Published online Sep 14, 2015. doi: 10.3748/wjg.v21.i34.9863
Peer-review started: April 4, 2015
First decision: April 23, 2015
Revised: May 15, 2015
Accepted: July 18, 2015
Article in press: July 18, 2015
Published online: September 14, 2015
Processing time: 164 Days and 1 Hours
Gastric cancer (GC) is the fourth most common cancer and the third leading cause of cancer mortality worldwide. MicroRNAs (miRNAs) and long non-coding RNAs (lncRNAs) are the most popular non-coding RNAs in cancer research. To date, the roles of miRNAs and lncRNAs have been extensively studied in GC, suggesting that miRNAs and lncRNAs represent a vital component of tumor biology. Furthermore, circulating miRNAs and lncRNAs are found to be dysregulated in patients with GC compared with healthy individuals. Circulating miRNAs and lncRNAs may function as promising biomarkers to improve the early detection of GC. Multiple possibilities for miRNA secretion have been elucidated, including active secretion by microvesicles, exosomes, apoptotic bodies, high-density lipoproteins and protein complexes as well as passive leakage from cells. However, the mechanism underlying lncRNA secretion and the functions of circulating miRNAs and lncRNAs have not been fully illuminated. Concurrently, to standardize results of global investigations of circulating miRNAs and lncRNAs biomarker studies, several recommendations for pre-analytic considerations are put forward. In this review, we summarize the known circulating miRNAs and lncRNAs for GC diagnosis. The possible mechanism of miRNA and lncRNA secretion as well as methodologies for identification of circulating miRNAs and lncRNAs are also discussed. The topics covered here highlight new insights into GC diagnosis and screening.
Core tip: MicroRNAs (miRNAs) and long non-coding RNAs (lncRNAs) are the most popular non-coding RNAs in cancer research. The roles of miRNAs and lncRNAs have been extensively studied in gastric cancer (GC). Concurrently, circulating miRNAs and lncRNAs may function as promising biomarkers to improve the early detection of GC. In this review, we summarize the known circulating miRNAs and lncRNAs for GC diagnosis. The possible mechanism of miRNA and lncRNA secretion as well as methodologies for identification of circulating miRNAs and lncRNAs are also discussed. The topics covered here highlight new insights into GC diagnosis and screening.
- Citation: Huang YK, Yu JC. Circulating microRNAs and long non-coding RNAs in gastric cancer diagnosis: An update and review. World J Gastroenterol 2015; 21(34): 9863-9886
- URL: https://www.wjgnet.com/1007-9327/full/v21/i34/9863.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i34.9863
Gastric cancer (GC) was the fourth most common cancer and the third leading cause of cancer mortality worldwide in 2012[1]. To date, the development of diagnostic methods and surgical techniques has remarkably improved the prognosis of patients with GC[2]. However, for advanced GC, the 5-year survival rate is 30%-50%, and the most common forms of recurrence are lymph node metastasis, hematogenous spread and peritoneal dissemination. The prognosis for individuals with advanced disease remains poor[3]. Early-stage diagnosis of GC potentially reduces the mortality of this disease. Many non-invasive methods for GC detection are available, such as serum pepsinogen and MG-7. However, the sensitivity and specificity of these methods remain unsatisfactory. Endoscopic screening for GC is currently the most reliable diagnostic tool, but its cost and invasive nature have limited its use. Therefore, novel, non-invasive, cost-effective and highly sensitive biomarkers are urgently needed to improve GC detection.
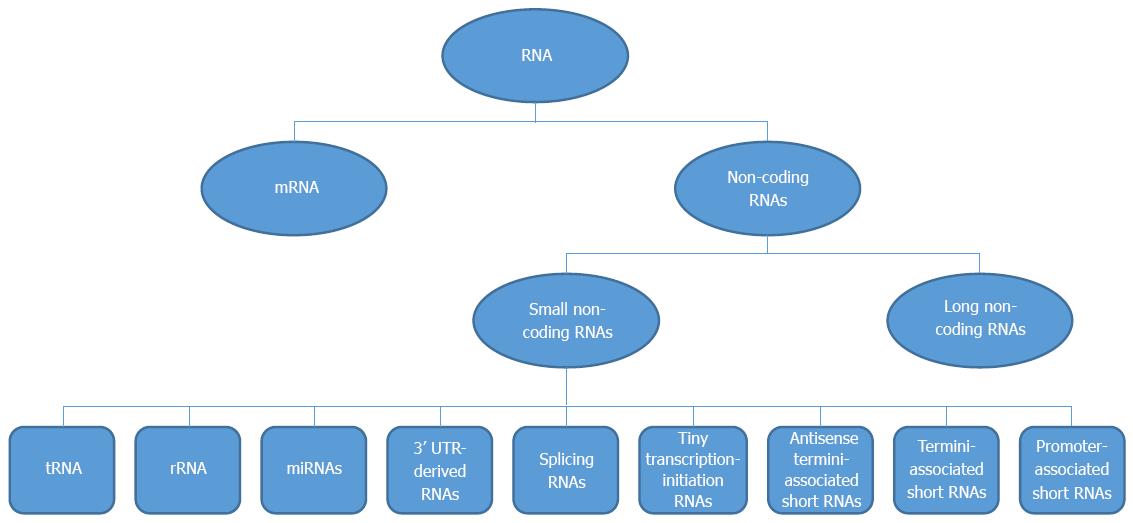
RNA transcripts include mRNA and non-coding RNAs; non-coding RNAs are defined as RNA transcripts that do not encode a protein. Non-coding RNAs are divided into two primary categories: small non-coding RNAs (< 200 nt) and long non-coding RNAs (lncRNAs; > 200 nt) (Figure 1). Some small non-coding RNAs are housekeeping RNAs, such as rRNA and tRNA, which are essential for cell physiology, while others, such as microRNAs (miRNAs), splicing RNAs, promoter-associated short RNAs, tiny transcription-initiation RNAs, termini-associated short RNAs, antisense termini-associated short RNAs and 3’-untranslated region (UTR)-derived RNAs, are associated with protein-coding gene regulation.
To date, miRNAs are the most extensively studied small non-coding RNAs. miRNAs are endogenous, single-stranded, non-coding, small RNAs of 21-22 nucleotides that are involved in regulating gene expression by incorporating into the RNA-induced silencing complex (RISC) and preferential binding to specific sequences in the 3’-UTR of their target mRNAs to suppress translation or to induce mRNA degradation[4]. Since their discovery in 1993, accumulating evidence has suggested that ectopic expression of miRNAs is responsible for a variety of biological processes including embryonic development and carcinogenesis[5-8]. Ectopic expression of miRNAs involved in tumorigenesis has been well described in most tumor types. Up-regulation, down-regulation or silencing of specific miRNAs has been described previously in the carcinogenesis of GC. A series of miRNAs have been identified to be down-regulated and act as tumor suppressors in GC by restraining oncogene expression or interfering with pathways that regulate cellular proliferation. Conversely, a number of miRNAs have been detected that are up-regulated and that may exhibit oncogenic functions in GC by targeting tumor suppressor genes.
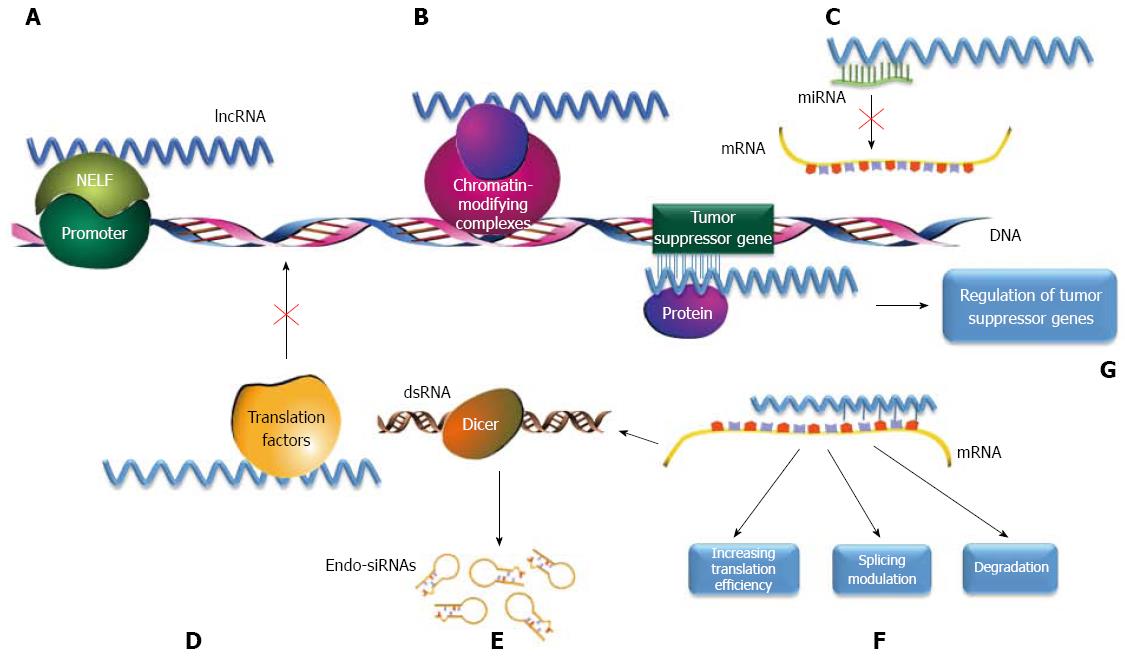
lncRNAs have also gained increasing popularity in the current research climate. lncRNAs are defined as RNA transcripts of more than 200 nucleotides in length with no protein-coding capability and are sometimes referred to as lincRNAs for long intergenic non-coding RNAs. lncRNAs play crucial roles in controlling gene expression during various physiological processes. Emerging evidence has suggested that ectopic lncRNA expression constitutes an important component of tumorigenesis and is involved in angiogenesis and cell proliferation, migration, and apoptosis[9-11]. lncRNAs exhibit a wide spectrum of biological functions and may work in conjunction with mRNAs and miRNAs to serve the following functions (Figure 2): (1) serve as enhancer RNAs (eRNAs) to compete with nascent mRNA for negative elongation factor (NELF) complex binding at the target promoter for transcriptional regulation[12]; (2) serve as a scaffolding base for the coordination of epigenetic or chromatin-modifying complexes[13]; (3) directly modulate tumor suppressor signaling by either transcriptional regulation of tumor suppressor genes or mediation of tumor suppressor target gene activation[14,15]; (4) serve as molecular sponges for miRNAs[16]; (5) gene expression regulation by direct lncRNA-mRNA interactions[17]; (6) function as a decoy for miRNA or translation factors to regulate gene expression[16]; and (7) hybridize with their corresponding spliced mRNAs to form dsRNAs, which are cleaved by Dicer to generate endogenous small interfering RNAs (endo-siRNAs) to modulate gene expression[18,19]. Growing evidence suggests that lncRNA dysregulation correlates with GC tumorigenesis, metastasis and prognosis. H19, HOTAIR, CCAT1, GHET1, CDKN2B LSINCT-5, CUDR, LINC00152 and MALAT1 can exhibit oncogenic functions in GC, whereas GAS5, MEG3, BM742401 and FER1L4 may function as tumor suppressors; their critical effects on tumor growth may be mediated through competition for targeted miRNAs or positive regulation of epithelial-mesenchymal transition[20].
miRNAs that are protected by exosomes and other microparticles have been detected in plasma, serum, urine, saliva, and milk[21-24]. lncRNAs are also detected in multiple body fluids. Circulating miRNAs and lncRNAs meet the basic conditions for utility as biomarkers that may be measured repeatedly and non-invasively in a wide array of cancer types to distinguish patients from health individuals[8,25-27]. Circulating miRNAs and lncRNAs may play an important role in the diagnosis of various types of cancer. In this review, the diagnostic applications of circulating miRNAs and lncRNAs in GC are assessed.
miR-1, miR-20a, miR-27a, miR-34a, and miR-423-5p: Liu et al[28] used Solexa sequencing to identify a profile of five serum miRNAs, i.e., miR-1, miR-20a, miR-27a, miR-34 and miR-423-5p, as a biomarker for GC detection, and the expression levels of these five miRNAs were associated with tumor stage. Receiver operating characteristic (ROC) curves were used to evaluate the diagnostic value of the five-serum miRNA signature with a sensitivity of 80% and specificity of 81%. The areas under the ROC curves (AUCs) of serum miRNAs in sets I and II were 0.879 (95%CI: 0.822-0.936) and 0.831 (95%CI: 0.767-0.898), respectively, which were higher than those of carcinoembryonic antigen (CEA; 0.503; 95%CI: 0.411-0.595) and carbohydrate antigen 19-9 (CA19-9; 0.600; 95%CI: 0.507-0.684). The expression levels of the five serum miRNAs differed significantly in the GC patients at different tumor progression stages. miR-20a, miR-27a and miR-423-5p were up-regulated in GC tissues, miR-20a promoted GC progression by directly targeting early growth response 2[29], miR-27a suppression inhibited GC cell growth by targeting prohibitin[30], and miRNA-423-5p might represent a promising therapeutic target for GC because of its regulation of tumor cell proliferation and invasion by targeting trefoil factor 1[31]. Taken together, current research suggests that the onco-miRNA roles of miR-20a, miR-27a and miR-423-5p in the tumorigenesis of GC are important. miR-1 has been suggested to function as a tumor suppressor miRNA because of its down-regulated expression level in GC tissues, and the ability of miR-1 to target MET has been validated[32]. However, changes in miR-34a expression were inconsistent in GC tissues[33,34]. In 2009, miR-34a was first identified to be significantly up-regulated in GC[34]. Subsequently, Kim et al[33] used a custom-designed Agilent microarray to determine that miR-34a was down-regulated in GC tissues, and PDGFR and the transcription factor Yin Yang 1 (YY1) were identified as targets of the miR-34 family[35,36]. However, the function of miR-34a in GC may be fully elucidated in the future. Moreover, these five miRNAs have also been reported to play a potent role in the development of other types of cancer, such as bladder cancer, squamous cell lung carcinoma, hepatocarcinoma, colorectal carcinoma, prostate cancer and glioblastoma, indicating the potential utility of these five miRNAs in cancer therapy in addition to their diagnostic value[37-41].
miR-200c: Valladares-Ayerbes et al[42] conducted a study that included 52 GC patients and 15 controls to explore the potential of using circulating miR-200c in whole blood as a biomarker for GC diagnosis and prognosis. In this study, miR-200c exhibited the highest expression level of all miR-200 cluster members in the GC cell lines MKN-45 and OE-19. Therefore, miR-200c was chosen for clinical validation. A high diagnostic value of circulating miR-200c was observed, with an AUC of 0.715, sensitivity of 65.4% and specificity of 100%, indicating that miR-200c may represent a novel biomarker for GC detection. The Cox multivariate regression model was used to further explore the relationship between circulating miR-200c and clinical outcomes of GC patients, and the result revealed that a high expression level of circulating miR-200c was an independent prognostic factor for progression free survival (PFS) and overall survival (OS) in patients with GC. In GC tissues, miR-200c was significantly down-regulated, which inhibited the migration and invasion of GC cells by decreasing ZEB1/2 expression[43]. In addition to GC, serum miR-200c was identified as an emerging metastasis-predictive and prognostic biomarker for patients with colorectal cancer[44].
miR-223, miR-16 and miR-100: Wang et al[45] demonstrated that serum miR-223, miR-16 and miR-100 play crucial roles in GC screening. The expression levels of serum miR-223, miR-16 and miR-100 were significantly elevated in the GC group relative to the controls, and these levels were associated with the tumor-node-metastasis (TNM) stage, tumor size, metastatic status and differentiation grade in GC patients. AUCs of 0.85 for miR-223, 0.90 for miR-16, and 0.71 for miR-100 were observed for GC detection. The sensitivity and specificity of miR-223 were 81% and 78%, respectively. miR-16 exhibited a sensitivity of 79% and specificity of 78%, whereas the maximal sensitivity and specificity of miR-100 were 0.71 and 0.58, respectively, implying that serum miR-223, miR-16 and miR-100 may serve as biomarker candidates for GC diagnosis. miR-223 and miR-100 were reported to exhibit elevated expression levels in GC tissues. miR-223 was identified to function as an oncogene that promotes GC invasion and metastasis by targeting FBXW7/hCdc4 and EPB41L3, which are tumor suppressor genes[46,47]. Yang et al[48] reported that silencing miR-100 expression initiated a robust apoptotic response in human GC cells and that HS3ST2 may be a target gene of miR-100, indicating its oncogenic role in GC. The expression level of miR-16 in GC tissues is inconsistent; Oh et al[49] observed that miR-16 was up-regulated in GC, whereas Xia et al[50] and Li et al[51] reported that miR-16 was down-regulated in GC tissue and GC cells. The reasons for this difference have not yet been determined.
miR-16, miR-25, miR-92a, miR-451 and miR-486-5p: Zhu et al[52] conducted a four-phase study to identify and evaluate a cluster of circulating miRNAs as novel biomarkers for GC diagnosis. During the discovery phase, the authors obtained the circulating miRNA expression profiles of gastric non-cardia adenocarcinoma (GNCA) patients and controls using TLDA chips with pooled samples, and differentially expressed circulating miRNAs (miR-16, miR-25, miR-92a, miR-451 and miR-486-5p) were validated by real-time quantitative polymerase chain reaction (qRT-PCR). During the independent validation phase, the five identified miRNAs were examined in a larger independent sample and found to be consistent with the previous phase, suggesting that these five plasma miRNAs might serve as promising biomarkers for early GNCA detection with a sensitivity of 72.9%, specificity of 89.2% and AUC of 0.812. In addition, miR-16, miR-92a and miR-25, but not miR-486-5p or miR-451, were identified as secretory miRNAs in BGC823 and MGC803 cell lines. miR-25 and miR-92a were also found to be up-regulated in GC tissues. miR-25 promotes GC cell motility and proliferation by targeting LATS2 and RECK[53,54], and miR-92a promotes GC cell growth by targeting E2F1 and HIPK1[55], supporting the onco-miRNA roles of miR-25 and miR-92a in GC. miR-16, miR-451 and miR-486-5p have exhibited inconsistent expression levels in GC tissues in different research studies[33,51,56,57] potentially due to different methodologies for miRNA detection. In addition to GC, aberrant expression of miR-16, miR-92, miR-25, miR-451 and miR-486-5p was also found to participate in the development of many other types of cancer[58-69].
miR-16 and miR-93 were also identified by Song et al[70] as suitable reference genes for serum miRNA analysis in GC patients and healthy controls. In their study, Song et al[71] identified seven candidate reference miRNAs from the sera of GC patients. miR-16 and miR-93 were identified as the top two most stable miRNAs serving as reference genes for serum miRNA analysis for GC detection. miR-93 was significantly up-regulated in GC specimens compared with healthy controls, and the prognostic value of miR-93 was observed in resectable gastric adenocarcinomas[71]. In addition to GC, circulating miR-93 also exhibits potential diagnostic value for breast cancer detection[72]. Du et al[73] demonstrated that miR-93 functioned as a potent suppressor of DAB2, which was identified as a tumor suppressor gene in multiple cancer types, and miR-93 overexpression correlated with poor survival of patients with lung cancer, indicating the vital role of miR-93 in cancer development and progression and in GC diagnosis.
miR-421: Zhou et al[74] reported that circulating miR-421 in mononuclear cells (MNCs) could serve as a distinctive biomarker for circulating tumor cells (CTCs). In their study, the authors used reverse transcription-polymerase chain reaction (RT-PCR) to evaluate the diagnostic value of miR-421 in peripheral blood for GC detection, and the sensitivity, specificity and AUC of miR-421 were 94.12%, 62.50% and 0.773, respectively. In addition, the authors observed that the miR-421 inhibitor repressed the proliferation and viability of GC cells in vivo and in vitro, indicating that miR-421 might act an onco-miRNA in GC. Wu et al[75] also validated the high diagnostic efficiency of serum miR-421 in a large sample set composed of 90 GC patients and 90 controls with a maximal sensitivity of 95.5%, maximal specificity of 89.1% and maximal AUC of 0.821, which was superior to cancer antigen 125 (CA125) and CEA for GC detection. The expression level of miR-421 in GC tissues was up-regulated, which was consistent with the circulating levels. Furthermore, one of the targets of miR-421 was identified as caspase-3[76], which is important for apoptosis. In addition, miR421 was identified to play a crucial role in the tumorigenesis and development of many other cancer types, including breast cancer, nasopharyngeal carcinoma, and SKX squamous cell carcinoma[76-80].
miR-378: Liu et al[81] used microarrays to obtain serum genome-wide miRNA profiles and identified serum miR-378 as a biomarker for the early detection of GC. In their article, three miRNAs (miR-187, miR-371-5p, and miR-378) were detected in all 30 samples and exhibited significantly higher expression in GC serum than in controls or in the colorectal cancer group. Further, the authors performed qRT-PCR in another independent set composed of 40 GC serum samples and 41 healthy controls to verify the diagnostic value of the three miRNAs in discriminating GC patients from controls. Subsequently, ROC curve and multivariate logistic regression analyses indicated that miR-378 was a potential biomarker for GC detection with a sensitivity of 87.5%, specificity of 70.73% and AUC of 0.861, whereas serum miR-187 and miR-371-5p could not improve GC diagnosis. Serum miR-378 expression levels did not differ significantly among the different TNM stages. Meanwhile, the expression level of miR-378 in GC tissues was down-regulated relative to normal tissues, indicating that miR-378 might serve as a tumor suppressor miRNA in GC. MAPK1 may be the target of miR-378 to assist miR-378 in inhibiting the progression of human GC cells[82]. However, miR-378 was overexpressed in ovarian cancer cells and tumor specimens vs normal ovarian epithelial cells[83], indicating that the expression pattern of miR-378 was dependent on the type of cancer. Moreover, miR-378 was identified as an ideal biomarker candidate for breast cancer, renal cell carcinoma, non-small cell lung cancer and colorectal cancer[84-86].
miR-199a-3p: Li et al[87] conducted a study to explore the expression levels of tumor-associated miRNAs in the plasma of patients with early GC. The authors determined that the expression of miR-199a-3p in the plasma of 30 early GC patients was significantly higher relative to healthy controls and patients with gastric precancerous diseases (GPDs). Meanwhile, the expression level of plasma miR-199a-3p in the postoperative patients was significantly reduced compared with the preoperative patients. Moreover, the AUC of plasma miR-199a-3p for early GC diagnosis was 0.818, which was significantly higher than that of combined tumor markers such as CEA, CA125, CA724, CA199, CA242 and CA50. The sensitivity, specificity, and accuracy of plasma miR-199a-3p for early GC diagnosis were 76%, 74%, and 75%, respectively. Peng et al[88] suggested that miR-199a-3p was down-regulated in gastric carcinoma tissues and regulated human GC cell proliferation by targeting the mTOR signaling pathway. However, Wang et al[89] demonstrated that miR-199a-3p was significantly up-regulated in GC tissues and cell lines and that its oncogenic activity in GC might involve the direct targeting of ZHX1. The expression level of miR-199a-3p in GC tissues is inconsistent, and the controversy may be resolved by stricter and more reliable studies.
miR-106a and miR-17: Zhou et al[90] performed a study to identify circulating miRNAs to detect circulating tumor cells in peripheral blood for GC diagnosis. The authors observed that the expression levels of miR-106a and miR-17 in the circulation were higher in GC patients compared with controls. Ninety GC patients and 27 controls were recruited to evaluate the diagnostic value of miR-106a and miR-17 in peripheral blood, and the result revealed that the AUC, sensitivity and specificity of miR-106a were 0.684, 48.15% and 90.24%, respectively, whereas the AUC, sensitivity and specificity of miR-17 were 0.743, 51.85% and 92.68%, respectively. Additionally, the AUC, sensitivity and specificity of the combination of miR-106a and miR-17 were 0.741, 62.96%, 80.49%, respectively. Zeng et al[91] suggested that the expression level of serum miR-17 was significantly reduced in both GC and benign gastric disease patients compared with healthy controls. The AUC for serum miR-17 to discriminate GC patients from controls was 0.879, with a sensitivity of 90.6% and specificity of 57.5%. To discriminate benign gastric disease patients from controls, the AUC, sensitivity and specificity of serum miR-17 were 0.725, 62.9% and 81.2%, respectively. In GC tissues, miR-17 was overexpressed to function as an onco-miRNA to promote cell proliferation, and UBE2C and FBXO31 were identified as the targets of miR-17[92,93]. Concurrently, miR-106a overexpression was observed in GC tissues, and miR-106a might play an oncogenic role in GC by targeting TIMP2 and FAS[94,95]. miR-106a in gastric juice was also demonstrated to exhibit potential diagnostic value for GC detection, with a sensitivity of 73.8%, specificity of 89.3%, and AUC of 0.871[96], which was consistent with the diagnostic value of circulating levels. In addition to GC, miR-106a and miR-17 were also found to be widely used as promising diagnostic or prognostic biomarkers for various types of cancer such as hepatocellular carcinoma, non-small cell lung cancer, prostate cancer, breast cancer and nasopharyngeal carcinoma[97-100].
miR-103, miR-107, miR-194 and miR-210: Rotkrua et al[101] established a mouse line to recapitulate human diffuse-type GC (DGC) morphologically and molecularly by E-cadherin/p53 double conditional knockout (DCKO). Subsequently, the authors employed miRNA microarrays to screen candidate miRNAs in individual mouse samples at different ages. miR-103, miR-107, miR-194, miR-210 and miR-291b-5p were identified as DGC-related miRNAs. To evaluate the accuracy of the microarray data, the authors measured the expression levels of these five miRNAs in serum samples using TaqMan qRT-PCR. miR-107, miR-103, miR-194 and miR-210 exhibited significantly increased expression levels in the sera of 6-12-mo-old DCKO mice (early-stage DGC) with histologically proven intramucosal DGC compared with the control group, suggesting that the four miRNAs had the potential to identify DGC at an early stage. In 12-mo-old DCKO mice (advanced-stage DGC), the expression levels of miR-107, miR-103 and miR-194 in the sera were significantly increased compared with controls, whereas the expression level of miR-210 was unchanged, indicating that miR-210 might be an inappropriate biomarker of advanced DGC. To discriminate DGC cases from controls, miR-103 exhibited a sensitivity of 81.8%, specificity of 95.7% and AUC of 0.881; miR-107 exhibited a sensitivity of 90.9%, specificity of 95.7% and AUC of 0.909; and miR-194 exhibited a sensitivity of 90.9%, specificity of 95.7% and AUC of 0.925. For differentiating early early-stage DGC cases from controls, miR-210 exhibited a sensitivity of 72.7%, specificity of 87.0% and AUC of 0.846. The expression levels of miR-107, miR-103 and miR-210 in human GC tissues were inconsistent. miR-107 expression was up-regulated in eight studies and down-regulated in one study[33,34,47,49,51,57,102-104], miR-103 expression was up-regulated in seven studies and down-regulated in one study[33,34,47,49,57,102-104], and miR-210 expression was up-regulated in one study and down-regulated in two studies[30,33,47]. However, these differences may be due to the different detection methods and small patient samples. miR-194 was significantly down-regulated in GC tissues, indicating its tumor suppressor role in GC development[105]. miR-103, miR-107, miR-194 and miR-210 were also found to modulate the proliferation, migration or multidrug resistance of GC cells[106-109]. Moreover, these four miRNAs were identified to play crucial roles in the oncogenesis of many other malignant tumor types, such as cervical cancer, colorectal cancer, pancreatic cancer, non-small cell lung carcinoma and endometrial cancer[69,109-113].
miR-18a: Tsujiura et al[114] assessed plasma miR-18 as a biomarker in patients with GC. The authors determined that the expression level of miR-18 increased significantly in GC tissues compared with normal gastric tissues. The diagnostic value of circulating miR-18 in plasma was evaluated, and the results indicated that the AUC of miR-18 was 0.8059, with a sensitivity of 0.846 and specificity of 0.692. Concurrently, plasma miR-18 exhibited significantly lower expression levels in postoperative patients compared with preoperative patients, indicating that circulating miR-18 in the plasma might function as a biomarker for GC screening and tumor dynamic monitoring. Su et al[115] also validated the diagnostic value of plasma miR-18a for GC detection and reported AUC, sensitivity and specificity values for discriminating GC patients from healthy controls to be 0.907, 80.5% and 84.6%, respectively. STAT3 was identified as the target of miR-18a to promote its onco-miRNA activity in GC[116]. In addition to GC, plasma/serum miR-18a also exhibited potential as a next-generation biomarker in the screening of other types of cancer, such as pancreatic cancer (AUC = 0.936), esophageal cancer (AUC = 0.944), hepatocellular cancer (AUC = 0.881) and colorectal cancer with a clinically satisfactory degree of specificity and sensitivity[117].
miR-223 and miR-21: Li et al[118] reported that the expression levels of miR-223 and miR-21 in the plasma were significantly higher in GC patients than in healthy controls, whereas miR-218 was significantly lower. The study they conducted comprised of 60 GC patients and 60 healthy controls in the validation stage. The combination of miR-223, miR-21 and miR-218 yielded an AUC of 0.9531 with a sensitivity of 84.29% and specificity of 92.86% in differentiating GC patients from healthy controls. The use of a single miRNA in the plasma as biomarker for GC detection produced sensitivity values of 84.29% and 74.29%; specificity values of 88.57% and 75.71%; and AUC values of 0.9089 and 0.7944 for miR-223 and miR-21, respectively. In addition to this, the expression level of miR-223 in the plasma was associated with Helicobacter pylori (H. pylori) infection status. miR-21 overexpression in the circulation also correlates with lymph node metastasis, tumor size and TNM stage in GC patients[119,120]. A meta-analysis suggested that the AUC of plasma miR-21 as a biomarker for GC diagnosis was 0.80, with a pooled sensitivity of 66.5% and pooled specificity of 83.1%[121]. Concurrently, miR-21 in gastric juice exhibited high diagnostic values for GC detection, with a sensitivity of 85.7%, specificity of 97.8%, and AUC of 0.969[96], consistent with those in the circulation. The expression levels of miR-223 and miR-21 in GC tissues were elevated, consistent with their expression levels in the circulation[46,122,123]. miR-223 functions as an oncogene in GC by targeting FBXW7/hCdc4, EPB41L3 and Stathmin1[46,47,124], and miR-21 promotes tumor proliferation and invasion or confers cisplatin resistance in GC by targeting NF-κB and PTEN[125-127]. In addition to GC, the two miRNAs were found to participate in tumorigenesis and enhance chemosensitivity in various other types of cancer, including prostate cancer, esophageal cancer, osteosarcoma, hepatocellular carcinoma and lung adenocarcinoma[128-132].
miR-21 and miR-106b: Shiotani et al[133] investigated the expression levels of serum miRNAs in high-risk GC patients before and after H. pylori eradication. The study recruited 87 early-stage, non-cardia, intestinal-type GC patients without lymph node metastasis and 114 sex- and age-matched healthy controls. Serum miR-106b was significantly up-regulated in the GC group before and after H. pylori eradication compared with healthy controls, whereas serum miR-21 also manifested higher expression levels in the GC group relative to healthy controls immediately after H. pylori eradication. ROC analyses revealed that the AUC, sensitivity and specificity values of miR-106b for early GC screening were 0.61, 55.6% and 70.3% before H. pylori eradication, respectively. For early GC screening after H. pylori eradication, the AUC values of miR-106b and miR-21 were 0.70 and 0.72; the sensitivity values of miR-106b, miR-21 and the combination of miR-106b and miR-21 were 75.8%, 58.6% and 69.0%; and the specificity values of miR-106b, miR-21 and the combination of miR-106b and miR-21 were 51.4%, 51.4% and 69.4%, respectively, superior to serum pepsinogen I and II. Zeng et al[91] also suggested that serum miR-106b yielded an AUC of 0.856, sensitivity of 75.0% and specificity of 92.5% for discriminating GC patients from healthy controls; an AUC of 0.700, sensitivity of 87.2% and specificity of 45.0% for discriminating GC patients from benign gastric disease patients; and an AUC of 0.739, sensitivity of 75.0% and specificity of 68.7% for discriminating benign gastric disease patients from healthy controls. In addition, the expression level of the miR-106b~25 cluster (miR-106b, miR-93 and miR-25) in the plasma was suggestive of significant correlation with TNM stage, tumor size and Bormann type of GC[134]. Recent observations also demonstrated that miR-106b is up-regulated in GC tissues to function as an oncogene by targeting PTEN, p21, E2F5 and p57[135-137].
miR-221, miR-376c and miR-744: Song et al[138] conducted a multi-stage, nested case-control study from two large cohorts to explore the potential use of serum miRNAs in GC detection. The study was composed of four stepwise phases, and 82 GC patients, 46 dysplasia patients and 128 controls with superficial gastritis or atrophic gastritis were recruited. The results indicated that a combination of miR-221, miR-376c and miR-744 was an efficient biomarker for GC diagnosis with a sensitivity of 82.4% and specificity of 58.8%. For early GC detection, the three-miRNA panel exhibited an interesting diagnostic value with a sensitivity of 73.3%. Concurrently, the three-miRNA panel could also discriminate dysplasia from controls with a sensitivity of 56.5% and specificity of 47.8%. Furthermore, a significant positive correlation between the expression levels of two serum miRNAs (miR-221, miR-376c) and the poor differentiation of GC was observed. In GC tissues and cell lines, miR-221 is up-regulated to function as an oncogene to promote GC invasion and metastasis by targeting PTEN, and its expression level is associated with the TNM stage and venous invasion[139,140]. However, miR-376c and miR-744 have not been well elucidated in GC. In addition to GC, serum miR-376c exhibits potential in the detection of breast cancer, and miR-221, miR-376c and miR-744 play pivotal roles in the development of multiple other types of cancer[141].
let-7a: Wang et al[142] demonstrated that let-7a exhibited a lower expression level in the sera of GC patients than in healthy individuals, corresponding with lower let-7a expression levels in gastric adenocarcinoma tissues relative to peritumoral tissues. The expression levels of let-7a in serum samples and tumor tissues were significantly correlated, indicating that let-7a could be a diagnostic biomarker of GC. In that study, in vitro experiments revealed that let-7a might function as a tumor suppressor miRNA in GC by suppressing CCR7. Tsujiura et al[143] found that the expression levels of miR-17-5p, miR-106b, miR-106a and miR-21 were significantly elevated in GC patients compared with controls, whereas let-7a exhibited a lower plasma concentration in GC patients compared with controls. To evaluate the diagnostic value of these circulating miRNAs for GC detection, large-scale validation including 69 GC patients and 30 controls was performed, and the result indicated that the ratio of miR-106a/let-7a exhibited a sensitivity of 85.5%, specificity of 80.0% and maximum AUC of 0.879. Tchernitsa et al[102] used miRNA microarrays and reported that let-7a was up-regulated in GC compared with non-neoplastic tissue, which was inconsistent with the research by Wang et al[142]. However, the result of Oleg Tchernitsa’s study revealed marginal significance with a P-value of 0.0474, which should be validated in a larger scale study. In addition to GC, let-7a also exhibits a potent role in regulating cell proliferation or chemosensitivity in other types of cancer such as hepatocellular carcinoma, breast cancer, lung cancer, bladder cancer, colorectal cancer, nasopharyngeal carcinoma, prostate cancer, esophageal cancer, pancreatic cancer and ovarian cancer[144-154], indicating its extensive and universal function in tumor development.
miR-375: Zhang et al[155] conducted a study to explore the miRNA profile of distal gastric adenocarcinoma (DGAC) and identified miR-375 as significantly down-regulated in DGAC tissue and GC cell lines (MGC-803, BGC-823, and SGC-7901) relative to normal gastric tissues and cell lines (GES-1). Concurrently, in an independent set composed of 20 patients with preoperative DGAC and 20 age-matched healthy individuals, serum miR-375 was identified as a biomarker candidate for discriminating patients with DGAC from healthy controls. The AUC of serum miR-375 was 0.835, with a sensitivity of 80% and specificity of 85%. Previous studies of miR-375 in GC had emphasized that miR-375 was frequently down-regulated in GC tissues. JAK2, ERBB2, STAT3 and p53 were identified as targets of miR-375, suggesting its tumor suppressor role in GC[156-160]. In addition to GC, miR-375 also exhibits a tumor suppressor miRNA role in many other malignant tumors, and circulating miR-375 is a potential diagnostic marker for other malignant tumors such as hepatocellular carcinoma[161], prostate cancer[162], non-small-cell lung cancer[163] and pancreatic cancer[164].
miR-218: The expression levels of miR-218 was identified to be significantly lower in GC patients than in healthy controls by Li et al[118] as well. As a single biomarker in plasma for GC detection, miR-218 produced a sensitivity of 94.29%, specificity of 44.29%, and AUC of 0.7432. In addition to this, miR-218 in the sera of patients with GC was also up-regulated after cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC), and its expression level in the sera correlated with GC tumor grade, stage and metastasis[165,166], suggesting that circulating miR-218 might exhibit a prognostic value for GC patients. miR-218 expression in GC tissues was reduced significantly, and miR-218 overexpression notably inhibited GC cell proliferation, indicating the tumor suppressor role of miR-218 in GC. NF-κB, Robo1 and VOPP1 were identified as targets of miR-218[122,167,168]. In addition to GC, miR-218 were found to participate in tumorigenesis in various other types of cancer[131,169-171].
Cai et al[172] demonstrated that plasma miR-106b, miR-20a and miR-221 might serve as potential non-invasive biomarkers for GC detection with AUCs of 0.7733, 0.8593 and 0.7960, respectively. Tsai et al[173] suggested that serum miR-196a is significantly down-regulated in postoperative GC patients compared with preoperative GC patients and that the expression level of serum miR-196a is significantly increased when patients experience recurrence, indicating that miR-196a might serve as a promising biomarker for GC detection and disease relapse. Plasma miR-192 and miR-122 were also identified to exhibit potential in distinguishing GC patients with distant metastasis (GC/DM) from those with non-distant metastasis (GC/NDM), and the results indicated that the AUCs for discriminating GC/DM from GC/NDM were 0.732 for miR-192 and 0.808 for miR-122 and that the AUCs to discriminate GC patients from healthy controls were 0.818 for miR-192 and 0.815 for miR-122[174]. Gorur et al[175] stated that plasma miR-195-5p was most significantly down-regulated, with more than 13-fold changes in the expression in GC patients compared with control groups, suggesting its potential as a biomarker for GC detection. Moreover, the expression levels of serum miR-146a, miR-21 and miR-148a were associated with GC pN stage, and the combination of these three miRNAs could represent a biomarker candidate for differentiating LN metastasis of GC patients from LN-negative GC patients with an AUC of 0.764[120]. Wang et al[176] demonstrated that the expression levels of plasma miR-17-5p and miR-20a were significantly associated with the differentiation status and TNM stage of GC and that the elevated miR-17-5p/20a levels in plasma indicated poorer overall survival of patients with GC.
The characteristics of circulating miRNAs in GC detection are summarized in Table 1. The AUC, sensitivity and specificity ranges were 0.684-0.9531, 48.15%-94.29%, and 44.29%-95.7%, respectively. The combination of miR-223, miR-21 and miR-218 exhibited the highest diagnostic value for GC detection in humans, regardless of tumor site, tumor stage and pathological type, with an AUC of 0.9531, sensitivity of 84.29% and specificity of 92.86%. Among these circulating miRNAs, the combination of miR-16, miR-25, miR-92a, miR-451 and miR-486-5p; miR-210; the combination of miR-221, miR-376c and miR-744; miR-199a-3p; and the combination of miR-21 and miR-106b were observed to exert effective diagnostic values for the detection of early GC with an AUC range of 0.7-0.846, sensitivity range of 72%-75.8% and specificity range of 51.4%-89.2%. Of all of these combinations, the combination of miR-16, miR-25, miR-92a, miR-451 and miR-486-5p is indicative of the highest diagnostic value, with an AUC of 0.89, sensitivity of 84.1% and specificity of 90.8%. For non-cardiac early GC detection specifically, miRNA-199a-3p demonstrates the highest AUC of 0.818, with a sensitivity of 74% and specificity of 75%. Improvement of the early diagnosis of GC is the best way to improve the prognosis of patients with GC. For non-cardiac GC diagnosis, the combination of miR-16, miR-25, miR-92a, miR-451 and miR-486-5p produces the highest AUC value of 0.89, with a sensitivity of 84.1% and specificity of 90.8%. Concurrently, miR-375 has been identified to have potential in discriminating distal gastric adenocarcinoma (DGAC) patients from healthy individuals, with an AUC of 0.835, sensitivity of 80% and specificity of 85%. However, few studies have been conducted to examine the diagnostic value of circulating miRNAs for cardiac GC detection. miR-103, miR-107, miR-194 and miR-210 are utilized for the detection of diffuse-type GC (DGC), with a sensitivity range of 72.7%-90.9%, specificity range of 87.0%-95.7% and AUC range of 0.846-0.925. Of these miRNAs, miR-194 exhibits the highest diagnostic value with an AUC of 0.925, sensitivity of 90.9% and specificity of 95.7%; however, these miRNAs were detected in mouse lines rather than in humans, and the sample size was small (5 advanced-stage DGCs, 6 early-stage DGCs and 18 healthy controls)[101]. Therefore, these results must be validated in GC patients on a larger scale. As presented in Table 2, miR-1, miR-17-5p, miR-16, miR-18a, miR-20a, miR-21, miR-25, miR-27a, miR-34a, miR-92a, miR-100, miR-103, miR-106a, miR-106b, miR-107, miR-146a, miR-148a, miR-192, miR-194, miR-196a, miR-199a-3p, miR-200c, miR-210, miR-221, miR-223, miR-376c, miR-378, miR-423-5p, miR-421, miR-451, miR-486-5p, miR-744 and miR-93 are up-regulated in the circulation of GC patients; on the contrary, miR-195-5p, let-7a, miR-218, miR-375 and miR-122 are down-regulated in the circulation. Among these miRNAs, miR-17-5p, miR-18a, miR-20a, miR-21, miR-25, miR-27a, miR-92a, miR-100, miR-106a, miR-106b, miR-192, miR-221, miR-223, miR-423-5p, miR-421, and miR-93 are overexpressed in GC tissues, and these data suggest that this group of miRNAs represent onco-miRNAs in GC. In contrast, the expression levels of miR-1, miR-146a, miR-148a, miR-194, miR-200c, miR-218, miR-375 and miR-378 are down-regulated in GC tissues, indicating that these miRNAs act as tumor suppressor miRNAs. miR-16, miR-34a, miR-103, miR-107, miR-196a, miR-199a-3p, miR-210, miR-451, miR-486-5p and let-7a manifest inconsistent expression in GC tissues among different research studies, which may be the result of different inclusion or exclusion criteria of study subjects, different accuracies of profiling platforms, different protocols for specimen collection and processing, previous cytotoxic treatments, underestimated hypoxia and infection and tumor heterogeneity. However, the reason for these differences should be elucidated sufficiently in the future. In all the circulating miRNAs, the expression levels of miR-1, miR-146a, miR-148a, miR-194, miR-200c, miR-218, miR-375 and miR-378 in GC tissues are inconsistent with their circulating levels, suggesting that these miRNAs are actively secreted into the circulation or by other mechanisms rather than by being passively released. Although the function and origin of circulating miRNAs in patients with GC have not been systematically clarified, they have exhibited superior diagnostic value as biomarkers for GC detection compared with conventional tumor markers such as CA199, CA125, CA724, CA242, CA50 and CEA. To standardize the results of global investigations of circulating miRNA biomarker studies, formal recommendations for pre-analytic considerations have been put forward[177]. Circulating miRNAs have gained in popularity as biomarker candidates for GC diagnosis and screening.
| Circulating miRNA | Species | Samples | Sensitivity | Specificity | AUC | Population |
| miR-1, miR-20a, miR-27a, miR-34a and miR-423-5p[28] | Human | Serum | 80% of miR-1 + miR-20a + miR-27a + miR-34a + miR-423-5p | 81% of miR-1 + miR-20a + miR-27a + miR-34a + miR-423-5p | 0.879 of miR-1 + miR-20a + miR-27a + miR-34a + miR-423-5p | 82 GC patients and 63 healthy controls |
| miR-16, miR-25, miR-92a, miR-451 and miR-486-5p[52] | Human | Plasma | 84.1% of miR-16 + miR-25 + miR-92a + miR-451 + miR-486-5p | 90.8% of miR-16 + miR-25 + miR-92a + miR-451 + miR-486-5p | 0.89 of miR-16 + miR-25 + miR-92a + miR-451 + miR-486-5p | 106 non-cardiac early GC patients and 160 healthy controls |
| miR-223, miR-16 and miR-100[45] | Human | Serum | 81% of miR-223, 79% of miR-16 and 71% of miR-100 | 78% of miR-223, 78% of miR-16 and 58% of miR-100 | 0.85 of miR-223, 0.9 of miR-16 and 0.71 of miR-100 | 50 GC patients and 47 healthy controls |
| miR-18a[114] | Human | Plasma | 84.6% | 69.2% | 0.8059 | 104 GC patients and 65 healthy controls |
| miR-18a[115] | Human | Plasma | 80.5% | 84.6% | 0.907 | 82 GC patients and 65 healthy controls |
| miR-106a and miR-17[90] | Human | Whole blood | 62.96% of miR-106a + miR-17, 48.15% of miR-106a and 51.85% of miR-17 | 80.49% of miR-106a + miR-17, 90.24% of miR-106a and 92.68% of miR-17 | 0.741 of miR-106a + miR-17, 0.684 of miR-106a and 0.74 of miR-17 | 90 GC patients and 27 healthy controls |
| The ratio of miR-106a/let-7a[90] | Human | Serum | 85.5% | 80% | 0.879 | 69 GC patients and 30 healthy controls |
| miR-103, miR-107, miR-194 and miR-210[101] | Mouse | Serum | 81.8% of miR-103, 90.9% of miR-107, 90.9% of miR-194 and 72.7% of miR-210 only for early-stage DGC | 95.7% of miR-103, 95.7% of miR-107, 95.7% of miR-194 and 87.0% of miR-210 only for early-stage DGC | 0.881 of miR-103, 0.909 of miR-107, 0.925 of miR-194 and 0.846 of miR-210 only for early-stage DGC | 5 advanced-stage DGC patients, 6 early-stage DGC patients and 18 healthy controls |
| miR-223, miR-21 and miR-218[118] | Human | Plasma | 84.29% of miR-223 + miR-21 + miR-218, 84.29% of miR-223, 74.29% of miR-21 and 94.29% of miR-218 | 92.86% of miR-223 + miR-21 + miR-218, 88.57% of miR-223, 75.71% of miR-21 and 44.29% of miR-218 | 0.9531 of miR-223 + miR-21 + miR-218, 0.9089 of miR-223, 0.7944 of miR-21 and 0.7432 of miR-218 | 60 GC patients and 60 healthy controls |
| miR-21[121] | Human | Plasma | 66.5% | 83.1% | 0.80 | 251 GC patients and 184 controls |
| miR-21 and miR-106b[133] | Human | Serum | 69% of miR-21 + miR-106b, 58.6% of miR-21 and 75.8% of miR-106b | 69.4% of miR-21 + miR-106b, 86.1% of miR-21 and 51.4% of miR-106b | 0.72 of miR-21 and 0.7 of miR-106b | 87 non-cardiac early GC patients and 114 healthy controls |
| miR-17 and miR-106b[91] | Human | Serum | 83.3% of miR-17 + miR-106b, 80.6% of miR-17 and 75.0% of miR-106b | 87.5% of miR-17 + miR-106b, 87.5% of miR-17 and 92.5% of miR-106b | 0.913 of miR-17 + miR-106b, 0.879 of miR-17 and 0.856 of miR-106b | 40 GC patients, 32 BGD patients and 36 healthy controls |
| miR-106b, miR-20a and miR-221[172] | Human | Plasma | Null | Null | 0.773 of miR-106b, 0.8593 of miR-20a and 0.7960 of miR-221 | 60 GC patients and 60 healthy controls |
| miR-122 and miR-192[174] | Human | Plasma | Null | Null | 0.808 of miR-122 for discriminating GC/DM from GC/NDM and 0.815 of miR-122 for discriminating GC/DM patients from healthy controls; 0.732 of miR-192 for discriminating GC/DM from GC/NDM and 0.818 of miR-192 for discriminating GC/DM patients from healthy controls | 36 GC/DM patients, 36 GC/NDM patients and 36 healthy controls |
| miR-195-5p[175] | Human | Plasma | Null | Null | Null | 20 GC patients and 190 healthy controls |
| miR-196a[173] | Human | Serum | Null | Null | Null | 20 Pre- and post-operative GC patients |
| miRNA-199a-3p[87] | Human | Plasma | 74% | 75% | 0.818 | 80 early GC patients, 20 patients with gastric precancerous |
| diseases and 70 healthy controls | ||||||
| miR-200c[42] | Human | Whole blood | 65.4% | 100% | 0.715 | 52 GC patients and 15 healthy controls |
| miR-221, miR-376c and miR-744[138] | Human | Serum | 82.4% of miR-221 + miR-376c + miR-744 | 58.8% of miR-221 + miR-376c + miR-744 | 0.7 of miR-221, 0.74 of miR-744 and 0.71 of miR-376c | 38 advanced GC patients, 30 early GC patients, 46 DYS patients and 128 healthy controls |
| 73.3% of miR-221 + miR-376c + miR-744 for early GC detection | ||||||
| miR-375[155] | Human | Serum | 80% | 85% | 0.835 | 20 DGAC patients and 20 healthy controls |
| miR-378[81] | Human | Serum | 87.5% | 70.73% | 0.861 | 40 GC patients and 41 healthy controls |
| miR-421[74] | Human | Whole blood | 94.12% | 62.5% | 0.773 | 40 GC patients and 17 healthy controls |
| miR-421[75] | Human | Whole blood | 95.5% | 89.1% | 0.821 | 90 GC patients and 90 healthy controls |
| Circulating miRNA | Expression in the circulation | Expression in tissue | Validated target genes |
| miR-1 | Up-regulation | Down-regulation | MET, Coronin1C, API-5, Slug, PIK3CA, NRF2, CCND2, CXCR4, SDF-1α, TAGLN2 |
| miR-17-5p | Up-regulation | Up-regulation | YES1, STAT3, SMAD7, SOCS6, Beclin1, P21, P130, PCAF, TP53INP1, VDUP1, HBP1, AIB1, |
| miR-16 | Up-regulation | Inconsistent direction of expression | ABCF2, ABHD10, ACP2, ACTR1A, ACVR2A, ADSS, ALG3, ANAPC16, ARHGDIA, ARL2, ASXL2, ATG9A, AURKB, BCL2, BMI1, BRCA1, C14orf109, C17orf80, C2orf43, C2orf74, C4orf27, C9orf114, C9orf167, C9orf89, CA12, CACNA2D1, CADM1, CAPRIN1, CARD8, CCDC109A, CCDC111, CCDC76, CCND1, CCND3, CCNE1, CCNT2, CDC14B, CDK5RAP1, CDK6, CENPJ, CEP63, CFL2, CHORDC1, CHUK, CREBL2, CRHBP, CSHL1, DNAJB4, ECHDC1, EGFR, EIF4E, EPT1, FAM122C, FAM69A, FGF2, FNDC3B, GALNT7, GFM1, GFPT1, GNL3L, GOLGA5, GOLPH3L, GPAM, GSTM4, GTF2H1, H3F3B, HACE1, HARS, HARS2, HBXIP, HDHD2 |
| HERC6, HMGA1, HMOX1, HRSP12, HSDL2, HSP90B1, HSPA1A, IFRD1, IFRD2, IGF2R, IPO4, ITGA2, JUN, KCNN4, KPNA3, LAMC1, LAMTOR2, LAMTOR3, LUZP1, LYPLA2, MCL1, MLLT11, MMS19, MRPL20, MSH2, MYB, NAA15, NAA25, NAPG, NIPAL2, NOB1, NOTCH2, NPR3, NT5DC1, OMA1, OSGEPL1, PAFAH1B2, PANX1, PDCD4, PDCD6IP, PHKB, PHLDB2, PISD, PLK1, PMS1, PNN, PNPLA6, PPIF, PPM1D, PPP2R5C, PRIM1, PSAT1, PTCD3, PTGS2, PURA, PWWP2A, RAB21, RAB30, RAB9B, RAD51C, RARS, RFT1, RHOT1, RNASEL, RTN4, SEC24A, SERPINE2, SHOC2, SKAP2, SLC12A2, SLC16A3, SLC25A22, SLC35A1, SLC35B3, SLC38A1, SLC38A5, SLC7A1, SPTLC1, SQSTM1, SRPR, SRPRB, TIA1, TMEM109, TMEM43, TNFSF9, TOMM34, TP53, TPI1, TPM3, TPPP3, TXN2, UBE2S, UBE2V1, UBE4A, UGDH, UGP2, UTP15, VEGFA, VPS45, VTI1B, WIPF1, WNT3A, WT1, YIF1B, ZNF384, ZNF559, ZNF622, NFKB1, ZYX | |||
| miR-17 | Inconsistent direction of expression | Up-regulation | APP, BCL2, BCL2L11, BMPR2, CCL1, CCND1, CCND2, CDKN1A, DNAJC27, E2F1, E2F3, FBXO31, GPR137B, ICAM1, JAK1, MAP3K12, MAPK9, MEF2D, MUC17, MYC, NCOA3, NPAT, OBFC2A, PKD2, PTEN, PTPRO, RB1, RBL1, RBL2, RUNX1, SELE, SMAD4, TGFBR2, THBS1, TNFSF12, VEGFA, WEE1, YES1, ZNFX1, PTEN |
| miR-18a | Up-regulation | Up-regulation | CCNL1, CSRNP3, CTGF, ESR1, HSF2, NCOA3, NR3C1, PTEN, SMAD4, SPPL3, TGFBR2, THRA, TNFSF11, TSC22D3, ATM |
| miR-20a | Up-regulation | Up-regulation | APP, BCL2, BMPR2, BNIP2, CCND1, CCND2, CDKN1A, E2F1, E2F3, HIF1A, MAP3K12, MAPK9, MEF2D, MUC17, MYC, NRAS, PTEN, RB1, RBL1, RBL2, RUNX1, SMAD4, TGFBR2, THBS1, VEGFA, WEE1, EGLN3, IRF2, KIT |
| miR-21 | Up-regulation | Up-regulation | ANKRD46, APAF1, BASP1, BCL2, BMPR2, BTG2, CCR1, CDC25A, CDK2AP1, DAXX, DERL1, E2F1, E2F2, EGFR, EIF2S1, EIF4A2, ERBB2, FMOD, HNRNPK, ICAM1, IL1BISCU, JAG1, JMY, LRRFIP1, MARCKS, MEF2C, MSH2, MSH6, MTAP, MYC, NCAPG, NCOA3, NFIB, PCBP1, PDCD4, PDHA2, PLAT, PLOD3, PPIF, PTEN, PTX3, RASA1, RASGRP1, RECK, REST, RHOB, RPS7, RTN4, SERPINB5, SOX5, SPATS2L, SPRY2, TGFBI, TGFBR2, TGFBR3, TGIF1, TIAM1, TIMP3, TM9SF3, TNFAIP3, TOPORS, TP53BP2, TP63, TPM1, WFS1, WIBG, ANP32A, CCL20, DOCK4, DOCK5, DOCK7, DUSP10, NFKB1, PIAS3, PPARA, SMARCA4, SP1, DAPK1, EZH2, MAP2K3, MAPK8, PLXNB1, POU4F2, PTEN, SRGAP1, ING4, XBP1, ACVR2B, WNK1 |
| miR-25 | Up-regulation | Up-regulation | BCL2L11, CDKN1C, KAT2B, KLF4, PRMT5, TP53, CCL26, CDH1, WDR4 |
| miR-27a | Up-regulation | Up-regulation | FOXO1, HIPK2, MYT1, PHB, SP1, SP3, SP4, SPRY2, THRB, ZBTB10, APC, FBXW7, IGF1, MMP13, PAX3, WEE1 |
| miR-34a | Up-regulation | Inconsistent direction of expression | AXIN2, BCL2, BIRC3, CCND1, CCND3, CCNE2, CD44, CDC25A, CDC25C, CDK4, CDK6, CEBPB, DLL1, E2F1, E2F3, E2F5, EMP1, FOXP1, GRM7, HMGA2, HNF4A, IFNB1, JAG1, MAGEA12, MAGEA2, MAGEA3, MAGEA6, MAP2K1, MAP3K9, MDM4, MET, MYB, MYC, MYCN, NOTCH1, NOTCH2, PEA15, SIRT1, SPI1, TNFRSF6B, VAMP2, VEGFA, WNT1, YY1, ZAP70, EPHA5, FOSL1, IMPA1, IMPDH2, MET, NANOG, PDGFRA, SOX2, STX1A, SYT1, ULBP2 |
| miR-92a | Up-regulation | Up-regulation | ARID4B, BMPR2, CPEB2, ESR2, HIPK3, ITGA5, KAT2B, MYLIP, SMAD4, TGFBR2, THBS1, TP63, BCL2L11, CDH1, KLF2 |
| miR-100 | Up-regulation | Up-regulation | ATM, EGR2, FGFR3, ID1, MMP13, PLK1, IGF1R |
| miR-103 | Up-regulation | Inconsistent direction of expression | CCNE1, CDK2, CREB1, DICER1, GPD1, CAV1 |
| miR-106a | Up-regulation | Up-regulation | APP, ARID4B, CDKN1A, E2F1, HIPK3, IL10, MYLIP, RB1, RUNX1, VEGFA, FAS |
| miR-106b | Up-regulation | Up-regulation | APP, CCND1, CCND2, CDKN1A, E2F1, E2F3, ITCH, KAT2B, MAPK9, PTEN, RB1, RBL1, RBL2, TCEAL1, VEGFA, WEE1, EOMES |
| miR-107 | Up-regulation | Inconsistent direction of expression | ARNT, BACE1, CCNE1, CDCA4, CDK6, CRKL, DICER1, FBXW7, GRN, HIF1A, MYB, NFIA, PLAG1, RAB1B, VEGFA |
| miR-146a | Up-regulation | Down-regulation | BRCA1, BRCA2, CCNA2, CD40LG, CDKN1A, CDKN3, CFH, CXCR4, ERBB4, FADD, FAF1, FAS, IL8, IRAK1, IRAK2, KIF22, MTA2, NFKB1, PA2G4, ROCK1, TLR2, TRAF6, EGFR, SMAD4, TLR4 |
| miR-148a | Up-regulation | Down-regulation | CCKBR, DNMT1, DNMT3B, HLA-G, NR1I2, RPS6KA5, TGIF2 |
| miR-192 | Up-regulation | Up-regulation | ABCA8, ABCC3, ABCG2, AKAP9, ALCAM, ATP10D, ATXN7, B3GALNT1, BARD1, BCL2, BRD3, C1D, CADM1, CD164, CDC7, CDKN1B, CLIC1, CUL3, CUL5, DDOST, DDX3X, DLG5, DTL, E2F5, EGR1, ENOSF1, ENTPD3, ERCC3, ERLIN2, GOLGA6A, GRIA1, HOXA10, HRH1, HSP90B1, KIDINS220, KIF20B, LMNB2, LOXL2, MAD2L1, MAP3K1, MCM10, MFSD10, MIS12, MSN, ODC1, PANX1, PDE2A, PERP, PIK3R4, PIM1, PRPF38A, PTP4A3, RAB2A, RABGAP1, RACGAP1, RANBP3, RBL2, RRM1, SEMA4D, SEPT10, SETD4, SMARCB1, SPARC, STX7, TFG, TRAPPC2P1, WDR44, WNK1, XPA, ACVR2B, ERCC4, RB1 |
| miR-194 | Up-regulation | Down-regulation | CDH2, DNMT3A, EP300, HBEGF, IGF1R, ITGA9, PTPN12, PTPN13, RAC1, SOCS2, ACVR2B, SOX5 |
| miR-196a | Up-regulation | Inconsistent direction of expression | Annexin A1, HOXB7, HOXB8, NME4, p-JNK, TIMP1, MMP1/9, NFKBIA, IκBα, FOXO1, p27Kip1, NTN4, HOXA5, NOBOX, HOXA9, HOX-C8, HOX-B7, BMP4 |
| miR-199a-3p | Up-regulation | Inconsistent direction of expression | mTOR, MET, AXL, ZHX1, COX2, NLK, Aurora kinase A, c-Met, LKB1, Caveolin-2, STAT3, CD44, ZNF217, ZEB1 |
| miR-200c | Up-regulation | Down-regulation | ADAM12-L, CYP1B1, Zeb2, Snail1, BMI-1, E2F3, HO-1, ZEB1, ZEB2, UBQLN1, USP25, HMGB1, PTEN, KRAS, VEGFR2, MUC4, MUC16, ERG, DLC1, ATRX, HFE, TUBB3, TRKB, ETS1, FLT1, FHOD1, PPM1F, BRD7, PPP2R1B |
| miR-210 | Up-regulation | Inconsistent direction of expression | ABCB9, ACVR1B, AIFM3, APC, ATP11C, BDNF, CASP8AP2, CBX1, CDK10, CHD9, CLASP2, CPEB2, DDAH1, E2F3, EFNA3, ELK3, FAM116A, FGFRL1, GPD1L, HECTD1, HOXA1, HOXA3, HOXA9, ISCU, KIAA1161, MDGA1, MIB1, MID1IP1, MNT, MRE11A, NCAM1, NIPBL, NPTX1, P4HB, PIM1, PTAR1, PTPN1, RAD52, SEH1L, SERTAD2, SMCHD1, TNPO1, TP53I11, UBQLN1, XIST, XPA |
| miR-221 | Up-regulation | Up-regulation | BBC3, BMF, BNIP3, CDKN1B, CDKN1C, CORO1A, DDIT4, ESR1, FOS, FOXO3, HOXB5, ICAM1, KIT, NAIP, PTEN, SELE, SSSCA1, TCEAL1, TICAM1, TNFSF10, TP53, DICER1, DIRAS3, ETS1, TIMP3 |
| miR-223 | Up-regulation | Up-regulation | CHUK, E2F1, LMO2, MEF2C, NFIA, NFIX, RHOB, STMN1, EPB41L3, FBXW7, IGF1R, SLC2A4 |
| miR-376c | Up-regulation | Null | SMAD4, PC-TP, GRB2, TGFα, ALK5, ALK7, IGF-1R, ALK7 |
| miR-378 | Up-regulation | Down-regulation | GALNT7, MYC, NPNT, TOB2, VEGFA |
| miR-423-5p | Up-regulation | Up-regulation | TFF1 |
| miR-421 | Up-regulation | Up-regulation | Caspase-3, Menin, ATM, FOXO4, SERPINE 1, FXR, DPC4/Smad4 |
| miR-451 | Up-regulation | Inconsistent direction of expression | ABCB1, AKT1, BCL2, CAB39, MIF, MMP2, MMP9, SSSCA1, MYC, RAB14 |
| miR-486-5p | Up-regulation | Inconsistent direction of expression | CD40, Pim-1, ARHGAP5 |
| miR-744 | Up-regulation | Null | Zbed4, Lrsam1, Ddx21, AW555464, c-Myc, eEF1A2, TGF-β1, Ccnb1 |
| miR-93 | Up-regulation | Up-regulation | CDKN1A, E2F1, ITGB8, KAT2B, MAPK9, TP53INP1, TUSC2, VEGFA, PTEN |
| miR-195-5p | Down-regulation | Null | CDK4, CDK6, CCNE1, GLUT3 |
| Let-7a | Down-regulation | Inconsistent direction of expression | AMMECR1, APP, BCL2, CASP3, CASP8, CASP9, CCND2, DICER1, E2F1, E2F2, EGR3, EIF2C4, FOXA1, HMGA1, HMGA2, HNRPDL, HRAS, IGF2, IL6, ITGB3, KRAS, LIN28A, MEIS1, MYC, NEFM, NF2, NFKB1, NKIRAS2, NR1I2, NRAS, PRDM1, RAVER2, SLC20A1, THBS1, TRIM71, TUSC2, UHRF2, VDR, ZFP36L1, CDC34, EWSR1, IGF2BP1, MPL |
| miR-218 | Down-regulation | Down-regulation | ACTN1, BIRC6, CDKN1B, EBP, EFNA1, IKBKB, LAMB3, LASP1, MAFG, MBNL2, MRPS27, NACC1, NFE2L1, NUP93, SP1, STAM2, VOPP1, BIRC5, GJA1, RICTOR, ROBO1 |
| miR-375 | Down-regulation | Down-regulation | ELAVL4, JAK2, MTDH, PDK1, PLAG1, RASD1, TIMM8A, YAP1, YY1AP1 |
| miR-122 | Down-regulation | Null | DLX4, Cyclin G1, MEF2D, TRAIL, PKM2, Wnt/β-catenin, AKT3, PI3K/Akt, c-Myc, CAT1, PBF, IGF1R, p53, ALDOA, ARHGAP1, BCAT2, CS, GNPDA2, IQGAP1, LAMC1, LMNB2, MTHFD2 |
H19: Arita et al[178] suggested that plasma H19 represents an important complementary biomarker for GC detection. The expression level of plasma H19 was significantly higher in GC patients than in healthy controls. When GC patients underwent surgery, the expression level of plasma H19 decreased significantly, indicating that plasma H19 was potentially able to discriminate GC patients from healthy individuals, with a sensitivity of 74%, specificity of 58% and AUC of 0.64. H19 was reported to be up-regulated in GC tissues[179]. Recent studies have suggested that H19 targets RUNX1 and ISM1 to promote GC cell proliferation, migration and invasion[180,181], indicating that H19 functions as an oncogene in GC. In addition to GC, aberrant H19 was also found to play a crucial role in the development of multiple other types of cancer such as adrenocortical carcinoma, ovarian cancer, prostate cancer, glioma breast cancer, renal cell carcinoma, pancreatic cancer and bladder cancer[182-186].
LINC00152: Several novel studies have demonstrated that long intergenic non-protein-coding RNA 152 (LINC00152) is overexpressed in GC tissues[187,188]. Li et al[189] analyzed the level of circulating LINC00152 in plasma samples from 79 GC patients and 81 healthy volunteers by qRT-PCR. Their results suggested that plasma LINC00152 had significantly higher expression levels in GC patients compared with healthy individuals and had potential as a promising non-invasive biomarker for GC screening, with an AUC of 0.657, sensitivity of 48.1% and specificity of 85.2%. Gastric juice can also be a source of biomarkers for GC detection; Pang et al[187] found that LINC00152 levels were significantly increased in gastric juice from patients with GC compared with healthy controls. Accordingly, we speculate that LINC00152 may function as an oncogene to promote tumorigenesis in GC. However, the function of LINC00152 in GC should be investigated further.
FERL4: Liu et al[190] demonstrated that FER1L4, a newly identified lncRNA, is down-regulated in GC tissues and that low expression levels of FER1L4 are associated with histological grade, tumor diameter, TNM stage, lymphatic metastasis, perineural invasion, venous invasion, and serum CA724. The authors further examined the expression level of plasma FER1L4 and observed no differences in the levels between preoperative GC patients and healthy individuals; however, a sharp decline in expression levels was observed in GC patients two weeks after surgery, suggesting that FER1L4 might serve as a potential biomarker for clinical prognosis evaluation of patients with GC. Song et al[191] were the first to report that FER1L4 is down-regulated in GC tissues relative to normal gastric specimens. Subsequently, Xia et al[192] suggested that FER1L4 is involved in a cancer-associated ceRNA network containing eight lncRNAs (FER1L4, GACAT1, GACAT3, AC009499.1, H19, LINC00152, RP4-620F22and 3AP000288.2.) and nine miRNAs (miR-106a-5p, miR-106b-5p, miR-139-5p, miR-18a-5p, miR-18b-5p, miR-19a-3p, miR-195-5p miR-20b-5p and miR-31-5p) to modulate tumor development and invasion in GC, thereby suggesting that FER1L4 functions as a tumor suppressor lncRNA in GC. However, the mechanism and function of FER1L4 should be further explored.
CUDR, LSINCT-5 and PTENP1: Dong et al[193] measured the expression levels of 39 candidate cancer-associated circulating lncRNAs by RT-qPCR in the sera of 110 patients with GC, 15 patients with benign gastric ulcer and 106 healthy individuals. Their results suggested that CUDR, LSINCT-5 and PTENP1 in the sera were the most significantly down-regulated in GC patients compared with the control group. During the validation phase of this study, the combination of CUDR, LSINCT-5 and PTENP1 provided the greatest predictive ability to distinguish GC patients from healthy controls, with an AUC of 0.92, sensitivity of 74.1% and specificity of 100%. For early GC detection, the three-lncRNA panel indicated a strong diagnostic value, with an AUC of 0.832, sensitivity of 77.8% and specificity of 97.0%. In addition, the three serum lncRNAs were also identified to be sufficiently sensitive and specific to distinguish benign peptic ulcers from GC patients, with an AUC of 0.902, sensitivity of 91.7% and specificity of 83.3%, indicating that the three-lncRNA serum signature might represent a novel biomarker candidate for GC detection. Concurrently, this study reported that a lower expression level of the three-lncRNA panel predicted higher survival rates in patients with GC, implying that these three serum lncRNAs can predict the prognosis of GC patients. However, CUDR, also termed UCA1, was observed to be overexpressed in GC tissues and gastric juice, indicating its potential as an onco-lncRNA and as a diagnostic biomarker in GC[194,195]. LSINCT-5 has also been reported to be up-regulated in GC tissues and to exhibit oncogenic activity to promote cellular proliferation. Concurrently, LSINCT-5 overexpression predicts the negative prognosis of GC patients[196]. The expression levels of CUDR and LSINCT-5 in GC tissues are inconsistent with their circulating levels, which should be validated by more reliable studies in the future. PTENP1 has not been well characterized in GC; however, previous studies have emphasized PTENP1 down-regulation in hepatocellular carcinoma, endometrial cancer and clear cell renal cell carcinoma, suggesting that it serves as a tumor suppressor to inhibit tumorigenesis[197-199]. Nonetheless, little is known regarding the functions and mechanisms of CUDR, LSINCT-5 and PTENP1 in GC; these lncRNAs should be thoroughly examined in future studies.
In addition to miRNAs, human blood may contain abundant lncRNAs, which can be detected by RNA-seq deep-sequencing technologies or microarrays and validated by RT-PCR, implying the potential application of circulating lncRNAs in GC detection; the AUC, sensitivity and specificity ranges were 0.64-0.92, 48.1%-77.8%, and 58%-100%, respectively (Table 3). The combination of CUDR, LSINCT-5 and PTENP1 provides the strongest diagnostic value of GC detection among all of the circulating lncRNAs, with an AUC of 0.92, sensitivity of 74.1% and specificity of 100%. For early-stage GC detection, the combination of CUDR, LSINCT-5 and PTENP1 reveals an AUC of 0.832 with a sensitivity of 77.8% and specificity of 97%, but the sample size is small (9 early-stage GC patients, 18 healthy subjects and 15 gastric peptic ulcer subjects), and the result must be validated in GC patients on a larger scale. However, few studies have focused on the diagnosis of GC classified by different tumor sites, different tumor stages or different pathological types (e.g., early-stage or advanced-stage GC, cardiac or non-cardiac GC, and intestinal-type, diffuse-type or mixed-type GC). To promote the prognosis of GC, studies of circulating lncRNAs for different subtypes of GC, particularly for early-stage GC detection, should be a focus of future research. Taken together, the sensitivity and specificity of a single tumor-associated circulating lncRNA as a biomarker remain poor; however, a circulating lncRNA signature, especially the combination of various circulating lncRNAs, can promote the diagnostic efficiency of GC detection considerably. Among these lncRNAs (Table 4), H19 and LINC00152 are up-regulated in the circulation, while FER1L4, LSINCT-5, PTENP1 and CUDR (UCA1) are down-regulated. FER1L4 is also down-regulated in GC tissues, indicating that it acts as a tumor suppressor in GC. In contrast, H19, LINC00152, LSINCT-5 and CUDR (UCA1) manifest up-regulated expression levels in GC tissues, suggesting that the oncogene roles they play in GC. However, the expression levels of LSINCT-5 and CUDR (UCA1) in tissues are inconsistent with their circulating levels, active secretion may underlie this discrepancy, but the inconsistent direction of expression should be validated in future studies. lncRNAs in gastric juice also exhibit potential in the early diagnosis of GC. Shao et al[200] suggested that lncRNA-AA174084 in gastric juice might serve as a promising biomarker for GC detection, with an AUC of 0.848. The rapidly expanding catalog of circulating lncRNAs strongly supports their clinical utility in GC diagnosis.
| Circulating lncRNA | Species | Samples | Sensitivity | Specificity | AUC | Population |
| FER1L4[190] | Human | Plasma | 67.2% | 80.3% | 0.778 | 83 GCs (Pre- and post-operative patients) |
| H19[178] | Human | Plasma | 74% | 58% | 0.64 | 43 GCs and 34 healthy controls |
| CUDR, LSINCT-5 and PTENP1[193] | Human | Serum | 74.1% of CUDR + LSINCT-5 + PTENP1 for GC detection, | 100% of CUDR + LSINCT-5 + PTENP1 for GC detection, 97.0% of CUDR + LSINCT-5 + PTENP1 for early GC detection | 0.92 of CUDR + LSINCT-5 + PTENP1 for GC detection, 0.832 of CUDR + LSINCT-5 + PTENP1 for early GC detection | 9 early-stage GCs, 64 advanced-stage GCs, 15 benign Peptic ulcer patients and 86 healthy controls |
| 77.8% of CUDR + LSINCT-5 + PTENP1 for early GC detection | ||||||
| LINC00152[189] | Human | Plasma | 48.1% | 85.2% | 0.657 | 79 GCs and 81 healthy controls |
| Circulating miRNA | Expression in the circulation | Expression in tissue | Validated targeted miRNAs | Putative targeted miRNAs in GC |
| H19 | Up-regulation | Up-regulation | miR-675, let-7 and miR-200 family | miR-17-5p, miR-18a-5p, miR-18b-5p, miR-19a-3p, miR-20a-5p, miR-20b-5p, miR-106a-5p, miR-106b-5, |
| LINC00152 | Up-regulation | Up-regulation | miR-130b-3p, miR-217 | miR-18a-5p, miR-18b-5p, miR-31-5p, miR-139-5p, miR-195-5p, miR-497-5p |
| FER1L4 | Down-regulation | Down-regulation | Null | miR-18a-5p, miR-18b-5p, miR-106a-5p, miR-133b, miR-139-5p, miR-195-5p, miR-497-5p |
| LSINCT-5 | Down-regulation | Up-regulation | Null | Null |
| CUDR (UCA1) | Down-regulation | Up-regulation | miR-216b, miR-1, miR-143 | Null |
| PTENP1 | Down-regulation | Null | miR-17, miR-19b, miR-20a, miR21 and miR-26a | Null |
Many studies have explored the mechanism underlying the secretion of miRNAs and lncRNAs. Multiple possibilities for miRNA secretion have been elucidated: (1) Active secretion by microvesicles (MVs), exosomes and apoptotic bodies. To be resistant to RNase activity, miRNAs destined for release are ultimately present in a stable, protected size that is much smaller than that of a typical epithelial cell such as exosomes and membrane-bound particles[25]. Skog et al[201] demonstrated that microvesicles are useful delivery vehicles for miRNA secretion in glioblastoma. The authors isolated microvesicles from glioblastoma-conditioned medium, performed microarrays and quantitative miRNA RT-PCR to identify 11 miRNAs that were abundant in donor cells and microvesicles to promote tumor growth and invasion, suggesting an intriguing insight into miRNA secretion. Kosaka et al[202] suggested that miRNAs are secreted within CD63-positive exosomes via a ceramide-dependent pathway but without ESCRT machinery. Ceramide is a key molecule that triggers the secretion of small membrane vesicles called exosomes, and the biosynthesis of ceramide is modulated by neutral sphingomyelinase 2 (nSMase2). The authors stated that the expression of exogenous miR-146a was attenuated by nSMase2 siRNA compared with control but was not affected by siRNA targeting Alix, an ESCRT-associated protein. Another underlying mechanism of miRNA secretion may be apoptotic bodies. Zernecke et al[203] reported that miR-126 is enriched in apoptotic bodies and that endothelial cell-derived apoptotic bodies transmitted paracrine alarm signals to recipient vascular cells to trigger CXCL12 generation, which is regulated by miR-126; (2) miRNA secretion associated with other molecules. Vickers et al[204] demonstrated that a novel intercellular communication pathway is involved in miRNA transportation and cellular communication by high-density lipoproteins. miRNAs are also released via protein complexes. Argonaute (Ago) complexes that mediate mRNA silencing activity were identified to serve as significant carriers of miRNAs in plasma[24]. Nucleophosmin 1 (NPM1), an RNA-binding protein, was found to protect miRNAs from degradation and to play a crucial role in packaging and exporting extracellular miRNAs[205]; and (3) Passive leakage from cells due to chronic inflammation, necrosis or injury. Previous studies have suggested that the secretion of some circulating miRNAs was due to tissue injury, such as myocardial injury, liver injury and kidney injury[206-209].
The mechanism underlying lncRNA secretion has not been systematically investigated. Circulating lncRNAs may be secreted in the same manner as extracellular miRNAs. In recent studies, circulating lncRNAs have been detected in exosomes, where they are protected from RNase[189,210]. Other types of extracellular vesicles, including apoptotic bodies and microvesicles, may also participate in the secretion pathway[189,211]. Despite recent research exploring the potential mechanism of miRNA and lncRNA secretion, significant work remains to be performed. Circulating miRNAs and lncRNAs might play general and pivotal roles as signal-conducting molecules in multiple physiological and pathological processes. Additional studies will be needed to explore these exciting mysteries and their mechanisms.
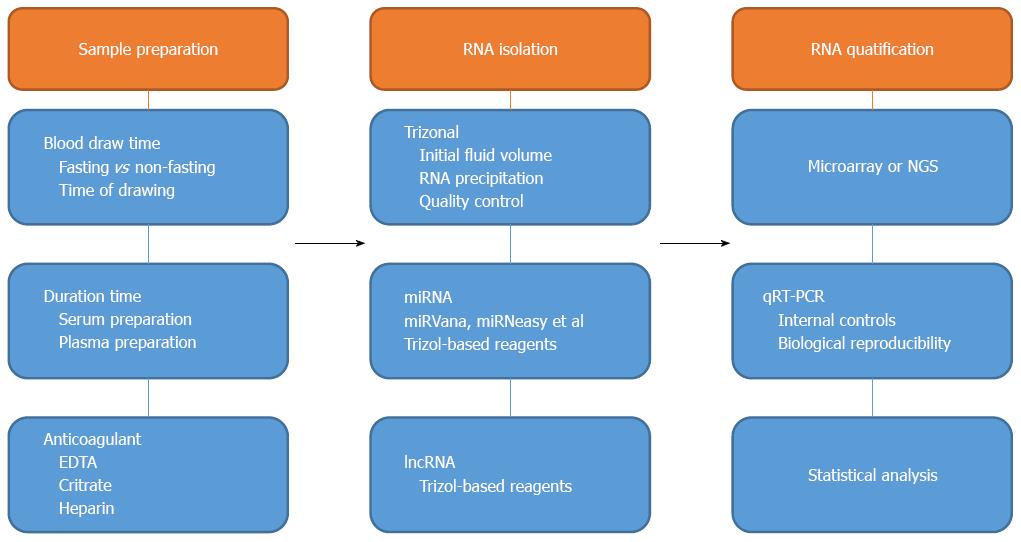
Increasing evidence suggests that cell-free circulating miRNAs and lncRNAs could serve as diagnostic and prognostic biomarkers for multiple types of cancer. Reliable methodologies and strategies for the quantification of circulating miRNAs and lncRNAs are urgently needed for clinical utility or experimental purposes (Figure 3). Different matrixes may exhibit different expression profiles of these circulating miRNAs and lncRNAs. Most studies use serum and plasma samples, with few reports utilizing whole blood or peripheral blood mononuclear cell samples. Conclusive research has suggested that the levels of miRNAs or lncRNAs detected in the serum are consistent with the levels in plasma in the same pathology if cellular contaminants are avoided; miRNAs and lncRNAs are both generally detected in hemocytes such as thrombocytes and erythrocytes[26]. Consequently, because the blood cell fraction may generate background noise, using serum or plasma will decrease the background noise and promote higher accuracy for circulating miRNAs and lncRNAs detection over whole blood[212]. Nonetheless, hemolysis frequently occurs at low levels during the sampling and processing of blood. To estimate the suitability of serum/plasma samples for analysis, the extent of hemolysis should be estimated by measuring the free hemoglobin and endogenous RNAs[213].
miRNAs and lncRNAs can be extracted using different approaches. Acid guanidinium thiocyanate-phenol-chloroform (AGPC) extraction is used primarily for RNA isolation, followed by either column-based RNA purification from the aqueous solution or precipitation of the RNA-containing aqueous solution. RNAs can be precipitated from aqueous solutions by adding 2-isopropanol and then stored overnight at -20 °C or -80 °C. Currently, standard protocols, including commercial kits directing small RNA enrichment or TRIzol-based reagents, have garnered wide acceptance[214]. If we target miRNAs, a miRVana extraction kit may improve the detection precision. However, discrepancies may be observed between different methods of extraction; most of the variabilities discovered between replicates are due to the different RNA extraction processes[213]. Therefore, to reduce the heterogeneity of circulating miRNA and lncRNA profiles, selective and consistent extraction methods throughout an entire study are strongly recommended[215].
Microarrays and next-generation sequencing (NGS) are performed to obtain large-scale profiles of circulating miRNAs and lncRNAs following the extraction of miRNAs and lncRNAs. miRNA qPCR arrays and lncRNA PCR arrays are also utilized to capture miRNA and lncRNA profiles. Each method has its own advantages and limitations. NGS can identify novel miRNAs and lncRNAs but is less cost-effective and less efficient compared with microarrays. Git et al[216] analyzed three biological samples across six miRNA microarray platforms and compared their hybridization performance. Only 53 miRNAs were consistent among the 4 microarrays. The sensitivity and specificity of the different platforms vary significantly. Different methods for the detection of circulating lncRNAs and miRNAs can lead to discrepancies in GC diagnoses. A large sample size is recommended to improve the reliability of the results.
After large-scale profiling of GC, the differential circulating miRNAs and lncRNAs between patients and healthy individuals must be validated in larger cohorts[217]. The standard reference for miRNA and lncRNA qualification is qRT-PCR. With low detection limits and high sensitivity, this method is suitable for circulating miRNA and lncRNA identification. Endogenous controls are important for qRT-PCR. U6, miR-93 and miR-16 are the most frequently used endogenous controls for miRNA normalization in qRT-PCR for GC detection. Chen et al[218] stated that the combination of let-7g, let-7d and let-7i could function as a reference gene superior to U6 and miR-16 for the normalization of circulating miRNAs. Kraus et al[219] investigated the expression stability of 90 lncRNAs in 30 tissue specimens, including anaplastic astrocytoma, human diffuse astrocytoma, glioblastoma and normal white matter. Their results indicated that 4 lncRNAs (BC200, Zfhx2as, H19 upstream conserved 1 and 2, and HOXA6as) were suitable for use as normalizers of lncRNA expression profiling in glioma and normal brain. Nonetheless, no current consensus exists with respect to a reference for the normalization of circulating lncRNAs. cDNA synthesis of miRNAs and lncRNAs is crucial for qRT-PCR, and universal poly-A tailing and stem-loop primer extension are two current methods. Dunnett et al[220] suggested that poly-A tail extension exhibited more amplification than stem-loop primer extension and that using poly-A tail extension might be best, particularly when low traces of sample are available. Universal poly-A tail extension may be an interesting method for miRNA identification in bodily fluids. Chugh et al[221] suggested that sequence-specific stem-loop primer extension failed to generate a signal (CT) for low-abundance miRNAs compared with universal poly-A tailing assays; however, universal poly-A tailing generated a quantitative signal with significant non-specific amplification. cDNA synthesis of lncRNA has not been extensively studied at present; most researchers use commercial kits. Speth et al[222] suggested a simple and sensitive protocol that allows the quantification of mRNAs, selected sRNAs, and lncRNAs in one cDNA sample by qRT-PCR.
GC was the world’s third leading cause of cancer mortality in 2012 and was responsible for 723000 deaths[223]. Delayed diagnosis is the largest obstacle to the treatment of GC; thus, the effective means to improve the prognosis of GC is to search for ideal biomarkers for early GC detection. Many serum markers, including CA199, CA125, CA724, CA242, CA50, CEA, and pepsinogen, are widely utilized in the clinic for GC detection. However, low sensitivity or low specificity diminishes their clinical value for GC diagnosis. A large number of non-protein-coding transcripts were formerly regarded as “noise” or “garbage” within the genome. With the progression of high-throughput sequencing technologies (microarray or RNA-seq), something important that had been hidden in the diverse non-coding RNAs has attracted the attention of many researchers and has driven them to search for promising functions of non-coding RNAs in the pathogenesis of diseases[224]. More importantly, the study of miRNAs and lncRNAs has gradually become prominent in RNA biology. Emerging evidence has suggested that the dysregulated expression of miRNAs and lncRNAs in cancer alters the spectrum of disease progression and might serve as an independent biomarker of multiple types of cancer[14]. The detection of circulating miRNAs and lncRNAs may be valuable for improving the diagnostic and prognostic value of GC. In this review, we have highlighted circulating miRNAs and lncRNAs in GC detection and summarized the potential mechanisms underlying extracellular miRNA and lncRNA secretion. The use of miRNAs and lncRNAs has several limitations such as the diverse methodologies for miRNA and lncRNA detection and the small cohort size for the validation steps in the present studies. Nonetheless, circulating miRNAs and lncRNAs exhibit higher diagnostic values relative to conventional tumor markers such as CA199, CA125, CA724, CA242, CA50, CEA, and pepsinogen. The combination of biomarkers may improve the diagnostic accuracy, and the combination of circulating miRNAs, lncRNAs and other screening methods may be particularly useful in cancer detection. Prospective studies should validate the feasibility of using circulating miRNAs and lncRNAs as diagnostic biomarkers for GC detection in different populations. These circulating small nucleic acids (miRNAs, lncRNAs and others) may have potential in distinguishing patients with GC from healthy individuals, with the advantages of practicability, non-invasiveness and cost-effectiveness.
P- Reviewer: Chiurillo MA, Park WS S- Editor: Ma YJ L- Editor: Wang TQ E- Editor: Wang CH
| 1. | Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359-E386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20108] [Cited by in RCA: 20516] [Article Influence: 2051.6] [Reference Citation Analysis (20)] |
| 2. | Zhu X, Lv M, Wang H, Guan W. Identification of circulating microRNAs as novel potential biomarkers for gastric cancer detection: a systematic review and meta-analysis. Dig Dis Sci. 2014;59:911-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 3. | Nilsson PJ, van Etten B, Hospers GA, Påhlman L, van de Velde CJ, Beets-Tan RG, Blomqvist L, Beukema JC, Kapiteijn E, Marijnen CA. Short-course radiotherapy followed by neo-adjuvant chemotherapy in locally advanced rectal cancer--the RAPIDO trial. BMC Cancer. 2013;13:279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 209] [Cited by in RCA: 215] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 4. | Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281-297. [PubMed] |
| 5. | Ebert MS, Sharp PA. Roles for microRNAs in conferring robustness to biological processes. Cell. 2012;149:515-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1144] [Cited by in RCA: 1260] [Article Influence: 96.9] [Reference Citation Analysis (0)] |
| 7. | Mendell JT, Olson EN. MicroRNAs in stress signaling and human disease. Cell. 2012;148:1172-1187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1233] [Cited by in RCA: 1371] [Article Influence: 105.5] [Reference Citation Analysis (0)] |
| 8. | Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5705] [Cited by in RCA: 6032] [Article Influence: 317.5] [Reference Citation Analysis (0)] |
| 9. | Prensner JR, Iyer MK, Balbin OA, Dhanasekaran SM, Cao Q, Brenner JC, Laxman B, Asangani IA, Grasso CS, Kominsky HD. Transcriptome sequencing across a prostate cancer cohort identifies PCAT-1, an unannotated lincRNA implicated in disease progression. Nat Biotechnol. 2011;29:742-749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 769] [Cited by in RCA: 841] [Article Influence: 60.1] [Reference Citation Analysis (0)] |
| 10. | Gutschner T, Diederichs S. The hallmarks of cancer: a long non-coding RNA point of view. RNA Biol. 2012;9:703-719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1254] [Cited by in RCA: 1504] [Article Influence: 115.7] [Reference Citation Analysis (0)] |
| 11. | Brunner AL, Beck AH, Edris B, Sweeney RT, Zhu SX, Li R, Montgomery K, Varma S, Gilks T, Guo X. Transcriptional profiling of long non-coding RNAs and novel transcribed regions across a diverse panel of archived human cancers. Genome Biol. 2012;13:R75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 194] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 12. | Schaukowitch K, Joo JY, Liu X, Watts JK, Martinez C, Kim TK. Enhancer RNA facilitates NELF release from immediate early genes. Mol Cell. 2014;56:29-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 335] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 13. | Gibb EA, Brown CJ, Lam WL. The functional role of long non-coding RNA in human carcinomas. Mol Cancer. 2011;10:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1127] [Cited by in RCA: 1340] [Article Influence: 95.7] [Reference Citation Analysis (0)] |
| 14. | Prensner JR, Chinnaiyan AM. The emergence of lncRNAs in cancer biology. Cancer Discov. 2011;1:391-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1266] [Cited by in RCA: 1470] [Article Influence: 113.1] [Reference Citation Analysis (0)] |
| 15. | Yap KL, Li S, Muñoz-Cabello AM, Raguz S, Zeng L, Mujtaba S, Gil J, Walsh MJ, Zhou MM. Molecular interplay of the noncoding RNA ANRIL and methylated histone H3 lysine 27 by polycomb CBX7 in transcriptional silencing of INK4a. Mol Cell. 2010;38:662-674. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1163] [Cited by in RCA: 1090] [Article Influence: 72.7] [Reference Citation Analysis (0)] |
| 16. | Poliseno L, Salmena L, Zhang J, Carver B, Haveman WJ, Pandolfi PP. A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature. 2010;465:1033-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1939] [Cited by in RCA: 1875] [Article Influence: 125.0] [Reference Citation Analysis (0)] |
| 17. | Gong C, Maquat LE. lncRNAs transactivate STAU1-mediated mRNA decay by duplexing with 3’ UTRs via Alu elements. Nature. 2011;470:284-288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1072] [Cited by in RCA: 1005] [Article Influence: 71.8] [Reference Citation Analysis (0)] |
| 18. | Tam OH, Aravin AA, Stein P, Girard A, Murchison EP, Cheloufi S, Hodges E, Anger M, Sachidanandam R, Schultz RM. Pseudogene-derived small interfering RNAs regulate gene expression in mouse oocytes. Nature. 2008;453:534-538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 877] [Cited by in RCA: 825] [Article Influence: 48.5] [Reference Citation Analysis (0)] |
| 19. | Watanabe T, Totoki Y, Toyoda A, Kaneda M, Kuramochi-Miyagawa S, Obata Y, Chiba H, Kohara Y, Kono T, Nakano T. Endogenous siRNAs from naturally formed dsRNAs regulate transcripts in mouse oocytes. Nature. 2008;453:539-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 837] [Cited by in RCA: 836] [Article Influence: 49.2] [Reference Citation Analysis (0)] |
| 20. | Zhao Y, Guo Q, Chen J, Hu J, Wang S, Sun Y. Role of long non-coding RNA HULC in cell proliferation, apoptosis and tumor metastasis of gastric cancer: a clinical and in vitro investigation. Oncol Rep. 2014;31:358-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 221] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 21. | Sun Y, Zhang K, Fan G, Li J. Identification of circulating microRNAs as biomarkers in cancers: what have we got? Clin Chem Lab Med. 2012;50:2121-2126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 22. | Zandberga E, Kozirovskis V, Ābols A, Andrējeva D, Purkalne G, Linē A. Cell-free microRNAs as diagnostic, prognostic, and predictive biomarkers for lung cancer. Genes Chromosomes Cancer. 2013;52:356-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 23. | Taylor DD, Gercel-Taylor C. MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol Oncol. 2008;110:13-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1730] [Cited by in RCA: 1858] [Article Influence: 109.3] [Reference Citation Analysis (0)] |
| 24. | Arroyo JD, Chevillet JR, Kroh EM, Ruf IK, Pritchard CC, Gibson DF, Mitchell PS, Bennett CF, Pogosova-Agadjanyan EL, Stirewalt DL. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc Natl Acad Sci USA. 2011;108:5003-5008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2345] [Cited by in RCA: 2643] [Article Influence: 188.8] [Reference Citation Analysis (0)] |
| 25. | Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O’Briant KC, Allen A. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA. 2008;105:10513-10518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5636] [Cited by in RCA: 6313] [Article Influence: 371.4] [Reference Citation Analysis (0)] |
| 26. | Chen X, Ba Y, Ma L, Cai X, Yin Y, Wang K, Guo J, Zhang Y, Chen J, Guo X. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008;18:997-1006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3218] [Cited by in RCA: 3554] [Article Influence: 209.1] [Reference Citation Analysis (0)] |
| 27. | Wang J, Huang SK, Zhao M, Yang M, Zhong JL, Gu YY, Peng H, Che YQ, Huang CZ. Identification of a circulating microRNA signature for colorectal cancer detection. PLoS One. 2014;9:e87451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 136] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 28. | Liu R, Zhang C, Hu Z, Li G, Wang C, Yang C, Huang D, Chen X, Zhang H, Zhuang R. A five-microRNA signature identified from genome-wide serum microRNA expression profiling serves as a fingerprint for gastric cancer diagnosis. Eur J Cancer. 2011;47:784-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 328] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 29. | Li X, Zhang Z, Yu M, Li L, Du G, Xiao W, Yang H. Involvement of miR-20a in promoting gastric cancer progression by targeting early growth response 2 (EGR2). Int J Mol Sci. 2013;14:16226-16239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 30. | Liu T, Tang H, Lang Y, Liu M, Li X. MicroRNA-27a functions as an oncogene in gastric adenocarcinoma by targeting prohibitin. Cancer Lett. 2009;273:233-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 289] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 31. | Liu J, Wang X, Yang X, Liu Y, Shi Y, Ren J, Guleng B. miRNA423-5p regulates cell proliferation and invasion by targeting trefoil factor 1 in gastric cancer cells. Cancer Lett. 2014;347:98-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 32. | Han C, Zhou Y, An Q, Li F, Li D, Zhang X, Yu Z, Zheng L, Duan Z, Kan Q. MicroRNA-1 (miR-1) inhibits gastric cancer cell proliferation and migration by targeting MET. Tumour Biol. 2015;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 33. | Kim CH, Kim HK, Rettig RL, Kim J, Lee ET, Aprelikova O, Choi IJ, Munroe DJ, Green JE. miRNA signature associated with outcome of gastric cancer patients following chemotherapy. BMC Med Genomics. 2011;4:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 117] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 34. | Yao Y, Suo AL, Li ZF, Liu LY, Tian T, Ni L, Zhang WG, Nan KJ, Song TS, Huang C. MicroRNA profiling of human gastric cancer. Mol Med Rep. 2009;2:963-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 147] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 35. | Wang AM, Huang TT, Hsu KW, Huang KH, Fang WL, Yang MH, Lo SS, Chi CW, Lin JJ, Yeh TS. Yin Yang 1 is a target of microRNA-34 family and contributes to gastric carcinogenesis. Oncotarget. 2014;5:5002-5016. [PubMed] |
| 36. | Peng Y, Guo JJ, Liu YM, Wu XL. MicroRNA-34A inhibits the growth, invasion and metastasis of gastric cancer by targeting PDGFR and MET expression. Biosci Rep. 2014;34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 37. | Hu Z, Chen X, Zhao Y, Tian T, Jin G, Shu Y, Chen Y, Xu L, Zen K, Zhang C. Serum microRNA signatures identified in a genome-wide serum microRNA expression profiling predict survival of non-small-cell lung cancer. J Clin Oncol. 2010;28:1721-1726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 591] [Cited by in RCA: 637] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 38. | Xu L, Zhang Y, Wang H, Zhang G, Ding Y, Zhao L. Tumor suppressor miR-1 restrains epithelial-mesenchymal transition and metastasis of colorectal carcinoma via the MAPK and PI3K/AKT pathway. J Transl Med. 2014;12:244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 76] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 39. | Stope MB, Stender C, Schubert T, Peters S, Weiss M, Ziegler P, Zimmermann U, Walther R, Burchardt M. Heat-shock protein HSPB1 attenuates microRNA miR-1 expression thereby restoring oncogenic pathways in prostate cancer cells. Anticancer Res. 2014;34:3475-3480. [PubMed] |
| 40. | Han C, Yu Z, Duan Z, Kan Q. Role of microRNA-1 in human cancer and its therapeutic potentials. Biomed Res Int. 2014;2014:428371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 41. | Bronisz A, Wang Y, Nowicki MO, Peruzzi P, Ansari KI, Ogawa D, Balaj L, De Rienzo G, Mineo M, Nakano I. Extracellular vesicles modulate the glioblastoma microenvironment via a tumor suppression signaling network directed by miR-1. Cancer Res. 2014;74:738-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 192] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 42. | Valladares-Ayerbes M, Reboredo M, Medina-Villaamil V, Iglesias-Díaz P, Lorenzo-Patiño MJ, Haz M, Santamarina I, Blanco M, Fernández-Tajes J, Quindós M. Circulating miR-200c as a diagnostic and prognostic biomarker for gastric cancer. J Transl Med. 2012;10:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 123] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 43. | Zhou X, Wang Y, Shan B, Han J, Zhu H, Lv Y, Fan X, Sang M, Liu XD, Liu W. The downregulation of miR-200c/141 promotes ZEB1/2 expression and gastric cancer progression. Med Oncol. 2015;32:428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 44. | Toiyama Y, Hur K, Tanaka K, Inoue Y, Kusunoki M, Boland CR, Goel A. Serum miR-200c is a novel prognostic and metastasis-predictive biomarker in patients with colorectal cancer. Ann Surg. 2014;259:735-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 244] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 45. | Wang H, Wang L, Wu Z, Sun R, Jin H, Ma J, Liu L, Ling R, Yi J, Wang L. Three dysregulated microRNAs in serum as novel biomarkers for gastric cancer screening. Med Oncol. 2014;31:298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 46. | Li J, Guo Y, Liang X, Sun M, Wang G, De W, Wu W. MicroRNA-223 functions as an oncogene in human gastric cancer by targeting FBXW7/hCdc4. J Cancer Res Clin Oncol. 2012;138:763-774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 130] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 47. | Li X, Zhang Y, Zhang H, Liu X, Gong T, Li M, Sun L, Ji G, Shi Y, Han Z. miRNA-223 promotes gastric cancer invasion and metastasis by targeting tumor suppressor EPB41L3. Mol Cancer Res. 2011;9:824-833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 293] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 48. | Yang G, Gong Y, Wang Q, Wang Y, Zhang X. The role of miR-100-mediated Notch pathway in apoptosis of gastric tumor cells. Cell Signal. 2015;27:1087-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 49. | Oh HK, Tan AL, Das K, Ooi CH, Deng NT, Tan IB, Beillard E, Lee J, Ramnarayanan K, Rha SY. Genomic loss of miR-486 regulates tumor progression and the OLFM4 antiapoptotic factor in gastric cancer. Clin Cancer Res. 2011;17:2657-2667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 165] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 50. | Xia L, Zhang D, Du R, Pan Y, Zhao L, Sun S, Hong L, Liu J, Fan D. miR-15b and miR-16 modulate multidrug resistance by targeting BCL2 in human gastric cancer cells. Int J Cancer. 2008;123:372-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 562] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 51. | Li X, Luo F, Li Q, Xu M, Feng D, Zhang G, Wu W. Identification of new aberrantly expressed miRNAs in intestinal-type gastric cancer and its clinical significance. Oncol Rep. 2011;26:1431-1439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 52. | Zhu C, Ren C, Han J, Ding Y, Du J, Dai N, Dai J, Ma H, Hu Z, Shen H. A five-microRNA panel in plasma was identified as potential biomarker for early detection of gastric cancer. Br J Cancer. 2014;110:2291-2299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 187] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 53. | Zhang M, Wang X, Li W, Cui Y. miR-107 and miR-25 simultaneously target LATS2 and regulate proliferation and invasion of gastric adenocarcinoma (GAC) cells. Biochem Biophys Res Commun. 2015;460:806-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 54. | Zhao H, Wang Y, Yang L, Jiang R, Li W. MiR-25 promotes gastric cancer cells growth and motility by targeting RECK. Mol Cell Biochem. 2014;385:207-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 55. | Wu Q, Yang Z, Wang F, Hu S, Yang L, Shi Y, Fan D. MiR-19b/20a/92a regulates the self-renewal and proliferation of gastric cancer stem cells. J Cell Sci. 2013;126:4220-4229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 56. | Ueda T, Volinia S, Okumura H, Shimizu M, Taccioli C, Rossi S, Alder H, Liu CG, Oue N, Yasui W. Relation between microRNA expression and progression and prognosis of gastric cancer: a microRNA expression analysis. Lancet Oncol. 2010;11:136-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 644] [Cited by in RCA: 676] [Article Influence: 45.1] [Reference Citation Analysis (0)] |
| 57. | Tsukamoto Y, Nakada C, Noguchi T, Tanigawa M, Nguyen LT, Uchida T, Hijiya N, Matsuura K, Fujioka T, Seto M. MicroRNA-375 is downregulated in gastric carcinomas and regulates cell survival by targeting PDK1 and 14-3-3zeta. Cancer Res. 2010;70:2339-2349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 346] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 58. | He Z, Liu Y, Xiao B, Qian X. miR-25 modulates NSCLC cell radio-sensitivity through directly inhibiting BTG2 expression. Biochem Biophys Res Commun. 2015;457:235-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 59. | Li Z, Sun Y, Chen X, Squires J, Nowroozizadeh B, Liang C, Huang J. p53 Mutation Directs AURKA Overexpression via miR-25 and FBXW7 in Prostatic Small Cell Neuroendocrine Carcinoma. Mol Cancer Res. 2015;13:584-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 60. | Ju X, Li D, Shi Q, Hou H, Sun N, Shen B. Differential microRNA expression in childhood B-cell precursor acute lymphoblastic leukemia. Pediatr Hematol Oncol. 2009;26:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 61. | Bandres E, Bitarte N, Arias F, Agorreta J, Fortes P, Agirre X, Zarate R, Diaz-Gonzalez JA, Ramirez N, Sola JJ. microRNA-451 regulates macrophage migration inhibitory factor production and proliferation of gastrointestinal cancer cells. Clin Cancer Res. 2009;15:2281-2290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 284] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 62. | Wang P, Yang D, Zhang H, Wei X, Ma T, Cheng Z, Hong Q, Hu J, Zhuo H, Song Y. Early Detection of Lung Cancer in Serum by a Panel of MicroRNA Biomarkers. Clin Lung Cancer. 2015;16:313-319.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 76] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 63. | Langhe R, Norris L, Saadeh FA, Blackshields G, Varley R, Harrison A, Gleeson N, Spillane C, Martin C, O’Donnell DM. A novel serum microRNA panel to discriminate benign from malignant ovarian disease. Cancer Lett. 2015;356:628-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 64. | Zhou X, Zhang X, Yang Y, Li Z, Du L, Dong Z, Qu A, Jiang X, Li P, Wang C. Urinary cell-free microRNA-106b as a novel biomarker for detection of bladder cancer. Med Oncol. 2014;31:197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 65. | Komatsu S, Ichikawa D, Hirajima S, Kawaguchi T, Miyamae M, Okajima W, Ohashi T, Arita T, Konishi H, Shiozaki A. Plasma microRNA profiles: identification of miR-25 as a novel diagnostic and monitoring biomarker in oesophageal squamous cell carcinoma. Br J Cancer. 2014;111:1614-1624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 66. | Wang Y, Gao Y, Shi W, Zhai D, Rao Q, Jia X, Liu J, Jiao X, Du Z. Profiles of differential expression of circulating microRNAs in hepatitis B virus-positive small hepatocellular carcinoma. Cancer Biomark. 2015;15:171-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 67. | Chang CC, Lin CC, Hsieh WL, Lai HW, Tsai CH, Cheng YW. MicroRNA expression profiling in PBMCs: a potential diagnostic biomarker of chronic hepatitis C. Dis Markers. 2014;2014:367157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 68. | Ralfkiaer U, Lindahl LM, Litman T, Gjerdrum LM, Ahler CB, Gniadecki R, Marstrand T, Fredholm S, Iversen L, Wasik MA. MicroRNA expression in early mycosis fungoides is distinctly different from atopic dermatitis and advanced cutaneous T-cell lymphoma. Anticancer Res. 2014;34:7207-7217. [PubMed] |
| 69. | Tafsiri E, Darbouy M, Shadmehr MB, Zagryazhskaya A, Alizadeh J, Karimipoor M. Expression of miRNAs in non-small-cell lung carcinomas and their association with clinicopathological features. Tumour Biol. 2015;36:1603-1612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 70. | Song J, Bai Z, Han W, Zhang J, Meng H, Bi J, Ma X, Han S, Zhang Z. Identification of suitable reference genes for qPCR analysis of serum microRNA in gastric cancer patients. Dig Dis Sci. 2012;57:897-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 223] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 71. | Chen L, Jiang M, Yuan W, Tang H. Prognostic value of miR-93 overexpression in resectable gastric adenocarcinomas. Acta Gastroenterol Belg. 2012;75:22-27. [PubMed] |
| 72. | Si H, Sun X, Chen Y, Cao Y, Chen S, Wang H, Hu C. Circulating microRNA-92a and microRNA-21 as novel minimally invasive biomarkers for primary breast cancer. J Cancer Res Clin Oncol. 2013;139:223-229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 195] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 73. | Du L, Zhao Z, Ma X, Hsiao TH, Chen Y, Young E, Suraokar M, Wistuba I, Minna JD, Pertsemlidis A. miR-93-directed downregulation of DAB2 defines a novel oncogenic pathway in lung cancer. Oncogene. 2014;33:4307-4315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 74. | Zhou H, Xiao B, Zhou F, Deng H, Zhang X, Lou Y, Gong Z, Du C, Guo J. MiR-421 is a functional marker of circulating tumor cells in gastric cancer patients. Biomarkers. 2012;17:104-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 75. | Wu J, Li G, Yao Y, Wang Z, Sun W, Wang J. MicroRNA-421 is a new potential diagnosis biomarker with higher sensitivity and specificity than carcinoembryonic antigen and cancer antigen 125 in gastric cancer. Biomarkers. 2015;20:58-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 76. | Wu JH, Yao YL, Gu T, Wang ZY, Pu XY, Sun WW, Zhang X, Jiang YB, Wang JJ. MiR-421 regulates apoptosis of BGC-823 gastric cancer cells by targeting caspase-3. Asian Pac J Cancer Prev. 2014;15:5463-5468. [PubMed] |
| 77. | Li Y, Li W, Zhang JG, Li HY, Li YM. Downregulation of tumor suppressor menin by miR-421 promotes proliferation and migration of neuroblastoma. Tumour Biol. 2014;35:10011-10017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 78. | Wang B, Li J, Sun M, Sun L, Zhang X. miRNA expression in breast cancer varies with lymph node metastasis and other clinicopathologic features. IUBMB Life. 2014;66:371-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 79. | Chen L, Tang Y, Wang J, Yan Z, Xu R. miR-421 induces cell proliferation and apoptosis resistance in human nasopharyngeal carcinoma via downregulation of FOXO4. Biochem Biophys Res Commun. 2013;435:745-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 80. | Mansour WY, Bogdanova NV, Kasten-Pisula U, Rieckmann T, Köcher S, Borgmann K, Baumann M, Krause M, Petersen C, Hu H. Aberrant overexpression of miR-421 downregulates ATM and leads to a pronounced DSB repair defect and clinical hypersensitivity in SKX squamous cell carcinoma. Radiother Oncol. 2013;106:147-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 59] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 81. | Liu H, Zhu L, Liu B, Yang L, Meng X, Zhang W, Ma Y, Xiao H. Genome-wide microRNA profiles identify miR-378 as a serum biomarker for early detection of gastric cancer. Cancer Lett. 2012;316:196-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 208] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 82. | Fei B, Wu H. MiR-378 inhibits progression of human gastric cancer MGC-803 cells by targeting MAPK1 in vitro. Oncol Res. 2012;20:557-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 83. | Chan JK, Kiet TK, Blansit K, Ramasubbaiah R, Hilton JF, Kapp DS, Matei D. MiR-378 as a biomarker for response to anti-angiogenic treatment in ovarian cancer. Gynecol Oncol. 2014;133:568-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 84. | Yin JY, Deng ZQ, Liu FQ, Qian J, Lin J, Tang Q, Wen XM, Zhou JD, Zhang YY, Zhu XW. Association between mir-24 and mir-378 in formalin-fixed paraffin-embedded tissues of breast cancer. Int J Clin Exp Pathol. 2014;7:4261-4267. [PubMed] |
| 85. | Redova M, Poprach A, Nekvindova J, Iliev R, Radova L, Lakomy R, Svoboda M, Vyzula R, Slaby O. Circulating miR-378 and miR-451 in serum are potential biomarkers for renal cell carcinoma. J Transl Med. 2012;10:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 213] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 86. | Chen LT, Xu SD, Xu H, Zhang JF, Ning JF, Wang SF. MicroRNA-378 is associated with non-small cell lung cancer brain metastasis by promoting cell migration, invasion and tumor angiogenesis. Med Oncol. 2012;29:1673-1680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 124] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 87. | Li C, Li JF, Cai Q, Qiu QQ, Yan M, Liu BY, Zhu ZG. MiRNA-199a-3p: A potential circulating diagnostic biomarker for early gastric cancer. J Surg Oncol. 2013;108:89-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 88. | Peng W, Chen ZY, Wang L, Wang Z, Li J. MicroRNA-199a-3p is downregulated in gastric carcinomas and modulates cell proliferation. Genet Mol Res. 2013;12:3038-3047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 89. | Wang Z, Ma X, Cai Q, Wang X, Yu B, Cai Q, liu B, Zhu Z, Li C. MiR-199a-3p promotes gastric cancer progression by targeting ZHX1. FEBS Lett. 2014;588:4504-4512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 90. | Zhou H, Guo JM, Lou YR, Zhang XJ, Zhong FD, Jiang Z, Cheng J, Xiao BX. Detection of circulating tumor cells in peripheral blood from patients with gastric cancer using microRNA as a marker. J Mol Med (Berl). 2010;88:709-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 115] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 91. | Zeng Q, Jin C, Chen W, Xia F, Wang Q, Fan F, Du J, Guo Y, Lin C, Yang K. Downregulation of serum miR-17 and miR-106b levels in gastric cancer and benign gastric diseases. Chin J Cancer Res. 2014;26:711-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 92. | Zhang Y, Han T, Wei G, Wang Y. Inhibition of microRNA-17/20a suppresses cell proliferation in gastric cancer by modulating UBE2C expression. Oncol Rep. 2015;33:2529-2536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 93. | Zhang X, Kong Y, Xu X, Xing H, Zhang Y, Han F, Li W, Yang Q, Zeng J, Jia J. F-box protein FBXO31 is down-regulated in gastric cancer and negatively regulated by miR-17 and miR-20a. Oncotarget. 2014;5:6178-6190. [PubMed] |
| 94. | Wang Z, Liu M, Zhu H, Zhang W, He S, Hu C, Quan L, Bai J, Xu N. miR-106a is frequently upregulated in gastric cancer and inhibits the extrinsic apoptotic pathway by targeting FAS. Mol Carcinog. 2013;52:634-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 95. | Zhu M, Zhang N, He S, Lui Y, Lu G, Zhao L. MicroRNA-106a targets TIMP2 to regulate invasion and metastasis of gastric cancer. FEBS Lett. 2014;588:600-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 96. | Cui L, Zhang X, Ye G, Zheng T, Song H, Deng H, Xiao B, Xia T, Yu X, Le Y. Gastric juice MicroRNAs as potential biomarkers for the screening of gastric cancer. Cancer. 2013;119:1618-1626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 124] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 97. | Oksuz Z, Serin MS, Kaplan E, Dogen A, Tezcan S, Aslan G, Emekdas G, Sezgin O, Altintas E, Tiftik EN. Serum microRNAs; miR-30c-5p, miR-223-3p, miR-302c-3p and miR-17-5p could be used as novel non-invasive biomarkers for HCV-positive cirrhosis and hepatocellular carcinoma. Mol Biol Rep. 2015;42:713-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 98. | Zeng X, Xiang J, Wu M, Xiong W, Tang H, Deng M, Li X, Liao Q, Su B, Luo Z. Circulating miR-17, miR-20a, miR-29c, and miR-223 combined as non-invasive biomarkers in nasopharyngeal carcinoma. PLoS One. 2012;7:e46367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 122] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 99. | Eichelser C, Flesch-Janys D, Chang-Claude J, Pantel K, Schwarzenbach H. Deregulated serum concentrations of circulating cell-free microRNAs miR-17, miR-34a, miR-155, and miR-373 in human breast cancer development and progression. Clin Chem. 2013;59:1489-1496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 153] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 100. | Lin HM, Castillo L, Mahon KL, Chiam K, Lee BY, Nguyen Q, Boyer MJ, Stockler MR, Pavlakis N, Marx G. Circulating microRNAs are associated with docetaxel chemotherapy outcome in castration-resistant prostate cancer. Br J Cancer. 2014;110:2462-2471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 117] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 101. | Rotkrua P, Shimada S, Mogushi K, Akiyama Y, Tanaka H, Yuasa Y. Circulating microRNAs as biomarkers for early detection of diffuse-type gastric cancer using a mouse model. Br J Cancer. 2013;108:932-940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 102. | Tchernitsa O, Kasajima A, Schäfer R, Kuban RJ, Ungethüm U, Györffy B, Neumann U, Simon E, Weichert W, Ebert MP. Systematic evaluation of the miRNA-ome and its downstream effects on mRNA expression identifies gastric cancer progression. J Pathol. 2010;222:310-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 106] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 103. | Petrocca F, Visone R, Onelli MR, Shah MH, Nicoloso MS, de Martino I, Iliopoulos D, Pilozzi E, Liu CG, Negrini M. E2F1-regulated microRNAs impair TGFbeta-dependent cell-cycle arrest and apoptosis in gastric cancer. Cancer Cell. 2008;13:272-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 678] [Cited by in RCA: 693] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 104. | Volinia S, Calin GA, Liu CG, Ambs S, Cimmino A, Petrocca F, Visone R, Iorio M, Roldo C, Ferracin M. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc Natl Acad Sci USA. 2006;103:2257-2261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4471] [Cited by in RCA: 4523] [Article Influence: 238.1] [Reference Citation Analysis (0)] |
| 105. | Chen X, Wang Y, Zang W, Du Y, Li M, Zhao G. miR-194 targets RBX1 gene to modulate proliferation and migration of gastric cancer cells. Tumour Biol. 2015;36:2393-2401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 106. | Zhang Y, Qu X, Li C, Fan Y, Che X, Wang X, Cai Y, Hu X, Liu Y. miR-103/107 modulates multidrug resistance in human gastric carcinoma by downregulating Cav-1. Tumour Biol. 2015;36:2277-2285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 107. | Li F, Liu B, Gao Y, Liu Y, Xu Y, Tong W, Zhang A. Upregulation of microRNA-107 induces proliferation in human gastric cancer cells by targeting the transcription factor FOXO1. FEBS Lett. 2014;588:538-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 108. | Kiga K, Mimuro H, Suzuki M, Shinozaki-Ushiku A, Kobayashi T, Sanada T, Kim M, Ogawa M, Iwasaki YW, Kayo H. Epigenetic silencing of miR-210 increases the proliferation of gastric epithelium during chronic Helicobacter pylori infection. Nat Commun. 2014;5:4497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 112] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 109. | Zhou C, Li G, Zhou J, Han N, Liu Z, Yin J. miR-107 activates ATR/Chk1 pathway and suppress cervical cancer invasion by targeting MCL1. PLoS One. 2014;9:e111860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 110. | Molina-Pinelo S, Carnero A, Rivera F, Estevez-Garcia P, Bozada JM, Limon ML, Benavent M, Gomez J, Pastor MD, Chaves M. MiR-107 and miR-99a-3p predict chemotherapy response in patients with advanced colorectal cancer. BMC Cancer. 2014;14:656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 111. | Geng L, Sun B, Gao B, Wang Z, Quan C, Wei F, Fang XD. MicroRNA-103 promotes colorectal cancer by targeting tumor suppressor DICER and PTEN. Int J Mol Sci. 2014;15:8458-8472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 112. | Wang ZH, Ren LL, Zheng P, Zheng HM, Yu YN, Wang JL, Lin YW, Chen YX, Ge ZZ, Chen XY. miR-194 as a predictor for adenoma recurrence in patients with advanced colorectal adenoma after polypectomy. Cancer Prev Res (Phila). 2014;7:607-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 113. | Zhai H, Karaayvaz M, Dong P, Sakuragi N, Ju J. Prognostic significance of miR-194 in endometrial cancer. Biomark Res. 2013;1:pii 12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 114. | Tsujiura M, Komatsu S, Ichikawa D, Shiozaki A, Konishi H, Takeshita H, Moriumura R, Nagata H, Kawaguchi T, Hirajima S. Circulating miR-18a in plasma contributes to cancer detection and monitoring in patients with gastric cancer. Gastric Cancer. 2015;18:271-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 115. | Su ZX, Zhao J, Rong ZH, Wu YG, Geng WM, Qin CK. Diagnostic and prognostic value of circulating miR-18a in the plasma of patients with gastric cancer. Tumour Biol. 2014;35:12119-12125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 116. | Wu W, Takanashi M, Borjigin N, Ohno SI, Fujita K, Hoshino S, Osaka Y, Tsuchida A, Kuroda M. MicroRNA-18a modulates STAT3 activity through negative regulation of PIAS3 during gastric adenocarcinogenesis. Br J Cancer. 2013;108:653-661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 86] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 117. | Komatsu S, Ichikawa D, Takeshita H, Morimura R, Hirajima S, Tsujiura M, Kawaguchi T, Miyamae M, Nagata H, Konishi H. Circulating miR-18a: a sensitive cancer screening biomarker in human cancer. In Vivo. 2014;28:293-297. [PubMed] |
| 118. | Li BS, Zhao YL, Guo G, Li W, Zhu ED, Luo X, Mao XH, Zou QM, Yu PW, Zuo QF. Plasma microRNAs, miR-223, miR-21 and miR-218, as novel potential biomarkers for gastric cancer detection. PLoS One. 2012;7:e41629. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 188] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 119. | Song J, Bai Z, Zhang J, Meng H, Cai J, Deng W, Bi J, Ma X, Zhang Z. Serum microRNA-21 levels are related to tumor size in gastric cancer patients but cannot predict prognosis. Oncol Lett. 2013;6:1733-1737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 120. | Kim SY, Jeon TY, Choi CI, Kim DH, Kim DH, Kim GH, Ryu DY, Lee BE, Kim HH. Validation of circulating miRNA biomarkers for predicting lymph node metastasis in gastric cancer. J Mol Diagn. 2013;15:661-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 121. | Zeng Z, Wang J, Zhao L, Hu P, Zhang H, Tang X, He D, Tang S, Zeng Z. Potential role of microRNA-21 in the diagnosis of gastric cancer: a meta-analysis. PLoS One. 2013;8:e73278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 122. | Gao C, Zhang Z, Liu W, Xiao S, Gu W, Lu H. Reduced microRNA-218 expression is associated with high nuclear factor kappa B activation in gastric cancer. Cancer. 2010;116:41-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 64] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 123. | Li L, Zhou L, Li Y, Lin S, Tomuleasa C. MicroRNA-21 stimulates gastric cancer growth and invasion by inhibiting the tumor suppressor effects of programmed cell death protein 4 and phosphatase and tensin homolog. J BUON. 2014;19:228-236. [PubMed] |
| 124. | Kang W, Tong JH, Chan AW, Lung RW, Chau SL, Wong QW, Wong N, Yu J, Cheng AS, To KF. Stathmin1 plays oncogenic role and is a target of microRNA-223 in gastric cancer. PLoS One. 2012;7:e33919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 80] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 125. | Shin VY, Jin H, Ng EK, Cheng AS, Chong WW, Wong CY, Leung WK, Sung JJ, Chu KM. NF-κB targets miR-16 and miR-21 in gastric cancer: involvement of prostaglandin E receptors. Carcinogenesis. 2011;32:240-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 128] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 126. | Zhang BG, Li JF, Yu BQ, Zhu ZG, Liu BY, Yan M. microRNA-21 promotes tumor proliferation and invasion in gastric cancer by targeting PTEN. Oncol Rep. 2012;27:1019-1026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 231] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 127. | Yang SM, Huang C, Li XF, Yu MZ, He Y, Li J. miR-21 confers cisplatin resistance in gastric cancer cells by regulating PTEN. Toxicology. 2013;306:162-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 173] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 128. | Wei Y, Yang J, Yi L, Wang Y, Dong Z, Liu Z, Ou-yang S, Wu H, Zhong Z, Yin Z. MiR-223-3p targeting SEPT6 promotes the biological behavior of prostate cancer. Sci Rep. 2014;4:7546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 129. | Yang T, Zheng ZM, Li XN, Li ZF, Wang Y, Geng YF, Bai L, Zhang XB. MiR-223 modulates multidrug resistance via downregulation of ABCB1 in hepatocellular carcinoma cells. Exp Biol Med (Maywood). 2013;238:1024-1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 130. | Li P, Mao WM, Zheng ZG, Dong ZM, Ling ZQ. Down-regulation of PTEN expression modulated by dysregulated miR-21 contributes to the progression of esophageal cancer. Dig Dis Sci. 2013;58:3483-3493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 131. | Sher YP, Wang LJ, Chuang LL, Tsai MH, Kuo TT, Huang CC, Chuang EY, Lai LC. ADAM9 up-regulates N-cadherin via miR-218 suppression in lung adenocarcinoma cells. PLoS One. 2014;9:e94065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 132. | Xu J, Yao Q, Hou Y, Xu M, Liu S, Yang L, Zhang L, Xu H. MiR-223/Ect2/p21 signaling regulates osteosarcoma cell cycle progression and proliferation. Biomed Pharmacother. 2013;67:381-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 133. | Shiotani A, Murao T, Kimura Y, Matsumoto H, Kamada T, Kusunoki H, Inoue K, Uedo N, Iishi H, Haruma K. Identification of serum miRNAs as novel non-invasive biomarkers for detection of high risk for early gastric cancer. Br J Cancer. 2013;109:2323-2330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 78] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 134. | Zhang R, Wang W, Li F, Zhang H, Liu J. MicroRNA-106b~25 expressions in tumor tissues and plasma of patients with gastric cancers. Med Oncol. 2014;31:243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 135. | Yao YL, Wu XY, Wu JH, Gu T, Chen L, Gu JH, Liu Y, Zhang QH. Effects of microRNA-106 on proliferation of gastric cancer cell through regulating p21 and E2F5. Asian Pac J Cancer Prev. 2013;14:2839-2843. [PubMed] |
| 136. | Yang TS, Yang XH, Chen X, Wang XD, Hua J, Zhou DL, Zhou B, Song ZS. MicroRNA-106b in cancer-associated fibroblasts from gastric cancer promotes cell migration and invasion by targeting PTEN. FEBS Lett. 2014;588:2162-2169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 137. | Wang J, Wang Q, Liu H, Hu B, Zhou W, Cheng Y. MicroRNA expression and its implication for the diagnosis and therapeutic strategies of gastric cancer. Cancer Lett. 2010;297:137-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 138. | Song MY, Pan KF, Su HJ, Zhang L, Ma JL, Li JY, Yuasa Y, Kang D, Kim YS, You WC. Identification of serum microRNAs as novel non-invasive biomarkers for early detection of gastric cancer. PLoS One. 2012;7:e33608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 159] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 139. | Chun-Zhi Z, Lei H, An-Ling Z, Yan-Chao F, Xiao Y, Guang-Xiu W, Zhi-Fan J, Pei-Yu P, Qing-Yu Z, Chun-Sheng K. MicroRNA-221 and microRNA-222 regulate gastric carcinoma cell proliferation and radioresistance by targeting PTEN. BMC Cancer. 2010;10:367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 321] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 140. | Wang M, Zhao C, Shi H, Zhang B, Zhang L, Zhang X, Wang S, Wu X, Yang T, Huang F. Deregulated microRNAs in gastric cancer tissue-derived mesenchymal stem cells: novel biomarkers and a mechanism for gastric cancer. Br J Cancer. 2014;110:1199-1210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 190] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 141. | Cuk K, Zucknick M, Madhavan D, Schott S, Golatta M, Heil J, Marmé F, Turchinovich A, Sinn P, Sohn C. Plasma microRNA panel for minimally invasive detection of breast cancer. PLoS One. 2013;8:e76729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 105] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 142. | Wang WN, Chen Y, Zhang YD, Hu TH. The regulatory mechanism of CCR7 gene expression and its involvement in the metastasis and progression of gastric cancer. Tumour Biol. 2013;34:1865-1871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 143. | Tsujiura M, Ichikawa D, Komatsu S, Shiozaki A, Takeshita H, Kosuga T, Konishi H, Morimura R, Deguchi K, Fujiwara H. Circulating microRNAs in plasma of patients with gastric cancers. Br J Cancer. 2010;102:1174-1179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 449] [Cited by in RCA: 509] [Article Influence: 33.9] [Reference Citation Analysis (0)] |
| 144. | Li Y, Liu H, Lai C, Du X, Su Z, Gao S. The Lin28/let-7a/c-Myc pathway plays a role in non-muscle invasive bladder cancer. Cell Tissue Res. 2013;354:533-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 145. | Luu C, Heinrich EL, Duldulao M, Arrington AK, Fakih M, Garcia-Aguilar J, Kim J. TP53 and let-7a micro-RNA regulate K-Ras activity in HCT116 colorectal cancer cells. PLoS One. 2013;8:e70604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 146. | Wang YY, Ren T, Cai YY, He XY. MicroRNA let-7a inhibits the proliferation and invasion of nonsmall cell lung cancer cell line 95D by regulating K-Ras and HMGA2 gene expression. Cancer Biother Radiopharm. 2013;28:131-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 147. | Cai K, Wan Y, Sun G, Shi L, Bao X, Wang Z. Let-7a inhibits proliferation and induces apoptosis by targeting EZH2 in nasopharyngeal carcinoma cells. Oncol Rep. 2012;28:2101-2106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 148. | Wu XQ, Huang C, Liu XH, Li J. MicroRNA let-7a: a novel therapeutic candidate in prostate cancer. Asian J Androl. 2014;16:327-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 149. | Lu L, Schwartz P, Scarampi L, Rutherford T, Canuto EM, Yu H, Katsaros D. MicroRNA let-7a: a potential marker for selection of paclitaxel in ovarian cancer management. Gynecol Oncol. 2011;122:366-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 150. | Yuan R, Zhi Q, Zhao H, Han Y, Gao L, Wang B, Kou Z, Guo Z, He S, Xue X. Upregulated expression of miR-106a by DNA hypomethylation plays an oncogenic role in hepatocellular carcinoma. Tumour Biol. 2015;36:3093-3100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 151. | Huh JH, Kim TH, Kim K, Song JA, Jung YJ, Jeong JY, Lee MJ, Kim YK, Lee DH, An HJ. Dysregulation of miR-106a and miR-591 confers paclitaxel resistance to ovarian cancer. Br J Cancer. 2013;109:452-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 94] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 152. | Chen Y, Jacamo R, Konopleva M, Garzon R, Croce C, Andreeff M. CXCR4 downregulation of let-7a drives chemoresistance in acute myeloid leukemia. J Clin Invest. 2013;123:2395-2407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 161] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 153. | Hamano R, Miyata H, Yamasaki M, Kurokawa Y, Hara J, Moon JH, Nakajima K, Takiguchi S, Fujiwara Y, Mori M. Overexpression of miR-200c induces chemoresistance in esophageal cancers mediated through activation of the Akt signaling pathway. Clin Cancer Res. 2011;17:3029-3038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 200] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 154. | Bhutia YD, Hung SW, Krentz M, Patel D, Lovin D, Manoharan R, Thomson JM, Govindarajan R. Differential processing of let-7a precursors influences RRM2 expression and chemosensitivity in pancreatic cancer: role of LIN-28 and SET oncoprotein. PLoS One. 2013;8:e53436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 155. | Zhang WH, Gui JH, Wang CZ, Chang Q, Xu SP, Cai CH, Li YN, Tian YP, Yan L, Wu B. The identification of miR-375 as a potential biomarker in distal gastric adenocarcinoma. Oncol Res. 2012;20:139-147. [PubMed] |
| 156. | Xu Y, Jin J, Liu Y, Huang Z, Deng Y, You T, Zhou T, Si J, Zhuo W. Snail-regulated MiR-375 inhibits migration and invasion of gastric cancer cells by targeting JAK2. PLoS One. 2014;9:e99516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 157. | Shen ZY, Zhang ZZ, Liu H, Zhao EH, Cao H. miR-375 inhibits the proliferation of gastric cancer cells by repressing ERBB2 expression. Exp Ther Med. 2014;7:1757-1761. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 158. | Miao L, Liu K, Xie M, Xing Y, Xi T. miR-375 inhibits Helicobacter pylori-induced gastric carcinogenesis by blocking JAK2-STAT3 signaling. Cancer Immunol Immunother. 2014;63:699-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 159. | Liu Y, Xing R, Zhang X, Dong W, Zhang J, Yan Z, Li W, Cui J, Lu Y. miR-375 targets the p53 gene to regulate cellular response to ionizing radiation and etoposide in gastric cancer cells. DNA Repair (Amst). 2013;12:741-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 160. | Ding L, Xu Y, Zhang W, Deng Y, Si M, Du Y, Yao H, Liu X, Ke Y, Si J. MiR-375 frequently downregulated in gastric cancer inhibits cell proliferation by targeting JAK2. Cell Res. 2010;20:784-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 281] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 161. | Yin J, Hou P, Wu Z, Wang T, Nie Y. Circulating miR-375 and miR-199a-3p as potential biomarkers for the diagnosis of hepatocellular carcinoma. Tumour Biol. 2015;36:4501-4507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 162. | Kachakova D, Mitkova A, Popov E, Popov I, Vlahova A, Dikov T, Christova S, Mitev V, Slavov C, Kaneva R. Combinations of serum prostate-specific antigen and plasma expression levels of let-7c, miR-30c, miR-141, and miR-375 as potential better diagnostic biomarkers for prostate cancer. DNA Cell Biol. 2015;34:189-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 94] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 163. | Yu H, Jiang L, Sun C, Li Guo L, Lin M, Huang J, Zhu L. Decreased circulating miR-375: a potential biomarker for patients with non-small-cell lung cancer. Gene. 2014;534:60-65. [PubMed] |
| 164. | Kawaguchi T, Komatsu S, Ichikawa D, Morimura R, Tsujiura M, Konishi H, Takeshita H, Nagata H, Arita T, Hirajima S. Clinical impact of circulating miR-221 in plasma of patients with pancreatic cancer. Br J Cancer. 2013;108:361-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 183] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 165. | Zhang XL, Shi HJ, Wang JP, Tang HS, Wu YB, Fang ZY, Cui SZ, Wang LT. MicroRNA-218 is upregulated in gastric cancer after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy and increases chemosensitivity to cisplatin. World J Gastroenterol. 2014;20:11347-11355. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 37] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 166. | Xin SY, Feng XS, Zhou LQ, Sun JJ, Gao XL, Yao GL. Reduced expression of circulating microRNA-218 in gastric cancer and correlation with tumor invasion and prognosis. World J Gastroenterol. 2014;20:6906-6911. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 35] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 167. | Tie J, Pan Y, Zhao L, Wu K, Liu J, Sun S, Guo X, Wang B, Gang Y, Zhang Y. MiR-218 inhibits invasion and metastasis of gastric cancer by targeting the Robo1 receptor. PLoS Genet. 2010;6:e1000879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 370] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 168. | Gao C, Pang M, Zhou Z, Long S, Dong D, Yang J, Cao M, Zhang C, Han S, Li L. Epidermal growth factor receptor-coamplified and overexpressed protein (VOPP1) is a putative oncogene in gastric cancer. Clin Exp Med. 2014;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 169. | Tian H, Hou L, Xiong YM, Huang JX, She YJ, Bi XB, Song XR. miR-218 suppresses tumor growth and enhances the chemosensitivity of esophageal squamous cell carcinoma to cisplatin. Oncol Rep. 2015;33:981-989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 170. | Wang HT, Liu AG, Luo DS, Zhou ZN, Lin HG, Chen RZ, He JS, Chen K. miR-218 expression in osteosarcoma tissues and its effect on cell growth in osteosarcoma cells. Asian Pac J Trop Med. 2014;7:1000-1004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 171. | Han G, Fan M, Zhang X. microRNA-218 inhibits prostate cancer cell growth and promotes apoptosis by repressing TPD52 expression. Biochem Biophys Res Commun. 2015;456:804-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 172. | Cai H, Yuan Y, Hao YF, Guo TK, Wei X, Zhang YM. Plasma microRNAs serve as novel potential biomarkers for early detection of gastric cancer. Med Oncol. 2013;30:452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 79] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 173. | Tsai KW, Liao YL, Wu CW, Hu LY, Li SC, Chan WC, Ho MR, Lai CH, Kao HW, Fang WL. Aberrant expression of miR-196a in gastric cancers and correlation with recurrence. Genes Chromosomes Cancer. 2012;51:394-401. [PubMed] |
| 174. | Chen Q, Ge X, Zhang Y, Xia H, Yuan D, Tang Q, Chen L, Pang X, Leng W, Bi F. Plasma miR-122 and miR-192 as potential novel biomarkers for the early detection of distant metastasis of gastric cancer. Oncol Rep. 2014;31:1863-1870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 78] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 175. | Gorur A, Balci Fidanci S, Dogruer Unal N, Ayaz L, Akbayir S, Yildirim Yaroglu H, Dirlik M, Serin MS, Tamer L. Determination of plasma microRNA for early detection of gastric cancer. Mol Biol Rep. 2013;40:2091-2096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 176. | Wang M, Gu H, Wang S, Qian H, Zhu W, Zhang L, Zhao C, Tao Y, Xu W. Circulating miR-17-5p and miR-20a: molecular markers for gastric cancer. Mol Med Rep. 2012;5:1514-1520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 76] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 177. | Duttagupta R, Jiang R, Gollub J, Getts RC, Jones KW. Impact of cellular miRNAs on circulating miRNA biomarker signatures. PLoS One. 2011;6:e20769. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 248] [Cited by in RCA: 249] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 178. | Arita T, Ichikawa D, Konishi H, Komatsu S, Shiozaki A, Shoda K, Kawaguchi T, Hirajima S, Nagata H, Kubota T. Circulating long non-coding RNAs in plasma of patients with gastric cancer. Anticancer Res. 2013;33:3185-3193. [PubMed] |
| 179. | Yang F, Bi J, Xue X, Zheng L, Zhi K, Hua J, Fang G. Up-regulated long non-coding RNA H19 contributes to proliferation of gastric cancer cells. FEBS J. 2012;279:3159-3165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 325] [Cited by in RCA: 376] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 180. | Li H, Yu B, Li J, Su L, Yan M, Zhu Z, Liu B. Overexpression of lncRNA H19 enhances carcinogenesis and metastasis of gastric cancer. Oncotarget. 2014;5:2318-2329. [PubMed] |
| 181. | Zhuang M, Gao W, Xu J, Wang P, Shu Y. The long non-coding RNA H19-derived miR-675 modulates human gastric cancer cell proliferation by targeting tumor suppressor RUNX1. Biochem Biophys Res Commun. 2014;448:315-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 171] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 182. | Zhu M, Chen Q, Liu X, Sun Q, Zhao X, Deng R, Wang Y, Huang J, Xu M, Yan J. lncRNA H19/miR-675 axis represses prostate cancer metastasis by targeting TGFBI. FEBS J. 2014;281:3766-3775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 258] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 183. | Shi Y, Wang Y, Luan W, Wang P, Tao T, Zhang J, Qian J, Liu N, You Y. Long non-coding RNA H19 promotes glioma cell invasion by deriving miR-675. PLoS One. 2014;9:e86295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 202] [Cited by in RCA: 250] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 184. | Luo M, Li Z, Wang W, Zeng Y, Liu Z, Qiu J. Long non-coding RNA H19 increases bladder cancer metastasis by associating with EZH2 and inhibiting E-cadherin expression. Cancer Lett. 2013;333:213-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 400] [Cited by in RCA: 461] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 185. | Glover AR, Zhao JT, Ip JC, Lee JC, Robinson BG, Gill AJ, Soon PS, Sidhu SB. Long noncoding RNA profiles of adrenocortical cancer can be used to predict recurrence. Endocr Relat Cancer. 2015;22:99-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 186. | Medrzycki M, Zhang Y, Zhang W, Cao K, Pan C, Lailler N, McDonald JF, Bouhassira EE, Fan Y. Histone h1.3 suppresses h19 noncoding RNA expression and cell growth of ovarian cancer cells. Cancer Res. 2014;74:6463-6473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 187. | Pang Q, Ge J, Shao Y, Sun W, Song H, Xia T, Xiao B, Guo J. Increased expression of long intergenic non-coding RNA LINC00152 in gastric cancer and its clinical significance. Tumour Biol. 2014;35:5441-5447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 142] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 188. | Cao WJ, Wu HL, He BS, Zhang YS, Zhang ZY. Analysis of long non-coding RNA expression profiles in gastric cancer. World J Gastroenterol. 2013;19:3658-3664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 144] [Cited by in RCA: 170] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 189. | Li Q, Shao Y, Zhang X, Zheng T, Miao M, Qin L, Wang B, Ye G, Xiao B, Guo J. Plasma long noncoding RNA protected by exosomes as a potential stable biomarker for gastric cancer. Tumour Biol. 2015;36:2007-2012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 320] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 190. | Liu Z, Shao Y, Tan L, Shi H, Chen S, Guo J. Clinical significance of the low expression of FER1L4 in gastric cancer patients. Tumour Biol. 2014;35:9613-9617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 191. | Song H, Sun W, Ye G, Ding X, Liu Z, Zhang S, Xia T, Xiao B, Xi Y, Guo J. Long non-coding RNA expression profile in human gastric cancer and its clinical significances. J Transl Med. 2013;11:225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 197] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 192. | Xia T, Liao Q, Jiang X, Shao Y, Xiao B, Xi Y, Guo J. Long noncoding RNA associated-competing endogenous RNAs in gastric cancer. Sci Rep. 2014;4:6088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 246] [Cited by in RCA: 331] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 193. | Dong L, Qi P, Xu MD, Ni SJ, Huang D, Xu QH, Weng WW, Tan C, Sheng WQ, Zhou XY. Circulating CUDR, LSINCT-5 and PTENP1 long noncoding RNAs in sera distinguish patients with gastric cancer from healthy controls. Int J Cancer. 2015;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 133] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 194. | Zheng Q, Wu F, Dai WY, Zheng DC, Zheng C, Ye H, Zhou B, Chen JJ, Chen P. Aberrant expression of UCA1 in gastric cancer and its clinical significance. Clin Transl Oncol. 2015;17:640-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 108] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 195. | Lin XC, Zhu Y, Chen WB, Lin LW, Chen DH, Huang JR, Pan K, Lin Y, Wu BT, Dai Y. Integrated analysis of long non-coding RNAs and mRNA expression profiles reveals the potential role of lncRNAs in gastric cancer pathogenesis. Int J Oncol. 2014;45:619-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 196. | Xu MD, Qi P, Weng WW, Shen XH, Ni SJ, Dong L, Huang D, Tan C, Sheng WQ, Zhou XY. Long non-coding RNA LSINCT5 predicts negative prognosis and exhibits oncogenic activity in gastric cancer. Medicine (Baltimore). 2014;93:e303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 197. | Yu G, Yao W, Gumireddy K, Li A, Wang J, Xiao W, Chen K, Xiao H, Li H, Tang K. Pseudogene PTENP1 functions as a competing endogenous RNA to suppress clear-cell renal cell carcinoma progression. Mol Cancer Ther. 2014;13:3086-3097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 175] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 198. | Chen CL, Tseng YW, Wu JC, Chen GY, Lin KC, Hwang SM, Hu YC. Suppression of hepatocellular carcinoma by baculovirus-mediated expression of long non-coding RNA PTENP1 and MicroRNA regulation. Biomaterials. 2015;44:71-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 182] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 199. | Kovalenko TF, Sorokina AV, Ozolinia LA, Patrushev LI. Pseudogene PTENP1 5’-region methylation in endometrial cancer and hyperplasias. Bioorg Khim. 2013;39:445-453. [PubMed] |
| 200. | Shao Y, Ye M, Jiang X, Sun W, Ding X, Liu Z, Ye G, Zhang X, Xiao B, Guo J. Gastric juice long noncoding RNA used as a tumor marker for screening gastric cancer. Cancer. 2014;120:3320-3328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 149] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 201. | Skog J, Würdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, Curry WT, Carter BS, Krichevsky AM, Breakefield XO. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol. 2008;10:1470-1476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4100] [Cited by in RCA: 3944] [Article Influence: 232.0] [Reference Citation Analysis (0)] |
| 202. | Kosaka N, Iguchi H, Yoshioka Y, Takeshita F, Matsuki Y, Ochiya T. Secretory mechanisms and intercellular transfer of microRNAs in living cells. J Biol Chem. 2010;285:17442-17452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1382] [Cited by in RCA: 1590] [Article Influence: 106.0] [Reference Citation Analysis (0)] |
| 203. | Zernecke A, Bidzhekov K, Noels H, Shagdarsuren E, Gan L, Denecke B, Hristov M, Köppel T, Jahantigh MN, Lutgens E. Delivery of microRNA-126 by apoptotic bodies induces CXCL12-dependent vascular protection. Sci Signal. 2009;2:ra81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 959] [Cited by in RCA: 1053] [Article Influence: 65.8] [Reference Citation Analysis (0)] |
| 204. | Vickers KC, Palmisano BT, Shoucri BM, Shamburek RD, Remaley AT. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat Cell Biol. 2011;13:423-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2292] [Cited by in RCA: 2188] [Article Influence: 156.3] [Reference Citation Analysis (0)] |
| 205. | Wang K, Zhang S, Weber J, Baxter D, Galas DJ. Export of microRNAs and microRNA-protective protein by mammalian cells. Nucleic Acids Res. 2010;38:7248-7259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 750] [Cited by in RCA: 813] [Article Influence: 54.2] [Reference Citation Analysis (0)] |
| 206. | Ji X, Takahashi R, Hiura Y, Hirokawa G, Fukushima Y, Iwai N. Plasma miR-208 as a biomarker of myocardial injury. Clin Chem. 2009;55:1944-1949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 364] [Cited by in RCA: 371] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 207. | Wang K, Zhang S, Marzolf B, Troisch P, Brightman A, Hu Z, Hood LE, Galas DJ. Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proc Natl Acad Sci USA. 2009;106:4402-4407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 891] [Cited by in RCA: 946] [Article Influence: 59.1] [Reference Citation Analysis (0)] |
| 208. | Hornby RJ, Starkey Lewis P, Dear J, Goldring C, Park BK. MicroRNAs as potential circulating biomarkers of drug-induced liver injury: key current and future issues for translation to humans. Expert Rev Clin Pharmacol. 2014;7:349-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 209. | Ramachandran K, Saikumar J, Bijol V, Koyner JL, Qian J, Betensky RA, Waikar SS, Vaidya VS. Human miRNome profiling identifies microRNAs differentially present in the urine after kidney injury. Clin Chem. 2013;59:1742-1752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 107] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 210. | Huang X, Yuan T, Tschannen M, Sun Z, Jacob H, Du M, Liang M, Dittmar RL, Liu Y, Liang M. Characterization of human plasma-derived exosomal RNAs by deep sequencing. BMC Genomics. 2013;14:319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 765] [Cited by in RCA: 811] [Article Influence: 67.6] [Reference Citation Analysis (0)] |
| 211. | Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200:373-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4900] [Cited by in RCA: 6160] [Article Influence: 513.3] [Reference Citation Analysis (0)] |
| 212. | Mo MH, Chen L, Fu Y, Wang W, Fu SW. Cell-free Circulating miRNA Biomarkers in Cancer. J Cancer. 2012;3:432-448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 118] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 213. | McDonald JS, Milosevic D, Reddi HV, Grebe SK, Algeciras-Schimnich A. Analysis of circulating microRNA: preanalytical and analytical challenges. Clin Chem. 2011;57:833-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 492] [Cited by in RCA: 521] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 214. | Accerbi M, Schmidt SA, De Paoli E, Park S, Jeong DH, Green PJ. Methods for isolation of total RNA to recover miRNAs and other small RNAs from diverse species. Methods Mol Biol. 2010;592:31-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 215. | Baker M. MicroRNA profiling: separating signal from noise. Nat Methods. 2010;7:687-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 174] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 216. | Git A, Dvinge H, Salmon-Divon M, Osborne M, Kutter C, Hadfield J, Bertone P, Caldas C. Systematic comparison of microarray profiling, real-time PCR, and next-generation sequencing technologies for measuring differential microRNA expression. RNA. 2010;16:991-1006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 521] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 217. | De Guire V, Robitaille R, Tétreault N, Guérin R, Ménard C, Bambace N, Sapieha P. Circulating miRNAs as sensitive and specific biomarkers for the diagnosis and monitoring of human diseases: promises and challenges. Clin Biochem. 2013;46:846-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 174] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 218. | Chen X, Liang H, Guan D, Wang C, Hu X, Cui L, Chen S, Zhang C, Zhang J, Zen K. A combination of Let-7d, Let-7g and Let-7i serves as a stable reference for normalization of serum microRNAs. PLoS One. 2013;8:e79652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 89] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 219. | Kraus TF, Greiner A, Guibourt V, Lisec K, Kretzschmar HA. Identification of Stably Expressed lncRNAs as Valid Endogenous Controls for Profiling of Human Glioma. J Cancer. 2015;6:111-119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 220. | Dunnett H, van der Meer D, Williams GA. Evaluation of stem-loop reverse transcription and poly-A tail extension in microRNA analysis of body fluids. Microrna. 2014;3:150-154. [PubMed] |
| 221. | Chugh P, Dittmer DP. Potential pitfalls in microRNA profiling. Wiley Interdiscip Rev RNA. 2012;3:601-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 146] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 222. | Speth C, Laubinger S. Rapid and parallel quantification of small and large RNA species. Methods Mol Biol. 2014;1158:93-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 223. | Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5015] [Cited by in RCA: 4858] [Article Influence: 441.6] [Reference Citation Analysis (2)] |
| 224. | Zhang XM, Ma ZW, Wang Q, Wang JN, Yang JW, Li XD, Li H, Men TY. A new RNA-seq method to detect the transcription and non-coding RNA in prostate cancer. Pathol Oncol Res. 2014;20:43-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |