Published online Oct 28, 2014. doi: 10.3748/wjg.v20.i40.14831
Revised: March 16, 2014
Accepted: June 14, 2014
Published online: October 28, 2014
Processing time: 272 Days and 5.1 Hours
Abdominal tuberculosis is an increasingly common disease that poses diagnostic challenge, as the nonspecific features of the disease which may lead to diagnostic delays and development of complications. This condition is regarded as a great mimicker of other abdominal pathology. A high index of suspicion is an important factor in early diagnosis. Abdominal involvement may occur in the gastrointestinal tract, peritoneum, lymphnodes or solid viscera. Various investigative methods have been used to aid in the diagnosis of abdominal tuberculosis. Early diagnosis and initiation of antituberculous therapy and surgical treatment are essential to prevent morbidity and mortality. Most of the patients respond very well to standard antitubercular therapy and surgery is required only in a minority of cases. Imaging plays an important role in diagnosis of abdominal tuberculosis because early recognition of this condition is important. We reviewed our experience with the findings on various imaging modalities for diagnosis of this potentially treatable disease.
Core tip: Tuberculosis has become a resurgent global problem with increasing numbers of immunocompromised patients, largely related to the global acquired immunodeficiency syndrome pandemic. The spread of the disease is further aided by poverty, overcrowding, and drug resistance. Abdominal tuberculosis rates are rising, consistent with the overall trend. Nonspecific features of the abdominal tuberculosis result in difficulty in establishing a diagnosis. After a diagnosis has been established, prompt initiation of treatment helps prevent morbidity and mortality as it is a treatable disease. This article should alert the clinician to consider abdominal tuberculosis in the correct clinical setting to ensure timely diagnosis and enable appropriate treatment.
- Citation: Debi U, Ravisankar V, Prasad KK, Sinha SK, Sharma AK. Abdominal tuberculosis of the gastrointestinal tract: Revisited. World J Gastroenterol 2014; 20(40): 14831-14840
- URL: https://www.wjgnet.com/1007-9327/full/v20/i40/14831.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i40.14831
Tuberculosis (TB) is a life threatening disease which can virtually affect any organ system[1]. Global burden of tuberculosis is nearly 12 million. According to World Health Organization report 2013, there were an estimated 8.6 million annual incidence of TB globally and 1.3 million people died from disease in 2012[2]. India has the world’s largest tuberculosis cases which is around 26% of the world TB cases, followed by China and South Africa. There were an estimated 0.45 million new cases of multi-drug resistant TB worldwide in 2012. More than half of these cases were in India, China and the Russian Federation. In addition, there is increase in the incidence of TB in developed countries due to increasing prevalence of immunocompromised individuals mainly due to acquired immunodeficiency syndrome (AIDS) pandemic, immigrant’s population, deteriorating social conditions and cutbacks in public health services[3-5]. The prevalence of TB has increased in both immunocompetent and immunocompromised and it can affect virtually any organ. The primary site of TB is usually lung, from which it can get disseminated into other parts of the body. The other routes of spread can be contiguous involvement from adjacent tuberculous lymphadenopathy or primary involvement of extrapulmonary organ. The diagnosis of extrapulmonary TB can be difficult as it presents with nonspecific clinical and radiological features and requires high degree of suspicion for diagnosis. The abdominal TB, which is not so commonly seen as pulmonary TB, can be a source of significant morbidity and mortality and is usually diagnosed late due to its nonspecific clinical presentation[6]. Approximately 15%-25% of cases with abdominal TB have concomitant pulmonary TB[7,8]. Hence, it is quite important in identifying these lesions with high index of suspicion especially in endemic areas. The abdominal TB usually occurs in four forms: tuberculous lymphadenopathy, peritoneal tuberculosis, gastrointestinal (GI) tuberculosis and visceral tuberculosis involving the solid organs. Usually a combination of these findings occurs in any individual patient. Generally, computed tomography (CT) appears to be the imaging modality of choice in the detection and assessment of abdominal TB, other than gastrointestinal TB. Barium studies remain superior for demonstrating intestinal mucosal lesions[9]. In this review we emphasize on the various radiological features of the gastrointestinal TB with a mention on the other types of abdominal TB as well.
There are several ways by which tuberculosis can involve abdomen (Table 1)[10,11]. Firstly, the tubercle bacilli may enter the intestinal tract through the ingestion of infected milk or sputum. The mucosal layer of the GI tract can be infected with the bacilli with formation of epithelioid tubercles in the lymphoid tissue of the submucosa. After 2-4 wk, caseous necrosis of the tubercles leads to ulceration of the overlying mucosa which can later spread into the deeper layers and into the adjacent lymphnodes and into peritoneum. Rarely, these bacilli can enter into the portal circulation or into hepatic artery to involve solid organs like liver, pancreas and spleen[8]. The second pathway is hematogenous spread from tubercular focus from elsewhere in the body to abdominal solid organs, kidneys, lymphnodes and peritoneum. The third pathway includes direct spread to the peritoneum from infected adjacent foci, including the fallopian tubes or adnexa, or psoas abscess, secondary to tuberculous spondylitis. Lastly it can spread through lymphatic channels from infected nodes.
| By ingestion |
| Infected food or milk - Primary intestinal tuberculosis |
| Infected sputum - Secondary intestinal tuberculosis |
| Hematogenous spread from distant tubercular focus |
| Contagious spread from infected adjacent foci |
| Through lymphatic channel |
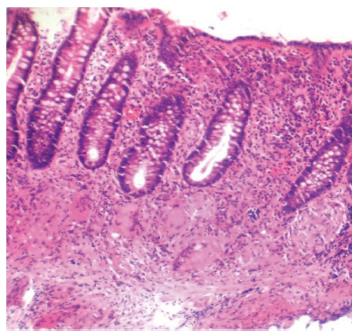
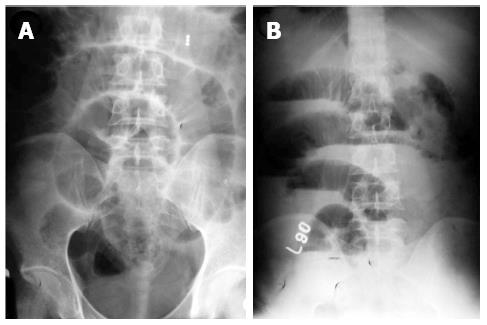
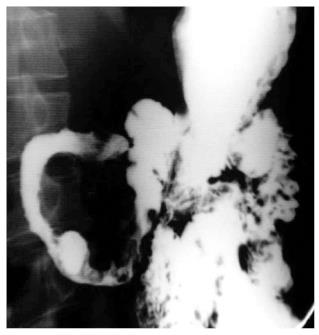
Although neither clinical signs, laboratory, endoscopic findings and radiological signs nor the bacteriological and histopathological findings are gold standard for the diagnosis of abdominal TB, the diagnosis of abdominal TB is usually made by adequate radiological and histopathological studies. The methods of biopsy include endoscopic GI mucosal biopsy, image-guided percutanous biopsy, endoscopic ultrasound guided biopsy, and surgical (open or laparoscopic) biopsy. The caseation necrosis in granulomas is the histologic hallmark of TB. In intestinal tuberculosis the granulomas are multiple, larger (more than 200 μm) and coalescent in mucosa and submucosa (Figure 1). Hematologic findings are nonspecific and include raised erythrocyte sedimentation rate, anemia and hypoalbuminemia. The tubercular ascitic fluid has protein more than 3 g/dL, with a total cell count of 150-4000/μL and consists predominantly of lymphocytes[12]. The ascitic fluid to blood glucose ratio is less than 0.96 and serum ascitic albumin gradient is less than 1.1 g/dL[13]. The yield of organisms on smear and culture is low. Staining for acid fast bacilli is positive in less than 3% of cases and a positive culture is seen in only 20% of cases. Ascitic fluid adenosine deaminase (ADA) levels are elevated in tubercular ascites. Serum ADA level above 54 U/L, ascitic fluid ADA level above 36 U/L and an ascitic fluid to serum ADA ratio more than 0.98 are suggestive of tuberculosis[14]. However, in cases of co-infection with HIV, ascitic ADA levels can be normal or low. Also, falsely high values can be seen in malignant ascites. Interferon-γ levels are also elevated in tubercular ascites[15]. The sensitivity and specificity increased by combining ascitic fluid ADA and interferon-γ assay. The various radiological studies are used for the diagnosis of abdominal TB include ultrasonography (USG), CT, barium studies and magnetic resonance imaging (MRI). Ultrasound is an initial modality of choice which is useful in picking up lymphadenopathy, tubercular ascites, peritoneal thickening, omental thickening and bowel wall thickening in some cases. Plain radiographs may show enteroliths, perforation and features of intestinal obstruction (Figure 2A and B). Barium studies are still gold standard in diagnosing strictures, fistulae, erosions, etc. Contrast enhanced CT and CT enterography provide adequate cross sectional imaging in depicting various forms of abdominal TB.
In the recent years various molecular and immunological techniques are used as a new approach for the rapid diagnosis of abdominal TB[16,17]. The clinical samples can be ascitic fluid, lymphnode, thickened omentum or mesentery. The insertion sequence IS6110 is used as a target for polymerase chain reaction (PCR) amplification in these samples[16]. Real time assay using fluorescence resonance energy transfer hybridization probes show a positivity index of 36% in those patients with clinical and radiological suspicion of TB, but with negative acid fast bacilli (AFB) and culture[17]. In addition, immune characterization show depleted CD4+ count and increased levels of interferon-γ and tumor necrosis factor-α cytokines. Multiplex PCR using MPB64 and IS6110 are useful in rapid diagnosis of gastrointestinal TB[18]. Multiplex PCR has sensitivity and specificity of 90% and 100%, respectively in confirmed (AFB/culture/histopathology) cases of gastrointestinal TB and positive results in 72.41% of the suspected gastrointestinal TB cases[18].
The abdominal TB is of four types: tubercular lymphadenopathy, peritoneal TB, gastrointestinal TB and visceral TB (Table 2).
| Tubercular lymphadenopathy |
| Peritoneal tuberculosis |
| Acute |
| Chronic |
| Wet ascitic type |
| Fixed fibrotic type |
| Dry plastic type |
| Encysted/loculated type |
| Visceral tuberculosis |
| Liver, pancreas, spleen etc. |
| Gastrointestinal tuberculosis |
| Esophageal tuberculosis |
| Gastric tuberculosis |
| Duodenal tuberculosis |
| Jejunal and ileocecal tuberculosis |
| Colorectal tuberculosis |
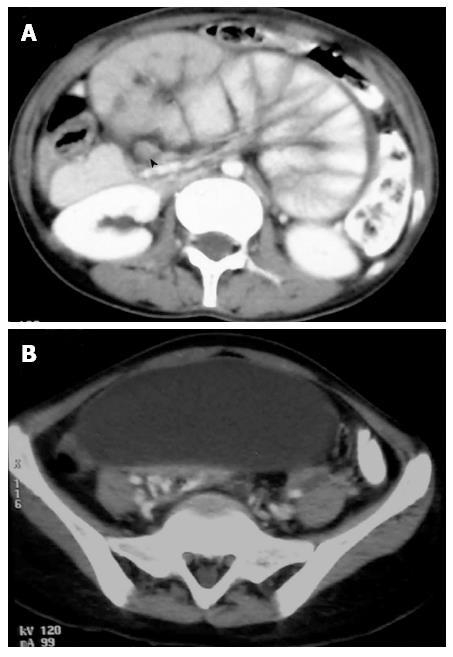
Abdominal lymphadenopathy is the most common manifestation of the abdominal TB. It usually follows the drainage of the affected organs, though it can affect any lymphnode in abdomen. The most commonly involved lymphnodes are the mesenteric nodes, omental nodes, those at porta hepatis, along the celiac axis and in peripancreatic location. The commonest route of transmission is usually secondary to ingestion of the infected material along with associated intestinal TB. Hematogenous route of transmission and contiguous spread from adjacent affected abdominal organ can also occur. On imaging, the appearance of affected lymphnodes can vary from increase in the number of the normal sized nodes to increase in the size of the nodes to formation of large conglomerate lymphnodal masses. However, the most common presentation is presence of multiple mildly enlarged nodes in clusters which are circular or ovoid. They usually have central areas of caseous necrosis with peripheral enhancement (Figure 3).
Traditionally the peritoneal TB is divided into three types: (1) The wet ascitic type is more common and is associated with large amounts of free or loculated fluid in abdomen; the ascites is usually of high density due to increased protein content of the inflammatory exudate. Associated peritoneal enhancement is usually present; (2) The fixed fibrotic type is relatively less common and is characterized by involvement of omentum and mesentery and is characterized by presence of matted bowel loops on imaging. Loculated ascites can be occasionally present; and (3) The dry plastic type is characterized by fibrous peritoneal reaction, peritoneal nodules and presence of adhesions. However, this classification is usually not adequate and a combination of features are usually noted (Figures 4 and 5).
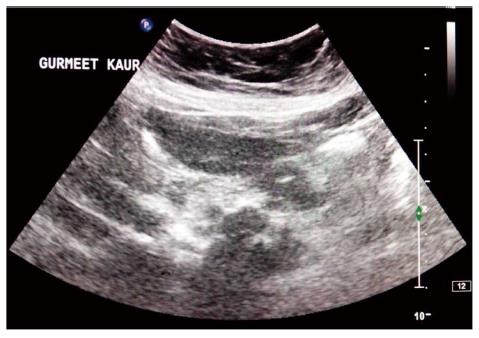
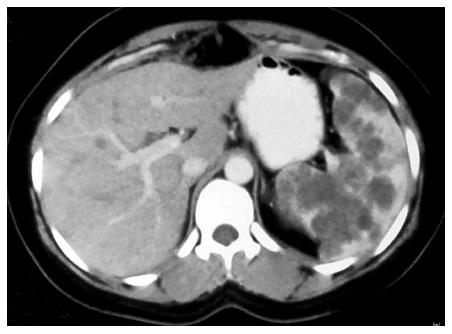
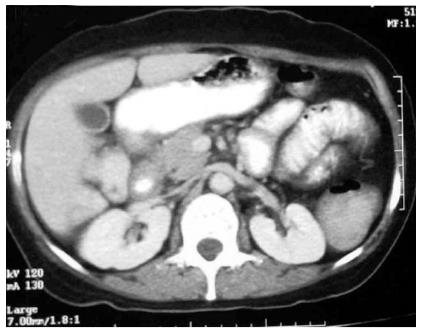
Isolated involvement of abdominal solid organs is relatively uncommon and occurs in 15%-20% of all patients with abdominal TB[19]. The genitourinary system is the most commonly involved followed by liver, spleen and pancreas. The mode of spread is through hematogenous route. Only 15% of patients have concomitant pulmonary TB[20]. Moreover, these patients present with nonspecific symptoms, all of which leads to difficulty in making prospective diagnosis. Cross sectional imaging with USG, multidetector CT, and MRI plays an important role in the diagnosis and post treatment follow up of tuberculosis (Figure 6)[19].
The most common site of the gastrointestinal TB is ileocecal location (ileocecal TB), followed by jejunum and colon. The esophagus, stomach and duodenum are rarely involved.
Esophageal TB: The esophageal involvement is extremely rare in immunocompetent patients and account for only 0.2%-1% of gastrointestinal TB[20,21], but it is more commonly seen in AIDS patients[10,22]. The symptoms are usually retrosternal pain, dysphagia and odynophagia. In esophagus, TB usually occurs in the middle third and manifests as ulcerative lesion or a tumor like lesion. The esophageal involvement is usually secondary to a contiguous tubercular mediastinal nodal involvement (extrinsic TB). Hematogenous spread from a small endobronchial lesion can occur as well. Primary involvement of esophagus (intrinsic TB) is extremely rare and can be attributed to ingestion of the infected sputum.
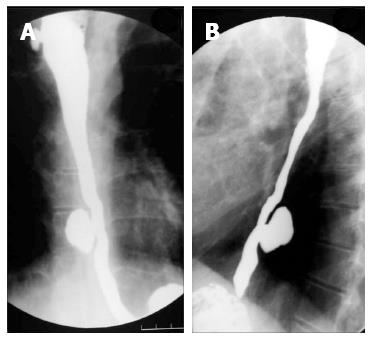
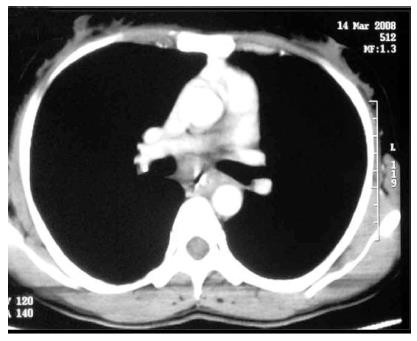
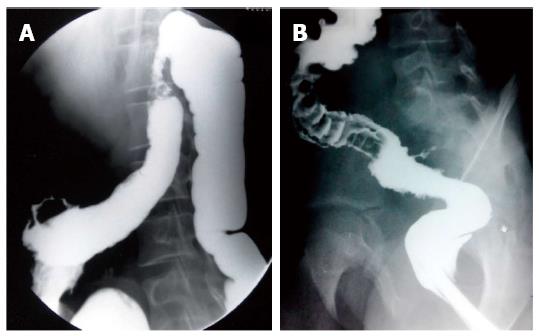
On barium studies, extrinsic TB is seen as narrowing or displacement of esophagus especially at the level of carina[23]. In late stages, the patients may present with formation of ulcers, strictures and fistulae. In fibrotic mediastinal disease, traction diverticulae can occur in the adjoining esophagus (Figure 7)[24]. The diagnosis of primary esophageal TB is usually made on histopathological examination as the radiological features are nonspecific (Figure 8) and are driven by high degree of suspicion. Since the tuberculous granulomas are located deep in the submucosal layer of esophagus, multiple and deep esophageal endoscopic biopsies should be obtained for optimum diagnosis[21].
Gastric TB: The gastric involvement is usually associated with pulmonary tuberculosis or immunodeficient state[20]. Primary involvement of the stomach is rare (0.4%-2%) because of the bactericidal property of the gastric acid, scarcity of lymphoid tissue in gastric wall and thick intact gastric mucosa. The other routes of spread can be either hematogenous or from adjacent lymphnodes. The patients may present with nonspecific symptoms of vague epigastric discomfort, weight loss and fever or may present with features of gastric outlet obstruction. Morphologically many types of gastric involvement can be seen. The most common type is ulcerative lesion along the lesser curvature and pylorus[25]. The other types are hypertrophic variety, presence of multiple miliary tubercles and tubercular pyloric stenosis in late stages. Associated tubercular lymphadenitis is usually present.
On imaging these lesions can appear as benign ulcers or erosions. In late stages features of pyloric stenosis with distorted antropyloric region can be present (Figure 9). The differential diagnosis of gastric TB can be carcinoma, lymphoma and other infections like syphilis.
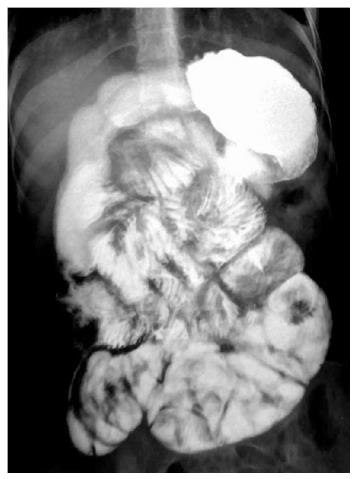
Duodenal TB: Duodenal involvement is seen in 2%-2.5% of all gastrointestinal TB cases[26]. As in stomach, primary involvement is rare. The most common site of primary involvement is the third part of duodenum[27]. It can be intrinsic, extrinsic or both[28]. Extrinsic form, which is more common, is usually secondary to the lymphadenopathy in the C-loop of the duodenum. The intrinsic form can be ulcerative, hypertrophic or ulcero-hypertrophic. It is usually complicated by formation of strictures or fistulas. These patients usually present late with features of obstruction where strictures are identified on imaging or an extrinsic impression by lymphadenopathy is noted. Barium studies demonstrate presence of narrowing either extrinsic or intrinisic (Figure 10)[29]. Band like narrowing of third part of duodenum may mimic superior mesenteric artery syndrome. A persistent narrow stream of barium in the bowel suggestive of the ‘‘string sign’’ may be seen. Formation of fistulous communication is readily identified by real time barium studies. Cross sectional imaging like CT demonstrates luminal narrowing, mural thickening of duodenum (Figure 11) and also adjacent lymphnodal masses causing compression. The differential diagnosis of duodenal TB can be lymphoma, malignancy, atypical peptic ulcer disease or carcinoma of head of pancreas. Since the features of duodenal TB can be nonspecific, a high index of suspicion is necessary for diagnosis.
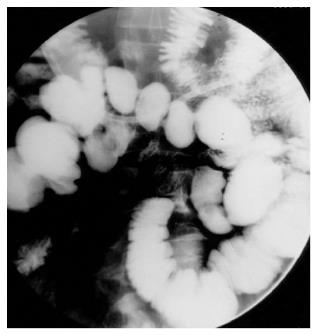
Jejunal and ileocecal TB: The most common site of GI involvement is the ileocecal region which is involved in 64% of cases of gastrointestinal TB[30]. The terminal ileum is more commonly involved because of the various contributing factors like stasis, presence of abundant lymphoid tissue, increased rate of absorption at this site and closer contact of the bacilli with the mucosa[7,31,32]. Concomitant jejunal involvement may be seen in the form of single or multiple short or long segment strictures. Isolated jejunal involvement is rare, and if present, may mimic Crohn’s disease (CD)[33]. The clinical features of small intestinal TB are protean, and the patients usually present with colicky abdominal pain, borborygmi and vomiting[30]. The most common complication is bowel obstruction secondary to hyperplastic mural thickening, stricture formation or due to adhesions[9]. Tuberculosis accounts for 5%-9% of all small intestinal perforations in India, and is the second commonest cause after typhoid fever[30].
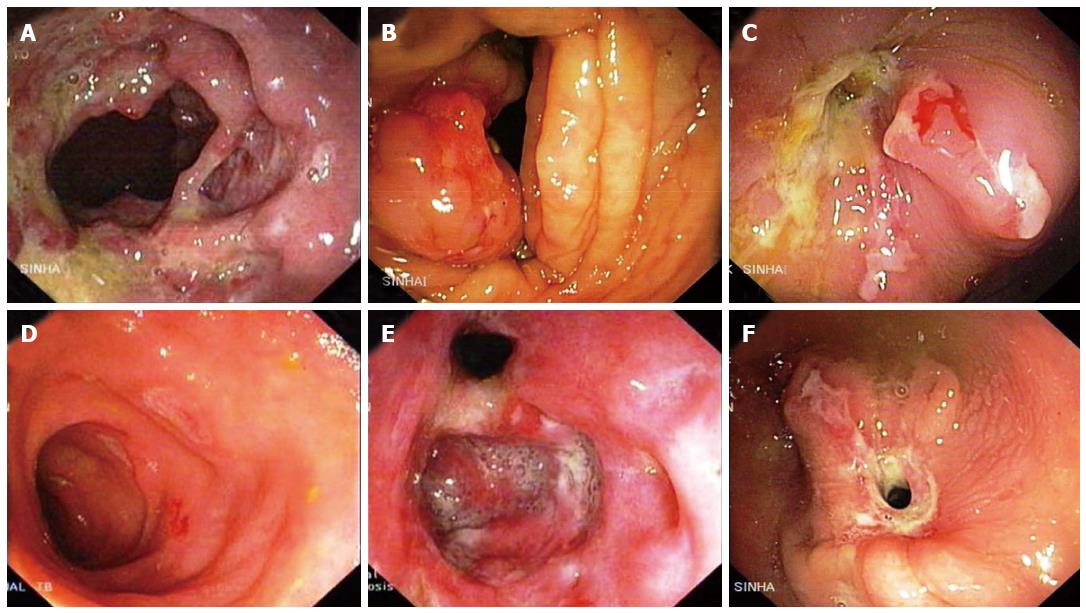
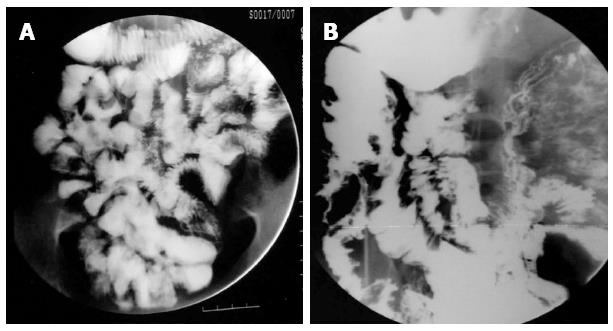
Morphologically, the lesions in intestinal tuberculosis are classified into ulcerative and ulcero-hypertrophic varieties. The distinction between these two lesions is not sharp and the two types may co-exist (Figure 12A and B). The macroscopic features present a very wide range and at times distinction from CD may be difficult. Confluent granulomas, presence of caseation necrosis, presence of granulomas in lymphnode in the absence of granulomatous lesions in the intestine, absence of transmural cracks and fissures (present in CD) serve to distinguish intestinal tuberculosis from CD[30]. Caseation necrosis remains a very important criterion for the histological diagnosis of intestinal TB. The terminal ileum, ileocecal (IC) junction and the cecum are concomitantly involved in the majority of cases (Figure 12C-F).
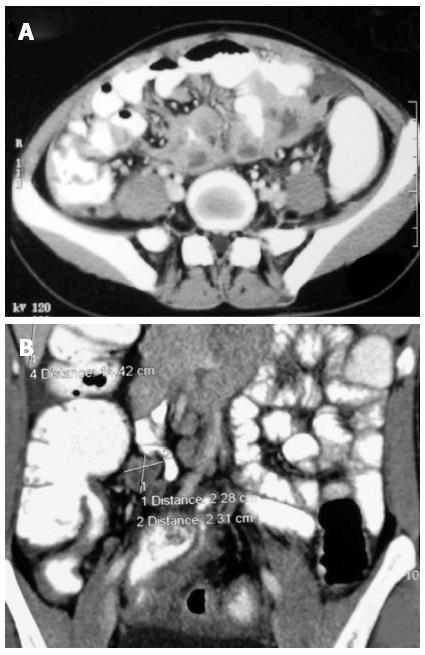
Plain X-ray abdomen may show enteroliths, features of obstruction like dilated bowel loops with multiple air fluid levels (Figure 2A and B), or presence of air under diaphragm in case of perforation. In addition, there may be calcified lymphnodes, calcified granulomas and hepatosplenomegaly. The features which may be seen in barium studies are accelerated intestinal transit; hypersegmentation of the barium column (“chicken intestine”), precipitation, flocculation and dilution of the barium; stiffened and thickened folds; luminal stenosis with smooth but stiff contours (“hourglass stenosis”), multiple strictures with segmental dilatation of bowel loops; and fixity and matting of bowel loops (Figure 13A). Various signs have been described in the ileocecal TB are the “Fleischner” or “inverted umbrella” sign, in which there is thickening of the lips of the ileocecal valve and/or wide gaping of the valve with narrowing of the terminal ileum. The “Goose neck deformity” signifies loss of normal ileocaecal angle and dilated terminal ileum, appearing suspended from a retracted, fibrosed cecum. The “Purse string stenosis” signifies localized stenosis opposite the ileocecal valve with a rounded off smooth cecum and a dilated terminal ileum. The two other signs are “Stierlin sign” and “string sign.” The “Stierlin sign” is a manifestation of acute inflammation superimposed on a chronically involved segment and is characterized by lack of barium retention in the inflamed segments of the ileum, cecum and variable length of the ascending colon, with a normal configured column of barium on either side. It appears as a narrowing of the terminal ileum with rapid emptying into a shortened, rigid or obliterated cecum. The “String sign” shows persistent narrow stream of barium indicating stenosis (Figure 13B). Both Stierlin and String signs can also be seen in CD and hence are not specific for TB[34]. Computed tomography may show circumferential wall thickening up to 3 cm in the cecum and terminal ileum with associated mesenteric lymphadenopathy (Figure 14A and B)[22,35]. Typically, asymmetric thickening of the IC valve and medial wall of the cecum, exophytic and engulfing the terminal ileum is seen. The associated lymphadenopathy is usually massive and central areas of low attenuation are indicative of caseous necrosis. Multiple small segment concentric strictures involving the jejunum and ileum can also be seen (Figure 15). Enteroclysis followed by a barium enema may still be the best protocol for evaluation of intestinal TB[23].
Colorectal TB: Isolated involvement of colon is 10.8%. The incidence is increased in the immunocompromised patients and patients with AIDS. The cecum is the most common site of involvement of colon but it is usually involved in contiguous involvement with the terminal ileum and IC junction. Apart from cecum, the most common site of isolated or segmental colon involvement varies between different studies. In one study, the most common site of involvement was transverse colon followed closely by rectum and ascending colon[36]. In another study, the most common site of colorectal TB was ascending colon followed by transverse colon and descending colon[6]. Multifocal involvement is seen in 28%-44% of cases with colorectal TB[30].
The most common clinical features are abdominal pain followed by loss of weight and appetite and altered bowel habits. During colonoscopy, the most common finding of colorectal TB is presence of ulcers, which are linear/fissured, transverse or circumferential and are covered with dull white or yellow exudates[6]. These ulcers can be differentiated from those of CD by presence of abnormal mucosa surrounding these ulcers which show features like erythema, edema, mucosal irregularity and nodularity. In contrast, the ulcers in the CD are usually surrounded by normal appearing mucosa. Moreover, aphthous ulcers are seen in CD, are not usually seen in TB[31-36]. The parameters like anorectal lesions, longitudinal ulcers, aphthous ulcers and cobblestone appearance are significantly more common in patients with CD than in those with TB[37].
The imaging in earlier stage can be nonspecific and include spasm and hypermotility of the intestine. Since the clinical features of the colorectal TB can be nonspecific, the patients usually present in a later stage. So the more common radiological features are presence of strictures (Figure 16A), followed by features of colitis and polypoidal lesions[35]. Complications in the form of perforations and fistulae can be seen in upto 18.9% of cases (Figure 16). Various conditions like CD, amebic colitis, pseudomembranous and ischemic colitis, and malignancy form the differential diagnosis of TB colitis. So a diagnosis of colorectal TB should be based on high index of suspicion and should be proven by colonoscopy guided biopsy and demonstration of caseating granulomas in the tissue. Majority of the lesions of colorectal TB show resolution following antitubercular therapy and therefore a repeat colonoscopy may not be required if the patient is asymptomatic after treatment[6].
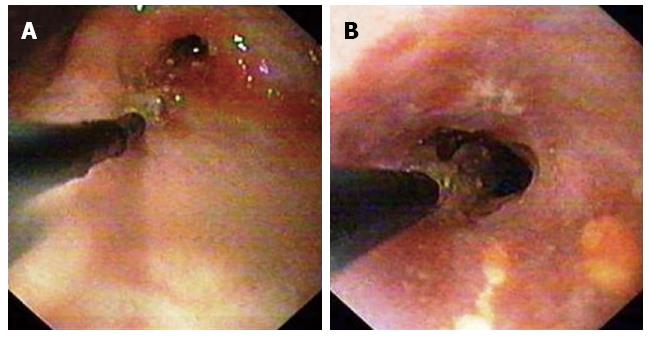
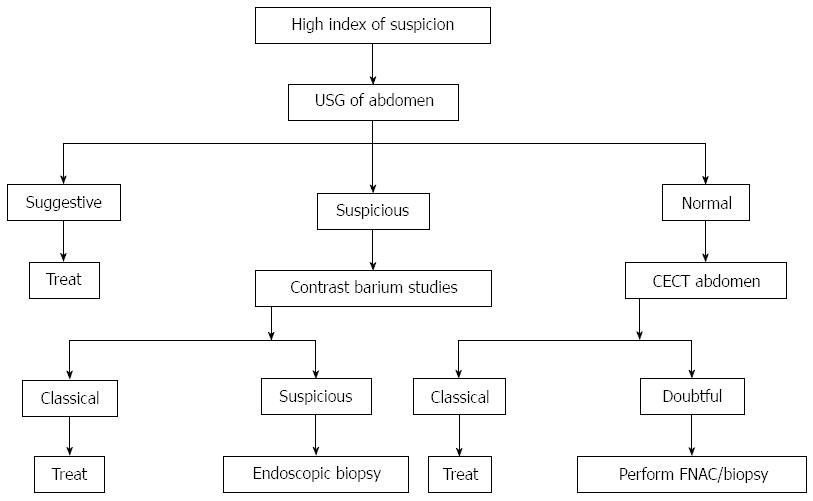
Abdominal TB is generally responsive to medical treatment alone, so early diagnosis can prevent unnecessary surgical intervention[38]. In general, with advent of antituberculous therapy, surgery is usually reserved for those cases where it is absolutely indicated as in cases of non-resolving intestinal obstruction, perforation and abscess or fistula formation. Even in cases of tuberculous strictures medical management with antituberculous drugs will result in significant resolution of symptoms in most of patients[39]. Endoscopic balloon dilation offers an alternative to the surgical management of GI strictures (Figure 9)[40]. Management algorithm for the abdominal TB is presented in (Figure 17)[41].
All the diagnosed cases of gastrointestinal TB should receive at least 6 mo of antituberculous therapy which includes initial two months of therapy with isoniazid, rifampicin, pyrazinamide and ethambutol thrice weekly[12]. Although 6 mo treatment regimen is recommended as per the revised national tuberculosis program guidelines, many clinicians extend the treatment regimen to 9 or 12 mo. However, no difference seen in effectiveness between the 6 mo short course therapy regimen with rifampicin, isoniazid and pyrazinamide for 2 mo followed by rifampicin with isoniazid for another 4 mo (6R series) and 12 mo standard regimen of ethambutol and isoniazid with streptomycin supplemented for 2 wk[33].
The surgeries performed in the gastrointestinal TB are of three types[12]. The first type is the surgery which is done to bypass the involved segments of bowel as in case of an enteroenterostomy or an ileotransverse colostomy. These surgeries are usually complicated by blind loop syndrome, fistula formation and recurrent disease in the remaining segments and hence usually not performed routinely. The second type of surgeries are those involving the radical resection like a right hemicolectomy which are feasible with the advent of effective antituberculous drugs so as to eradicate the disease completely. However, these surgeries are also hindered by malnourished status of the most of the patients which make them poor surgical candidates. Also the lesions can be widely placed and radical resection may not be possible in all the cases. The third type of surgeries is usually conservative like a strictureplasty in those strictures which cause more than 50% luminal compromise[42]. These kinds of conservative surgeries are usually preferred nowadays. The tuberculous bowel perforations are usually treated with resection of involved segments with primary anastomosis.
Abdominal TB can be of various forms like peritoneal TB, tuberculous lymphadenopathy, gastrointestinal TB and visceral TB. Though gastrointestinal TB usually involves the ileocecal region, it can virtually affect any part of GI tract. The symptoms of abdominal TB can be nonspecific. Various imaging features and radiological signs are useful in making a diagnosis of abdominal TB. A high degree of clinical suspicion is required to make a diagnosis of TB in those areas which are not commonly involved. Various molecular and immunological techniques are increasingly used for rapid diagnosis in suspected cases of abdominal TB. Gastrointestinal TB is generally managed with medical therapy with antituberculous drugs and surgeries are usually conservative and are done only if absolutely indicated.
P- Reviewer: Boonsarngsuk V, Nagata T S- Editor: Gou SX L- Editor: A E- Editor: Wang CH
| 1. | Rosado E, Penha D, Paixao P, Costa AMD, Amadora PT. Abdominal tuberculosis - Imaging findings. Educational exhibit; ECR. 2013;C-0549. [DOI] [Full Text] |
| 2. | World Health Organization. Global tuberculosis report 2013. Geneva: WHO. 23 Oct 2013; Available from: http://apps.who.int/iris/bitstream/10665/91355/1/9789241564656_eng.pdf. |
| 3. | Pop M, Pop C, Homorodean D, Itu C, Man M, Goron M, Gherasim R, Coroiu G. Abdominal miliary tuberculosis in a patient with AIDS: a case report. Rom J Gastroenterol. 2003;12:231-234. [PubMed] |
| 4. | Albalak R, O’Brien RJ, Kammerer JS, O’Brien SM, Marks SM, Castro KG, Moore M. Trends in tuberculosis/human immunodeficiency virus comorbidity, United States, 1993-2004. Arch Intern Med. 2007;167:2443-2452. [PubMed] |
| 5. | Burzynski J, Schluger NW. The epidemiology of tuberculosis in the United States. Semin Respir Crit Care Med. 2008;29:492-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 6. | Mukewar S, Mukewar S, Ravi R, Prasad A, S Dua K. Colon tuberculosis: endoscopic features and prospective endoscopic follow-up after anti-tuberculosis treatment. Clin Transl Gastroenterol. 2012;3:e24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 7. | Horvath KD, Whelan RL. Intestinal tuberculosis: return of an old disease. Am J Gastroenterol. 1998;93:692-696. [PubMed] |
| 8. | Akhan O, Pringot J. Imaging of abdominal tuberculosis. Eur Radiol. 2002;12:312-323. [PubMed] |
| 9. | Marshall JB. Tuberculosis of the gastrointestinal tract and peritoneum. Am J Gastroenterol. 1993;88:989-999. [PubMed] |
| 10. | McGuinness FE, Hamilton D, Al Nabulsi J. Tuberculosis of the gastrointestinal tract and peritoneum. Clinical imaging of non-pulmonary tuberculosis. Berlin: Springer 2000; 107-138. |
| 11. | Hopewell PC. A clinical view of tuberculosis. Radiol Clin North Am. 1995;33:641-653. [PubMed] |
| 12. | Sharma MP, Bhatia V. Abdominal tuberculosis. Indian J Med Res. 2004;120:305-315. [PubMed] |
| 13. | Wilkins EG. Tuberculous peritonitis: diagnostic value of the ascitic/blood glucose ratio. Tubercle. 1984;65:47-52. [PubMed] |
| 14. | Bhargava DK, Gupta M, Nijhawan S, Dasarathy S, Kushwaha AK. Adenosine deaminase (ADA) in peritoneal tuberculosis: diagnostic value in ascitic fluid and serum. Tubercle. 1990;71:121-126. [PubMed] |
| 15. | Sathar MA, Simjee AE, Coovadia YM, Soni PN, Moola SA, Insam B, Makumbi F. Ascitic fluid gamma interferon concentrations and adenosine deaminase activity in tuberculous peritonitis. Gut. 1995;36:419-421. [PubMed] |
| 16. | Mimidis K, Ritis K, Kartalis G. Peritoneal tuberculosis. Ann Gastroenterol. 2005;18:325-329. |
| 17. | Mishra PK, Bhargava A, Punde RP, Pathak N, Desikan P, Jain A, Varshney S, Maudar KK. Diagnosis of gastrointestinal tuberculosis: Using cytomorphological, microbiological, immunological and molecular techniques - A study from Central India. Indian J Clin Biochem. 2010;25:158-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 18. | Sharma K, Sinha SK, Sharma A, Nada R, Prasad KK, Goyal K, Rana SS, Bhasin DK, Sharma M. Multiplex PCR for rapid diagnosis of gastrointestinal tuberculosis. J Glob Infect Dis. 2013;5:49-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Tirumani SH, Ojili V, Gunabushanam G, Shanbhogue AK, Nagar A, Fasih N, Chintapalli KN. Imaging of tuberculosis of the abdominal viscera: beyond the intestines. J Clin Imaging Sci. 2013;3:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 20. | Padma V, Anand NN, Rajendran SM, Gurukal S. Primary tuberculosis of stomach. J Indian Med Assoc. 2012;110:187-188. [PubMed] |
| 21. | Welzel TM, Kawan T, Bohle W, Richter GM, Bosse A, Zoller WG. An unusual cause of dysphagia: esophageal tuberculosis. J Gastrointestin Liver Dis. 2010;19:321-324. [PubMed] |
| 22. | Leder RA, Low VH. Tuberculosis of the abdomen. Radiol Clin North Am. 1995;33:691-705. [PubMed] |
| 23. | Vanhoenacker FM, De Backer AI, Op de BB, Maes M, Van Altena R, Van Beckevoort D, Kersemans P, De Schepper AM. Imaging of gastrointestinal and abdominal tuberculosis. Eur Radiol. 2004;14 Suppl 3:E103-E115. [PubMed] |
| 24. | Kaman L, Kundel B, Sinha SK, Singh R. True epiphrenic diverticulum of esophagus secondary to tubercular adenitis. Indian J Gastroenterol. 2003;22:65-66. [PubMed] |
| 25. | Chetri K, Prasad KK, Jain M, Choudhuri G. Gastric tuberculosis presenting as non-healing ulcer: case report. Trop Gastroenterol. 2000;21:180-181. [PubMed] |
| 26. | Paustin FF, Marshall JB. Intestinal tuberculosis. Bockus Gastroenterology. 4th ed. Philadelphia: WB Saunders 1985; 2018-2036. |
| 27. | Reader MM, Philip ESP. Infections and infestations. Alimentary tract radiology. St Louis: CV Mosby 1989; 1478-1479. |
| 28. | Bhatti A, Hussain M, Kumar D, Samo KA. Duodenal tuberculosis. J Coll Physicians Surg Pak. 2012;22:111-112. [PubMed] |
| 29. | Chavhan GE, Ramakantan R. Duodenal tuberculosis: radiological features on barium studies and their clinical correlation in 28 cases. J Postgrad Med. 2003;49:214-217. [PubMed] |
| 30. | Sharma R. Abdominal Tuberculosis. Imaging Science Today 2009: 146. Available from: http://www.imagingsciencetoday.com/node/146. |
| 31. | Alvares JF, Devarbhavi H, Makhija P, Rao S, Kottoor R. Clinical, colonoscopic, and histological profile of colonic tuberculosis in a tertiary hospital. Endoscopy. 2005;37:351-356. [PubMed] |
| 32. | Das HS, Rathi P, Sawant P, Chodankar CM, Vyas K, Patrawala V, Dhadphale S. Colonic tuberculosis: colonoscopic appearance and clinico-pathologic analysis. J Assoc Physicians India. 2000;48:708-710. [PubMed] |
| 33. | Balasubramanian R, Nagarajan M, Balambal R, Tripathy SP, Sundararaman R, Venkatesan P, Paramasivam CN, Rajasambandam P, Rangabashyam N, Prabhakar R. Randomised controlled clinical trial of short course chemotherapy in abdominal tuberculosis: a five-year report. Int J Tuberc Lung Dis. 1997;1:44-51. [PubMed] |
| 34. | Harisinghani MG, McLoud TC, Shepard JA, Ko JP, Shroff MM, Mueller PR. Tuberculosis from head to toe. Radiographics. 2000;20:449-470; quiz 528-529, 532. [PubMed] |
| 35. | Nagi B, Kochhar R, Bhasin DK, Singh K. Colorectal tuberculosis. Eur Radiol. 2003;13:1907-1912. [PubMed] |
| 36. | Shah S, Thomas V, Mathan M, Chacko A, Chandy G, Ramakrishna BS, Rolston DD. Colonoscopic study of 50 patients with colonic tuberculosis. Gut. 1992;33:347-351. [PubMed] |
| 37. | Lee YJ, Yang SK, Byeon JS, Myung SJ, Chang HS, Hong SS, Kim KJ, Lee GH, Jung HY, Hong WS. Analysis of colonoscopic findings in the differential diagnosis between intestinal tuberculosis and Crohn’s disease. Endoscopy. 2006;38:592-597. [PubMed] |
| 38. | Uzunkoy A, Harma M, Harma M. Diagnosis of abdominal tuberculosis: experience from 11 cases and review of the literature. World J Gastroenterol. 2004;10:3647-3649. [PubMed] |
| 39. | Anand BS, Nanda R, Sachdev GK. Response of tuberculous stricture to antituberculous treatment. Gut. 1988;29:62-69. [PubMed] |
| 40. | Bhasin DK, Sharma BC, Dhavan S, Sethi A, Sinha SK, Singh K. Endoscopic balloon dilation of ileal stricture due to tuberculosis. Endoscopy. 1998;30:S44. [PubMed] |
| 41. | Sood R, Sethu Madhavan M. Diagnostic approach to abdominal tuberculosis. Clinical Medicine: A Practical manual for students and practitioners. India: Jaypee Brothers Medical Publishers (P) Ltd 2007; 249. |