Revised: December 8, 2012
Accepted: December 15, 2012
Published online: January 18, 2013
Processing time: 136 Days and 4.1 Hours
AIM: To investigate whether the finger movement at birth is a better predictor of the brachial plexus birth injury.
METHODS: We conducted a retrospective study reviewing pre-surgical records of 87 patients with residual obstetric brachial plexus palsy in study 1. Posterior subluxation of the humeral head (PHHA), and glenoid retroversion were measured from computed tomography or Magnetic resonance imaging, and correlated with the finger movement at birth. The study 2 consisted of 141 obstetric brachial plexus injury patients, who underwent primary surgeries and/or secondary surgery at the Texas Nerve and Paralysis Institute. Information regarding finger movement was obtained from the patient’s parent or guardian during the initial evaluation.
RESULTS: Among 87 patients, 9 (10.3%) patients who lacked finger movement at birth had a PHHA > 40%, and glenoid retroversion < -12°, whereas only 1 patient (1.1%) with finger movement had a PHHA > 40%, and retroversion < -8° in study 1. The improvement in glenohumeral deformity (PHHA, 31.8% ± 14.3%; and glenoid retroversion 22.0°± 15.0°) was significantly higher in patients, who have not had any primary surgeries and had finger movement at birth (group 1), when compared to those patients, who had primary surgeries (nerve and muscle surgeries), and lacked finger movement at birth (group 2), (PHHA 10.7% ± 15.8%; Version -8.0°± 8.4°, P = 0.005 and P = 0.030, respectively) in study 2. No finger movement at birth was observed in 55% of the patients in this study group.
CONCLUSION: Posterior subluxation and glenoid retroversion measurements indicated significantly severe shoulder deformities in children with finger movement at birth, in comparison with those lacked finger movement. However, the improvement after triangle tilt surgery was higher in patients who had finger movement at birth.
- Citation: Nath RK, Benyahia M, Somasundaram C. Finger movement at birth in brachial plexus birth palsy. World J Orthop 2013; 4(1): 24-28
- URL: https://www.wjgnet.com/2218-5836/full/v4/i1/24.htm
- DOI: https://dx.doi.org/10.5312/wjo.v4.i1.24
Normal shoulder development requires balanced dynamic muscle environment between the humeral head and the glenoid. Initial damage during birth to the brachial plexus, and its incomplete recovery results in full or partial paralysis of shoulder muscles during the child’s development. The most common muscle imbalances after partial recovery occur between the internal and the external rotators, and the abductors and adductors of the shoulder. The chronic evolution of the muscle imbalance causes changes to the developing bony structures, and formation of scapular and glenohumeral joint deformities.
Lack of finger movement at birth in obstetric brachial plexus injury (OBPI) represents a pejorative sign of prognosis. In these patients, the shoulder muscles are all weakened, and there is no muscle balance, indicating a severe initial injury that mostly affects the entire brachial plexus. However, the presence of finger movement at birth in asymmetrical brachial plexus injury (initial damage to C5-C6 or C5-C7) also predicts the development of severe bony deformities caused by severe muscle imbalance on the growing bony structures of the infant shoulde[1,2]. This progress to a posterior subluxation or complete dislocation of the humeral head. These secondary deformities, including internal rotator and adductor contractures, glenohumeral dysplasia, cause major long-term morbidity requiring surgical correction to improve limb function.
The severity of glenohumeral dysplasia and shoulder function associated with nerve repair in OBPI patients has been recently demonstrated[3]. In this report, we further evaluated the severity of glenohumeral dysplasia in OBPI patients with and without finger movement at birth, and correlated the outcome of primary and secondary surgeries in this patient population.
We conducted a retrospective study reviewing pre-surgical records of 87 patients with residual obstetric brachial plexus palsy. Their ages at the time of computed tomography (CT) or magnetic resonance imaging (MRI) scan were between 4 mo and 16 years (average 4.6 years). All the patients in this study have a CT/MRI of bilateral shoulders prior to any surgical procedure. We compared and correlated the pre-surgical results of posterior subluxation of the humeral head (PHHA), and glenoscapular version angle to the finger movement at birth. In studies that quantify obstetric brachial plexus deformities, the most common measurements are PHHA and glenoid retroversion.
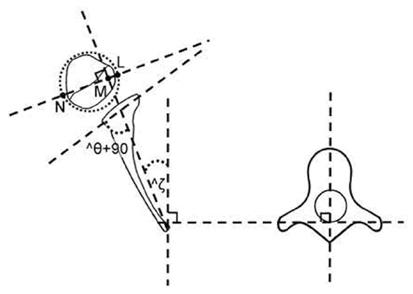
Radiological measurements were taken using patients CT or MRI on the transverse sections at the level of the scapular spine as follows: (1) PHHA[4] calculated as percent humeral head anterior to the scapular line (Figure 1); and (2) Glenoscapular version angle (theta;-angle difference between the glenoid and a line 90° to scapular line), was measured from either CT or MRI scans[5] as previously described[6,7], and in the figure legend (Figure 1).
This study consisted of 141 OBPI patients, who underwent primary surgeries and/or secondary surgery at the Texas Nerve and Paralysis Institute. All the patients in this study were injured severely enough to develop shoulder deformities that required surgical reconstruction. All surgeries were performed by the same surgeon (Nath RK), whose practice has focused on reconstructive surgery in this population for the past 15 years. The age of these patients ranges from 5 mo to 20 years at the time of visit. One group included 50 patients who underwent nerve reconstruction and secondary surgeries (muscle and bony), the second group included 82 patients, who underwent only secondary surgeries (no nerve surgery), and the third group included 9 patients, who have had only bony (triangle tilt) surgery.
Nerve repair, modified Quad, and triangle tilt surgeries were performed on these patients by the senior author and the surgeon (Nath RK) as described previously[8-11]. Information regarding finger movement was obtained from the patient’s parent or guardian during the initial evaluation.
Statistical analysis was performed using Analyse-It plugin (Leeds, United Kingdom) for Microsoft Excel 2003 software. A P-value of < 0.05 was considered as statistically significant.
No finger movement at birth was observed in 56% of the patients. Among 87 patients, 9 (10.3%) patients who lacked finger movement at birth had a PHHA > 40%, and glenoid retroversion < -12°, whereas only 1 patient (1.1%) with finger movement had a PHHA > 40%, and retroversion < -8º.
The improvement in glenohumeral deformity (PHHA, 31.8% ± 14.3%; and glenoid retroversion 22.0% ± 15.0%) was significantly higher in patients, who have not had any primary surgeries and had finger movement at birth (group 1), when compared to those patients, who had primary surgeries (nerve and muscle surgeries) and lacked finger movement at birth (group 2), (PHHA, 10.7% ± 15.8%; Version -8.0% ± 8.4%, P = 0.005 and P = 0.030, respectively).
The change in radiological measurments was not statistically significantly different in patients who have had primary surgeries (data not shown) with reference to finger movement at birth. No finger movement at birth was observed in 55% of the patients in this study group.
The integrity of the motor cortex and the corticospinal tract is critical for the movements of the extremities, and for the control of finger movements[12-17]. Finger movement at birth is an important indication of the functional and anatomical integrity of the brachial plexus.
There are numerous reports in the literature relating finger movements to brain region and brain damage[18-22], stroke[23-25], cerebral palsy[26-28], Parkinson’s disease[29,30], carpal tunnel syndrome[31,32], traumatic injury[21,33-37]. However, there are only few reports correlating finger movements and obstetric brachial plexus injury[2,38] and hand injuries, despite the hands are important in performing daily activities[36].
Finger movement at birth was evaluated as one of the potential risk factors for permanent injury and predictors of future osseous shoulder deformity[2]. Glenoid retroversion was significantly more severe in patients with finger movement at birth, and thus associated with the development of a worse glenohumeral deformity. Posterior subluxation was also more severe in these patients, however not significantly.
Although, the mean radiological scores show that lack of finger movement at birth is actually protective against bony deformities of the shoulder, yet, some patients in this group faced severe bony deformities (up to PHHA-31, and version-16, data not shown). Therefore, these patients also suffer extensive functional impairment that necessitated for surgical treatment.
Permanently injured patients with finger movement at birth develop more severe bony deformities of the shoulder than patients without finger movement at birth due, in part, to asymmetrical muscle action on growing bony elements, also underwent surgical treatment at the Texas Nerve and Paralysis Institute.
The outcome of triangle tilt surgery in terms of radiological scores (PHHA and version) was significantly higher in patients who have not had any primary surgeries and had finger movement at birth (group 1), when compared to patients who had primary surgeries (nerve, nerve and muscle surgeries) and lacked finger movement at birth (group 2). Other investigators have reported that some OBPI patients achieved voluntary finger movement with double free-muscle transfer[38].
Our present study is unique in that it evaluates the relationship between finger movement at birth, and the outcome of the primary and secondary surgeries in OBPI patients. Finger movement at birth, may be used as a simple and rapid clinical test, as a predictor of the outcome. The finger movement data in this study is based on retrospective information which was obtained from patient families. The limitation of this study is that a population of transiently injured patients was not available for comparison. In addition, there are not many reports in the literature to compare the finger movement at birth and the surgical outcome in OBPI patients.
The severity of glenohumeral dysplasia and shoulder function associated with nerve repair in obstetric brachial plexus injury (OBPI) patients has been recently demonstrated. In this report, authors further evaluated the severity of glenohumeral dysplasia in OBPI patients with and without finger movement at birth, and correlated the outcome of primary and secondary surgeries in this patient population.
Finger movement at birth has been evaluated as one of the potential risk factors for permanent brachial plexus injury, and predictors of future osseous shoulder deformity.
Although, there are numerous reports in the literature relating finger movements to brain region and brain damage, stroke, cerebral palsy, Parkinson’s disease, carpal tunnel syndrome, traumatic injury, this is the first report proposing that finger movement may be used as a simple and rapid clinical test, and as a predictor of the surgical outcome in obstetric brachial plexus injury.
Finger movement at birth, may be used as a simple and rapid clinical test, as a predictor of the surgical outcome.
Triangle tilt surgery: This operative technique includes osteotomies of the clavicle, neck of the acromion and scapula in order to release the distal acromioclavicular triangle and allow it to reorient itself in a more neutral position into the glenoid. The modified Quad procedure: Transfer of the latissimus dorsi and teres major muscles, release of contractures of subscapularis pectoralis major and minor and axillary nerve decompression and neurolysis.
The limitation of this study is that a population of transiently injured patients was not available for comparison. In addition, there are not many reports in the literature to compare the finger movement at birth and the surgical outcome in OBPI patients.
P- Reviewer NAS K S- Editor Gou SX L- Editor A E- Editor Zhang DN
| 1. | Troum S, Floyd WE, Waters PM. Posterior dislocation of the humeral head in infancy associated with obstetrical paralysis. A case report. J Bone Joint Surg Am. 1993;75:1370-1375. [PubMed] |
| 2. | Nath RK, Kumar N, Avila MB, Nath DK, Melcher SE, Eichhorn MG, Somasundaram C. Risk factors at birth for permanent obstetric brachial plexus injury and associated osseous deformities. ISRN Pediatr. 2012;2012:307039. [PubMed] |
| 3. | Nath RK, Liu X. Nerve reconstruction in patients with obstetric brachial plexus injury results in worsening of glenohumeral deformity: a case-control study of 75 patients. J Bone Joint Surg Br. 2009;91:649-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Waters PM, Smith GR, Jaramillo D. Glenohumeral deformity secondary to brachial plexus birth palsy. J Bone Joint Surg Am. 1998;80:668-677. [PubMed] |
| 5. | Friedman RJ, Hawthorne KB, Genez BM. The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am. 1992;74:1032-1037. [PubMed] |
| 6. | Nath RK, Humphries AD. Computed tomography of the shoulders in patients with obstetric brachial plexus injuries: a retrospective study. Ann Surg Innov Res. 2008;2:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 7. | Nath RK, Paizi M. Scapular deformity in obstetric brachial plexus palsy: a new finding. Surg Radiol Anat. 2007;29:133-140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Nath RK, Somasundaram C, Melcher SE, Bala M, Wentz MJ. Arm rotated medially with supination - the ARMS variant: description of its surgical correction. BMC Musculoskelet Disord. 2009;10:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 9. | Nath RK. Obstetric brachial plexus injuries-Erb’s palsy: The Nath method of diagnosis and treatment. College Station, TX: VirtualBookworm Publishing 2007; . |
| 10. | Nath RK, Paizi M. Improvement in abduction of the shoulder after reconstructive soft-tissue procedures in obstetric brachial plexus palsy. J Bone Joint Surg Br. 2007;89:620-626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 11. | Nath RK, Amrani A, Melcher SE, Wentz MJ, Paizi M. Surgical normalization of the shoulder joint in obstetric brachial plexus injury. Ann Plast Surg. 2010;65:411-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Bennett KM, Lemon RN. Corticomotoneuronal contribution to the fractionation of muscle activity during precision grip in the monkey. J Neurophysiol. 1996;75:1826-1842. [PubMed] |
| 13. | Maier MA, Bennett KM, Hepp-Reymond MC, Lemon RN. Contribution of the monkey corticomotoneuronal system to the control of force in precision grip. J Neurophysiol. 1993;69:772-785. [PubMed] |
| 14. | Lang CE, Schieber MH. Reduced muscle selectivity during individuated finger movements in humans after damage to the motor cortex or corticospinal tract. J Neurophysiol. 2004;91:1722-1733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 119] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 15. | Lawrence DG, Kuypers HG. The functional organization of the motor system in the monkey. I. The effects of bilateral pyramidal lesions. Brain. 1968;91:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 858] [Cited by in RCA: 808] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 16. | Baker JT, Donoghue JP, Sanes JN. Gaze direction modulates finger movement activation patterns in human cerebral cortex. J Neurosci. 1999;19:10044-10052. [PubMed] |
| 17. | Eyre JA, Miller S, Clowry GJ, Conway EA, Watts C. Functional corticospinal projections are established prenatally in the human foetus permitting involvement in the development of spinal motor centres. Brain. 2000;123:51-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 201] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 18. | Milak MS, Shimansky Y, Bracha V, Bloedel JR. Effects of inactivating individual cerebellar nuclei on the performance and retention of an operantly conditioned forelimb movement. J Neurophysiol. 1997;78:939-959. [PubMed] |
| 19. | Thach WT, Goodkin HP, Keating JG. The cerebellum and the adaptive coordination of movement. Annu Rev Neurosci. 1992;15:403-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 659] [Cited by in RCA: 653] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 20. | Konishi Y, Prechtl HF. Finger movements and fingers postures in pre-term infants are not a good indicator of brain damage. Early Hum Dev. 1994;36:89-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 21. | Prigatano GP, Borgaro SR. Qualitative features of finger movement during the Halstead finger oscillation test following traumatic brain injury. J Int Neuropsychol Soc. 2003;9:128-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Solodkin A, Hlustik P, Noll DC, Small SL. Lateralization of motor circuits and handedness during finger movements. Eur J Neurol. 2001;8:425-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 168] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 23. | Leistner S, Sander T, Wachs M, Burghoff M, Curio G, Trahms L, Mackert BM. Differential infraslow (<0.1 Hz) cortical activations in the affected and unaffected hemispheres from patients with subacute stroke demonstrated by noninvasive DC-magnetoencephalography. Stroke. 2009;40:1683-1686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 24. | Lange G, Waked W, Kirshblum S, DeLuca J. Organizational strategy influence on visual memory performance after stroke: cortical/subcortical and left/right hemisphere contrasts. Arch Phys Med Rehabil. 2000;81:89-94. [PubMed] |
| 25. | Kitamura J, Shabasaki H, Terashi A, Tashima K. Cortical potentials preceding voluntary finger movement in patients with focal cerebellar lesion. Clin Neurophysiol. 1999;110:126-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 26. | Yang H, Einspieler C, Shi W, Marschik PB, Wang Y, Cao Y, Li H, Liao YG, Shao XM. Cerebral palsy in children: movements and postures during early infancy, dependent on preterm vs. full term birth. Early Hum Dev. 2012;88:837-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 48] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 27. | Xu SG, Xu L, Cao X, Jiao Y. [Cervical perivascular sympathectomy for the treatment of athetoid cerebral palsy]. Zhongguo Gu Shang. 2010;23:291-293. [PubMed] |
| 28. | Wilton J. Casting, splinting, and physical and occupational therapy of hand deformity and dysfunction in cerebral palsy. Hand Clin. 2003;19:573-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 29. | Singh A, Kammermeier S, Mehrkens JH, Bötzel K. Movement kinematic after deep brain stimulation associated microlesions. J Neurol Neurosurg Psychiatry. 2012;83:1022-1026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 30. | Vanbellingen T, Kersten B, Bellion M, Temperli P, Baronti F, Müri R, Bohlhalter S. Impaired finger dexterity in Parkinson’s disease is associated with praxis function. Brain Cogn. 2011;77:48-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 31. | Lin YC, Chen CH, Fu YC, Lin GT, Chang JK, Hu ST. Carpal tunnel syndrome and finger movement dysfunction caused by tophaceous gout: a case report. Kaohsiung J Med Sci. 2009;25:34-39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 32. | Gehrmann S, Tang J, Kaufmann RA, Goitz RJ, Windolf J, Li ZM. Variability of precision pinch movements caused by carpal tunnel syndrome. J Hand Surg Am. 2008;33:1069-1075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 33. | Yii NW, Urban M, Elliot D. A prospective study of flexor tendon repair in zone 5. J Hand Surg Br. 1998;23:642-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 34. | Mano Y, Chuma T, Watanabe I. Cortical reorganization in training. J Electromyogr Kinesiol. 2003;13:57-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Sawant N, Kulikov Y, Giddins GE. Outcome following conservative treatment of metacarpophalangeal collateral ligament avulsion fractures of the finger. J Hand Surg Eur Vol. 2007;32:102-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 36. | Yildirim A, Nas K. Evaluation of postoperative early mobilization in patients with repaired flexor tendons of the wrist, the spaghetti wrist. J Back Musculoskelet Rehabil. 2010;23:193-200. [PubMed] |
| 37. | Werdin F, Schaller HE. [Combined flexor tendon and nerve injury of the hand]. Orthopade. 2008;37:1202-1209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 38. | Doi K, Hattori Y, Kuwata N, Soo-heong T, Kawakami F, Otsuka K, Watanabe M. Free muscle transfer can restore hand function after injuries of the lower brachial plexus. J Bone Joint Surg Br. 1998;80:117-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (0)] |