Published online Sep 19, 2025. doi: 10.5498/wjp.v15.i9.107726
Revised: April 15, 2025
Accepted: June 18, 2025
Published online: September 19, 2025
Processing time: 151 Days and 15.8 Hours
Suicidality is a complex phenomenon influenced by genetic, environmental, and epigenetic factors. Current tools to estimate suicide risk are insufficient, and there is an increasing need for reliable biomarkers to complement clinical approaches. Growing evidence suggests that immune system dysregulation contributes to the pathophysiology of psychiatric disorders and suicidal behavior. Epigenetic mechanisms, including DNA methylation, histone modifications, and non-coding RNAs, regulate gene expression and may act as a bridge between environmental stressors and (neuro)inflammatory responses. In this review, we examine the evidence of peripheral and central inflammation in suicide completers and individuals with suicidal behavior. Next, we review current knowledge from va
Core Tip: Suicidality is heterogeneous and cannot be fully accounted for by psychiatric comorbidities. To understand its biological background, it is important to consider epigenetic regulation as a mediator of suicide risk in response to environmental stimuli, particularly early life adversity. Chronic inflammation has been implicated in neuropsychiatric disorders, and further research should examine whether suicidality is associated with a specific inflammatory profile. The interactions between immune system dysregulation, stress response, and epigenetics hold promise for biomarker discovery but are insufficiently explored. To accelerate the development of targeted suicide prevention strategies, stringent sample selection and integrative multi-omics approaches should be applied.
- Citation: Kouter K, Šmon J, Videtič Paska A. Epigenetics and immunology: Under-recognized aspects of suicidality. World J Psychiatry 2025; 15(9): 107726
- URL: https://www.wjgnet.com/2220-3206/full/v15/i9/107726.htm
- DOI: https://dx.doi.org/10.5498/wjp.v15.i9.107726
Suicide is a major global public health challenge, accounting for more than 720000 deaths each year[1]. Suicide attempts may be up to 20 times more frequent and present a major risk factor for suicide[2]. Suicidal behavior, which includes preparatory behaviors, suicide attempts, and completed suicide, is a complex and poorly understood phenomenon[2]. While commonly associated with an underlying psychiatric disorder, most notably major depressive disorder (MDD), bipolar disorder, substance use disorder, and schizophrenia[3,4], approximately 10% of suicide completers do not have a psychiatric diagnosis at the time of their death[2]. Additionally, few psychiatric patients die due to suicide, making psychiatric diagnosis a poor predictor of suicide[5].
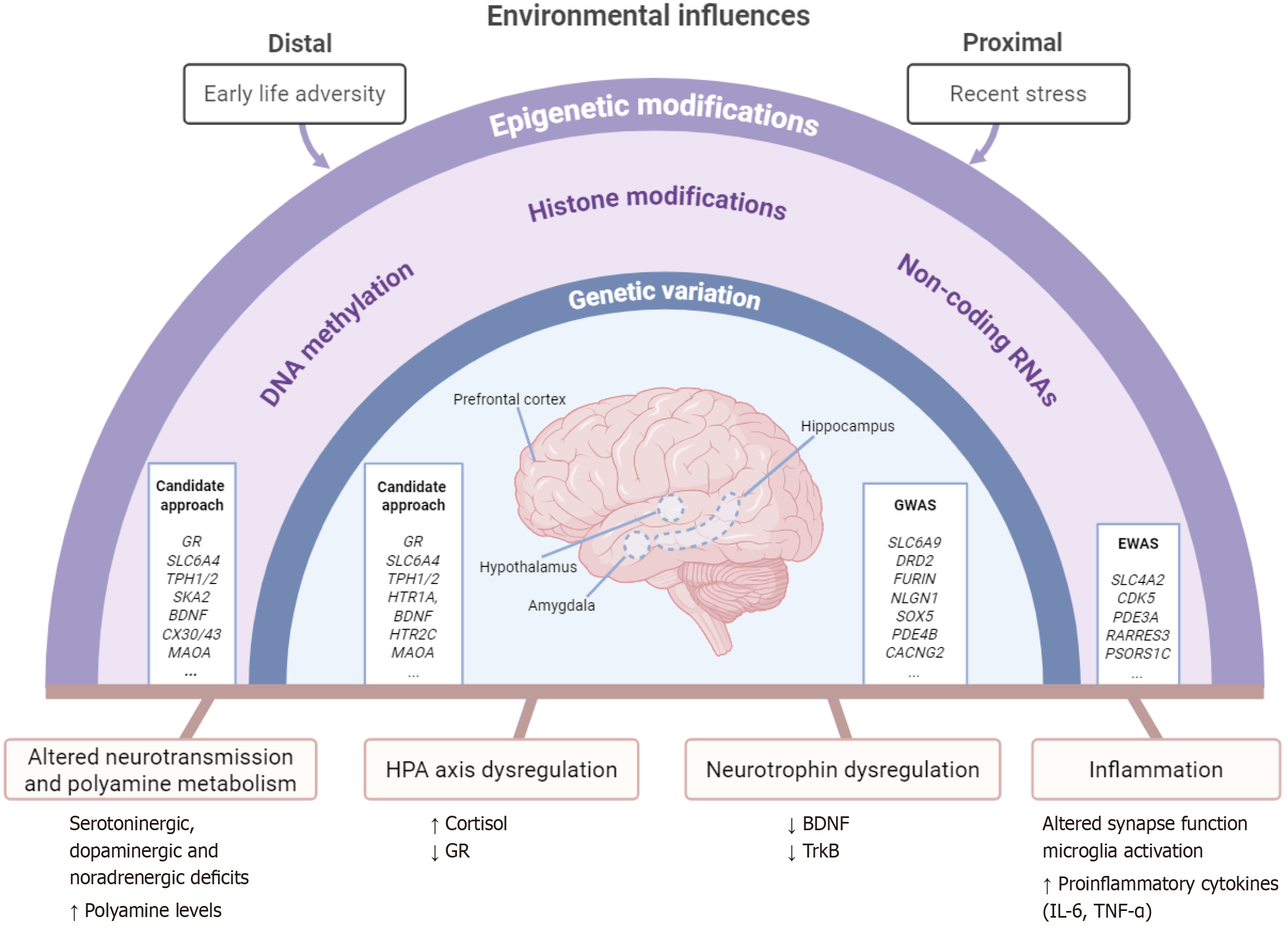
The stress-diathesis model describes suicide as a result of an interaction between environmental stressors and a susceptibility (diathesis), independent of psychiatric disorders[6]. While many molecular and physiological changes have been linked to suicidality, including inflammation, glial dysfunction and disruptions in neuroplasticity, neurotransmission, the hypothalamic-pituitary-adrenal (HPA) axis, and neurotrophic signaling, the biological correlates of this diathesis are still not fully understood[7]. Figure 1 summarizes environmental, genetic, and epigenetic influences on the pathophysiological changes associated with suicide.
Suicidal behavior exerts a heavy emotional and financial toll on society. However, suicide prevention efforts remain inadequate[8]. Various factors, including stigma, insufficient prediction power of clinical assessments, as well as the lack of reliable biomarkers of suicide risk, present challenges in implementing effective interventions[3,9]. Furthermore, suicidality research faces many obstacles, including the translational limitations of animal models[10], unavailability of in vivo brain sampling in humans, the high prevalence of psychiatric comorbidities accompanying suicidality, and limited access to patients’ past medical and personal history, which contributes to variabilities in study designs[11]. Nevertheless, technological advancements have made it possible to investigate various aspects of the neurobiological background of suicidal behavior, which may provide tools for improving suicide prevention practices[2].
While computationally demanding, the multi-omics approach has enabled simultaneous investigation of multiple potential biomarkers[12,13]. Promising targets have emerged in immune response and inflammation pathways, both peripherally and centrally[13]. In many cases, the mechanistic roles of putative biomarkers remain poorly understood[9]. The rapidly growing research on epigenetics has great potential to advance our understanding of suicidality, as it may provide a link between genetic predisposition and environmental influences[4]. While rarely examined in combination, the intersection of epigenetic regulation and immune system dysfunction may represent a critical dimension of suicidality that warrants further study. Epigenetic modifications may regulate immune system responses, while dysregulated immune responses may feedback to alter epigenetic marks, influencing suicide diathesis.
In this review, we critically examine current research on epigenetic and immune alterations in suicidality, covering pre-clinical and clinical studies. We aimed to highlight the evidence for the interplay between epigenetic modifications and (neuro)inflammation. Furthermore, we assessed the relevance for biomarker discovery and future therapeutic implementation, while also describing the pitfalls of past research and providing recommendations for future research.
Assessing an individual's suicide risk is challenging, as suicidal behavior is etiologically heterogeneous and is determined by biological characteristics, comorbid psychiatric diagnoses, personality traits, as well as economic, sociodemographic, and environmental influences[4]. Various models explaining suicidal behavior commonly classify risk factors into distal and proximal and, in some cases, developmental[4]. Distal factors, such as family history, early life adversity (ELA), and genotype, are thought to increase predisposition; proximal factors, which include recent life events, current psychiatric disorders, substance use, and the availability of means, are viewed as precipitants or triggers[7].
The genetic contribution to suicidality was first suggested by epidemiologic studies of twins, families[14], and adoptees[15]. Many studies associated the familial aggregation of impulsive-aggressive personality traits with an increased suicide risk[16,17]. The heritability of suicidal behavior was estimated at approximately 40%, independent of familial clustering of psychiatric disorders[18]. Early genetic research into suicide neurobiology utilized candidate gene associations that targeted neurotransmitter systems, most notably the serotonergic and, to a lesser extent, the dopaminergic and noradrenergic systems. While these studies identified variations in tryptophan hydroxylase 1 and serotonin transporter gene[19,20] as contributors to suicide risk, it was clear that suicidality is a polygenic condition, and this approach proved insufficient in determining its etiology.
The development of high-throughput genomic technologies led to the emergence of an unbiased approach[21]. With the ability to simultaneously examine numerous genetic variants in large cohorts, many genome-wide association studies (GWAS) have identified novel loci associated with suicide risk[22-24], further supporting the estimated heritability of suicidality[2]. These loci are often intergenic, associated with inflammatory pathways and the immune response, as well as with cell interactions, neurodevelopment, and synaptic plasticity[25]. While glucocorticoids, whose levels are regulated by the HPA axis, exert immunomodulatory functions in peripheral tissues and in the brain, the target candidate approach rarely focused directly on immunology-related targets[26]. GWAS results thus provided a new perspective on the role of immunity in neuropsychiatric disorders, suggesting that the genetic variability in immune and inflammatory pathways may present a vulnerability factor for suicidality[27].
The largest GWAS meta-analysis to date comprised 22 cohorts of various ancestries and connected 12 risk loci to suicidal behavior[28]. These loci were brain-enriched and overlapped with antidepressant targets[28]. However, due to modest loci effect size, the polygenic risk scores obtained by GWAS currently have low predictive power, and therefore lack clinical utility[29]. Furthermore, suicide and/or suicidal behavior were rarely examined independent of one another or independent of psychiatric disorders, which may have contributed to the scarcity of significant findings and inconsistent results of these studies[25,30,31]. While the genetics of suicide death and suicide attempt appear to strongly overlap, a locus mapped to neuroligin-1, involved in synapse function, may be specific to suicide[32]. Expanding GWAS study results with other modalities, such as measuring expression of genes surrounding the loci of interest, may prove beneficial in elucidating the functional role of these variants in suicidality[33]. Regarding the immune alterations in suicidality, combining GWAS with epigenetic approaches, such as genome-wide DNA methylation studies[34], is likely to provide insight into the mechanisms by which environmental exposures, including ELA, exert their effects on immune and stress-response genes[35]. Findings on the heritability of suicidal behavior underscore the need to further investigate the immunological dimension of suicidality, including the differences that may distinguish it from various neuropsychiatric conditions[36].
As implied by extensive evidence from genetic and epigenetic studies, many neuropsychological disorders, including depression and suicidality, are connected to alterations in the innate and adaptive immune systems, both peripherally and centrally[37-40]. Recently, a bidirectional relationship has been proposed between inflammation and depression in which both conditions exacerbate one another[41]. The ability of inflammation to trigger depressive symptoms was already identified in the 1990s through observations of patients receiving immunotherapy. As an adverse effect of interferon (IFN) treatment, up to 30%-70% increased risk of depression was observed[42], which did not always subside with treatment discontinuation. The effect of IFN on mood disorders was linked to its stimulation of pro-inflammatory cytokine production, with subsequent tryptophan depletion, excess reactive oxygen species formation and HPA axis dysregulation[43]. Various genetic variants were associated with IFN treatment-induced depression, including variants in genes coding for tryptophan-degrading enzyme, serotonin receptor[44], and glucocorticoid receptor (GR)[45]. The incidence of comorbid depression is elevated in patients with autoimmune disorders and other diseases associated with chronic inflammation[46], and increased CD4+/CD8+ T cell ratio was linked to depression[47]. However, increased anti-inflammatory processes have also been reported[40]. These findings led to further inquiries about the relationship between depression and inflammation, including the effects of cytokines that are specific for the central nervous system (CNS). It has been proposed that ELA and psychosocial stress may induce inflammatory responses, which alter microglial function and synaptic remodeling, thus influencing the predisposition to suicidal behavior[37].
Some studies suggest that the relationship between inflammation and depression is sex-specific[48], and the association between depression and elevated inflammatory marker levels seems to be stronger in females than males[49]. However, with the majority of postmortem suicidality research focusing solely on male subjects, these differences are likely underexplored. While women are twice as likely to develop depression compared to men[50] and more frequently display suicidal ideation (SI) and attempt suicide, men more often die due to suicide[51]. As early as neonatal de
Cytokines are small signaling proteins that are involved in cell-cell communication. They are produced and secreted by different cell types but are particularly important for immune cells, which use cytokines as messenger molecules. There are over 300 different cytokines, which are divided into chemokines, lymphokines, IFNs and growth factors. They can have pro-inflammatory or anti-inflammatory effects, and their action can promote or inhibit inflammatory reactions. Even very low concentrations of cytokines can have strong effects[57]. They act as messengers over shorter distances by binding to a cell surface receptor, which triggers signaling pathway cascades within the cell. A single type of cytokine can bind to different receptors, and different types of cytokines can also bind to the same type of receptor. When the same type of cytokine binds to different types of receptors, each binding event can lead to a different effect depending on the receptor type. Cytokine function is therefore complex and contributes to the maintenance of homeostasis[58].
In the context of neuropsychiatry, various brain regions, including the hypothalamus, hippocampus and amygdala, both react to and produce cytokines[37], which can also act as messengers between the peripheral and central immune systems[59]. The brain is separated from the peripheral immune system by the blood-brain barrier (BBB), which normally prevents peripheral immune cells from entering the brain under physiological conditions. Additional neuroimmune response mechanisms have evolved in the brain that are based on glial cells and have partially taken over the role of immune cells in the brain. It is not yet fully understood to what extent cytokines can enter the brain from other parts of the body, but there are known examples of cytokines that can cross the BBB (either due to increased BBB permeability or specialized transporters)[60].
Impaired cytokine function is associated with several mental and neurological disorders such as depression, bipolar disorder, schizophrenia and Alzheimer's disease, as well as suicidality[61]. Polymorphisms in interleukin genes are associated with completed suicide[62]. Notably, increased levels of pro-inflammatory cytokines, especially tumor necrosis factor α (TNF-α) and interleukin-6 (IL-6), have been consistently linked to depression in human studies[4,63,64]. Regarding suicidality, the inflammatory markers most strongly associated with increased risk are IL-6 and C-reactive protein (CRP), which were both elevated in the plasma of suicide attempters[65]. Plasma IL-6 is also associated with increased impulsivity and aggression, which are behavioral risk factors for suicidality[66].
Inflammatory cytokines are released by innate immune cells, including macrophages and microglia, typically in response to the binding of pathogen-associated molecules (e.g., lipopolysaccharide) to toll-like receptors (TLRs) and subsequent activation of the nuclear factor kappa B pathway[67]. IL-1β, a key cytokine that stimulates the production of other pro-inflammatory cytokines, also requires activation by the inflammasome, which is induced by extracellular ATP accumulation, tissue damage[37], and accumulation of protein aggregates[68]. Cytokines are also associated with neurotransmitter function, as they affect neurotransmitter synthesis, release and reuptake, thereby significantly shaping communication pathways in the brain. They are involved in the regulation of dopamine and glutamate[69]. Excessive cytokine signaling in the brain promotes corticotropin-releasing hormone (CRH) release and may have deleterious effects on neurogenesis, memory formation, circadian rhythms[37] and brain-derived neurotrophic factor (BDNF) signaling[70]. Furthermore, cytokine-induced increase in indoleamine 2,3-dioxygenase activity shifts tryptophan metabolism from serotonin production towards the kynurenine pathway, which increases the levels of the neurotoxin quinolinic acid and lowers serotonin levels[69,71]. Alterations in kyreunine metabolism in cerebrospinal fluid (CSF) of post-mortem brains and plasma from living subjects have been associated with depression and suicide[72-74].
Morphological and functional alterations in glial cells have also been implicated in suicidality[37]. Glial and neuronal cells can produce cytokines and they also have receptors on their surfaces to which cytokines can bind[75]. Aside from their role in immunity, microglia contribute to the regulation of BBB, neurodevelopment and synapse remodeling, and are therefore involved in the processes of learning and memory[76]. Upon activation, microglia alter their shape and start releasing inflammatory mediators, which can be either pro- or anti-inflammatory[77]. Peripheral inflammation can trigger neuroinflammation via HPA axis activation or T-cell effects on glia[78]. Serum markers of neuroinflammation, including neuron specific enolase and glial fibrillary acidic protein, have been associated with suicidality[79]. Some studies have documented increased levels of activated microglia in post-mortem brain of depressed suicide completers compared to controls[80,81].
Studies investigating genetic predisposition to cytokine-related genes are scarce. Kaushik et al[82] analyzed five single nucleotide polymorphisms (SNPs) in the IL-1β, IL-4, IL-6 and IL-10 genes in a study group of Indian suicide completers and controls (n = 490). Significant differences between the two groups were observed for genetic variation in IL-1β (rs16944), IL-4 (rs2070874), and IL-6 (rs1800795)[82]. Noroozi et al[62] genotyped three SNPs within the IL-8 gene, which codes for an interleukin with neuroprotective function, in individuals who attempted suicide, suicide completers, and healthy controls. The T allele of rs4073 was significantly associated with suicide attempts, and specific IL-8 haplotypes showed distinct associations with method and severity of suicidal behavior[62]. Focusing on gene expression, Rengasamy et al[83] aimed to differentiate between suicide attempters, suicide ideators, and healthy controls based on cytokine gene expression. While they found a significant association between the severity of depression and expression of IL-10 and TNF-α, such results were not identified for the severity of SI[83].
A larger amount of data is provided by studies that examined cytokines as biomarker molecules, accessible in both living and post-mortem samples (Table 1)[84-93]. Most of the studies described in Table 1 use a targeted approach, examining selected cytokines previously associated with neuroinflammation and mental/neurological disorders. As blood is a relatively accessible tissue, the studies predominantly investigated cytokine concentration in peripheral blood as the sample of choice. Only a few studies use CSF, which is a more invasive procedure for the patient. Brain tissue is only available for post-mortem studies. Suicidality is a complex phenomenon often associated with other mental disorders, which also have an impact on study participants. Table 1 shows that the groups studied are quite heterogeneous and exhibit various mental disorders in combination with suicidality. A distinction must be made for the term suicidality, as its meaning can range from SI, behavior, attempt to completed suicide. With such complex behaviors, we can also expect differences in the time course of the behavior, which could also likely affect the temporal course of the cytokines present and measured. This may represent one of the possibilities for study results that are sometimes difficult to compare. Examples of such cytokines are the pro-inflammatory IL-2 and the anti-inflammatory IL-10. Studies of both cytokines show mixed results, with some researchers describing decreased levels[84-86] and others reporting increased levels[87,88]. For example, Tian et al[87] observed increased IL-2 Levels, while Yang et al[84] and Hoprekstad et al[85] observed decreased IL-2 Levels. Looking more closely, multiple differences between studies can be observed. Tian et al[87] examined first-episode drug-naïve MDD patients, while Hoprekstad et al[85] specifically observed schizophrenia spectrum disorder patients on antipsychotic therapy. Differences can also be observed in the severity or definition of suicidality of the studied patients: SI in the past week, Tian et al[87] vs low suicide risk - mild suicide risk - moderate suicide risk - severe suicide risk, Yang et al[84] vs low - high risk, Hoprekstad et al[85].
| Studied biomarker | Tissue | Subjects | Number | Method | Main findings | Ref. |
| IL-1β, IL-2, IL-6, IL-8, IL-10, IFN-γ, TNF-α, CRP, CXCL-1, CCL2 | Blood | MDD patients and healthy controls | n = 260 | ELISA | Association between suicide risk and increased levels of IL-6, CRP, TNF-α, CXCL-2, and IFN-γ. Association between suicide risk and decreased levels of IL-2 and IL-8 | Yang et al[84], 2024 |
| CRP, TNF-α, IL-2, IL-6 | Blood | MDD patients with severe or mild suicidal symptoms, healthy controls | n = 179 | ELISA | Association between severe suicidal symptoms and increased levels of CRP and TNF-α | Chen et al[90], 2024 |
| IL-1β, IL-2, IL-6, IL-8, IL-10, TNF-α | Blood | First-episode drug-naïve MDD patients: With suicidal ideation and without suicidal ideation | n = 88 | Luminex | Association between suicidal ideation and increased levels of IL-2 | Tian et al[87], 2024 |
| IL-1β, IL-2, IL-4, IL-6, IL-10, IFN-γ, TNF-α | Blood | Schizophrenia spectrum disorder patients: Suicidality low risk and suicidality low risk | n = 141 | Luminex | Association between suicidality and decreased levels of IL-2 and IL-10 | Hoprekstad et al[85], 2024 |
| TNF-α, IL-1β, IL-6 | Blood | BD patients with or without suicide attempt, healthy controls | n = 64 | Luminex | Association between suicidal behavior and increased levels of IL-6 | Jiang et al[91], 2022 |
| IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12p70, IL-13, IFN-γ, TNF-α | Blood | Mood and anxiety disorders patients | n = 66 | V-PLEX | Association between suicidal risk and increased levels of IL-6. Association between suicidal risk and decreased levels of IL-8 | Keaton et al[92], 2019 |
| IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12P70, IL-17 A, IL-17 F, IL-22, TNF-α, TNF-β, IFN-γ | Blood | Depression disorder patients with or without suicidal behavior | n = 82 | Flow cytometry | Association between suicidal ideation and increased levels of TNF-α | Liu et al[89], 2024 |
| 40 chemokines (Bio-Plex Pro™ Human Chemokine 40-Plex Panel) | Brain (dorsolateral prefrontal cortex) | Suicide completers and control | n = 39 | Bio-Plex | Association between completed suicide and decreased levels of CCL1, CCL8, CCL13, CCL15, CCL17, CCL19, CCL20, CXCL11, and IL-10. Association between completed suicide and increased levels of IL-16 | Shinko et al[86], 2020 |
| IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-13, TNF-α VEGF | Blood, CSF | Suicide death subjects and non-suicidal death subjects | n = 65 | Luminex | Association between suicide and increased levels of plasma IL-6 and IL-10. Association between suicide and decreased levels of CSF VEGF | Kaushik et al[88], 2025 |
| IL-1β, IL-6, IL-8, TNF-α | CSF | Suicide attempters and healthy control subjects | n = 110 | ELISA | Association between suicide attempt/Level of suicide violence method and increased levels of IL-6 | Lindqvist et al[93], 2009 |
Looking more broadly, the occurrence and representation of cytokines can also be influenced by pharmacological treatment. Studies suggest that antidepressant use can reduce cytokine levels, while some antidepressants (such as mirtazapine) increase cytokine production[94]. Cytokine expression is also controlled by the biological circadian rhythm - this can be observed by disease severity or symptoms in some chronic inflammatory diseases. This indicates that the time of day when the sample is taken may potentially affect cytokine levels[95]. In addition, numerous environmental, socio-psychological and biological factors are also associated with changes in cytokines. All of these factors further increase the non-specificity of differences in cytokines across subjects[94].
Nevertheless, what does appear to be frequently observed are increased levels of pro-inflammatory cytokines such as IL-6, CRP and TNF. IL-6, which shows the most conclusive results (Table 1), is a major mediator of the acute phase reaction and acts directly on cells to synthesize CRP, complement components, and other acute phase proteins. Excessive IL-6 activation has also been observed in other chronic inflammatory diseases[96]. A disrupted balance between pro- and anti-inflammatory cytokines influences HPA dysregulation, as cytokines can affect the hypothalamus, stimulating the HPA, releasing CRH, adrenocorticotropic hormone and glucocorticoids[89]. Additionally, cytokines affect neurotransmission, altering the levels of both neurotransporters and neurotransmitters[97], and dysregulate neurogenesis[98].
As suicidality studies per se are relatively scarce, it is worth examining studies on depression, as they are repeatedly studied together. Coexistence of MDD and systemic immune activation (changes in inflammatory markers levels, immune cell count and antibody titers) has already been shown in several studies. Köhler et al[64] performed the most extensive meta-analysis of 82 studies on depression, cytokine and chemokine levels, and determined increased levels of IL-6, TNF, IL-10, sIL-2, C-C motif ligand (CCL) 2, IL-13, IL-18, IL-12, IL-1 receptor antagonist, and soluble TNF receptor 2, and decreased levels of IFN-γ in MDD patients compared to healthy controls. When studying the effect of antidepressant treatment on base levels of inflammatory biomarkers, it was found that antidepressants can decrease IL-6, IL-10, TNF-α and CCL2 Levels[99,100]. While these findings hold implications for future therapeutic opportunities, deeper investigation into the regulatory mechanisms of (neuro)inflammation in depression and suicide is needed.
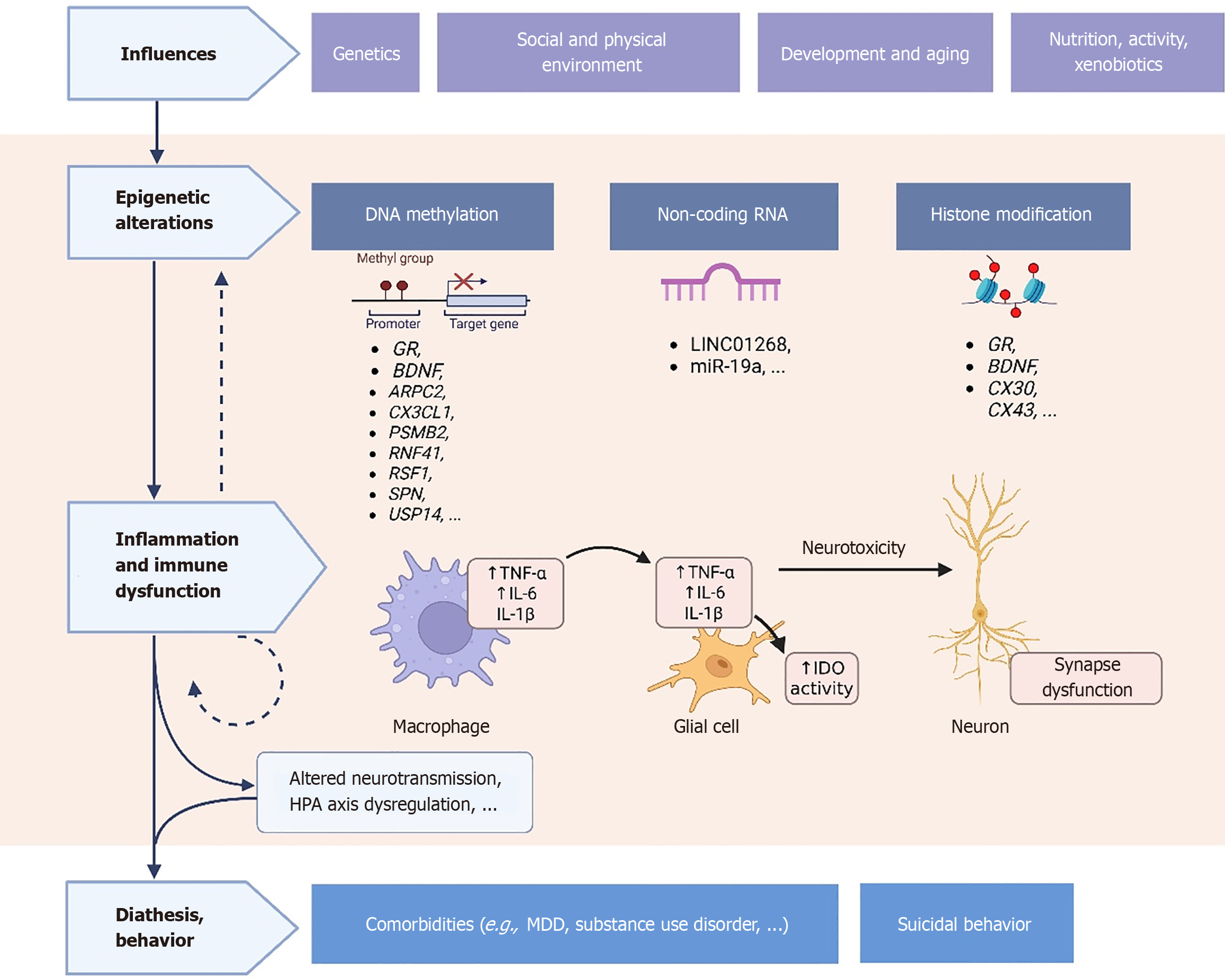
As genetic characterization could not provide a comprehensive understanding of the neurobiological basis for suicide diathesis, including the contribution of immunity and inflammation towards suicide risk, research has expanded towards other molecular factors, including epigenetic processes[2]. Epigenetic alterations are defined as heritable DNA modifications that regulate gene function and do not involve nucleotide sequence changes[101]. Environmental signals, including social environment, can trigger epigenetic responses, which include DNA methylation, histone modifications and non-coding RNA (ncRNA)-mediated changes of gene activity[101]. In suicidality, epigenetic modulation has been proposed to mediate the connection between genetic predisposition and environmental influences, most notably ELA[35], and may be involved both in distal (stable epigenetic modifications) and proximal (dynamic epigenetic modifications) factors[34]. While epigenetic changes may dysregulate immune system function, inflammation can in turn induce further epigenetic alterations, influencing the vulnerability to psychiatric disorders. For example, increased gastrointestinal permeability and subsequent alterations in microbiota-derived metabolites with immunomodulatory functions may influence brain neurogenesis and contribute to depression through epigenetic modifications[102]. Figure 2 represents the proposed interplay between different types of epigenetic alterations and the immune system, with its consequent contribution to suicide risk.
Most epigenetic studies in suicidality have focused on DNA methylation, which is usually viewed as a stable epigenetic mark and thus reflective of long-term effects of environmental influences[34]. To date, various studies have been conducted on post-mortem brain tissue or blood, utilizing both candidate gene and -omics approaches[103-112], and in some cases multi-omics[113]. Common candidate genes include those involved in the HPA axis, neurotransmission and neurotrophin signaling[34]. In a study that did not account for the subjects’ history of ELA, differences in promotor methylation of the BDNF gene were found in Wernicke’s area of suicide completers compared to controls[114]. Hypermethylation of BDNF and the glucocorticoid receptor gene (NR3C1) promoters has been linked to increased risk of depression[115]. In the hippocampus and prefrontal cortex of adolescent suicide completers, a correlation between altered DNA methylation and decreased GR expression was found, as well as altered expression of methylating and de
To date, few studies have examined posttranslational histone modifications in connection to suicidality. Histones are proteins involved in chromatin structure and gene transcription regulation[121]. Post-translational modifications of histone tails influence the level of chromatin compaction, and thus, the accessibility of DNA for transcriptional machinery. Specific histone modification patterns are associated with different genomic regions: Dimethylation and trimethylation of histone 3 Lysine 4 (H3K4) is associated with active promoters, while dimethylation and trimethylation of H3K9 and H3K27 are associated with repressed promoters[7]. Epigenetic regulation of BDNF via histone modifications has been implicated in rodent models of depression[122].
In humans, studies focusing on specific genes have connected histone modifications in post-mortem brain samples to suicidality, with a various degree of success. In the orbitofrontal cortex, increased H3K27 trimethylation in suicide completers corresponded to decreased expression of the astrocyte-expressed tropomycin receptor kinase B gene (TrkB.T1), which functions as a BDNF receptor[123]. Disturbances in astrocytic networks were connected to increased H3K9me3 in connexin 30 (CX30) and 43 (CX43) promoters in the prefrontal cortex[124]. In the frontal cortex and hippocampus of suicide completers, increased H3K27me2 and decreased H3K9/14ac were linked to deceased BDNF levels[125], which are associated with a proinflammatory profile[126]. Alterations in polyamine metabolism have been implicated in suicide neurobiology[127] and the overexpression of OAZ1, a gene involved in the polyamine system, was linked to increased H3K4me3 in its promoter region in the prefrontal cortex of suicide completers[121]. A recent study that utilized chromatin immunoprecipitation followed by high-throughput DNA sequencing demonstrated decreased H3K14ac levels in the hippocampus of suicide completers compared to controls, which associated with the differential expression of ADORA2A, B4GALT2 and MMP14[128]; ADORA2A has been implicated in mood disorders[129], MMP14 in inflammation[130], while B4GALT was associated with suicide in blood proteomics[131].
NcRNAs are a heterogenous group of functional RNA molecules that are not translated into proteins but participate in post-transcriptional regulation of gene expression[132]. In neuropsychiatry, the most examined ncRNA type are mi
LncRNAs are RNA molecules that are over 200 base pairs long, abundant in the brain and less evolutionarily conserved than other ncRNAs. They have been implicated in numerous inflammatory diseases[134], which makes them interesting candidates for suicide research. However, they are difficult to study due to the variety of effects they can exert on gene expression and are therefore poorly examined[7]. Downregulation of certain lncRNAs has been found in peripheral blood mononuclear cells of MDD patients compared to controls, with the presence of SI and previous suicide attempt(s) associated with greater alterations[135]. Furthermore, the use of violent methods for suicide has been associated with alterations in the brain expression levels of the human-specific lncRNA LINC01268, which may regulate genes associated with immunity[136], and LOC285758, which maps next to MARCKS, a gene upregulated in suicide. Recently, a novel lncRNA regulating MAOA gene expression was characterized in the brain of suicide completers, and its upregulation was associated with increased impulsivity and aggression[137,138]. In another study, altered expression of 23 LncRNAs, as well as their overlapping or antisense protein coding genes, was found in the rostral anterior cingulate cortex of depressed suicide completers compared to controls. Many of their corresponding genes were associated with the innate immune response, specifically IFN signaling[139].
MiRNAs are noncoding RNA molecules approximately 22 nucleotides long that regulate numerous cellular functions via binding to partially complementary mRNA targets and repressing translation[140]. Seventy percent of the total human miRNAs are estimated to be neuronal miRNAs, which makes them interesting targets for neuropsychiatric research[141]. Proper miRNA function is important for neurodevelopment, and altered miRNA expression has been associated with neurodegenerative and psychiatric disorders including depression, (neuro)inflammation and suicidal behavior[142,143]. Importantly, animal model studies have shown that ELA may contribute to suicidal behavior in adults by inducing abnormal miRNA expression[144], which may be detected in brain and blood[145,146].
Most suicidality studies have focused on miRNA expression in the prefrontal cortex of post-mortem brain, where global downregulation of miRNA was reported in suicide completers compared to controls[147], as well as increased expression of certain miRNAs, including those presumably involved in serotonin receptor 5-HT1AR signaling pathway[148] and TNF-α upregulation[149]. Downregulation of the miRNA miR-218 that targets deleted in colorectal cancer, which as
Some studies have reported similar miRNA expression patterns in blood and brain tissue, which is promising because the detection of miRNAs in peripheral samples has greater potential for clinical implementation[154]. Downregulation of miR-1202, a brain-enriched miRNA that targets the metabotropic glutamate receptor 4 transcript, was found both in the prefrontal cortex of suicide completers with MDD and in blood samples from MDD patients, and may be predictive of antidepressant response[155]. In living subjects, expression of miRNA in the blood of patients with treatment-resistant depression was compared to that of controls, and the downregulation of two miRNAs, let-7b and let-7c, was identified as a possible biomarker[156]. In the peripheral blood of depressed patients, increased levels of TNF-α associated with a pro-inflammatory miRNA profile (upregulated miR-342, and downregulated miR-146a and miR-155) compared to controls[157]. Another recent study associated elevated serum levels of miR-30a, miR-30e, and miR-200a in suicidal MDD patients with acute suicide risk compared to MDD patients without acute suicide risk. Early growth response protein 1 was indicated as a transcriptional regulator of these miRNAs[158]. Furthermore, there is evidence that some miRNA levels are responsive to antidepressant treatment, which makes them an attractive biomarker for monitoring treatment success[159]. However, due to miRNA pleiotropy, selection of miRNAs that could function as suicide biomarkers is difficult[160], and in silico analysis algorithms are being developed for selection of brain-expressed miRNAs that target suicidality- and inflammation-associated genes[161].
To further understand the mechanisms by which the epigenetic-immune interaction contributes to suicidality, we must consider the context of stress and trauma as predisposing factors. When CNS homeostasis is disrupted, there is a neuroinflammatory response, which is appropriate as long as it does not cause a chronic inflammatory state[162]. As the immune system is closely linked to the stress response, it is not surprising that suicidality research has correlated dysregulated stress responses with increased inflammation[163].
In the context of psychoneuroimmunology, the most often studied cytokines are IL-6, TNF, IL-1b, and INFs, associated with inflammation, and IL-10 which is associated with an anti-inflammatory effect[40]. Several of these were changed in suicide completers and attempters[164], as demonstrated in Table 1, and in most cases, they have also been linked to stress. Preclinical studies have demonstrated increased brain IL-1β levels in response to stress[165], as well as the reversal of depressive-like behaviors after administration of IL-1β antagonists in rodents[166]. Furthermore, the activation of the hippocampal melacontin receptor MC4R reversed the deleterious effects of IL-1β on cognitive function[167]. In suicidal adolescents, ELA was associated with altered expression of glucocorticoid response elements in peripheral mononuclear cells[168].
Stress can be perceived as acute psychosocial stress, chronic social stress or ELA, which have all been linked to suicidality. Acute stress is associated with glucocorticoids and microglia activation, which leads to cytokine synthesis. In the case of ELA, the abnormal cortisol stress response and elevated levels of pro-inflammatory biomarkers such as CRP, IL-6 and TNF-α are present in subjects with chronic stress, GR resistance is observed. This resistance can also be induced by inflammatory cytokines, and elevated levels of IL-6 have been associated with chronic stress[163]. Events like traumatic brain injury (TBI) are also linked to immune system activation and are associated with microglia activation and increased inflammatory factor levels, such as IL-1 and TNF-α[169]. In a large study of TBI patients, the odds of dying due to suicide were three times higher in TBI patients than controls[170]. TNF-α levels positively correlated to SI[171].
Exposure to ELA has been robustly associated with an increased risk of maladaptive behavioral phenotypes[172], psychiatric disorders[173] and suicidality[174], as well as with structural and functional alterations in the brain[175] and HPA and immune system dysregulation[36]. The long-lasting effects of stress, including the influence of early-life social environment on stress reactivity in adulthood, has been extensively demonstrated in animal studies[176-179] and translated to humans[177,180-182].
As previously noted, poor adaptation to stress and dysfunction of the stress axis can be associated with changes in the regulation of certain genes. The candidate genes of the HPA axis, SKA2, FKBP5, NR3C1 and CRH genes, are of particular interest; of the latter, SKA2 has been linked to lifetime trauma history. The most evident results, however, are available for GR, which has an anti-inflammatory effect after cortisol binding. For this gene, lower expression[183] was associated with stress in rodents and humans due to increased DNA methylation[178]. In adult rats, insufficient maternal care promoted exaggerated stress response by inducing methylation changes in the NR3C1 promoter in the hippocampus[177]. Furthermore, altered histone acetylation and transcription factor binding to the NR3C1 promoter was observed[178]. Most importantly, however, the DNA methylation patterns could be modified by exposure to a different en
Similar effects of ELA and trauma have been observed in humans[180,181]: In suicide completers with a history of childhood abuse, decreased hippocampal expression and increased methylation in the NR3C1 promoter was reported[182,187]. Furthermore, intrauterine stress induced by maternal stress can dysregulate the HPA axis[188], activate the immune system and may present a risk factor for the development of depression[189]. A similar picture of increased DNA methylation levels of NR3C1 in the hippocampus was also observed in those who died by suicide when compared with the DNA methylation status of a control group of those who died from other causes. The effect was especially strong in those who died by suicide and who had experienced childhood abuse, suggesting that DNA methylation changes acquired in childhood remained active in adulthood. They also detected decreased levels of NR3C1 expression, which is consistent with expectations, as increased levels of DNA methylation often lead to reduced gene expression[187,190]. An epigenome-wide association study identified the interactions between methylation patterns in the SKA2 gene, whose product interacts with GR, and traumatic experiences as predictive of lifetime suicide risk[111]. This result was confirmed by candidate gene studies[191,192].
While it is well established that HPA axis dysfunction accompanies prolonged inflammation, there is little research on epigenetic alterations targeting immune-related genes in association with ELA and psychopathologies. Aside from stress-related genes, epigenetic mechanisms also regulate the expression of cytokines and TLRs, and environmental stressors such as ELA may contribute to a pro-inflammatory phenotype. Reduced IL-6 promoter methylation has been associated with childhood trauma in a cohort of African American men[193]. Lam et al[194] examined peripheral blood DNA methylation patterns in over 14000 gene promoters from individuals with variations in early-life socioeconomic status. DNA methylation patterns were connected to leukocyte composition, demographics, cortisol levels, early-life poverty and psychosocial stress in adulthood. Interestingly, DNA methylation patterns were associated with inflammatory responses of peripheral blood mononuclear cells to ex vivo TLR stimulation, while the relationship to gene expression was weaker, which may suggest that epigenetic changes induced by ELA may create a poised transcriptional state, priming the immune system for altered response to future environmental challenges[194].
The risk for psychiatric disorders, including depression, is increased by stressful events, particularly ELA, and it can be further associated with a more damaging disease course and with poor treatment outcome and suicidality[173], which is closely intertwined with epigenetic alterations and (neuro)inflammation. In a study by Miller and Cole[195], healthy adolescents with a high risk of depression were followed for 2.5 years. Neuroinflammation became present in subjects exposed to ELA, such as parental separation, low socioeconomic status, and familial psychopathology. Elevated levels of IL-6 and CRP were present in their blood. On the other hand, there were no changes in these biomarkers among the subjects not experiencing adversities[195]. When looking at DNA methylation, several genes that are associated with inflammatory and immune processes, such as LTA, GFI1, ARID5B, CD52, TNFSF13, SLFN13, LST1, TNFRSF1A, CCRL2, and IL-32, as well as NF receptor and TNF signaling pathway, were found differentially methylated in ELA patients that received trauma-focused psychotherapy. These findings supported the hypothesis of inflammatory/immune pa
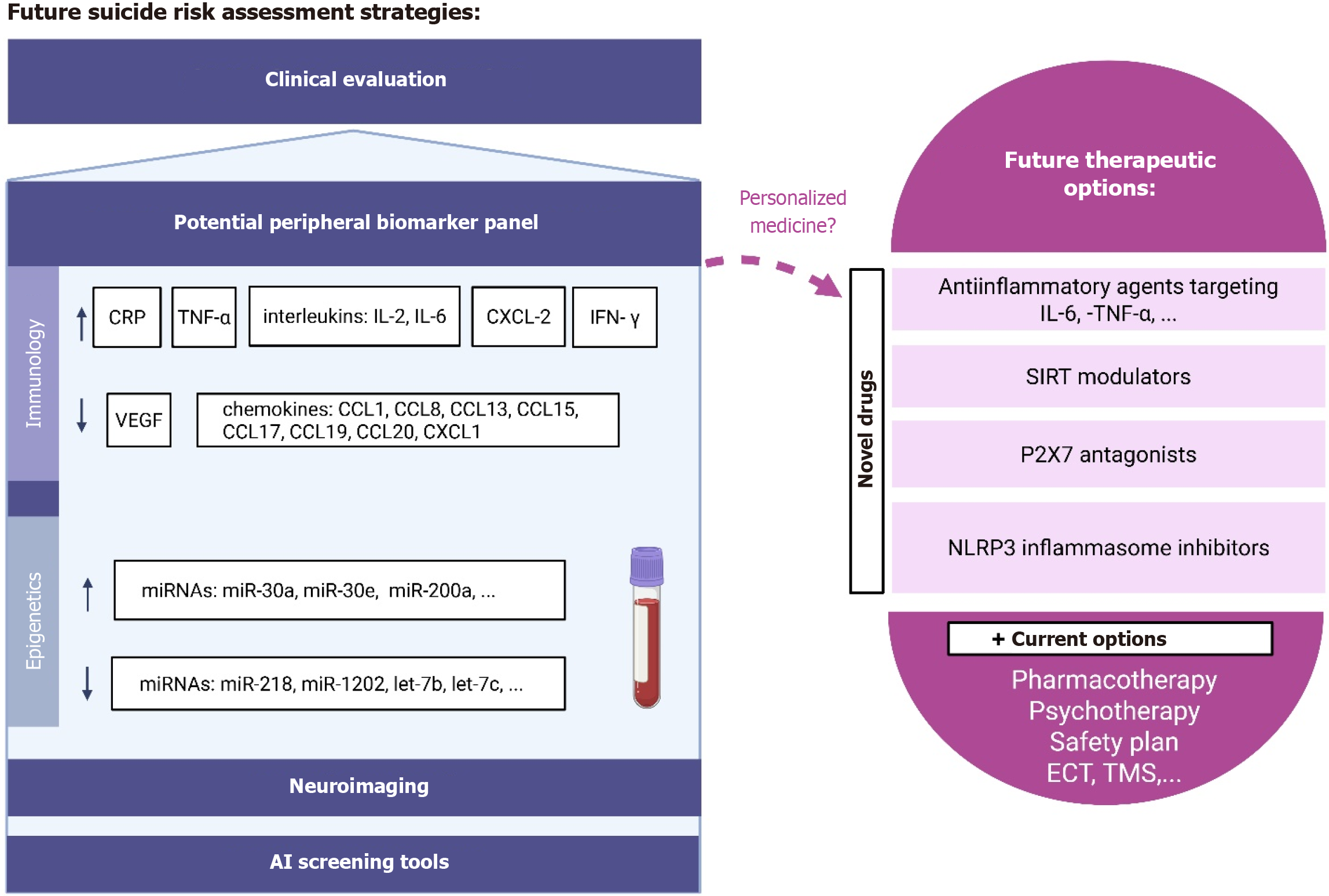
With increasing insight into the neurobiological background of suicidality, new diagnostic and therapeutic avenues are possible. Identification of objective biological markers associated with suicide risk is particularly promising, as it could lead to the development of minimally invasive risk stratification tools and increase the prediction value of clinical assessments[4]. However, no such quantifiable indicators are currently available[198]. Possible targets include the molecular components of neurotransmitter systems, the HPA axis, the immune response and inflammation, as well as endocannabinoids and lipids[198]. As some of these targets may be affected on different levels (genetic and epigenetic alterations, as well as altered mRNA or protein concentrations), multi-omic research has the potential to facilitate biomarker discovery[13]. Epigenetic markers of immune response are particularly compelling candidates, as they reflect the interplay between genetic vulnerability and environmental exposures[103], and are important in mediating the influence of ELA and stress on suicide risk[199]. Furthermore, inflammation may induce epigenetic changes[200], which has therapeutic implications, especially for antidepressant non-responders. Figure 3 highlights the potential future applications of peripheral immunology and/or epigenetics- related biomarkers in assessing suicide risk and predicting effective treatment.
Regarding epigenetic biomarkers of neuroinflammation in the context of suicidality, ncRNAs are especially promising due to their accessibility in peripheral body fluids[141,198], high specificity for cell of origin and possibly being in comparison to novel protein biomarkers, cheaper to detect[201]. In a randomized clinical trial, some miRNAs in combination with clinical testing, have predicted worsening of SI in patients receiving antidepressant treatment[202]. However, miRNA detection has certain limitations: Levels of neuronal miRNAs in peripheral tissues largely reflect the integrity of the BBB, which may be altered in various inflammatory conditions. Furthermore, it is difficult to identify the freely circulating miRNAs’ tissue of origin. Therefore, EV-derived miRNAs may be advantageous because they can be traced to their (e.g., neuronal) tissue of origin via surface protein markers[203]. Exosomal miRNAs are secreted physiologically in response to stress[204] and as previously mentioned, their CSF levels have been linked to suicide in post-mortem studies[153]. However, their levels appear to be low in peripheral fluids, requiring sensitive detection methods[205], which raises the question of cost-effectiveness. Further studies examining EV-derived miRNA in peripheral fluids of living subjects with suicide risk, in combination with inflammation marker assessment, are necessary to evaluate their biomarker potential.
Many studies have also examined protein expression levels as potential suicidality biomarkers[9]. For example, an analysis of matched post-mortem blood and brain samples of depressed patients associated the differential expression of four genes with suicide completion, of which two (decreased SOX9 and increased PER3 levels) showed the same pattern of change in both tissues[206]. A recent study investigated the genetic basis of SI in patients with MDD via a weighted gene co-expression network analysis on peripheral blood RNA-sequencing data. These results suggest that gene expression regulation, immune response, and inflammation may be linked to SI among MDD patients, while innate immunity, mitochondrial function, and neurodegeneration are more broadly associated with MDD[207].
Peripheral markers of inflammation are consistently being reported as possible biomarkers of suicide risk[208]. Elevated neutrophil-to-lymphocyte ratio, primarily driven by higher neutrophil counts, has recently emerged as a potential marker of suicidality[209]. A recent 2-year follow-up study evaluated 39 molecular analytes in peripheral blood of patients with mood disorders. Elevated CRP and plasma thrombospondin-2 were linked to recent suicide attempts, while low plasma serotonin levels were linked to SI. Thrombospondin-1 and platelet-derived growth factor were predictive of future suicidal events. Interestingly, there was no overlap between markers of past and future suicidal events, suggesting the possibility of distinct molecular components influencing SI development of and attempt[210]. Abnormal expression of inflammation-related genes in monocytes was found in MDD patients, and patients with upregulated gene profiles had a higher prevalence of ELA and high suicide risk. These alterations were not observed in controls with a history of ELA, suggesting that inflammation specifically occurs in MDD-predisposed individuals exposed to trauma[211].
Despite the increasing attempts at elucidating molecular changes associated with suicide risk, including epigenetic and immune alterations, there are currently many drawbacks that limit the utility of these findings. Among studies, there are pronounced differences in cohorts and methodologies, as well as the criteria for assessing suicidal behavior (ideation, attempts, differences in means used for suicide)[9]. The influence of sex, age, psychiatric and other comorbidities, medications, socioeconomic environment and other confounding factors may contribute to the heterogeneity of participants and the lack of replicable results by obscuring true biomarker effects[4]. For example, some potential biomarkers seem to be sex-specific, such as SAT1 expression in blood[206]. It is challenging to distinguish molecular aspects unique to suicidality from the influence of disorders commonly associated with it, including MDD and substance abuse[4], as most suicide completers have comorbid psychiatric diagnoses[2]. Similarly, many studies suffer from small sample sizes and therefore low statistical power. Furthermore, most potential biomarkers lack accuracy and most notably specificity; for example, blood levels of CRP and IL-6 are associated with suicide risk[65], however, they are also elevated in numerous other disorders and show high variability among individuals[212]. While peripheral tissues and fluids, including blood, saliva and hair, may present possible biomarker sources[213], most suicidality research is performed on post-mortem brain, which undergoes rapid degradation, and variations in the post-mortem interval present an important confounding factor[11]. It is also not yet clear how post-mortem brain findings translate to other post-mortem or living tissues, such as venous blood which is the preferred clinical sample due to ease of access, or even among different brain regions[11]. Furthermore, post-mortem samples do not provide longitudinal insight into the development of suicidality, or real-time assessment of suicide risk. This is problematic, as identifying suicide risk factors that characterize the early stages in the progression from predisposition towards suicidal behavior is crucial for timely intervention[2]. To overcome at least some of these challenges, future research should prioritize large, well-characterized cohorts, standardized methodologies and, if possible, longitudinal designs. Complementing post-mortem studies with approaches that target living participants, and integrating multi-omics approaches with clinical and behavioral data, would enhance replicability and predictive value, which would facilitate the discovery and clinical implementation of reliable suicidality biomarkers.
Due to the heterogeneity of suicidality, characterization of distinct biological subtypes (e.g., the impulsive and planned behavior subtypes) would be an important future goal in suicidality research, and continuing with integrative studies is important for the realization of personalized medicine in the psychiatric field[214]. Statistical and machine learning approaches may aid in subtype identification[215]. Controlling for confounding factors is a challenging but important step in planning future research to increase reproducibility. With technological advancement, it is likely that a panel of multiple biomarkers, including immune and epigenetic alterations, in concert with clinical assessment and computerized adaptive tests, will be used for risk stratification in the future[4]. Combining clinical and biochemical modalities with brain imaging studies such as functional magnetic resonance imaging would further contribute to improved risk assessment, however, cost- and time-effectiveness may present barriers to their clinical implementation[216].
Despite methodological challenges in suicidality research and the current lack of reliable biomarkers, the growing insights into the inflammatory background of suicidality are driving the development of novel therapeutic strategies for alleviating suicide risk and treating suicide-associated pathologies, most notably, MDD. High inflammation and insufficient anti-inflammatory response have been associated with resistance to antidepressant treatment in MDD[40]. In randomized clinical trials, various anti-inflammatory agents, including omega 3 fatty acids, non-steroidal anti-inflammatory drugs (NSAIDs), minocycline and statins have exhibited limited efficacy in alleviating depressive symptoms, with differences in patient subpopulations[217]. Furthermore, glial dysfunction and neuroinflammation are being targeted via purine receptor antagonists, which also mediate IL-1β release[218], and inflammasome inhibitors[219]. Among more promising potential anti-inflammatory agents for depression treatment are sirtuin modulators[220], which have recently shown promise in Phase 1 clinical trial[221]. While anti-inflammatory drugs are increasingly being explored in the treatment of depression[38], it is more difficult to assess their potential in suicide prevention, as subjects with increased suicide risk are rarely included in clinical trials[222]. Still, a reduction in SI was noted in patients with depression that were taking NSAIDs[223]. Certain medications used in depression treatment, e.g., ketamine, also exert an anti-inflammatory effect, especially in patients with higher inflammation levels, and a rapid reduction of suicidal thoughts has been observed in patients treated with ketamine[224]. Glutamatergic system modulation and the activation of the neuroprotective aspects of the kynurenine pathway are thought to contribute to the anti-inflammatory effects of ketamine. Therefore, clinical trials are examining the potential of dual (antidepressant and anti-inflammatory) therapy in treatment-resistant depression[225]. It is likely that future therapeutic strategies will utilize a similar, but increasingly personalized antidepressant and anti-inflammatory combination approach.
Despite the growing evidence of epigenetic dysregulation in suicidality, therapeutic approaches targeting epigenetic modifications have not yet been examined due to their effects on global gene expression and safety concerns. Currently, the use of demethylating agents and histone deacetylase inhibitors is limited to hematologic malignancies[226]. However, animal studies have suggested that histone deacetylase inhibitors may have antidepressant-like effects in animals, most likely by upregulating BDNF expression[227]. Furthermore, epigenetic alterations may help in predicting treatment efficacy[228]. Non-pharmacological approaches, including psychotherapy[197] and lifestyle interventions such as mindfulness, exercise and diet, may also exert anti-inflammatory effects via epigenetic mechanisms, highlighting the importance of lifestyle medicine in treatment and prevention of depressive symptoms[229]. In MDD patients, aerobic exercise was shown to influence the concentration and miRNA cargo of EVs, stimulating their neuroprotective effect[230].
Recent advancements in artificial intelligence (AI) methods have shown promise for improving suicidality risk assessment[231]. However, current machine learning approaches are limited, as they do not include neurobiological and neuroimaging data[232]. The development of AI-driven models harnessing large-scale datasets, including multi-omics data, may facilitate the discovery of complex relationships between molecular alterations and suicidal behaviors[233]. In future research, a collaborative, interdisciplinary approach is required to adequately address the heterogeneity of suicidality, as well as to ensure adherence to ethical standards in utilizing large datasets[231].
Suicidality is a complex and multifactorial phenomenon. There is increasing evidence of the involvement of epigenetic and immune alterations in the development of suicide risk, particularly in mediating the influences of stress and early-life adversity. While the interplay between epigenetic regulation and (neuro)inflammatory processes, including cytokine imbalance and glial dysfunction, remains underexplored in neuropsychiatric research, there is compelling evidence that advancing our understanding of this field may enable the development of personalized approaches to suicide risk assessment, monitoring and treatment of psychiatric comorbidities. Continued interdisciplinary research integrating multi-omic approaches, applying well-characterized cohorts, is essential to ensure reproducibility of findings.
The authors would like to thank Dr. John Hancock for critical appraisal and scientific English editing of the manuscript.
| 1. | World Health Organization. Suicide worldwide in 2019: global health estimates. Global health estimates. Geneva: World Health Organization; 2021. |
| 2. | Abou Chahla MN, Khalil MI, Comai S, Brundin L, Erhardt S, Guillemin GJ. Biological Factors Underpinning Suicidal Behaviour: An Update. Brain Sci. 2023;13:505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 3. | Baldessarini RJ. Epidemiology of suicide: recent developments. Epidemiol Psychiatr Sci. 2019;29:e71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 4. | Turecki G, Brent DA, Gunnell D, O'Connor RC, Oquendo MA, Pirkis J, Stanley BH. Suicide and suicide risk. Nat Rev Dis Primers. 2019;5:74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 509] [Article Influence: 84.8] [Reference Citation Analysis (0)] |
| 5. | O'Connor RC, Nock MK. The psychology of suicidal behaviour. Lancet Psychiatry. 2014;1:73-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 610] [Cited by in RCA: 656] [Article Influence: 59.6] [Reference Citation Analysis (0)] |
| 6. | van Heeringen K, Mann JJ. The neurobiology of suicide. Lancet Psychiatry. 2014;1:63-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 350] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 7. | Turecki G. The molecular bases of the suicidal brain. Nat Rev Neurosci. 2014;15:802-816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 172] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 8. | Oquendo MA, Porras-Segovia A. Barriers for the Research, Prevention, and Treatment of Suicidal Behavior. Curr Top Behav Neurosci. 2020;46:25-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 9. | Costanza A, D'Orta I, Perroud N, Burkhardt S, Malafosse A, Mangin P, La Harpe R. Neurobiology of suicide: do biomarkers exist? Int J Legal Med. 2014;128:73-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 10. | Gould TD, Georgiou P, Brenner LA, Brundin L, Can A, Courtet P, Donaldson ZR, Dwivedi Y, Guillaume S, Gottesman II, Kanekar S, Lowry CA, Renshaw PF, Rujescu D, Smith EG, Turecki G, Zanos P, Zarate CA Jr, Zunszain PA, Postolache TT. Animal models to improve our understanding and treatment of suicidal behavior. Transl Psychiatry. 2017;7:e1092. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 11. | Pandey GN, Dwivedi Y. What can post-mortem studies tell us about the pathoetiology of suicide? Future Neurol. 2010;5:701-720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 12. | Pereira CA, Reis-de-Oliveira G, Pierone BC, Martins-de-Souza D, Kaster MP. Depicting the molecular features of suicidal behavior: a review from an "omics" perspective. Psychiatry Res. 2024;332:115682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 13. | Kouter K, Videtic Paska A. 'Omics' of suicidal behaviour: A path to personalised psychiatry. World J Psychiatry. 2021;11:774-790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 3] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Voracek M, Loibl LM. Genetics of suicide: a systematic review of twin studies. Wien Klin Wochenschr. 2007;119:463-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 183] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 15. | von Borczyskowski A, Lindblad F, Vinnerljung B, Reintjes R, Hjern A. Familial factors and suicide: an adoption study in a Swedish National Cohort. Psychol Med. 2011;41:749-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 16. | McGirr A, Alda M, Séguin M, Cabot S, Lesage A, Turecki G. Familial aggregation of suicide explained by cluster B traits: a three-group family study of suicide controlling for major depressive disorder. Am J Psychiatry. 2009;166:1124-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 112] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 17. | Diaconu G, Turecki G. Family history of suicidal behavior predicts impulsive-aggressive behavior levels in psychiatric outpatients. J Affect Disord. 2009;113:172-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 18. | Fu Q, Heath AC, Bucholz KK, Nelson EC, Glowinski AL, Goldberg J, Lyons MJ, Tsuang MT, Jacob T, True MR, Eisen SA. A twin study of genetic and environmental influences on suicidality in men. Psychol Med. 2002;32:11-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 160] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 19. | Bondy B, Buettner A, Zill P. Genetics of suicide. Mol Psychiatry. 2006;11:336-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 194] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 20. | Antypa N, Serretti A, Rujescu D. Serotonergic genes and suicide: a systematic review. Eur Neuropsychopharmacol. 2013;23:1125-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 88] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 21. | Uffelmann E, Huang QQ, Munung NS, de Vries J, Okada Y, Martin AR, Martin HC, Lappalainen T, Posthuma D. Genome-wide association studies. Nat Rev Methods Primers. 2021;1:59. [DOI] [Full Text] |
| 22. | Docherty AR, Shabalin AA, DiBlasi E, Monson E, Mullins N, Adkins DE, Bacanu SA, Bakian AV, Crowell S, Chen D, Darlington TM, Callor WB, Christensen ED, Gray D, Keeshin B, Klein M, Anderson JS, Jerominski L, Hayward C, Porteous DJ, McIntosh A, Li Q, Coon H. Genome-Wide Association Study of Suicide Death and Polygenic Prediction of Clinical Antecedents. Am J Psychiatry. 2020;177:917-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 23. | Strawbridge RJ, Ward J, Ferguson A, Graham N, Shaw RJ, Cullen B, Pearsall R, Lyall LM, Johnston KJA, Niedzwiedz CL, Pell JP, Mackay D, Martin JL, Lyall DM, Bailey MES, Smith DJ. Identification of novel genome-wide associations for suicidality in UK Biobank, genetic correlation with psychiatric disorders and polygenic association with completed suicide. EBioMedicine. 2019;41:517-525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 24. | Erlangsen A, Appadurai V, Wang Y, Turecki G, Mors O, Werge T, Mortensen PB, Starnawska A, Børglum AD, Schork A, Nudel R, Bækvad-Hansen M, Bybjerg-Grauholm J, Hougaard DM, Thompson WK, Nordentoft M, Agerbo E. Genetics of suicide attempts in individuals with and without mental disorders: a population-based genome-wide association study. Mol Psychiatry. 2020;25:2410-2421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 105] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 25. | Galfalvy H, Haghighi F, Hodgkinson C, Goldman D, Oquendo MA, Burke A, Huang YY, Giegling I, Rujescu D, Bureau A, Turecki G, Mann JJ. A genome-wide association study of suicidal behavior. Am J Med Genet B Neuropsychiatr Genet. 2015;168:557-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 77] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 26. | Bellavance MA, Rivest S. The HPA - Immune Axis and the Immunomodulatory Actions of Glucocorticoids in the Brain. Front Immunol. 2014;5:136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 226] [Cited by in RCA: 295] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 27. | Iakunchykova O, Leonardsen EH, Wang Y. Genetic evidence for causal effects of immune dysfunction in psychiatric disorders: where are we? Transl Psychiatry. 2024;14:63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 28. | Docherty AR, Mullins N, Ashley-Koch AE, Qin X, Coleman JRI, Shabalin A, Kang J, Murnyak B, Wendt F, Adams M, Campos AI, DiBlasi E, Fullerton JM, Kranzler HR, Bakian AV, Monson ET, Rentería ME, Walss-Bass C, Andreassen OA, Behera C, Bulik CM, Edenberg HJ, Kessler RC, Mann JJ, Nurnberger JI Jr, Pistis G, Streit F, Ursano RJ, Polimanti R, Dennis M, Garrett M, Hair L, Harvey P, Hauser ER, Hauser MA, Huffman J, Jacobson D, Madduri R, McMahon B, Oslin DW, Trafton J, Awasthi S, Berrettini WH, Bohus M, Chang X, Chen HC, Chen WJ, Christensen ED, Crow S, Duriez P, Edwards AC, Fernández-Aranda F, Galfalvy H, Gandal M, Gorwood P, Guo Y, Hafferty JD, Hakonarson H, Halmi KA, Hishimoto A, Jain S, Jamain S, Jiménez-Murcia S, Johnson C, Kaplan AS, Kaye WH, Keel PK, Kennedy JL, Kim M, Klump KL, Levey DF, Li D, Liao SC, Lieb K, Lilenfeld L, Marshall CR, Mitchell JE, Okazaki S, Otsuka I, Pinto D, Powers A, Ramoz N, Ripke S, Roepke S, Rozanov V, Scherer SW, Schmahl C, Sokolowski M, Starnawska A, Strober M, Su MH, Thornton LM, Treasure J, Ware EB, Watson HJ, Witt SH, Woodside DB, Yilmaz Z, Zillich L, Adolfsson R, Agartz I, Alda M, Alfredsson L, Appadurai V, Artigas MS, Van der Auwera S, Azevedo MH, Bass N, Bau CHD, Baune BT, Bellivier F, Berger K, Biernacka JM, Bigdeli TB, Binder EB, Boehnke M, Boks MP, Braff DL, Bryant R, Budde M, Byrne EM, Cahn W, Castelao E, Cervilla JA, Chaumette B, Corvin A, Craddock N, Djurovic S, Foo JC, Forstner AJ, Frye M, Gatt JM, Giegling I, Grabe HJ, Green MJ, Grevet EH, Grigoroiu-Serbanescu M, Gutierrez B, Guzman-Parra J, Hamshere ML, Hartmann AM, Hauser J, Heilmann-Heimbach S, Hoffmann P, Ising M, Jones I, Jones LA, Jonsson L, Kahn RS, Kelsoe JR, Kendler KS, Kloiber S, Koenen KC, Kogevinas M, Krebs MO, Landén M, Leboyer M, Lee PH, Levinson DF, Liao C, Lissowska J, Mayoral F, McElroy SL, McGrath P, McGuffin P, McQuillin A, Mehta D, Melle I, Mitchell PB, Molina E, Morken G, Nievergelt C, Nöthen MM, O'Donovan MC, Ophoff RA, Owen MJ, Pato C, Pato MT, Penninx BWJH, Potash JB, Power RA, Preisig M, Quested D, Ramos-Quiroga JA, Reif A, Ribasés M, Richarte V, Rietschel M, Rivera M, Roberts A, Roberts G, Rouleau GA, Rovaris DL, Sanders AR, Schofield PR, Schulze TG, Scott LJ, Serretti A, Shi J, Sirignano L, Sklar P, Smeland OB, Smoller JW, Sonuga-Barke EJS, Trzaskowski M, Tsuang MT, Turecki G, Vilar-Ribó L, Vincent JB, Völzke H, Walters JTR, Weickert CS, Weickert TW, Weissman MM, Williams LM, Wray NR, Zai CC, Agerbo E, Børglum AD, Breen G, Demontis D, Erlangsen A, Gelernter J, Glatt SJ, Hougaard DM, Hwu HG, Kuo PH, Lewis CM, Li QS, Liu CM, Martin NG, McIntosh AM, Medland SE, Mors O, Nordentoft M, Olsen CM, Porteous D, Smith DJ, Stahl EA, Stein MB, Wasserman D, Werge T, Whiteman DC, Willour V; VA Million Veteran Program (MVP); MVP Suicide Exemplar Workgroup; Suicide Working Group of the Psychiatric Genomics Consortium; Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium; Bipolar Disorder Working Group of the Psychiatric Genomics Consortium; Schizophrenia Working Group of the Psychiatric Genomics Consortium; Eating Disorder Working Group of the Psychiatric Genomics Consortium; German Borderline Genomics Consortium, Coon H, Beckham JC, Kimbrel NA, Ruderfer DM. GWAS Meta-Analysis of Suicide Attempt: Identification of 12 Genome-Wide Significant Loci and Implication of Genetic Risks for Specific Health Factors. Am J Psychiatry. 2023;180:723-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 65] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 29. | DiBlasi E, Kang J, Docherty AR. Genetic contributions to suicidal thoughts and behaviors. Psychol Med. 2021;51:2148-2155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 30. | Coon H, Darlington TM, DiBlasi E, Callor WB, Ferris E, Fraser A, Yu Z, William N, Das SC, Crowell SE, Chen D, Anderson JS, Klein M, Jerominski L, Cannon D, Shabalin A, Docherty A, Williams M, Smith KR, Keeshin B, Bakian AV, Christensen E, Li QS, Camp NJ, Gray D. Genome-wide significant regions in 43 Utah high-risk families implicate multiple genes involved in risk for completed suicide. Mol Psychiatry. 2020;25:3077-3090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 31. | Otsuka I, Akiyama M, Shirakawa O, Okazaki S, Momozawa Y, Kamatani Y, Izumi T, Numata S, Takahashi M, Boku S, Sora I, Yamamoto K, Ueno Y, Toda T, Kubo M, Hishimoto A. Genome-wide association studies identify polygenic effects for completed suicide in the Japanese population. Neuropsychopharmacology. 2019;44:2119-2124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 32. | Li QS, Shabalin AA, DiBlasi E, Gopal S, Canuso CM; FinnGen, International Suicide Genetics Consortium, Palotie A, Drevets WC, Docherty AR, Coon H. Genome-wide association study meta-analysis of suicide death and suicidal behavior. Mol Psychiatry. 2023;28:891-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 33. | Fiori L, Chawla A, Nagy C, Turecki G. Characterization of a suicidal behavior GWAS locus on chromosome 7. Eur Neuropsychopharmacol. 2024;87:19. [DOI] [Full Text] |
| 34. | Turecki G. Epigenetics and suicidal behavior research pathways. Am J Prev Med. 2014;47:S144-S151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 35. | Roy B, Dwivedi Y. Understanding epigenetic architecture of suicide neurobiology: A critical perspective. Neurosci Biobehav Rev. 2017;72:10-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 36. | Segerstrom SC, Miller GE. Psychological stress and the human immune system: a meta-analytic study of 30 years of inquiry. Psychol Bull. 2004;130:601-630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2019] [Cited by in RCA: 1774] [Article Influence: 84.5] [Reference Citation Analysis (0)] |
| 37. | Jones KA, Thomsen C. The role of the innate immune system in psychiatric disorders. Mol Cell Neurosci. 2013;53:52-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 209] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 38. | Bai Y, Cai Y, Chang D, Li D, Huo X, Zhu T. Immunotherapy for depression: Recent insights and future targets. Pharmacol Ther. 2024;257:108624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 39. | Medina-Rodriguez EM, Lowell JA, Worthen RJ, Syed SA, Beurel E. Involvement of Innate and Adaptive Immune Systems Alterations in the Pathophysiology and Treatment of Depression. Front Neurosci. 2018;12:547. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 40. | Beurel E, Toups M, Nemeroff CB. The Bidirectional Relationship of Depression and Inflammation: Double Trouble. Neuron. 2020;107:234-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 1257] [Article Influence: 251.4] [Reference Citation Analysis (0)] |
| 41. | Chan VKY, Luo H, Chan SSM, Lau CS, Yeung WWY, Peng K, Tong X, Lam MPS, Wong ICK, Li X. Treatment-resistant depression and risk of autoimmune diseases: evidence from a population-based cohort and nested case-control study. Transl Psychiatry. 2023;13:76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 42. | Pinto EF, Andrade C. Interferon-Related Depression: A Primer on Mechanisms, Treatment, and Prevention of a Common Clinical Problem. Curr Neuropharmacol. 2016;14:743-748. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 43. | Chiu WC, Su YP, Su KP, Chen PC. Recurrence of depressive disorders after interferon-induced depression. Transl Psychiatry. 2017;7:e1026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 44. | Lemonde S, Turecki G, Bakish D, Du L, Hrdina PD, Bown CD, Sequeira A, Kushwaha N, Morris SJ, Basak A, Ou XM, Albert PR. Impaired repression at a 5-hydroxytryptamine 1A receptor gene polymorphism associated with major depression and suicide. J Neurosci. 2003;23:8788-8799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 477] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 45. | Udina M, Navinés R, Egmond E, Oriolo G, Langohr K, Gimenez D, Valdés M, Gómez-Gil E, Grande I, Gratacós M, Kapczinski F, Artigas F, Vieta E, Solà R, Martín-Santos R. Glucocorticoid Receptors, Brain-Derived Neurotrophic Factor, Serotonin and Dopamine Neurotransmission are Associated with Interferon-Induced Depression. Int J Neuropsychopharmacol. 2016;19:pyv135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 46. | Pryce CR, Fontana A. Depression in Autoimmune Diseases. Curr Top Behav Neurosci. 2017;31:139-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 47. | Wang F, Zhu D, Cao L, Wang S, Tong Y, Xie F, Zhang X, Su P, Wang G. Peripheral CD4(+) T helper lymphocytes alterations in major depressive disorder: A systematic review and meta-analysis. Neuroscience. 2024;555:145-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 48. | Birur B, Amrock EM, Shelton RC, Li L. Sex Differences in the Peripheral Immune System in Patients with Depression. Front Psychiatry. 2017;8:108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 49. | Jarkas DA, Villeneuve AH, Daneshmend AZB, Villeneuve PJ, McQuaid RJ. Sex differences in the inflammation-depression link: A systematic review and meta-analysis. Brain Behav Immun. 2024;121:257-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 50. | Salk RH, Hyde JS, Abramson LY. Gender differences in depression in representative national samples: Meta-analyses of diagnoses and symptoms. Psychol Bull. 2017;143:783-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1293] [Cited by in RCA: 1424] [Article Influence: 178.0] [Reference Citation Analysis (0)] |
| 51. | Carretta RF, McKee SA, Rhee TG. Gender Differences in Risks of Suicide and Suicidal Behaviors in the USA: A Narrative Review. Curr Psychiatry Rep. 2023;25:809-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 52. | Hodes GE, Walker DM, Labonté B, Nestler EJ, Russo SJ. Understanding the epigenetic basis of sex differences in depression. J Neurosci Res. 2017;95:692-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 53. | Issler O, van der Zee YY, Ramakrishnan A, Xia S, Zinsmaier AK, Tan C, Li W, Browne CJ, Walker DM, Salery M, Torres-Berrío A, Futamura R, Duffy JE, Labonte B, Girgenti MJ, Tamminga CA, Dupree JL, Dong Y, Murrough JW, Shen L, Nestler EJ. The long noncoding RNA FEDORA is a cell type- and sex-specific regulator of depression. Sci Adv. 2022;8:eabn9494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 54. | Zhang J, Jia CX, Wang LL. Testosterone differs between suicide attempters and community controls in men and women of China. Physiol Behav. 2015;141:40-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 55. | Wang W, Liang W, Sun C, Liu S. Sex Differences in Depression: Insights from Multimodal Gray Matter Morphology and Peripheral Inflammatory Factors. Int J Mol Sci. 2024;25:13412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 56. | Cabrera-Mendoza B, Fresno C, Monroy-Jaramillo N, Fries GR, Walss-Bass C, Glahn DC, Ostrosky-Wegman P, Mendoza-Morales RC, García-Dolores F, Díaz-Otañez CE, González-Sáenz EE, Genis-Mendoza AD, Martínez-Magaña JJ, Romero-Pimentel AL, Flores G, Vázquez-Roque RA, Nicolini H. Sex differences in brain gene expression among suicide completers. J Affect Disord. 2020;267:67-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 57. | Becher B, Spath S, Goverman J. Cytokine networks in neuroinflammation. Nat Rev Immunol. 2017;17:49-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 486] [Article Influence: 54.0] [Reference Citation Analysis (0)] |
| 58. | Kany S, Vollrath JT, Relja B. Cytokines in Inflammatory Disease. Int J Mol Sci. 2019;20:6008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 536] [Cited by in RCA: 1154] [Article Influence: 192.3] [Reference Citation Analysis (0)] |
| 59. | Tian L, Ma L, Kaarela T, Li Z. Neuroimmune crosstalk in the central nervous system and its significance for neurological diseases. J Neuroinflammation. 2012;9:155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 143] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 60. | Pan W, Kastin AJ. TNFalpha transport across the blood-brain barrier is abolished in receptor knockout mice. Exp Neurol. 2002;174:193-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 161] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 61. | Stuart MJ, Singhal G, Baune BT. Systematic Review of the Neurobiological Relevance of Chemokines to Psychiatric Disorders. Front Cell Neurosci. 2015;9:357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 123] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 62. | Noroozi R, Omrani MD, Ayatollahi SA, Sayad A, Ata A, Fallah H, Taheri M, Ghafouri-Fard S. Interleukin (IL)-8 polymorphisms contribute in suicide behavior. Cytokine. 2018;111:28-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 63. | Hiles SA, Baker AL, de Malmanche T, Attia J. A meta-analysis of differences in IL-6 and IL-10 between people with and without depression: exploring the causes of heterogeneity. Brain Behav Immun. 2012;26:1180-1188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 218] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 64. | Köhler CA, Freitas TH, Maes M, de Andrade NQ, Liu CS, Fernandes BS, Stubbs B, Solmi M, Veronese N, Herrmann N, Raison CL, Miller BJ, Lanctôt KL, Carvalho AF. Peripheral cytokine and chemokine alterations in depression: a meta-analysis of 82 studies. Acta Psychiatr Scand. 2017;135:373-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 700] [Cited by in RCA: 1002] [Article Influence: 125.3] [Reference Citation Analysis (0)] |
| 65. | Neupane SP, Daray FM, Ballard ED, Galfalvy H, Itzhaky L, Segev A, Shelef A, Tene O, Rizk MM, Mann JJ, Zalsman G. Immune-related biomarkers and suicidal behaviors: A meta-analysis. Eur Neuropsychopharmacol. 2023;75:15-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 28] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 66. | Isung J, Aeinehband S, Mobarrez F, Nordström P, Runeson B, Asberg M, Piehl F, Jokinen J. High interleukin-6 and impulsivity: determining the role of endophenotypes in attempted suicide. Transl Psychiatry. 2014;4:e470. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 63] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 67. | Liu T, Zhang L, Joo D, Sun SC. NF-κB signaling in inflammation. Signal Transduct Target Ther. 2017;2:17023-17023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2830] [Cited by in RCA: 5562] [Article Influence: 695.3] [Reference Citation Analysis (0)] |
| 68. | Voet S, Srinivasan S, Lamkanfi M, van Loo G. Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol Med. 2019;11:e10248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 530] [Article Influence: 106.0] [Reference Citation Analysis (0)] |
| 69. | Miller AH, Haroon E, Raison CL, Felger JC. Cytokine targets in the brain: impact on neurotransmitters and neurocircuits. Depress Anxiety. 2013;30:297-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 487] [Cited by in RCA: 585] [Article Influence: 48.8] [Reference Citation Analysis (0)] |
| 70. | Tong L, Balazs R, Soiampornkul R, Thangnipon W, Cotman CW. Interleukin-1 beta impairs brain derived neurotrophic factor-induced signal transduction. Neurobiol Aging. 2008;29:1380-1393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 177] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 71. | Clark SM, Pocivavsek A, Nicholson JD, Notarangelo FM, Langenberg P, McMahon RP, Kleinman JE, Hyde TM, Stiller J, Postolache TT, Schwarcz R, Tonelli LH. Reduced kynurenine pathway metabolism and cytokine expression in the prefrontal cortex of depressed individuals. J Psychiatry Neurosci. 2016;41:386-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 72. | Brown SJ, Christofides K, Weissleder C, Huang XF, Shannon Weickert C, Lim CK, Newell KA. Sex- and suicide-specific alterations in the kynurenine pathway in the anterior cingulate cortex in major depression. Neuropsychopharmacology. 2024;49:584-592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 73. | Trepci A, Sellgren CM, Pålsson E, Brundin L, Khanlarkhani N, Schwieler L, Landén M, Erhardt S. Central levels of tryptophan metabolites in subjects with bipolar disorder. Eur Neuropsychopharmacol. 2021;43:52-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 74. | Messaoud A, Rym M, Wahiba D, Neffati F, Najjar MF, Gobbi G, Manchia M, Valtorta F, Lotfi G, Comai S. Investigation of the Relationship among Cortisol, Pro-inflammatory Cytokines, and the Degradation of Tryptophan into Kynurenine in Patients with Major Depression and Suicidal Behavior. Curr Top Med Chem. 2022;22:2119-2125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 75. | Bourgognon JM, Cavanagh J. The role of cytokines in modulating learning and memory and brain plasticity. Brain Neurosci Adv. 2020;4:2398212820979802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 141] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 76. | Verkhratsky A, Butt A, Li B, Illes P, Zorec R, Semyanov A, Tang Y, Sofroniew MV. Astrocytes in human central nervous system diseases: a frontier for new therapies. Signal Transduct Target Ther. 2023;8:396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 154] [Article Influence: 77.0] [Reference Citation Analysis (0)] |
| 77. | Baharikhoob P, Kolla NJ. Microglial Dysregulation and Suicidality: A Stress-Diathesis Perspective. Front Psychiatry. 2020;11:781. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 78. | West PK, McCorkindale AN, Guennewig B, Ashhurst TM, Viengkhou B, Hayashida E, Jung SR, Butovsky O, Campbell IL, Hofer MJ. The cytokines interleukin-6 and interferon-α induce distinct microglia phenotypes. J Neuroinflammation. 2022;19:96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 79. | Bagheri M, Ghaneialvar H, Oshnokhah M, Salari S. GFAP and Neuron Specific Enolase (NSE) in the Serum of Suicide Attempters. Med J Islam Repub Iran. 2022;36:103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 80. | Torres-Platas SG, Cruceanu C, Chen GG, Turecki G, Mechawar N. Evidence for increased microglial priming and macrophage recruitment in the dorsal anterior cingulate white matter of depressed suicides. Brain Behav Immun. 2014;42:50-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 397] [Article Influence: 36.1] [Reference Citation Analysis (0)] |
| 81. | Schnieder TP, Trencevska I, Rosoklija G, Stankov A, Mann JJ, Smiley J, Dwork AJ. Microglia of prefrontal white matter in suicide. J Neuropathol Exp Neurol. 2014;73:880-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 131] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 82. | Kaushik R, Nayak B, Patra BN, Docherty AR, Shabalin A, Behera C. Cytokine gene polymorphisms and suicide risk in an Indian ancestral population: A case-control study. World J Biol Psychiatry. 2024;25:547-559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 83. | Rengasamy M, Zhong Y, Marsland A, Chen K, Douaihy A, Brent D, Melhem NM. Signaling networks in inflammatory pathways and risk for suicidal behavior. Brain Behav Immun Health. 2020;7:100122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 84. | Yang Y, Gu K, Li J. Relationship between serum inflammatory cytokines and suicide risk in patients with major depressive disorder. Front Psychiatry. 2024;15:1422511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 85. | Hoprekstad GE, Skrede S, Bartz-Johannessen C, Joa I, Reitan SK, Steen VM, Torsvik A, Johnsen E, Kroken RA, Rettenbacher M. Association between cytokines and suicidality in patients with psychosis: A multicentre longitudinal analysis. Brain Behav Immun Health. 2024;37:100756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 86. | Shinko Y, Otsuka I, Okazaki S, Horai T, Boku S, Takahashi M, Ueno Y, Sora I, Hishimoto A. Chemokine alterations in the postmortem brains of suicide completers. J Psychiatr Res. 2020;120:29-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 87. | Tian X, Dong YQ, Yuan JY, Gao Y, Zhang CH, Li MJ, Li J. Association between peripheral plasma cytokine levels and suicidal ideation in first-episode, drug-naïve major depressive disorder. Psychoneuroendocrinology. 2024;165:107042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 88. | Kaushik R, Nayak B, Satapathy S, Bharti DR, Pandey S, Behera C. Association of peripheral and cerebral inflammatory markers with suicidal deaths in Indian population. Psychoneuroendocrinology. 2025;172:107268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 89. | Liu F, Yang Y, Fan XW, Zhang N, Wang S, Shi YJ, Hu WJ, Wang CX. Impacts of inflammatory cytokines on depression: a cohort study. BMC Psychiatry. 2024;24:195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 90. | Chen MH, Bai YM, Hsu JW, Huang KL, Tsai SJ. Proinflammatory cytokine levels, cognitive function, and suicidal symptoms of adolescents and young adults with major depressive disorder. Eur Arch Psychiatry Clin Neurosci. 2024;274:1681-1687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 91. | Jiang X, Guo Y, Jia L, Zhu Y, Sun Q, Kong L, Wu F, Tang Y. Altered Levels of Plasma Inflammatory Cytokines and White Matter Integrity in Bipolar Disorder Patients With Suicide Attempts. Front Psychiatry. 2022;13:861881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 92. | Keaton SA, Madaj ZB, Heilman P, Smart L, Grit J, Gibbons R, Postolache TT, Roaten K, Achtyes ED, Brundin L. An inflammatory profile linked to increased suicide risk. J Affect Disord. 2019;247:57-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 93. | Lindqvist D, Janelidze S, Hagell P, Erhardt S, Samuelsson M, Minthon L, Hansson O, Björkqvist M, Träskman-Bendz L, Brundin L. Interleukin-6 is elevated in the cerebrospinal fluid of suicide attempters and related to symptom severity. Biol Psychiatry. 2009;66:287-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 396] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 94. | Himmerich H, Patsalos O, Lichtblau N, Ibrahim MAA, Dalton B. Cytokine Research in Depression: Principles, Challenges, and Open Questions. Front Psychiatry. 2019;10:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 185] [Cited by in RCA: 182] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 95. | Nakao A. Temporal regulation of cytokines by the circadian clock. J Immunol Res. 2014;2014:614529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 96. | Ihan A. Osnove medicinske imunologije. 2nd ed. Ljubljana: Medicinska fakulteta, Inštitut za mikrobiologijo in imunologijo, Katedra za mikrobiologijo in imunologijo, 2020: 223. |
| 97. | Tilleux S, Hermans E. Neuroinflammation and regulation of glial glutamate uptake in neurological disorders. J Neurosci Res. 2007;85:2059-2070. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 295] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 98. | Koo JW, Russo SJ, Ferguson D, Nestler EJ, Duman RS. Nuclear factor-kappaB is a critical mediator of stress-impaired neurogenesis and depressive behavior. Proc Natl Acad Sci U S A. 2010;107:2669-2674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 401] [Cited by in RCA: 498] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 99. | Köhler CA, Freitas TH, Stubbs B, Maes M, Solmi M, Veronese N, de Andrade NQ, Morris G, Fernandes BS, Brunoni AR, Herrmann N, Raison CL, Miller BJ, Lanctôt KL, Carvalho AF. Peripheral Alterations in Cytokine and Chemokine Levels After Antidepressant Drug Treatment for Major Depressive Disorder: Systematic Review and Meta-Analysis. Mol Neurobiol. 2018;55:4195-4206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 185] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 100. | Liu JJ, Wei YB, Strawbridge R, Bao Y, Chang S, Shi L, Que J, Gadad BS, Trivedi MH, Kelsoe JR, Lu L. Peripheral cytokine levels and response to antidepressant treatment in depression: a systematic review and meta-analysis. Mol Psychiatry. 2020;25:339-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 278] [Article Influence: 55.6] [Reference Citation Analysis (0)] |
| 101. | Cavalli G, Heard E. Advances in epigenetics link genetics to the environment and disease. Nature. 2019;571:489-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 895] [Article Influence: 149.2] [Reference Citation Analysis (0)] |
| 102. | Nohesara S, Abdolmaleky HM, Zhou JR, Thiagalingam S. Microbiota-Induced Epigenetic Alterations in Depressive Disorders Are Targets for Nutritional and Probiotic Therapies. Genes (Basel). 2023;14:2217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 103. | Cheung S, Woo J, Maes MS, Zai CC. Suicide epigenetics, a review of recent progress. J Affect Disord. 2020;265:423-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 104. | Policicchio S, Washer S, Viana J, Iatrou A, Burrage J, Hannon E, Turecki G, Kaminsky Z, Mill J, Dempster EL, Murphy TM. Genome-wide DNA methylation meta-analysis in the brains of suicide completers. Transl Psychiatry. 2020;10:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 105. | Kimbrel NA, Garrett ME, Evans MK, Mellows C, Dennis MF, Hair LP, Hauser MA; VA Mid-Atlantic MIRECC Workgroup, Ashley-Koch AE, Beckham JC. Large epigenome-wide association study identifies multiple novel differentially methylated CpG sites associated with suicidal thoughts and behaviors in veterans. Front Psychiatry. 2023;14:1145375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 106. | Labonté B, Suderman M, Maussion G, Lopez JP, Navarro-Sánchez L, Yerko V, Mechawar N, Szyf M, Meaney MJ, Turecki G. Genome-wide methylation changes in the brains of suicide completers. Am J Psychiatry. 2013;170:511-520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 109] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 107. | Haghighi F, Xin Y, Chanrion B, O'Donnell AH, Ge Y, Dwork AJ, Arango V, Mann JJ. Increased DNA methylation in the suicide brain. Dialogues Clin Neurosci. 2014;16:430-438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 108. | Schneider E, El Hajj N, Müller F, Navarro B, Haaf T. Epigenetic Dysregulation in the Prefrontal Cortex of Suicide Completers. Cytogenet Genome Res. 2015;146:19-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 109. | Cabrera-Mendoza B, Martínez-Magaña JJ, Genis-Mendoza AD, Monroy-Jaramillo N, Walss-Bass C, Fries GR, García-Dolores F, López-Armenta M, Flores G, Vázquez-Roque RA, Nicolini H. Brain Gene Expression-DNA Methylation Correlation in Suicide Completers: Preliminary Results. Rev Invest Clin. 2020;73. [PubMed] |
| 110. | Nagy C, Suderman M, Yang J, Szyf M, Mechawar N, Ernst C, Turecki G. Astrocytic abnormalities and global DNA methylation patterns in depression and suicide. Mol Psychiatry. 2015;20:320-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 236] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 111. | Guintivano J, Brown T, Newcomer A, Jones M, Cox O, Maher BS, Eaton WW, Payne JL, Wilcox HC, Kaminsky ZA. Identification and replication of a combined epigenetic and genetic biomarker predicting suicide and suicidal behaviors. Am J Psychiatry. 2014;171:1287-1296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 122] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 112. | Kouter K, Zupanc T, Videtič Paska A. Genome-wide DNA methylation in suicide victims revealing impact on gene expression. J Affect Disord. 2019;253:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 113. | Zhou Y, Xiong L, Chen J, Wang Q. Integrative Analyses of scRNA-seq, Bulk mRNA-seq, and DNA Methylation Profiling in Depressed Suicide Brain Tissues. Int J Neuropsychopharmacol. 2023;26:840-855. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 114. | Keller S, Sarchiapone M, Zarrilli F, Videtic A, Ferraro A, Carli V, Sacchetti S, Lembo F, Angiolillo A, Jovanovic N, Pisanti F, Tomaiuolo R, Monticelli A, Balazic J, Roy A, Marusic A, Cocozza S, Fusco A, Bruni CB, Castaldo G, Chiariotti L. Increased BDNF promoter methylation in the Wernicke area of suicide subjects. Arch Gen Psychiatry. 2010;67:258-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 257] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 115. | Zhu JH, Bo HH, Liu BP, Jia CX. The associations between DNA methylation and depression: A systematic review and meta-analysis. J Affect Disord. 2023;327:439-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 116. | Rizavi HS, Khan OS, Zhang H, Bhaumik R, Grayson DR, Pandey GN. Methylation and expression of glucocorticoid receptor exon-1 variants and FKBP5 in teenage suicide-completers. Transl Psychiatry. 2023;13:53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 117. | Sha Q, Fu Z, Escobar Galvis ML, Madaj Z, Underwood MD, Steiner JA, Dwork A, Simpson N, Galfalvy H, Rozoklija G, Achtyes ED, Mann JJ, Brundin L. Integrative transcriptome- and DNA methylation analysis of brain tissue from the temporal pole in suicide decedents and their controls. Mol Psychiatry. 2024;29:134-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 118. | Romero-Pimentel AL, Almeida D, Muñoz-Montero S, Rangel C, Mendoza-Morales R, Gonzalez-Saenz EE, Nagy C, Chen G, Aouabed Z, Theroux JF, Turecki G, Martinez-Levy G, Walss-Bass C, Monroy-Jaramillo N, Fernández-Figueroa EA, Gómez-Cotero A, García-Dolores F, Morales-Marin ME, Nicolini H. Integrative DNA Methylation and Gene Expression Analysis in the Prefrontal Cortex of Mexicans Who Died by Suicide. Int J Neuropsychopharmacol. 2021;24:935-947. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 119. | Crawford B, Craig Z, Mansell G, White I, Smith A, Spaull S, Imm J, Hannon E, Wood A, Yaghootkar H, Ji Y; Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium, Mullins N, Lewis CM, Mill J, Murphy TM. DNA methylation and inflammation marker profiles associated with a history of depression. Hum Mol Genet. 2018;27:2840-2850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 120. | Clark SL, Hattab MW, Chan RF, Shabalin AA, Han LKM, Zhao M, Smit JH, Jansen R, Milaneschi Y, Xie LY, van Grootheest G, Penninx BWJH, Aberg KA, van den Oord EJCG. A methylation study of long-term depression risk. Mol Psychiatry. 2020;25:1334-1343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 121. | Fiori LM, Gross JA, Turecki G. Effects of histone modifications on increased expression of polyamine biosynthetic genes in suicide. Int J Neuropsychopharmacol. 2012;15:1161-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 122. | Su CL, Su CW, Hsiao YH, Gean PW. Epigenetic regulation of BDNF in the learned helplessness-induced animal model of depression. J Psychiatr Res. 2016;76:101-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 123. | Ernst C, Chen ES, Turecki G. Histone methylation and decreased expression of TrkB.T1 in orbital frontal cortex of suicide completers. Mol Psychiatry. 2009;14:830-832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 124. | Nagy C, Torres-Platas SG, Mechawar N, Turecki G. Repression of Astrocytic Connexins in Cortical and Subcortical Brain Regions and Prefrontal Enrichment of H3K9me3 in Depression and Suicide. Int J Neuropsychopharmacol. 2017;20:50-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 125. | Misztak P, Pańczyszyn-Trzewik P, Nowak G, Sowa-Kućma M. Epigenetic marks and their relationship with BDNF in the brain of suicide victims. PLoS One. 2020;15:e0239335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 126. | Porter GA, O'Connor JC. Brain-derived neurotrophic factor and inflammation in depression: Pathogenic partners in crime? World J Psychiatry. 2022;12:77-97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 48] [Cited by in RCA: 105] [Article Influence: 35.0] [Reference Citation Analysis (3)] |
| 127. | Fiori LM, Turecki G. Association of the SAT1 in/del polymorphism with suicide completion. Am J Med Genet B Neuropsychiatr Genet. 2010;153B:825-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 128. | Arčan IŠ, Kouter K, Zupanc T, Paska AV. Epigenetics and suicide: investigating altered H3K14ac unveiled differential expression in ADORA2A, B4GALT2 and MMP14. Epigenomics. 2024;16:701-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 129. | Oliveira S, Ardais AP, Bastos CR, Gazal M, Jansen K, de Mattos Souza L, da Silva RA, Kaster MP, Lara DR, Ghisleni G. Impact of genetic variations in ADORA2A gene on depression and symptoms: a cross-sectional population-based study. Purinergic Signal. 2019;15:37-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 130. | Aguirre A, Blázquez-Prieto J, Amado-Rodriguez L, López-Alonso I, Batalla-Solís E, González-López A, Sánchez-Pérez M, Mayoral-Garcia C, Gutiérrez-Fernández A, Albaiceta GM. Matrix metalloproteinase-14 triggers an anti-inflammatory proteolytic cascade in endotoxemia. J Mol Med (Berl). 2017;95:487-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 131. | Zhao H, Liu Y, Zhang X, Liao Y, Zhang H, Han X, Guo L, Fan B, Wang W, Lu C. Identifying novel proteins for suicide attempt by integrating proteomes from brain and blood with genome-wide association data. Neuropsychopharmacology. 2024;49:1255-1265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 132. | Nemeth K, Bayraktar R, Ferracin M, Calin GA. Non-coding RNAs in disease: from mechanisms to therapeutics. Nat Rev Genet. 2024;25:211-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 340] [Article Influence: 340.0] [Reference Citation Analysis (0)] |
| 133. | Yoshino Y, Dwivedi Y. Non-Coding RNAs in Psychiatric Disorders and Suicidal Behavior. Front Psychiatry. 2020;11:543893. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 134. | Zhang Y, Liu H, Niu M, Wang Y, Xu R, Guo Y, Zhang C. Roles of long noncoding RNAs in human inflammatory diseases. Cell Death Discov. 2024;10:235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 135. | Cui X, Niu W, Kong L, He M, Jiang K, Chen S, Zhong A, Li W, Lu J, Zhang L. Long noncoding RNA expression in peripheral blood mononuclear cells and suicide risk in Chinese patients with major depressive disorder. Brain Behav. 2017;7:e00711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 136. | Punzi G, Ursini G, Viscanti G, Radulescu E, Shin JH, Quarto T, Catanesi R, Blasi G, Jaffe AE, Deep-Soboslay A, Hyde TM, Kleinman JE, Bertolino A, Weinberger DR. Association of a Noncoding RNA Postmortem With Suicide by Violent Means and In Vivo With Aggressive Phenotypes. Biol Psychiatry. 2019;85:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 137. | Labonté B, Abdallah K, Maussion G, Yerko V, Yang J, Bittar T, Quessy F, Golden SA, Navarro L, Checknita D, Gigek C, Lopez JP, Neve RL, Russo SJ, Tremblay RE, Côté G, Meaney MJ, Mechawar N, Nestler EJ, Turecki G. Regulation of impulsive and aggressive behaviours by a novel lncRNA. Mol Psychiatry. 2021;26:3751-3764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 138. | Punzi G, Ursini G, Shin JH, Kleinman JE, Hyde TM, Weinberger DR. Increased expression of MARCKS in post-mortem brain of violent suicide completers is related to transcription of a long, noncoding, antisense RNA. Mol Psychiatry. 2014;19:1057-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 139. | Zhou Y, Lutz PE, Wang YC, Ragoussis J, Turecki G. Global long non-coding RNA expression in the rostral anterior cingulate cortex of depressed suicides. Transl Psychiatry. 2018;8:224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 140. | Ambros V. The functions of animal microRNAs. Nature. 2004;431:350-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7919] [Cited by in RCA: 8601] [Article Influence: 409.6] [Reference Citation Analysis (0)] |
| 141. | Slota JA, Booth SA. MicroRNAs in Neuroinflammation: Implications in Disease Pathogenesis, Biomarker Discovery and Therapeutic Applications. Noncoding RNA. 2019;5:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 168] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 142. | Cao DD, Li L, Chan WY. MicroRNAs: Key Regulators in the Central Nervous System and Their Implication in Neurological Diseases. Int J Mol Sci. 2016;17:842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 140] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 143. | Dwivedi Y. MicroRNAs in depression and suicide: Recent insights and future perspectives. J Affect Disord. 2018;240:146-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 144. | Allen L, Dwivedi Y. MicroRNA mediators of early life stress vulnerability to depression and suicidal behavior. Mol Psychiatry. 2020;25:308-320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 145. | Torres-Berrío A, Nouel D, Cuesta S, Parise EM, Restrepo-Lozano JM, Larochelle P, Nestler EJ, Flores C. MiR-218: a molecular switch and potential biomarker of susceptibility to stress. Mol Psychiatry. 2020;25:951-964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 146. | Torres-Berrío A, Morgunova A, Giroux M, Cuesta S, Nestler EJ, Flores C. miR-218 in Adolescence Predicts and Mediates Vulnerability to Stress. Biol Psychiatry. 2021;89:911-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 147. | Smalheiser NR, Lugli G, Rizavi HS, Torvik VI, Turecki G, Dwivedi Y. MicroRNA expression is down-regulated and reorganized in prefrontal cortex of depressed suicide subjects. PLoS One. 2012;7:e33201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 217] [Cited by in RCA: 256] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 148. | Gorinski N, Bijata M, Prasad S, Wirth A, Abdel Galil D, Zeug A, Bazovkina D, Kondaurova E, Kulikova E, Ilchibaeva T, Zareba-Koziol M, Papaleo F, Scheggia D, Kochlamazashvili G, Dityatev A, Smyth I, Krzystyniak A, Wlodarczyk J, Richter DW, Strekalova T, Sigrist S, Bang C, Hobuß L, Fiedler J, Thum T, Naumenko VS, Pandey G, Ponimaskin E. Attenuated palmitoylation of serotonin receptor 5-HT1A affects receptor function and contributes to depression-like behaviors. Nat Commun. 2019;10:3924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 149. | Wang Q, Roy B, Turecki G, Shelton RC, Dwivedi Y. Role of Complex Epigenetic Switching in Tumor Necrosis Factor-α Upregulation in the Prefrontal Cortex of Suicide Subjects. Am J Psychiatry. 2018;175:262-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 150. | Torres-Berrío A, Lopez JP, Bagot RC, Nouel D, Dal Bo G, Cuesta S, Zhu L, Manitt C, Eng C, Cooper HM, Storch KF, Turecki G, Nestler EJ, Flores C. DCC Confers Susceptibility to Depression-like Behaviors in Humans and Mice and Is Regulated by miR-218. Biol Psychiatry. 2017;81:306-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 120] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 151. | Roy B, Wang Q, Palkovits M, Faludi G, Dwivedi Y. Altered miRNA expression network in locus coeruleus of depressed suicide subjects. Sci Rep. 2017;7:4387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 152. | Ibrahim P, Denniston R, Mitsuhashi H, Yang J, Fiori LM, Żurawek D, Mechawar N, Nagy C, Turecki G. Profiling Small RNA From Brain Extracellular Vesicles in Individuals With Depression. Int J Neuropsychopharmacol. 2024;27:pyae013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 153. | Šalamon Arčan I, Katrašnik M, Kouter K, Zupanc T, Videtič Paska A. Extracellular vesicles from cerebrospinal fluid revealed changes in miR-19a-3p and miR-4516 expression in Slovene male suicides. Genes Brain Behav. 2023;22:e12868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 154. | Roy B, Ochi S, Dwivedi Y. Potential of Circulating miRNAs as Molecular Markers in Mood Disorders and Associated Suicidal Behavior. Int J Mol Sci. 2023;24:4664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 155. | Lopez JP, Lim R, Cruceanu C, Crapper L, Fasano C, Labonte B, Maussion G, Yang JP, Yerko V, Vigneault E, El Mestikawy S, Mechawar N, Pavlidis P, Turecki G. miR-1202 is a primate-specific and brain-enriched microRNA involved in major depression and antidepressant treatment. Nat Med. 2014;20:764-768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 211] [Cited by in RCA: 246] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 156. | Gururajan A, Naughton ME, Scott KA, O'Connor RM, Moloney G, Clarke G, Dowling J, Walsh A, Ismail F, Shorten G, Scott L, McLoughlin DM, Cryan JF, Dinan TG. MicroRNAs as biomarkers for major depression: a role for let-7b and let-7c. Transl Psychiatry. 2016;6:e862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 108] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 157. | Brás JP, Pinto S, von Doellinger O, Prata J, Coelho R, Barbosa MA, Almeida MI, Santos SG. Combining inflammatory miRNA molecules as diagnostic biomarkers for depression: a clinical study. Front Psychiatry. 2023;14:1227618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 158. | Stapel B, Xiao K, Gorinski N, Schmidt K, Pfanne A, Fiedler J, Richter I, Vollbrecht AL, Thum T, Kahl KG, Ponimaskin E. MicroRNAs as novel peripheral markers for suicidality in patients with major depressive disorder. Front Psychiatry. 2022;13:1020530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 159. | Lopez JP, Fiori LM, Cruceanu C, Lin R, Labonte B, Cates HM, Heller EA, Vialou V, Ku SM, Gerald C, Han MH, Foster J, Frey BN, Soares CN, Müller DJ, Farzan F, Leri F, MacQueen GM, Feilotter H, Tyryshkin K, Evans KR, Giacobbe P, Blier P, Lam RW, Milev R, Parikh SV, Rotzinger S, Strother SC, Lewis CM, Aitchison KJ, Wittenberg GM, Mechawar N, Nestler EJ, Uher R, Kennedy SH, Turecki G. MicroRNAs 146a/b-5 and 425-3p and 24-3p are markers of antidepressant response and regulate MAPK/Wnt-system genes. Nat Commun. 2017;8:15497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 149] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 160. | Wang KR, Hecker J, McGeachie MJ. Quantifying the massive pleiotropy of microRNA: a human microRNA-disease causal association database generated with ChatGPT. bioRxiv. 2024;2024.07.08.602488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 161. | Videtič Paska A, Alič U, Zupanc T, Kouter K. Suicide and Changes in Expression of Neuronal miRNA Predicted by an Algorithm Search through miRNA Databases. Genes (Basel). 2022;13:562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 162. | Kölliker-Frers R, Udovin L, Otero-Losada M, Kobiec T, Herrera MI, Palacios J, Razzitte G, Capani F. Neuroinflammation: An Integrating Overview of Reactive-Neuroimmune Cell Interactions in Health and Disease. Mediators Inflamm. 2021;2021:9999146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 163. | Ganança L, Oquendo MA, Tyrka AR, Cisneros-Trujillo S, Mann JJ, Sublette ME. The role of cytokines in the pathophysiology of suicidal behavior. Psychoneuroendocrinology. 2016;63:296-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 152] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 164. | Brundin L, Bryleva EY, Thirtamara Rajamani K. Role of Inflammation in Suicide: From Mechanisms to Treatment. Neuropsychopharmacology. 2017;42:271-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 175] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 165. | Nguyen KT, Deak T, Will MJ, Hansen MK, Hunsaker BN, Fleshner M, Watkins LR, Maier SF. Timecourse and corticosterone sensitivity of the brain, pituitary, and serum interleukin-1beta protein response to acute stress. Brain Res. 2000;859:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 109] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 166. | Goshen I, Kreisel T, Ben-Menachem-Zidon O, Licht T, Weidenfeld J, Ben-Hur T, Yirmiya R. Brain interleukin-1 mediates chronic stress-induced depression in mice via adrenocortical activation and hippocampal neurogenesis suppression. Mol Psychiatry. 2008;13:717-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 539] [Cited by in RCA: 579] [Article Influence: 34.1] [Reference Citation Analysis (0)] |
| 167. | Gonzalez PV, Schiöth HB, Lasaga M, Scimonelli TN. Memory impairment induced by IL-1beta is reversed by alpha-MSH through central melanocortin-4 receptors. Brain Behav Immun. 2009;23:817-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 168. | Goltser-Dubner T, Benarroch F, Lavon M, Amer R, Canetti L, Giesser R, Kianski E, Martin J, Pevzner D, Blum Weinberg P, Ben-Ari A, Bar-Nitsan M, Alon S, Yshai S, Lotan A, Galili-Weisstub E, Segman R, Shalev A. Childhood trauma cortisol and immune cell glucocorticoid transcript levels are associated with increased risk for suicidality in adolescence. Mol Psychiatry. 2025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 169. | Block ML, Hong JS. Microglia and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Prog Neurobiol. 2005;76:77-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1090] [Cited by in RCA: 1197] [Article Influence: 59.9] [Reference Citation Analysis (0)] |
| 170. | Fazel S, Wolf A, Pillas D, Lichtenstein P, Långström N. Suicide, fatal injuries, and other causes of premature mortality in patients with traumatic brain injury: a 41-year Swedish population study. JAMA Psychiatry. 2014;71:326-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 116] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 171. | Juengst SB, Kumar RG, Arenth PM, Wagner AK. Exploratory associations with tumor necrosis factor-α, disinhibition and suicidal endorsement after traumatic brain injury. Brain Behav Immun. 2014;41:134-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 172. | Shenk CE, Noll JG, Putnam FW, Trickett PK. A prospective examination of the role of childhood sexual abuse and physiological asymmetry in the development of psychopathology. Child Abuse Negl. 2010;34:752-761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 173. | Lippard ETC, Nemeroff CB. The Devastating Clinical Consequences of Child Abuse and Neglect: Increased Disease Vulnerability and Poor Treatment Response in Mood Disorders. Am J Psychiatry. 2020;177:20-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 275] [Article Influence: 55.0] [Reference Citation Analysis (0)] |
| 174. | Angelakis I, Gillespie EL, Panagioti M. Childhood maltreatment and adult suicidality: a comprehensive systematic review with meta-analysis. Psychol Med. 2019;49:1057-1078. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 237] [Article Influence: 39.5] [Reference Citation Analysis (0)] |
| 175. | Teicher MH, Khan A. Childhood Maltreatment, Cortical and Amygdala Morphometry, Functional Connectivity, Laterality, and Psychopathology. Child Maltreat. 2019;24:458-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 176. | Elliott E, Ezra-Nevo G, Regev L, Neufeld-Cohen A, Chen A. Resilience to social stress coincides with functional DNA methylation of the Crf gene in adult mice. Nat Neurosci. 2010;13:1351-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 320] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 177. | Zhang TY, Labonté B, Wen XL, Turecki G, Meaney MJ. Epigenetic mechanisms for the early environmental regulation of hippocampal glucocorticoid receptor gene expression in rodents and humans. Neuropsychopharmacology. 2013;38:111-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 242] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 178. | Weaver IC, Cervoni N, Champagne FA, D'Alessio AC, Sharma S, Seckl JR, Dymov S, Szyf M, Meaney MJ. Epigenetic programming by maternal behavior. Nat Neurosci. 2004;7:847-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4264] [Cited by in RCA: 3785] [Article Influence: 180.2] [Reference Citation Analysis (0)] |
| 179. | Brivio P, Sbrini G, Tarantini L, Parravicini C, Gruca P, Lason M, Litwa E, Favero C, Riva MA, Eberini I, Papp M, Bollati V, Calabrese F. Stress Modifies the Expression of Glucocorticoid-Responsive Genes by Acting at Epigenetic Levels in the Rat Prefrontal Cortex: Modulatory Activity of Lurasidone. Int J Mol Sci. 2021;22:6197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 180. | Suderman M, McGowan PO, Sasaki A, Huang TC, Hallett MT, Meaney MJ, Turecki G, Szyf M. Conserved epigenetic sensitivity to early life experience in the rat and human hippocampus. Proc Natl Acad Sci U S A. 2012;109 Suppl 2:17266-17272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 204] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 181. | Czamara D, Tissink E, Tuhkanen J, Martins J, Awaloff Y, Drake AJ, Khulan B, Palotie A, Winter SM, Nemeroff CB, Craighead WE, Dunlop BW, Mayberg HS, Kinkead B, Mathew SJ, Iosifescu DV, Neylan TC, Heim CM, Lahti J, Eriksson JG, Räikkönen K, Ressler KJ, Provençal N, Binder EB. Combined effects of genotype and childhood adversity shape variability of DNA methylation across age. Transl Psychiatry. 2021;11:88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 182. | Labonte B, Yerko V, Gross J, Mechawar N, Meaney MJ, Szyf M, Turecki G. Differential glucocorticoid receptor exon 1(B), 1(C), and 1(H) expression and methylation in suicide completers with a history of childhood abuse. Biol Psychiatry. 2012;72:41-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 239] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 183. | Liu D, Diorio J, Tannenbaum B, Caldji C, Francis D, Freedman A, Sharma S, Pearson D, Plotsky PM, Meaney MJ. Maternal care, hippocampal glucocorticoid receptors, and hypothalamic-pituitary-adrenal responses to stress. Science. 1997;277:1659-1662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2263] [Cited by in RCA: 2120] [Article Influence: 75.7] [Reference Citation Analysis (0)] |
| 184. | McGowan PO, Suderman M, Sasaki A, Huang TC, Hallett M, Meaney MJ, Szyf M. Broad epigenetic signature of maternal care in the brain of adult rats. PLoS One. 2011;6:e14739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 316] [Cited by in RCA: 291] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 185. | Zaidan H, Wnuk A, Aderka IM, Kajta M, Gaisler-Salomon I. Pre-reproductive stress in adolescent female rats alters maternal care and DNA methylation patterns across generations. Stress. 2023;26:2201325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 186. | King SE, Schatz NA, Babenko O, Ilnytskyy Y, Kovalchuk I, Metz GAS. Prenatal maternal stress in rats alters the epigenetic and transcriptomic landscape of the maternal-fetal interface across four generations. Commun Biol. 2025;8:38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 187. | McGowan PO, Sasaki A, D'Alessio AC, Dymov S, Labonté B, Szyf M, Turecki G, Meaney MJ. Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nat Neurosci. 2009;12:342-348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2696] [Cited by in RCA: 2130] [Article Influence: 133.1] [Reference Citation Analysis (0)] |
| 188. | Wadhwa PD, Entringer S, Buss C, Lu MC. The contribution of maternal stress to preterm birth: issues and considerations. Clin Perinatol. 2011;38:351-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 318] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 189. | Heim C, Newport DJ, Mletzko T, Miller AH, Nemeroff CB. The link between childhood trauma and depression: insights from HPA axis studies in humans. Psychoneuroendocrinology. 2008;33:693-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1122] [Cited by in RCA: 1140] [Article Influence: 67.1] [Reference Citation Analysis (0)] |
| 190. | Labonté B, Suderman M, Maussion G, Navaro L, Yerko V, Mahar I, Bureau A, Mechawar N, Szyf M, Meaney MJ, Turecki G. Genome-wide epigenetic regulation by early-life trauma. Arch Gen Psychiatry. 2012;69:722-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 297] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 191. | Kaminsky Z, Wilcox HC, Eaton WW, Van Eck K, Kilaru V, Jovanovic T, Klengel T, Bradley B, Binder EB, Ressler KJ, Smith AK. Epigenetic and genetic variation at SKA2 predict suicidal behavior and post-traumatic stress disorder. Transl Psychiatry. 2015;5:e627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 192. | Sadeh N, Wolf EJ, Logue MW, Hayes JP, Stone A, Griffin LM, Schichman SA, Miller MW. Epigenetic variation at ska2 predicts suicide phenotypes and internalizing psychopathology. Depress Anxiety. 2016;33:308-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 193. | Janusek LW, Tell D, Gaylord-Harden N, Mathews HL. Relationship of childhood adversity and neighborhood violence to a proinflammatory phenotype in emerging adult African American men: An epigenetic link. Brain Behav Immun. 2017;60:126-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 96] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 194. | Lam LL, Emberly E, Fraser HB, Neumann SM, Chen E, Miller GE, Kobor MS. Factors underlying variable DNA methylation in a human community cohort. Proc Natl Acad Sci U S A. 2012;109 Suppl 2:17253-17260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 344] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 195. | Miller GE, Cole SW. Clustering of depression and inflammation in adolescents previously exposed to childhood adversity. Biol Psychiatry. 2012;72:34-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 266] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 196. | Carvalho Silva R, Martini P, Hohoff C, Mattevi S, Bortolomasi M, Menesello V, Gennarelli M, Baune BT, Minelli A. DNA methylation changes in association with trauma-focused psychotherapy efficacy in treatment-resistant depression patients: a prospective longitudinal study. Eur J Psychotraumatol. 2024;15:2314913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 197. | Maffioletti E, Bocchio-Chiavetto L, Perusi G, Carvalho Silva R, Sacco C, Bazzanella R, Zampieri E, Bortolomasi M, Gennarelli M, Minelli A. Inflammation-related microRNAs are involved in stressful life events exposure and in trauma-focused psychotherapy in treatment-resistant depressed patients. Eur J Psychotraumatol. 2021;12:1987655. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 198. | Johnston JN, Campbell D, Caruncho HJ, Henter ID, Ballard ED, Zarate CA. Suicide Biomarkers to Predict Risk, Classify Diagnostic Subtypes, and Identify Novel Therapeutic Targets: 5 Years of Promising Research. Int J Neuropsychopharmacol. 2022;25:197-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 199. | Wisłowska-Stanek A, Kołosowska K, Maciejak P. Neurobiological Basis of Increased Risk for Suicidal Behaviour. Cells. 2021;10:2519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 200. | Wielscher M, Mandaviya PR, Kuehnel B, Joehanes R, Mustafa R, Robinson O, Zhang Y, Bodinier B, Walton E, Mishra PP, Schlosser P, Wilson R, Tsai PC, Palaniswamy S, Marioni RE, Fiorito G, Cugliari G, Karhunen V, Ghanbari M, Psaty BM, Loh M, Bis JC, Lehne B, Sotoodehnia N, Deary IJ, Chadeau-Hyam M, Brody JA, Cardona A, Selvin E, Smith AK, Miller AH, Torres MA, Marouli E, Gào X, van Meurs JBJ, Graf-Schindler J, Rathmann W, Koenig W, Peters A, Weninger W, Farlik M, Zhang T, Chen W, Xia Y, Teumer A, Nauck M, Grabe HJ, Doerr M, Lehtimäki T, Guan W, Milani L, Tanaka T, Fisher K, Waite LL, Kasela S, Vineis P, Verweij N, van der Harst P, Iacoviello L, Sacerdote C, Panico S, Krogh V, Tumino R, Tzala E, Matullo G, Hurme MA, Raitakari OT, Colicino E, Baccarelli AA, Kähönen M, Herzig KH, Li S; BIOS consortium, Conneely KN, Kooner JS, Köttgen A, Heijmans BT, Deloukas P, Relton C, Ong KK, Bell JT, Boerwinkle E, Elliott P, Brenner H, Beekman M, Levy D, Waldenberger M, Chambers JC, Dehghan A, Järvelin MR. DNA methylation signature of chronic low-grade inflammation and its role in cardio-respiratory diseases. Nat Commun. 2022;13:2408. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 61] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 201. | Condrat CE, Thompson DC, Barbu MG, Bugnar OL, Boboc A, Cretoiu D, Suciu N, Cretoiu SM, Voinea SC. miRNAs as Biomarkers in Disease: Latest Findings Regarding Their Role in Diagnosis and Prognosis. Cells. 2020;9:276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 583] [Cited by in RCA: 859] [Article Influence: 171.8] [Reference Citation Analysis (0)] |
| 202. | Belzeaux R, Fiori LM, Lopez JP, Boucekine M, Boyer L, Blier P, Farzan F, Frey BN, Giacobbe P, Lam RW, Leri F, MacQueen GM, Milev R, Müller DJ, Parikh SV, Rotzinger S, Soares CN, Uher R, Foster JA, Kennedy SH, Turecki G. Predicting Worsening Suicidal Ideation With Clinical Features and Peripheral Expression of Messenger RNA and MicroRNA During Antidepressant Treatment. J Clin Psychiatry. 2019;80:18m12556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 203. | Roy B, Yoshino Y, Allen L, Prall K, Schell G, Dwivedi Y. Exploiting Circulating MicroRNAs as Biomarkers in Psychiatric Disorders. Mol Diagn Ther. 2020;24:279-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 204. | Saeedi S, Israel S, Nagy C, Turecki G. The emerging role of exosomes in mental disorders. Transl Psychiatry. 2019;9:122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 287] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 205. | Mustapic M, Eitan E, Werner JK Jr, Berkowitz ST, Lazaropoulos MP, Tran J, Goetzl EJ, Kapogiannis D. Plasma Extracellular Vesicles Enriched for Neuronal Origin: A Potential Window into Brain Pathologic Processes. Front Neurosci. 2017;11:278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 310] [Cited by in RCA: 335] [Article Influence: 41.9] [Reference Citation Analysis (0)] |
| 206. | Mamdani F, Weber MD, Bunney B, Burke K, Cartagena P, Walsh D, Lee FS, Barchas J, Schatzberg AF, Myers RM, Watson SJ, Akil H, Vawter MP, Bunney WE, Sequeira A. Identification of potential blood biomarkers associated with suicide in major depressive disorder. Transl Psychiatry. 2022;12:159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 207. | Wang M, Xiang H, Wei J, Dou Y, Yan Y, Du Y, Fan H, Zhao L, Ni R, Yang X, Ma X. Identification of blood transcriptome modules associated with suicidal ideation in patients with major depressive disorder. Sci Rep. 2025;15:1067. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 208. | Ganança L, Galfalvy HC, Cisneros-Trujillo S, Basseda Z, Cooper TB, Ren X, Figueira ML, Oquendo MA, Mann JJ, Sublette ME. Relationships between inflammatory markers and suicide risk status in major depression. J Psychiatr Res. 2021;134:192-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 209. | Daray FM, Chiapella LC, Grendas LN, Casiani RIÁ, Olaviaga A, Robetto J, Prokopez CR, Carrera Silva EA, Errasti AE, Neupane SP. Peripheral blood cellular immunophenotype in suicidal ideation, suicide attempt, and suicide: a systematic review and meta-analysis. Mol Psychiatry. 2024;29:3874-3892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 210. | Lengvenyte A, Belzeaux R, Olié E, Hamzeh-Cognasse H, Sénèque M, Strumila R, Cognasse F, Courtet P. Associations of potential plasma biomarkers with suicide attempt history, current suicidal ideation and subsequent suicidal events in patients with depression: A discovery study. Brain Behav Immun. 2023;114:242-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 211. | Schiweck C, Claes S, Van Oudenhove L, Lafit G, Vaessen T, de Beeck GO, Berghmans R, Wijkhuijs A, Müller N, Arolt V, Drexhage H, Vrieze E. Childhood trauma, suicide risk and inflammatory phenotypes of depression: insights from monocyte gene expression. Transl Psychiatry. 2020;10:296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 212. | Del Giudice M, Gangestad SW. Rethinking IL-6 and CRP: Why they are more than inflammatory biomarkers, and why it matters. Brain Behav Immun. 2018;70:61-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 503] [Article Influence: 71.9] [Reference Citation Analysis (0)] |
| 213. | Melhem NM, Munroe S, Marsland A, Gray K, Brent D, Porta G, Douaihy A, Laudenslager ML, DePietro F, Diler R, Driscoll H, Gopalan P. Blunted HPA axis activity prior to suicide attempt and increased inflammation in attempters. Psychoneuroendocrinology. 2017;77:284-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 214. | Stanley B, Itzhaky L, Oquendo MA. Identifying Neurobiological Underpinnings of Two Suicidal Subtypes. J Psychiatr Brain Sci. 2021;6:e210016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 215. | Feczko E, Miranda-Dominguez O, Marr M, Graham AM, Nigg JT, Fair DA. The Heterogeneity Problem: Approaches to Identify Psychiatric Subtypes. Trends Cogn Sci. 2019;23:584-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 264] [Article Influence: 44.0] [Reference Citation Analysis (0)] |
| 216. | Dobbertin M, Blair KS, Carollo E, Blair JR, Dominguez A, Bajaj S. Neuroimaging alterations of the suicidal brain and its relevance to practice: an updated review of MRI studies. Front Psychiatry. 2023;14:1083244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 217. | Bai S, Guo W, Feng Y, Deng H, Li G, Nie H, Guo G, Yu H, Ma Y, Wang J, Chen S, Jing J, Yang J, Tang Y, Tang Z. Efficacy and safety of anti-inflammatory agents for the treatment of major depressive disorder: a systematic review and meta-analysis of randomised controlled trials. J Neurol Neurosurg Psychiatry. 2020;91:21-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 147] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 218. | Recourt K, de Boer P, van der Ark P, Benes H, van Gerven JMA, Ceusters M, van Nueten L, Drevets WC, Bhatacharya A, Browning M, Jacobs GE. Characterization of the central nervous system penetrant and selective purine P2X7 receptor antagonist JNJ-54175446 in patients with major depressive disorder. Transl Psychiatry. 2023;13:266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 219. | Yamanashi T, Iwata M, Kamiya N, Tsunetomi K, Kajitani N, Wada N, Iitsuka T, Yamauchi T, Miura A, Pu S, Shirayama Y, Watanabe K, Duman RS, Kaneko K. Beta-hydroxybutyrate, an endogenic NLRP3 inflammasome inhibitor, attenuates stress-induced behavioral and inflammatory responses. Sci Rep. 2017;7:7677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 151] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 220. | Zhang Y, Anoopkumar-Dukie S, Davey AK. SIRT1 and SIRT2 Modulators: Potential Anti-Inflammatory Treatment for Depression? Biomolecules. 2021;11:353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 221. | Rigdon G, Prescott Y, Hall J, Abernathy K, Raskin J, Wargin W. Phase 1, Single-Center, Double-Blind, Randomized, Placebo-Controlled Studies of the Safety, Tolerability, and Pharmacokinetics of Single and Multiple Ascending Oral Doses of the Sirtuin 6 Activator SP-624 in Healthy Adults. Clin Pharmacol Drug Dev. 2025;14:18-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 222. | Capuzzi E, Caldiroli A, Capellazzi M, Tagliabue I, Buoli M, Clerici M. Biomarkers of suicidal behaviors: A comprehensive critical review. Adv Clin Chem. 2020;96:179-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 223. | Lehrer S, Rheinstein PH. Nonsteroidal anti-inflammatory drugs (NSAIDs) reduce suicidal ideation and depression. Discov Med. 2019;28:205-212. [PubMed] |
| 224. | Nikkheslat N. Targeting inflammation in depression: Ketamine as an anti-inflammatory antidepressant in psychiatric emergency. Brain Behav Immun Health. 2021;18:100383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 225. | Jóźwiak-Bębenista M, Sokołowska P, Wiktorowska-Owczarek A, Kowalczyk E, Sienkiewicz M. Ketamine - A New Antidepressant Drug with Anti-Inflammatory Properties. J Pharmacol Exp Ther. 2024;388:134-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 226. | Yu X, Zhao H, Wang R, Chen Y, Ouyang X, Li W, Sun Y, Peng A. Cancer epigenetics: from laboratory studies and clinical trials to precision medicine. Cell Death Discov. 2024;10:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 92] [Reference Citation Analysis (0)] |
| 227. | Wei Y, Melas PA, Wegener G, Mathé AA, Lavebratt C. Antidepressant-like effect of sodium butyrate is associated with an increase in TET1 and in 5-hydroxymethylation levels in the Bdnf gene. Int J Neuropsychopharmacol. 2014;18:pyu032. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 109] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 228. | Paoli C, Misztak P, Mazzini G, Musazzi L. DNA Methylation in Depression and Depressive-Like Phenotype: Biomarker or Target of Pharmacological Intervention? Curr Neuropharmacol. 2022;20:2267-2291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 229. | Lee J, Papa F, Jaini PA, Alpini S, Kenny T. An Epigenetics-Based, Lifestyle Medicine-Driven Approach to Stress Management for Primary Patient Care: Implications for Medical Education. Am J Lifestyle Med. 2020;14:294-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 230. | Khoury R, Nagy C. Running from stress: a perspective on the potential benefits of exercise-induced small extracellular vesicles for individuals with major depressive disorder. Front Mol Biosci. 2023;10:1154872. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 231. | Atmakuru A, Shahini A, Chakraborty S, Seoni S, Salvi M, Hafeez-baig A, Rashid S, Tan RS, Barua PD, Molinari F, Acharya UR. Artificial intelligence-based suicide prevention and prediction: A systematic review (2019-2023). Inf Fusion. 2025;114:102673. [DOI] [Full Text] |
| 232. | Pigoni A, Delvecchio G, Turtulici N, Madonna D, Pietrini P, Cecchetti L, Brambilla P. Machine learning and the prediction of suicide in psychiatric populations: a systematic review. Transl Psychiatry. 2024;14:140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 22] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 233. | Hamamoto R, Komatsu M, Takasawa K, Asada K, Kaneko S. Epigenetics Analysis and Integrated Analysis of Multiomics Data, Including Epigenetic Data, Using Artificial Intelligence in the Era of Precision Medicine. Biomolecules. 2019;10:62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (0)] |