Published online Aug 21, 2013. doi: 10.3748/wjg.v19.i31.5035
Revised: March 20, 2013
Accepted: May 16, 2013
Published online: August 21, 2013
Processing time: 178 Days and 23.3 Hours
Bleeding from esophageal varices is a life threatening complication of portal hypertension. Primary prevention of bleeding in patients at risk for a first bleeding episode is therefore a major goal. Medical prophylaxis consists of non-selective beta-blockers like propranolol or carvedilol. Variceal endoscopic band ligation is equally effective but procedure related morbidity is a drawback of the method. Therapy of acute bleeding is based on three strategies: vasopressor drugs like terlipressin, antibiotics and endoscopic therapy. In refractory bleeding, self-expandable stents offer an option for bridging to definite treatments like transjugular intrahepatic portosystemic shunt (TIPS). Treatment of bleeding from gastric varices depends on vasopressor drugs and on injection of varices with cyanoacrylate. Strategies for primary or secondary prevention are based on non-selective beta-blockers but data from large clinical trials is lacking. Therapy of refractory bleeding relies on shunt-procedures like TIPS. Bleeding from ectopic varices, portal hypertensive gastropathy and gastric antral vascular ectasia-syndrome is less common. Possible medical and endoscopic treatment options are discussed.
Core tip: Gastrointestinal bleeding is a life threatening complication of portal hypertension. Primary prevention of bleeding in patients at risk for a first bleeding episode is therefore a major goal. The article gives a concise overview of possible bleeding sites in patients with portal hypertension. The diagnosis, prevention, therapy of acute bleeding and secondary prophylaxis of bleeding from esophageal and gastric varices, portal hypertensive gastropathy gastric antral vascular ectasia and ectopic varices are discussed.
- Citation: Biecker E. Portal hypertension and gastrointestinal bleeding: Diagnosis, prevention and management. World J Gastroenterol 2013; 19(31): 5035-5050
- URL: https://www.wjgnet.com/1007-9327/full/v19/i31/5035.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i31.5035
One of the major complications of portal hypertension is bleeding from esophageal varices. Bleeding from gastric or duodenal varices as well as bleeding from colonic varices or from portal hypertensive gastropathy is less common.
A lot of studies investigating prophylaxis and therapy of bleeding in portal hypertension have been published in the last years. This paper gives a concise overview of the current knowledge.
Primary prophylaxis of bleeding from esophageal varices is defined as a therapeutic intervention that aims at the prevention of the first variceal hemorrhage.
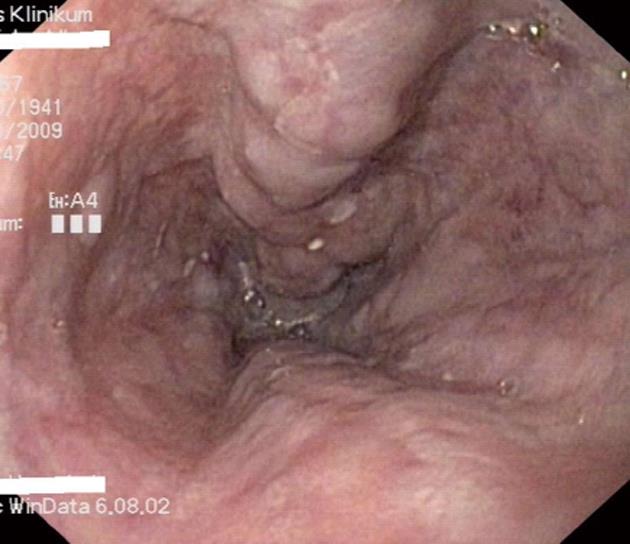
At the time of the first diagnosis, about half of the patients with liver cirrhosis have esophageal varices (Figure 1)[1,2]. During the course of the disease about 90% of the patients develop esophageal varices. Variceal hemorrhage still carries a significant mortality of 7%-15%[3-5]. The identification and prophylactic treatment of patients at risk for esophageal bleeding is therefore mandatory[6].
Risk factors for variceal bleeding are the diameter of the varix, presence of red wale signs and an impaired liver function[7-10]. Hemodynamic studies point at a close association of the hepatic venous pressure gradient (HVPG) and the bleeding risk[9].
Every patient with newly diagnosed liver cirrhosis should underwent upper endoscopy for screening of esophageal and/or gastric varices[6]. In patients with esophageal varices with a diameter of more than 5 mm, prophylactic treatment should be initiated.
Prophylactic treatment is not necessary when only small varices (diameter below 5 mm) are present. Nevertheless, endoscopic follow-up is mandatory[6]. The overall incidence of esophageal varices is 5% per year[11,12]. Esophageal varices tend to increase in size in a linear fashion. One study including 258 patients with small varices and without a history of variceal bleeding found an increase in variceal size in 21%, 45% and 66% of the patients after 1.5, 3 and 4.5 years, respectively[13]. However, it has to be kept in mind that the course of the underlying liver disease is a major determinant of variceal progression[7,13]. The actual recommendation for surveillance in patients with compensated liver disease and small varices at the screening endoscopy is a follow-up examination after 1-2 years[6,14]. If the screening endoscopy showed no varices, a follow-up examination after 2-3 years is sufficient in patients with compensated liver disease[6,13,14].
Non-selective beta-blockers cause vasoconstriction of the splanchnic circulation by β2-receptor inhibition and decrease cardiac output by β1-receptor blockade. This leads to a decrease in portal venous inflow and thereby lowers portal pressure.
Beta-blocker therapy is not effective in preventing gastro-esophageal varices in patients with cirrhosis[15]. There is only one study that showed that prophylaxis with a non-selective beta-blocker is effective in preventing the enlargement of small varices[16]. Patients with varices at risk of bleeding (diameter > 5 mm, presence of red-color-signs) should receive prophylactic treatment (see below), since the risk of bleeding is 30%-35% in two years. Effective prophylactic treatment reduces the risk of bleeding by about 50%[17].
A major drawback of beta-blocker therapy is that not all patients respond to beta-blockers with a reduction of the HVPG[18]. Clinical studies have shown that at most 50% of beta-blocker treated patients achieved a reduction of the HVPG below 12 mmHg or > 20% from baseline levels[18]. However, other effects of beta-blocker therapy besides the reduction of HVPG like a decrease in azygos blood flow or a decrease in bacterial translocation from the gut[19] may play a role in the prevention of variceal hemorrhage[20].
Endoscopic sclerotherapy and shunt procedures are obsolete in primary prophylaxis. Standard modalities are drug therapy with non-selective beta-blockers and endoscopic variceal band ligation (VBL) of varices.
Non-selective beta-blockers like propranolol and nadolol were introduced for primary prophylaxis almost 30 years ago[17]. In recent years, the non-cardioselective vasodilating beta-blocker with mild intrinsic anti-α(1)-adrenergic activity carvedilol was shown to be at least as effective in lowering HVPG as propranolol[21] or nadolol plus nitrate[22] and to be as effective as VBL for primary prophylaxis of variceal bleeding[23]. A monotherapy with nitrates or a combination of beta-blockers and nitrates compared to beta-blockers alone has no benefit in primary prophylaxis[17,24]. Meta-analysis have shown a reduction of the bleeding risk by a non-selective beta-blocker of about 50%. Around 20% of patients suffer from intolerable side effects that require discontinuation of the drug. After discontinuation, the bleeding risk is not different from an untreated population. That makes an indefinite prophylactic therapy necessary[25]. The most important predictor for variceal bleeding in patients on a therapy with beta-blockers is the dose of the drug[26]. Patients should therefore receive the highest tolerated dose.
An effective alternative treatment for primary prophylaxis is endoscopic VBL[27-30]. One meta-analysis has shown, that compared with untreated controls, prophylactic VBL reduces the risks of variceal bleeding and mortality[31]. Several studies compared endoscopic VBL with propranolol for primary prophylaxis of variceal bleeding[27-30]. Only one study that is controversially discussed because of some methodological flaws found a significant benefit for endoscopic VBL[29]. The other studies found no difference between beta-blockers and VBL concerning prophylaxis of bleeding[27,29,30]. A recently published Cochrane analysis that included 19 randomized trials found a slight beneficial effect for VBL, but that effect was not present when only full published paper articles were analyzed[32]. In terms of efficacy, VBL and non-selective beta-blocker therapy are considered to be equivalent.
Because of the low costs, ease of administration as well as the absence of procedure-related mortality, non-selective beta-blockers are recommended as first-line treatment for the primary prophylaxis of esophageal variceal bleeding[17].
VBL is recommended in patients with serious side effects or intolerability of beta-blocker therapy as well as in patients with contraindications for drug therapy.
Acute variceal bleeding is defined as: (1) active bleeding from esophageal varices at the moment of endoscopy; or (2) non-bleeding varices and blood in the esophagus/stomach are present and no other source of bleeding is found[33]. Recurrent bleeding is defined as rebleeding after 24-h of clinical absence of bleeding.
Acute bleeding from esophageal varices is often a dramatic event. Most patients vomit blood but hematochezia and melena might be the only symptoms. Dependent on the amount of lost blood, patients might be hemodynamic instable and present in hemorrhagic shock. Today only 40% of patients die from exsanguinating bleeding. Most deaths are caused by complications of bleeding like liver failure, infections and hepatorenal syndrome[34,35]. Risk factors for an adverse course are the degree of liver dysfunction, creatinine, hypovolemic shock, active bleeding on endoscopy and presence of hepatocellular carcinoma[4,34-37]. Thus, the management of patients with acute variceal bleeding includes not only treatment and control of active bleeding but also the prevention of rebleeding, infections and renal failure[38].
If variceal bleeding is suspected, patients should be hemodynamically stabilized and receive medical treatment with vasopressors and antibiotic treatment[39-43]. In uncomplicated patients antibiotic therapy is done using quinolones[44]. High-risk patients with advanced liver disease (ascites, encephalopathy, jaundice, malnutrition) or previous therapy with quinolones should receive ceftriaxone[41]. Antibiotic treatment of patients with acute variceal bleeding does not only decreases mortality but also decreases the probability of rebleeding[42]. Transfusion of blood should be done with caution with a target hemoglobin level between 7 to 8 g/dL, since higher hemoglobin levels can increase portal pressure[45] and restrictive transfusion strategies are associated with better survival[46]. Patients with massive bleeding and/or patients who are somnolent should undergo endotracheal intubation and mechanical ventilation prior endoscopy to prevent aspiration pneumonia.
Available therapy options include medical and endoscopic treatment, balloon tamponade, placement of fully covered self-expandable metallic stents, transjugular intrahepatic portosystemic shunt (TIPS) and surgical shunts. Nowadays, the initial approach is a combination of vasoactive drugs, antibiotics and endoscopic therapy[47].
The aim of medical therapy is to reduce splanchnic blood flow and portal pressure. Drugs currently in use are vasopressin, somatostatin and, most important in Europe, terlipressin. Due to its short half-life, vasopressin has to be given as a continuous iv infusion. Relevant adverse effects include systemic vasoconstriction with serious implications like mesenteric or myocardial ischemia[48]. Application of vasopressin in combination with nitrates reduces the side effects associated with vasoconstriction[49,50]. Several studies have shown that the vasopressin treatment is effective in terms of bleeding control but does not affect mortality[48,51-53]. Terlipressin is a synthetic vasopressin analogue with a longer half-life and less adverse effects. Several studies have shown that terlipressin is effective in bleeding control and has a positive impact on survival[54-56]. Terlipressin achieves control of bleeding in 75%-80% and 67% of patients at 48 h and at 5 d, respectively[56,57]. It is given at a dose of 2 mg every 4 h for the first 48 h and could be continued for prevention of early rebleeding at a dose of 1 mg every 4 h for up to 5 d[57,58]. A recent study has shown a drop of serum sodium in the range of > 5 mEq/L in 67% of patients and of > 10 mEq/L in 36% of patients treated with terlipressin[59]. Therefore, serum sodium should be monitored in patients receiving terlipressin. Compared to vasopressin, terlipressin is more effective in control of esophageal bleeding[60,61] and compared to vasopressin plus nitrate[62] as well as compared to somatostatin it is comparable effective[63,64].
Somatostatin is given as an initial bolus of 250 μg followed by a 250 to 500 μg/h continuous infusion until a bleed-free period of 24 h is achieved[38]. Octreotide is a synthetic analogue of somatostatin with longer half-life. It is administered as an initial bolus of 25 μg, followed by an infusion of 25 to 50 μg/h[65]. Both, somatostatin and octreotide, have a good safety profile. Possible adverse effects include mild hyperglycemia and abdominal craps. Somatostatin is as effective as vasopressin in control of variceal bleeding; the safety profile is superior to vasopressin[66]. The combination of terlipressin and octreotide is not superior to a monotherapy with terlipressin[67].
In summary, the available data is most convincing for terlipressin, however, the direct comparison of terlipressin and octreotide revealed no superiority of terlipressin[68,69].
About 80%-90% of acute variceal bleeding episodes are successfully controlled by endoscopic therapy[70]. Nowadays, most important is endoscopic VBL, injection therapy using sclerosing agents like ethoxysklerol or cyanoacrylate is less commonly used. Ethoxysklerol is injected next to - not into - the varix. It causes local inflammation and scaring and thereby thrombosis and obliteration of the vessel. On the opposite, cyanoacrylate is injected directly into the varix, causing immediate obliteration of the vessel. Endoscopic band ligation is done using a transparent cap that is attached to the tip of the endoscope. By applying suction, the varix is then pulled into the cap and a rubber ring is thrown over the varix causing thrombosis and scaring of the vessel (Figure 2).
Before the introduction of VBL, ethoxysklerol injection was widely used in the treatment of acute esophageal variceal bleeding. Studies have shown that sclerotherapy was at least as effective as balloon tamponade[71,72]. The injection of cyanoacrylate is used as a second line therapy when VBL of variceal bleeding fails.
Endoscopic VBL was first carried out in 1988[73]. The method is now widely available and complications are - compared to sclerotherapy - less common[74]. The most frequent complications are superficial ulcerations and esophageal strictures. Bleeding after the rubber rings have been fallen off is less common. A disadvantage of the method is the impaired sight that is caused by the ligation system. Costs are - compared to sclerotherapy - higher. Mortality rates after VBL are lower as compared to sclerotherapy[75,76].
The use of balloon tamponade for the treatment of acute esophageal variceal bleeding was introduced by Sengstaken et al[77] The Minnesota-tube is a modified version with an aspiration channel above the esophageal balloon. For uncontrolled bleeding from gastric varices, the Linton-Nachlas tube is preferred[78]. In the hand of the experienced user the method allows control of bleeding in most patients[79]. A major drawback of the method is the high amount of possible serious complications like necrosis and/or rupture of the esophagus as well as aspiration pneumonia[80]. Deflating of the balloon after six hours reduces the risk of complications. Due to the serious risks, balloon tamponade should only be applied by an experienced physician under fluoroscopic control. After all, balloon tamponade is only a bridging procedure until other, definite therapy options are available.
The placement of fully covered self-expandable metal stents (SEMS) is an alternative to balloon tamponade. The SEMS is inserted over an endoscopic placed guidewire using a stent delivery device without the need of fluoroscopy[81]. SEMS controls bleeding by compression of the bleeding varices[81]. The stent can be left in place for up to two weeks and can be easily removed by endoscopy. The effectiveness in the control of refractory esophageal variceal bleeding has been shown in four case series[82-85]. The procedure is save with minor complications like esophageal ulcerations, compression of the bronchial system and stent migration into the stomach being described[82-85]. Like balloon tamponade, the procedure is reserved for patients with bleeding refractory to medical and endoscopic treatment. It does not allow definite treatment of variceal bleeding due to the high percentage of patients with rebleeding after the SEMS has been removed, but has to be considered as an effective and safe bridging procedure that allows stabilization of the patient until definite treatment is possible.
By TIPS placement a functional portacaval side-to-side shunt is established. TIPS is indicated in patients with refractory acute variceal bleeding that could not be sufficiently controlled by endoscopic and/or medical therapy and in patients with recurrent bleeding despite optimal endoscopic therapy. After TIPS insertion, bleeding is stopped in almost all of the affected patients[86-88]. The rate of recurrent bleeding after one year is 8%-18%[89-91]. However, TIPS insertion is a problem in patients with multi-organ failure and/or in patients with decompensated liver disease. In these patients, the 30-d-mortality rate is as high as 100%[86,88,92]. Disadvantages of the procedure are the risk of hepatic encephalopathy as well as TIPS dysfunction with the risk of recurrent bleeding[93,94]. A major improvement was the introduction of polytetrafluoroethylene (PTFE) covered stents. These stents have higher rates of patency over time and mortality rates are lower[95]. A recently published trial has investigated the role of early TIPS in high-risk patients[96,97]. The multicenter study including 63 patients with esophageal hemorrhage and a high risk of treatment failure (Child B with active bleeding or Child C < 14 points) demonstrated that insertion of a PTFE covered TIPS within 72 h (preferable within 24 h) compared to combined endoscopic and vasoactive drug treatment decreased rebleeding (50% patients without rebleeding in the non-TIPS vs 97% in the TIPS group) and 1-year mortality (86% survival in the TIPS vs 61% in the non-TIPS group)[96].
Surgical procedures in patients with acute or recurrent variceal bleeding are limited to a very small portion of patients in whom medical and/or endoscopic control of bleeding was not achievable and TIPS was no option because of technical or anatomical problems (e.g., complete thrombosis of the portal vein). Possible procedures are porto-systemic shunt operations[98] or staple transection of the esophagus[99]. Survival of patients who have undergone surgery is dependent on liver function but the mortality rate is as high as 80%.
In patients who survive the first episode of esophageal hemorrhage, the risk of recurrent bleeding is as high as 60% with a mortality rate of up to 33%[100]. Prevention of rebleeding is therefore a major goal in patients in whom the initial bleeding episode has been successfully controlled.
Secondary prophylaxis of variceal bleeding is defined as the prevention of rebleeding from varices.
Several studies are available that compared the non-selective beta-blockers propranolol or nadolol with no prophylaxis after initial bleeding[101-107]. Most of the studies found a reduction of the rebleeding risk as well as a reduction in mortality. Addition of nitrates further increased this positive effect[108]. Essential is a reduction of the HVPG of at least 20%, even if a reduction below 12 mmHg could not be achieved[26,109-111].
Several groups studied the effect of sclerotherapy for secondary prophylaxis of variceal bleeding[105,112-114]. The comparison of sclerotherapy to medical therapy with a non-selective beta-blocker found a benefit for patients treated with sclerotherapy in two studies[115,116] and a slight but statistically not significant benefit for beta-blocker therapy[105,117,118]. Three more studies did not find a difference between the two treatment modalities[115,116,119].
For prophylaxis of recurrent bleeding, sclerotherapy is now replaced widely by VBL. Several studies have shown the superiority of VBL over sclerotherapy[74,76,120-124].
Comparing VBL to medical therapy with non-selective beta-blockers in combination with nitrates, two studies found medical therapy to be as effective[110] or more effective[125] than VBL. In contrast, one study found VBL to be advantageous over medical therapy[126]. From the pathophysiological point of view, the combination of VBL and medical therapy is an even more promising approach for secondary prophylaxis. This has been investigated in five studies[127-131]. Whereas two studies found combination therapy to be more effective than VBL alone[127,131] two more recent studies, that compared nadolol plus nitrates with combination treatment of drugs and VBL failed to demonstrate superiority of combination treatment[128,130]. Therefore, it seems that a clear recommendation for medial treatment alone, VBL alone or combination treatment of drugs and VBL cannot be made at the moment. A reasonable approach is to perform VBL alone in patients with contraindications for beta-blocker therapy or in patients who suffer from side effects of beta-blocker therapy. Patients who tolerate drug treatment well should be placed on a combination therapy.
TIPS was compared to sclerotherapy[90,132-137] as well as to VBL[89]. In all but two studies[136,138] patients treated with TIPS had lower rates of recurrent bleeding. Three meta-analysis[139-141] summarized the available studies and found a significant lower probability of rebleeding in the TIPS treated patients. The incidence of hepatic encephalopathy was higher in the TIPS-group. A difference in mortality was not evident.
Shunt surgery has been shown to be effective in the prophylaxis of rebleeding from esophageal varices. This has been shown for non-selective as well as for selective shunts (e.g., distal spleno-renal shunt) comparing operative shunts with no therapy or endoscopic sclerotherapy[99,142-147]. As in TIPS, the most important side effect was the incidence of hepatic encephalopathy.
One study[148] compared non-covered TIPS with a small diameter prosthetic porta-caval H-shunt. Both shunts led to an adequate reduction in portal pressure, but patency rates of the operative shunts were higher over time. This led to a lower rate of rebleeding as well as to a decrease in mortality in patients with the surgical shunt. A meta-analysis compared different porto-systemic shunts (TIPS, diverse surgical shunts) with endoscopic treatment[149]. All shunts were equally effective in reducing the risk of rebleeding. The incidence of hepatic encephalopathy was higher in patients who received a shunt procedure. TIPS was complicated by a high incidence of shunt dysfunction. Comparing the different shunt procedures, there was no difference in survival.
In contrast to esophageal variceal bleeding, prevention and treatment of bleeding from gastric varices and from portal gastropathy is less well evaluated in clinical studies.
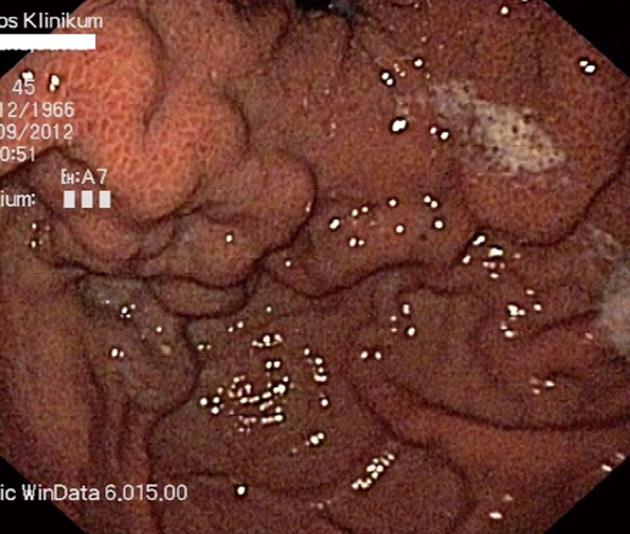
According to Sarin et al[150] gastric varices (Figure 3) are endoscopically classified as gastro-esophageal varices type I (lesser curvature), gastro-esophageal varices type II (greater curvature), isolated gastric varices type I (located in the gastric fundus) or isolated gastric varices type II (any location in the stomach except the gastric fundus).
The diagnosis of gastric varices is made by endoscopy. In case of doubt of the diagnosis, endosonography with Doppler sonography allows further differentiation. If only isolated gastric varices are present, the exclusion of portal or splenic vein thrombosis as the underlying cause is mandatory.
About on fifth of the patients with portal hypertension develop gastric varices[150]. In patients with gastrointestinal bleeding due to portal hypertension, bleeding from gastric varices is the cause in 5%-10% of patients[151]. The risk of the first bleeding from gastric varices is lower than the risk of first bleeding from esophageal varices (4% in one and 9% in three years)[152]. The risk of recurrent bleeding is dependent on the localization of the varix: isolated varices in the gastric fundus (53%) bear the highest risk of recurrent bleeding, followed by varices of the greater curvature (19%) and lesser curvature (6%)[150]. The prophylactic treatment of esophageal varices by VBL does not increase the risk of secondary gastric varices compared to propranolol[153].
Almost no data is available whether medical treatment for the primary prophylaxis of bleeding from gastric varices is effective. Pathophysiological considerations warrant the use of non-selective beta-blockers for this indication[151]. One trial including 27 patients with gastric varices studied the injection of cyanoacrylate for primary prophylaxis of bleeding from large gastric varices and found the injection of cyanoacrylate to be safe and effective in primary prophylaxis[154]. However, before recommending cyanoacrylate injection as prophylactic therapy, more studies are necessary.
Data for the treatment of acute bleeding from gastric varices is sparse. Therapy with terlipressin or somatostatin is recommend although controlled studies are lacking. The endoscopic treatment of choice is injection with cyanoacrylate[155-157]. Control of bleeding is as high as 90% and more effective than sclerotherapy or band ligation in one trial[158], whereas another study found VBL and cyanoacrylate injection equally effective in terms of control of acute bleeding but reported higher rebleeding rates in the VBL group[159]. Known complications of cyanoacrylate injection include mucosal ulcerations as well as thromboembolism. TIPS insertion is highly effective with control of bleeding in more than 90% of patients[160,161] and should be considered in patients in whom endoscopic therapy fails.
The use of non-selective beta-blockers and nitrates for prophylaxis of rebleeding was shown in one study to be not effective[162]. The comparison of cyanoacrylate with propranolol for secondary prophylaxis has shown no difference between the two treatment modalities in terms of rebleeding or mortality but found more complications in the cyanoacrylate group[163]. Another study compared TIPS with cyanoacrylate in patients with bleeding from gastric varices. TIPS was shown to be more effective for prevention of recurrent bleeding, with no difference in mortality[164]. These results are in contrast to a retrospective analysis that found TIPS and cyanoacrylate equally effective in controlling and preventing gastric variceal hemorrhage with no significant differences in survival[165]. Patients who received TIPS experienced significantly more long-term morbidity[165]. Nevertheless, the above mentioned studies have to be interpreted with caution, since they included patients with different types of gastric varices.
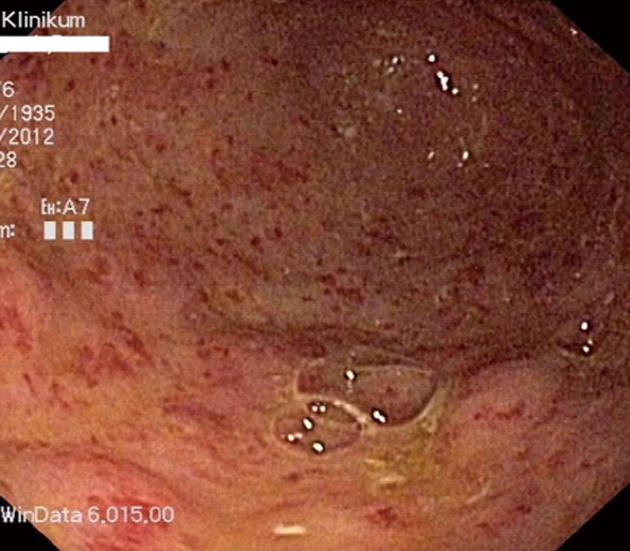
The diagnosis of portal hypertensive gastropathy is made by endoscopy. Typical signs are mosaic, also called “snakeskin”, pattern of erythema. More severe forms present with red punctuate erythema, diffuse hemorrhagic lesions and/or brown spots that indicate submucosal hemorrhage[166]. Histopathologic features of portal hypertensive gastropathy are vascular ectasia of the mucosal and submucosal veins and capillaries[166]. The exact pathogenesis of portal hypertensive gastropathy is unknown. Important factors in the pathogenesis are the presence of portal hypertension as well as hyperemia of the gastric mucosa. Several authors assumed that the endoscopic treatment of esophageal varices aggravates portal hypertensive gastropathy[167]. The worsening is often transient and portal hypertensive gastropathy shows regression in more than 40% of patients after VBL[168]. The incidence of portal hypertensive gastropathy is around 80% in patients with liver cirrhosis[169]. Acute bleeding from portal hypertensive gastropathy (Figure 4) is a rare event, with an incidence of less than 3% in three years. One study that evaluated the cause of GI-bleeding in 1496 patients found bleeding from portal hypertensive gastropathy the cause in 0.8% of patients, accounting for 8% of non-variceal bleeding in patients with liver disease[170]. The probability of chronic bleeding is around 10%-15% in three years[6].
There is only one small trial that studied the effect of non-selective beta-blockers on portal hypertensive gastropathy[171]. Twenty-four patients with non-bleeding portal hypertensive gastropathy received 160 mg propranolol per day in a double-blind placebo controlled cross-over trial. Endoscopic grading of portal hypertensive gastropathy improved after propranolol in nine patients compared to three after placebo[171].
The therapy of acute bleeding from portal hypertensive gastropathy is mainly based on drugs that decrease portal pressure. In one study, 14 portal hypertensive patients with heavy diffuse bleeding from portal hypertensive gastropathy received propranolol in a dose of 24 to 480 mg per day. Within 3 d, bleeding ceased in 13 (93%) of patients[171]. Since the study did not have a control group of untreated patients, the results have to be interpreted with caution. A small study compared octreotide, vasopressin and omeprazole for therapy of acute bleeding. In this setting, octreotide was more effective than omeprazole or vasopressin[172]. Terlipressin was also shown to be effective in acute bleeding from portal hypertensive gastropathy[173].
No studies that investigated the role of endoscopic treatment using argon-plasma-coagulation in acute or recurrent bleeding from portal hypertensive gastropathy are available. If medical therapy fails, TIPS insertion or surgical shunt are an option[6,174,175].
In the secondary prophylaxis of bleeding from portal hypertensive gastropathy, one study including 54 patients showed that propranolol is effective in the prevention of rebleeding[176]. In the group of the propranolol treated patients 65% were free of rebleeding after one year compared to 38% in the control group. After 30 mo of follow-up, 52% of the patients in the propranolol group were free of rebleeding compared to 7% of the untreated patients[176].
In summary, the risk of bleeding from portal hypertensive gastropathy is low and primary prophylaxis is therefore not necessary. In patients with recurrent bleeding from portal hypertensive gastropathy, propranolol should be considered for secondary prophylaxis.
Bleeding from gastric antral vascular ectasia (GAVE) is an uncommon but sometimes severe cause of upper gastrointestinal bleeding. It accounts for 4% of non-variceal upper GI-bleeding[177].
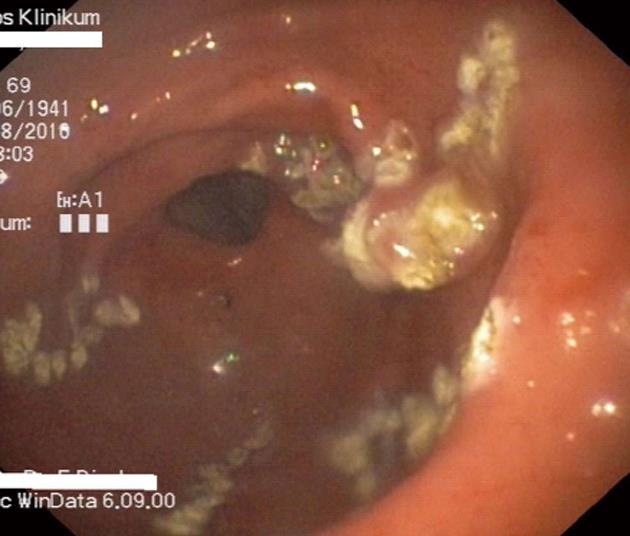
Gastric antral vascular ectasia (GAVE-syndrome, also known as “watermelon stomach” or “honeycomb stomach”) is endoscopically as well as histologically distinguished form portal hypertensive gastropathy. In most patients, the diagnosis of GAVE is easily made on endoscopy. In case of diagnostic uncertainty, the so called GAVE-score that defines histological changes helps to distinguish the both entities[177]. GAVE-syndrome is most often found in older women and is associated with autoimmune disorders in about 60% of patients[178]. Liver disease is a risk factor for the development of GAVE-syndrome, but only 30% of affected patients suffer from liver cirrhosis[179]. On endoscopy (Figure 5), linear red streaks running longitudinally in the gastric antrum are apparent (“watermelon stomach”). In patients with liver cirrhosis the mucosal pattern is often more diffuse (“honeycomb stomach”)[180]. The lesion consists of ectatic vessels of the mucosa with focal thrombosis surrounded by fibromuscular hyperplasia[181]. The pathogenesis of GAVE is not well known. Hypothesis for the pathogenesis include mechanical stress[182], humoural[183] and autoimmune factors[184]. Portal hypertension per se does not seem to be a risk factor for GAVE[185,186].
Different drugs have been used in the treatment of bleeding from GAVE. A small controlled cross-over trial has shown estrogen-progesterone to be highly effective in GAVE related bleeding[187]. Another study confirmed these findings[188]. However, the therapy has to be maintained on a long-term basis since a dose reduction results in recurrent bleeding[189]. Moreover, long-term hormonal treatment is associated with an increased risk for breast and endometrial cancer[190]. One small trial showed octreotide to be effective in bleeding from GAVE[191], but another study failed to confirm the efficacy of octreotide[192].
Treatment consists mainly of endoscopic measures like argon plasma coagulation (APC) (Figure 6), or laser photoablation of the lesions[193,194]. Endoscopic treatment using (Nd: YAG) laser has been shown to be effective in bleeding from GAVE in several studies[195-198]. The treatment is relatively safe, complications like perforation or pyloric stenosis are infrequent[199]. Disadvantages of the method are the high costs and the need for a long training period. Argon plasma coagulation has therefore widely replaced laser therapy in the treatment of GAVE related bleeding. The procedure is easy to use, relatively cheap and widely available as well as safe. The efficacy of APC in the treatment of bleeding from GAVE is very high (90%-100% in two studies[194,200]). On average, 2.5 sessions are necessary for successful eradication of the lesions[193,194,201]. Three studies using endoscopic band ligation for the treatment of GAVE related bleeding are available[202-204]. Band ligation was shown to be effective in all trials but a study with sufficient patient numbers comparing band ligation to APC treatment is missing. Lowering portal pressure by TIPS-insertion is not effective in chronic bleeding from GAVE[179,205]. Surgery (antrectomy) is efficient in bleeding from GAVE[206] but bears a significant morbidity and mortality and is therefore reserved for patients with recurrent bleeding despite therapy with argon plasma coagulation.
Ectopic varices are dilated porto-venous vessels of the gastrointestinal mucosa that are located outside of the esophagus or the stomach.
Ectopic varices have their origin from preexisting small veins of the gastrointestinal mucosa that are porto-systemic collaterals between the portal vein and the inferior vena cava. In the majority of cases, portal hypertension or an extrahepatic obliteration of the portal vein are the cause for the development of ectopic varices.
Bleeding from ectopic varices is a rare event. It accounts for 1%-5% of all gastrointestinal bleeding episodes in patients with portal hypertension[207,208]. Endoscopy is the most important diagnostic tool. In patients with portal hypertension, acute bleeding and negative findings on upper endoscopy, bleeding from ectopic varices has to be considered. In these patients, accurate examination of the duodenum is mandatory. Examination of the jejunum makes double-balloon enteroscopy necessary.
Colonoscopy is the principal method for the diagnosis of colonic varices. One study found rectal varices via endoscopy in 43% and via EUS in 75% of patients with portal hypertension, pointing out that rectal varices might be overlooked by conventional endoscopy[209].
In patients in whom bleeding from ectopic varices is assumed but endoscopy was negative, nuclear magnetic resonance (NMR) with NMR-angiography is the diagnostic tool of choice and allows the identification of ectopic varices in most patients.
Sclerotherapy/injection therapy: Therapy of ectopic varices is mainly based on sclerotherapy or injection therapy. Controlled studies which method is best are not available but case reports showed that both sclerotherapy with ethoxysklerol as well as injection of the varix with cyanoacrylate are feasible[210-213].
Band ligation may be useful for temporary hemostasis[209,214] in duodenal varices but rebleeding of duodenal varices is a problem with ligation therapy. Additional treatment following band ligation for duodenal varices is therefore mandatory.
Surgery and TIPS: Porta-caval shunts are effective therapy measures in recurrent bleeding from ectopic varices[147,215,216]. Another option in patients without portal vein thrombosis is TIPS-insertion. Several case reports that show that TIPS is an effective option in the treatment of ectopic varices have been published[217-220].
Interventional radiology: Balloon-occluded retrograde transvenous obliteration (B-RTO) was successfully performed for patients with duodenal varices[221,222]. B-RTO can obliterate not only varices but also the afferent and efferent veins and should be considered for treating duodenal varices.
Medical therapy: From a pathophysiological point of view the application of beta-blockers does makes sense in patients with ectopic varices, but no data from controlled trials that investigate the role of non-selective beta-blockers and/or nitrates are available.
P- Reviewers Fausto C, Issa IA, Karaman A S- Editor Wen LL L- Editor A E- Editor Ma S
| 1. | Calès P, Pascal JP. Natural history of esophageal varices in cirrhosis (from origin to rupture). Gastroenterol Clin Biol. 1988;12:245-254. [PubMed] |
| 2. | D’Amico G, Pagliaro L, Bosch J. The treatment of portal hypertension: a meta-analytic review. Hepatology. 1995;22:332-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 454] [Cited by in RCA: 467] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 3. | Abraldes JG, Villanueva C, Bañares R, Aracil C, Catalina MV, Garci A-Pagán JC, Bosch J. Hepatic venous pressure gradient and prognosis in patients with acute variceal bleeding treated with pharmacologic and endoscopic therapy. J Hepatol. 2008;48:229-236. [PubMed] |
| 4. | Augustin S, Altamirano J, González A, Dot J, Abu-Suboh M, Armengol JR, Azpiroz F, Esteban R, Guardia J, Genescà J. Effectiveness of combined pharmacologic and ligation therapy in high-risk patients with acute esophageal variceal bleeding. Am J Gastroenterol. 2011;106:1787-1795. [PubMed] |
| 5. | Villanueva C, Piqueras M, Aracil C, Gómez C, López-Balaguer JM, Gonzalez B, Gallego A, Torras X, Soriano G, Sáinz S. A randomized controlled trial comparing ligation and sclerotherapy as emergency endoscopic treatment added to somatostatin in acute variceal bleeding. J Hepatol. 2006;45:560-567. [PubMed] |
| 6. | de Franchis R. Updating consensus in portal hypertension: report of the Baveno III Consensus Workshop on definitions, methodology and therapeutic strategies in portal hypertension. J Hepatol. 2000;33:846-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 373] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 7. | North Italian Endoscopic Club for the Study and Treatment of Esophageal Varices. Prediction of the first variceal hemorrhage in patients with cirrhosis of the liver and esophageal varices. A prospective multicenter study. N Engl J Med. 1988;319:983-989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 996] [Cited by in RCA: 841] [Article Influence: 22.7] [Reference Citation Analysis (1)] |
| 8. | Kleber G, Sauerbruch T, Ansari H, Paumgartner G. Prediction of variceal hemorrhage in cirrhosis: a prospective follow-up study. Gastroenterology. 1991;100:1332-1337. [PubMed] |
| 9. | Merkel C, Bolognesi M, Bellon S, Zuin R, Noventa F, Finucci G, Sacerdoti D, Angeli P, Gatta A. Prognostic usefulness of hepatic vein catheterization in patients with cirrhosis and esophageal varices. Gastroenterology. 1992;102:973-979. [PubMed] |
| 10. | Sauerbruch T, Wotzka R, Köpcke W, Härlin M, Heldwein W, Bayerdörffer E, Sander R, Ansari H, Starz I, Paumgartner G. Prophylactic sclerotherapy before the first episode of variceal hemorrhage in patients with cirrhosis. N Engl J Med. 1988;319:8-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 91] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 11. | Christensen E, Fauerholdt L, Schlichting P, Juhl E, Poulsen H, Tygstrup N. Aspects of the natural history of gastrointestinal bleeding in cirrhosis and the effect of prednisone. Gastroenterology. 1981;81:944-952. [PubMed] |
| 12. | D’Amico G, Luca A. Natural history. Clinical-haemodynamic correlations. Prediction of the risk of bleeding. Baillieres Clin Gastroenterol. 1997;11:243-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 144] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 13. | Zoli M, Merkel C, Magalotti D, Gueli C, Grimaldi M, Gatta A, Bernardi M. Natural history of cirrhotic patients with small esophageal varices: a prospective study. Am J Gastroenterol. 2000;95:503-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 68] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Biecker E, Classen L, Sauerbruch T, Schepke M. Does therapy of oesophageal varices influences the progression of varices? Eur J Gastroenterol Hepatol. 2009;21:751-755. [PubMed] |
| 15. | Groszmann RJ, Garcia-Tsao G, Bosch J, Grace ND, Burroughs AK, Planas R, Escorsell A, Garcia-Pagan JC, Patch D, Matloff DS. Beta-blockers to prevent gastroesophageal varices in patients with cirrhosis. N Engl J Med. 2005;353:2254-2261. [PubMed] |
| 16. | Merkel C, Marin R, Angeli P, Zanella P, Felder M, Bernardinello E, Cavallarin G, Bolognesi M, Donada C, Bellini B. A placebo-controlled clinical trial of nadolol in the prophylaxis of growth of small esophageal varices in cirrhosis. Gastroenterology. 2004;127:476-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 167] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 17. | Garcia-Tsao G. Current management of the complications of cirrhosis and portal hypertension: variceal hemorrhage, ascites, and spontaneous bacterial peritonitis. Gastroenterology. 2001;120:726-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 284] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 18. | Turnes J, Garcia-Pagan JC, Abraldes JG, Hernandez-Guerra M, Dell’Era A, Bosch J. Pharmacological reduction of portal pressure and long-term risk of first variceal bleeding in patients with cirrhosis. Am J Gastroenterol. 2006;101:506-512. [PubMed] |
| 19. | Thalheimer U, Triantos CK, Samonakis DN, Patch D, Burroughs AK. Infection, coagulation, and variceal bleeding in cirrhosis. Gut. 2005;54:556-563. [PubMed] |
| 20. | Feu F, Bordas JM, Luca A, García-Pagán JC, Escorsell A, Bosch J, Rodés J. Reduction of variceal pressure by propranolol: comparison of the effects on portal pressure and azygos blood flow in patients with cirrhosis. Hepatology. 1993;18:1082-1089. [PubMed] |
| 21. | Hobolth L, Møller S, Grønbæk H, Roelsgaard K, Bendtsen F, Feldager Hansen E. Carvedilol or propranolol in portal hypertension? A randomized comparison. Scand J Gastroenterol. 2012;47:467-474. [PubMed] |
| 22. | Lo GH, Chen WC, Wang HM, Yu HC. Randomized, controlled trial of carvedilol versus nadolol plus isosorbide mononitrate for the prevention of variceal rebleeding. J Gastroenterol Hepatol. 2012;27:1681-1687. [PubMed] |
| 23. | Tripathi D, Ferguson JW, Kochar N, Leithead JA, Therapondos G, McAvoy NC, Stanley AJ, Forrest EH, Hislop WS, Mills PR. Randomized controlled trial of carvedilol versus variceal band ligation for the prevention of the first variceal bleed. Hepatology. 2009;50:825-833. [PubMed] |
| 24. | García-Pagán JC, Morillas R, Bañares R, Albillos A, Villanueva C, Vila C, Genescà J, Jimenez M, Rodriguez M, Calleja JL. Propranolol plus placebo versus propranolol plus isosorbide-5-mononitrate in the prevention of a first variceal bleed: a double-blind RCT. Hepatology. 2003;37:1260-1266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 106] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 25. | Abraczinskas DR, Ookubo R, Grace ND, Groszmann RJ, Bosch J, Garcia-Tsao G, Richardson CR, Matloff DS, Rodés J, Conn HO. Propranolol for the prevention of first esophageal variceal hemorrhage: a lifetime commitment? Hepatology. 2001;34:1096-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 71] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 26. | Abraldes JG, Tarantino I, Turnes J, Garcia-Pagan JC, Rodés J, Bosch J. Hemodynamic response to pharmacological treatment of portal hypertension and long-term prognosis of cirrhosis. Hepatology. 2003;37:902-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 367] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 27. | Lo GH, Chen WC, Chen MH, Lin CP, Lo CC, Hsu PI, Cheng JS, Lai KH. Endoscopic ligation vs. nadolol in the prevention of first variceal bleeding in patients with cirrhosis. Gastrointest Endosc. 2004;59:333-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 102] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 28. | Lui HF, Stanley AJ, Forrest EH, Jalan R, Hislop WS, Mills PR, Finlayson ND, Macgilchrist AJ, Hayes PC. Primary prophylaxis of variceal hemorrhage: a randomized controlled trial comparing band ligation, propranolol, and isosorbide mononitrate. Gastroenterology. 2002;123:735-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 108] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 29. | Sarin SK, Lamba GS, Kumar M, Misra A, Murthy NS. Comparison of endoscopic ligation and propranolol for the primary prevention of variceal bleeding. N Engl J Med. 1999;340:988-993. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 222] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 30. | Schepke M, Kleber G, Nürnberg D, Willert J, Koch L, Veltzke-Schlieker W, Hellerbrand C, Kuth J, Schanz S, Kahl S. Ligation versus propranolol for the primary prophylaxis of variceal bleeding in cirrhosis. Hepatology. 2004;40:65-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 132] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 31. | Imperiale TF, Chalasani N. A meta-analysis of endoscopic variceal ligation for primary prophylaxis of esophageal variceal bleeding. Hepatology. 2001;33:802-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 184] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 32. | Gluud LL, Krag A. Banding ligation versus beta-blockers for primary prevention in oesophageal varices in adults. Cochrane Database Syst Rev. 2012;8:CD004544. [PubMed] |
| 33. | Jalan R, Hayes PC. UK guidelines on the management of variceal haemorrhage in cirrhotic patients. British Society of Gastroenterology. Gut. 2000;46 Suppl 3-4:III1-III15. [PubMed] |
| 34. | Augustin S, Muntaner L, Altamirano JT, González A, Saperas E, Dot J, Abu-Suboh M, Armengol JR, Malagelada JR, Esteban R. Predicting early mortality after acute variceal hemorrhage based on classification and regression tree analysis. Clin Gastroenterol Hepatol. 2009;7:1347-1354. [PubMed] |
| 35. | D’Amico G, De Franchis R. Upper digestive bleeding in cirrhosis. Post-therapeutic outcome and prognostic indicators. Hepatology. 2003;38:599-612. [PubMed] |
| 36. | Bernard B, Cadranel JF, Valla D, Escolano S, Jarlier V, Opolon P. Prognostic significance of bacterial infection in bleeding cirrhotic patients: a prospective study. Gastroenterology. 1995;108:1828-1834. [PubMed] |
| 37. | Cárdenas A, Ginès P, Uriz J, Bessa X, Salmerón JM, Mas A, Ortega R, Calahorra B, De Las Heras D, Bosch J. Renal failure after upper gastrointestinal bleeding in cirrhosis: incidence, clinical course, predictive factors, and short-term prognosis. Hepatology. 2001;34:671-676. [PubMed] |
| 38. | García-Pagán JC, Reverter E, Abraldes JG, Bosch J. Acute variceal bleeding. Semin Respir Crit Care Med. 2012;33:46-54. [PubMed] |
| 39. | Bernard B, Grangé JD, Khac EN, Amiot X, Opolon P, Poynard T. Antibiotic prophylaxis for the prevention of bacterial infections in cirrhotic patients with gastrointestinal bleeding: a meta-analysis. Hepatology. 1999;29:1655-1661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 594] [Cited by in RCA: 461] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 40. | Blaise M, Pateron D, Trinchet JC, Levacher S, Beaugrand M, Pourriat JL. Systemic antibiotic therapy prevents bacterial infection in cirrhotic patients with gastrointestinal hemorrhage. Hepatology. 1994;20:34-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 94] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 41. | Fernández J, Ruiz del Arbol L, Gómez C, Durandez R, Serradilla R, Guarner C, Planas R, Arroyo V, Navasa M. Norfloxacin vs ceftriaxone in the prophylaxis of infections in patients with advanced cirrhosis and hemorrhage. Gastroenterology. 2006;131:1049-1056; quiz 1285. [PubMed] |
| 42. | Hou MC, Lin HC, Liu TT, Kuo BI, Lee FY, Chang FY, Lee SD. Antibiotic prophylaxis after endoscopic therapy prevents rebleeding in acute variceal hemorrhage: a randomized trial. Hepatology. 2004;39:746-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 245] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 43. | Soares-Weiser K, Brezis M, Tur-Kaspa R, Paul M, Yahav J, Leibovici L. Antibiotic prophylaxis of bacterial infections in cirrhotic inpatients: a meta-analysis of randomized controlled trials. Scand J Gastroenterol. 2003;38:193-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 59] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 44. | Rimola A, García-Tsao G, Navasa M, Piddock LJ, Planas R, Bernard B, Inadomi JM. Diagnosis, treatment and prophylaxis of spontaneous bacterial peritonitis: a consensus document. International Ascites Club. J Hepatol. 2000;32:142-153. [PubMed] |
| 45. | Castañeda B, Morales J, Lionetti R, Moitinho E, Andreu V, Pérez-Del-Pulgar S, Pizcueta P, Rodés J, Bosch J. Effects of blood volume restitution following a portal hypertensive-related bleeding in anesthetized cirrhotic rats. Hepatology. 2001;33:821-825. [PubMed] |
| 46. | Villanueva C, Colomo A, Bosch A, Concepción M, Hernandez-Gea V, Aracil C, Graupera I, Poca M, Alvarez-Urturi C, Gordillo J. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med. 2013;368:11-21. [PubMed] |
| 47. | Bañares R, Albillos A, Rincón D, Alonso S, González M, Ruiz-del-Arbol L, Salcedo M, Molinero LM. Endoscopic treatment versus endoscopic plus pharmacologic treatment for acute variceal bleeding: a meta-analysis. Hepatology. 2002;35:609-615. [PubMed] |
| 48. | Conn HO, Ramsby GR, Storer EH, Mutchnick MG, Joshi PH, Phillips MM, Cohen GA, Fields GN, Petroski D. Intraarterial vasopressin in the treatment of upper gastrointestinal hemorrhage: a prospective, controlled clinical trial. Gastroenterology. 1975;68:211-221. [PubMed] |
| 49. | Bosch J, Groszmann RJ, García-Pagán JC, Terés J, García-Tsao G, Navasa M, Mas A, Rodés J. Association of transdermal nitroglycerin to vasopressin infusion in the treatment of variceal hemorrhage: a placebo-controlled clinical trial. Hepatology. 1989;10:962-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 58] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 50. | Gimson AE, Westaby D, Hegarty J, Watson A, Williams R. A randomized trial of vasopressin and vasopressin plus nitroglycerin in the control of acute variceal hemorrhage. Hepatology. 1986;6:410-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 123] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 51. | Fogel MR, Knauer CM, Andres LL, Mahal AS, Stein DE, Kemeny MJ, Rinki MM, Walker JE, Siegmund D, Gregory PB. Continuous intravenous vasopressin in active upper gastrointestinal bleeding. Ann Intern Med. 1982;96:565-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 94] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 52. | Mallory A, Schaefer JW, Cohen JR, Holt SA, Norton LW. Selective intra-arterial vasopression in fusion for upper gastrointestinal tract hemorrhage: a controlled trial. Arch Surg. 1980;115:30-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 53. | Merigan TC, Plotkin GR, Davidson CS. Effect of intravenously administered posterior pituitary extract on hemorrhage from bleeding esophageal varices. A controlled evaluation. N Engl J Med. 1962;266:134-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 97] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 54. | Levacher S, Letoumelin P, Pateron D, Blaise M, Lapandry C, Pourriat JL. Early administration of terlipressin plus glyceryl trinitrate to control active upper gastrointestinal bleeding in cirrhotic patients. Lancet. 1995;346:865-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 174] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 55. | Söderlund C, Magnusson I, Törngren S, Lundell L. Terlipressin (triglycyl-lysine vasopressin) controls acute bleeding oesophageal varices. A double-blind, randomized, placebo-controlled trial. Scand J Gastroenterol. 1990;25:622-630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 73] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 56. | Ioannou GN, Doust J, Rockey DC. Systematic review: terlipressin in acute oesophageal variceal haemorrhage. Aliment Pharmacol Ther. 2003;17:53-64. [PubMed] |
| 57. | Escorsell A, Ruiz del Arbol L, Planas R, Albillos A, Bañares R, Calès P, Pateron D, Bernard B, Vinel JP, Bosch J. Multicenter randomized controlled trial of terlipressin versus sclerotherapy in the treatment of acute variceal bleeding: the TEST study. Hepatology. 2000;32:471-476. [PubMed] |
| 58. | Escorsell A, Bandi JC, Moitinho E, Feu F, García-Pagan JC, Bosch J, Rodés J. Time profile of the haemodynamic effects of terlipressin in portal hypertension. J Hepatol. 1997;26:621-627. [PubMed] |
| 59. | Solà E, Lens S, Guevara M, Martín-Llahí M, Fagundes C, Pereira G, Pavesi M, Fernández J, González-Abraldes J, Escorsell A. Hyponatremia in patients treated with terlipressin for severe gastrointestinal bleeding due to portal hypertension. Hepatology. 2010;52:1783-1790. [PubMed] |
| 60. | Chiu KW, Sheen IS, Liaw YF. A controlled study of glypressin versus vasopressin in the control of bleeding from oesophageal varices. J Gastroenterol Hepatol. 1990;5:549-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 61. | Freeman JG, Cobden I, Record CO. Placebo-controlled trial of terlipressin (glypressin) in the management of acute variceal bleeding. J Clin Gastroenterol. 1989;11:58-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 62] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 62. | D’Amico G, Traina M, Vizzini G, Tinè F, Politi F, Montalbano L, Luca A, Pasta L, Pagliaro L, Morabito A. Terlipressin or vasopressin plus transdermal nitroglycerin in a treatment strategy for digestive bleeding in cirrhosis. A randomized clinical trial. Liver Study Group of V. Cervello Hospital. J Hepatol. 1994;20:206-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 41] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 63. | Silvain C, Carpentier S, Sautereau D, Czernichow B, Métreau JM, Fort E, Ingrand P, Boyer J, Pillegand B, Doffël M. Terlipressin plus transdermal nitroglycerin vs. octreotide in the control of acute bleeding from esophageal varices: a multicenter randomized trial. Hepatology. 1993;18:61-65. [PubMed] |
| 64. | Walker S, Kreichgauer HP, Bode JC. Terlipressin (glypressin) versus somatostatin in the treatment of bleeding esophageal varices--final report of a placebo-controlled, double-blind study. Z Gastroenterol. 1996;34:692-698. [PubMed] |
| 65. | Abraldes JG, Bosch J. Somatostatin and analogues in portal hypertension. Hepatology. 2002;35:1305-1312. [PubMed] |
| 66. | Bagarani M, Albertini V, Anzà M, Barlattani A, Bracci F, Cucchiara G, Gizzonio D, Grassini G, Mari T, Procacciante F. Effect of somatostatin in controlling bleeding from esophageal varices. Ital J Surg Sci. 1987;17:21-26. [PubMed] |
| 67. | Lin HC, Yang YY, Hou MC, Huang YT, Lee WC, Lee FY, Chang FY, Lee SD. Hemodynamic effects of a combination of octreotide and terlipressin in patients with viral hepatitis related cirrhosis. Scand J Gastroenterol. 2002;37:482-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 68. | Gotzsche PC. Somatostatin or octreotide for acute bleeding oesophageal varices. (Cochrane review). Oxford: Update Software 2001; . |
| 69. | Joannon G, Doust J, Rockey DC. Terlipressin for acute esophageal variceal hemorrhage. (Cochrane review). Oxford: Update Software 2001; . |
| 70. | Lo GH, Lai KH, Ng WW, Tam TN, Lee SD, Tsai YT, Lo KJ. Injection sclerotherapy preceded by esophageal tamponade versus immediate sclerotherapy in arresting active variceal bleeding: a prospective randomized trial. Gastrointest Endosc. 1992;38:421-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 71. | Moretó M, Zaballa M, Bernal A, Ibáñez S, Ojembarrena E, Rodriguez A. A randomized trial of tamponade or sclerotherapy as immediate treatment for bleeding esophageal varices. Surg Gynecol Obstet. 1988;167:331-334. [PubMed] |
| 72. | Paquet KJ, Feussner H. Endoscopic sclerosis and esophageal balloon tamponade in acute hemorrhage from esophagogastric varices: a prospective controlled randomized trial. Hepatology. 1985;5:580-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 189] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 73. | Van Stiegmann G, Goff JS. Endoscopic esophageal varix ligation: preliminary clinical experience. Gastrointest Endosc. 1988;34:113-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 100] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 74. | Laine L, el-Newihi HM, Migikovsky B, Sloane R, Garcia F. Endoscopic ligation compared with sclerotherapy for the treatment of bleeding esophageal varices. Ann Intern Med. 1993;119:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 203] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 75. | Lo GH, Lai KH, Cheng JS, Lin CK, Huang JS, Hsu PI, Chiang HT. Emergency banding ligation versus sclerotherapy for the control of active bleeding from esophageal varices. Hepatology. 1997;25:1101-1104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 135] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 76. | Stiegmann GV, Goff JS, Michaletz-Onody PA, Korula J, Lieberman D, Saeed ZA, Reveille RM, Sun JH, Lowenstein SR. Endoscopic sclerotherapy as compared with endoscopic ligation for bleeding esophageal varices. N Engl J Med. 1992;326:1527-1532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 504] [Cited by in RCA: 415] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 77. | Sengstaken RW, Blakemore AH. Balloon tamponage for the control of hemorrhage from esophageal varices. Ann Surg. 1950;131:781-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 150] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 78. | Terés J, Cecilia A, Bordas JM, Rimola A, Bru C, Rodés J. Esophageal tamponade for bleeding varices. Controlled trial between the Sengstaken-Blakemore tube and the Linton-Nachlas tube. Gastroenterology. 1978;75:566-569. [PubMed] |
| 79. | Panés J, Terés J, Bosch J, Rodés J. Efficacy of balloon tamponade in treatment of bleeding gastric and esophageal varices. Results in 151 consecutive episodes. Dig Dis Sci. 1988;33:454-459. [PubMed] |
| 80. | Cook D, Laine L. Indications, technique, and complications of balloon tamponade for variceal gastrointestinal bleeding. J Intensive Care Med. 1992;7:212-218. [PubMed] |
| 81. | Maufa F, Al-Kawas FH. Role of self-expandable metal stents in acute variceal bleeding. Int J Hepatol. 2012;2012:418369. [PubMed] |
| 82. | Dechêne A, El Fouly AH, Bechmann LP, Jochum C, Saner FH, Gerken G, Canbay A. Acute management of refractory variceal bleeding in liver cirrhosis by self-expanding metal stents. Digestion. 2012;85:185-191. [PubMed] |
| 83. | Hubmann R, Bodlaj G, Czompo M, Benkö L, Pichler P, Al-Kathib S, Kiblböck P, Shamyieh A, Biesenbach G. The use of self-expanding metal stents to treat acute esophageal variceal bleeding. Endoscopy. 2006;38:896-901. [PubMed] |
| 84. | Wright G, Lewis H, Hogan B, Burroughs A, Patch D, O’Beirne J. A self-expanding metal stent for complicated variceal hemorrhage: experience at a single center. Gastrointest Endosc. 2010;71:71-78. [PubMed] |
| 85. | Zehetner J, Shamiyeh A, Wayand W, Hubmann R. Results of a new method to stop acute bleeding from esophageal varices: implantation of a self-expanding stent. Surg Endosc. 2008;22:2149-2152. [PubMed] |
| 86. | Jalan R, Elton RA, Redhead DN, Finlayson ND, Hayes PC. Analysis of prognostic variables in the prediction of mortality, shunt failure, variceal rebleeding and encephalopathy following the transjugular intrahepatic portosystemic stent-shunt for variceal haemorrhage. J Hepatol. 1995;23:123-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 129] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 87. | McCormick PA, Dick R, Chin J, Irving JD, McIntyre N, Hobbs KE, Burroughs AK. Transjugular intrahepatic portosystemic stent-shunt. Br J Hosp Med. 1993;49:791-793, 796-797. [PubMed] |
| 88. | Sanyal AJ, Freedman AM, Luketic VA, Purdum PP, Shiffman ML, Tisnado J, Cole PE. Transjugular intrahepatic portosystemic shunts for patients with active variceal hemorrhage unresponsive to sclerotherapy. Gastroenterology. 1996;111:138-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 159] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 89. | Jalan R, Forrest EH, Stanley AJ, Redhead DN, Forbes J, Dillon JF, MacGilchrist AJ, Finlayson ND, Hayes PC. A randomized trial comparing transjugular intrahepatic portosystemic stent-shunt with variceal band ligation in the prevention of rebleeding from esophageal varices. Hepatology. 1997;26:1115-1122. [PubMed] |
| 90. | Rössle M, Deibert P, Haag K, Ochs A, Olschewski M, Siegerstetter V, Hauenstein KH, Geiger R, Stiepak C, Keller W. Randomised trial of transjugular-intrahepatic-portosystemic shunt versus endoscopy plus propranolol for prevention of variceal rebleeding. Lancet. 1997;349:1043-1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 171] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 91. | Sanyal AJ, Freedman AM, Luketic VA, Purdum PP, Shiffman ML, DeMeo J, Cole PE, Tisnado J. The natural history of portal hypertension after transjugular intrahepatic portosystemic shunts. Gastroenterology. 1997;112:889-898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 199] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 92. | Jabbour N, Zajko AB, Orons PD, Irish W, Bartoli F, Marsh WJ, Dodd GD, Aldreghitti L, Colangelo J, Rakela J. Transjugular intrahepatic portosystemic shunt in patients with end-stage liver disease: results in 85 patients. Liver Transpl Surg. 1996;2:139-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 93. | Freedman AM, Sanyal AJ, Tisnado J, Cole PE, Shiffman ML, Luketic VA, Purdum PP, Darcy MD, Posner MP. Complications of transjugular intrahepatic portosystemic shunt: a comprehensive review. Radiographics. 1993;13:1185-1210. [PubMed] |
| 94. | Sanyal AJ, Freedman AM, Purdum PP. TIPS-associated hemolysis and encephalopathy. Ann Intern Med. 1992;117:443-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 95. | Bureau C, Garcia-Pagan JC, Otal P, Pomier-Layrargues G, Chabbert V, Cortez C, Perreault P, Péron JM, Abraldes JG, Bouchard L, Bilbao JI, Bosch J, Rousseau H, Vinel JP. Improved clinical outcome using polytetrafluoroethylene-coated stents for TIPS: results of a randomized study. Gastroenterology. 2004;126:469-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 370] [Cited by in RCA: 338] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 96. | García-Pagán JC, Caca K, Bureau C, Laleman W, Appenrodt B, Luca A, Abraldes JG, Nevens F, Vinel JP, Mössner J, Bosch J. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362:2370-2379. [PubMed] |
| 97. | Thabut D, Rudler M, Lebrec D. Early TIPS with covered stents in high-risk patients with cirrhosis presenting with variceal bleeding: are we ready to dive into the deep end of the pool? J Hepatol. 2011;55:1148-1149. [PubMed] |
| 98. | Rosemurgy AS, Goode SE, Zwiebel BR, Black TJ, Brady PG. A prospective trial of transjugular intrahepatic portasystemic stent shunts versus small-diameter prosthetic H-graft portacaval shunts in the treatment of bleeding varices. Ann Surg. 1996;224:378-384; discussion 384-386. [PubMed] |
| 99. | Terés J, Baroni R, Bordas JM, Visa J, Pera C, Rodés J. Randomized trial of portacaval shunt, stapling transection and endoscopic sclerotherapy in uncontrolled variceal bleeding. J Hepatol. 1987;4:159-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 70] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 100. | Bari K, Garcia-Tsao G. Treatment of portal hypertension. World J Gastroenterol. 2012;18:1166-1175. [PubMed] |
| 101. | Buchwalow IB, Podzuweit T, Bocker W, Samoilova VE, Thomas S, Wellner M, Baba HA, Robenek H, Schnekenburger J, Lerch MM. Vascular smooth muscle and nitric oxide synthase. FASEB J. 2002;16:500-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 118] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 102. | Burroughs AK, Jenkins WJ, Sherlock S, Dunk A, Walt RP, Osuafor TO, Mackie S, Dick R. Controlled trial of propranolol for the prevention of recurrent variceal hemorrhage in patients with cirrhosis. N Engl J Med. 1983;309:1539-1542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 211] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 103. | Colombo M, de Franchis R, Tommasini M, Sangiovanni A, Dioguardi N. Beta-blockade prevents recurrent gastrointestinal bleeding in well-compensated patients with alcoholic cirrhosis: a multicenter randomized controlled trial. Hepatology. 1989;9:433-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 73] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 104. | Queuniet AM, Czernichow P, Lerebours E, Ducrotte P, Tranvouez JL, Colin R. Controlled study of propranolol in the prevention of recurrent hemorrhage in cirrhotic patients. Gastroenterol Clin Biol. 1987;11:41-47. [PubMed] |
| 105. | Rossi V, Calès P, Burtin P, Charneau J, Person B, Pujol P, Valentin S, D’Aubigny N, Joubaud F, Boyer J. Prevention of recurrent variceal bleeding in alcoholic cirrhotic patients: prospective controlled trial of propranolol and sclerotherapy. J Hepatol. 1991;12:283-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 47] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 106. | Sheen IS, Chen TY, Liaw YF. Randomized controlled study of propranolol for prevention of recurrent esophageal varices bleeding in patients with cirrhosis. Liver. 1989;9:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 107. | Villeneuve JP, Pomier-Layrargues G, Infante-Rivard C, Willems B, Huet PM, Marleau D, Viallet A. Propranolol for the prevention of recurrent variceal hemorrhage: a controlled trial. Hepatology. 1986;6:1239-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 104] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 108. | García-Pagán JC, Feu F, Bosch J, Rodés J. Propranolol compared with propranolol plus isosorbide-5-mononitrate for portal hypertension in cirrhosis. A randomized controlled study. Ann Intern Med. 1991;114:869-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 172] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 109. | Feu F, García-Pagán JC, Bosch J, Luca A, Terés J, Escorsell A, Rodés J. Relation between portal pressure response to pharmacotherapy and risk of recurrent variceal haemorrhage in patients with cirrhosis. Lancet. 1995;346:1056-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 457] [Cited by in RCA: 391] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 110. | Patch D, Sabin CA, Goulis J, Gerunda G, Greenslade L, Merkel C, Burroughs AK. A randomized, controlled trial of medical therapy versus endoscopic ligation for the prevention of variceal rebleeding in patients with cirrhosis. Gastroenterology. 2002;123:1013-1019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 147] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 111. | Villanueva C, Balanzó J, Novella MT, Soriano G, Sáinz S, Torras X, Cussó X, Guarner C, Vilardell F. Nadolol plus isosorbide mononitrate compared with sclerotherapy for the prevention of variceal rebleeding. N Engl J Med. 1996;334:1624-1629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 213] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 112. | Sclerotherapy after first variceal hemorrhage in cirrhosis. A randomized multicenter trial. The Copenhagen Esophageal Varices Sclerotherapy Project. N Engl J Med. 1984;311:1594-1600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 229] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 113. | Korula J, Balart LA, Radvan G, Zweiban BE, Larson AW, Kao HW, Yamada S. A prospective, randomized controlled trial of chronic esophageal variceal sclerotherapy. Hepatology. 1985;5:584-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 177] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 114. | Westaby D, Macdougall BR, Williams R. Improved survival following injection sclerotherapy for esophageal varices: final analysis of a controlled trial. Hepatology. 1985;5:827-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 202] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 115. | Dasarathy S, Dwivedi M, Bhargava DK, Sundaram KR, Ramachandran K. A prospective randomized trial comparing repeated endoscopic sclerotherapy and propranolol in decompensated (Child class B and C) cirrhotic patients. Hepatology. 1992;16:89-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 116. | Westaby D, Polson RJ, Gimson AE, Hayes PC, Hayllar K, Williams R. A controlled trial of oral propranolol compared with injection sclerotherapy for the long-term management of variceal bleeding. Hepatology. 1990;11:353-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 47] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 117. | Dollet JM, Champigneulle B, Patris A, Bigard MA, Gaucher P. Endoscopic sclerotherapy versus propranolol after hemorrhage caused by rupture of esophageal varices in patients with cirrhosis. Results of a 4-year randomized study. Gastroenterol Clin Biol. 1988;12:234-239. [PubMed] |
| 118. | Martin T, Taupignon A, Lavignolle A, Perrin D, Le Bodic L. Prevention of recurrent hemorrhage in patients with cirrhosis. Results of a controlled trial of propranolol versus endoscopic sclerotherapy. Gastroenterol Clin Biol. 1991;15:833-837. [PubMed] |
| 119. | Terés J, Bosch J, Bordas JM, Garcia Pagán JC, Feu F, Cirera I, Rodés J. Propranolol versus sclerotherapy in preventing variceal rebleeding: a randomized controlled trial. Gastroenterology. 1993;105:1508-1514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 37] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 120. | Gimson AE, Ramage JK, Panos MZ, Hayllar K, Harrison PM, Williams R, Westaby D. Randomised trial of variceal banding ligation versus injection sclerotherapy for bleeding oesophageal varices. Lancet. 1993;342:391-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 186] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 121. | Hashizume M, Ohta M, Ueno K, Tanoue K, Kitano S, Sugimachi K. Endoscopic ligation of esophageal varices compared with injection sclerotherapy: a prospective randomized trial. Gastrointest Endosc. 1993;39:123-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 55] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 122. | Hou MC, Lin HC, Kuo BI, Chen CH, Lee FY, Lee SD. Comparison of endoscopic variceal injection sclerotherapy and ligation for the treatment of esophageal variceal hemorrhage: a prospective randomized trial. Hepatology. 1995;21:1517-1522. [PubMed] |
| 123. | Laine L, Cook D. Endoscopic ligation compared with sclerotherapy for treatment of esophageal variceal bleeding. A meta-analysis. Ann Intern Med. 1995;123:280-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 500] [Cited by in RCA: 417] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 124. | Lo GH, Lai KH, Cheng JS, Hwu JH, Chang CF, Chen SM, Chiang HT. A prospective, randomized trial of sclerotherapy versus ligation in the management of bleeding esophageal varices. Hepatology. 1995;22:466-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 146] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 125. | Villanueva C, Miñana J, Ortiz J, Gallego A, Soriano G, Torras X, Sáinz S, Boadas J, Cussó X, Guarner C. Endoscopic ligation compared with combined treatment with nadolol and isosorbide mononitrate to prevent recurrent variceal bleeding. N Engl J Med. 2001;345:647-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 206] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 126. | Lo GH, Chen WC, Chen MH, Hsu PI, Lin CK, Tsai WL, Lai KH. Banding ligation versus nadolol and isosorbide mononitrate for the prevention of esophageal variceal rebleeding. Gastroenterology. 2002;123:728-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 99] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 127. | de la Peña J, Brullet E, Sanchez-Hernández E, Rivero M, Vergara M, Martin-Lorente JL, Garcia Suárez C. Variceal ligation plus nadolol compared with ligation for prophylaxis of variceal rebleeding: a multicenter trial. Hepatology. 2005;41:572-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 130] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 128. | García-Pagán JC, Villanueva C, Albillos A, Bañares R, Morillas R, Abraldes JG, Bosch J. Nadolol plus isosorbide mononitrate alone or associated with band ligation in the prevention of recurrent bleeding: a multicentre randomised controlled trial. Gut. 2009;58:1144-1150. [PubMed] |
| 129. | Kumar A, Jha SK, Sharma P, Dubey S, Tyagi P, Sharma BC, Sarin SK. Addition of propranolol and isosorbide mononitrate to endoscopic variceal ligation does not reduce variceal rebleeding incidence. Gastroenterology. 2009;137:892-901, 901.e1. [PubMed] |
| 130. | Lo GH, Chen WC, Chan HH, Tsai WL, Hsu PI, Lin CK, Chen TA, Lai KH. A randomized, controlled trial of banding ligation plus drug therapy versus drug therapy alone in the prevention of esophageal variceal rebleeding. J Gastroenterol Hepatol. 2009;24:982-987. [PubMed] |
| 131. | Lo GH, Lai KH, Cheng JS, Chen MH, Huang HC, Hsu PI, Lin CK. Endoscopic variceal ligation plus nadolol and sucralfate compared with ligation alone for the prevention of variceal rebleeding: a prospective, randomized trial. Hepatology. 2000;32:461-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 164] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 132. | Cabrera J, Maynar M, Granados R, Gorriz E, Reyes R, Pulido-Duque JM, Rodriguez SanRoman JL, Guerra C, Kravetz D. Transjugular intrahepatic portosystemic shunt versus sclerotherapy in the elective treatment of variceal hemorrhage. Gastroenterology. 1996;110:832-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 155] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 133. | Cello JP, Ring EJ, Olcott EW, Koch J, Gordon R, Sandhu J, Morgan DR, Ostroff JW, Rockey DC, Bacchetti P. Endoscopic sclerotherapy compared with percutaneous transjugular intrahepatic portosystemic shunt after initial sclerotherapy in patients with acute variceal hemorrhage. A randomized, controlled trial. Ann Intern Med. 1997;126:858-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 143] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 134. | García-Villarreal L, Martínez-Lagares F, Sierra A, Guevara C, Marrero JM, Jiménez E, Monescillo A, Hernández-Cabrero T, Alonso JM, Fuentes R. Transjugular intrahepatic portosystemic shunt versus endoscopic sclerotherapy for the prevention of variceal rebleeding after recent variceal hemorrhage. Hepatology. 1999;29:27-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 97] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 135. | Merli M, Salerno F, Riggio O, de Franchis R, Fiaccadori F, Meddi P, Primignani M, Pedretti G, Maggi A, Capocaccia L. Transjugular intrahepatic portosystemic shunt versus endoscopic sclerotherapy for the prevention of variceal bleeding in cirrhosis: a randomized multicenter trial. Gruppo Italiano Studio TIPS (G.I.S.T.). Hepatology. 1998;27:48-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 125] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 136. | Sanyal AJ, Freedman AM, Luketic VA, Purdum PP, Shiffman ML, Cole PE, Tisnado J, Simmons S. Transjugular intrahepatic portosystemic shunts compared with endoscopic sclerotherapy for the prevention of recurrent variceal hemorrhage. A randomized, controlled trial. Ann Intern Med. 1997;126:849-857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 145] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 137. | Sauer P, Theilmann L, Stremmel W, Benz C, Richter GM, Stiehl A. Transjugular intrahepatic portosystemic stent shunt versus sclerotherapy plus propranolol for variceal rebleeding. Gastroenterology. 1997;113:1623-1631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 126] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 138. | Gülberg V, Schepke M, Geigenberger G, Holl J, Brensing KA, Waggershauser T, Reiser M, Schild HH, Sauerbruch T, Gerbes AL. Transjugular intrahepatic portosystemic shunting is not superior to endoscopic variceal band ligation for prevention of variceal rebleeding in cirrhotic patients: a randomized, controlled trial. Scand J Gastroenterol. 2002;37:338-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 139. | Luca A, D’Amico G, La Galla R, Midiri M, Morabito A, Pagliaro L. TIPS for prevention of recurrent bleeding in patients with cirrhosis: meta-analysis of randomized clinical trials. Radiology. 1999;212:411-421. [PubMed] |
| 140. | Papatheodoridis GV, Goulis J, Leandro G, Patch D, Burroughs AK. Transjugular intrahepatic portosystemic shunt compared with endoscopic treatment for prevention of variceal rebleeding: A meta-analysis. Hepatology. 1999;30:612-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 187] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 141. | Zheng M, Chen Y, Bai J, Zeng Q, You J, Jin R, Zhou X, Shen H, Zheng Y, Du Z. Transjugular intrahepatic portosystemic shunt versus endoscopic therapy in the secondary prophylaxis of variceal rebleeding in cirrhotic patients: meta-analysis update. J Clin Gastroenterol. 2008;42:507-516. [PubMed] |
| 142. | Grace ND, Conn HO, Resnick RH, Groszmann RJ, Atterbury CE, Wright SC, Gusberg RJ, Vollman R, Garcia-Tsao G, Fisher RL. Distal splenorenal vs. portal-systemic shunts after hemorrhage from varices: a randomized controlled trial. Hepatology. 1988;8:1475-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 74] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 143. | Henderson JM, Kutner MH, Millikan WJ, Galambos JT, Riepe SP, Brooks WS, Bryan FC, Warren WD. Endoscopic variceal sclerosis compared with distal splenorenal shunt to prevent recurrent variceal bleeding in cirrhosis. A prospective, randomized trial. Ann Intern Med. 1990;112:262-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 81] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 144. | Planas R, Boix J, Broggi M, Cabré E, Gomes-Vieira MC, Morillas R, Armengol M, De León R, Humbert P, Salvá JA. Portacaval shunt versus endoscopic sclerotherapy in the elective treatment of variceal hemorrhage. Gastroenterology. 1991;100:1078-1086. [PubMed] |
| 145. | Reynolds TB, Donovan AJ, Mikkelsen WP, Redeker AG, Turrill FL, Weiner JM. Results of a 12-year randomized trial of portacaval shunt in patients with alcoholic liver disease and bleeding varices. Gastroenterology. 1981;80:1005-1011. [PubMed] |
| 146. | Spina GP, Santambrogio R, Opocher E, Cosentino F, Zambelli A, Passoni GR, Cucchiaro G, Macrì M, Morandi E, Bruno S. Distal splenorenal shunt versus endoscopic sclerotherapy in the prevention of variceal rebleeding. First stage of a randomized, controlled trial. Ann Surg. 1990;211:178-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 47] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 147. | Rikkers LF, Jin G, Burnett DA, Buchi KN, Cormier RA. Shunt surgery versus endoscopic sclerotherapy for variceal hemorrhage: late results of a randomized trial. Am J Surg. 1993;165:27-32; discussion 32-33. [PubMed] |
| 148. | Rosemurgy AS, Serafini FM, Zweibel BR, Black TJ, Kudryk BT, Nord HJ, Goode SE. Transjugular intrahepatic portosystemic shunt vs. small-diameter prosthetic H-graft portacaval shunt: extended follow-up of an expanded randomized prospective trial. J Gastrointest Surg. 2000;4:589-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 60] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 149. | Khan S, Tudur Smith C, Williamson P, Sutton R. Portosystemic shunts versus endoscopic therapy for variceal rebleeding in patients with cirrhosis. Cochrane Database Syst Rev. 2006;CD000553. [PubMed] |
| 150. | Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology. 1992;16:1343-1349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 789] [Cited by in RCA: 846] [Article Influence: 25.6] [Reference Citation Analysis (42)] |
| 151. | Bosch J, Abraldes JG, Groszmann R. Current management of portal hypertension. J Hepatol. 2003;38 Suppl 1:S54-S68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 169] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 152. | Tajiri T, Onda M, Yoshida H, Mamada Y, Taniai N, Yamashita K. The natural history of gastric varices. Hepatogastroenterology. 2002;49:1180-1182. [PubMed] |
| 153. | Schepke M, Biecker E, Appenrodt B, Heller J, Sauerbruch T. Coexisting gastric varices should not preclude prophylactic ligation of large esophageal varices in cirrhosis. Digestion. 2009;80:165-169. [PubMed] |
| 154. | Kang EJ, Jeong SW, Jang JY, Cho JY, Lee SH, Kim HG, Kim SG, Kim YS, Cheon YK, Cho YD. Long-term result of endoscopic Histoacryl (N-butyl-2-cyanoacrylate) injection for treatment of gastric varices. World J Gastroenterol. 2011;17:1494-1500. [PubMed] |
| 155. | Huang YH, Yeh HZ, Chen GH, Chang CS, Wu CY, Poon SK, Lien HC, Yang SS. Endoscopic treatment of bleeding gastric varices by N-butyl-2-cyanoacrylate (Histoacryl) injection: long-term efficacy and safety. Gastrointest Endosc. 2000;52:160-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 177] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 156. | Lee YT, Chan FK, Ng EK, Leung VK, Law KB, Yung MY, Chung SC, Sung JJ. EUS-guided injection of cyanoacrylate for bleeding gastric varices. Gastrointest Endosc. 2000;52:168-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 128] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 157. | Sarin SK, Jain AK, Jain M, Gupta R. A randomized controlled trial of cyanoacrylate versus alcohol injection in patients with isolated fundic varices. Am J Gastroenterol. 2002;97:1010-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 208] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 158. | Lo GH, Lai KH, Cheng JS, Chen MH, Chiang HT. A prospective, randomized trial of butyl cyanoacrylate injection versus band ligation in the management of bleeding gastric varices. Hepatology. 2001;33:1060-1064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 332] [Cited by in RCA: 304] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 159. | Tan PC, Hou MC, Lin HC, Liu TT, Lee FY, Chang FY, Lee SD. A randomized trial of endoscopic treatment of acute gastric variceal hemorrhage: N-butyl-2-cyanoacrylate injection versus band ligation. Hepatology. 2006;43:690-697. [PubMed] |
| 160. | Barange K, Péron JM, Imani K, Otal P, Payen JL, Rousseau H, Pascal JP, Joffre F, Vinel JP. Transjugular intrahepatic portosystemic shunt in the treatment of refractory bleeding from ruptured gastric varices. Hepatology. 1999;30:1139-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 119] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 161. | Chau TN, Patch D, Chan YW, Nagral A, Dick R, Burroughs AK. “Salvage” transjugular intrahepatic portosystemic shunts: gastric fundal compared with esophageal variceal bleeding. Gastroenterology. 1998;114:981-987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 189] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 162. | Wu CY, Yeh HZ, Chen GH. Pharmacologic efficacy in gastric variceal rebleeding and survival: including multivariate analysis. J Clin Gastroenterol. 2002;35:127-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 163. | Evrard S, Dumonceau JM, Delhaye M, Golstein P, Devière J, Le Moine O. Endoscopic histoacryl obliteration vs. propranolol in the prevention of esophagogastric variceal rebleeding: a randomized trial. Endoscopy. 2003;35:729-735. [PubMed] |
| 164. | Lo GH, Liang HL, Chen WC, Chen MH, Lai KH, Hsu PI, Lin CK, Chan HH, Pan HB. A prospective, randomized controlled trial of transjugular intrahepatic portosystemic shunt versus cyanoacrylate injection in the prevention of gastric variceal rebleeding. Endoscopy. 2007;39:679-685. [PubMed] |
| 165. | Procaccini NJ, Al-Osaimi AM, Northup P, Argo C, Caldwell SH. Endoscopic cyanoacrylate versus transjugular intrahepatic portosystemic shunt for gastric variceal bleeding: a single-center U.S. analysis. Gastrointest Endosc. 2009;70:881-887. [PubMed] |
| 166. | Cubillas R, Rockey DC. Portal hypertensive gastropathy: a review. Liver Int. 2010;30:1094-1102. [PubMed] |
| 167. | Sarin SK, Sreenivas DV, Lahoti D, Saraya A. Factors influencing development of portal hypertensive gastropathy in patients with portal hypertension. Gastroenterology. 1992;102:994-999. [PubMed] |
| 168. | Sarin SK, Shahi HM, Jain M, Jain AK, Issar SK, Murthy NS. The natural history of portal hypertensive gastropathy: influence of variceal eradication. Am J Gastroenterol. 2000;95:2888-2893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 80] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 169. | Primignani M, Carpinelli L, Preatoni P, Battaglia G, Carta A, Prada A, Cestari R, Angeli P, Gatta A, Rossi A. Natural history of portal hypertensive gastropathy in patients with liver cirrhosis. The New Italian Endoscopic Club for the study and treatment of esophageal varices (NIEC). Gastroenterology. 2000;119:181-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 157] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 170. | Gostout CJ, Viggiano TR, Balm RK. Acute gastrointestinal bleeding from portal hypertensive gastropathy: prevalence and clinical features. Am J Gastroenterol. 1993;88:2030-2033. [PubMed] |
| 171. | Hosking SW, Kennedy HJ, Seddon I, Triger DR. The role of propranolol in congestive gastropathy of portal hypertension. Hepatology. 1987;7:437-441. [PubMed] |
| 172. | Zhou Y, Qiao L, Wu J, Hu H, Xu C. Comparison of the efficacy of octreotide, vasopressin, and omeprazole in the control of acute bleeding in patients with portal hypertensive gastropathy: a controlled study. J Gastroenterol Hepatol. 2002;17:973-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 58] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 173. | Bruha R, Marecek Z, Spicak J, Hulek P, Lata J, Petrtyl J, Urbanek P, Taimr P, Volfova M, Dite P. Double-blind randomized, comparative multicenter study of the effect of terlipressin in the treatment of acute esophageal variceal and/or hypertensive gastropathy bleeding. Hepatogastroenterology. 2002;49:1161-1166. [PubMed] |
| 174. | Orloff MJ, Orloff MS, Orloff SL, Haynes KS. Treatment of bleeding from portal hypertensive gastropathy by portacaval shunt. Hepatology. 1995;21:1011-1017. [PubMed] |
| 175. | Urata J, Yamashita Y, Tsuchigame T, Hatanaka Y, Matsukawa T, Sumi S, Matsuno Y, Takahashi M. The effects of transjugular intrahepatic portosystemic shunt on portal hypertensive gastropathy. J Gastroenterol Hepatol. 1998;13:1061-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 56] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 176. | Pérez-Ayuso RM, Piqué JM, Bosch J, Panés J, González A, Pérez R, Rigau J, Quintero E, Valderrama R, Viver J. Propranolol in prevention of recurrent bleeding from severe portal hypertensive gastropathy in cirrhosis. Lancet. 1991;337:1431-1434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 166] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 177. | Dulai GS, Jensen DM, Kovacs TO, Gralnek IM, Jutabha R. Endoscopic treatment outcomes in watermelon stomach patients with and without portal hypertension. Endoscopy. 2004;36:68-72. [PubMed] |
| 178. | Gostout CJ, Viggiano TR, Ahlquist DA, Wang KK, Larson MV, Balm R. The clinical and endoscopic spectrum of the watermelon stomach. J Clin Gastroenterol. 1992;15:256-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 169] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 179. | Burak KW, Lee SS, Beck PL. Portal hypertensive gastropathy and gastric antral vascular ectasia (GAVE) syndrome. Gut. 2001;49:866-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 96] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 180. | Ito M, Uchida Y, Kamano S, Kawabata H, Nishioka M. Clinical comparisons between two subsets of gastric antral vascular ectasia. Gastrointest Endosc. 2001;53:764-770. [PubMed] |
| 181. | Fuccio L, Mussetto A, Laterza L, Eusebi LH, Bazzoli F. Diagnosis and management of gastric antral vascular ectasia. World J Gastrointest Endosc. 2013;5:6-13. [PubMed] |
| 182. | Quintero E, Pique JM, Bombi JA, Bordas JM, Sentis J, Elena M, Bosch J, Rodes J. Gastric mucosal vascular ectasias causing bleeding in cirrhosis. A distinct entity associated with hypergastrinemia and low serum levels of pepsinogen I. Gastroenterology. 1987;93:1054-1061. [PubMed] |
| 183. | Payen JL, Calès P, Voigt JJ, Barbe S, Pilette C, Dubuisson L, Desmorat H, Vinel JP, Kervran A, Chayvialle JA. Severe portal hypertensive gastropathy and antral vascular ectasia are distinct entities in patients with cirrhosis. Gastroenterology. 1995;108:138-144. [PubMed] |
| 184. | Watson M, Hally RJ, McCue PA, Varga J, Jiménez SA. Gastric antral vascular ectasia (watermelon stomach) in patients with systemic sclerosis. Arthritis Rheum. 1996;39:341-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 68] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 185. | Spahr L, Villeneuve JP, Dufresne MP, Tassé D, Bui B, Willems B, Fenyves D, Pomier-Layrargues G. Gastric antral vascular ectasia in cirrhotic patients: absence of relation with portal hypertension. Gut. 1999;44:739-742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 116] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 186. | Vincent C, Pomier-Layrargues G, Dagenais M, Lapointe R, Létourneau R, Roy A, Paré P, Huet PM. Cure of gastric antral vascular ectasia by liver transplantation despite persistent portal hypertension: a clue for pathogenesis. Liver Transpl. 2002;8:717-720. [PubMed] |
| 187. | van Cutsem E, Rutgeerts P, Vantrappen G. Treatment of bleeding gastrointestinal vascular malformations with oestrogen-progesterone. Lancet. 1990;335:953-955. [PubMed] |
| 188. | Manning RJ. Estrogen/progesterone treatment of diffuse antral vascular ectasia. Am J Gastroenterol. 1995;90:154-156. [PubMed] |
| 189. | Moss SF, Ghosh P, Thomas DM, Jackson JE, Calam J. Gastric antral vascular ectasia: maintenance treatment with oestrogen-progesterone. Gut. 1992;33:715-717. [PubMed] |
| 190. | Narod SA. Hormone replacement therapy and the risk of breast cancer. Nat Rev Clin Oncol. 2011;8:669-676. [PubMed] |
| 191. | Nardone G, Rocco A, Balzano T, Budillon G. The efficacy of octreotide therapy in chronic bleeding due to vascular abnormalities of the gastrointestinal tract. Aliment Pharmacol Ther. 1999;13:1429-1436. [PubMed] |
| 192. | Barbara G, De Giorgio R, Salvioli B, Stanghellini V, Corinaldesi R. Unsuccessful octreotide treatment of the watermelon stomach. J Clin Gastroenterol. 1998;26:345-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 193. | Sebastian S, McLoughlin R, Qasim A, O’Morain CA, Buckley MJ. Endoscopic argon plasma coagulation for the treatment of gastric antral vascular ectasia (watermelon stomach): long-term results. Dig Liver Dis. 2004;36:212-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 194. | Yusoff I, Brennan F, Ormonde D, Laurence B. Argon plasma coagulation for treatment of watermelon stomach. Endoscopy. 2002;34:407-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 80] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 195. | Bourke MJ, Hope RL, Boyd P, Gillespie PE, Ward M, Cowen AE, Williams SJ. Endoscopic laser therapy for watermelon stomach. J Gastroenterol Hepatol. 1996;11:832-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 29] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 196. | Gostout CJ, Ahlquist DA, Radford CM, Viggiano TR, Bowyer BA, Balm RK. Endoscopic laser therapy for watermelon stomach. Gastroenterology. 1989;96:1462-1465. [PubMed] |
| 197. | Potamiano S, Carter CR, Anderson JR. Endoscopic laser treatment of diffuse gastric antral vascular ectasia. Gut. 1994;35:461-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 198. | Sargeant IR, Loizou LA, Rampton D, Tulloch M, Bown SG. Laser ablation of upper gastrointestinal vascular ectasias: long term results. Gut. 1993;34:470-475. [PubMed] |
| 199. | Liberski SM, McGarrity TJ, Hartle RJ, Varano V, Reynolds D. The watermelon stomach: long-term outcome in patients treated with Nd: YAG laser therapy. Gastrointest Endosc. 1994;40:584-587. [PubMed] |
| 200. | Roman S, Saurin JC, Dumortier J, Perreira A, Bernard G, Ponchon T. Tolerance and efficacy of argon plasma coagulation for controlling bleeding in patients with typical and atypical manifestations of watermelon stomach. Endoscopy. 2003;35:1024-1028. [PubMed] |
| 201. | Wahab PJ, Mulder CJ, den Hartog G, Thies JE. Argon plasma coagulation in flexible gastrointestinal endoscopy: pilot experiences. Endoscopy. 1997;29:176-181. [PubMed] |
| 202. | Sato T, Yamazaki K, Akaike J. Endoscopic band ligation versus argon plasma coagulation for gastric antral vascular ectasia associated with liver diseases. Dig Endosc. 2012;24:237-242. [PubMed] |
| 203. | Sinha SK, Udawat HP, Varma S, Lal A, Rana SS, Bhasin DK. Watermelon stomach treated with endoscopic band ligation. Gastrointest Endosc. 2006;64:1028-1031. [PubMed] |
| 204. | Wells CD, Harrison ME, Gurudu SR, Crowell MD, Byrne TJ, Depetris G, Sharma VK. Treatment of gastric antral vascular ectasia (watermelon stomach) with endoscopic band ligation. Gastrointest Endosc. 2008;68:231-236. [PubMed] |
| 205. | Kamath PS, Lacerda M, Ahlquist DA, McKusick MA, Andrews JC, Nagorney DA. Gastric mucosal responses to intrahepatic portosystemic shunting in patients with cirrhosis. Gastroenterology. 2000;118:905-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 118] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 206. | Novitsky YW, Kercher KW, Czerniach DR, Litwin DE. Watermelon stomach: pathophysiology, diagnosis, and management. J Gastrointest Surg. 2003;7:652-661. [PubMed] |
| 207. | Kinkhabwala M, Mousavi A, Iyer S, Adamsons R. Bleeding ileal varicosity demonstrated by transhepatic portography. AJR Am J Roentgenol. 1977;129:514-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 61] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 208. | Sato T, Akaike J, Toyota J, Karino Y, Ohmura T. Clinicopathological features and treatment of ectopic varices with portal hypertension. Int J Hepatol. 2011;2011:960720. [PubMed] |
| 209. | Dhiman RK, Saraswat VA, Choudhuri G, Sharma BC, Pandey R, Naik SR. Endosonographic, endoscopic, and histologic evaluation of alterations in the rectal venous system in patients with portal hypertension. Gastrointest Endosc. 1999;49:218-227. [PubMed] |
| 210. | Bhasin DK, Sharma BC, Sriram PV, Makharia G, Singh K. Endoscopic management of bleeding ectopic varices with histoacryl. HPB Surg. 1999;11:171-173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 211. | Chen WC, Hou MC, Lin HC, Chang FY, Lee SD. An endoscopic injection with N-butyl-2-cyanoacrylate used for colonic variceal bleeding: a case report and review of the literature. Am J Gastroenterol. 2000;95:540-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 212. | Gertsch P, Blumgart LH. Cure of a bleeding duodenal varix by sclerotherapy. Br J Surg. 1988;75:717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 213. | Tsuji H, Okano H, Fujino H, Satoh T, Kodama T, Takino T, Yoshimura N, Aikawa I, Oka T, Tsuchihashi Y. A case of endoscopic injection sclerotherapy for a bleeding duodenal varix. Gastroenterol Jpn. 1989;24:60-64. [PubMed] |
| 214. | Yoshida Y, Imai Y, Nishikawa M, Nakatukasa M, Kurokawa M, Shibata K, Shimomukai H, Shimano T, Tokunaga K, Yonezawa T. Successful endoscopic injection sclerotherapy with N-butyl-2-cyanoacrylate following the recurrence of bleeding soon after endoscopic ligation for ruptured duodenal varices. Am J Gastroenterol. 1997;92:1227-1229. [PubMed] |
| 215. | Rahmani O, Wolpert LM, Drezner AD. Distal inferior mesenteric veins to renal vein shunt for treatment of bleeding anorectal varices: case report and review of literature. J Vasc Surg. 2002;36:1264-1266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 216. | Wang CS, Jeng LB, Chen MF. Duodenal variceal bleeding--successfully treated by mesocaval shunt after failure of sclerotherapy. Hepatogastroenterology. 1995;42:59-61. [PubMed] |
| 217. | Johnson PA, Laurin J. Transjugular portosystemic shunt for treatment of bleeding stomal varices. Dig Dis Sci. 1997;42:440-442. [PubMed] |
| 218. | McChesney L, Jensen D, Matalon T, Ganger D, Sankary H, Foster P, Williams JW. Duodenal varices: a case report and review of the literature. HPB Surg. 1995;9:31-35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 219. | Wong RC, Berg CL. Portal hypertensive stomapathy: a newly described entity and its successful treatment by placement of a transjugular intrahepatic portosystemic shunt. Am J Gastroenterol. 1997;92:1056-1057. [PubMed] |
| 220. | Shibata D, Brophy DP, Gordon FD, Anastopoulos HT, Sentovich SM, Bleday R. Transjugular intrahepatic portosystemic shunt for treatment of bleeding ectopic varices with portal hypertension. Dis Colon Rectum. 1999;42:1581-1585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 85] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 221. | Akazawa Y, Murata I, Yamao T, Yamakawa M, Kawano Y, Nomura N, Isomoto H, Mizuta Y, Murase K, Kohno S. Successful management of bleeding duodenal varices by endoscopic variceal ligation and balloon-occluded retrograde transvenous obliteration. Gastrointest Endosc. 2003;58:794-797. [PubMed] |