Published online May 28, 2012. doi: 10.3748/wjg.v18.i20.2586
Revised: February 15, 2012
Accepted: March 9, 2012
Published online: May 28, 2012
A 69-year-old man was admitted to our hospital in October 2003, for further examination of two liver tumors. He was diagnosed with hepatocellular carcinoma (HCC) arising from decompensated hepatitis B virus (HBV)-related cirrhosis. Long-term lamivudine administration improved liver function dramatically despite repeated treatment for HCC. His Child-Pugh score was 9 points at start of lamivudine treatment, improving to 5 points after 1 year. His indocyanine green at 15 min after injection test score was 48% before lamivudine treatment, improving to 22% after 2 years and to 5% after 4 years. Radiofrequency ablation controlled the HCC foci and maintained his liver function. In April 2009, abdominal computed tomography revealed a tumor thrombus in the right portal vein. Since his indocyanine green test results had improved to less than 10%, we performed a right hepatectomy, which was successful. To our knowledge, there have been no documented reports of patients undergoing successful right hepatectomy for HCC arising from decompensated cirrhosis. The findings observed in our patient indicate the importance of nucleoside analogs for treating HBV-related HCC.
- Citation: Honda K, Seike M, Maehara SI, Tahara K, Anai H, Moriuchi A, Muro T. Lamivudine treatment enabling right hepatectomy for hepatocellular carcinoma in decompensated cirrhosis. World J Gastroenterol 2012; 18(20): 2586-2590
- URL: https://www.wjgnet.com/1007-9327/full/v18/i20/2586.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i20.2586
The hepatitis B virus (HBV) infects more than 400 million people worldwide[1] and is an important risk factor for the development of hepatocellular carcinoma (HCC). In Japan, about 1% of individuals in the general population are HBV carriers, accounting for about 14% of patients with liver cirrhosis[2] and 15%-20% of those with HCC[3,4]. The prognosis of patients with HCC arising from chronic liver disease is dependent not only on tumor factors but on hepatic functional reserve. Depending on patient age, liver transplantation may be a good therapeutic option in patients with poor functional reserve. Lamivudine treatment is beneficial in patients with HBV-related HCC because it contributes to improvement of remnant liver function. We describe here a patient with decompensated HBV-related cirrhosis who developed HCC. Lamivudine therapy improved liver function and enabled a right hepatectomy 5 years later.
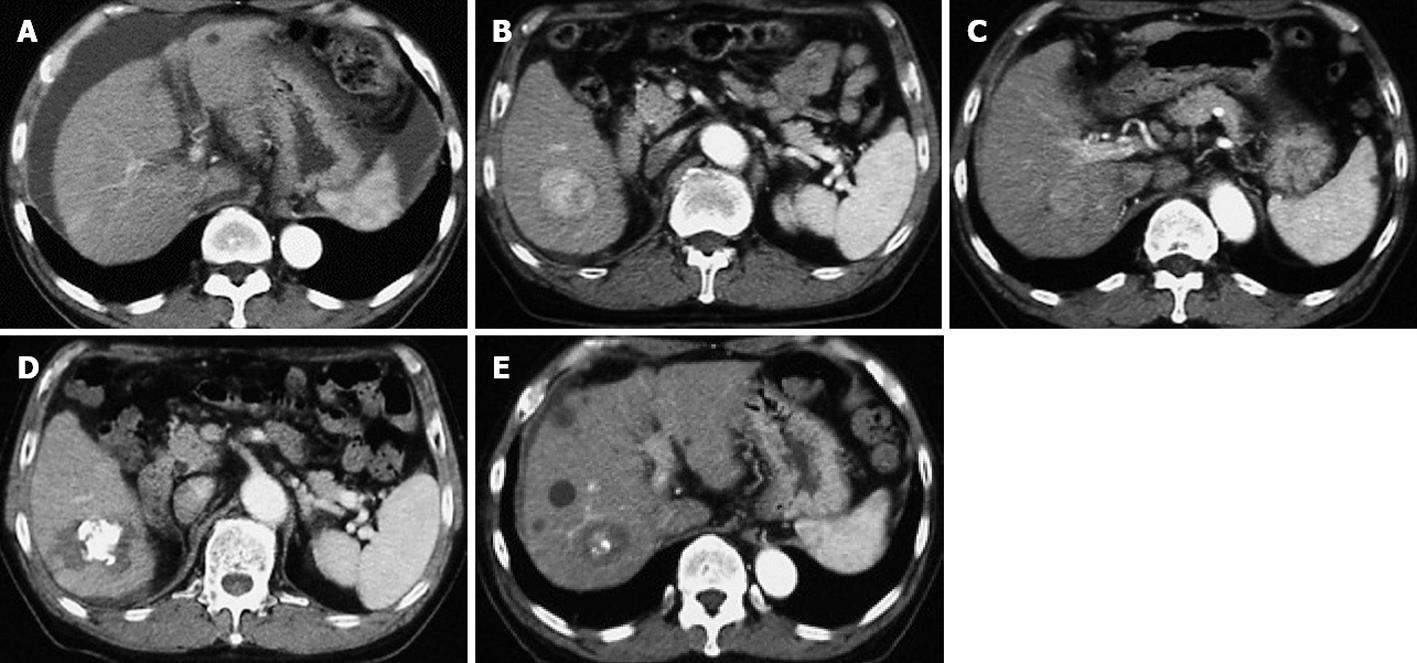
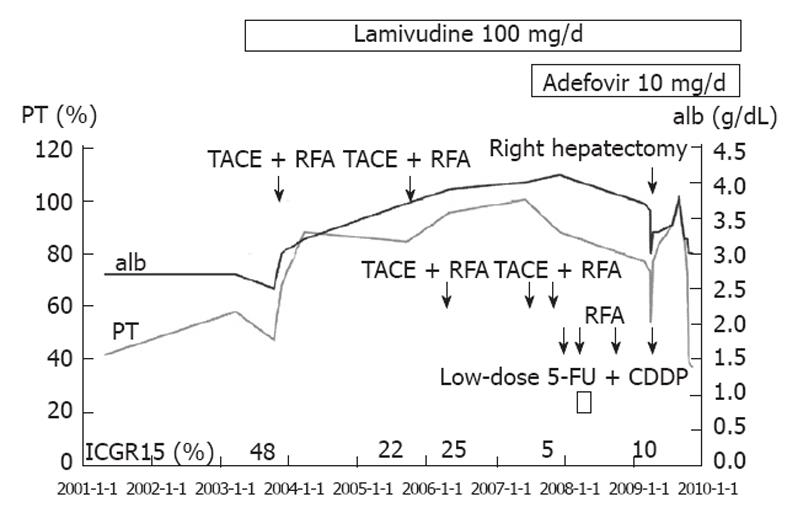
A 69-year old man was admitted to our hospital in October 2003 for examination of two liver tumors. He had been diagnosed with hepatitis B in 1994 and treated with glycyrrhizin. His liver function deteriorated gradually, with ascites appearing in May 2001. He was first admitted to our hospital for treatment of intractable ascites (Figure 1A). Laboratory tests showed that his serum albumin (alb) concentration was 2.7 g/dL, his total bilirubin (T-Bil) was 2.8 mg/dL, his aspartate aminotransferase (AST) was 54 IU/L, his alanine aminotransferase (ALT) was 43 IU/L, and his prothrombin time (PT) was 42%. Administration of diuretic drugs was not effective, but treatment with a preparation of albumin resulted in the disappearance of ascites 1 mo later. Afterward, the ascites was kept under control by administration of diuretics. In October 2003, a computed tomographic (CT) scan of the abdomen revealed two HCCs (4.5 and 2.5 cm in diameter) in the right hepatic lobe (Figure 1B, C). Laboratory tests showed alb 2.5 g/dL, T-Bil 2.4 mg/dL, AST 152 IU/L, ALT 98 IU/L, PT 47%, indocyanine green at 15 min after injection (ICGR15) 48%, alpha-fetoprotein 444 ng/mL, and protein induced by vitamine K absence or antagonist II <10 mAU/mL. He was positive for HBe antigen, negative for HBe antibody, and had an HBV-DNA viral load of 6.7 log copies/mL. Beginning in November 2003, he was treated with 100 mg/d lamivudine. The two HCCs were treated by transcatheter arterial chemoembolization (TACE) and radiofrequency ablation (RFA) (Figure 1D, E). Both tumors were treated successfully and the patient’s liver function recovered gradually after initiation of lamivudine treatment. In September 2005, an abdominal CT scan revealed a recurrent HCC, located near one of the previously treated tumors; this lesion was treated successfully with TACE and RFA. At this time, laboratory tests showed alb 3.7 g/dL, T-Bil 0.7 mg/dL, AST 23 IU/L, ALT 22 IU/L, PT 84% and ICGR15 22%. All HCC treatments were based on clinical practice guidelines in Japan[5], with the patient providing informed consent.
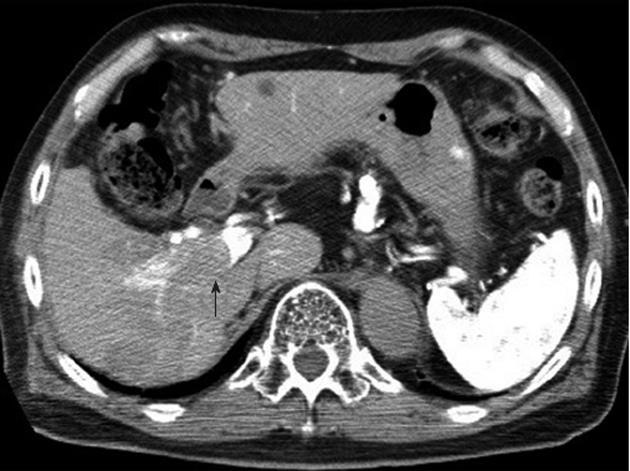
In May 2006, two HCC recurrences were detected in the right liver lobe and treated with TACE and RFA. Laboratory tests showed good liver function, alb 3.9 g/dL, T-Bil 1.1 mg/dL, AST 22 IU/L, ALT 11 IU/L, PT 95% and ICGR15 25%. In June 2007, a recurrent HCC was treated with TACE and RFA. Liver function was also excellent at this time (alb 4.0 g/dL, T-Bil 0.7 mg/dL and PT 100%). In September 2007, his viral load had again increased, with breakthrough hepatitis, and the YMDD mutation was detected. Treatment with adefovir dipivoxil plus lamivudine resulted in a gradual reduction in viral load. In December 2007, abdominal CT revealed five HCCs in the right lobe; these were treated by TACE and RFA. The patient was then treated with low-dose cisplatin and 5-fluorouracil infused through the hepatic artery. Laboratory tests showed alb 4.1 g/dL, T-Bil 1.0 mg/dL, AST 38 IU/L, ALT 30 IU/L, PT 88% and ICGR15 5%. Due to the development of a pseudoaneurysm in his hepatic artery, infusion of chemotherapy was discontinued. In March 2009, two HCCs were detected in the right lobe and were treated by RFA. Laboratory tests showed alb 3.7 g/dL, T-Bil 0.7 mg/dL, AST 35 IU/L, ALT 31 IU/L, PT 77% and ICGR15 10%. In April 2009, abdominal CT and CT angiography revealed a tumor thrombus in the right portal vein, but no lesion could be detected in the left lobe (Figure 2). Although he was diagnosed with decompensated cirrhosis, of Child-Pugh C, when first hospitalized, lamivudine treatment improved his liver function sufficiently, with an improvement of ICGR15 results to < 10%, to allow the successful performance of a right hepatectomy in April 2009 (Figure 3A). After liver resection, his AST rose to 1220 IU/L, his T-Bil to 1.9 mg/dL, and his PT decreased to 54%, followed by gradual recovery of liver function. He recovered well and left the hospital 1 mo after surgery.
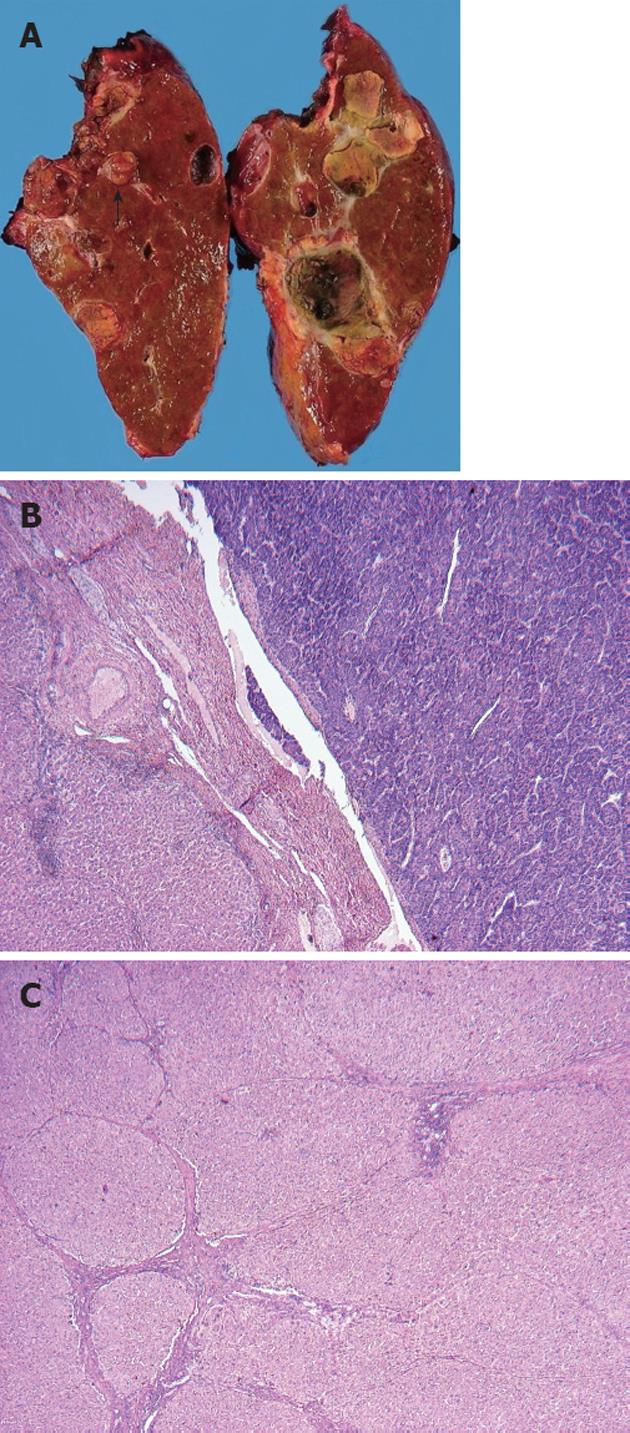
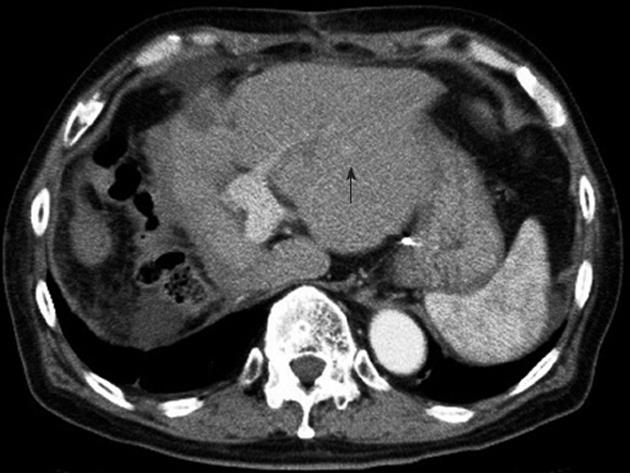
Histologic examination of the extracted specimen showed a moderately differentiated HCC with portal tumor thrombus (Figure 3B) and multiple intrahepatic metastases. Fibrosis of varying extent was observed in the cancer-free area, with some areas showing severe fibrosis with pseudolobules and others showing mild fibrosis (Figure 3C). Abdominal CT in June 2009 suggested the recurrence of HCC in the left lobe (Figure 4), and abdominal angiography revealed multiple HCCs. These tumors were treated by TACE, but this was not effective. The left lobe tumors subsequently enlarged and the patient’s liver function deteriorated gradually. The patient died in December 2009 (Clinical course Figure 5).
Chronic hepatitis B is a progressive liver disease, leading to cirrhosis and HCC[6]. Before antiviral agents became established as effective treatments for hepatitis B, the prognosis of patients with end-stage HBV infection was generally poor. The 5-year survival rates of patients with compensated and decompensated cirrhosis have been reported to be 55%-84%[7,8] and 14%[9], respectively. Lamivudine, an antiviral drug, is an oral nucleoside analog that inhibits DNA synthesis by terminating the nascent proviral DNA chain. It rapidly reduces both serum HBV-DNA and transaminase concentrations[10]. Prolonged viral suppression can result in histological improvement, including the regression of fibrosis[10-13]. Although a subgroup of individuals with extremely advanced disease require urgent transplantation, lamivudine treatment can achieve significant improvement in liver function and reduce the morbidity of many of patients with decompensated cirrhosis[14-19]. At present entecavir is recommended as the primary oral agent for hepatitis B because of its strong antiviral effects and low resistance rate, as well as being effective in treating decompensated cirrhosis[20]. Long-term lamivudine monotherapy can induce the emergence of resistant viruses with an amino acid substitution in the YMDD motif of the viral DNA polymerase[21]. Particularly in patients with decompensated cirrhosis, breakthrough hepatitis resulting from such a mutation may lead to hepatic failure[22] if other antiviral drugs such as adefovir dipivoxil[23,24] are not administered. Long-term treatment with lamivudine has been reported to reduce the incidence of HCC[25,26]. In addition, lamivudine has been found to improve liver function[27,28] and survival[29] in patients with HBV-related HCC after initial treatment of HCC. We have described a patient with HCC arising from decompensated HBV-related cirrhosis. Long-term lamivudine treatment improved his remnant liver function dramatically, despite repeated TACE and RFA sessions for HCC. Although his Child-Pugh score at the start of lamivudine treatment was 9 points, it improved up to 5 points 1 year later. Moreover, he scored 48% on an ICGR15 test performed before his first treatment for HCC, but this score improved to 22% after 2 years and to 5% after 4 years, with the latter considered safe for the performance of a right hepatectomy[30].
Despite repeated RFA, the liver function of this patient was well maintained. Generally, RFA has been regarded as safe and effective for HCC, and has been found to maintain liver function[31-33]. In our patient, lamivudine and RFA were effective in maintaining liver function. A previous case report described a patient with decompensated HBV-related cirrhosis, who, following lamivudine treatment, underwent a hepatectomy for HCC after liver function had improved[34]. That patient, however, underwent a partial hepatectomy for a small HCC. To our knowledge, no prior report has described a successful right hepatectomy for HCC arising from decompensated HBV-associated liver cirrhosis. The findings reported in this patient indicate the importance of nucleoside analogs for treating HBV-related HCC.
In conclusion, we found that lamivudine treatment was beneficial for our patient with decompensated HBV-related cirrhosis and HCC, increasing the likelihood of treatment for HCC.
Peer reviewer: Dimitrios E Giakoustidis, Professor, Department of Transplantation, Aristotle University of Thessaloniki, 49 Konstantinoupoleos Street, 54642 Thessaloniki, Greece
S- Editor Gou SX L- Editor A E- Editor Li JY
| 1. | Lai CL, Ratziu V, Yuen MF, Poynard T. Viral hepatitis B. Lancet. 2003;362:2089-2094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 595] [Cited by in RCA: 590] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 2. | Michitaka K, Nishiguchi S, Aoyagi Y, Hiasa Y, Tokumoto Y, Onji M. Etiology of liver cirrhosis in Japan: a nationwide survey. J Gastroenterol. 2010;45:86-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 107] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 3. | Edamoto Y, Tani M, Kurata T, Abe K. Hepatitis C and B virus infections in hepatocellular carcinoma. Analysis of direct detection of viral genome in paraffin embedded tissues. Cancer. 1996;77:1787-1791. [PubMed] |
| 4. | Taura N, Fukushima N, Yastuhashi H, Takami Y, Seike M, Watanabe H, Mizuta T, Sasaki Y, Nagata K, Tabara A. The incidence of hepatocellular carcinoma associated with hepatitis C infection decreased in Kyushu area. Med Sci Monit. 2011;17:PH7-P11. [PubMed] |
| 5. | Makuuchi M, Kokudo N. Clinical practice guidelines for hepatocellular carcinoma: the first evidence based guidelines from Japan. World J Gastroenterol. 2006;12:828-829. [PubMed] |
| 6. | Park BK, Park YN, Ahn SH, Lee KS, Chon CY, Moon YM, Park C, Han KH. Long-term outcome of chronic hepatitis B based on histological grade and stage. J Gastroenterol Hepatol. 2007;22:383-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 74] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 7. | Weissberg JI, Andres LL, Smith CI, Weick S, Nichols JE, Garcia G, Robinson WS, Merigan TC, Gregory PB. Survival in chronic hepatitis B. An analysis of 379 patients. Ann Intern Med. 1984;101:613-616. [PubMed] |
| 8. | Realdi G, Fattovich G, Hadziyannis S, Schalm SW, Almasio P, Sanchez-Tapias J, Christensen E, Giustina G, Noventa F. Survival and prognostic factors in 366 patients with compensated cirrhosis type B: a multicenter study. The Investigators of the European Concerted Action on Viral Hepatitis (EUROHEP). J Hepatol. 1994;21:656-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 206] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 9. | de Jongh FE, Janssen HL, de Man RA, Hop WC, Schalm SW, van Blankenstein M. Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology. 1992;103:1630-1635. [PubMed] |
| 10. | Lai CL, Chien RN, Leung NW, Chang TT, Guan R, Tai DI, Ng KY, Wu PC, Dent JC, Barber J. A one-year trial of lamivudine for chronic hepatitis B. Asia Hepatitis Lamivudine Study Group. N Engl J Med. 1998;339:61-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1381] [Cited by in RCA: 1347] [Article Influence: 49.9] [Reference Citation Analysis (0)] |
| 11. | Honkoop P, de Man RA, Zondervan PE, Schalm SW. Histological improvement in patients with chronic hepatitis B virus infection treated with lamivudine. Liver. 1997;17:103-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 50] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 12. | Suzuki Y, Kumada H, Ikeda K, Chayama K, Arase Y, Saitoh S, Tsubota A, Kobayashi M, Koike M, Ogawa N. Histological changes in liver biopsies after one year of lamivudine treatment in patients with chronic hepatitis B infection. J Hepatol. 1999;30:743-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 101] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 13. | Dienstag JL, Goldin RD, Heathcote EJ, Hann HW, Woessner M, Stephenson SL, Gardner S, Gray DF, Schiff ER. Histological outcome during long-term lamivudine therapy. Gastroenterology. 2003;124:105-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 574] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 14. | Kapoor D, Guptan RC, Wakil SM, Kazim SN, Kaul R, Agarwal SR, Raisuddin S, Hasnain SE, Sarin SK. Beneficial effects of lamivudine in hepatitis B virus-related decompensated cirrhosis. J Hepatol. 2000;33:308-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 113] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Villeneuve JP, Condreay LD, Willems B, Pomier-Layrargues G, Fenyves D, Bilodeau M, Leduc R, Peltekian K, Wong F, Margulies M. Lamivudine treatment for decompensated cirrhosis resulting from chronic hepatitis B. Hepatology. 2000;31:207-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 295] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 16. | Yao FY, Bass NM. Lamivudine treatment in patients with severely decompensated cirrhosis due to replicating hepatitis B infection. J Hepatol. 2000;33:301-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 138] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 17. | Fontana RJ, Hann HW, Perrillo RP, Vierling JM, Wright T, Rakela J, Anschuetz G, Davis R, Gardner SD, Brown NA. Determinants of early mortality in patients with decompensated chronic hepatitis B treated with antiviral therapy. Gastroenterology. 2002;123:719-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 180] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 18. | Hann HW, Fontana RJ, Wright T, Everson G, Baker A, Schiff ER, Riely C, Anschuetz G, Gardner SD, Brown N. A United States compassionate use study of lamivudine treatment in nontransplantation candidates with decompensated hepatitis B virus-related cirrhosis. Liver Transpl. 2003;9:49-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 19. | Tseng PL, Lu SN, Tung HD, Wang JH, Changchien CS, Lee CM. Determinants of early mortality and benefits of lamivudine therapy in patients with hepatitis B virus-related decompensated liver cirrhosis. J Viral Hepat. 2005;12:386-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Shim JH, Lee HC, Kim KM, Lim YS, Chung YH, Lee YS, Suh DJ. Efficacy of entecavir in treatment-naïve patients with hepatitis B virus-related decompensated cirrhosis. J Hepatol. 2010;52:176-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 210] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 21. | Liaw YF, Chien RN, Yeh CT, Tsai SL, Chu CM. Acute exacerbation and hepatitis B virus clearance after emergence of YMDD motif mutation during lamivudine therapy. Hepatology. 1999;30:567-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 403] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 22. | Nishida T, Kobashi H, Fujioka S, Fujio K, Takaguchi K, Ikeda H, Kawaguchi M, Ando M, Araki Y, Higashi T. A prospective and comparative cohort study on efficacy and drug resistance during long-term lamivudine treatment for various stages of chronic hepatitis B and cirrhosis. J Gastroenterol Hepatol. 2008;23:794-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 23. | Perrillo R, Hann HW, Mutimer D, Willems B, Leung N, Lee WM, Moorat A, Gardner S, Woessner M, Bourne E. Adefovir dipivoxil added to ongoing lamivudine in chronic hepatitis B with YMDD mutant hepatitis B virus. Gastroenterology. 2004;126:81-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 305] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 24. | Peters MG, Hann Hw H, Martin P, Heathcote EJ, Buggisch P, Rubin R, Bourliere M, Kowdley K, Trepo C, Gray Df D. Adefovir dipivoxil alone or in combination with lamivudine in patients with lamivudine-resistant chronic hepatitis B. Gastroenterology. 2004;126:91-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 423] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 25. | Liaw YF, Sung JJ, Chow WC, Farrell G, Lee CZ, Yuen H, Tanwandee T, Tao QM, Shue K, Keene ON, Dixon JS, Gray DF, Sabbat J. Lamivudine for patients with chronic hepatitis B and advanced liver disease. N Engl J Med. 2004;351:1521-1531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1739] [Cited by in RCA: 1740] [Article Influence: 82.9] [Reference Citation Analysis (0)] |
| 26. | Matsumoto A, Tanaka E, Rokuhara A, Kiyosawa K, Kumada H, Omata M, Okita K, Hayashi N, Okanoue T, Iino S. Efficacy of lamivudine for preventing hepatocellular carcinoma in chronic hepatitis B: A multicenter retrospective study of 2795 patients. Hepatol Res. 2005;32:173-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 149] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 27. | Kuzuya T, Katano Y, Kumada T, Toyoda H, Nakano I, Hirooka Y, Itoh A, Ishigami M, Hayashi K, Honda T. Efficacy of antiviral therapy with lamivudine after initial treatment for hepatitis B virus-related hepatocellular carcinoma. J Gastroenterol Hepatol. 2007;22:1929-1935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 93] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 28. | Kim JH, Park JW, Koh DW, Lee WJ, Kim CM. Efficacy of lamivudine on hepatitis B viral status and liver function in patients with hepatitis B virus-related hepatocellular carcinoma. Liver Int. 2009;29:203-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 29. | Kubo S, Tanaka H, Takemura S, Yamamoto S, Hai S, Ichikawa T, Kodai S, Shinkawa H, Sakaguchi H, Tamori A. Effects of lamivudine on outcome after liver resection for hepatocellular carcinoma in patients with active replication of hepatitis B virus. Hepatol Res. 2007;37:94-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 75] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 30. | Imamura H, Sano K, Sugawara Y, Kokudo N, Makuuchi M. Assessment of hepatic reserve for indication of hepatic resection: decision tree incorporating indocyanine green test. J Hepatobiliary Pancreat Surg. 2005;12:16-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 255] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 31. | Livraghi T, Solbiati L, Meloni MF, Gazelle GS, Halpern EF, Goldberg SN. Treatment of focal liver tumors with percutaneous radio-frequency ablation: complications encountered in a multicenter study. Radiology. 2003;226:441-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1051] [Cited by in RCA: 932] [Article Influence: 42.4] [Reference Citation Analysis (0)] |
| 32. | Lu DS, Yu NC, Raman SS, Lassman C, Tong MJ, Britten C, Durazo F, Saab S, Han S, Finn R. Percutaneous radiofrequency ablation of hepatocellular carcinoma as a bridge to liver transplantation. Hepatology. 2005;41:1130-1137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 269] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 33. | N'Kontchou G, Mahamoudi A, Aout M, Ganne-Carrié N, Grando V, Coderc E, Vicaut E, Trinchet JC, Sellier N, Beaugrand M. Radiofrequency ablation of hepatocellular carcinoma: long-term results and prognostic factors in 235 Western patients with cirrhosis. Hepatology. 2009;50:1475-1483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 356] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 34. | Nakanishi S, Michitaka K, Miyake T, Hidaka S, Yoshino I, Konishi I, Iuchi H, Horiike N, Onji M. Decompensated hepatitis B virus-related cirrhosis successfully treated with lamivudine allowing surgery for hepatocellular carcinoma. Intern Med. 2003;42:416-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |