Published online Aug 28, 2015. doi: 10.3748/wjg.v21.i32.9614
Peer-review started: April 4, 2015
First decision: April 23, 2015
Revised: May 21, 2015
Accepted: July 8, 2015
Article in press: July 8, 2015
Published online: August 28, 2015
Processing time: 147 Days and 17.3 Hours
AIM: To establish a new model for predicting survival in acute-on-chronic liver failure (ACLF) patients treated with an artificial liver support system.
METHODS: One hundred and eighty-one ACLF patients who were admitted to the hospital from January 1, 2012 to December 31, 2014 and were treated with an artificial liver support system were enrolled in this retrospective study, including a derivation cohort (n = 113) and a validation cohort (n = 68). Laboratory parameters at baseline were analyzed and correlated with clinical outcome. In addition to standard medical therapy, ACLF patients underwent plasma exchange (PE) or plasma bilirubin adsorption (PBA) combined with plasma exchange. For the derivation cohort, Kaplan-Meier methods were used to estimate survival curves, and Cox regression was used in survival analysis to generate a prognostic model. The performance of the new model was tested in the validation cohort using a receiver-operator curve.
RESULTS: The mean overall survival for the derivation cohort was 441 d (95%CI: 379-504 d), and the 90- and 270-d survival probabilities were 70.3% and 58.3%, respectively. The mean survival times of patients treated with PBA plus PE and patients treated with PE were 531 d (95%CI: 455-605 d) and 343 d (95%CI: 254-432 d), respectively, which were significantly different (P = 0.012). When variables with bivariate significance were selected for inclusion into the multivariate Cox regression model, number of complications, age, scores of the model for end-stage liver disease (MELD) and type of artificial liver support system were defined as independent risk factors for survival in ACLF patients. This new prognostic model could accurately discriminate the outcome of patients with different scores in this cohort (P < 0.001). The model also had the ability to assign a predicted survival probability for individual patients. In the validation cohort, the new model remained better than the MELD.
CONCLUSION: A novel model was constructed to predict prognosis and accurately discriminate survival in ACLF patients treated with an artificial liver support system.
Core tip: Liver failure has a high mortality. The current prognostic model to estimate the survival in acute-on-chronic liver failure (ACLF) patients treated with an artificial liver support system (ALSS) is not fully characterized. The aim of this study was to establish a new scoring model and to test its ability to predict the survival of ACLF patients treated with ALSS. This prognostic model accurately differentiated the outcome of ACLF patients with different risk scores and also had the ability to assign a predicted survival probability for individual patients.
- Citation: Zhou PQ, Zheng SP, Yu M, He SS, Weng ZH. Prognosis of acute-on-chronic liver failure patients treated with artificial liver support system. World J Gastroenterol 2015; 21(32): 9614-9622
- URL: https://www.wjgnet.com/1007-9327/full/v21/i32/9614.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i32.9614
Liver failure (LF) can be classified as acute LF occurring without any potential liver diseases, acute-on-chronic liver failure (ACLF), which is caused by an acute exacerbation of chronic liver diseases resulting from virus, alcohol or drugs, and chronic decompensation in any type of end-stage liver disease. ACLF, the most common type of LF, constitutes a serious condition with a sophisticated etiology, diversified manifestations and a high short-term mortality[1]. So far, liver transplantation is identified as the most useful approach for ACLF; however, few patients benefit from this treatment due to the extreme lack of healthy livers and/or the costly operation[2].
In the past five decades, a variety of different types of artificial liver support systems (ALSS) have been developed to bridge patients with LF to liver transplantation or to support the failing liver temporarily, until it is able to regenerate. Although ALSS cannot substitute for the whole spectrum of liver functions, these methods can take the place of a few basic hepatic functions[3-7]. It has been demonstrated that some types of ALSS, such as plasma exchange (PE) and plasma bilirubin adsorption (PBA), are able to remove toxic substances, improve coagulopathy, and prevent bleeding. Although some studies have reported success in prolonging survival, the survival of patients with LF is variable, approximately 21%-60%[8-11].
Prognostic models have been used for estimating disease severity and survival and are of great importance for doctors to make therapeutic decisions. The current prognosis models for ACLF were only analyzing ACLF caused by HBV and might not be ideal for clinical practice because of the diversified etiologies of ACLF in Asia[12-14]. Moreover, these models were established based on data from standard medical treatment (SMT) and without considering the impact of ALSS on the prognosis of patients. Therefore, a feasible prognostic model is urgently needed to estimate the outcomes of patients with ACLF.
We have assessed the overall survival and the possible prognostic predictors in a cohort of ACLF patients treated with SMT together with ALSS. The aim of this study was to develop a novel model to supply reliable predictive information about these patients.
The present study was based on a retrospective cohort, including 181 patients with ACLF hospitalized between January 1, 2012 and December 31, 2014 at the Department of Infectious Diseases, Union Hospital, Wuhan, China. The ACLF patients hospitalized from January 1, 2012 to December 31, 2013 were included in a derivation cohort (n = 113). This dataset was used to create the prognostic model. Then, the new model was validated in another 68 patients hospitalized from January 1, 2014 to December 31, 2014.
Patients who met the diagnostic criteria for ACLF were hospitalized, and besides SMT, they were treated with PE or PBA combined with PE. In this study, liver transplantation was not available for ACLF patients owing to the extreme deficiency of healthy livers and/or the costly operation. ACLF patients with persistent bleeding, circulatory shock, severe bacterial infection, pregnancy, international normalized ratio (INR) ≥ 3.0 or platelet count ≤ 30000/μL were excluded for ALSS.
ACLF patients were randomly divided into either a PBA plus PE group or a PE only group. The follow-up began at the date of initial treatment of ALSS. In the derivation cohort, patients were followed until death or censored at the end point of January 31, 2014. While in the validation cohort, patients were followed until the end point of January 31, 2015. Medical history, physical examination, and auxiliary investigations, such as laboratory test, abdominal ultrasound or computed tomography (CT) scan, were finished at admission. Laboratory parameters included serum total bilirubin (TB), aspartate aminotransferase (AST), alanine aminotransferase (ALT), creatinine, and INR, and others. Adverse events and drugs received were documented during the whole study period.
The data of ACLF patients were analyzed anonymously based on the Declaration of Helsinki. This study was approved by the Ethics Committee of the Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, and all enrolled patients gave their written consent, which was collected in the hospital and could be used for research.
According to the recommendations generated by the Asian Pacific Association for the Study of the Liver, ACLF was defined as acute liver injury emerging as jaundice and coagulopathy, complicated by ascites and/or encephalopathy within 4 wk in a patient with known or unknown chronic liver disease[15]. The definition of LF in ACLF was as follows: severe jaundice (total serum bilirubin ≥ 5 mg/dL) and coagulopathy (INR ≥ 1.5 or prothrombin activity < 40%), which were indispensable, and ascites and/or encephalopathy, which were diagnosed by physical examination[15].
The diagnosis of hepatitis E virus (HEV) infection was made according to the detection of anti-HEV IgM and/or HEV RNA in serum[16]. Liver cirrhosis (LC) was diagnosed by the medical history, physical examination, and laboratory tests, together with ultrasonography or CT[17]. Hepatic encephalopathy (HE) was defined as neuropsychiatric abnormalities including the cognitive, affective, behavior and consciousness. It was diagnosed by clinical manifestations and brain edema identified by magnetic resonance imaging (MRI) or CT[18]. Hepatorenal syndrome (HRS) was defined as a functional renal failure according to the criteria created by the International Ascites Club[19]. Spontaneous bacterial peritonitis (SBP) was diagnosed by the examination of ascites[20]. Upper gastrointestinal bleeding commonly arose from the esophagus, stomach, or duodenum. In some cases, blood could be observed in vomit or in tarry stool[21].
SMT: SMT was intended to support the liver and to prevent and treat complications of ACLF. All patients were administered according to the following recommendations: absolute bed rest, the use of hepatocyte growth factor and adenosylmethionine to regenerate liver cells, the infusion of albumin, maintaining electrolyte or acid-base equilibrium and preventing and curing complications. Oral antiviral drugs including lamivudine, telbivudine, or entecavir were ordered for the patients with activated replication of hepatitis B virus.
In addition to SMT, ACLF patients in this group underwent treatment of PE, which was performed with a membrane plasma filter (Plasmaflo OP-08W, Asahi Kasei Kuraray Medical Co., Ltd., Tokyo, Japan). A double-lumen catheter was inserted into the right femoral vein of ACLF patients, and approximately 3000 mL of plasma was exchanged per time at a blood flow rate of 20 to 25 mL/min. Each patient in the derivation cohort received PE 1 to 4 times, and 93 times in total were performed in 54 patients. In the validation cohort, each patient received PE 1 to 4 times, and 62 times in total were performed in 33 patients.
Blood was separated by Plasmaflo OP-08W. Then, the plasma was passed through an adsorbent column (Plasorba BR-350, Asahi Kasei Kuraray Medical Co., Ltd., Tokyo, Japan). Approximately 2000-2500 mL of plasma was separated per time at a blood flow rate of 20 mL/min. Perfused over the adsorbent column, the plasma was returned to the patient after being merged with hemocytes coming from the plasma filter. After the PBA process, the patient was treated with PE using a total of 1500-2000 mL of fresh, frozen plasma. In the derivation cohort, each patient received PBA plus PE 1 to 4 times, and 135 times in total were performed in 59 patients. In the validation cohort, each patient received PBA plus PE 1 to 4 times, and 76 times in total were performed in 35 patients.
Continuous variables are shown as mean ± SD or median, and categorical variables are expressed by count. The period from the date of the initial ALSS to the date of death or loss to follow-up was defined as survival time. The comparison of the survival distributions in different groups of patients was determined by the Kaplan-Meier analysis. The model for end-stage liver disease (MELD) scores were calculated by the formula: 9.57 × ln[creatinine (mg/dL)] + 3.78 × ln[total bilirubin (mg/dL)] + 11.2 × ln(INR) + 6.43 (etiology: 0 if cholestatic or alcoholic, 1 otherwise)[22].
The variables acquired at baseline before the initial ALSS with P < 0.05 in the bivariate analysis were introduced to create a multivariate Cox regression analysis with a P value < 0.1 (using the backward conditional stepwise regression manner). P < 0.05 was considered significant with a CI of 95%. The receiver-operating characteristic (ROC) curve was used to describe the MELD and the Cox regression model. The performance of the model was determined by the concordance statistic (c-statistic), which was equal to the area under the ROC curve (AUC). A c-statistic > 0.7 was considered useful[23]. The predictive accuracy of the new model was examined in the validation cohort by calculating the c-statistic[23]. The AUCs were compared by the z-test. SPSS 18.0 (SPSS, Chicago, IL, United States) and MedCalc 11.4 (Mariakerke, Belgium) software programs were used for data analyses.
In the derivation cohort, a total of 113 ACLF patients were reviewed and registered into this study. Table 1 shows the demographics at baseline. The median age of these patients was 44.6 years, and 83% of patients were male. In this Asian cohort, hepatitis B virus was the prevailing etiology of liver disease. The average level of HBV DNA of patients infected with hepatitis B virus was (5.03 ± 2.11) log10 IU/mL. More than half of the patients (50.4%) had one complication, such as HE, SBP, HRS, upper gastrointestinal hemorrhage, or electrolyte disturbances, while others suffered with two or more complications. The median MELD score of these patients was 24.
| Variable | Derivation cohort | Validation cohort | P value |
| (n = 113) | (n = 68) | ||
| Age (yr) | 44.6 (22-81) | 46.1 (20-79) | 0.529 |
| Men | 94 (83) | 52 (76) | 0.268 |
| Etiology of liver failure | |||
| HBV | 64 (56.6) | 40 (59) | 0.773 |
| HBV + HEV | 36 (31.9) | 18 (26) | 0.443 |
| Other causes1 | 13 (11.5) | 10 (15) | 0.531 |
| MELD score | 24 (12-44) | 26 (13-46) | 0.659 |
| Total bilirubin (mg/dL) | 24.8 (5.9-51.8) | 23.1 (5.7-50.1) | 0.841 |
| ALT (IU/mL) | 262 (12-1757) | 211 (23-1519) | 0.169 |
| AST (IU/mL) | 232 (17-1634) | 206 (20-1301) | 0.326 |
| Creatinine (mg/dL) | 0.9 (0.3-6.8) | 1.0 (0.3-7.1) | 0.533 |
| INR | 2.4 (0.8-11.4) | 2.6 (1.0-16.1) | 0.285 |
| Cirrhosis | 58 (51.3) | 45 (66.2) | 0.051 |
| Number of complications | |||
| 1 | 57 (50.4) | 39 (57.4) | 0.367 |
| 2 | 27 (23.9) | 13 (19.1) | 0.453 |
| 3 | 20 (17.7) | 10 (14.7) | 0.600 |
| 4 | 8 (7.1) | 4 (5.9) | 0.996 |
| 5 | 1 (0.9) | 2 (2.9) | 0.654 |
| Type of ALSS | |||
| PE | 54 (47.8) | 33 (48.5) | 0.923 |
| PE + PBA | 59 (52.2) | 35 (51.5) | 0.923 |
One hundred and thirteen ACLF patients were treated with SMT plus PBA and/or PE therapy. No therapy-related adverse events, including severe hemorrhage, shock, or hypersensitivity, occurred in the PBA process, but a few allergies such as rash occurred in cases during PE. At the end point of this study, 65 patients were alive, 42 (37.2%) patients died, and 5 were lost to follow-up.
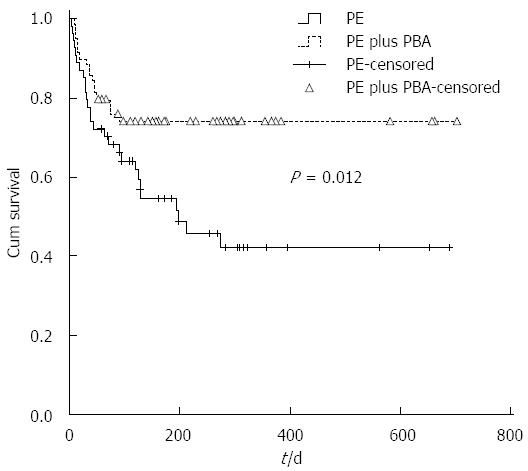
For the derivation cohort, the mean overall survival was 441 d (95%CI: 379-504 d), and the 90- and 270-d survival probabilities were 70.3% and 58.3%, respectively. The mean survival times of patients treated with SMT together with PBA plus PE and patients treated with SMT plus PE were 531 d (95%CI: 455-605 d) and 343 d (95%CI: 254-432 d), respectively, which were significantly different (P = 0.012, Figure 1).
Moreover, we investigated the correlation between survival time and clinical data, such as gender, age, etiology, numbers of complications, type of ALSS, and serum biomarkers tested at baseline, including TB, ALT, AST, INR and creatinine. In the bivariate analysis, some factors evaluated at baseline showed a predictive impact on overall survival, comprising the type of ALSS, age, number of complications, MELD score, TB, ALT, AST, and INR (Table 2).
| Variable | Univariate | Multivariate | |||
| HR (95%CI) | P value | HR (95%CI) | P value | Coefficient | |
| Age | 1.045 (1.024-1.067) | < 0.001 | 1.031 (1.008-1.054) | 0.008 | 0.030 |
| Gender1 | 0.704 (0.296-1.676) | 0.428 | |||
| Etiology2 | 0.736 (0.178-3.049) | 0.673 | |||
| MELD score | 1.120 (1.080-1.161) | < 0.001 | 1.102 (1.056-1.150) | < 0.001 | 0.097 |
| Total bilirubin | 1.002 (1.000-1.003) | 0.047 | |||
| ALT | 1.001 (1.000-1.002) | 0.022 | |||
| AST | 1.001 (1.000-1.002) | 0.011 | |||
| Creatinine | 1.003 (1.000-1.005) | 0.055 | |||
| BUN | 1.083 (1.015-1.157) | 0.017 | |||
| INR | 1.369 (1.209-1.550) | < 0.001 | |||
| Times3 | 0.753 (0.549-1.034) | 0.080 | |||
| Complication4 | 2.246 (1.721-2.933) | < 0.001 | 1.694 (1.224-2.344) | 0.001 | 0.527 |
| ALSS5 | 0.455 (0.242-0.856) | 0.015 | 0.454 (0.232-0.889) | 0.021 | -0.790 |
Significant variables in the bivariate analysis were selected into a multivariate Cox regression analysis, such as type of ALSS, age, number of complications and MELD score, to define independent predictive factors for survival (Table 2). According to the multivariate Cox regression analysis, a risk score (R) can be calculated by the equation: R = 0.03*(age) + 0.097*(MELD score) + 0.527*(the number of complications) - 0.79*(the type of ALSS).
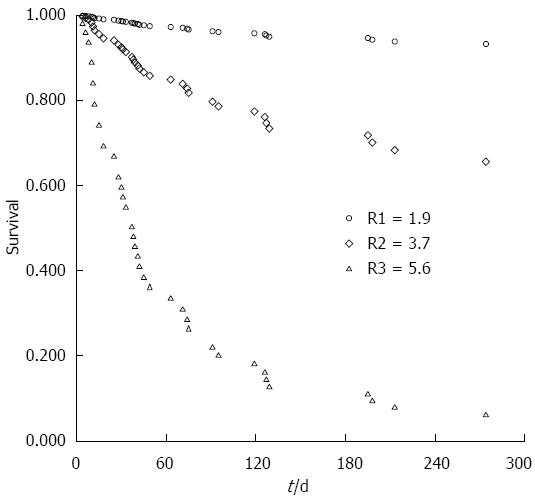
Further, using the means of covariates for (age, type of ALSS, MELD and number of complications) all of the patients in the derivation cohort, a mean R value of 3.7 could be calculated based on the formula. Simultaneously, the survival probabilities for an individual with an R value of 3.7 were estimated by the survival table created by SPSS (Table 3). If S0(t) was assigned to the estimated survival probabilities for a patient with an R value of 3.7, we can calculate the survival probabilities at t days for any patient by using the formula: S(t) = S0(t)exp(score-3.7), which was previously described[24]. Particularly, based on the score, the expected survival probability for an individual patient can be calculated. For instance, the 90- and 270-d survival probabilities for ACLF patients in the lowest quartile (R = 1.9) were 96.3% and 93.3%, respectively. However, in the highest quartile (R = 5.6), the survival probabilities sharply decreased to 21.9% and 6.0% at 90 and 270 d, respectively (Figure 2).
| Days | 30 | 60 | 90 | 120 | 180 | 270 |
| S0 (t) | 92.50% | 84.90% | 79.70% | 77.40% | 71.80% | 65.60% |
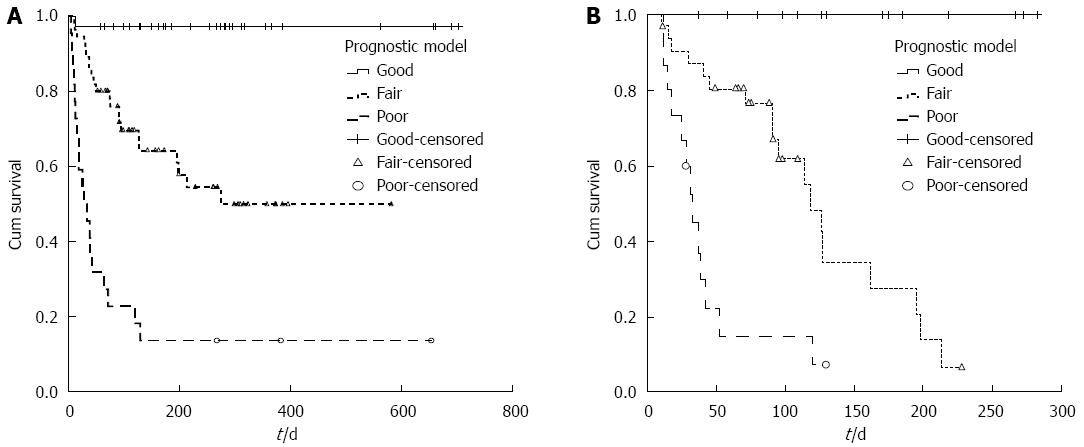
The new prognostic model also illustrated that ACLF patients who had lower R values might have a better survival probability. If the outcomes of patients with R values less than 3.0 were assigned as “Good”, patients with an R value more than 5.0 were “Poor”, and the rest with an R value between 3.0 and 5.0 were defined as “Fair”. There were significant differences in the survival probabilities of patients with “Good”, “Fair”, and “Poor” prognoses (P < 0.001, Figure 3A), and the mean survival times of these patients were 684, 344 and 120 d, respectively (P < 0.001).
The predictive accuracy of the new model was validated in another cohort of 68 ACLF patients. The clinical characteristics of the patients in the validation cohort are shown in Table 1. For the validation cohort, the c-statistic for the new prognostic model was 0.879 (95%CI: 0.799-0.959) (Figure 3B).
The classification of the outcome of patients in the derivation cohort based on the R value resulted in the assignment of 31.9% of the patients to the “Good” group (R < 3.0), 48.7% to the “Fair” group (3.0 ≤ R ≤ 5.0), and 19.4% to the “Poor” group (R > 5.0) (Table 4). Similar results were also found in the validation cohort: 30.9% of the patients were in the “Good” group, 47.1% in the “Fair” group, and 22.0% in the “Poor” group.
| Prognosis | Derivation cohort | Validation cohort | P value |
| (n = 113) | (n = 68) | ||
| Good | 36 (31.9) | 21 (30.9) | 0.891 |
| Fair | 55 (48.7) | 32 (47.1) | 0.833 |
| Poor | 22 (19.4) | 15 (22.0) | 0.676 |
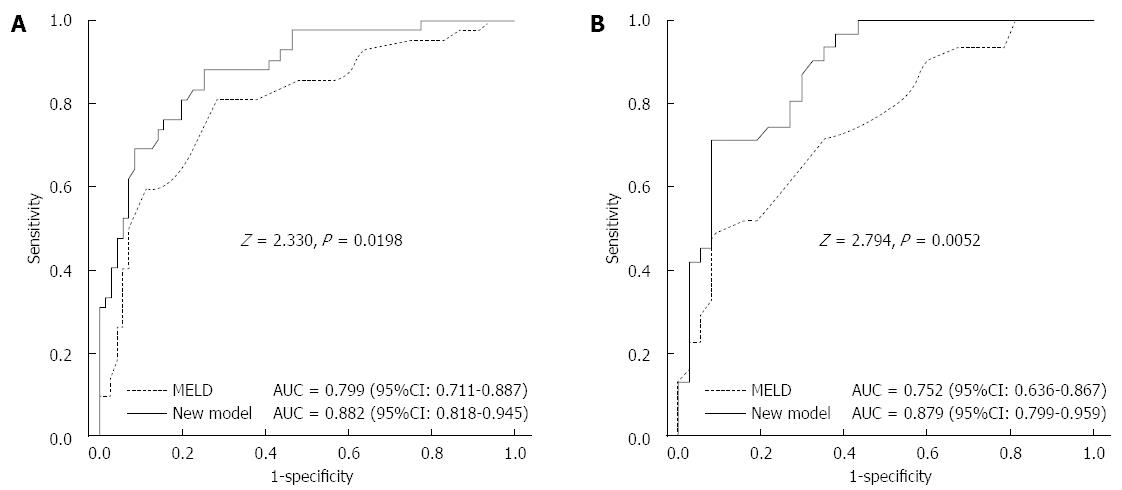
We further evaluated the predictive values of MELD scores and the new Cox model. The results showed that the c-statistics were 0.799 (95%CI: 0.711-0.887) and 0.882 (95%CI: 0.818-0.945), respectively, for these two scoring systems used in the derivation cohort. Furthermore, the AUC was obviously greater in the new prognostic model than in the MELD (z = 2.330, P = 0.0198) (Figure 4A). In the validation cohort, the c-statistics were 0.752 (95%CI: 0.636-0.867) and 0.879 (95%CI: 0.799-0.959), respectively, and the AUC was also greater than in the MELD (z = 2.794, P = 0.0052) (Figure 4B).
ACLF is distinguished by the acute exacerbation of liver function in patients with pre-existing chronic liver disease that occurs due to acute episodes, including both infectious and noninfectious causes. Alcohol and drugs make up the main acute events in the West; however, infectious causes are common in Asia. The reactivation of HBV infection is the major etiology of ACLF in Asia[25,26]. Another significant infectious cause of the acute episode is superinfection with HEV[27,28]. Because there are still no effective therapies available, except liver transplantation, ACLF is correlated with a poor prognosis[29]. Thus, ALSS had been developed to bridge patients with LF to liver transplantation. Early estimation of outcome is significant for differentiating ACLF patients who need liver transplantation from patients who would survive followed by ALSS. Current prognostic models based on parameters of clinical characteristics and the degree of liver dysfunction had been generated to evaluate the short-term survival probability of ACLF patients[12-14]. However, these models were usually created on the basis of the data at baseline from patients without ALSS treatment. Specifically, in the present study, we had screened the prognostic factors in a cohort of ACLF patients treated with ALSS besides SMT and established a scoring model that could accurately predict the prognosis in different groups of these patients.
It was reported that some types of ALSS such as PE or PBA could supply a beneficial internal environment for liver cells to restore liver functions in ACLF patients[30-33]. PE can separate and discard the plasma of LF patients to get rid of toxic substances and replenish it with normal plasma to supply several essential substances, such as coagulation factors and immunoglobulin[34,35]. However, PE requires exchanging plenty of fresh plasma and bears the risk of some potential infections. PBA could absorb conjugated and albumin-bound bilirubin from the plasma and had proved to be an effective treatment for LF patients[36,37]. PE and PBA have similar effects in lowering bilirubin and inflammatory cytokines, but PBA has the advantages of no risk of blood-transmissible diseases and rare side effects[32,33]. Recently, due to a shortage of plasma, the combination therapy of PBA plus PE, which needs less plasma than PE, is only widely used for ACLF patients in China. Our results showed that ACLF patients treated with PBA plus PE had better outcomes than patients treated with PE alone.
In this study, the MELD score served as an independent predictor of survival in accordance with previous reports[12,13]. In addition to MELD, we encompassed the clinical and biochemical variables into a Cox regression analysis to identify which were potential predictors of survival. As previously described, age was significantly associated with 3-mo mortality in ACLF patients[12,13]. Here, older age had an unfavorable prognostic relevance. Complications such as HE[12] and HRS[12,13] were also determined to be significantly associated with mortality in ACLF patients. The multivariate analysis in the present study showed that age, number of complications, MELD score and type of ALSS independently determined the outcomes of patients suffering with ACLF. Then, a predictive scoring system was created based on the above variables. As a successive score, the scoring model could precisely distinguish the prognosis of ACLF patients with different scores. Furthermore, high-score patients who are estimated to have a poor outcome probability could be recognized at baseline and considered for early alternative treatments, such as liver transplantation. Specifically, the model could estimate a forecasted survival probability for each individual by calculating the risk score based on the model.
As the prognostic ability of the MELD scoring system had been reported in many studies[38], we further verified the validity of MELD in patients with ACLF. The results showed that MELD did well in categorizing patients based on their risk scores. The c-statistic was 0.799, indicating that the MELD scoring system was useful in forecasting survival in ACLF patients. However, the c-statistic in the new model created in our study was 0.882. By statistical analysis, we found that the new scoring model had a higher predictive capability than MELD. MELD scoring system was originally developed to determine the priority of liver transplantation objectively and was built with only subjective parameters. Therefore, this new prognostic model including some other clinical variables besides MELD had a better performance than MELD scoring system.
However, there are several limitations in this study. First, it was a retrospective study, and the patients only came from a single medical center. Second, the clinical characteristics of the derivation cohort may limit the new model to be applied in other populations, such as patients in Western countries, where alcohol and drugs are the main cause of ACLF. Studies of more heterogeneous groups of patients from geographically diverse areas are needed. Third, several widely accepted prognostic models for LF were not selected, and only MELD was compared in this study. ALSS is not actually proven to be effective in prolonging the patient’s survival by a well-designed randomized controlled trial. Though the new model could predict the patient’s survival better than MELD, it can only be applied to ACLF patients who undergo ALSS besides SMT.
In summary, based on a cohort of patients with ACLF, we have established and validated a new prognostic model for ACLF patients. It was the first to explore an approach to estimate the prognosis of patients treated with ALSS in Asia, and the feasibility of this novel scoring system should be validated by additional larger prospective studies.
Liver failure has a high mortality. The current prognostic model to estimate the survival in acute-on-chronic liver failure (ACLF) patients treated with artificial liver support system (ALSS) is not fully characterized.
Current prognostic models based on parameters of clinical characteristics and the degree of liver dysfunction had been generated to evaluate the short-term survival probability of ACLF patients. However, these models were usually created on the basis of the data at baseline from patients without ALSS treatment.
In the present study, the authors had screened for the predictive factors in a cohort of ACLF patients treated with ALSS, besides standard medical therapy, and established a scoring model which could accurately predict the prognosis in different groups of these patients.
This prognostic model could accurately differentiate the outcome of patients with different risk scores for ACLF patients and also had the ability to assign a predicted survival probability for individual patients.
ACLF was defined as acute liver injury emerging as jaundice and coagulopathy, complicated by ascites and/or encephalopathy within 4 wk in a patient with known or unknown chronic liver disease.
The authors of this paper established a model to predict the prognosis of patients with ACLF treated with artificial liver support system (plasma exchange or plasma bilirubin adsorption combined with plasma exchange). The model seems to be accurate in predicting patients’ survival.
P- Reviewer: Dehghani SM, Garcia-Compean D, Gong ZJ, Liu ZW Mizuguchi T S- Editor: Ma YJ L- Editor: Wang TQ E- Editor: Ma S
| 1. | Katoonizadeh A, Laleman W, Verslype C, Wilmer A, Maleux G, Roskams T, Nevens F. Early features of acute-on-chronic alcoholic liver failure: a prospective cohort study. Gut. 2010;59:1561-1569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 200] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 2. | Wlodzimirow KA, Eslami S, Abu-Hanna A, Nieuwoudt M, Chamuleau RA. A systematic review on prognostic indicators of acute on chronic liver failure and their predictive value for mortality. Liver Int. 2013;33:40-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 125] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 3. | Iwai H, Nagaki M, Naito T, Ishiki Y, Murakami N, Sugihara J, Muto Y, Moriwaki H. Removal of endotoxin and cytokines by plasma exchange in patients with acute hepatic failure. Crit Care Med. 1998;26:873-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 77] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 4. | Silk DB, Trewby PN, Chase RA, Mellon PJ, Hanid MA, Davies M, Langley PG, Wheeler PG, Williams R. Treatment of fulminant hepatic failure by polyacrylonitrile-membrane haemodialysis. Lancet. 1977;2:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 111] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Redeker AG, Yamahiro HS. Controlled trial of exchange-transfusion therapy in fulminant hepatitis. Lancet. 1973;1:3-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 98] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 6. | O’Grady JG, Gimson AE, O’Brien CJ, Pucknell A, Hughes RD, Williams R. Controlled trials of charcoal hemoperfusion and prognostic factors in fulminant hepatic failure. Gastroenterology. 1988;94:1186-1192. [PubMed] |
| 7. | Kondrup J, Almdal T, Vilstrup H, Tygstrup N. High volume plasma exchange in fulminant hepatic failure. Int J Artif Organs. 1992;15:669-676. [PubMed] |
| 8. | Chen CZ, Yu YT, Song JC, Yuan P, Li T, Du Z. Clinical trials for removal of bilirubin by high-capacity nonionic adsorbent. Artif Organs. 1993;17:76-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 9. | Malchesky PS. Nonbiological liver support: historic overview. Artif Organs. 1994;18:342-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 10. | Morimoto T, Matsushima M, Sowa N, Ide K, Sawanishi K. Plasma adsorption using bilirubin-adsorbent materials as a treatment for patients with hepatic failure. Artif Organs. 1989;13:447-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 11. | Hughes RD. Review of methods to remove protein-bound substances in liver failure. Int J Artif Organs. 2002;25:911-917. [PubMed] |
| 12. | Zheng MH, Shi KQ, Fan YC, Li H, Ye C, Chen QQ, Chen YP. A model to determine 3-month mortality risk in patients with acute-on-chronic hepatitis B liver failure. Clin Gastroenterol Hepatol. 2011;9:351-356.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 13. | Sun QF, Ding JG, Xu DZ, Chen YP, Hong L, Ye ZY, Zheng MH, Fu RQ, Wu JG, Du QW. Prediction of the prognosis of patients with acute-on-chronic hepatitis B liver failure using the model for end-stage liver disease scoring system and a novel logistic regression model. J Viral Hepat. 2009;16:464-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 14. | Yang WB, Chen EQ, Bi HX, Bai L, Chen XB, Feng P, Tang H. Different models in predicting the short-term prognosis of patients with hepatitis B virus-related acute-on-chronic liver failure. Ann Hepatol. 2012;11:311-319. [PubMed] |
| 15. | Sarin SK, Kumar A, Almeida JA, Chawla YK, Fan ST, Garg H, de Silva HJ, Hamid SS, Jalan R, Komolmit P. Acute-on-chronic liver failure: consensus recommendations of the Asian Pacific Association for the study of the liver (APASL). Hepatol Int. 2009;3:269-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 630] [Cited by in RCA: 643] [Article Influence: 40.2] [Reference Citation Analysis (0)] |
| 16. | Candido A, Taffon S, Chionne P, Pisani G, Madonna E, Dettori S, Hamza A, Valdarchi C, Bruni R, Ciccaglione AR. Diagnosis of HEV infection by serological and real-time PCR assays: a study on acute non-A-C hepatitis collected from 2004 to 2010 in Italy. BMC Res Notes. 2012;5:297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Schuppan D, Afdhal NH. Liver cirrhosis. Lancet. 2008;371:838-851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1686] [Cited by in RCA: 1565] [Article Influence: 92.1] [Reference Citation Analysis (0)] |
| 18. | Ferenci P, Lockwood A, Mullen K, Tarter R, Weissenborn K, Blei AT. Hepatic encephalopathy--definition, nomenclature, diagnosis, and quantification: final report of the working party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology. 2002;35:716-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1594] [Cited by in RCA: 1410] [Article Influence: 61.3] [Reference Citation Analysis (0)] |
| 19. | Salerno F, Gerbes A, Ginès P, Wong F, Arroyo V. Diagnosis, prevention and treatment of hepatorenal syndrome in cirrhosis. Gut. 2007;56:1310-1318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 356] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 20. | Căruntu FA, Benea L. Spontaneous bacterial peritonitis: pathogenesis, diagnosis, treatment. J Gastrointestin Liver Dis. 2006;15:51-56. [PubMed] |
| 21. | Feinman M, Haut ER. Upper gastrointestinal bleeding. Surg Clin North Am. 2014;94:43-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Wiesner R, Edwards E, Freeman R, Harper A, Kim R, Kamath P, Kremers W, Lake J, Howard T, Merion RM, Wolfe RA, Krom R; United Network for Organ Sharing Liver Disease Severity Score Committee. Model for end-stage liver disease (MELD) and allocation of donor livers. Gastroenterology. 2003;124:91-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1825] [Cited by in RCA: 1865] [Article Influence: 84.8] [Reference Citation Analysis (0)] |
| 23. | Altman DG, Royston P. What do we mean by validating a prognostic model? Stat Med. 2000;19:453-473. [PubMed] |
| 24. | Yang JD, Kim WR, Park KW, Chaiteerakij R, Kim B, Sanderson SO, Larson JJ, Pedersen RA, Therneau TM, Gores GJ. Model to estimate survival in ambulatory patients with hepatocellular carcinoma. Hepatology. 2012;56:614-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 80] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 25. | Flink HJ, Sprengers D, Hansen BE, van Zonneveld M, de Man RA, Schalm SW, Janssen HL; HBV 99-01 Study Group. Flares in chronic hepatitis B patients induced by the host or the virus? Relation to treatment response during Peg-interferon {alpha}-2b therapy. Gut. 2005;54:1604-1609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 88] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 26. | Perceau G, Diris N, Estines O, Derancourt C, Lévy S, Bernard P. Late lethal hepatitis B virus reactivation after rituximab treatment of low-grade cutaneous B-cell lymphoma. Br J Dermatol. 2006;155:1053-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 69] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 27. | Hamid SS, Atiq M, Shehzad F, Yasmeen A, Nissa T, Salam A, Siddiqui A, Jafri W. Hepatitis E virus superinfection in patients with chronic liver disease. Hepatology. 2002;36:474-478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 152] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 28. | Ramachandran J, Eapen CE, Kang G, Abraham P, Hubert DD, Kurian G, Hephzibah J, Mukhopadhya A, Chandy GM. Hepatitis E superinfection produces severe decompensation in patients with chronic liver disease. J Gastroenterol Hepatol. 2004;19:134-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 85] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Jalan R, Williams R. Acute-on-chronic liver failure: pathophysiological basis of therapeutic options. Blood Purif. 2002;20:252-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 223] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 30. | Li LJ, Yang Q, Huang JR, Xu XW, Chen YM, Fu SZ. Effect of artificial liver support system on patients with severe viral hepatitis: a study of four hundred cases. World J Gastroenterol. 2004;10:2984-2988. [PubMed] |
| 31. | Du WB, Li LJ, Huang JR, Yang Q, Liu XL, Li J, Chen YM, Cao HC, Xu W, Fu SZ. Effects of artificial liver support system on patients with acute or chronic liver failure. Transplant Proc. 2005;37:4359-4364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 32. | Pless G. Artificial and bioartificial liver support. Organogenesis. 2007;3:20-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 33. | Zheng Z, Li X, Li Z, Ma X. Artificial and bioartificial liver support systems for acute and acute-on-chronic hepatic failure: A meta-analysis and meta-regression. Exp Ther Med. 2013;6:929-936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 34. | Nakae H, Yonekawa T, Narita K, Endo S. Are proinflammatory cytokine concentrations reduced by plasma exchange in patients with severe acute hepatic failure? Res Commun Mol Pathol Pharmacol. 2001;109:65-72. [PubMed] |
| 35. | Nakamura T, Ushiyama C, Suzuki S, Shimada N, Ebihara I, Suzaki M, Takahashi T, Koide H. Effect of plasma exchange on serum tissue inhibitor of metalloproteinase 1 and cytokine concentrations in patients with fulminant hepatitis. Blood Purif. 2000;18:50-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 36. | Geiger H, Klepper J, Lux P, Heidland A. Biochemical assessment and clinical evaluation of a bilirubin adsorbent column (BR-350) in critically ill patients with intractable jaundice. Int J Artif Organs. 1992;15:35-39. [PubMed] |
| 37. | Senf R, Klingel R, Kurz S, Tullius S, Sauer I, Frei U, Schindler R. Bilirubin-adsorption in 23 critically ill patients with liver failure. Int J Artif Organs. 2004;27:717-722. [PubMed] |
| 38. | Said A, Williams J, Holden J, Remington P, Gangnon R, Musat A, Lucey MR. Model for end stage liver disease score predicts mortality across a broad spectrum of liver disease. J Hepatol. 2004;40:897-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 282] [Article Influence: 13.4] [Reference Citation Analysis (0)] |