Published online Jan 19, 2022. doi: 10.5498/wjp.v12.i1.24
Peer-review started: March 31, 2021
First decision: July 15, 2021
Revised: August 1, 2021
Accepted: December 21, 2021
Article in press: December 21, 2021
Published online: January 19, 2022
Processing time: 292 Days and 12.6 Hours
We review the still scarce but growing literature on resilience to the effects of social stress on the rewarding properties of drugs of abuse. We define the concept of resilience and how it is applied to the field of drug addiction research. We also describe the internal and external protective factors associated with resilience, such as individual behavioral traits and social support. We then explain the physiological response to stress and how it is modulated by resilience factors. In the subsequent section, we describe the animal models commonly used in the study of resilience to social stress, and we focus on the effects of chronic social defeat (SD), a kind of stress induced by repeated experience of defeat in an agonistic encounter, on different animal behaviors (depression- and anxiety-like behavior, cognitive impairment and addiction-like symptoms). We then summarize the current knowledge on the neurobiological substrates of resilience derived from studies of resilience to the effects of chronic SD stress on depression- and anxiety-related behaviors in rodents. Finally, we focus on the limited studies carried out to explore resilience to the effects of SD stress on the rewarding properties of drugs of abuse, describing the current state of knowledge and suggesting future research directions.
Core Tip: Preclinical research on drug addiction has focused on the factors that enhance vulnerability to develop drug addiction. Recent studies of resilience have determined the neurobehavioral traits that confer protection against developing an addictive disorder after stress exposure. Active coping strategies to face the stressor and the absence of depression-like symptoms are consistently associated with resilience to the stress-induced potentiation of the rewarding effects of cocaine and alcohol. Unravelling the neurobiological substrates of resilience is key to developing pharmacological and psychological interventions to enhance stress resilience in order to prevent the development of addiction and other stress-related disorders.
- Citation: Calpe-López C, Martínez-Caballero MA, García-Pardo MP, Aguilar MA. Resilience to the effects of social stress on vulnerability to developing drug addiction. World J Psychiatry 2022; 12(1): 24-58
- URL: https://www.wjgnet.com/2220-3206/full/v12/i1/24.htm
- DOI: https://dx.doi.org/10.5498/wjp.v12.i1.24
The noun resilience derives from the Latin resiliens, the present participle of resilire (re- "back" + salire "to jump"), and was first used by Cicero and Francis Bacon (among others) as a synonym of rebound[1]. From the nineteen century on, material science has also used the word resilience to indicate the flexibility of a material or its ability to resist stress (force being applied) without permanent deformation. In the context of psychology, resilience can be defined as “the process of adapting well in the face of adversity, trauma, or other significant sources of stress”[2,3]. Besides the rebound of the equilibrium, resilience often implies an increase in mental resistance.
Although resilience is sometimes considered an extraordinary capacity of some individuals, research indicates that it is a common trait. The majority of individuals exposed to trauma or stressful events adapt to and overcome stress and maintain normal psychological and physical functioning without developing stress-related disorders[4]. Approximately 50% of people experience trauma in their life, but the prevalence of post-traumatic stress disorder (PTSD) is about 8%[5]. Resilience is an innate capacity, although it is not a stable trait, it is a dynamic process[6,7] that changes through a life span and can be enhanced by different factors.
Most research on resilience has focused on the biological and behavioral profile of individuals who are resilient to developing psychiatric illnesses such as depression and PTSD after exposure to stress. However, studies on resilience to the effects of stress on the initiation, maintenance and relapse to addictive disorders are very limited. In fact, almost all research regarding substance use disorders (SUD) has focused on risk; i.e., the factors that predispose an individual to develop an addictive disorder. Vulnerability to the effects of drugs of abuse depends on multiple factors, including biologic factors such as genetic load, which are modified by life experiences and the environment in which the individual lives. Stressful experiences have a profound impact on the brain[8], for this reason, stress can increase vulnerability to addiction. Exposure to stress, especially in early life and adolescence, induces long-term modifications in the physiological response to stress, emotional reactivity, the brain reward system and cognitive processing, all of which contribute to the increased vulnerability to develop a SUD[9]. However, as commented on before, most people are resilient to stress. Consequently, only a small percentage of individuals that experience a traumatic event or are exposed to chronic stress develop an addictive disorder.
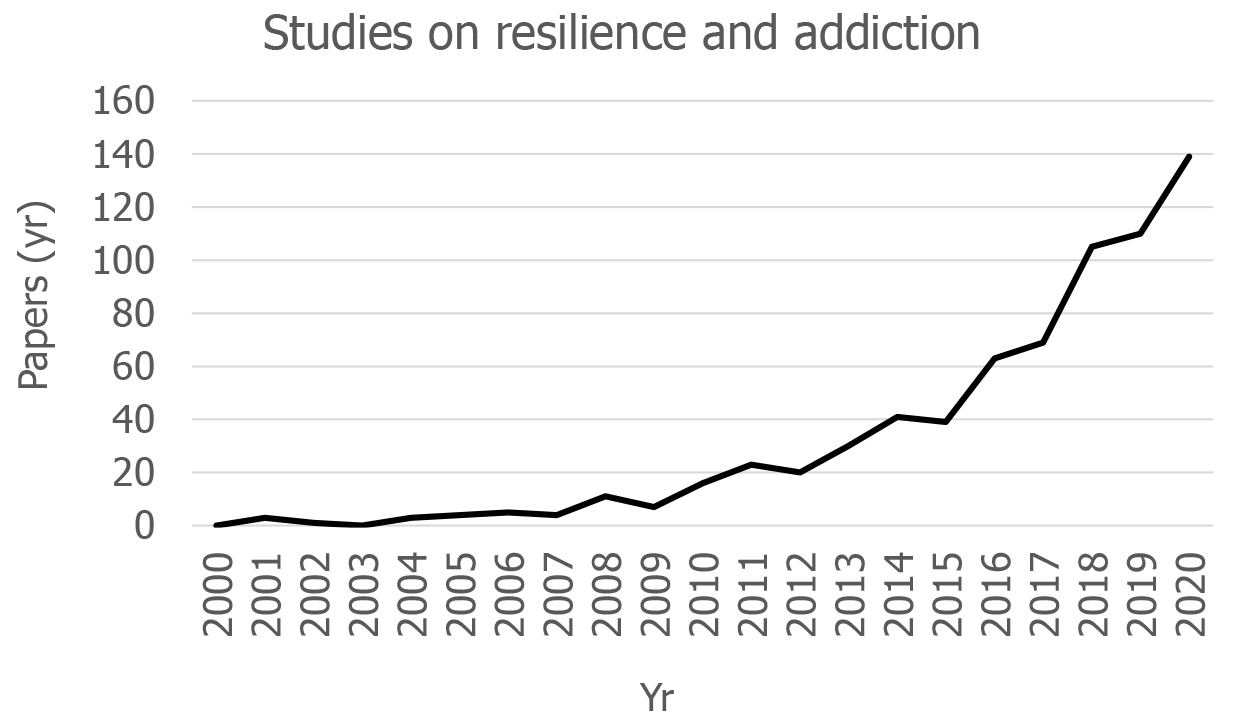
In recent years there has been an important impulse in the study of resilience to develop a SUD or an addictive behavior. In fact, until 2010, literature related to resilience and addiction was scarce, while in the last ten years the number of works on the subject has increased exponentially (Figure 1). Epidemiologic studies indicate a clear association between low resilience (often during adolescence) and the increment of addictive behaviors[10,11]. Resilience is a factor moderating the relationship between stress and alcohol use disorders (AUD)[12] and is strongly associated with a reduction in the risk of AUD, though there is not a direct causal relationship but rather an overlapping of genetic and environmental influences[13]. Animal models used to study the impact of stress on drug addiction[14,15] are also being incorporated into research to identify the behavioral and physiological traits that characterize animals’ resilience to the effects of stress on the rewarding properties of drugs of abuse, as well as the neurobiological substrates of the resilience process.
Addressing the perspective of resilience in the study of addictive behaviors is promising as a way of enhancing knowledge regarding the neuroscience of addiction. However, as Rudzinski et al[16] noted, there are difficulties in the use of the concept of resilience in the field of drug addiction, especially regarding its definition and operationalization as a trait, as a process or, as is more common, as an outcome (for example, the absence of SUD). In addition, it is important to distinguish between resilience (a concept with multiple meanings) and resiliency, which is a personality trait that has been linked to alcohol/drug problems and is defined as “the ability to flexibly adapt impulse control relative to contextual demand”[17]. Some studies define resilience as the capacity to maintain abstinence and not relapse to drug use during a recovery period[18,19]. In this sense, neuroimaging studies have shown that conserved prefrontal cortex (PFC) morphology and heightened neural PFC engagement are linked to abstinence and resilience against relapse in alcohol-dependent patients[19]. Other studies consider drug use as a stressor or risk factor for resilience (for example[20]), while some studies do not evaluate resilience to stress. In the last case, the concept of resilience is used to design a reduced response to the drugs of abuse in rodents that have been exposed to a genetic or pharmacological manipulation[21,22].
In the present review we mainly focus on research that has studied resilience to the effects of stressful experiences on subsequent drug use/abuse in animal models. First, we succinctly comment on the protective factors associated with resilience in studies with humans and explain the relationship between the physiological response to stress and resilience, since most human studies have focused on the neuroendocrine changes that are predictive of resilience. We then discuss the main animal models used to study resilience to social stress and review advances concerning the neurobiological substrates of resilience in said studies. Finally, we discuss research that specifically addresses resilience to the effects of repeated social defeat (SD) on the rewarding properties of drugs of abuse and lay out future research directions and conclusions.
Different protective factors associated with resilience can be identified on biological, psychological, and social levels. Among the internal factors are stable predispositions (such as genotype or personality traits) and the influence of skills or capacities acquired through interaction with stressors (emotion-regulation abilities, appraisal styles, etc.). Resilient people are more prone to experience positive emotions, realistic optimism, cognitive reappraisal (ability to replace negative thoughts with more positive ones), secure attachment, an active coping with stress, high coping self-efficacy, self-esteem, empathy, prosocial behavior and altruism, a healthy lifestyle (for example, physical exercise) and a sense of coherence (moral compass that gives meaning of life)[4,16,23,24].
There are also external factors related with resilience at three levels[16]: Family (parental supervision, setting boundaries, bonding, support, etc.), school (positive environment, good relationships with teachers and peers, school engagement and extra-curricular activities involvement, etc.) and community (positive relationships with friends or neighbors, participation in religious practices, community engagement, community support, etc.). All these internal strengths and external resources help to prevent maladaptive responses to adversity[9]. Longitudinal studies have indicated several key factors related with resilience and a successful transition from childhood and adolescence to adulthood, such as social support (family, peer relationships, romantic partners, etc.), self-discipline, and good cognitive and executive functioning (planification, cognitive flexibility, etc.)[25]. Children exposed to war show increased risk of PTSD in adulthood, but some protective factors against the deleterious impact of war have been identified, including a loving and supportive environment (family, peers, teachers, etc.), a shared sense of values and religious beliefs, positive thinking and generosity[26]. Similarly, in patients with psychiatric disorders (depression and/or anxiety), factors predictive of low resilience include lack of purpose in life, less frequent physical exercise and low spirituality[27]. A study with fire-workers indicated that the trait of mindfulness (concentration on and moment-to-moment awareness of bodily activities and feelings) contributed to resilience, thus reducing avoidant coping in response to stress. Fire-workers with this trait reported less alcohol problems and reduced physical, depressive and PTSD symptoms[28].
As mentioned before, resilience is a dynamic process that raises individuals up from life’s adversities and allows them to successfully overcome stressful events. The phenomenon known as “stress inoculation” occurs when a person exposed to mild or moderate stressors develops an adaptive stress response and shows a higher resilience to the negative effects of a variety of subsequent stressors[5,29]. As demonstrated by the group of McEwen, the behavioral effects of stress follow an inverted U–shape curve; low and high stress levels induced impairing effects, but intermediate levels promote better coping responses[30]. In the same way as a vaccine induces immunity against disease[2], stress inoculation is a form of immunity or protection against later stress that may be a result of neuroplasticity in the PFC[29]. The “Systematic Self-reflection model” proposes that engaging with moderate stressors can have positive consequences on mental health if scaffolded in self-reflection, a meta-cognitive skill (consisting of an honest reflection on the individual’s coping and emotion regulatory practices) that leads to a cognitive maturity and on-going adaptation of the capacity of resilience[31]. On the other hand, substance use and other adjustment problems (depression, anxiety, rule-breaking, etc.) have been observed in adolescents from affluent families that have not been exposed to identified stressful experiences. These individuals are now considered as a group at risk that needs to build resilience through positive changes in parenting, construction and maintenance of supportive social networks, promotion of coping self-efficacy and self-esteem, etc[32].
Exposure to a physically or psychologically stressful stimulus immediately activates a physiological response characterized by a cascade of hormones in the hypothalamus-pituitary-adrenal (HPA) axis that prepare the body for fight or flight. The paraventricular nucleus of the hypothalamus releases corticotropin-releasing factor (CRF), which leads to the release of adrenocorticotropic hormone (ACTH) by the adenohypophysis, which in turn stimulates the release of glucocorticoids (cortisol in humans and corticosterone in rodents) by the cortex of adrenal glands. There are negative feedback mechanisms in the HPA axis; for example, glucocorticoids suppress CRF and ACTH production. In addition, stress activates the sympathetic nervous system (SNS), which induces the adrenomedullary release of noradrenaline (NA). Stress also stimulates the brain’s noradrenergic system, resulting in the release of NA from the locus coeruleus (LC) to the amygdala, hippocampus, hypothalamus and PFC[33,34]. Dopamine (DA) release is also altered by stress, with an increase in the PFC and a reduction in the nucleus accumbens (NAcc)[35], and acute stress increases serotonin turnover in the amygdala, hippocampus, PFC and NAcc[36-38], although other studies have shown a lack of an effect of acute stress on serotonin turnover in the amygdala, NAcc, striatum[39] and hypothalamic paraventricular nucleus[38].
Glucocorticoid elevation may cause damage and atrophy of neurons in different brain areas involved in memory and emotional behavior, such as the hippocampus and amygdala, inducing physical and psychological problems. Moreover, chronic stress interferes with the activity of neurotrophic factors that are responsible for the formation and strengthening of new neurons and synaptic connections, especially in the hippocampus, such as brain-derived neural factor (BDNF). The volume of this structure and the levels of BDNF are reduced in subjects exposed to prolonged stress, which could be a risk factor for the development of PTSD[40-42]. Resilience can avoid these negative effects of stress, for example, through the release of substances that block the physiological stress response. Neuropeptide Y (NPY) and dehydroepiandrosterone (DHEA) counteract CRF and cortisol, respectively[43,44]. Higher levels of NPY in response to acute stress predict less psychological distress and fewer symptoms of dissociation[45]. Furthermore, the brain of resilient people produces more BDNF, which also decreases levels of glucocorticoids in the hippocampus, and BDNF-mediated plasticity increases attention and memory and accelerates recovery from adversity. Resilient people have been shown to exhibit an adaptive stress response, rapid stress recovery (levels of cortisol decreasing fast after adversity) and lower susceptibility to stress-related physical and mental pathology[4].
There is an interface between the endocrine stress response and the immune system. Communication between neural, hormonal and immune systems is mediated by cytokines and chemokines, small molecules that mediate inflammatory processes, corticosteroids, pituitary hormones, catecholamines and neuropeptides[46,47]. Feedback between the peripheral immune system and the brain contributes to individual differences in the behavioral response to stress[48,49]. Resilient subjects display reduced neuroinflammation, which facilitates habituation to and recovery from stressful events and explains the lower incidence of medical and psychiatric diseases amongst these individuals[49,50,51]. Resilient people have lower systemic inflammation, and the psychosocial factors associated with resilience mitigate the impact of stress on systemic inflammation[51]. These bidirectional relationships between resilience and immunity are modulated by the gut microbiota[52]. There is an interaction between the gut and the brain that involves neural, endocrine, and immune pathways. It seems that the stress-induced activation of the HPA axis stimulates the immune system and causes changes in microbial diversity[53]. The gut microbiota has been associated with a wide range of physiological processes, including the response to stress[54]. Oral intake of Bifidobacterium was shown to significantly increase the number of mice that were resilient after repeated SD stress with respect to control animals not receiving treatment[55]. Moreover, administration of Lactobacillus was found to decrease anxiety-like behavior induced by repeated SD stress and to improve the immune response[56].
Animal models are necessary to understand the different aspects of human resilience, such as physiological or behavioral changes. As mentioned before, after exposure to stress, some humans develop a psychopathological disorder, such as depression or anxiety, while others are resilient to such effects. These disorders are complex and multifactorial and affect many aspects of human life; thus, no animal model can mimic the complexity of human disorder. However, animal models are useful for simulating some of the psychiatric symptoms[57] or behavioral dimensions that characterize a disorder[58]. After exposure to chronic stress, some animals develop depression– and anxiety–like symptoms and other behavioral alterations (susceptible or vulnerable animals), while others exhibit clear resistance to at least some of the maladaptive sequelae of stress (resilient animals). In addition, animal models also contribute to our understanding of the mechanisms underlying the development of resilience, such as the therapeutic effects of the inoculation of stress[59].
In this section, we first describe the animal models and behavioral tests used to study resilience to the symptoms of stress-related disorders, such as anxiety, depression, cognitive impairment or drug addiction and then the models used to induce stress in experimental animals. This is not an exhaustive review of these models, but only a brief description of the main paradigms used in preclinical studies of resilience. We focus on the model of SD stress in rodents, and on the behavioral paradigms that have been used to evaluate its short- and long-term consequences.
There are multiple techniques to induce stress in experimental rodents. Some of them use pharmacological stressors, such as daily administration of corticosterone[60], or physical stressors, such as restraint or immobilization[61]. Another model is based on a combination of physical and psychosocial stressors (chronic unpredictable stress (UCS) or chronic “mild” stress (CMS) paradigm)[62]. In the CMS, most animals (about 70%) show anhedonia–like symptoms (less sucrose consumption), reduction of hippocampal volume and alterations in glutamate metabolism, although there is a subset of resilient animals that do not exhibit these changes[62]. Resilience to stress has also been studied with the model known as “predator odor”, in which the stress response is induced by exposing animals to the odor of a predator[63]. Usually, rats are classified into 3 groups according to the number and type of behavioral deficits observed as extremely, partially, or minimally disrupted. Anxiety–like symptoms, increased acoustic startle responses and reductions in NPY are observed in animals that are extremely disrupted, while partially and minimally disrupted animals exhibit mixed deficits within these domains[63].
The paradigm of learned helplessness is an animal model of depression that is also employed to induce stress and study resilience by exposing animals to the stress induced by an inescapable, unpredictable and uncontrollable foot shock[60,64,65]. After such exposure to stress, a subset of susceptible animals develops learned helplessness (coping deficits to deal with the inescapable shocks), while another subset of resilient animals displays escape responses with latencies similar to those of non-stressed animals[64]. Results are in function of the severity, duration and control over cessation of the footshock, the last variable of which promotes resilience[65].
As commented on before, in the present work we focus on the model of chronic SD stress because it is the most used animal model to study resilience to the effects of stress and has more ethological and ecological validity. In fact, the most frequent type of stress faced by humans is the chronic social stress derived from problems with social interaction (family or friend relationships, work-place stress, bullying, etc.). In the chronic SD model, brief episodes of aggression from a more aggressive conspecific in the resident-intruder paradigm result in the defeat of the experimental animal (intruder), which usually shows anxiety- and depression-like symptoms[15,66-69]. In the most widely employed SD model, rats or mice are exposed to SD for 10 days. Each day, the experimental animal undergoes 10 min of physical attack by the aggressive opponent, followed by 24 h of sensory contact. The consequences of this kind of stress are also a function of the severity and duration of the defeat episodes but chronic SD exposure induced an escalation of cocaine and alcohol consumption. To study resilience, genetically inbred C57BL6/J male mice are usually employed. Following chronic SD stress, all mice exhibit heightened reactivity of the HPA axis, deficits in exploration (interpreted as increased anxiety) and polydipsia[70]. However, there are differences between susceptible and resilient mice regarding other consequences of chronic SD. Resilient mice do not exhibit social avoidance, hyperthermia elicited by social interactions, anhedonia–like symptoms, or metabolic syndrome, characterized by over–eating, obesity, and leptin resistance[70,71]. Approximately 35% of C57BL6/J mice are resilient, although the relative distribution of resilience differs across strains[72]. Similarly, wild-type Groningen rats have better coping strategies and are more resilient to SD stress than Wistar rats[73].
A variation of the classical 10-day SD paradigm consists of exposing animals to intermittent repeated SD (IRSD); usually, four episodes of defeat separated by intervals of 72 h. The IRSD model is frequently employed in studies on the influence of social stress on vulnerability to developing drug addiction. Exposure to IRSD has also been shown to increase the rewarding effects of drugs of abuse[14,74,75,76]. In our laboratory, mice exposed to IRSD during adolescence or adulthood exhibit a long-term enhanced sensitivity to the rewarding effects of drug of abuse such as cocaine and MDMA[77,78,79].
To study the phenomenon of “stress inoculation” several types of moderate stressors have been used, including exposure to intermittent foot shocks[80] and brief intermittent maternal separations during early periods of life[81] or a combination of maternal separation and UCS[81]. Infant rats exposed to intermittent foot shocks subsequently respond more effectively than non-stressed control rats when confronted with novel situations[80]. The combination of maternal deprivation during early life with UCS during adolescence promotes greater resilience in adulthood than maternal deprivation alone or when combined with UCS[81].
During chronic exposure to stress, behavioral strategies that limit the experience of stress may promote resilience[5]. During chronic SD, animals that engage in less submissive postures when threatened and attacked by the opponent show less social avoidance, suggesting that this behavioral coping strategy reduces the effects of the stress[82]. Behavioral manipulations have also been used to reduce the effects of stress and increase resilience; for example, exposure to physical exercise[83,84] or environmental enrichment[85].
The forced swim test (FST) is a classic behavioral test of depression-like symptomatology in which animals are placed into a cylinder filled with water and forced to swim during a period lasting a few minutes. Initially animals attempt to escape and swim, but afterwards they stop fighting and become passive. Immobility (passive floating with minor movements necessary to keep the head above water) is interpreted as a failure to persist in escape-directed behavior, hopelessness, negative mood and depressive-like behavior. The FST is frequently used to evaluate resilience since SD increases immobility in this test in susceptible but not in resilient animals[60,86,87,88]. Similar to the FST, the tail suspension test (TST) measures immobility, which is considered to represent despair and depressive-like behavior[89]. Rodents are hung in an uncontrollable fashion by their tail for a few minutes[90] and, after initial escape-oriented movements, develop an immobile posture. The effects of SD exposure in the TST are unclear, and it has been suggested that this paradigm models the stress-coping strategy from which depressive-like behavior is inferred[91]. An increase in immobi
Anhedonia– or depressive-like symptoms are also frequently evaluated by measuring sucrose consumption. During training, after some hours of food and water deprivation, a bottle containing a sucrose solution is made available in the home cage. Sucrose intake is measured at different intervals during stress exposure and is reduced in vulnerable but not resilient stressed animals[70,71,88,94,95]. The splash test consists of spraying a 10% sucrose solution on the dorsal coat of a rodent to stimulate grooming behavior. An increase in the latency of grooming and a decrease in the time and/or frequency of grooming is interpreted as depressive-like behavior[96]. This test has also been used to evaluate resilience to the consequences of SD stress[60,93].
In the social interaction test, animals are placed within an open field in two trials (2.5-10 min), in the absence (no target) and presence (target) of a conspecific animal contained in a perforated Plexiglas cage, in order to allow for social interaction while preventing confrontation. Social avoidance is considered to take place when the experimental animal spends less time in the area immediately surrounding the enclosure containing the opponent (interaction zone) and more time in the corners of the open field. Social avoidance is associated with depressive-like behaviors and is frequently observed after SD exposure in susceptible but not in resilient animals[70,71,93,97,98].
The novelty suppressed feeding test is based on the innate fear of rodents of novelty and the inhibition of feeding behavior when exposed to a novel environment. Animals’ access to food is restricted for 12-24 h. Animals are placed in a corner of a box containing a pellet of food and the latency to begin eating is recorded. Immedi
The elevated plus maze (EPM) is one of the most used paradigms to measure anxiety in rodents. This test is based on the natural aversion of rodents to open elevated areas and the exploratory behavior that they exhibit in novel environments. The apparatus, elevated about 50 cm above floor level, consists of two open arms and two enclosed arms, and the junction of the four arms forms a central platform. Subjects are placed on the central platform and allowed to explore the maze for 5 min. The total time spent in and the number of entries into the open (and closed) arms, and the percentage of time and entries into the open arms are measured. Anxiety levels are considered to be lower when the measurements in the open arms are higher and those in the closed arms are lower, and vice versa[100,101]. Mice exposed to chronic SD exhibit higher anxiety levels in this paradigm[93,97,102]. The EPM is also frequently used in studies of resilience to the effects of social stress on anxiety[60,92,102]. In a recent study in our laboratory, we observed that mice that were resilient to the effects of stress on cocaine reward spent less time in the open arms[93].
In the open field/exploration test, the animal is placed into an open-field arena for several min and its locomotor activity is evaluated by measuring distance travelled and velocity. A reduction of these measures is indicative of anxiety[103]. Sometimes the open field is divided into a center and a surrounding area, with thigmotaxis being indicative of anxiety. Maternal separation decreases the time that mice remain in the center of the open field[104]. SD induces deficits in exploration that are not observed in resilient animals[60,70,71,102].
The hole-board test is used to evaluate anxiety-related and novelty-seeking behavior of rodents. This test is carried out in a square box with equidistant holes in the floor. The animal is placed in a corner of the box and is allowed to freely explore the apparatus for a few minutes. Head-dipping represents exploratory tendencies that are distinct from general locomotor activity; thus, the latency to perform the first head-dip and the frequency of dips is recorded. Stress exposure elevates anxiety-related behavior in the hole-board test in rats and mice[105,106]. In our laboratory, we have observed that mice with low novelty-seeking are resilient to the effects of SD on cocaine reward[93].
The novel object recognition test evaluates episodic memory in rodents[107] and has been used to measure cognitive dysfunction according to deficits in object-context identification[108]. The task is performed in an open field box and consists of three phases: habituation (free exploration of the empty box), training (exploration of the box, which contains two small river stones) and test (one of the stones is replaced with a small plastic toy). In the training and test sessions, separated for a memory retention interval (1 min), the exploration of the objects is measured for 3 min. It is assumed that if the animal recognizes the stone, it has spent more time exploring the new object. Exposure to different paradigms of stress induces cognitive deficits in recognition memory[94,104,105,109]. Acute[110] and chronic[97] SD impairs performance of the object recognition task. This task has also been used to study resilience to the impairing effects of social stress on cognition[88,94,97,111,112].
The Morris water maze task measures spatial memory that is dependent on the hippocampus[113]. The apparatus consists of a circular swimming pool, divided into 4 equal quadrants (NW, NE, SE and SW), with an escape platform placed 1 cm below the water surface. Several visual cues surrounding the maze are placed on the walls. During the training phase the animal is placed in the water inside one of the quadrants and allowed to swim freely until it locates and climbs onto the platform. If the animal fails to locate the platform, it is guided to the platform by the experimenter and allowed to stand on it for several seconds. The training is performed over 4-5 consecutive days (3 trials per day), measuring the escape latency (the time taken to locate the platform in each trial). The test is performed 24 h after the last training session (the platform is removed and the time spent in each quadrant is measured). If the animal recalls the placement of the hidden platform it will spend more time in that quadrant. Unpredicted CMS impairs performance of the water maze[114], but chronic SD stress does not affect this task[102]. The water maze has also been used to study resilience to the effects of stress on cognitive processes[114,115]. An interesting study[115] showed that rats that emitted ultrasonic vocalizations during intermittent swim stress later showed resilience in the Morris water maze and an instrumental swim escape test.
The Y-maze is a spatial task that requires intact hippocampal function[116]. The Y-maze apparatus has three identical and symmetrical arms that radiate out from the center. Explicit cues are presented outside the maze (located on the walls around the room). In the first trial, the animal is placed in one arm, designated as the “start” arm, while another arm is blocked so that the animal can only explore the start and the other arm. After 4 h, in the second trial, animals are placed in the start arm and can freely explore all three arms. The number of entries and the time spent in each arm is measured. If the animal recalls the arms previously explored in trial 1, it will spend more time in the “novel” arm in trial 2 (discrimination performance). CMS induces deficits in the performance of the Y-maze among vulnerable anhedonic-rats[88]. Acute[110] but not chronic[117] SD stress also impairs performance in the Y-maze. However, the combination of chronic SD with a slight peripheral infection (produced by injection of a sub-threshold of LPS) impairs the performance of susceptible mice in the Y-maze[111].
The radial arm maze is a model of hippocampus-dependent memory. Animals are food-restricted (approximately 85.0% of their previous body weight) and pre-trained to associate the maze with a food reward placed at the end of all 8 arms. Subsequently, the animals are trained for several consecutive days. In each trial the animal is placed in the central chamber of the maze for habituation and can then freely explore the arms until it consumes all food reward or until a maximum time. The measurement of memory is the number of errors committed, defined as entries in a previously visited arm[118,119]. Chronic stress induced by visual and olfactory exposure to a predator (Long Evan rat) without direct physical contact impairs performance in the radial maze[118]. Similarly, maternal separation induces an overall impairment in the performance of the radial maze in adulthood; however, this impairment is observed in susceptible, but not in resilient mice[119]. On the other hand, adult rats exposed to maternal deprivation perform better in the radial maze, an effect probably related with the phenomenon of inoculation of stress[120].
The radial arm water maze also evaluates spatial ability in rodents[121,122,123]. In this case, the radial arm maze is filled with cloudy water to conceal a platform placed in one of the eight arms, and there are prominent extra-maze cues on the walls of the room. The animals perform several trials in three days, which consists of placing the animal into an arm (the start arm, which does not contain the platform). When the animal reaches the hidden platform it remains on it for several seconds to visualize the room spatially. If the animal fails to find the hidden platform, it is guided there by the experimenter. The number of entrances is measured in each trial. Two types of errors are considered in each trial; reference memory errors (number of first-time entries into arms that did not contain the platform) and working memory errors (number of repeat entries into an arm that did not contain the platform). Chronic restraint stress impairs radial arm water maze performance[122,123], but this effect recedes with time[124] and is prevented by environmental enrichment[85].
It is important to note that chronic stressors do not affect the performance of females in most of these tests (spatial object recognition, radial arm maze, Morris water maze, Y-maze), while males show stress-induced impairments in all of them[125]. These sex-dependent differences include the use of different strategies by the sexes to solve cognitive tasks and may be related to estradiol levels[87].
The animal models of drug reward and addiction-like symptoms are essential to progress in understanding the biological basis of SUD and for the identification of new therapeutic targets. Drug addiction is a neuropsychiatric disorder characterized by loss of control over drug-seeking and drug-taking, the presence of a negative emotional state and an intense craving for the drug when it is not available, and a high propensity to relapse even after long-term periods of abstinence[126]. Drug addiction represents a profound disruption of different neural circuits, including a deficit of the brain reward system, an over-activation of the stress systems, aberrant associative learning (which confers exaggerated incentive salience to stimuli or contexts associated with the drug), and a dysfunction of the PFC, resulting in the inability to inhibit drug-taking behavior. The transition from an initial recreational and controlled drug use to compulsive consumption is also related with a change from the ventral to the dorsal striatum in the control of drug use behavior, with the consequent development of rigid stimulus-response habits[127,128].
Drug addiction has a multifactorial nature, since environmental and biological factors interact to confer vulnerability or resilience to the development of this disorder. The complexity of addictive behavior cannot be captured by an animal model, but they are useful in modelling some specific aspects of drug addiction. The two main models to study vulnerability or resilience to drug addiction are the self-administration (SA) paradigm, which is based on the primary hedonic effects produced by the consump
The intravenous SA paradigm is the most important procedure for assessing the primary intrinsic reinforcing effect of drugs, and is the most commonly used in rodents[129,130]. In this paradigm animals are trained in daily sessions to obtain the drug by performing an operant response; for example, by pressing a lever or performing a nose-poke. This response is reinforced by injection of the drug, usually according to a fixed response (FR) program in which the animal must perform a fixed number of responses in order to obtain the dose of the drug. Variable or progressive response programs are also used to measure motivation of the animal for the drug. The oral SA paradigm, frequently used for alcohol, is similar regardless of the way in which the substance is ingested by the animal. Pharmacological and methodological factors may influence the results obtained with the SA paradigm, such as the drug, dose and rate of infusion, duration of the SA session, the requirements of response, the sex and age of the animal, etc.
The SA paradigm is also used to study extinction and reinstatement of drug-seeking behavior. During the extinction phase, the drug of abuse is not presented after responding, and as a consequence, a progressive decrease in the operant response takes place[131-133]. When extinction has been completed, reinstatement of the operant response by several stimuli is observed. Reinstatement of drug SA is a model of relapse to drug consumption after a period of abstinence. As in humans, administration of the drug of abuse (priming), re-exposure to drug-associated stimuli, or exposure to stress reinstates the initially learned operant response[134]. Indeed, some researchers have adapted the SA paradigm in order to model the main features of addiction in humans based on the DSM-5 criteria: loss of control or persistence in drug seeking (active responses during periods in which the reinforcer is not available), high motivation for the drug (using a progressive reinforcement schedule), and mainte
Using the SA paradigm, it has been demonstrated that exposure to social stress increases the reinforcing effects of drugs of abuse[136-139]. Recently, resilience to these effects has also been studied using different types of social stress and drugs of abuse, such as cocaine[140-143], methamphetamine[144] and alcohol[145-147].
The CPP is a paradigm that evaluates the conditioned rewarding effects of a drug of abuse, since some contextual stimuli acquire appetitive properties when associated with the drug[148-151]. This paradigm is characterized by its methodological simplicity and is thus frequently used. Animals are conditioned in a box with two or three compartments that are clearly distinct in terms of the stimuli present in each compartment; for example, they have different colored walls and floor textures. Before conditioning, a pre-conditioning phase takes place to evaluate the time spent by the animal in each compartment without any treatment. During conditioning the animal receives the drug (usually 4 injections in 4 or 8 days) in a specific compartment (without access to the other compartment) and physiological saline in the opposite compartment. Later, in the post-conditioning phase (equal to pre-conditioning) it is evaluated whether the animal has learned to associate the rewarding effects of the drug with the environmental cues present in the drug-paired compartment. If the animal spends more time in this compartment (in comparison to the time spent in pre-conditioning or to the time spent in the saline-paired compartment), it is considered that the animal has acquired CPP. All drugs abused by humans induce CPP in rodents[150].
As described for the SA paradigm, the CPP procedure can also be used to evaluate other processes besides acquisition, such as extinction and reinstatement of motivated behavior[148]. To induce extinction, animals are placed in the CPP box and perform daily or weekly sessions similarly to pre- and post-conditioning (i.e., they are exposed to the previously drug-paired compartment without administration of the drug). Progressively, the association between the reinforcing value and environmental cues weakens, and the CPP is finally extinguished. The period needed for extinction of CPP is influenced by different factors, including exposure to stressful events. For example, exposure to SD before each acquisition session[152], or 3-weeks before the initiation of the CPP procedure[78], slows the extinction of MDMA-induced CPP. After extinction, an injection of the drug of abuse (priming) or exposure to stress induces the reinstatement of CPP. In this paradigm, reinstatement refers to the recovery of the conditioned response and involves renewed memory of the association - learned during conditioning - between the reinforcing effect of the drug and the environmental cues associated with its pleasant effects. In our laboratory we have observed that SD exposure induces reinstatement of the CPP induced by cocaine[153,154].
The CPP has been widely used to evaluate the influence of social stress on the conditioned rewarding effects of different drugs of abuse, including alcohol, cocaine and MDMA[15]. In our laboratory, the animals are exposed to SD three weeks before initiation of the CPP procedure. We have seen that exposure to SD induces a long-term increase in the rewarding effects of cocaine, since defeated mice acquire CPP with doses that are ineffective in inducing place conditioning in control mice[77]. Furthermore, we have observed how SD induces a long-term enhancement in the vulnerability of mice to priming-induced reinstatement of the CPP induced by cocaine[155] and MDMA[78]. In addition, the CPP model has been used to study resilience to the effects of social stress on the rewarding effects of methamphetamine[156], MDMA[157] and cocaine[93,143,158,159,160-163].
Finally, the effects of social stress on alcohol intake and resilience to these effects have been studied in the two-bottle choice test, a paradigm of voluntary consumption, in which animals can choose freely, during a limited time, to drink from one of the two bottles placed in the home cage: one containing water and the other containing alcohol[92,164].
The study of the neurobiology of resilience is a relatively young area of scientific investigation[24,35]. Research carried out with animal models in the last decade has identified several behavioral, hormonal, neural and molecular mechanisms underlying the development and enhancement of resilience, mainly in relation with the reduced susceptibility to develop psychiatric disorders, such as depression or PTSD, after stress or trauma (Figure 2). As Russo et al[5] noted, resilience is mediated not only by the absence of neurobiological abnormalities that occur in susceptible animals after stress exposure (passive resilience), but also by the presence of neuroadaptations which occur in individuals that are resilient to stress, which help them to maintain normal functioning (active resilience). In this section we review the main results obtained in studies using electrophysiological, optogenetic, pharmacological, and molecular profiling techniques to unravel the neurobiological substrates of resilience to the negative consequences of chronic SD stress, mainly social avoidance and anhedonia. Advances in this field may guide ongoing research regarding the neurobiological substrates of resilience to the effects of SD on addiction disorders.
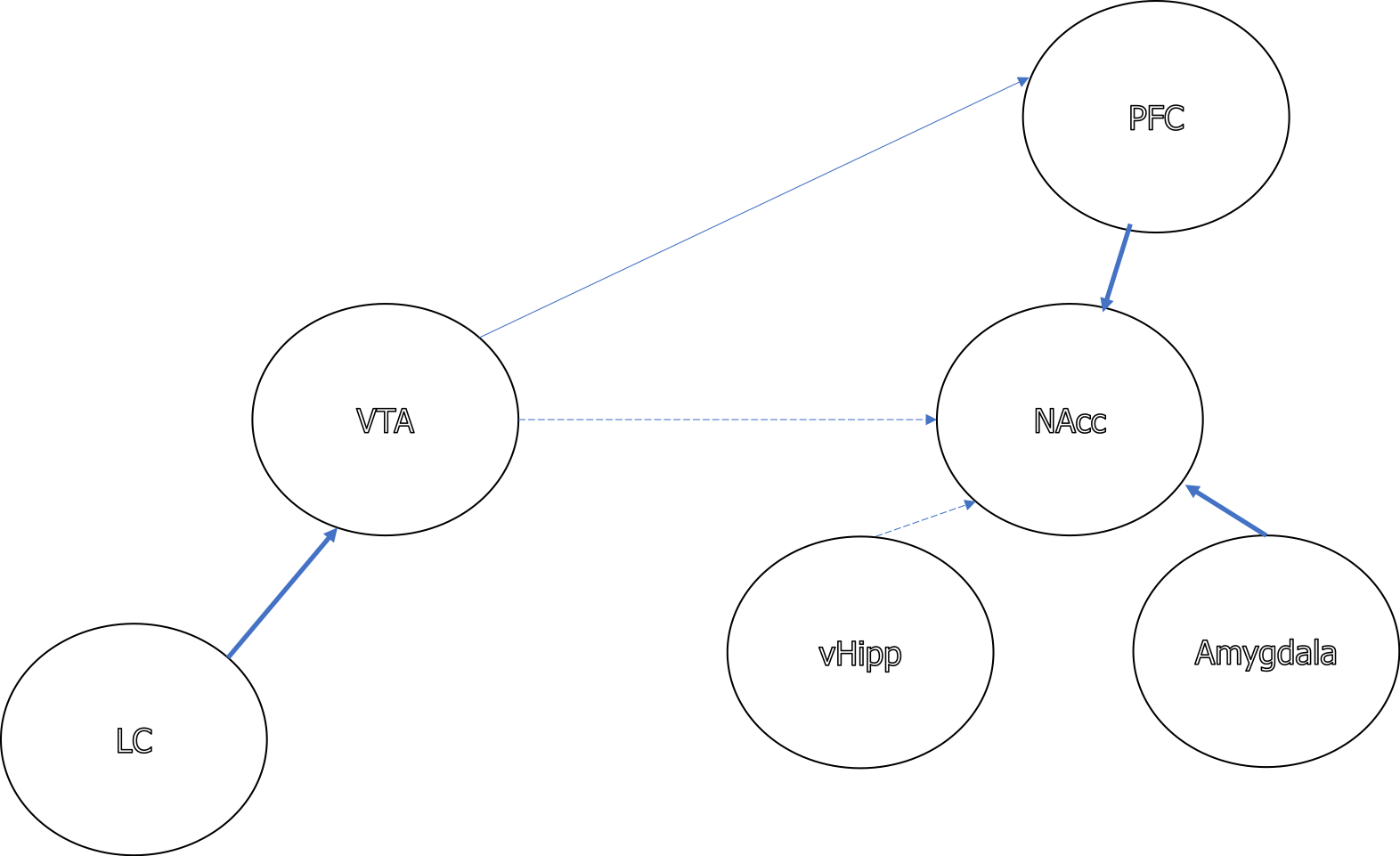
The glutamatergic system seems to play an important role in resilience to stress[165]. Chronic stress reduces the dendritic spine density of glutamatergic neurons in the PFC and hippocampus, while it increases it in the amygdala and NAcc[166]. In the chronic predator and SD stress paradigms, resilient mice show greater expression of immediate early genes (c–Fos, FosB, or ΔFosB) in glutamatergic neurons of the medial PFC[106,167,168] and in medium spiny neurons (MSN) of the NAcc, inducing expression of the AMPA glutamate receptor subunit GluA2[169]. Optogenetic stimulation of either medial PFC or amygdala glutamate afferents to the NAcc induces resilience[168,170], while attenuation of glutamatergic transmission from the ventral hippocampus to the NAcc is pro-resilient, and reduced activity in the ventral hippocampus is observed in mice that are resilient to the effects of chronic SD[170]. Furthermore, several environmental manipulations that promote resilience to stress–induced depression– and anxiety–like behaviors, such as early intermittent maternal separation and environmental enrichment, increase the volume of ventromedial PFC[171], the dendritic spine density of hippocampal and PFC neurons[172], and expression of FosB and ΔFosB in medial PFC[167]. All these results suggest that increased neuronal activation of mPFC represent pro–resilience adaptation[5].
NMDA receptors have been implicated in stress resilience[165]. Mice susceptible to chronic SD stress exhibit low activity of hippocampal extrasynaptic NMDA receptors, and enhancement in the function of these receptors prevents social avoidance behavior in defeated mice[173]. The NMDA antagonist ketamine protects against the long-term consequences of different types of stress in animal models[165,174]. For example, administration of ketamine protects mice against SD-induced depressive symptomatology in the FST and against learned helplessness-induced coping deficits when dealing with inescapable shocks, although it did not protect against the anxiety-like phenotype in the EPM[60]. Reducing brain D-serine, an endogenous co-agonist at the glycine site of the NMDA receptors, may also improve stress resilience[175], and NMDA receptor blockade in the right medial PFC facilitates resilience to SDS-induced anxiety in mice[176]. Furthermore, we have observed that the NMDA antagonist memantine increases resilience to the effects of IRSD on the CPP induced by cocaine in mice[77].
Some subunits of AMPA receptors might be involved in resilience. For instance, mice resilient to developing social avoidance after chronic SD show increased GluR2 mRNA expression compared to control mice, while susceptible mice display a decrease in GluR2 levels in the NAcc[169]. In addition, AMPA agonists prevent increases in corticosterone and latency to feed in the novelty-suppressed feeding induced by chronic stress[177].
The role of metabotropic glutamate receptors in stress resilience remains uncertain[165]. After 3 days exposure to learned helplessness or SD, mGluR5 KO mice exhibit enhanced susceptibility to stress-induced depression, social avoidance, and anhedonia. In addition, susceptible mice exhibit less mGluR5 in the NAcc than both resilient and control mice[178]. Finally, blockade of mGlu2/3 and deletion of mGlu2, but not mGlu3, promotes stress resilience, including protection against stress-induced depressive-like symptoms[179].
There are a limited number of studies on the role of GABA in resilience to the effects of chronic SD, and the effects observed to date have been in the function of the brain area containing GABA neurons and the subtype of receptor studied.
Chronic SD defeat activates GABA neurons of the dorsal raphe nucleus (DRN) and strengthens inhibition of 5-HT neurons in susceptible mice, but this effect is not observed in resilient mice without a social interaction deficit; accordingly, optogenetic inhibition of DRN GABA neurons was shown to disinhibit 5-HT neurons and promote resilience[180]. Conversely, SD stress impairs the inhibitory tone in the NAcc. Stress-susceptible mice exhibit reduced levels of inhibitory synaptic markers and protein expression (vesicular GABA transporters (vGAT) and gephyrin) in the NAcc that are not observed in resilient mice[181]. GABA (B) receptors in the habenular nuclei are also down-regulated in susceptible mice, which display elevated c-Fos expression in this structure; furthermore, intra-habenular injection of baclofen and CGP36216 (GABA (B) agonist and antagonist, respectively) reverses social avoidance[182]. Studies with KO mice have also indicated the role of GABA in resilience to the effects of SD. GAT-1-deficient mice demonstrate an increase in resilience to the effects of acute stress on depressive- and anxiety-like symptoms[183,184]. Moreover, GABA(B1a) KO mice are more susceptible, whereas GABA(B1b) KO mice are more resilient to both stress-induced anhedonia and psychosocial stress-induced social avoidance[185].
Adaptations within the brain reward system, and in particular in the mesolimbic DA circuit, are closely associated with resilience to the effects of chronic SD stress. The firing rate of ventral tegmental area (VTA) DA neurons has been shown to be increased in susceptible animals exposed to chronic SD; conversely, resilient mice show an increase in K + channels that normalizes hyperexcitability of VTA DA neurons and prevents social avoidance and sucrose preference deficit[70,186,187]. A further increase in the hyperactivity of VTA DA neurons in susceptible mice produced by optogenetics or pharmacological treatments induces homeostatic plasticity and reverses depression-related behaviors[187,188]. Such studies bring to light the self-stabilizing capacity of midbrain DA neurons of the brain reward system[187]. A recent study has demonstrated that a baseline level of physical activity (voluntary wheel running), mediated by the tyrosine hydroxylase (TH) neurons in the VTA, affects susceptibility and resilience to chronic SD. Mice with low levels of physical activity showed lower TH expression in the VTA and were susceptible to SD, while mice with high levels of activity showed higher TH expression and were resilient to SD; activation of TH neurons in the VTA of mice with lower levels of activity increased resilience, while inhibition of these neurons increased susceptibility to SD[189].
Different MSN subtypes of the NAcc (D1-MSN and D2-MSN, with predominant expression of DA D1 and D2 receptors, respectively) are also involved in susceptibility and resilience to chronic SD stress. Susceptible mice that develop depression-like behaviors after SD showed decreased frequency of excitatory synaptic input in D1-MSN (but an increase in D2-MSN); in addition, enhancing the activity[190] or the spine density[191] of D1-MSN has been shown to induce resilience. Fosb-targeted histone methylation in D1-MSN or histone acetylation in D2-MSN promote a susceptible, depressive-like phenotype, while histone acetylation in D1-MSN or histone methylation in D2-MSN increase resilience[192]. Resilient animals also display an upregulation of synaptic strength at dendritic spines of D1-MSN and a concomitant downregulation in D2-MSN[193]. In addition, chronic SD selectively reduces NLGN-2, a neuronal postsynaptic cell adhesion protein, in DA D1-MSN of susceptible mice[181]. D1-MSN activity prior to stress is also a predictor of resilience, as mice that will later become resilient display increased baseline D1-MSN activity[194].
Single and repeated SD stress induces D1 receptor-mediated changes in medial PFC neurons. A single SD was shown to increase arborization and the spines of apical dendrites of pyramidal neurons in the medial PFC, whereas repeated SD reduced dendritic lengths of these neurons[195]. Optogenetic inhibition of the DA VTA neurons projecting to the medial PFC promotes susceptibility[188]. DA D1 receptors in medial PFC excitatory neurons plays a role in suppressing susceptibility to stress, since repeated SD reduces the expression of these receptors in susceptible mice, while its genetic deletion facilitates the induction of social avoidance[195].
DA transmission in other brain areas is also involved in susceptibility or resilience to stress, although results are contradictory. Vulnerable mice were reported to display increased expression of DA D2 receptors in the amygdala[102,196] and increased levels of DA in the hippocampus and PFC[197]. However, another study found that hippocampal dopaminergic activity was inversely correlated with the level of social avoidance induced by SD and chronic treatment with hop bitter acids enhanced stress resilience[198]. Similarly, treatment with caffeine (from 14 days before until the end of SD) reverses social avoidance and anhedonia, and this pro-resilience effect of caffeine is reversed by the antagonism of DA D1 (but not D2) receptors[199].
Noradrenergic (NA) neurons in the LC have direct connections within the VTA and regulate vulnerability to SD through inhibitory control of VTA DA neurons[200]. NA LC neurons projecting to the VTA exhibit enhanced firing activity in resilient, but not susceptible, mice, and optogenetic activation of LC neurons in susceptible mice reverses depression-related behaviors[201]. α1- and β3-adrenergic receptors are highly expressed in VTA neurons projecting to the NAcc, and the antagonism of these receptors blocks the effects of the optogenetic and pharmacologic activation of LC neurons; i.e., it reverses hyperactivity and homeostatic plasticity in the VTA-NAcc pathway in susceptible mice[201].
Plasticity of the serotonergic system also contributes to susceptibility or resilience to the effects of SD stress, although the role of serotonin depends on the brain area under consideration. As commented on before, inhibition of GABA neurons of DRN disinhibits 5-HT neurons and promotes resilience to social avoidance induced by SD in mice[180]. In fact, the mechanism underlying SD-induced social avoidance is a hyposerotonergic state in the DRN, which results from the activation of p38α mitogen-activated protein kinase (MAPK), the consequent translocation of the SERT to the membrane, and the increase in the rate of serotonin uptake[202]. Down-regulation of the 5-HT1A auto-receptors in 5-HT neurons of DRN (which can result in increased 5HT release), improves behavioral resilience to SD[203]. On the other hand, rats susceptible to stress-induced anhedonia, but not resilient rats, display an increased number of neurons expressing tryptophan-hydroxylase-2 (TPH2, the enzyme for serotonin synthesis) in the ventral subnucleus of the DRN (DRNv), while activation of the CRF containing neurons of the amygdala induce resilience, suppressing the increase of TPH2 positive neurons in the DRNv and ameliorating anhedonia in susceptible rats[204]. Mice resilient to the effects of chronic SD also display a reduction of serotonin in the hippocampus[197].
ACh signaling in the hippocampus may be related with differential responses to SD stress. Interference with hippocampal AChE activity increases anxiety- and depre
Chronic SD stress increases μ and κ opioid receptors and reduces δ opioid receptors in the PFC of susceptible mice (with social avoidance), while resilient mice show no alteration in the levels of opioid receptors and increased p38 MAPK phosphorylation[207]. Besides the increased mRNA expression of the opioid μ and κ receptors in the frontal cortex, susceptible mice also show a reduction in the expression of μ receptors in the hippocampus and a reduction of κ receptors in the basolateral amygdala[208,209]. Conversely, mRNA of dynorphin is increased in the shell of NAcc in susceptible rats and in the striatum of resilient animals[208].
Chronic SD also decreases mRNA levels of δ opioid receptors and enkephalins in the basolateral amygdala and in the ventral hippocampus (CA1) of vulnerable mice[209]. Administration of an agonist of δ receptors increases resilience and reduces oxidative stress markers in CA1 neurons, a mechanism that may be involved in the pro-resilient effect of enkephalin signaling[210]. Similarly, susceptible animals display reduced enkephalin levels in the NAcc and enkephalinase inhibitors, while intra-NAcc infusion of a δ receptor agonist induces resilience and increases phosphorylation of extracellular signal-regulated kinase (ERK), which is downregulated by SD stress[211].
μ-opioid receptor G-allele carriers express less submissive behavior and exhibit resilience to SD, demonstrated by a lack of subsequent social avoidance and reductions in anhedonia; moreover, the resilience in question was associated with a greater induction of c-fos in the NAcc and periaqueductal gray[212].
Neuropeptide Y (NPY) is a neuropeptide that is widely distributed in the brain and promotes protective responses in the face of stress[213,214] by inducing anxiolytic effects and counteracting the anxiogenic effects of CRF. Multiple studies indicate a positive correlation between NPY levels and resilience to the deleterious effects of stress in humans and animal models. A significant down-regulation of NPY in the amygdala and hippocampus has been observed in animals with PTSD-like symptoms, and administration of NPY reversed the negative behavioral effects of predator-scent stress[63]. Mice susceptible to the effects of chronic SD also show a down-regulation of NPY and NPY2R in the hippocampus[215]. Administration of NPY significantly reduces submissive/defensive behaviors in socially defeated hamsters, although this effect is not mediated by the Y1 receptor[216]. Such results demonstrate that NPY may function as an important factor in resilience against the impairing effects of SD, and a recent study has suggested that deficiency of NPY plays a role in the impairing effects of stress on hippocampal function and the processes mediated by this structure[217].
Orexins (OX) produced in the lateral hypothalamus also play an important role in the response to stress[218,219]. Chronic SD stress-susceptible and -resilient mice (with or without deficits in social interaction) display different levels of prepro-OX in the hypothalamus[220] and basolateral amygdala, with increased OX1 and decreased OX2 observed in susceptible mice[221]. Brain infusion of OX A was found to induce an antidepressant-like effect only in susceptible mice, while co-infusion of OX A and B induced an anxiogenic effect only in resilient mice[220]. In addition, knocking down the OX2 receptors in the basolateral amygdala increases social avoidance and reduces the time spent in the center of an open field[221]. Similarly, after SD stress, resilient (actively coping) rats express lower prepro-OX mRNA levels than passively coping rats, while inhibition of OX before each SD episode increases social interaction and decreases depressive-like behavior in vulnerable rats[222]. These results suggest that lower levels of OX contribute to resilience to repeated SD, although in this context it is important to consider the different types of OX receptors. A recent study indicated that OX1 and OX2 receptors exert opposite functions and that the agonism of OX2 receptors promotes resilience to the anxiety and depression induced by exposure to SD stress in mice[223,224].
Neurotrophic factors and their signaling pathways, such as BDNF or ERK1/2, have been implicated in the neuroadaptations that take place in response to stress.
ERK is reduced after SD stress in both susceptible and resilient mice[207]. SD also decreases phosphorylation of ERK[211] and the pERK/ERK ratio[225]. Overexpression of ERK2 in the VTA increases susceptibility to SD stress in mice, while blockade of VTA ERK2 activity promotes behavioral resilience and decreases the frequency of firing of the VTA DA neurons, an important electrophysiological hallmark of resilience[226]. Phosphorylation of ERK is enhanced by treatments that induce resilience, such as the intra-NAcc infusion of a delta opioid receptor agonist or enkephalinase inhibitors[211].
BDNF is expressed in the amygdala, hippocampus, PFC and basal forebrain, and acts through its two main receptors, TrkB and p75[227]. BDNF has antidepressant-like effects and enhances hippocampal neurogenesis[228,229], which suggests an important role of this factor in the potentiation of resilience. Chronic SD stress decreases BDNF/TrkB in the PFC, the dentate gyrus (DG), and the CA3 region of the hippocampus, but increases BDNF/TrkB in the NAcc[175,225]. A differential expression of BDNF has been observed in susceptible and resilient mice in function of the brain area studied. Susceptible mice have higher levels of BDNF mRNA in the VTA than resilient and control mice, suggesting that this increase is associated with depressive-like behavior induced by SD[230]. An increase of BDNF-4 has been observed in the PFC of susceptible mice exposed to chronic SD, but the same animals also showed a selective reduction of BDNF-6 transcript in the hippocampus[231]. Conversely, in another study with mice exposed to chronic SD stress, levels of BDNF in the medial PFC and hippocampus were lower in susceptible mice than in control and resilient animals[232]. Finally, several studies support the contribution of hippocampal BDNF expression to resilience to chronic stress[233]. In rodents exposed to SD, activation of hippocampal BDNF/TRKB signaling (by means of branched-chain amino acids, exercise and high protein diets) induces resilience to social avoidance[234,235,236]. In addition, enhancement of BDNF and TRKB levels and signaling has been implicated in the nicotine-induced resilience to the social deficit induced by SD[237].
Stress activates the HPA axis and the release of stress hormones that regulate the individual response to stress. SD stress induces hypercortisolemia and adrenal hypertrophy in susceptible mice, but not in resilient rodents[48,238]. In addition, susceptible mice exhibit reduced glucocorticoid (GR) receptor expression in the hippocampus in comparison to resilient mice, suggesting that up-regulation of GR and enhancement of GR nuclear translocation in the hippocampus play an important role in resilience to chronic SD stress[238]. Susceptible mice show higher plasma corticosterone concentrations 2 h and 48 h after single and chronic SD stress, respectively; and administration of corticosterone via drinking water enhances susceptibility while a GR antagonist alleviates the negative consequences of chronic stress[239]. A single dose of ketamine that improved depressive-like behaviors was shown to decrease plasma corticosterone levels and rescue GR expression and nuclear translocation in the hippocampus of susceptible mice[239].
Resilient rats (with proactive behavior in resisting defeat) show decreased efficacy of CRF[82]. Similarly, mice in which CRF is deleted from GABAergic forebrain neurons were found to display a resilient phenotype[240], and PFC mRNA expression of CRF was stronger in susceptible mice than in resilient counterparts[48]. However, another study showed that increasing CRF neuronal activity in a subtype of GABAergic inhibitory interneurons in the medial PFC promoted lasting resilience to SD stress[241,226].
A wide variety of genetic factors - polymorphisms of genes of NPY, CRFR1, catecho
HDAC inhibitors may also regulate stress-related behaviors independently of their action on histones, through prevention of glucocorticoid signaling in serotonin pathways. Deletion of HDAC6 in serotonin neurons prevents the electrophysiological and morphological changes induced by chronic SD in these neurons and blocks the expression of social avoidance[247]. In one study, lower acetylated Hsp90 levels, higher GR-Hsp90 association, and enhanced GR translocation were observed in the DRN of vulnerable mice after chronic SD stress, and a HDAC6-selective inhibitor or the serotonin-selective viral overexpression of the acetylation-mimic mutant of Hsp90 in DRN neurons promoted resilience to chronic SD stress[248].
Inflammation may underlie individual differences in vulnerability and resilience to chronic SD stress[249,250].
Exposure to SD increases inflammatory markers, but the enhancement of proinflammatory proteins is more pronounced in susceptible rats (with passive coping during defeats and anhedonia) than in active coping rats[236]. In addition, only susceptible rats exhibit elevated levels of inflammatory proteins (IL-1β, TNF-α, GM-CSF) in the LC[251], and higher systemic levels of interleukin-6 (IL-6)[252]. Rats with short-defeat latencies (vulnerable rats) exhibit increased anxiety- and depression-like behaviors, and inflammation in the ventral hippocampus[253]. On the other hand, selective KO of the miR-106b~25 cluster in peripheral leukocytes promotes behavioral resilience to chronic SD stress[254]. Preexisting individual differences in the sensitivity of the peripheral immune system (IL-6) may predict vulnerability or resilience to social stress[250].
Gut microbiota, important activators of inflammatory substances, have emerged as a putative mechanism for promoting stress vulnerability[253]. For example, in one study, mice that were most susceptible to the behavioral effects of chronic SD (reflected by severe social avoidance behaviors) displayed the greatest changes within particular sets of bacteria in the phylum and genus taxonomic ranks[255].
There is a well-known link between stress and the development of AUD/SUD, and preclinical studies have shown that early life stress, social rank stress, and SD stress impact on vulnerability and resilience to alcohol, cocaine and other drugs of abuse[14,15,256]. However, as mentioned previously, there are few works studying resilience to the effects of social stress on the rewarding properties of drugs of abuse. For example, in the search “social defeat, addiction, resilience” in PubMed we identified only 18 papers, and some of these studies did not employ any paradigm of drug reward or addiction. After an exhaustive search and review of the literature we found only 8 papers on resilience to the consequences of repeated or chronic SD for the rewarding effects of cocaine, alcohol or methamphetamine.
In a classic preclinical study of resilience, Krishnan et al[70] demonstrated for the first time that, following exposure to chronic SD stress, mice can be classified as susceptible or resilient according to their differential response to stress. Susceptible mice exhibited anhedonia, social avoidance and anxiety-like behavior in the EPM, while resilient mice did not show such symptoms. This study was also pioneering in demonstrating differences between susceptible and resilient mice in sensitivity to the rewarding effects of cocaine. Only susceptible mice showed CPP after conditioning with a low dose of cocaine, while resilient or non-stressed mice did not acquire CPP[70]. Surprisingly, until recently, no other studies have addressed this issue.
In our laboratory, we have studied the influence of IRSD stress on the rewarding properties of cocaine in the CPP paradigm. Exposure to four episodes of SD during late adolescence (on post-natal day (PND) 47-56) subsequently increased the sensitivity of adult mice to a low dose of cocaine. In particular, 1 mg/kg of cocaine induced CPP in defeated mice but not in non-stressed control mice[77]. In a recent study, we evaluated whether some animals were resilient to the effects of IRSD. Overall, exposure to SD decreased all measurements related to the open arms of the EPM, immobility in the TST, social contact in the social interaction test, and grooming in the splash test. IRSD exposure also increased the sensitivity of the mice to the rewarding effects of cocaine, since only defeated animals acquired CPP. However, the potentiation of cocaine CPP was not observed in all the defeated mice, as some of them were resilient to the effects of IRSD on cocaine reward[93]. In the same study we characterized the behavioral profile of vulnerable and resilient mice during defeat episodes and in several behavioral paradigms shortly after SD. Vulnerable mice that showed CPP also exhibited depressive-like behavior, in line with the results of Krishnan et al[70]. In comparison with vulnerable mice, resilient mice displayed different behavioral traits, such as less submissive behavior during episodes of defeat, a lower percentage of time in the open arms of an EPM, lower novelty-seeking in the hole board, higher social interaction, greater immobility in the TST, and higher frequency of grooming in the splash test. These results indicate that the behavioral profile of resilient mice is characterized by an active coping response during defeat episodes, a reduced short-term response to SD (lesser reactivity to moderate unavoidable stress, enhanced concern in a potentially dangerous environment and absence of depressive symptoms), and a lack of long-term responses to SD, as eviden
Two similar studies also showed that control mice do not develop CPP with a low dose of cocaine, while defeated mice did overall develop a preference for the drug-paired compartment[143,162]. Among the defeated animals, two populations could be distinguished - resilient (did not develop preference) and susceptible mice (developed preference) - and they differed in their active or passive behavior during the SD sessions. As the authors stated, “resilient animals showed less flight and submission behaviors than susceptible mice and they presented attack behaviors towards the residents, thereby showing their resistance to being defeated”[162]. Besides passive coping behavior during SD episodes, susceptible mice (which showed cocaine CPP) also displayed social avoidance and higher IL-6 levels in the striatum and hippo
Rats exposed to repeated SD (five episodes) and social isolation (approximately 12 wk) can also be classified as SD-prone or SD-resilient, based on their affective (depression-like behavior) and cognitive performance. In one study, although SD was shown to increase alcohol SA in both groups, only SD-prone rats displayed heightened motivation for alcohol, persistent alcohol-seeking despite unavailability, resistance to extinction, and increased cue-induced reinstatement of alcohol SA[145]. Similarly, among rats exposed to SD stress, there was a subpopulation in which SD exposure increased anxiety-like behavior and induced escalation of alcohol SA. In comparison with resilient rats, vulnerable rats showed a strong upregulation of vasopressin and oxytocin that correlated positively with the magnitude of the anxiety-like behavior and alcohol SA[146]. These studies suggest that proneness to depression or anxiety enhances vulnerability to AUD, while resilience to mental disorders induced by stress can protect the individual from the development of AUD.
No studies have evaluated resilience to the effects of SD on the rewarding effects of drugs of abuse other than cocaine and alcohol, although one did show that a single SD episode combined with drug priming potentiated the reinstatement of methamphe
Although scarce, there is research on resilience to the effects of stress on drug reward carried out with other paradigms of social stress. Mice segregated according to whether they are vulnerable or resilient (socially-submissive or socially-dominant mice, respectively) were exposed to CMS for 4 wk; vulnerable, submissive mice displayed a marked increase in cocaine preference after stress, whereas the preference of resilient, dominant mice did not change. In addition, vulnerable mice displayed an increase in the expression of CRF and a reduction in the expression of DA D1 and D2 receptors in the hippocampus[159]. Following exposure to predator odor stress, animals were classified as susceptible or resilient based on EPM behavior and context avoidance; as expected, susceptible animals showed heightened motivation to self-administer cocaine[141]. With the same model, female and male rats were classified according to their stress-reactive behavior (digging and immobility during exposure to the predator odor); no different subgroups could be distinguished in males because all presented the same profile, but female rats were composed of two different populations - high digging/low immobility vs low digging/high immobility - and the former showed increased alcohol SA[147].
Early-life adversity consisting of rearing mouse pups in a limited bedding and nesting environment facilitates the escalation of ethanol intake in males at an earlier stage of exposure to alcohol, while females are insensitive to both stress and alcohol. In the study in question, stressed males exposed to alcohol showed reduced open arm exploration in the EPM and increased immobility in the TST compared to alcohol-naïve mice, although they did not differ in grooming response in the splash test, novel object recognition test or corticosterone levels. There were also no differences among control-reared males exposed or not to alcohol. The authors concluded that early stress accelerates the transition from moderate to excessive alcohol drinking and produces anxiety- and depression-like symptoms during alcohol withdrawal[92].
Finally, foot shock stress has been shown to increase two-bottle drinking in some mice, although others show resilience to this effect, displaying higher G-CSF, IL-13, and leptin levels[164]. All these studies suggest that differences in the ability to cope with stressful situations or in the response to stress results in varying tendencies to develop addictive behaviors.
Stress and drug use can lead to common alterations in synaptic plasticity that may contribute to the ability of stress to elicit relapse. For example, disruption of PKC-mediated GluA2 phosphorylation increases vulnerability to both SD-induced enhancement of social avoidance and stress-induced reinstatement of cocaine AA and CPP[258]. Study of resilience to the reinstating effects of SD stress may help to identify therapies that prevent stress-induced relapse. In line with this, Bruchas et al[202] demonstrated that SD stress produced reinstatement of cocaine-induced CPP in wild-type mice, but not in mice with a selective deletion of p38α MAPK in DRN serotonergic neurons. The antagonism of DA D3 receptors also prevents the SD-induced reinstatement of cocaine CPP and the increase in corticosterone provoked by SD[259]. Similarly, elimination of Rgs7 (a regulator of G-protein-coupled receptors) in striatal neurons induces a resilient phenotype, since mice do not show SD-induced reinstatement of cocaine CPP and exhibit an anxiolytic- and antidepressant-like profile[163]. Finally, our group has demonstrated that cannabidiol can prevent SD-induced reinstatement of cocaine CPP[154]. Altogether, these studies suggest that resilience to the effects of social stress on relapse to cocaine seeking can be pharmacologically enhanced.
An important variable in the development of resilience is sex. Studies on resilience to stress in animal models have been performed almost exclusively in males, although the prevalence of stress-related disorders clearly differ between males and females[260]. Some studies have shown sex differences in vulnerability and resilience to stress through a lifetime. Prenatal or early stress affects males more than females, inducing problems in social interaction, attention and cognition; conversely, adolescent stress induces more effects in females, increasing the risk of depression, anxiety and PTSD[261]. Thus, research suggests that hormonal activation during puberty, pregnancy or perimenopause highlights the risk associated with stress in females. Furthermore, in comparison with males, female rodents are more resilient to the effects of stress on cognitive processes (for example, in the object recognition task), but are more susceptible to the effects of stress in emotional domains (for example, in the sucrose preference and the FST)[86,87]. In fact, the effects of stress on cognition depend on both sex and the learning task. For example, stress improves the performance of radial arm maze, Morris water maze, Y–maze, non–associative learning, and object placement tasks in females, but impairs it in males[262-264]. Conversely, stress enhances learning of aversive conditioning in males but impairs it in females[265,266]. These data point to the possibility that males and females use different coping strategies in the face of stress[5]. Females are more resilient than males to the impairing effects of chronic unpredictable intermittent restraint on spatial memory, suggesting that chronic stress negatively impairs hippocampal-dependent function in males, but not in females[125]. Sexual differences in the consequences of stress on corticolimbic areas[267], including the brain reward system and the NAcc[202,250], and on the NA LC system[109] have also been reported. In fact, there are sex differences in the effects of ketamine on resilience to chronic stress, as this drug is only effective in males[268]. Research on sex differences in vulnerability and resilience to stress in general and in the field of drug abuse in particular should be a priority of future research. The knowledge obtained by studies with females is critical to the development of effective treatments customized for each sex, which may improve psychological disorders derived from or related with stress, including drug addiction.
Age is another essential variable to be taken into account in the study of resilience to the effects of stress. Adolescents are particularly sensitive to environmental influences, since DA circuitries in the PFC undergo maturational changes at this age. This can render adolescents more vulnerable to the effects of SD. For example, we have observed how SD exposure during adolescence induces a long-term increase in vulnerability to reinstatement[78,155]. A recent study has compared adolescent and adult mice according to their resilience or susceptibility to social avoidance behavior after SD exposure. Although the majority of adolescent mice were resilient, they exhibited risk-taking behavior, alterations in PFC DA connectivity and deficits in inhibitory control when they reached adulthood. Conversely, the majority of adult mice were susceptible (they exhibited social avoidance), but did not show alterations in anxiety-like traits, PFC connectivity or cognition[269]. Chronic SD stress in adolescent mice has a profound impact on the development and plasticity of reward circuity, inducing alterations in the glutamatergic development of the NAcc and mesolimbic DA system[270]. In our laboratory we are now studying the behavioral traits that characterize resilient mice exposed to IRSD stress during early adolescence, with the objective of comparing the results with those of our previous study in adult mice. As maternal experience promotes resilience to the effects of stress on cognition[271], we believe it could be interesting to evaluate how maternity modifies resilience to the effects of SD on drug reward and other behavioral outcomes. The study of stress resilience in the context of aging is very limited[165] but necessary, because the likelihood of the mood and cognitive disorders frequently associated with drug addiction increases in older adults. Thus, future research on resilience must be extended to cover the whole lifespan, with a special focus on critical periods such as prenatal or early life, adolescence, maternity and old age. In addition, it is essential to determine if resilience is a stable trait or changes with time. In this context, a recent study has analyzed the behavior of stressed mice after chronic SD at early and late stages of their lives and has found very dynamic courses of behavior: there are those that are consistently resilient or susceptible over time; those that are susceptible in the short term after stress, but recover with time; and there are animals that are initially resilient but develop vulnerability at a later date[112].
Another area of future research in the field of resilience to the effects of stress on drug addiction is that which explores the behavioral or pharmacological manipulations that increase or promote resilience to the effects of stress on drug reward. Regarding behavioral manipulations, we have observed that mice allowed to perform voluntary physical exercise before exposure to IRSD become resilient to the effects of stress on cocaine-induced CPP (Calpe-López et al[93], in preparation). Positive social conditions, such as paired housing, also increase stress resilience and reverse the potentiation of cocaine reward induced by IRSD, effects that are mediated by oxytocin[161]. In addition, it is essential to improve our understanding of the phenomenon of stress inoculation in order to define the right type and level of stress and the influence of age and sex variables. In recent studies we have tested the effects of different types of stress inoculation, which can modulate the subsequent response of mice to IRSD. In particular, a brief maternal separation (6 h on PND 9) or exposure during adolescence (PND 27) to immobilization, to a single SD or a vicarious defeat experience all induced resilience, and mice did not show cocaine-induced CPP (Calpe-López et al[93], in preparation).
Regarding pharmacological manipulations, preclinical studies have shown the effectiveness of several pharmacological treatments in models of resilience to the effects of stress on depressive- and anxiety-like symptoms and in animal models of drug addiction. These studies have highlighted the potential of several drugs that may increase resilience to some of the effects of social stress, including cannabidiol, NMDA antagonists and NOS inhibitors, NPY, galanin, and OX receptor modulators. Thus, it may be interesting to test whether these compounds are a therapeutic target to increase resilience to the effects of stress on drug addiction. Among these compounds, we would highlight cannabidiol, which is effective in reducing PTSD[272] and the effects of cocaine and other drugs of abuse[273,274,275]. A recent study in our laboratory demonstrated that cannabidiol prevents the stress-induced reinstatement of cocaine CPP[154]. Thus, it would be interesting to test the potential of cannabidiol to promote resilience to the effects of stress on the rewarding properties of drugs of abuse. We consider this drug to be a promising pro-resilience compound. Similarly, several studies have demonstrated that the NMDA antagonist ketamine exerts protective effects against the long-term consequences of different kinds of stress in animal models[165,174]. We believe this drug may prevent the long-term effects of stress on drug addiction, as we have demonstrated that another NMDA antagonist, memantine, induces resilience to the effects of social stress on the CPP induced by cocaine in mice[77]. In the same way, the effects of nitric oxide (NO) modulators need to be evaluated, since exposure to stressful events activates NO synthase (NOS), while pharmacological inhibition of NOS reduces depressive and anxious behaviors in animal models[276]. Furthermore, previous studies have demonstrated that NO is also involved in the rewarding effects of drugs of abuse[79,277], and NOS inhibition has been shown to prevent the effects of social stress on the rewarding properties of MDMA in the CPP paradigm[157].
The role of several neuropeptides in the effects of stress in animal models of drug addiction must also be evaluated. As commented on in the previous section, NPY is involved in the regulation of stress responses and plays an important role in emotional behaviors, mediating PTSD and addictive disorders[213-214]. Thus, NPY, as well as galanin[83, 84], could induce resilience and prevent the effects of stress on drug addiction. OX play an important role in the response to stress and drugs of abuse[219]. In particular, OX-A activates the HPA axis and induces ACTH and corticosterone release[218]. Furthermore, the antagonism of OX1 receptor blocks the stress-induced reinstatement of cocaine seeking[224,278], and the genetic manipulation of animals to induce a deficiency of OX has been shown to reduce cocaine-seeking after a withdrawal period and responsivity to cocaine-associated cues[22]. Conversely, agonism of the OX2 receptor promotes resilience to the anxiety- and depression-like symptoms induced by SD[224]. Thus, it would be of interest to test whether OX1 antagonists or OX2 agonists increase resilience to the effects of SD in animal models of drug addiction. Finally, p38 MAPKs are key signaling molecules in response to stress, regulation of pro-inflammatory cytokines and drug addiction. For these reasons, p38 MAPK and HDAC6 inhibitors are promising drugs, because they might increase resilience against stress and addiction relapse induced by adverse experiences[160].
It is vital that future research focus on other drugs of abuse, since, with the exception of one study on methamphetamine, cocaine and alcohol are the only drugs to have been evaluated. It is also important to study the relationship between different aspects of resilience (for example, between resilience to the development of depressive symptomatology and resilience against developing drug addiction after stress exposure). Frequently, resilience to a particular effect of stress does not imply resilience to another effect. For example, inhibition of 5-HT synthesis provides resilience against the effects of CMS in the open field, but not in the EPM[103]. Pre-treatment with ketamine before SD protects mice against depressive-like behavior in the FST but does not prevent anxiety-like behavior in the EPM[60]. A recent study showed that susceptibility to SD-induced social avoidance is unrelated to susceptibility to develop a deficit in appetitive, goal-directed motivation after SD; however, motivational impairments were related to ventral hippocampus hyperactivity, since successful task completion in resilient animals was associated with suppression of ventral hippocampal neural activity[279]. Similarly, rats displaying high cognitive competence in the Y-maze and radial arm maze are also resilient to the negative effects of the FST[280]. Resilience to the effects of SD on cocaine and alcohol reward frequently correlates with the absence of depressive-like symptoms[70,93,143,145].
Finally, translational studies of potential cognitive treatments to increase resilience are essential. For example, cognitive training in humans reduces vulnerability in the face of environmental stress. Similarly, it has been observed that a brief 9-day cognitive training can promote long-term resilience to the CPP induced by cocaine in mice, thus accelerating the extinction of CPP[158]. Coordination between human and animal studies is also required to understand the neural circuit of resilience, and neuroimaging techniques in humans can be combined with classic or more innovative methods in animals.
In the fields of medicine and psychology, the concept of resilience has implied a change of paradigm, placing the focus on factors that maintain health and promote wellness. The majority of research on drug addiction aims to identify individual and environmental factors that enhance the vulnerability of a subject to drug addiction. From our point of view, the incorporation of the concept of resilience - a complex, multidimensional construct - will allow scientists to unravel the neurobehavioral traits that confer protection against developing an addictive disorder after exposure to stressful or traumatic events, as well as permitting the underlying neurobiological substrates of resilience to be determined.
Overall, our understanding of the neurobiology of resilience is still at an early stage, but research in the last decade has made leaps and bounds by identifying genetic, epigenetic, molecular, neurochemical, psychological and environmental factors that protect individuals from the neuropsychiatric disorders related to stress[9,281-284]. Currently, resilience is considered an active and dynamic process that can be enhanced to allow individuals to adapt positively to a stressful context that, in other cases, could increase the risk of developing a psychiatric disorder. This concept of resilience has fueled the number of studies focused on specific protective factors and how the neurobiological mechanisms of resilience (HPA axis, GABA, serotonin, glutamate, DA, NA, acetylcholine, endocannabinoids, BDNF-TrkB, OX/hypocretin, NPY, galanin, etc.) can be manipulated to increase stress resilience in high-risk individuals and thus prevent the development of psychiatric disorders related to stress[165,219,281,284-286]. However, as stated before, most studies to date have focused on resilience to the development of emotional and anxiety disorders, while those on resilience to addictive disorders are few and far between. The members of our research team are pioneers in the study of resilience to stress in the context of drug addiction. Besides identifying behavioral traits that predict resilience in mice[93], we have highlighted behavioral manipulations (papers in preparation) and pharmacological treatments that increase resilience to the effects of stress in preclinical models of drug addiction. In particular, antagonism of glutamate receptors and inhibition of NOS reverses the effects of IRSD on cocaine and MDMA CPP[77,157,287], while cannabidiol reduces the effects of cocaine[154,273,274] and blocks SD-induced reinstatement of cocaine CPP[154].
Although promising, research on resilience to developing drug addiction after stress in animal models is not devoid of limitations; namely, the difficulty of determining the intensity and duration of exposure to adversity, and the definition of a concrete criterion to consider an animal resilient (absence or reduction of substance use; resistance to developing a SUD or to reinstatement of drug seeking, etc.). In addition, the incorporation of females and rodents at different developmental ages is crucial if the realities of resilience are to be fully understood.
From a translational point of view, understanding how an individual develops resilience is of paramount relevance to the design of training programs that increase this ability and promote coping mechanisms, especially in subjects with a maladaptive stress response. There is a well-known link between stress and the development of AUD/SUD, anxiety and depression disorders. Comorbidity between these disorders is frequent and associated with more severe symptoms and poor treatment outcomes. Besides reducing addictive behaviors, resilience training may have positive effects on mental health, reducing vulnerability to the development of anxiety, depressive, and cognitive disorders. Advances in the identification of neurobiological substrates of resilience will help in the development of pharmacological and psychological interventions for enhancing resilience to adversity and stress.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Psychiatry
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Gunlu A, Pesarico A S-Editor: Wang LL L-Editor: Webster JR P-Editor: Wang LL
| 1. | Alexander DE. Resilience and disaster risk reduction: an etymological journey. Nat Hazards Earth Syst Sci. 2013;13: 2707-2716. [DOI] [Full Text] |
| 2. | Rutter M. Resilience: some conceptual considerations. J Adolesc Health. 1993;14:626-631, 690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 473] [Cited by in RCA: 313] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 3. | Rutter M. Annual Research Review: Resilience-clinical implications. J Child Psychol Psychiatry. 2013;54:474-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 4. | Wu G, Feder A, Cohen H, Kim JJ, Calderon S, Charney DS, Mathé AA. Understanding resilience. Front Behav Neurosci. 2013;7:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 235] [Cited by in RCA: 306] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 5. | Russo SJ, Murrough JW, Han MH, Charney DS, Nestler EJ. Neurobiology of resilience. Nat Neurosci. 2012;15:1475-1484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 804] [Cited by in RCA: 742] [Article Influence: 57.1] [Reference Citation Analysis (0)] |
| 6. | Rutter M. Resilience as a dynamic concept. Dev Psychopathol. 2012;24:335-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 724] [Cited by in RCA: 631] [Article Influence: 48.5] [Reference Citation Analysis (0)] |
| 7. | Kalisch R, Cramer AOJ, Binder H, Fritz J, Leertouwer I, Lunansky G, Meyer B, Timmer J, Veer IM, van Harmelen AL. Deconstructing and Reconstructing Resilience: A Dynamic Network Approach. Perspect Psychol Sci. 2019;14:765-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 129] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 8. | McEwen BS. In pursuit of resilience: stress, epigenetics, and brain plasticity. Ann N Y Acad Sci. 2016;1373:56-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 186] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 9. | al'Absi M. The influence of stress and early life adversity on addiction: Psychobiological mechanisms of risk and resilience. Int Rev Neurobiol. 2020;152:71-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 10. | Kennedy B, Chen R, Fang F, Valdimarsdottir U, Montgomery S, Larsson H, Fall K. Low stress resilience in late adolescence and risk of smoking, high alcohol consumption and drug use later in life. J Epidemiol Community Health. 2019;73:496-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 11. | Yen JY, Lin HC, Chou WP, Liu TL, Ko CH. Associations Among Resilience, Stress, Depression, and Internet Gaming Disorder in Young Adults. Int J Environ Res Public Health. 2019;16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 76] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 12. | Morgan JK, Brown J, Bray RM. Resilience as a moderating factor between stress and alcohol-related consequences in the Army National Guard. Addict Behav. 2018;80:22-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 13. | Long EC, Lönn SL, Ji J, Lichtenstein P, Sundquist J, Sundquist K, Kendler KS. Resilience and Risk for Alcohol Use Disorders: A Swedish Twin Study. Alcohol Clin Exp Res. 2017;41:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 14. | Aguilar MA, García-Pardo MP, Montagud-Romero S, Miñarro J, Do Couto BR. Impact of social stress in addiction to psychostimulants: what we know from animal models. Curr Pharm Des. 2013;19:7009-7025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Vannan A, Powell GL, Scott SN, Pagni BA, Neisewander JL. Animal Models of the Impact of Social Stress on Cocaine Use Disorders. Int Rev Neurobiol. 2018;140:131-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Rudzinski K, McDonough P, Gartner R, Strike C. Is there room for resilience? Subst Abuse Treat Prev Policy. 2017;12:41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 17. | Weiland BJ, Nigg JT, Welsh RC, Yau WY, Zubieta JK, Zucker RA, Heitzeg MM. Resiliency in adolescents at high risk for substance abuse: flexible adaptation via subthalamic nucleus and linkage to drinking and drug use in early adulthood. Alcohol Clin Exp Res. 2012;36:1355-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Harris KJ, Smock SA, Wilkes MT. Relapse resilience: a process model of addiction and recovery. J Fam Psychother. 2011;22: 265-74. [RCA] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | Charlet K, Rosenthal A, Lohoff FW, Heinz A, Beck A. Imaging resilience and recovery in alcohol dependence. Addiction. 2018;113:1933-1950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Pearce ME, Jongbloed KA, Richardson CG, Henderson EW, Pooyak SD, Oviedo-Joekes E, Christian WM, Schechter MT, Spittal PM; Cedar Project Partnership. The Cedar Project: resilience in the face of HIV vulnerability within a cohort study involving young Indigenous people who use drugs in three Canadian cities. BMC Public Health. 2015;15:1095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 21. | Kallupi M, Scuppa G, de Guglielmo G, Calò G, Weiss F, Statnick MA, Rorick-Kehn LM, Ciccocioppo R. Genetic Deletion of the Nociceptin/Orphanin FQ Receptor in the Rat Confers Resilience to the Development of Drug Addiction. Neuropsychopharmacology. 2017;42:695-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 22. | Steiner N, Rossetti C, Sakurai T, Yanagisawa M, de Lecea L, Magistretti PJ, Halfon O, Boutrel B. Hypocretin/orexin deficiency decreases cocaine abuse liability. Neuropharmacology. 2018;133:395-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 23. | Gloria CT, Steinhardt MA. Relationships Among Positive Emotions, Coping, Resilience and Mental Health. Stress Health. 2016;32:145-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 239] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 24. | Southwick SM, Vythilingam M, Charney DS. The psychobiology of depression and resilience to stress: implications for prevention and treatment. Annu Rev Clin Psychol. 2005;1:255-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 651] [Cited by in RCA: 660] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 25. | Burt KB, Paysnick AA. Resilience in the transition to adulthood. Dev Psychopathol. 2012;24:493-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 26. | Werner EE. Children and war: risk, resilience, and recovery. Dev Psychopathol. 2012;24:553-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 27. | Min JA, Jung YE, Kim DJ, Yim HW, Kim JJ, Kim TS, Lee CU, Lee C, Chae JH. Characteristics associated with low resilience in patients with depression and/or anxiety disorders. Qual Life Res. 2013;22:231-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 28. | Smith BW, Ortiz JA, Steffen LE, Tooley EM, Wiggins KT, Yeater EA, Montoya JD, Bernard ML. Mindfulness is associated with fewer PTSD symptoms, depressive symptoms, physical symptoms, and alcohol problems in urban firefighters. J Consult Clin Psychol. 2011;79:613-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 153] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 29. | Southwick SM, Charney DS. The science of resilience: implications for the prevention and treatment of depression. Science. 2012;338:79-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 505] [Article Influence: 38.8] [Reference Citation Analysis (0)] |
| 30. | McEwen BS, Gianaros PJ. Stress- and allostasis-induced brain plasticity. Annu Rev Med. 2011;62:431-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 650] [Cited by in RCA: 733] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 31. | Crane MF, Searle BJ, Kangas M, Nwiran Y. How resilience is strengthened by exposure to stressors: the systematic self-reflection model of resilience strengthening. Anxiety Stress Coping. 2019;32:1-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 84] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 32. | Luthar SS, Barkin SH. Are affluent youth truly "at risk"? Dev Psychopathol. 2012;24:429-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 33. | Aston-Jones G, Cohen JD. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu Rev Neurosci. 2005;28:403-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2497] [Cited by in RCA: 2809] [Article Influence: 140.5] [Reference Citation Analysis (0)] |
| 34. | Strawn JR, Geracioti TD Jr. Noradrenergic dysfunction and the psychopharmacology of posttraumatic stress disorder. Depress Anxiety. 2008;25:260-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 164] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 35. | Charney DS. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am J Psychiatry. 2004;161:195-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 957] [Cited by in RCA: 876] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 36. | Feder A, Nestler EJ, Charney DS. Psychobiology and molecular genetics of resilience. Nat Rev Neurosci. 2009;10:446-457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 900] [Cited by in RCA: 770] [Article Influence: 48.1] [Reference Citation Analysis (0)] |
| 37. | Miura H, Qiao H, Ohta T. Attenuating effects of the isolated rearing condition on increased brain serotonin and dopamine turnover elicited by novelty stress. Brain Res. 2002;926:10-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 38. | Culman J, Mühlenhoff S, Blume A, Hedderich J, Lützen U, Hunt SP, Rupniak NMJ, Zhao Y. The Hypothalamic-Pituitary-Adrenal Axis and Serotonin Metabolism in Individual Brain Nuclei of Mice with Genetic Disruption of the NK1 Receptor Exposed to Acute Stress. Cell Mol Neurobiol. 2018;38:1271-1281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 39. | De La Garza R 2nd, Mahoney JJ 3rd. A distinct neurochemical profile in WKY rats at baseline and in response to acute stress: implications for animal models of anxiety and depression. Brain Res. 2004;1021:209-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 115] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 40. | Duman RS. Neuronal damage and protection in the pathophysiology and treatment of psychiatric illness: stress and depression. Dialogues Clin Neurosci. 2009;11:239-255. [PubMed] |
| 41. | Tural Ü, Aker AT, Önder E, Sodan HT, Ünver H, Akansel G. Neurotrophic factors and hippocampal activity in PTSD. PLoS One. 2018;13:e0197889. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 42. | Kozlovsky N, Matar MA, Kaplan Z, Kotler M, Zohar J, Cohen H. Long-term down-regulation of BDNF mRNA in rat hippocampal CA1 subregion correlates with PTSD-like behavioural stress response. Int J Neuropsychopharmacol. 2007;10:741-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 107] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 43. | Sajdyk TJ, Shekhar A, Gehlert DR. Interactions between NPY and CRF in the amygdala to regulate emotionality. Neuropeptides. 2004;38:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 130] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 44. | Jones T, Moller MD. Implications of hypothalamic-pituitary-adrenal axis functioning in posttraumatic stress disorder. J Am Psychiatr Nurses Assoc. 2011;17:393-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 45. | Morgan CA 3rd, Rasmusson AM, Wang S, Hoyt G, Hauger RL, Hazlett G. Neuropeptide-Y, cortisol, and subjective distress in humans exposed to acute stress: replication and extension of previous report. Biol Psychiatry. 2002;52:136-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 146] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 46. | Bottaccioli AG, Bottaccioli F, Minelli A. Stress and the psyche-brain-immune network in psychiatric diseases based on psychoneuroendocrineimmunology: a concise review. Ann N Y Acad Sci. 2019;1437:31-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 47. | Verburg-van Kemenade BM, Van der Aa LM, Chadzinska M. Neuroendocrine-immune interaction: regulation of inflammation via G-protein coupled receptors. Gen Comp Endocrinol. 2013;188:94-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 48. | Gururajan A, van de Wouw M, Boehme M, Becker T, O'Connor R, Bastiaanssen TFS, Moloney GM, Lyte JM, Ventura Silva AP, Merckx B, Dinan TG, Cryan JF. Resilience to chronic stress is associated with specific neurobiological, neuroendocrine and immune responses. Brain Behav Immun. 2019;80:583-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 49. | Tsyglakova M, McDaniel D, Hodes GE. Immune mechanisms of stress susceptibility and resilience: Lessons from animal models. Front Neuroendocrinol. 2019;54:100771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 50. | Ménard C, Pfau ML, Hodes GE, Russo SJ. Immune and Neuroendocrine Mechanisms of Stress Vulnerability and Resilience. Neuropsychopharmacology. 2017;42:62-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 251] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 51. | Feder A, Fred-Torres S, Southwick SM, Charney DS. The Biology of Human Resilience: Opportunities for Enhancing Resilience Across the Life Span. Biol Psychiatry. 2019;86:443-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 131] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 52. | Dantzer R, Cohen S, Russo SJ, Dinan TG. Resilience and immunity. Brain Behav Immun. 2018;74:28-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 139] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 53. | Makris AP, Karianaki M, Tsamis KI, Paschou SA. The role of the gut-brain axis in depression: endocrine, neural, and immune pathways. Hormones (Athens). 2021;20:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 59] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 54. | Cathomas F, Murrough JW, Nestler EJ, Han MH, Russo SJ. Neurobiology of Resilience: Interface Between Mind and Body. Biol Psychiatry. 2019;86:410-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 194] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 55. | Yang C, Fujita Y, Ren Q, Ma M, Dong C, Hashimoto K. Bifidobacterium in the gut microbiota confer resilience to chronic social defeat stress in mice. Sci Rep. 2017;7:45942. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 146] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 56. | Bharwani A, Mian MF, Surette MG, Bienenstock J, Forsythe P. Oral treatment with Lactobacillus rhamnosus attenuates behavioural deficits and immune changes in chronic social stress. BMC Med. 2017;15:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 162] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 57. | Harris JC. Experimental animal modeling of depression and anxiety. Psychiatr Clin North Am. 1989;12:815-836. [PubMed] |
| 58. | Frazer A, Morilak DA. What should animal models of depression model? Neurosci Biobehav Rev. 2005;29:515-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 92] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 59. | Ayash S, Schmitt U, Lyons DM, Müller MB. Stress inoculation in mice induces global resilience. Transl Psychiatry. 2020;10:200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 60. | Brachman RA, McGowan JC, Perusini JN, Lim SC, Pham TH, Faye C, Gardier AM, Mendez-David I, David DJ, Hen R, Denny CA. Ketamine as a Prophylactic Against Stress-Induced Depressive-like Behavior. Biol Psychiatry. 2016;79:776-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 199] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 61. | Ono Y, Lin HC, Tzen KY, Chen HH, Yang PF, Lai WS, Chen JH, Onozuka M, Yen CT. Active coping with stress suppresses glucose metabolism in the rat hypothalamus. Stress. 2012;15:207-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 62. | Delgado y Palacios R, Campo A, Henningsen K, Verhoye M, Poot D, Dijkstra J, Van Audekerke J, Benveniste H, Sijbers J, Wiborg O, Van der Linden A. Magnetic resonance imaging and spectroscopy reveal differential hippocampal changes in anhedonic and resilient subtypes of the chronic mild stress rat model. Biol Psychiatry. 2011;70:449-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 97] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 63. | Cohen H, Liu T, Kozlovsky N, Kaplan Z, Zohar J, Mathé AA. The neuropeptide Y (NPY)-ergic system is associated with behavioral resilience to stress exposure in an animal model of post-traumatic stress disorder. Neuropsychopharmacology. 2012;37:350-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 211] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 64. | Berton O, Covington HE 3rd, Ebner K, Tsankova NM, Carle TL, Ulery P, Bhonsle A, Barrot M, Krishnan V, Singewald GM, Singewald N, Birnbaum S, Neve RL, Nestler EJ. Induction of deltaFosB in the periaqueductal gray by stress promotes active coping responses. Neuron. 2007;55:289-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 102] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 65. | Fleshner M, Maier SF, Lyons DM, Raskind MA. The neurobiology of the stress-resistant brain. Stress. 2011;14:498-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 63] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 66. | Bartolomucci A, Cabassi A, Govoni P, Ceresini G, Cero C, Berra D, Dadomo H, Franceschini P, Dell'Omo G, Parmigiani S, Palanza P. Metabolic consequences and vulnerability to diet-induced obesity in male mice under chronic social stress. PLoS One. 2009;4:e4331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 124] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 67. | Nestler EJ, Hyman SE. Animal models of neuropsychiatric disorders. Nat Neurosci. 2010;13:1161-1169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1530] [Cited by in RCA: 1489] [Article Influence: 99.3] [Reference Citation Analysis (0)] |
| 68. | Hollis F, Kabbaj M. Social defeat as an animal model for depression. ILAR J. 2014;55:221-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 162] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 69. | Czéh B, Fuchs E, Wiborg O, Simon M. Animal models of major depression and their clinical implications. Prog Neuropsychopharmacol Biol Psychiatry. 2016;64:293-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 265] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 70. | Krishnan V, Han MH, Graham DL, Berton O, Renthal W, Russo SJ, Laplant Q, Graham A, Lutter M, Lagace DC, Ghose S, Reister R, Tannous P, Green TA, Neve RL, Chakravarty S, Kumar A, Eisch AJ, Self DW, Lee FS, Tamminga CA, Cooper DC, Gershenfeld HK, Nestler EJ. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell. 2007;131:391-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1425] [Cited by in RCA: 1751] [Article Influence: 103.0] [Reference Citation Analysis (0)] |
| 71. | Lutter M, Sakata I, Osborne-Lawrence S, Rovinsky SA, Anderson JG, Jung S, Birnbaum S, Yanagisawa M, Elmquist JK, Nestler EJ, Zigman JM. The orexigenic hormone ghrelin defends against depressive symptoms of chronic stress. Nat Neurosci. 2008;11:752-753. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 504] [Cited by in RCA: 478] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 72. | Golden SA, Covington HE 3rd, Berton O, Russo SJ. A standardized protocol for repeated social defeat stress in mice. Nat Protoc. 2011;6:1183-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 835] [Cited by in RCA: 1148] [Article Influence: 82.0] [Reference Citation Analysis (0)] |
| 73. | Vidal J, Buwalda B, Koolhaas JM. Male Wistar rats are more susceptible to lasting social anxiety than Wild-type Groningen rats following social defeat stress during adolescence. Behav Processes. 2011;88:76-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 74. | Ellenbroek BA, van der Kam EL, van der Elst MC, Cools AR. Individual differences in drug dependence in rats: the role of genetic factors and life events. Eur J Pharmacol. 2005;526:251-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 75. | Burke AR, Watt MJ, Forster GL. Adolescent social defeat increases adult amphetamine conditioned place preference and alters D2 dopamine receptor expression. Neuroscience. 2011;197:269-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 65] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 76. | Newman EL, Leonard MZ, Arena DT, de Almeida RMM, Miczek KA. Social defeat stress and escalation of cocaine and alcohol consumption: Focus on CRF. Neurobiol Stress. 2018;9:151-165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 77. | García-Pardo MP, Calpe-López C, Miñarro J, Aguilar MA. Role of N-methyl-D-aspartate receptors in the long-term effects of repeated social defeat stress on the rewarding and psychomotor properties of cocaine in mice. Behav Brain Res. 2019;361:95-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 78. | García-Pardo MP, Blanco-Gandía MC, Valiente-Lluch M, Rodríguez-Arias M, Miñarro J, Aguilar MA. Long-term effects of repeated social stress on the conditioned place preference induced by MDMA in mice. Prog Neuropsychopharmacol Biol Psychiatry. 2015;63:98-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 79. | García-Pardo MP, De la Rubia Ortí JE, Aguilar Calpe MA. Differential effects of MDMA and cocaine on inhibitory avoidance and object recognition tests in rodents. Neurobiol Learn Mem. 2017;146:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 80. | LEVINE S. Plasma-free corticosteroid response to electric shock in rats stimulated in infancy. Science. 1962;135:795-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 190] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 81. | Ricon T, Toth E, Leshem M, Braun K, Richter-Levin G. Unpredictable chronic stress in juvenile or adult rats has opposite effects, respectively, promoting and impairing resilience. Stress. 2012;15:11-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 82. | Wood SK, Walker HE, Valentino RJ, Bhatnagar S. Individual differences in reactivity to social stress predict susceptibility and resilience to a depressive phenotype: role of corticotropin-releasing factor. Endocrinology. 2010;151:1795-1805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 196] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 83. | Holmes PV. Trophic Mechanisms for Exercise-Induced Stress Resilience: Potential Role of Interactions between BDNF and Galanin. Front Psychiatry. 2014;5:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 84. | Sciolino NR, Smith JM, Stranahan AM, Freeman KG, Edwards GL, Weinshenker D, Holmes PV. Galanin mediates features of neural and behavioral stress resilience afforded by exercise. Neuropharmacology. 2015;89:255-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 85. | Hutchinson KM, McLaughlin KJ, Wright RL, Bryce Ortiz J, Anouti DP, Mika A, Diamond DM, Conrad CD. Environmental enrichment protects against the effects of chronic stress on cognitive and morphological measures of hippocampal integrity. Neurobiol Learn Mem. 2012;97:250-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 86. | Luine V. Sex differences in chronic stress effects on memory in rats. Stress. 2002;5:205-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 184] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 87. | Luine V, Gomez J, Beck K, Bowman R. Sex differences in chronic stress effects on cognition in rodents. Pharmacol Biochem Behav. 2017;152:13-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 122] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 88. | Fu W, Xie H, Laudon M, Zhou S, Tian S, You Y. Piromelatine ameliorates memory deficits associated with chronic mild stress-induced anhedonia in rats. Psychopharmacology (Berl). 2016;233:2229-2239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 89. | Pollak DD, Rey CE, Monje FJ. Rodent models in depression research: classical strategies and new directions. Ann Med. 2010;42:252-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 136] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 90. | Cryan JF, Mombereau C, Vassout A. The tail suspension test as a model for assessing antidepressant activity: review of pharmacological and genetic studies in mice. Neurosci Biobehav Rev. 2005;29:571-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 989] [Cited by in RCA: 1191] [Article Influence: 59.6] [Reference Citation Analysis (0)] |
| 91. | Commons KG, Cholanians AB, Babb JA, Ehlinger DG. The Rodent Forced Swim Test Measures Stress-Coping Strategy, Not Depression-like Behavior. ACS Chem Neurosci. 2017;8: 955-960. [RCA] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 362] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 92. | Okhuarobo A, Bolton JL, Igbe I, Zorrilla EP, Baram TZ, Contet C. A novel mouse model for vulnerability to alcohol dependence induced by early-life adversity. Neurobiol Stress. 2020;13:100269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 93. | Calpe-López C, García-Pardo MP, Martínez-Caballero MA, Santos-Ortíz A, Aguilar MA. Behavioral Traits Associated With Resilience to the Effects of Repeated Social Defeat on Cocaine-Induced Conditioned Place Preference in Mice. Front Behav Neurosci. 2019;13:278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 94. | Rimmerman N, Schottlender N, Reshef R, Dan-Goor N, Yirmiya R. The hippocampal transcriptomic signature of stress resilience in mice with microglial fractalkine receptor (CX3CR1) deficiency. Brain Behav Immun. 2017;61:184-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 95. | Park J, Lee J, Choi K, Kang HJ. Regulation of behavioral response to stress by microRNA-690. Mol Brain. 2021;14:7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 96. | Bergner CL, Smolinsky AN, Hart PC, Dufour BD, Egan RJ, LaPorte JL, Kalueff AV. Mouse Models for Studying Depression-Like States and Antidepressant Drugs. Methods Mol Biol. 2016;1438:255-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 97. | Duque A, Vinader-Caerols C, Monleón S. Indomethacin counteracts the effects of chronic social defeat stress on emotional but not recognition memory in mice. PLoS One. 2017;12: e0173182. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 98. | Andoh C, Nishitani N, Hashimoto E, Nagai Y, Takao K, Miyakawa T, Nakagawa T, Mori Y, Nagayasu K, Shirakawa H, Kaneko S. TRPM2 confers susceptibility to social stress but is essential for behavioral flexibility. Brain Res. 2019;1704:68-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 99. | Blasco-Serra A, González-Soler EM, Cervera-Ferri A, Teruel-Martí V, Valverde-Navarro AA. A standardization of the Novelty-Suppressed Feeding Test protocol in rats. Neurosci Lett. 2017;658:73-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 81] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 100. | Rodgers RJ, Johnson NJ. Factor analysis of spatiotemporal and ethological measures in the murine elevated plus-maze test of anxiety. Pharmacol Biochem Behav. 1995;52:297-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 566] [Cited by in RCA: 584] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 101. | Rodgers RJ, Dalvi A. Anxiety, defence and the elevated plus-maze. Neurosci Biobehav Rev. 1997;21:801-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 656] [Cited by in RCA: 726] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 102. | Prabhu VV, Nguyen TB, Cui Y, Oh YE, Lee KH, Bagalkot TR, Chung YC. Effects of social defeat stress on dopamine D2 receptor isoforms and proteins involved in intracellular trafficking. Behav Brain Funct. 2018;14:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 103. | Gutknecht L, Popp S, Waider J, Sommerlandt FM, Göppner C, Post A, Reif A, van den Hove D, Strekalova T, Schmitt A, Colaςo MB, Sommer C, Palme R, Lesch KP. Interaction of brain 5-HT synthesis deficiency, chronic stress and sex differentially impact emotional behavior in Tph2 knockout mice. Psychopharmacology (Berl). 2015;232:2429-2441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 73] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 104. | Hidaka C, Kashio T, Uchigaki D, Mitsui S. Vulnerability or resilience of motopsin knockout mice to maternal separation stress depending on adulthood behaviors. Neuropsychiatr Dis Treat. 2018;14:2255-2268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 105. | Binder E, Malki K, Paya-Cano JL, Fernandes C, Aitchison KJ, Mathé AA, Sluyter F, Schalkwyk LC. Antidepressants and the resilience to early-life stress in inbred mouse strains. Pharmacogenet Genomics. 2011;21:779-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 106. | Adamec R, Toth M, Haller J, Halasz J, Blundell J. A comparison of activation patterns of cells in selected prefrontal cortical and amygdala areas of rats which are more or less anxious in response to predator exposure or submersion stress. Physiol Behav. 2012;105:628-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 107. | Ennaceur A, Delacour J. A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behav Brain Res. 1988;31:47-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2209] [Cited by in RCA: 2458] [Article Influence: 66.4] [Reference Citation Analysis (0)] |
| 108. | Broadbent NJ, Squire LR, Clark RE. Spatial memory, recognition memory, and the hippocampus. Proc Natl Acad Sci U S A. 2004;101:14515-14520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 687] [Cited by in RCA: 720] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 109. | Atmore KH, Stein DJ, Harvey BH, Russell VA, Howells FM. Differential effects of social isolation rearing on glutamate- and GABA-stimulated noradrenaline release in the rat prefrontal cortex and hippocampus. Eur Neuropsychopharmacol. 2020;36:111-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 110. | Jene T, Gassen NC, Opitz V, Endres K, Müller MB, van der Kooij MA. Temporal profiling of an acute stress-induced behavioral phenotype in mice and role of hippocampal DRR1. Psychoneuroendocrinology. 2018;91:149-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 111. | Tzeng WY, Su CC, Sun LH, Cherng CG, Yu L. Synergistic Effects of Psychosocial Stress and Mild Peripheral Infection on Inducing Microglial Activation in the Hippocampal Dentate Gyrus and Long-Lasting Deficits in Hippocampus-Related Memory. Chin J Physiol. 2018;61:106-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 112. | Wendelmuth M, Willam M, Todorov H, Radyushkin K, Gerber S, Schweiger S. Dynamic longitudinal behavior in animals exposed to chronic social defeat stress. PLoS One. 2020;15:e0235268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 113. | Vorhees CV, Williams MT. Value of water mazes for assessing spatial and egocentric learning and memory in rodent basic research and regulatory studies. Neurotoxicol Teratol. 2014;45:75-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 114. | Qu N, Wang XM, Zhang T, Zhang SF, Li Y, Cao FY, Wang Q, Ning LN, Tian Q. Estrogen Receptor α Agonist is Beneficial for Young Female Rats Against Chronic Unpredicted Mild Stress-Induced Depressive Behavior and Cognitive Deficits. J Alzheimers Dis. 2020;77:1077-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 115. | Drugan RC, Warner TA, Papallo TA, Castracane LL, Stafford NP. Ultrasonic vocalizations during intermittent swim stress forecasts resilience in subsequent forced swim and spatial learning tests. Behav Brain Res. 2014;259:41-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 116. | Conrad CD, Galea LA, Kuroda Y, McEwen BS. Chronic stress impairs rat spatial memory on the Y maze, and this effect is blocked by tianeptine pretreatment. Behav Neurosci. 1996;110:1321-1334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 57] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 117. | McAllister BB, Wright DK, Wortman RC, Shultz SR, Dyck RH. Elimination of vesicular zinc alters the behavioural and neuroanatomical effects of social defeat stress in mice. Neurobiol Stress. 2018;9:199-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 118. | Bhakta A, Gavini K, Yang E, Lyman-Henley L, Parameshwaran K. Chronic traumatic stress impairs memory in mice: Potential roles of acetylcholine, neuroinflammation and corticotropin releasing factor expression in the hippocampus. Behav Brain Res. 2017;335:32-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 119. | Tractenberg SG, Orso R, Creutzberg KC, Malcon LMC, Lumertz FS, Wearick-Silva LE, Viola TW, Riva MA, Grassi-Oliveira R. Vulnerable and resilient cognitive performance related to early life stress: The potential mediating role of dopaminergic receptors in the medial prefrontal cortex of adult mice. Int J Dev Neurosci. 2020;80:13-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 120. | Kambali MY, Anshu K, Kutty BM, Muddashetty RS, Laxmi TR. Effect of early maternal separation stress on attention, spatial learning and social interaction behaviour. Exp Brain Res. 2019;237:1993-2010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 121. | Diamond DM, Park CR, Heman KL, Rose GM. Exposing rats to a predator impairs spatial working memory in the radial arm water maze. Hippocampus. 1999;9:542-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 122. | Hoffman AN, Krigbaum A, Ortiz JB, Mika A, Hutchinson KM, Bimonte-Nelson HA, Conrad CD. Recovery after chronic stress within spatial reference and working memory domains: correspondence with hippocampal morphology. Eur J Neurosci. 2011;34:1023-1030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 123. | Ortiz JB, Mathewson CM, Hoffman AN, Hanavan PD, Terwilliger EF, Conrad CD. Hippocampal brain-derived neurotrophic factor mediates recovery from chronic stress-induced spatial reference memory deficits. Eur J Neurosci. 2014;40:3351-3362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 124. | Ortiz JB, Anglin JM, Daas EJ, Paode PR, Nishimura K, Conrad CD. BDNF and TrkB Mediate the Improvement from Chronic Stress-induced Spatial Memory Deficits and CA3 Dendritic Retraction. Neuroscience. 2018;388:330-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 125. | Peay DN, Saribekyan HM, Parada PA, Hanson EM, Badaruddin BS, Judd JM, Donnay ME, Padilla-Garcia D, Conrad CD. Chronic unpredictable intermittent restraint stress disrupts spatial memory in male, but not female rats. Behav Brain Res. 2020;383:112519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 126. | Koob GF, Volkow ND. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry. 2016;3:760-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1666] [Cited by in RCA: 2127] [Article Influence: 236.3] [Reference Citation Analysis (0)] |
| 127. | Everitt BJ. Neural and psychological mechanisms underlying compulsive drug seeking habits and drug memories--indications for novel treatments of addiction. Eur J Neurosci. 2014;40:2163-2182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 258] [Cited by in RCA: 239] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 128. | Everitt BJ, Robbins TW. From the ventral to the dorsal striatum: devolving views of their roles in drug addiction. Neurosci Biobehav Rev. 2013;37:1946-1954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 434] [Cited by in RCA: 482] [Article Influence: 40.2] [Reference Citation Analysis (0)] |
| 129. | Moser P, Wolinsky T, Duxon M, Porsolt RD. How good are current approaches to nonclinical evaluation of abuse and dependence? J Pharmacol Exp Ther. 2011;336:588-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 130. | Yahyavi-Firouz-Abadi N, See RE. Anti-relapse medications: preclinical models for drug addiction treatment. Pharmacol Ther. 2009;124:235-247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 131. | Epstein DH, Preston KL, Stewart J, Shaham Y. Toward a model of drug relapse: an assessment of the validity of the reinstatement procedure. Psychopharmacology (Berl). 2006;189:1-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 465] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 132. | Shaham Y, Shalev U, Lu L, de Wit H, Stewart J. The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology (Berl). 2003;168:3-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1191] [Cited by in RCA: 1206] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 133. | Stewart J. Pathways to relapse: the neurobiology of drug- and stress-induced relapse to drug-taking. J Psychiatry Neurosci. 2000;25:125-136. [PubMed] |
| 134. | Soria G, Barbano MF, Maldonado R, Valverde O. A reliable method to study cue-, priming-, and stress-induced reinstatement of cocaine self-administration in mice. Psychopharmacology (Berl). 2008;199:593-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 50] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 135. | Deroche-Gamonet V, Piazza PV. Psychobiology of cocaine addiction: Contribution of a multi-symptomatic animal model of loss of control. Neuropharmacology. 2014;76 Pt B:437-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 136. | Rodriguez-Arias M, Navarrete F, Blanco-Gandia MC, Arenas MC, Bartoll-Andrés A, Aguilar MA, Rubio G, Miñarro J, Manzanares J. Social defeat in adolescent mice increases vulnerability to alcohol consumption. Addict Biol. 2016;21:87-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 137. | Reguilón MD, Ferrer-Pérez C, Ballestín R, Miñarro J, Rodríguez-Arias M. Voluntary wheel running protects against the increase in ethanol consumption induced by social stress in mice. Drug Alcohol Depend. 2020;212:108004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 138. | Reguilón MD, Ferrer-Pérez C, Miñarro J, Rodríguez-Arias M. Oxytocin reverses ethanol consumption and neuroinflammation induced by social defeat in male mice. Horm Behav. 2021;127:104875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 139. | Montagud-Romero S, Reguilón MD, Pascual M, Blanco-Gandía MC, Guerri C, Miñarro J, Rodríguez-Arias M. Critical role of TLR4 in uncovering the increased rewarding effects of cocaine and ethanol induced by social defeat in male mice. Neuropharmacology. 2021;182:108368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 140. | Bock R, Shin JH, Kaplan AR, Dobi A, Markey E, Kramer PF, Gremel CM, Christensen CH, Adrover MF, Alvarez VA. Strengthening the accumbal indirect pathway promotes resilience to compulsive cocaine use. Nat Neurosci. 2013;16:632-638. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 208] [Cited by in RCA: 237] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 141. | Brodnik ZD, Black EM, Clark MJ, Kornsey KN, Snyder NW, España RA. Susceptibility to traumatic stress sensitizes the dopaminergic response to cocaine and increases motivation for cocaine. Neuropharmacology. 2017;125:295-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 142. | Lee JH, Ribeiro EA, Kim J, Ko B, Kronman H, Jeong YH, Kim JK, Janak PH, Nestler EJ, Koo JW, Kim JH. Dopaminergic Regulation of Nucleus Accumbens Cholinergic Interneurons Demarcates Susceptibility to Cocaine Addiction. Biol Psychiatry. 2020;88:746-757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 143. | Ballestín R, Alegre-Zurano L, Ferrer-Pérez C, Cantacorps L, Miñarro J, Valverde O, Rodríguez-Arias M. Neuroinflammatory and behavioral susceptibility profile of mice exposed to social stress towards cocaine effects. Prog Neuropsychopharmacol Biol Psychiatry. 2021;105:110123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 144. | Blouin AM, Pisupati S, Hoffer CG, Hafenbreidel M, Jamieson SE, Rumbaugh G, Miller CA. Social stress-potentiated methamphetamine seeking. Addict Biol. 2019;24:958-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 145. | Riga D, Schmitz LJM, van Mourik Y, Hoogendijk WJG, De Vries TJ, Smit AB, Spijker S. Stress vulnerability promotes an alcohol-prone phenotype in a preclinical model of sustained depression. Addict Biol. 2020;25:e12701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 146. | Barchiesi R, Chanthongdee K, Domi E, Gobbo F, Coppola A, Asratian A, Toivainen S, Holm L, Augier G, Xu L, Augier E, Heilig M, Barbier E. Stress-induced escalation of alcohol self-administration, anxiety-like behavior, and elevated amygdala Avp expression in a susceptible subpopulation of rats. Addict Biol. 2021;e13009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 147. | Ornelas LC, Tyler RE, Irukulapati P, Paladugu S, Besheer J. Increased alcohol self-administration following exposure to the predator odor TMT in active coping female rats. Behav Brain Res. 2021;402:113068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 148. | Aguilar MA, Rodríguez-Arias M, Miñarro J. Neurobiological mechanisms of the reinstatement of drug-conditioned place preference. Brain Res Rev. 2009;59:253-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 208] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 149. | Bardo MT, Bevins RA. Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology (Berl). 2000;153:31-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 944] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 150. | Tzschentke TM. Measuring reward with the conditioned place preference paradigm: a comprehensive review of drug effects, recent progress and new issues. Prog Neurobiol. 1998;56:613-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 935] [Cited by in RCA: 941] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 151. | Tzschentke TM. Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addict Biol. 2007;12:227-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 966] [Cited by in RCA: 1036] [Article Influence: 57.6] [Reference Citation Analysis (0)] |
| 152. | García-Pardo MP, Rodríguez-Arias M, Maldonado C, Manzanedo C, Miñarro J, Aguilar MA. Effects of acute social stress on the conditioned place preference induced by MDMA in adolescent and adult mice. Behav Pharmacol. 2014;25:532-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 153. | Titomanlio F, Manzanedo C, Rodríguez-Arias M, Mattioli L, Perfumi M, Miñarro J, Aguilar MA. Rhodiola rosea Impairs Acquisition and Expression of Conditioned Place Preference Induced by Cocaine. Evid Based Complement Alternat Med. 2013;2013:697632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 154. | Calpe-López C, Gasparyan A, Navarrete F, Manzanares J, Miñarro J, Aguilar MA. Cannabidiol prevents priming- and stress-induced reinstatement of the conditioned place preference induced by cocaine in mice. J Psychopharmacol. 2021;35:864-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 155. | Montagud-Romero S, Aguilar MA, Maldonado C, Manzanedo C, Miñarro J, Rodríguez-Arias M. Acute social defeat stress increases the conditioned rewarding effects of cocaine in adult but not in adolescent mice. Pharmacol Biochem Behav. 2015;135:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 156. | Ueno M, Yamada K, Ichitani Y. The relationship between fear extinction and resilience to drug-dependence in rats. Neurosci Res. 2017;121:37-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 157. | García-Pardo MP, LLansola M, Felipo V, De la Rubia Ortí JE, Aguilar MA. Blockade of nitric oxide signalling promotes resilience to the effects of social defeat stress on the conditioned rewarding properties of MDMA in mice. Nitric Oxide. 2020;98:29-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 158. | Boivin JR, Piscopo DM, Wilbrecht L. Brief cognitive training interventions in young adulthood promote long-term resilience to drug-seeking behavior. Neuropharmacology. 2015;97:404-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 159. | Yanovich C, Kirby ML, Michaelevski I, Yadid G, Pinhasov A. Social rank-associated stress vulnerability predisposes individuals to cocaine attraction. Sci Rep. 2018;8:1759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 160. | El Rawas R, Amaral IM, Hofer A. Is p38 MAPK Associated to Drugs of Abuse-Induced Abnormal Behaviors? Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 161. | Ferrer-Pérez C, Reguilón MD, Miñarro J, Rodríguez-Arias M. Endogenous oxytocin is essential for the buffering effects of pair housing against the increase in cocaine reward induced by social stress. Physiol Behav. 2020;221:112913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 162. | Ródenas-González F, Blanco-Gandía MDC, Miñarro López J, Rodriguez-Arias M. Behavioral and neuroimmune characterization of resilience to social stress: rewarding effects of cocaine. Adicciones. 2020;0:1348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 163. | Sutton LP, Khalatyan N, Savas JN, Martemyanov KA. Striatal RGS7 Regulates Depression-Related Behaviors and Stress-Induced Reinstatement of Cocaine Conditioned Place Preference. eNeuro. 2021;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 164. | Steinman MQ, Kirson D, Wolfe SA, Khom S, D'Ambrosio SR, Spierling Bagsic SR, Bajo M, Vlkolinský R, Hoang NK, Singhal A, Sureshchandra S, Oleata CS, Messaoudi I, Zorrilla EP, Roberto M. Importance of sex and trauma context on circulating cytokines and amygdalar GABAergic signaling in a comorbid model of posttraumatic stress and alcohol use disorders. Mol Psychiatry. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 165. | Faye C, Mcgowan JC, Denny CA, David DJ. Neurobiological Mechanisms of Stress Resilience and Implications for the Aged Population. Curr Neuropharmacol. 2018;16:234-270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 166. | Christoffel DJ, Golden SA, Russo SJ. Structural and synaptic plasticity in stress-related disorders. Rev Neurosci. 2011;22:535-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 253] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 167. | Lehmann ML, Herkenham M. Environmental enrichment confers stress resiliency to social defeat through an infralimbic cortex-dependent neuroanatomical pathway. J Neurosci. 2011;31:6159-6173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 187] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 168. | Covington HE 3rd, Lobo MK, Maze I, Vialou V, Hyman JM, Zaman S, LaPlant Q, Mouzon E, Ghose S, Tamminga CA, Neve RL, Deisseroth K, Nestler EJ. Antidepressant effect of optogenetic stimulation of the medial prefrontal cortex. J Neurosci. 2010;30:16082-16090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 528] [Cited by in RCA: 485] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 169. | Vialou V, Robison AJ, Laplant QC, Covington HE 3rd, Dietz DM, Ohnishi YN, Mouzon E, Rush AJ 3rd, Watts EL, Wallace DL, Iñiguez SD, Ohnishi YH, Steiner MA, Warren BL, Krishnan V, Bolaños CA, Neve RL, Ghose S, Berton O, Tamminga CA, Nestler EJ. DeltaFosB in brain reward circuits mediates resilience to stress and antidepressant responses. Nat Neurosci. 2010;13:745-752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 413] [Cited by in RCA: 385] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 170. | Bagot RC, Parise EM, Peña CJ, Zhang HX, Maze I, Chaudhury D, Persaud B, Cachope R, Bolaños-Guzmán CA, Cheer JF, Deisseroth K, Han MH, Nestler EJ. Ventral hippocampal afferents to the nucleus accumbens regulate susceptibility to depression. Nat Commun. 2015;6:7062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 249] [Cited by in RCA: 349] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 171. | Katz M, Liu C, Schaer M, Parker KJ, Ottet MC, Epps A, Buckmaster CL, Bammer R, Moseley ME, Schatzberg AF, Eliez S, Lyons DM. Prefrontal plasticity and stress inoculation-induced resilience. Dev Neurosci. 2009;31:293-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 172. | Kozorovitskiy Y, Gross CG, Kopil C, Battaglia L, McBreen M, Stranahan AM, Gould E. Experience induces structural and biochemical changes in the adult primate brain. Proc Natl Acad Sci U S A. 2005;102:17478-17482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 144] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 173. | Tse YC, Lopez J, Moquin A, Wong SA, Maysinger D, Wong TP. The susceptibility to chronic social defeat stress is related to low hippocampal extrasynaptic NMDA receptor function. Neuropsychopharmacology. 2019;44:1310-1318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 174. | Rincón-Cortés M, Grace AA. Antidepressant effects of ketamine on depression-related phenotypes and dopamine dysfunction in rodent models of stress. Behav Brain Res. 2020;379:112367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 175. | Dong C, Zhang JC, Ren Q, Ma M, Qu Y, Zhang K, Yao W, Ishima T, Mori H, Hashimoto K. Deletion of serine racemase confers D-serine -dependent resilience to chronic social defeat stress. Neurochem Int. 2018;116:43-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 176. | Victoriano G, Santos-Costa N, Mascarenhas DC, Nunes-de-Souza RL. Inhibition of the left medial prefrontal cortex (mPFC) prolongs the social defeat-induced anxiogenesis in mice: Attenuation by NMDA receptor blockade in the right mPFC. Behav Brain Res. 2020;378:112312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 177. | Schmidt MV, Trümbach D, Weber P, Wagner K, Scharf SH, Liebl C, Datson N, Namendorf C, Gerlach T, Kühne C, Uhr M, Deussing JM, Wurst W, Binder EB, Holsboer F, Müller MB. Individual stress vulnerability is predicted by short-term memory and AMPA receptor subunit ratio in the hippocampus. J Neurosci. 2010;30:16949-16958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 178. | Shin S, Kwon O, Kang JI, Kwon S, Oh S, Choi J, Kim CH, Kim DG. mGluR5 in the nucleus accumbens is critical for promoting resilience to chronic stress. Nat Neurosci. 2015;18:1017-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 97] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 179. | Highland JN, Zanos P, Georgiou P, Gould TD. Group II metabotropic glutamate receptor blockade promotes stress resilience in mice. Neuropsychopharmacology. 2019;44:1788-1796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 180. | Challis C, Boulden J, Veerakumar A, Espallergues J, Vassoler FM, Pierce RC, Beck SG, Berton O. Raphe GABAergic neurons mediate the acquisition of avoidance after social defeat. J Neurosci. 2013;33:13978-13988, 13988a. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 156] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 181. | Heshmati M, Christoffel DJ, LeClair K, Cathomas F, Golden SA, Aleyasin H, Turecki G, Friedman AK, Han MH, Menard C, Russo SJ. Depression and Social Defeat Stress Are Associated with Inhibitory Synaptic Changes in the Nucleus Accumbens. J Neurosci. 2020;40:6228-6233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 182. | Li ZL, Wang Y, Zou HW, Jing XY, Liu YJ, Li LF. GABA(B) receptors within the lateral habenula modulate stress resilience and vulnerability in mice. Physiol Behav. 2021;230:113311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 183. | Liu GX, Cai GQ, Cai YQ, Sheng ZJ, Jiang J, Mei Z, Wang ZG, Guo L, Fei J. Reduced anxiety and depression-like behaviors in mice lacking GABA transporter subtype 1. Neuropsychopharmacology. 2007;32:1531-1539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 105] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 184. | Gong X, Shao Y, Li B, Chen L, Wang C, Chen Y. γ-aminobutyric acid transporter-1 is involved in anxiety-like behaviors and cognitive function in knockout mice. Exp Ther Med. 2015;10:653-658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 185. | O'Leary OF, Felice D, Galimberti S, Savignac HM, Bravo JA, Crowley T, El Yacoubi M, Vaugeois JM, Gassmann M, Bettler B, Dinan TG, Cryan JF. GABAB(1) receptor subunit isoforms differentially regulate stress resilience. Proc Natl Acad Sci U S A. 2014;111:15232-15237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 186. | Cao JL, Covington HE 3rd, Friedman AK, Wilkinson MB, Walsh JJ, Cooper DC, Nestler EJ, Han MH. Mesolimbic dopamine neurons in the brain reward circuit mediate susceptibility to social defeat and antidepressant action. J Neurosci. 2010;30:16453-16458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 340] [Cited by in RCA: 307] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 187. | Friedman AK, Walsh JJ, Juarez B, Ku SM, Chaudhury D, Wang J, Li X, Dietz DM, Pan N, Vialou VF, Neve RL, Yue Z, Han MH. Enhancing depression mechanisms in midbrain dopamine neurons achieves homeostatic resilience. Science. 2014;344:313-319. [PubMed] |
| 188. | Chaudhury D, Walsh JJ, Friedman AK, Juarez B, Ku SM, Koo JW, Ferguson D, Tsai HC, Pomeranz L, Christoffel DJ, Nectow AR, Ekstrand M, Domingos A, Mazei-Robison MS, Mouzon E, Lobo MK, Neve RL, Friedman JM, Russo SJ, Deisseroth K, Nestler EJ, Han MH. Rapid regulation of depression-related behaviours by control of midbrain dopamine neurons. Nature. 2013;493:532-536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 718] [Cited by in RCA: 865] [Article Influence: 66.5] [Reference Citation Analysis (0)] |
| 189. | Zhang J, He ZX, Qu YS, Li LF, Wang LM, Yuan W, Hou WJ, Zhu YQ, Cai WQ, Zhang XN, Guo QQ, An SC, Jia R, Tai FD. Different baseline physical activity predicts susceptibility and resilience to chronic social defeat stress in mice: Involvement of dopamine neurons. Eur Neuropsychopharmacol. 2021;45:15-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 190. | Francis TC, Chandra R, Friend DM, Finkel E, Dayrit G, Miranda J, Brooks JM, Iñiguez SD, O'Donnell P, Kravitz A, Lobo MK. Nucleus accumbens medium spiny neuron subtypes mediate depression-related outcomes to social defeat stress. Biol Psychiatry. 2015;77:212-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 281] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 191. | Francis TC, Gaynor A, Chandra R, Fox ME, Lobo MK. The Selective RhoA Inhibitor Rhosin Promotes Stress Resiliency Through Enhancing D1-Medium Spiny Neuron Plasticity and Reducing Hyperexcitability. Biol Psychiatry. 2019;85:1001-1010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 192. | Hamilton PJ, Burek DJ, Lombroso SI, Neve RL, Robison AJ, Nestler EJ, Heller EA. Cell-Type-Specific Epigenetic Editing at the Fosb Gene Controls Susceptibility to Social Defeat Stress. Neuropsychopharmacology. 2018;43:272-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 80] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 193. | Khibnik LA, Beaumont M, Doyle M, Heshmati M, Slesinger PA, Nestler EJ, Russo SJ. Stress and Cocaine Trigger Divergent and Cell Type-Specific Regulation of Synaptic Transmission at Single Spines in Nucleus Accumbens. Biol Psychiatry. 2016;79:898-905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 194. | Muir J, Lorsch ZS, Ramakrishnan C, Deisseroth K, Nestler EJ, Calipari ES, Bagot RC. In Vivo Fiber Photometry Reveals Signature of Future Stress Susceptibility in Nucleus Accumbens. Neuropsychopharmacology. 2018;43:255-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 87] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 195. | Shinohara R, Taniguchi M, Ehrlich AT, Yokogawa K, Deguchi Y, Cherasse Y, Lazarus M, Urade Y, Ogawa A, Kitaoka S, Sawa A, Narumiya S, Furuyashiki T. Dopamine D1 receptor subtype mediates acute stress-induced dendritic growth in excitatory neurons of the medial prefrontal cortex and contributes to suppression of stress susceptibility in mice. Mol Psychiatry. 2018;23:1717-1730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 79] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 196. | Bagalkot TR, Jin HM, Prabhu VV, Muna SS, Cui Y, Yadav BK, Chae HJ, Chung YC. Chronic social defeat stress increases dopamine D2 receptor dimerization in the prefrontal cortex of adult mice. Neuroscience. 2015;311:444-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 197. | Xu K, He Y, Chen X, Tian Y, Cheng K, Zhang L, Wang Y, Yang D, Wang H, Wu Z, Li Y, Lan T, Dong Z, Xie P. Validation of the targeted metabolomic pathway in the hippocampus and comparative analysis with the prefrontal cortex of social defeat model mice. J Neurochem. 2019;149:799-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 198. | Ano Y, Kitaoka S, Ohya R, Kondo K, Furuyashiki T. Hop Bitter Acids Increase Hippocampal Dopaminergic Activity in a Mouse Model of Social Defeat Stress. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 199. | Yin YQ, Zhang C, Wang JX, Hou J, Yang X, Qin J. Chronic caffeine treatment enhances the resilience to social defeat stress in mice. Food Funct. 2015;6:479-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 200. | Isingrini E, Perret L, Rainer Q, Amilhon B, Guma E, Tanti A, Martin G, Robinson J, Moquin L, Marti F, Mechawar N, Williams S, Gratton A, Giros B. Resilience to chronic stress is mediated by noradrenergic regulation of dopamine neurons. Nat Neurosci. 2016;19:560-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 122] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 201. | Zhang H, Chaudhury D, Nectow AR, Friedman AK, Zhang S, Juarez B, Liu H, Pfau ML, Aleyasin H, Jiang C, Crumiller M, Calipari ES, Ku SM, Morel C, Tzavaras N, Montgomery SE, He M, Salton SR, Russo SJ, Nestler EJ, Friedman JM, Cao JL, Han MH. α1- and β3-Adrenergic Receptor-Mediated Mesolimbic Homeostatic Plasticity Confers Resilience to Social Stress in Susceptible Mice. Biol Psychiatry. 2019;85:226-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 202. | Bruchas MR, Schindler AG, Shankar H, Messinger DI, Miyatake M, Land BB, Lemos JC, Hagan CE, Neumaier JF, Quintana A, Palmiter RD, Chavkin C. Selective p38α MAPK deletion in serotonergic neurons produces stress resilience in models of depression and addiction. Neuron. 2011;71:498-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 221] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 203. | Xie L, Chen J, Ding YM, Gui XW, Wu LX, Tian S, Wu W. MicroRNA-26a-2 maintains stress resiliency and antidepressant efficacy by targeting the serotonergic autoreceptor HTR1A. Biochem Biophys Res Commun. 2019;511:440-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 204. | Prakash N, Stark CJ, Keisler MN, Luo L, Der-Avakian A, Dulcis D. Serotonergic Plasticity in the Dorsal Raphe Nucleus Characterizes Susceptibility and Resilience to Anhedonia. J Neurosci. 2020;40:569-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 205. | Mineur YS, Obayemi A, Wigestrand MB, Fote GM, Calarco CA, Li AM, Picciotto MR. Cholinergic signaling in the hippocampus regulates social stress resilience and anxiety- and depression-like behavior. Proc Natl Acad Sci U S A. 2013;110:3573-3578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 285] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 206. | Mineur YS, Fote GM, Blakeman S, Cahuzac EL, Newbold SA, Picciotto MR. Multiple Nicotinic Acetylcholine Receptor Subtypes in the Mouse Amygdala Regulate Affective Behaviors and Response to Social Stress. Neuropsychopharmacology. 2016;41:1579-1587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 207. | Rosa SG, Pesarico AP, Nogueira CW. m-Trifluoromethyl-diphenyl diselenide promotes resilience to social avoidance induced by social defeat stress in mice: Contribution of opioid receptors and MAPKs. Prog Neuropsychopharmacol Biol Psychiatry. 2018;82:123-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 208. | Bérubé P, Laforest S, Bhatnagar S, Drolet G. Enkephalin and dynorphin mRNA expression are associated with resilience or vulnerability to chronic social defeat stress. Physiol Behav. 2013;122:237-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 209. | Browne CA, Falcon E, Robinson SA, Berton O, Lucki I. Reversal of Stress-Induced Social Interaction Deficits by Buprenorphine. Int J Neuropsychopharmacol. 2018;21:164-174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 210. | Henry MS, Bisht K, Vernoux N, Gendron L, Torres-Berrio A, Drolet G, Tremblay MÈ. Delta Opioid Receptor Signaling Promotes Resilience to Stress Under the Repeated Social Defeat Paradigm in Mice. Front Mol Neurosci. 2018;11:100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 211. | Nam H, Chandra R, Francis TC, Dias C, Cheer JF, Lobo MK. Reduced nucleus accumbens enkephalins underlie vulnerability to social defeat stress. Neuropsychopharmacology. 2019;44:1876-1885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 212. | Briand LA, Hilario M, Dow HC, Brodkin ES, Blendy JA, Berton O. Mouse model of OPRM1 (A118G) polymorphism increases sociability and dominance and confers resilience to social defeat. J Neurosci. 2015;35:3582-3590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 213. | Wu G, Feder A, Wegener G, Bailey C, Saxena S, Charney D, Mathé AA. Central functions of neuropeptide Y in mood and anxiety disorders. Expert Opin Ther Targets. 2011;15:1317-1331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 122] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 214. | Schmeltzer SN, Herman JP, Sah R. Neuropeptide Y (NPY) and posttraumatic stress disorder (PTSD): A translational update. Exp Neurol. 2016;284:196-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 215. | He Y, Li W, Tian Y, Chen X, Cheng K, Xu K, Li C, Wang H, Qu C, Wang C, Li P, Chen H, Xie P. iTRAQ-based proteomics suggests LRP6, NPY and NPY2R perturbation in the hippocampus involved in CSDS may induce resilience and susceptibility. Life Sci. 2018;211:102-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 216. | Lacey T, Sweeting J, Kingston R, Smith M, Markham CM. Neuropeptide Y impairs the acquisition of conditioned defeat in Syrian hamsters. Neurosci Lett. 2019;690:214-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 217. | Alviña K, Jodeiri Farshbaf M, Mondal AK. Long term effects of stress on hippocampal function: Emphasis on early life stress paradigms and potential involvement of neuropeptide Y. J Neurosci Res. 2021;99:57-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 218. | Kagerer SM, Jöhren O. Interactions of orexins/hypocretins with adrenocortical functions. Acta Physiol (Oxf). 2010;198:361-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 219. | Srinivasan S, Shariff M, Bartlett SE. The role of the glucocorticoids in developing resilience to stress and addiction. Front Psychiatry. 2013;4:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 220. | Chung HS, Kim JG, Kim JW, Kim HW, Yoon BJ. Orexin administration to mice that underwent chronic stress produces bimodal effects on emotion-related behaviors. Regul Pept. 2014;194-195:16-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 221. | Arendt DH, Hassell J, Li H, Achua JK, Guarnieri DJ, Dileone RJ, Ronan PJ, Summers CH. Anxiolytic function of the orexin 2/hypocretin A receptor in the basolateral amygdala. Psychoneuroendocrinology. 2014;40:17-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 222. | Grafe LA, Eacret D, Dobkin J, Bhatnagar S. Reduced Orexin System Function Contributes to Resilience to Repeated Social Stress. eNeuro. 2018;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 223. | Staton CD, Yaeger JDW, Khalid D, Haroun F, Fernandez BS, Fernandez JS, Summers BK, Summers TR, Sathyanesan M, Newton SS, Summers CH. Orexin 2 receptor stimulation enhances resilience, while orexin 2 inhibition promotes susceptibility, to social stress, anxiety and depression. Neuropharmacology. 2018;143:79-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 224. | Summers CH, Yaeger JDW, Staton CD, Arendt DH, Summers TR. Orexin/hypocretin receptor modulation of anxiolytic and antidepressive responses during social stress and decision-making: Potential for therapy. Brain Res. 2020;1731:146085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 225. | Pesarico AP, Rosa SG, Martini F, Goulart TA, Zeni G, Nogueira CW. Brain-derived neurotrophic factor signaling plays a role in resilience to stress promoted by isoquinoline in defeated mice. J Psychiatr Res. 2017;94:78-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 226. | Iñiguez SD, Vialou V, Warren BL, Cao JL, Alcantara LF, Davis LC, Manojlovic Z, Neve RL, Russo SJ, Han MH, Nestler EJ, Bolaños-Guzmán CA. Extracellular signal-regulated kinase-2 within the ventral tegmental area regulates responses to stress. J Neurosci. 2010;30:7652-7663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 89] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 227. | Castrén E, Rantamäki T. The role of BDNF and its receptors in depression and antidepressant drug action: Reactivation of developmental plasticity. Dev Neurobiol. 2010;70:289-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 642] [Article Influence: 42.8] [Reference Citation Analysis (0)] |
| 228. | Li Y, Luikart BW, Birnbaum S, Chen J, Kwon CH, Kernie SG, Bassel-Duby R, Parada LF. TrkB regulates hippocampal neurogenesis and governs sensitivity to antidepressive treatment. Neuron. 2008;59:399-412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 509] [Cited by in RCA: 493] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 229. | Autry AE, Monteggia LM. Brain-derived neurotrophic factor and neuropsychiatric disorders. Pharmacol Rev. 2012;64:238-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1096] [Cited by in RCA: 1026] [Article Influence: 78.9] [Reference Citation Analysis (0)] |
| 230. | Pagliusi MOF Jr, Bonet IJM, Dias EV, Vieira AS, Tambeli CH, Parada CA, Sartori CR. Social defeat stress induces hyperalgesia and increases truncated BDNF isoforms in the nucleus accumbens regardless of the depressive-like behavior induction in mice. Eur J Neurosci. 2018;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 231. | Mallei A, Ieraci A, Popoli M. Chronic social defeat stress differentially regulates the expression of BDNF transcripts and epigenetic modifying enzymes in susceptible and resilient mice. World J Biol Psychiatry. 2019;20:555-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 232. | Yao W, Lin S, Su J, Cao Q, Chen Y, Chen J, Zhang Z, Hashimoto K, Qi Q, Zhang JC. Activation of BDNF by transcription factor Nrf2 contributes to antidepressant-like actions in rodents. Transl Psychiatry. 2021;11:140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 73] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 233. | Taliaz D, Loya A, Gersner R, Haramati S, Chen A, Zangen A. Resilience to chronic stress is mediated by hippocampal brain-derived neurotrophic factor. J Neurosci. 2011;31:4475-4483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 211] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 234. | Nasrallah P, Haidar EA, Stephan JS, El Hayek L, Karnib N, Khalifeh M, Barmo N, Jabre V, Houbeika R, Ghanem A, Nasser J, Zeeni N, Bassil M, Sleiman SF. Branched-chain amino acids mediate resilience to chronic social defeat stress by activating BDNF/TRKB signaling. Neurobiol Stress. 2019;11:100170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 235. | Lagace DC, Donovan MH, DeCarolis NA, Farnbauch LA, Malhotra S, Berton O, Nestler EJ, Krishnan V, Eisch AJ. Adult hippocampal neurogenesis is functionally important for stress-induced social avoidance. Proc Natl Acad Sci U S A. 2010;107:4436-4441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 244] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 236. | Duclot F, Kabbaj M. Individual differences in novelty seeking predict subsequent vulnerability to social defeat through a differential epigenetic regulation of brain-derived neurotrophic factor expression. J Neurosci. 2013;33:11048-11060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 237. | Khalifeh M, Hobeika R, El Hayek L, Saad J, Eid F, El-Khoury R, Ghayad LM, Jabre V, Nasrallah P, Barmo N, Stephan JS, Khnayzer R, Khalil C, Sleiman SF. Nicotine induces resilience to chronic social defeat stress in a mouse model of water pipe tobacco exposure by activating BDNF signaling. Behav Brain Res. 2020;382:112499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 238. | Han QQ, Yang L, Huang HJ, Wang YL, Yu R, Wang J, Pilot A, Wu GC, Liu Q, Yu J. Differential GR Expression and Translocation in the Hippocampus Mediates Susceptibility vs. Resilience to Chronic Social Defeat Stress. Front Neurosci. 2017;11:287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 239. | Wang W, Liu L, Yang X, Gao H, Tang QK, Yin LY, Yin XY, Hao JR, Geng DQ, Gao C. Ketamine improved depressive-like behaviors via hippocampal glucocorticoid receptor in chronic stress induced- susceptible mice. Behav Brain Res. 2019;364:75-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 240. | Dedic N, Kühne C, Gomes KS, Hartmann J, Ressler KJ, Schmidt MV, Deussing JM. Deletion of CRH From GABAergic Forebrain Neurons Promotes Stress Resilience and Dampens Stress-Induced Changes in Neuronal Activity. Front Neurosci. 2019;13:986. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 241. | Chen P, Lou S, Huang ZH, Wang Z, Shan QH, Wang Y, Yang Y, Li X, Gong H, Jin Y, Zhang Z, Zhou JN. Prefrontal Cortex Corticotropin-Releasing Factor Neurons Control Behavioral Style Selection under Challenging Situations. Neuron. 2020;106:301-315.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 242. | Nestler EJ, Waxman SG. Resilience to Stress and Resilience to Pain: Lessons from Molecular Neurobiology and Genetics. Trends Mol Med. 2020;26:924-935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 243. | Dudek KA, Kaufmann FN, Lavoie O, Menard C. Central and peripheral stress-induced epigenetic mechanisms of resilience. Curr Opin Psychiatry. 2021;34:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 244. | Wilkinson MB, Xiao G, Kumar A, LaPlant Q, Renthal W, Sikder D, Kodadek TJ, Nestler EJ. Imipramine treatment and resiliency exhibit similar chromatin regulation in the mouse nucleus accumbens in depression models. J Neurosci. 2009;29:7820-7832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 196] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 245. | Kenworthy CA, Sengupta A, Luz SM, Ver Hoeve ES, Meda K, Bhatnagar S, Abel T. Social defeat induces changes in histone acetylation and expression of histone modifying enzymes in the ventral hippocampus, prefrontal cortex, and dorsal raphe nucleus. Neuroscience. 2014;264:88-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 246. | Covington HE 3rd, Maze I, Sun H, Bomze HM, DeMaio KD, Wu EY, Dietz DM, Lobo MK, Ghose S, Mouzon E, Neve RL, Tamminga CA, Nestler EJ. A role for repressive histone methylation in cocaine-induced vulnerability to stress. Neuron. 2011;71:656-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 226] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 247. | Espallergues J, Teegarden SL, Veerakumar A, Boulden J, Challis C, Jochems J, Chan M, Petersen T, Deneris E, Matthias P, Hahn CG, Lucki I, Beck SG, Berton O. HDAC6 regulates glucocorticoid receptor signaling in serotonin pathways with critical impact on stress resilience. J Neurosci. 2012;32:4400-4416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 109] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 248. | Jochems J, Teegarden SL, Chen Y, Boulden J, Challis C, Ben-Dor GA, Kim SF, Berton O. Enhancement of stress resilience through histone deacetylase 6-mediated regulation of glucocorticoid receptor chaperone dynamics. Biol Psychiatry. 2015;77:345-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 249. | Ambrée O, Ruland C, Scheu S, Arolt V, Alferink J. Alterations of the Innate Immune System in Susceptibility and Resilience After Social Defeat Stress. Front Behav Neurosci. 2018;12:141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 250. | Hodes GE, Pfau ML, Leboeuf M, Golden SA, Christoffel DJ, Bregman D, Rebusi N, Heshmati M, Aleyasin H, Warren BL, Lebonté B, Horn S, Lapidus KA, Stelzhammer V, Wong EH, Bahn S, Krishnan V, Bolaños-Guzman CA, Murrough JW, Merad M, Russo SJ. Individual differences in the peripheral immune system promote resilience vs susceptibility to social stress. Proc Natl Acad Sci U S A. 2014;111:16136-16141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 419] [Cited by in RCA: 523] [Article Influence: 47.5] [Reference Citation Analysis (0)] |
| 251. | Finnell JE, Lombard CM, Melson MN, Singh NP, Nagarkatti M, Nagarkatti P, Fadel JR, Wood CS, Wood SK. The protective effects of resveratrol on social stress-induced cytokine release and depressive-like behavior. Brain Behav Immun. 2017;59:147-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 73] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 252. | Nasca C, Menard C, Hodes G, Bigio B, Pena C, Lorsch Z, Zelli D, Ferris A, Kana V, Purushothaman I, Dobbin J, Nassim M, DeAngelis P, Merad M, Rasgon N, Meaney M, Nestler EJ, McEwen BS, Russo SJ. Multidimensional Predictors of Susceptibility and Resilience to Social Defeat Stress. Biol Psychiatry. 2019;86:483-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 253. | Pearson-Leary J, Zhao C, Bittinger K, Eacret D, Luz S, Vigderman AS, Dayanim G, Bhatnagar S. The gut microbiome regulates the increases in depressive-type behaviors and in inflammatory processes in the ventral hippocampus of stress vulnerable rats. Mol Psychiatry. 2020;25:1068-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 144] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 254. | Pfau ML, Menard C, Cathomas F, Desland F, Kana V, Chan KL, Shimo Y, LeClair K, Flanigan ME, Aleyasin H, Walker DM, Bouchard S, Mack M, Hodes GE, Merad MM, Russo SJ. Role of Monocyte-Derived MicroRNA106b∼25 in Resilience to Social Stress. Biol Psychiatry. 2019;86:474-482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 255. | Szyszkowicz JK, Wong A, Anisman H, Merali Z, Audet MC. Implications of the gut microbiota in vulnerability to the social avoidance effects of chronic social defeat in male mice. Brain Behav Immun. 2017;66:45-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 84] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 256. | Aguilar MA, Cannella N, Ferragud A, Spanagel R. Editorial: Neurobehavioural Mechanisms of Resilience and Vulnerability in Addictive Disorders. Front Behav Neurosci. 2020;14:644495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 257. | Manvich DF, Stowe TA, Godfrey JR, Weinshenker D. A Method for Psychosocial Stress-Induced Reinstatement of Cocaine Seeking in Rats. Biol Psychiatry. 2016;79:940-946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 258. | Ellis AS, Fosnocht AQ, Lucerne KE, Briand LA. Disruption of GluA2 phosphorylation potentiates stress responsivity. Behav Brain Res. 2017;333:83-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 259. | Guerrero-Bautista R, Do Couto BR, Hidalgo JM, Cárceles-Moreno FJ, Molina G, Laorden ML, Núñez C, Milanés MV. Modulation of stress- and cocaine prime-induced reinstatement of conditioned place preference after memory extinction through dopamine D3 receptor. Prog Neuropsychopharmacol Biol Psychiatry. 2019;92:308-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 260. | Fallon IP, Tanner MK, Greenwood BN, Baratta MV. Sex differences in resilience: Experiential factors and their mechanisms. Eur J Neurosci. 2020;52:2530-2547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 261. | Hodes GE, Epperson CN. Sex Differences in Vulnerability and Resilience to Stress Across the Life Span. Biol Psychiatry. 2019;86:421-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 282] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 262. | Conrad CD, Grote KA, Hobbs RJ, Ferayorni A. Sex differences in spatial and non-spatial Y-maze performance after chronic stress. Neurobiol Learn Mem. 2003;79:32-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 166] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 263. | Galea LA, McEwen BS, Tanapat P, Deak T, Spencer RL, Dhabhar FS. Sex differences in dendritic atrophy of CA3 pyramidal neurons in response to chronic restraint stress. Neuroscience. 1997;81:689-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 418] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 264. | Bowman RE, Beck KD, Luine VN. Chronic stress effects on memory: sex differences in performance and monoaminergic activity. Horm Behav. 2003;43:48-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 221] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 265. | Wood GE, Shors TJ. Stress facilitates classical conditioning in males, but impairs classical conditioning in females through activational effects of ovarian hormones. Proc Natl Acad Sci U S A. 1998;95:4066-4071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 272] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 266. | Wood GE, Beylin AV, Shors TJ. The contribution of adrenal and reproductive hormones to the opposing effects of stress on trace conditioning in males vs females. Behav Neurosci. 2001;115:175-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 157] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 267. | Wellman CL, Bangasser DA, Bollinger JL, Coutellier L, Logrip ML, Moench KM, Urban KR. Sex Differences in Risk and Resilience: Stress Effects on the Neural Substrates of Emotion and Motivation. J Neurosci. 2018;38:9423-9432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 268. | Okine T, Shepard R, Lemanski E, Coutellier L. Sex Differences in the Sustained Effects of Ketamine on Resilience to Chronic Stress. Front Behav Neurosci. 2020;14:581360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 269. | Vassilev P, Pantoja-Urban AH, Giroux M, Nouel D, Hernandez G, Orsini T, Flores C. Unique effects of social defeat stress in adolescent male mice on the Netrin-1/DCC pathway, prefrontal cortex dopamine and cognition (Social stress in adolescent vs. adult male mice). eNeuro. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 270. | Bath KG, Russo SJ, Pleil KE, Wohleb ES, Duman RS, Radley JJ. Circuit and synaptic mechanisms of repeated stress: Perspectives from differing contexts, duration, and development. Neurobiol Stress. 2017;7:137-151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 271. | Douglas AJ, Brunton PJ, Bosch OJ, Russell JA, Neumann ID. Neuroendocrine responses to stress in mice: hyporesponsiveness in pregnancy and parturition. Endocrinology. 2003;144:5268-5276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 98] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 272. | Bitencourt RM, Takahashi RN. Cannabidiol as a Therapeutic Alternative for Post-traumatic Stress Disorder: From Bench Research to Confirmation in Human Trials. Front Neurosci. 2018;12:502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 273. | Calpe-López C, García-Pardo MP, Aguilar MA. Cannabidiol Treatment Might Promote Resilience to Cocaine and Methamphetamine Use Disorders: A Review of Possible Mechanisms. Molecules. 2019;24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 274. | Ledesma JC, Manzanedo C, Aguilar MA. Cannabidiol prevents several of the behavioral alterations related to cocaine addiction in mice. Prog Neuropsychopharmacol Biol Psychiatry. 2021;111:110390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 275. | Nona CN, Hendershot CS, Le Foll B. Effects of cannabidiol on alcohol-related outcomes: A review of preclinical and human research. Exp Clin Psychopharmacol. 2019;27:359-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 276. | Kumar A, Chanana P. Role of Nitric Oxide in Stress-Induced Anxiety: From Pathophysiology to Therapeutic Target. Vitam Horm. 2017;103:147-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 277. | Liddie S, Balda MA, Itzhak Y. Nitric oxide (NO) signaling as a potential therapeutic modality against psychostimulants. Curr Pharm Des. 2013;19:7092-7102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 278. | Boutrel B, Kenny PJ, Specio SE, Martin-Fardon R, Markou A, Koob GF, de Lecea L. Role for hypocretin in mediating stress-induced reinstatement of cocaine-seeking behavior. Proc Natl Acad Sci U S A. 2005;102:19168-19173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 370] [Cited by in RCA: 409] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 279. | Yoshida K, Drew MR, Kono A, Mimura M, Takata N, Tanaka KF. Chronic social defeat stress impairs goal-directed behavior through dysregulation of ventral hippocampal activity in male mice. Neuropsychopharmacology. 2021;46:1606-1616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 280. | Atesyakar N, Canbeyli R, Unal G. Low cognitive competence as a vulnerability factor for behavioral despair in rats. Behav Processes. 2020;174:104103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 281. | Averill LA, Averill CL, Kelmendi B, Abdallah CG, Southwick SM. Stress Response Modulation Underlying the Psychobiology of Resilience. Curr Psychiatry Rep. 2018;20:27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 282. | Baratta MV, Maier SF. New tools for understanding coping and resilience. Neurosci Lett. 2019;693:54-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 283. | Ersche KD, Meng C, Ziauddeen H, Stochl J, Williams GB, Bullmore ET, Robbins TW. Brain networks underlying vulnerability and resilience to drug addiction. Proc Natl Acad Sci U S A. 2020;117:15253-15261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 284. | Osório C, Probert T, Jones E, Young AH, Robbins I. Adapting to Stress: Understanding the Neurobiology of Resilience. Behav Med. 2017;43:307-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 115] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 285. | Albrecht A, Redavide E, Regev-Tsur S, Stork O, Richter-Levin G. Hippocampal GABAergic interneurons and their co-localized neuropeptides in stress vulnerability and resilience. Neurosci Biobehav Rev. 2021;122:229-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 286. | Stainton A, Chisholm K, Kaiser N, Rosen M, Upthegrove R, Ruhrmann S, Wood SJ. Resilience as a multimodal dynamic process. Early Interv Psychiatry. 2019;13:725-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 126] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 287. | García-Pardo MP, Miñarro J, Llansola M, Felipo V, Aguilar MA. Role of NMDA and AMPA glutamatergic receptors in the effects of social defeat on the rewarding properties of MDMA in mice. Eur J Neurosci. 2019;50:2623-2634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |