Published online May 8, 2016. doi: 10.5409/wjcp.v5.i2.228
Peer-review started: November 2, 2015
First decision: December 4, 2015
Revised: January 19, 2016
Accepted: March 9, 2016
Article in press: March 14, 2016
Published online: May 8, 2016
Processing time: 188 Days and 22.9 Hours
Aural atresia is a rare congenital malformation of the external and middle ear. There are several syndromic associations of this anomaly with those involving the first and second branchial arches. Occurrence of aural atresia with sclerosing skeletal dysplasia is unknown and has never been reported. The co-existence of a sclerosing dysplasia can make the surgical treatment in aural atresia difficult and risky; and the auditory improvement may not be as expected. Moreover, internal auditory canal narrowing and hence sensorineural hearing loss in sclerosing dysplasia might add to the already existing conductive hearing loss in such patients. In this case report we have described an unknown association of bilateral microtia with sclerosing skeletal dysplasia (autosomal dominant osteopetrosis) and clinical implications of these two conditions occurring together leading to a change in the management plan.
Core tip: Congenital aural atresia and microtia are one of the most challenging surgeries for an ear, nose, and throat surgeon. It is imperative to know when not to operate a patient. Improper patient selection may not benefit the patient in terms of hearing improvement rather it may further add to complications like chronic cavity infection and potential risk of facial nerve injury. Imaging plays an important role in preoperative evaluation and selection of appropriate surgical candidates. This case shows incidental detection of previously unsuspected osteopetrosis in a child having profound congenital hearing loss due to congenital bilateral aural atresia that posed difficulty in treatment and required change in management from surgery to bone anchored hearing aid.
- Citation: Verma R, Jana M, Bhalla AS, Kumar A, Kumar R. Diagnosis of osteopetrosis in bilateral congenital aural atresia: Turning point in treatment strategy. World J Clin Pediatr 2016; 5(2): 228-233
- URL: https://www.wjgnet.com/2219-2808/full/v5/i2/228.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v5.i2.228
Aural atresia is a rare congenital malformation affecting the external, middle and rarely the inner ear structures with an incidence of 1:10000 to 1:20000 live births[1]. It is more often unilateral, and male to female ratio is 2:1. Though the entity is usually sporadic, upto 30%-40% cases may have syndromic association with Goldenhar’s syndrome, Treacher Collin syndrome, De Grouchy, Crouzon syndrome, Branchio-oto-renal syndrome and hemifacial macrosomia[2]. However, none of the described associated syndromes are known to have generalised increased bone density, and alternatively none of the sclerosing skeletal dysplasia is known to be associated with microtia.
We present an unusual case of incidental radiological detection of autosomal dominant osteopetrosis (ADOP) during pre-operative evaluation of a child with congenital bilateral microtia and its clinical implications in terms of patient management and treatment measures.
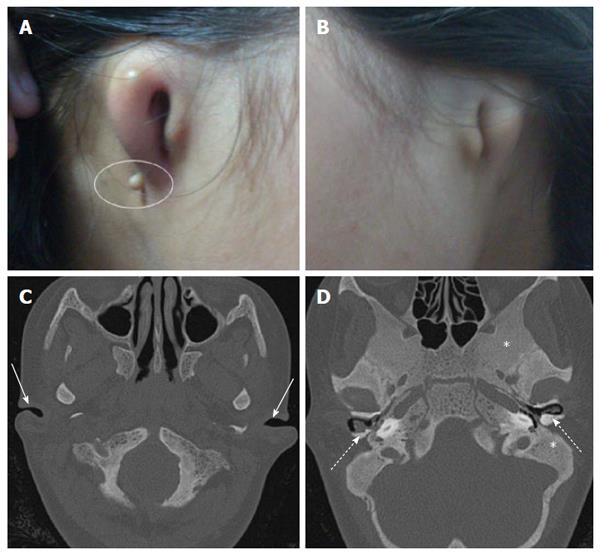
A 12-year-old adolescent female was referred to our department for High resolution computed tomography (HRCT) of temporal bone as a part of routine microtia work-up. Her both pinnae were severely malformed (Figure 1) and she had profound hearing loss since birth. She was not using any hearing aids. There was no history of similar condition in immediate or extended family members. There was no history of consanguineous marriage and drug exposure during pregnancy.
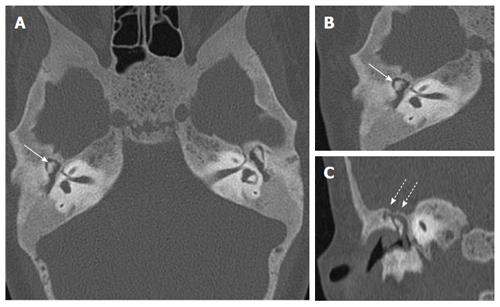
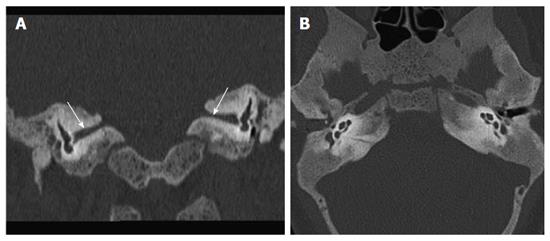
Audiometry revealed bilateral mixed (conductive as well as sensorineural) hearing loss. HRCT revealed that both cartilaginous external auditory canal (EAC) were narrowed while bony EAC and middle ear cavity were normal in size (Figure 1). Stapes was not visualised and oval window was shallow on both sides with bilateral poor pneumatisation of mastoids. There was Eustachian tube block with resultant soft tissue opacification of both the middle ears. Soft tissue opacification, ossicular erosion and erosion of tegmen tympani was also noted on right side suggesting congenital cholesteatoma (Figure 2). Besides the external and middle ear anomalies related to aural atresia the additional striking finding on CT was generalised bone thickening and sclerosis with loss of definition of the inner and outer table involving skull base and calvarial bones. There was bilateral internal auditory canal (IAC) narrowing consequent to calvarial thickening but vestibule, cochlea and the semicircular canals were normal (Figure 3).
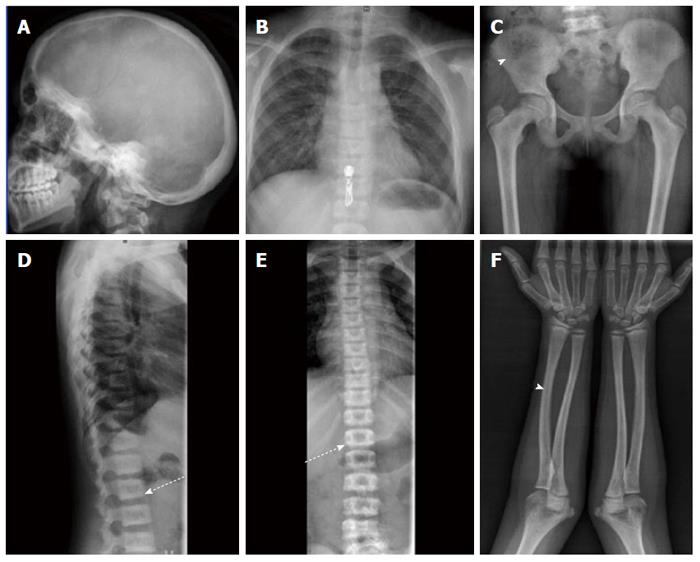
In view of sclerosis of skull bones skeletal survey was done for further evaluation. On skeletal survey, both axial and appendicular skeleton were affected, and there was mild generalised increase in bone density with sclerosis noted along superior as well as inferior vertebral endplates. The cortico-medullary differentiation of long bones was lost (Figure 4). There was no premature sutural closure, wormian bones and the shape of skull was normal. No endobones were seen and there was no abnormal soft tissue calcification or ossification of interosseous membranes. Based on these skeletal findings metabolic cause or a sclerosing skeletal dysplasia was suspected.
Biochemical workup revealed normal renal functions (Urea = 22 mg/dL, creatinine = 0.7) and blood counts (Hb = 13.4 g%). Serum calcium (9.6 mg%), phosphorus (4 mg/dL), alkaline phosphatase (90 IU/L), parathormone (PTH = 50 pg/mL) and fluoride levels (1.5 μmol/L) were within normal limits. This ruled out the diagnosis of metabolic bone disease and a final impression of sclerosing skeletal dysplasia likely ADOP was made (Figure 4).
Microtia refers to a malformed external ear or pinna, anotia denotes a more severe degree of deformity. EAC atresia refers to a narrowed or hypoplastic external auditory canal. Aural atresia is a birth defect that is characterised by hypoplasia of the EAC; often with pinna deformity and malformations of the middle ear, and rarely the inner ear structures.
Development of external and middle ear starts at six weeks of pregnancy from first and second branchial arches and is completed by twenty weeks[3]. EAC develops from the ectodermal components of the first branchial pouch. Meatal plug (mesodermal derivative) recanalizes and forms the EAC at the end of 28th week. Failure of recanalization leads to congenital aural atresia. Owing to common developmental origin, microtia and aural atresia can be associated with other craniofacial malformations resulting from abnormal development of first and second branchial arch structures. In addition to the syndromic associations described above, various non-syndromic associations known are facial asymmetry, facial nerve weakness, cleft lip and cleft palate, urogenital defects, and craniovertebral malformations[2,4].
Although sclerosing skeletal dysplasia like osteopetrosis can cause hearing loss by EAC and IAC narrowing due to bony involvement the deformity of pinna can not be explained. To the best of our knowledge, associations of microtia with sclerosing skeletal dysplasia has never been described in English medical literature.
Diffuse skeletal sclerosis can be caused by sclerosing skeletal dysplasias; and secondary to various other conditions such as renal osteodystrophy, hypoparathyroidism, fluorosis; Beryllium, lead and bismuth poisoning; myelofibrosis; Paget’s disease (sclerosing form); and malignancies (lymphoma, osteoblastic cancer metastases)[5].
Osteopetrosis is the prototype of sclerosing skeletal dysplasia caused by deficient osteoclastic resorption of the primary spongiosa. It can be inherited both as autosomal dominant and autosomal recessive forms. Autosomal recessive osteopetrosis (AROP) has a more severe disease course and early onset of disease. ADOP is ten times more common than AROP; is usually asymptomatic, and is usually discovered incidentally on plain radiographs[6,7]. It classically displays the radiographic sign of “sandwich vertebrae”. The main complications are confined to the skeleton, including fractures, scoliosis, hip osteoarthritis and osteomyelitis, particularly affecting the mandible in association with dental abscess or caries. The mainstay of diagnosis is clinical and largely depends on the radiographic appearance of the skeleton. In the absence of typical radiographic findings, raised concentrations of the creatine kinase BB isoenzyme and tartrate resistant acid phosphatase (TRAP) can be helpful in making the diagnosis of ADOP[8].
The goals of management in aural atresia are: (1) cosmetic reconstruction of the pinna; (2) canal reconstruction; and (3) hearing restoration.
Successful restoration of hearing in aural atresia after surgery depends on an intact sensorineural hearing. Contraindications to surgery are significant sensory neural hearing loss (SNHL) or inner ear malformation, limited middle ear-mastoid pneumatisation or a significantly hypoplastic middle-ear cleft. Pre-surgical work-up includes evaluation of sensorineural hearing by auditory brainstem response; and structural evaluation by HRCT of temporal bone. HRCT temporal bone is the most important imaging for pre-surgical evaluation; it differentiates surgical from non-surgical candidates and detects conditions that might increase the surgical risk (for example, anomalous facial nerve course, aberrant vascular structures, low lying tegmen tympani[9-11]). The status of atretic plate, pneumatisation of middle ear and mastoid, anatomy of ear ossicles, course of facial nerve and inner ear morphology are other important part of assessment[6]. Radiologically the most frequently followed scoring system is the Jahrsdoerfer scoring system, lesser the score poorer is the surgical outcome[10].
Causes of hearing loss in patients with osteopetrosis may vary[12]. Hearing is less commonly affected than vision, with approximately one third of patients having some degree of hearing loss. The deafness is probably secondary to a combination of bony compression of the nerve due to narrowing of IAC, sclerosis of the middle ear ossicles, chronic middle ear effusion due to Eustachian tube block, EAC and middle ear cavity narrowing. Our patient had associated pinna deformity, which can not be explained by osteopetrosis itself, and it implies that the EAC atresia in our patient is unrelated to ADOP.
Our case had microtia with bilateral cartilaginous EAC atresia and diffuse osteosclerosis. The secondary causes of osteosclerosis were excluded as she had a normal blood work-up, no history of exposure to heavy metals, normal facies, dentition, lip and palate. There was no pallor and hepatosplenomegaly. The combined clinical, radiological and biochemical picture led to diagnosis of sclerosing skeletal dysplasia which was further categorised as ADOP.
The management of our patient was difficult for multiple reasons. The expected auditory outcome was not good; firstly as she presented quite late (post-lingual deafness) and had not used hearing aids till then; secondly as there was mixed hearing loss with a sensorineural component resulting from the IAC narrowing because of ADOP. Additionally, the sclerosis of petrous and mastoid temporal bone because of ADOP made the surgery demanding; posing a high risk of facial nerve and cochlear injury resulting from excessive drilling. Since cosmetic reconstruction was still a feasible option, the patient was counselled for cosmetic reconstruction of the pinnae and Bone anchored hearing aid (BAHA) for hearing improvement. Considering poor prognosis and treatment cost patient refused the treatment and was lost to follow up.
Careful review of literature did not reveal any association of microtia and generalised increased bone density. To the best of our knowledge this is the first case describing bilateral microtia in a child with sclerosing skeletal dysplasia that was unsuspected prior to CT evaluation for microtia. The occurrence of two conditions together may be incidental or there may be some unknown unidentified association. But the importance of this diagnosis lies not on the etiological cause, rather on the treatment plan. Such co-existence can mandate a modification of treatment and careful reconsideration of the potential surgical risks. The auditory result should also be explained well to the candidate and the family.
Twelve-year-old female with neglected congenital profound hearing loss and malformed both pinnae.
Microtia and aural atresia.
Normal limits.
Incidental generalised increased bone density. Skeletal survey consistent with autosomal dominant osteopetrosis (ADOP).
Bone anchored hearing aid offered and cosmetic reconstruction of pinnae but refused.
Turning point in treatment strategy as undiagnosed ADOP precluded normal surgical management due to associated changes and potential risks.
Imaging diagnosed previously unsuspected skeletal dysplasia and teaches the lesson of proper patient selection.
The paper is well written.
P- Reviewer: Abulezz TA, Ciuman R, Mostafa BE, Nakashima T S- Editor: Qiu S L- Editor: A E- Editor: Wang CH
| 1. | Mastroiacovo P, Corchia C, Botto LD, Lanni R, Zampino G, Fusco D. Epidemiology and genetics of microtia-anotia: a registry based study on over one million births. J Med Genet. 1995;32:453-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 140] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 2. | Alasti F, Van Camp G. Genetics of microtia and associated syndromes. J Med Genet. 2009;46:361-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 3. | Sadler TW. Langman’s medical embryology. 7th ed. Baltimore: Williams and Wilkins 1995; . |
| 4. | Luquetti DV, Cox TC, Lopez-Camelo J, Dutra Mda G, Cunningham ML, Castilla EE. Preferential associated anomalies in 818 cases of microtia in South America. Am J Med Genet A. 2013;161A:1051-1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Stark Z, Savarirayan R. Osteopetrosis. Orphanet J Rare Dis. 2009;4:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 277] [Cited by in RCA: 308] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 6. | Bollerslev J, Andersen PE. Radiological, biochemical and hereditary evidence of two types of autosomal dominant osteopetrosis. Bone. 1988;9:7-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 109] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 7. | Bénichou OD, Laredo JD, de Vernejoul MC. Type II autosomal dominant osteopetrosis (Albers-Schönberg disease): clinical and radiological manifestations in 42 patients. Bone. 2000;26:87-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 146] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 8. | Waguespack SG, Hui SL, White KE, Buckwalter KA, Econs MJ. Measurement of tartrate-resistant acid phosphatase and the brain isoenzyme of creatine kinase accurately diagnoses type II autosomal dominant osteopetrosis but does not identify gene carriers. J Clin Endocrinol Metab. 2002;87:2212-2217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Gassner EM, Mallouhi A, Jaschke WR. Preoperative evaluation of external auditory canal atresia on high-resolution CT. AJR Am J Roentgenol. 2004;182:1305-1312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Yeakley JW, Jahrsdoerfer RA. CT evaluation of congenital aural atresia: what the radiologist and surgeon need to know. J Comput Assist Tomogr. 1996;20:724-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 59] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 11. | Patil AR, Bhalla A, Gupta P, Goyal D, Vishnubhatla S, Ramavat A, Sharma S. HRCT evaluation of microtia: A retrospective study. Indian J Radiol Imaging. 2012;22:188-194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Stocks RM, Wang WC, Thompson JW, Stocks MC, Horwitz EM. Malignant infantile osteopetrosis: otolaryngological complications and management. Arch Otolaryngol Head Neck Surg. 1998;124:689-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.0] [Reference Citation Analysis (0)] |