Copyright
©The Author(s) 2021.
World J Clin Cases. Jul 26, 2021; 9(21): 6091-6101
Published online Jul 26, 2021. doi: 10.12998/wjcc.v9.i21.6091
Published online Jul 26, 2021. doi: 10.12998/wjcc.v9.i21.6091
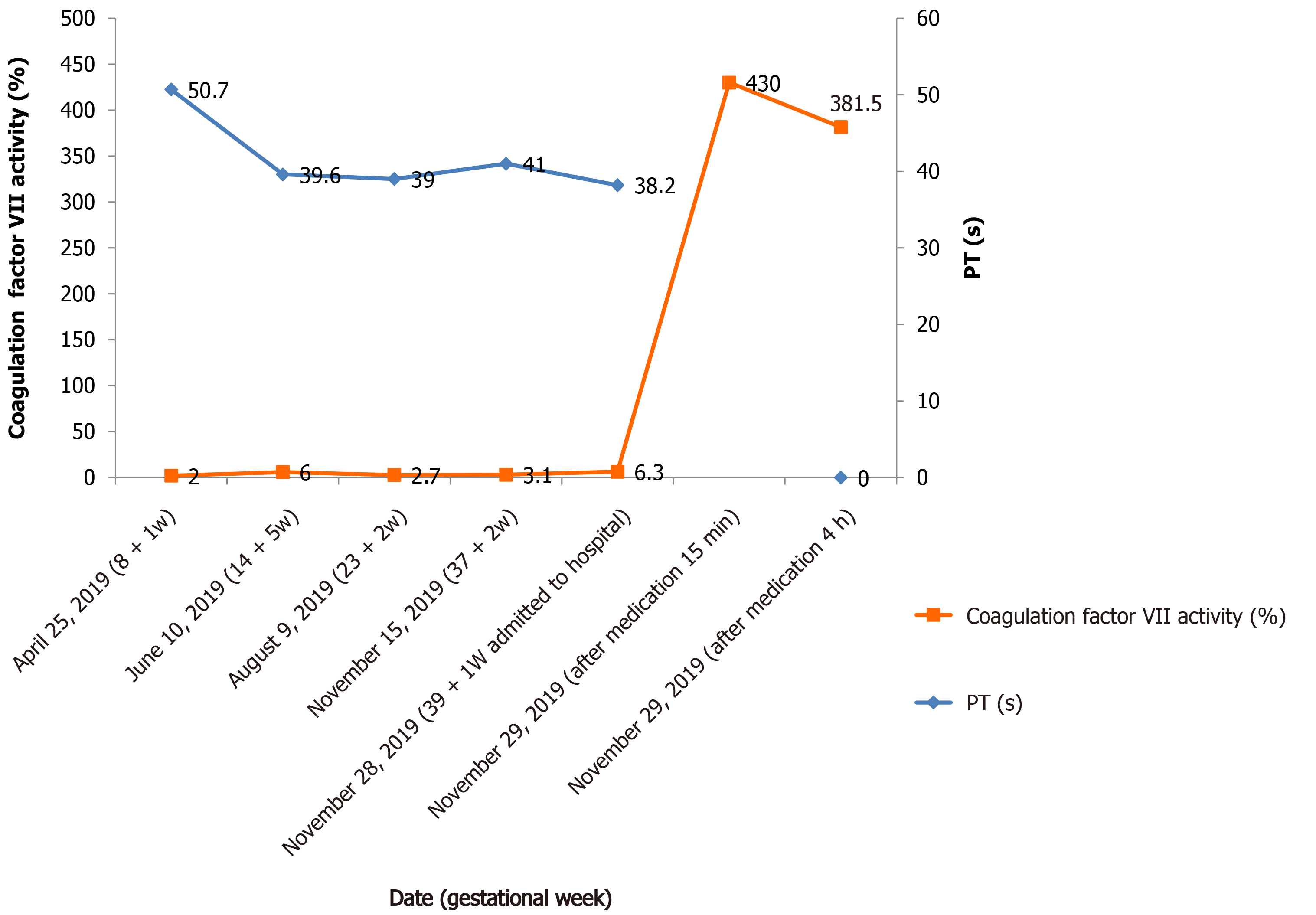
Figure 1 Fluctuation of factor VII activity and corresponding prothrombin time value in the pregnant woman.
Medication: Intravenous injection of recombinant factor VIIa (NovoSeven®, Novo Nordisk A/S, Bagsvaerd, Denmark) 5 mg, (90 μg/kg). PT: Prothrombin time.
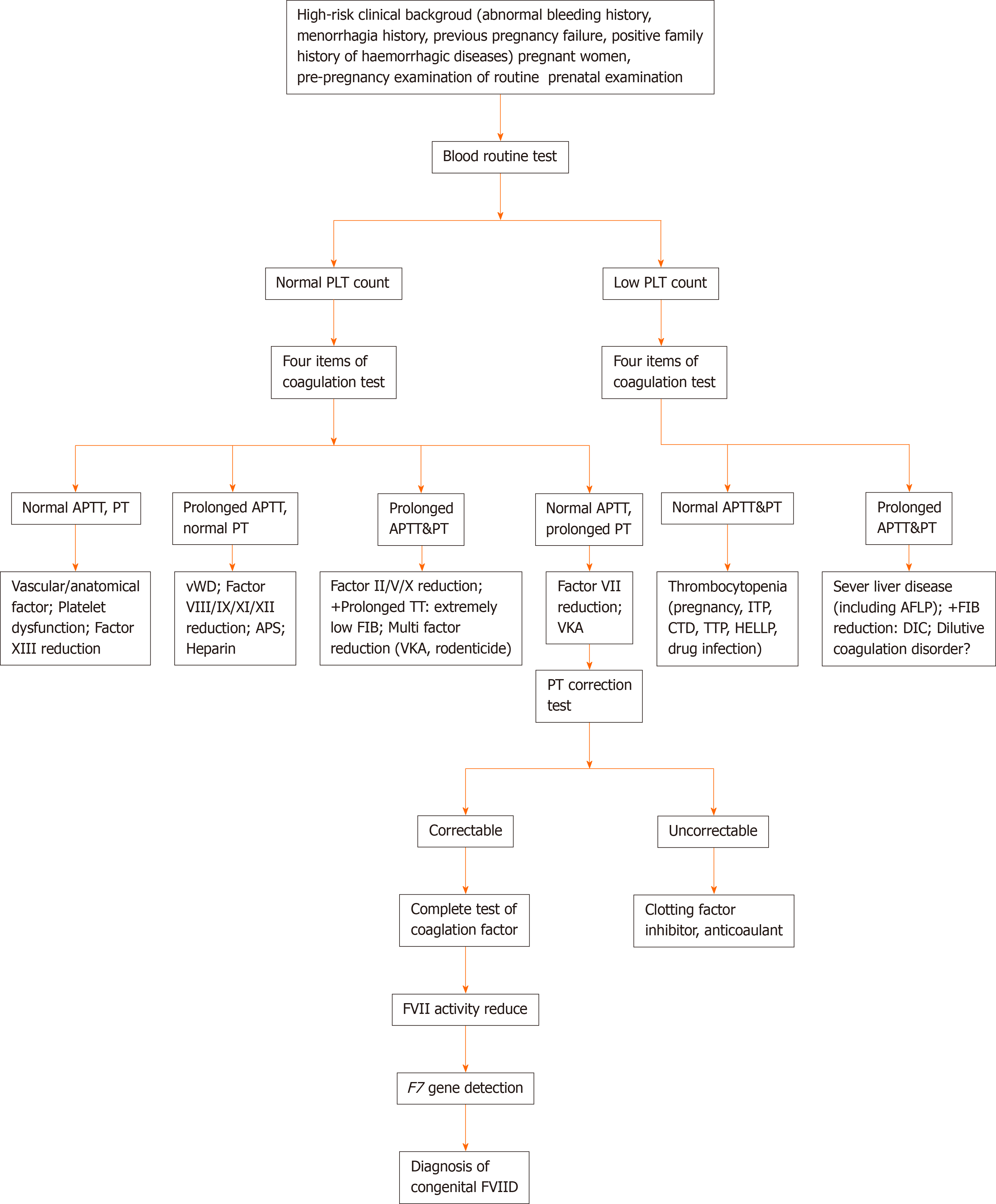
Figure 2 Diagnosis flow chart of pregnancy with congenital factor VII deficiency.
vWD: Von Willebrand disease; APS: Antiphospholipid antibody syndrome; PLT: Platelet; APTT: Activated partial thromboplastin time; PT: Prothrombin time; TT: Plasma thrombin time; FIB: Fibrinogen; VKA: Vitamin K antagonist; ITP: Idiopathic thrombocytopenic purpura; CTD: Diffuse connective tissue disease; TTP: Thrombocytopenic purpura; HELLP: HELLP syndrome; AFLP: Acute fatty liver in pregnancy; DIC: Diffuse intravascular coagulation; FVII: Factor VII; FVIID: Factor VII deficiency.
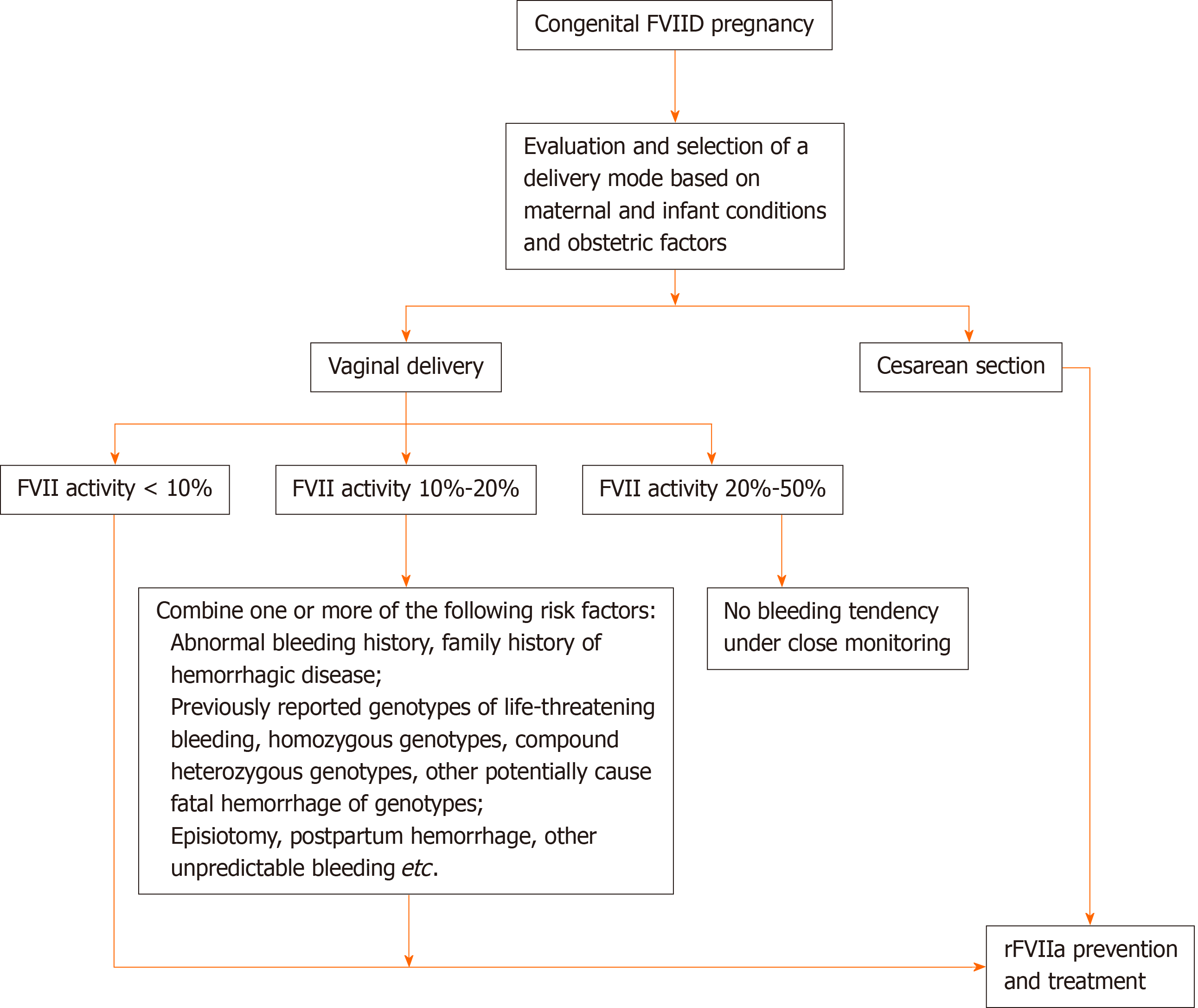
Figure 3 Selection of delivery mode and corresponding recombinant factor VIIa medication scheme of congenital factor VII deficiency pregnant women during delivery.
FVIID: Factor VII deficiency; FVII: Factor VII; rFVIIa: Recombinant factor VIIa.
- Citation: Yang Y, Zeng YC, Rumende P, Wang CG, Chen Y. Diagnosis and treatment discussion of congenital factor VII deficiency in pregnancy: A case report. World J Clin Cases 2021; 9(21): 6091-6101
- URL: https://www.wjgnet.com/2307-8960/full/v9/i21/6091.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i21.6091