Revised: January 28, 2013
Accepted: February 5, 2013
Published online: February 20, 2013
Processing time: 195 Days and 7.5 Hours
Dental materials’ choice of patients has considerably changed. Whereas cast gold and amalgam have been the predominant biomaterials for decades, today tooth-colored materials like resin-based composites and ceramics are more and more successful. However, are we going to replace a good but biologically questionable material (amalgam) with an equal material (resin composite) being more esthetic but also biologically questionable For amalgam, long-term clinical studies reported some significant hints that in single cases amalgam may be a health hazard for patients, finally Norway banned amalgam completely. The main advantage of a resin-based composite over amalgam is its tooth-like appearance and more or less absence of extensive preparation rules. For many years it was believed that resin-based composites may cause pulpal injury. However, pulpal injury associated with the use of resin-based composites is not correlated with their cytotoxic properties. Nevertheless, resin-based composites and other dental materials require rigorous safety evaluation and continuous monitoring to prevent adverse events similar like with amalgam. Because of non-biocompatible pulp responses to resin-based composites and amalgam, they should not be placed in direct contact with the dental pulp. The less dentin remaining in the floor of preparations between resin-based composites or other dental materials is more likely to cause pulpitis. Percentage of patients and dental practitioners who display allergic reactions is between 0.7% and 2%. The release of cytotoxic monomers from resin-based materials is highest after polymerization and much lower after 1 wk. Substances released from resin-based composites have been shown to be toxic in cytotoxicity tests. Nevertheless, in vitro cytotoxicity assays have shown that amalgam has greater toxic effects than resin-based composites, sometime 100-700-fold higher. Altogether, the risk of side-effects is low, but not zero, especially for dental personnel.
- Citation: Frankenberger R, Garcia-Godoy F, Murray PE, Feilzer AJ, Krämer N. Risk aspects of dental restoratives: From amalgam to tooth-colored materials. World J Stomatol 2013; 2(1): 1-11
- URL: https://www.wjgnet.com/2218-6263/full/v2/i1/1.htm
- DOI: https://dx.doi.org/10.5321/wjs.v2.i1.1
The choice of dental materials has considerably changed during the last 20 years[1-3]. In former times, cast gold and amalgam have been the materials of choice for decades[4]. However, after amalgam was alleged to be inacceptably toxic and simultaneously esthetic demands of patients were growing, tooth-colored materials like resin-based composites and ceramics took more and more parts of this huge market[5].
In terms of biocompatibility and exposure, cast gold may be still the best restorative material, however, it is non-esthetic when it is used in visible areas such as premolars and also here some health concerns in terms of gold allergies are present[6]. Furthermore, the gold prize considerably increased from < $ 200 to > $ 1000 per ounce during the last decade which is consequently also transferred to restoration costs and therefore being detrimental for cost effectiveness as well. Other highly biocompatible materials like phosphate or glass ionomer cements are too brittle and therefore not able to withstand intraoral occlusal forces in deciduous and permanent teeth over time[1,2].
Today’s restorative trend clearly answers the question “black or white” by more and more moving from metallic amalgam to resin-based composites[5]. The same is true for bonded all-ceramic restorations such as ceramic inlays and onlays, because they have to be adhesively luted with the same adhesives and resin-based composite luting cements. So we face the interesting question whether we are replacing a clinically good but biologically questionable material (amalgam) with an equal material being more esthetic (resin composite) but also (or even more) biologically questionable.
Amalgam is one of the by far most successful dental restoratives which has been used all over the world since more than 150 years[7-10]. Long-term data are sufficient and long-term costs due to repair, refurbishment and tooth hard tissue loss during replacement are favourable[1-3,11-15]. Disadvantages are a compromised esthetic appearance due to an argentic to black color and especially biocompatibility concerns[7,16-24]. Dental silver amalgam consists of 50% mercury (in a complex mixture of copper, tin, silver, and zinc) and therefore this material was always suspected to be a considerable hazard for both patient and environment[25-35].
In the literature of the field, two opposing groups are identified: Primarily toxicologists are arguing against the health risks of mercury vapor being released from amalgam restorations and potentially threatening health of both patients and dentists and moreover polluting the environment by dental mixing and application processes[25-50]. On the other hand many authors with clinical dental background repeatedly state that amalgam per se is one of the most successful restorative materials[3,5,8,11,13,14,23,24,51-65]. So what is the real threat with amalgam It is common knowledge that high-dose exposure to elemental mercury vapor cause several diseases like emotional dysfunction[53]. However, it is not fully understood to the date whether smaller amounts like being released from amalgam restorations are a considerable health hazard as well[51-53,66,67].
In a retrospective cohort study involving 20 000 participants over 20 years (1977-1997) in the New Zealand defense force[28]. The cohort was linked with morbidity records by use of a time-varying exposure unit of 100 amalgam surface-years. Multiple sclerosis had an adjusted hazard ratio of 1.24, but there was no association with chronic fatigue syndrome (0.98), or kidney diseases[28]. Also Aminzadeh et al[25] reported some hints for a possible correlation of amalgam restorations and multiple sclerosis, however, also stating that more clinical studies are needed.
One of the most intensive clinical trials so far was the New England’s Children Amalgam Trial (NECAT) giving clinical result. 534 children (6-10 years old) with carious primary molars received either amalgam or resin composite restorations. Evaluated parameters were neuropsychological outcome (Full-Scale IQ score, General Memory Index, Visual-Motor Composite of the Wide Range Assessment of Visual Motor Abilities) and renal glomerular function with no statistical differences between resin composite and amalgam groups in any of the investigated criteria[51,52,68]. Furthermore, parent-completed child behavior checklists and children’s self-reports were collected. Children’s psychosocial status was evaluated in relation to three indices of mercury exposure: treatment assignment, surface-years of amalgam, and urinary mercury excretion. Again, there was no evidence that exposure to mercury from dental amalgam was associated with adverse psychosocial outcomes[53]. In another part of NECAT, longitudinal amalgam exposure data in children randomized to amalgam restorations were analyzed. Amalgam and U-Hg were moderately correlated with the total of amalgam surfaces having been a good predictor of current U-Hg and posterior occlusal surface-years for cumulative U-Hg. One additional amalgam surface caused a 9% increase in current U-Hg, and one posterior occlusal surface-year resulted in a 3% increase in cumulative U-Hg excretion[61]. Finally it could be shown that daily chewing gum use resulted in higher urinary Hg levels[69].
Halbach et al[56] measured internal exposure to amalgam-related mercury in plasma and erythrocytes after amalgam removal and estimated the amalgam-related absorbed dose in 82 patients. Post-removal steady-state Hg concentrations were taken for 18 mo for three groups: Removal of the fillings/removal and non-specific detoxification/health promotion program without removal. After amalgam removal, inorganic Hg was decreased, leveling at 27% of pre-removal levels after 60 d. Organic Hg in plasma did not change. Organic Hg in red cells of group A was lower in the early post-removal phase and higher in the late post-removal phase, being higher than the pre-removal control. A protracted increase in organic Hg was also found in red cells of group B after 60 d. In all groups, time profiles of urinary concentration and excretion of total-Hg were similar to those of inorganic-Hg levels in plasma. It was estimated that the amalgam-related inhalation and ingestion of Hg species were within the limits proposed by the World Health Organization (WHO), Agency for Toxic Substances and Desease Registry, and Environmental Protection Agency. The integrated daily Hg dose absorbed from amalgam was estimated < 3 μg for an average number of fillings and 7.4 μg for high amalgam load, with 30 μg being the tolerable dose according to the WHO[56].
On the other hand, there is no doubt that amalgam restorations release small amounts of mercury during clinical service which is absorbed by several body tissues in human subjects[32-35,64,70-75]. The daily dose is found to be 14% of the threshold above which observable adverse neurological symptoms are expected[75]. It has reported that methyl mercury and inorganic mercury levels in blood and cortex of autopsy bodies with a significant correlation between methyl mercury in blood and occipital cortex. Inorganic mercury in blood and occipital cortex, as well as total-Hg in pituitary and thyroid were strongly associated with the number of dental amalgam surfaces at the time of death[46,67]. Mutter et al[33] repeatedly stated that some of the clinical studies reporting low to no risk connected with dental amalgam may be methodically flawed which may lead to inadequate conclusions about the safety of dental amalgam. He also identified mercury vapor as potential reason for autism or Kawasaki’s disease[34,35,76]. It is also controversially discussed whether carbamide peroxide tooth bleaching agents lead to an increased release of mercury[77,78].
Another important point in the amalgam issue is occupational exposure for dentists and dental nurses[26,31,79-87]. It was found that a correlation between total Hg-U and duration of dental practice exists[87]. However, a cytogenetic damage in oral health professions dealing with amalgam was not reported[26,87]. Farahat et al[82] showed that dental staff have significant exposure to mercury vapor, furthermore indicating a negative impact of mercury on thymus gland functions[82,87]. Jones et al[84] investigated possible residual adverse effects from occupational mercury exposure in dentistry in 115 graduates of a dental nurse school from 1968-1971 because 30 years ago, dental nurses worked with amalgam without protective gloves or a ventilation system, resulting in chronic mercury exposure. Significant differences were found in current health experience and reproductive health, especially early hysterectomy experience. Reporting of Occupational Overuse Syndrome was strongly positively correlated with years of work.
Finally, also environmental aspects of mercury pollution by amalgam waste of dental practices and clinics have to be considered. Mercury occurs in nature as sulfides and in some minerals. All over the world every year 20 000-30 000 tons of mercury are discharged into the environment. Less than 50% of freshly triturated amalgam is inserted in cavities, more than 50% is waste. Extracted teeth with preexisting amalgams, amalgam-contaminated capsules and cotton rolls are discharged with the solid waste. However, dental mercury contamination makes only 3%-4% of global mercury being insignificant compared with industrial pollution[30,80,88,89]. With proper amalgam separators it could be even more reduced[30,80,88,89].
Despite all hints towards side-effects caused by mercury vapor of dental amalgam restorations, unproportionally many patients suffer amalgam incompatibility. Gottwald et al[55] conducted an interdisciplinary case-control study with special focus on toxicological, allergic, psychological and psychiatric aspects. Patients with amalgam-associated complaints (n = 40) were compared to amalgam bearers without complaints (n = 40) regarding quantity, surface area and quality of amalgam fillings, mercury load in blood and urine, allergy examination, and psychometric assessment with questionnaires noting coping strategies, interpersonal problems and self-consciousness. Patients and controls did not reveal different mercury concentrations in body fluids with patients having higher levels of psychic distress, higher incidence of depression and somatization disorders as well as different styles of coping with anxiety compared to controls. So the theory of amalgam-related complaints as an expression of underlying psychic problems was confirmed. A socio-economically important issue is that a ban of dental amalgam would also have some economic impact. Beazoglou et al[90] calculated the economic costs of an amalgam ban in the United States with total expenditures for restorations increasing from $ 46.2 billion to $ 49.7 billion and with consequently 15 444 021 fewer restorations inserted per year. An estimated first-year impact of an amalgam ban means an increase in expenditures of $ 8.2 billion.
Altogether it can be summarized that long-term clinical studies primarily demonstrated that amalgam can be safely used for patients, dental staff, and environment. However, there are some significant hints that in single cases amalgam may be a health hazard for patients. From 2008, Norway banned amalgam completely which is another hint[58]. So, amalgam remains an excellent restorative material with centuries of clinical success and decades of significant problems in biocompatibility.
A remarkable change in restorative dentistry has been the dramatic drop in the use of amalgam to restore teeth[91]. Patient and practitioner demand for a tooth-colored material as an alternative to amalgam was addressed in 1955 by Dr. Buonocore who described the use of a plastic material to restore teeth[92]. Later in 1950s, the first tooth-colored direct restorative material called Sevitron was produced by L.D. Caulk[93]. In the 1960s, several resin-based composite dental restorative materials (resin-based composites) were introduced[94]. The main advantage of a resin-based composite over amalgam is that it can be made in a wide range of tooth colors allowing the almost invisible restoration of teeth. However, the benefits of resin-based composites in comparison with amalgam and other dental materials have proved to be controversial. Normally resin-based composites can be used to restore teeth and repair or replace failing restorations with less removal of vital tooth structure in comparison with amalgam[95].
Unlike amalgam, resin-based composites must be bonded to teeth using an adhesive, which makes them more expensive and more technique-sensitive. Without meticulous placement, resin-based composite restorations can fail quickly. Nevertheless, even with the most meticulous placement, the longevity of resin-based composite restorations placed in posterior teeth has been shown to be significantly less than amalgam restorations[96]. The main reasons for the inferior clinical longevity of composite restorations in comparison with amalgam are marginal discoloration and a loss of adhesion[97].
Resin-based composites shrink by approximately 5% upon light-curing, which can create gaps for bacterial microleakage along the cavity margins[98]. These examples indicate that many of the problems patients have suffered with resin-based composites does not appear to be directly caused by the chemicals within the formulation of the material, but because of the shortcomings of the material when it is used to restore teeth. The short-comings of resin-based composites, particularly their polymerization shrinkage, are an active area of research and new lower shrinkage materials are under development to help improve their clinical performance similar to amalgam restorations.
The earliest resin-based composites had the worst longevity because they were prone to breakage and leakage due to their weak compressive strength[99]. The initial techniques to etch enamel to bond dental restorations were also not very successful, so many restorations suffered a loss of adhesion and were lost[100]. Many clinicians were initially reluctant to bond to dentin because they feared the high acid content of the etchant would cause a necrosis of underlying pulp tissue[101]. Subsequently, it was discovered that the buffering capacity of dentin, along with an improved quality of sealing to reduce microleakage, reduced the pulp irritation beneath resin-based composite restorations[102]. As research progressed, the concept of the “hybrid layer” was created to explain the physical and chemical interactions of the adhesive, resin-based composite, and tooth structure[103]. The “hybrid layer” concept has proved to be useful to develop research strategies to increase the quality of sealing and bonding of resin-based composites to tooth structure[104]. Improvements to the process of accomplishing resin-based composite bonding to tooth structure progressed through a number of “generations”. Each new generation of resin-based composite materials have had improved bonding and physical properties which are beneficial to patients through their increased longevity[105]. The current, 7th generation of resin-based composite adhesives can accomplish very high bond strengths to tooth structure[106]. The newest generations of “one-step” resin-based composite materials are generally easier for practitioners to use, and help reduce the exposure of patients to failed restorations.
For many years it was believed that the toxicity of the chemicals in the resin-based composite materials was responsible for pulpal injury. However, pulpal injury associated with the use of resin-based composites could not be correlated with their cytotoxic properties[107]. The discovery of the effect of bacterial contamination on the vitality of the tooth pulp, was a major milestone in dental research. In general, resin-based composites and other dental materials do not provide a hermetic seal with the tooth structure. Bacterial leakage may subsequently occur. The presence of bacteria and their toxic products can evoke an inflammatory response in the underlying pulp. Suh et al[108] demonstrated that the growth of bacteria in cavity restorations was directly correlated with pulpal inflammatory responses in the adjacent pulp tissue. As yet no permanent filling material has shown to consistently provide a perfect marginal seal, so leakage and bacterial contamination are always a threat to the integrity of the pulp. Therefore, the antibacterial properties of restorative materials are of considerable importance, and this explains the clinical success of some cytotoxic restorative materials, such as zinc oxide eugenol[109]. Despite these findings, it must be acknowledged that generally it is preferable to use dental materials which have the least potential to be toxic to patients and dental professionals. Similar to amalgam, resin-based composites and other dental materials require rigorous safety evaluation and continuous monitoring[110] to prevent adverse events.
Dentin and enamel have different physical properties and elemental compositions which have complicated the resin-based composite bonding to tooth structure[111]. It was discovered that the inclusion of hydrophobic monomers in adhesives could not penetrate the aqueous environment of demineralized dentin. Thus, methacrylate-based priming agents were used to create a permeable interface for the formation of a hybrid layer[112] which can increase micromechanical retention of the resin-based composite[113]. Thus, the need for “wet bonding” arose, and techniques for preparing the interface for increasingly hydrophobic monomers were developed[114]. Wet bonding systems have been successful[115]. However, they require the handling of multiple components which must be used in multiple steps. To facilitate the ease and speed with which bonding can be accomplished, the latest generation of “one-step adhesive systems” have been introduced which don’t have a separate acid etching step. Instead, acrylic resin monomers themselves provide the acidity needed for demineralization and simultaneously penetrate exposed and uplifted collagen fibrils[116]. A dental composite typically consists of a resin-based oligomer matrix, such as a bisphenol A-glycidyl methacrylate (Bis-GMA) or urethane dimethacrylate (UDMA), and an inorganic filler such as silicon dioxide silica. Compositions vary widely, with proprietary mixes of resins forming the matrix, as well as engineered filler glasses and glass ceramics. The filler gives the composite wear resistance and translucency. A coupling agent such as silane is used to enhance the bond between these two components. An initiator package (such as: Camphorquinone, Phenylpropanedion or Lucirin) begins the polymerization reaction of the resins. A catalyst is added in varying concentrations to control the speed of polymerization[117]. Resin-based composite materials are all capable of causing moderate to severe cytotoxicity when placed in contact with in vitro cell lines[118]. Resin-based composite materials may also cause severe pulp necrosis when used for direct-pulp capping[119]. The migration of adhesive and resin-based composite particles into pulp tissue can stimulate inflammatory responses[120]. Because of these non-biocompatible pulp responses to resin-based composites and amalgam, they should not be placed in direct contact with the dental pulp. A biocompatible liner such as Ca(OH)2 or preferably; mineral trioxide aggregate (MTA) must be used as a liner to help prevent unfavorable responses to direct pulp capping with resin-based composite[121] or amalgam. An MTA or Ca(OH)2 liner is not needed in shallow indirect pulp capping restorations because the buffering effect of dentin can prevent the diffusion of chemicals from resin-based composites and amalgam from entering the pulp tissue, particularly when the dentin thickness is above 0.5 mm[122]. The less dentin remaining in the floor of preparations between resin-based composites or other dental materials is more likely to cause pulpitis[122].
A number of local and systemic reactions to resin-based composite materials have been reported. The incidence of patients and dental practitioners who display allergic reactions is between 0.7% and 2%[123-126]. The main source of cellular and molecular cytotoxic injury from resin-based materials is claimed to be the leaching of unpolymerized monomers from the restoration during and after polymerization[127] which can reduce pulp vitality and cause a retraction of the gingival margin[128,129]. The release of cytotoxic monomers from resin-based materials is highest after polymerization and much lower after 1 wk[130]. Which may suggest the health risks to patients and practitioners are highest when in contact with newly polymerized resin-based composite materials, and the health risk diminishes over time.
Erosion and saliva degradation of resin-based composites may cause the release of leachable substances. Human-saliva derived esterases can biodegrade resin-based composites, causing the release of (Bis-GMA) monomers and (UDMA-type) comonomer[131]. The substances released from resin-based composites, particularly the (Bis-GMA) monomers have been shown to be toxic in cytotoxicity tests[132]. The presence of leached compounds is dependent on the formulation of resin-based composite[133]. The more flowable resin-based composites are more toxic than the traditional resin-based composites[134]. The relative in vitro cytotoxicity of resin-based composite monomers measured using a bromodeoxyuridine assay discovered that the Hg2+ amalgam component was four-fold more toxic than Bis-GMA to human gingival fibroblasts[135]. Almost all the in vitro cytotoxicity assays have shown that amalgam has greater toxic effects than resin-based composites, sometime 100-700-fold higher[136]. A problem is the general lack of resin-based composite biocompatibility data in comparison with amalgam. The results from systemic toxicity tests of resin-based composites do not indicate any unacceptable risk to the patient’s general health[137]. The in vitro screening of some components of resin-based composites are mutagenic[138]. Due to the limitations of the in vitro genotoxicity test systems and the comparatively high concentrations needed to elicit the reactions, no unacceptable risk can yet be derived from those data for the patient[139]. Most of the available data suggests that amalgam is relatively more hazardous to patients and dental professionals, than resin-based composites.
Skin and mucosa which come into contact with resin-based composites and bonding agents can become slightly inflamed which is commonly observed as a reddening of the affected area. However, if a patient or dental professional is allergic to a compound within the resin-based composite their reactions may be more severe and allergic irritant contact dermatitis can be observed. Contact urticaria, pigmentary changes, and photoallergic contact dermatitis may occasionally occur. Rarely other health effects, such as respiratory and neurologic signs and symptoms have been reported, but none have been linked to dental resin-based composites[140]. The concentrations are probably too minute to cause systemic reactions[137]. The most common resin-based composites to cause contact dermatitis, are (meth)acrylics, polyurethanes, phenol-formaldehydes, polyesters, amino resins (melamine-formaldehydes, urea-formaldehydes), polyvinyls, polystyrenes, polyolefins, polyamides and polycarbonates[140]. Contact dermatitis usually presents on the hands, fingers, and forearms, while facial, eyelid, and neck involvement may occur through indirect contact, e.g., via the hands, or from airborne exposure[140]. Patch testing with commercially available materials is important for a diagnosis of an allergy[141]. In some countries, occupational dermatoses are relatively common among dental staff, sometimes entailing occupational disability and re-schooling[142]. The risk of occupational dermatoses can be reduced by the development of new bonding techniques and careful risk-benefit assessments in the formulation of new dental composites. To protect patients from potential hazards of light-cured monomers released from resin-based composites it is important to use an effective curing unit and to applying the light-curing for the recommended length of time[142]. To protect dental professionals from the potential hazards of monomers released from resin-based composites, gloves should always been worn to prevent direct skin contact.
Dental restorative materials represent the most frequent replacement materials in the human body[143]. Despite that fact, biocompatibility issues regarding dental materials (especially amalgam) have not been scientifically evaluated until the early 1980s[141]. During the last two decades, however, amalgam lost its unique feature because adhesively bonded resin composites got suitable even for stress-bearing posterior restorations[144]. The paradigm shift towards minimally invasive restorations additionally supported this trend[145]. However, in many cases there is almost no patient or dental staff knowledge of hazards by the use of dental restoratives[146]. Furthermore it is of significant interest whether recently used dental materials changed the use-risk ratio.
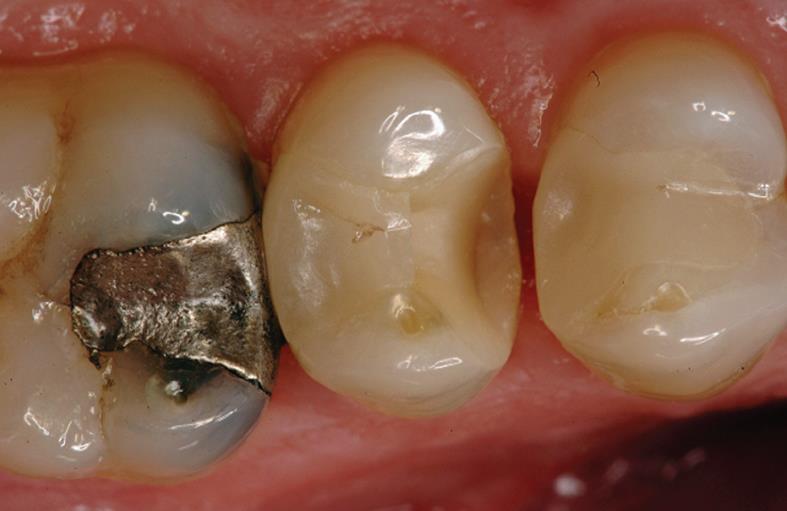
Fundamental judgement tool of dental materials is a risk analysis. Schmalz et al[145] defined the term “risk” concerning biocompatibility of dental restoratives as “the probability of a side effect and the severity of that side effect”. Risk analysis implies the description of indication ranges of a medical product, analysis of tissue exposure, and potential hazard[147]. So risk analyses try to determine the probability and severity of side effects for human health by exact knowledge of their composition. The consecutive risk assessment clarifies under estimation of usefulness and risk, whether a medical product may enter the market. Here it is decisive to compare the advantages of the material with the frequency and severity of side-effects[148]. In restorative dentistry, primarily a potential hazard by release of ingredients is discussed. Dental biomaterials are medical products with medium hazard potential. This means that clinical investigations are not mandatory in Europe, manufacturers just have to meet minimum requirements[147]. Especially in the post-amalgam era in the middle of the 1990s, some restoratives diminished from the market because minimum requirements were not achieved (Figure 1)[149].
Systematic epidemiological studies concerning frequency of side-effects with dental biomaterials are missing. Mjör[147] reported possible side-effects with different materials with 13 325 sessions (done by 137 dentists) and 24 cases of subjective discomfort, 7 cases of acute nature, and 15 cases of long-standing effects. In eight cases, amalgam was the reason for patients’ complaints[150]. So altogether the risk of side-effects is low, but not zero[148].
Dental personnel is much more under risk than patients. Geukens et al[148] observed 13 000 patients with contact dermatitis. In 31 patients (meth) acrylates were responsible for the complaints, and almost 50% of these group was working as dentist or dental nurse or dental technician[151]. Unfortunately, latex or vinyl gloves do not guarantee for safety due to their permeability at least after some minutes. This should be one of the reasons that dental personnel reveal increased rates of contact dermatitis of the fingers or allergic reactions following contact with monomers[152-154]. Thus, it is clearly recommended to completely avoid contact with unpolymerized resins[147].
Side-effects of dental materials are primarily of a local nature (e.g., gingivitis, mucosal alterations, pulpitis, etc.) or allergic (type I: immediate reaction or type IV: delayed reaction). Contact allergies have been observed for nickel sulfate, potassium dichromate, cobalt chloride, palladium chloride and gold sodium thiosulfate in patients with presence of metal allergy[155].
Other systemic effects (e.g., mutagenic, cancerogenic or teratogenic) are more of a theoretical nature[147]. Although there is a proven amount of substance release, this does not automatically mean an inacceptable health hazard[137]. Bacsik et al[156] could show estrogenic effects by bisphenol-A in mice, however, it is a matter of clinical relevance when substances are directly injected into the stomach of the animals instead of investigating true release from restorations. So also here clinical studies remain the ultimate instrument for risk assessment[157]. In the course of prospective clinical studies with dental biomaterials, the risk aspect plays a minor role. Main focus here are longevity aspects such as marginal integrity, restoration integrity, hypersensitivities, and recurrent caries. Moreover, patient numbers in dentistry e.g., for studies with posterior restorations are normally in the range of 30 patients[158] which may not be of sufficient power to describe side-effects. This is clearly reflected by evaluations of side-effects with local anesthesia. Despite 0.5 million local anesthetic injections which are administered in the United States daily, the actual risks of toxicity from these local anesthetic injections remain more or less unknown[159]. Therefore, prospective clinical studies mainly concentrate on local risks such as pulp reactions, compatibility with gingiva/periodontium, irritation of the oral mucosa, or biofilm accumulation[157].
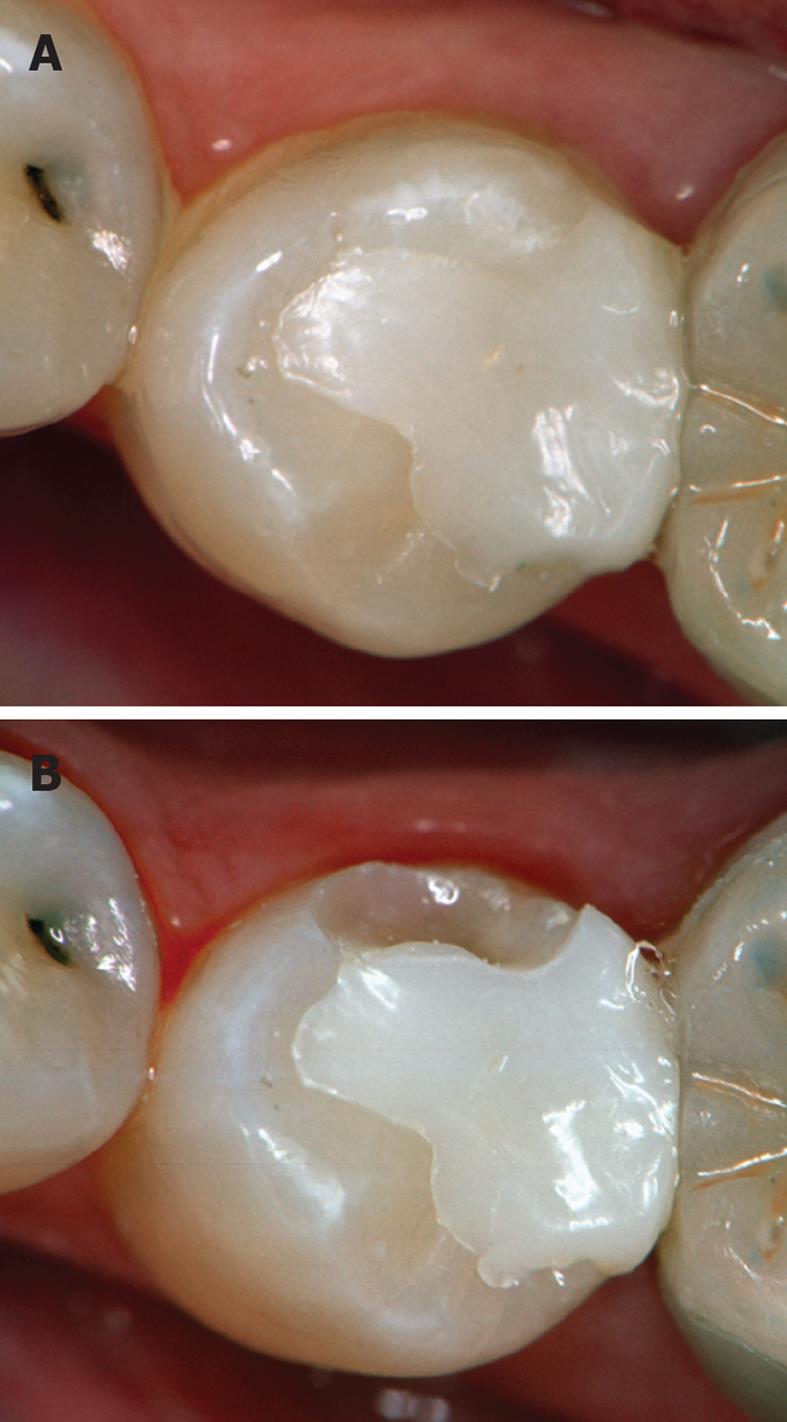
The benefit of dental biomaterials is still related to longevity. Kaplan-Meier survival curves and the associated nonparametric log rank test statistic are methods of choice for estimation of survival and therefore also failure risk[159]. This risk is appropriately reflected by annual failure rates[160,161]. Quality assessment of dental restorations is carried out according to modified USPHS critera with clinical examinations and analysis of replicas[162]. Main failure reasons are related to crucial criteria “marginal quality”, “restoration integrity”, “tooth integrity”, and “hypersensitivities” (Figure 2)[162]. Concerning clinical success, ADA criteria of 1996 are still valid. Failure rates < 10% after 4 years are defined as acceptable.
P-Reviewer Eugenia KK S- Editor Huang XZ L- Editor A E- Editor Zheng XM
| 1. | Hickel R, Kaaden C, Paschos E, Buerkle V, García-Godoy F, Manhart J. Longevity of occlusally-stressed restorations in posterior primary teeth. Am J Dent. 2005;18:198-211. [PubMed] |
| 2. | Manhart J, Chen H, Hamm G, Hickel R. Buonocore Memorial Lecture. Review of the clinical survival of direct and indirect restorations in posterior teeth of the permanent dentition. Oper Dent. 2004;29:481-508. [PubMed] |
| 3. | Mjör IA, Moorhead JE, Dahl JE. Selection of restorative materials in permanent teeth in general dental practice. Acta Odontol Scand. 1999;57:257-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 50] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 4. | Erpenstein H, Kerschbaum T, Halfin T. Long-term survival of cast-gold inlays in a specialized dental practice. Clin Oral Investig. 2001;5:162-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 5. | De Moor R, Delmé K. [Black or white--Which choice for the molars Part 2. Which does one choose for the restoration of posterior teeth: amalgam or composite]. Rev Belge Med Dent (1984). 2008;63:135-146. [PubMed] |
| 6. | Ahnlide I, Ahlgren C, Björkner B, Bruze M, Lundh T, Möller H, Nilner K, Schütz A. Gold concentration in blood in relation to the number of gold restorations and contact allergy to gold. Acta Odontol Scand. 2002;60:301-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 35] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 7. | Osborne JW. Safety of dental amalgam. J Esthet Restor Dent. 2004;16:377-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Smith D. Mercury pollution: fact or fiction. J Okla Dent Assoc. 2004;95:5. [PubMed] |
| 9. | Kostyniak PJ. Mercury and dentistry. Alpha Omegan. 2003;96:53-56. [PubMed] |
| 10. | Soler JI, Ellacuria J, Triana R, Guinea E, Osborne JW. A history of dental amalgam. J Hist Dent. 2002;50:109-116. [PubMed] |
| 11. | Fuks AB. Status of amalgams in pediatric dentistry: pros and cons. Alpha Omegan. 2005;98:26-32. [PubMed] |
| 12. | Maserejian NN, Tavares MA, Hayes C, Soncini JA, Trachtenberg FL. Prospective study of 5-year caries increment among children receiving comprehensive dental care in the New England children‘s amalgam trial. Community Dent Oral Epidemiol. 2009;37:9-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Sjögren P, Halling A. Survival time of Class II molar restorations in relation to patient and dental health insurance costs for treatment. Swed Dent J. 2002;26:59-66. [PubMed] |
| 14. | Sjögren P, Halling A. Long-term cost of direct Class II molar restorations. Swed Dent J. 2002;26:107-114. [PubMed] |
| 15. | Trachtenberg F, Maserejian NN, Tavares M, Soncini JA, Hayes C. Extent of tooth decay in the mouth and increased need for replacement of dental restorations: the New England Children’s Amalgam Trial. Pediatr Dent. 2008;30:388-392. [PubMed] |
| 16. | Nur Ozdabak H, Karaoğlanoğlu S, Akgül N, Polat F, Seven N. The effects of amalgam restorations on plasma mercury levels and total antioxidant activity. Arch Oral Biol. 2008;53:1101-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Puriene A, Janulyte V, Musteikyte M, Bendinskaite R. General health of dentists. Literature review. Stomatologija. 2007;9:10-20. [PubMed] |
| 19. | St John KR. Biocompatibility of dental materials. Dent Clin North Am. 2007;51:747-760, viii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Sweeney M, Creanor SL, Smith RA, Foye RH. The release of mercury from dental amalgam and potential neurotoxicological effects. J Dent. 2002;30:243-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 21. | Udoye C, Aguwa E. Amalgam safety and dentists’ attitude: a survey among a Subpopulation of Nigerian dentists. Oper Dent. 2008;33:467-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | van Zyl I. Mercury amalgam safety: a review. J Mich Dent Assoc. 1999;81:40-48, 50, 52. [PubMed] |
| 23. | Wahl MJ. Amalgam--Resurrection and redemption. Part 1: the clinical and legal mythology of anti-amalgam. Quintessence Int. 2001;32:525-535. [PubMed] |
| 24. | Yip HK, Li DK, Yau DC. Dental amalgam and human health. Int Dent J. 2003;53:464-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 25. | Aminzadeh KK, Etminan M. Dental amalgam and multiple sclerosis: a systematic review and meta-analysis. J Public Health Dent. 2007;67:64-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 26. | Atesagaoglu A, Omurlu H, Ozcagli E, Sardas S, Ertas N. Mercury exposure in dental practice. Oper Dent. 2006;31:666-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Balevi B. Are dental amalgams toxic to children Comment on 2 recently published randomized controlled trials. J Can Dent Assoc. 2007;73:51-54. [PubMed] |
| 28. | Bates MN, Fawcett J, Garrett N, Cutress T, Kjellstrom T. Health effects of dental amalgam exposure: a retrospective cohort study. Int J Epidemiol. 2004;33:894-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 29. | Bates MN. Mercury amalgam dental fillings: an epidemiologic assessment. Int J Hyg Environ Health. 2006;209:309-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 30. | Hiltz M. The environmental impact of dentistry. J Can Dent Assoc. 2007;73:59-62. [PubMed] |
| 31. | Hörsted-Bindslev P. Amalgam toxicity--environmental and occupational hazards. J Dent. 2004;32:359-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 32. | Mutter J, Naumann J. Mercury and the risk of myocardial infarction. N Engl J Med. 2003;348:2151-2154; author reply 2151-2154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 33. | Mutter J, Naumann J, Sadaghiani C, Walach H, Drasch G. Amalgam studies: disregarding basic principles of mercury toxicity. Int J Hyg Environ Health. 2004;207:391-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 34. | Mutter J, Naumann J, Schneider R, Walach H, Haley B. Mercury and autism: accelerating evidence. Neuro Endocrinol Lett. 2005;26:439-446. [PubMed] |
| 35. | Mutter J, Yeter D. Kawasaki’s disease, acrodynia, and mercury. Curr Med Chem. 2008;15:3000-3010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 36. | af Geijersstam E, Sandborgh-Englund G, Jonsson F, Ekstrand J. Mercury uptake and kinetics after ingestion of dental amalgam. J Dent Res. 2001;80:1793-1796. [PubMed] |
| 37. | Barregard L. Mercury from dental amalgam: looking beyond the average. Occup Environ Med. 2005;62:352-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 38. | Barregard L. Exposure to inorganic mercury: from dental amalgam to artisanal gold mining. Environ Res. 2008;107:4-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 39. | Barregard L, Trachtenberg F, McKinlay S. Renal effects of dental amalgam in children: the New England children’s amalgam trial. Environ Health Perspect. 2008;116:394-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 40. | Clarkson TW, Magos L. The toxicology of mercury and its chemical compounds. Crit Rev Toxicol. 2006;36:609-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1498] [Cited by in RCA: 1399] [Article Influence: 77.7] [Reference Citation Analysis (0)] |
| 41. | Clarkson TW, Vyas JB, Ballatori N. Mechanisms of mercury disposition in the body. Am J Ind Med. 2007;50:757-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 221] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 42. | Edlich RF, Greene JA, Cochran AA, Kelley AR, Gubler KD, Olson BM, Hudson MA, Woode DR, Long WB, McGregor W. Need for informed consent for dentists who use mercury amalgam restorative material as well as technical considerations in removal of dental amalgam restorations. J Environ Pathol Toxicol Oncol. 2007;26:305-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 43. | Edlich RF, Cross CL, Dahlstrom JJ, Long WB, Newkirk AT. Implementation of revolutionary legislation for informed consent for dental patients receiving amalgam restorations. J Environ Pathol Toxicol Oncol. 2008;27:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 44. | Edlich RF, Cochran AA, Cross CL, Wack CA, Long WB, Newkirk AT. Legislation and informed consent brochures for dental patients receiving amalgam restorations. Int J Toxicol. 2008;27:313-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 45. | Guzzi G, Minoia C, Pigatto PD, Severi G. Methylmercury, amalgams, and children’s health. Environ Health Perspect. 2006;114:A149; author reply A149-A150. [PubMed] [DOI] [Full Text] |
| 46. | Guzzi G, Grandi M, Cattaneo C, Calza S, Minoia C, Ronchi A, Gatti A, Severi G. Dental amalgam and mercury levels in autopsy tissues: food for thought. Am J Forensic Med Pathol. 2006;27:42-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 47. | Guzzi G, Pigatto PD. Occupational exposure to mercury from amalgams during pregnancy. Occup Environ Med. 2007;64:715-716; discussion 715-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 48. | Guzzi G, Minoia C. Biological detoxification and mercury dental amalgam. J Dent Res. 2008;87:800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 49. | Guzzi G, Fogazzi GB, Cantù M, Minoia C, Ronchi A, Pigatto PD, Severi G. Dental amalgam, mercury toxicity, and renal autoimmunity. J Environ Pathol Toxicol Oncol. 2008;27:147-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 50. | Guzzi G, Pigatto PD. Urinary mercury levels in children with amalgam fillings. Environ Health Perspect. 2008;116:A286-A287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 51. | Bellinger DC, Trachtenberg F, Daniel D, Zhang A, Tavares MA, McKinlay S. A dose-effect analysis of children’s exposure to dental amalgam and neuropsychological function: the New England Children’s Amalgam Trial. J Am Dent Assoc. 2007;138:1210-1216. [PubMed] |
| 52. | Bellinger DC, Daniel D, Trachtenberg F, Tavares M, McKinlay S. Dental amalgam restorations and children’s neuropsychological function: the New England Children’s Amalgam Trial. Environ Health Perspect. 2007;115:440-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 53. | Bellinger DC, Trachtenberg F, Zhang A, Tavares M, Daniel D, McKinlay S. Dental amalgam and psychosocial status: the New England Children’s Amalgam Trial. J Dent Res. 2008;87:470-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 54. | Giangrego E. Amalgam: has junk science caused dentists to pull it. CDS Rev. 2006;99:10-13. [PubMed] |
| 55. | Gottwald B, Kupfer J, Traenckner I, Ganss C, Gieler U. Psychological, allergic, and toxicological aspects of patients with amalgam-related complaints. Psychother Psychosom. 2002;71:223-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 56. | Halbach S, Vogt S, Köhler W, Felgenhauer N, Welzl G, Kremers L, Zilker T, Melchart D. Blood and urine mercury levels in adult amalgam patients of a randomized controlled trial: interaction of Hg species in erythrocytes. Environ Res. 2008;107:69-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 57. | Hyson JM. Amalgam: Its history and perils. J Calif Dent Assoc. 2006;34:215-229. [PubMed] |
| 58. | Jones DW. A Scandinavian tragedy. Br Dent J. 2008;204:233-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 59. | Jones DW. Has dental amalgam been torpedoed and sunk. J Dent Res. 2008;87:101-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 60. | Martin MD, Woods JS. The safety of dental amalgam in children. Expert Opin Drug Saf. 2006;5:773-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 61. | Maserejian NN, Trachtenberg FL, Assmann SF, Barregard L. Dental amalgam exposure and urinary mercury levels in children: the New England Children’s Amalgam Trial. Environ Health Perspect. 2008;116:256-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 62. | Maserejian NN, Tavares MA, Hayes C, Soncini JA, Trachtenberg FL. Rural and urban disparities in caries prevalence in children with unmet dental needs: the New England Children‘s Amalgam Trial. J Public Health Dent. 2008;68:7-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 63. | Mitchell RJ, Koike M, Okabe T. Posterior amalgam restorations--usage, regulation, and longevity. Dent Clin North Am. 2007;51:573-589, v. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 64. | Needleman HL. Mercury in dental amalgam--a neurotoxic risk. JAMA. 2006;295:1835-1836. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 65. | Wahl MJ. Amalgam--resurrection and redemption. Part 2: The medical mythology of anti-amalgam. Quintessence Int. 2001;32:696-710. [PubMed] |
| 66. | Berglund A. Release of mercury vapor from dental amalgam. Swed Dent J Suppl. 1992;85:1-52. [PubMed] |
| 67. | Björkman L, Lundekvam BF, Laegreid T, Bertelsen BI, Morild I, Lilleng P, Lind B, Palm B, Vahter M. Mercury in human brain, blood, muscle and toenails in relation to exposure: an autopsy study. Environ Health. 2007;6:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 132] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 68. | Bellinger DC, Trachtenberg F, Barregard L, Tavares M, Cernichiari E, Daniel D, McKinlay S. Neuropsychological and renal effects of dental amalgam in children: a randomized clinical trial. JAMA. 2006;295:1775-1783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 147] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 69. | Dunn JE, Trachtenberg FL, Barregard L, Bellinger D, McKinlay S. Scalp hair and urine mercury content of children in the Northeast United States: the New England Children’s Amalgam Trial. Environ Res. 2008;107:79-88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 70. | Larose P, Basciano M. Dental mercury and Norway. J Dent Res. 2008;87:413; author reply 413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 71. | Leistevuo J, Leistevuo T, Helenius H, Pyy L, Osterblad M, Huovinen P, Tenovuo J. Dental amalgam fillings and the amount of organic mercury in human saliva. Caries Res. 2001;35:163-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 72. | Lindbohm ML, Ylöstalo P, Sallmén M, Henriks-Eckerman ML, Nurminen T, Forss H, Taskinen H. Occupational exposure in dentistry and miscarriage. Occup Environ Med. 2007;64:127-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 73. | Luglie PF, Campus G, Chessa G, Spano G, Capobianco G, Fadda GM, Dessole S. Effect of amalgam fillings on the mercury concentration in human amniotic fluid. Arch Gynecol Obstet. 2005;271:138-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 74. | Magos L, Clarkson TW. Overview of the clinical toxicity of mercury. Ann Clin Biochem. 2006;43:257-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 150] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 75. | Mitchell RJ, Osborne PB, Haubenreich JE. Dental amalgam restorations: daily mercury dose and biocompatibility. J Long Term Eff Med Implants. 2005;15:709-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 76. | Geier DA, Kern JK, Geier MR. A prospective study of prenatal mercury exposure from maternal dental amalgams and autism severity. Acta Neurobiol Exp (Wars). 2009;69:189-197. [PubMed] |
| 77. | Al-Salehi SK, Hatton PV, Miller CA, Mcleod C, Joiner A. The effect of carbamide peroxide treatment on metal ion release from dental amalgam. Dent Mater. 2006;22:948-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 78. | Rotstein I, Dogan H, Avron Y, Shemesh H, Steinberg D. Mercury release from dental amalgam after treatment with 10% carbamide peroxide in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;89:216-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 79. | Burk JW. The impact of mercury on the environment. J Calif Dent Assoc. 2004;32:885; discussion 885. [PubMed] |
| 80. | Chin G, Chong J, Kluczewska A, Lau A, Gorjy S, Tennant M. The environmental effects of dental amalgam. Aust Dent J. 2000;45:246-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 81. | Costa RD, Cossich ES, Tavares CR. Influence of the temperature, volume and type of solution in the mercury vaporization of dental amalgam residue. Sci Total Environ. 2008;407:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 82. | Farahat SA, Rashed LA, Zawilla NH, Farouk SM. Effect of occupational exposure to elemental mercury in the amalgam on thymulin hormone production among dental staff. Toxicol Ind Health. 2009;25:159-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 83. | Fasunloro A, Owotade FJ. Occupational hazards among clinical dental staff. J Contemp Dent Pract. 2004;5:134-152. [PubMed] |
| 84. | Jones L, Bunnell J, Stillman J. A 30-year follow-up of residual effects on New Zealand School Dental Nurses, from occupational mercury exposure. Hum Exp Toxicol. 2007;26:367-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 85. | Joshi A, Douglass CW, Kim HD, Joshipura KJ, Park MC, Rimm EB, Carino MJ, Garcia RI, Morris JS, Willett WC. The relationship between amalgam restorations and mercury levels in male dentists and nondental health professionals. J Public Health Dent. 2003;63:52-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 86. | Paksoy CS, Görgün S, Nalçaci R, Yagbasan A. Assessment of blood mercury levels in practicing Turkish clinicians, dental students, and dental nurses. Quintessence Int. 2008;39:e173-e178. [PubMed] |
| 87. | Trzcinka-Ochocka M, Gazewski A, Brodzka R. Exposure to mercury vapors in dental workers in Poland. Int J Occup Med Environ Health. 2007;20:147-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 88. | Kao RT, Dault S, Pichay T. Understanding the mercury reduction issue: the impact of mercury on the environment and human health. J Calif Dent Assoc. 2004;32:574-579. [PubMed] |
| 89. | Lubick N. Dental offices contribute to methylmercury burden. Environ Sci Technol. 2008;42:2712. [PubMed] [DOI] [Full Text] |
| 90. | Beazoglou T, Eklund S, Heffley D, Meiers J, Brown LJ, Bailit H. Economic impact of regulating the use of amalgam restorations. Public Health Rep. 2007;122:657-663. [PubMed] |
| 91. | Hickel R. Trends in materials science from the point of view of a practicing dentist. J Eur Ceram Soc. 2009;29:1283-1289. [DOI] [Full Text] |
| 92. | BUONOCORE MG. A simple method of increasing the adhesion of acrylic filling materials to enamel surfaces. J Dent Res. 1955;34:849-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1460] [Cited by in RCA: 1227] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 93. | Buonocore MG. Retrospections on bonding. Dent Clin North Am. 1981;25:241-255. [PubMed] |
| 94. | Suh BI. All-Bond--fourth generation dentin bonding system. J Esthet Dent. 1991;3:139-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 95. | Bernardo M, Luis H, Martin MD, Leroux BG, Rue T, Leitão J, DeRouen TA. Survival and reasons for failure of amalgam versus composite posterior restorations placed in a randomized clinical trial. J Am Dent Assoc. 2007;138:775-783. [PubMed] |
| 96. | Kugel G, Ferrari M. The science of bonding: from first to sixth generation. J Am Dent Assoc. 2000;131 Suppl:20S-25S. [PubMed] |
| 97. | Gwinnett AJ. Acid etching for composite resins. Dent Clin North Am. 1981;25:271-289. [PubMed] |
| 98. | Cox CF, Suzuki S. Re-evaluating pulp protection: calcium hydroxide liners vs. cohesive hybridization. J Am Dent Assoc. 1994;125:823-831. [PubMed] |
| 99. | Perdigão J, Lopes M. Dentin bonding--state of the art 1999. Compend Contin Educ Dent. 1999;20:1151-1158, 1160-1162; quiz 1164. [PubMed] |
| 100. | Nakabayashi N, Pashley DH. Hybrid layer formation. Hybridization of hard dental tissues. Tokyo: Quintessence 1998; 8-9. |
| 101. | Kanca J. Improving bond strength through acid etching of dentin and bonding to wet dentin surfaces. J Am Dent Assoc. 1992;123:35-43. [PubMed] |
| 102. | Perdigão J, Lopes M. Dentin bonding--questions for the new millennium. J Adhes Dent. 1999;1:191-209. [PubMed] |
| 103. | Kallenos TN, Al-Badawi E, White GE. An in vitro evaluation of microleakage in class I preparations using 5th, 6th and 7th generation composite bonding agents. J Clin Pediatr Dent. 2005;29:323-328. [PubMed] |
| 104. | Watts A, Paterson RC. Cellular responses in the dental pulp: a review. Int Endod J. 1981;14:10-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 105. | Brännström M, Vojinovic O, Nordenvall KJ. Bacteria and pulpal reactions under silicate cement restorations. J Prosthet Dent. 1979;41:290-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 106. | Wright KJ, Barbosa SV, Araki K, Spångberg LS. In vitro antimicrobial and cytotoxic effects of Kri 1 paste and zinc oxide-eugenol used in primary tooth pulpectomies. Pediatr Dent. 1994;16:102-106. [PubMed] |
| 107. | Food and Drug Administration, HHS . Dental devices: classification of dental amalgam, reclassification of dental mercury, designation of special controls for dental amalgam, mercury, and amalgam alloy. Final rule. Fed Regist. 2009;74:38685-38714. [PubMed] |
| 108. | Suh BI, Cincione FA. All-bond 2: The fourth generation bonding system. Esthet Dent Update. 1992;3:61-66. |
| 109. | Kenshima S, Francci C, Reis A, Loguercio AD, Filho LE. Conditioning effect on dentin, resin tags and hybrid layer of different acidity self-etch adhesives applied to thick and thin smear layer. J Dent. 2006;34:775-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 52] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 110. | Kwong SM, Cheung GS, Kei LH, Itthagarun A, Smales RJ, Tay FR, Pashley DH. Micro-tensile bond strengths to sclerotic dentin using a self-etching and a total-etching technique. Dent Mater. 2002;18:359-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 76] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 111. | White KC, Cox CF, Kanka J, Dixon DL, Farmer JB, Snuggs HM. Pulpal response to adhesive resin systems applied to acid-etched vital dentin: damp versus dry primer application. Quintessence Int. 1994;25:259-268. [PubMed] |
| 112. | Ferracane JL. Current trends in dental composites. Crit Rev Oral Biol Med. 1995;6:302-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 247] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 113. | Suh BI. A 4th generation universal bonding system. Asian J Aesthet Dent. 1994;2:19-25. [PubMed] |
| 114. | Griggs JA, Shen C, Anusavice KJ. Sensitivity of catalyst/base ratio on curing of resin luting agents: polymerization exotherm analysis. Dent Mater. 1994;10:314-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 115. | Geurtsen W, Lehmann F, Spahl W, Leyhausen G. Cytotoxicity of 35 dental resin composite monomers/additives in permanent 3T3 and three human primary fibroblast cultures. J Biomed Mater Res. 1998;41:474-480. [PubMed] |
| 116. | Pameijer CH, Stanley HR. The disastrous effects of the “total etch” technique in vital pulp capping in primates. Am J Dent. 1998;11 Spec No:S45-S54. [PubMed] |
| 117. | Kitasako Y, Shibata S, Tagami J. Migration and particle clearance from hard-setting Ca(OH)2 and self-etching adhesive resin following direct pulp capping. Am J Dent. 2006;19:370-375. [PubMed] |
| 118. | Bogen G, Kim JS, Bakland LK. Direct pulp capping with mineral trioxide aggregate: an observational study. J Am Dent Assoc. 2008;139:305-315; quiz 305-315. [PubMed] |
| 119. | Murray PE, Lumley PJ, Smith AJ. Preserving the vital pulp in operative dentistry: 3. Thickness of remaining cavity dentine as a key mediator of pulpal injury and repair responses. Dent Update. 2002;29:172-178. [PubMed] |
| 120. | Carmichael AJ, Gibson JJ, Walls AW. Allergic contact dermatitis to bisphenol-A-glycidyldimethacrylate (BIS-GMA) dental resin associated with sensitivity to epoxy resin. Br Dent J. 1997;183:297-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 121. | Hensten-Pettersen A. Skin and mucosal reactions associated with dental materials. Eur J Oral Sci. 1998;106:707-712. [PubMed] |
| 122. | Munksgaard EC, Hansen EK, Engen T, Holm U. Self-reported occupational dermatological reactions among Danish dentists. Eur J Oral Sci. 1996;104:396-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 39] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 123. | Ortengren U, Andreasson H, Karlsson S, Meding B, Barregård L. Prevalence of self-reported hand eczema and skin symptoms associated with dental materials among Swedish dentists. Eur J Oral Sci. 1999;107:496-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 124. | Goldberg M. In vitro and in vivo studies on the toxicity of dental resin components: a review. Clin Oral Investig. 2008;12:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 282] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 125. | Geurtsen- W, Leyhausen G. Chemical-Biological Interactions of the resin monomer triethyleneglycol-dimethacrylate (TEGDMA). J Dent Res. 2001;80:2046-2050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 115] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 126. | Peumans M, Van Meerbeek B, Lambrechts P, Vanherle G, Quirynen M. The influence of direct composite additions for the correction of tooth form and/or position on periodontal health. A retrospective study. J Periodontol. 1998;69:422-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 127. | Franz A, König F, Lucas T, Watts DC, Schedle A. Cytotoxic effects of dental bonding substances as a function of degree of conversion. Dent Mater. 2009;25:232-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 128. | Tabatabaee MH, Mahdavi H, Zandi S, Kharrazi MJ. HPLC analysis of eluted monomers from two composite resins cured with LED and halogen curing lights. J Biomed Mater Res B Appl Biomater. 2009;88:191-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 129. | Darmani H, Al-Hiyasat AS, Milhem MM. Cytotoxicity of dental composites and their leached components. Quintessence Int. 2007;38:789-795. [PubMed] |
| 130. | Müller H, Olsson S, Söderholm KJ. The effect of comonomer composition, silane heating, and filler type on aqueous TEGDMA leachability in model resin composites. Eur J Oral Sci. 1997;105:362-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 131. | Al-Hiyasat AS, Darmani H, Milhem MM. Cytotoxicity evaluation of dental resin composites and their flowable derivatives. Clin Oral Investig. 2005;9:21-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 82] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 132. | Reichl FX, Simon S, Esters M, Seiss M, Kehe K, Kleinsasser N, Hickel R. Cytotoxicity of dental composite (co)monomers and the amalgam component Hg(2+) in human gingival fibroblasts. Arch Toxicol. 2006;80:465-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 133. | Reichl FX, Walther UI, Durner J, Kehe K, Hickel R, Kunzelmann KH, Spahl W, Hume WR, Benschop H, Forth W. Cytotoxicity of dental composite components and mercury compounds in lung cells. Dent Mater. 2001;17:95-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 134. | Schmalz G. The biocompatibility of non-amalgam dental filling materials. Eur J Oral Sci. 1998;106:696-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 79] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 135. | Schweikl H, Hiller KA, Bolay C, Kreissl M, Kreismann W, Nusser A, Steinhauser S, Wieczorek J, Vasold R, Schmalz G. Cytotoxic and mutagenic effects of dental composite materials. Biomaterials. 2005;26:1713-1719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 50] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 136. | Schweikl H, Spagnuolo G, Schmalz G. Genetic and cellular toxicology of dental resin monomers. J Dent Res. 2006;85:870-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 386] [Cited by in RCA: 392] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 137. | Cao LY, Sood A, Taylor JS. Hand/face/neck localized pattern: sticky problems--resins. Dermatol Clin. 2009;27:227-249, v. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 138. | Schedle A, Ortengren U, Eidler N, Gabauer M, Hensten A. Do adverse effects of dental materials exist What are the consequences, and how can they be diagnosed and treated. Clin Oral Implants Res. 2007;18 Suppl 3:232-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 139. | Tang AT, Björkman L, Ekstrand J. New filling materials--an occupational health hazard. Ann R Australas Coll Dent Surg. 2000;15:102-105. [PubMed] |
| 140. | Knezevic A, Zeljezic D, Kopjar N, Tarle Z. Influence of curing mode intensities on cell culture cytotoxicity/genotoxicity. Am J Dent. 2009;22:43-48. [PubMed] |
| 141. | Tillberg A, Järvholm B, Berglund A. Risks with dental materials. Dent Mater. 2008;24:940-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 142. | Statement on posterior resin-based composites. ADA Council on Scientific Affairs; ADA Council on Dental Benefit Programs. J Am Dent Assoc. 1998;129:1627-1628. [PubMed] |
| 143. | Murdoch-Kinch CA, McLean ME. Minimally invasive dentistry. J Am Dent Assoc. 2003;134:87-95. [PubMed] |
| 144. | Schmalz G, Arenholt-Bindslev D. Biocompatibility of dental materials. Heidelberg: Springer 2009; . |
| 145. | Schmalz G, Geurtsen W. Unwanted biological side effects. Dental Biomaterials and their application. II: Clinical Aspects. Stuttgart: Thieme 2008; 2-31. |
| 146. | Krämer N, García-Godoy F, Frankenberger R. Evaluation of resin composite materials. Part II: in vivo investigations. Am J Dent. 2005;18:75-81. [PubMed] |
| 147. | Mjör IA. Biological side effects to materials used in dentistry. J R Coll Surg Edinb. 1999;44:146-149. [PubMed] |
| 148. | Geukens S, Goossens A. Occupational contact allergy to (meth)acrylates. Contact Dermatitis. 2001;44:153-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 74] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 149. | Afsahi SP, Sydiskis RJ, Davidson WM. Protection by latex or vinyl gloves against cytotoxicity of direct bonding adhesives. Am J Orthod Dentofacial Orthop. 1988;93:47-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 150. | Aalto-Korte K, Alanko K, Kuuliala O, Jolanki R. Methacrylate and acrylate allergy in dental personnel. Contact Dermatitis. 2007;57:324-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 86] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 151. | Kieć-Swierczyńska M, Krecisz B. Allergic contact dermatitis in a dental nurse induced by methacrylates. Int J Occup Med Environ Health. 2003;16:73-74. [PubMed] |
| 152. | Marcusson JA. Contact allergies to nickel sulfate, gold sodium thiosulfate and palladium chloride in patients claiming side-effects from dental alloy components. Contact Dermatitis. 1996;34:320-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 52] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 153. | Al-Hiyasat AS, Darmani H, Elbetieha AM. Leached components from dental composites and their effects on fertility of female mice. Eur J Oral Sci. 2004;112:267-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 72] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 154. | Geurtsen W. Biocompatibility of resin-modified filling materials. Crit Rev Oral Biol Med. 2000;11:333-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 269] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 155. | Posterior composite resins. Council on Dental Materials, Instruments, and Equipment. J Am Dent Assoc. 1986;112:707-709. [PubMed] |
| 156. | Bacsik CJ, Swift JQ, Hargreaves KM. Toxic systemic reactions of bupivacaine and etidocaine. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79:18-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 157. | Cole SR, Hernán MA. Adjusted survival curves with inverse probability weights. Comput Methods Programs Biomed. 2004;75:45-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 645] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 158. | Hickel R, Manhart J. Longevity of restorations in posterior teeth and reasons for failure. J Adhes Dent. 2001;3:45-64. [PubMed] |
| 159. | Ryge G, Snyder M. Evaluating the clinical quality of restorations. J Am Dent Assoc. 1973;87:369-377. [PubMed] |
| 160. | Hujoel PP. Design and analysis issues in split mouth clinical trials. Community Dent Oral Epidemiol. 1998;26:85-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 161. | Krämer N, Frankenberger R, Pelka M, Petschelt A. IPS Empress inlays and onlays after four years--a clinical study. J Dent. 1999;27:325-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 59] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 162. | Eichmiller FC, Marjenhoff WA. Posterior restorative materials research. J Calif Dent Assoc. 1996;24:73-76. [PubMed] |