Published online Feb 4, 2016. doi: 10.5492/wjccm.v5.i1.7
Peer-review started: July 16, 2015
First decision: September 28, 2015
Revised: October 22, 2015
Accepted: December 8, 2015
Article in press: December 11, 2015
Published online: February 4, 2016
Processing time: 191 Days and 18.6 Hours
Point-of-care ultrasound has been increasingly used in evaluating shocked patients including the measurement of inferior vena cava (IVC) diameter. Operators should standardize their technique in scanning IVC. Relative changes are more important than absolute numbers. We advise using the longitudinal view (B mode) to evaluate the gross collapsibility, and the M mode to measure the IVC diameter. Combining the collapsibility and diameter size will increase the value of IVC measurement. This approach has been very useful in the resuscitation of shocked patients, monitoring their fluid demands, and predicting recurrence of shock. Pitfalls in measuring IVC diameter include increased intra-thoracic pressure by mechanical ventilation or increased right atrial pressure by pulmonary embolism or heart failure. The IVC diameter is not useful in cases of increased intra-abdominal pressure (abdominal compartment syndrome) or direct pressure on the IVC. The IVC diameter should be combined with focused echocardiography and correlated with the clinical picture as a whole to be useful.
Core tip: Bedside measurement of inferior vena cava is useful in evaluating and resuscitating shocked patients. To achieve that, the operator should be well-trained, use standardized techniques, understand ultrasound limitations, and finally correlate the findings with the clinical picture as a whole.
- Citation: Abu-Zidan FM. Optimizing the value of measuring inferior vena cava diameter in shocked patients. World J Crit Care Med 2016; 5(1): 7-11
- URL: https://www.wjgnet.com/2220-3141/full/v5/i1/7.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v5.i1.7
Point-of-care ultrasound has been increasingly used in evaluating shocked patients including the measurement of inferior vena cava diameter (IVC)[1-3]. Nevertheless, there have been conflicting results regarding its value[4-6]. It is important to highlight the technical and clinical difficulties that may be encountered in measuring the IVC diameter as these limit its use. There are four components that affect the outcome of ultrasound studies. These are the effectiveness and technical limitations of the ultrasound machine, the experience of the operator, the body built of the patient, and the pathology studied.
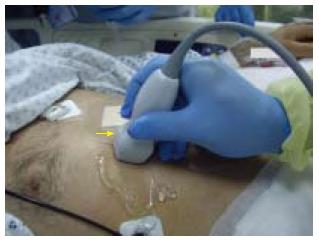
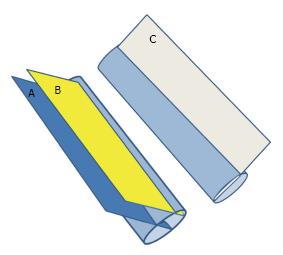
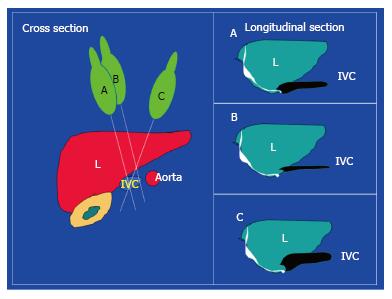
Operators should standardize their technique in scanning the IVC. IVC can be measured through different approaches including the subxiphoid or subcostal approach[7,8]. We prefer to measure the IVC directly through a trans-hepatic approach using a portable machine and a small print convex array probe with a frequency of 3-5 MHZ while the patient is in supine position. The probe is located in the mid-clavicular line between the ribs of the right lower chest wall at 90 degrees perpendicular to the skin. The marker points proximally towards the head (Figure 1). The probe may be slightly directed towards the right to be parallel to the IVC. The probe is then shifted slowly transversely to get the best longitudinal perpendicular view. We think that this is better than the subxiphoid approach[7] as the IVC is located slightly to right and the diameter of the IVC may be overestimated by getting an oblique section (Figure 2).
The ultrasound cross section should be vertical to the IVC. Common pitfalls in measurement include measuring the IVC obliquely or peripherally (Figures 2 and 3). In general, it is advised to use the B mode to evaluate the gross collapsibility of the IVC and the M mode to accurately measure the changes in IVC diameter. The IVC can be measured in both longitudinal and transverse sections.
Pitfalls in measuring IVC include increased intra-thoracic pressure resulting from mechanical ventilation or increased right atrial pressure resulting from heart failure or pulmonary embolism. These conditions will increase the diameter of the abdominal IVC[3]. We have recently reported that IVC diameter was not useful in guiding resuscitation, and was even misleading in abdominal compartment syndrome[9]. The increased pressure in abdominal compartment syndrome will compress the IVC and reduce its antero-posterior diameter. The unexperienced clinician may increase the fluid resuscitation which would further decrease the diameter. Furthermore, direct pressure on the IVC as in late pregnancy and acute gastric dilatation[10] can affect the measurement. The IVC diameter should be combined with focused echocardiography and correlated with the clinical picture as a whole to be useful.
IVC measurement can be used as part of defined protocols in diagnosing shocked patients to optimize its value. These protocols evaluate the heart, IVC, chest, and the abdomen to try defining the cause of the shock. Our group follows the RUSH protocol which examines the pump (heart), tubes (great vessels) and reservoir (free intra-peritoneal or intra-thoracic fluid)[2]. Vegas et al[7] use the same principles but in a different approach, whereby they classify the shocked patients into those with (1) reduced mean systemic venous pressure; (2) increased right atrial pressure; and (3) increased resistance to the venous return. They study the size of IVC, respiratory variation of the IVC, and the hepatic venous flow to define the type of shock[7].
In a study of 47 patients having septic shock, Coen et al[11] used the variability of IVC diameter to decide the volume of fluid resuscitation. They gave boluses of 500 mL of crystalloids as needed to reach an IVC index of 30%-50% which was defined as [(maximum IVC diameter - minimum IVC diameter)/maximum IVC diameter] × 100. IVC measurement was feasible in 92% of the cases and central venous catheter was avoided in more than one third of the patients. The IVC index was significantly higher in shocked patients compared with non shocked patients[12].
The IVC diameter was negatively correlated with the lactate level and positively correlated with the base excess level during hemorrhagic shock resuscitation indicating its good clinical value[8]. Furthermore, Yanagawa et al[13] prospectively studied 30 trauma patients and found that the relative change of IVC diameter is effective in differentiating stable resuscitation responders from transient responders who develop recurrent shock. These findings were supported by Feissel et al[14] who found the same results in ventilated septic patients. Furthermore, Schefold et al[15] found that IVC diameter was highly correlated with the central venous pressure and extravascular lung water index in septic ventilated intensive care unit (ICU) patients. This may be helpful in avoiding unnecessary volume expansion in these sick patients. In contrast, Corl et al[5] found that measuring the IVC index was not a good marker for proper fluid responsiveness in the emergency department and questioned its value.
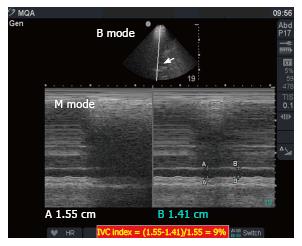
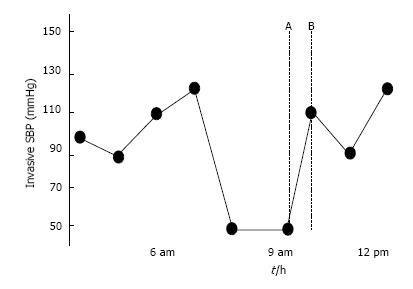
Weekes et al[4] prospectively evaluated the gross appearance of IVC and correlated it with the actual measured size in 24 hypotensive patients. They developed a three point scale of visual appearance of IVC as follows: (1) decreased IVC index of ≤ 0.3; (2) normal range (0.31-0.69); and (3) increased index ≥ 0.7. Serial gross evaluation of IVC agreed with the actual measured IVC during fluid resuscitation. This study supports the opinion that relative changes are more important than absolute numbers. Gross collapsibility is a more useful marker for hypovolemia than IVC collapsibility index[3]. We advise using the longitudinal view (B mode) to evaluate the gross collapsibility, and the M mode to measure the diameter of IVC. Combining the collapsibility and diameter will increase the value of IVC measurement. This approach has been very useful in our hands (Figures 4-6).
There is no doubt that this area needs more evidence based approach. Dipti et al[6] in a meta-analysis that was published in 2012, studied the value of IVC diameter in estimating volume status in adults. They searched 5 major databases and combined 5 prospective studies on this topic. The meta-analysis included 86 hypovolemic patients and 189 controls. IVC diameter was significantly less in hypovolemic patients compared with controls. These studies stemmed from 4 countries having good external validity and a spectrum of different disease severity. Nevertheless, the analysis had a very high heterogeneity (I2 = 99%), no randomized controlled study was included, and the sample size was small.
A recent prospective randomized controlled trial in injured patients having hypotension or tachycardia treated in a level I trauma center in United States has just been published[16]. It compared transthoracic limited echocardiography including measuring IVC (106 patients) with usual care (134 patients). The outcome variables were fluid requirement, time to surgery, percentage of ICU admission, and mortality. This study shows that limited echocardiography significantly reduced the IV fluid requirements (average of 1.5 L compared with 2.5 L) and significantly reduced the time to the operating theatre (by 50%). It also alerted the physicians to the seriousness of some cases and increased the likelihood of ICU admission of such cases (from 67% to 80%).
It may be argued that the statements expressed in this editorial are biased. I have been interested in and passionate about point of care ultrasound for a quarter of a century, since time when it was not yet commonly used by surgeons or intensivists. I have observed the dramatic improvement in this field over time including the huge progress in point-of-care ultrasound and the development of acute care surgery as a special entity. I have been measuring the IVC diameter in critically-ill and trauma shocked patients as an acute care surgeon over the last 8 years and using it in making very critical decisions in a busy acute care hospital. From personal experience, I am confident that bedside measurement of IVC is to stay. It is useful in evaluating and resuscitating shocked patients. To achieve that, the operator should be well-trained, use standardized techniques, understand ultrasound limitations, and finally correlate the findings with the clinical picture as a whole.
The author thanks Ms. Geraldine Kershaw, Lecturer, Medical Communication and Study Skills, Department of Medical Education, College of Medicine and Health Sciences, UAE University for language and grammar corrections.
P- Reviewer: Boucek C, Inaba H, Lin J, Willms D S- Editor: Gong XM L- Editor: A E- Editor: Lu YJ
| 1. | Abu-Zidan FM. Point-of-care ultrasound in critically ill patients: Where do we stand? J Emerg Trauma Shock. 2012;5:70-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 2. | Seif D, Perera P, Mailhot T, Riley D, Mandavia D. Bedside ultrasound in resuscitation and the rapid ultrasound in shock protocol. Crit Care Res Pract. 2012;2012:503254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 3. | Seif D, Mailhot T, Perera P, Mandavia D. Caval sonography in shock: a noninvasive method for evaluating intravascular volume in critically ill patients. J Ultrasound Med. 2012;31:1885-1890. [PubMed] |
| 4. | Weekes AJ, Tassone HM, Babcock A, Quirke DP, Norton HJ, Jayarama K, Tayal VS. Comparison of serial qualitative and quantitative assessments of caval index and left ventricular systolic function during early fluid resuscitation of hypotensive emergency department patients. Acad Emerg Med. 2011;18:912-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 5. | Corl K, Napoli AM, Gardiner F. Bedside sonographic measurement of the inferior vena cava caval index is a poor predictor of fluid responsiveness in emergency department patients. Emerg Med Australas. 2012;24:534-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 6. | Dipti A, Soucy Z, Surana A, Chandra S. Role of inferior vena cava diameter in assessment of volume status: a meta-analysis. Am J Emerg Med. 2012;30:1414-1419.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 173] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 7. | Vegas A, Denault A, Royse C. A bedside clinical and ultrasound-based approach to hemodynamic instability - Part II: bedside ultrasound in hemodynamic shock: continuing professional development. Can J Anaesth. 2014;61:1008-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 8. | Akilli B, Bayir A, Kara F, Ak A, Cander B. Inferior vena cava diameter as a marker of early hemorrhagic shock: a comparative study. Ulus Travma Acil Cerrahi Derg. 2010;16:113-118. [PubMed] |
| 9. | Abu-Zidan FM, Idris K. Sonographic Measurement of the IVC Diameter as an Indicator for Fluid Resuscitation: Beware of the Intra-abdominal Pressure. World J Surg. 2015;39:2608-2609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Abu-Zidan FM, Hefny AF, Saadeldinn YA, El-Ashaal YI. Sonographic findings of superior mesenteric artery syndrome causing massive gastric dilatation in a young healthy girl. Singapore Med J. 2010;51:e184-e186. [PubMed] |
| 11. | Coen D, Cortellaro F, Pasini S, Tombini V, Vaccaro A, Montalbetti L, Cazzaniga M, Boghi D. Towards a less invasive approach to the early goal-directed treatment of septic shock in the ED. Am J Emerg Med. 2014;32:563-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 12. | Sefidbakht S, Assadsangabi R, Abbasi HR, Nabavizadeh A. Sonographic measurement of the inferior vena cava as a predictor of shock in trauma patients. Emerg Radiol. 2007;14:181-185. [PubMed] |
| 13. | Yanagawa Y, Sakamoto T, Okada Y. Hypovolemic shock evaluated by sonographic measurement of the inferior vena cava during resuscitation in trauma patients. J Trauma. 2007;63:1245-1248; discussion 1248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 75] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 14. | Feissel M, Michard F, Faller JP, Teboul JL. The respiratory variation in inferior vena cava diameter as a guide to fluid therapy. Intensive Care Med. 2004;30:1834-1837. [PubMed] |
| 15. | Schefold JC, Storm C, Bercker S, Pschowski R, Oppert M, Krüger A, Hasper D. Inferior vena cava diameter correlates with invasive hemodynamic measures in mechanically ventilated intensive care unit patients with sepsis. J Emerg Med. 2010;38:632-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 16. | Ferrada P, Evans D, Wolfe L, Anand RJ, Vanguri P, Mayglothling J, Whelan J, Malhotra A, Goldberg S, Duane T. Findings of a randomized controlled trial using limited transthoracic echocardiogram (LTTE) as a hemodynamic monitoring tool in the trauma bay. J Trauma Acute Care Surg. 2014;76:31-37; discussion 37-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |