Published online Dec 21, 2013. doi: 10.3748/wjg.v19.i47.8873
Revised: November 20, 2013
Accepted: December 5, 2013
Published online: December 21, 2013
Processing time: 112 Days and 23.5 Hours
High-throughput short-read sequencing of exomes and whole cancer genomes in multiple human hepatocellular carcinoma (HCC) cohorts confirmed previously identified frequently mutated somatic genes, such as TP53, CTNNB1 and AXIN1, and identified several novel genes with moderate mutation frequencies, including ARID1A, ARID2, MLL, MLL2, MLL3, MLL4, IRF2, ATM, CDKN2A, FGF19, PIK3CA, RPS6KA3, JAK1, KEAP1, NFE2L2, C16orf62, LEPR, RAC2, and IL6ST. Functional classification of these mutated genes suggested that alterations in pathways participating in chromatin remodeling, Wnt/β-catenin signaling, JAK/STAT signaling, and oxidative stress play critical roles in HCC tumorigenesis. Nevertheless, because there are few druggable genes used in HCC therapy, the identification of new therapeutic targets through integrated genomic approaches remains an important task. Because a large amount of HCC genomic data genotyped by high density single nucleotide polymorphism arrays is deposited in the public domain, copy number alteration (CNA) analyses of these arrays is a cost-effective way to reveal target genes through profiling of recurrent and overlapping amplicons, homozygous deletions and potentially unbalanced chromosomal translocations accumulated during HCC progression. Moreover, integration of CNAs with other high-throughput genomic data, such as aberrantly coding transcriptomes and non-coding gene expression in human HCC tissues and rodent HCC models, provides lines of evidence that can be used to facilitate the identification of novel HCC target genes with the potential of improving the survival of HCC patients.
Core tip: In addition to detecting somatic mutations in cancer genomes with high-throughput short-read sequencing technologies, analysis of copy number alteration in hepatocellular carcinoma (HCC) cancer genomes genotyped by high density single nucleotide polymorphism arrays is a cost-effective approach to reveal genome-wide somatic alterations accumulated during tumorigenesis. Integration with other genomic data from HCC tissues derived from high-throughput short-read sequencing, proteomics, epigenomics and transcriptomics could provide lines of evidence to identify common and novel HCC genes for potential clinical applications.
- Citation: Gu DL, Chen YH, Shih JH, Lin CH, Jou YS, Chen CF. Target genes discovery through copy number alteration analysis in human hepatocellular carcinoma. World J Gastroenterol 2013; 19(47): 8873-8879
- URL: https://www.wjgnet.com/1007-9327/full/v19/i47/8873.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i47.8873
Human hepatocellular carcinoma (HCC) is the fifth leading cause of cancer mortality, causing an estimated half a million deaths annually[1,2]. Risk factors for developing HCC include hepatitis infection, obesity, alcoholism and consumption of aflatoxin-contaminated food. Due to the rising incidence of hepatitis C infection, HCC is one of the fastest-growing cancers in the United States and Western countries, and the incidence is expected to continue to increase[3]. Surgical resection is the most successful treatment for early stage HCC. However, fewer than 30% of HCC patients are qualified for curative resection owing to liver dysfunction and cirrhosis. Moreover, frequent tumor recurrence is observed even after curative resection.
Recent successes in cancer targeted therapy arising from the identification of somatic alterations and their specific inhibitors are associated with reduced side effects and prolonged patient survival. Many of these FDA-approved inhibitors are small molecules or monoclonal antibodies against cancer-specific tyrosine kinase mutations, including Imatinib mesylate (Gleevec) for fusion oncogene Bcr/Abl-positive chronic myelogenous leukemia[4], Gefitinib (Iressa) or Erlotinib (Tarceva) for epidermal growth factor receptor mutated non-small cell lung cancer[5] and Trastuzumab (Herceptin) for HER2/neu receptor amplified and overexpressed breast cancer patients[6]. Although no specific drug target has been identified for HCC, FDA approved the multi-kinase inhibitor sorafenib for treatment of advanced HCC, due to a favorable overall patient survival[7]. However, HCC patients receiving sorafenib showed marginal benefits, with a prolonged survival of 3-4 mo on average[8,9]. With limited improvement of HCC patient survival, identification of recurrent and altered somatic genes through integrated genomic approaches is vital to better understand HCC molecular tumorigenesis, to develop early diagnostic markers and methods, and to find additional druggable targets for the improvement of HCC management.
In HCC, many tumor suppressor genes and oncogenes were identified based on recurrent genetic lesions, including loss of TP53 (17p13)[10], RB and BRCA2 (13q)[11], and amplification of c-myc (8q24)[12] and ERBB2 (17q12-q21)[13]. Epigenetic mechanisms also contribute to HCC progression, such as CpG island hyper- methylation of p16 (INK4a) and COX2[14-16], as well as altered expression of microRNAs[17,18]. Conventional point mutation is another common mechanism to alter cancer gene functions. In HCC, frequent point mutations of TP53 and β-catenin are involved in key pathways of hepatocarcinogenesis[19,20]. Other studies have reported mutations in M6P/IGF2R[21], BRCA2[22], Smad2/4[23], HCCS1[24], PTEN[25] and Axin1[26].
Recently developed high-throughput short-read sequencing technologies were used to identify somatic mutations in HCC cancer genomes at genome-wide scales. These studies confirmed that TP53 and CTNNB1 (encoding for β-catenin) are the most frequent recurrent mutations in human HCC. In addition, moderate mutation frequencies were identified in multiple HCC cohorts for several novel genes, including epigenetic and chromatin remodeling genes (ARID1A, ARID2, MLL and MLL3) and members of a number of oncogenic pathways (RPS6KA3, JAK1 and KEAP1)[27-32]. These results suggested that aberrant pathways involved in cell cycle regulation, oxidative stress, chromatin remodeling and oncogenic signaling, such as Wnt/β-catenin, JAK/STAT and Akt/mTOR, play critical roles in the process of HCC tumorigenesis. Nevertheless, HCC remains a highly lethal cancer due to the lack of biomarkers for early diagnosis, molecular classification and efficient therapeutic interventions. Efforts to develop specific inhibitors for these aberrant pathways and reveal better therapeutic targets in HCC are urgently needed.
Copy number alterations (CNAs), distinguished from germ line transmitted copy number variations, account for some of the genetic diversity of populations, in addition to the accumulated genomic DNA changes during tumor progression. CNAs are important subclasses of somatic mutations, with aberrant chromosomal regions of amplification or deletion commonly associated with overexpressed oncogenes or loss of tumor suppressor genes, respectively[33]. With the comprehensive annotation of human genome in the last decade, the mutated cancer genes could be aberrant protein-coding and non-coding genes such as small microRNAs or long non-coding RNAs within the CNA regions[34].
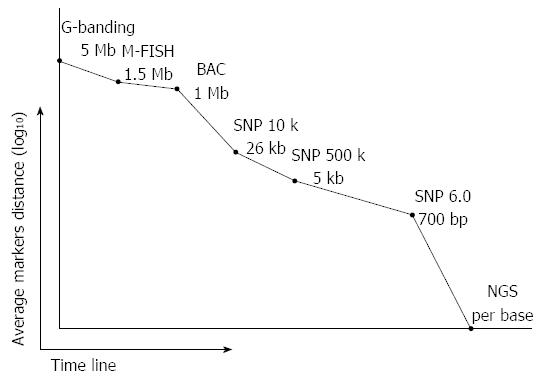
Copy number alterations in cancer cells can be detected by conventional karyotyping and chromosomal in situ hybridization technologies. To profile CNAs in cancer genomes compared to the genomes of adjacent normal cells, comparative genome hybridization (CGH) technology was used to identify copy number changes in karyotypes from breast cancer cell lines and primary bladder tumors[35]. With the availability of genomic resources (e.g., BAC clones) and array technologies (e.g., high density oligonucleotide probes), array-based CGH (aCGH) technologies not only examine genome-wide CNAs in high resolution but also allow researchers to pinpoint and profile the non-random CNAs for identification of novel aberrant cancer genes (Figure 1)[36-38]. The recently developed high-throughput short-read sequencing technology might become an alternative and effective approach to simultaneously detect CNAs and other classes of somatic mutations at the single nucleotide level[39,40]. Nevertheless, the availability of thousands of cancer genomes genotyped by high-density SNP arrays from various tumor samples and cancer cell lines at both NCBI GEO (Gene Expression Omnibus) and EBI ArrayExpress databases is a critical resource for in silico analysis of CNAs[41,42]. Moreover, integrated genomic analysis with both high-throughput short-read sequencing technology and high-density SNP genotyping arrays to comprehensively profile and validate recurrent CNAs of cancer genomes are promising approaches for the identification of novel cancer genes[40].
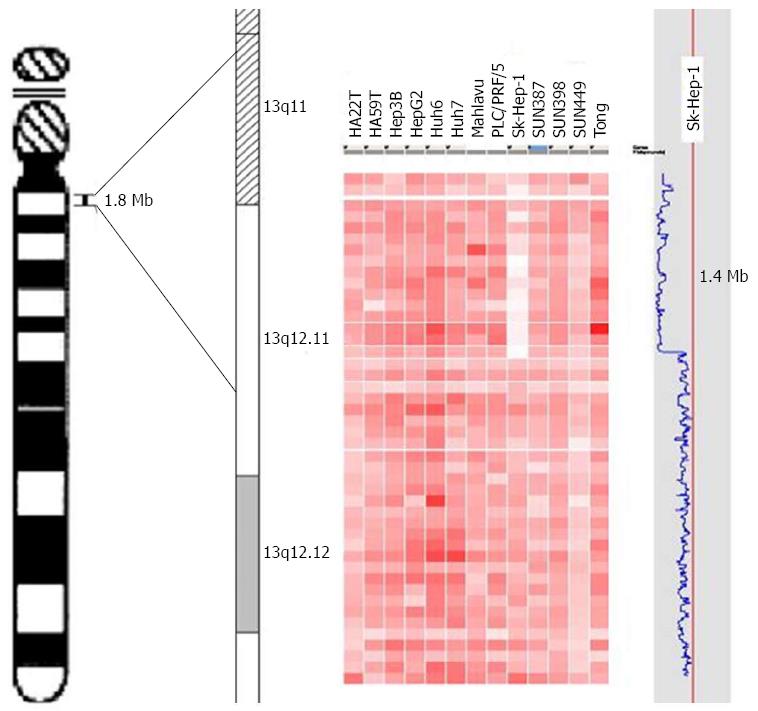
To identify novel diagnostic and therapeutic target genes, CNA analysis of cancer genomes genotyped using commercial high-density SNP arrays from your own experiments or downloaded from public domains is a powerful and cost-effective approach. First, to discover putative tumor suppressor genes, we overlapped homozygous deleted regions from multiple samples to narrow down the common deleted regions by using high-density SNP genotyping arrays. As shown in Figure 2, the homozygous deleted region at chromosome 13q12.11 in SK-hep1 cells could be refined from 1.88 to 1.46 Mb to facilitate the identification of candidate tumor suppressor genes[38,43]. Second, for the identification of candidate oncogenes in HCC, the most common approach is to integrate data from genomic experiments in order to reveal genes residing in overlapping amplicons with up-regulated gene expression. For instance, FNDC3B, SLC29A2, Ago2, IER3 and many others were identified as putative oncogenes due to their genomic DNA amplification and mRNA overexpression in HCC tissues[38,44-47]. When ectopically expressed, these putative oncogenes in HCC cells commonly show malignant phenotypes using various functional assays and facilitated tumor progression in vitro and in vivo.
Third, CNA analysis allows the identification of HCC genes with attributes of genomic DNA amplification, mRNA overexpression and recurrent point mutations, such as the putative metastatic HCC oncogene with LMCD1 mutations at E135K (in 3/48 cases) and K237R (in PLC/PRF/5 cells)[44]. When these mutations were expressed in HCC cells, HCC cell migration capability was enhanced in association with cortical actin accumulation and lamellipodial extension. Moreover, the overexpression of the LMCD1 E135K mutation in HCC cells significantly promoted systemic lung metastasis in a murine tail vein injection model. Table 1 summarizes some novel HCC genes in association with overlapping amplicons and homozygous deletions in HCC cell lines. Finally, CNA analysis detects differences in copy number (i.e., dosages), such as amplifications and deletions. Therefore, it will not reveal balanced translocation but will detect sudden dosage changes for unbalanced translocation. Using CNA analysis and high-density SNP arrays, PAX5 fusion genes were identified with a variety of partner genes, including ETV6, FOXP1, AUTS2, and C20orf112, in pediatric acute lymphoblastic leukemia (ALL)[48].
| Cytoband | Start (Mb) | End (Mb) | Known cancer genes | Novel candidates |
| Amplicons | ||||
| 1q21.2-22 | 150.07 | 151.89 | SHC1, CKS1B, ADAM15 | CREB3L4, RAB1, mir190b, S100A14 |
| 3p26.1-25.3 | 6.90 | 9.43 | LMCD1 | |
| 3q26.2-26.31 | 170.07 | 170.24 | ||
| 170.28 | 170.99 | EVI1, MDS1, TERC | ||
| 171.21 | 173.50 | FNDC3B | ||
| 5p15.33-12 | 0.40 | 45.14 | TRIO, AMACR, DAB2 | LPCAT1, SEMA5A, CDH12 |
| 7p22.2-14.3 | 4.15 | 32.10 | RAC1, ETV1, CHN2 | |
| 7p12.1-11.2 | 52.79 | 55.17 | EGFR | |
| 56.00 | 56.53 | |||
| 8p11.21 | 40.44 | 40.62 | ||
| 8q24.21 | 129.21 | 129.29 | ||
| 11q13.2-13.3 | 65.85 | 66.44 | RIN1, BRMS1 | SLC29A2 |
| 67.58 | 67.71 | |||
| 67.91 | 69.35 | LRP5, CCND1, ORAOV1 | FGF4, FGF3 | |
| 12p12.1 | 24.36 | 25.54 | BCAS1, K-ras | |
| 20q13.31 | 53.94 | 53.96 | ||
| Homozygous deletions | ||||
| 2q22.1 | 141.72 | 141.80 | LRP1B | |
| 7q21.11 | 77.96 | 78.04 | MAGI2 | |
| 9p23 | 9.42 | 9.46 | PTPRD | |
| 11.90 | 12.00 | |||
| 9p21.3 | 21.85 | 21.90 | MTAP, CDKN2A | |
| 24.27 | 24.84 | |||
| 13q12.11 | 18.98 | 20.44 | TPTE2, Tg737 | |
Integrated data derived from multiple genomic approaches could potentially avoid pitfalls of data inconsistency usual with the single genomic approach and provide lines of evidence to validate target genes embraced in the aberrant genomic loci from the level of DNA and RNA to protein. For these advantages, several user-friendly HCC databases were constructed, including OncoDB.HCC, HCCnet, dbHCCvar, CellMinerHCC, HCC-M, and EHCO[49-54]. However, only OncoDB.HCC integrated genomic alteration data to prioritize HCC cancer genes for further expression and functional validations in HCC cell lines and tissues. Nevertheless, recent international efforts at applying high-throughput short-read sequencing technologies and CNA analysis of cancer genomes in multiple cancer types, including HCC, comprehensively cataloged different types of somatic mutations and revealed genetic heterogeneity even from the same subtype of cancer. Table 2 lists common open-access integrated cancer genome databases for downloading and visualizing cancer genomic data[55,56].
| Database | Project | Website | Ref. |
| cBioPortal for cancer genome | Project provides visualization, analysis and download of large-scale cancer genomic data sets | http://www.cbioportal.org/public-portal/ | Cerami et al[57] |
| COSMIC | Catalogue of somatic mutations in cancer | http://cancer.sanger.ac.uk/cancergenome/projects/cosmic/ | Forbes et al[58] |
| ICGC | International Cancer Genome Consortium provides tools for visualizing, querying and downloading the data. | http://dcc.icgc.org/ | Joly et al[59] |
| TCGA data portal | A platform for researchers to search, download, and analyze data sets generated by TCGA | https://tcga-data.nci.nih.gov/tcga/tcgaHome2.jsp | |
| Tumorscape | High-resolution copy number data collected from multiple cancer types | http://www.broadinstitute.org/tumorscape/pages/portalHome.jsf | |
| UCSC cancer genome browser | A set of web-based tools to display, investigate and analyze cancer genomic data and associated clinical information | https://genome-cancer.ucsc.edu/proj/site/hgHeatmap/ | Goldman et al[60] |
As discussed in this review article, an integrated genomic approach is an effective and essential method of identifying novel HCC genes. With the availability of a tremendous amount of high-throughput short-read sequencing data and SNP array data from cancer genomes deposited in the public domain, integrated genomic approaches, including CNA analysis, are the most cost-effective approach for revealing HCC driver genes for improving HCC therapy.
P- Reviewers: Arezoo A, Matthews V, Yu DY S- Editor: Ma YJ L- Editor: A E- Editor: Ma S
| 1. | Bruix J, Boix L, Sala M, Llovet JM. Focus on hepatocellular carcinoma. Cancer Cell. 2004;5:215-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 455] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 2. | Bosch FX, Ribes J, Díaz M, Cléries R. Primary liver cancer: worldwide incidence and trends. Gastroenterology. 2004;127:S5-S16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1799] [Cited by in RCA: 1816] [Article Influence: 86.5] [Reference Citation Analysis (0)] |
| 3. | Benowitz S. Liver cancer biomarkers struggling to succeed. J Natl Cancer Inst. 2007;99:590-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 658] [Cited by in RCA: 624] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 5. | Pao W, Miller V, Zakowski M, Doherty J, Politi K, Sarkaria I, Singh B, Heelan R, Rusch V, Fulton L. EGF receptor gene mutations are common in lung cancers from “never smokers” and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc Natl Acad Sci USA. 2004;101:13306-13311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3303] [Cited by in RCA: 3438] [Article Influence: 163.7] [Reference Citation Analysis (0)] |
| 6. | Hudis CA. Trastuzumab--mechanism of action and use in clinical practice. N Engl J Med. 2007;357:39-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1728] [Cited by in RCA: 1922] [Article Influence: 106.8] [Reference Citation Analysis (0)] |
| 7. | Frenette C, Gish R. Targeted systemic therapies for hepatocellular carcinoma: clinical perspectives, challenges and implications. World J Gastroenterol. 2012;18:498-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 37] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 8. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10209] [Article Influence: 600.5] [Reference Citation Analysis (2)] |
| 9. | Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3854] [Cited by in RCA: 4626] [Article Influence: 272.1] [Reference Citation Analysis (0)] |
| 10. | Wang G, Zhao Y, Liu X, Wang L, Wu C, Zhang W, Liu W, Zhang P, Cong W, Zhu Y. Allelic loss and gain, but not genomic instability, as the major somatic mutation in primary hepatocellular carcinoma. Genes Chromosomes Cancer. 2001;31:221-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 11. | Knuutila S, Aalto Y, Autio K, Björkqvist AM, El-Rifai W, Hemmer S, Huhta T, Kettunen E, Kiuru-Kuhlefelt S, Larramendy ML. DNA copy number losses in human neoplasms. Am J Pathol. 1999;155:683-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 289] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 12. | Kusano N, Shiraishi K, Kubo K, Oga A, Okita K, Sasaki K. Genetic aberrations detected by comparative genomic hybridization in hepatocellular carcinomas: their relationship to clinicopathological features. Hepatology. 1999;29:1858-1862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 154] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 13. | Niketeghad F, Decker HJ, Caselmann WH, Lund P, Geissler F, Dienes HP, Schirmacher P. Frequent genomic imbalances suggest commonly altered tumour genes in human hepatocarcinogenesis. Br J Cancer. 2001;85:697-704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 50] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Matsuda Y, Ichida T, Matsuzawa J, Sugimura K, Asakura H. p16(INK4) is inactivated by extensive CpG methylation in human hepatocellular carcinoma. Gastroenterology. 1999;116:394-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 144] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 15. | Murata H, Tsuji S, Tsujii M, Sakaguchi Y, Fu HY, Kawano S, Hori M. Promoter hypermethylation silences cyclooxygenase-2 (Cox-2) and regulates growth of human hepatocellular carcinoma cells. Lab Invest. 2004;84:1050-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Breuhahn K, Gores G, Schirmacher P. Strategies for hepatocellular carcinoma therapy and diagnostics: lessons learned from high throughput and profiling approaches. Hepatology. 2011;53:2112-2121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 17. | Giordano S, Columbano A. MicroRNAs: new tools for diagnosis, prognosis, and therapy in hepatocellular carcinoma? Hepatology. 2013;57:840-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 296] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 18. | Khare S, Zhang Q, Ibdah JA. Epigenetics of hepatocellular carcinoma: role of microRNA. World J Gastroenterol. 2013;19:5439-5445. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 19. | Ozturk M. p53 mutation in hepatocellular carcinoma after aflatoxin exposure. Lancet. 1991;338:1356-1359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 299] [Cited by in RCA: 271] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 20. | de La Coste A, Romagnolo B, Billuart P, Renard CA, Buendia MA, Soubrane O, Fabre M, Chelly J, Beldjord C, Kahn A. Somatic mutations of the beta-catenin gene are frequent in mouse and human hepatocellular carcinomas. Proc Natl Acad Sci USA. 1998;95:8847-8851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 835] [Cited by in RCA: 840] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 21. | De Souza AT, Hankins GR, Washington MK, Orton TC, Jirtle RL. M6P/IGF2R gene is mutated in human hepatocellular carcinomas with loss of heterozygosity. Nat Genet. 1995;11:447-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 257] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 22. | Katagiri T, Nakamura Y, Miki Y. Mutations in the BRCA2 gene in hepatocellular carcinomas. Cancer Res. 1996;56:4575-4577. [PubMed] |
| 23. | Yakicier MC, Irmak MB, Romano A, Kew M, Ozturk M. Smad2 and Smad4 gene mutations in hepatocellular carcinoma. Oncogene. 1999;18:4879-4883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 117] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 24. | Zhao X, Li J, He Y, Lan F, Fu L, Guo J, Zhao R, Ye Y, He M, Chong W. A novel growth suppressor gene on chromosome 17p13.3 with a high frequency of mutation in human hepatocellular carcinoma. Cancer Res. 2001;61:7383-7387. [PubMed] |
| 25. | Yao YJ, Ping XL, Zhang H, Chen FF, Lee PK, Ahsan H, Chen CJ, Lee PH, Peacocke M, Santella RM. PTEN/MMAC1 mutations in hepatocellular carcinomas. Oncogene. 1999;18:3181-3185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 85] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Satoh S, Daigo Y, Furukawa Y, Kato T, Miwa N, Nishiwaki T, Kawasoe T, Ishiguro H, Fujita M, Tokino T. AXIN1 mutations in hepatocellular carcinomas, and growth suppression in cancer cells by virus-mediated transfer of AXIN1. Nat Genet. 2000;24:245-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 703] [Cited by in RCA: 721] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 27. | Li M, Zhao H, Zhang X, Wood LD, Anders RA, Choti MA, Pawlik TM, Daniel HD, Kannangai R, Offerhaus GJ. Inactivating mutations of the chromatin remodeling gene ARID2 in hepatocellular carcinoma. Nat Genet. 2011;43:828-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 343] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 28. | Guichard C, Amaddeo G, Imbeaud S, Ladeiro Y, Pelletier L, Maad IB, Calderaro J, Bioulac-Sage P, Letexier M, Degos F. Integrated analysis of somatic mutations and focal copy-number changes identifies key genes and pathways in hepatocellular carcinoma. Nat Genet. 2012;44:694-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 962] [Cited by in RCA: 1146] [Article Influence: 88.2] [Reference Citation Analysis (0)] |
| 29. | Fujimoto A, Totoki Y, Abe T, Boroevich KA, Hosoda F, Nguyen HH, Aoki M, Hosono N, Kubo M, Miya F. Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat Genet. 2012;44:760-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 631] [Cited by in RCA: 711] [Article Influence: 54.7] [Reference Citation Analysis (0)] |
| 30. | Huang J, Deng Q, Wang Q, Li KY, Dai JH, Li N, Zhu ZD, Zhou B, Liu XY, Liu RF. Exome sequencing of hepatitis B virus-associated hepatocellular carcinoma. Nat Genet. 2012;44:1117-1121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 307] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 31. | Cleary SP, Jeck WR, Zhao X, Chen K, Selitsky SR, Savich GL, Tan TX, Wu MC, Getz G, Lawrence MS. Identification of driver genes in hepatocellular carcinoma by exome sequencing. Hepatology. 2013;May 31; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 240] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 32. | Kan Z, Zheng H, Liu X, Li S, Barber TD, Gong Z, Gao H, Hao K, Willard MD, Xu J. Whole-genome sequencing identifies recurrent mutations in hepatocellular carcinoma. Genome Res. 2013;23:1422-1433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 355] [Cited by in RCA: 411] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 33. | Pinkel D, Albertson DG. Array comparative genomic hybridization and its applications in cancer. Nat Genet. 2005;37 Suppl:S11-S17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 374] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 34. | Djebali S, Davis CA, Merkel A, Dobin A, Lassmann T, Mortazavi A, Tanzer A, Lagarde J, Lin W, Schlesinger F. Landscape of transcription in human cells. Nature. 2012;489:101-108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3924] [Cited by in RCA: 3963] [Article Influence: 304.8] [Reference Citation Analysis (0)] |
| 35. | Kallioniemi A, Kallioniemi OP, Sudar D, Rutovitz D, Gray JW, Waldman F, Pinkel D. Comparative genomic hybridization for molecular cytogenetic analysis of solid tumors. Science. 1992;258:818-821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2226] [Cited by in RCA: 2049] [Article Influence: 62.1] [Reference Citation Analysis (0)] |
| 36. | Solinas-Toldo S, Lampel S, Stilgenbauer S, Nickolenko J, Benner A, Döhner H, Cremer T, Lichter P. Matrix-based comparative genomic hybridization: biochips to screen for genomic imbalances. Genes Chromosomes Cancer. 1997;20:399-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 37. | Lin M, Wei LJ, Sellers WR, Lieberfarb M, Wong WH, Li C. dChipSNP: significance curve and clustering of SNP-array-based loss-of-heterozygosity data. Bioinformatics. 2004;20:1233-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 301] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 38. | Chen CF, Hsu EC, Lin KT, Tu PH, Chang HW, Lin CH, Chen YJ, Gu DL, Lin CH, Wu JY. Overlapping high-resolution copy number alterations in cancer genomes identified putative cancer genes in hepatocellular carcinoma. Hepatology. 2010;52:1690-1701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 39. | Shendure J, Ji H. Next-generation DNA sequencing. Nat Biotechnol. 2008;26:1135-1145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2905] [Cited by in RCA: 2465] [Article Influence: 145.0] [Reference Citation Analysis (0)] |
| 40. | Stratton MR, Campbell PJ, Futreal PA. The cancer genome. Nature. 2009;458:719-724. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2798] [Cited by in RCA: 2397] [Article Influence: 149.8] [Reference Citation Analysis (0)] |
| 41. | Brazma A, Parkinson H, Sarkans U, Shojatalab M, Vilo J, Abeygunawardena N, Holloway E, Kapushesky M, Kemmeren P, Lara GG. ArrayExpress--a public repository for microarray gene expression data at the EBI. Nucleic Acids Res. 2003;31:68-71. [PubMed] |
| 42. | Barrett T, Suzek TO, Troup DB, Wilhite SE, Ngau WC, Ledoux P, Rudnev D, Lash AE, Fujibuchi W, Edgar R. NCBI GEO: mining millions of expression profiles--database and tools. Nucleic Acids Res. 2005;33:D562-D566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 748] [Cited by in RCA: 792] [Article Influence: 39.6] [Reference Citation Analysis (0)] |
| 43. | Chen CF, Yeh SH, Chen DS, Chen PJ, Jou YS. Molecular genetic evidence supporting a novel human hepatocellular carcinoma tumor suppressor locus at 13q12.11. Genes Chromosomes Cancer. 2005;44:320-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 44. | Chang CY, Lin SC, Su WH, Ho CM, Jou YS. Somatic LMCD1 mutations promoted cell migration and tumor metastasis in hepatocellular carcinoma. Oncogene. 2012;31:2640-2652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 45. | Cheng N, Li Y, Han ZG. Argonaute2 promotes tumor metastasis by way of up-regulating focal adhesion kinase expression in hepatocellular carcinoma. Hepatology. 2013;57:1906-1918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 46. | Wang K, Lim HY, Shi S, Lee J, Deng S, Xie T, Zhu Z, Wang Y, Pocalyko D, Yang WJ. Genomic landscape of copy number aberrations enables the identification of oncogenic drivers in hepatocellular carcinoma. Hepatology. 2013;58:706-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 153] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 47. | Kwon SM, Kim DS, Won NH, Park SJ, Chwae YJ, Kang HC, Lee SH, Baik EJ, Thorgeirsson SS, Woo HG. Genomic copy number alterations with transcriptional deregulation at 6p identify an aggressive HCC phenotype. Carcinogenesis. 2013;34:1543-1550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 48. | Kawamata N, Ogawa S, Zimmermann M, Niebuhr B, Stocking C, Sanada M, Hemminki K, Yamatomo G, Nannya Y, Koehler R. Cloning of genes involved in chromosomal translocations by high-resolution single nucleotide polymorphism genomic microarray. Proc Natl Acad Sci USA. 2008;105:11921-11926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 49. | Liang RC, Neo JC, Lo SL, Tan GS, Seow TK, Chung MC. Proteome database of hepatocellular carcinoma. J Chromatogr B Analyt Technol Biomed Life Sci. 2002;771:303-328. [PubMed] |
| 50. | Su WH, Chao CC, Yeh SH, Chen DS, Chen PJ, Jou YS. OncoDB.HCC: an integrated oncogenomic database of hepatocellular carcinoma revealed aberrant cancer target genes and loci. Nucleic Acids Res. 2007;35:D727-D731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 71] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 51. | Hsu CN, Lai JM, Liu CH, Tseng HH, Lin CY, Lin KT, Yeh HH, Sung TY, Hsu WL, Su LJ. Detection of the inferred interaction network in hepatocellular carcinoma from EHCO (Encyclopedia of Hepatocellular Carcinoma genes Online). BMC Bioinformatics. 2007;8:66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 52. | He B, Qiu X, Li P, Wang L, Lv Q, Shi T. HCCNet: an integrated network database of hepatocellular carcinoma. Cell Res. 2010;20:732-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 53. | Yu XJ, Fang F, Tang CL, Yao L, Yu L, Yu L. dbHCCvar: a comprehensive database of human genetic variations in hepatocellular carcinoma. Hum Mutat. 2011;32:E2308-E2316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 54. | Staib F, Krupp M, Maass T, Itzel T, Weinmann A, Lee JS, Schmidt B, Müller M, Thorgeirsson SS, Galle PR. CellMinerHCC: a microarray-based expression database for hepatocellular carcinoma cell lines. Liver Int. 2013;Jul 31; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 55. | Schroeder MP, Gonzalez-Perez A, Lopez-Bigas N. Visualizing multidimensional cancer genomics data. Genome Med. 2013;5:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 115] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 56. | Su G, Burant CF, Beecher CW, Athey BD, Meng F. Integrated metabolome and transcriptome analysis of the NCI60 dataset. BMC Bioinformatics. 2011;12 Suppl 1:S36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 57. | Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2:401-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9144] [Cited by in RCA: 12625] [Article Influence: 971.2] [Reference Citation Analysis (0)] |
| 58. | Forbes SA, Tang G, Bindal N, Bamford S, Dawson E, Cole C, Kok CY, Jia M, Ewing R, Menzies A. COSMIC (the Catalogue of Somatic Mutations in Cancer): a resource to investigate acquired mutations in human cancer. Nucleic Acids Res. 2010;38:D652-D657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 426] [Cited by in RCA: 451] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 59. | Joly Y, Dove ES, Knoppers BM, Bobrow M, Chalmers D. Data sharing in the post-genomic world: the experience of the International Cancer Genome Consortium (ICGC) Data Access Compliance Office (DACO). PLoS Comput Biol. 2012;8:e1002549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 72] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 60. | Goldman M, Craft B, Swatloski T, Ellrott K, Cline M, Diekhans M, Ma S, Wilks C, Stuart J, Haussler D. The UCSC Cancer Genomics Browser: update 2013. Nucleic Acids Res. 2013;41:D949-D954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 132] [Article Influence: 10.2] [Reference Citation Analysis (0)] |