Published online Feb 19, 2024. doi: 10.5498/wjp.v14.i2.276
Peer-review started: November 23, 2023
First decision: December 6, 2023
Revised: December 13, 2023
Accepted: January 8, 2024
Article in press: January 8, 2024
Published online: February 19, 2024
Processing time: 74 Days and 18.2 Hours
Major depression disorder (MDD) constitutes a significant mental health concern. Epidemiological surveys indicate that the lifetime prevalence of depression in adolescents is much higher than that in adults, with a corresponding increased risk of suicide. In studying brain dysfunction associated with MDD in adole-scents, research on brain white matter (WM) is sparse. Some researchers even mistakenly regard the signals generated by the WM as noise points. In fact, studies have shown that WM exhibits similar blood oxygen level-dependent signal fluctuations. The alterations in WM signals and their relationship with disease severity in adolescents with MDD remain unclear.
To explore potential abnormalities in WM functional signals in adolescents with MDD.
This study involved 48 adolescent patients with MDD and 31 healthy controls (HC). All participants were assessed using the Patient Health Questionnaire-9 Scale and the mini international neuropsychiatric interview (MINI) suicide inventory. In addition, a Siemens Skyra 3.0T magnetic resonance scanner was used to obtain the subjects' image data. The DPABI software was utilized to calculate the WM signal of the fractional amplitude of low frequency fluctuations (fALFF) and regional homogeneity, followed by a two-sample t-test between the MDD and HC groups. Independent component analysis (ICA) was also used to evaluate the WM functional signal. Pearson’s correlation was performed to assess the relationship between statistical test results and clinical scales.
Compared to HC, individuals with MDD demonstrated a decrease in the fALFF of WM in the corpus callosum body, left posterior limb of the internal capsule, right superior corona radiata, and bilateral posterior corona radiata [P < 0.001, family-wise error (FWE) voxel correction]. The regional homogeneity of WM increased in the right posterior limb of internal capsule and left superior corona radiata, and decreased in the left superior longitudinal fasciculus (P < 0.001, FWE voxel correction). The ICA results of WM overlapped with those of regional homo-geneity. The fALFF of WM signal in the left posterior limb of the internal capsule was negatively correlated with the MINI suicide scale (P = 0.026, r = -0.32), and the right posterior corona radiata was also negatively correlated with the MINI suicide scale (P = 0.047, r = -0.288).
Adolescents with MDD involves changes in WM functional signals, and these differences in brain regions may increase the risk of suicide.
Core Tip: This groundbreaking study investigates white matter (WM) functional signals in adolescents with major depressive disorder (MDD), an area often overlooked in research. Utilizing advanced imaging techniques, the study identifies specific abnormalities in WM signals, revealing decreased fractional amplitude of low frequency fluctuations in key regions and altered regional homogeneity and independent component analysis patterns. Notably, these changes correlate with suicidality scales, indicating a potential link between WM anomalies and severity of depression. The study pioneers a crucial shift in understanding MDD's neuropathogenesis, offering novel insights and support for future research and predictive measures.
- Citation: Huang XL, Gao J, Wang YM, Zhu F, Qin J, Yao QN, Zhang XB, Sun HY. Neuropathological characteristics of abnormal white matter functional signaling in adolescents with major depression. World J Psychiatry 2024; 14(2): 276-286
- URL: https://www.wjgnet.com/2220-3206/full/v14/i2/276.htm
- DOI: https://dx.doi.org/10.5498/wjp.v14.i2.276
Major depressive disorder (MDD) is a widespread psychiatric condition across the globe, with an estimated lifetime prevalence rate of approximately 11% among adolescents[1,2]. It has been reported that over half of adolescent suicide victims had a depressive disorder[3]. MDD in adolescents is associated with an increased risk of suicide, and the disorder often persists into adulthood[4]. Therefore, the increasing incidence of depression among adolescents requires urgent attention[5]. The recognition and treatment of MDD in adolescence are crucial; however, our current grasp of the physiological and pathological underpinnings of the condition remains incomplete.
Functional magnetic resonance imaging (fMRI) is a non-invasive technique, and it can be used to indirectly measure neuronal activity via the blood oxygenation level-dependent (BOLD) signal[6]. In recent years, the development and progress of magnetic resonance imaging (MRI) technology have provided an opportunity to study the pathophysiology of MDD using various resting state fMRI (rs-fMRI) techniques. These techniques have been extensively utilized in the exploration of the physiological and pathological foundations of the brain in mental disorders. The changes in the resting state of gray matter (GM) found in most studies are often used as biomarkers for neuropsychiatric disorders[7-9], while white matter (WM) tends to be ignored[10,11], even though the volume of WM in the human brain accounts for approximately 40%-45%[12].
To date, various approaches are used to analyze spontaneous BOLD signals, such as the amplitude of low frequency fluctuations (ALFF), the fractional ALFF (fALFF), regional homogeneity (ReHo), and independent component analysis (ICA)[13]. The fALFF[14] is a metric believed to capture spontaneous neural activity and has been demonstrated to correlate with regional brain glucose metabolism[15-17], which effectively mitigates physiological noise when compared to ALFF[14]. ReHo, a data-driven approach, implies that the time series of spatially adjacent voxels exhibit greater temporal similarity when the brain region is engaged in a specific condition[18]. ICA is also a data-driven method, it can decompose fMRI data into spatially independent and functionally connected brain networks[19]. ReHo and ICA can acquire a greater amount of information than methods driven by models[18,20]. Using these data analysis methods, Liu et al[21] discovered the alterations in ReHo and ALFF in the precentral gyrus, postcentral gyrus, and paracentral gyrus in MDD[21]. In addition, a meta-analysis found a correlation between amygdala activity and depression[22]. These findings may indicate the pathological and physiological processes associated with MDD.
Recent studies have shown the presence of functional brain activity related to neuronal activity in the WM, including connectivity and interconnection functions[23,24]. In task related studies, activation of brain regions can be detected in the inner capsule and corpus callosum (CC)[25,26]. Additional studies showed that fMRI activity within particular WM pathways is remarkably consistent during the resting state, and indicated that these WM signals exhibit features remi-niscent of hemodynamic (BOLD) alterations linked to neuronal activity[27,28]. Furthermore, a recent study employing ICA and hierarchical clustering revealed the presence of clusters of correlated activity within the WM[29]. These findings reveal that WM may play a crucial role in resting states. We hope to provide more information in order to understand the underlying pathological mechanisms of adolescent severe depression by revealing the fluctuation characteristics of WM functional signals.
Therefore, we decided to use the GM analysis method to explore changes in WM, using a combination of multiple features, including fALFF, ReHo, and ICA. These three analytical methods present a progressive relationship layer by layer, from individual, to local, and finally to component networks. The combination of multiple analytical methods is used to explore the functional differences of WM in adolescents with MDD. This method is expected to reveal the characteristics and potential biological mechanisms of abnormal brain activity in adolescents with MDD during the resting state, filling the current incomplete understanding of the physiological and pathological basis of the disease. From this study, we may not only better understand the pathogenesis of MDD in adolescents, but also provide more targeted methods for future diagnosis and treatment, thereby more effectively addressing this global health problem.
A total of 84 subjects, including 51 adolescents with MDD and 33 healthy controls (HC) matched by age, gender, education and right-handedness were initially recruited from Suzhou Guangji Hospital. Using the mini international neuropsychiatric interview (MINI), patients were diagnosed by two trained psychiatrists above the attending level who conducted a structured interview. The inclusion criteria were: (1) The patients had not received systematic medication treatment prior to the MRI scan; (2) Patient Health Questionnaire-9 (PHQ-9) scores ≥ 20; (3) Right-handed; (4) Met the criteria of the Diagnostic and Statistical Manual of Mental Disorders, 5th edition; and (5) Aged 11 to 18 years. The exclusion criteria were: (1) Contrain-dications to MRI; (2) Any other psychiatric disorders such as bipolar disorder and schizophrenia; (3) Individuals with organic brain diseases, as identified by imaging; and (4) 32-item Hypomania Checklist (HCL-32) scores < 14.
This study was approved by the Ethics Committee of Suzhou Guangji Hospital, and each subject signed a written informed consent form. For participants under age 18, at least one legal guardian signed an informed consent form on their behalf.
Two attending psychiatrists with specialized training assessed the clinical symptoms of the participants and performed a reliability assessment. The PHQ-9 is a reliable and valid screening tool for depression[30,31], derived from the depression section of the Patient Health Questionnaire developed by Spitzer et al[32]. All participants completed the MINI suicidality subscale (MINISS), a user-friendly and highly accurate tool for predicting suicide risk[33] and the HCL-32, a self-assessment tool for hypomanic symptoms[34].
All participants’ image data were obtained on a 3.0T Siemens Skyra scanner at Suzhou Guangji Hospital, equipped with a head/neck 20 channel coil. The scanning parameters of rs-fMRI are as follows: Repetition time = 2000 ms, echo time = 30 ms, slice thickness = 3.5 mm, 32 slices, slice gap = 0.875 mm, filed of view = 224 mm × 224 mm, flip angle = 90°, matrix size = 64 voxels × 64 voxels, acquisition time = 8.04 min. During the scan procedure, each subject was asked to lie flat in the machine, and close their eyes but not to fall asleep. While the subjects were being scanned, a sponge pad was placed on each person's head to prevent head movement and obtain clear images.
The MRI data of each subject were preprocessed by The Data Processing & Analysis for (Resting-state) Brain Imaging (DPABI)[35] and Statistical Parametric Map-ping (SPM12, http://www.fil.ion.ucl.ac.uk/spm) toolkit in MATLAB 2016b. This was based on the following steps: (1) Conversion data format from DICOM to NIFTI; (2) the first 10 time points were removed in order to stabilize the data; (3) slice timing and realignment of head motion correction (any participant whose head motion exceeded 2.0 mm or rotation exceeded 2.0° was excluded); (4) the T1 images were registered to functional images and segmentation into WM and GM and cerebrospinal fluid using the New Segment algorithm; (5) apply white mask to functional images; (6) normalize the functional image space to a standard space (Montreal Neurological Institute) using DARTEL, and resampled to a voxel size of 3 mm × 3 mm × 3 mm; and (7) extraction of individual-level WM 4D images[36]. Five participants were excluded due to head motion > 2 mm or 2°. Finally, 48 MDD and 31 HC were included for further analysis in the current study.
Using a fast Fourier transform at each voxel, we computed the power of the BOLD signal of WM within the low-frequency range of 0.01–0.10 Hz and subsequently divided it by the entire frequency range to calculate the fALFF of WM[14].
The calculation of ReHo of WM values was performed as follows: Firstly, a low-pass filter (0.01–0.1 Hz) was used to mitigate the effects of high-frequency noise and low-frequency drift. Then, Kendall's Coefficient of Concordance[18], also known as the ReHo value, was utilized to assess the similarity between an individual voxel and its neighboring 27 voxels. Subsequently, in order to minimize individual variance, the ReHo value for each voxel was normalized by dividing it by the global mean ReHo value.
On the basis of the aforementioned WM signal preprocessing, the preprocessed imaging data in each group was used to perform group ICA analysis using the fMRI toolbox (GIFT, version 3.0C, http://mialab.mrn.org/software/gift)[37]. Firstly, the pre-processed data's dimensionality was reduced, which was followed by application of the Infomax algorithm for spatial ICA on this data. In addition, component stability was attained by running 100 iterations in a software package called ICASSO[38]. A total of 6 components were estimated. Finally, we selected an interesting independent component that was related to the alteration of WM in MDD for further evaluation.
Differences in demographics between the two participant groups were assessed using SPSS version 29.0, sex differences were assessed using the chi-squared test, while other parameters were compared between the two groups using a two-sample t-test.
Voxel-based comparisons of fALFF and ReHo of WM maps between the patient and control groups were performed using the two-sample t-test in DPABI software with age, sex, education, head-motion included as covariates, and with a threshold setting at P < 0.001, FWE voxel correction for multiple comparisons. Additionally, sex, age, education, and head motion were controlled for during the analysis of ICA maps. The selected component of interest was then compared between the patient and control groups (FWE voxel correction for multiple comparisons).
Correlational analyses were performed to investigate the association between fALFF, ReHo values of MW in regions exhibiting group differences and clinical variables, including the PHQ-9 and the MINI Suicide Inventory.
The demographic and clinical characteristics of the adolescents with MDD and HC are summarized in Table 1. There were no significant differences in sex, age, education and head-motion between the two groups.
| MDD (n = 48) | HC (n = 31) | χ²/t | P value | |
| Sex (male/female) | 8/40 | 6/25 | 0.093 | 0.771 |
| Age (yr) | 14.15 (1.79) | 14.71 (2.00) | -1.306 | 0.195 |
| Education (yr) | 8.44 (1.73) | 8.87 (1.77) | -1.081 | 0.283 |
| Head-motion | 0.08 (0.04) | 0.07 (0.03) | 0.234 | 0.815 |
| PHQ-9 scores | 23.02 (2.39) | - | - | - |
| HCL-32 scores | 6.94 (2.77) | - | - | - |
| MINISS scores | 22.44 (9.61) | - | - | - |
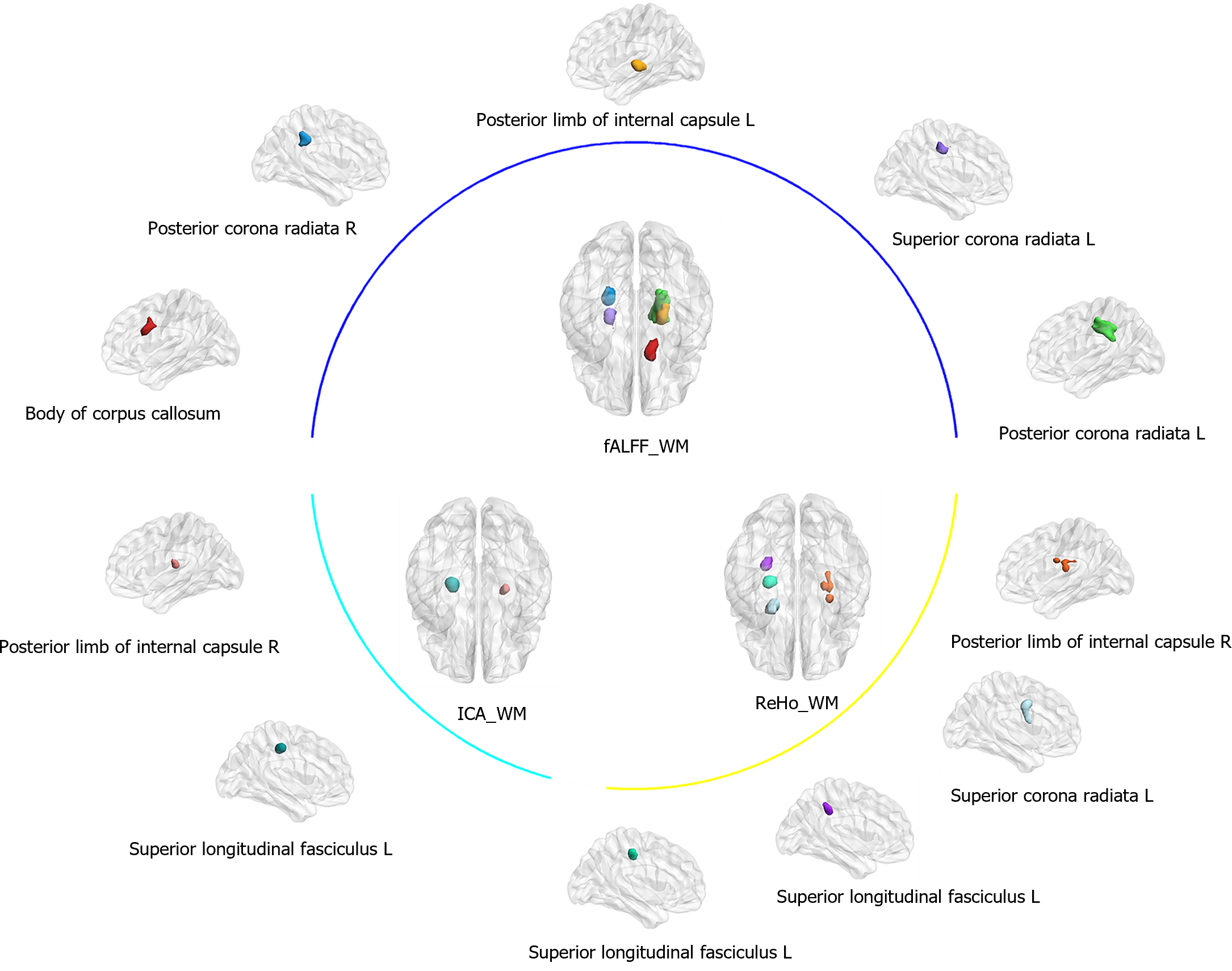
WM brain regions that exhibited disparities between the groups in the fALFF and ReHo analyses were identified and reported using the JHU-ICBM WM label atlas (provided by Wakana et al[39] and Hua et al[40] from the Laboratory of Brain Anatomical MRI at Johns Hopkins University)[39,40]. With regard to the fALFF calculation, patients with MDD showed decreased fALFF in the left posterior limb of the internal capsule (PLIC), posterior corona radiata (PCR), right PCR, superior corona radiata (SCR), and CC body (P < 0.001, FWE voxel correction for multiple comparisons). With regard to the ReHo computation, MDD patients displayed decreased ReHo values in the left superior longitudinal fasciculus (SLF), and higher ReHo values in the right PLIC and the left SCR (P < 0.001, FWE voxel correction for multiple comparisons). These results are presented in Table 2 and Figure 1.
| Indices | Anatomical region | MNI coordinates, x, y, z | Peak intensity | Cluster size |
| fALFF of WM | Posterior limb of the internal capsule L | 21, -18, 3 | -6.12 | 27 |
| Posterior corona radiata R | -24, -39, 33 | -5.57 | 36 | |
| Body of the corpus callosum | 12, 15, 30 | -5.75 | 62 | |
| Superior corona radiata R | -18, -21, 51 | -5.44 | 71 | |
| Posterior corona radiata L | 24, -36, 39 | -6.39 | 106 | |
| ReHo of WM | Posterior limb of the internal capsule R | 27, 0, 18 | 5.40 | 60 |
| Superior corona radiata L | -27, 3, 39 | 4.55 | 23 | |
| Superior longitudinal fasciculus L | -30, -39, 42 | -5.33 | 18 | |
| Superior longitudinal fasciculus L | -24, -24, 48 | -4.76 | 14 | |
| ICA of WM | Posterior limb of the internal capsule R | 27, -15, 12 | 4.30 | 26 |
| Superior longitudinal fasciculus L | -24, -24, 45 | -5.80 | 25 |
We investigated the spatiotemporal patterns in rs-WM-fMRI data using ICA. Six spatially independent components were estimated and extracted from the time series of all WM voxels. Subsequent analyses of the 6 components showed decreased connectivity of the left SLF and higher connectivity of the right PLIC in MDD patients relative to HC (P < 0.001, FWE voxel correction for multiple comparisons; Table 2 and Figure 1).
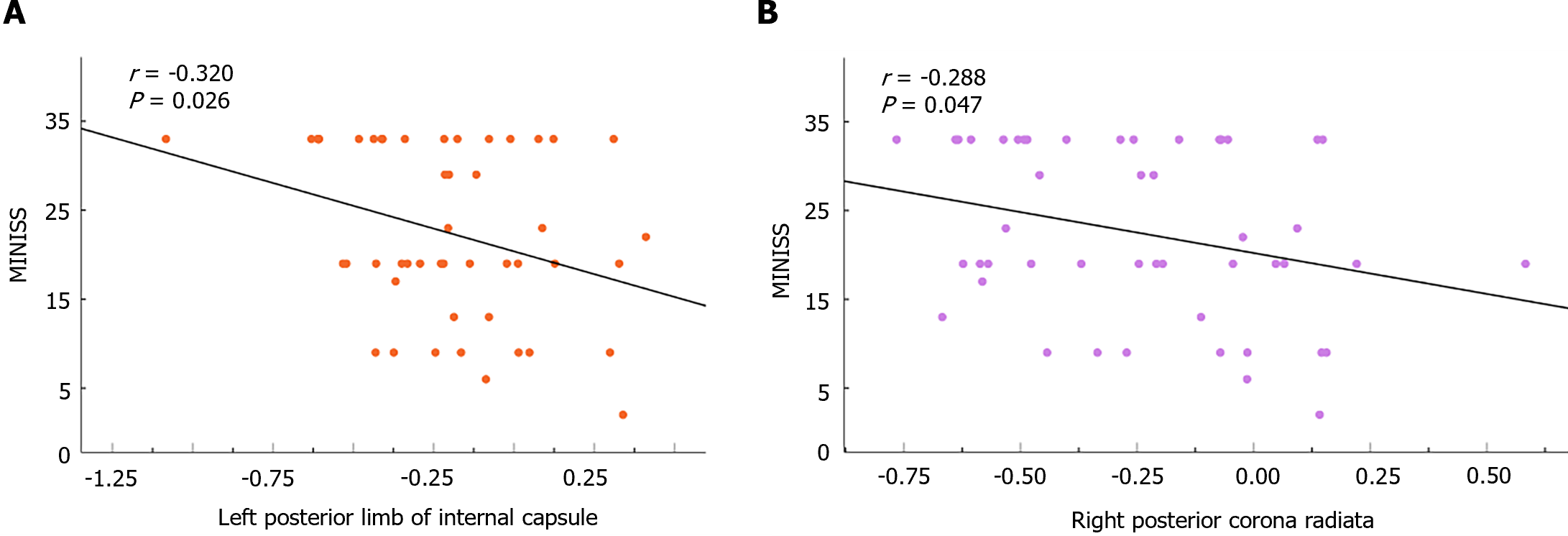
The fALFF and ReHo values were extracted from WM regions that displayed significant differences between adolescents with MDD and the HC group, and correlation analyses between these region’s values and clinical characteristics were conducted. We found there is no correlation between significant differential brain regions and PHQ-9 scales. A negative correlation between the fALFF values in the left PLIC and the MINI suicide scale (P = 0.026, r = -0.320; Figure 2A), as well as between the right PCR and the MINI suicide scale (P = 0.047, r = -0.288; Figure 2B).
In this study, we demonstrated functional changes of WM by employing several distinct rs-fMRI techniques (ReHo, fALFF, and ICA), for the first time, on datasets from healthy subjects, and MDD patients. We found that adolescent patients with MDD showed significant differences in the CC body, left SLF, bilateral PLIC, PCR and SCR compared with the HC group. We also investigated the relationship between functional changes in regions of WM and the clinical features in patients with MDD. These results indicated that resting state functional metrics of WM can be valuable in investigating the pathophysiologic basis of MDD.
In the fALFF results, significant differences in MDD patients were mainly observed in the CC, PLIC, and corona radiata, when compared with the HC group. The CC is a dense bundle of nerve fibers that plays a crucial role in connecting different regions of the neocortex. It facilitates neural circuits involved in cognitive and emotional processing[41,42]. In addition, the CC body contains fibers connecting the cingulate cortex, insular cortex, and temporal cortex[43], these areas are often associated with depression[44-46]. A lower fALFF value indicates a decrease in WM integrity of the CC, which may hinder the interaction between the cerebral hemispheres and cause emotional processing disorders in depression. It is worth noting that in this study, we found that adolescent patients with severe depression experienced abnormal activation of brain regions in the posterior limbs of the inner capsule using various analytical methods. This may be because the core symptom of MDD is low mood, and the PLIC participates in the formation of a neural network by connecting structures such as the cerebral cortex and hypothalamus. Its subcortical area belongs to the frontal striatal circuit[47], and these two circuits play a crucial role in emotional, cognitive, and motor functions[48]. Interestingly, in this study, MDD patients showed significantly lower fALFF values in the PLIC. In addition, Sisti et al[49] also found that cognitive decline was not only related to local brain lesions, but may also be related to the destruction of WM fibers and impaired connectivity in these brain regions[49]. Therefore, this discovery may explain the cognitive style of adolescent patients with depression. The corona radiata is composed of ascending and descending fibers that transmit information to the cerebral cortex and functionally involve emotions and executive processing[50,51]. Furthermore, in the correlation analysis, there was a negative correlation between the fALFF values of the PLIC and the PCR and the suicide scale. Some studies have found that impaired executive function may be a risk factor for suicide[52]. This may indicate that as brain dysfunction increases, the risk of suicide also increases.
The upper longitudinal bundle is considered the largest associative fiber bundle system in the brain[53], connecting the frontal and parietal lobes[54]. It is considered a higher-order multi-sensory associative system and is often reported to be related to executive function and emotions[55]. Previous studies have shown that the degree of damage to the SLF in patients with anxiety related depression may be more severe than in patients with non-anxiety related depression, which may lead to cognitive and emotional impairment[56]. This is consistent with the findings of this study in relation to WM ReHo. In a study of mild cognitive impairment (MCI), the left upper corona showed a lower fractional anisotropy value, suggesting that changes in WM in this brain area may be a potential biomarker of MCI[57]. Therefore, an abnormality of the SLF may indicate that patients with depression have more severe depression.
In this study, the ICA method was used to calculate the differences in the brain network of the components of interest. The brain regions overlapped with ReHo, and the direction of changes in the signal values of the brain regions was consistent. This may be because ReHo measures the local connectivity of spontaneous fMRI signals[18,58], and ICA studies measure inter-regional connectivity. These two methods are complementary to each other in a sense, which is why there is an abnormal overlap of activated brain regions. MDD patients also showed an increase in ReHo values in the left WM and a decrease in fALFF values on the right side in bilateral corona radiata lesions in this study, which may be due to the non-flow coupling metabolism of fALFF and ReHo.
Correlation analysis showed that significant correlations were observed between fALFF values, ReHo values, and clinical features in several WM regions. The left PLIC and the right PCR were negatively correlated with suicide. Research found that the bilateral PCR was associated with cognitive impairment in several different diseases[59-61]. There are many studies on the relationship between cognitive impairment and suicidal behavior[62,63]. In the study of suicidal ideation in schizophrenia, it is mentioned that the PCR may be associated with biological processes leading to depression and increased suicidal ideation[64]. This is consistent with the correlation between the PCR and the MINISS found in our study. In addition, studies have shown that impaired executive function may be a risk factor for suicide[52]. Therefore, we speculate that the abnormal activity of the right PCR and the left posterior limb brain area of the inner capsule may be potential biomarkers that trigger suicidal ideation in patients. There are multiple neural circuits within the inner capsule, and the corona radiata and fiber bundles within the capsule project from the cortex to the thalamus and pons nuclei[65]. The thalamus plays an important role in emotional regulation[66], and changes in the inner capsule may interfere with the connection between the thalamus and cortex, leading to abnormal emotional regulation and increasing the occurrence of manic symptoms.
There are some limitations in this study. Firstly, this was a cross-sectional study that failed to reveal the dynamic changes in WM functional signals over time in adolescents with severe depression. Further longitudinal research will help us understand the principles of this disease. Secondly, the sample size in this study was relatively small, and further research and verification are needed in a larger sample size. In addition, studies have also indicated that the relationship between BOLD signals observed in WM and neuronal related activities is still unclear[67]. We require more evidence in future work to demonstrate the importance of BOLD signals observed in WM.
Our research findings suggest that changes in WM functional signals may provide new insights into the neuro-physiological mechanisms of severe depression in adolescents, and that changes in WM functional signals may serve as biomarkers for predicting future trends in suicide in this disease.
White matter (WM) is composed of various functional nerve fibers and plays an indispensable role in the central nervous system. However, the WM signal changes and their correlation with major depression disorder (MDD) in adolescents are still unclear.
An increasing number of studies have confirmed the functional organization of WM by the resting state functional magnetic imaging (rs-fMRI), indicating its feasibility of studying WM function in adolescents with MDD.
The purpose of this study is to explore the functional changes in the WM of adolescents with MDD.
We collected rs-fMRI data and clinical scale information from the adolescent group with MDD and the healthy control group, and analyzed the correlation between WM function signals and clinical scales in the two groups.
We found significant changes in the functional signals of WM in adolescents with MDD, using the fractional amplitude of low frequency fluctuations, regional homogeneity, and independent component analysis. There are two brain regions, the left posterior limb of the inner capsule and the right posterior corona radiata, which are negatively correlated with the mini international neuropsychiatric interview suicide scales.
The discovery of changes in WM functional signals in adolescents with MDD is of great significance for understanding the neuropathogenesis of depression.
Our research findings may serve as biomarkers for predicting the risk of MDD and suicide in adolescents.
The corresponding author would like to thank all participants and all authors in the study for guidance and support.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Psychiatry
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Bernstein HG, Germany S-Editor: Li L L-Editor: A P-Editor: Chen YX
| 1. | Stallwood E, Monsour A, Rodrigues C, Monga S, Terwee C, Offringa M, Butcher NJ. Systematic Review: The Measurement Properties of the Children's Depression Rating Scale-Revised in Adolescents With Major Depressive Disorder. J Am Acad Child Adolesc Psychiatry. 2021;60:119-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 2. | Avenevoli S, Swendsen J, He JP, Burstein M, Merikangas KR. Major depression in the national comorbidity survey-adolescent supplement: prevalence, correlates, and treatment. J Am Acad Child Adolesc Psychiatry. 2015;54:37-44.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 612] [Cited by in RCA: 765] [Article Influence: 76.5] [Reference Citation Analysis (0)] |
| 3. | Hawton K, van Heeringen K. Suicide. Lancet. 2009;373:1372-1381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1310] [Cited by in RCA: 1289] [Article Influence: 80.6] [Reference Citation Analysis (0)] |
| 4. | Weissman MM, Wolk S, Goldstein RB, Moreau D, Adams P, Greenwald S, Klier CM, Ryan ND, Dahl RE, Wickramaratne P. Depressed adolescents grown up. JAMA. 1999;281:1707-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 574] [Cited by in RCA: 559] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 5. | Barbeito S, Vega P, Sánchez-Gutiérrez T, Becerra JA, González-Pinto A, Calvo A. A systematic review of suicide and suicide attempts in adolescents with psychotic disorders. Schizophr Res. 2021;235:80-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 6. | Ogawa S, Lee TM, Kay AR, Tank DW. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc Natl Acad Sci U S A. 1990;87:9868-9872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4233] [Cited by in RCA: 3761] [Article Influence: 107.5] [Reference Citation Analysis (0)] |
| 7. | Reuveni I, Bonne O, Giesser R, Shragai T, Lazarovits G, Isserles M, Schreiber S, Bick AS, Levin N. Anatomical and functional connectivity in the default mode network of post-traumatic stress disorder patients after civilian and military-related trauma. Hum Brain Mapp. 2016;37:589-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 8. | Finke C, Kopp UA, Scheel M, Pech LM, Soemmer C, Schlichting J, Leypoldt F, Brandt AU, Wuerfel J, Probst C, Ploner CJ, Prüss H, Paul F. Functional and structural brain changes in anti-N-methyl-D-aspartate receptor encephalitis. Ann Neurol. 2013;74:284-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 115] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 9. | Yan CG, Chen X, Li L, Castellanos FX, Bai TJ, Bo QJ, Cao J, Chen GM, Chen NX, Chen W, Cheng C, Cheng YQ, Cui XL, Duan J, Fang YR, Gong QY, Guo WB, Hou ZH, Hu L, Kuang L, Li F, Li KM, Li T, Liu YS, Liu ZN, Long YC, Luo QH, Meng HQ, Peng DH, Qiu HT, Qiu J, Shen YD, Shi YS, Wang CY, Wang F, Wang K, Wang L, Wang X, Wang Y, Wu XP, Wu XR, Xie CM, Xie GR, Xie HY, Xie P, Xu XF, Yang H, Yang J, Yao JS, Yao SQ, Yin YY, Yuan YG, Zhang AX, Zhang H, Zhang KR, Zhang L, Zhang ZJ, Zhou RB, Zhou YT, Zhu JJ, Zou CJ, Si TM, Zuo XN, Zhao JP, Zang YF. Reduced default mode network functional connectivity in patients with recurrent major depressive disorder. Proc Natl Acad Sci U S A. 2019;116:9078-9083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 594] [Cited by in RCA: 521] [Article Influence: 86.8] [Reference Citation Analysis (0)] |
| 10. | Marchitelli R, Paillère-Martinot ML, Bourvis N, Guerin-Langlois C, Kipman A, Trichard C, Douniol M, Stordeur C, Galinowski A, Filippi I, Bertschy G, Weibel S, Granger B, Limosin F, Cohen D, Martinot JL, Artiges E. Dynamic Functional Connectivity in Adolescence-Onset Major Depression: Relationships With Severity and Symptom Dimensions. Biol Psychiatry Cogn Neurosci Neuroimaging. 2022;7:385-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 11. | Hu L, Xiao M, Ai M, Wang W, Chen J, Tan Z, Cao J, Kuang L. Disruption of resting-state functional connectivity of right posterior insula in adolescents and young adults with major depressive disorder. J Affect Disord. 2019;257:23-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 45] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 12. | Black SE. Imaging white matter and the burden of small vessel disease. Brain Cogn. 2007;63:192-193. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Lan H, Suo X, Li W, Li N, Li J, Peng J, Lei D, Sweeney JA, Kemp GJ, Peng R, Gong Q. Abnormalities of intrinsic brain activity in essential tremor: A meta-analysis of resting-state functional imaging. Hum Brain Mapp. 2021;42:3156-3167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 14. | Zou QH, Zhu CZ, Yang Y, Zuo XN, Long XY, Cao QJ, Wang YF, Zang YF. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J Neurosci Methods. 2008;172:137-141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1781] [Cited by in RCA: 1610] [Article Influence: 94.7] [Reference Citation Analysis (0)] |
| 15. | Jiao F, Gao Z, Shi K, Jia X, Wu P, Jiang C, Ge J, Su H, Guan Y, Shi S, Zang YF, Zuo C. Frequency-Dependent Relationship Between Resting-State fMRI and Glucose Metabolism in the Elderly. Front Neurol. 2019;10:566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | Aiello M, Salvatore E, Cachia A, Pappatà S, Cavaliere C, Prinster A, Nicolai E, Salvatore M, Baron JC, Quarantelli M. Relationship between simultaneously acquired resting-state regional cerebral glucose metabolism and functional MRI: a PET/MR hybrid scanner study. Neuroimage. 2015;113:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 161] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 17. | Marchitelli R, Aiello M, Cachia A, Quarantelli M, Cavaliere C, Postiglione A, Tedeschi G, Montella P, Milan G, Salvatore M, Salvatore E, Baron JC, Pappatà S. Simultaneous resting-state FDG-PET/fMRI in Alzheimer Disease: Relationship between glucose metabolism and intrinsic activity. Neuroimage. 2018;176:246-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 18. | Zang Y, Jiang T, Lu Y, He Y, Tian L. Regional homogeneity approach to fMRI data analysis. Neuroimage. 2004;22:394-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1520] [Cited by in RCA: 1987] [Article Influence: 94.6] [Reference Citation Analysis (0)] |
| 19. | Pamilo S, Malinen S, Hlushchuk Y, Seppä M, Tikka P, Hari R. Functional subdivision of group-ICA results of fMRI data collected during cinema viewing. PLoS One. 2012;7:e42000. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 20. | La C, Mossahebi P, Nair VA, Bendlin BB, Birn R, Meyerand ME, Prabhakaran V. Age-Related Changes in Inter-Network Connectivity by Component Analysis. Front Aging Neurosci. 2015;7:237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 21. | Liu P, Tu H, Zhang A, Yang C, Liu Z, Lei L, Wu P, Sun N, Zhang K. Brain functional alterations in MDD patients with somatic symptoms: A resting-state fMRI study. J Affect Disord. 2021;295:788-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 72] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 22. | Nawaz H, Shah I, Ali S. The amygdala connectivity with depression and suicide ideation with suicide behavior: A meta-analysis of structural MRI, resting-state fMRI and task fMRI. Prog Neuropsychopharmacol Biol Psychiatry. 2023;124:110736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 23. | Nasrallah IM, Pajewski NM, Auchus AP, Chelune G, Cheung AK, Cleveland ML, Coker LH, Crowe MG, Cushman WC, Cutler JA, Davatzikos C, Desiderio L, Doshi J, Erus G, Fine LJ, Gaussoin SA, Harris D, Johnson KC, Kimmel PL, Kurella Tamura M, Launer LJ, Lerner AJ, Lewis CE, Martindale-Adams J, Moy CS, Nichols LO, Oparil S, Ogrocki PK, Rahman M, Rapp SR, Reboussin DM, Rocco MV, Sachs BC, Sink KM, Still CH, Supiano MA, Snyder JK, Wadley VG, Walker J, Weiner DE, Whelton PK, Wilson VM, Woolard N, Wright JT Jr, Wright CB, Williamson JD, Bryan RN; SPRINT MIND Investigators for the SPRINT Research Group. Association of Intensive vs Standard Blood Pressure Control With Cerebral White Matter Lesions. JAMA. 2019;322:524-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 301] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 24. | Zhao B, Li T, Yang Y, Wang X, Luo T, Shan Y, Zhu Z, Xiong D, Hauberg ME, Bendl J, Fullard JF, Roussos P, Li Y, Stein JL, Zhu H. Common genetic variation influencing human white matter microstructure. Science. 2021;372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 121] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 25. | Gawryluk JR, Mazerolle EL, Brewer KD, Beyea SD, D'Arcy RC. Investigation of fMRI activation in the internal capsule. BMC Neurosci. 2011;12:56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 26. | Mazerolle EL, D'Arcy RC, Beyea SD. Detecting functional magnetic resonance imaging activation in white matter: interhemispheric transfer across the corpus callosum. BMC Neurosci. 2008;9:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 27. | Ding Z, Newton AT, Xu R, Anderson AW, Morgan VL, Gore JC. Spatio-temporal correlation tensors reveal functional structure in human brain. PLoS One. 2013;8:e82107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 102] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 28. | Ding Z, Xu R, Bailey SK, Wu TL, Morgan VL, Cutting LE, Anderson AW, Gore JC. Visualizing functional pathways in the human brain using correlation tensors and magnetic resonance imaging. Magn Reson Imaging. 2016;34:8-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 29. | Marussich L, Lu KH, Wen H, Liu Z. Mapping white-matter functional organization at rest and during naturalistic visual perception. Neuroimage. 2017;146:1128-1141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 85] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 30. | Sun Y, Kong Z, Song Y, Liu J, Wang X. The validity and reliability of the PHQ-9 on screening of depression in neurology: a cross sectional study. BMC Psychiatry. 2022;22:98. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 31. | Kroenke K. PHQ-9: global uptake of a depression scale. World Psychiatry. 2021;20:135-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 32. | Spitzer RL, Kroenke K, Williams JB. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA. 1999;282:1737-1744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6086] [Cited by in RCA: 6826] [Article Influence: 262.5] [Reference Citation Analysis (0)] |
| 33. | Katz C, Roos LE, Wang Y, Bolton J, Hwang SW, Katz LY, Bourque J, Adair CE, Somers JM, Sareen J; At Home/Chez Soi Investigators. Predictive Validity of the MINI Suicidality Subscale for Suicide Attempts in a Homeless Population With Mental Illness. Suicide Life Threat Behav. 2019;49:1630-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 34. | Angst J, Adolfsson R, Benazzi F, Gamma A, Hantouche E, Meyer TD, Skeppar P, Vieta E, Scott J. The HCL-32: towards a self-assessment tool for hypomanic symptoms in outpatients. J Affect Disord. 2005;88:217-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 474] [Cited by in RCA: 476] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 35. | Yan CG, Wang XD, Zuo XN, Zang YF. DPABI: Data Processing & Analysis for (Resting-State) Brain Imaging. Neuroinformatics. 2016;14:339-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1581] [Cited by in RCA: 2656] [Article Influence: 295.1] [Reference Citation Analysis (0)] |
| 36. | Ma H, Xie Z, Huang L, Gao Y, Zhan L, Hu S, Zhang J, Ding Q. The White Matter Functional Abnormalities in Patients with Transient Ischemic Attack: A Reinforcement Learning Approach. Neural Plast. 2022;2022:1478048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 37. | Beckmann CF, DeLuca M, Devlin JT, Smith SM. Investigations into resting-state connectivity using independent component analysis. Philos Trans R Soc Lond B Biol Sci. 2005;360:1001-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2522] [Cited by in RCA: 2571] [Article Influence: 128.6] [Reference Citation Analysis (0)] |
| 38. | Himberg J, Hyvärinen A, Esposito F. Validating the independent components of neuroimaging time series via clustering and visualization. Neuroimage. 2004;22:1214-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 865] [Cited by in RCA: 932] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 39. | Wakana S, Caprihan A, Panzenboeck MM, Fallon JH, Perry M, Gollub RL, Hua K, Zhang J, Jiang H, Dubey P, Blitz A, van Zijl P, Mori S. Reproducibility of quantitative tractography methods applied to cerebral white matter. Neuroimage. 2007;36:630-644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1475] [Cited by in RCA: 1331] [Article Influence: 73.9] [Reference Citation Analysis (0)] |
| 40. | Hua K, Zhang J, Wakana S, Jiang H, Li X, Reich DS, Calabresi PA, Pekar JJ, van Zijl PC, Mori S. Tract probability maps in stereotaxic spaces: analyses of white matter anatomy and tract-specific quantification. Neuroimage. 2008;39:336-347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1302] [Cited by in RCA: 1191] [Article Influence: 70.1] [Reference Citation Analysis (0)] |
| 41. | Gazzaniga MS. Cerebral specialization and interhemispheric communication: does the corpus callosum enable the human condition? Brain. 2000;123:1293-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 815] [Cited by in RCA: 796] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 42. | Tamietto M, Adenzato M, Geminiani G, de Gelder B. Fast recognition of social emotions takes the whole brain: interhemispheric cooperation in the absence of cerebral asymmetry. Neuropsychologia. 2007;45:836-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 57] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 43. | Seltzer B, Pandya DN. Posterior parietal projections to the intraparietal sulcus of the rhesus monkey. Exp Brain Res. 1986;62:459-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 79] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 44. | Mayberg HS. Modulating dysfunctional limbic-cortical circuits in depression: towards development of brain-based algorithms for diagnosis and optimised treatment. Br Med Bull. 2003;65:193-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 807] [Cited by in RCA: 781] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 45. | Price JL, Drevets WC. Neurocircuitry of mood disorders. Neuropsychopharmacology. 2010;35:192-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1149] [Cited by in RCA: 1159] [Article Influence: 77.3] [Reference Citation Analysis (0)] |
| 46. | Caetano SC, Fonseca M, Hatch JP, Olvera RL, Nicoletti M, Hunter K, Lafer B, Pliszka SR, Soares JC. Medial temporal lobe abnormalities in pediatric unipolar depression. Neurosci Lett. 2007;427:142-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 47. | Zhang X, Yao S, Zhu X, Wang X, Zhong M. Gray matter volume abnormalities in individuals with cognitive vulnerability to depression: a voxel-based morphometry study. J Affect Disord. 2012;136:443-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 72] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 48. | Friedman A, Homma D, Gibb LG, Amemori K, Rubin SJ, Hood AS, Riad MH, Graybiel AM. A Corticostriatal Path Targeting Striosomes Controls Decision-Making under Conflict. Cell. 2015;161:1320-1333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 215] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 49. | Sisti HM, Geurts M, Gooijers J, Heitger MH, Caeyenberghs K, Beets IA, Serbruyns L, Leemans A, Swinnen SP. Microstructural organization of corpus callosum projections to prefrontal cortex predicts bimanual motor learning. Learn Mem. 2012;19:351-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 50. | Stave EA, De Bellis MD, Hooper SR, Woolley DP, Chang SK, Chen SD. Dimensions of Attention Associated With the Microstructure of Corona Radiata White Matter. J Child Neurol. 2017;32:458-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 51. | Karababa IF, Bayazıt H, Kılıçaslan N, Celik M, Cece H, Karakas E, Selek S. Microstructural Changes of Anterior Corona Radiata in Bipolar Depression. Psychiatry Investig. 2015;12:367-371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 52. | Fernández-Sevillano J, Alberich S, Zorrilla I, González-Ortega I, López MP, Pérez V, Vieta E, González-Pinto A, Saíz P. Cognition in Recent Suicide Attempts: Altered Executive Function. Front Psychiatry. 2021;12:701140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 53. | Schmahmann JD, Pandya DN, Wang R, Dai G, D'Arceuil HE, de Crespigny AJ, Wedeen VJ. Association fibre pathways of the brain: parallel observations from diffusion spectrum imaging and autoradiography. Brain. 2007;130:630-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 760] [Cited by in RCA: 762] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 54. | Caspers S, Zilles K. Microarchitecture and connectivity of the parietal lobe. Handb Clin Neurol. 2018;151:53-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 55. | Biesbroek JM, Kuijf HJ, van der Graaf Y, Vincken KL, Postma A, Mali WP, Biessels GJ, Geerlings MI; SMART Study Group. Association between subcortical vascular lesion location and cognition: a voxel-based and tract-based lesion-symptom mapping study. The SMART-MR study. PLoS One. 2013;8:e60541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 92] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 56. | Xia W, Zhou R, Zhao G, Wang F, Mao R, Peng D, Yang T, Wang Z, Chen J, Fang Y. Abnormal white matter integrity in Chinese young adults with first-episode medication-free anxious depression: a possible neurological biomarker of subtype major depressive disorder. Neuropsychiatr Dis Treat. 2018;14:2017-2026. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 57. | Zhuang L, Wen W, Zhu W, Trollor J, Kochan N, Crawford J, Reppermund S, Brodaty H, Sachdev P. White matter integrity in mild cognitive impairment: a tract-based spatial statistics study. Neuroimage. 2010;53:16-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 96] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 58. | Zou Q, Wu CW, Stein EA, Zang Y, Yang Y. Static and dynamic characteristics of cerebral blood flow during the resting state. Neuroimage. 2009;48:515-524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 163] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 59. | Li C, Dang C, Liu G, Chen L, Zhang J, Li J, Ou Z, Zhang Y, Xu A. Secondary damage in left-sided frontal white matter detected by diffusion tensor imaging is correlated with executive dysfunction in patients with acute infarction at the ipsilateral posterior corona radiata. Eur J Med Res. 2014;19:44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 60. | Poletti S, Mazza E, Bollettini I, Falini A, Smeraldi E, Cavallaro R, Benedetti F. The COMT Val158Met polymorphism moderates the association between cognitive functions and white matter microstructure in schizophrenia. Psychiatr Genet. 2016;26:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 61. | Kodl CT, Franc DT, Rao JP, Anderson FS, Thomas W, Mueller BA, Lim KO, Seaquist ER. Diffusion tensor imaging identifies deficits in white matter microstructure in subjects with type 1 diabetes that correlate with reduced neurocognitive function. Diabetes. 2008;57:3083-3089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 149] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 62. | Barrett EA, Sundet K, Simonsen C, Agartz I, Lorentzen S, Mehlum L, Mork E, Andreassen OA, Melle I. Neurocognitive functioning and suicidality in schizophrenia spectrum disorders. Compr Psychiatry. 2011;52:156-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 63. | Davey DK, Jurick SM, Crocker LD, Hoffman SN, Sanderson-Cimino M, Tate DF, Velez CS, Delano-Wood L, Jak AJ. White matter integrity, suicidal ideation, and cognitive dysfunction in combat-exposed Iraq and Afghanistan Veterans. Psychiatry Res Neuroimaging. 2021;317:111389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 64. | Long Y, Ouyang X, Liu Z, Chen X, Hu X, Lee E, Chen EYH, Pu W, Shan B, Rohrbaugh RM. Associations Among Suicidal Ideation, White Matter Integrity and Cognitive Deficit in First-Episode Schizophrenia. Front Psychiatry. 2018;9:391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 65. | Goh S, Bansal R, Xu D, Hao X, Liu J, Peterson BS. Neuroanatomical correlates of intellectual ability across the life span. Dev Cogn Neurosci. 2011;1:305-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 66. | Pessoa L. A Network Model of the Emotional Brain. Trends Cogn Sci. 2017;21:357-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 221] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 67. | Gawryluk JR, Mazerolle EL, D'Arcy RC. Does functional MRI detect activation in white matter? A review of emerging evidence, issues, and future directions. Front Neurosci. 2014;8:239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 140] [Cited by in RCA: 192] [Article Influence: 17.5] [Reference Citation Analysis (0)] |