Published online Jul 18, 2014. doi: 10.5312/wjo.v5.i3.247
Revised: May 19, 2014
Accepted: May 29, 2014
Published online: July 18, 2014
Processing time: 245 Days and 7.3 Hours
Vertebral and nonvertebral fractures prevention is the main goal for osteoporosis therapy by inhibiting bone resorption and/or stimulating bone formation. Antiresorptive drugs decrease the activation frequency, thereby determining a secondary decrease in bone formation rate and a low bone turnover. Bisphosphonates are today’s mainstay among antiresorptive treatment of osteoporosis. Also, oral selective estrogen receptor modulators and recently denosumab have a negative effect on bone turnover. Agents active on bone formation are considered a better perspective in the treatment of severe osteoporosis. Recombinant-human parathyroid hormone (PTH) has showed to increase bone formation and significantly decrease vertebral fractures in severe patients, but with a modest effect on nonvertebral fractures. The study of Wnt signaling pathway, that induces prevalently an osteoblastic activity, opens large possibilities to antagonists of Wnt-inhibitors, such as sclerostin antibodies and dickkopf-1 antagonists, with potential effects not only on trabecular bone but also on cortical bone.
Core tip: The study of agents active on bone formation is the main objective in the treatment of severe osteoporosis. rhparathyroid hormone (rhPTH) decreases vertebral, but not nonvertebral, fractures. On the contrary, antagonists of Wnt-inhibitors, that exert their effects mostly through a bone remodeling-independent mechanism, open new perspectives to improve not only trabecular bone but also cortical bone, with potential positive effect also on nonvertebral fractures incidence. The perspective in osteoporosis treatment should be more effective and better tolerated therapies aimed at minimizing individually fractures risk.
- Citation: Montagnani A. Bone anabolics in osteoporosis: Actuality and perspectives. World J Orthop 2014; 5(3): 247-254
- URL: https://www.wjgnet.com/2218-5836/full/v5/i3/247.htm
- DOI: https://dx.doi.org/10.5312/wjo.v5.i3.247
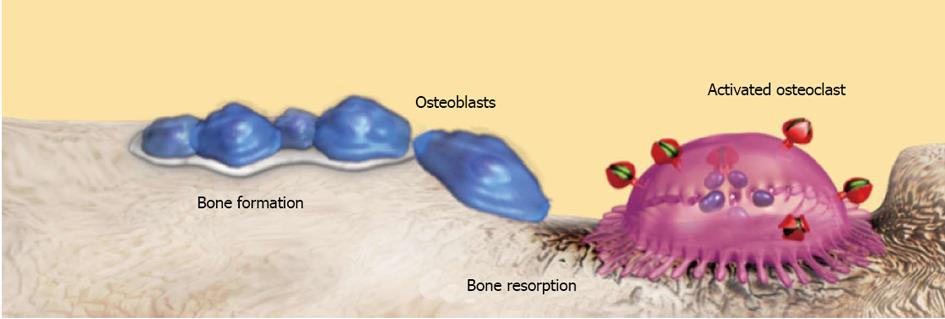
Osteoporosis is an emerging medical and socioeconomic threat characterized by a systemic impairment of bone mass, strength and microarchitecture, which increases the propensity of fragility fractures[1]. Osteoporosis results by a dysfunction of physiological bone turnover and cells in bone by endocrine and/or autocrine/paracrine factors (Figure 1), negatively affecting peak bone mass and/or skeletal homeostasis. Patient with osteoporosis show a higher propensity to spine and femur fractures, even if other bones could be also involved.
Osteoporotic fractures of the hip and spine increase mortality and are related to important medical complications that, such as pneumonia or thromboembolic disease due to chronic immobilization with a negative economic impact on public health[2].
Osteoporosis is considered a global public health concern and result to have great socioeconomic burden[3], worthy to be addressed in an evidence-based and cost-effective manner[4-6], taking into account several risk factors[7]. Taking into account these preliminary considerations is resulting that osteoporosis therapy is considered an important field of study where to converge most of the efforts.
Osteoporosis therapy should prevent both vertebral (mostly dependent on trabecular bone density and architecture) and nonvertebral (mostly dependent on cortical thickness and porosity) fractures. This could be achieved by inhibiting bone resorption and/or by stimulating bone formation.
Bone remodeling or modeling activity is different between cortical and trabecular bone sites and this difference could mostly explain the relative lack of efficacy of antiresorptive drugs on nonvertebral fractures since their effect is higher on trabecular than cortical bone.
Bisphosphonates are the most prescribed drugs for osteoporosis treatment. They have a high affinity for bone and inhibit bone resorption reducing fracture risk. Alendronate, risedronate, and zoledronate were shown to reduce the risk of new vertebral, non-vertebral, and hip fractures[8-12], showing a prevalent effect on axial with respect to appendicular skeletal site, a relative risk reduction of 50% for spine vs 20%. Although, long term treatment with bisphosphonate has been associated with a potential risk of osteonecrosis of the jaw and of atypical subtrochanteric femoral fractures, their use for at least 10 years has shown good safety[13,14].
Raloxifene, bazedoxifene and subcutaneous denosumab, a human monoclonal antibody that inhibits RANKL, have showed convincing evidences to reduce osteoporotic fractures. Raloxifene have a positive effect on vertebral fracture and on breast cancer risk worsening the thrombotic risk[15,16]. Denosumab, instead, reduced vertebral, non-vertebral and hip fracture risk in postmenopausal women with osteoporosis by the same order of magnitude as bisphosphonates without significant adverse events[17]. A particular behavior seems to have strontium ranelate (SR), which has a double effect, anabolic, inducing an increase of osteoblast activity, and at the same time antiresorptive, inhibiting osteoclasts activity[18]. In a recent meta-analysis Kanis et al[19] reported positive effect on clinical and morphometric vertebral fractures. Since SR has shown to have a reduced safety in patients with venous thromboembolism and ischaemic heart diseases, such a drug should not be administered to patients with a higher risk of atherothrombotic events.
In synthesis, antiresorptive drugs reduce the activation frequency, acting mostly on osteoclast and only indirectly on osteoblast activity, with e final slight gain in trabecular bone mass.
Anabolic therapies, instead, directly stimulate bone formation through activation of bone modeling, independently of resorption activity, suggesting a potential positive effect on non-vertebral other than vertebral fractures.
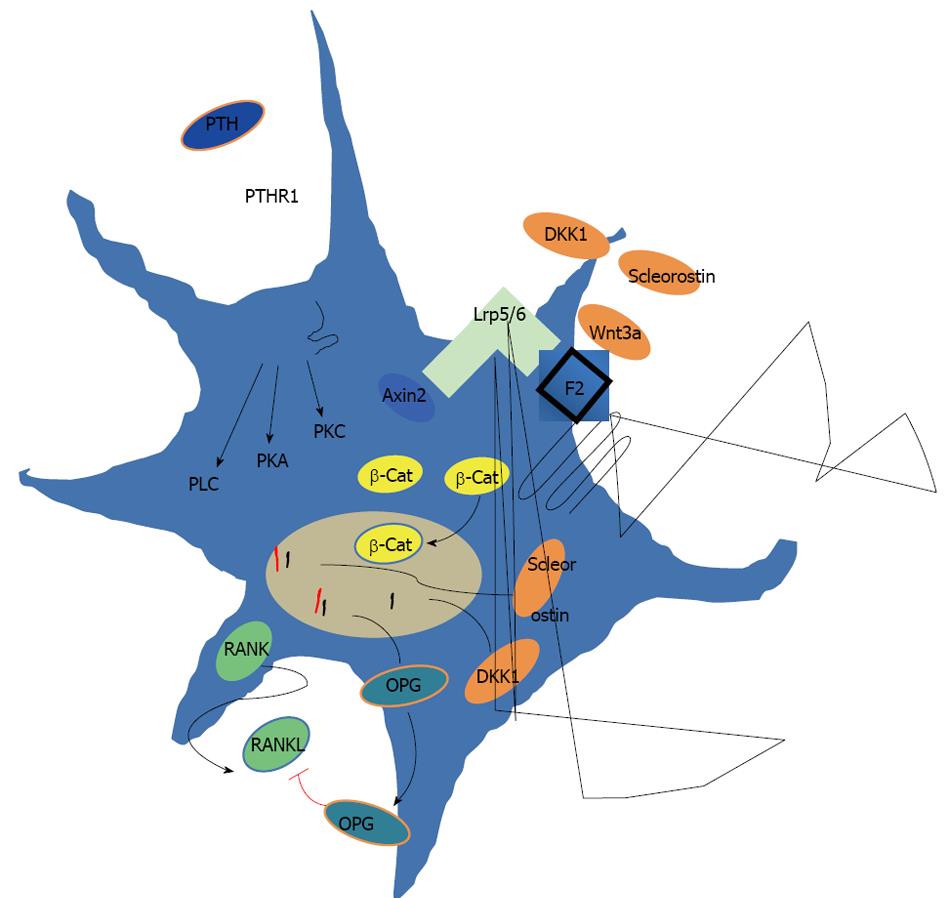
In Figure 2 are reported the two main bone anabolic pathways: one linked to parathyroid hormone (PTH) signaling and the second dependent on canonical wingless-int (Wnt) signaling (Figure 2). The main difference between this two pathways is that Wnt-signaling acts increasing bone mass independently of bone remodeling, as it does PTH induces an increase of osteoblastic and osteoclastic activity. This could explain why PTH shows a closer therapeutic windows.
The secretion of human PTH, an 84-amino acid peptide, by parathyroid cells is closely controlled by serum calcium levels through the calcium-sensing receptors (CaSR). This hormone plays an important role in calcium homeostasis. PTH determines an increase of serum calcium by mobilization of skeletal stores, increasing intestinal and renal calcium absorption[20]. When PTH is administered by intermittent subcutaneous via, it has an anabolic effect on bone, influencing osteoblastic activity directly and indirectly with the regulation of some growth factors[21].
To date, injectable forms of recombinant-human PTH (rhPTH) are the only approved osteoanabolic drugs on the market for the treatment of osteoporosis. It exists an intact form (rhPTH 1-84) and an other bioactive N-terminal 34-amino acid fragment rhPTH 1-34 (teriparatide). rhPTH showed a higher effects on trabecular bone reducing more the relative risk of vertebral than nonvertebral fractures, confirming that rhPTH has a prevalent effect on trabecular rather than on cortical bone[22].
Osteoblasts, activated by rhPTH, produce several paracrine factors, which in turn stimulate osteoclast activity. This, when the rhPTH intermittent treatment is prolonged, could enhance activation frequency and thereby increase bone resorption. Although the initial net effect is positive with a gain of trabecular bone mass, the anabolic effect could show a plateau curve when the treatment is prolonged beyond two years[22]. Such limit could be overcome by a co-administration of an antiresorptive drug able to limit the rhPTH-activated bone resorption. Some experiences did not report consistent evidence that confirm such hypothesis[23,24], however, a recent study has reported that one single administration of zoledronic acid combined with daily sc injections of rhPTH could reduced fracture risk in patients with a high risk profile[25]. On the other hand, sequential administration of antiresorptive drugs after rhPTH is already an established treatment protocol that limit bone resorption after withdraw of rhPTH treatment[26].
Although, rhPTH is usually well tolerated, some adverse effects, such as hypercalcemia, nausea, headache, dizziness, and leg cramps, could be associated to rhPTH treatment with a lower risk of hypercalcemia for the rhPTH 1-84[26].
To improve the rhPTH safety profile some attractive options for the alternative delivery have been tested. One is transdermal self-administration using coated microneedle patches[27] whereas other are inhaled and oral delivery[28]. In the first case PTH interestingly showed an increased of trabecular bone to the same extent whereas the gain of total hip BMD was much greater than those obtained with sc administered rhPTH 1-34[27]. Oral and inhaled administrations are being investigated in phase I studies, showing interesting data.
Since rhPTH use is limited by a low effect on non-vertebral fractures, by the osteoclasts activation and by the loss of efficacy in a prolonged treatment, it seems to need to search new molecule which show a better profile.
PTH related peptide (PTHrP) shows a similar sequence to PTH in its first 36 amino acids and activates PTH1R. In rats and in humans PTHrP has demonstrated similar effect to rhPTH on bone mass, improving mechanical strength of bone tissue in rats[29]. However, PTHrP appeared to stimulate only bone formation as a pure bone anabolic agent; as showed by bone turnover markers variations with an increase of bone formation markers, such as osteocalcin and P1NP associated to unchanged levels of bone resorption markers[30]. In a phase 2 study the administration of PTHrP in postmenopausal women determined an 4%-5%/year increase of BMD without serious adverse effects[31]. On this basis, some phase 3 studies are ongoing and could give further information on efficacy and safety of this interesting molecule, namely in comparison with PTH (http://www.clinicaltrials.gov).
PTH is synthesized and secreted by parathyroid glands cells expressing on their surface calcium-sensing receptor (CaSR). Serum low levels of Ca2+ determine a low bond with CaSR decreasing its activity, and in turn stimulating PTH release. On the contrary, activation of the CaSR decreases PTH synthesis and secretion[32].
Antagonists of the CaSR bind and inhibit the receptor determining a short pulse of PTH secretion. A rapid increase of PTH secretion followed by rapid normalization should cause an anabolic effect in bone. Unfortunately, calcilytics, considered a new class of bone-forming agents, have showed an unfavorable pharmacokinetics[33]. In fact, a close therapeutic window between the effect on bone and hypercalcemia, the fact that CaSR are also expressed in other organs besides the parathyroid glands and finally, that together to PTH other products, with potential negative effects on PTH secretion itself, represent actual limits to use of these new anabolic drugs[34]. Although the mechanism of action, calcilytics remain an interesting opportunity for treatment of a reduced bone mass. However, these drugs are worthy of furthers studies to clarify their role in osteoporosis therapy.
In the last decade, some genetic study of the low-density lipoprotein receptor-related protein 5 (Lrp5) associated to low or high bone mass, suggested a potential role of the Wnt pathway as an important player influencing bone mass and as possible target to the PTH signaling pathway (Figure 2).
To date two endogenous inhibitors of the Wnt/β-catenin pathway specific to bone have been known: sclerostin (SOST) and dickkopf-1 (dkk1). These molecule inhibit Wnt signal stopping β-catenin degradation and osteoblast differentiation. When SOST and dkk1 are blocked by specific antibodies bone formation increases with an anabolic effect.
Binding of Wnt to Lrp5/6 prevents the phosphorylation and the proteasomal degradation of β-catenin, stimulates the production of osteoprotegerin (OPG), an osteoblast-derived inhibitor of osteoclast differentiation[35] that acts by binding to RANKL and preventing it from binding to its receptor, RANK.
The fact that Wnt signaling pathway is blocked by endogenous inhibitor factors, represents an important opportunity in the field of osteoporosis therapy.
Sclerostin expression is prevalently restricted to late osteoblasts and osteocytes[36], and therefore could represent a favorable target of osteoporosis treatment. In studies in animals, SOST antibodies significantly improved the healing of fractures with an increase in bone formation, bone mass, and bone strength[37]. Similar findings were made in sclerostin knockout mice and in ovariectomized rats treated with sclerostin antibodies[38].
The first human phase I clinical trial, studying a humanized monoclonal sclerostin antibody in healthy men and postmenopausal women, showed that this new compound had effects on bone formation and resorption after a single month similar to those showed by rhPTH after 6 months, but with greater effects on bone mass than rhPTH[39]. Such data are, then confirmed, in a phase II study in more than 400 postmenopausal osteoporotic women who showed a significantly increase in BMD at the lumbar spine compared with placebo and teriparatide[40].
Antagonists to inhibitors of the Wnt pathway have show to have positive effect on osteoblast activity regardless of osteoclasts and bone resorption. This their characteristic may be useful not only in osteoporosis but also in other pathologic conditions, such as bone repair after fracture and in low bone turnover diseases.
Dkk1 is a further endogenous inhibitor of Wnt signaling. Its neutralization by antibodies is still limited to preclinical trials which have showed an inhibited bone loss in a model of rheumatoid[41] and the prevention of the formation of osteolytic lesions with an increased bone formation rate in a myeloma model[42].
These antibodies could also play a role in the treatment of diseases characterized by a low bone mass, first of all osteoporosis. Some concerns may exist about the possibility that Dkk1 is less selective for bone than SOST with possible more off-target effects.
The possibility to induce the Wnt signaling pathway is a very promising, however, some doubt exist regarding possible important adverse-effects, namely oncogenic effects and a possible uncontrolled process of bone formation with important neurological consequences at cranial and spine levels. Therefore, a particular attention must be taken in long-term use of Wnt antagonists inhibitors.
Activin A, a transforming growth factor-β (TGF-β) superfamily member, has showed to be an antagonist to human osteoblast differentiation[43] and to induce osteoclast formation and bone resorption[44]. On this basis, an antagonist of activin should shift the balance of bone turnover in favor of bone formation. In fact, as showed by a phase I trial, using an activin antagonist increased markers of bone formation[45] of similar extent determined by rhPTH or antagonists of Wnt signaling inhibitors.
Some evidence indicate that prostaglandin E2 (PGE2) play a role in bone metabolism by stimulating bone turnover with a prevalence of bone formation and thereby an increasing bone mass and bone strength[46]. A study in OVX rats animal models has showed that a subcutaneous administration of PGE2 E4 receptor agonist stimulates bone formation by increasing osteoblast recruitment activity on periosteal, endocortical, and trabecular surfaces[47]. The PGE2 effect seems to be present on both smooth and scalloped endocortical and trabecular surface, suggesting an effect both on bone modeling and remodeling-dependent bone formation.
Statins have a well-know hypocholesterolemic effect by reducing 3-hydroxy-3-glutaryl-coenzyme A (HMG-CoA) reductase activity. However, the blocking of such enzyme causes the depletion of farnesyl diphosphate or geranyl diphosphate synthesis and in turn the reduction of protein prenylation, which plays a role in bone cells activity by preventing the post-translational modifications of small GTPases.
However, the main proposed mechanism by which statins stimulate bone formation involves an increase in expression and synthesis of BMP-2[48] and osteocalcin[49].
Evidence regarding the effects of statins on BMD[50,51] and fracture risk are not completely consistent but do suggest the anabolic potential of these drugs. In fact, a meta-analysis conclude that statins reduce hip fracture risk and, to a lesser extent, nonspine fracture risk[52].
Unfortunately, statin shows a high affinity for the liver and only very low concentration reach the bone as potential target. Therefore, to overcome the liver first-pass effect, statins would be administered in a suitable delivery system aimed to allow the major concentration in fracture sites. In such sense, a perspective could be a different copolymerization with ethylene glycol that covalently incorporates into hydrogel networks[53] or a different administration route, as a transdermal application, which bypasses the first-pass liver effect[54].
Administration of insulin-growth-factor I (IGF-I) determines an increase of bone mass with an anabolic effect by inducing bone remodeling both in healthy and in subjects with GH deficiency or IGF-I deficiency[55]. Although, recombinant human IGF-I is used currently for the treatment of short stature genetic syndromes secondary to caused by mutations of the GH receptor or the IGF1 gene, the long-term efficacy and safety of IGF-I in patients with osteoporosis remain to be determined.
An interesting suggestion to identify a novel future anabolic therapy of osteoporosis seems to come from the study of marrow cultures from the proline-rich tyrosine kinase 2 (PYK2)-null mice, which showed enhanced osteogenesis[56]. Blocking PYK2 activity may be hypothesized to have an osteogenic effect also in humans. However, no evidence for such effect in humans has been reported and therefore up to date, this remains only an interesting field of study.
All antiresorptive drugs share a minor effect on nonvertebral fracture and this remains the biggest limit of severe osteoporosis therapy inducing an important research to identify an agent able to induce bone formation rather than block resorption[57].
To date, only some drugs have demonstrated to have an anabolic effect on bone; one of these, rhPTH, increases bone formation and significantly decreases vertebral fractures in severe patients, but it is less effective on nonvertebral fractures, probably because rhPTH action is mostly based on bone remodeling, that induces an increase both osteoblasts and osteoclasts activity. On the contrary, the agents influencing Wnt signaling pathway, mostly linked to a bone remodeling-independent mechanism (modeling-based), prevalently affect osteoblastic activity, thereby with a major improvement of trabecular than cortical bone. This action may be thought that Antagonists of Wnt-inhibitors may reduce the incidence of nonvertebral other than of vertebral fractures.
In the next years, several clinical trials could give further data making available more effective and better tolerated therapies allowing tailor-made approaches aimed at minimizing individually fractures risk.
P- Reviewers: Belaya ZE, Bruyere O, Faienza MF, Hung SH S- Editor: Song XX L- Editor: A E- Editor: Lu YJ
| 1. | NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA. 2001;285:785-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3072] [Cited by in RCA: 3079] [Article Influence: 128.3] [Reference Citation Analysis (0)] |
| 2. | Center JR, Nguyen TV, Schneider D, Sambrook PN, Eisman JA. Mortality after all major types of osteoporotic fracture in men and women: an observational study. Lancet. 1999;353:878-882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1338] [Cited by in RCA: 1333] [Article Influence: 51.3] [Reference Citation Analysis (0)] |
| 3. | Harvey N, Dennison E, Cooper C. Osteoporosis: impact on health and economics. Nat Rev Rheumatol. 2010;6:99-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 392] [Cited by in RCA: 491] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 4. | Hodgson SF, Watts NB, Bilezikian JP, Clarke BL, Gray TK, Harris DW, Johnston CC, Kleerekoper M, Lindsay R, Luckey MM. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the prevention and treatment of postmenopausal osteoporosis: 2001 edition, with selected updates for 2003. Endocr Pract. 2003;9:544-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 250] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 5. | Compston J, Cooper A, Cooper C, Francis R, Kanis JA, Marsh D, McCloskey EV, Reid DM, Selby P, Wilkins M. Guidelines for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK. Maturitas. 2009;62:105-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 264] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 6. | Brown JP, Josse RG. 2002 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada. CMAJ. 2002;167:S1-34. [PubMed] |
| 7. | Kanis JA. Diagnosis of osteoporosis and assessment of fracture risk. Lancet. 2002;359:1929-1936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1444] [Cited by in RCA: 1433] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 8. | Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, Cosman F, Lakatos P, Leung PC, Man Z. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356:1809-1822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2034] [Cited by in RCA: 1967] [Article Influence: 109.3] [Reference Citation Analysis (0)] |
| 9. | Black DM, Thompson DE, Bauer DC, Ensrud K, Musliner T, Hochberg MC, Nevitt MC, Suryawanshi S, Cummings SR. Fracture risk reduction with alendronate in women with osteoporosis: the Fracture Intervention Trial. FIT Research Group. J Clin Endocrinol Metab. 2000;85:4118-4124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 530] [Cited by in RCA: 554] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 10. | Cummings SR, Black DM, Thompson DE, Applegate WB, Barrett-Connor E, Musliner TA, Palermo L, Prineas R, Rubin SM, Scott JC. Effect of alendronate on risk of fracture in women with low bone density but without vertebral fractures: results from the Fracture Intervention Trial. JAMA. 1998;280:2077-2082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1643] [Cited by in RCA: 1476] [Article Influence: 54.7] [Reference Citation Analysis (0)] |
| 11. | McClung MR, Geusens P, Miller PD, Zippel H, Bensen WG, Roux C, Adami S, Fogelman I, Diamond T, Eastell R. Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group. N Engl J Med. 2001;344:333-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1386] [Cited by in RCA: 1245] [Article Influence: 51.9] [Reference Citation Analysis (0)] |
| 12. | Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut CH, Brown J, Eriksen EF, Hoseyni MS. Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA. 1999;282:1344-1352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1762] [Cited by in RCA: 1582] [Article Influence: 60.8] [Reference Citation Analysis (0)] |
| 13. | Black DM, Schwartz AV, Ensrud KE, Cauley JA, Levis S, Quandt SA, Satterfield S, Wallace RB, Bauer DC, Palermo L. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA. 2006;296:2927-2938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1000] [Cited by in RCA: 942] [Article Influence: 49.6] [Reference Citation Analysis (0)] |
| 14. | Rizzoli R, Akesson K, Bouxsein M, Kanis JA, Napoli N, Papapoulos S, Reginster JY, Cooper C. Subtrochanteric fractures after long-term treatment with bisphosphonates: a European Society on Clinical and Economic Aspects of Osteoporosis and Osteoarthritis, and International Osteoporosis Foundation Working Group Report. Osteoporos Int. 2011;22:373-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 161] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 15. | Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J. Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) Investigators. JAMA. 1999;282:637-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2393] [Cited by in RCA: 2058] [Article Influence: 79.2] [Reference Citation Analysis (0)] |
| 16. | Martino S, Cauley JA, Barrett-Connor E, Powles TJ, Mershon J, Disch D, Secrest RJ, Cummings SR. Continuing outcomes relevant to Evista: breast cancer incidence in postmenopausal osteoporotic women in a randomized trial of raloxifene. J Natl Cancer Inst. 2004;96:1751-1761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 496] [Cited by in RCA: 446] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 17. | Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, Delmas P, Zoog HB, Austin M, Wang A. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361:756-765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2264] [Cited by in RCA: 2278] [Article Influence: 142.4] [Reference Citation Analysis (0)] |
| 18. | Canalis E, Giustina A, Bilezikian JP. Mechanisms of anabolic therapies for osteoporosis. N Engl J Med. 2007;357:905-916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 499] [Cited by in RCA: 470] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 19. | Kanis JA, Johansson H, Oden A, McCloskey EV. A meta-analysis of the effect of strontium ranelate on the risk of vertebral and non-vertebral fracture in postmenopausal osteoporosis and the interaction with FRAX(®). Osteoporos Int. 2011;22:2347-2355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 20. | Hodsman AB, Bauer DC, Dempster DW, Dian L, Hanley DA, Harris ST, Kendler DL, McClung MR, Miller PD, Olszynski WP. Parathyroid hormone and teriparatide for the treatment of osteoporosis: a review of the evidence and suggested guidelines for its use. Endocr Rev. 2005;26:688-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 489] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 21. | Jilka RL. Molecular and cellular mechanisms of the anabolic effect of intermittent PTH. Bone. 2007;40:1434-1446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 575] [Cited by in RCA: 507] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 22. | Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001;344:1434-1441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3315] [Cited by in RCA: 3048] [Article Influence: 127.0] [Reference Citation Analysis (0)] |
| 23. | Black DM, Greenspan SL, Ensrud KE, Palermo L, McGowan JA, Lang TF, Garnero P, Bouxsein ML, Bilezikian JP, Rosen CJ. The effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med. 2003;349:1207-1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 926] [Cited by in RCA: 794] [Article Influence: 36.1] [Reference Citation Analysis (0)] |
| 24. | Finkelstein JS, Wyland JJ, Lee H, Neer RM. Effects of teriparatide, alendronate, or both in women with postmenopausal osteoporosis. J Clin Endocrinol Metab. 2010;95:1838-1845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 194] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 25. | Cosman F, Eriksen EF, Recknor C, Miller PD, Guañabens N, Kasperk C, Papanastasiou P, Readie A, Rao H, Gasser JA. Effects of intravenous zoledronic acid plus subcutaneous teriparatide [rhPTH(1-34)] in postmenopausal osteoporosis. J Bone Miner Res. 2011;26:503-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 235] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 26. | Black DM, Bilezikian JP, Ensrud KE, Greenspan SL, Palermo L, Hue T, Lang TF, McGowan JA, Rosen CJ. One year of alendronate after one year of parathyroid hormone (1-84) for osteoporosis. N Engl J Med. 2005;353:555-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 409] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 27. | Cosman F, Lane NE, Bolognese MA, Zanchetta JR, Garcia-Hernandez PA, Sees K, Matriano JA, Gaumer K, Daddona PE. Effect of transdermal teriparatide administration on bone mineral density in postmenopausal women. J Clin Endocrinol Metab. 2010;95:151-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 95] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 28. | Shoyele SA, Sivadas N, Cryan SA. The effects of excipients and particle engineering on the biophysical stability and aerosol performance of parathyroid hormone (1-34) prepared as a dry powder for inhalation. AAPS PharmSciTech. 2011;12:304-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 29. | Stewart AF, Cain RL, Burr DB, Jacob D, Turner CH, Hock JM. Six-month daily administration of parathyroid hormone and parathyroid hormone-related protein peptides to adult ovariectomized rats markedly enhances bone mass and biomechanical properties: a comparison of human parathyroid hormone 1-34, parathyroid hormone-related protein 1-36, and SDZ-parathyroid hormone 893. J Bone Miner Res. 2000;15:1517-1525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 103] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 30. | Horwitz MJ, Tedesco MB, Garcia-Ocaña A, Sereika SM, Prebehala L, Bisello A, Hollis BW, Gundberg CM, Stewart AF. Parathyroid hormone-related protein for the treatment of postmenopausal osteoporosis: defining the maximal tolerable dose. J Clin Endocrinol Metab. 2010;95:1279-1287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 31. | Horwitz MJ, Tedesco MB, Gundberg C, Garcia-Ocana A, Stewart AF. Short-term, high-dose parathyroid hormone-related protein as a skeletal anabolic agent for the treatment of postmenopausal osteoporosis. J Clin Endocrinol Metab. 2003;88:569-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 130] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 32. | John MR, Widler L, Gamse R, Buhl T, Seuwen K, Breitenstein W, Bruin GJ, Belleli R, Klickstein LB, Kneissel M. ATF936, a novel oral calcilytic, increases bone mineral density in rats and transiently releases parathyroid hormone in humans. Bone. 2011;49:233-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 33. | Fukumoto S. [Antagonist for calcium-sensing receptor. JTT-305/MK-5442]. Clin Calcium. 2011;21:89-93. [PubMed] [DOI] [Full Text] |
| 34. | Cohn DV, Fasciotto BH, Reese BK, Zhang JX. Chromogranin A: a novel regulator of parathyroid gland secretion. J Nutr. 1995;125:2015S-2019S. [PubMed] |
| 35. | MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17:9-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 36. | van Bezooijen RL, ten Dijke P, Papapoulos SE, Löwik CW. SOST/sclerostin, an osteocyte-derived negative regulator of bone formation. Cytokine Growth Factor Rev. 2005;16:319-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 256] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 37. | Ominsky MS, Li C, Li X, Tan HL, Lee E, Barrero M, Asuncion FJ, Dwyer D, Han CY, Vlasseros F. Inhibition of sclerostin by monoclonal antibody enhances bone healing and improves bone density and strength of nonfractured bones. J Bone Miner Res. 2011;26:1012-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 202] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 38. | Li X, Warmington KS, Niu QT, Asuncion FJ, Barrero M, Grisanti M, Dwyer D, Stouch B, Thway TM, Stolina M. Inhibition of sclerostin by monoclonal antibody increases bone formation, bone mass, and bone strength in aged male rats. J Bone Miner Res. 2010;25:2647-2656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 171] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 39. | Padhi D, Jang G, Stouch B, Fang L, Posvar E. Single-dose, placebo-controlled, randomized study of AMG 785, a sclerostin monoclonal antibody. J Bone Miner Res. 2011;26:19-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 578] [Cited by in RCA: 570] [Article Influence: 40.7] [Reference Citation Analysis (0)] |
| 40. | Baron R, Hesse E. Update on bone anabolics in osteoporosis treatment: rationale, current status, and perspectives. J Clin Endocrinol Metab. 2012;97:311-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 250] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 41. | Diarra D, Stolina M, Polzer K, Zwerina J, Ominsky MS, Dwyer D, Korb A, Smolen J, Hoffmann M, Scheinecker C. Dickkopf-1 is a master regulator of joint remodeling. Nat Med. 2007;13:156-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 934] [Cited by in RCA: 977] [Article Influence: 54.3] [Reference Citation Analysis (0)] |
| 42. | Heath DJ, Chantry AD, Buckle CH, Coulton L, Shaughnessy JD, Evans HR, Snowden JA, Stover DR, Vanderkerken K, Croucher PI. Inhibiting Dickkopf-1 (Dkk1) removes suppression of bone formation and prevents the development of osteolytic bone disease in multiple myeloma. J Bone Miner Res. 2009;24:425-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 196] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 43. | Eijken M, Swagemakers S, Koedam M, Steenbergen C, Derkx P, Uitterlinden AG, van der Spek PJ, Visser JA, de Jong FH, Pols HA. The activin A-follistatin system: potent regulator of human extracellular matrix mineralization. FASEB J. 2007;21:2949-2960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 142] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 44. | Sugatani T, Alvarez UM, Hruska KA. Activin A stimulates IkappaB-alpha/NFkappaB and RANK expression for osteoclast differentiation, but not AKT survival pathway in osteoclast precursors. J Cell Biochem. 2003;90:59-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 84] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 45. | Ruckle J, Jacobs M, Kramer W, Pearsall AE, Kumar R, Underwood KW, Seehra J, Yang Y, Condon CH, Sherman ML. Single-dose, randomized, double-blind, placebo-controlled study of ACE-011 (ActRIIA-IgG1) in postmenopausal women. J Bone Miner Res. 2009;24:744-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 169] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 46. | Jee WS, Ma YF. The in vivo anabolic actions of prostaglandins in bone. Bone. 1997;21:297-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 134] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 47. | Ke HZ, Crawford DT, Qi H, Simmons HA, Owen TA, Paralkar VM, Li M, Lu B, Grasser WA, Cameron KO. A nonprostanoid EP4 receptor selective prostaglandin E2 agonist restores bone mass and strength in aged, ovariectomized rats. J Bone Miner Res. 2006;21:565-575. [PubMed] |
| 48. | Mundy G, Garrett R, Harris S, Chan J, Chen D, Rossini G, Boyce B, Zhao M, Gutierrez G. Stimulation of bone formation in vitro and in rodents by statins. Science. 1999;286:1946-1949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1289] [Cited by in RCA: 1254] [Article Influence: 48.2] [Reference Citation Analysis (0)] |
| 49. | Ohnaka K, Shimoda S, Nawata H, Shimokawa H, Kaibuchi K, Iwamoto Y, Takayanagi R. Pitavastatin enhanced BMP-2 and osteocalcin expression by inhibition of Rho-associated kinase in human osteoblasts. Biochem Biophys Res Commun. 2001;287:337-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 152] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 50. | Montagnani A, Gonnelli S, Cepollaro C, Pacini S, Campagna MS, Franci MB, Lucani B, Gennari C. Effect of simvastatin treatment on bone mineral density and bone turnover in hypercholesterolemic postmenopausal women: a 1-year longitudinal study. Bone. 2003;32:427-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 91] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 51. | Edwards CJ, Hart DJ, Spector TD. Oral statins and increased bone-mineral density in postmenopausal women. Lancet. 2000;355:2218-2219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 216] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 52. | Bauer DC, Mundy GR, Jamal SA, Black DM, Cauley JA, Ensrud KE, van der Klift M, Pols HA. Use of statins and fracture: results of 4 prospective studies and cumulative meta-analysis of observational studies and controlled trials. Arch Intern Med. 2004;164:146-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 138] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 53. | Benoit DS, Nuttelman CR, Collins SD, Anseth KS. Synthesis and characterization of a fluvastatin-releasing hydrogel delivery system to modulate hMSC differentiation and function for bone regeneration. Biomaterials. 2006;27:6102-6110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 114] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 54. | Gutierrez GE, Lalka D, Garrett IR, Rossini G, Mundy GR. Transdermal application of lovastatin to rats causes profound increases in bone formation and plasma concentrations. Osteoporos Int. 2006;17:1033-1042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 71] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 55. | Langlois JA, Rosen CJ, Visser M, Hannan MT, Harris T, Wilson PW, Kiel DP. Association between insulin-like growth factor I and bone mineral density in older women and men: the Framingham Heart Study. J Clin Endocrinol Metab. 1998;83:4257-4262. [PubMed] |
| 56. | Buckbinder L, Crawford DT, Qi H, Ke HZ, Olson LM, Long KR, Bonnette PC, Baumann AP, Hambor JE, Grasser WA. Proline-rich tyrosine kinase 2 regulates osteoprogenitor cells and bone formation, and offers an anabolic treatment approach for osteoporosis. Proc Natl Acad Sci USA. 2007;104:10619-10624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 122] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 57. | Rachner TD, Khosla S, Hofbauer LC. Osteoporosis: now and the future. Lancet. 2011;377:1276-1287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1770] [Cited by in RCA: 1903] [Article Influence: 135.9] [Reference Citation Analysis (0)] |