Published online May 7, 2009. doi: 10.3748/wjg.15.2049
Revised: March 30, 2009
Accepted: April 6, 2009
Published online: May 7, 2009
Intrahepatic cholestasis of pregnancy (ICP) is a pregnancy-specific liver disorder characterized by maternal pruritus in the third trimester, raised serum bile acids and increased rates of adverse fetal outcomes. The etiology of ICP is complex and not fully understood, but it is likely to result from the cholestatic effects of reproductive hormones and their metabolites in genetically susceptible women. Equally unclear are the mechanisms by which the fetal complications occur. This article reviews the epidemiology, clinical features, diagnosis, etiology and management of ICP.
- Citation: Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol 2009; 15(17): 2049-2066
- URL: https://www.wjgnet.com/1007-9327/full/v15/i17/2049.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.2049
Intrahepatic cholestasis of pregnancy (ICP), which is also known as obstetric cholestasis, is a liver disease of pregnancy associated with raised serum bile acids and increased rates of adverse fetal outcomes.
ICP was originally described in 1883 by Ahlfeld as recurrent jaundice in pregnancy that resolved following delivery. Pruritus was not mentioned in this report, but in subsequent case reports published in the 1950s, severe pruritus with or without jaundice was reported in conjunction with the condition, in addition to complete resolution following delivery and high recurrence rates in subsequent pregnancies[12].
Over the years, ICP has also been described as jaundice in pregnancy, recurrent jaundice in pregnancy, idiopathic jaundice of pregnancy, obstetric hepatosis, hepatosis gestationalis or obstetric cholestasis.
Most authors now agree that ICP should be defined as pruritus with onset in pregnancy, which is associated with abnormal liver function in the absence of other liver disease and which resolves following delivery.
The incidence of ICP varies widely with geographical location and ethnicity (Table 1)[3–27]. It is most common in South America, particularly in Chile, where early reports described an overall incidence of 10%, with higher rates seen in women of Araucanian Indian descent[6]. More recently, this has fallen to approximately 1.5%-4%[28]. The reasons for this decline are unclear but do not appear to reflect changing diagnostic criteria which have become more inclusive in recent studies; while early reports commonly used jaundice to diagnose ICP, more recently, any abnormality in liver function has been used. Instead, it has been proposed that the decline is due to changes in environmental factors, which will be discussed in more detail later in this article. The incidence of ICP is lower in Europe (approximately 1%) and has been stable for many years.
| Country | Prevalence (%) | Year of study | Diagnostic criteria | References |
| Australia | 0.2 | 1964-1966 | P, J, LFT, PR1 | [5] |
| Australia | 1.5 | 1968-1970 | P, J, LFT, R | [8] |
| Australia | 0.2 | 1975-1984 | P, J, LFT, PR; SBA (from 1982) | [3] |
| Bolivia | 9.2 | 1976 | P, J, B, LD | [7] |
| Aimaras | 13.8 | |||
| Quechas | 4.3 | |||
| Caucasians | 7.8 | |||
| Mixed Indian | 7.3 | |||
| Canada | 0.07 | 1963-1976 | J, P, LFT, LD, R | [4] |
| Chile | 1974-1975 | P, J, B, LD | [6] | |
| Aimaras | 11.8 | |||
| Araucanian | 27.6 | |||
| Caucasian | 15.1 | |||
| Chile | 4.7 | NA | P, SBA (10 &mgr;mol/L), B, LFT | [10] |
| Chile | 6.5 | 1988-1990 | P, LD2 | [18] |
| China | 0.32 | 1981-1983 | J, LFT, B, SBA, LD, R3 | [12] |
| Chongqing | ||||
| China | 0.05 | 2003-2005 | LFT, SBA (11 &mgr;mol/L), B, LD | [14] |
| Hong Kong | ||||
| Finland | 1.1 | 1971-1972 | P, LFT | [13] |
| Finland | 0.54 | 1990-1996 | P, LFT, SBA (8 &mgr;mol/L), LD | [11] |
| Finland | 0.54 | 1994-1998 | P, LFT, SBA (8 &mgr;mol/L), LD | [9] |
| France | 0.2 | 1953-1961 | P, J, LFT, PR, R | [16] |
| France | 0.53 | 1988-1989 | LFT, B, SBA (6 &mgr;mol/L) | [19] |
| India | 0.08 | 2002-2004 | P, LD, LFT | [17] |
| Italy | 0.96 | 1996-1999 | P, LFT, SBA4 | [15] |
| Italy | 1 | 1989-1997 | P, PR, LFT ± SBA | [20] |
| Poland | 1.5 | NA | P, LFT, B, LD | [21] |
| Portugal | 1 | NA | P, SBA, LFT, B, LD5 | [24] |
| Sweden | 1.5 | 1971-1974 | P, LFT, LD | [23] |
| 1 | 1980-1982 | |||
| Sweden | 1.5 | 1999-2002 | P, SBA (10 &mgr;mol/L), LFT, LD | [25] |
| USA | 0.32 | 1997-1999 | P, SBA or LFT, PR6 | [26] |
| USA | 5.6 | 1997-1998 | P, SBA (20 &mgr;mol/L) | [27] |
| Latina | ||||
| UK | 0.7 | 1995-1997 | P, SBA (14 &mgr;mol/L), LFT, LD | [22] |
| Caucasian | 0.62 | |||
| Indian | 1.24 | |||
| Pakistani | 1.46 |
ICP is more common in the winter months in Finland, Sweden, Chile and Portugal[2324]. A higher incidence is seen in twin pregnancies (20%-22%)[1018] and following in vitro fertilization treatment (2.7% vs 0.7%)[29]. One study has suggested that it is more common in women over the age of 35 years[11]. There is a higher incidence of gallstones in both affected women and their families[3031]. Hepatitis C seropositivity has been reported to be a risk factor for ICP, and may be associated with early onset of the condition[1532]. It has also been suggested that women with ICP have more severe and prolonged emesis, and higher rates of drug sensitivities[33].
The most common presenting symptom of ICP is pruritus that usually presents in the third trimester. This becomes progressively more severe as the pregnancy advances and typically resolves within 48 h of delivery. Pruritus is defined as an unpleasant sensation that evokes the desire to scratch. It most frequently affects the palms of the hands and soles of the feet but it can be generalized or affect other areas of the body. There are no associated dermatological features other than excoriation marks, which may be severe. Many women report that their pruritus worsens at night and may become so extreme that it causes insomnia.
Approximately 80% of affected women present after 30 wk of gestation[3034], but ICP has been reported as early as 8 wk[23].
The relationship between onset of pruritus and development of deranged liver function is not clear. It has been reported that itch may be present either prior to or after abnormal liver function is detected[35], and this may reflect the heterogeneous nature of the condition.
Clinical jaundice is rare, affecting approximately 10%-15% of pregnant women with ICP, and if it does occur, it tends to be mild with bilirubin levels rarely exceeding 100 &mgr;mol/L. Unlike the pruritus, it does not typically deteriorate with advancing gestation[36].
Constitutional symptoms of cholestasis may also be present, including anorexia, malaise and abdominal pain. Pale stools and dark urine have been reported and steatorrhea may occur[37]. Theoretically steatorrhea is associated with an increased risk of post-partum haemorrhage as a result of malabsorption of vitamin K, although there are only a small number of reports of this complication in the literature[38]. Steatorrhea may respond to treatment with pancreatic enzymes.
There have been some reports of the co-existence of ICP with other pregnancy-related disorders including pre-eclampsia[1439–41], acute fatty liver of pregnancy[3042], and gestational diabetes[39]. This reflects the etiological heterogeneity of the condition and thus it is important to exclude other causes of hepatic impairment in women who present with cholestasis in pregnancy.
ICP is not typically associated with ongoing hepatic impairment after pregnancy and the biochemical abnormalities normally resolve within 2-8 wk of delivery. There are a few case reports of a more prolonged course with biochemical abnormalities lasting up to 34, 45 and 82 wk postpartum[4344]. In women with continued liver dysfunction it is important to exclude alternative underlying diagnoses. In the majority of women, ICP recurs in subsequent pregnancies, but disease severity cannot be predicted by the course in previous pregnancies.
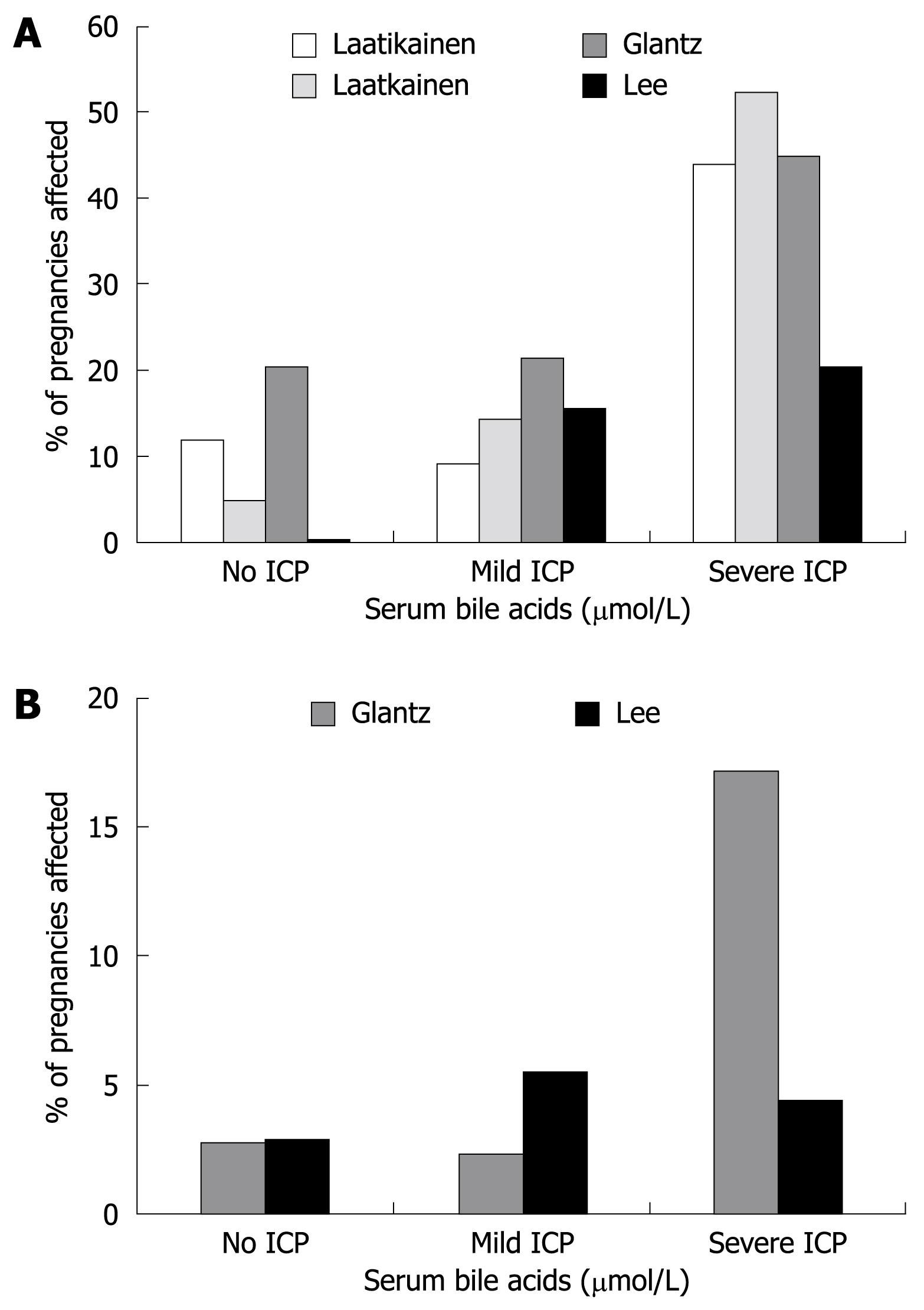
There is considerable debate in the literature about the extent of the ICP-associated fetal risk. There are consistent reports of adverse fetal outcomes in association with the condition[4546], although most studies are not sufficiently large to allow accurate quantification of the frequency of the complications. Many studies have tried to correlate maternal serum biochemistry with fetal outcomes and one series reported higher rates of fetal complications in women with jaundice compared to those with pruritus alone[38]. Bile acids have been repeatedly implicated in the etiology of the fetal disease, and the sensitivity of bile acids as a predictive marker of fetal risk has been examined in several studies involving small numbers of cases[18394748]. A recent, more definitive study from Sweden reported that there was a 1%-2% increase in risk of spontaneous preterm labour, asphyxial events (defined as operative delivery due to asphyxia, Apgar score < 7 at 5 min or arterial cord pH < 7.05) or meconium staining of the amniotic fluid and/or placenta and membranes for every additional µmol/L of maternal serum bile acids[25]. This study also reported no increase in adverse outcomes if the maternal fasting serum bile acids were below 40 &mgr;mol/L, leading the authors to suggest there is no increased risk to the fetus with mild ICP according to this definition. This result was generally consistent with the findings of other smaller studies in Finnish and American Latina populations[47–49] (Figure 1), although the magnitude of the effect varied in different studies, possibly relating to variations in management strategy, maternal ethnicity and study design. The reported incidence of each adverse fetal outcome is shown in Table 2.
| Study period | Number of cases (controls) | Preterm delivery (< 37 wk) | Abnormal CTG (timing) | Apgar score ≤7 (min) | Meconium staining of amniotic fluid | PPH (> 500 mL) | References |
| 1951-1983 | 100 (156) | 38% | - | - | - | 7% | [12] |
| 38% U | |||||||
| 1963-1976 | 42 (42) | 39% | - | - | - | 19% | [4] |
| 39% S | |||||||
| 1965-1974 | 56 | 36% | 14% (NS) | - | 27% | 9% | [38] |
| 36% S | 8 B | NS | |||||
| 1971-1972 | 116 (116) | - | 19% (labour) | 7% (1) | 28% | - | [13] |
| 3% LD | 3% (5) | 7% < 37 wk | |||||
| 16% B or T | 21% > 37 wk | ||||||
| 1971-1974 | 100 (100) | - | - | 10% (1) | 12% | - | [23] |
| 8% (5) | NS | ||||||
| 1975-1984 | 83 | 44% | - | - | 45% | - | [46] |
| 44% S | 10% < 37 wk | ||||||
| 35% > 37 wk | |||||||
| 1979-1981 | 18 | - | - | - | 58.30% | 22% | [39] |
| NS | |||||||
| 1980–1981 | 117 | - | 14% (antepartum) | 10% (NS) | 16.20% | - | [48] |
| 4% (labour) | NS | ||||||
| 1988-1990 | 320 (320) | 19% | 12.8% (NS) | 8% (1) | 38% | - | [18] |
| 12% S | 2% (5) | 13% < 37 wk | |||||
| 7% I | 25% > 37 wk | ||||||
| 1988-1995 | 79 (792) | 14% | 2% (NS) | None | 44% | - | [57] |
| 14% S | 2% B | NS | |||||
| 1989-1995 | 50 | 60% | - | - | - | 2% | [56] |
| 60% U | |||||||
| 1989-1997 | 206 | 27% | - | 1% (5) | 21% | - | [20] |
| 4% S | 5% < 37 wk | ||||||
| 23% I | 16% > 37 wk | ||||||
| 1990-1996 | 91 (16, 818) | 14% | 20% (labour) | 8% (1) | 15% | - | [11] |
| 14% U | 2% (5) | NS | |||||
| 1999-2001 | 70 | 17% | - | - | 14% | 17% | [30] |
| 6% S | 4% < 37 wk | ||||||
| 11% I | 10% > 37 wk | ||||||
| 1999-2002 | 690 (44, 792) | 12% | 7%3 | 7%3 | 25% | - | [25] |
| 12% U | |||||||
| 1999-20031 | 352 | 38% | - | - | - | - | [31] |
| 16% S | |||||||
| 22% I | |||||||
| 2000-2007 | 122 | 4% | - | 8% (1) | 13% | 7% | [49] |
| 4% S | 2% (5) | ||||||
| 2003-2005 | 8 | 50% | - | - | 62% | - | [14] |
| 50% U | NS |
Meconium staining of the amniotic fluid: The incidence of meconium staining of amniotic fluid (MSAF) in normal term pregnancies is approximately 15% and is considered to be a sign of fetal distress. In ICP, MSAF has been reported in 16%-58%[3839] of all cases and up to 100% of cases affected by intrauterine death (IUD)[45]. The group in which the incidence of MSAF is particularly striking is women who had amnioscopy or amniocentesis at approximately 37 wk of gestation, as the rates are significantly higher than in controls at this time[20]. The frequency of MSAF is greater in pregnancies with higher reported levels of maternal serum bile acids[2547–49] (Figure 1).
Cardiotocography (CTG) abnormalities: Both ante- and intrapartum CTG abnormalities have been reported in association with ICP, including reduced fetal heart rate variability, tachycardia and bradycardia (< 100 bpm)[13384850]. More recently, a case report has described fetal tachyarrhythmia (220-230 bpm) leading to atrial flutter during labour at 37 wk gestation[51].
Preterm labour: There is an increased risk of spontaneous preterm labour, which has been seen in as many as 60% of deliveries in some studies[4], but most studies report rates of 30%-40% in ICP cases without active management. Reid et al[38] found an overall incidence of 36%, but interestingly this rose to 48% in women with raised bilirubin. Two studies have related the maternal serum bile acid level to the rate of spontaneous preterm delivery[2549]. The rate of this complication was significantly higher in ICP pregnancies with maternal fasting serum bile acids > 40 &mgr;mol/L in the larger study of Swedish ICP cases[25]. However it was not higher in pregnancies with mildly raised (< 20 &mgr;mol/L) or more markedly raised maternal serum bile acids in a study of American Latina cases[49].
In more recent studies, the majority of preterm deliveries are iatrogenic (Table 2), which reflects the relatively frequent practice of electively delivering ICP cases at around 37 wk, with the aim of reducing the risk of fetal complications. There have been concerns raised over whether this in itself carries an increased risk of neonatal morbidity for the fetus. Studies have shown that there is an increased risk of respiratory distress syndrome (RDS) with either induction of labour or elective cesarean section at this stage of gestation[52]. It should be noted that the risk of neonatal respiratory distress is considerably higher with elective cesarean section, and it should be borne in mind that labour is induced in the majority of women with ICP. Also, there are some data to suggest that neonatal respiratory distress following ICP may be a consequence of the disease process. A recent series reported unexpected respiratory distress in association with maternal cholestasis in three infants delivered between 36 and 37 wk gestation with good indices of lung maturity in the amniotic fluid and negative blood, urine and cerebrospinal fluid cultures[53]. The authors therefore proposed that the development of RDS was as a direct consequence of ICP. In follow-up studies, RDS was found to affect 28.6% of newborns from cholestatic pregnancies and high levels of bile acids were found in the bronchoalveolar fluid of 10 infants with RDS[5455].
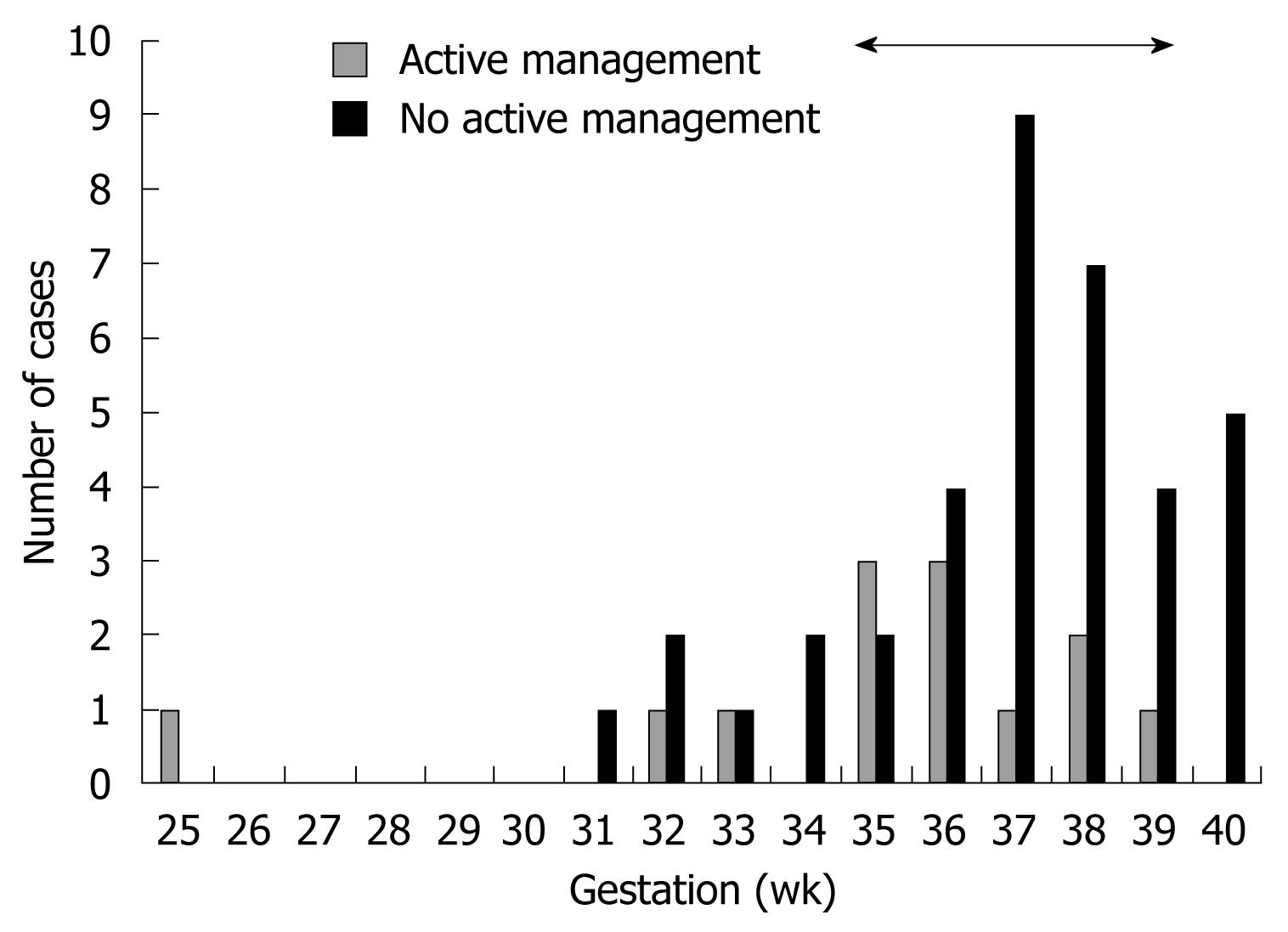
Sudden IUD: Older studies using biochemical abnormalities to diagnose ICP have reported a perinatal mortality rate of 10%-15%[1338]. This has been reduced to 3.5% or less in more recent studies employing policies of active management[11131820253039464856]. The term active management may encompass many different clinical practices, including increased fetal monitoring, frequent biochemical testing, pharmacotherapy with ursodeoxycholic acid (UDCA) or delivery at 37-38 wk gestation. These management protocols are based on evidence showing that stillbirths in ICP tend to cluster around 37-39 wk (Figure 2)[13182325313845–4856–58]. However, there have been reports of stillbirths at less than 37 wk; in one series a fetus died at 32 wk[45] and in another one, twin fetuses died at 31 wk[31].
The risk of adverse fetal outcomes is thought to relate to the maternal serum bile acid level, and a recent study has shown that there is a 1%-2% increased risk for every &mgr;mol/L of bile acid above 40 &mgr;mol/L[25]. It is therefore likely that the risk of IUD is higher in ICP pregnancies with more severe hypercholanemia. However, the same study reported a stillbirth with maternal serum bile acid levels of 27 &mgr;mol/L and there are additional case reports of stillbirths at 39 wk with bile acids of 15 and 21 &mgr;mol/L[4858]. It is not clear how close to the fetal death these blood specimens were taken, and maternal serum bile acid level is high in the majority of IUDs reported in the literature[2547], but these observations illustrate the difficulty encountered by clinicians in using biochemical measurements to dictate delivery strategies for ICP cases.
Other findings: Several studies have shown that there is no increase in the number of small for gestational age infants born to women with ICP[1859]. However, lower mean birth weight has been noted in three studies[41138], although this does not appear to be due to intrauterine growth restriction. One study reported an increased placental/fetal mass, i.e. larger placentas in ICP[11].
The diagnosis of ICP is one of exclusion and alternative causes of hepatic impairment or pruritus should be considered before the diagnosis is made.
Liver function in normal pregnancy: Liver function does not change in normal pregnancy, although it is recommended that adjusted upper limits of normal are used. The upper limit of the normal reference range for serum alanine transaminase (ALT) and aspartate transaminase (AST) should be reduced by 20%[60] and the γ-glutamyl transpeptidase (GGT) level is reduced by a similar amount in later pregnancy[61]. Total and free bilirubin is also lower during all three trimesters, and conjugated bilirubin is lower in the second and third trimesters[61].
LFTs in ICP: The transaminase enzymes are located within hepatocytes and raised serum levels are thus indicative of hepatocellular damage. In ICP, ALT and AST may rise before or after serum bile acids[3962]. Of the two, ALT is thought to be a more sensitive marker of ICP; there is a 2-10-fold increase in serum levels that is generally more marked than the rise in AST[34762].
Bilirubin is normal in the majority of ICP cases and is of limited value in diagnosis or follow up. If raised, it tends to be a conjugated hyperbilirubinemia[62].
GGT has been shown to be raised in some studies[245663] but is more commonly normal[47]. It has been proposed that elevations in GGT are associated with a greater impairment in other LFTs[63], and that they can provide insights into the genetic etiology of the condition.
Alkaline phosphatase (ALP) levels may rise in ICP but production of large amounts of the placental isoform render this biochemical marker of limited diagnostic value.
Glutathione S-transferase alpha (GSTA) is a phase II detoxification enzyme that is rapidly released into the circulation following acute hepatic damage. It is reported to be a more sensitive and specific marker of hepatic integrity than standard LFTs[64–66]. A longitudinal study comparing serum levels from ICP, control and pruritus gravidarum cases demonstrated higher GSTA in ICP at all gestations from 24 wk to term[67]. A recent study confirmed that GSTA levels are elevated in ICP cases[68], and the authors of both studies proposed that it may be a useful tool for early diagnosis of the condition.
Bile acids: The primary bile acids cholic acid (CA) and chenodeoxycholic acid (CDCA) are the end products of hepatic cholesterol metabolism and represent the major route for excretion of cholesterol. Following synthesis, CA and CDCA are conjugated with taurine or glycine (in a ratio of approximately 1:3) before export across the canalicular membrane to enter the bile. In the terminal ileum and colon, CA and CDCA undergo bacterial modification, mainly deconjugation and 7α-dehydroxylation, to form the secondary bile acids deoxycholic acid (DCA) and lithocholic acid (LCA), respectively. UDCA is a tertiary bile acid as it results from bacterial modification followed by hepatic metabolism. It is normally detectable in trace amounts in normal serum. Bile acids are reabsorbed in the terminal ileum and enter the portal vein for transport back to the liver. The enterohepatic circulation is highly efficient and 95% of all bile acids are reabsorbed. A more detailed account of bile acid synthesis, canalicular secretion, sinusoidal uptake and homeostasis is given in a number of recent reviews[6970].
Bile acids in normal pregnancy: In healthy pregnancy, there is a minimal rise in total serum bile acids as gestation advances[7172]. Studies in which individual bile acid levels have been measured show that there is no change in DCA but CDCA doubles by term[73]. The data concerning CA are not so conclusive, with some studies reporting a significant increase in the third trimester compared to the first[5972], and others showing no change[73]. However, in all studies the level stayed well within the normal range (< 1.5 &mgr;mol/L). Perhaps a more informative measure is the ratio of the two primary bile acids (CA:CDCA), which is reported to be between 0.68 and 1.9 at term (38-40 wk)[7273].
The reference range used for total serum bile acids in pregnancy varies, but most authors accept an upper limit of normal of between 10 and 14 &mgr;mol/L.
The phenomenon of asymptomatic hypercholanemia of pregnancy (AHP) has recently been reported. AHP is defined as raised serum bile acids in pregnancy in the absence of symptoms and other biochemical markers of ICP[74]. Specifically, they describe a change in the profiles of serum bile acids in women with AHP with increased CA and relatively unchanged CDCA levels. AHP is reported to affect approximately 10% of the pregnant population, and 2%-3% of women with AHP during the second trimester subsequently develop ICP.
More recently, Castaño et al[75] reported that AHP affects 40% of pregnant women in Argentina and that the pregnancy outcome is similar to that of normal pregnancies, suggesting that mild hypercholanemia may fall within the biochemical spectrum of normal pregnancy.
Bile acids in ICP: Serum bile acid measurement is now considered to be the most suitable biochemical marker for both the diagnosis and monitoring of ICP[76], with the cholic acid level[59] or the CA:CDCA ratio proposed as being the most sensitive indicator for the early diagnosis of the condition[2462]. Levels of the secondary bile acid DCA also rise[73], although to a lesser extent, and indicate impairment of the enterohepatic circulation.
There are reports in the literature of elevations in serum bile acids as high as 100 times the upper limit of normal[7377].
In addition, maternal cholestasis results in elevated levels of bile acids in the fetal circulation and a reversal in the normal feto-maternal transplacental bile acid gradient[397879].
There is as yet no consensus on whether a rise in serum bile acids precedes the onset of symptoms. There are reports of women with elevated serum bile acids prior to the onset of symptoms or the appearance of other biochemical abnormalities[62]. Equally, there is no agreement on whether serum bile acids should be measured in the fasted or post-prandial state. In ICP, consumption of a standard test meal causes a more dramatic and prolonged post-prandial rise in serum bile acids, particularly CA, than in control women, suggesting that the use of a standard test meal may help to distinguish mild forms of the disease from normal[8081]. However, this approach would be costly and time-consuming if used in routine obstetric practice.
The biliary bile acid profile is also altered in ICP. Although CA remains the predominant bile acid, the proportion is greatly increased, with a consequent reduction in the proportion of CDCA. DCA is markedly diminished, suggesting a significant impairment of the enterohepatic circulation in ICP[82].
Lipids: Cross-sectional studies have reported deranged lipid profiles in association with ICP[348384], and in a prospective longitudinal study LDL cholesterol, apolipoprotein B-100 and total cholesterol concentrations were found to be elevated[85]. However, it should be noted that this study was performed in a group of patients who were not fasted at the time of sample collection.
Glucose: One study has shown that ICP is associated with impaired glucose tolerance. Although there was no difference in fasting glucose levels between cases and controls, both the 2 h post-prandial glucose and oral glucose tolerance tests were higher in ICP[21].
Clotting: One study reported a prolonged prothrombin time in 20% of patients[12]. However this is not consistent with the author’s experience. In 65 ICP cases with raised serum bile acids managed in our hospital from 1996-2003, there were none with an abnormal clotting profile.
Other biochemical markers investigated in ICP are summarized in Table 3[86–100].
| Number of cases (controls) | Gestation | Parameter investigated | Main findings | References |
| 76 | NR | Serum human placental lactogen and AFP | ↑ hPL, no difference in AFP | [88] |
| 68 | 37-38/40 | Pregnancy specific β-1-glycoprotein | ↓ Levels | [89] |
| 76 (150) | T3 | Prolactin | ↑ Prolactin from 33 wk | [94] |
| 10 (288) | T3 | Placental protein 10 | ↓ Placental protein 10 in ICP-negatively correlated with AST and SBA | [98] |
| NR | T3 + PN | Serum copper and zinc | ↑ Copper in ICP | [91] |
| No difference in zinc | ||||
| NR | T3 | Serum 25(OH)D, 24, 25(OH)D, 1, 25(OH)D, total protein, calcium, phosphorus, magnesium and alkaline phosphatase | 25(OH)D initially higher in patients than controls, but decreased by delivery | [92] |
| No change in any other parameter | ||||
| 12 (12) | T3 | Serum selenium and glutathione peroxidase | ↓ Selenium in ICP | [90] |
| ↓ Glutathione peroxidase activity | ||||
| 33 (5680) | T2 | Serum AFP and hCG | No differences | [87] |
| 26 (13) | NR | Thyroid hormones | ↓ T3 | [93] |
| 21 (98) | T3 | Serum selenium, zinc and copper | ↓ Selenium in ICP | [96] |
| ↑ (Double) copper in ICP | ||||
| No difference in zinc | ||||
| 72 (30) | T3 | Renal function tests: uric acid, urea, potassium, sodium, creatinine | ↑ Uric acid and creatinine | [97] |
| 22 (21) | NR | Maternal-fetal mixed lymphocyte reaction | ↓ Transformation rate of lymphocytes | [86] |
| 24 (1148) | T3 | Serum AFP and β-hCG | No differences | [95] |
| 30 (30) | T3 | Serum neopterin and soluble interleukin 2 receptor (sIL-2R) | ↑ Neopterin and sIL-2R | [99] |
| 58 (42) | T3 | Alpha-hydroxybutyrate dehydrogenase (α-HBDH) activity | ↑α-HBDH | [100] |
Analysis of the urine from women with ICP shows an increased excretion of total bile acids, with a 10- to 100-fold increase in CA and CDCA, but decreased excretion of DCA and LCA. The reduced excretion of the secondary bile acids supports the hypothesis that a canalicular defect is a primary feature of ICP, and is consistent with an impaired enterohepatic circulation. The bile acid profile shows a shift from glycine to taurine conjugation and an increased proportion of sulfated species[101]. In a longitudinal study of two women, the first detectable change in urinary bile acids was the appearance of tetrahydroxylated species, which are known products of CA metabolism. This change occurred before the rise in total bile acid excretion[102].
Gallstones are reported in 13% of women with ICP[31]. While it is likely that affected women have increased susceptibility to cholelithiasis, there have been no robust studies of ICP cases and controls, and pregnancy itself is also associated with an increased incidence of asymptomatic gallstones[103104]. First-degree relatives of affected women also have higher rates of cholelithiasis (26% of relatives of 227 ICP cases compared to 9% of 234 controls, P < 0.001, χ2) (unpublished data, C. Williamson).
In ICP, the intrahepatic bile ducts appear normal, but the fasting and ejection volumes of the gallbladder are greater, possibly predisposing these women to the formation of gallstones[105–107]. However, ICP has been described in women with previous cholecystectomy[108], suggesting that the presence of gallstones is not causative of ICP.
Several studies have reported that there is normal liver structure with no evidence of liver cell damage and only mildly dilated bile ducts, bile stasis in canaliculi, bile plugs and mild portal tract inflammation in liver biopsies from women with ICP[5109]. Electron microscopy findings show generally well-preserved architecture with dilated bile capillaries, distorted microvilli and granular deposits (bile thrombi)[5109].
The etiology of ICP is complex and not fully understood. Evidence from ICP pedigrees suggests that there is a genetic component to the disease[110], and the relative risk for parous sisters of affected women is 12[9111]. Insights into the genetic etiology come from studies of the familial cholestasis syndromes progressive familial cholestasis (PFIC) and benign recurrent cholestasis (BRIC). These autosomal recessive syndromes are caused by homozygous mutations in the genes encoding biliary transport proteins, and case reports have described ICP in the heterozygous mothers of affected children.
The most extensively studied candidate gene in ICP is ABCB4, which encodes the multidrug resistance protein 3, a floppase that transports phosphatidylcholine from the inner to the outer leaflet of the hepatocyte canalicular membrane[112–114]. Homozygous mutations result in a spectrum of phenotypes that include PFIC type 3[115] and cholelithiasis[116]. ICP-associated variants were first described in a case report published in 1999, in which the mother of a child with PFIC type 3 was found to have a heterozygous single nucleotide deletion (1712delT). She and five of her female relatives had a history of ICP[117]. However, a second study screening 57 Finnish cases for this mutation concluded that it does not play a significant role in the etiology of ICP in this population[118]. Subsequent to this, there have been 12 different genetic variants and four splicing mutations in ABCB4 reported in ICP cases[119–126]. The first ABCB4 mutations were described in patients with elevated serum GGT levels, a biochemical phenotype that is also found in PFIC type 3, and not in types 1 and 2. However, some recent studies have described ABCB4 variants in ICP patients with normal GGT[120126]. A recent study has also described an ABCB4 haplotype which is associated with the “severe” phenotype of serum bile acids > 40 &mgr;mol/L[127].
Homozygous mutations in the familial intrahepatic cholestasis one gene (FIC1, ATP8B1) cause PFIC type 1 and BRIC. The function of the FIC1 protein is disputed, but it is hypothesized to be an aminophospholipid translocase which transports phosphatidylserine from the canaliculus into the hepatocyte, thus maintaining membrane asymmetry and the function of the other biliary transporters embedded within the membrane. Variation in FIC1 occurs in a small number of ICP cases, although the functional consequences are currently not known[128129].
Genetic variation in the bile salt export pump (BSEP), encoded by ABCB11 has also been reported in ICP. BSEP is located exclusively in the hepatocyte canalicular membrane and is the primary export pump for bile acids. Homozygous mutations in ABCB11 are associated with PFIC type 2. Two studies investigated the role of ABCB11 variation in Finnish ICP cases. One study reported that single nucleotide polymorphisms in exons 28 and 19 were susceptibility loci for ICP[130]. However, a subsequent study in a larger number of cases with a more diverse ethnic background failed to confirm these findings, suggesting that ICP is a genetically heterogeneous disease[131]. Further evidence for genetic heterogeneity was provided by a study of 16 individuals from two affected Finnish families. Segregation of haplotypes and multipoint linkage analysis of microsatellite markers in ABCB11, ABCB4 and ATP8B1 excluded genetic variation in these genes from playing a role in the etiology of ICP[132]. Other BSEP variants reported to be associated with ICP include N591S and the V444A polymorphism[123126133]. The latter of these variants is particularly interesting as it is also reported to be a susceptibility factor for estrogen-induced cholestasis[133]. A recent UK study demonstrated that two common PFIC2-associated mutations (E279G and D482G) and N591S are present as heterozygous variants in a small proportion of ICP cases. This study of 491 Caucasian ICP cases and 261 controls also demonstrated that the V444A allele is a significant risk locus for ICP in this population[134].
Genetic variation has also been reported in another biliary transporter, ABCC2, which encodes the multidrug resistance related protein 2 (MRP2). MRP2 exports organic anions including bilirubin into the bile. A study from South America has suggested that a polymorphism in exon 28 is associated with ICP[135].
The farnesoid X receptor encoded by NR1H4 is the principal bile acid receptor and is responsible for the regulation of bile acid synthesis and transport within the liver. A recent study has described four heterozygous variants within FXR that are associated with ICP, three of which were shown to have functional effects[136].
A variety of other genetic loci have been investigated in ICP cases and the reader is referred to a recent review for full details of all studies to date[111].
Several studies provide evidence that reproductive hormones play a role in the etiology of ICP. The disease is more common in multiple than singleton pregnancies (20.9% vs 4.7% in one study)[10], and the symptoms may recur in a subgroup of affected women when taking the combined oral contraceptive pill[31]. In addition, most women present with symptoms of ICP in the third trimester when estrogen and progesterone levels are highest.
Most studies have focussed on estrogen rather than progesterone. In clinical studies, administration of ethinylestradiol to both men and women results in a decreased clearance of sulfobromophthalein, and this is further reduced in women with a personal history of ICP and their male relatives[137]. Furthermore, administration of the depot estrogen, ethinylestradiol propanolsulphonate to 20 healthy women significantly increased the total serum bile acid concentration, and in particular, the proportion of taurine conjugates[138]. In vitro studies have demonstrated that the cholestatic estrogen metabolite, 17-β-estradiol glucuronide, transinhibits the BSEP following excretion into the bile canaliculus by MRP2[139]. Supporting this hypothesis, administration of 17-β-estradiol glucuronide to rats causes endocytic internalization of BSEP in an MRP2 (Mrp2; Abcc2)-dependent manner[140]. Estrogen also impairs the expression and/or function of, BSEP and MRP2 in rodent studies[141–143].
However, progesterone may play an even greater role in the pathogenesis of ICP. Bacq et al[144] observed that administration of natural progestin to women with threatened preterm labour resulted in ICP in 11 of the 12 women treated. This finding was confirmed by two subsequent studies[56145]. Although total progesterone does not rise in comparison to normal pregnancies, the profile of metabolites is considerably different. An excess of monosulfated and disulfated (in particular 3α and 5α) isomers has been reported in the serum and urine of women with ICP, and this may reflect impaired excretion of these metabolites at the canalicular membrane, or abnormal synthesis[146147]. Studies of umbilical cord serum have also shown that disulfated progesterone metabolites are increased in the fetal compartment of affected pregnancies compared with normal pregnancies. Moreover, the level of steroid sulfates synthesized by the fetus, e.g. 16α-hydroxydeh-ydroepiandrosterone sulfate are decreased, suggesting that maternal cholestasis is associated with impaired fetal steroid synthesis[148].
There are few in vitro studies that have investigated the mechanism of progesterone metabolite induced cholestasis. One study has shown that sulfated progesterone metabolites, but not progesterone itself, cause reduced bile flow in the rat[149]. The same authors demonstrated that these metabolites inhibit BSEP-mediated bile acid efflux in Xenopus oocytes.
Selenium: Serum levels of selenium usually decrease with advancing gestation, but normal serum levels are maintained if dietary intake is adequate[96]. Dietary selenium intake is lower in Finland and Chile, and serum levels have been shown to be reduced in women with ICP compared to pregnant controls in both countries[9096]. Glutathione peroxidase is a powerful antioxidant that is dependent on selenium. Estrogens and bile acids cause oxidative stress, and it has been proposed that reduced serum selenium levels may contribute to the etiology of ICP and may also provide an explanation for the geographic variation in the prevalence of the condition.
Seasonal variation: The incidence of ICP peaks in the winter months in Scandinavia and Chile, suggesting a possible association with an environmental trigger. Interestingly, serum selenium levels have also been reported to be significantly higher in the summer than winter[96].
Infection: There is an increased incidence of hepatitis C infection in women with ICP, and one study has reported that affected women develop cholestasis at earlier gestations[32]. There have also been reports of an increased incidence of urinary tract infection and pyelonephritis in the early stages of pregnancy than in controls[13].
Drug cholestasis: Johnston et al[4] reported higher rates of drug sensitivities among the ICP population, especially to antibiotics. There may be overlapping etiological factors that influence susceptibility to ICP and drug-induced cholestasis. For example, the V444A allele of BSEP that confers increased risk for ICP is also found more commonly in individuals with drug-induced cholestasis[150].
Leaky gut: Increased gut permeability as measured by the urinary lactulose/mannitol ratio (L/M) has been reported in a subgroup of ICP patients (five of the 20 women in the study), and the authors postulate that this may participate in the pathogenesis of the condition by enhancing the absorption of bacterial endotoxins[151]. However, levels of anti-lipopolysaccharide antibodies and pro-inflammatory cytokines [tumor necrosis factor-α, interleukin (IL)-1β, IL-6 and IL-10] were measured and no significant differences were shown, suggesting that this may not be the case. It should also be noted that the abnormal excretion of lactulose and mannitol persisted for up to 2 years in four of the five women identified, and thus it is possible that a leaky gut is a permanent abnormality in these women rather than a result of ICP.
Pruritus: The itch associated with ICP is often the most troubling symptom for affected women. It has been speculated that it is due to accumulation of bile acids in the interstitial fluid of the skin. However, serum bile acid levels do not correlate well with maternal symptoms and, while application of bile acids to blister bases or intradermal injection of bile acids results in pruritus[152153], absolute concentrations of bile acids in the skin do not correlate well with the sensation of itch[154]. Furthermore, some studies have reported pruritus before the onset of biochemical abnormalities[35]. These findings suggest that an alternative compound acts as a pruritogen in this and possibly other forms of cholestatic liver disease. Candidates include reproductive hormone metabolites, and a recent study has reported that serum sulfated progesterone metabolites were reduced following treatment with UDCA, concurrently with a reduction of pruritus[155]. Interestingly, a 5-hydroxytrytamine 3 receptor agonist has been reported to rapidly reduce pruritus in both ICP and other liver diseases, raising the possibility that serotonin is involved in the etiology of pruritus[156157].
The etiology of the fetal complications associated with ICP is poorly understood, but is thought to relate to an increased flux of bile acids into the fetal circulation, as indicated by elevated levels in amniotic fluid, cord serum and meconium. In vitro studies of isolated placental vesicles have shown that vectorial transfer of bile acids from fetus to mother is impaired in ICP, and that this is specifically the result of decreased efficiency of ATP-independent transport[158159]. Taken together, these findings suggest that bile acids accumulate in the fetal compartment and thus are likely to exacerbate fetal risk. Furthermore, a recent study of fetal outcomes in ICP has shown that the risk of adverse fetal outcomes increases with increasing levels of maternal serum bile acids[25].
Evidence for the involvement of bile acids in the etiology of MSAF comes from studies of fetal sheep infused with CA, in which 100% of the treated lambs were born with MSAF[160]. The mechanism by which bile acids cause this effect is not clear from this study:the lambs did not show any signs of fetal distress. However, bile acids are known to cause an increase in colonic motility[161162] and this is a possible explanation. Alternatively, the bile acids may cause fetal distress and subsequent meconium passage.
Individual neonatal rat cardiomyocytes treated with taurocholic acid show a decrease in the rate of contraction, which is reversible. Furthermore, cells in a network lose their ability to beat synchronously after the addition of taurocholic acid and have abnormal calcium dynamics, suggesting that elevated levels of bile acids in ICP may be responsible for the CTG abnormalities observed[163].
Rodent studies have shown that the non-pregnant rat myometrium displays a dose-dependent increase in contractility in response to CA[164], and sheep infused with this bile acid have an increased incidence of spontaneous preterm labour[160]. Furthermore, it has been suggested that the myometrium of ICP patients may be more responsive to the effects of oxytocin[165166].
Bile acid aspiration or accumulation within the fetal circulation is thought to be responsible for the increased incidence of RDS seen in association with ICP. In animal models, bile acids have been shown to cause severe chemical pneumonitis and pulmonary edema[167168]. Furthermore, intra-tracheal injection of bile acids in rabbits resulted in atelectasis, eosinophilic infiltration and formation of hyaline membrane, which could be reversed by the administration of surfactant[169]. It has therefore been hypothesized that elevated levels of bile acids in the fetal circulation cause a reversal of the action of phospholipase A2, thereby causing the degradation of phosphatidylcholine and a lack of surfactant[53]. Interestingly, administration of intra-tracheal surfactant to two of the infants reported in a recent series resulted in some improvement in their condition[53].
The mechanisms causing sudden IUD are poorly understood. At autopsy, the majority of the stillborn babies are of normal weight and have no signs of chronic utero-placental insufficiency, but do have signs of acute anoxia[38]. However, several studies have reported non-specific morphological changes in the placenta, including increased syncytial knot formation and villous edema, which are suggestive of hypoxic insults[47170171]. These findings are comparable to the morphological appearance of placentas from a rodent model of ICP, which is also associated with increased placental oxidative stress[172].
As previously discussed, there is MSAF in up to 100% of ICP-associated stillbirths. Studies of meconium from healthy pregnancies has shown that it penetrates deep into placental and umbilical cord tissue in less than 3 h[173], and can cause vasoconstriction of the placental and umbilical vessels. The mechanism is not known, but heat-inactivated meconium did not induce an effect in one study[174], suggesting that a peptide or prostaglandin produces the effect. In ICP, the meconium contains significantly elevated levels of bile acids (13.5 ± 5.1 &mgr;mol/g vs 2.0 ± 0.5 &mgr;mol/g)[175], and as bile acids are known to cause vasoconstriction of the placental chorionic vessels[176], it is possible that placental vasoconstriction is a mechanism that contributes to the risk of IUD in ICP.
The aims of management are to reduce symptoms and biochemical abnormalities in the mother and to reduce the risk of fetal distress, preterm delivery and sudden fetal death.
There are several case reports of normal CTG and/or normal fetal movements in the hours preceding fetal loss[1848177178]. Thus, the general consensus is that these forms of fetal surveillance do not prevent IUD. However, they may be reassuring to women with ICP and the clinicians responsible for their care at the time they are performed. One study reported good fetal and neonatal outcomes with a policy of routine amnioscopy at 36 wk to assess amniotic fluid color in addition to standard monitoring for fetal wellbeing[20]. However, this approach may be considered overly invasive by many obstetricians.
Some studies have reported good outcomes with a policy of induction of labour at 37 or 38 wk gestation[2046]. Many clinicians in the UK have adopted this practice as the IUDs appear to cluster at later gestations (Figure 2). However there have been very few reports of the gestational week at which the IUD occurs, nor have there been any large prospective studies of whether drug treatment or early delivery prevents adverse fetal outcomes.
UDCA: UDCA is a naturally occurring hydrophilic bile acid that constitutes < 3% of the physiological bile acid pool in humans. It has been used with positive effects in the management of primary biliary cirrhosis and other cholestatic disorders for several years, and is gaining popularity as a treatment for ICP. There is evidence that UDCA stimulates biliary secretion by post-transcriptional regulation of BSEP and the alternative exporters MRP4 and MRP3. In addition, it has antiapoptotic effects and has been shown to reduce the mitochondrial membrane permeability to ions and cytochrome c expression[179180]. Finally, UDCA lowers serum levels of ethinyl-estradiol 17β-glucuronide, a major cholestatic metabolite of estrogen.
The first reported use of UDCA in ICP was by Palma et al[181] in 1992. In an uncontrolled series of eight cases, UDCA was prescribed at a dose of 1 g/d either continuously for 20 d or for two 20-d periods interrupted by a 14-d drug-free period. Both groups had a significant improvement in serum biochemistry and symptoms after 20 d treatment, but relapse was seen after the first week of the drug-free period in the latter group. Subsequently, UDCA was used to treat three patients with recurrent ICP, all of whom had rapid symptomatic and biochemical improvements with no adverse fetal outcomes[45]. This was followed by three small randomized, controlled trials (maximum of eight patients in each arm), the first of which showed that 20 d of UDCA treatment (600 mg/d) resulted in a significant reduction of pruritus and LFTs, including bile acids, compared to baseline[182]. One other study failed to show any reduction in pruritus compared with placebo, and the final one showed a reduction that did not reach statistical significance because of the small numbers of women treated[183184]. In both studies, UDCA caused a significant reduction in serum transaminases and bilirubin compared to placebo. One study also showed a significant reduction in serum bile acids[183]. There have been several additional case series demonstrating that UDCA treatment results in clinical and biochemical improvement in ICP[26185–188].
More recently, a randomized placebo-controlled trial comparing the efficacy of UDCA and dexamethasone therapy in ICP reported that UDCA, but not dexamethasone, significantly reduced ALT and bilirubin in all women treated. Furthermore, there was a significant reduction of pruritus and bile acids in women with serum bile acid levels exceeding 40 &mgr;mol/L at inclusion[189].
Studies examining the bile acid pool composition have shown that, in addition to a reduction in the serum bile acid concentration, treatment with UDCA results in a normalization of the CA:CDCA and glycine:taurine ratios[190], and a reduction in urinary excretion of sulfated progesterone metabolites, which the authors propose is associated with a concurrent reduction in pruritus[155].
There have been no reports of fetal morbidity or mortality resulting from UDCA treatment, although no study has had sufficiently large numbers to allow this to be fully evaluated. However, UDCA treatment has been shown to reduce the bile acid level in cord blood[187], amniotic fluid[187191192] and colostrum[193], and it reduced cord blood bilirubin levels in one study[194]. As previously discussed, the level of bile acids in meconium is considerably elevated in ICP, and this is not influenced by treatment with UDCA[175]. However, this may be because bile acids had already accumulated in the meconium prior to UDCA treatment. It is likely that, if UDCA reduces the maternal serum bile acid level, and thus placental transfer of bile acids, then there should be a corresponding reduction in the level in meconium from the time of treatment.
Finally, UDCA has been shown to correct the impaired bile acid transfer kinetics observed in ICP placentas[159] and to reverse the morphological changes seen in the placentas of a rodent model of ICP[195]. In addition, placental MRP2 protein and mRNA expression were significantly increased in patients treated with UDCA compared to controls[194]. UDCA also protects cardiomyocytes from bile acid-induced arrhythmias in an in vitro model[196].
There are very few side effects reported with UDCA treatment. At higher doses women may complain of gastrointestinal upset and diarrhea, but this is rare.
Dexamethasone: Dexamethasone inhibits placental estrogen synthesis by reducing secretion of the precursor, dehydroepiandrosterone sulfate, from the fetal adrenal glands[197198]. An early observational study of 10 affected women from Finland suggested a beneficial effect with reduced serum estriol and estradiol levels and symptomatic improvement in all cases. In addition liver biochemistry, including the serum bile acid level, was improved and symptoms did not recur on cessation of treatment[199]. However, this was not supported by subsequent studies[189200201].
In addition to the conflicting reports of efficacy, there are concerns over safety. Dexamethasone has been widely used to promote fetal lung maturity and is reported to be safe in this context. However, it crosses the placenta easily, and animal and human data suggest that repeated high doses are associated with decreased birth weight[202] and abnormal neuronal development[203].
Rifampicin: Although there are no published studies reporting the use of rifampicin in ICP, it has been used with good results in several other liver diseases, including gallstones and primary biliary cirrhosis[204–206]. In these studies treatment with rifampicin resulted in significant decreases in serum levels of transaminases and total bile acids, as well as an improvement in pruritus, suggesting that it might also be useful in the treatment of ICP. A recent study investigating the molecular mechanism by which rifampicin works has shown that it enhances bile acid detoxification, an effect that is complementary to the up-regulation of bile acid export induced by UDCA, suggesting that the two drugs used in combination may be more effective than monotherapy[204]. The authors are aware of several ICP cases that have not responded to monotherapy with UDCA, but have responded to combined treatment with rifampicin and UDCA.
Vitamin K: ICP is associated with a risk of malabsorption of fat soluble vitamins due to reduced enterohepatic circulation of bile acids and subsequent reduction of uptake in the terminal ileum. Therefore many clinicians opt to treat women with oral vitamin K to guard against the theoretical risk of fetal antepartum and maternal intra- or postpartum hemorrhage. However, there have been no studies to support or refute this practice.
Others: S-Adenosyl-L-methionine (SAMe) is the principal methyl group donor involved in the synthesis of phosphatidylcholine, and therefore, it influences the composition and fluidity of hepatic membranes and hence biliary excretion of hormone metabolites[207]. It reverses estrogen-induced impairment of bile flow in rats[208–210]. Furthermore, in a human study of estrogen-induced cholestasis in women with a history of ICP, SAMe was shown to prevent ethinylestradiol-induced elevations in AST/ALT, bile acids and bilirubin[211]. Early studies of SAMe in the treatment of ICP reported that it improved both symptoms and biochemistry[212213], and these findings were confirmed by a subsequent placebo controlled study in which 15 women were treated with high dose SAMe (800 mg/d iv)[214]. However, a double-blind, placebo-controlled trial showed no improvement in symptoms or biochemistry following treatment with SAMe[215]. Finally, the efficacy of SAMe has been compared to that of UDCA, combination therapy with UDCA and SAMe, and placebo in one study[183]. Women treated with SAMe had a larger reduction in pruritus score and biochemical parameters than women in the placebo group, but this was not as large in the UDCA group. Furthermore, treatment with a combination of UDCA and SAMe was more effective than SAMe alone in reducing bile acid levels. Some patients have reported problems with peripheral veins following prolonged intravenous administration[215]. No other adverse maternal or fetal effects have been reported and SAMe seems to be well tolerated.
Cholestyramine is an anion exchange resin which acts by binding bile acids in the gut, thereby inhibiting the enterohepatic circulation and increasing fecal excretion of bile acids. There have been several studies suggesting that cholestyramine is effective at reducing pruritus in ICP[216217]. However, it has no effect on serum bile acid levels or other biochemical markers of cholestasis[216]. Furthermore, it may reduce the intestinal absorption of fat-soluble vitamins, thus depleting the levels of vitamin K and increasing the risk of hemorrhage for the mother and fetus[218]. Cholestyramine is therefore no longer considered a first-line therapy for ICP.
Guar gum is a dietary fiber that acts in a similar manner to cholestyramine. One small study has reported the use of guar gum in the treatment of ICP and reported no effect on serum bile acids or bilirubin and only a minimal reduction in pruritus score[219]. Subsequently, a randomized controlled trial has shown that guar gum is no more effective than placebo in improving pruritus or reducing serum bile acids[220].
Peroral activated charcoal has been shown to reduce serum bile acids in seven of nine women treated in one study. However, there was no improvement in symptoms[221].
Topical treatment with aqueous cream with 2% menthol is of value in the relief of pruritus, but does not improve biochemical abnormalities.
Most women have no lasting hepatic damage, but ICP recurs in the majority of cases, with variations in intensity in subsequent pregnancies[10222]. Recurrence is less likely following multiple pregnancy. Women with a history of ICP may also develop symptoms if taking the combined oral contraceptive pill or in the second half of the menstrual cycle[31]. Long-term follow-up studies have shown an increased risk of gallstones, non-alcoholic cirrhosis and pancreatitis, hepatitis C and autoimmune hepatitis[223224].
ICP is a relatively common cause of hepatic impairment in pregnancy. It has a complex etiology with genetic, endocrine and environmental components. ICP causes maternal pruritus with impaired liver function and raised serum bile acids. The maternal cholestasis is transient with postnatal resolution, although affected women have increased rates of hepatobiliary disorders in later life. ICP is associated with adverse fetal outcomes. The risk of meconium-stained liquor, fetal asphyxia and spontaneous preterm delivery is greater in pregnancies with more marked elevations in maternal serum bile acid levels. The condition is also associated with IUD. The most effective pharmacological therapy for improvement of maternal symptoms and biochemical abnormalities is UDCA, and this has also been shown to reduce placental abnormalities and to improve placental bile acid transport in in vitro studies. Fetal outcomes are improved with a variety of strategies of active management, although the most effective intervention has not currently been established. A common practice is induction of labour at 37-38 wk of gestation with the aim of reducing the risk of IUD as many deaths occur at later gestations. Large therapeutic trials are required to establish which specific drug treatments or management strategies are effective at reducing the rates of adverse fetal outcomes.
| 1. | Svanborg A. A study of recurrent jaundice in pregnancy. Acta Obstet Gynecol Scand. 1954;33:434-444. [Cited in This Article: ] |
| 2. | Thorling L. Jaundice in pregnancy; a clinical study. Acta Med Scand Suppl. 1955;302:1-123. [Cited in This Article: ] |
| 3. | Fisk NM, Bye WB, Storey GN. Maternal features of obstetric cholestasis: 20 years experience at King George V Hospital. Aust N Z J Obstet Gynaecol. 1988;28:172-176. [Cited in This Article: ] |
| 4. | Johnston WG, Baskett TF. Obstetric cholestasis. A 14 year review. Am J Obstet Gynecol. 1979;133:299-301. [Cited in This Article: ] |
| 5. | Kater RM, Mistilis SP. Obstetric cholestasis and pruritus of pregnancy. Med J Aust. 1967;1:638-640. [Cited in This Article: ] |
| 6. | Reyes H, Gonzalez MC, Ribalta J, Aburto H, Matus C, Schramm G, Katz R, Medina E. Prevalence of intrahepatic cholestasis of pregnancy in Chile. Ann Intern Med. 1978;88:487-493. [Cited in This Article: ] |
| 7. | Reyes H, Taboada G, Ribalta J. Prevalence of intrahepatic cholestasis of pregnancy in La Paz, Bolivia. J Chronic Dis. 1979;32:499-504. [Cited in This Article: ] |
| 8. | Steel R, Parker ML. Jaundice in pregnancy. Med J Aust. 1973;1:461. [Cited in This Article: ] |
| 9. | Eloranta ML, Heinonen S, Mononen T, Saarikoski S. Risk of obstetric cholestasis in sisters of index patients. Clin Genet. 2001;60:42-45. [Cited in This Article: ] |
| 10. | Gonzalez MC, Reyes H, Arrese M, Figueroa D, Lorca B, Andresen M, Segovia N, Molina C, Arce S. Intrahepatic cholestasis of pregnancy in twin pregnancies. J Hepatol. 1989;9:84-90. [Cited in This Article: ] |
| 11. | Heinonen S, Kirkinen P. Pregnancy outcome with intrahepatic cholestasis. Obstet Gynecol. 1999;94:189-193. [Cited in This Article: ] |
| 12. | Jiang ZH, Qiu ZD, Liu WW, Liu YH, Wang QN, Miao HZ, Zhou ZC, Wu XL, Xu BY, Gu CH. Intrahepatic cholestasis of pregnancy and its complications. Analysis of 100 cases in Chongqing area. Chin Med J (Engl). 1986;99:957-960. [Cited in This Article: ] |
| 13. | Laatikainen T, Ikonen E. Fetal prognosis in obstetric hepatosis. Ann Chir Gynaecol Fenn. 1975;64:155-164. [Cited in This Article: ] |
| 14. | Lo TK, Lau WL, Lam HS, Leung WC, Chin RK. Obstetric cholestasis in Hong Kong--local experience with eight consecutive cases. Hong Kong Med J. 2007;13:387-391. [Cited in This Article: ] |
| 15. | Paternoster DM, Fabris F, Palù G, Santarossa C, Bracciante R, Snijders D, Floreani A. Intra-hepatic cholestasis of pregnancy in hepatitis C virus infection. Acta Obstet Gynecol Scand. 2002;81:99-103. [Cited in This Article: ] |
| 16. | Perreau P, Rouchy R. [Recurrent cholostatic jaundice of pregnancy.]. Gynecol Obstet (Paris). 1961;60:161-179. [Cited in This Article: ] |
| 17. | Rathi U, Bapat M, Rathi P, Abraham P. Effect of liver disease on maternal and fetal outcome--a prospective study. Indian J Gastroenterol. 2007;26:59-63. [Cited in This Article: ] |
| 18. | Rioseco AJ, Ivankovic MB, Manzur A, Hamed F, Kato SR, Parer JT, Germain AM. Intrahepatic cholestasis of pregnancy: a retrospective case-control study of perinatal outcome. Am J Obstet Gynecol. 1994;170:890-895. [Cited in This Article: ] |
| 19. | Roger D, Vaillant L, Fignon A, Pierre F, Bacq Y, Brechot JF, Grangeponte MC, Lorette G. Specific pruritic diseases of pregnancy. A prospective study of 3192 pregnant women. Arch Dermatol. 1994;130:734-739. [Cited in This Article: ] |
| 20. | Roncaglia N, Arreghini A, Locatelli A, Bellini P, Andreotti C, Ghidini A. Obstetric cholestasis: outcome with active management. Eur J Obstet Gynecol Reprod Biol. 2002;100:167-170. [Cited in This Article: ] |
| 21. | Wójcicka-Jagodzińska J, Kuczyńska-Sicińska J, Czajkowski K, Smolarczyk R. Carbohydrate metabolism in the course of intrahepatic cholestasis in pregnancy. Am J Obstet Gynecol. 1989;161:959-964. [Cited in This Article: ] |
| 22. | Abedin P, Weaver JB, Egginton E. Intrahepatic cholestasis of pregnancy: prevalence and ethnic distribution. Ethn Health. 1999;4:35-37. [Cited in This Article: ] |
| 23. | Berg B, Helm G, Petersohn L, Tryding N. Cholestasis of pregnancy. Clinical and laboratory studies. Acta Obstet Gynecol Scand. 1986;65:107-113. [Cited in This Article: ] |
| 24. | Brites D, Rodrigues CM, van-Zeller H, Brito A, Silva R. Relevance of serum bile acid profile in the diagnosis of intrahepatic cholestasis of pregnancy in an high incidence area: Portugal. Eur J Obstet Gynecol Reprod Biol. 1998;80:31-38. [Cited in This Article: ] |
| 25. | Glantz A, Marschall HU, Mattsson LA. Intrahepatic cholestasis of pregnancy: Relationships between bile acid levels and fetal complication rates. Hepatology. 2004;40:467-474. [Cited in This Article: ] |
| 26. | Laifer SA, Stiller RJ, Siddiqui DS, Dunston-Boone G, Whetham JC. Ursodeoxycholic acid for the treatment of intrahepatic cholestasis of pregnancy. J Matern Fetal Med. 2001;10:131-135. [Cited in This Article: ] |
| 27. | Lee RH, Goodwin TM, Greenspoon J, Incerpi M. The prevalence of intrahepatic cholestasis of pregnancy in a primarily Latina Los Angeles population. J Perinatol. 2006;26:527-532. [Cited in This Article: ] |
| 28. | Reyes H. Sex hormones and bile acids in intrahepatic cholestasis of pregnancy. Hepatology. 2008;47:376-379. [Cited in This Article: ] |
| 29. | Koivurova S, Hartikainen AL, Karinen L, Gissler M, Hemminki E, Martikainen H, Tuomivaara L, Järvelin MR. The course of pregnancy and delivery and the use of maternal healthcare services after standard IVF in Northern Finland 1990-1995. Hum Reprod. 2002;17:2897-2903. [Cited in This Article: ] |
| 30. | Kenyon AP, Piercy CN, Girling J, Williamson C, Tribe RM, Shennan AH. Obstetric cholestasis, outcome with active management: a series of 70 cases. BJOG. 2002;109:282-288. [Cited in This Article: ] |
| 31. | Williamson C, Hems LM, Goulis DG, Walker I, Chambers J, Donaldson O, Swiet M, Johnston DG. Clinical outcome in a series of cases of obstetric cholestasis identified via a patient support group. BJOG. 2004;111:676-681. [Cited in This Article: ] |
| 32. | Locatelli A, Roncaglia N, Arreghini A, Bellini P, Vergani P, Ghidini A. Hepatitis C virus infection is associated with a higher incidence of cholestasis of pregnancy. Br J Obstet Gynaecol. 1999;106:498-500. [Cited in This Article: ] |
| 33. | Johnson P, Samsioe G, Gustafson A. Studies in cholestasis of pregnancy. I. Clinical aspects and liver function tests. Acta Obstet Gynecol Scand. 1975;54:77-84. [Cited in This Article: ] |
| 34. | Reyes H. The spectrum of liver and gastrointestinal disease seen in cholestasis of pregnancy. Gastroenterol Clin North Am. 1992;21:905-921. [Cited in This Article: ] |
| 35. | Kenyon AP, Piercy CN, Girling J, Williamson C, Tribe RM, Shennan AH. Pruritus may precede abnormal liver function tests in pregnant women with obstetric cholestasis: a longitudinal analysis. BJOG. 2001;108:1190-1192. [Cited in This Article: ] |
| 36. | Lunzer MR. Jaundice in pregnancy. Baillieres Clin Gastroenterol. 1989;3:467-483. [Cited in This Article: ] |
| 37. | Reyes H, Radrigan ME, Gonzalez MC, Latorre R, Ribalta J, Segovia N, Alvarez C, Andresen M, Figueroa D, Lorca B. Steatorrhea in patients with intrahepatic cholestasis of pregnancy. Gastroenterology. 1987;93:584-590. [Cited in This Article: ] |
| 38. | Reid R, Ivey KJ, Rencoret RH, Storey B. Fetal complications of obstetric cholestasis. Br Med J. 1976;1:870-872. [Cited in This Article: ] |
| 39. | Shaw D, Frohlich J, Wittmann BA, Willms M. A prospective study of 18 patients with cholestasis of pregnancy. Am J Obstet Gynecol. 1982;142:621-625. [Cited in This Article: ] |
| 40. | Atabey S, Duvan CI, Eren U, Turhan NO. Intrahepatic cholestasis and eclampsia: a case report. Hypertens Pregnancy. 2007;26:363-369. [Cited in This Article: ] |
| 41. | Goulis DG, Walker IA, de Swiet M, Redman CW, Williamson C. Preeclampsia with abnormal liver function tests is associated with cholestasis in a subgroup of cases. Hypertens Pregnancy. 2004;23:19-27. [Cited in This Article: ] |
| 42. | Vanjak D, Moreau R, Roche-Sicot J, Soulier A, Sicot C. Intrahepatic cholestasis of pregnancy and acute fatty liver of pregnancy. An unusual but favorable association? Gastroenterology. 1991;100:1123-1125. [Cited in This Article: ] |
| 43. | Aytaç S, Kargili A, Türkay C. A prolonged gestational intrahepatic cholestasis: a case report. Turk J Gastroenterol. 2006;17:206-208. [Cited in This Article: ] |
| 44. | Olsson R, Tysk C, Aldenborg F, Holm B. Prolonged postpartum course of intrahepatic cholestasis of pregnancy. Gastroenterology. 1993;105:267-271. [Cited in This Article: ] |
| 45. | Davies MH, da Silva RC, Jones SR, Weaver JB, Elias E. Fetal mortality associated with cholestasis of pregnancy and the potential benefit of therapy with ursodeoxycholic acid. Gut. 1995;37:580-584. [Cited in This Article: ] |
| 46. | Fisk NM, Storey GN. Fetal outcome in obstetric cholestasis. Br J Obstet Gynaecol. 1988;95:1137-1143. [Cited in This Article: ] |
| 47. | Laatikainen T, Ikonen E. Serum bile acids in cholestasis of pregnancy. Obstet Gynecol. 1977;50:313-318. [Cited in This Article: ] |
| 48. | Laatikainen T, Tulenheimo A. Maternal serum bile acid levels and fetal distress in cholestasis of pregnancy. Int J Gynaecol Obstet. 1984;22:91-94. [Cited in This Article: ] |
| 49. | Lee RH, Kwok KM, Ingles S, Wilson ML, Mullin P, Incerpi M, Pathak B, Goodwin TM. Pregnancy outcomes during an era of aggressive management for intrahepatic cholestasis of pregnancy. Am J Perinatol. 2008;25:341-345. [Cited in This Article: ] |
| 50. | Ammälä P, Kariniemi V. Short-term variability of fetal heart rate in cholestasis of pregnancy. Am J Obstet Gynecol. 1981;141:217-220. [Cited in This Article: ] |
| 51. | Al Inizi S, Gupta R, Gale A. Fetal tachyarrhythmia with atrial flutter in obstetric cholestasis. Int J Gynaecol Obstet. 2006;93:53-54. [Cited in This Article: ] |
| 52. | Morrison JJ, Rennie JM, Milton PJ. Neonatal respiratory morbidity and mode of delivery at term: influence of timing of elective caesarean section. Br J Obstet Gynaecol. 1995;102:101-106. [Cited in This Article: ] |
| 53. | Zecca E, Costa S, Lauriola V, Vento G, Papacci P, Romagnoli C. Bile acid pneumonia: a "new" form of neonatal respiratory distress syndrome? Pediatrics. 2004;114:269-272. [Cited in This Article: ] |
| 54. | Zecca E, De Luca D, Baroni S, Vento G, Tiberi E, Romagnoli C. Bile acid-induced lung injury in newborn infants: a bronchoalveolar lavage fluid study. Pediatrics. 2008;121:e146-e149. [Cited in This Article: ] |
| 55. | Zecca E, De Luca D, Marras M, Caruso A, Bernardini T, Romagnoli C. Intrahepatic cholestasis of pregnancy and neonatal respiratory distress syndrome. Pediatrics. 2006;117:1669-1672. [Cited in This Article: ] |
| 56. | Bacq Y, Sapey T, Bréchot MC, Pierre F, Fignon A, Dubois F. Intrahepatic cholestasis of pregnancy: a French prospective study. Hepatology. 1997;26:358-364. [Cited in This Article: ] |
| 57. | Alsulyman OM, Ouzounian JG, Ames-Castro M, Goodwin TM. Intrahepatic cholestasis of pregnancy: perinatal outcome associated with expectant management. Am J Obstet Gynecol. 1996;175:957-960. [Cited in This Article: ] |
| 58. | Sentilhes L, Verspyck E, Pia P, Marpeau L. Fetal death in a patient with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:458-460. [Cited in This Article: ] |
| 59. | Lunzer M, Barnes P, Byth K, O'Halloran M. Serum bile acid concentrations during pregnancy and their relationship to obstetric cholestasis. Gastroenterology. 1986;91:825-829. [Cited in This Article: ] |
| 60. | Girling JC, Dow E, Smith JH. Liver function tests in pre-eclampsia: importance of comparison with a reference range derived for normal pregnancy. Br J Obstet Gynaecol. 1997;104:246-250. [Cited in This Article: ] |
| 61. | Bacq Y, Zarka O, Bréchot JF, Mariotte N, Vol S, Tichet J, Weill J. Liver function tests in normal pregnancy: a prospective study of 103 pregnant women and 103 matched controls. Hepatology. 1996;23:1030-1034. [Cited in This Article: ] |
| 62. | Heikkinen J. Serum bile acids in the early diagnosis of intrahepatic cholestasis of pregnancy. Obstet Gynecol. 1983;61:581-587. [Cited in This Article: ] |
| 63. | Milkiewicz P, Gallagher R, Chambers J, Eggington E, Weaver J, Elias E. Obstetric cholestasis with elevated gamma glutamyl transpeptidase: incidence, presentation and treatment. J Gastroenterol Hepatol. 2003;18:1283-1286. [Cited in This Article: ] |
| 64. | Beckett GJ, Hayes JD. Glutathione S-transferases: biomedical applications. Adv Clin Chem. 1993;30:281-380. [Cited in This Article: ] |
| 65. | Hayes PC, Hussey AJ, Keating J, Bouchier IA, Williams R, Beckett GJ, Hayes JD. Glutathione S-transferase levels in autoimmune chronic active hepatitis: a more sensitive index of hepatocellular damage than aspartate transaminase. Clin Chim Acta. 1988;172:211-216. [Cited in This Article: ] |
| 66. | Knapen MF, Peters WH, Mulder TP, Steegers EA. A marker for hepatocellular damage. Lancet. 2000;355:1463-1464. [Cited in This Article: ] |
| 67. | Dann AT, Kenyon AP, Seed PT, Poston L, Shennan AH, Tribe RM. Glutathione S-transferase and liver function in intrahepatic cholestasis of pregnancy and pruritus gravidarum. Hepatology. 2004;40:1406-1414. [Cited in This Article: ] |
| 68. | Joutsiniemi T, Leino R, Timonen S, Pulkki K, Ekblad U. Hepatocellular enzyme glutathione S-transferase alpha and intrahepatic cholestasis of pregnancy. Acta Obstet Gynecol Scand. 2008;87:1280-1284. [Cited in This Article: ] |
| 69. | Russell DW. The enzymes, regulation, and genetics of bile acid synthesis. Annu Rev Biochem. 2003;72:137-174. [Cited in This Article: ] |
| 70. | Trauner M, Boyer JL. Bile salt transporters: molecular characterization, function, and regulation. Physiol Rev. 2003;83:633-671. [Cited in This Article: ] |
| 71. | Carter J. Serum bile acids in normal pregnancy. Br J Obstet Gynaecol. 1991;98:540-543. [Cited in This Article: ] |
| 72. | Fulton IC, Douglas JG, Hutchon DJ, Beckett GJ. Is normal pregnancy cholestatic? Clin Chim Acta. 1983;130:171-176. [Cited in This Article: ] |
| 73. | Heikkinen J, Mäentausta O, Ylöstalo P, Jänne O. Changes in serum bile acid concentrations during normal pregnancy, in patients with intrahepatic cholestasis of pregnancy and in pregnant women with itching. Br J Obstet Gynaecol. 1981;88:240-245. [Cited in This Article: ] |
| 74. | Pascual MJ, Serrano MA, El-Mir MY, Macias RI, Jiménez F, Marin JJ. Relationship between asymptomatic hypercholanaemia of pregnancy and progesterone metabolism. Clin Sci (Lond). 2002;102:587-593. [Cited in This Article: ] |
| 75. | Castaño G, Lucangioli S, Sookoian S, Mesquida M, Lemberg A, Di Scala M, Franchi P, Carducci C, Tripodi V. Bile acid profiles by capillary electrophoresis in intrahepatic cholestasis of pregnancy. Clin Sci (Lond). 2006;110:459-465. [Cited in This Article: ] |
| 76. | Walker IA, Nelson-Piercy C, Williamson C. Role of bile acid measurement in pregnancy. Ann Clin Biochem. 2002;39:105-113. [Cited in This Article: ] |
| 77. | Sjövall K, Sjövall J. Serum bile acid levels in pregnancy with pruritus (bile acids and steroids 158). Clin Chim Acta. 1966;13:207-211. [Cited in This Article: ] |
| 78. | Laatikainen TJ. Fetal bile acid levels in pregnancies complicated by maternal intrahepatic cholestasis. Am J Obstet Gynecol. 1975;122:852-856. [Cited in This Article: ] |
| 79. | Colombo C, Roda A, Roda E, Buscaglia M, dell'Agnola CA, Filippetti P, Ronchi M, Sereni F. Correlation between fetal and maternal serum bile acid concentrations. Pediatr Res. 1985;19:227-231. [Cited in This Article: ] |
| 80. | Heikkinen J. Effect of a standard test meal on serum bile acid levels in healthy nonpregnant and pregnant women and in patients with intrahepatic cholestasis of pregnancy. Ann Clin Res. 1983;15:183-188. [Cited in This Article: ] |
| 81. | Laatikainen T. Postprandial serum bile acids in cholestasis of pregnancy. Ann Clin Res. 1978;10:307-312. [Cited in This Article: ] |
| 82. | Laatikainen T, Lehtonen P, Hesso A. Biliary bile acids in uncomplicated pregnancy and in cholestasis of pregnancy. Clin Chim Acta. 1978;85:145-150. [Cited in This Article: ] |
| 83. | Johnson P. Studies in cholestasis of pregnancy with special reference to lipids and lipoproteins. Acta Obstet Gynecol Scand Suppl. 1973;27:1-80. [Cited in This Article: ] |
| 84. | Nikkilä K, Riikonen S, Lindfors M, Miettinen TA. Serum squalene and noncholesterol sterols before and after delivery in normal and cholestatic pregnancy. J Lipid Res. 1996;37:2687-2695. [Cited in This Article: ] |
| 85. | Dann AT, Kenyon AP, Wierzbicki AS, Seed PT, Shennan AH, Tribe RM. Plasma lipid profiles of women with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:106-114. [Cited in This Article: ] |
| 86. | Dong M, Xie X, Wang Z, He J, Zhou J, Cheng Q. Impaired mixed lymphocyte reaction in intrahepatic cholestasis of pregnancy. Gynecol Obstet Invest. 2002;54:191-195. [Cited in This Article: ] |
| 87. | Eloranta ML, Heinonen S, Kirkinen P. Intrahepatic cholestasis of pregnancy has no effect on maternal serum second trimester alpha-fetoprotein and hCG. Acta Obstet Gynecol Scand. 2000;79:548-552. [Cited in This Article: ] |
| 88. | Garoff L. Prediction of fetal outcome by urinary estriol, maternal serum placental lactogen, and alpha-fetoprotein in diabetes and hepatosis of pregnancy. Obstet Gynecol. 1976;48:659-666. [Cited in This Article: ] |
| 89. | Heikinheimo M, Unnérus HA, Ranta T, Jalanko H, Seppälä M. Pregnancy-specific beta-1-glycoprotein levels in cholestasis of pregnancy. Obstet Gynecol. 1978;52:276-278. [Cited in This Article: ] |
| 90. | Kauppila A, Korpela H, Mäkilä UM, Yrjänheikki E. Low serum selenium concentration and glutathione peroxidase activity in intrahepatic cholestasis of pregnancy. Br Med J. 1987;294:150-152. [Cited in This Article: ] |
| 91. | Kiilholma P. Serum copper and zinc concentrations in intrahepatic cholestasis of pregnancy: a controlled study. Eur J Obstet Gynecol Reprod Biol. 1986;21:207-212. [Cited in This Article: ] |
| 92. | Kuoppala T, Tuimala R, Parviainen M, Koskinen T. Vitamin D and mineral metabolism in intrahepatic cholestasis of pregnancy. Eur J Obstet Gynecol Reprod Biol. 1986;23:45-51. [Cited in This Article: ] |
| 93. | Pineda G, Aguayo J, Ribalta J, González M, Reyes H. [Thyroid function tests in normal pregnant women (third trimester) and in pregnant women with pregnancy cholestasis or with acute hepatitis]. Rev Med Chil. 2000;128:35-43. [Cited in This Article: ] |
| 94. | Ranta T, Unnérus HA, Rossi J, Seppälä M. Elevated plasma prolactin concentration in cholestasis of pregnancy. Am J Obstet Gynecol. 1979;134:1-3. [Cited in This Article: ] |
| 95. | Räty R, Anttila L, Virtanen A, Koskinen P, Laitinen P, Mörsky P, Tiitinen A, Martikainen H, Ekblad U. Maternal midtrimester free beta-HCG and AFP serum levels in spontaneous singleton pregnancies complicated by gestational diabetes mellitus, pregnancy-induced hypertension or obstetric cholestasis. Prenat Diagn. 2003;23:1045-1048. [Cited in This Article: ] |
| 96. | Reyes H, Báez ME, González MC, Hernández I, Palma J, Ribalta J, Sandoval L, Zapata R. Selenium, zinc and copper plasma levels in intrahepatic cholestasis of pregnancy, in normal pregnancies and in healthy individuals, in Chile. J Hepatol. 2000;32:542-549. [Cited in This Article: ] |
| 97. | Smolarczyk R, Wójcicka-Jagodzińska J, Piekarski P, Romejko E, Czajkowski K. The biochemical functions of the renal tubules and glomeruli in the course of intrahepatic cholestasis in pregnancy. Eur J Obstet Gynecol Reprod Biol. 2000;89:35-39. [Cited in This Article: ] |
| 98. | Tiitinen A, Laatikainen T, Rutanen EM, Ranta T, Koistinen R, Bohn H, Seppälä M. Placental protein 10 (PP10) in normal pregnancy and cholestasis of pregnancy. Br J Obstet Gynaecol. 1985;92:1137-1140. [Cited in This Article: ] |
| 99. | Wang Z, Dong M, Chu H, He J. Increased serum levels of neopterin and soluble interleukin-2 receptor in intrahepatic cholestasis of pregnancy. Acta Obstet Gynecol Scand. 2004;83:1067-1070. [Cited in This Article: ] |
| 100. | Wojcicka J, Sienko J, Smolarczyk R, Romejko E, Grymowicz M, Czajkowski K. Alpha-hydroxybutyrate dehydrogenase activity in intrahepatic cholestasis of pregnancy. Int J Gynaecol Obstet. 2005;89:247-250. [Cited in This Article: ] |
| 101. | Thomassen PA. Urinary bile acids in late pregnancy and in recurrent cholestasis of pregnancy. Eur J Clin Invest. 1979;9:425-432. [Cited in This Article: ] |
| 102. | Thomassen PA. Urinary bile acids during development of recurrent cholestasis of pregnancy. Eur J Clin Invest. 1979;9:417-423. [Cited in This Article: ] |
| 103. | Ko CW, Beresford SA, Schulte SJ, Matsumoto AM, Lee SP. Incidence, natural history, and risk factors for biliary sludge and stones during pregnancy. Hepatology. 2005;41:359-365. [Cited in This Article: ] |
| 104. | Tsimoyiannis EC, Antoniou NC, Tsaboulas C, Papanikolaou N. Cholelithiasis during pregnancy and lactation. Prospective study. Eur J Surg. 1994;160:627-631. [Cited in This Article: ] |
| 105. | Kirkinen P, Ylöstalo P, Heikkinen J, Mäentausta O. Gallbladder function and maternal bile acids in intrahepatic cholestasis of pregnancy. Eur J Obstet Gynecol Reprod Biol. 1984;18:29-34. [Cited in This Article: ] |
| 106. | Ylöstalo P, Kirkinen P, Heikkinen J, Mäentausta O, Järvinen PA. Gall bladder volume and serum bile acids in cholestasis of pregnancy. Br J Obstet Gynaecol. 1982;89:59-61. [Cited in This Article: ] |
| 107. | Ylötalo P, Kirkinen P, Heikkinen J, Mäentausta O. Gallbladder volume in cholestasis of pregnancy. N Engl J Med. 1981;304:359. [Cited in This Article: ] |
| 108. | Ikonen E. Jaundice in Late Pregnancy. Acta Obstet Gynecol Scand. 1964;43 Suppl 5:1-130. [Cited in This Article: ] |
| 109. | Eliakim M, Sadovsky E, Stein O, Shenkar YG. Recurrent cholestatic jaundice of pregnancy. Report of five cases and electron microscopic observations. Arch Intern Med. 1966;117:696-705. [Cited in This Article: ] |
| 110. | Reyes H, Ribalta J, González-Cerón M. Idiopathic cholestasis of pregnancy in a large kindred. Gut. 1976;17:709-713. [Cited in This Article: ] |
| 111. | Dixon PH, Williamson C. The molecular genetics of intrahepatic cholestasis of pregnancy. Obstet Med. 2008;1:65-71. [Cited in This Article: ] |
| 112. | Ruetz S, Gros P. Phosphatidylcholine translocase: a physiological role for the mdr2 gene. Cell. 1994;77:1071-1081. [Cited in This Article: ] |
| 113. | Smith AJ, Timmermans-Hereijgers JL, Roelofsen B, Wirtz KW, van Blitterswijk WJ, Smit JJ, Schinkel AH, Borst P. The human MDR3 P-glycoprotein promotes translocation of phosphatidylcholine through the plasma membrane of fibroblasts from transgenic mice. FEBS Lett. 1994;354:263-266. [Cited in This Article: ] |
| 114. | van Helvoort A, Smith AJ, Sprong H, Fritzsche I, Schinkel AH, Borst P, van Meer G. MDR1 P-glycoprotein is a lipid translocase of broad specificity, while MDR3 P-glycoprotein specifically translocates phosphatidylcholine. Cell. 1996;87:507-517. [Cited in This Article: ] |
| 115. | de Vree JM, Jacquemin E, Sturm E, Cresteil D, Bosma PJ, Aten J, Deleuze JF, Desrochers M, Burdelski M, Bernard O. Mutations in the MDR3 gene cause progressive familial intrahepatic cholestasis. Proc Natl Acad Sci USA. 1998;95:282-287. [Cited in This Article: ] |
| 116. | Rosmorduc O, Hermelin B, Poupon R. MDR3 gene defect in adults with symptomatic intrahepatic and gallbladder cholesterol cholelithiasis. Gastroenterology. 2001;120:1459-1467. [Cited in This Article: ] |
| 117. | Jacquemin E, Cresteil D, Manouvrier S, Boute O, Hadchouel M. Heterozygous non-sense mutation of the MDR3 gene in familial intrahepatic cholestasis of pregnancy. Lancet. 1999;353:210-211. [Cited in This Article: ] |
| 118. | Eloranta ML, Heiskanen JT, Hiltunen MJ, Mannermaa AJ, Punnonen KR, Heinonen ST. Multidrug resistance 3 gene mutation 1712delT and estrogen receptor alpha gene polymorphisms in Finnish women with obstetric cholestasis. Eur J Obstet Gynecol Reprod Biol. 2002;105:132-135. [Cited in This Article: ] |
| 119. | Dixon PH, Weerasekera N, Linton KJ, Donaldson O, Chambers J, Egginton E, Weaver J, Nelson-Piercy C, de Swiet M, Warnes G. Heterozygous MDR3 missense mutation associated with intrahepatic cholestasis of pregnancy: evidence for a defect in protein trafficking. Hum Mol Genet. 2000;9:1209-1217. [Cited in This Article: ] |
| 120. | Floreani A, Carderi I, Paternoster D, Soardo G, Azzaroli F, Esposito W, Montagnani M, Marchesoni D, Variola A, Rosa Rizzotto E. Hepatobiliary phospholipid transporter ABCB4, MDR3 gene variants in a large cohort of Italian women with intrahepatic cholestasis of pregnancy. Dig Liver Dis. 2008;40:366-370. [Cited in This Article: ] |
| 121. | Floreani A, Carderi I, Paternoster D, Soardo G, Azzaroli F, Esposito W, Variola A, Tommasi AM, Marchesoni D, Braghin C. Intrahepatic cholestasis of pregnancy: three novel MDR3 gene mutations. Aliment Pharmacol Ther. 2006;23:1649-1653. [Cited in This Article: ] |
| 122. | Gendrot C, Bacq Y, Brechot MC, Lansac J, Andres C. A second heterozygous MDR3 nonsense mutation associated with intrahepatic cholestasis of pregnancy. J Med Genet. 2003;40:e32. [Cited in This Article: ] |
| 123. | Keitel V, Vogt C, Häussinger D, Kubitz R. Combined mutations of canalicular transporter proteins cause severe intrahepatic cholestasis of pregnancy. Gastroenterology. 2006;131:624-629. [Cited in This Article: ] |
| 124. | Lucena JF, Herrero JI, Quiroga J, Sangro B, Garcia-Foncillas J, Zabalegui N, Sola J, Herraiz M, Medina JF, Prieto J. A multidrug resistance 3 gene mutation causing cholelithiasis, cholestasis of pregnancy, and adulthood biliary cirrhosis. Gastroenterology. 2003;124:1037-1042. [Cited in This Article: ] |
| 125. | Müllenbach R, Linton KJ, Wiltshire S, Weerasekera N, Chambers J, Elias E, Higgins CF, Johnston DG, McCarthy MI, Williamson C. ABCB4 gene sequence variation in women with intrahepatic cholestasis of pregnancy. J Med Genet. 2003;40:e70. [Cited in This Article: ] |
| 126. | Pauli-Magnus C, Lang T, Meier Y, Zodan-Marin T, Jung D, Breymann C, Zimmermann R, Kenngott S, Beuers U, Reichel C. Sequence analysis of bile salt export pump (ABCB11) and multidrug resistance p-glycoprotein 3 (ABCB4, MDR3) in patients with intrahepatic cholestasis of pregnancy. Pharmacogenetics. 2004;14:91-102. [Cited in This Article: ] |
| 127. | Wasmuth HE, Glantz A, Keppeler H, Simon E, Bartz C, Rath W, Mattsson LA, Marschall HU, Lammert F. Intrahepatic cholestasis of pregnancy: the severe form is associated with common variants of the hepatobiliary phospholipid transporter ABCB4 gene. Gut. 2007;56:265-270. [Cited in This Article: ] |
| 128. | Müllenbach R, Bennett A, Tetlow N, Patel N, Hamilton G, Cheng F, Chambers J, Howard R, Taylor-Robinson SD, Williamson C. ATP8B1 mutations in British cases with intrahepatic cholestasis of pregnancy. Gut. 2005;54:829-834. [Cited in This Article: ] |
| 129. | Painter JN, Savander M, Ropponen A, Nupponen N, Riikonen S, Ylikorkala O, Lehesjoki AE, Aittomäki K. Sequence variation in the ATP8B1 gene and intrahepatic cholestasis of pregnancy. Eur J Hum Genet. 2005;13:435-439. [Cited in This Article: ] |
| 130. | Eloranta ML, Häkli T, Hiltunen M, Helisalmi S, Punnonen K, Heinonen S. Association of single nucleotide polymorphisms of the bile salt export pump gene with intrahepatic cholestasis of pregnancy. Scand J Gastroenterol. 2003;38:648-652. [Cited in This Article: ] |
| 131. | Painter JN, Savander M, Sistonen P, Lehesjoki AE, Aittomäki K. A known polymorphism in the bile salt export pump gene is not a risk allele for intrahepatic cholestasis of pregnancy. Scand J Gastroenterol. 2004;39:694-695. [Cited in This Article: ] |
| 132. | Savander M, Ropponen A, Avela K, Weerasekera N, Cormand B, Hirvioja ML, Riikonen S, Ylikorkala O, Lehesjoki AE, Williamson C. Genetic evidence of heterogeneity in intrahepatic cholestasis of pregnancy. Gut. 2003;52:1025-1029. [Cited in This Article: ] |
| 133. | Meier Y, Zodan T, Lang C, Zimmermann R, Kullak-Ublick GA, Meier PJ, Stieger B, Pauli-Magnus C. Increased susceptibility for intrahepatic cholestasis of pregnancy and contraceptive-induced cholestasis in carriers of the 1331T>C polymorphism in the bile salt export pump. World J Gastroenterol. 2008;14:38-45. [Cited in This Article: ] |
| 134. | Dixon PH, van Mil SW, Chambers J, Strautnieks S, Thompson RJ, Lammert F, Kubitz R, Keitel V, Glantz A, Mattsson LA. Contribution of variant alleles of ABCB11 to susceptibility to intrahepatic cholestasis of pregnancy. Gut. 2009;58:537-544. [Cited in This Article: ] |
| 135. | Sookoian S, Castaño G, Burgueño A, Gianotti TF, Pirola CJ. Association of the multidrug-resistance-associated protein gene (ABCC2) variants with intrahepatic cholestasis of pregnancy. J Hepatol. 2008;48:125-132. [Cited in This Article: ] |
| 136. | Van Mil SW, Milona A, Dixon PH, Mullenbach R, Geenes VL, Chambers J, Shevchuk V, Moore GE, Lammert F, Glantz AG. Functional variants of the central bile acid sensor FXR identified in intrahepatic cholestasis of pregnancy. Gastroenterology. 2007;133:507-516. [Cited in This Article: ] |
| 137. | Reyes H, Ribalta J, González MC, Segovia N, Oberhauser E. Sulfobromophthalein clearance tests before and after ethinyl estradiol administration, in women and men with familial history of intrahepatic cholestasis of pregnancy. Gastroenterology. 1981;81:226-231. [Cited in This Article: ] |
| 138. | Barth A, Klinger G, Rost M. Influence of ethinyloestradiol propanolsulphonate on serum bile acids in healthy volunteers. Exp Toxicol Pathol. 2003;54:381-386. [Cited in This Article: ] |
| 139. | Stieger B, Fattinger K, Madon J, Kullak-Ublick GA, Meier PJ. Drug- and estrogen-induced cholestasis through inhibition of the hepatocellular bile salt export pump (Bsep) of rat liver. Gastroenterology. 2000;118:422-430. [Cited in This Article: ] |
| 140. | Crocenzi FA, Mottino AD, Cao J, Veggi LM, Pozzi EJ, Vore M, Coleman R, Roma MG. Estradiol-17beta-D-glucuronide induces endocytic internalization of Bsep in rats. Am J Physiol Gastrointest Liver Physiol. 2003;285:G449-G459. [Cited in This Article: ] |
| 141. | Simon FR, Fortune J, Iwahashi M, Qadri I, Sutherland E. Multihormonal regulation of hepatic sinusoidal Ntcp gene expression. Am J Physiol Gastrointest Liver Physiol. 2004;287:G782-G794. [Cited in This Article: ] |
| 142. | Yamamoto Y, Moore R, Hess HA, Guo GL, Gonzalez FJ, Korach KS, Maronpot RR, Negishi M. Estrogen receptor alpha mediates 17alpha-ethynylestradiol causing hepatotoxicity. J Biol Chem. 2006;281:16625-16631. [Cited in This Article: ] |
| 143. | Geier A, Dietrich CG, Gerloff T, Haendly J, Kullak-Ublick GA, Stieger B, Meier PJ, Matern S, Gartung C. Regulation of basolateral organic anion transporters in ethinylestradiol-induced cholestasis in the rat. Biochim Biophys Acta. 2003;1609:87-94. [Cited in This Article: ] |
| 144. | Bacq Y, Myara A, Brechot MC, Hamon C, Studer E, Trivin F, Metman EH. Serum conjugated bile acid profile during intrahepatic cholestasis of pregnancy. J Hepatol. 1995;22:66-70. [Cited in This Article: ] |
| 145. | Benifla JL, Dumont M, Levardon M, Foucher E, Cadiot G, Crenn-Hebert C, Heid M, Lelaidier C, Rosenbaum A, Bernuau J. [Effects of micronized natural progesterone on the liver during the third trimester of pregnancy]. Contracept Fertil Sex. 1997;25:165-169. [Cited in This Article: ] |
| 146. | Meng LJ, Reyes H, Axelson M, Palma J, Hernandez I, Ribalta J, Sjövall J. Progesterone metabolites and bile acids in serum of patients with intrahepatic cholestasis of pregnancy: effect of ursodeoxycholic acid therapy. Hepatology. 1997;26:1573-1579. [Cited in This Article: ] |
| 147. | Meng LJ, Reyes H, Palma J, Hernandez I, Ribalta J, Sjövall J. Profiles of bile acids and progesterone metabolites in the urine and serum of women with intrahepatic cholestasis of pregnancy. J Hepatol. 1997;27:346-357. [Cited in This Article: ] |
| 148. | Laatikainen TJ, Peltonen JI, Nylander PL. Effect of maternal intrahepatic cholestasis on fetal steroid metabolism. J Clin Invest. 1974;53:1709-1715. [Cited in This Article: ] |
| 149. | Vallejo M, Briz O, Serrano MA, Monte MJ, Marin JJ. Potential role of trans-inhibition of the bile salt export pump by progesterone metabolites in the etiopathogenesis of intrahepatic cholestasis of pregnancy. J Hepatol. 2006;44:1150-1157. [Cited in This Article: ] |
| 150. | Lang C, Meier Y, Stieger B, Beuers U, Lang T, Kerb R, Kullak-Ublick GA, Meier PJ, Pauli-Magnus C. Mutations and polymorphisms in the bile salt export pump and the multidrug resistance protein 3 associated with drug-induced liver injury. Pharmacogenet Genomics. 2007;17:47-60. [Cited in This Article: ] |
| 151. | Reyes H, Zapata R, Hernández I, Gotteland M, Sandoval L, Jirón MI, Palma J, Almuna R, Silva JJ. Is a leaky gut involved in the pathogenesis of intrahepatic cholestasis of pregnancy? Hepatology. 2006;43:715-722. [Cited in This Article: ] |
| 152. | Kirby J, Heaton KW, Burton JL. Pruritic effect of bile salts. Br Med J. 1974;4:693-695. [Cited in This Article: ] |
| 153. | Varadi DP. Pruritus induced by crude bile and purified bile acids. Experimental production of pruritus in human skin. Arch Dermatol. 1974;109:678-681. [Cited in This Article: ] |
| 154. | Ghent CN, Bloomer JR, Klatskin G. Elevations in skin tissue levels of bile acids in human cholestasis: relation to serum levels and topruritus. Gastroenterology. 1977;73:1125-1130. [Cited in This Article: ] |
| 155. | Glantz A, Reilly SJ, Benthin L, Lammert F, Mattsson LA, Marschall HU. Intrahepatic cholestasis of pregnancy: Amelioration of pruritus by UDCA is associated with decreased progesterone disulphates in urine. Hepatology. 2008;47:544-551. [Cited in This Article: ] |
| 156. | Schumann R, Hudcova J. Cholestasis of pregnancy, pruritus and 5-hydroxytryptamine 3 receptor antagonists. Acta Obstet Gynecol Scand. 2004;83:861-862. [Cited in This Article: ] |
| 157. | Schwörer H, Ramadori G. Improvement of cholestatic pruritus by ondansetron. Lancet. 1993;341:1277. [Cited in This Article: ] |
| 158. | Marin JJ, Serrano MA, el-Mir MY, Eleno N, Boyd CA. Bile acid transport by basal membrane vesicles of human term placental trophoblast. Gastroenterology. 1990;99:1431-1438. [Cited in This Article: ] |
| 159. | Serrano MA, Brites D, Larena MG, Monte MJ, Bravo MP, Oliveira N, Marin JJ. Beneficial effect of ursodeoxycholic acid on alterations induced by cholestasis of pregnancy in bile acid transport across the human placenta. J Hepatol. 1998;28:829-839. [Cited in This Article: ] |
| 160. | Campos GA, Guerra FA, Israel EJ. Effects of cholic acid infusion in fetal lambs. Acta Obstet Gynecol Scand. 1986;65:23-26. [Cited in This Article: ] |
| 161. | Falconer JD, Smith AN, Eastwood MA. The effects of bile acids on colonic motility in the rabbit. Q J Exp Physiol Cogn Med Sci. 1980;65:135-144. [Cited in This Article: ] |
| 162. | Kirwan WO, Smith AN, Mitchell WD, Falconer JD, Eastwood MA. Bile acids and colonic motility in the rabbit and the human. Gut. 1975;16:894-902. [Cited in This Article: ] |
| 163. | Williamson C, Gorelik J, Eaton BM, Lab M, de Swiet M, Korchev Y. The bile acid taurocholate impairs rat cardiomyocyte function: a proposed mechanism for intra-uterine fetal death in obstetric cholestasis. Clin Sci (Lond). 2001;100:363-369. [Cited in This Article: ] |
| 164. | Campos GA, Castillo RJ, Toro FG. [Effect of bile acids on the myometral contractility of the isolated pregnant uterus]. Rev Chil Obstet Ginecol. 1988;53:229-233. [Cited in This Article: ] |
| 165. | Germain AM, Kato S, Carvajal JA, Valenzuela GJ, Valdes GL, Glasinovic JC. Bile acids increase response and expression of human myometrial oxytocin receptor. Am J Obstet Gynecol. 2003;189:577-582. [Cited in This Article: ] |
| 166. | Israel EJ, Guzman ML, Campos GA. Maximal response to oxytocin of the isolated myometrium from pregnant patients with intrahepatic cholestasis. Acta Obstet Gynecol Scand. 1986;65:581-582. [Cited in This Article: ] |
| 167. | Brown ES. Aspiration and lung surfactant. Anesth Analg. 1967;46:665-672. [Cited in This Article: ] |
| 168. | Porembka DT, Kier A, Sehlhorst S, Boyce S, Orlowski JP, Davis K Jr. The pathophysiologic changes following bile aspiration in a porcine lung model. Chest. 1993;104:919-924. [Cited in This Article: ] |
| 169. | Kaneko T, Sato T, Katsuya H, Miyauchi Y. Surfactant therapy for pulmonary edema due to intratracheally injected bile acid. Crit Care Med. 1990;18:77-83. [Cited in This Article: ] |
| 170. | Costoya AL, Leontic EA, Rosenberg HG, Delgado MA. Morphological study of placental terminal villi in intrahepatic cholestasis of pregnancy: histochemistry, light and electron microscopy. Placenta. 1980;1:361-368. [Cited in This Article: ] |
| 171. | Liebhart M, Wójcicka J. Microscopic patterns of placenta in cases of pregnancy complicated by intrahepatic cholestasis (idiopathic jaundice). Pol Med J. 1970;9:1589-1600. [Cited in This Article: ] |
| 172. | Perez MJ, Macias RI, Marin JJ. Maternal cholestasis induces placental oxidative stress and apoptosis. Protective effect of ursodeoxycholic acid. Placenta. 2006;27:34-41. [Cited in This Article: ] |
| 173. | Miller PW, Coen RW, Benirschke K. Dating the time interval from meconium passage to birth. Obstet Gynecol. 1985;66:459-462. [Cited in This Article: ] |
| 174. | Altshuler G, Hyde S. Meconium-induced vasocontraction: a potential cause of cerebral and other fetal hypoperfusion and of poor pregnancy outcome. J Child Neurol. 1989;4:137-142. [Cited in This Article: ] |
| 175. | Rodrigues CM, Marín JJ, Brites D. Bile acid patterns in meconium are influenced by cholestasis of pregnancy and not altered by ursodeoxycholic acid treatment. Gut. 1999;45:446-452. [Cited in This Article: ] |
| 176. | Sepúlveda WH, González C, Cruz MA, Rudolph MI. Vasoconstrictive effect of bile acids on isolated human placental chorionic veins. Eur J Obstet Gynecol Reprod Biol. 1991;42:211-215. [Cited in This Article: ] |
| 177. | Matos A, Bernardes J, Ayres-de-Campos D, Patrício B. Antepartum fetal cerebral hemorrhage not predicted by current surveillance methods in cholestasis of pregnancy. Obstet Gynecol. 1997;89:803-804. [Cited in This Article: ] |
| 178. | Medina Lomelí JM, Medina Castro N. [Intrahepatic cholestasis of pregnancy, an unpredictable fetal risk: report of a case and review of the literature]. Ginecol Obstet Mex. 2000;68:486-488. [Cited in This Article: ] |
| 179. | Lazaridis KN, Gores GJ, Lindor KD. Ursodeoxycholic acid 'mechanisms of action and clinical use in hepatobiliary disorders'. J Hepatol. 2001;35:134-146. [Cited in This Article: ] |
| 180. | Rodrigues CM, Fan G, Wong PY, Kren BT, Steer CJ. Ursodeoxycholic acid may inhibit deoxycholic acid-induced apoptosis by modulating mitochondrial transmembrane potential and reactive oxygen species production. Mol Med. 1998;4:165-178. [Cited in This Article: ] |
| 181. | Palma J, Reyes H, Ribalta J, Iglesias J, Gonzalez MC, Hernandez I, Alvarez C, Molina C, Danitz AM. Effects of ursodeoxycholic acid in patients with intrahepatic cholestasis of pregnancy. Hepatology. 1992;15:1043-1047. [Cited in This Article: ] |
| 182. | Diaferia A, Nicastri PL, Tartagni M, Loizzi P, Iacovizzi C, Di Leo A. Ursodeoxycholic acid therapy in pregnant women with cholestasis. Int J Gynaecol Obstet. 1996;52:133-140. [Cited in This Article: ] |
| 183. | Nicastri PL, Diaferia A, Tartagni M, Loizzi P, Fanelli M. A randomised placebo-controlled trial of ursodeoxycholic acid and S-adenosylmethionine in the treatment of intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol. 1998;105:1205-1207. [Cited in This Article: ] |
| 184. | Palma J, Reyes H, Ribalta J, Hernández I, Sandoval L, Almuna R, Liepins J, Lira F, Sedano M, Silva O. Ursodeoxycholic acid in the treatment of cholestasis of pregnancy: a randomized, double-blind study controlled with placebo. J Hepatol. 1997;27:1022-1028. [Cited in This Article: ] |
| 185. | Berkane N, Cocheton JJ, Brehier D, Merviel P, Wolf C, Lefèvre G, Uzan S. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy. A retrospective study of 19 cases. Acta Obstet Gynecol Scand. 2000;79:941-946. [Cited in This Article: ] |
| 186. | Floreani A, Paternoster D, Grella V, Sacco S, Gangemi M, Chiaramonte M. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol. 1994;101:64-65. [Cited in This Article: ] |
| 187. | Mazzella G, Rizzo N, Azzaroli F, Simoni P, Bovicelli L, Miracolo A, Simonazzi G, Colecchia A, Nigro G, Mwangemi C. Ursodeoxycholic acid administration in patients with cholestasis of pregnancy: effects on primary bile acids in babies and mothers. Hepatology. 2001;33:504-508. [Cited in This Article: ] |
| 188. | Zapata R, Sandoval L, Palma J, Hernández I, Ribalta J, Reyes H, Sedano M, Tohá D, Silva JJ. Ursodeoxycholic acid in the treatment of intrahepatic cholestasis of pregnancy. A 12-year experience. Liver Int. 2005;25:548-554. [Cited in This Article: ] |
| 189. | Glantz A, Marschall HU, Lammert F, Mattsson LA. Intrahepatic cholestasis of pregnancy: a randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology. 2005;42:1399-1405. [Cited in This Article: ] |
| 190. | Brites D, Rodrigues CM, Oliveira N, Cardoso M, Graça LM. Correction of maternal serum bile acid profile during ursodeoxycholic acid therapy in cholestasis of pregnancy. J Hepatol. 1998;28:91-98. [Cited in This Article: ] |
| 191. | Brites D, El-Mir MY, Oliveira N, Marín JJG. Amniotic fluid bile acid changes in the course of ursodeoxycholic acid therapy in intrahepatic cholestasis of pregnancy [Abstract]. J Hepatol. 1997;26:164A. [Cited in This Article: ] |
| 192. | Brites D, El-Mir MY, Rodrigues CMP, van-Zeller H, Marín JJG. Bile acid composition of amniotic fluid and maternal serum in cholestasis of pregnancy and effect of ursodeoxycholic acid [Abstract]. J Hepatol. 1998;28:125A. [Cited in This Article: ] |
| 193. | Brites D, Rodrigues CM. Elevated levels of bile acids in colostrum of patients with cholestasis of pregnancy are decreased following ursodeoxycholic acid therapy [see comemnts]. J Hepatol. 1998;29:743-751. [Cited in This Article: ] |
| 194. | Azzaroli F, Mennone A, Feletti V, Simoni P, Baglivo E, Montagnani M, Rizzo N, Pelusi G, DE Aloysio D, Lodato F. Clinical trial: modulation of human placental multidrug resistance proteins in cholestasis of pregnancy by ursodeoxycholic acid. Aliment Pharmacol Ther. 2007;26:1139-1146. [Cited in This Article: ] |
| 195. | Serrano MA, Macias RI, Vallejo M, Briz O, Bravo A, Pascual MJ, St-Pierre MV, Stieger B, Meier PJ, Marin JJ. Effect of ursodeoxycholic acid on the impairment induced by maternal cholestasis in the rat placenta-maternal liver tandem excretory pathway. J Pharmacol Exp Ther. 2003;305:515-524. [Cited in This Article: ] |
| 196. | Gorelik J, Shevchuk AI, Diakonov I, de Swiet M, Lab M, Korchev Y, Williamson C. Dexamethasone and ursodeoxycholic acid protect against the arrhythmogenic effect of taurocholate in an in vitro study of rat cardiomyocytes. BJOG. 2003;110:467-474. [Cited in This Article: ] |
| 197. | Kauppila A, Jouppila P, Karvonen P, Tuimala R, Ylikorkala O. Effect of dexamethasone on blood levels of ACTH, cortisol, progesterone, estradiol and estriol during late pregnancy. Int J Gynaecol Obstet. 1976;14:177-181. [Cited in This Article: ] |
| 198. | Kauppila A, Tuimala R, Ylikorkala O, Reinilä M, Ylöstalo P. Placental steroid synthesis from DHEAS during dexamethasone therapy. Obstet Gynecol. 1979;54:39-42. [Cited in This Article: ] |
| 199. | Hirvioja ML, Tuimala R, Vuori J. The treatment of intrahepatic cholestasis of pregnancy by dexamethasone. Br J Obstet Gynaecol. 1992;99:109-111. [Cited in This Article: ] |
| 200. | Diac M, Kenyon A, Nelson-Piercy C, Girling J, Cheng F, Tribe RM, Goodman J, Shennan A, Williamson C. Dexamethasone in the treatment of obstetric cholestasis: a case series. J Obstet Gynaecol. 2006;26:110-114. [Cited in This Article: ] |
| 201. | Kretowicz E, McIntyre HD. Intrahepatic cholestasis of pregnancy, worsening after dexamethasone. Aust N Z J Obstet Gynaecol. 1994;34:211-213. [Cited in This Article: ] |
| 202. | Bloom SL, Sheffield JS, McIntire DD, Leveno KJ. Antenatal dexamethasone and decreased birth weight. Obstet Gynecol. 2001;97:485-490. [Cited in This Article: ] |
| 203. | Modi N, Lewis H, Al-Naqeeb N, Ajayi-Obe M, Doré CJ, Rutherford M. The effects of repeated antenatal glucocorticoid therapy on the developing brain. Pediatr Res. 2001;50:581-585. [Cited in This Article: ] |
| 204. | Marschall HU, Wagner M, Zollner G, Fickert P, Diczfalusy U, Gumhold J, Silbert D, Fuchsbichler A, Benthin L, Grundström R. Complementary stimulation of hepatobiliary transport and detoxification systems by rifampicin and ursodeoxycholic acid in humans. Gastroenterology. 2005;129:476-485. [Cited in This Article: ] |
| 205. | Bachs L, Parés A, Elena M, Piera C, Rodés J. Comparison of rifampicin with phenobarbitone for treatment of pruritus in biliary cirrhosis. Lancet. 1989;1:574-576. [Cited in This Article: ] |
| 206. | Hoensch HP, Balzer K, Dylewizc P, Kirch W, Goebell H, Ohnhaus EE. Effect of rifampicin treatment on hepatic drug metabolism and serum bile acids in patients with primary biliary cirrhosis. Eur J Clin Pharmacol. 1985;28:475-477. [Cited in This Article: ] |
| 207. | Boelsterli UA, Rakhit G, Balazs T. Modulation by S-adenosyl-L-methionine of hepatic Na+,K+-ATPase, membrane fluidity, and bile flow in rats with ethinyl estradiol-induced cholestasis. Hepatology. 1983;3:12-17. [Cited in This Article: ] |
| 208. | Stramentinoli G, Di Padova C, Gualano M, Rovagnati P, Galli-Kienle M. Ethynylestradiol-induced impairment of bile secretion in the rat: protective effects of S-adenosyl-L-methionine and its implication in estrogen metabolism. Gastroenterology. 1981;80:154-158. [Cited in This Article: ] |
| 209. | Stramentinoli G, Gualano M, Di Padova C. Effect of S-adenosyl-L-methionine on ethynylestradiol-induced impairment of bile flow in female rats. Experientia. 1977;33:1361-1362. [Cited in This Article: ] |
| 210. | Stramentinoli G, Gualano M, Rovagnati P, Di Padova C. Influence of S-adenosyl-L-methionine on irreversible binding of ethynylestradiol to rat liver microsomes, and its implication in bile secretion. Biochem Pharmacol. 1979;28:981-984. [Cited in This Article: ] |
| 211. | Frezza M, Tritapepe R, Pozzato G, Di Padova C. Prevention of S-adenosylmethionine of estrogen-induced hepatobiliary toxicity in susceptible women. Am J Gastroenterol. 1988;83:1098-1102. [Cited in This Article: ] |
| 212. | Frezza M, Pozzato G, Chiesa L, Stramentinoli G, di Padova C. Reversal of intrahepatic cholestasis of pregnancy in women after high dose S-adenosyl-L-methionine administration. Hepatology. 1984;4:274-278. [Cited in This Article: ] |
| 213. | Bonfirraro G, Chieffi O, Quinti R, Tedesco R, LeGrazie C, Bortolini M. S-adenosyl-L-methionine (SAMe)-induced amelioration of intrahepatic cholestasis of pregnancy. Results of an open study. Drug Invest. 1990;2:125-128. [Cited in This Article: ] |
| 214. | Frezza M, Centini G, Cammareri G, Le Grazie C, Di Padova C. S-adenosylmethionine for the treatment of intrahepatic cholestasis of pregnancy. Results of a controlled clinical trial. Hepatogastroenterology. 1990;37 Suppl 2:122-125. [Cited in This Article: ] |
| 215. | Ribalta J, Reyes H, Gonzalez MC, Iglesias J, Arrese M, Poniachik J, Molina C, Segovia N. S-adenosyl-L-methionine in the treatment of patients with intrahepatic cholestasis of pregnancy: a randomized, double-blind, placebo-controlled study with negative results. Hepatology. 1991;13:1084-1089. [Cited in This Article: ] |
| 216. | Heikkinen J, Mäentausta O, Ylöstalo P, Jänne O. Serum bile acid levels in intrahepatic cholestasis of pregnancy during treatment with phenobarbital or cholestyramine. Eur J Obstet Gynecol Reprod Biol. 1982;14:153-162. [Cited in This Article: ] |
| 217. | Lutz EE, Margolis AJ. Obstetric hepatosis: treatment with cholestyramine and interim response to steroids. Obstet Gynecol. 1969;33:64-71. [Cited in This Article: ] |
| 218. | Sadler LC, Lane M, North R. Severe fetal intracranial haemorrhage during treatment with cholestyramine for intrahepatic cholestasis of pregnancy. Br J Obstet Gynaecol. 1995;102:169-170. [Cited in This Article: ] |
| 219. | Gylling H, Riikonen S, Nikkilä K, Savonius H, Miettinen TA. Oral guar gum treatment of intrahepatic cholestasis and pruritus in pregnant women: effects on serum cholestanol and other non-cholesterol sterols. Eur J Clin Invest. 1998;28:359-363. [Cited in This Article: ] |
| 220. | Riikonen S, Savonius H, Gylling H, Nikkilä K, Tuomi AM, Miettinen TA. Oral guar gum, a gel-forming dietary fiber relieves pruritus in intrahepatic cholestasis of pregnancy. Acta Obstet Gynecol Scand. 2000;79:260-264. [Cited in This Article: ] |
| 221. | Kaaja RJ, Kontula KK, Räihä A, Laatikainen T. Treatment of cholestasis of pregnancy with peroral activated charcoal. A preliminary study. Scand J Gastroenterol. 1994;29:178-181. [Cited in This Article: ] |
| 222. | Arrese M, Reyes H. Intrahepatic cholestasis of pregnancy: a past and present riddle. Ann Hepatol. 2006;5:202-205. [Cited in This Article: ] |
| 223. | Furhoff AK. Itching in pregnancy. A 15-year follow-up study. Acta Med Scand. 1974;196:403-410. [Cited in This Article: ] |
| 224. | Ropponen A, Sund R, Riikonen S, Ylikorkala O, Aittomäki K. Intrahepatic cholestasis of pregnancy as an indicator of liver and biliary diseases: a population-based study. Hepatology. 2006;43:723-728. [Cited in This Article: ] |