Published online Dec 22, 2012. doi: 10.5498/wjp.v2.i6.114
Revised: September 19, 2012
Accepted: November 17, 2012
Published online: December 22, 2012
Major depression is an independent risk factor for cardiovascular mortality and morbidity. The exact mechanisms linking depression and increased cardiovascular risk remain poorly understood. Several mechanisms have been proposed including increased platelet reactivity. This review focuses on the current literature that examines the platelet hypothesis of depression. To date studies show increased serotonin response, increased platelet serotonin receptor density, decreased serotonin transporter binding, and decreased platelet serotonin levels in individuals with depression. However other studies have shown no change in serotonin uptake. In addition to platelet serotonin specific pathways, other platelet pathways that have shown significant changes in depressed individuals include blunting of the platelet adenosine response, increased platelet thrombin response, increased glycoprotein Ib expression, increased P-selectin, β thromboglobulin, and platelet factor four, as well as decreased platelet brain derived neurotrophic factor. However there are other studies that show conflicting evidence of increased platelet activation as measured by integrin receptor α2bβ3. Other conflicting data include α adrenergic density and platelet response to augmented serotonin. The direction of future research in platelet functional changes in depression and coronary artery disease should continue to focus on serotonin specific pathways with emphasis on potential mechanisms of specific pathway changes.
- Citation: Williams MS. Platelets and depression in cardiovascular disease: A brief review of the current literature. World J Psychiatr 2012; 2(6): 114-123
- URL: https://www.wjgnet.com/2220-3206/full/v2/i6/114.htm
- DOI: https://dx.doi.org/10.5498/wjp.v2.i6.114
There are approximately 26.8 million people within the United States with heart disease[1]. Although the incidence of depression is 5% in the general population, after myocardial infarction the incidence of depression jumps to 20%[2]. Major depression is an independent risk factor for cardiovascular mortality and morbidity[3-6]. Depression increases the risk of cardiac mortality in patients with and without baseline cardiac disease[7]. The exact mechanisms linking depression and increased cardiovascular risk remain poorly understood. Several mechanisms have been proposed such as medication noncompliance, smoking, and physical inactivity[8-12]. More traditional risks factors for heart disease have also been proposed such as hypertension, diabetes, and insulin resistance. In addition several nontraditional risk factors for heart disease have also been proposed which include increased platelet reactivity[13], procoagulopathy, inflammation, obesity, and dysregulation of the autonomic nervous system leading to increased arrhythmias. The following review will focus on the platelet hypothesis of depression.
Given the established link between serotonin and depression and the fact that platelets contain 99% of the body’s serotonin along with the fact that increased platelet reactivity is firmly linked to cardiovascular complications, platelet dysfunction is a logical mechanism linking depression and cardiovascular disease[12-15].
Platelets are aneuclear cells derived from their precursor cells, megakaryocytes, and circulate in the blood able to detect and respond to vessel injury. They circulate for an average of seven to 10 d. Platelets contain mitochondria, lysosomes, and dense and α granules[16]. Dysfunctional platelets can lead to either bleeding or thrombotic complications.
Compared to non-depressed controls, depressed patients exhibit enhanced platelet reactivity and increased expression of activated glycoprotein IIb/IIIa and P selectin when patients are given an orthostatic challenge[17]. This has led researchers to conclude that “Heightened susceptibility to platelet activation may be a mechanism by which depression is a significant risk factor for ischemic heart and cerebrovascular disease and/or mortality after myocardial infarction”. Markovitz et al[18] have found that patients with major depression have increased collagen induced platelet secretion when compared to age and gender matched controls without depression. Plasma levels of platelet β-thromboglobulin and platelet factor four (PF4) are significantly elevated in patients with depression when compared with those of healthy control subjects[19,20]. Increased platelet aggregation to both collagen and thrombin has been found in patients with major depression[21]. The SADHART trial examined the effects of 24 wk of treatment with sertraline, a selective serotonin reuptake inhibitor (SSRI), in patients with acute coronary syndrome and depression and found that platelet activation is suppressed by this SSRI as evidence by decreased platelet release of β thromboglobulin and P-selectin[22,23]. Shimbo et al[24] have shown that increased platelet reactivity to serotonin, as measured by serotonin-augmented epinephrine platelet impedance aggregometry, is present in patients with depression without coronary artery disease (CAD). Interestingly, these same patients with depression had no difference in adenosine diphosphate (ADP) - induced aggregation compared to non-depressed controls. In another study no significant difference between depressed patients and healthy volunteers (HV) was found when ADP - induced aggregation was measured[25].
The above studies have contributed to the hypothesis that depression is associated with increased serotonin-induced platelet activation, rather than a more general increase in platelet activation; however, this has by no means been unequivocally established as Gómez-Gil found no significant difference in serotonin-amplified platelet aggregation to ADP between patients with untreated major depression and non-depressed controls. There was also no correlation of serotonin-amplified platelet aggregation with the severity of depression as assessed by the Hamilton scale[26].
Despite the above findings of increased platelet activation in patients with depression, the association between depression and platelets is not completely clear. A review by Parakh et al[27] revealed that although many studies have shown exaggerated platelet activation in patients with depression, several have shown no such relationship. They concluded that larger, carefully designed, adequately powered studies with standardized methods of assessing platelet function are needed.
The evidence that supports a key role of serotonin (5-HT) in depression include: (1) reduced cerebrospinal fluid concentrations of 5-hydroxyindoleacetic acid (5-HIAA) which is a major metabolite of 5-HT; (2) reduced concentrations of 5-HT and 5-HIAA in postmortem brain tissue of depressed and/or suicidal patients; (3) clinically effective antidepressants all affect 5-HT neurotransmission; and (4) decreased number of serotonin transporter binding sites in postmortem brains and platelets of depressed patients and suicide victims[28].
Platelets store 99% of the body’s serotonin in their dense granules[15]. Upon release of serotonin from the dense granules at sites of endothelial damage, platelet aggregation is promoted.
Platelet serotonin levels have been found to be decreased in patients with depression and further decreased after SSRI use when compared to normal controls[29-31]. In a sample of 791 outpatients with stable coronary heart disease and no antidepressant use, Wulsin et al[32] have found that current (past month) major depression was associated with higher levels of whole blood serotonin, independent of demographic factors, medical comorbidities, medication use, platelet count, and cardiac disease severity.
The function of the serotonin transporter (5-HTT, SERT) is to regulate extracellular 5-HT by taking up a single molecule of 5-HT along with one sodium ion into the presynaptic 5-HT terminal effectively reducing synaptic cleft concentrations of 5-HT and inhibiting/terminating post-synaptic neurotransmission[28]. The SERT transports 5-HT into the platelet where it is stored in the dense granules. Studies that have examined the SERT in suicide and depression have shown a decrease in the number of serotonin binding sites[28,33,34]. Serotonin transporters play an important role during depression. The efficacy of both the inward and outward transport of serotonin by the SERT has been found to be enhanced in patients with depression and both inward and outward transport normalizes after therapy[35]. The effect of monoamine oxidase inhibitors and the newer SSRIs are to bind to the serotonin transporter and inhibit the uptake of serotonin into the cell[36]. The human brain and the platelet SERT are identical with only slightly different extents of glycosylation[36]. The maximal velocity of the platelet SERT has been found to be decreased in patients with depression when compared to controls[37]. In another study, drug-naïve depressed patients had serotonin uptake parameters similar to controls; However, after citalopram treatment Fisar et al[38] demonstrated that although there is an expected decreased 5HT uptake with therapy, patients who have a more pronounced decrease in uptake have an increased response to therapy and thus results from 5HT uptake changes can be used to predict response to antidepressant medications.
The 5-HT receptor subtype 2A (5-HT2A) is a G-protein coupled receptor. Upon 5-HT binding, activation of the G-protein occurs, which stimulates membrane bound phospholipase C causing phosphorylation of phosphatidylinositol 4,5-bisphosphate resulting in the formation of diacylglycerol and inositol 1,4,5-triphosphate. Both inositol 1,4,5-triphosphate and diacylglycerol induce calcium release from intracellular stores. Calcium mobilization then leads to platelet activation[39,40]. The 5-HT2A has long been implicated in depression[41]. There is evidence of increased density of the 5-HT2A receptor in platelets of depressed suicide victims[42-44]. Post mortem studies of the brains of several suicide victims show an increase in the number of 5-HT2A receptors[45]. There is also evidence that indicates that the up regulation of the platelet 5-HT receptor found in patients with depression persists even after therapy with antidepressants and clinical improvement[46]. Serotonin receptors and the serotonin transporter on platelets share many physiologic properties with those in the brain. The coding cDNA sequences for the platelet 5-HT2A is identical to the neuronal receptors leading many researchers to infer similar functional findings between platelet and neuronal receptors[47]. Gómez-Gil et al[26] have noted that the platelet 5-HT2A receptor provides a reliable non-invasive means of studying the serotonergic properties of the 5-HT2A receptor in patients with depression. Hrdina et al[42] have found a significant 52% increase in the density of 5-HT2A receptors in platelets from depressed patients, particularly females. This suggests that up-regulation of platelet 5-HT2A receptors may be a useful biological marker in major depression. In vivo studies to date suggest that there is up regulation of the 5-HT receptor, decreased serotonin transporter binding, and decreased rate of 5-HT uptake in patients with depression[39].
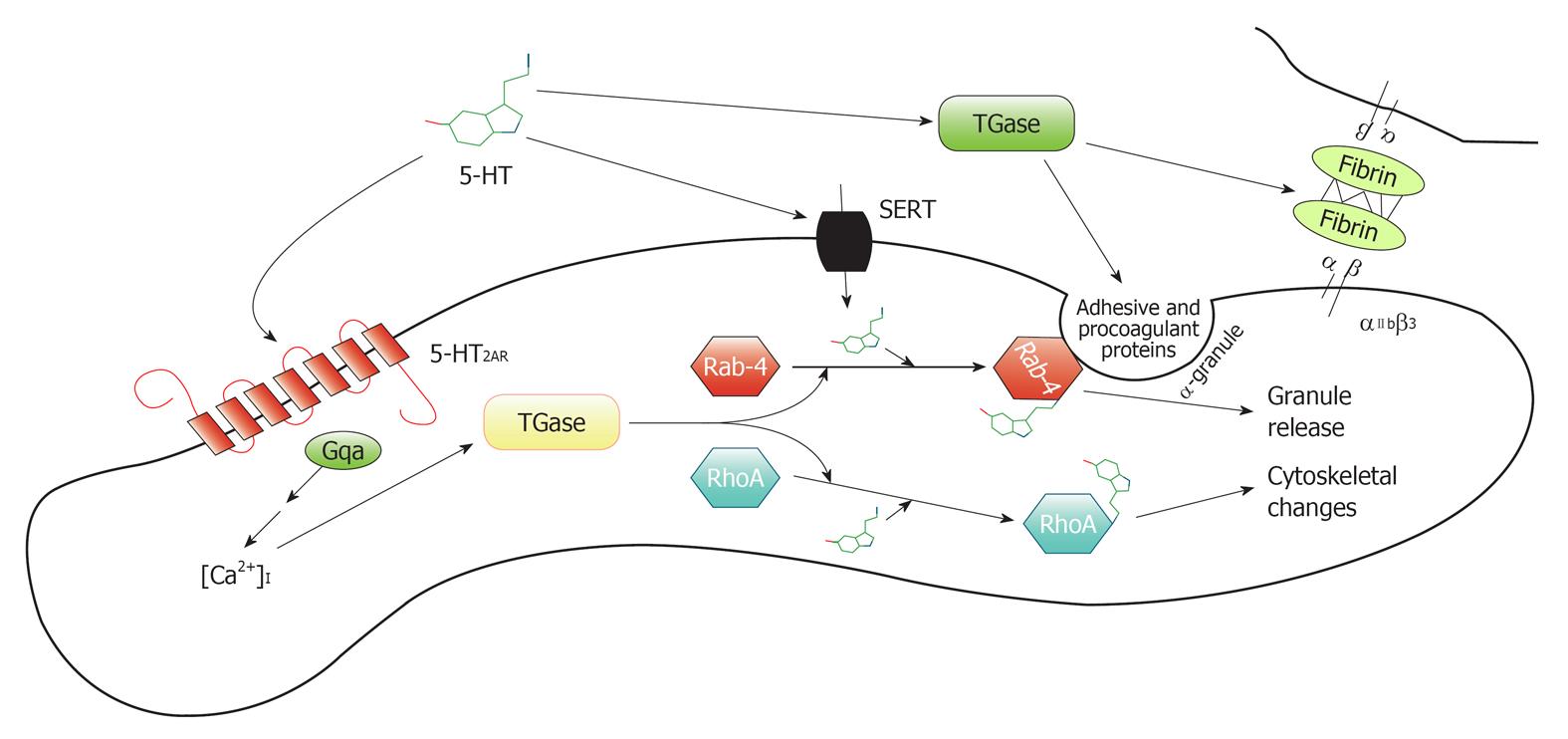
Figure 1 depicts the sequence of activation within the platelet once serotonin binds to the 5-HT2A receptor. 5-HT is actively imported into platelets by the serotonin transporter (5-HTT) and stored in dense granules. 5-HT-mediated stimulation of the 5-HT2A results in a rise in intracellular calcium [Ca2+]I as depicted in Figure 1, taken from Heger et al[40]. This calcium activates transglutaminase (which is the activated form of coagulation factor XIII) and uses 5-HT to transamidate (transfer of an amide molecule) RhoA or Rab4. RhoA leads to cytoskeletal rearrangements and Rab4 is a small Guanosine-5’-triphosphate binding protein that regulates vesicular traffic[48]. Activation of Rab4 leads to α-granule exocytosis. These granules release proaggregatory factors such as von Willebrand factor, Factor V, and fibrinogen, all of which have been shown to be substrates for transamidation. Transamidation of these factors with 5-HT enhances their adhesive properties. Once on the platelet membrane, these factors then participate in further platelet activation events. Extracellular transaminase (Factor XIIIa) also cross-links fibrin bound to its receptor αIIbβ3 which stabilizes the newly-formed platelet plug[40].
In part due to the effectiveness of SSRIs in the treatment of depression and the above evidence for platelet serotonin variations, the serotonin system is a logical candidate for examining genetic polymorphisms and their effects on depression[49].
One described polymorphism of the 5-HT2A gene is a T for C substitution. The T for C substitution at position 102 is located in a noncoding DNA region of the 5-HT2A gene. The 102T/C polymorphism does not result in changes in the amino acid sequence of the 5-HT2A receptor protein but has been shown to be in absolute linkage disequilibrium with a polymorphism (-1438A/G) in the promoter region of 5-HT2A receptor gene. There is no evidence for a functional significance of the 1438A/G polymorphism[50]. Platelet aggregation has been found to be increased by 38% in subjects with T102 homozygosity (T/T genotype) when compared with the carriers of the 102C-allele (T/C and the C/C genotypic groups)[51].
After studying 120 patients with major depression and 131 age, gender, and ethnically controlled controls, Du et al[52] found a significant association between the 102 C allele and major depression. The homozygous allele C frequency was significantly higher in patients than in controls, 35% vs 18%, P= 0.003. The odds ratio in patients with genotype C/C was 2.4 with a 95% confidence interval from 1.3 to 4.3. They found that patients with a 102 C/C genotype had a significantly higher mean Hamilton Rating Scale for Depression item 3 score (indication of suicidal ideation) than T/C or T/T genotype patients. However a subsequent study showed no association between the 5-HT2A polymorphism and major depression[53]. The C allele frequency has been found to be significantly higher in patients with depressive disorders than in those with bipolar disorders and healthy control subjects[54]. Eley et al[55] have found that the T allele to be protective in the major depressive disorder (MDD).
A genetic predisposition to increased stress reactivity is thought to underlie the development of depressive symptoms after a stressful event[56]. SSRIs bind to the 5-HT transporter and inhibit 5-HT uptake. The most widely reported serotonergic marker in major depression and suicide involves the serotonin transporter[57]. The promoter activity of the 5-HTT has several polymorphisms located within the 5’ regulatory region and is known as the 5-HTTLPR[49]. 5-HTT maps to chromosome 17 at position 17-q11.17-q12. The 5-HTTLPR polymorphism is functional, and consists of varying numbers of copies of a 20-23 base pair repeat sequence that has two variants (short-14 repeats and long-16 repeats). The short (S) allele is associated with decreased transcriptional efficiency of the promoter when compared to the long (L) allele. The S allele leads to lower expression of 5-HTT mRNA and lower expression of 5-HT transporter in membranes with decreased 5-HT uptake. The S allele has a dominant mode of action[58]. The 5-HTTLPR is thought to be an excellent candidate allele because it has known biochemical effects. The short allele has been found to interact with stressful life events to predict depression in otherwise healthy individuals; this allele is also associated with less rapid platelet serotonin uptake[59,60].
Several studies have examined the role of the 5-HT transporter polymorphism 5-HTTPLR with varying findings[61]. One study has revealed that individuals with one or two copies of the short allele of the 5-HTTLPR (SL or SS) exhibit more depressive symptoms, diagnosable depression, and suicidality in relation to stressful life events than individuals homozygous for the long allele (LL)[49]. Hayden et al[62] have shown that children homozygous for the short allele of the 5-HTTPLR gene show greater negative schematic processing following a negative mood prime than those with other genotypes. Among patients with chronic illness, carriers of the S allele of 5-HTTLPR are more vulnerable to depression, perceived stress, and high norepinephrine secretion. These factors are thought to contribute to worse cardiovascular outcomes in these patients[63]. A study done among 70 white European psychiatric outpatients suffering from MDD revealed an increased frequency of the 5-HTTLPR-S allele (P < 0.05) when compared to Healthy Volunteers (HV). The odds ratio associated with 5-HTTLPR-S allele was 2.03 for the MDD patients in comparison with the HV group[64]. However, no association between the 5-HTTLPR polymorphism and either unipolar or bipolar depression was found in a large European multicenter case-control sample, including 539 unipolar, 572 bipolar patients, and 821 controls[65]. Minov et al[53] found a trend toward a higher frequency of the SS allele of the 5-HTTLPR in patients with major depression (23.1%) vs controls (14%) but since this did not reach statistical significance, the authors concluded that the 5-HTTLPR polymorphism is not a major susceptibility factor in the etiology of major depression.
Platelet functional implications have been studied in relation to the 5-HTTLPR gene. Platelet factor 4 and β thromboglobulin were found to be higher in elderly patients with depression and the 5-HTTLPR LL genotype[66]. A prospective study was done to investigate whether the 5-HT transporter polymorphism increases the bleeding time in paroxetine users. There were no significant difference seen between the SS, SL, LL genotypes of the 5-HT transporter and the Platelet Function Assay (PFA)-closure time, a measure of platelet agglutination[67].
Several studies have examined the role of 5-HTTLPR in depression but have obtained inconsistent results[68]. Among white individuals, the frequency of the L allele is thought to be 0.60 and the frequency of the S allele is thought to be 0.40[58]. Reports have raised the possibility that there exist variants of both the S and L alleles[69,70]. Hu et al[68] have found an A for G single nucleotide polymorphism (SNP) within the 5-HTTLPR insertion that leads to only the A variant of the L allele denoted LA which yields high amounts of 5-HTT mRNA levels while the G variant LG leads to low levels of 5-HTT mRNA. LG behaves equivalent to the low expressing S allele. This has prompted investigators to reconsider the established subdivision of 5-HTTLPR into S and L allele[71]. Hu and colleagues have found the frequency of the LG allele to be 0.14 with the LA frequency of 0.49 with S being 0.37 in a population of US whites. Given the fact that the LG allele is functionally equivalent to the S allele, the ratio of alleles which lead to low expression of 5-HTT mRNA changes with reappraisal of 5-HTTLPR polymorphism. With this information the 5-HTTLPR is functionally triallelic with the functional alleles being: S, LG, and LA. The gain of function genotype LA LA genotype is twice as common among 169 whites with obsessive compulsive disorders compared to 253 controls leading to the conclusion that the 5-HTTLPR LA LA genotype exerts a 1.8 fold effect on the risk of obsessive compulsive disorder. In another study, the LA variant allele of the 5-HTTLPR polymorphism was found to modify the bleeding effects of SSRIs. Platelet serotonin levels are further decreased in patients without LA alleles on SSRIs. PFA closure time and PF4 levels are increased significantly in patients without LA alleles[72].
Brockmann et al[73] have demonstrated that cerebral blood flow is higher in mood modulating areas of the brain in MDD patients with lower serotonin expression as assess by the 5-HTTLPR gene polymorphism (i.e., S/S, L/S or Non-LALA genetic groups). These investigators feel that the need to monitor and to account for individual 5-HTTLPR-status in future MDD imaging studies is highly recommended.
Patients with major depression have been found to have significantly higher intracellular calcium responses to thrombin stimulation than control subjects[74]. Since both serotonin (5-HT2A) and α(2)-adrenergic receptors are linked to the phosphoinositide (PI) signaling system in platelets and brain, this system has been investigated as possible mechanistic pathways in depression. Depressed patients have been found to have significantly higher thrombin-stimulated inositol phosphate formation in platelets when compared to non depressed controls indicating a hyperactive PI signaling system[75]. Investigators concluded that this hyperactive system may be related either to an increased number of thrombin receptors or to a generalized overstimulation of this pathway. However other investigators have shown that thrombin receptor coupled G protein function is unaltered in patients with unipolar and bipolar depression[76]. It has also been found that patients with anxiety and depression on serotonergic antidepressants have decreased procoagulant effects when compared to similar patients not on serotonergic antidepressants[77].
Reduced norepinephrine function at neuronal adrenoreceptors is a well accepted mechanism of depression. Several studies have shown that major depression is associated with up regulation of the α-adrenergic receptor in the brain[78]. Therefore platelet α-adrenergic receptor density has been studied in patients with major depression and when compared to healthy controls patients with MDD have significantly higher α(2A) adrenergic receptor density, particularly in the high-conformational state, than control subjects[79,80]. However other studies have not demonstrated a change in α (2)-adrenoreceptor density as measured by [3H] rauwolscine binding in patients with major depression[81]. An interesting finding, however, in this study was that patients with a higher density of α 2 adrenergic receptors have increased severity of depression as measured using the Hamilton Rating Scale of Depression.
There are four subtypes of the platelet adenosine receptor A1, A2a, A2b, and A3. Stimulation of the platelet A2a receptor leads to increased intracellular cyclic AMP, inhibition of serotonin release, and inhibition of platelet aggregation. In a study examining the response to stimulation, patients with major depression have been found to have a blunted intracellular calcium response to A2a stimulation suggesting dysregulation of the platelet adenosine receptor in major depression[82].
The platelet glutamate receptors has been found to be supersensitive in depression[83]. Most recently the platelet glutamate receptor has been proposed as potentially playing a role in major depression and cardiovascular disease[84].
Patients with major depression have been shown to have increased mean platelet volume when compared to healthy controls[85,86]. In addition citalopram has been shown to decrease mean platelet volume after 8 wk of open label treatment.
Brain derived neurotrophic factor (BDNF) is a member of the nerve growth factor and as such plays a critical role in the survival, differentiation, neuronal strength, and morphology of neurons[87]. BDNF is also found in both serum and plasma. Serum levels are 100-200 times higher than in plasma[88]. Human platelets contain a significant amount of the plasma BDNF and due to the ease of measuring serum and platelet BDNF over neuronal BDNF, both serum and platelets have been used to measure BDNF levels. Stress decreases the expression of BDNF while antidepressants increase the expression. It has been proposed that decreased BDNF could lead to atrophy of limbic structures that have been found in depressed patients[89,90]. Platelet BDNF levels have been measured in depressed patients and have been found to be decreased when compared to normal controls. However plasma BDNF levels have been found to be increased in these same patients when compared to controls[91]. After treatment of depression with Citalopram, BDNF levels normalize to the level of controls. SNP in the BDNF gene leading to a G (Val) to A (Met) substitution at position 196 has been found to be associated with neuropsychiatric disorders including depression and bipolar disorder[92,93]. Investigators have shown a higher frequency of the AA genotype in CAD patients with depression while the GG genotype is lower in these patients[94].
One potential reason for the many discrepancies in platelet function when it comes to depression and platelet function may lie with the various methods used to assess platelet function. There are several areas for variability in testing results. Whether or not a subject is fasting effects the amount of lipids in the plasma thereby changing the results. The method of blood drawing is crucial in the initial steps of preparing the sample for analysis. Excessive shear forces with rapid withdrawal of blood into a syringe can cause platelet activation[95]. Even tourniquet pressure if excessive can cause platelet activation. The amount and type of anticoagulant used can also vary results. Patients with higher hematocrits require lower concentrations of anticoagulant[96]. The container into which blood is drawn is also a very important factor since platelets can stick to glass. There are biological variations in platelet function (increased aggregation in the early morning) that affect the timing of blood draw[97]. Drawing blood at relatively the same time of day when comparing patient results to themselves as well as to others is important. Platelet count is also crucial when comparing different results, because higher platelet count changes results dramatically. Finally, platelets become nonfunctional approximately 4 h after blood draw therefore functional studies should always be time dependant.
Another major source of variability is intra-observer and inter-observer variability. The precision of the technician is extremely crucial in measuring platelet aggregation by light transmission. Methods used to determine intra-observer variability include, performing identical repetitions of assays by the same technician. Careful blood draw using a 19 or 21 gauge needle each time reduces the unintended activation of platelets. Blood should also be drawn as close to the same time of day each time if a study is being performed to compare the results of platelet function. Functional assays should be performed within 4 h of blood draw. Other factors to take into consideration are the presence of other antiplatelet medications which may alter platelet function. In particular when platelet aggregation is measured in patients, knowing which agonists stimulate which platelet pathway is essential in the interpretation of results. When working with platelet genotyping a pivotal aspect of data interpretation is the appropriate and thorough phenotyping of patient cohorts. Individuals with stable CAD, acute coronary syndrome, and patients without CAD all have different platelet function[98]. Interpretation of functional studies in light of genotype differences have been demonstrated in various studies[99,100]. Therefore careful considerations of expected and actual platelet functional results are equally as important. Finally the exact characterization of the population being studied is essential in the interpretation of platelet functional results. Whether patients have MDD vs depressive symptoms can lead to different platelet functional observations.
The current literature on platelet function and depression has highlighted the fact that platelet function is increased in patients with depression and CAD potentially explaining the increased rate of morbidity and mortality in these individuals. What is less consistent is the etiology of this increased platelet function. With the success of SSRIs in the treatment of depression and the similarity between the platelet serotonin receptor and the brain serotonin receptor, investigators have postulated that platelet serotonin function would be altered. To date studies show increased serotonin response, increase platelet serotonin density, decreased serotonin transporter binding, and decreased platelet serotonin levels in individuals with depression. However other studies have shown no change in serotonin uptake. In addition to platelet serotonin specific pathways, other platelet pathways that have shown significant changes in depressed individuals include blunting of the platelet adenosine response, increased platelet thrombin response, increased glycoprotein Ib expression, increased P-selectin, β thromboglobulin, and PF4, as well as decreased platelet BDNF. However there are other studies that show conflicting evidence of increased platelet activation as measured by integrin receptor α2bβ3 which is generally increased in states of platelet activation. Other conflicting data include α adrenergic density and platelet response to augmented serotonin. Table 1 demonstrates a summary of major specific platelet pathway findings in depression. When functional assays are completed they should be performed ideally in the morning hours, with appropriate attention to proper blood drawing techniques. Functional assays that appear to be most consistent with depression biochemistry include serotonin and serotonin augmented platelet function. As always platelet function should be performed by individuals knowledgeable in the performance of platelet functional assays. The direction of future research in platelet functional changes in depression and CAD should continue to focus on serotonin specific pathways with emphasis on potential mechanisms of specific pathway changes keeping in mind that the examination of platelet reactivity can often lead to the discovery of novel platelet associated proteins/pathways.
| Platelet pathway | Results | Population studied | Ref. |
| Integrin receptor | No change in expression | Depressed-Hamilton scale | [25] |
| α2bβ3 (GP IIb/IIIa) | Increased expression with orthostatic challenge | Depressed | [17] |
| Integrin Glycoprotein Ib | Increased expression | Depressed-Hamilton scale | [25] |
| P-selectin | Increased with orthostatic challenge | Depressed | [17] |
| P-selectin | Increased | Depressed-Hamilton scale | [25] |
| β TG + PF4 | Increased expression | Depressed with CAD - SCID, HDRS ≥ 15, CGI ≥ 4 | [19,20,22] |
| 5HT amplified Epinephrine whole blood platelet aggregation | Increased | MDD-Diagnostic and Statistical Manual of Mental Disorders | [24] |
| ADP Induced PRP aggregation | No change | Depressed | [24,25] |
| 5HT amplified ADP platelet aggregation | No change | MDD- Hamilton | [26] |
| Platelet 5-HT level | decreased | Depression | [29-31] |
| 5-HT2A | Increased density/Upregulation | Depressed/Suicide | [42-44,46] |
| 5HT-Transporter (SERT) | Decreased binding | Depressed | [37] |
| 5HT Uptake | Unchanged | Depressed | [38] |
| Thrombin | Increased thrombin stimulated Inositol phosphate formation | Depressed | [74,75] |
| α adrenergic receptor | Increased density | MDD | [79,80] |
| No change | MDD | [81] | |
| Adenosine | Blunted intracellular calcium response | MDD | [82] |
| BDNF | Decreased platelet levels | Depression | [91] |
Peer reviewer: John E Zeber, PhD, Scott and White Healthcare, Center for Applied Health Research, Co-director, Health Outcomes Core, Investigator, Central Texas Veterans Healthcare System, Associate Professor, Texas A and M HSC College of Medicine, 2102 Birdcreek Drive, Temple, TX 76501, United States
S- Editor Wang JL L- Editor A E- Editor Zheng XM
| 1. | Pleis JR, Ward BW, Lucas JW. Summary health statistics for U.S. adults: National Health Interview Survey, 2009. Vital Health Stat 10. 2010;249:1-207. [PubMed] |
| 2. | Forrester AW, Lipsey JR, Teitelbaum ML, DePaulo JR, Andrzejewski PL. Depression following myocardial infarction. Int J Psychiatry Med. 1992;22:33-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 95] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 3. | Frasure-Smith N, Lespérance F, Talajic M. Depression following myocardial infarction. Impact on 6-month survival. JAMA. 1993;270:1819-1825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1162] [Cited by in RCA: 986] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 4. | Thombs BD, Bass EB, Ford DE, Stewart KJ, Tsilidis KK, Patel U, Fauerbach JA, Bush DE, Ziegelstein RC. Prevalence of depression in survivors of acute myocardial infarction. J Gen Intern Med. 2006;21:30-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 566] [Cited by in RCA: 577] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 5. | Lespérance F, Frasure-Smith N, Talajic M, Bourassa MG. Five-year risk of cardiac mortality in relation to initial severity and one-year changes in depression symptoms after myocardial infarction. Circulation. 2002;105:1049-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 480] [Cited by in RCA: 453] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 6. | Bush DE, Ziegelstein RC, Tayback M, Richter D, Stevens S, Zahalsky H, Fauerbach JA. Even minimal symptoms of depression increase mortality risk after acute myocardial infarction. Am J Cardiol. 2001;88:337-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 422] [Cited by in RCA: 411] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 7. | Penninx BW, Beekman AT, Honig A, Deeg DJ, Schoevers RA, van Eijk JT, van Tilburg W. Depression and cardiac mortality: results from a community-based longitudinal study. Arch Gen Psychiatry. 2001;58:221-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 647] [Cited by in RCA: 603] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 8. | Parissis JT, Fountoulaki K, Filippatos G, Adamopoulos S, Paraskevaidis I, Kremastinos D. Depression in coronary artery disease: novel pathophysiologic mechanisms and therapeutic implications. Int J Cardiol. 2007;116:153-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 9. | Carney RM, Freedland KE, Eisen SA, Rich MW, Jaffe AS. Major depression and medication adherence in elderly patients with coronary artery disease. Health Psychol. 1995;14:88-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 349] [Cited by in RCA: 313] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 10. | Whooley MA, de Jonge P, Vittinghoff E, Otte C, Moos R, Carney RM, Ali S, Dowray S, Na B, Feldman MD. Depressive symptoms, health behaviors, and risk of cardiovascular events in patients with coronary heart disease. JAMA. 2008;300:2379-2388. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 641] [Cited by in RCA: 652] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 11. | Lehto S, Koukkunen H, Hintikka J, Viinamäki H, Laakso M, Pyörälä K. Depression after coronary heart disease events. Scand Cardiovasc J. 2000;34:580-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 12. | Nemeroff CB, Musselman DL. Are platelets the link between depression and ischemic heart disease. Am Heart J. 2000;140:57-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 141] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 13. | Markovitz JH, Matthews KA. Platelets and coronary heart disease: potential psychophysiologic mechanisms. Psychosom Med. 1991;53:643-668. [PubMed] |
| 14. | Musselman DL, Marzec U, Davidoff M, Manatunga AK, Gao F, Reemsnyder A, Duggirala S, Larsen H, Taylor RW, Hanson S. Platelet activation and secretion in patients with major depression, thoracic aortic atherosclerosis, or renal dialysis treatment. Depress Anxiety. 2002;15:91-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Skop BP, Brown TM. Potential vascular and bleeding complications of treatment with selective serotonin reuptake inhibitors. Psychosomatics. 1996;37:12-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 127] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 17. | Musselman DL, Tomer A, Manatunga AK, Knight BT, Porter MR, Kasey S, Marzec U, Harker LA, Nemeroff CB. Exaggerated platelet reactivity in major depression. Am J Psychiatry. 1996;153:1313-1317. [PubMed] |
| 18. | Markovitz JH, Shuster JL, Chitwood WS, May RS, Tolbert LC. Platelet activation in depression and effects of sertraline treatment: An open-label study. Am J Psychiatry. 2000;157:1006-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 103] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 19. | Pollock BG, Laghrissi-Thode F, Wagner WR. Evaluation of platelet activation in depressed patients with ischemic heart disease after paroxetine or nortriptyline treatment. J Clin Psychopharmacol. 2000;20:137-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 137] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 20. | Laghrissi-Thode F, Wagner WR, Pollock BG, Johnson PC, Finkel MS. Elevated platelet factor 4 and beta-thromboglobulin plasma levels in depressed patients with ischemic heart disease. Biol Psychiatry. 1997;42:290-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 225] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 21. | Lederbogen F, Gilles M, Maras A, Hamann B, Colla M, Heuser I, Deuschle M. Increased platelet aggregability in major depression. Psychiatry Res. 2001;102:255-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 55] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Serebruany VL, Glassman AH, Malinin AI, Nemeroff CB, Musselman DL, van Zyl LT, Finkel MS, Krishnan KR, Gaffney M, Harrison W. Platelet/endothelial biomarkers in depressed patients treated with the selective serotonin reuptake inhibitor sertraline after acute coronary events: the Sertraline AntiDepressant Heart Attack Randomized Trial (SADHART) Platelet Substudy. Circulation. 2003;108:939-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 249] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 23. | Serebruany VL, Glassman AH, Malinin AI, Sane DC, Finkel MS, Krishnan RR, Atar D, Lekht V, O'Connor CM. Enhanced platelet/endothelial activation in depressed patients with acute coronary syndromes: evidence from recent clinical trials. Blood Coagul Fibrinolysis. 2003;14:563-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 73] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 24. | Shimbo D, Child J, Davidson K, Geer E, Osende JI, Reddy S, Dronge A, Fuster V, Badimon JJ. Exaggerated serotonin-mediated platelet reactivity as a possible link in depression and acute coronary syndromes. Am J Cardiol. 2002;89:331-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Walsh MT, Dinan TG, Condren RM, Ryan M, Kenny D. Depression is associated with an increase in the expression of the platelet adhesion receptor glycoprotein Ib. Life Sci. 2002;70:3155-3165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | Gómez-Gil E, Gastó C, Díaz-Ricart M, Carretero M, Salamero M, Catalán R, Escolar G. Platelet 5-HT2A-receptor-mediated induction of aggregation is not altered in major depression. Hum Psychopharmacol. 2002;17:419-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 27. | Parakh K, Sakhuja A, Bhat U, Ziegelstein RC. Platelet function in patients with depression. South Med J. 2008;101:612-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Owens MJ, Nemeroff CB. Role of serotonin in the pathophysiology of depression: focus on the serotonin transporter. Clin Chem. 1994;40:288-295. [PubMed] |
| 29. | Maurer-Spurej E, Pittendreigh C, Solomons K. The influence of selective serotonin reuptake inhibitors on human platelet serotonin. Thromb Haemost. 2004;91:119-128. [PubMed] |
| 30. | Muck-Seler D, Pivac N, Mustapic M, Crncevic Z, Jakovljevic M, Sagud M. Platelet serotonin and plasma prolactin and cortisol in healthy, depressed and schizophrenic women. Psychiatry Res. 2004;127:217-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 71] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 31. | Maurer-Spurej E, Pittendreigh C, Misri S. Platelet serotonin levels support depression scores for women with postpartum depression. J Psychiatry Neurosci. 2007;32:23-29. [PubMed] |
| 32. | Wulsin LR, Musselman D, Otte C, Bruce E, Ali S, Whooley MA. Depression and whole blood serotonin in patients with coronary heart disease from the Heart and Soul Study. Psychosom Med. 2009;71:260-265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 33. | Leake A, Fairbairn AF, McKeith IG, Ferrier IN. Studies on the serotonin uptake binding site in major depressive disorder and control post-mortem brain: neurochemical and clinical correlates. Psychiatry Res. 1991;39:155-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 59] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 34. | Malison RT, Price LH, Berman R, van Dyck CH, Pelton GH, Carpenter L, Sanacora G, Owens MJ, Nemeroff CB, Rajeevan N. Reduced brain serotonin transporter availability in major depression as measured by [123I]-2 beta-carbomethoxy-3 beta-(4-iodophenyl)tropane and single photon emission computed tomography. Biol Psychiatry. 1998;44:1090-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 344] [Cited by in RCA: 309] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 35. | Willeit M, Sitte HH, Thierry N, Michalek K, Praschak-Rieder N, Zill P, Winkler D, Brannath W, Fischer MB, Bondy B. Enhanced serotonin transporter function during depression in seasonal affective disorder. Neuropsychopharmacology. 2008;33:1503-1513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 36. | Owens MJ, Nemeroff CB. The serotonin transporter and depression. Depress Anxiety. 1998;8 Suppl 1:5-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 37. | Stain-Malmgren R, Khoury AE, Aberg-Wistedt A, Tham A. Serotonergic function in major depression and effect of sertraline and paroxetine treatment. Int Clin Psychopharmacol. 2001;16:93-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 38. | Fisar Z, Kalisová L, Paclt I, Anders M, Vevera J. Platelet serotonin uptake in drug-naïve depressive patients before and after treatment with citalopram. Psychiatry Res. 2008;161:185-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 39. | Schins A, Honig A, Crijns H, Baur L, Hamulyák K. Increased coronary events in depressed cardiovascular patients: 5-HT2A receptor as missing link. Psychosom Med. 2003;65:729-737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 40. | Heger CD, Collins RN. Platelet activation and "crossover appeal": Rab and Rho families united by common links to serotonin. Mol Interv. 2004;4:79-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 41. | Mann JJ. Role of the serotonergic system in the pathogenesis of major depression and suicidal behavior. Neuropsychopharmacology. 1999;21:99S-105S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 310] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 42. | Hrdina PD, Bakish D, Chudzik J, Ravindran A, Lapierre YD. Serotonergic markers in platelets of patients with major depression: upregulation of 5-HT2 receptors. J Psychiatry Neurosci. 1995;20:11-19. [PubMed] |
| 43. | McBride PA, Mann JJ, Polley MJ, Wiley AJ, Sweeney JA. Assessment of binding indices and physiological responsiveness of the 5-HT2 receptor on human platelets. Life Sci. 1987;40:1799-1809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 44. | Pandey GN, Pandey SC, Janicak PG, Marks RC, Davis JM. Platelet serotonin-2 receptor binding sites in depression and suicide. Biol Psychiatry. 1990;28:215-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 139] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 45. | Hrdina PD, Demeter E, Vu TB, Sótónyi P, Palkovits M. 5-HT uptake sites and 5-HT2 receptors in brain of antidepressant-free suicide victims/depressives: increase in 5-HT2 sites in cortex and amygdala. Brain Res. 1993;614:37-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 208] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 46. | Hrdina PD, Bakish D, Ravindran A, Chudzik J, Cavazzoni P, Lapierre YD. Platelet serotonergic indices in major depression: up-regulation of 5-HT2A receptors unchanged by antidepressant treatment. Psychiatry Res. 1997;66:73-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 73] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 47. | Cook EH, Fletcher KE, Wainwright M, Marks N, Yan SY, Leventhal BL. Primary structure of the human platelet serotonin 5-HT2A receptor: identify with frontal cortex serotonin 5-HT2A receptor. J Neurochem. 1994;63:465-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 100] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 48. | Walther DJ, Peter JU, Winter S, Höltje M, Paulmann N, Grohmann M, Vowinckel J, Alamo-Bethencourt V, Wilhelm CS, Ahnert-Hilger G. Serotonylation of small GTPases is a signal transduction pathway that triggers platelet alpha-granule release. Cell. 2003;115:851-862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 362] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 49. | Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H, McClay J, Mill J, Martin J, Braithwaite A. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science. 2003;301:386-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5478] [Cited by in RCA: 4740] [Article Influence: 215.5] [Reference Citation Analysis (0)] |
| 50. | Spurlock G, Heils A, Holmans P, Williams J, D'Souza UM, Cardno A, Murphy KC, Jones L, Buckland PR, McGuffin P. A family based association study of T102C polymorphism in 5HT2A and schizophrenia plus identification of new polymorphisms in the promoter. Mol Psychiatry. 1998;3:42-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 160] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 51. | Ozdener F, Gülbas Z, Erol K, Ozdemir V. 5-Hydroxytryptamine-2A receptor gene (HTR 2 A) candidate polymorphism (T 102 C): Role for human platelet function under pharmacological challenge ex vivo. Methods Find Exp Clin Pharmacol. 2005;27:395-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 52. | Du L, Bakish D, Lapierre YD, Ravindran AV, Hrdina PD. Association of polymorphism of serotonin 2A receptor gene with suicidal ideation in major depressive disorder. Am J Med Genet. 2000;96:56-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 53. | Minov C, Baghai TC, Schüle C, Zwanzger P, Schwarz MJ, Zill P, Rupprecht R, Bondy B. Serotonin-2A-receptor and -transporter polymorphisms: lack of association in patients with major depression. Neurosci Lett. 2001;303:119-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 125] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 54. | Zhang HY, Ishigaki T, Tani K, Chen K, Shih JC, Miyasato K, Ohara K, Ohara K. Serotonin2A receptor gene polymorphism in mood disorders. Biol Psychiatry. 1997;41:768-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 66] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 55. | Eley TC, Sugden K, Corsico A, Gregory AM, Sham P, McGuffin P, Plomin R, Craig IW. Gene-environment interaction analysis of serotonin system markers with adolescent depression. Mol Psychiatry. 2004;9:908-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 439] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 56. | Kendler KS, Kessler RC, Walters EE, MacLean C, Neale MC, Heath AC, Eaves LJ. Stressful life events, genetic liability, and onset of an episode of major depression in women. Am J Psychiatry. 1995;152:833-842. [PubMed] |
| 57. | Mann JJ, Huang YY, Underwood MD, Kassir SA, Oppenheim S, Kelly TM, Dwork AJ, Arango V. A serotonin transporter gene promoter polymorphism (5-HTTLPR) and prefrontal cortical binding in major depression and suicide. Arch Gen Psychiatry. 2000;57:729-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 414] [Cited by in RCA: 391] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 58. | Lesch KP, Bengel D, Heils A, Sabol SZ, Greenberg BD, Petri S, Benjamin J, Müller CR, Hamer DH, Murphy DL. Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region. Science. 1996;274:1527-1531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3630] [Cited by in RCA: 3446] [Article Influence: 118.8] [Reference Citation Analysis (0)] |
| 59. | Glatz K, Mössner R, Heils A, Lesch KP. Glucocorticoid-regulated human serotonin transporter (5-HTT) expression is modulated by the 5-HTT gene-promotor-linked polymorphic region. J Neurochem. 2003;86:1072-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 89] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 60. | Figueras G, Pérez V, San Martino O, Alvarez E, Artigas F. Pretreatment platelet 5-HT concentration predicts the short-term response to paroxetine in major depression. Grupo de Trastornos Afectivos. Biol Psychiatry. 1999;46:518-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 61. | Glatt CE, Freimer NB. Association analysis of candidate genes for neuropsychiatric disease: the perpetual campaign. Trends Genet. 2002;18:307-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 62. | Hayden EP, Dougherty LR, Maloney B, Olino TM, Sheikh H, Durbin CE, Nurnberger JI, Lahiri DK, Klein DN. Early-emerging cognitive vulnerability to depression and the serotonin transporter promoter region polymorphism. J Affect Disord. 2008;107:227-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 63. | Otte C, McCaffery J, Ali S, Whooley MA. Association of a serotonin transporter polymorphism (5-HTTLPR) with depression, perceived stress, and norepinephrine in patients with coronary disease: the Heart and Soul Study. Am J Psychiatry. 2007;164:1379-1384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 104] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 64. | Dorado P, Peñas-Lledó EM, González AP, Cáceres MC, Cobaleda J, Llerena A. Increased risk for major depression associated with the short allele of the serotonin transporter promoter region (5-HTTLPR-S) and the CYP2C9*3 allele. Fundam Clin Pharmacol. 2007;21:451-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 65. | Mendlewicz J, Massat I, Souery D, Del-Favero J, Oruc L, Nöthen MM, Blackwood D, Muir W, Battersby S, Lerer B. Serotonin transporter 5HTTLPR polymorphism and affective disorders: no evidence of association in a large European multicenter study. Eur J Hum Genet. 2004;12:377-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 64] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 66. | Whyte EM, Pollock BG, Wagner WR, Mulsant BH, Ferrell RE, Mazumdar S, Reynolds CF. Influence of serotonin-transporter-linked promoter region polymorphism on platelet activation in geriatric depression. Am J Psychiatry. 2001;158:2074-2076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 67. | Hougardy DM, Egberts TC, van der Graaf F, Brenninkmeijer VJ, Derijks LJ. Serotonin transporter polymorphism and bleeding time during SSRI therapy. Br J Clin Pharmacol. 2008;65:761-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 68. | Hu XZ, Lipsky RH, Zhu G, Akhtar LA, Taubman J, Greenberg BD, Xu K, Arnold PD, Richter MA, Kennedy JL. Serotonin transporter promoter gain-of-function genotypes are linked to obsessive-compulsive disorder. Am J Hum Genet. 2006;78:815-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 890] [Cited by in RCA: 885] [Article Influence: 46.6] [Reference Citation Analysis (0)] |
| 69. | Nakamura M, Ueno S, Sano A, Tanabe H. The human serotonin transporter gene linked polymorphism (5-HTTLPR) shows ten novel allelic variants. Mol Psychiatry. 2000;5:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 438] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 70. | Sakai K, Nakamura M, Ueno S, Sano A, Sakai N, Shirai Y, Saito N. The silencer activity of the novel human serotonin transporter linked polymorphic regions. Neurosci Lett. 2002;327:13-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 71. | Wendland JR, Martin BJ, Kruse MR, Lesch KP, Murphy DL. Simultaneous genotyping of four functional loci of human SLC6A4, with a reappraisal of 5-HTTLPR and rs25531. Mol Psychiatry. 2006;11:224-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 430] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 72. | Abdelmalik N, Ruhé HG, Barwari K, van den Dool EJ, Meijers JC, Middeldorp S, Büller HR, Schene AH, Kamphuisen PW. Effect of the selective serotonin reuptake inhibitor paroxetine on platelet function is modified by a SLC6A4 serotonin transporter polymorphism. J Thromb Haemost. 2008;6:2168-2174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 73. | Brockmann H, Zobel A, Schuhmacher A, Daamen M, Joe A, Biermann K, Schwab SG, Biersack HJ, Maier W, Boecker H. Influence of 5-HTTLPR polymorphism on resting state perfusion in patients with major depression. J Psychiatr Res. 2011;45:442-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 74. | Berk M, Plein H. Platelet supersensitivity to thrombin stimulation in depression: a possible mechanism for the association with cardiovascular mortality. Clin Neuropharmacol. 2000;23:182-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 75. | Pandey GN, Ren X, Pandey SC, Dwivedi Y, Sharma R, Janicak PG. Hyperactive phosphoinositide signaling pathway in platelets of depressed patients: effect of desipramine treatment. Psychiatry Res. 2001;105:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 76. | Odagaki Y, Koyama T. Epinephrine- and thrombin-stimulated high-affinity GTPase activity in platelet membranes from patients with psychiatric disorders. Psychiatry Res. 2002;112:111-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 77. | Geiser F, Conrad R, Imbierowicz K, Meier C, Liedtke R, Klingmüller D, Oldenburg J, Harbrecht U. Coagulation activation and fibrinolysis impairment are reduced in patients with anxiety and depression when medicated with serotonergic antidepressants. Psychiatry Clin Neurosci. 2011;65:518-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 78. | García-Sevilla JA, Escribá PV, Ozaita A, La Harpe R, Walzer C, Eytan A, Guimón J. Up-regulation of immunolabeled alpha2A-adrenoceptors, Gi coupling proteins, and regulatory receptor kinases in the prefrontal cortex of depressed suicides. J Neurochem. 1999;72:282-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 117] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 79. | Gurguis GN, Vo SP, Griffith JM, Rush AJ. Platelet alpha2A-adrenoceptor function in major depression: Gi coupling, effects of imipramine and relationship to treatment outcome. Psychiatry Res. 1999;89:73-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 80. | García-Sevilla JA, Ventayol P, Pérez V, Rubovszky G, Puigdemont D, Ferrer-Alcón M, Andreoli A, Guimón J, Alvarez E. Regulation of platelet alpha 2A-adrenoceptors, Gi proteins and receptor kinases in major depression: effects of mirtazapine treatment. Neuropsychopharmacology. 2004;29:580-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 81. | Marazziti D, Baroni S, Masala I, Di Nasso E, Giannaccini G, Betti L, Lucacchini A, Cassano GB. Correlation between platelet alpha(2)-adrenoreceptors and symptom severity in major depression. Neuropsychobiology. 2001;44:122-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 82. | Berk M, Plein H, Ferreira D, Jersky B. Blunted adenosine A2a receptor function in platelets in patients with major depression. Eur Neuropsychopharmacol. 2001;11:183-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 83. | Berk M, Plein H, Ferreira D. Platelet glutamate receptor supersensitivity in major depressive disorder. Clin Neuropharmacol. 2001;24:129-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 47] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 84. | Chen H. Possible Role of Platelet GluR1 Receptors in Comorbid Depression and Cardiovascular Disease. Cardiovasc Psychiatry Neurol. 2009;2009:424728. [PubMed] |
| 85. | Ataoglu A, Canan F. Mean platelet volume in patients with major depression: effect of escitalopram treatment. J Clin Psychopharmacol. 2009;29:368-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 86. | Canan F, Dikici S, Kutlucan A, Celbek G, Coskun H, Gungor A, Aydin Y, Kocaman G. Association of mean platelet volume with DSM-IV major depression in a large community-based population: the MELEN study. J Psychiatr Res. 2012;46:298-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 87. | Duman RS. Novel therapeutic approaches beyond the serotonin receptor. Biol Psychiatry. 1998;44:324-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 171] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 88. | Fujimura H, Altar CA, Chen R, Nakamura T, Nakahashi T, Kambayashi J, Sun B, Tandon NN. Brain-derived neurotrophic factor is stored in human platelets and released by agonist stimulation. Thromb Haemost. 2002;87:728-734. [PubMed] |
| 89. | Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 2006;59:1116-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2372] [Cited by in RCA: 2477] [Article Influence: 130.4] [Reference Citation Analysis (0)] |
| 90. | Liu CY, Jiang XX, Zhu YH, Wei DN. Metabotropic glutamate receptor 5 antagonist 2-methyl-6-(phenylethynyl)pyridine produces antidepressant effects in rats: role of brain-derived neurotrophic factor. Neuroscience. 2012;223:219-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 91. | Serra-Millàs M, López-Vílchez I, Navarro V, Galán AM, Escolar G, Penadés R, Catalán R, Fañanás L, Arias B, Gastó C. Changes in plasma and platelet BDNF levels induced by S-citalopram in major depression. Psychopharmacology (Berl). 2011;216:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 92. | Ventriglia M, Bocchio Chiavetto L, Benussi L, Binetti G, Zanetti O, Riva MA, Gennarelli M. Association between the BDNF 196 A/G polymorphism and sporadic Alzheimer's disease. Mol Psychiatry. 2002;7:136-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 177] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 93. | Sen S, Nesse RM, Stoltenberg SF, Li S, Gleiberman L, Chakravarti A, Weder AB, Burmeister M. A BDNF coding variant is associated with the NEO personality inventory domain neuroticism, a risk factor for depression. Neuropsychopharmacology. 2003;28:397-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 246] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 94. | Bozzini S, Gambelli P, Boiocchi C, Schirinzi S, Falcone R, Buzzi P, Storti C, Falcone C. Coronary artery disease and depression: possible role of brain-derived neurotrophic factor and serotonin transporter gene polymorphisms. Int J Mol Med. 2009;24:813-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 95. | Harrison MJ, Emmons PR, Mitchell JR. The variability of human platelet aggregation. J Atheroscler Res. 1967;7:197-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 96. | Hardisty RM, Hutton RA, Montgomery D, Rickard S, Trebilcock H. Secondary platelet aggregation: a quantitative study. Br J Haematol. 1970;19:307-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 97. | Cooke-Ariel H. Circadian variations in cardiovascular function and their relation to the occurrence and timing of cardiac events. Am J Health Syst Pharm. 1998;55 Suppl 3:S5-11. [PubMed] |
| 98. | Williams MS, Kickler TS, Vaidya D, Ng'alla LS, Bush DE. Evaluation of platelet function in aspirin treated patients with CAD. J Thromb Thrombolysis. 2006;21:241-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 99. | Michelson AD, Furman MI, Goldschmidt-Clermont P, Mascelli MA, Hendrix C, Coleman L, Hamlington J, Barnard MR, Kickler T, Christie DJ. Platelet GP IIIa Pl(A) polymorphisms display different sensitivities to agonists. Circulation. 2000;101:1013-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 215] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 100. | Bennett JS, Catella-Lawson F, Rut AR, Vilaire G, Qi W, Kapoor SC, Murphy S, FitzGerald GA. Effect of the Pl(A2) alloantigen on the function of beta(3)-integrins in platelets. Blood. 2001;97:3093-3099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 52] [Article Influence: 2.2] [Reference Citation Analysis (0)] |