Published online Nov 21, 2014. doi: 10.3748/wjg.v20.i43.16311
Revised: February 22, 2014
Accepted: July 16, 2014
Published online: November 21, 2014
Processing time: 314 Days and 1.4 Hours
AIM: To evaluate the effectiveness of trimodal imaging endoscopy (TME) to detect another lesion after endoscopic submucosal dissection (ESD) for superficial gastric neoplasia (SGN).
METHODS: Surveillance esophagogastroduodenoscopy (EGD) using a TME was conducted in 182 patients that had undergone ESD for SGN. Autofluorescence imaging (AFI) was conducted after white-light imaging (WLI). When SGN was suspicious, magnifying endoscopy with narrow-band imaging (ME-NBI) was conducted. Final diagnoses were made by histopathologic findings of biopsy specimens. The detection rates of lesions in WLI, AFI, and NBI, and the characteristics of lesions detected by WLI and ones missed by WLI but detected by AFI were examined. The sensitivity, specificity, and accuracy of endoscopic diagnosis using WLI, AFI and ME-NBI were evaluated.
RESULTS: In 242 surveillance EGDs, 27 lesions were determined pathologically to be neoplasias. Sixteen early gastric cancers and 6 gastric adenomas could be detected by WLI. Sixteen lesions were reddish and 6 were whitish. Five gastric neoplasias were missed by WLI but were detected by AFI, and all were whitish and protruded gastric adenomas. There was a significant difference in color and pathology between the two groups (P = 0.006). Sensitivity, specificity and accuracy in ME-NBI were higher than those in both WLI and AFI. Specificity and accuracy in AFI were lower than those in WLI.
CONCLUSION: Surveillance using trimodal imaging endoscopy might be useful for detecting another lesion after endoscopic submucosal dissection for superficial gastric neoplasia.
Core tip: We report surveillance esophagogastroduodenoscopy (EGD) using trimodal imaging videoendoscopy (TME) to detect another lesion after endoscopic submucosal dissection (ESD) for superficial gastric neoplasia (SGN). In 242 surveillance EGDs, 16 early gastric cancers and 11 gastric adenomas were detected, 5 of which were missed by white-light imaging (WLI) but were detected by autofluorescence imaging (AFI). The five were whitish and protruded gastric adenomas. Sensitivity, specificity and accuracy in magnifying endoscopy with narrow-band imaging were higher compared to those in both WLI and AFI. Surveillance using TME is useful for detecting another lesion after ESD for SGN.
- Citation: Imaeda H, Hosoe N, Kashiwagi K, Ida Y, Nakamura R, Suzuki H, Saito Y, Yahagi N, Iwao Y, Kitagawa Y, Hibi T, Ogata H, Kanai T. Surveillance using trimodal imaging endoscopy after endoscopic submucosal dissection for superficial gastric neoplasia. World J Gastroenterol 2014; 20(43): 16311-16317
- URL: https://www.wjgnet.com/1007-9327/full/v20/i43/16311.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i43.16311
Endoscopic submucosal dissection (ESD) for early gastric cancer (EGC) has been developed recently[1-3]. Since en bloc resection is needed for accurate histopathologic diagnosis, ESD has become the standard endoscopic treatment for EGC in Japan. Metachronous multiple gastric cancers, however, developed within 3-5 years in 2.7%-14% of patients after endoscopic resection (ER) for EGC[4-7]. The frequency of metachronous gastric cancers in patients after ER for EGC is higher than that of gastric cancers in the general population. Eradication of Helicobacter pylori (H. pylori) infection was reported to decrease the incidence of metachronous gastric cancers in patients who had undergone ER for EGC, but about 3% of them had metachronous lesions at 3-year follow-up[8]. Surveillance using esophagogastroduodenoscopy (EGD) after ER for EGC is therefore very important for detecting such lesions at the early stage, when they are still resectable by ESD.
Exogenous fluorophores such as 5-aminolevulinic acid[9] and endogenous fluorophores such as collagens, nicotinamides, adenine dinucleotides, flavins, and porphyrins[10-12] are used in autofluorescence (AF) endoscopy for examining the gastrointestinal (GI) tract. When the mucosal layer and the submucosal layer are illuminated with blue excitation light, green AF light is emitted from endogenous fluorophores in the submucosal layer. The intensity of this AF was diminished by abnormal vessels, the thick mucosa, fibrosis, inflammation, and the changes of endogenous fluorophores correlated with the neoplasia[10-13]. AF videoendoscopy (GIF-FQ260Z, Olympus Medical Systems Corp., Tokyo, Japan) using combination of autofluorescence imaging (AFI) and reflectance imaging has been reported in observation of superficial neoplasias in the esophagus[14-17], stomach[18,19], and colon[19]. Kato et al[20] reported that AFI detected 10% more neoplasms than did white-light imaging (WLI) and that although the sensitivities of WLI and AFI are comparable, clinical value of AFI is limited because its specificity is considerably lower than that of WLI (i.e., its use resulted in a large number of false positives). The GIF-FQ260Z videoendoscopy combines not only AFI endoscopy but also magnifying endoscopy with WLI and narrow-band imaging (NBI) in a single endoscope with two charge-coupled devices. It is a trimodal imaging endoscopy (TME) system, and TME is effective for detecting early-stage neoplasias in the esophagus[21], stomach[20] and colon[22].
The aim of this study was to evaluate the ability of surveillance EGD using TME system to detect another lesion after ESD for SGN.
One hundred eighty-two patients (147 male and 35 female; mean age 70.2 years) who had undergone ESD for SGN at Keio University Hospital were enrolled between January 2010 and December 2011 in this prospective study.
The study protocol was approved by the institutional review board at our hospital, and it was registered with the UMIN Clinical Trials Registry (UMIN000003120). Informed consent was obtained from each patient.
We used a high-resolution magnifying endoscopy with 80-fold optical magnification (GIF-FQ260Z), that is able to be easily switched from WLI mode to AFI or NBI mode and the standard videoendoscopy system (LUCERA CV-260SL; Olympus Medical Systems Corp.). The TME system conducts illumination of red, green, and blue light, which were used for WLI and NBI and illumination of an excitation/reflected light, which were used for AFI. Hemoglobin absorbs short-wavelength blue light, therefore, the appearance of microvessels in the superficial mucosal layer is enhanced. NBI mode was performed with magnification. The AFI allowed the light emitted from a xenon lamp to direct to a rotating filter wheel that passed 390-470 nm excitation light and 540-560 nm green light. A pseudocolor image based on the AF signals and reflected light was reconstructed, therefore high-intensity of AF showed green color and low-intensity of AF showed magenta color. The AFI system displayed the pyloric mucosa or the atrophic mucosa as green, blood vessels as dark green, and the fundic mucosa or neoplasia as magenta.
Patients underwent surveillance EGD using TME system under conscious sedation with flunitrazepam (0.2-0.4 mg). First, an examiner performed endoscopic observation using WLI, second, did that using AFI. Third, if lesions were suspected by WLI or AFI, endoscopic observation using magnifying endoscopy with narrow-band imaging (ME-NBI) was performed. All lesions identified by WLI or AFI modalities that seemed to be neoplasias were recorded as endoscopic neoplasias and biopsies were performed. A lesion identified by WLI endoscopy was recorded as a neoplasia if it was, from its color and appearance, recognized as or suspected to be an EGC or adenoma. A lesion identified by AFI endoscopy was recorded as a neoplasia if its color was different from that of the surrounding mucosa and its circumferential margin was definite. A lesion in ME-NBI was recorded as a neoplasia if it could be demarcated from the surrounding mucosa by irregular microsurface pattern and/or irregular microvascular pattern. The histopathologic finding obtained from the biopsy specimens were deemed to be the gold standard for the diagnosis. The examinations were performed by one of four endoscopists. AFI and ME-NBI were considerably different from WLI, the AFI and ME-NBI examinations were, therefore, performed by endoscopists who had previously experienced diagnosis of EGC by TME system using AFI and ME-NBI mode for more than 20 lesions.
Characteristics of SGNs detected by WLI and ones missed by WLI but detected by AFI were analysed. The sensitivity, specificity, and accuracy of endoscopic diagnosis using WLI, AFI and ME-NBI of pathology-proven neoplasia were evaluated. Assessment of the spread of atrophic gastritis evident by AFI mode was based on Kimura-Takemoto classification: closed-type gastric mucosal atrophy has an atrophic border in the antrum or the lesser curvature of the gastric body, and open-type gastric mucosal atrophy has that in the lateral wall or the greater curvature of it[23]. If the mucosa in the lesser curvature of the gastric body was purple, the diagnosis was closed-type atrophic gastritis; if the mucosa in the lateral wall or the greater curvature of it was green, the diagnosis was open-type atrophic gastritis.
Biopsy specimens were immersed in 10% formalin, embedded in paraffin, sectioned serially, and stained with hematoxylin and eosin. The diagnostic histopathologic criteria were based on the revised Vienna classification[24]. Category 3, which was mucosal low-grade neoplasia was classified as gastric adenoma, and category 4, which was mucosal high-grade neoplasia was classified as gastric carcinoma. Expert GI pathologists diagnosed the histopathologic findings of all biopsy specimens.
The data were analysed using SPSS software, version 17 (SPSS Inc., Chicago, IL), and statistical significance was assessed by Fischer’s exact test, Student’s t-test and Mann-Whitney’s U test. All probability values calculated in this analysis were sided, and P values of less than 0.05 were considered to denote statistical significance.
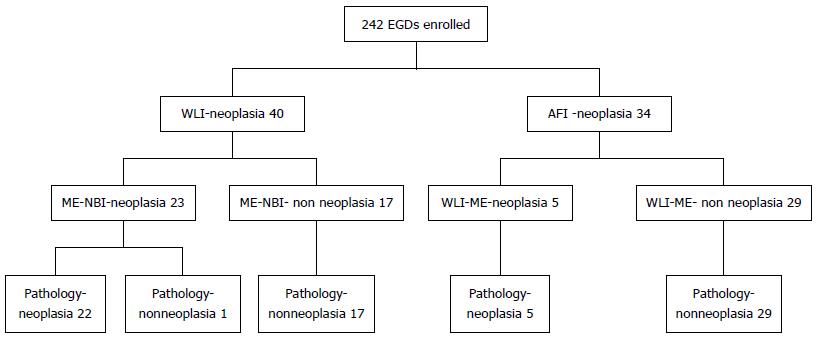
Two hundred and forty-two surveillance EGDs using the TME system were conducted in 182 patients. EGD was performed once in 129 patients, twice in 46 patients, and three times in 7 patients. Forty lesions detected by WLI were suspected to be neoplasias; of these lesions, 23 were identified as possible neoplasias by ME-NBI. Twenty-two of these 23 lesions were determined pathologically to be neoplasias. Thirty-four lesions detected by AFI were suspected to be neoplasias, five of them were identified as possible neoplasias by ME-NBI. All five were determined pathologically to be neoplasias (Figure 1). All of the 27 lesions determined pathologically to be neoplasias were detected by ME-NBI. Sixteen lesions in 16 patients were determined pathologically to be EGC (15 intramucosal lesions and one slightly submucosal lesion, and 15 well-differentiated adenocarcinomas and one moderately-differentiated adenocarcinoma). Eleven lesions in 10 patients were pathologically defined as gastric adenoma. SGN was detected in 2 of the 28 patients (7.1%) with closed-type atrophic gastritis and in 24 of the 154 patients (15.6%) with open-type atrophic gastritis. The number of lesions did not differ significantly between patients with the closed and open types of atrophic gastritis (P = 0.38).
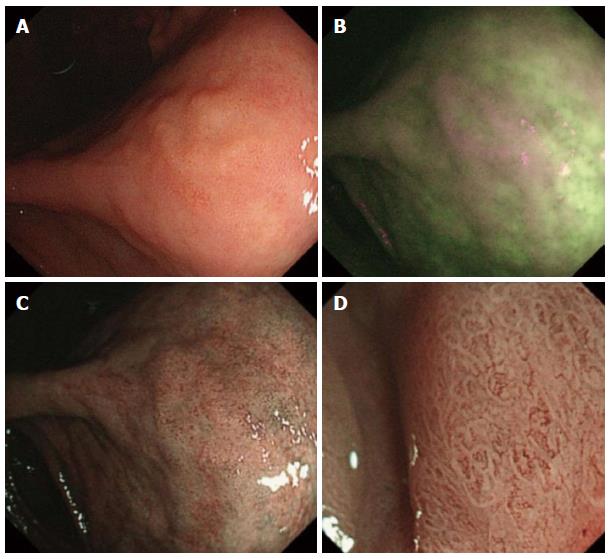
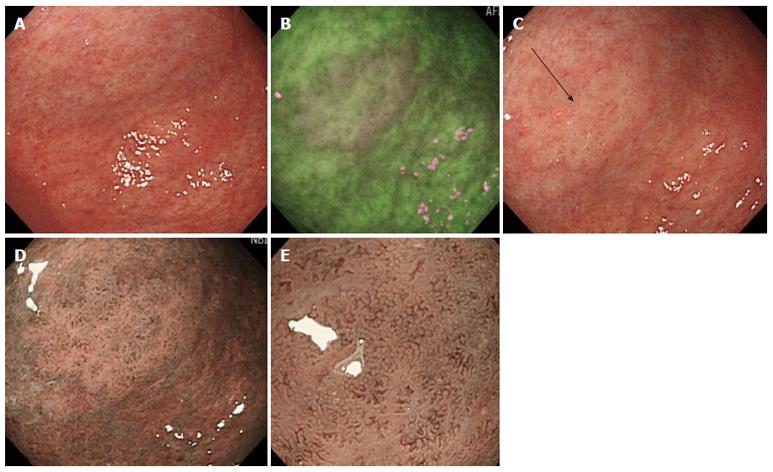
Twenty-two of 27 lesions (81%) were detected by WLI (Figure 2) and five (19%) were missed by WLI but were detected by AFI. All of the five lesions were, however, detected in the second WLI after AFI (Figure 3). There was no significant differences in size, structure, or location between lesions detected by WLI and ones missed by the first WLI but detected by AFI (Table 1). Sixteen of 22 lesions detected by WLI were reddish and 6 were whitish, but all of five lesions missed by the first WLI but detected by AFI were whitish. There was a significant difference in color between the two groups (P = 0.006). Sixteen of 22 lesions detected by WLI were EGCs and 6 were gastric adenomas, but 5 missed by the first WLI but detected by AFI were gastric adenomas that were slightly protruded. There was a significant difference in pathology (carcinoma/ adenoma) between the two groups (P = 0.006).
| Detection mode | WLI | not WLI but AFI | P value |
| n (%) | 22 (81) | 5 (19) | |
| Mean tumor size (mm) | 12.1 (5-60) | 11.6 (5-20) | 0.400 |
| Color (reddish: whitish) | 16:6 | 0:5 | 0.006 |
| Type (protruded:depressed) | 14:8 | 5:0 | 0.280 |
| Location (U:M:L) | 2:9:11 | 0:2:3 | 0.640 |
| Pathology (carcinoma:adenoma) | 16:6 | 0:5 | 0.006 |
Sensitivity in ME-NBI was higher than that in WLI (P = 0.02) and that in AFI (P = 0.02). Specificity in ME-NBI was higher than that in WLI (P < 0.001) and that in AFI (P < 0.001). Accuracy in ME-NBI was higher than that in WLI (P < 0.001) and that in AFI (P < 0.001). In addition, both specificity and accuracy in AFI were lower than those in WLI (P < 0.001) (Table 2).
EGC is a good indication for ER and surveillance EGD is considered to reduce the mortality correlated with gastric neoplasia[1-3]. Moreover, surveillance EGD after ESD for SGN is important to detect metachronous SGNs. Twenty-two lesions (81%) were detected by WLI, 5 lesions (19%) were missed by WLI but were detected by AFI in this study, therefore approximately one fifth could not be correctly diagnosed in WLI mode. Kato et al[18] reported that the detection rate of EGC was more than 10% not only by WLI but also by AFI. In our study, the detection by AFI was also similar to that reported by Kato et al[18]. Our study found AFI videoendoscopy to be effective for identifying gastric neoplasias missed by WLI videoendoscopy. All five lesions missed by the first WLI were able to be detected in the second WLI. All five lesions missed by WLI were, however, gastric adenomas in this study.
Sixteen of the neoplasias detected by WLI were reddish and six were whitish, and all five missed by WLI but detected by AFI were whitish. All five lesions were located in the atrophic mucosa, and they resembled the surrounding relatively whitish atrophic mucosa, because there was little color difference. It thus might be difficult to detect gastric neoplasias that were minimally protruded and/or were small. All five lesions were minimally protruded, and three of 5 were small (7 mm or less). Moreover, two of 5 lesions missed by the first WLI were located in the anterior wall behind the angle, which were necessary to be observed carefully not to be missed.
ME-NBI can reduce large false-positive numbers detected by WLI and/or AFI, it is, therefore, deemed to be the mainstay of TME system in increase of diagnostic accuracy for SGN[20]. Diagnoses based on WLI and AFI depend on the changes in the color and configuration of lesions, that are not always specific for gastric neoplasias. In our data, the specificity in AFI was especially low. ME-NBI, in contrast, has the advantage of yielded clearer images of the microsurface pattern and microvascular pattern, which show abnormalities specific for neoplasia and enables an endoscopically pathologic diagnosis[20]. Therefore, ME-NBI reveals high diagnostic specificity for SGN and makes a correct diagnosis of the false-positive lesion detected by WLI and AFI in the TME system. Recently, the number of patients being administered antithrombotic drugs is increasing, therefore, endoscopic biopsy might increase the risk of post-biopsy hemorrhage in such patients. Histological findings by biopsy specimen might not be sufficient for evaluating the whole region in some gastric neoplasias, while endoscopic findings of the TME system could evaluate the whole region, due to high accuracy of the TME system. The biopsy samples are deemed to be the gold standard for diagnosis, however, they might not always be necessary for some gastric neoplasias.
Ezoe et al[25] reported that sensitivities in WLI and WLI + ME-NBI were respectively 40.0% and 95.0%. In our study the sensitivity in WLI was 81.5%. In the study reported by Ezoe et al[25], all lesions were depressed-type gastric cancers less than 10 mm in diameter, whereas 19 of the 27 lesions in our study protruded and 10 were over 10 mm in diameter. This may be the reason the WLI-mode sensitivity in our study was higher than that reported by Ezoe et al[25]. The specificities in WLI and ME-NBI reported by Ezoe et al[25] were similar to those in our study.
Hanaoka et al[26] reported that the recurrence rate after ESD for EGCs was higher in the open-type atrophic gastritis than in the closed-type one. No significant difference between the two groups was noted in our study, perhaps because our data were analysed at only one point in only about 70% of the patients. Hanaoka et al[26], however, reported results obtained when surveillance EGD was scheduled annually after ESD and eradication of H. pylori infection, and in their study the median follow-up duration was 55 mo.
There are some limitations to our study. It was performed in a single center and the sample size was small. About one-fifth of the neoplasias in this study were found by AFI rather than WLI, all of them were not, however, adenocarcinomas but gastric adenomas. Three gastric adenomas which were missed by WLI but were detected by AFI were histologically diagnosed by only biopsy specimens, because the patients did not hope to undergo ESD. Therefore, those histopathologic findings might be not accurate, because it is sometimes difficult to differentiate gastric cancers from gastric adenomas by biopsy specimens. AFI videoendoscopy has low specificity in detecting gastric neoplasias, and it has large numbers of false positives. TME using ME-NBI as well as WLI and AFI, takes more time than endoscopic observation using only WLI but eliminates the need to take biopsy specimens so false positive lesions can be excluded pathologically. In this study, four well-trained endoscopists performed the TME videoendoscopy. The advanced techniques of ME-NBI are necessary to obtain clearer images of lesions and to clarify the advantages. Tada et al[27] reported that AFI improved sensitivity of endoscopic diagnosis of EGC by less experienced endoscopists. In the near future, we need additional study using TME system by less experienced endoscopists.
In conclusion, surveillance using trimodal imaging endoscopy might be useful for detecting another lesion after endoscopic submucosal dissection for superficial gastric neoplasia.
The endoscope (FQ260Z) was provided by Olympus Medical Systems Corp., Tokyo, Japan.
COMMENTS
Metachronous multiple gastric cancers sometimes develop in patients who underwent endoscopic resection for superficial gastric neoplasia (SGN). Surveillance after endoscopic resection for SGN is very important.
The specificity of autofluorescence imaging is lower than that of white light imaging. Trimodal endoscopy with magnifying endoscopy with narrow band imaging is effective for detecting early-stage gastric cancers.
Surveillance using trimodal imaging endoscopy might be useful for detecting another lesion after endoscopic submucosal dissection for superficial gastric neoplasia.
This is a well done and interesting clinical study for evaluation of the effectiveness of trimodal imaging endoscopy after endoscopic submucosal dissection for SGN.
P- Reviewer: Takeno S, Vorobjova T S- Editor: Gou SX L- Editor: A E- Editor: Liu XM
| 1. | Ohkuwa M, Hosokawa K, Boku N, Ohtu A, Tajiri H, Yoshida S. New endoscopic treatment for intramucosal gastric tumors using an insulated-tip diathermic knife. Endoscopy. 2001;33:221-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 309] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 2. | Gotoda T, Yamamoto H, Soetikno RM. Endoscopic submucosal dissection of early gastric cancer. J Gastroenterol. 2006;41:929-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 507] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 3. | Isomoto H, Shikuwa S, Yamaguchi N, Fukuda E, Ikeda K, Nishiyama H, Ohnita K, Mizuta Y, Shiozawa J, Kohno S. Endoscopic submucosal dissection for early gastric cancer: a large-scale feasibility study. Gut. 2009;58:331-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 470] [Cited by in RCA: 520] [Article Influence: 32.5] [Reference Citation Analysis (1)] |
| 4. | Arima N, Adachi K, Katsube T, Amano K, Ishihara S, Watanabe M, Kinoshita Y. Predictive factors for metachronous recurrence of early gastric cancer after endoscopic treatment. J Clin Gastroenterol. 1999;29:44-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Nasu J, Doi T, Endo H, Nishina T, Hirasaki S, Hyodo I. Characteristics of metachronous multiple early gastric cancers after endoscopic mucosal resection. Endoscopy. 2005;37:990-993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 123] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 6. | Nakajima T, Oda I, Gotoda T, Hamanaka H, Eguchi T, Yokoi C, Saito D. Metachronous gastric cancers after endoscopic resection: how effective is annual endoscopic surveillance? Gastric Cancer. 2006;9:93-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 170] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 7. | Uedo N, Iishi H, Tatsuta M, Ishihara R, Higashino K, Takeuchi Y, Imanaka K, Yamada T, Yamamoto S, Yamamoto S. Longterm outcomes after endoscopic mucosal resection for early gastric cancer. Gastric Cancer. 2006;9:88-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 123] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 8. | Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, Terao S, Amagai K, Hayashi S, Asaka M. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 876] [Cited by in RCA: 935] [Article Influence: 55.0] [Reference Citation Analysis (0)] |
| 9. | Bhunchet E, Hatakawa H, Sakai Y, Shibata T. Fluorescein electronic endoscopy: a novel method for detection of early stage gastric cancer not evident to routine endoscopy. Gastrointest Endosc. 2002;55:562-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Ohkawa A, Miwa H, Namihisa A, Kobayashi O, Nakaniwa N, Ohkusa T, Ogihara T, Sato N. Diagnostic performance of light-induced fluorescence endoscopy for gastric neoplasms. Endoscopy. 2004;36:515-521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 11. | Kobayashi M, Tajiri H, Seike E, Shitaya M, Tounou S, Mine M, Oba K. Detection of early gastric cancer by a real-time autofluorescence imaging system. Cancer Lett. 2001;165:155-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Abe S, Izuishi K, Tajiri H, Kinoshita T, Matsuoka T. Correlation of in vitro autofluorescence endoscopy images with histopathologic findings in stomach cancer. Endoscopy. 2000;32:281-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Imaeda H, Hosoe N, Kashiwagi K, Ida Y, Saito Y, Suzuki H, Aiura K, Ogata H, Kumai K, Hibi T. Autofluorescence videoendoscopy system using the SAFE-3000 for assessing superficial gastric neoplasia. J Gastroenterol Hepatol. 2010;25:706-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 14. | Kara MA, Peters FP, Ten Kate FJ, Van Deventer SJ, Fockens P, Bergman JJ. Endoscopic video autofluorescence imaging may improve the detection of early neoplasia in patients with Barrett’s esophagus. Gastrointest Endosc. 2005;61:679-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 158] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 15. | Borovicka J, Fischer J, Neuweiler J, Netzer P, Gschossmann J, Ehmann T, Bauerfeind P, Dorta G, Zürcher U, Binek J. Autofluorescence endoscopy in surveillance of Barrett’s esophagus: a multicenter randomized trial on diagnostic efficacy. Endoscopy. 2006;38:867-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 16. | Uedo N, Iishi H, Tatsuta M, Yamada T, Ogiyama H, Imanaka K, Sugimoto N, Higashino K, Ishihara R, Narahara H. A novel videoendoscopy system by using autofluorescence and reflectance imaging for diagnosis of esophagogastric cancers. Gastrointest Endosc. 2005;62:521-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 85] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 17. | Ishihara R, Inoue T, Hanaoka N, Takeuchi Y, Tsujii Y, Kanzaki H, Oota T, Hanafusa M, Yamamoto S, Nagai K. Autofluorescence imaging endoscopy for screening of esophageal squamous mucosal high-grade neoplasia: a phase II study. J Gastroenterol Hepatol. 2012;27:86-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 18. | Kato M, Kaise M, Yonezawa J, Yoshida Y, Tajiri H. Autofluorescence endoscopy versus conventional white light endoscopy for the detection of superficial gastric neoplasia: a prospective comparative study. Endoscopy. 2007;39:937-941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Matsuda T, Saito Y, Fu KI, Uraoka T, Kobayashi N, Nakajima T, Ikehara H, Mashimo Y, Shimoda T, Murakami Y. Does autofluorescence imaging videoendoscopy system improve the colonoscopic polyp detection rate?--a pilot study. Am J Gastroenterol. 2008;103:1926-1932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 86] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 20. | Kato M, Kaise M, Yonezawa J, Goda K, Toyoizumi H, Yoshimura N, Yoshida Y, Kawamura M, Tajiri H. Trimodal imaging endoscopy may improve diagnostic accuracy of early gastric neoplasia: a feasibility study. Gastrointest Endosc. 2009;70:899-906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | Curvers WL, Alvarez Herrero L, Wallace MB, Wong Kee Song LM, Ragunath K, Wolfsen HC, Prasad GA, Wang KK, Subramanian V, Weusten BL. Endoscopic tri-modal imaging is more effective than standard endoscopy in identifying early-stage neoplasia in Barrett’s esophagus. Gastroenterology. 2010;139:1106-1114. [PubMed] |
| 22. | Kuiper T, van den Broek FJ, Naber AH, van Soest EJ, Scholten P, Mallant-Hent RCh, van den Brande J, Jansen JM, van Oijen AH, Marsman WA. Endoscopic trimodal imaging detects colonic neoplasia as well as standard video endoscopy. Gastroenterology. 2011;140:1887-1894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 23. | Kimura K, Takemoto T. An endoscopic recognition of the atrophy border and its significance in chronic gastritis. Endoscopy. 1969;1:87-97. |
| 24. | The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003;58:S3-S43. [PubMed] |
| 25. | Ezoe Y, Muto M, Uedo N, Doyama H, Yao K, Oda I, Kaneko K, Kawahara Y, Yokoi C, Sugiura Y. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology. 2011;141:2017-2025.e3. [PubMed] |
| 26. | Hanaoka N, Uedo N, Shiotani A, Inoue T, Takeuchi Y, Higashino K, Ishihara R, Iishi H, Haruma K, Tatsuta M. Autofluorescence imaging for predicting development of metachronous gastric cancer after Helicobacter pylori eradication. J Gastroenterol Hepatol. 2010;25:1844-1849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 27. | Tada K, Oda I, Yokoi C, Taniguchi T, Sakamoto T, Suzuki H, Nonaka S, Yoshinaga S, Saito Y, Gotoda T. Pilot study on clinical effectiveness of autofluorescence imaging for early gastric cancer diagnosis by less experienced endoscopists. Diagn Ther Endosc. 2011;2011:419136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |