Published online Nov 7, 2014. doi: 10.3748/wjg.v20.i41.15454
Revised: April 8, 2014
Accepted: May 25, 2014
Published online: November 7, 2014
Processing time: 271 Days and 1.3 Hours
Gangliocytic paraganglioma (GP) is rare and has been regarded as benign in general with a good prognosis. We present a patient with duodenal GP showing a malignant and lethal clinical course. A 47-year-old male patient was found to have a duodenal tumor and enlarged regional lymph nodes. The patient initially underwent a pancreaticoduodenectomy to resect the tumor and involved lymph nodes completely. Histological and immunohistochemical analyses showed findings typical of GP. However, the distant metastatic lesions in the liver and pelvic cavity were rapidly observed after surgery. The patient underwent chemotherapy and radiotherapy, as well as a second surgery to partly remove the metastatic mass in the pelvic cavity. The histological examination revealed no significant difference in histological features between the primary duodenal tumor and the metastatic pelvic mass. However, the patient finally died of the tumor due to the recurrence of the residual pelvic lesion and increased liver mass. To our knowledge, this is the first report of lethal GP with multifocal metastases. Our case confirms that GP should be regarded as a malignant potential tumor with behavior code of “1”, rather than a benign tumor of “0”.
Core tip: This case for the first time presents a patient with duodenalgangliocytic paraganglioma showing distant metastases and a lethal clinical course. It indicates that gangliocytic paraganglioma should be regarded as a malignant potential tumor, rather than a benign tumor.
- Citation: Li B, Li Y, Tian XY, Luo BN, Li Z. Malignant gangliocytic paraganglioma of the duodenum with distant metastases and a lethal course. World J Gastroenterol 2014; 20(41): 15454-15461
- URL: https://www.wjgnet.com/1007-9327/full/v20/i41/15454.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i41.15454
Gangliocytic paraganglioma (GP) is a rare tumor, which occurs nearly exclusively in the second portion of the duodenum and is characterized by its triphasic cellular differentiation, consisting of epithelioid neuroendocrine cells, spindle-shaped cells with Schwannian cell differentiation, and ganglion-like cells[1]. Since this tumor was first described by Dahl et al[2], there have been no more than 200 cases reported in the literature in the world to date. According to the World Health Organization (WHO) classification, this lesion has been regarded as benign, but a few cases with regional lymph node metastasis and even distant metastasis have been reported. To the best of our knowledge, so far only 18 cases of GP with regional lymph node metastases[3-20] and two with bone or liver metastasis[21,22] have been described. Surprisingly, almost all patients with GP, including those having lymph node or distant metastasis, gain a good outcome without recurrence. Moreover, there is no record of a patient dying of GP although only one patient received irradiation intervention because of aggressive behavior of the tumor[12]. Herein, we report a unique case of this clinical entity in a middle-aged male patient. In contrast to previous cases, our case presents prominent aggressive biological behavior with regional lymph node, liver and pelvic cavity metastases. The patient died of GP finally after radiotherapy and chemotherapy. This is the first presentation of a malignant clinical course and poor prognosis of GP. The literature on this rare tumor is reviewed and its clinicopathological characteristics are discussed.
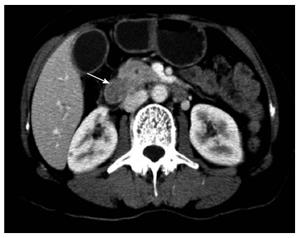
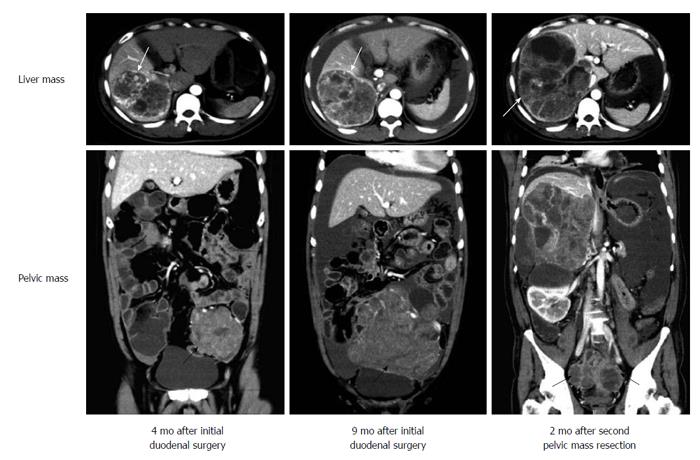
A 47-year-old male patient presented with complaints of mild upper abdominal pain for 3 mo. During this period, he was referred to a local hospital for radiological examination. Computed tomographic (CT) scans revealed a mass in the duodenum, near the duodenal papilla. A gastrointestinal endoscopy was performed in the local hospital, but biopsy examination was negative because only inflammatory mucosa was observed under the microscopy. As a result, the patient was referred to our hospital for examination and treatment. Physical examination results were normal. The laboratory results, including blood count and liver and renal function, were within the normal range. The CT images acquired at the local hospital showed a 3.0 cm duodenal mass in the medial wall of the subpapillary duodenum without signs of pancreatic invasion (Figure 1). An enlarged lymph node of the pancreatic posterior group, measuring 1.0 cm, was also found. The lesion was preoperatively diagnosed as a gastrointestinal stromal tumor (GIST) of the duodenum with lymph node metastasis. The patient underwent a pancreaticoduodenectomy since the lymph node could be metastatic, and the mass of the duodenum was gross totally resected. Postoperative recovery was uneventful without surgical complications. After diagnosis, the patient received no radiotherapy/chemotherapy and was only on a regular follow-up. Follow-up CT scans at 4 mo showed that two masses grew in the liver and pelvic cavity, respectively. Considering that distant metastases could occur, the patient decided to receive radiotherapy and chemotherapy. The patient was treated with intensity-modulated radiotherapy over 30 elapsed days, and the total dose was 5040 cGy. However, the tumors did not show remarkable radiosensitivity. Therefore, systemic chemotherapy with the combination of cyclophosphamide, vincristine and dacarbazine was also performed. Regretfully, the masses did not regress. After chemotherapy, that was nine months after initial surgical resection of duodenal mass, CT scans revealed that the mass in the pelvic cavity was larger and the surrounding organs were pressed. A second laparotomy was performed and the majority of the pelvic mass was resected. However, two months later, CT scans showed that a mass re-grew at the site of original tumor location in the pelvic cavity and the liver mass became larger, which lost opportunities for surgical treatment (Figure 2). The patient developed persistent ascites and fever. Finally, he died 13 mo after the initial surgery.
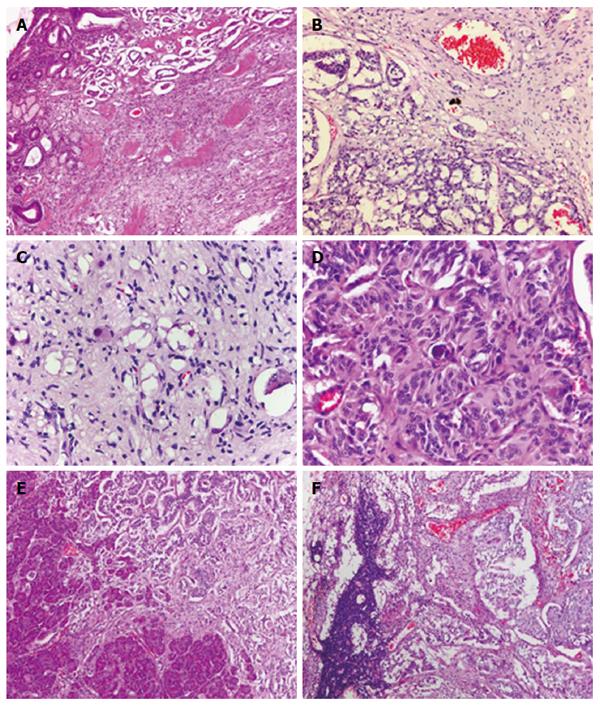
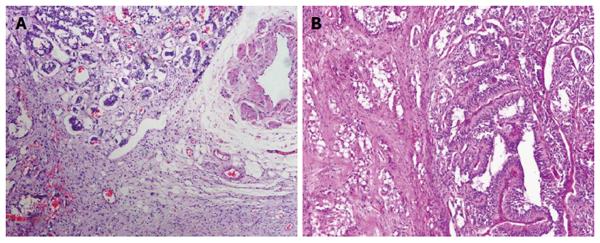
On microscopic examination, the duodenal mass was located in the submucosa with extension to the muscularis propria. The mass was nonencapsulated and had an infiltrative margin, and the pancreas was observed to be involved. The tumor was composed of three morphologically distinct cell populations: epithelioid cells, spindle cells, and scattered ganglion-like cells. The epithelioid cells, arranged in nests and gland-like structures, had granular eosinophilic cytoplasm and round to oval-shaped nucleus with an inconspicuous nucleolus. They were cytologically bland with minimal to mild nuclear pleomorphism. The spindle cells formed slender fascicles wrapping around nests of epithelioid cells. These spindle-shaped cells had an elongated and plump nucleus, including an attenuated eosinophilic cytoplasm without marked atypia. Tumor cells of a ganglion-like cell type were rarely seen and had a round nucleus with conspicuous nucleolus. There was no mitotic figure or necrosis found in the mass. The metastatic tumor was found in seven of sixteen lymph nodes, and the metastatic foci showed the presence of the three cellular components identified in the primary tumor (Figure 3). Like the duodenal mass, the mass in the pelvic cavity also exhibited similar histological appearance with three identical cellular components. The tumor appeared to have an infiltrative margin and mild to moderate atypia. No necrosis or hemorrhage was found in the tumor (Figure 4).
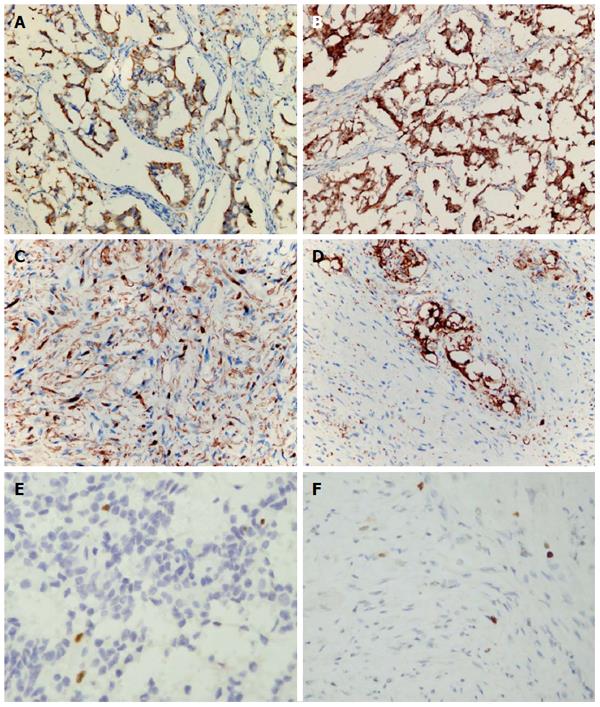
By immunohistochemical staining, the epithelioid cells were positive for cytokeratin (AE1/AE3), neuron-specific enolase (NSE), synaptophysin (Syn), Chromogranin A (CgA) and CD56. The ganglion-like cells were immunoreactive for Syn and NSE. In contrast, the spindle shaped cell type showed positive reactivity for S-100 protein, but not for Syn or CgA. Neither cell type was positive for CD117, CD34, Dog-1, SMA, desmin, Myo D1 or myogenenin. MIB-1 (Ki-67) labeling index estimated less than 1% in both primary and metastatic foci (Figure 5). In order to inspect the lymphatic and vascular invasion of tumor cells, D2-40 and CD31 were used to highlight the lymphatic vessels and blood vessels, respectively. However, there was no certain vascular invasion identified in tumor tissues.
Based on the pathological findings, the tumor was diagnosed as a malignant duodenal GP with regional lymph node and distant metastases.
GP has been known as a rare and benign neuroendocrine tumor. It was first described as “duodenal ganglioneuroma” in 1957 by Dahl et al[2]. In that case, only a very small marginal area of epithelioid cells was found, where the greatest portion of the lesion was a ganglioneuroma and the authors classified it as such[2]. In 1962, Taylor et al[23] described a group of unusual polypoid tumors in the duodenum which they called “benign nonchromaffin paragangliomas”, mostly because they found the nests of epithelioid cells to be indistinguishable from the “Zellballen” of carotid body tumors and other chemoreceptor structures. In 1971, Kepes and Zacharias[24] suggested the term of “gangliocytic paraganglioma” for those duodenal lesions, because these tumors had light and electron microscopic features seen in paragangliomas as well as ganglioneuromas, indicating a transitional or hybrid form of tumor between gangliocytomas and nonchromaffin paragangliomas. In 1989, WHO accepted GP as a new entity of benign tumors of “neurogenic tumors” because of its distinct morphological and clinical characteristics[25]. However, the 3rd edition[26] and the latest edition (4th edition)[1] of the WHO classification revised GP to “endocrine tumor” and “neurodendocrine neoplasm”, respectively, which further emphasized neuroendocrine differentiation of the tumor.
GPs have been regarded as benign in general, but a few cases showing lymph node metastasis are well known which required extensive surgical removal and adjuvant radiotherapy[7,12,13]. However, the penetrative growth pattern of the tumor and even lymph node metastasis are not indicative of aggressive behavior or malignancy. Unlike previously reported cases, our case was characterized clinically by multifocal distant metastases and a rapidly aggressive clinical course. The patient died of the disease after surgical resection, chemotherapy and radiotherapy. To our knowledge, this is the first case of lethal GP with a malignant clinical course, but without obvious histological atypia of all three elements of the tumor, and high mitotic figures or proliferative index. We have compared the histological difference between the primary tumor of the duodenum and the metastatic pelvic mass in the present case, and tried to establish the histological prognostic indicators of malignant GP. However, the cytological bland spindle cells along with mild atypia of epithelioid cells were observed in both masses. No necrosis or high mitotic activity was found in tumors, which was consistent with all of reported GPs, even those with lymph node or distant metastasis. Our findings indicate that histological evaluation has limited prognostic value in GPs. We postulated that some important factors are involved in the process of GP invasion and metastasis, which cannot bring obvious morphological changes but indeed induce aggressive biological behavior, and result in the discrepancy between malignant clinical course and pathological findings. Further molecular or cytogenetic analysis is needed to elucidate the related mechanisms.
Although most of GPs exhibited good outcomes, our case suggested that it should not be considered as a benign tumor. Because of its potentially metastatic behavior, we prefer to label GP as a tumor with “uncertain malignant potential”, at least for those with evidence of lymph node and/or distant metastasis. The behavior code of the tumor is probably best to be regarded as “1 (unspecified, borderline or uncertain behavior)”, rather than as “0 (benign)”. We focused on the significant factors associated with the malignant behavior of the tumor in previously reported cases. To date, there have been no more than 200 cases of GP reported in the literature in the world, but only 20 bona fide cases reported as GP with regional lymph node metastasis or distant metastasis (Table 1). The primary tumors of GP were usually large (> 2 cm in diameter), relatively well-circumscribed, had an infiltrative growth margin and frequently extend into the muscularis propria. However, almost all patients showed good outcomes without recurrence. Indeed, only one patient required additional surgical intervention due to a residue of the tumor at her first endoscopic procedure[13], one patient has been reported as showing a recurrence due to a residue of a previous tumor at his initial surgical intervention[7], one patient received radiotherapy after surgical intervention[12], and two patients had distant liver and bone metastasis, respectively[21,22]. GP patients with metastasis have extremely good prognosis with long survival periods (6 mo to 11 years), and there was no record of a patient dying of GP to date except for our presenting case. Furthermore, despite of metastatic locations, the neoplastic elements of GP in metastatic foci were not observed to influence the prognosis of the patients, although epithelioid component sometimes exhibited as the sole element in metastatic foci[3,5,6]. Some researchers tried to establish the immunohistochemical prognostic indicators of GP using bcl-2, p53, and Ki-67, which were acceptable prognostic indicators in several kinds of neuroendocrine tumors. However, bcl-2 and p53 showed negative reactivity in all of reported cases[15,27], and Ki-67 labeling index value was extremely low either in primary tumors or in metastatic tumors. Therefore, the immunohistochemical evaluation using these markers may have limited prognostic value in GPs at present.
| No. | Ref. | Age (yr)/gender | Tumor site | Tumor size (cm) | Regional lymph node metastasis | Distant metastasis | Treatment | Outcome |
| 1 | Büchler et al[3] (1985) | 50/male | Ampulla of Vater | 3.0 | Yes | No | LR | NED 20 mo |
| 2 | Korbi et al[4] (1987) | 73/female | Duodenum | NA | Yes | No | WP | NA |
| 3 | Inai et al[5] (1989) | 17/male | Duodenum | 2.0 | Yes | No | WP | NED 32 mo |
| 4 | Hashimoto et al[6] (1992) | 47/male | Second portion of the duodenum | 6.5 | Yes | No | WP | NED 14 mo |
| 5 | Dookhan et al[7] (1993) | 41/male | Duodenum | 2.5 | Yes | No | LR + WP | Recurrence 11 years after first local resection |
| 6 | Takabayashi et al[8] (1993) | 63/female | Papilla of Vater | 3.2 | Yes | No | WP | NED 24 mo |
| 7 | Tomic et al[9] (1996) | 74/female | Pancreas | 4.0 | Yes | No | WP | NED 19 mo |
| 8 | Henry et al[21] (2003) | 50/male | Pancreas | 2.5 | Yes | Yes (bone) | WP | NA |
| 9 | Sundararajan et al[10] (2003) | 67/female | Second portion of the duodenum | 5.0 | Yes | No | WP | NED 9 mo |
| 10 | Bucher et al[11] (2004) | 31/female | Papilla of Vater | 3.0 | Yes | No | WP | NED 44 mo |
| 11 | Wong et al[12] (2005) | 49/female | Duodenum | 1.4 | Yes | No | WP + RT | NED 12 mo |
| 12 | Witkiewicz et al[13] (2007) | 38/female | Papilla of Vater | 1.5 | Yes | No | LR + WP | NA |
| 13 | Mann et al[14] (2009) | 17/female | Duodenum | NA | Yes | No | WP | NED 12 mo |
| 14 | Okubo et al[15] (2010) | 61/male | Papilla of Vater | 3.0 | Yes | No | WP | NED 6 mo |
| 15 | Saito et al[16] (2010) | 28/male | Papilla of Vater | NA | Yes | No | WP | NA |
| 16 | Sandmann et al[17] (2010) | 62/female | Ampulla of Vater | 5.0 | Yes | No | LR | NA |
| 17 | Uchida et al[18] (2010) | 67/female | Second portion of the duodenum | NA | Yes | No | WP | NA |
| 18 | Rowsell et al[22] (2011) | 52/female | Duodenum | 1.0 | Yes | Yes (liver) | LR | NED 27 mo |
| 19 | Ogata et al[19] (2011) | 16/male | Ampulla of Vater | 2.5 | Yes | No | WP | NED 36 mo |
| 20 | Barret et al[20] (2012) | 51/female | Duodenal papilla | 3.5 | Yes | No | WP | NED 8 yr |
| 21 | Present case | 47/male | Duodenal papilla | 3.0 | Yes | Yes (liver and pelvic cavity) | WP + RT + CT + pelvic mass resection | Die 13 moafter initial surgery |
Patients with GP usually have favorable prognosis when they receive total resection, although a residual mass can recur after a long time[7]. At present, no chemotherapy was advised due to the rarity of distant metastases and the lack of response of this tumor to conventional systemic therapy. The present case was also not responsive to conventional chemotherapeutic regimens, such as cyclophosphamide and vincristine, which were associated with response rates of 50%-55% for malignant paraganglioma. A few cases had been described to apply radiotherapy for the lesions that had regional lymph node metastasis, but the efficacy of treatment was difficult to ascertain[12]. Recent reports suggested that radiotherapy might be effective in the management of locally advanced malignant paraganglioma when used in conjunction with radionuclide therapy (131I-MIBG), but they have not been used in GP treatment yet. Some authors suggested that the presence of lymph node and liver metastases should not result in over-treatment as such behavior was not necessarily fraught with a bad clinical outcome[22]. So far there have been no acceptable prognostic factors for malignant GPs. But complete resection of the tumor combined with adjuvant chemo- or radiotherapy is suggested to apply for GP patients with lymph node and/or distant metastasis to avoid the potentially rapid progression of the disease. Of course, more effective chemotherapeutic regimens and long-term careful follow-up are necessary for these patients.
In conclusion, we report a rare case of duodenal GP with regional lymph node and distant metastases occurring in an adult patient. In contrast to previously reported GPs with metastasis, our presenting case exhibits marked malignancy with a rapid aggressive clinical course. The patient finally died of GP after surgery, chemotherapy and radiation intervention, which has not been previously described. To our knowledge, this is the first case of lethal GP with multifocal metastases. Despite the lack of acceptable prognostic indicators for GPs at present, especially for those with regional lymph node and/or distant metastasis, our case indicates that GP is indeed a tumor with uncertain malignant potential. We suggest adjuvant therapies for the patients with metastasis after complete excision of the tumor, although the consensus on the adjuvant chemotherapy and/or radiotherapy for this tumor has not been reached yet.
A 47-years-old male patient with duodenal gangliocytic paraganglioma (GP) showed distant metastases and a lethal clinical course.
A mass was observed in the duodenum, near the duodenal papilla.
Gastrointestinal stromal tumors (GISTs) and poorly differentiated carcinoma of Vater.
The laboratory results, including blood count and liver and renal function, were within the normal range.
Computed tomography (CT) scans revealed a mass in the duodenum, near the duodenal papilla.
The tumor was composed of three morphologically distinct cell populations: epithelioid cells, spindle cells, and scattered ganglion-like cells.
The duodenal tumor was totally resected and the metastatic lesions of liver and pelvic cavity were treated with adjuvant radiotherapy and chemotherapy.
GP has been regarded as benign, but a few cases with regional lymph node metastasis and even distant metastasis have been reported. However, there has been no record of a patient dying of GP to date except for our presenting case.
The biological behavior of GP is unpredictable, and long-term careful follow-up is necessary for these patients.
GP should be regarded as a malignant potential tumor with behavior code of “1”, rather than a benign tumor of “0”.
P- Reviewer: Daphan CE, Nishida T, Wu DC S- Editor: Nan J L- Editor: Wang TQ E- Editor: Liu XM
| 1. | Kloppel G, Arnold R, Capella C, Klimstra DS, Albores-Saavedra J, Solcia E, Rindi G, Komminoth R. Neruoendocrine neoplasms of the ampullary region. WHO classification of the tumors of the digestive system. Lyon: IARC press 2010; 92-94. |
| 2. | Dahl EV, Waugh JM, Dahlin DC. Gastrointestinal ganglioneuromas; brief review with report of a duodenal ganglioneuroma. Am J Pathol. 1957;33:953-965. [PubMed] |
| 3. | Büchler M, Malfertheiner P, Baczako K, Krautzberger W, Beger HG. A metastatic endocrine-neurogenic tumor of the ampulla of Vater with multiple endocrine immunoreaction--malignant paraganglioma? Digestion. 1985;31:54-59. [PubMed] |
| 4. | Korbi S, Kapanci Y, Widgren S. [Malignant paraganglioma of the duodenum. Immunohistochemical and ultrastructural study of a case]. Ann Pathol. 1987;7:47-55. [PubMed] |
| 5. | Inai K, Kobuke T, Yonehara S, Tokuoka S. Duodenal gangliocytic paraganglioma with lymph node metastasis in a 17-year-old boy. Cancer. 1989;63:2540-2545. [PubMed] |
| 6. | Hashimoto S, Kawasaki S, Matsuzawa K, Harada H, Makuuchi M. Gangliocytic paraganglioma of the papilla of Vater with regional lymph node metastasis. Am J Gastroenterol. 1992;87:1216-1218. [PubMed] |
| 7. | Dookhan DB, Miettinen M, Finkel G, Gibas Z. Recurrent duodenal gangliocytic paraganglioma with lymph node metastases. Histopathology. 1993;22:399-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Takabayashi N, Kimura T, Yoshida M, Sakuramachi S, Harada Y, Kino I. A case report of duodenal gangliocytic paraganglioma with lymph node metastasis. Jpn Soc Gastroenterol Surg. 1993;26:2444-2448. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 9. | Tomic S, Warner T. Pancreatic somatostatin-secreting gangliocytic paraganglioma with lymph node metastases. Am J Gastroenterol. 1996;91:607-608. [PubMed] |
| 10. | Sundararajan V, Robinson-Smith TM, Lowy AM. Duodenal gangliocytic paraganglioma with lymph node metastasis: a case report and review of the literature. Arch Pathol Lab Med. 2003;127:e139-e141. [PubMed] |
| 11. | Bucher P, Mathe Z, Bühler L, Chilcott M, Gervaz P, Egger JF, Morel P. Paraganglioma of the ampulla of Vater: a potentially malignant neoplasm. Scand J Gastroenterol. 2004;39:291-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 12. | Wong A, Miller AR, Metter J, Thomas CR. Locally advanced duodenal gangliocytic paraganglioma treated with adjuvant radiation therapy: case report and review of the literature. World J Surg Oncol. 2005;3:15. [PubMed] |
| 13. | Witkiewicz A, Galler A, Yeo CJ, Gross SD. Gangliocytic paraganglioma: case report and review of the literature. J Gastrointest Surg. 2007;11:1351-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 14. | Mann CM, Bramhall SR, Buckels JA, Taniere P. An unusual case of duodenal obstruction-gangliocytic paraganglioma. J Hepatobiliary Pancreat Surg. 2009;16:562-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Okubo Y, Yokose T, Tuchiya M, Mituda A, Wakayama M, Hasegawa C, Sasai D, Nemoto T, Shibuya K. Duodenal gangliocytic paraganglioma showing lymph node metastasis: a rare case report. Diagn Pathol. 2010;5:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Saito J, Hirata N, Furuzono M, Nakaji S, Inase M, Nagano H, Iwata M, Tochitani S, Fukatsu K, Fujii H. A case of duodenal gangliocytic paraganglioma with lymph node metastasis. Nihon Shokakibyo Gakkai Zasshi. 2010;107:639-648. [PubMed] |
| 17. | Sandmann M, Fähndrich M, Lorenzen J, Heike M. [Gangliocytic paraganglioma--a rare cause of an upper gastrointestinal bleeding]. Z Gastroenterol. 2010;48:1297-1300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 18. | Uchida D, Ogawa T, Ueki T, Kominami Y, Numata N, Matsusita H, Morimoto Y, Nakarai A, Ota S, Nanba S. [A case of gangliocytic paraganglioma with lymphoid metastasis]. Nihon Shokakibyo Gakkai Zasshi. 2010;107:1456-1465. [PubMed] |
| 19. | Ogata S, Horio T, Sugiura Y, Aiko S, Aida S. Duodenal gangliocytic paraganglioma with regional lymph node metastasis and a glandular component. Pathol Int. 2011;61:104-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Barret M, Rahmi G, Duong van Huyen JP, Landi B, Cellier C, Berger A. Duodenal gangliocytic paraganglioma with lymph node metastasis and an 8-year follow-up: a case report. Eur J Gastroenterol Hepatol. 2012;24:90-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Henry C, Ghalel-Méchaoui H, Bottero N, Pradier T, Moindrot H. [Gangliocytic paraganglioma of the pancreas with bone metastasis]. Ann Chir. 2003;128:336-338. [PubMed] |
| 22. | Rowsell C, Coburn N, Chetty R. Gangliocytic paraganglioma: a rare case with metastases of all 3 elements to liver and lymph nodes. Ann Diagn Pathol. 2011;15:467-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 23. | Taylor HB, Helwig EB. Benign nonchromaffin paragangliomas of the duodenum. Virchows Arch Pathol Anat Physiol Klin Med. 1962;335:356-366. [PubMed] |
| 24. | Kepes JJ, Zacharias DL. Gangliocytic paragangliomas of the duodenum. A report of two cases with light and electron microscopic examination. Cancer. 1971;27:61-67. [PubMed] |
| 25. | Jass JR, Sobin LH. Histological typing of intestinal tumors. Berlin: Springer-Verlag 1989; 31. |
| 26. | Capella C, Solcia E, Sobin LH, Arnold R. Endocrine tumors of the small intestine. Pathology and Genetics of tumor of the digestive system. Lyon: IARC Press 2000; 78-79. |
| 27. | Okubo Y, Wakayama M, Nemoto T, Kitahara K, Nakayama H, Shibuya K, Yokose T, Yamada M, Shimodaira K, Sasai D. Literature survey on epidemiology and pathology of gangliocytic paraganglioma. BMC Cancer. 2011;11:187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |