Published online Mar 6, 2021. doi: 10.12998/wjcc.v9.i7.1696
Peer-review started: October 13, 2020
First decision: December 13, 2020
Revised: December 28, 2020
Accepted: January 22, 2021
Article in press: January 22, 2021
Published online: March 6, 2021
Thrombocytopenia is a serious complication in the medical practice of numerous drugs. Vancomycin is frequently used for the prophylaxis and treatment of suspected or identified methicillin-resistant positive infections. Several cases with vancomycin-induced thrombocytopenia (VIT) have been reported. However, these have rarely been extensively reviewed. The present report describes a case of VIT in endocarditis, and reviews all VIT cases reported in the literature.
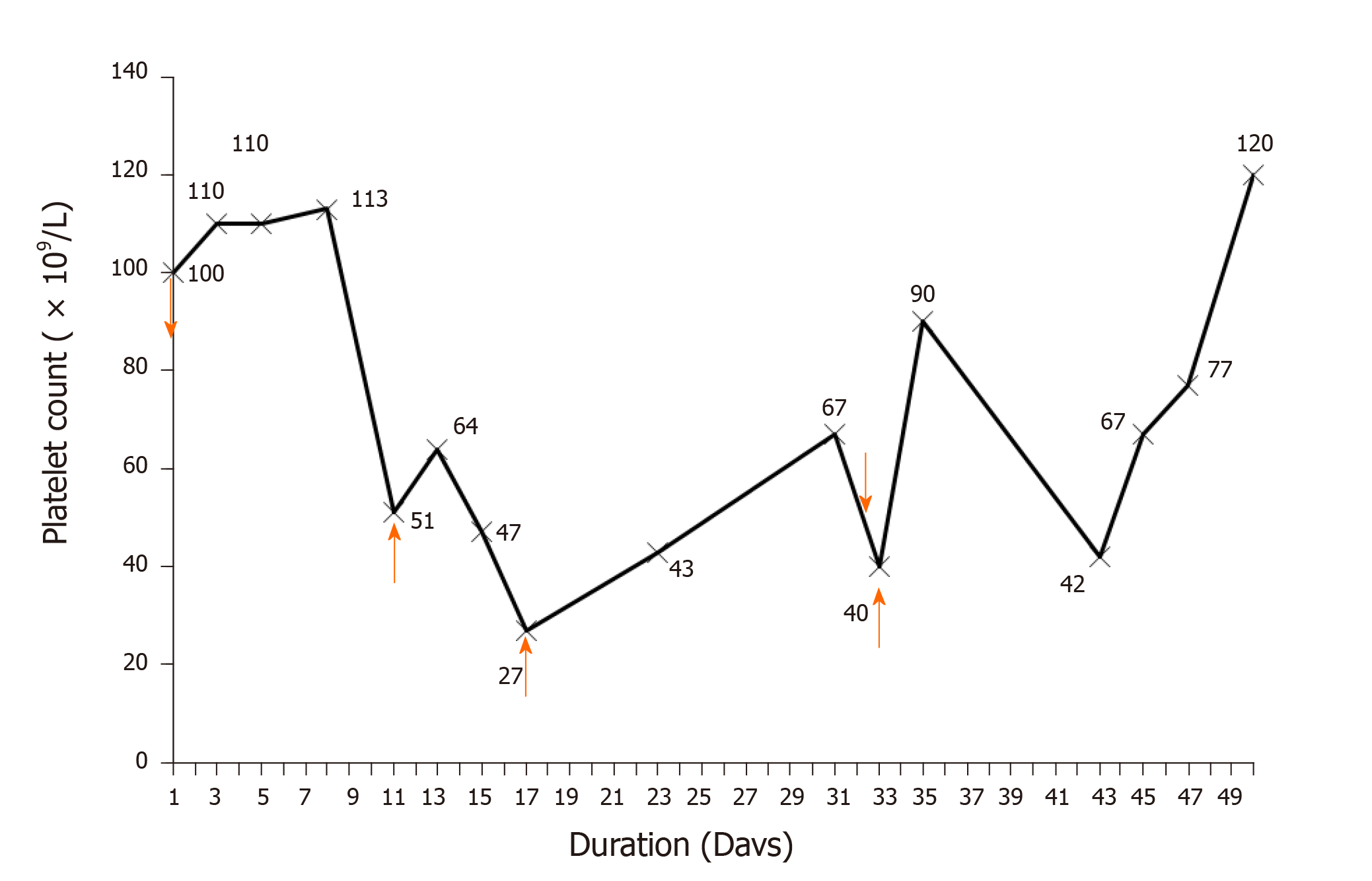
A 26-year-old male diagnosed with infective endocarditis was admitted. The patient was treated with multiple drugs, including vancomycin, which was initially intravenously given at 1000 mg every 12 h and subsequently at 500 mg every 8 h on day 3. On day 11, the platelet count decreased to 51 × 109/L, vancomycin was switched to 500 mg every 12 h, and platelet transfusion was given. On day 17, the platelet count dropped to 27 × 109/L, and platelet transfusion was administered again. On day 23, vancomycin was adjusted to 500 mg every 8 h as the trough concentration dropped to the minimum effective concentration. On day 33, the platelet count declined to approximately 40 × 109/L. After platelet transfusion, the platelet count rebounded to 90 × 109/L on day 35 but dropped again to 42 × 109/L on day 43. Based on the time-to-platelet count curve and Naranjo’s Adverse Drug Reaction Probability Scale score, VIT was suspected. After vancomycin discontinuation and platelet transfusion, the platelet count gradually normalized.
The diagnosis of VIT can be achieved through the time-to-platelet count curve and Naranjo’s Adverse Drug Reaction Probability Scale score. The platelet count cannot be normalized simply by platelet transfusion alone, and vancomycin discontinuation is essential.
Core Tip: The present report describes a case of vancomycin-induced thrombocytopenia (VIT) in endocarditis and reviews all VIT cases reported in the literature, in terms of indications, diagnosis, management, and potential molecular mechanisms. The present case highlights the importance of accurate diagnosis of VIT in endocarditis, which can be achieved through the time-to-platelet count curve and the Naranjo’s Adverse Drug Reaction Probability Scale score. The platelet count cannot be normalized simply by platelet transfusion alone, and the discontinuation of vancomycin is essential. Due to the anamnestic response to vancomycin re-exposure and the formation of vancomycin-dependent antiplatelet antibodies, the re-administration of vancomycin should be avoided.
- Citation: Guleng SR, Wu RH, Guo XB. Vancomycin-induced thrombocytopenia in endocarditis: A case report and review of literature . World J Clin Cases 2021; 9(7): 1696-1704
- URL: https://www.wjgnet.com/2307-8960/full/v9/i7/1696.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i7.1696
Thrombocytopenia, which is generally defined as a platelet count of less than 100 × 109/L of blood or a decrease in the platelet count of 20%-50% from baseline[1,2], has been considered as an adverse reaction correlated to drugs used in the treatment of various diseases[3]. Drug-induced thrombocytopenia is mostly caused by immune-mediated platelet degradation and is always drug dependent, indicating that the platelet count can return to baseline levels after the discontinuation of medications[4]. Although it has been reported that approximately 10 million persons per year are suspected to suffer from drug-induced thrombocytopenia[5], its incidence has not been well-defined[4].
Antibiotic-induced thrombocytopenia has been documented[3]. The commonly reported antibiotics that induce thrombocytopenia are β-lactam antibiotics agents (such as penicillin, nafcillin, ticarcillin, cefazolin, cefuroxime, ceftriaxone, and piperacillin) and linezolid[6]. Vancomycin is a first-generation glycopeptide antibiotic that is often used for the prophylaxis and treatment of suspected or identified methicillin-resistant infections, such as Staphylococcus aureus infection[7], and has been considered as an uncommon cause of thrombocytopenia[6]. Several clinical observational studies have suggested that the incidence of vancomycin-induced thrombocytopenia (VIT) might be higher than that induced by linezolid[1]. However, the incidence of VIT may have been overestimated, because the definition for thrombocytopenia used among these studies vary. A platelet count of less than 150 × 109/L was used in two studies[8,9], and a decrease in platelet count of at least 50% from baseline was used in another study[1]. In addition, the clinical manifestations, diagnosis, and management of VIT have not been well-established, and the underlying molecular mechanisms by which vancomycin induces thrombocytopenia needs to be further elucidated[10]. Over the past two decades, cases with VIT have been continuously reported[11]. However, these cases have rarely been extensively reviewed.
In June 2019, a young male patient with endocarditis was admitted to our hospital, and he developed VIT after vancomycin therapy during the treatment for endocarditis. The present report describes this case and extensively reviews all VIT cases reported in the literature, in terms of indications, diagnosis, management, and potential molecular mechanisms.
A 26-year-old Chinese male presented with dyspnea, fatigue, arrhythmias, fever, and cough.
The patient was admitted to the hospital on June 6, 2019 and diagnosed with infective endocarditis, heart failure, and ventricular septal defect, with a series of manifestations of inflammatory response syndrome.
He had a medical history of uncontrolled hypotension and rheumatic heart disease.
He denied any family history.
He had a body temperature of 39.1 °C and a heart rate of 100 beats per min.
Initial laboratory testing showed no abnormality.
The electrocardiosignal data revealed a high echo of the tricuspid valve, suggesting a neoplasm.
Infective endocarditis, heart failure, and ventricular septal defect.
During hospitalization, the patient was treated with multiple courses of drug therapy, which included vancomycin, omeprazole (40 mg q.d. for 10 d), ceftazidime (2 g t.i.d. for 9 d) and metoprolol (50 mg q.d. for 11 d). Based on the bacterial culture and drug susceptibility test, methicillin-resistant Staphylococcus aureus was identified with the minimum inhibitory concentration of less than 2 mg/L. Therefore, according to The Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-Resistant Staphylococcus aureus Infections in Adults and Children[12], vancomycin was prescribed for the patient with infective endocarditis. Initially, the patient was intravenously treated with 1000 mg of vancomycin every 12 h and 960 mg of benzylpenicillin every 8 h on day 2 for suspected gram-positive infections. The blood cultures were negative for two consecutive tests. Then, the intravenous administration of vancomycin was changed to 500 mg for every 8 h on day 3, for a trough concentration of 25.87 µg/mL beyond the upper limit of 20 µg/mL, and the platelet count was maintained within 100-110 × 109/L for 7 d. On day 11, the administration of vancomycin was switched to 500 mg every 12 h, because the platelet count decreased to 51 × 109/L and the peak concentration of vancomycin reached 57.2 µg/mL. Platelet transfusion (1 U) was given on day 11, and the platelet count slightly increased to 64 × 109/L on day 14, but dramatically dropped afterwards to 27 × 109/L on day 17. Then, platelet transfusion (1 U) was given again on day 17, and the platelet count steadily increased up to 67 × 109/L on day 31. The dose of vancomycin was adjusted to 500 mg every 8 h on day 23 when the trough concentration of vancomycin dropped to 8.86 µg/mL in order to ensure the minimal effective treatment concentration (10 µg/mL). However, on day 33, the hemoglobin level sharply dropped from 101 g/L to 54 g/L, the platelet count continuously declined to approximately 40 × 109/L, and platelet transfusion (1 U) was given. The platelet count rebounded to 90 × 109/L on day 35 but dropped to 42 × 109/L again on day 43. Based on these above observations, and along with the time-to-platelet count curve that illustrated the decline in platelet level after the administration of vancomycin (Figure 1), VIT was suspected, vancomycin was discontinued and replaced with daptomycin, and platelet transfusion (1 U) was given on the same day. The platelet count level increased and reached up to 120 × 109/L on day 50 and remained stable thereafter.
The concurrent antibiotic, benzylpenicillin, was continuously used during the period with little effect on the platelet count. The respiratory rate, white blood cell count, and hemodynamic variables remained stable, and the microbiological cultures from the sputum and urine revealed negative results, implying that no infection was correlated with thrombocytopenia for the present patient. The other causes of thrombocytopenia, including thrombotic thrombocytopenic purpura, hemolytic anemia, disseminated intravascular coagulation, and platelet-clumping pseudothrombocytopenia, were also excluded, because the results of the blood smear, bilirubin, and hemoglobin were normal. There have been no reports on the occurrence of drug-induced thrombocytopenia caused by other medications during treatment, including omeprazole, ceftazidime, and metoprolol. Therefore, a diagnosis of VIT was initially established using the time-to-platelet count curve. In addition, the Naranjo’s Adverse Drug Reaction Probability Scale score (Naranjo score) for vancomycin was 8, indicating the probable correlation between thrombocytopenia and vancomycin.
The patient was discharged from the hospital.
A literature search for all case reports on VIT published in English from April 1985 to June 2020 was performed on PMC/PubMed, Scopus, and Web of Science. Keywords, which included vancomycin and thrombocytopenia, were used to search the titles and abstracts. References of relevant case reports, according to the brief research, were also carefully checked. The demographic data (e.g., age and gender), platelet count, diagnosis, and treatment were recorded and analyzed.
A total of 36 articles that reported 37 cases with VIT were identified in the literature. Thus, the data of 38 cases, which included the case described in the present report, were reviewed and are summarized in Table 1. Among these cases, 14 cases were females and 24 cases were males. The mean age of these cases was 51.19 ± 26.98 years-old (range: 3 mo to 81-years-old). Furthermore, four children were under 2-years-old, and 36 cases were adults 26-81 years-old. The most common indications for the treatment with vancomycin were endocarditis (n = 6), sepsis (n = 6), pneumonia (n = 5), and cellulitis (n = 2). These were followed by other various indications, such as prosthetic infection, hemodialysis, bone graft, acute pancreatitis, etc. The diagnosis of VIT mainly relied on the platelet count, with or without the Naranjo score[13]. The test results for the vancomycin-dependent antibodies were available for 19 patients, and all except three were positive. Among the 27 patients with available bleeding information, 21 patients exhibited bleeding signs. The treatments for these cases comprised of platelet transfusion, prednisone, dexamethasone, immunoglobulin, and daptomycin, and the discontinuation of vancomycin. For the six cases with endocarditis, VIT was diagnosed as probable by the Naranjo score. The information on bleeding signs was available for four cases, and three of these cases presented with bleeding signs. The presence of vancomycin-dependent antibodies was examined only in two patients, and positive results were obtained in one of them. After vancomycin discontinuation, VIT was generally resolved for all patients within 4-15 d. Despite clinical significance revealed by the present literature review, it must be clearly stated that this is only a narrative review, which possesses all the limitations of the selection of papers. Thus, a methodologically acceptable systematic review should be carried out when more data with high quality are available in order to draw a firm conclusion.
| Author (publication year) | Age (yr) | Gender | Indication | Platelet count (109/L)1 | Naranjo Score | Treatment | Time for resolution (d)2 | Vancomycin-dependentantibodies | Bleeding sign |
| Present case | 26 | M | Endocarditis | 27 | Probable | Platelet transfusion and daptomycin | 12 | NA | + |
| MacDougall et al[14] (2020) | 81 | M | Prosthetic infection | 2 | NA | Prednisone, immunoglobulin, and daptomycin | 10 | + | - |
| Ajit et al[27] (2019) | 61 | M | Hemodialysis | < 10 | NA | Prednisone, immunoglobulin, eltrombopag and plasma exchange | 15 | + | + |
| Getz et al[18] (2019) | 71 | M | Transverse myelitis | 8.6 | Definite | Dexamethasone and immunoglobulin | 8 | + | + |
| Chen et al[16] (2018) | 0 | F | Purulent meningitis | 8 | Probable | Methylprednisolone | 4 | + | + |
| Danieletto et al[17] (2017) | 57 | F | Bone graft | 9 | Probable | Tigecycline | 7 | - | - |
| Kalra et al[28] (2016) | 0.7 | M | Sepsis | 30 | NA | Platelet transfusion | 3 | NA | - |
| Schueler et al[15] (2016) | 55 | M | Purpura | 1 | NA | Prednisone and immunoglobulin | 3 | NA | + |
| Yamanouchi et al[29] (2016) | 72 | F | Sepsis | 2 | NA | Steroid therapy and carbapenem | 7 | + | + |
| Lobo et al[13] (2015) | 67 | M | Pneumonia | 2 | Probable | Platelet transfusion, methylprednisolone and immunoglobulin | 3 | + | + |
| Ahmed et al[30] (2015) | 63 | M | Diabetic foot | 2 | Probable | Daptomycin | 10 | + | - |
| Candemir et al[10] (2013) | 54 | F | Hematoma | 42 | Probable | Platelet transfusion and daptomycin | 4 | NA | NA |
| Wetzel et al[31] (2013) | 64 | F | Sepsis | 7 | Probable | Prednisone and immunoglobulin | 8 | NA | + |
| Ruggero et al[32] (2012) | 41 | M | Pneumonia | 15 | Definite | Daptomycin | 5 | NA | + |
| Arnold et al[23] (2013) | 66 | F | Endocarditis | 4 | Probable | Platelet transfusion and immunoglobulin | 5 | + | + |
| Rowland et al[33] (2013) | 51 | M | Acute pancreatitis | 9 | Probable | Only vancomycin discontinuation | 5 | NA | + |
| Anand et al[34] (2011) | 54 | M | Cellulitis | 1 | Possible | Corticosteroids and daptomycin | 2 | + | + |
| Ganly et al[22] (2011) | 67 | M | Sepsis | 2 | NA | Platelet transfusion and immunoglobulin | 8 | NA | + |
| Shah et al[11] (2009) | 60 | M | Shoulder infection | 9 | Possible | Platelet transfusion | 3 | + | + |
| Lee et al[35] (2009) | 76 | M | Diabetic foot | < 15 | Possible | Platelet transfusion and teicoplanin. | 5 | + | - |
| Apiwattanakul et al[36] (2008) | 16 | F | Endocarditis | 123 | Possible | Prednisone and cefotaxime | 5 | NA | - |
| Kenney et al[20] (2008) | 61 | M | Gangrene and bacteremia | 3 | Probable | Platelet transfusion and immunoglobulin | 4 | + | + |
| Pauldine et al[37] (2008) | 60 | M | Pneumonia | 10 | Possible | Platelet transfusion | 12 | + | + |
| Dilli et al[38] (2008) | 0.6 | M | Neonatal respiratory distress | 41 | NA | Only vancomycin discontinuation | 3 | - | NA |
| O’Donnell et al[21] (2007) | 56 | F | Prosthetic infection | 2 | Possible | Immunoglobulin | 7 | + | + |
| Bay et al[39] (2006) | 2 | M | Pneumonia | 11 | NA | Only vancomycin discontinuation | 4 | NA | NA |
| Winteroll et al[24] (2004) | 72 | M | Sepsis | 3 | Probable | Platelet transfusion | 10 | + | + |
| Peel et al[40] (2003) | 45 | M | Peritonitis | 5 | Probable | Platelet transfusion and prednisone | 4 | NA | + |
| Marraffa et al[41] (2003) | 50 | M | Endocarditis | 10 | Probable | Clindamycin and methylprednisolone | 5 | NA | + |
| Rocha et al[42] (2002) | 38 | F | Prosthetic infection | 68 | Possible | Only vancomycin discontinuation | 4 | NA | NA |
| Shahar et al[43] (2000) | 43 | F | Surgery-site infection | 118 | Probable | Only vancomycin discontinuation | 3 | NA | NA |
| Govindarajan et al[44] (1999) | 72 | M | Epidural abscess | 13 | Probable | Trimethoprim/sulfmethoxazole | 10 | NA | NA |
| Kuruppu et al[45] (1999) | 72 | F | Endocarditis | 14 | Possible | Only vancomycin discontinuation | 5 | - | NA |
| Mizon et al[46] (1997) | 71 | F | Sepsis | < 10 | Probable | Antibiotics withdrawn | 4 | + | + |
| Zenon et al[47] (1991) | 54 | M | Cellulitis | 17 | Probable | Antibiotics withdrawn | 7 | NA | NA |
| Christie et al[48] (1990) | 73 | F | Pneumonia | 14 | Probable | Platelet transfusion | 2 | + | NA |
| Christie et al[48] (1990) | 31 | M | Fever | < 10 | Possible | Platelet transfusion | 11 (not continued) | + | NA |
| Carmichael et al[49] (1986) | 42 | F | Endocarditis | 82 | Possible | NA | 9 | NA | NA |
The present patient with infective endocarditis was diagnosed with VIT, which rapidly progressed. The early diagnosis of VIT was performed using the time-to-platelet count curve and Naranjo score. During treatment, the repeated platelet transfusions failed to increase the platelet levels. Subsequently, simply switching vancomycin to daptomycin returned the platelet count close to the baseline level.
The early diagnosis of VIT is quite difficult due to many suspected causes, such as severe infection, disseminated intravascular coagulation, heparin, and other medications. The correct diagnosis can be masked by the simultaneous administration of medications, which can cause drug-induced immune thrombocytopenia[14] or a complicating disease, such as primary idiopathic thrombocytopenic purpura or chronic hepatitis C[15]. Therefore, the early recognition of VIT was of key importance for the following treatment. According to previous studies[6], the investigators used the platelet count at different time points as the initial diagnosis of VIT, which can reveal a definite time-independent relationship with drug administration. All other causes of thrombocytopenia were excluded. Furthermore, the Naranjo score, which has been applied to help with the diagnosis of VIT[16-18], also indicated a probable correlation between thrombocytopenia and vancomycin. Noticeably, endocarditis represents the most common indication that is associated with VIT. The probable correlation between thrombocytopenia and vancomycin can be made through the Naranjo score for all cases, indicating that the Naranjo score is a useful tool to initially diagnose VIT in patients with endocarditis. A bleeding sign is also a useful clinical manifestation to which attention should be given in order to make a definite diagnosis, since most cases would present bleeding signs when VIT occurs. This is particularly true in patents with endocarditis, based on the analyzed data. However, the value of the detection of vancomycin-dependent antibodies in the diagnosis of VIT in patients with endocarditis remains to be elucidated due to the very limited data available.
The critical management after suspicions of VIT is to determine whether to continue or discontinue the vancomycin therapy. The seriousness of the bleeding and the dropping trend of the platelet count should be carefully considered and closely monitored when continuing the vancomycin therapy. However, once VIT is diagnosed, the vancomycin must be stopped, and the platelet transfusion should be taken into consideration as a supplemental treatment for some patients with various success rates, although there are still some transfusion-resistant patients[10,13,19,20]. If vancomycin is not stopped, the survival time of the transfused platelets would be obviously reduced, and the platelet transfusion will not always expectedly increase the platelet count of affected patients[3]. In any of these circumstances, platelet transfusion should be considered for severe thrombocytopenia with a platelet count of below 20 × 109/L and bleeding[19]. In addition to platelet transfusion, therapies with intravenous corticosteroids, intravenous immunoglobulins, rituximab, and plasma exchange have been shown to be beneficial in some cases after vancomycin discontinuation[3,21-23]. However, these medical approaches may not always be effective. In the present case, VIT was effectively resolved by platelet transfusion with the switching of vancomycin to daptomycin.
The mechanisms of VIT remains unclear. The formation of different drug-dependent platelet antibodies, including hapten-dependent, quinine-type, fiban-type and drug-specific antibodies, and other platelet destruction antibodies, are the widely documented mechanisms for VIT[11,21,24]. In contrast, naturally occurring antibodies may not contribute to VIT[25]. It has been postulated that vancomycin can bind to platelet glycoproteins and induce the generation of antibodies, which can attach to the drug-platelet complex, resulting in cell lysis[26]. In the present case, the platelet count gradually dropped after the administration of vancomycin (1000 mg per day) for 7 d. Although the vancomycin was changed to a low-dose (500 mg per day), the platelet count continued to drop. It was postulated that the persistent thrombocytopenia might be due to the anamnestic response for vancomycin re-exposure, and vancomycin-dependent antiplatelet antibodies were formed once vancomycin was re-used.
The present case highlights the importance of the accurate diagnosis of VIT in patients with endocarditis. The time-to-platelet count curve and Naranjo score are useful tools for the diagnosis of VIT. The platelet count cannot be normalized simply by platelet transfusion alone. Instead, the discontinuation of vancomycin or switching vancomycin to other antibiotics, such as daptomycin, is essential for effectively treating VIT. Due to the anamnestic response to vancomycin re-exposure and the formation of vancomycin-dependent antiplatelet antibodies, the re-administration of vancomycin should be avoided.
Manuscript source: Unsolicited manuscript
Specialty type: Pharmacology and pharmacy
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Momčilović S S-Editor: Zhang H L-Editor: Filipodia P-Editor: Xing YX
| 1. | Patel N, VanDeWall H, Tristani L, Rivera A, Woo B, Dihmess A, Li HK, Smith R, Lodise TP. A comparative evaluation of adverse platelet outcomes among Veterans' Affairs patients receiving linezolid or vancomycin. J Antimicrob Chemother. 2012;67:727-735. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 2. | Rao N, Ziran BH, Wagener MM, Santa ER, Yu VL. Similar hematologic effects of long-term linezolid and vancomycin therapy in a prospective observational study of patients with orthopedic infections. Clin Infect Dis. 2004;38:1058-1064. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 73] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 3. | van den Bemt PM, Meyboom RH, Egberts AC. Drug-induced immune thrombocytopenia. Drug Saf. 2004;27:1243-1252. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 77] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 4. | Aster RH, Bougie DW. Drug-induced immune thrombocytopenia. N Engl J Med. 2007;357:580-587. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 393] [Cited by in F6Publishing: 343] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 5. | Aster RH, Curtis BR, McFarland JG, Bougie DW. Drug-induced immune thrombocytopenia: pathogenesis, diagnosis, and management. J Thromb Haemost. 2009;7:911-918. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 238] [Cited by in F6Publishing: 215] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 6. | Loo AS, Gerzenshtein L, Ison MG. Antimicrobial drug-induced thrombocytopenia: a review of the literature. Semin Thromb Hemost. 2012;38:818-829. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 7. | Levine DP. Vancomycin: understanding its past and preserving its future. South Med J. 2008;101:284-291. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Rao N, Ziran BH, Hall RA, Santa ER. Successful treatment of chronic bone and joint infections with oral linezolid. Clin Orthop Relat Res. 2004: 67-71. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 65] [Cited by in F6Publishing: 65] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 9. | Nasraway SA, Shorr AF, Kuter DJ, O'Grady N, Le VH, Cammarata SK. Linezolid does not increase the risk of thrombocytopenia in patients with nosocomial pneumonia: comparative analysis of linezolid and vancomycin use. Clin Infect Dis. 2003;37:1609-1616. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 60] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 10. | Candemir B, Aribuca A, Koca C, Ozcan OU, Gerede M, Kaya CT. An unusual case of vancomycin-related systemic reaction accompanied with severe thrombocytopenia mimicking pacemaker-related infective endocarditis: a case report and review of literature. J Interv Card Electrophysiol. 2013;38:143-145. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Shah RA, Musthaq A, Khardori N. Vancomycin-induced thrombocytopenia in a 60-year-old man: a case report. J Med Case Rep. 2009;3:7290. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Liu C, Bayer A, Cosgrove SE, Daum RS, Fridkin SK, Gorwitz RJ, Kaplan SL, Karchmer AW, Levine DP, Murray BE, J Rybak M, Talan DA, Chambers HF; Infectious Diseases Society of America. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52:e18-e55. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1692] [Cited by in F6Publishing: 1848] [Article Influence: 142.2] [Reference Citation Analysis (0)] |
| 13. | Lobo N, Ejiofor K, Thurairaja R, Khan MS. Life-threatening haematuria caused by vancomycin-induced thrombocytopenia. BMJ Case Rep. 2015;2015. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | MacDougall KN, Parylo S, Sokoloff A. A Case of Vancomycin-Induced Immune Thrombocytopenia. Cureus. 2020;12:e7940. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 15. | Schueler SA, Shet NS, Stienstra N, Chen DC. A Masked Case of Vancomycin-induced Immune Thrombocytopenia. Am J Med Sci. 2016;351:636. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 16. | Chen QY, Wan J, Yang JH, Lin M, Chen Y. Vancomycin-induced severe thrombocytopenia in a young infant. Rev Soc Bras Med Trop. 2018;51:873-875. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 17. | Danieletto CF, Ferreira GZ, Farah GJ, Cuman RK. Vancomycin-induced thrombocytopenia: a rare adverse effect in a patient -submitted to bone graft in the jaw. Spec Care Dentist. 2017;37:38-42. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Getz TM, Packer CD. Rapid-Onset Vancomycin-Induced Thrombocytopenia With Reexposure. Ann Pharmacother. 2019;53:1259-1261. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Mohammadi M, Jahangard-Rafsanjani Z, Sarayani A, Hadjibabaei M, Taghizadeh-Ghehi M. Vancomycin-Induced Thrombocytopenia: A Narrative Review. Drug Saf. 2017;40:49-59. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Kenney B, Tormey CA. Acute vancomycin-dependent immune thrombocytopenia as an anamnestic response. Platelets. 2008;19:379-383. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | O'Donnell E, Shepherd C, Neff A. Immune thrombocytopenia from vancomycin in orthopedic cement. Am J Hematol. 2007;82:1122. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 22. | Ganly P, Downing J, Stiven P, Frizelle F, Badami K. Clinical and serological diagnoses of a patient with vancomycin-induced thrombocytopenia. Transfus Med. 2011;21:137-139. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Arnold DM, Nazi I, Warkentin TE, Smith JW, Toltl LJ, George JN, Kelton JG. Approach to the diagnosis and management of drug-induced immune thrombocytopenia. Transfus Med Rev. 2013;27:137-145. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 100] [Cited by in F6Publishing: 104] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 24. | Winteroll S, Kerowgan M, Vahl CF, Leo A. Vancomycin-mediated drug-induced immune thrombocytopenia. Transfus Med Hemother. 2004;32:20-23. [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 25. | Von Drygalski A, Curtis BR, Bougie DW, McFarland JG, Ahl S, Limbu I, Baker KR, Aster RH. Vancomycin-induced immune thrombocytopenia. N Engl J Med. 2007;356:904-910. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 149] [Cited by in F6Publishing: 138] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 26. | Rondina MT, Walker A, Pendleton RC. Drug-induced thrombocytopenia for the hospitalist physician with a focus on heparin-induced thrombocytopenia. Hosp Pract (1995). 2010;38:19-28. [PubMed] [Cited in This Article: ] |
| 27. | Ajit NE, Devarashetty SP, Master S. Vancomycin Induced Thrombocytopenia - Protracted Course in a Hemodialysis Patient. Case Rep Oncol. 2019;12:749-754. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 28. | Kalra K, Mittal HG, Maria A. Vancomycin-induced thrombocytopenia in a newborn. Drug Metab Pers Ther. 2016;31:235-237. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 29. | Yamanouchi J, Hato T, Shiraishi S, Takeuchi K, Yakushijin Y, Yasukawa M. Vancomycin-induced Immune Thrombocytopenia Proven by the Detection of Vancomycin-dependent Anti-platelet Antibody with Flow Cytometry. Intern Med. 2016;55:3035-3038. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Ahmed Y, Sartin C, Umer I. Vancomycin-induced severe asymptomatic immune thrombocytopenia: a rare cause. Southwest Respir Crit Care Chronicles. 2015;3:42-45. [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 31. | Wetzel DR, Njathi CW, Teleszm BJ, Stewartm TM, Smischney NJ. Thrombocytopenia of unusual etiology in the intensive care unit. J Med Cases. 2013;4:792-795. [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 32. | Ruggero MA, Abdelghany O, Topal JE. Vancomycin-induced thrombocytopenia without isolation of a drug-dependent antibody. Pharmacotherapy. 2012;32:e321-5. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 33. | Rowland SP, Rankin I, Sheth H. Vancomycin-induced thrombocytopaenia in a patient with severe pancreatitis. BMJ Case Rep. 2013;2013. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 34. | Anand A, Chauhan HK. Piperacillin and vancomycin induced severe thrombocytopenia in a hospitalized patient. Platelets. 2011;22:294-301. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 35. | Lee JH, Kim DS, Lee HS, Choi SI, Cho YG. A case of vancomycin induced thrombocytopenia. Korean J Hematol. 2009;44:294-297. [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 36. | Apiwattanakul N, Wanitkun S, Chongtrakool P, Sirinavin S. A patient with penicillin-resistant viridans group streptococcal endocarditis and unusual reactions to vancomycin. Southeast Asian J Trop Med Public Health. 2008;39:1088-1091. [PubMed] [Cited in This Article: ] |
| 37. | Pauldine R, Pustavoitau A. Case report: vancomycin-induced thrombocytopenia in a burn patient. Eplasty. 2008;8:e39. [PubMed] [Cited in This Article: ] |
| 38. | Dilli D, Oğuz SS, Dilmen U. A newborn with vancomycin-induced thrombocytopenia. Pharmacology. 2008;82:285-286. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 39. | Bay A, Oner AF, Dogan M, Caksen H. A child with vancomycin-induced thrombocytopenia. J Emerg Med. 2006;30:99-100. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 40. | Peel RK, Sykes A, Ashmore S, Turney JH, Woodrow G. A case of immune thrombocytopenic purpura from intraperitoneal vancomycin use. Perit Dial Int. 2003;23:506-508. [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 41. | Marraffa J, Guharoy R, Duggan D, Rose F, Nazeer S. Vancomycin-induced thrombocytopenia: a case proven with rechallenge. Pharmacotherapy. 2003;23:1195-1198. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 22] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 42. | Rocha JL, Kondo W, Baptista MI, Da Cunha CA, Martins LT. Uncommon vancomycin-induced side effects. Braz J Infect Dis. 2002;6:196-200. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 53] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 43. | Shahar A, Berner Y, Levi S. Fever, rash, and pancytopenia following vancomycin rechallenge in the presence of ceftazidime. Ann Pharmacother. 2000;34:263-264. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 44. | Govindarajan R, Baxter D, Wilson C, Zent C. Vancomycin-induced thrombocytopenia. Am J Hematol. 1999;62:122-123. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 45. | Kuruppu JC, Le TP, Tuazon CU. Vancomycin-associated thrombocytopenia: case report and review of the literature. Am J Hematol. 1999;60:249-250. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 46. | Mizon P, Kiefel V, Mannessier L, Mueller-Eckhardt C, Goudemand J. Thrombocytopenia induced by vancomycin-dependent platelet antibody. Vox Sang. 1997;73:49-51. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 47. | Zenon GJ, Cadle RM, Hamill RJ. Vancomycin-induced thrombocytopenia. Arch Intern Med. 1991;151:995-996. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 28] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 48. | Christie DJ, van Buren N, Lennon SS, Putnam JL. Vancomycin-dependent antibodies associated with thrombocytopenia and refractoriness to platelet transfusion in patients with leukemia. Blood. 1990;75:518-523. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 48] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 49. | Carmichael A, Al-Zahawi M. Drug points: pancytopenia associated with vancomycin. Br Med J. 1986;293:1103. [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |