Published online Jun 18, 2016. doi: 10.5312/wjo.v7.i6.361
Peer-review started: February 14, 2016
First decision: March 21, 2016
Revised: April 6, 2016
Accepted: April 21, 2016
Article in press: April 22, 2016
Published online: June 18, 2016
Processing time: 131 Days and 1.9 Hours
Infections can hinder orthopedic implant function and retention. Current implant-based antimicrobial strategies largely utilize coating-based approaches in order to reduce biofilm formation and bacterial adhesion. Several emerging antimicrobial technologies that integrate a multidisciplinary combination of drug delivery systems, material science, immunology, and polymer chemistry are in development and early clinical use. This review outlines orthopedic implant antimicrobial technology, its current applications and supporting evidence, and clinically promising future directions.
Core tip: Infections can hinder orthopedic implant function and retention. Current implant-based antimicrobial strategies largely utilize coating-based approaches in order to reduce biofilm formation and bacterial adhesion. Several emerging antimicrobial technologies that integrate a multidisciplinary combination of drug delivery systems, material science, immunology, and polymer chemistry are in development and early clinical use. This review outlines the latest orthopedic implant antimicrobial technologies-including updates on chitosan coatings, photoactive-based coatings, electrospinning technology, integrated biofilms-highlighting the current applications, supporting evidence, and clinically-promising future directions.
- Citation: Eltorai AE, Haglin J, Perera S, Brea BA, Ruttiman R, Garcia DR, Born CT, Daniels AH. Antimicrobial technology in orthopedic and spinal implants. World J Orthop 2016; 7(6): 361-369
- URL: https://www.wjgnet.com/2218-5836/full/v7/i6/361.htm
- DOI: https://dx.doi.org/10.5312/wjo.v7.i6.361
Orthopedic implants are commonly used in spine surgery, arthroplasty, arthrodesis, as well for applications in treating fractures and nonunions[1]. Typically formulated from titanium, stainless steel, cobalt-chromium, or polyethylene polymers, orthopedic implants can serve as niduses for infection and may hinder infection clearance due to biofilm formation on the implant surface[2]. Orthopedic implant-associated infections are challenging complications which can lead to delayed healing, implant loosening, implant removal, amputation, or even death[3].
In many infections, bacteria will form a biofilm on the implant, increasing their resistance to antibiotics and resulting in infection persistence despite aggressive surgical debridement and prolonged antibiotic treatments[4,5]. A biofilm is an aggregated mass of bacteria that can form on the surface of an orthopedic implant, providing the ideal environment for bacteria to flourish. Such bacterial growths are difficult to eliminate and present a serious challenge in implant development[6,7]. In the United States, orthopedic implants are associated with an approximate 5% infection rate, representing 100000 infections per year[8]. This frequency represents a notable economic burden on both patients and health care providers. Although exact figures are elusive, even with the existence of antibiotic prophylactic it is estimated that implant infections increase the overall cost of hospitalization up to 45% on average[9,10].
Current antimicrobial strategies have largely focused on coating-based approaches-each of which aims to prevent infection by mitigating biofilm formation[11]. Key coatings include antibiotic, antiseptic, nano-silver, and photoactive-based coatings[11].
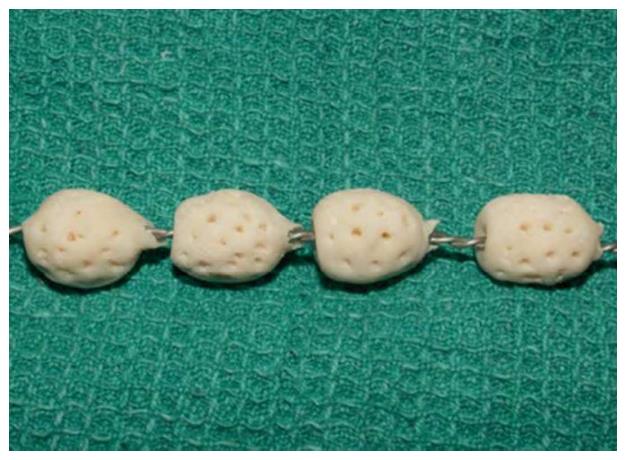
Antibiotic coatings allow for local delivery of antibiotics with a sustained release based on the drug carrier pharmacokinetics[12]. While various antibiotics have been studied (e.g., amoxicillin, vancomycin, cephalothin, and tobramycin), the most widely studied antibiotic for such coatings has been gentamicin[11]. Common biocompatible drug carriers for the coatings include polymethylmethacrylate (PMMA), poly(lactic-co-glycolic acid) (PLGA), poly(lactic acid), polyethylene glycol, and poly(D,L)lactide (Figure 1)[7]. Hydroxyapatite (HA) was recently shown to be an effective drug carrier of gentamicin[13,14].
Neut et al[15] demonstrated the wide-spectrum antibacterial efficacy of a gentamicin coating in vitro through investigating infection prophylaxis of Staphylococcus aureus (S. aureus) in cementless total-hip arthroplasty. In a rabbit model, Alt et al[16] found that the gentamicin-HA composite provided a statistically significant reduction in infection rate when compared to uncoated total joint replacements. In patient trials, gentamicin-coated implants have displayed promising preliminary results (Figure 1)[17-20]. Limitations of antibiotic coatings include the use of fixed, predetermined antibiotics; limited duration of drug elution; and the risk of developing drug resistance[21].
To overcome the limited duration of drug elution, Ambrose et al[22-24] developed antibiotic-impregnated bioresorbable microspheres for sustained release of antibiotics over several weeks-which have been shown to reduce infection rates in animal models. Antiseptic-based coatings have emerged to address antibiotic coatings fixed bactericidal spectrum and possible drug resistance limitations. Antibiotic-based coatings are currently the most commonly utilized local antimicrobial clinical delivery method due to the well characterized nature of the antimicrobial agents. These coatings are limited by antibiotic classes, which are compatible with the chemistry of the coating matrix. Asides from pharmacokinetic limitations, antibiotic-based coatings represent the most accepted antimicrobial option available.
In contrast to antibiotic coatings, which are formulated to work against specific bacterial strains, antiseptic-based coatings are intended to combat a wide range of bacteria by way of more general chemical agents. For this reason antiseptic coatings are less likely to induce bacterial resistance compared to antibiotics[25,26]. Common antiseptics include chlorhexidine and chloroxylenol, which are thought to act through the interaction of their natural cationic nature with the anionic phosphate residue of the lipid molecules in bacterial cell membranes. This ionic adsorption damages cell membranes and limits bacterial adhesion (Figure 2)[27,28]. In 1998, Darouiche[8] first demonstrated the effectiveness of antiseptic coatings on titanium cylinders studied in vitro with human serum before DeJong et al[29] tested chlorhexidine and chloroxylenol in a goat model, finding that these two antiseptics reduced external fixator pin tract infections. Ho et al[30] demonstrated in vivo efficacy of antiseptic coatings in humans by reducing vascular and epidural catheter infection with application of a chlorhexidine-impregnated dressing. Due to their broad spectrum efficacy, antiseptic-based coatings are not without some level of generalized toxicity. Because of their general toxicity, antiseptic based coatings are more commonly utilized as topical dressings.
Chitosan is a polymer of chitin that exhibits active antimicrobial properties. Recent pre-clinical studies have provided evidence that several composites of chitosan may act as effective antimicrobial agents suited for titanium orthopedic implants. Yang et al[31] tested a vancomycin-chitosan composite by monitoring the proliferation of human osteoblast cells in vitro using methyl thiazole tetrazolium and cell adhesion using FEMSEM. They found that vancomycin-chitosan coated implants displayed lesser biofilm formation, a result corroborated by in vivo experiments in a rabbit model[31]. In fact, some results indicate that a simple mixtures of 2%-3% chitosan and 2% cinnamon oil may also hold antimicrobial properties against Staphylococcus epidermidis (S. epidermidis) on titanium implants[32]. Most recently, Qin et al[33] revealed preliminary in vitro results suggesting that chitosan-casein phosphopeptides coatings could provide antimicrobial benefits for cobalt matrix orthopedic implants. Other studies have suggested that chitosan alone may not be sufficiently potent as an antimicrobial agent and suffers from poor release kinetics. More current studies have focused on the synergistic use of chitosan and antibacterial agents with more promising results. As yet we are not aware of any clinical trials incorporating chitosan-based coats.
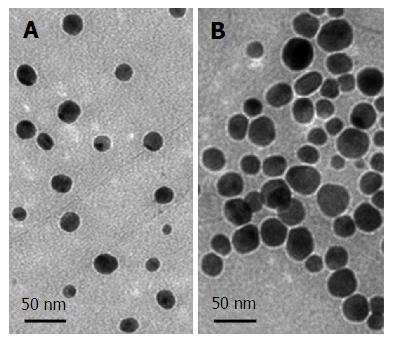
The antimicrobial properties of silver particles are well-established[34-38]. Silver particles have several known mechanisms of action including binding to thiol groups of enzymes, cell membranes, and nucleic acids, resulting in structural abnormalities, a damaged cell envelope, and inhibition of cell division[39-41]. Silver nanoparticles (Figure 3)[42] are typically incorporated into titanium surfaces or polymeric coating to control the release rate and duration of the bioactive silver[11,43-45]. Electrical currents are established when silver nanoparticles (cathode) embedded in a titanium matrix (anode) are exposed to electrolytes[45] - this galvanic coupling can cause changes in bacterial membrane morphology and DNA, leading to cell death[37]. Silver-based coatings have antimicrobial efficacy against a broad spectrum of pathogens, including Escherichia coli, S. aureus and S. epidermidis[46-48]. Using an in vivo model for osteomyelitis, Tran et al[48] inoculated S. aureus into fractured goat tibias and found after 5 wk silver-doped coated intramedullary nails led to better clinical and histology outcomes than the controls fixed with uncoated nails.
Early clinical studies have shown promising results with regard to reducing periprosthetic infections. Wafa et al[49] retrospectively compared 85 patients with silver-coated tumor prostheses to 85 tumor patients with non-silver tumor prostheses. The authors found that the average infection rate among silver-coated implant patients was 10.6% lower than that of their uncoated counterparts. In a similar prospective study by Hardes et al[50], silver-coated prosthetic tumor implants were shown to have an 11.7% lower infection rate over a five-year period than uncoated implants. Despite these encouraging clinical results, clinical use of silver-coated implants has been limited by concerns of mammalian bone cell cytotoxicity[51,52]. While this cytotoxic level is much lower than the anti-microbial threshold used for implant coatings, there is evidence to suggest that prolonged exposure to even low doses of nano-silver may result in mild toxicity in rats[53]. The long-term implications of such toxicity are yet undetermined. Because of its long history of usage, and relatively low toxicity, silver-based antimicrobial coatings represent a very promising tool against antibiotic-resistant pathogens. The effectiveness of the technology has been shown to be largely dependent on the ability of the coating matrix to provide efficacious release kinetics and formulation of silver nanoparticles or ions.
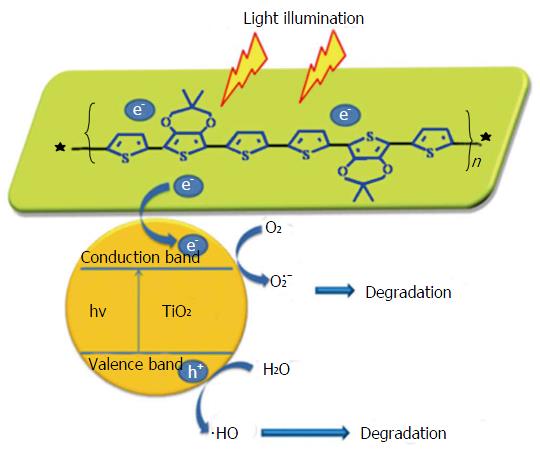
Photocatalyst coatings are composed of titanium alloys and display bactericidal effects via membrane degradation after activating exposure to ultraviolet irradiation (Figure 4)[54,55]. Titanium oxide (TiO2) is a commonly used photocatalytic agent due to its strong oxidizing power, lack of toxicity, and long-term chemical stability[56]. Villatte et al[56] demonstrated TiO2-based photoactive coatings were able to withstand mechanical stress from inserting stainless steel pins in cow femurs, had antibacterial effectiveness against S. aureus and S. epidermis cultures, and has the added benefit of low cost and easy scalability. Photocatalysts as antimicrobial agents in orthopedic implants remain to be tested in vivo.
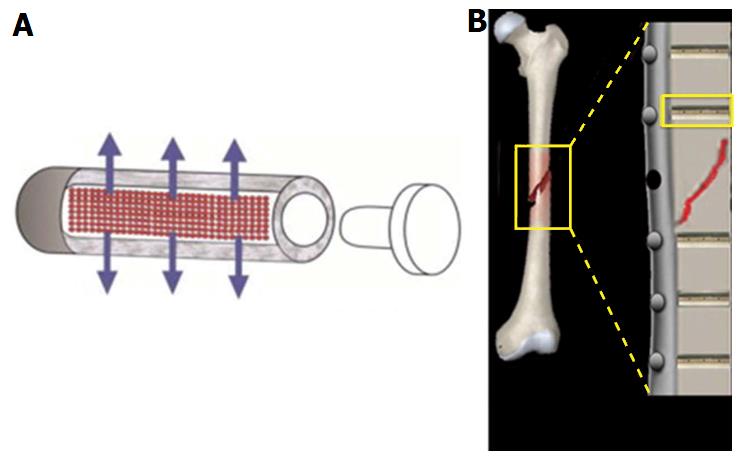
In addition to coatings, several other antimicrobial orthopedic implant technologies are being evaluated. Antibiotic-loaded bone cement (ALBC), such as PMMA, is widely used by orthopedic surgeons to help secure arthroplasty implants, to fill bone voids, and to treat vertebral compression fractures (Figure 5)[57,58]. ALBC has been in use since first being developed in 1970 as a potential method for in situ drug release[59]. Despite its widespread use, the antimicrobial efficacy of ALBC is debated[60,61]. Due to irregular release of antibiotic, only 5%-8% of the drug typically elutes properly[62]. Therefore, the high doses needed for a therapeutic effect have been shown to produce pathogen resistance[57].
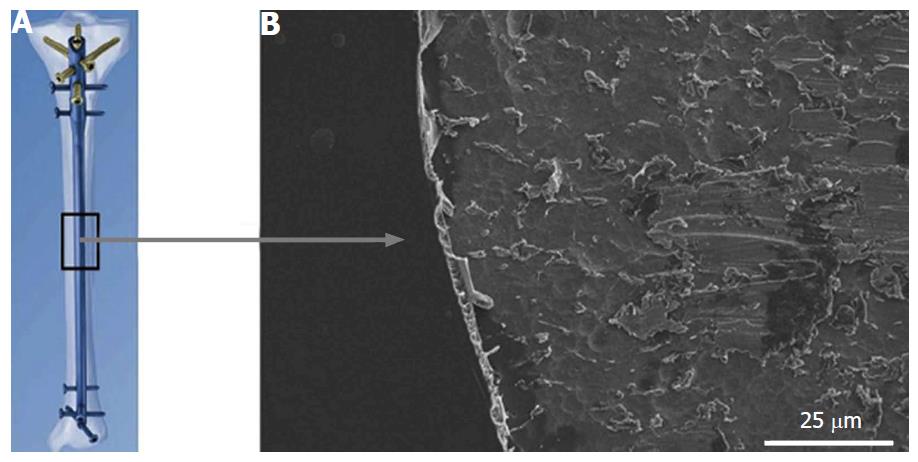
A novel system utilizes antibiotic-loaded reservoirs within the steel implant itself to enable a more controlled, localized release of drug when compared to coatings[63]. Initial in vivo testing by Gimeno et al[64] demonstrated that sheep infected with a biofilm-forming S. aureus strain showed no signs of infection of pre-placed tibia implants 7-9 d post introduction of S. aureus. Gimeno et al[65] subsequently proposed a design detailing fixation pins with tubular reservoirs for loading of antibiotics, allowing for more controlled release of the antibiotic based on number and size of release orifices (Figure 6).
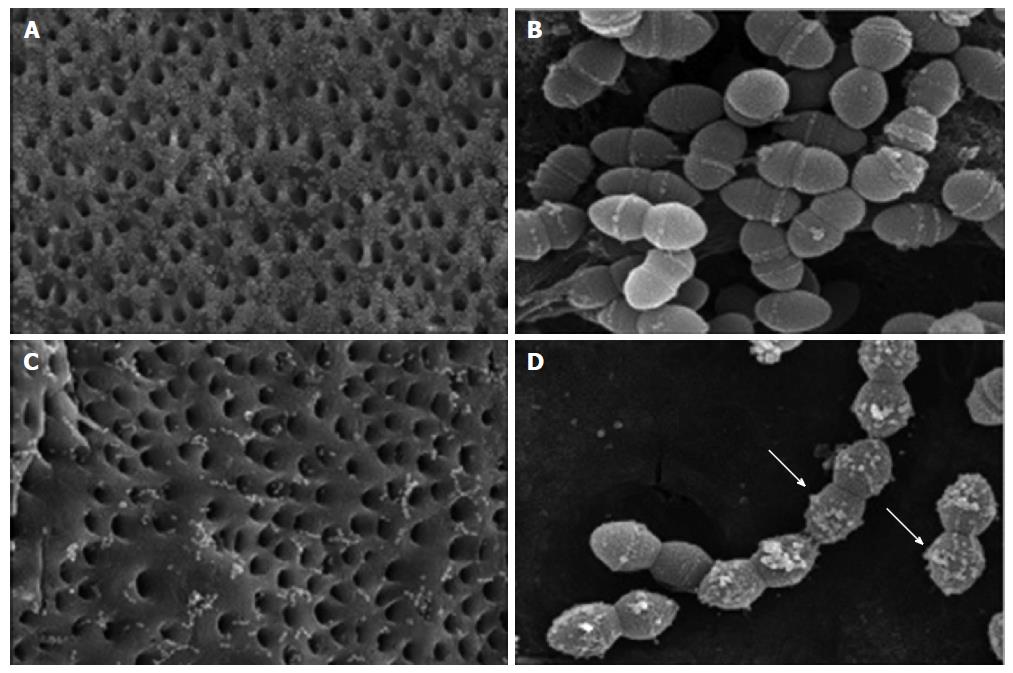
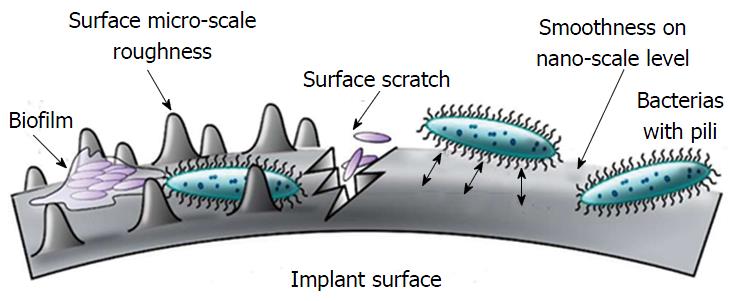
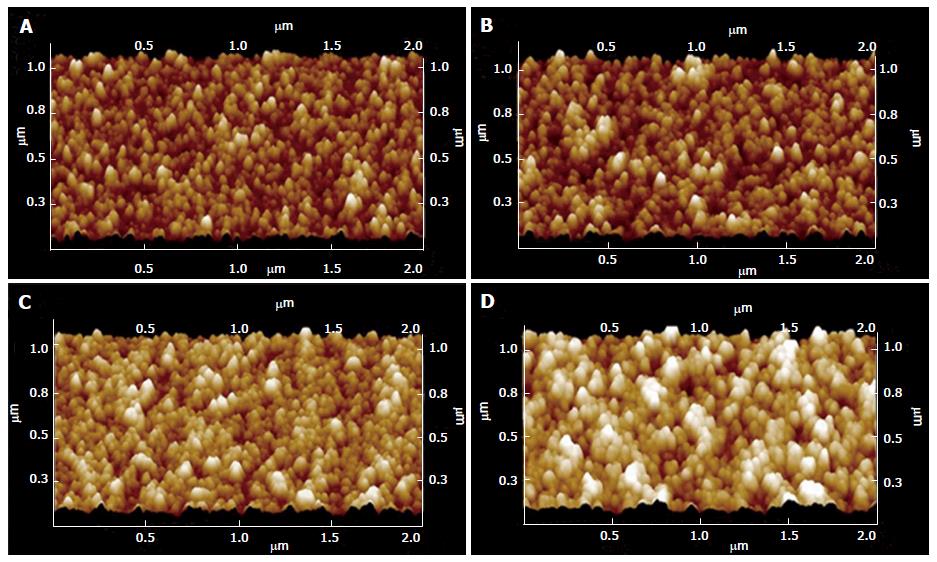
Modifying implant surface characteristics have are also been investigated as a means of reducing biofilm. For example, mixtures of polyethylene oxide and protein-repelling polyethylene glycol have shown significant bacterial inhibition when applied implant surfaces[66,67]. Singh et al[68] demonstrated that modifying surface roughness (Figures 7[69] and 8) of a material at the nanoscale level could provide antibacterial properties. Surface characteristic modification has been shown to interfere with osseointegration of the implants, challenging its clinical application[70]. Other studies have shown that certain pathogens are able to adhere, proliferate, and form biofilms more readily on rough surfaces. The data available suggests there is threshold where modified surface microtopography can be an effective means of reducing biofilm, or encouraging bacterial growth.
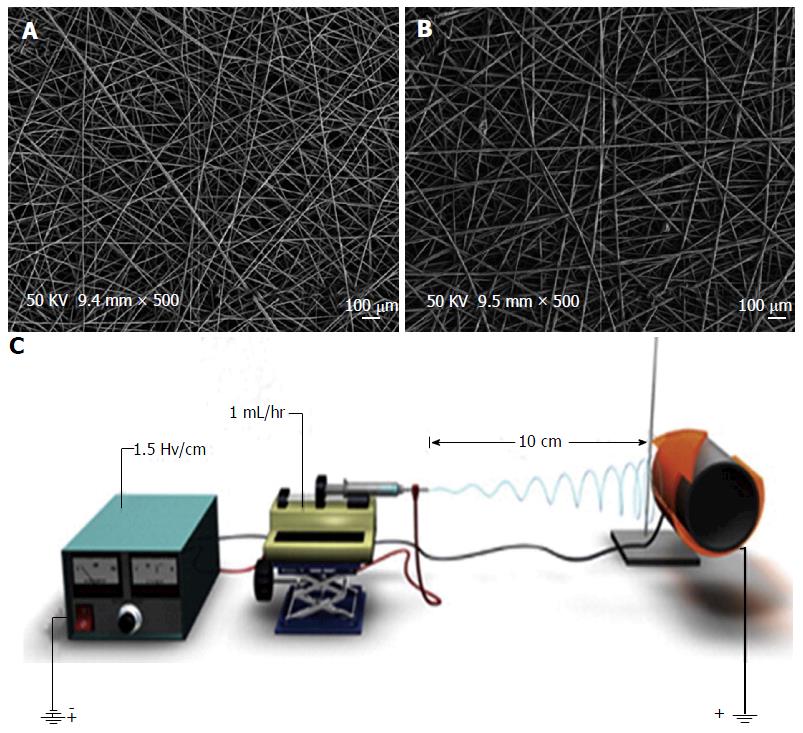
Electrospun matrices of PLGA nano-fibers have recently been proposed as a promising antimicrobial approach to orthopedic implant-associated infections[71]. In electrospinning, ultrafine fibers with nanometer diameters form a matrix with a very high surface-area-to-volume ratio[72]. Produced by syringe-pumping various drug and polymer solutions in the presence of a high electrical field potential[73], the resulting drug loaded, non-woven PLGA membranes are flexible, porous, and enable controlled drug release (Figure 9)[71,74]. Like coating, the matrices adhere directly to orthopedic implants.
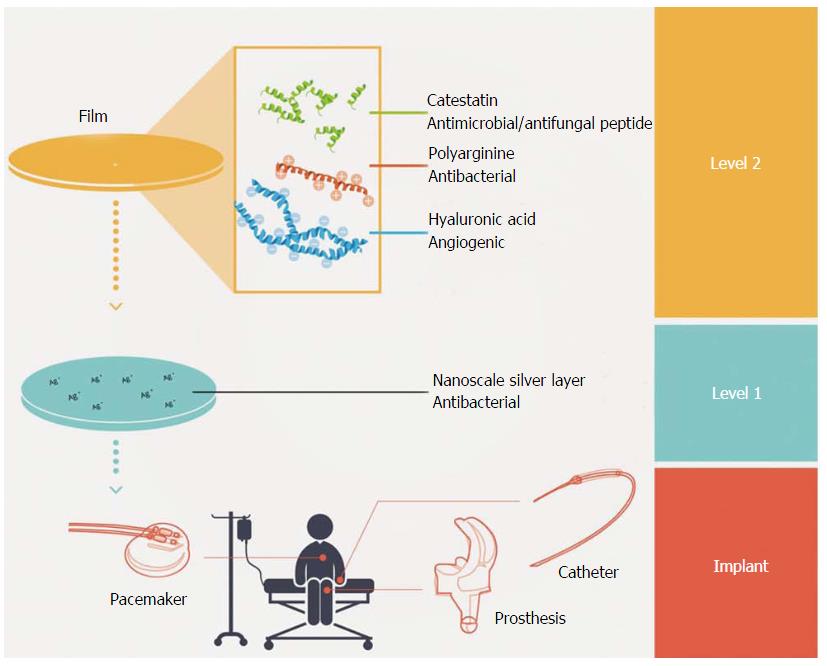
Özçelik et al[75] proposed a novel polyelectrolyte multilayer film approach using combined antimicrobial and immunomodulatory strategies (Figure 10). Composed of polyarginine and hyaluronic acid, the film inhibits the production of inflammatory cytokines, combats bacteria using a nanoscale silver coating, and opens the opportunity for bacteria-specific customization via embedded antimicrobial peptides. Although development of such films is far from clinical practice, microfilms are a promising look into the benefits of combining existing approaches for limiting implant-related complications to develop the composite technology of the future.
Several imperfect options exist for reducing the risk of orthopaedic implant infections. Despite technological advancement, orthopedic implant-associated infections remain as an important clinical problem, necessitating additional improvement. With promising technology on the horizon, it seems that the answer for reduced infection may not lie in solely one device or technology but in the synergy of many.
P- Reviewer: Kelesidis T, Rouabhia M S- Editor: Ji FF L- Editor: A E- Editor: Li D
| 1. | Goodman SB, Yao Z, Keeney M, Yang F. The future of biologic coatings for orthopaedic implants. Biomaterials. 2013;34:3174-3183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 568] [Cited by in RCA: 444] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 2. | Simon JP, Fabry G. An overview of implant materials. Acta Orthop Belg. 1991;57:1-5. [PubMed] |
| 3. | Moriarty TF, Schlegel U, Perren S, Richards RG. Infection in fracture fixation: can we influence infection rates through implant design? J Mater Sci Mater Med. 2010;21:1031-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Donlan RM. Biofilms: microbial life on surfaces. Emerg Infect Dis. 2002;8:881-890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3145] [Cited by in RCA: 2595] [Article Influence: 112.8] [Reference Citation Analysis (0)] |
| 5. | Stewart PS, Costerton JW. Antibiotic resistance of bacteria in biofilms. Lancet. 2001;358:135-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3133] [Cited by in RCA: 2947] [Article Influence: 122.8] [Reference Citation Analysis (0)] |
| 6. | Jefferson KK, Goldmann DA, Pier GB. Use of confocal microscopy to analyze the rate of vancomycin penetration through Staphylococcus aureus biofilms. Antimicrob Agents Chemother. 2005;49:2467-2473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 216] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 7. | Luo J, Chen Z, Sun Y. Controlling biofilm formation with an N-halamine-based polymeric additive. J Biomed Mater Res A. 2006;77:823-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 48] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Darouiche RO. Treatment of infections associated with surgical implants. N Engl J Med. 2004;350:1422-1429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1677] [Cited by in RCA: 1582] [Article Influence: 75.3] [Reference Citation Analysis (0)] |
| 9. | Kirkland KB, Briggs JP, Trivette SL, Wilkinson WE, Sexton DJ. The impact of surgical-site infections in the 1990s: attributable mortality, excess length of hospitalization, and extra costs. Infect Control Hosp Epidemiol. 1999;20:725-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1168] [Cited by in RCA: 1176] [Article Influence: 45.2] [Reference Citation Analysis (0)] |
| 10. | Bryan CS, Morgan SL, Caton RJ, Lunceford EM. Cefazolin versus cefamandole for prophylaxis during total joint arthroplasty. Clin Orthop Relat Res. 1988;117-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Veerachamy S, Yarlagadda T, Manivasagam G, Yarlagadda PK. Bacterial adherence and biofilm formation on medical implants: a review. Proc Inst Mech Eng H. 2014;228:1083-1099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 333] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 12. | Wu P, Grainger DW. Drug/device combinations for local drug therapies and infection prophylaxis. Biomaterials. 2006;27:2450-2467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 332] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 13. | Avés EP, Estévez GF, Sader MS, Sierra JC, Yurell JC, Bastos IN, Soares GD. Hydroxyapatite coating by sol-gel on Ti-6Al-4V alloy as drug carrier. J Mater Sci Mater Med. 2009;20:543-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 14. | Geesink RG, de Groot K, Klein CP. Bonding of bone to apatite-coated implants. J Bone Joint Surg Br. 1988;70:17-22. [PubMed] |
| 15. | Neut D, Dijkstra RJ, Thompson JI, van der Mei HC, Busscher HJ. A gentamicin-releasing coating for cementless hip prostheses-Longitudinal evaluation of efficacy using in vitro bio-optical imaging and its wide-spectrum antibacterial efficacy. J Biomed Mater Res A. 2012;100:3220-3226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | Alt V, Bitschnau A, Osterling J, Sewing A, Meyer C, Kraus R, Meissner SA, Wenisch S, Domann E, Schnettler R. The effects of combined gentamicin-hydroxyapatite coating for cementless joint prostheses on the reduction of infection rates in a rabbit infection prophylaxis model. Biomaterials. 2006;27:4627-4634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 114] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Schmidmaier G, Lucke M, Wildemann B, Haas NP, Raschke M. Prophylaxis and treatment of implant-related infections by antibiotic-coated implants: a review. Injury. 2006;37 Suppl 2:S105-S112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 205] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 18. | Fuchs T, Stange R, Schmidmaier G, Raschke MJ. The use of gentamicin-coated nails in the tibia: preliminary results of a prospective study. Arch Orthop Trauma Surg. 2011;131:1419-1425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 124] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 19. | Metsemakers WJ, Reul M, Nijs S. The use of gentamicin-coated nails in complex open tibia fracture and revision cases: A retrospective analysis of a single centre case series and review of the literature. Injury. 2015;46:2433-2437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 79] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 20. | ter Boo GJ, Grijpma DW, Moriarty TF, Richards RG, Eglin D. Antimicrobial delivery systems for local infection prophylaxis in orthopedic- and trauma surgery. Biomaterials. 2015;52:113-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 147] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 21. | Arciola CR, Campoccia D, An YH, Baldassarri L, Pirini V, Donati ME, Pegreffi F, Montanaro L. Prevalence and antibiotic resistance of 15 minor staphylococcal species colonizing orthopedic implants. Int J Artif Organs. 2006;29:395-401. [PubMed] |
| 22. | Ambrose CG, Clyburn TA, Mika J, Gogola GR, Kaplan HB, Wanger A, Mikos AG. Evaluation of antibiotic-impregnated microspheres for the prevention of implant-associated orthopaedic infections. J Bone Joint Surg Am. 2014;96:128-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 23. | Ambrose CG, Gogola GR, Clyburn TA, Raymond AK, Peng AS, Mikos AG. Antibiotic microspheres: preliminary testing for potential treatment of osteomyelitis. Clin Orthop Relat Res. 2003;415:279-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Ambrose CG, Clyburn TA, Louden K, Joseph J, Wright J, Gulati P, Gogola GR, Mikos AG. Effective treatment of osteomyelitis with biodegradable microspheres in a rabbit model. Clin Orthop Relat Res. 2004;421:293-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 62] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Reading AD, Rooney P, Taylor GJ. Quantitative assessment of the effect of 0.05% chlorhexidine on rat articular cartilage metabolism in vitro and in vivo. J Orthop Res. 2000;18:762-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 26. | Russell AD, Day MJ. Antibacterial activity of chlorhexidine. J Hosp Infect. 1993;25:229-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 113] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 27. | Cheung HY, Wong MM, Cheung SH, Liang LY, Lam YW, Chiu SK. Differential actions of chlorhexidine on the cell wall of Bacillus subtilis and Escherichia coli. PLoS One. 2012;7:e36659. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 28. | Kim HS, Woo Chang S, Baek SH, Han SH, Lee Y, Zhu Q, Kum KY. Antimicrobial effect of alexidine and chlorhexidine against Enterococcus faecalis infection. Int J Oral Sci. 2013;5:26-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 29. | DeJong ES, DeBerardino TM, Brooks DE, Nelson BJ, Campbell AA, Bottoni CR, Pusateri AE, Walton RS, Guymon CH, McManus AT. Antimicrobial efficacy of external fixator pins coated with a lipid stabilized hydroxyapatite/chlorhexidine complex to prevent pin tract infection in a goat model. J Trauma. 2001;50:1008-1014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 75] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 30. | Ho KM, Litton E. Use of chlorhexidine-impregnated dressing to prevent vascular and epidural catheter colonization and infection: a meta-analysis. J Antimicrob Chemother. 2006;58:281-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 125] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 31. | Yang CC, Lin CC, Liao JW, Yen SK. Vancomycin-chitosan composite deposited on post porous hydroxyapatite coated Ti6Al4V implant for drug controlled release. Mater Sci Eng C Mater Biol Appl. 2013;33:2203-2212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 32. | Magetsari PhD R, Dewo PhD P, Saputro Md BK, Lanodiyu Md Z. Cinnamon Oil and Chitosan Coating on Orthopaedic Implant Surface for Prevention of Staphylococcus Epidermidis Biofilm Formation. Malays Orthop J. 2014;8:11-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 33. | Qin L, Dong H, Mu Z, Zhang Y, Dong G. Preparation and bioactive properties of chitosan and casein phosphopeptides composite coatings for orthopedic implants. Carbohydr Polym. 2015;133:236-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Sondi I, Salopek-Sondi B. Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. J Colloid Interface Sci. 2004;275:177-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3981] [Cited by in RCA: 3006] [Article Influence: 150.3] [Reference Citation Analysis (0)] |
| 35. | Kim JS, Kuk E, Yu KN, Kim JH, Park SJ, Lee HJ, Kim SH, Park YK, Park YH, Hwang CY. Antimicrobial effects of silver nanoparticles. Nanomedicine. 2007;3:95-101. [PubMed] |
| 36. | Shrivastava S, Bera T, Roy A, Singh G, Ramachandrarao P, Dash D. Characterization of enhanced antibacterial effects of novel silver nanoparticles. Nanotechnology. 2007;18:225103. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1206] [Cited by in RCA: 792] [Article Influence: 44.0] [Reference Citation Analysis (0)] |
| 37. | Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, Ramírez JT, Yacaman MJ. The bactericidal effect of silver nanoparticles. Nanotechnology. 2005;16:2346-2353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4690] [Cited by in RCA: 3385] [Article Influence: 169.3] [Reference Citation Analysis (0)] |
| 38. | Pal S, Tak YK, Song JM. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the Gram-negative bacterium Escherichia coli. Appl Environ Microbiol. 2007;73:1712-1720. [PubMed] |
| 39. | Gosheger G, Hardes J, Ahrens H, Streitburger A, Buerger H, Erren M, Gunsel A, Kemper FH, Winkelmann W, Von Eiff C. Silver-coated megaendoprostheses in a rabbit model--an analysis of the infection rate and toxicological side effects. Biomaterials. 2004;25:5547-5556. [PubMed] |
| 40. | Lee D, Cohen RE, Rubner MF. Antibacterial properties of Ag nanoparticle loaded multilayers and formation of magnetically directed antibacterial microparticles. Langmuir. 2005;21:9651-9659. [PubMed] |
| 41. | Jung WK, Koo HC, Kim KW, Shin S, Kim SH, Park YH. Antibacterial activity and mechanism of action of the silver ion in Staphylococcus aureus and Escherichia coli. Appl Environ Microbiol. 2008;74:2171-2178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1249] [Cited by in RCA: 1073] [Article Influence: 63.1] [Reference Citation Analysis (0)] |
| 42. | Dal Lago V, de Oliveira LF, de Almeida Gonçalves K, Kobargb J, Cardoso MB. Size-selective silver nanoparticles: future of biomedical devices with enhanced bactericidal properties. J Mater Chem. 2011;21:12267-12273. [RCA] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 43. | Knetsch ML, Koole LH. New strategies in the development of antimicrobial coatings: the example of increasing usage of silver and silver nanoparticles. Polymer. 2011;3:340-366. [RCA] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 360] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 44. | Zheng Y, Li J, Liu X, Sun J. Antimicrobial and osteogenic effect of Ag-implanted titanium with a nanostructured surface. Int J Nanomedicine. 2012;7:875-884. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 45. | Cao H, Liu X, Meng F, Chu PK. Biological actions of silver nanoparticles embedded in titanium controlled by micro-galvanic effects. Biomaterials. 2011;32:693-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 191] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 46. | Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J Biomed Mater Res. 2000;52:662-668. [PubMed] |
| 47. | Tran N, Kelley MN, Tran PA, Garcia DR, Jarrell JD, Hayda RA, Born CT. Silver doped titanium oxide-PDMS hybrid coating inhibits Staphylococcus aureus and Staphylococcus epidermidis growth on PEEK. Mater Sci Eng C Mater Biol Appl. 2015;49:201-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 48. | Tran N, Tran PA, Jarrell JD, Engiles JB, Thomas NP, Young MD, Hayda RA, Born CT. In vivo caprine model for osteomyelitis and evaluation of biofilm-resistant intramedullary nails. Biomed Res Int. 2013;2013:674378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 49. | Wafa H, Grimer RJ, Reddy K, Jeys L, Abudu A, Carter SR, Tillman RM. Retrospective evaluation of the incidence of early periprosthetic infection with silver-treated endoprostheses in high-risk patients: case-control study. Bone Joint J. 2015;97-B:252-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 137] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 50. | Hardes J, von Eiff C, Streitbuerger A, Balke M, Budny T, Henrichs MP, Hauschild G, Ahrens H. Reduction of periprosthetic infection with silver-coated megaprostheses in patients with bone sarcoma. J Surg Oncol. 2010;101:389-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 235] [Article Influence: 15.7] [Reference Citation Analysis (1)] |
| 51. | Park MV, Neigh AM, Vermeulen JP, de la Fonteyne LJ, Verharen HW, Briedé JJ, van Loveren H, de Jong WH. The effect of particle size on the cytotoxicity, inflammation, developmental toxicity and genotoxicity of silver nanoparticles. Biomaterials. 2011;32:9810-9817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 706] [Cited by in RCA: 657] [Article Influence: 46.9] [Reference Citation Analysis (0)] |
| 52. | AshaRani PV, Low Kah Mun G, Hande MP, Valiyaveettil S. Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano. 2009;3:279-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2712] [Cited by in RCA: 2316] [Article Influence: 144.8] [Reference Citation Analysis (0)] |
| 53. | Kim YS, Kim JS, Cho HS, Rha DS, Kim JM, Park JD, Choi BS, Lim R, Chang HK, Chung YH. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhal Toxicol. 2008;20:575-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 599] [Cited by in RCA: 539] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 54. | Matsunaga T, Tomoda R, Nakajima T, Wake H. Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol Lett. 1985;29:211-214. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1069] [Cited by in RCA: 1079] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 55. | Jamal R, Osman Y, Rahman A, Ali A, Zhang Y, Abdiryim T. Solid-State Synthesis and Photocatalytic Activity of Polyterthiophene Derivatives/TiO2 Nanocomposites. Materials. 2014;7:3786-3801. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 56. | Villatte G, Massard C, Descamps S, Sibaud Y, Forestier C, Awitor KO. Photoactive TiO2 antibacterial coating on surgical external fixation pins for clinical application. Int J Nanomedicine. 2015;10:3367-3375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 57. | Passuti N, Gouin F. Antibiotic-loaded bone cement in orthopedic surgery. Joint Bone Spine. 2003;70:169-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 58. | Samuel S. Antibiotic Loaded Acrylic Bone Cement in Orthopaedic Trauma. In: Bapatista MS, editor. Osteomyelitis. InTech Publishers, Rijeka, Croatia. |
| 59. | Buchholz HW, Engelbrecht H. [Depot effects of various antibiotics mixed with Palacos resins]. Chirurg. 1970;41:511-515. [PubMed] |
| 60. | Yi Z, Bin S, Jing Y, Zongke Z, Pengde K, Fuxing P. No decreased infection rate when using antibiotic-impregnated cement in primary total joint arthroplasty. Orthopedics. 2014;37:839-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 61. | Kendall RW, Duncan CP, Smith JA, Ngui-Yen JH. Persistence of bacteria on antibiotic loaded acrylic depots. A reason for caution. Clin Orthop Relat Res. 1996;329:273-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 116] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 62. | van de Belt H, Neut D, Schenk W, van Horn JR, van der Mei HC, Busscher HJ. Gentamicin release from polymethylmethacrylate bone cements and Staphylococcus aureus biofilm formation. Acta Orthop Scand. 2000;71:625-629. [PubMed] |
| 63. | Perez LM, Lalueza P, Monzon M, Puertolas JA, Arruebo M, Santamaría J. Hollow porous implants filled with mesoporous silica particles as a two-stage antibiotic-eluting device. Int J Pharm. 2011;409:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 64. | Gimeno M, Pinczowski P, Vázquez FJ, Pérez M, Santamaría J, Arruebo M, Luján L. Porous orthopedic steel implant as an antibiotic eluting device: prevention of post-surgical infection on an ovine model. Int J Pharm. 2013;452:166-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 65. | Gimeno M, Pinczowski P, Pérez M, Giorello A, Martínez MÁ, Santamaría J, Arruebo M, Luján L. A controlled antibiotic release system to prevent orthopedic-implant associated infections: An in vitro study. Eur J Pharm Biopharm. 2015;96:264-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 66. | Kaper HJ, Busscher HJ, Norde W. Characterization of poly (ethylene oxide) brushes on glass surfaces and adhesion of Staphylococcus epidermidis. J Biomater Sci Polym Ed. 2003;14:313-324. [PubMed] |
| 67. | Zhang F, Zhang Z, Zhu X, Kang ET, Neoh KG. Silk-functionalized titanium surfaces for enhancing osteoblast functions and reducing bacterial adhesion. Biomaterials. 2008;29:4751-4759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 135] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 68. | Singh AV, Vyas V, Patil R, Sharma V, Scopelliti PE, Bongiorno G, Podestà A, Lenardi C, Gade WN, Milani P. Quantitative characterization of the influence of the nanoscale morphology of nanostructured surfaces on bacterial adhesion and biofilm formation. PLoS One. 2011;6:e25029. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 179] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 69. | Gallo J, Holinka M, Moucha CS. Antibacterial surface treatment for orthopaedic implants. Int J Mol Sci. 2014;15:13849-13880. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 188] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 70. | Braem A, Van Mellaert L, Mattheys T, Hofmans D, De Waelheyns E, Geris L, Anné J, Schrooten J, Vleugels J. Staphylococcal biofilm growth on smooth and porous titanium coatings for biomedical applications. J Biomed Mater Res A. 2014;102:215-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 71. | Gilchrist SE, Lange D, Letchford K, Bach H, Fazli L, Burt HM. Fusidic acid and rifampicin co-loaded PLGA nanofibers for the prevention of orthopedic implant associated infections. J Control Release. 2013;170:64-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 77] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 72. | Reneker DH, Chun I. Nanometre diameter fibers of polymer, produced by electrospinning. Nanotechnology. 1996;7:216-223. [DOI] [Full Text] |
| 73. | Reneker DH, Yarin AL, Fong H, Koombhongse S. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J Appl Phys. 2000;87:4531-4547. [DOI] [Full Text] |
| 74. | Zhang L, Yan J, Yin Z, Tang C, Guo Y, Li D, Wei B, Xu Y, Gu Q, Wang L. Electrospun vancomycin-loaded coating on titanium implants for the prevention of implant-associated infections. Int J Nanomedicine. 2014;9:3027-3036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 75. | Özçelik H, Vrana NE, Gudima A, Riabov V, Gratchev A, Haikel Y, Metz-Boutigue MH, Carradò A, Faerber J, Roland T. Harnessing the multifunctionality in nature: a bioactive agent release system with self-antimicrobial and immunomodulatory properties. Adv Healthc Mater. 2015;4:2026-2036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |