gasbarrini G, Jaup B, Misiewicz JJ. Current European concepts in the management of Helicobacter pylori infection--the Maastricht Consensus Report. The European Helicobacter Pylori Study Group (EHPSG). Eur J Gastroenterol Hepatol. 1997;9:1-2. [PubMed] |
| 97. | Gisbert JP, Calvet X, O’Connor A, Mégraud F, O’Morain CA. Sequential therapy for Helicobacter pylori eradication: a critical review. J Clin Gastroenterol. 2010;44:313-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 143] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 98. | Vaira D, Zullo A, Vakil N, Gatta L, Ricci C, Perna F, Hassan C, Bernabucci V, Tampieri A, Morini S. Sequential therapy versus standard triple-drug therapy for Helicobacter pylori eradication: a randomized trial. Ann Intern Med. 2007;146:556-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 211] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 99. | Essa AS, Kramer JR, Graham DY, Treiber G. Meta-analysis: four-drug, three-antibiotic, non-bismuth-containing “concomitant therapy” versus triple therapy for Helicobacter pylori eradication. Helicobacter. 2009;14:109-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 100. | Gisbert JP, Calvet X. Review article: non-bismuth quadruple (concomitant) therapy for eradication of Helicobater pylori. Aliment Pharmacol Ther. 2011;34:604-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 101. | Kim JS, Park SM, Kim BW. Sequential or concomitant therapy for eradication of Helicobacter pylori infection: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2015;30:1338-1345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 102. | Laine L, Hunt R, El-Zimaity H, Nguyen B, Osato M, Spénard J. Bismuth-based quadruple therapy using a single capsule of bismuth biskalcitrate, metronidazole, and tetracycline given with omeprazole versus omeprazole, amoxicillin, and clarithromycin for eradication of Helicobacter pylori in duodenal ulcer patients: a prospective, randomized, multicenter, North American trial. Am J Gastroenterol. 2003;98:562-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 103. | O’Morain C, Borody T, Farley A, De Boer WA, Dallaire C, Schuman R, Piotrowski J, Fallone CA, Tytgat G, Mégraud F, Spénard J; International multicentrestudy. Efficacy and safety of single-triple capsules of bismuth biskalcitrate, metronidazole and tetracycline, given with omeprazole, for the eradication of Helicobacter pylori: an international multicentre study. Aliment Pharmacol Ther. 2003;17:415-420. [PubMed] [DOI] [Full Text] |
| 104. | Malfertheiner P, Bazzoli F, Delchier JC, Celiñski K, Giguère M, Rivière M, Mégraud F; Pylera Study Group. Helicobacter pylori eradication with a capsule containing bismuth subcitrate potassium, metronidazole, and tetracycline given with omeprazole versus clarithromycin-based triple therapy: a randomised, open-label, non-inferiority, phase 3 trial. Lancet. 2011;377:905-913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 373] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 105. | Hsu PI, Wu DC, Wu JY, Graham DY. Modified sequential Helicobacter pylori therapy: proton pump inhibitor and amoxicillin for 14 days with clarithromycin and metronidazole added as a quadruple (hybrid) therapy for the final 7 days. Helicobacter. 2011;16:139-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 106. | Kim SY, Jung SW, Kim JH, Koo JS, Yim HJ, Park JJ, Chun HJ, Lee SW, Choi JH. Effectiveness of three times daily lansoprazole/amoxicillin dual therapy for Helicobacter pylori infection in Korea. Br J Clin Pharmacol. 2012;73:140-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 107. | Sugimoto M, Furuta T, Shirai N, Kodaira C, Nishino M, Ikuma M, Ishizaki T, Hishida A. Evidence that the degree and duration of acid suppression are related to Helicobacter pylori eradication by triple therapy. Helicobacter. 2007;12:317-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 108. | Yoon JH, Baik GH, Sohn KM, Kim DY, Kim YS, Suk KT, Kim JB, Kim DJ, Kim JB, Shin WG. Trends in the eradication rates of Helicobacter pylori infection for eleven years. World J Gastroenterol. 2012;18:6628-6634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 109. | Berning M, Krasz S, Miehlke S. Should quinolones come first in Helicobacter pylori therapy? Therap Adv Gastroenterol. 2011;4:103-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 110. | Cammarota G, Cianci R, Cannizzaro O, Cuoco L, Pirozzi G, Gasbarrini A, Armuzzi A, Zocco MA, Santarelli L, Arancio F. Efficacy of two one-week rabeprazole/levofloxacin-based triple therapies for Helicobacter pylori infection. Aliment Pharmacol Ther. 2000;14:1339-1343. [PubMed] [DOI] [Full Text] |
| 111. | Perna F, Zullo A, Ricci C, Hassan C, Morini S, Vaira D. Levofloxacin-based triple therapy for Helicobacter pylori re-treatment: role of bacterial resistance. Dig Liver Dis. 2007;39:1001-1005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 85] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 112. | Pilotto A, Franceschi M, Rassu M, Furlan F, Scagnelli M. In vitro activity of rifabutin against strains of Helicobacter pylori resistant to metronidazole and clarithromycin. Am J Gastroenterol. 2000;95:833-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 113. | Gisbert JP, Castro-Fernandez M, Perez-Aisa A, Cosme A, Molina-Infante J, Rodrigo L, Modolell I, Cabriada JL, Gisbert JL, Lamas E. Fourth-line rescue therapy with rifabutin in patients with three Helicobacter pylori eradication failures. Aliment Pharmacol Ther. 2012;35:941-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 48] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 114. | Gisbert JP, Calvet X. Review article: rifabutin in the treatment of refractory Helicobacter pylori infection. Aliment Pharmacol Ther. 2012;35:209-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 115. | Perri F, Festa V, Clemente R, Villani MR, Quitadamo M, Caruso N, Bergoli ML, Andriulli A. Randomized study of two “rescue” therapies for Helicobacter pylori-infected patients after failure of standard triple therapies. Am J Gastroenterol. 2001;96:58-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 116. | Cammarota G, Martino A, Pirozzi G, Cianci R, Branca G, Nista EC, Cazzato A, Cannizzaro O, Miele L, Grieco A. High efficacy of 1-week doxycycline- and amoxicillin-based quadruple regimen in a culture-guided, third-line treatment approach for Helicobacter pylori infection. Aliment Pharmacol Ther. 2004;19:789-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 117. | Savarino V, Zentilin P, Pivari M, Bisso G, Raffaella Mele M, Bilardi C, Borro P, Dulbecco P, Tessieri L, Mansi C. The impact of antibiotic resistance on the efficacy of three 7-day regimens against Helicobacter pylori. Aliment Pharmacol Ther. 2000;14:893-900. [PubMed] [DOI] [Full Text] |
| 118. | Kim JJ, Kim JG, Kwon DH. Mixed-infection of antibiotic susceptible and resistant Helicobacter pylori isolates in a single patient and underestimation of antimicrobial susceptibility testing. Helicobacter. 2003;8:202-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 119. | Padol S, Yuan Y, Thabane M, Padol IT, Hunt RH. The effect of CYP2C19 polymorphisms on H. pylori eradication rate in dual and triple first-line PPI therapies: a meta-analysis. Am J Gastroenterol. 2006;101:1467-1475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 120. | Sheu BS, Fock KM. CYP2C19 genotypes and Helicobacter pylori eradication. J Gastroenterol Hepatol. 2008;23:1163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 121. | Zhang MM, Qian W, Qin YY, He J, Zhou YH. Probiotics in Helicobacter pylori eradication therapy: a systematic review and meta-analysis. World J Gastroenterol. 2015;21:4345-4357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 108] [Cited by in RCA: 115] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 122. | Aiba Y, Suzuki N, Kabir AM, Takagi A, Koga Y. Lactic acid-mediated suppression of Helicobacter pylori by the oral administration of Lactobacillus salivarius as a probiotic in a gnotobiotic murine model. Am J Gastroenterol. 1998;93:2097-2101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 123. | Sgouras D, Maragkoudakis P, Petraki K, Martinez-Gonzalez B, Eriotou E, Michopoulos S, Kalantzopoulos G, Tsakalidou E, Mentis A. In vitro and in vivo inhibition of Helicobacter pylori by Lactobacillus casei strain Shirota. Appl Environ Microbiol. 2004;70:518-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 210] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 124. | Ruggiero P. Use of probiotics in the fight against Helicobacter pylori. World J Gastrointest Pathophysiol. 2014;5:384-391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 57] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 125. | Nseir W, Diab H, Mahamid M, Abu-Elheja O, Samara M, Abid A, Mograbi J. Randomised clinical trial: simvastatin as adjuvant therapy improves significantly the Helicobacter pylori eradication rate--a placebo-controlled study. Aliment Pharmacol Ther. 2012;36:231-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 126. | Gotoh A, Akamatsu T, Shimizu T, Shimodaira K, Kaneko T, Kiyosawa K, Ishida K, Ikeno T, Sugiyama A, Kawakami Y. Additive effect of pronase on the efficacy of eradication therapy against Helicobacter pylori. Helicobacter. 2002;7:183-191. [PubMed] [DOI] [Full Text] |
| 127. | Kimura K, Ido K, Saifuku K, Taniguchi Y, Kihira K, Satoh K, Takimoto T, Yoshida Y. A 1-h topical therapy for the treatment of Helicobacter pylori infection. Am J Gastroenterol. 1995;90:60-63. [PubMed] |
| 128. | Graham DY, Lew GM, Malaty HM, Evans DG, Evans DJ, Klein PD, Alpert LC, Genta RM. Factors influencing the eradication of Helicobacter pylori with triple therapy. Gastroenterology. 1992;102:493-496. [PubMed] |
| 129. | De Francesco V, Hassan C, Ridola L, Giorgio F, Ierardi E, Zullo A. Sequential, concomitant and hybrid first-line therapies for Helicobacter pylori eradication: a prospective randomized study. J Med Microbiol. 2014;63:748-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 130. | Wu DC, Hsu PI, Wu JY, Opekun AR, Kuo CH, Wu IC, Wang SS, Chen A, Hung WC, Graham DY. Sequential and concomitant therapy with four drugs is equally effective for eradication of H pylori infection. Clin Gastroenterol Hepatol. 2010;8:36-41.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 190] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 131. | Greenberg ER, Anderson GL, Morgan DR, Torres J, Chey WD, Bravo LE, Dominguez RL, Ferreccio C, Herrero R, Lazcano-Ponce EC. 14-day triple, 5-day concomitant, and 10-day sequential therapies for Helicobacter pylori infection in seven Latin American sites: a randomised trial. Lancet. 2011;378:507-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 206] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 132. | McNicholl AG, Marin AC, Molina-Infante J, Castro M, Barrio J, Ducons J, Calvet X, de la Coba C, Montoro M, Bory F. Randomised clinical trial comparing sequential and concomitant therapies for Helicobacter pylori eradication in routine clinical practice. Gut. 2014;63:244-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 95] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 133. | Lim JH, Lee DH, Choi C, Lee ST, Kim N, Jeong SH, Kim JW, Hwang JH, Park YS, Lee SH. Clinical outcomes of two-week sequential and concomitant therapies for Helicobacter pylori eradication: a randomized pilot study. Helicobacter. 2013;18:180-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 134. | Huang YK, Wu MC, Wang SS, Kuo CH, Lee YC, Chang LL, Wang TH, Chen YH, Wang WM, Wu DC. Lansoprazole-based sequential and concomitant therapy for the first-line Helicobacter pylori eradication. J Dig Dis. 2012;13:232-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 135. | Hsu PI, Wu DC, Chen WC, Tseng HH, Yu HC, Wang HM, Kao SS, Lai KH, Chen A, Tsay FW. Randomized controlled trial comparing 7-day triple, 10-day sequential, and 7-day concomitant therapies for Helicobacter pylori infection. Antimicrob Agents Chemother. 2014;58:5936-5942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 136. | Davidson GP, Whyte PB, Daniels E, Franklin K, Nunan H, McCloud PI, Moore AG, Moore DJ. Passive immunisation of children with bovine colostrum containing antibodies to human rotavirus. Lancet. 1989;2:709-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 137. | Mitra AK, Mahalanabis D, Ashraf H, Unicomb L, Eeckels R, Tzipori S. Hyperimmune cow colostrum reduces diarrhoea due to rotavirus: a double-blind, controlled clinical trial. Acta Paediatr. 1995;84:996-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 73] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 138. | van Dissel JT, de Groot N, Hensgens CM, Numan S, Kuijper EJ, Veldkamp P, van ‘t Wout J. Bovine antibody-enriched whey to aid in the prevention of a relapse of Clostridium difficile-associated diarrhoea: preclinical and preliminary clinical data. J Med Microbiol. 2005;54:197-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 86] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 139. | Casswall TH, Nilsson HO, Björck L, Sjöstedt S, Xu L, Nord CK, Borén T, Wadström T, Hammarström L. Bovine anti-Helicobacter pylori antibodies for oral immunotherapy. Scand J Gastroenterol. 2002;37:1380-1385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 140. | Marnila P, Rokka S, Rehnberg-Laiho L, Kärkkäinen P, Kosunen TU, Rautelin H, Hänninen ML, Syväoja EL, Korhonen H. Prevention and suppression of Helicobacter felis infection in mice using colostral preparation with specific antibodies. Helicobacter. 2003;8:192-201. [PubMed] [DOI] [Full Text] |
| 141. | Thomas JE, Austin S, Dale A, McClean P, Harding M, Coward WA, Weaver LT. Protection by human milk IgA against Helicobacter pylori infection in infancy. Lancet. 1993;342:121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 76] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 142. | Bitzan MM, Gold BD, Philpott DJ, Huesca M, Sherman PM, Karch H, Lissner R, Lingwood CA, Karmali MA. Inhibition of Helicobacter pylori and Helicobacter mustelae binding to lipid receptors by bovine colostrum. J Infect Dis. 1998;177:955-961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 143. | Oona M. Helicobacter pylori in children with abdominal complaints: Has immuni bovine colostrum some influence on gastritis? Alpe Adria Microbiol J. 1997;6:49-57. |
| 144. | Opekun AR, El-Zaimaity HM, Osato MS, Gilger MA, Malaty HM, Terry M, Headon DR, Graham DY. Novel therapies for Helicobacter pylori infection. Aliment Pharmacol Ther. 1999;13:35-42. [PubMed] [DOI] [Full Text] |
| 145. | Casswall TH, Sarker SA, Albert MJ, Fuchs GJ, Bergström M, Björck L, Hammarström L. Treatment of Helicobacter pylori infection in infants in rural Bangladesh with oral immunoglobulins from hyperimmune bovine colostrum. Aliment Pharmacol Ther. 1998;12:563-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 146. | den Hoed CM, de Vries AC, Mensink PB, Dierikx CM, Suzuki H, Capelle L, van Dekken H, Ouwendijk R, Kuipers EJ. Bovine antibody-based oral immunotherapy for reduction of intragastric Helicobacter pylori colonization: a randomized clinical trial. Can J Gastroenterol. 2011;25:207-213. [PubMed] |
| 147. | Nyström J, Svennerholm AM. Oral immunization with HpaA affords therapeutic protective immunity against H. pylori that is reflected by specific mucosal immune responses. Vaccine. 2007;25:2591-2598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 148. | Sanchez V, Gimenez S, Haensler J, Geoffroy C, Rokbi B, Seguin D, Lissolo L, Harris B, Rizvi F, Kleanthous H. Formulations of single or multiple H. pylori antigens with DC Chol adjuvant induce protection by the systemic route in mice. Optimal prophylactic combinations are different from therapeutic ones. FEMS Immunol Med Microbiol. 2001;30:157-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 149. | Li Y, Jiang Y, Xi Y, Zhang L, Luo J, He D, Zeng S, Ning Y. Identification and characterization of H-2d restricted CD4+ T cell epitopes on Lpp20 of Helicobacter pylori. BMC Immunol. 2012;13:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 150. | O’Riordan AA, Morales VA, Mulligan L, Faheem N, Windle HJ, Kelleher DP. Alkyl hydroperoxide reductase: a candidate Helicobacter pylori vaccine. Vaccine. 2012;30:3876-3884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 151. | Stent A, Every AL, Ng GZ, Chionh YT, Ong LS, Edwards SJ, Sutton P. Helicobacter pylori thiolperoxidase as a protective antigen in single- and multi-component vaccines. Vaccine. 2012;30:7214-7220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 152. | Corthésy-Theulaz IE, Hopkins S, Bachmann D, Saldinger PF, Porta N, Haas R, Zheng-Xin Y, Meyer T, Bouzourène H, Blum AL. Mice are protected from Helicobacter pylori infection by nasal immunization with attenuated Salmonella typhimurium phoPc expressing urease A and B subunits. Infect Immun. 1998;66:581-586. [PubMed] |
| 153. | Gómez-Duarte OG, Lucas B, Yan ZX, Panthel K, Haas R, Meyer TF. Protection of mice against gastric colonization by Helicobacter pylori by single oral dose immunization with attenuated Salmonella typhimurium producing urease subunits A and B. Vaccine. 1998;16:460-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 109] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 154. | Chen J, Lin M, Li N, Lin L, She F. Therapeutic vaccination with Salmonella-delivered codon-optimized outer inflammatory protein DNA vaccine enhances protection in Helicobacter pylori infected mice. Vaccine. 2012;30:5310-5315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 155. | Liu KY, Shi Y, Luo P, Yu S, Chen L, Zhao Z, Mao XH, Guo G, Wu C, Zou QM. Therapeutic efficacy of oral immunization with attenuated Salmonella typhimurium expressing Helicobacter pylori CagA, VacA and UreB fusion proteins in mice model. Vaccine. 2011;29:6679-6685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 156. | Novak MJ, Smythies LE, McPherson SA, Smith PD, Morrow CD. Poliovirus replicons encoding the B subunit of Helicobacter pylori urease elicit a Th1 associated immune response. Vaccine. 1999;17:2384-2391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 157. | Li HX, Mao XH, Shi Y, Ma Y, Wu YN, Zhang WJ, Luo P, Yu S, Zhou WY, Guo Y. Screening and identification of a novel B-cell neutralizing epitope from Helicobacter pylori UreB. Vaccine. 2008;26:6945-6949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 158. | Li HB, Zhang JY, He YF, Chen L, Li B, Liu KY, Yang WC, Zhao Z, Zou QM, Wu C. Systemic immunization with an epitope-based vaccine elicits a Th1-biased response and provides protection against Helicobacter pylori in mice. Vaccine. 2012;31:120-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 159. | Mori J, Vranac T, Smrekar B, Cernilec M, Serbec VČ, Horvat S, Ihan A, Benčina M, Jerala R. Chimeric flagellin as the self-adjuvanting antigen for the activation of immune response against Helicobacter pylori. Vaccine. 2012;30:5856-5863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 160. | Marchetti M, Rossi M, Giannelli V, Giuliani MM, Pizza M, Censini S, Covacci A, Massari P, Pagliaccia C, Manetti R. Protection against Helicobacter pylori infection in mice by intragastric vaccination with H. pylori antigens is achieved using a non-toxic mutant of E. coli heat-labile enterotoxin (LT) as adjuvant. Vaccine. 1998;16:33-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 123] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 161. | Zawahir S, Czinn SJ, Nedrud JG, Blanchard TG. Vaccinating against Helicobacter pylori in the developing world. Gut Microbes. 2013;4:568-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 162. | Czinn SJ, Blanchard T. Vaccinating against Helicobacter pylori infection. Nat Rev Gastroenterol Hepatol. 2011;8:133-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 163. | Kreiss C, Buclin T, Cosma M, Corthésy-Theulaz I, Michetti P. Safety of oral immunisation with recombinant urease in patients with Helicobacter pylori infection. Lancet. 1996;347:1630-1631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 164. | Banerjee S, Medina-Fatimi A, Nichols R, Tendler D, Michetti M, Simon J, Kelly CP, Monath TP, Michetti P. Safety and efficacy of low dose Escherichia coli enterotoxin adjuvant for urease based oral immunisation against Helicobacter pylori in healthy volunteers. Gut. 2002;51:634-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 68] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 165. | Michetti P, Kreiss C, Kotloff KL, Porta N, Blanco JL, Bachmann D, Herranz M, Saldinger PF, Corthésy-Theulaz I, Losonsky G. Oral immunization with urease and Escherichia coli heat-labile enterotoxin is safe and immunogenic in Helicobacter pylori-infected adults. Gastroenterology. 1999;116:804-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 231] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 166. | Kotloff KL, Sztein MB, Wasserman SS, Losonsky GA, DiLorenzo SC, Walker RI. Safety and immunogenicity of oral inactivated whole-cell Helicobacter pylori vaccine with adjuvant among volunteers with or without subclinical infection. Infect Immun. 2001;69:3581-3590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 156] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 167. | Angelakopoulos H, Hohmann EL. Pilot study of phoP/phoQ-deleted Salmonella enterica serovar typhimurium expressing Helicobacter pylori urease in adult volunteers. Infect Immun. 2000;68:2135-2141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 126] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 168. | Bumann D, Metzger WG, Mansouri E, Palme O, Wendland M, Hurwitz R, Haas G, Aebischer T, von Specht BU, Meyer TF. Safety and immunogenicity of live recombinant Salmonella enterica serovar Typhi Ty21a expressing urease A and B from Helicobacter pylori in human volunteers. Vaccine. 2001;20:845-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 169. | Aebischer T, Bumann D, Epple HJ, Metzger W, Schneider T, Cherepnev G, Walduck AK, Kunkel D, Moos V, Loddenkemper C. Correlation of T cell response and bacterial clearance in human volunteers challenged with Helicobacter pylori revealed by randomised controlled vaccination with Ty21a-based Salmonella vaccines. Gut. 2008;57:1065-1072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 170. | Ferrero RL, Thiberge JM, Kansau I, Wuscher N, Huerre M, Labigne A. The GroES homolog of Helicobacter pylori confers protective immunity against mucosal infection in mice. Proc Natl Acad Sci USA. 1995;92:6499-6503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 207] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 171. | Malfertheiner P, Schultze V, Rosenkranz B, Kaufmann SH, Ulrichs T, Novicki D, Norelli F, Contorni M, Peppoloni S, Berti D. Safety and immunogenicity of an intramuscular Helicobacter pylori vaccine in noninfected volunteers: a phase I study. Gastroenterology. 2008;135:787-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 136] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 172. | Malfertheiner P, Selgrad M, Wex T, Bornschein J, Palla E, Del Giudice G, Graham D, Heaton PM. Efficacy of an investigational recombinant antigen based vaccine against a CagA H. pylori infectious challenge in healthy volunteers. Gastroenterology. 2012;142:S-184. |
| 173. | Choudhari SP, Pendleton KP, Ramsey JD, Blanchard TG, Picking WD. A systematic approach toward stabilization of CagL, a protein antigen from Helicobacter pylori that is a candidate subunit vaccine. J Pharm Sci. 2013;102:2508-2519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 174. | Nedrud JG, Bagheri N, Schön K, Xin W, Bergroth H, Eliasson DG, Lycke NY. Subcomponent vaccine based on CTA1-DD adjuvant with incorporated UreB class II peptides stimulates protective Helicobacter pylori immunity. PLoS One. 2013;8:e83321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 175. | Chionh YT, Arulmuruganar A, Venditti E, Ng GZ, Han JX, Entwisle C, Ang CS, Colaco CA, McNulty S, Sutton P. Heat shock protein complex vaccination induces protection against Helicobacter pylori without exogenous adjuvant. Vaccine. 2014;32:2350-2358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 176. | Sjökvist Ottsjö L, Flach CF, Clements J, Holmgren J, Raghavan S. A double mutant heat-labile toxin from Escherichia coli, LT(R192G/L211A), is an effective mucosal adjuvant for vaccination against Helicobacter pylori infection. Infect Immun. 2013;81:1532-1540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 177. | Altman E, Chandan V, Harrison B. The potential of dextran-based glycoconjugates for development of Helicobacter pylori vaccine. Glycoconj J. 2014;31:13-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 178. | Zhang HX, Qiu YY, Zhao YH, Liu XT, Liu M, Yu AL. Immunogenicity of oral vaccination with Lactococcus lactis derived vaccine candidate antigen (UreB) of Helicobacter pylori fused with the human interleukin 2 as adjuvant. Mol Cell Probes. 2014;28:25-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 179. | Shirai Y, Wakatsuki Y, Kusumoto T, Nakata M, Yoshida M, Usui T, Iizuka T, Kita T. Induction and maintenance of immune effector cells in the gastric tissue of mice orally immunized to Helicobacter pylori requires salivary glands. Gastroenterology. 2000;118:749-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 180. | Ng GZ, Chionh YT, Sutton P. Vaccine-mediated protection against Helicobacter pylori is not associated with increased salivary cytokine or mucin expression. Helicobacter. 2014;19:48-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 181. | Sutton P, Wilson J, Kosaka T, Wolowczuk I, Lee A. Therapeutic immunization against Helicobacter pylori infection in the absence of antibodies. Immunol Cell Biol. 2000;78:28-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 182. | Blanchard TG, Czinn SJ, Redline RW, Sigmund N, Harriman G, Nedrud JG. Antibody-independent protective mucosal immunity to gastric helicobacter infection in mice. Cell Immunol. 1999;191:74-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 113] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 183. | Ermak TH, Giannasca PJ, Nichols R, Myers GA, Nedrud J, Weltzin R, Lee CK, Kleanthous H, Monath TP. Immunization of mice with urease vaccine affords protection against Helicobacter pylori infection in the absence of antibodies and is mediated by MHC class II-restricted responses. J Exp Med. 1998;188:2277-2288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 329] [Cited by in RCA: 321] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 184. | Pappo J, Torrey D, Castriotta L, Savinainen A, Kabok Z, Ibraghimov A. Helicobacter pylori infection in immunized mice lacking major histocompatibility complex class I and class II functions. Infect Immun. 1999;67:337-341. [PubMed] |
| 185. | Freire de Melo F, Rocha GA, Rocha AM, Teixeira KN, Pedroso SH, Pereira Junior JB, Fonseca de Castro LP, Cabral MM, Carvalho SD, Bittencourt PF. Th1 immune response to H. pylori infection varies according to the age of the patients and influences the gastric inflammatory patterns. Int J Med Microbiol. 2014;304:300-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 186. | Harris PR, Wright SW, Serrano C, Riera F, Duarte I, Torres J, Peña A, Rollán A, Viviani P, Guiraldes E. Helicobacter pylori gastritis in children is associated with a regulatory T-cell response. Gastroenterology. 2008;134:491-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 185] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 187. | Raghavan S, Fredriksson M, Svennerholm AM, Holmgren J, Suri-Payer E. Absence of CD4+CD25+ regulatory T cells is associated with a loss of regulation leading to increased pathology in Helicobacter pylori-infected mice. Clin Exp Immunol. 2003;132:393-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 188. | Rad R, Brenner L, Bauer S, Schwendy S, Layland L, da Costa CP, Reindl W, Dossumbekova A, Friedrich M, Saur D. CD25+/Foxp3+ T cells regulate gastric inflammation and Helicobacter pylori colonization in vivo. Gastroenterology. 2006;131:525-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 206] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 189. | Arnold IC, Lee JY, Amieva MR, Roers A, Flavell RA, Sparwasser T, Müller A. Tolerance rather than immunity protects from Helicobacter pylori-induced gastric preneoplasia. Gastroenterology. 2011;140:199-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 234] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 190. | Anderson KM, Czinn SJ, Redline RW, Blanchard TG. Induction of CTLA-4-mediated anergy contributes to persistent colonization in the murine model of gastric Helicobacter pylori infection. J Immunol. 2006;176:5306-5313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 61] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 191. | Ding H, Nedrud JG, Blanchard TG, Zagorski BM, Li G, Shiu J, Xu J, Czinn SJ. Th1-mediated immunity against Helicobacter pylori can compensate for lack of Th17 cells and can protect mice in the absence of immunization. PLoS One. 2013;8:e69384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 192. | Velin D, Favre L, Bernasconi E, Bachmann D, Pythoud C, Saiji E, Bouzourene H, Michetti P. Interleukin-17 is a critical mediator of vaccine-induced reduction of Helicobacter infection in the mouse model. Gastroenterology. 2009;136:2237-2246.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 111] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 193. | Muhsen K, Pasetti MF, Reymann MK, Graham DY, Levine MM. Helicobacter pylori infection affects immune responses following vaccination of typhoid-naive U.S. adults with attenuated Salmonella typhi oral vaccine CVD 908-htrA. J Infect Dis. 2014;209:1452-1458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 194. | Muhsen K, Lagos R, Reymann MK, Graham DY, Pasetti MF, Levine MM. Age-dependent association among Helicobacter pylori infection, serum pepsinogen levels and immune response of children to live oral cholera vaccine CVD 103-HgR. PLoS One. 2014;9:e83999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |