Published online Aug 6, 2014. doi: 10.5315/wjh.v3.i3.115
Revised: February 8, 2014
Accepted: July 12, 2014
Published online: August 6, 2014
Processing time: 298 Days and 12.3 Hours
Cytogenetic abnormalities are the hallmark of leukemias. We report here two cases of unusual cytogenetic abnormalities associated with Philadelphia chromosome, one with mixed phenotypic acute leukemia showing monosomy 7 and t(9;22) (q34;q11.2) and the other with chronic myeloid leukemia and additional translocation involving chromosomes 10 and 13. Both patients achieved complete remission following imatinib based treatment.
Core tip: Cytogenetic abnormalities are the hallmark of leukemias. We report here two cases of unusual cytogenetic abnormalities associated with Philadelphia chromosome, one with mixed phenotypic acute leukemia showing monosomy 7 and t(9;22) (q34;q11.2) and the other with chronic myeloid leukemia and additional translocation involving chromosomes 10 and 13. Both patients achieved complete remission following imatinib based treatment.These two cases highlight the novel additional cytogenetic abnormalities associated with Ph chromosome.
- Citation: Sharma SK, Handoo A, Choudhary D, Gupta N. Unusual cytogenetic abnormalities associated with Philadelphia chromosome. World J Hematol 2014; 3(3): 115-117
- URL: https://www.wjgnet.com/2218-6204/full/v3/i3/115.htm
- DOI: https://dx.doi.org/10.5315/wjh.v3.i3.115
Cytogenetic abnormalities are the hallmark of leukemias. Translocation (9;22) is the characteristic feature of chronic myeloid leukemia and is also seen in variable number of patients with acute lymphoblastic leukemia. Monosomy 7 is seen in patients with myelodysplastic syndrome, acute myeloid leukemia and few cases of acute lymphoblastic leukemia. Mixed phenotypic acute leukemia (MPAL) is commonly associated with t(9;22) and sometimes with monosomy or deletion 7, but combined cytogenetic abnormality involving t(9;22) and monosomy 7 has rarely been reported in MPAL. We report here two cases, one of MPAL showing monosomy 7 and t(9;22) (q34;q11.2) and the other with chronic myeloid leukemia and additional translocation involving chromosomes 10 and 13. Both patients achieved complete remission following imatinib based treatment.
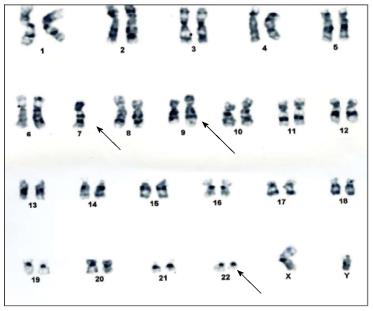
A 27 years old man was admitted with weakness and low grade fever for 20 d. On examination he was found to have pallor without lymphadenopathy or hepato-splenomegaly. Hemogram showed hemoglobin 5.8 g/dL, total leukocyte count 62 × 109/L with 60% blasts and platelet count 170 × 109/L. Flowcytometry revealed two distinct clusters of cells, one expressing CD34, HLD-DR, MPO, CD117, CD13 and CD33. The other cluster of blasts expressed cytoplasmic CD79a, CD19 and CD10. The blasts were negative for surface and cytoplasmic CD3, CD5, CD20 and CD16. The patient was diagnosed as a case of MPAL (Blymphoid/myeloid). Karyotyping revealed 45,XY, monosomy7, t(9;22)(q34;q11.2) [18]/46,XY[2] (Figure 1). RT-PCR for BCR-ABL detected genomic breakpoint at e13a2 corresponding to p210. Patient was treated with hyper-CVAD chemotherapy along with imatinib, which resulted in complete remission.
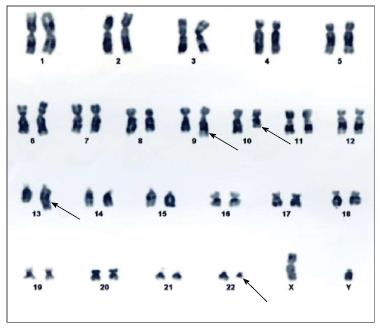
A 50 years old man presented with complaints of mild weakness for 3 wk. His hemoglobin was 10.5 g/dL, total leukocyte count 46.7 × 109/L and platelet count 3.6 × 109/L, differential count showed neutrophils 62%, myelocytes 10%, metamyelocytes 8%, lymphocytes 12%, monocytes 3%, eosinophils 2% and basophils 3%. Leukocyte alkaline phosphatase was low. Karyotyping revealed 46 XY, t(9;22)(q34;11.2), t(10;13)(q23;q34)[20] (Figure 2). Real-time polymerase chain reaction for BCR-ABL was positive for p210. He was diagnosed as chronic myeloid leukemia-chronic phase and treated with imatinib. He achieved complete hematological remission in 2 mo.
Philadelphia (Ph) chromosome results from reciprocal translocation of chromosome 9 and 22. This translocation leads to the generation of a chimeric gene that results from the fusion of the ABL gene on chromosome 9 with the BCR gene on chromosome 22. MPAL is a rare leukemia arising from a hematopoietic pluripotent stem cell with a frequency of 0.5%-1%[1,2]. In a study by Matutes et al[2], comprising 100 patients of MPAL, cytogenetics revealed t(9;22) in 20%, 11q23/MLL-rearrangements in 8%, complex in 32%, aberrant in 27% and normal in 13% karyotypes. Three cases of CML[3,4] and one case of T-cell/myeloid MPAL[5] associated with del 7, t(9;22)(q34;q11) have been reported. Deletion/monosomy 7 is associated with poor prognosis in AML and t(9;22) confers a bad prognosis in ALL. Additional chromosomal abnormalities in CML may appear in about 5% of cases[6-8]. In a study by Luatti et al[9], 21 patients (5.6%) had additional chromosomal abnormalities; the overall cytogenetic and molecular response rates in these patients were significantly lower. None of these patients had translocations involving chromosome 10 and 13. Our patient with MPAL demonstrated characteristic bilineage leukemia and showed complete remission following hyper-CVAD plus imatinib therapy. The patient with CML also responded to imatinib. These two cases highlight the novel additional cytogenetic abnormalities associated with Ph chromosome. Whether this association of Philadelphia chromosome with these additional cytogenetic abnormalities adversely affect the prognosis needs to be evaluated. Though some studies have shown poor outcome with additional chromosomal abnormalities[9], our patients showed good initial response to imatinib based therapy.
A 27 years old man was admitted with weakness and low grade fever for 20 d. A 50 years old man presented with complaints of mild weakness for 3 wk.
Case 1: Hemogram showed hemoglobin 5.8 g/dL, total leukocyte count 62 × 109/L with 60% blasts and platelet count 170 × 109/L. Case 2: His hemoglobin was 10.5 g/dL, total leukocyte count 46.7 × 109/L and platelet count 3.6 × 109/L, differential count showed neutrophils 62%, myelocytes 10%, metamyelocytes 8%, lymphocytes 12%, monocytes 3%, eosinophils 2% and basophils 3%.
Real-time polymerase chain reaction for BCR-ABL was positive for p210.
Patient was treated with hyper-CVAD chemotherapy along with imatinib, which resulted in complete remission.
The manuscript demonstrates good initial response to imatinib based therapy of two rare cases of leukemia patients one with mixed phenotypic acute leukemia bilineage (B-lymphoid/myeloid) and the second with CML bearing an additional to t(9;22) chromosomal abnormality.
P- Reviewer: Demonacos C, Huang SA S- Editor: Gou SX L- Editor: A E- Editor: Lu YJ
| 1. | Borowitz M, Bene MC, Harris NL, Porwit A, Matutes E. Acute leukemias of ambiguous lineage. World Health Organization classification of tumors. Pathology and genetics of tumors of hematopoietic and lymphoid tissues. Lyon, France: IARC Press 2008; 150-155. |
| 2. | Matutes E, Pickl WF, Van’t Veer M, Morilla R, Swansbury J, Strobl H, Attarbaschi A, Hopfinger G, Ashley S, Bene MC. Mixed-phenotype acute leukemia: clinical and laboratory features and outcome in 100 patients defined according to the WHO 2008 classification. Blood. 2011;117:3163-3171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 257] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 3. | Fischer K, Fröhling S, Scherer SW, McAllister Brown J, Scholl C, Stilgenbauer S, Tsui LC, Lichter P, Döhner H. Molecular cytogenetic delineation of deletions and translocations involving chromosome band 7q22 in myeloid leukemias. Blood. 1997;89:2036-2041. [PubMed] |
| 4. | Christodoulidou F, Silver RT, Macera MJ, Verma RS. Disappearance of a highly unusual clone, 46,XY,del(7)(p12),t(9; 22)(q34; q11) in chronic myeloid leukemia after treatment with recombinant interferon and cytosine arabinoside. Cancer Genet Cytogenet. 1992;64:174-177. [PubMed] |
| 5. | Monma F, Nishii K, Ezuki S, Miyazaki T, Yamamori S, Usui E, Sugimoto Y, Lorenzo V F, Katayama N, Shiku H. Molecular and phenotypic analysis of Philadelphia chromosome-positive bilineage leukemia: possibility of a lineage switch from T-lymphoid leukemic progenitor to myeloid cells. Cancer Genet Cytogenet. 2006;164:118-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 6. | Marktel S, Marin D, Foot N, Szydlo R, Bua M, Karadimitris A, De Melo VA, Kotzampaltiris P, Dazzi F, Rahemtulla A. Chronic myeloid leukemia in chronic phase responding to imatinib: the occurrence of additional cytogenetic abnormalities predicts disease progression. Haematologica. 2003;88:260-267. [PubMed] |
| 7. | Sokal JE, Gomez GA, Baccarani M, Tura S, Clarkson BD, Cervantes F, Rozman C, Carbonell F, Anger B, Heimpel H. Prognostic significance of additional cytogenetic abnormalities at diagnosis of Philadelphia chromosome-positive chronic granulocytic leukemia. Blood. 1988;72:294-298. [PubMed] |
| 8. | Kantarjian HM, Smith TL, McCredie KB, Keating MJ, Walters RS, Talpaz M, Hester JP, Bligham G, Gehan E, Freireich EJ. Chronic myelogenous leukemia: a multivariate analysis of the associations of patient characteristics and therapy with survival. Blood. 1985;66:1326-1335. [PubMed] |
| 9. | Luatti S, Castagnetti F, Marzocchi G, Baldazzi C, Gugliotta G, Iacobucci I, Specchia G, Zanatta L, Rege-Cambrin G, Mancini M. Additional chromosomal abnormalities in Philadelphia-positive clone: adverse prognostic influence on frontline imatinib therapy: a GIMEMA Working Party on CML analysis. Blood. 2012;120:761-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 89] [Article Influence: 6.8] [Reference Citation Analysis (0)] |