Published online Mar 28, 2015. doi: 10.3748/wjg.v21.i12.3759
Peer-review started: August 3, 2014
First decision: August 27, 2014
Revised: September 11, 2014
Accepted: November 18, 2014
Article in press: November 19, 2014
Published online: March 28, 2015
Processing time: 241 Days and 13.7 Hours
Gastritis cystica profunda is a relatively rare disease, usually observed at anastomotic sites in stomachs of patients that have undergone gastric procedures. We present the rare case of an elevated lesion in the anterior wall of the gastric antrum of a 43-year-old Chinese woman who had never undergone gastric surgery and had no gastrointestinal tract symptoms. Although the physical examination and laboratory data showed no abnormalities, endoscopic ultrasonography revealed an anechoic cystic structure. Abdominal computed tomography and magnetic resonance imaging showed the gastric wall of the greater curvature of the antrum was markedly and irregularly thickened, and mild to moderate enhancement was observed around the lesion with no enhancement in the central portion, suggestive of a gastrointestinal stromal tumor. The patient underwent a distal gastric resection of the 2.5 cm × 1.5 cm lesion. A postoperative pathologic examination showed dilated cystic glands in the muscularis mucosa and submucosal layers and erosion of the mucosal surface of the tumor, confirming the diagnosis of gastritis cystica profunda without malignancy.
Core tip: Gastritis cystica profunda is a rare disease characterized by polypoid hyperplasia and cystic dilatation of the gastric glands extending into the submucosa of the stomach. It is typically only found in the stomach after gastric surgery, however, we encountered a rare case of gastritis cystica profunda in a 43-year-old Chinese woman who had never undergone gastric surgery. The elevated lesion in the anterior wall of the gastric antrum was discovered by endoscopic ultrasonography, and marked and irregular thickening of the gastric wall was observed with computed tomography and magnetic resonance imaging.
- Citation: Yu XF, Guo LW, Chen ST, Teng LS. Gastritis cystica profunda in a previously unoperated stomach: A case report. World J Gastroenterol 2015; 21(12): 3759-3762
- URL: https://www.wjgnet.com/1007-9327/full/v21/i12/3759.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i12.3759
Gastritis cystica profunda (GCP) is a relatively rare, benign disease characterized by polypoid hyperplasia and cystic dilatation of the gastric glands that extend into the submucosa of the stomach[1]. GCP can present as upper gastrointestinal symptoms, such as upper abdominal pain, acid reflux, nausea, anorexia, or bleeding, though some patients may experience no symptoms at all[2], and can cause massive upper gastrointestinal hemorrhage and gastric outlet obstruction in some cases[3-5]. GCP usually emerges as giant gastric folds, submucosal tumors, or isolated polyps and typically occurs at anastomotic sites of previous surgeries[6]. Here we report a unique case of GCP in an asymptomatic patient who had no history of gastric surgery.
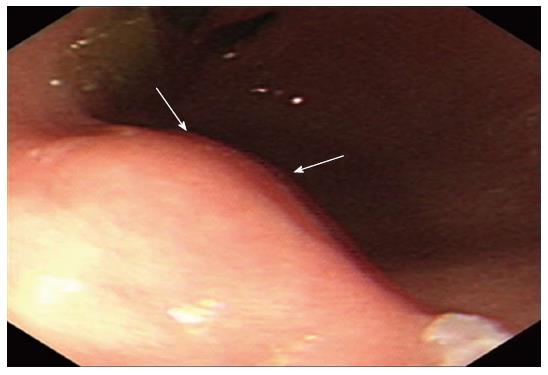
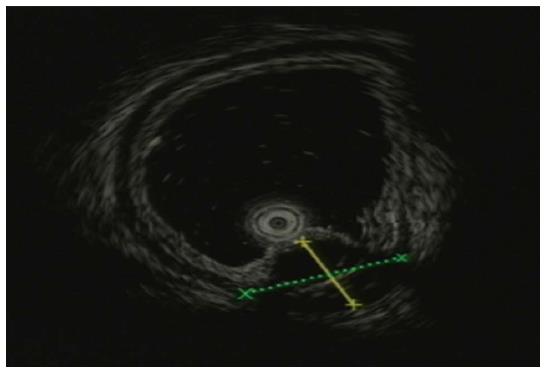
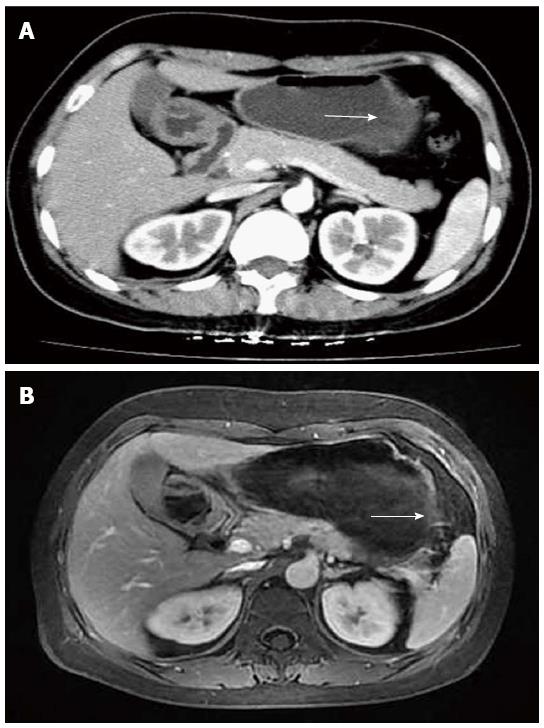
A 43-year-old Chinese woman was admitted to our hospital for an abnormality of the stomach that was discovered during a routine medical check-up. Gastroscopy revealed a mass-like lesion protruding into the gastric lumen (Figure 1). She had no gastrointestinal symptoms and no history of abdominal surgery. Physical examination and laboratory data showed no abnormalities. Endoscopic ultrasonography revealed an anechoic cystic structure in the irregularly thickened stomach wall of the greater curvature of the antrum (Figure 2). Additional abdominal computed tomography and magnetic resonance imaging also showed that the gastric wall was markedly and irregularly thickened, and enhanced scanning showed mild to moderate enhancement around the lesion, suggestive of a gastrointestinal stromal tumor (Figure 3).
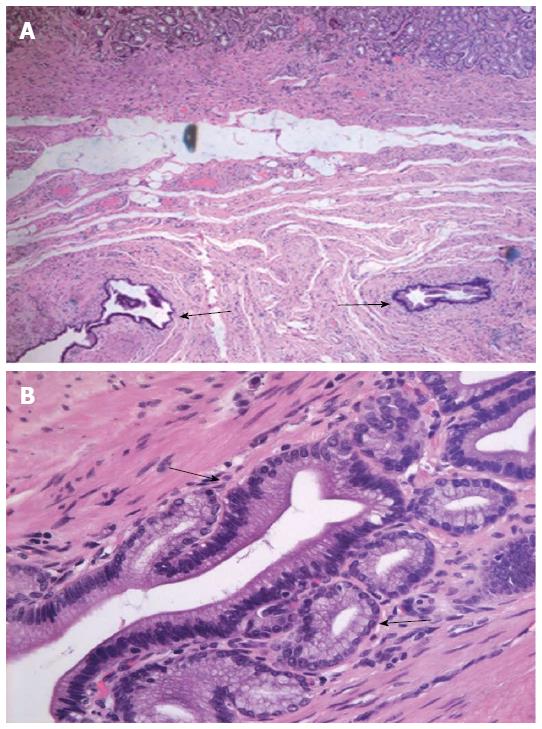
A distal gastric resection (Billroth I) was performed to remove the 2.5 cm × 1.5 cm tumor, which had an eroded mucosal surface. A histologic examination revealed dilated cystic glands in the muscularis mucosa and submucosa (Figure 4), consistent with a diagnosis of GCP without malignancy.
The etiology and pathogenesis of GCP have not been established, however, lesions are often accompanied by severe diffuse chronic gastritis[7] and Helicobacter pylori infection[8]. It is thought that inflammation causes the erosion of the muscularis mucosa, consequently leading to the formation of submucosal cysts. The potassium voltage-gated channel subfamily E member 2 (KCNE2), an essential subunit of apical potassium channels in parietal cells, may also be implicated, as knockout mice exhibit a severe gastric preneoplastic phenotype[9], and the expression of KCNE2 is negatively correlated with gastric cancer formation in humans[10]. GCP is also considered to be precancerous[8], as a few cases have been associated with gastric cancers[11-13] and gastric carcinoma with lymphoid stroma[14]. There is also evidence that the development of GCP is influenced by infection with the Epstein-Barr virus, which was observed in both dysplastic and carcinoma areas[14], and is more prevalent in gastric cancer tissues from patients with GCP[11].
As GCP is typically found at sites of previous surgeries, it was suggested that mucosal injury caused by surgery or the suture technique itself promotes mucosal prolapse and herniation of glands into the submucosa[15,16]. Interestingly, the patient in the present case had no history of abdominal surgery, thus additional factors must have contributed. Mukaisho et al[17] found GCP-like mucosal changes in an animal model of duodenal reflux, of which 76.2% developed adenocarcinomas at week 80. Repeated erosion of the stomach lining may give rise to heterotopic cysts, the surface mucosa of which may be more prone to further erosion. Additionally, the regeneration of mucosal epithelium may lead to the development of carcinoma[8]. Indeed, GCP lesions may be precancerous, as Ochiai et al[13] found that cell kinetics were accelerated and gene mutations were enhanced in GCP lesions, and foveolar glands in GCP can be hyperplastic and highly proliferative[18]. Therefore, patients with non-malignant GCP should continue to be monitored.
The main treatment for GCP is surgical removal of the lesion, while minimizing bile reflux into the stomach. Due to the premalignant nature of the disease, histopathologic evaluation of the lesion is recommended. In addition, GCP recurrence after surgical resection has been reported[19]. Therefore, careful long-term follow-up is needed. The case reported here demonstrates that GCP is not limited to patients who have previously undergone gastric surgery, and should therefore be considered for cases with abnormalities of the stomach mucosa.
A 43-year-old Chinese woman who had no history of gastric surgery presented with an abnormality of the stomach by gastroscopy during a routine medical check-up.
Endoscopic ultrasonography revealed an anechoic cystic structure in the irregularly thickened stomach wall in the greater curvature of the antrum.
Complete blood count, serum tumor markers (carcinoembryonic antigen, alpha-fetoprotein, carbohydrate antigen 19-9), metabolic panel and liver function test were within normal limits.
Abdominal computed tomography and magnetic resonance imaging showed the gastric wall on the greater curvature of the antrum was markedly and irregularly thickened; enhanced scanning showed mild to moderate enhancement of the lesion periphery, with no enhancement in the central portion of the lesion.
Postoperative pathologic examination showed a 2.5 cm × 1.5 cm lesion with an eroded mucosal surface, confirming the diagnosis of gastritis cystica profunda (GCP) without malignancy.
The patient underwent a distal gastric resection (Billroth I).
GCP usually emerges as giant gastric folds, submucosal tumors, or isolated polyps at the anastomotic sites of previous surgeries. GCP is also more prevalent in patients with gastric cancer, and can be considered as a precancerous lesion.
GCP is a rare disease characterized by polypoid hyperplasia and cystic dilatation of the gastric glands extending into the submucosa of the stomach.
Although GCP is typically only seen in stomachs of patients who have undergone gastric procedures, it can also develop in stomachs of patients with no history of abdominal surgery.
In this article, the authors report a rare case of GCP that developed in that stomach of patient who had not previously undergone gastric surgery. The authors also review the clinical pathologic features of GCP. It is a case report that is of interest to the readership.
P- Reviewer: Caboclo JLF, Chetty R, Kai K, Park WS S- Editor: Gou SX L- Editor: A E- Editor: Zhang DN
| 1. | Littler ER, Gleibermann E. Gastritis cystica polyposa. (Gastric mucosal prolapse at gastroenterostomy site, with cystic and infiltrative epithelial hyperplasia). Cancer. 1972;29:205-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 2. | Xu G, Qian J, Ren G, Shan G, Wu Y, Ruan L. A case of gastritis cystica profunda. Ir J Med Sci. 2011;180:929-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (96)] |
| 3. | Itte V, Mallick IH, Moore PJ. Massive gastrointestinal haemorrhage due to gastritis cystica profunda. Cases J. 2008;1:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Kurland J, DuBois S, Behling C, Savides T. Severe upper-GI bleed caused by gastritis cystica profunda. Gastrointest Endosc. 2006;63:716-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 5. | Matsumoto T, Wada M, Imai Y, Inokuma T. A rare cause of gastric outlet obstruction: gastritis cystica profunda accompanied by adenocarcinoma. Endoscopy. 2012;44 Suppl 2 UCTN:E138-E139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (111)] |
| 6. | Ozenç AM, Ruacan S, Aran O. Gastritis cystica polyposa. Arch Surg. 1988;123:372-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 7. | Franzin G, Novelli P. Gastritis cystica profunda. Histopathology. 1981;5:535-547. [PubMed] |
| 8. | Iwanaga T, Koyama H, Takahashi Y, Taniguchi H, Wada A. Diffuse submucosal cysts and carcinoma of the stomach. Cancer. 1975;36:606-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 9. | Roepke TK, Purtell K, King EC, La Perle KM, Lerner DJ, Abbott GW. Targeted deletion of Kcne2 causes gastritis cystica profunda and gastric neoplasia. PLoS One. 2010;5:e11451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 65] [Article Influence: 4.3] [Reference Citation Analysis (105)] |
| 10. | Kuwahara N, Kitazawa R, Fujiishi K, Nagai Y, Haraguchi R, Kitazawa S. Gastric adenocarcinoma arising in gastritis cystica profunda presenting with selective loss of KCNE2 expression. World J Gastroenterol. 2013;19:1314-1317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (111)] |
| 11. | Choi MG, Jeong JY, Kim KM, Bae JM, Noh JH, Sohn TS, Kim S. Clinical significance of gastritis cystica profunda and its association with Epstein-Barr virus in gastric cancer. Cancer. 2012;118:5227-5233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (113)] |
| 12. | Lee SJ, Park JK, Seo HI, Han KH, Kim YD, Jeong WJ, Cheon GJ, Eom DW. A case of gastric inverted hyperplastic polyp found with gastritis cystica profunda and early gastric cancer. Clin Endosc. 2013;46:568-571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (111)] |
| 13. | Ochiai M, Matsubara T, Zhi LZ, Funabiki T, Sakurai Y, Hasegawa S, Imazu H, Suganuma M. Gastritis cystica polyposa associated with a gastric stump carcinoma, with special reference to cell kinetics and p53 gene aberrations. Gastric Cancer. 2000;3:165-170. [PubMed] |
| 14. | Kim L, Kim JM, Hur YS, Shin YW, Park IS, Choi SJ, Han JY, Chu YC, Kim KH. Extended gastritis cystica profunda associated with Epstein-Barr virus-positive dysplasia and carcinoma with lymphoid stroma. Pathol Int. 2012;62:351-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (113)] |
| 15. | Kondo K, Kojima H, Akiyama S, Ito K, Takagi H. Pathogenesis of adenocarcinoma induced by gastrojejunostomy in Wistar rats: role of duodenogastric reflux. Carcinogenesis. 1995;16:1747-1751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 16. | Lee TH, Lee JS, Jin SY. Gastritis cystica profunda with a long stalk. Gastrointest Endosc. 2013;77:821-82; discussion 822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (113)] |
| 17. | Mukaisho K, Miwa K, Kumagai H, Bamba M, Sugihara H, Hattori T. Gastric carcinogenesis by duodenal reflux through gut regenerative cell lineage. Dig Dis Sci. 2003;48:2153-2158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 18. | Aoyagi K, Koufuji K, Yano S, Murakami N, Terasaki Y, Yamasaki Y, Takeda J, Tanaka M, Shirouzu K. Two cases of cancer in the remnant stomach derived from gastritis cystica polyposa. Kurume Med J. 2000;47:243-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 19. | Wang L, Yan H, Cao DC, Huo L, Huo HZ, Wang B, Chen Y, Liu HL. Gastritis cystica profunda recurrence after surgical resection: 2-year follow-up. World J Surg Oncol. 2014;12:133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (111)] |