Published online Apr 7, 2015. doi: 10.3748/wjg.v21.i13.4014
Peer-review started: August 8, 2014
First decision: August 27, 2014
Revised: October 2, 2014
Accepted: November 18, 2014
Article in press: November 19, 2014
Published online: April 7, 2015
Processing time: 243 Days and 9.4 Hours
AIM: To investigate the accuracy of the strain histogram endoscopic ultrasound (EUS)-based method for the diagnostic differentiation of patients with pancreatic masses.
METHODS: In a prospective single center study, 149 patients were analyzed, 105 with pancreatic masses and 44 controls. Elastography images were recorded using commercially available ultrasound equipment in combination with EUS linear probes. Strain histograms (SHs) were calculated by machine integrated software in regions of interest and mean values of the strain histograms were expressed as Mode 1 (over the mass) and Mode 2 (over an adjacent part of pancreatic tissue, representing the reference area). The ratio between Mode 2 and Mode 1 was calculated later, representing a new variable, the strain histogram ratio. After the final diagnosis was established, two groups of patients were formed: a pancreatic cancer group with positive cytology achieved by fine needle aspiration puncture or histology after surgery (58 patients), and a mass-forming pancreatitis group with negative cytology and follow-up after 3 and 6 mo (47 patients). All statistical analyses were conducted in SPSS 14.0 (SPSS Inc., Chicago, IL, United States).
RESULTS: Results were obtained with software for strain histograms with reversed hue scale (0 represents the hardest tissue structure and 255 the softest). Based on the receiver operating characteristics (ROC) curve coordinates, the cut-off point for Mode 1 was set at the value of 86. Values under the cut-off point indicated the presence of pancreatic malignancy. Mode 1 reached 100% sensitivity and 45% specificity with overall accuracy of 66% (95%CI: 61%-66%) in detection of pancreatic malignant tumors among the patients with pancreatic masses. The positive and negative predictive values were 54% and 100%, respectively. The cut-off for the new calculated variable, the SH ratio, was set at the value 1.153 based on the ROC curve coordinates. Values equal or above the cut-off value were indicative of pancreatic malignancy. The SH ratio reached 98% sensitivity, 50% specificity and an overall accuracy of 69% (95%CI: 63%-70%). The positive and negative predictive values were 92% and 100%, respectively.
CONCLUSION: SH showed high sensitivity in pancreatic malignant tumor detection but disappointingly low specificity. Slight improvements in specificity and accuracy were achieved using the SH ratio.
Core tip: Endoscopic ultrasound quantitative elastography strain histograms (SHs) are a recently developed method for non-invasive differentiation of pancreatic masses. In a prospective single center study, 105 patients with pancreatic masses and 44 controls were evaluated. For SH measurements, Hitachi software was used and applied for the first time in a clinical study as far as we know. We determined the accuracy of the method and a cut-off value between malignant pancreatic tumors and mass-forming pancreatitis. A new variable, the SH ratio, is calculated in an attempt to improve the sensitivity, specificity and accuracy of the method.
- Citation: Opačić D, Rustemović N, Kalauz M, Markoš P, Ostojić Z, Majerović M, Ledinsky I, Višnjić A, Krznarić J, Opačić M. Endoscopic ultrasound elastography strain histograms in the evaluation of patients with pancreatic masses. World J Gastroenterol 2015; 21(13): 4014-4019
- URL: https://www.wjgnet.com/1007-9327/full/v21/i13/4014.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i13.4014
Despite numerous imaging techniques, differential diagnosis of focal pancreatic masses is still a diagnostic problem in a significant number of patients.
In this setting, endoscopic ultrasound-guided fine needle aspiration biopsy (EUS-FNA) has high accuracy and traditionally represents the gold diagnostic standard[1-3], but it is not without risks and complications[4,5].
Multiple pathological processes including cancer can induce alterations of tissue stiffness, changing the elastic properties in comparison with surrounding tissues. Elasticity imaging and measurement provide a significant adjunct to current imaging methods[6]. The elastic properties of tissue are assessed by comparing color images superimposed over B mode scans before and after compression, yielding the local index of tissue elasticity[7]. This method was implemented and used in endosonography with the goal of differentiating tissue changes without risks related to FNA.
Modules for qualitative EUS elastography were incorporated into the first generation of commercially available ultrasound platforms in combination with adequate EUS probes. Tissue stiffness analysis for pancreatic masses using a three-point scoring system was developed by Giovannini et al[8,9] and refined later to a five-point system, reaching better results with higher accuracy. A similar four-point system was proposed by Iglesias-Garcia[10]. The method demonstrated promising results in the evaluation of pancreatic masses[9,10], although poor accuracy was reported in one study[11] and the differentiation of chronic pancreatitis from hard tumors was not satisfactory in another[12].
Second-generation EUS elastography instruments allowed quantitative analysis of tissue stiffness using the strain ratio, numerically expressed elasticity in the target area relative to a reference soft tissue area. Several investigations have shown high but variable sensitivity and specificity of the strain ratio with a wide span of cut-off values delineating inflammatory form malignant pancreatic masses[13-18].
A quantitative technique based on artificial neural network processing of EUS elastography digitalized videos was first applied by Săftoiu et al[19], obtaining average value of the hue histograms with a special computer program. Published results have shown very good sensitivity with satisfactory accuracy[19-21].
Some ultrasound platforms of the latest generation have integrated software for strain (hue) histogram measurement, automatically calculating the graph in real time.
The aim of this study was to evaluate the diagnostic value of the strain histogram in the patients with pancreatic masses and to determine the cut-off value between pancreatic malignancy and focal pancreatitis using software integrated into the commercially available ultrasound platforms.
As the next step we intend to calculate a new variable, the strain histogram ratio, by analogy to the strain ratio, calculating the ratio between the SH over an adjacent part of pancreatic tissue outside the mass and the SH over the mass.
The investigation was organized as a prospective single center study from January 2011 to March 2013 in the gastroenterology department of the University Hospital Centre Zagreb, an academic center. The study was approved by the hospital ethics committee. Written informed consent for participation in the study was obtained from all patients included in the study. Two experienced EUS examiners performed all endosonographic procedures and elastography measurements. Elastography images were recorded using Pentax FG-38 UX and EG-3870 UTK EUS linear probes in combination with Hitachi 8500 and Hitachi Avius ultrasound machines.
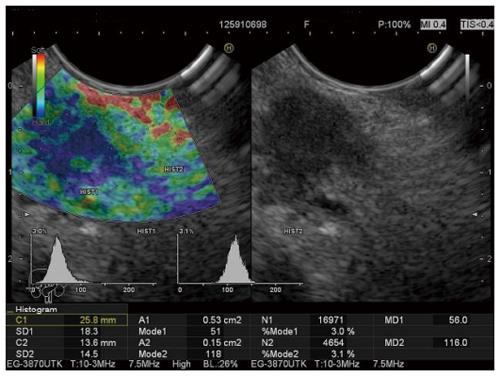
The inclusion criteria were: patients of both sexes over 18 years of age with solid pancreatic masses diagnosed with imaging techniques who accepted participation in the study. The exclusion criteria were: tumors with cystic or liquid components. In total 149 patients were included in the data analysis, 105 with pancreatic masses and 44 controls. Strain histograms were calculated automatically by integrated software in regions of interest manually selected by the endosonographer and expressed as the mean values (Mode 1 over the mass, and Mode 2 over an adjacent part of homogenous pancreatic tissue representing a reference area) (Figure 1). In the control group, two neighboring areas of pancreatic tissue were selected for histogram measurements (Figure 2). Three measurements were performed on the new elastography Figure recorded during the examination, and the average values of Mode 1 and Mode 2 for all three measurements were used for the statistical analysis.
The new variable, the strain histogram ratio, was calculated later by dividing the value of Mode 2 with the value of Mode 1 for each patient.
After the elastography measurements FNA was performed (Echotip, Cook Endoscopy) in all patients with pancreatic masses except in patients with lesions smaller than 3 cm without extrapancreatic or vascular invasion but with strong suspicion of malignancy. In these patients, histology was obtained after surgery. In the patients with negative cytology, follow-up FNA was performed after 3 mo and if necessary after 6 mo. Finally, the patients with pancreatic masses were divided into two groups according to cytology/histology and clinical course after a follow-up period of six months. The first group represents patients with verified pancreatic malignancy and the second group represents patients with mass-forming pancreatitis.
All statistical analyses were conducted in SPSS 14.0 (SPSS Inc., Chicago, IL, United States).
The diagnostic validity analysis included receiver operating characteristics (ROC) curve analysis, with specific values for the area under the curve, the significance of the area, and confidence intervals. Sensitivity (SS), specificity (SP), positive and negative predictive values, and positive and negative likelihood ratios are also expressed in the analysis. The calculation of the overall accuracy (ACC) of the prediction was based on previous parameters. The cut-off point for measurement the calculation of which is based on the coordinates of the ROC curve is also presented in the analysis. The positive likelihood ratio is calculated as sensitivity/1 - specificity. The negative likelihood ratio is calculated as 1- specificity/sensitivity. The overall accuracy is calculated as the sum of the positive and negative predictive values.
Statistical significance is set to P < 0.05, with all confidence intervals expressed at the 95% level.
The median age of the examined group of patients was 63 years (interquartile range: 54-70 years), of whom 76 were female.
Analyzing the sample according to cytology or histology after surgery and a follow-up period, 58/149 (39%) were finally considered to have pancreatic malignant tumors (53 with pancreatic carcinomas, 2 with neuroendocrine malignant tumors, 3 with metastatic carcinomas), 47/149 (31.5%) were classified as patients with pseudotumoral inflammatory masses, and 44/149 (29.5%) patients were control subjects without any clinical or endosonographic evidence of pancreatic disease.
The mass lesion diameter was 31.7 mm; SD = 13.2 mm. Focal lesions were most frequently located in the head of the pancreas (51.5%), less frequently in the body (39.5%), whereas only 9% of them were located in the pancreatic tail.
The Mode 1 variable representing the average strain histogram measure achieved the following diagnostic validity parameters: AUC = 0.815; P < 0.001; 95%CI: 0.749-0.881.
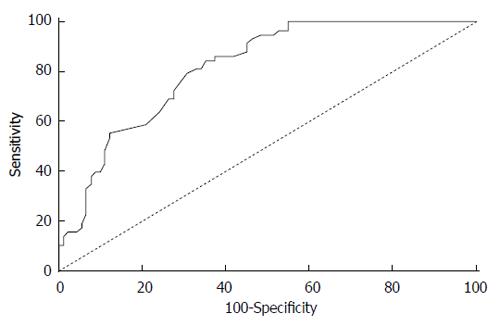
Based on the ROC curve coordinates in Figure 3, the cut-off point for Mode 1 was set at 86. Considering the direction of the variable, values under the cut-off point indicate the presence of malignancy.
Consequently, using this cut-off point, all tumors were correctly identified as such, but as many as 55% of the non-malignant pancreatic masses were incorrectly identified as malignant (Table 1). Using this cut-off value, the overall accuracy achieved with the Mode 1 measure was 66% (95%CI: 61%-66%).
| Malignant pancreatic tumor | |||
| Yes | No | ||
| Mode 1 | |||
| Indicative < 86 | True positive | False positive | Positive predictive value |
| 58/58 | 50/91 | 54% (50%-54%) | |
| Non indicative ≥ 86 | False negative | True negative | Negative predictive value |
| 0/58 | 41/91 | 100% (90%-100%) | |
| Sensitivity | Specificity | ||
| 100% (93%-100%) | 45% (41%-45%) | ||
According to the ROC curve coordinates in Figure 4, the cut-off point for the Mode 2/Mode 1 ratio was set at 1.153. We determined the significance of the diagnostic validity for the new measure derived from the Mode 2/Mode 1 ratio (AUC = 0.843; P < 0.001; 95%CI: 0.782-0.903). All values above the determined cut-off point were classified as malignant. The variable therefore achieves high sensitivity where only 2% of patients having a malignant tumor would not be recognized, but as much as 50% of patients would be incorrectly classified as having a malignant tumor. The overall accuracy achieved with the Mode2/Mode 1 ratio is 69% (95%CI: 63%-70%) (Table 2).
| Malignant pancreatic tumor | |||
| Yes | No | ||
| Mode 2/Mode 1 | |||
| Indicative ≥ 1.153 | True positive | False positive | Positive predictive value |
| 57/58 | 46/91 | 92% (87%-92%) | |
| Non indicative < 1.153 | False negative | True negative | Negative predictive value |
| 1/58 | 45/91 | 100% (96%-100%) | |
| Sensitivity | Specificity | ||
| 98% (91%-100%) | 50% (45%-50%) | ||
The strain ratio and strain histogram quantitative elastography methods are based on different principles[22], as mentioned previously, and that is the reason why we did not compare the sensitivity, specificity and accuracy obtained in our study with the results of published quantitative elastography studies using the strain ratio.
In the three published studies where strain histograms were used as the quantitative elastography method, post-processing software analysis of the elastography video was performed, and the average value of the strain histogram was calculated by a special computer program. A cut-off value was computed initially to distinguish malignant pancreatic masses from pseudotumoral or normal pancreatic tissue[19-21].
Hue histograms were calculated within manually selected regions of interest (ROIs) using a qualitative elastography image. The X-axis in a hue histogram represents tissue elasticity expressed numerically, and the y-axis represents the number of pixels in the ROI. The mean histogram value corresponds to the global hardness of the lesion expressed on a scale from 0 (softest) to 255 (hardest)[21].
In these studies, based on a cut-off of 175 sensitivity reached 91.4%, 84.8% and 93.4%, the specificity was 87.9%, 76.2% and 66.0%, with overall accuracy of 89.7%, 81.5% and 85.4%[19-21]. Data from the latest study[21] were analyzed in extended neural network analysis with automatic differentiation of benign from malignant lesions, and it achieved better sensitivity and overall accuracy (SS = 87.59%, SP = 82.4%, ACC = 91.4%)[23].
Our investigation represents quantitative elastography analysis with strain histograms of pancreatic masses using commercially available integrated ultrasound machine software for the first time. This software also uses a scale from 0 to 255, but the scale is reversed with 0 representing the hardest tissue structure and 255 the softest. The results were obtained as an average value of measurements from the three Figures selected by the endosonographer.
It is possible to compare the cut-off value from the previous studies with the cut-off value computed in our study. Expressed in the same way, the mean value from the previous studies[19-21] becomes 80, which is very close to 86, the value from our study.
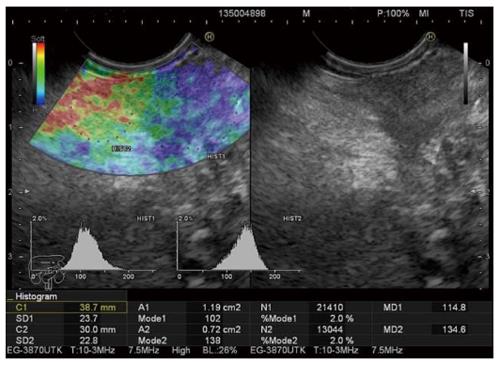
The Mode 1 histogram measured over the tumor in our study showed high sensitivity in cancer detection, but also disappointingly low specificity, with modest overall accuracy (Figure 1: Patient with Mode 1 = 51, and Mode2/Mode1 = 2.31. Both values suggesting a malignant tumor confirmed by surgery). Slight improvements in specificity and overall accuracy were achieved using the new variable, the Mode2/Mode 1 ratio (Figure 5. Patient with Mode 1 = 102 suggesting inflammatory mass and Mode2/Mode1 = 1.35 suggesting malignant mass. Malignancy was confirmed by EUS-FNA).
Explanations for the significant difference in specificity and accuracy achieved in our study in comparison with the results of previous studies using strain histograms as the measurement method could be just speculative at the moment. More studies on Hitachi ultrasound machines with integrated software are needed, comparing whether other analyses possible with the data processing method of Săftoiu et al[19-21] on the same group of patients.
A few recent studies have shown that use of endoscopic ultrasound-based quantitative hue histogram methodology represents a useful tool in the differentiation of pancreatic malignancies from mass-forming pancreatitis.
Previous studies used artificial neural network processing of digitalized endoscopic ultrasound elastography videos, obtaining average values for hue histograms with a special computer program. This method has shown very good sensitivity with satisfactory accuracy, but a limitation of this type of analysis is its availability to other investigators.
In this study, commercially available integrated ultrasound machine software was used for hue histogram analysis for the first time as far as we know. The authors’ determined the accuracy of the method and a cut-off value distinguishing between pancreatic malignant tumors and mass-forming pancreatitis. With the new calculated strain histogram ratio variable, the sensitivity, specificity and accuracy of the method were all improved.
In hospitals with adequate equipment this method could assist in distinguishing malignant from benign pancreatic lesions.
The study is very interesting. In this paper, a new evaluation approach named the strain histogram ratio was established. The cut-off, sensitivity and specificity for this method were calculated. It is a remarkable step forward. Good work.
P- Reviewer: Chisthi MM, Sun SY S- Editor: Ma YJ L- Editor: A E- Editor: Ma S
| 1. | Iglesias-Garcia J, Dominguez-Munoz E, Lozano-Leon A, Abdulkader I, Larino-Noia J, Antunez J, Forteza J. Impact of endoscopic ultrasound-guided fine needle biopsy for diagnosis of pancreatic masses. World J Gastroenterol. 2007;13:289-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 71] [Cited by in RCA: 83] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 2. | Turner BG, Cizginer S, Agarwal D, Yang J, Pitman MB, Brugge WR. Diagnosis of pancreatic neoplasia with EUS and FNA: a report of accuracy. Gastrointest Endosc. 2010;71:91-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 153] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 3. | Dumonceau JM, Polkowski M, Larghi A, Vilmann P, Giovannini M, Frossard JL, Heresbach D, Pujol B, Fernández-Esparrach G, Vazquez-Sequeiros E, Ginès A; European Society of Gastrointestinal Endoscopy. Indications, results, and clinical impact of endoscopic ultrasound (EUS)-guided sampling in gastroenterology: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2011;43:897-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 225] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 4. | Eloubeidi MA, Tamhane A, Varadarajulu S, Wilcox CM. Frequency of major complications after EUS-guided FNA of solid pancreatic masses: a prospective evaluation. Gastrointest Endosc. 2006;63:622-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 203] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 5. | Seicean A. Endoscopic ultrasound in chronic pancreatitis: where are we now? World J Gastroenterol. 2010;16:4253-4263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 37] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 6. | Gao L, Parker KJ, Lerner RM, Levinson SF. Imaging of the elastic properties of tissue--a review. Ultrasound Med Biol. 1996;22:959-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 383] [Cited by in RCA: 281] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 7. | Frey H. [Realtime elastography. A new ultrasound procedure for the reconstruction of tissue elasticity]. Radiologe. 2003;43:850-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 160] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 8. | Giovannini M, Hookey LC, Bories E, Pesenti C, Monges G, Delpero JR. Endoscopic ultrasound elastography: the first step towards virtual biopsy? Preliminary results in 49 patients. Endoscopy. 2006;38:344-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 211] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 9. | Giovannini M, Thomas B, Erwan B, Christian P, Fabrice C, Benjamin E, Geneviève M, Paolo A, Pierre D, Robert Y. Endoscopic ultrasound elastography for evaluation of lymph nodes and pancreatic masses: a multicenter study. World J Gastroenterol. 2009;15:1587-1593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 204] [Cited by in RCA: 198] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 10. | Iglesias-Garcia J, Larino-Noia J, Abdulkader I, Forteza J, Dominguez-Munoz JE. EUS elastography for the characterization of solid pancreatic masses. Gastrointest Endosc. 2009;70:1101-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 121] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 11. | Hirche TO, Ignee A, Barreiros AP, Schreiber-Dietrich D, Jungblut S, Ott M, Hirche H, Dietrich CF. Indications and limitations of endoscopic ultrasound elastography for evaluation of focal pancreatic lesions. Endoscopy. 2008;40:910-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 12. | Janssen J, Schlörer E, Greiner L. EUS elastography of the pancreas: feasibility and pattern description of the normal pancreas, chronic pancreatitis, and focal pancreatic lesions. Gastrointest Endosc. 2007;65:971-978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 176] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 13. | Iglesias-Garcia J, Larino-Noia J, Abdulkader I, Forteza J, Dominguez-Munoz JE. Quantitative endoscopic ultrasound elastography: an accurate method for the differentiation of solid pancreatic masses. Gastroenterology. 2010;139:1172-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 195] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 14. | Itokawa F, Itoi T, Sofuni A, Kurihara T, Tsuchiya T, Ishii K, Tsuji S, Ikeuchi N, Umeda J, Tanaka R. EUS elastography combined with the strain ratio of tissue elasticity for diagnosis of solid pancreatic masses. J Gastroenterol. 2011;46:843-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 113] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 15. | Dawwas MF, Taha H, Leeds JS, Nayar MK, Oppong KW. Diagnostic accuracy of quantitative EUS elastography for discriminating malignant from benign solid pancreatic masses: a prospective, single-center study. Gastrointest Endosc. 2012;76:953-961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 86] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 16. | Figuereido AF, Giovannini M, Bories E, Pesenti C, Caillol F, Monges G, Delpero JR. Endoscopic Ultrasonography Strain Ratio vs. Contrast Enhanced EUS for the Diagnosis of Focal Pancreatic Solid Lesions [abstract]. Gastrointest Endosc. 2009;69:AB129. |
| 17. | Badaoui A, Borbath I, Aouattah T. Evaluation of pancreatic tumors with contrast enhanced-endoscopic ultrasonography and EUS-strain ratio elastography [abstract]. Gastrointest Endosc. 2010;71:AB281. |
| 18. | Mayerle J, Simon P, Dickson EJ. The role of EUS guided elastography to diagnose solid pancreatic mass lesions [abstract]. Pancreas. 2010;39:1334. |
| 19. | Săftoiu A, Vilmann P, Gorunescu F, Gheonea DI, Gorunescu M, Ciurea T, Popescu GL, Iordache A, Hassan H, Iordache S. Neural network analysis of dynamic sequences of EUS elastography used for the differential diagnosis of chronic pancreatitis and pancreatic cancer. Gastrointest Endosc. 2008;68:1086-1094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 172] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 20. | Săftoiu A, Iordache SA, Gheonea DI, Popescu C, Maloş A, Gorunescu F, Ciurea T, Iordache A, Popescu GL, Manea CT. Combined contrast-enhanced power Doppler and real-time sonoelastography performed during EUS, used in the differential diagnosis of focal pancreatic masses (with videos). Gastrointest Endosc. 2010;72:739-747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 21. | Săftoiu A, Vilmann P, Gorunescu F, Janssen J, Hocke M, Larsen M, Iglesias-Garcia J, Arcidiacono P, Will U, Giovannini M, Dietrich C, Havre R, Gheorghe C, McKay C, Gheonea DI, Ciurea T; European EUS Elastography Multicentric Study Group. Accuracy of endoscopic ultrasound elastography used for differential diagnosis of focal pancreatic masses: a multicenter study. Endoscopy. 2011;43:596-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 117] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 22. | Popescu A, Săftoiu A. Can elastography replace fine needle aspiration? Endosc Ultrasound. 2014;3:109-117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 23. | Săftoiu A, Vilmann P, Gorunescu F, Janssen J, Hocke M, Larsen M, Iglesias-Garcia J, Arcidiacono P, Will U, Giovannini M. Efficacy of an artificial neural network-based approach to endoscopic ultrasound elastography in diagnosis of focal pancreatic masses. Clin Gastroenterol Hepatol. 2012;10:84-90.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |