Published online Apr 28, 2014. doi: 10.3748/wjg.v20.i16.4702
Revised: December 25, 2013
Accepted: February 26, 2014
Published online: April 28, 2014
Processing time: 186 Days and 12.1 Hours
AIM: To evaluate the performance of a novel non-invasive controlled attenuation parameter (CAP) to assess liver steatosis.
METHODS: This was a multi-center prospective cohort study. Consecutive patients (aged ≥ 18 years) who had undergone percutaneous liver biopsy and CAP measurement were recruited from three Chinese liver centers. Steatosis was categorized as S0: < 5%; S1: 5%-33%; S2: 34%-66%; or S3: ≥ 67%, according to the nonalcoholic fatty liver disease (NAFLD) activity score. The FibroScan® 502 equipped with the M probe (Echosens, Paris, France) was used to capture both CAP and liver stiffness measurement values simultaneously. Receiver operating characteristic curves were plotted, and the areas under the curves were calculated to determine the diagnostic efficacy. The accuracy of the CAP values at the optimal thresholds was defined by maximizing the sum of sensitivity and specificity (maximum Youden index).
RESULTS: A total of 152 patients were recruited, including 52 (34.2%) patients with NAFLD and 100 (65.8%) with chronic hepatitis B (CHB) virus infection. After adjustment, the steatosis grade (OR = 37.12; 95%CI: 21.63-52.60, P < 0.001) and body mass index (BMI, OR = 6.20; 95%CI: 2.92-9.48, P < 0.001) were found independently associated with CAP by multivariate linear regression analysis. CAP was not influenced by inflammation, fibrosis or aetiology. The median CAP values and interquartile ranges among patients with S0, S1, S2 and S3 steatosis were 211 (181-240) dB/m, 270 (253-305) dB/m, 330 (302-360) dB/m, and 346 (313-363) dB/m, respectively. The cut-offs for the CAP values in all patients with steatosis ≥ 5%, ≥ 34% and ≥ 67% were 253 dB/m, 285 dB/m and 310 dB/m, respectively. The areas under the curves were 0.92, 0.92 and 0.88 for steatosis ≥ 5%, ≥ 34% and ≥ 67%, respectively. No significant differences were found in the CAP values between the NAFLD group and the CHB group in each steatosis grade.
CONCLUSION: CAP appears to be a promising tool for the non-invasive detection and quantification of hepatic steatosis, but is limited by BMI.
Core tip: We introduced a novel controlled attenuation parameter (CAP) which was acquired using the FibroScan® equipped with the M probe. This multi-center prospective cohort study was performed in Chinese nonalcoholic fatty liver disease or chronic hepatitis B patients. Following multivariate linear regression analysis, we found that CAP was significantly correlated with steatosis grade and was not influenced by inflammation, fibrosis or aetiology. Although it is less effective in identifying moderate to severe steatosis and limited by body mass index, we believe that CAP values are more useful than the measurement of 5% steatosis and may be used as a substitute for ultrasonography in epidemiological investigations of fatty liver.
- Citation: Shen F, Zheng RD, Mi YQ, Wang XY, Pan Q, Chen GY, Cao HX, Chen ML, Xu L, Chen JN, Cao Y, Zhang RN, Xu LM, Fan JG. Controlled attenuation parameter for non-invasive assessment of hepatic steatosis in Chinese patients. World J Gastroenterol 2014; 20(16): 4702-4711
- URL: https://www.wjgnet.com/1007-9327/full/v20/i16/4702.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i16.4702
Hepatic steatosis is diagnosed when the accumulation of fatty droplets (mainly triglycerides) exceeds more than 5% of liver weight[1]. The prevalence of hepatic steatosis is rising in association with the global increase in obesity and type 2 diabetes and is currently present in 2%-40% of the general population[2], 50.9% of individuals with chronic hepatitis C (CHC)[3], 46.4% of heavy drinkers[4] and 50%-80% of obese individuals[5]. In the past, simple steatosis was regarded as benign and reversible. However, the presence of other aetiologies may act in synergy with steatosis to aggravate liver injury, enhance oxidative stress, produce inflammation, increase susceptibility to apoptosis and even promote the progression of fibrosis[6]. Therefore, it is necessary to accurately quantify the extent of hepatic steatosis and monitor its dynamic changes.
Although liver biopsy (LB) is regarded as the gold standard to assess hepatic steatosis, its use has several limitations, including sampling bias, intra- or inter-observer sampling variability, and the potential for severe complications[7]. Therefore, patients opt to avoid such an invasive procedure and frequently refuse to repeat it. As a result, there is a need for a simple and reliable non-invasive alternative that either complements or eliminates liver biopsy altogether.
Recently, a novel non-invasive tool based on ultrasound attenuation was developed to assess liver steatosis. The evaluation of ultrasound attenuation has been implemented with the FibroScan® (Echosens, Paris, France) using a novel proprietary algorithm called the controlled attenuation parameter (CAP)[1]. In the existing literature, CAP displayed good diagnostic value for chronic liver diseases such as viral hepatitis[8-9] and multi-aetiology cohorts[10-12]. However, there is no research using CAP values to assess hepatic steatosis in the Chinese population. CHB is the most prevalent liver disease in China, and nonalcoholic fatty liver disease (NAFLD) is also highly prevalent, especially in the more affluent regions[13]. Furthermore, the coexistence of hepatitis B virus (HBV) infection and NAFLD is a novel characteristic of liver disease in the Chinese population. Therefore, the aim of this study was to evaluate the performance of CAP measurements in assessing steatosis, in a cohort of consecutive NAFLD/CHB patients in China, using liver biopsy as the reference.
Adults (aged ≥ 18 years) with NAFLD or HBV infection (with or without steatosis) were eligible for the study. Patients were prospectively recruited from three Chinese liver centers (Xinhua Hospital, Shanghai; Dongnan Hospital, Fujian; and Tianjin Hospital of Infectious Diseases, Tianjin) between March 2012 and March 2013. The ethics committees of the three hospitals approved the study, and all patients gave their written informed consent before participation.
Each patient had undergone percutaneous liver biopsy and transient elastography (TE) within 4 wk and met the diagnostic criteria for either NAFLD[14] or CHB[15]. Exclusion criteria were: (1) alcohol intake per week greater than 140 g in men and 70 g in women in the past 12 months; (2) other diseases that lead to fatty liver (e.g., CHC, drug-induced liver disease, total parenteral nutrition, hepatolenticular degeneration, autoimmune liver disease, etc.); (3) previous liver transplantation; (4) other terminal disease or malignancy; (5) refusal to undergo LB or disqualified biopsy specimens; and (6) contraindications to FibroScan® examination (e.g., ascites, implanted pacemakers, non-healing wounds in the upper-right quadrant of the abdomen, pregnancy, etc.) or unreliable CAP measurements [e.g., success rate less than 60% or interquartile range (IQR) > 30%].
All patients received physical examinations at the time of TE measurement. Liver disease aetiology and anthropometric measurements, including body mass index [BMI, weight (kg)/height (m)2] and waist-to-hip ratio [WHR, waist circumference (cm)/hip circumference (cm)] were obtained. Demographic information, such as age, sex and medical/drinking history were obtained from patient interviews during screening. Laboratory data, including liver biochemistry, fasting glucose, total cholesterol (TC), triglycerides (TG), high density lipoprotein cholesterol (HDL-C) and low density lipoprotein cholesterol (LDL-C) within 4 wk of the liver biopsy and in fasting conditions were recorded. HBsAg, HBeAg and anti-HBe were determined with commercially available enzyme-linked immunosorbent assay kits. Serum HBV DNA was measured by the real-time PCR Cobas Taqman assay if HBV was positive.
Percutaneous LB was performed with an 18-gauge BARD Max-Core Disposable Biopsy Instrument (BARD Biopsy Systems, Tempe, AZ, United States) from the right lobe under real-time ultrasound guidance. Biopsy specimens were formalin-fixed, paraffin-embedded, sectioned, and stained with HE, Masson’s trichome stain and reticulin. Liver biopsy sections were interpreted by two experienced hepatopathologists who were blinded to the clinical data, and a consensus was required in the case of discordant results. The length of the sample was required to be ≥ 15 mm, and the sample was to contain at least 6 portal tracts (PTs). For both NAFLD and CHB samples, the liver sections were first evaluated for percentage of lipid deposition, and the presence of visible steatosis in ≥ 5% of hepatocytes was considered to represent fatty liver[16] which was evaluated by light microscopic examination of an HE liver section (4-5 μm thick) under a 10 × objective lens[17]. Steatosis was categorised as S0: < 5%; S1: 5%-33%; S2: 34%-66%; or S3: ≥ 67%, according to the NAFLD activity score (NAS)[16].
The grades and stages of the liver samples were dependent on the liver disease aetiology. The METAVIR classification was used for CHB and fibrosis was staged from F0 to F4: F0, no fibrosis; F1, portal fibrosis without septa; F2, portal fibrosis with few septa; F3, numerous septa without cirrhosis; and F4, cirrhosis[18]. Fibrosis in NAFLD was staged as follows: F0, no fibrosis; F1, perisinusoidal or periportal fibrosis; F2, perisinusoidal and portal/periportal fibrosis; F3, bridging fibrosis; and F4, cirrhosis. Because of the different diagnostic criteria, METAVIR grades of A2-A3 in patients with CHB and NAS scores of ≥ 5 in patients with NAFLD were classified as moderate to severe inflammation, and F ≥ 2 was classified as significant fibrosis.
One certified operator with experience of more than 200 cases in each centre performed the TE examinations and was blinded to the liver histology. The FibroScan® 502 equipped with the M probe (Echosens, Paris, France) was used to capture both CAP and liver stiffness measurement (LSM) values simultaneously. CAP values and LSM values were expressed in units of decibels per metre (dB/m) and kilopascal (kPa), ranging from 100 to 400 dB/m and 2.5 to 75 kPa, respectively. Details of the LSM and CAP measurement principle were provided in previous publications[1,19]. Fasted patients were placed in the supine position with their right hand on their head in order to extend the intercostal space. The tip of the transducer probe was placed on the surface of the skin between the ribs and over the right lobe of the liver.
A reliable LSM was defined as more than 10 valid shots, a success rate of at least 60%, and an IQR < 30% of the median LSM value[19]. Since there are no reliability criteria for CAP measurement, it was arbitrarily decided to use the reliability criteria for LSM. Therefore if the LSM was reliable according to those criteria, the corresponding median CAP value was also considered reliable.
Continuous variables and patient characteristics were expressed as either medians (IQR) or n (%), as appropriate. The χ2 test and Fisher’s exact test were used to compare categorical data. The Mann-Whitney test was used to compare two groups, and the Kruskal-Wallis test was used to compare more than three groups. Correlations between CAP values and continuous variables were assessed by Spearman correlation coefficients (ρ), and multivariate analysis was performed using linear regressions.
The receiver operating characteristic (ROC) curves were plotted, and the areas under the curves (AUC) were calculated with 95% confidence intervals (CIs) to determine the diagnostic efficacy of CAP to differentiate between those with hepatic steatosis ≥ 5%, ≥ 34% and ≥ 67% versus controls. The accuracy of the CAP values at the optimal thresholds was defined by maximizing the sum of sensitivity and specificity (maximum Youden index). For each optimal cut-off value, the sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were calculated. All statistical analyses were performed using SPSS version 13 for Windows (SPSS, Chicago, IL, United States). Two-sided P values < 0.05 indicated statistical significance.
A total of 189 consecutive patients were screened within the study period. Of these patients, 15 were excluded due to withdrawal of consent, 6 for excessive alcohol consumption, 5 for disqualified biopsy specimens, and 11 due to the inability to acquire qualified CAP and/or LSM data. A total of 152 (80.4%) patients were finally included in the statistical analysis, including 52 (34.2%) with NAFLD and 100 (65.8%) with CHB. Demographic, anthropometric, laboratory, and FibroScan examination characteristics of the study population are outlined in Table 1. The median BMI (26.0 kg/m2, IQR: 24.4-29.3), waist circumference (90.0 cm, IQR 86.0-97.5), hip circumference (97.0 cm, IQR: 93.0-102.8) and WHR (0.94, IQR: 0.91-0.97) in the NAFLD group were significantly higher than those in the CHB group.
| Characteristics | All patients | NAFLD | CHB | P value |
| (n = 152) | (n = 52) | (n = 100) | ||
| Demographics | ||||
| Male gender, n (%)1 | 106 (69.3) | 36 (69.2) | 70 (70.0) | 0.992 |
| Age (yr)2 | 35 (28-49) | 39 (29-50) | 35 (27-49) | 0.167 |
| Anthropometrics | ||||
| BMI (kg/m2)2 | 24.9 (22.5-27.7) | 26.0 (24.4-29.3) | 23.9 (21.8-26.6) | < 0.001 |
| < 18.5 | 7 (4.6) | 0 | 7 (7.0) | |
| 18.5-24.9 | 70 (46.1) | 18 (34.6) | 52 (52.0) | |
| 25-29.9 | 61 (40.1) | 24 (46.2) | 37 (37.0) | |
| ≥ 30 | 14 (9.2) | 10 (19.2) | 4 (4.0) | |
| WHR2 | 0.92 (0.88-0.96) | 0.94 (0.91-0.97) | 0.90 (0.86-0.95) | 0.001 |
| Laboratory findings | ||||
| ALT (U/L) | 64.8 (37.9-134.0) | 55.0 (31.3-104.4) | 69.0 (40.0-187.0) | 0.022 |
| AST (U/L) | 44.0 (26.0-77.5) | 33.4 (25.0-67.0) | 46.3 (28.7-93.5) | 0.042 |
| ALP (U/L) | 89.0 (68.0-109.0) | 84.0 (63.1-109.0) | 90.3 (73.3-109.0) | 0.320 |
| Γ-GT (U/L) | 54.3 (28.9-104.4) | 60.0 (31.6-97.1) | 52.0 (26.5-117.3) | 0.770 |
| Albumin (g/L) | 42.7 (39.8-45.1) | 43.5 (41.5-46.8) | 42.1 (38.6-44.8) | 0.017 |
| Total bilirubin (μmol/L) | 14.2 (11.6-20.2) | 12.6 (10.5-15.5) | 15.2 (12.1-22.3) | 0.003 |
| Direct bilirubin (μmol/L) | 4.9 (3.7-7.8) | 4.6 (3.8-5.6) | 5.2 (3.7-9.0) | 0.080 |
| Total cholesterol (mmol/L) | 4.6 (4.0-5.1) | 4.7 (4.3-5.1) | 4.3 (3.7-5.1) | 0.018 |
| Triglyceride (mmol/L) | 1.4 (0.9-2.2) | 2.0 (1.3-2.7) | 1.3 (0.87-2.0) | 0.001 |
| HDL-C (mmol/L) | 1.2 (1.0-1.3) | 1.2 (1.1-1.4) | 1.1 (1.0-1.3) | 0.166 |
| LDL-C (mmol/L) | 2.5 (1.9-2.9) | 2.8 (2.6-3.1) | 2.3 (1.8-2.6) | < 0.001 |
| Prothrombin time (s) | 12.3 (11.1-13.0) | 11.9 (10.4-12.4) | 12.7 (12.0-13.4) | < 0.001 |
| Fasting glucose (μmol/L) | 5.3 (4.7-5.8) | 5.2 (4.5-6.0) | 5.3 (4.8-5.7) | 0.819 |
| Log10 (HBV-DNA,IU/mL) | - | - | 3.3 (0.92-5.9) | |
| Liver histology | ||||
| Steatosis grade, n (%)1 | < 0.001 | |||
| S0 (< 5%) | 63 (41.4) | 0 | 63 (63.0) | |
| S1 (5%-33%) | 44 (28.9) | 19 (36.5) | 25 (25.0) | |
| S2 (34%-66%) | 32 (21.1) | 23 (44.2) | 9 (9.0) | |
| S3 (≥ 67%) | 13 (8.6) | 10 (19.2) | 3 (3.0) | |
| Fibrosis stage, n (%)1 | 0.234 | |||
| F0 | - | 28 (53.8) | 36 (36.0) | |
| F1 | - | 13 (25.0) | 30 (30.0) | |
| F2 | - | 6 (11.5) | 20 (20.0) | |
| F3 | - | 4 (7.7) | 8 (8.0) | |
| F4 | - | 1 (1.9) | 6 (6.0) | |
| Significant fibrosis(F ≥ 2), n (%) | 45 (29.6) | 11 (21.2) | 34 (34.0) | 0.100 |
| Moderate to severe inflammation, n (%)13 | 62 (40.8) | 21 (40.4) | 41 (41.0) | 0.942 |
| FibroScan® parameters | ||||
| Controlled attenuation parameter (dB/m) | 262 (215-310) | 310 (273-347) | 236 (199-281) | < 0.001 |
| Liver stiffness measurement (kPa) | 7.5 (5.5-12.9) | 6.1 (4.8-11.1) | 8.0 (5.9-13.9) | 0.025 |
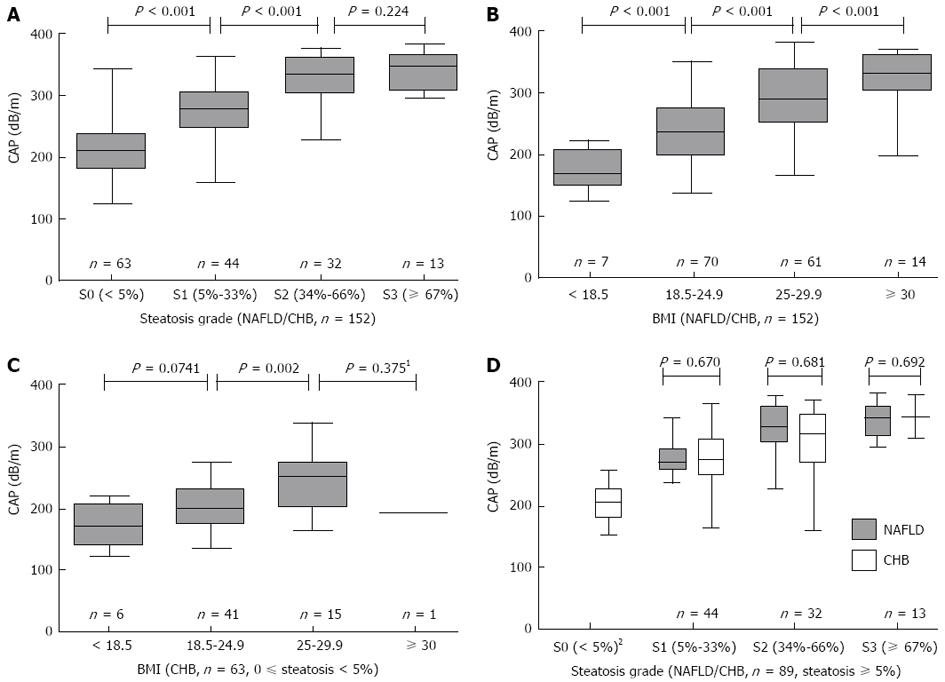
The characteristics of liver histology are also shown in Table 1. The median length and the PTs of the liver biopsy samples were 18 mm (IQR: 17-19) and 8 (IQR: 7-10), respectively. The 63 patients with a steatosis score of 0%-5% (S0) all had CHB.
BMI, WHR, albumin, TC, TG, LDL-C and steatosis grade showed significant positive associations with CAP, while alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin and direct bilirubin were negatively associated with CAP (Table 2). Parameters such as gender (P = 0.49), age (P = 0.93), alkaline phosphatase (P = 0.37), γ-glutamyl transpeptidase (P = 0.93), HDL-C (P = 0.24), fasting glucose (P = 0.20), HBV DNA levels (P = 0.42, for CHB patients), prothrombin time (P = 0.06), moderate to severe inflammation (P = 0.18), significant fibrosis (P = 0.55) and LSM (P = 0.43) were not significantly correlated with CAP.
| Spearman correlation | Multivariate linear regression | ||||
| Parameter | r | P value | OR | 95%CI: | P value |
| BMI | 0.49 | < 0.001 | 6.20 | 2.92-9.48 | < 0.001 |
| WHR | 0.32 | < 0.001 | - | ||
| ALT | -0.25 | 0.002 | - | ||
| AST | -0.25 | 0.002 | - | ||
| Albumin | 0.27 | 0.001 | - | ||
| Total bilirubin | -0.24 | 0.003 | - | ||
| Direct bilirubin | -0.17 | 0.039 | - | ||
| Total cholesterol | 0.18 | 0.034 | - | ||
| Triglyceride | 0.31 | < 0.001 | - | ||
| LDL-C | 0.39 | < 0.001 | - | ||
| Steatosis grade | 0.76 | < 0.001 | 37.12 | 21.63-52.60 | < 0.001 |
Parameters which were significantly associated with CAP were entered into a multivariate linear regression model (stepwise methods). After adjustment, only steatosis grade (OR = 37.12; 95%CI: 21.63-52.60, P < 0.001) and BMI (OR = 6.2; 95%CI: 2.92-9.48, P < 0.001) were independent predictive factors of CAP (Table 2).
The median CAP (IQR) values in patients with S0, S1, S2 and S3 steatosis were 211 (181-240) dB/m, 270 (253-305) dB/m, 330 (302-360) dB/m, and 346 (313-363) dB/m, respectively (P < 0.001; Figure 1A). CAP values were significantly different between S0 vs S1 (P < 0.001) and S1 vs S2 (P < 0.001), but not between S2 vs S3 (P = 0.224). Moreover, the median CAP (IQR) values were 169 (148-207) dB/m, 235 (200-275) dB/m, 288 (254-340) dB/m and 331 (304-360) dB/m for patients with a BMI (kg/m2) of < 18.5, 18.5-24.9, 25-29.9 and ≥ 30, respectively (P < 0.001; Figure 1B). To remove the confounding factors of liver steatosis and aetiology, we further analyzed the CAP values with BMI values in the 63 CHB patients with 0%-5% steatosis. The Kruskal-Wallis test was used, and significant differences (P = 0.004) were found between the CAP values and the different BMI levels (Figure 1C).
The CAP (IQR) values were 271 (258-291) dB/m for NAFLD patients and 270 (247-308) dB/m for CHB patients with S1 (P = 0.670), 328 (303-361) dB/m for NAFLD patients and 331 (289-350) dB/m for CHB patients with S2 (P = 0.681), and 341 (315-361) dB/m for NAFLD patients and 349 (310-378) dB/m for CHB patients with S3 (P = 0.692). Therefore, no significant differences were found between the CAP values of NAFLD and CHB patients in each steatosis grade (Figure 1D).
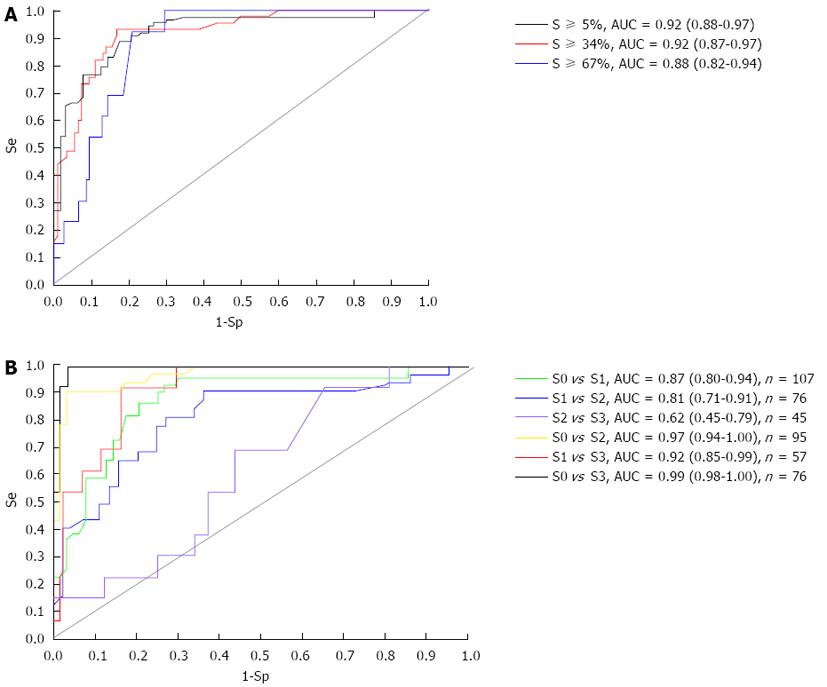
The ROC curves of CAP to differentiate between steatosis grades are displayed in Figure 2A. CAP was found to be excellent for predicting fatty liver (steatosis ≥ 5%; AUC = 0.92, 95%CI: 0.88-0.97), for the detection of steatosis ≥ 34% (AUC = 0.92, 95%CI: 0.87-0.97) and good for the detection of steatosis ≥ 67% (AUC = 0.88, 95%CI: 0.82-0.94). Using the maximum Youden index, optimal cut-off values with the sensitivity, specificity, PPV and NPV of CAP for hepatic steatosis ≥ 5%, ≥ 34% and ≥ 67% in patients were also calculated and are shown in Table 3.
| Steatosis | AUC (95%CI) | P value | Cut-off (dB/m) | Sensitivity (95%CI), % | Specificity (95%CI), % | PPV (95%CI), % | NPV (95%CI), % |
| S0 vs S1-S3 (≥ 5%) | 0.92 (0.88-0.97) | < 0.001 | 253 | 88.8 (79.9-94.2) | 82.5 (70.5-90.6) | 87.8 (78.8-93.4) | 83.9 (71.9-91.6) |
| S0-S1 vs S2-S3 (≥ 34%) | 0.92 (0.87-0.97) | < 0.000 | 285 | 93.3 (80.7-98.3) | 83.2 (74.4-89.5) | 70.0 (56.6-80.8) | 96.7 (90.1-99.2) |
| S0-S2 vs S3 (≥ 67%) | 0.88 (0.82-0.94) | < 0.001 | 310 | 92.3 (62.1-99.6) | 79.1 (71.3-85.4) | 29.3 (16.6-45.7) | 99.1 (94.4-99.9) |
ROC curves and AUCs of CAP values were also calculated between two steatosis grades to differentiate individual grades of steatosis: 1-grade difference, including S0 vs S1, S1 vs S2 and S2 vs S3; 2-grade difference, including S0 vs S2 and S1 vs S3; 3-grade difference, including S0 vs S3. All six potential pairs are shown in Figure 2B. CAP performance was excellent for differentiating between 2 or 3 grades, such as S0 vs S2 (AUC = 0.97), S1 vs S3 (AUC = 0.92) and S0 vs S3 (AUC = 0.99). However, it was poorer at differentiating between 1 grade than more than 2 grades, especially for S2 vs S3 (AUC = 0.62).
In China, it has been estimated that at least 10% of the general population is chronically infected with HBV, which is the most common cause of liver disease[20]. In recent years, with the increasing pandemic of obesity, NAFLD has now become a major cause of liver-related morbidity and mortality, with a prevalence of 15% in China[13]. Although clinical studies have found that HBV infection is associated with a lower prevalence of fatty liver, hypertriglyceridemia and metabolic syndrome[21], hepatic steatosis has been associated with Entecavir treatment failure[22]. Therefore, it is important to determine whether hepatic steatosis or steatohepatitis coexist in CHB patients.
The current “gold standard” diagnostic procedure is still LB. Due to the trauma, sampling error, complications and imperfect reproducibility of LB, its application is limited. Therefore, the development of a non-invasive quantitative measure of hepatic steatosis is necessary. The two existing methods mainly include serological and imaging methods. Serological methods such as the SteatoTest[23], the Fatty Liver Index (FLI)[24] and the Hepatic Steatosis Index (HSI)[25] combine a number of biochemical markers and/or anthropometric characteristics that have been extensively developed in the last decade to diagnose fatty liver. The M65 enzyme-linked immunosorbent assay, which detects both caspase-cleaved and uncleaved cytokeratin-18, may also differentiate patients with simple steatosis from healthy individuals[26]. However, the accuracy and diagnostic efficacy of these tests still need to be improved, thus, they are not yet recommended for use in clinical practice. The imaging methods include ultrasonography, computed tomography (CT) and magnetic resonance (MR)[27]. Ultrasonography is the most common technique, and is accepted as an initial tool for fatty liver as it is non-invasive, non-ionizing, inexpensive and widely available. However, the major weaknesses of ultrasound include high operator- and machine-dependency and the ability to detect only patients with more than 30% steatosis. CT provides an accurate and a reliable visualisation of the whole liver, enabling the diagnosis not only of diffuse, but also of focal fatty deposits. However, CT is associated with radiation exposure, which limits its use in longitudinal studies and in children. Moreover, iron accumulation plays an important role in steatohepatitis during NAFLD[28], and CT is strongly influenced by iron deposition in the liver, leading to misdiagnosis. MR, especially proton magnetic resonance spectroscopy (1H MRS), has emerged as a fast, safe, non-invasive alternative for the quantification of hepatic fat content. 1H MRS has been used in NAFLD patients with mild steatosis or advanced fibrosis, and can be performed easily without special devices[29]. However, its use is limited due to high costs, low availability and a lack of standardisation.
FibroScan® is now widely used to obtain LSMs, which relate to liver fibrosis, and has shown good results for the diagnosis of cirrhosis in chronic liver disease[30], but was incapable of assessing steatosis. In 2010, Sasso et al[10] first reported a novel attenuation parameter that was based on the ultrasonic properties of the radiofrequency back-propagated signals acquired by the FibroScan® guided by vibration-controlled transient elastography. This new parameter has the advantages of being a non-ionizing, relatively inexpensive, painless, and operator and machine independent method. The overall intraclass correlation coefficient for the determination of hepatic steatosis by means of CAP in HIV and/or hepatitis virus infection was 0.84 (95%CI: 0.77-0.88)[31], suggesting that CAP measurement represents an observer-independent method.
In the first publication[10], CAP values were significantly correlated with steatosis grade (Spearman correlation ρ = 0.81, P < 10-16), which were similar to our results (Ρ = 0.76, P < 0.001). Our study identified median (IQR) CAP values for the S0, S1, S2 and S3 steatosis grades to be 211 (181-240) dB/m, 270 (253-305) dB/m, 330 (302-360) dB/m, and 346 (313-363) dB/m, respectively. CAP values were significantly different between S0 vs S1 and S1 vs S2 groups, but were not significantly different between the S2 vs S3 groups. Combined with other clinical trial results[9,12,32], we believe that CAP values can identify more than 5% steatosis, but are less effective in identifying close steatosis grades, especially for moderate to severe steatosis. The reason for this may be that BMI was increasing together with the fat content in the liver, and the discriminability of the CAP measurement using the M probe was limited by BMI.
Inflammation and biochemical indicators such as ALT and bilirubin have been demonstrated to influence LSM measurement, and different liver disease aetiologies may lead to different cut-off values. Therefore, multivariate linear regression was used to evaluate which parameters are related to CAP values. After adjustment, only steatosis grade and BMI were independent predictive factors of CAP values. In this work, CAP values did not appear to be influenced by inflammation, fibrosis or aetiology. These results were similar to those obtained in other studies[9]. It was previously found that LSM values are significantly correlated with BMI, especially in NAFLD patients[19]. In our study, BMI was also independently associated with CAP, and elevated BMI may influence the accuracy of CAP values in detecting liver steatosis. To control the confounding factors, we determined the impact of BMI in 63 CHB patients with steatosis less than 5%. Significant differences were still found, especially between the normal and overweight groups. In other studies, the optimal cut-off of CAP values for significant steatosis (≥ 10%) in patients with BMIs ≥ 28 kg/m2 [12] was higher than in patients with low BMIs[10] (283 dB/m vs 237.7 dB/m). The reason for this phenomenon might be that subcutaneous fat is involved in the measurement using the M probe in patients with a skin-liver capsule depth distance greater than 25 mm, strengthening the degree of attenuation[33]. It is already known that subcutaneous adipose tissue may lead to overestimation of liver stiffness. Therefore, the choice of probe during an examination should depend on the distance between the probe and the liver. Compared with the M probe, the XL probe reduces TE failure and facilitates reliable LSM in NAFLD patients[34]. However, CAP measurements are currently available only using the M probe and under development for the XL probe.
In the present study which involved a Chinese population, we suggest CAP cut-off values of 253 dB/m, 285 dB/m, and 310 dB/m should be used for the diagnosis of S ≥ 5% (S0 vs S1-S3), S ≥ 34% (S0-S1 vs S2-S3), and S ≥ 67% (S0-S2 vs S3), respectively. The ROC curves and corresponding AUCs were also calculated to assess the CAP performance. The results are consistent with several other reports[8,10], suggesting that this non-invasive test has a high accuracy for the detection of steatosis. The performance of CAP was also excellent for the detection of significant steatosis (≥ 10%) with AUCs of 0.84[11], 0.81[12] or 0.80[8], higher than those for HSI (0.65),FLI (0.72) and M65 (0.68)[12,26]. CAP provides a high ability to identify steatosis (more than 5%) compared with unenhanced CT or ultrasonography (more than 30%). CAP performance between two steatosis grades for differentiating individual grades of steatosis was also excellent; however, it showed poor accuracy in differentiating between adjacent grades of steatosis, especially for S2 vs S3 (AUC = 0.62).
Our study has some limitations. First, liver biopsies were used as the gold standard and interpreted by two experienced hepatopathologists, however, biopsies are not the best reference for liver steatosis measurement. Determination of the percentage of hepatocytes containing lipid vesicles is highly subjective, and steatosis grading corresponds only to a semiquantitative scale. Therefore, objective tools such as MRS together with liver biopsies may be better for the assessment of hepatic steatosis. Second, our sample size was limited due to the difficulty in obtaining liver biopsies from NAFLD patients and valid CAP measurements from obese patients. Thus, further studies including large cohorts of patients are needed to validate our preliminary data in patients with NAFLD or CHB. Third, this clinical trial was performed only in the Chinese Han population, and more tests should be carried out between different ethnicities.
In conclusion, our study shows that the novel CAP appears to be a promising tool for the non-invasive detection and quantification of hepatic steatosis in patients with either NAFLD or CHB. CAP values can be evaluated simultaneously with LSM to assess hepatic fibrosis. This new parameter has the advantages of being a simple, non-invasive, inexpensive, painless, and operator and machine independent method, and displays good application prospects. More than 5% steatosis is an acceptable pathological diagnosis of fatty liver and this clinical trial has shown that CAP values can be identified. Therefore, we believe that CAP may be an alternative method to ultrasonography for epidemiological investigations of fatty liver. However, this new method is limited by BMI, and CAP values in obese patients may be overestimated. The CAP method requires further evaluation in studies using adequate references, including studies using large samples of patients with different aetiologies.
Although liver biopsy is regarded as the gold standard to assess hepatic steatosis, its use has several limitations. Recently, a novel non-invasive tool based on ultrasound attenuation was developed to assess liver steatosis. The aim of this study was to evaluate the performance of controlled attenuation parameter (CAP) measurements in assessing steatosis, in a cohort of consecutive nonalcoholic fatty liver disease (NAFLD)/chronic hepatitis B (CHB) patients in China, using liver biopsy as the reference.
In 2010, a novel attenuation parameter was reported which was based on the ultrasonic properties of the radiofrequency back-propagated signals acquired by the FibroScan® guided by vibration-controlled transient elastography. In the first publication, CAP values were significantly correlated with steatosis grade. In their clinical trial, they believe that CAP can identify more than 5% steatosis, but is less effective in identifying close steatosis grades, and this new method is limited by BMI.
This study shows that the novel CAP appears to be a promising tool for the non-invasive detection and quantification of hepatic steatosis in patients with either NAFLD or CHB. They firstly reported in the Chinese population, and suggested that CAP cut-off values of 253 dB/m, 285 dB/m, and 310 dB/m should be used for the diagnosis of S ≥ 5% (S0 vs S1-S3), S ≥ 34% (S0-S1 vs S2-S3), and S ≥ 67% (S0-S2 vs S3), respectively.
Ultrasonography is the most common technique, and is accepted as an initial tool for fatty liver. However, the major weaknesses of ultrasound include high operator- and machine-dependency and the ability to detect only patients with more than 30% steatosis. CAP can identify more than 5% steatosis and assess steatosis quantitatively and dynamically. Therefore, they believe that CAP may be an alternative method to ultrasonography for epidemiological investigations of fatty liver.
FibroScan® is now widely used to obtain LSMs, which relate to liver fibrosis, and has shown good results for the diagnosis of cirrhosis in chronic liver disease. The evaluation of ultrasound attenuation has been implemented with the FibroScan® using a novel proprietary algorithm called the CAP. The new FibroScan® 502 equipped with the M probe was used to capture both CAP and LSM values simultaneously.
A multi-center prospective cohort study involving patients from three Chinese liver centers who had undergone percutaneous liver biopsy shows that the novel CAP appears to be a promising tool for the non-invasive detection and quantification of hepatic steatosis in patients with either NAFLD or chronic hepatitis B. CAP values can be evaluated simultaneously with LSM. Though CAP was limited by BMI, it may be an alternative method to ultrasonography for epidemiological research of fatty liver. On the other hand, the CAP method requires further in-depth studies, also comparative analyses, involving similar observations from other research centers.
P- Reviewers: Kim SR, Lotowska MES S- Editor: Ma YJ L- Editor: Wang TQ E- Editor: Wang CH
| 1. | Sasso M, Miette V, Sandrin L, Beaugrand M. The controlled attenuation parameter (CAP): a novel tool for the non-invasive evaluation of steatosis using Fibroscan. Clin Res Hepatol Gastroenterol. 2012;36:13-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 217] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 2. | Blachier M, Leleu H, Peck-Radosavljevic M, Valla DC, Roudot-Thoraval F. The burden of liver disease in Europe: a review of available epidemiological data. J Hepatol. 2013;58:593-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 908] [Article Influence: 75.7] [Reference Citation Analysis (0)] |
| 3. | Leandro G, Mangia A, Hui J, Fabris P, Rubbia-Brandt L, Colloredo G, Adinolfi LE, Asselah T, Jonsson JR, Smedile A. Relationship between steatosis, inflammation, and fibrosis in chronic hepatitis C: a meta-analysis of individual patient data. Gastroenterology. 2006;130:1636-1642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 408] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 4. | Bellentani S, Saccoccio G, Masutti F, Crocè LS, Brandi G, Sasso F, Cristanini G, Tiribelli C. Prevalence of and risk factors for hepatic steatosis in Northern Italy. Ann Intern Med. 2000;132:112-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 842] [Cited by in RCA: 863] [Article Influence: 34.5] [Reference Citation Analysis (0)] |
| 5. | Farrell GC, Larter CZ. Nonalcoholic fatty liver disease: from steatosis to cirrhosis. Hepatology. 2006;43:S99-S112. [PubMed] |
| 6. | Powell EE, Jonsson JR, Clouston AD. Steatosis: co-factor in other liver diseases. Hepatology. 2005;42:5-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 257] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 7. | Grant A, Neuberger J. Guidelines on the use of liver biopsy in clinical practice. British Society of Gastroenterology. Gut. 1999;45 Suppl 4:IV1-IV11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 186] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 8. | Sasso M, Tengher-Barna I, Ziol M, Miette V, Fournier C, Sandrin L, Poupon R, Cardoso AC, Marcellin P, Douvin C. Novel controlled attenuation parameter for noninvasive assessment of steatosis using Fibroscan(®): validation in chronic hepatitis C. J Viral Hepat. 2012;19:244-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 157] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 9. | Kumar M, Rastogi A, Singh T, Behari C, Gupta E, Garg H, Kumar R, Bhatia V, Sarin SK. Controlled attenuation parameter for non-invasive assessment of hepatic steatosis: does etiology affect performance? J Gastroenterol Hepatol. 2013;28:1194-1201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 79] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 10. | Sasso M, Beaugrand M, de Ledinghen V, Douvin C, Marcellin P, Poupon R, Sandrin L, Miette V. Controlled attenuation parameter (CAP): a novel VCTE™ guided ultrasonic attenuation measurement for the evaluation of hepatic steatosis: preliminary study and validation in a cohort of patients with chronic liver disease from various causes. Ultrasound Med Biol. 2010;36:1825-1835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 564] [Cited by in RCA: 635] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 11. | de Lédinghen V, Vergniol J, Foucher J, Merrouche W, le Bail B. Non-invasive diagnosis of liver steatosis using controlled attenuation parameter (CAP) and transient elastography. Liver Int. 2012;32:911-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 267] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 12. | Myers RP, Pollett A, Kirsch R, Pomier-Layrargues G, Beaton M, Levstik M, Duarte-Rojo A, Wong D, Crotty P, Elkashab M. Controlled Attenuation Parameter (CAP): a noninvasive method for the detection of hepatic steatosis based on transient elastography. Liver Int. 2012;32:902-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 262] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 13. | Fan JG, Farrell GC. Epidemiology of non-alcoholic fatty liver disease in China. J Hepatol. 2009;50:204-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 437] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 14. | Fan JG, Jia JD, Li YM, Wang BY, Lu LG, Shi JP, Chan LY. Guidelines for the diagnosis and management of nonalcoholic fatty liver disease: update 2010: (published in Chinese on Chinese Journal of Hepatology 2010; 18: 163-166). J Dig Dis. 2011;12:38-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 224] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 15. | Ahn SH, Chan HL, Chen PJ, Cheng J, Goenka MK, Hou J, Lim SG, Omata M, Piratvisuth T, Xie Q. Chronic hepatitis B: whom to treat and for how long? Propositions, challenges, and future directions. Hepatol Int. 2010;4:386-395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 16. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6807] [Cited by in RCA: 8228] [Article Influence: 411.4] [Reference Citation Analysis (5)] |
| 17. | Sanyal AJ, Brunt EM, Kleiner DE, Kowdley KV, Chalasani N, Lavine JE, Ratziu V, McCullough A. Endpoints and clinical trial design for nonalcoholic steatohepatitis. Hepatology. 2011;54:344-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 573] [Cited by in RCA: 581] [Article Influence: 41.5] [Reference Citation Analysis (0)] |
| 18. | Bedossa P, Poynard T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology. 1996;24:289-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2860] [Cited by in RCA: 3081] [Article Influence: 106.2] [Reference Citation Analysis (0)] |
| 19. | Gaia S, Carenzi S, Barilli AL, Bugianesi E, Smedile A, Brunello F, Marzano A, Rizzetto M. Reliability of transient elastography for the detection of fibrosis in non-alcoholic fatty liver disease and chronic viral hepatitis. J Hepatol. 2011;54:64-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 201] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 20. | Williams R. Global challenges in liver disease. Hepatology. 2006;44:521-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 546] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 21. | Wong VW, Wong GL, Chu WC, Chim AM, Ong A, Yeung DK, Yiu KK, Chu SH, Chan HY, Woo J. Hepatitis B virus infection and fatty liver in the general population. J Hepatol. 2012;56:533-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 194] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 22. | Jin X, Chen YP, Yang YD, Li YM, Zheng L, Xu CQ. Association between hepatic steatosis and entecavir treatment failure in Chinese patients with chronic hepatitis B. PLoS One. 2012;7:e34198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 23. | Poynard T, Ratziu V, Naveau S, Thabut D, Charlotte F, Messous D, Capron D, Abella A, Massard J, Ngo Y. The diagnostic value of biomarkers (SteatoTest) for the prediction of liver steatosis. Comp Hepatol. 2005;4:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 227] [Cited by in RCA: 253] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 24. | Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, Castiglione A, Tiribelli C. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1238] [Cited by in RCA: 2029] [Article Influence: 106.8] [Reference Citation Analysis (0)] |
| 25. | Lee JH, Kim D, Kim HJ, Lee CH, Yang JI, Kim W, Kim YJ, Yoon JH, Cho SH, Sung MW. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis. 2010;42:503-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1102] [Cited by in RCA: 1062] [Article Influence: 70.8] [Reference Citation Analysis (0)] |
| 26. | Joka D, Wahl K, Moeller S, Schlue J, Vaske B, Bahr MJ, Manns MP, Schulze-Osthoff K, Bantel H. Prospective biopsy-controlled evaluation of cell death biomarkers for prediction of liver fibrosis and nonalcoholic steatohepatitis. Hepatology. 2012;55:455-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 136] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 27. | Schwenzer NF, Springer F, Schraml C, Stefan N, Machann J, Schick F. Non-invasive assessment and quantification of liver steatosis by ultrasound, computed tomography and magnetic resonance. J Hepatol. 2009;51:433-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 558] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 28. | Corradini E, Pietrangelo A. Iron and steatohepatitis. J Gastroenterol Hepatol. 2012;27 Suppl 2:42-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 29. | Hatta T, Fujinaga Y, Kadoya M, Ueda H, Murayama H, Kurozumi M, Ueda K, Komatsu M, Nagaya T, Joshita S. Accurate and simple method for quantification of hepatic fat content using magnetic resonance imaging: a prospective study in biopsy-proven nonalcoholic fatty liver disease. J Gastroenterol. 2010;45:1263-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 30. | Kim BK, Fung J, Yuen MF, Kim SU. Clinical application of liver stiffness measurement using transient elastography in chronic liver disease from longitudinal perspectives. World J Gastroenterol. 2013;19:1890-1900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 65] [Cited by in RCA: 63] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 31. | Recio E, Cifuentes C, Macías J, Mira JA, Parra-Sánchez M, Rivero-Juárez A, Almeida C, Pineda JA, Neukam K. Interobserver concordance in controlled attenuation parameter measurement, a novel tool for the assessment of hepatic steatosis on the basis of transient elastography. Eur J Gastroenterol Hepatol. 2013;25:905-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 32. | Masaki K, Takaki S, Hyogo H, Kobayashi T, Fukuhara T, Naeshiro N, Honda Y, Nakahara T, Ohno A, Miyaki D. Utility of controlled attenuation parameter measurement for assessing liver steatosis in Japanese patients with chronic liver diseases. Hepatol Res. 2013;43:1182-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 33. | Audière S, Charbit M, Angelini ED, Oudry J, Sandrin L. Measurement of the skin-liver capsule distance on ultrasound RF data for 1D transient elastography. Med Image Comput Comput Assist Interv. 2010;13:34-41. [PubMed] |
| 34. | Myers RP, Pomier-Layrargues G, Kirsch R, Pollett A, Duarte-Rojo A, Wong D, Beaton M, Levstik M, Crotty P, Elkashab M. Feasibility and diagnostic performance of the FibroScan XL probe for liver stiffness measurement in overweight and obese patients. Hepatology. 2012;55:199-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 349] [Cited by in RCA: 380] [Article Influence: 29.2] [Reference Citation Analysis (0)] |