Published online Dec 28, 2014. doi: 10.3748/wjg.v20.i48.18092
Revised: September 3, 2014
Accepted: October 14, 2014
Published online: December 28, 2014
Processing time: 181 Days and 14.1 Hours
Acute pancreatitis remains a clinical challenge, despite an exponential increase in our knowledge of its complex pathophysiological changes. Early fluid therapy is the cornerstone of treatment and is universally recommended; however, there is a lack of consensus regarding the type, rate, amount and end points of fluid replacement. Further confusion is added with the newer studies reporting better results with controlled fluid therapy. This review focuses on the pathophysiology of fluid depletion in acute pancreatitis, as well as the rationale for fluid replacement, the type, optimal amount, rate of infusion and monitoring of such patients. The basic goal of fluid epletion should be to prevent or minimize the systemic response to inflammatory markers. For this review, various studies and reviews were critically evaluated, along with authors’ recommendations, for predicted severe or severe pancreatitis based on the available evidence.
Core tip: Acute pancreatitis can manifest as a severe form, which has a high mortality rate. The treatment of AP is primarily supportive, and fluid replacement therapy has emerged as one of the key treatment strategies. There is a lack of randomized studies addressing the questions of the best type of fluid, amount of fluid and rate of fluid transfusion. This paper reviews the available literature and the controversies and attempts to frame guidelines for fluid therapy in acute pancreatitis.
- Citation: Aggarwal A, Manrai M, Kochhar R. Fluid resuscitation in acute pancreatitis. World J Gastroenterol 2014; 20(48): 18092-18103
- URL: https://www.wjgnet.com/1007-9327/full/v20/i48/18092.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i48.18092
Acute pancreatitis (AP) is acute inflammation of the pancreas, and has high morbidity and mortality rates[1]. AP displays a wide spectrum of disease presentation, ranging from self-limiting mild illness to rapidly progressive severe illness ending in multi-organ failure with a high risk of mortality. Different stages of severity have been described in the Atlanta classification[2]. It has been estimated that about 10% to 20% of AP patients develop the severe form, which has a 15% to 40% mortality rate[3].
A major factor complicating the appropriate management of AP is the failure to discriminate its mild and severe forms in the initial stages. This issue is critical, as about half of the patients with severe AP die within the first week due to the development of organ failure; the incidence of organ failure is maximal (17%) on the first day[4,5]. The causes for later mortality are development of infected necrosis and other complications. Thus, it is important to identify factors that can predict severity of the AP disease so as to guide early clinical management within the so-called interventional window[6,7].
Despite increased understanding of the underlying pathophysiology of both the disease and its complications, its management remains a clinical challenge and is primarily based on supportive therapy[8]. Fluid resuscitation is the current cornerstone of early management, although there is little consensus on the details of its application[9]. Widely accepted clinical practice guidelines recommend vigorous fluid resuscitation in the early management of AP[10-12] (Table 1). However, there is a lack of consensus on specific recommendations regarding the type of fluid, optimal rate of fluid administration and end points to indicate adequate resuscitation[13].
| Ref. | Type | Conclusion |
| Tenner et al[7], 2004 | Review | 250-500 mL/h or more for 48 h |
| Whitcomb et al[79], 2006 | Review | Fluid bolus: maintain hemodynamicsLater: 250-500 mL/h |
| Otsuki et al[91], 2006 | Review | 60-160 mL/kg per day1/3 to 1/2 to be given in 6 h |
| Forsmark et al[96], 2007 | Review | Use crystalloids first,Use colloids if hematocrit < 25% or albumin < 2 g/dL |
| Pandol et al[78], 2007 | Review | Severe volume depletion: 500-1000 mL/h; reduce later |
| Nasr et al[8], 2011 | Review | 20 mL/kg (1-2 L) in emergency; 150-300 mL/h (3 mL/kg per hour) for 24 h |
| Trikudanathan et al[49], 2012 | Review | Aggressive fluid resuscitation in patients with AP needs to be initiated with therapeutic intent |
| Haydock et al[49], 2013 | Review | Lack of quality evidence to guide most basic aspects of FT providing the equipoise necessary for further RCTs |
| Wu et al[31], 2013 | Review | Institutional protocols must be developed to help ensure adequate fluid resuscitation, particularly in initial 24 h |
Although aggressive fluid therapy is the cornerstone of treatment in AP, a few recent studies have suggested that non-aggressive fluid therapy may be better in reducing mortality and improving outcomes[14-18]. A recent systematic review has brought into focus the lack of evidence in literature on the type of fluid, rate of fluid infusion and the goals to be monitored[9]. Even the evidence that is available is of low or very low quality. Therefore, we reviewed the literature regarding fluid resuscitation in the course of AP, placing emphasis on goals, choice and amount of fluids to reduce complications such as pancreatic necrosis and organ failure (Table 2). The review’s findings, written up herein, are based on a detailed PubMed search which encompassed all published articles up to the end of June 2014.
| Ref. | Year | Type of study (sample size) | Conclusion |
| Mao et al[16] | 2010 | RCT (n = 155) | Rapid hemodilution increases incidence of sepsis within 28 d and in-hospital mortality. Hematocrit should be maintained between 30% and 40% in acute response stage |
| Mao et al[17] | 2009 | RCT (n = 76) | Controlled fluid resuscitation offers better prognosis in patients with severe volume deficit within72 h of severe acute pancreatitis onset |
| Eckerwall et al[15] | 2006 | Retrospective cohort (n = 99) | Patients receiving 4000 mL or more of fluid in first 24 h developed more respiratory complications |
| Madaria et al[14] | 2011 | Retrospective cohort (n = 247) | Administration of > 4.1 L but not < 3.1 L was significantly associated with more local and systemic complications |
| Kuwabara et al[75] | 2011 | Retrospective (n = 9489) | Fluid volume during first 48 h was higher in patients requiring ventilation and higher mortality in acute pancreatitis |
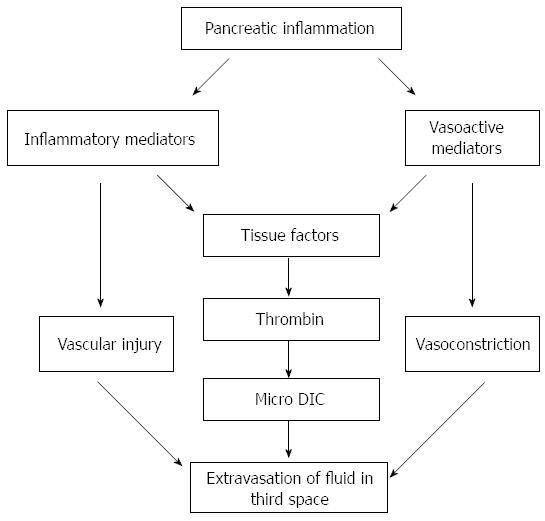
The major etiologic factors of AP are alcohol and biliary obstruction; other causes being autoimmunity, drugs, trauma, iatrogenic and idiopathic. A major complication of the disease is organ failure as a result of excessive activation of a systemic inflammatory response cascade[19]. Pancreatic insult due to any etiology leads to release of pro-inflammatory mediators, such as zymogens, cytokines and vasoactive factors. These mediators cause endothelial cell activation leading to arteriolar vasoconstriction, increased permeability and circulatory stasis, thereby inducing ischemia[20]. This increased permeability, related to capillary leakage, causes intravascular fluid loss and hypotension, and shock may ensue (Figure 1). Further, accumulation of inflammatory mediators with enhanced leucocytes and endothelial interaction results in the activation of the coagulation cascade and hyper-coagulation. Micro-vascular thrombosis further causes tissue hypoxia and, ultimately, systemic inflammatory response syndrome (SIRS)[20]. Organ dysfunction usually occurs quite early in the course of severe AP, usually the first four days[21,22], and unless aggressive management is performed, it causes mortality in about 50% of cases within the first week of its manifestation[23]. The first five days after the onset of acute disease are considered as the “therapeutic interventional window”[6], during which aggressive fluid resuscitation can correct the third space losses and increase tissue perfusion. SIRS may be averted with prevention of multiple organ failure and/or pancreatic necrosis.
Severity of AP can be classified as mild (having no local or systemic complications), moderate (in which local or systemic complications are associated with organ failure that resolves within 48 h) or severe (in which organ failure persists beyond 48 h)[3]. The risk of mortality is nil or minimal in patients with no or minimal organ failure, and it can be as high as 15% to 40% in those with severe disease[11]. Organ failure has emerged as a key determinant of the severity of AP. Organ failure itself is classified as transient or persistent depending upon the duration and is classified as per the modified Marshall score[24].
There are various scoring criteria described in the literature to assess the severity of pancreatitis[25-30]. However, these scoring systems require 48 h to complete. The first 12-24 h are critical, as the maximum incidence of development of organ failure fits within this time frame[31]. Hence, two new scoring systems have been proposed recently, each of which assesses severity in the first 24 h. The Bedside Index of Severity in Acute Pancreatitis (BISAP) is one and assesses 5 criteria: blood urea nitrogen (BUN) > 25 mg/dL, age > 60 years, impaired mental status, SIRS and pleural effusion[32]. For BISAP, a score of > 2 is associated with a 10-fold increase in mortality risk[27,33]. The second scoring system is the Harmless Acute Pancreatitis Score, which focuses on patients who are unlikely to develop severe pancreatitis[26].
Management of AP revolves around supportive care, adequate nutrition, and intravenous hydration. The rationale for hydration is based on the need to resolve the hypovolemia that occurs secondary to vomiting, reduced oral intake, third space extravasation, respiratory losses and diaphoresis. In addition, early hydration provides macrocirculatory and microcirculatory support to prevent the cascade of events leading to pancreatic necrosis[34].
Correction of hypovolemia, as assessed by changes in hematocrit, BUN and serum creatinine, has been documented to limit necrosis and improve outcome. Hemoconcentration, as a marker of hypovolemia and severity of pancreatitis, has been studied since the 1960s[35]. A hematocrit of ≥ 44%-47% on admission combined with failure of a decrease in the hematocrit at 24 h was reported as the best risk factor for development of necrosis[36]. Wu et al[37] showed that early hemoconcentration was associated with increased mortality only among hospital transferred cases, and not among non-transferred cases. This difference could be due to variations in the early management of the studied cases, further emphasizing the fact that fluid resuscitation should be instituted early. Similarly, changes in BUN and creatinine from baseline are indicative of intravascular volume depletion and both these markers are used to predict outcome. Monitoring of these parameters can gauge the effectiveness of initial resuscitative measures[38-43]. These parameters can, therefore, be used to optimize goal-directed resuscitation.
Microcirculatory disturbances in AP are different from simple hypovolemia of trauma or bleeding, as they are caused by SIRS with overexpression of inflammatory mediators which injure the endothelium and increase capillary permeability, leading to fluid sequestration and capillary leak syndrome[44]. Thus, the purpose of effective fluid resuscitation in severe AP is not only to replenish the blood volume but also to stabilize the capillary permeability, modulate the inflammatory reaction, and sustain intestinal barrier function[44].
A number of animal studies have shown the benefit of fluid resuscitation in AP, using both colloids and crystalloids. Juvonen et al[45] used a pig model of AP to show that signs of splanchnic hypoperfusion could be prevented with fluid resuscitation. In that study, the authors measured splanchnic perfusion by local pC02 gap, oxygen delivery and consumption, lactate production, and blood flow. On the other hand, Niederau et al[46] used a mouse model of AP to show that subcutaneous administration of fluid could normalize hemoconcentration and improve survival. High volume crystalloid infusion with Ringer’s lactate has also been shown to improve pancreatic microcirculation in a canine model[47] and with balanced salt solution in a rat model[48].
The primary aim of fluid therapy is to limit or prevent pancreatic necrosis. Any patient with AP has the potential to progress to severe disease. Patients with mild interstitial pancreatitis are commonly kept under observation in the emergency room, and once their pain settles they can be discharged. Patients with underlying comorbidities would, however, require closer observation. The Revised Atlanta guidelines[3] allow for patients to be triaged and evaluated for severity. Patients with moderate and severe AP require observation for organ failure and local or systemic complications, and should be started on fluid therapy. It must be recognized that at the time of first interface with the patient it may not be possible to gauge severity, which may evolve over the next 24-48 h.
Two types of fluids used frequently are colloids and crystalloids. Commonly used colloids are various formulations of dextran, hetastarch and albumin. Colloids are considered superior to crystalloids in optimization of the hemodynamic response[49]. They also have better retention in the intravascular compartment because of their larger size[34]. As they remain within the lumen, despite increased vascular permeability, they help to maintain better circulatory flow. In addition, they contribute to the correction of hypovolemia because of their osmotic effect in drawing fluid from the interstitium to the vascular compartment. However, colloids can cause intravascular volume overload, hyperoncotic renal impairment, coagulopathy, and anaphylactic reaction[50].
Commonly used crystalloids are normal saline (NS), lactated Ringer’s (RL) and Ringer’s ethyl pyruvate, with hypertonic saline being the so-called “new kid on the block”. Crystalloids are distributed in both the plasma and the interstitial compartments, and large spaces are therefore required to restore the circulation. Infusion of large amounts of crystalloids could lead to pulmonary edema[48]. Hypertonic saline effectively reduces the volume of isotonic fluid resuscitation, thereby reducing the risk of pulmonary edema[51]. However, there is a potential risk of central pontine myelinolysis with aggressive hypertonic saline therapy[52].
The ideal fluid for resuscitation in AP is yet to be determined. The choice is primarily between a colloid and a crystalloid. Variability in the results of initial studies with fluid resuscitation could be attributed to different types of fluids used. Colloids have been shown to be superior to crystalloids in animal experiments[34], which may be due to the fact that they are not as permeable to leakage in pancreatic microcirculation as crystalloids. By remaining in the lumen, circulatory blood flow is better maintained and inflammatory mediators are less likely to reach the acinus when colloids are used[34].
Most of the animal studies on fluid resuscitation have used colloid solutions and found them to be better than crystalloids. Schmidt et al[53] showed intra-aortic bolus infusion of high-dose dextran to be better than LR and NS with regards to necrosis and mortality in a rat model of pancreatitis. Other researchers have also shown beneficial effects of different dextran preparations[47,54-57]. Klar et al[55] quantified the effect of dextran on pancreatic microcirculation during experimental acute biliary pancreatitis and showed that stabilization of microcirculation was accomplished in 6 h. In yet another study, mortality was reduced from 60% in RL controls to 10% and 0% in groups treated with DEX-70 and DEX-160 respectively[57].
The use of colloids in humans has involved various preparations of dextran and albumin. In a phase I human study involving 13 patients with severe non-biliary pancreatitis, after hemodilution with dextran, progression of pancreatic necrosis was seen in 15% and mortality was seen in only 7.7%[55]. In another human study, 32 patients with severe AP were treated with 500-1000 mL dextran 40 and dexamethasone (0.5-1 mg/kg) daily along with standard therapy for one week. Twenty-seven patients improved, 5 required surgery and 4 ultimately succumbed to death. The authors concluded that short period use of dexamethasone can inhibit inflammatory mediators while dextran helps in correcting microcirculatory disturbances[58].
Hydroxyethyl starch (HES) is another colloid fluid that can preserve systemic oxygenation in patients with capillary leak. While it has been shown to reduce the risk of intra-abdominal hypertension in severe AP[59], no impact on organ failure and mortality has been observed. In a recent study comparing NS (group 1), HES and NS (group 2) and HES, NS and glutamine (group 3), Zhao et al[44] showed that patients in groups 2 and 3 had decreased rates of organ failure and infection. However, a Cochrane review has concluded that HES may actually increase mortality in critically ill patients[60].
In contrast to the animal studies, there is more data on beneficial effects of crystalloids in humans. Du et al[59] compared RL with a combination of RL and HES. The combination showed better results on intra-abdominal pressure in AP but no effect was seen on organ failure, in-hospital stay or mortality. A recent randomized controlled trial by Wu et al[61] compared NS vs RL as resuscitation fluid in AP and reported dampening of systemic inflammation after 24 h in subjects resuscitated with RL. A significant reduction in the prevalence of SIRS and levels of C-reactive protein was found in the RL group as compared to the NS group. This pilot trial of RL as the primary resuscitation fluid has become a landmark study for the potential utility of RL as a resuscitation fluid in early treatment of AP.
Recently, hypertonic saline was found in various animal studies to be useful in management of AP by modulation of cytokine expression[43,62,63]. It has been shown to significantly facilitate pancreatic microcirculation and to have favorable effects on cardiac contractility and peripheral tissue perfusion[51]. Aerosolized hypertonic saline has also been shown to reduce lung injury as well as the risk of lung edema, due to the lower volume used[51]. However, safety of hypertonic saline for volume repletion needs to be established as adverse potential effects, such as renal failure, have been documented in burn patients[64].
A combination of crystalloids and colloids has been suggested by some researchers to be better than either alone[17,43]. Moreover, some researchers have suggested using a ratio of 1:3 for colloids and crystalloids respectively[65]. In a recent study of 47 patients with AP, Chang et al[66] showed that a crystalloid to colloid ratio of 1.1-3.0 was superior to a ratio of < 1.5 or > 3.
Other solutions used in fluid resuscitation are a cell-free hemoglobin-based oxygen carrier[67], a combination of isovolemic hemodilution with HES and a plasmatic oxygen carrier[68], 6% hydroxyethyl starch and furosemide[69]. A recent Chinese study demonstrated the effects of 6% HES and furosemide on the outcome of AP, but only 4% of the patients with Ranson’s score of 3 or more and 7% with a Balthazar computed tomography (CT) score D developed pancreatic necrosis and organ dysfunction[69]. Another study demonstrated the effect of fresh frozen plasma (FFP) in AP and observed that the ability of FFP to reduce the fall in serum α-2 macroglobulin may have therapeutic implications in early treatment of AP[70].
In summary, there is a lack of high level evidence to guide the choice of fluid in AP. Crystalloids are recommended by the American Gastroenterological Association, and colloids (packed red blood cells) are considered in cases of low hematocrit (< 25%) and low serum albumin (< 2 g/dL). Among the crystalloids, RL solution is preferred over NS[71]. However, there is an urgent need of studies on this issue.
Aggressive fluid resuscitation was defined by the Mayo Clinic group to constitute ≥ 33% of the total volume in 72 h of infusion performed in the first 24 h[72]. Chinese researchers have used a more objective criteria of 15 mL/kg per hour infusion as aggressive resuscitation, as compared to controlled resuscitation, which they defined as 5-10 mL/kg per hour[17]. Although recommendations by various authors and reviews have suggested intensive fluid resuscitation, it was two retrospective studies from Mayo Clinic[72,73] that defined aggressive fluid resuscitation. In the first study, 28 patients in the non-aggressive group experienced higher mortality (17.9%) than the 17 patients in the aggressive group (0% mortality)[73]. In the second study, out of a total 73 patients, the 31 who were given non-aggressive fluid resuscitation had higher SIRS scores[38]. In a separate retrospective study, Wall et al[74] observed the mean rate of hydration fluid being higher (234 mL/h in first 6 h and 221 mL/h in first 12 h) in patients treated in 2008 as compared to those treated in 1988 (194 mL/h in first 6 h and 188 mL/h during first 12 h) with a decrease in mortality and necrosis in 2008 as compared to 1998.
There are, however, other studies suggesting that aggressive hydration may be associated with increased morbidity and mortality[14,15]. de-Madaria et al[14] analyzed 247 patients prospectively who were divided into three groups depending on the fluid received in the initial 24 h. Administration of > 4.1 L during the initial 24 h was found to be associated with persistent organ failure and acute collections, while administration between 3.1 and 4.1 L was associated with an excellent outcome. Eckerwall et al[15] reported that patients receiving > 4.0 L in the first 24 h developed more respiratory complications (66% vs 53%, P < 0.001) than those receiving less than 4.0 L/24 h. The latter group had decreased need for ICU care, as well. A Japanese study analyzed the demographics of fluid resuscitation in 9489 patients and found that those with higher volume infused in the first 48 h had higher respiratory complications and higher mortality[75].
Mao et al[17] found higher complication rates and mortality in patients given aggressive hydration (15 mL/kg per hour) when compared to those given controlled hydration (5-10 mL/kg per hour). Another study by Mao et al[16] showed that survival rate in the slow hemodilution group (84.7%) was better than that in the rapid hemodilution group (66%). These authors also found that rapid hemodilution can increase the incidence of sepsis within 28 d.
Since there has been a negative side effect reported for aggressive fluid resuscitation, some practitioners have proposed a more controlled fluid resuscitation rather than overzealous fluid therapy. A Chinese study of 83 patients confirmed the observations that survival rates improved significantly by controlling fluid resuscitation and preventing sequestration of body fluids[18].
It has been pointed out that there are an equal number of studies in favor of aggressive and non-aggressive fluid resuscitation[9]. One can argue that aggressive resuscitation restores the intravascular compartment depleted by “third spacing” and results in more effective end-organ tissue perfusion and reverses pancreatic ischemia[76]. Those in favor of non-aggressive hydration suggest that by the time we intervene in patients with AP, pancreatic necrosis is already non-reversible and aggressive fluid therapy will only lead to respiratory failure and increased intra-abdominal pressure, etc.[76]. Therefore, a “controlled” resuscitation aimed at reversing hypotension, and being able to maintain effective mean arterial pressure (MAP) and urine output > 0.5 mL/kg, is the best bet[76].
Recommendations from various societies and groups have suggested aggressive fluid resuscitation in AP[7,34,71,77]; however, these recommendations were not based on any hard data. Although it is well known that some patients with AP have an increased need for fluid therapy, it is not clear who should get fluids aggressively. In a recent study, de-Madaria et al[14] prospectively calculated fluid sequestration in AP by subtracting fluid output from fluid intake in the first 48 h. Fluid intake included all intravenous fluids, oral fluids and blood, while fluid output included volumes of urine, stool, vomitus, as well as insensible losses (10 mL/kg per day). The median fluid sequestration in the first 48 h after hospitalization was 3.2 L (1.4-5 L), 6.4 L (3.6-9.5 L) in those without necrosis and those with necrosis, and 7.5 L (4.4-12 L) in those with persistent organ failure. Fluid sequestration correlated with age, alcohol as an etiology, hematocrit, and SIRS score. This study provided the first data for the possible fluid requirement in AP and determinants of aggressive fluid resuscitation. In a retrospective study performed in 1974, Ranson et al[29] had reported a mean fluid sequestration at 48 h of 3.7 L vs 5.6 L in mild and severe pancreatitis. The data from de-Madaria et al[14] will be useful in planning future prospective trials.
Tenner had given the first recommendation of rate of fluid replacement as 250-500 mL/h for 48 h in 2004[7]. Subsequently, Pandol et al[78] suggested that in cases of severe volume depletion fluids can be infused at 500-1000 mL/h followed by 300-500 mL/h to replace non-pancreatic fluid loss. However, these recommendations would amount to up to 6-12 L of fluid in the first 24 h, which is beyond the volumes used in different studies, both prospective and retrospective. Even in studies in which early aggressive hydration had been used, the total volume in the first 24 h did not generally exceed 6 L[72,73]. Whitcomb[79] was the first one to suggest that fluid should be started as a bolus, followed by maintenance. Nasr et al[8] suggested that a bolus of 20 mL/kg should be given in the emergency room, followed by 150-300 mL/h (3 mL/kg per hour) for 24 h. In their recent study on goal-directed fluid resuscitation, Wu et al[61] used a bolus of 20 mL/kg, followed by another bolus and 3 mL/kg per hour (210 mL/h in a 70 kg patient) in fluid refractory and 1.5 mL/kg per hour (105 mL/h) without another bolus in fluid responsible patients. Thus, it seems reasonable to start with a bolus and follow it up with maintenance fluid infusion. This recommendation is close to the guidelines for septic shock, which recommend an initial bolus with crystalloids at the rate of 1000 mL/h, with a minimum of 30 mL/kg, which also includes use of vasopressor epinephrine to maintain adequate blood pressure. However, more data is needed from prospective studies to reach consensus on this issue. It must be mentioned here that the Fluid Expansion as Supportive Therapy (FEAST) trial of fluid bolus in African children with severe infection had actually resulted in greater mortality[80].
A recent review has pointed out there are reports favoring and opposing early aggressive fluid resuscitation[9]. It has also been acknowledged that benefits of early aggressive therapy have not been confirmed prospectively[14]. Early aggressive therapy with predetermined fluid infusion for the first 24 h to 48 h is based on the assumption that fluid sequestration and hence fluid requirement over this time frame is the same for all the patients. However, de-Madaria et al[14] have reasoned that patients who develop local complications after admission are prone to more fluid sequestration, so they require more fluids. They suggest that fluid resuscitation and its replacement is a dynamic process and patients with local complications should receive heightened fluid intake on the second and third days of admission.
All the recommendations and guidelines presume that patients would report within 24 h of onset of pancreatitis. Since this is not the case, generally, especially in referral centers, there is a need to take this factor into account as, the longer the delay in hospitalization, the more established the hemodynamic alterations become.
Patients with co-morbidities like renal failure, cardiac compromise and pulmonary disease need special attention. As per the Revised Atlanta classification system, comorbidities are important determinants of severity of AP[3]. Moreover, occurrence of organ failure as a consequence of AP calls for special attention in such patients. Management of such patients may be extrapolated from the Surviving Sepsis Campaign guidelines for management of septic shock[81]. Issues in such patients that need special considerations include the rate of fluid transfused, use of additional agents like vasopressors, and the need for specific monitoring. There may be a need to restrict fluid infusion in those patients who have renal dysfunction or cardiac dysfunction. As in patients with sepsis, vasopressor therapy is required to maintain perfusion pressure in the face of hypotension, even when hypovolemia has not yet been treated. Some patients require vasopressors to achieve a minimum perfusion pressure. In patients with sepsis, titration of norepinephrine to a MAP as low as 65 mmHg has been shown to preserve tissue perfusion[82]. Norepinephrine is the recommended vasopressor in such situations[82]. Management of adult respiratory distress syndrome is as per standard guidelines[82]. Recently, extracorporeal life support for acute pancreatitis-induced acute respiratory distress syndrome has been shown to be beneficial[83].
Since hypercytokinemia is believed to be pivotal in pathophysiology of severe AP, the role of continuous hemodiafiltration (CHDF) has been investigated for removal of pro-inflammatory cytokines[84]. CHDF with a polymethylmethacrylate (PMMA) membrane removes various cytokines from the bloodstream and is widely used in Japan for blood purification therapy in patients with morbid conditions and is thought to prevent organ failure. In a retrospective review of 10 years’ experience, Pupelis et al[85] summarized the clinical application of continuous veno-venous hemofiltration (CVVH) in patients with AP and concluded that it may help in balancing fluid replacement and removal of cytokines from the blood and tissue compartments. Early application of CVVH helped in achieving cumulative negative fluid balance starting at day 5 in patients with AP and intra-abdominal hypertension (IAH) in one study[85]. It has been investigated for decreasing intra-abdominal pressure (IAP) in patients with AP[86-88], and Japanese guidelines suggest blood purification therapy as recommendation C in severe AP which may prevent OF but not the mortality[89]. Xu et al[90] recently demonstrated a decrease in tumor necrosis factor-alpha (TNF-α) levels after initiation of CVVH; they also showed a positive correlation between blood levels of TNF-α and IAP. Pupelis et al[91] have documented that use of CVVH as a part of conservative treatment of AP decreases the need for surgical intervention from 41% to 19%. Even though neither CHDF nor CVVH has been recommended so far in the treatment of severe AP, due to non-availability of quality evidence, it is expected to contribute in the improvement of AP outcome in the future[85].
Fluid resuscitation needs to be monitored by periodic assessment of cardiovascular, renal and pulmonary functions, as well as electrolyte imbalances. A drop in hematocrit and BUN has often been recommended as a marker of hemoconcentration correction. Hematocrit has been used for over 50 years to guide fluid replacement in critically ill patients; in AP, it has been identified as a marker that correlates with development of pancreatic necrosis[36,92]. Failure of hematocrit decrease has been correlated with increased necrosis and poor outcome. Similarly, elevated BUN has been used as a marker of severe disease, and the failure of BUN to decrease has been reported in patients with increased necrosis. In a recent study, Wu et al[61] used BUN to define fluid responsiveness. At 8-12 h after of the start of resuscitation, if the BUN level remained unchanged or increased from its previous value, participants were considered refractory. The authors used this key parameter as a target for goal-directed fluid therapy.
The aim of monitoring is accurate prediction of the response to fluids before volume expansion occurs. The classic static parameters for monitoring are central venous pressure (CVP), pulmonary artery occlusion pressure (PAOP) and MAP. However, these may be fallacious in patients receiving mechanical ventilation and in those with IAH[93]. The dynamic parameters measuring cardiac response to changes in preload, such as systolic volume variation and pulse pressure variation (PPV), can be used in patients on mechanical ventilation but requires caution in those with IAH[93]. In a recent study of an experimental model of AP, Trepte et al[94] showed that goal-directed fluid resuscitation using stroke volume variation (SVV) led to better survival, tissue oxygenation, and microcirculatory perfusion when compared to CVP and MAP measurements. However, in an accompanying editorial, it was pointed out that in an experimental setting resuscitation can be started immediately after induction of AP, but in clinical practice patients usually reach the hospital with already established dramatic fluid shifts with hypovolemia, increased hematocrit, and IAH[95]. Moreover, the use of SVV indispensably requires controlled mechanical ventilation, which is not a realistic scenario at admission of such patients. Also SVV/PPV are less useful in patients with IAH[95]. Huber et al[95] suggest that strategies combining volumetric and dynamic parameters (where applicable) with functional tests (passive leg raising and expiratory occlusion), and also taking into account IAP and abdominal perfusion pressure, need to be evaluated prospectively.
Though CVP is often used in critical care units to monitor fluid resuscitation, it is not an ideal end point in patients with AP because there is a disconnect between the intracellular space and the intravascular compartment[23]. A high CVP in patients with severe AP may indicate intravascular repletion when the intracellular compartment is actually under-resuscitated.
Urine output measurement remains an easy end point to monitor. Adequate urine output confirms adequate intravascular repletion. While urine output of > 0.5 mL/kg per hour was considered adequate by a technical review[96], the Japanese recommend a urine output greater than 1 mL/kg per hour[5]. However, recent studies have used > 0.5 mL/kg per hour as the end point of fluid resuscitation[61]. In a recent editorial, the same has also been recommended[76]. Similar to other critical illnesses, serum lactate levels have also been recommended to monitor resuscitation in AP[97]; however, there is no evidence of its application in patients with AP.
Because of the compounding influence of mechanical ventilation, IAH, pleural effusion and mediastinal edema, the pressure-based parameters may not accurately reflect adequacy of fluid replacement in AP. Therefore, newer hemodynamic measurements, such as the intrathoracic blood volume index, global end diastolic volume index and extravascular lung water index, have been suggested[49]. However, there is need to have more data and the fact that these measures are invasive makes it less likely that they will be used routinely.
Responsiveness to fluid resuscitation can be assessed by measurement of hematocrit, BUN, urine output or intrathoracic volume index, among other parameters. As highlighted above, Wu et al[61] have used BUN to classify patients as responsive or refractory to fluid resuscitation. A simple maneuver of leg elevation has been suggested to determine fluid responsiveness in patients having spontaneous breathing. An increase in ≥ 10% in cardiac output during the first 60-90 s of leg elevation can predict response to fluid replacement[98].
Severe AP can become complicated by IAH, which can become worsened by aggressive fluid resuscitation. In healthy individuals, IAP ranges from 0 mmHg to 5 mmHg and varies with the respiratory cycle. IAH is a sustained increase in IAP > 12 mmHg. Abdominal compartment syndrome is a sustained increase in IAP > 20 mmHg, with new onset organ failure with or without a low abdominal perfusion pressure. The incidence of IAH in patients with severe AP is approximately 60% to 80%[97]. IAH is usually an early phenomenon, partly related to the effects of the inflammatory process causing retroperitoneal edema, fluid collection, ascites and ileus and is partly iatrogenic, resulting from aggressive fluid resuscitation[97]. It is associated with impaired organ dysfunction, especially of the cardiovascular, respiratory and renal systems[98].
Since it contributes to fluid non-responsiveness and carries a high surgical mortality, IAH should be monitored in all patients with severe AP. Presence of intra-abdominal pressure > 16 mmHg makes it difficult to correctly interpret results of passive leg raising, as well as of other static and dynamic pressure parameters. Abdominal perfusion pressure can serve as a good marker and resuscitation end point in patients with IAH[99]. Maintaining abdominal perfusion pressure above 50-60 mmHg is recommended in order to facilitate adequate perfusion of abdominal organs[97]. There is some evidence that use of HES resuscitation reduces the risk of IAH in severe AP[59]; however, HES may actually increase mortality in critically ill patients[60].
As the pathogenesis of AP is a dynamic process, with local and systemic complications compounding the clinical management, it is imperative to monitor fluid resuscitation closely. After an initial bolus of 20 mL/kg or 1000 mL over one hour, followed by controlled fluid infusion for the first 24 h, the patients should be evaluated closely in a high dependency unit. Hemodynamically, the goals chosen to monitor should be assessed at 8 h to 12 h intervals. Wu et al[61] evaluated their patients every 8 h for the first 24 h. Although there are no guidelines on this issue, it seems logical to follow the practice of Wu et al[61].
Fluid resuscitation has emerged as a key therapeutic strategy in patients with acute pancreatitis. It has to be acknowledged that fluid resuscitation in AP is a complex process, with need to take into account the dynamics of fluid sequestration during different stages of the disease. Current knowledge suggests that controlled fluid resuscitation (3.0-4.0 L/24 h) should be started after a bolus infusion of 20 mL/kg (1000 mL over one hour). Among the different fluids, lactated Ringers’ is the one recommended by most guidelines. There is a need to carry out fluid resuscitation with a goal-directed strategy, with urine output > 0.5 mL/kg and a drop in BUN as simple goals. There is a need for randomized controlled trials to generate data on all the three issues addressed (rate, type and end points of fluid resuscitation) before definite guidelines can be framed. The authors have summarized recommendations about fluid therapy in predicted severe or severe AP (Table 3).
| Parameter | Recommendation |
| Fluid resuscitation | Necessary: the earlier the resuscitation, the better the outcome |
| Type of fluid | Colloids and/or crystalloids: Among crystalloids, lactate Ringer’s better than normal salineUse colloids especially when albumin < 2.0 g/dL or hematocrit < 35% |
| Amount of fluid | Total fluid in first 24 h: between 3 and 4 L, Not to exceed 4 L |
| Rate of infusion | Initial bolus 1000 mL over one hour followed by 3 mL/kg per hour (200 mL/h) for 24-48 h |
| Monitoring | Urine output > 0.5 mL/kg/h, hematocrit = 25% to 35%, drop in BUNCVP: Not good for monitoring due to third space loss and hypoalbuminemia |
| Duration of resuscitation | 24-48 h, until signs of volume depletion disappear |
P- Reviewer: Fujino Y, Sakata n, Shehata MMM S- Editor: Ma YJ L- Editor: AmEditor E- Editor: Zhang DN
| 1. | Lowenfels AB, Maisonneuve P, Sullivan T. The changing character of acute pancreatitis: epidemiology, etiology, and prognosis. Curr Gastroenterol Rep. 2009;11:97-103. [PubMed] |
| 2. | Bradley EL. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, Ga, September 11 through 13, 1992. Arch Surg. 1993;128:586-590. [PubMed] |
| 3. | Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, Tsiotos GG, Vege SS. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4932] [Cited by in RCA: 4329] [Article Influence: 360.8] [Reference Citation Analysis (45)] |
| 4. | Halonen KI, Pettilä V, Leppäniemi AK, Kemppainen EA, Puolakkainen PA, Haapiainen RK. Multiple organ dysfunction associated with severe acute pancreatitis. Crit Care Med. 2002;30:1274-1279. [PubMed] |
| 5. | Wu BU, Conwell DL. Update in acute pancreatitis. Curr Gastroenterol Rep. 2010;12:83-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Norman J. The role of cytokines in the pathogenesis of acute pancreatitis. Am J Surg. 1998;175:76-83. [PubMed] |
| 7. | Tenner S. Initial management of acute pancreatitis: critical issues during the first 72 hours. Am J Gastroenterol. 2004;99:2489-2494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 68] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 8. | Nasr JY, Papachristou GI. Early fluid resuscitation in acute pancreatitis: a lot more than just fluids. Clin Gastroenterol Hepatol. 2011;9:633-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Haydock MD, Mittal A, van den Heever M, Rossaak JI, Connor S, Rodgers M, Petrov MS, Windsor JA. National survey of fluid therapy in acute pancreatitis: current practice lacks a sound evidence base. World J Surg. 2013;37:2428-2435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Gurusamy KS, Farouk M, Tweedie JH. UK guidelines for management of acute pancreatitis: is it time to change? Gut. 2005;54:1344-1345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 11. | Banks PA, Freeman ML. Practice guidelines in acute pancreatitis. Am J Gastroenterol. 2006;101:2379-2400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1181] [Cited by in RCA: 1150] [Article Influence: 60.5] [Reference Citation Analysis (0)] |
| 13. | Loveday BP, Srinivasa S, Vather R, Mittal A, Petrov MS, Phillips AR, Windsor JA. High quantity and variable quality of guidelines for acute pancreatitis: a systematic review. Am J Gastroenterol. 2010;105:1466-1476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 14. | de-Madaria E, Soler-Sala G, Sánchez-Payá J, Lopez-Font I, Martínez J, Gómez-Escolar L, Sempere L, Sánchez-Fortún C, Pérez-Mateo M. Influence of fluid therapy on the prognosis of acute pancreatitis: a prospective cohort study. Am J Gastroenterol. 2011;106:1843-1850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 143] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 15. | Eckerwall G, Olin H, Andersson B, Andersson R. Fluid resuscitation and nutritional support during severe acute pancreatitis in the past: what have we learned and how can we do better? Clin Nutr. 2006;25:497-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 69] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 16. | Mao EQ, Fei J, Peng YB, Huang J, Tang YQ, Zhang SD. Rapid hemodilution is associated with increased sepsis and mortality among patients with severe acute pancreatitis. Chin Med J (Engl). 2010;123:1639-1644. [PubMed] |
| 17. | Mao EQ, Tang YQ, Fei J, Qin S, Wu J, Li L, Min D, Zhang SD. Fluid therapy for severe acute pancreatitis in acute response stage. Chin Med J (Engl). 2009;122:169-173. [PubMed] |
| 18. | Mao EQ, Tang YQ, Li L, Qin S, Wu J, Liu W, Lei RQ, Zhang SD. [Strategy of controlling fluid resuscitation for severe acute pancreatitis in acute phase]. Zhonghua Wai Ke Za Zhi. 2007;45:1331-1334. [PubMed] |
| 19. | Mayer J, Rau B, Gansauge F, Beger HG. Inflammatory mediators in human acute pancreatitis: clinical and pathophysiological implications. Gut. 2000;47:546-552. [PubMed] |
| 20. | Hack CE, Zeerleder S. The endothelium in sepsis: source of and a target for inflammation. Crit Care Med. 2001;29:S21-S27. [PubMed] |
| 21. | Mentula P, Kylänpää-Bäck ML, Kemppainen E, Takala A, Jansson SE, Kautiainen H, Puolakkainen P, Haapiainen R, Repo H. Decreased HLA (human leucocyte antigen)-DR expression on peripheral blood monocytes predicts the development of organ failure in patients with acute pancreatitis. Clin Sci (Lond). 2003;105:409-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 53] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 22. | Mofidi R, Duff MD, Wigmore SJ, Madhavan KK, Garden OJ, Parks RW. Association between early systemic inflammatory response, severity of multiorgan dysfunction and death in acute pancreatitis. Br J Surg. 2006;93:738-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 363] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 23. | Mole DJ, Olabi B, Robinson V, Garden OJ, Parks RW. Incidence of individual organ dysfunction in fatal acute pancreatitis: analysis of 1024 death records. HPB (Oxford). 2009;11:166-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 24. | Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med. 1995;23:1638-1652. [PubMed] |
| 25. | Barreto SG, Rodrigues J. Comparison of APACHE II and Imrie Scoring Systems in predicting the severity of Acute Pancreatitis. World J Emerg Surg. 2007;2:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 26. | Lankisch PG, Weber-Dany B, Hebel K, Maisonneuve P, Lowenfels AB. The harmless acute pancreatitis score: a clinical algorithm for rapid initial stratification of nonsevere disease. Clin Gastroenterol Hepatol. 2009;7:702-75; quiz 607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 105] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 27. | Papachristou GI, Muddana V, Yadav D, O’Connell M, Sanders MK, Slivka A, Whitcomb DC. Comparison of BISAP, Ranson’s, APACHE-II, and CTSI scores in predicting organ failure, complications, and mortality in acute pancreatitis. Am J Gastroenterol. 2010;105:435-41; quiz 442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 331] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 28. | Petrov MS, Windsor JA. Classification of the severity of acute pancreatitis: how many categories make sense? Am J Gastroenterol. 2010;105:74-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 29. | Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet. 1974;139:69-81. [PubMed] |
| 30. | Vege SS, Gardner TB, Chari ST, Munukuti P, Pearson RK, Clain JE, Petersen BT, Baron TH, Farnell MB, Sarr MG. Low mortality and high morbidity in severe acute pancreatitis without organ failure: a case for revising the Atlanta classification to include “moderately severe acute pancreatitis”. Am J Gastroenterol. 2009;104:710-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 101] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 31. | Wu BU, Banks PA. Clinical management of patients with acute pancreatitis. Gastroenterology. 2013;144:1272-1281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 125] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 32. | Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA. The early prediction of mortality in acute pancreatitis: a large population-based study. Gut. 2008;57:1698-1703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 451] [Cited by in RCA: 516] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 33. | Singh VK, Wu BU, Bollen TL, Repas K, Maurer R, Johannes RS, Mortele KJ, Conwell DL, Banks PA. A prospective evaluation of the bedside index for severity in acute pancreatitis score in assessing mortality and intermediate markers of severity in acute pancreatitis. Am J Gastroenterol. 2009;104:966-971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 175] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 34. | Gardner TB, Vege SS, Pearson RK, Chari ST. Fluid resuscitation in acute pancreatitis. Clin Gastroenterol Hepatol. 2008;6:1070-1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 111] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 35. | Gray SH, Rosenman LD. Acute pancreatitis. The significance of hemoconcentration at admission to the hospital. Arch Surg. 1965;91:485-489. [PubMed] |
| 36. | Brown A, Orav J, Banks PA. Hemoconcentration is an early marker for organ failure and necrotizing pancreatitis. Pancreas. 2000;20:367-372. [PubMed] |
| 37. | Wu BU, Conwell DL, Singh VK, Repas K, Maurer R, Bollen TL, Mortele KJ, Banks PA. Early hemoconcentration is associated with pancreatic necrosis only among transferred patients. Pancreas. 2010;39:572-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 38. | Gardner TB. BUN level as a marker of severity in acute pancreatitis: simple, universal, and accurate: comment on “Blood urea nitrogen in the early assessment of acute pancreatitis”. Arch Intern Med. 2011;171:676-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 39. | Wu BU, Bakker OJ, Papachristou GI, Besselink MG, Repas K, van Santvoort HC, Muddana V, Singh VK, Whitcomb DC, Gooszen HG. Blood urea nitrogen in the early assessment of acute pancreatitis: an international validation study. Arch Intern Med. 2011;171:669-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 120] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 40. | Wu BU, Johannes RS, Sun X, Conwell DL, Banks PA. Early changes in blood urea nitrogen predict mortality in acute pancreatitis. Gastroenterology. 2009;137:129-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 142] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 41. | Baillargeon JD, Orav J, Ramagopal V, Tenner SM, Banks PA. Hemoconcentration as an early risk factor for necrotizing pancreatitis. Am J Gastroenterol. 1998;93:2130-2134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 110] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 42. | Muddana V, Whitcomb DC, Khalid A, Slivka A, Papachristou GI. Elevated serum creatinine as a marker of pancreatic necrosis in acute pancreatitis. Am J Gastroenterol. 2009;104:164-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 113] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 43. | Yang R, Uchiyama T, Alber SM, Han X, Watkins SK, Delude RL, Fink MP. Ethyl pyruvate ameliorates distant organ injury in a murine model of acute necrotizing pancreatitis. Crit Care Med. 2004;32:1453-1459. [PubMed] |
| 44. | Zhao G, Zhang JG, Wu HS, Tao J, Qin Q, Deng SC, Liu Y, Liu L, Wang B, Tian K. Effects of different resuscitation fluid on severe acute pancreatitis. World J Gastroenterol. 2013;19:2044-2052. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 64] [Cited by in RCA: 56] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 45. | Juvonen PO, Tenhunen JJ, Heino AA, Merasto M, Paajanen HE, Alhava EM, Takala JA. Splanchnic tissue perfusion in acute experimental pancreatitis. Scand J Gastroenterol. 1999;34:308-314. [PubMed] |
| 46. | Niederau C, Crass RA, Silver G, Ferrell LD, Grendell JH. Therapeutic regimens in acute experimental hemorrhagic pancreatitis. Effects of hydration, oxygenation, peritoneal lavage, and a potent protease inhibitor. Gastroenterology. 1988;95:1648-1657. [PubMed] |
| 47. | Knol JA, Inman MG, Strodel WE, Eckhauser FE. Pancreatic response to crystalloid resuscitation in experimental pancreatitis. J Surg Res. 1987;43:387-392. [PubMed] |
| 48. | Martin DT, Kopolovic R, Gower WR, Steinberg SM, Cloutier CT, Carey LC. Colloid vs. crystalloid resuscitation in experimental hemorrhagic pancreatitis. Curr Surg. 1984;41:189-192. [PubMed] |
| 49. | Trikudanathan G, Navaneethan U, Vege SS. Current controversies in fluid resuscitation in acute pancreatitis: a systematic review. Pancreas. 2012;41:827-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 50. | Ring J, Messmer K. Incidence and severity of anaphylactoid reactions to colloid volume substitutes. Lancet. 1977;1:466-469. [PubMed] |
| 51. | Shields CJ, Sookhai S, Winter DC, Dowdall JF, Kingston G, Parfrey N, Wang JH, Kirwan WO, Redmond HP. Attenuation of pancreatitis-induced pulmonary injury by aerosolized hypertonic saline. Surg Infect (Larchmt). 2001;2:215-23; discussion 223-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 52. | Sterns RH, Riggs JE, Schochet SS. Osmotic demyelination syndrome following correction of hyponatremia. N Engl J Med. 1986;314:1535-1542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 548] [Cited by in RCA: 464] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 53. | Schmidt J, Ferńandez-del Castillo C, Rattner DW, Lewandrowski KB, Messmer K, Warshaw AL. Hyperoncotic ultrahigh molecular weight dextran solutions reduce trypsinogen activation, prevent acinar necrosis, and lower mortality in rodent pancreatitis. Am J Surg. 1993;165:40-4; discussion 45. [PubMed] |
| 54. | Donaldson LA, Schenk WG. Experimental acute pancreatitis: the changes in pancreatic oxygen consumption and the effect of Dextran 40. Ann Surg. 1979;190:728-731. [PubMed] |
| 55. | Klar E, Foitzik T, Buhr H, Messmer K, Herfarth C. Isovolemic hemodilution with dextran 60 as treatment of pancreatic ischemia in acute pancreatitis. Clinical practicability of an experimental concept. Ann Surg. 1993;217:369-374. [PubMed] |
| 56. | Klar E, Herfarth C, Messmer K. Therapeutic effect of isovolemic hemodilution with dextran 60 on the impairment of pancreatic microcirculation in acute biliary pancreatitis. Ann Surg. 1990;211:346-353. [PubMed] |
| 57. | Schmidt J, Huch K, Mithöfer K, Hotz HG, Sinn HP, Buhr HJ, Warshaw AL, Herfarth C, Klar E. Benefits of various dextrans after delayed therapy in necrotizing pancreatitis of the rat. Intensive Care Med. 1996;22:1207-1213. [PubMed] |
| 58. | Wang ZF, Liu C, Lu Y, Dong R, Xu J, Yu L, Yao YM, Liu QG, Pan CE. Dexamethasone and dextran 40 treatment of 32 patients with severe acute pancreatitis. World J Gastroenterol. 2004;10:1333-1336. [PubMed] |
| 59. | Du XJ, Hu WM, Xia Q, Huang ZW, Chen GY, Jin XD, Xue P, Lu HM, Ke NW, Zhang ZD. Hydroxyethyl starch resuscitation reduces the risk of intra-abdominal hypertension in severe acute pancreatitis. Pancreas. 2011;40:1220-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 60. | Perel P, Roberts I, Ker K. Colloids versus crystalloids for fluid resuscitation in critically ill patients. Cochrane Database Syst Rev. 2013;2:CD000567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 186] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 61. | Wu BU, Hwang JQ, Gardner TH, Repas K, Delee R, Yu S, Smith B, Banks PA, Conwell DL. Lactated Ringer’s solution reduces systemic inflammation compared with saline in patients with acute pancreatitis. Clin Gastroenterol Hepatol. 2011;9:710-717.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 340] [Article Influence: 24.3] [Reference Citation Analysis (1)] |
| 62. | Horton JW, Dunn CW, Burnweit CA, Walker PB. Hypertonic saline-dextran resuscitation of acute canine bile-induced pancreatitis. Am J Surg. 1989;158:48-56. [PubMed] |
| 63. | Shields CJ, Winter DC, Sookhai S, Ryan L, Kirwan WO, Redmond HP. Hypertonic saline attenuates end-organ damage in an experimental model of acute pancreatitis. Br J Surg. 2000;87:1336-1340. [PubMed] |
| 64. | Huang PP, Stucky FS, Dimick AR, Treat RC, Bessey PQ, Rue LW. Hypertonic sodium resuscitation is associated with renal failure and death. Ann Surg. 1995;221:543-54; discussion 554-7. [PubMed] |
| 65. | Mayerle J, Hlouschek V, Lerch MM. Current management of acute pancreatitis. Nat Clin Pract Gastroenterol Hepatol. 2005;2:473-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 54] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 66. | Chang YS, Fu HQ, Zou SB, Yu BT, Liu JC, Xia L, Lv NH. [The impact of initial fluid resuscitation with different ratio of crystalloid-colloid on prognosis of patients with severe acute pancreatitis]. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2013;25:48-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 67. | Strate T, Mann O, Kleinhans H, Schneider C, Knoefel WT, Yekebas E, Standl T, Bloechle C, Izbicki JR. Systemic intravenous infusion of bovine hemoglobin significantly reduces microcirculatory dysfunction in experimentally induced pancreatitis in the rat. Ann Surg. 2003;238:765-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 68. | Freitag M, Standl TG, Kleinhans H, Gottschalk A, Mann O, Rempf C, Bachmann K, Gocht A, Petri S, Izbicki JR. Improvement of impaired microcirculation and tissue oxygenation by hemodilution with hydroxyethyl starch plus cell-free hemoglobin in acute porcine pancreatitis. Pancreatology. 2006;6:232-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 69. | Wang J, Chen Y, Dong Y, Hu W, Zhou P, Chang L, Feng S, Lin J, Zhao Y. [Role of 6% hydroxyethylstarch 130/0.4 and furosemide in the treatment of acute pancreatitis]. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2010;27:1138-141, 1145. [PubMed] |
| 71. | Tenner S, Baillie J, DeWitt J, Vege SS. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108:1400-115; 1416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1232] [Cited by in RCA: 1382] [Article Influence: 115.2] [Reference Citation Analysis (3)] |
| 72. | Gardner TB, Vege SS, Chari ST, Petersen BT, Topazian MD, Clain JE, Pearson RK, Levy MJ, Sarr MG. Faster rate of initial fluid resuscitation in severe acute pancreatitis diminishes in-hospital mortality. Pancreatology. 2009;9:770-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 135] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 73. | Warndorf MG, Kurtzman JT, Bartel MJ, Cox M, Mackenzie T, Robinson S, Burchard PR, Gordon SR, Gardner TB. Early fluid resuscitation reduces morbidity among patients with acute pancreatitis. Clin Gastroenterol Hepatol. 2011;9:705-709. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 151] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 74. | Wall I, Badalov N, Baradarian R, Iswara K, Li JJ, Tenner S. Decreased mortality in acute pancreatitis related to early aggressive hydration. Pancreas. 2011;40:547-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 75. | Kuwabara K, Matsuda S, Fushimi K, Ishikawa KB, Horiguchi H, Fujimori K. Early crystalloid fluid volume management in acute pancreatitis: association with mortality and organ failure. Pancreatology. 2011;11:351-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 76. | Sarr MG. Early fluid “resuscitation/therapy” in acute pancreatitis: which fluid? What rate? What parameters to gauge effectiveness? Ann Surg. 2013;257:189-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 77. | Whitcomb DC. Genetic polymorphisms in alcoholic pancreatitis. Dig Dis. 2005;23:247-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 78. | Pandol SJ, Saluja AK, Imrie CW, Banks PA. Acute pancreatitis: bench to the bedside. Gastroenterology. 2007;132:1127-1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 449] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 79. | Whitcomb DC. Clinical practice. Acute pancreatitis. N Engl J Med. 2006;354:2142-2150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 518] [Article Influence: 27.3] [Reference Citation Analysis (1)] |
| 80. | Maitland K, Kiguli S, Opoka RO, Engoru C, Olupot-Olupot P, Akech SO, Nyeko R, Mtove G, Reyburn H, Lang T. Mortality after fluid bolus in African children with severe infection. N Engl J Med. 2011;364:2483-2495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1259] [Cited by in RCA: 1084] [Article Influence: 77.4] [Reference Citation Analysis (0)] |
| 81. | Dellinger RP, Carlet JM, Gerlach H, Ramsey G, Levy M. The surviving sepsis guidelines: not another “groundhog day”. Crit Care Med. 2004;32:1601-1602. [PubMed] |
| 82. | Dellinger RP, Carlet JM, Masur H, Gerlach H, Calandra T, Cohen J, Gea-Banacloche J, Keh D, Marshall JC, Parker MM. Surviving Sepsis Campaign guidelines for management of severe sepsis and septic shock. Crit Care Med. 2004;32:858-873. [PubMed] |
| 83. | Bryner BS, Smith C, Cooley E, Bartlett RH, Mychaliska GB. Extracorporeal life support for pancreatitis-induced acute respiratory distress syndrome. Ann Surg. 2012;256:1073-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 84. | Oda S, Hirasawa H, Shiga H, Nakanishi K, Matsuda K, Nakamura M. Continuous hemofiltration/hemodiafiltration in critical care. Ther Apher. 2002;6:193-198. [PubMed] |
| 85. | Pupelis G, Plaudis H, Zeiza K, Drozdova N, Mukans M, Kazaka I. Early continuous veno-venous haemofiltration in the management of severe acute pancreatitis complicated with intra-abdominal hypertension: retrospective review of 10 years’ experience. Ann Intensive Care. 2012;2 Suppl 1:S21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 86. | Jiang HL, Xue WJ, Li DQ, Yin AP, Xin X, Li CM, Gao JL. Influence of continuous veno-venous hemofiltration on the course of acute pancreatitis. World J Gastroenterol. 2005;11:4815-4821. [PubMed] |
| 87. | Yan XW, Li WQ, Wang H, Zhang ZH, Li N, Li JS. Effects of high-volume continuous hemofiltration on experimental pancreatitis associated lung injury in pigs. Int J Artif Organs. 2006;29:293-302. [PubMed] |
| 88. | Mao EQ, Tang YQ, Zhang SD. Effects of time interval for hemofiltration on the prognosis of severe acute pancreatitis. World J Gastroenterol. 2003;9:373-376. [PubMed] |
| 89. | Mayumi T, Ura H, Arata S, Kitamura N, Kiriyama I, Shibuya K, Sekimoto M, Nago N, Hirota M, Yoshida M. Evidence-based clinical practice guidelines for acute pancreatitis: proposals. J Hepatobiliary Pancreat Surg. 2002;9:413-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 65] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 90. | Xu J, Tian X, Zhang C, Wang M, Li Y. Management of abdominal compartment syndrome in severe acute pancreatitis patients with early continuous veno-venous hemofiltration. Hepatogastroenterology. 2013;60:1749-1752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 91. | Pupelis G, Zeiza K, Plaudis H, Suhova A. Conservative approach in the management of severe acute pancreatitis: eight-year experience in a single institution. HPB (Oxford). 2008;10:347-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 92. | Lankisch PG, Mahlke R, Blum T, Bruns A, Bruns D, Maisonneuve P, Lowenfels AB. Hemoconcentration: an early marker of severe and/or necrotizing pancreatitis? A critical appraisal. Am J Gastroenterol. 2001;96:2081-2085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 111] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 93. | Maraví Poma E, Zubia Olascoaga F, Petrov MS, Navarro Soto S, Laplaza Santos C, Morales Alava F, Darnell Martin A, Gorraiz López B, Bolado Concejo F, Casi Villarroya M. SEMICYUC 2012. Recommendations for intensive care management of acute pancreatitis. Med Intensiva. 2013;37:163-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 94. | Trepte CJ, Bachmann KA, Stork JH, Friedheim TJ, Hinsch A, Goepfert MS, Mann O, Izbicki JR, Goetz AE, Reuter DA. The impact of early goal-directed fluid management on survival in an experimental model of severe acute pancreatitis. Intensive Care Med. 2013;39:717-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 95. | Huber W, Malbrain ML. Goal-directed fluid resuscitation in acute pancreatitis: shedding light on the penumbra by dynamic markers of preload? Intensive Care Med. 2013;39:784-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 96. | Forsmark CE, Baillie J. AGA Institute technical review on acute pancreatitis. Gastroenterology. 2007;132:2022-2044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 505] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 97. | Mentula P, Leppäniemi A. Position paper: timely interventions in severe acute pancreatitis are crucial for survival. World J Emerg Surg. 2014;9:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 98. | Préau S, Saulnier F, Dewavrin F, Durocher A, Chagnon JL. Passive leg raising is predictive of fluid responsiveness in spontaneously breathing patients with severe sepsis or acute pancreatitis. Crit Care Med. 2010;38:819-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 155] [Article Influence: 10.3] [Reference Citation Analysis (0)] |