Published online Nov 18, 2011. doi: 10.5312/wjo.v2.i11.102
Revised: October 2, 2011
Accepted: October 9, 2011
Published online: November 18, 2011
In peripheral nerve injury, end-to-side neurorrhaphy involves coaptation of the distal stump of a transected nerve to the trunk of an adjacent donor nerve. It has been proposed as an alternative technique when the proximal stump of an injured nerve is unavailable or the nerve gap is too long to be bridged by a nerve graft. Experimental and clinical data suggests that end-to-side neurorrhaphy can provide satisfactory functional recovery for the recipient nerve, without any deterioration of the donor nerve function. The most accepted mechanism of nerve regeneration following end-to-side neurorrhaphy is collateral sprouting. The source of the regenerating axons traveling in the epineurium of the donor nerve is thought to be the proximal Ranvier’s nodes at the site of end-to-side neurorrhaphy, however, histologic evidence is still lacking. Partial neurotomy of the donor nerve may enhance regeneration of motor neurons through end-to-side neurorrhaphy and reinnervation of motor targets.
- Citation: Lykissas MG. Current concepts in end-to-side neurorrhaphy. World J Orthop 2011; 2(11): 102-106
- URL: https://www.wjgnet.com/2218-5836/full/v2/i11/102.htm
- DOI: https://dx.doi.org/10.5312/wjo.v2.i11.102
Autologous nerve grafting remains the gold standard for the management of nerve gaps following peripheral nerve injury. Use of autologous nerve grafts is bounded by the limited amount of available tissue and the increased donor site morbidity. Several surgical alternatives have been reported with various success. These include the combination of nerve grafts and silicon tubes[1], the use of synthetic or biologic nerve conduits[2], tubes containing blood vessels[3],the application of cultured Schwann cells[4] and end-to-side neurorrhaphy.
It was not until 1992, when Viterbo et al[5] reintroduced end-to-side neurorrhaphy, an almost forgotten technique of nerve coaptation. End-to-side neurorrhaphy involves coaptation of the distal stump of a transected nerve to the trunk of an adjacent donor nerve. It has been proposed as an alternative technique in cases of peripheral nerve injury, when the proximal stump of an injured nerve is unavailable or obliterated or the nerve gap is too long to be bridged by a nerve graft[6-8].
End-to-side neurorrhaphy was first described by Letievant in 1873 as a reconstructive strategy of peripheral nerves in cases of large substance loss[9]. The pioneer’s idea was abandoned due to poor results that can be attributed to the use of conventional surgical instruments and non-microsurgical techniques without the use of a microscope. Almost nine decades later, many investigators have again experimented using this interesting technique[5,10-15]. The results this time were very promising and since then, many studies have been performed resulting in improvement of functional results and further understanding of nerve regeneration after end-to-side neurorrhaphy.
The most accepted mechanism of nerve regeneration following end-to-side neurorrhaphy is collateral sprouting, where regenerated axons emerge from the most proximal Ranvier’s node of the donor nerve to the coaptation site and travel in the epineurium of the donor nerve[16-25]. Before axonal development, schwann cells are organized into columns at the coaptation site[26]. At a later stage, these cells invade the epineurial layer of the recipient nerve. This is considered the critical step for the initiation of collateral axonal sprouting from the intact axons. It is supported that axons emerge from the Ranvier’s nodes of the donor nerve proximal to the coaptation site[25,27-29]. According to one study, Schwann cells were found to stimulate axonal regeneration from both the distal nerve stump and Ranvier’s nodes of the donor nerve[30].
The mechanism causing collateral sprouting after end-to-side neurorrhaphy may result from switching signals and/or switching factors, presumably neurotrophic[19]. Zhang et al[19] suggested that factors released from the Schwann cells, which have migrated to the epineurium, are transferred into the perineurium by diffusion and promote collateral sprouting from the closest to the injury site to Ranvier’s nodes of the donor nerve.
It is well known that Neurotrophine-3 (NT-3) plays a distinct role in the processes of nerve regeneration and muscle reinnervation[25]. NT-3 and its receptor Trk C are expressed in the coaptation site following end-to-side neurorrhaphy[31]. Growth-associated protein-43 (GAP-43), a marker of growth cone formation, brain-derived neurotrophic factor (BDNF) and Trk B (BDNF receptor) are also detected in the coaptation site in lower concentrations and after NT-3 expression[31]. In an end-to-side neurorrhaphy model using anti-GAP-43 antibody, growth cone direction was recorded from the donor nerve to the peripheral nerve segment of the injured nerve.
Many investigators have also shown the distinct role of nerve growth factor (NGF) during collateral sprouting[32-37]. NGF is produced in end-organs following nerve injury. The secreted NGF is taken up by the axon terminals and transported retrogradely to the nerve cell body stimulating a secondary response. It has been shown that the combination of NGF and ciliary neurotrophic factor (CNTF) promotes axonal regeneration after end-to-side neurorrhaphy[38].
Biological responses of the donor neuron to factors emanating from the transected nerve have been implicated in the initiation of collateral sprouting for both sensory and motor axons. According to previous studies, significant motor functional recovery after end-to-side neurorrhaphy can be achieved without donor nerve axotomy[39,40]. However, more recent studies suggest that donor nerve injury, such as axotomy or suturing, is required for motor reinnervation of the recipient nerve[41,42].
Bontioti et al[41] revealed increased expression of activating transcription factor 3 (ATF3), a marker of cell activation induced in sensory and motor neurons following peripheral nerve injury, after the creation of an epineurial window and/or suturing. According to these findings, an operative injury to the donor nerve during end-to-side neurorrhaphy is the main prerequisite for axonal sprouting.
A dose-response relationship between axotomy of the donor nerve and motor axons regeneration has been demonstrated[42]. Presumably, motor fibers from the donor nerve may enter the recipient nerve segment to supply muscles which were normally innervated by motor fibers from the recipient nerve[43].
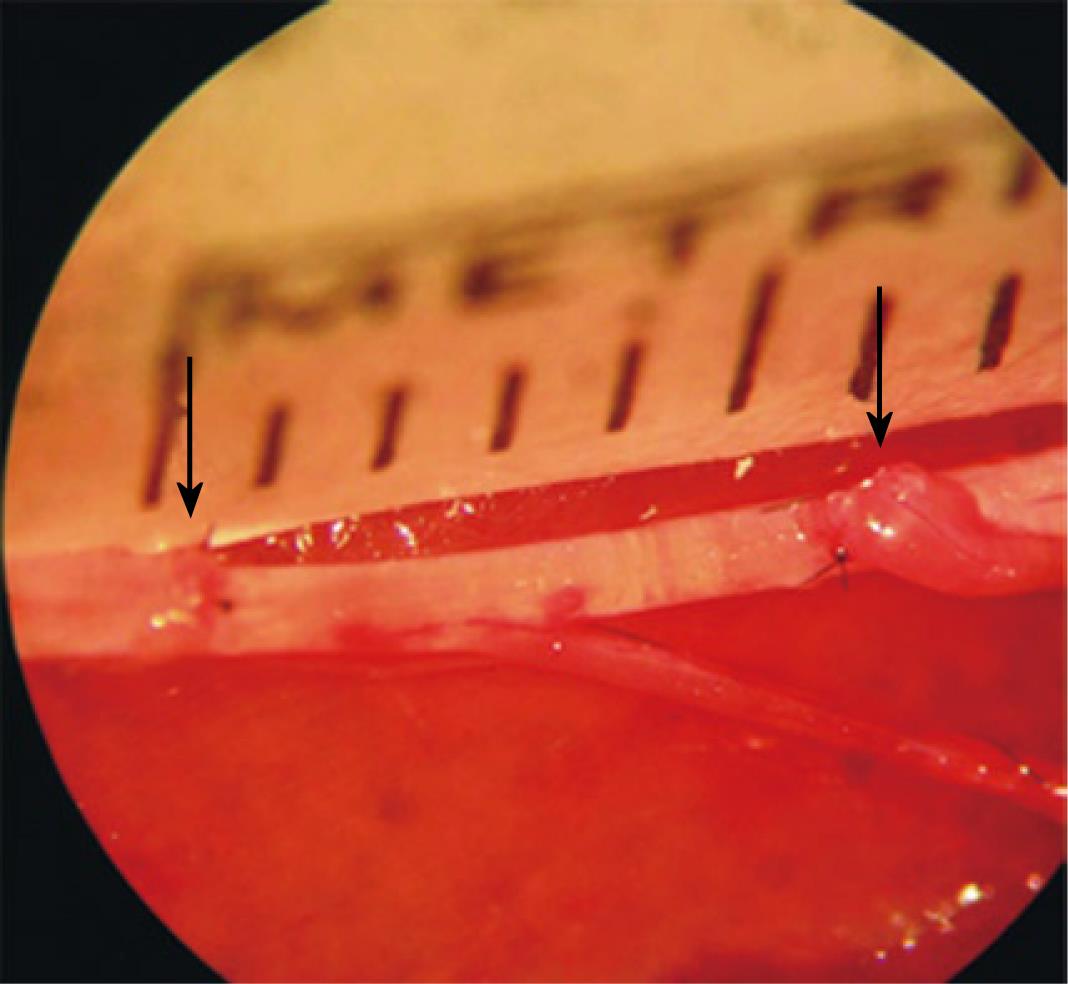
Viterbo et al[13] first described double end-to-side neurorrhaphy. In this technique, both proximal and distal stumps of the recipient nerve are coapted in an end-to-side fashion to the trunk of an adjacent donor nerve (Figure 1). The regenerated axons use the epineurium of the donor nerve as a bridge to find the distal stump. It has been suggested that this technique stimulates axonal growth by a supercharged effect compared with end-to-end repair. Interestingly, when double end-to-side neurorrhaphy was compared with the conventional end-to-side technique, the recipient nerve following the double terminolateral technique was found to contain a significantly larger number of myelinated nerve fibers distal to the neurorrhaphy site[44]. Two sources of axons may contribute to the increased number of regenerating nerve fibers, axons sprouted collaterally from myelinated nerve fibers at the node of Ranvier of the donor nerve, and axons that arise from the proximally coapted nerve segment.
Our experimental knowledge of double end-to-side neurorrhaphy, leads us to the belief that double end-to-side coaptation may be a valuable tool when the classic end-to-end technique is not possible. In our previous studies in rats, functional evaluation and axonal counting data demonstrated that nerve regeneration can be supported using the intact nerve bridge technique for a distance of 1.2 cm in a rat sciatic model[44].
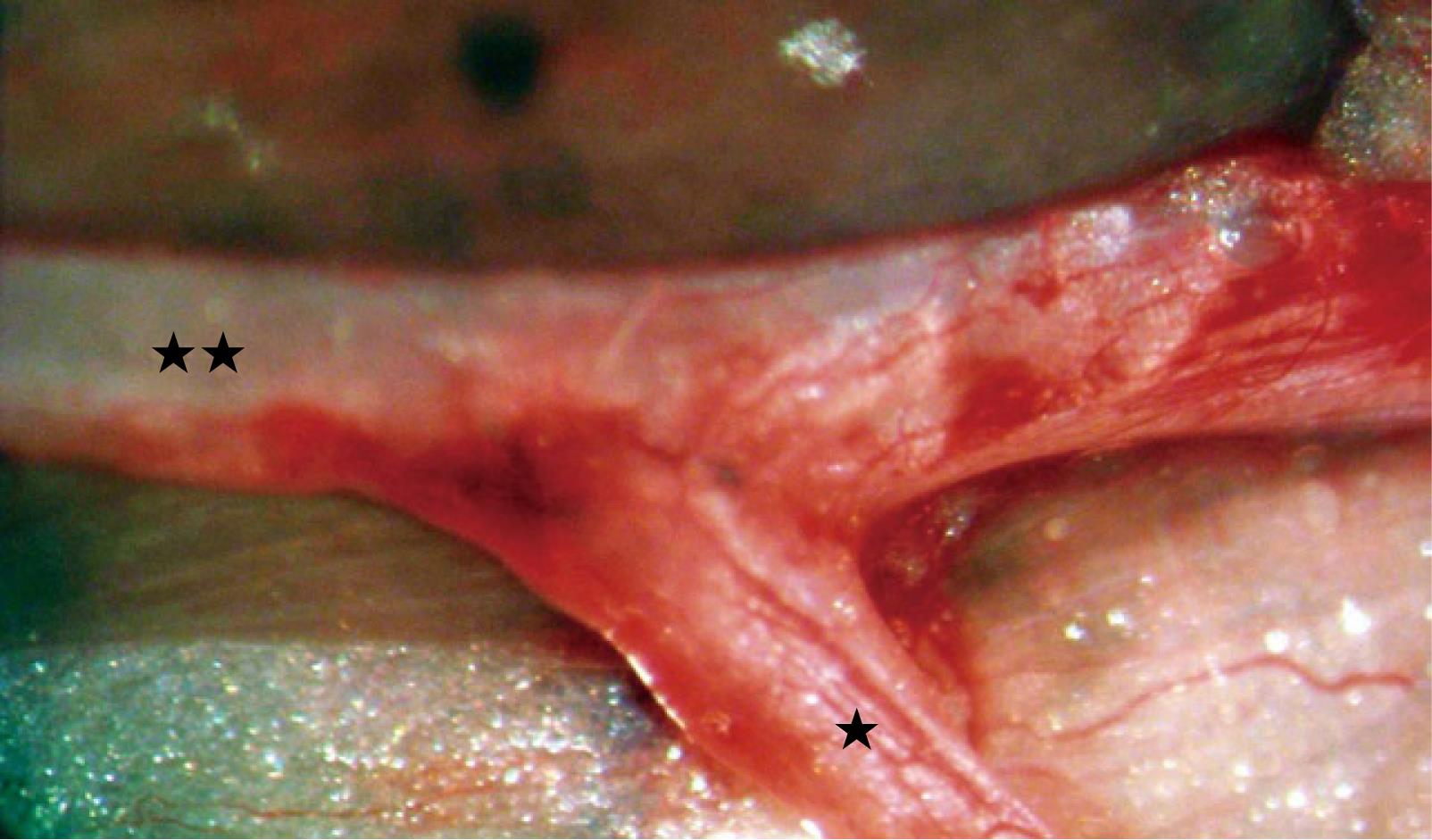
A technical parameter that may significantly affect axonal regeneration after end-to-side neurorrhaphy involves the application of epineurotomy or perineurotomy. Viterbo[5] and Cao[16] demonstrated no significant difference for end-to-side neurorrhaphy with and without epineurial window. Likewise, Viterbo et al[45] revealed no difference between neurorrhaphies with and without perineurial window. These observations may, in part, be explained by the finding that the regenerating axons following end-to-side neurorrhaphy can penetrate the endoneurium, perineurium, and epineurium[17].
According to some investigators, histologic results were better when a perineurial window was opened[19,20]. This can be attributed to the greater degree of axonal damage to the donor nerve and subsequently the enhanced axonal regeneration after perineurotomy. When fibrin glue is used as an alternative to end-to-side neurorrhaphy, no damage to the donor nerve trunk is produced. This may explain the absence of muscle reinnervation after end-to-side coaptation with fibrin glue, without removing the epineurium[46]. According to our studies, resection of a small part of the epineurium and placement of epineurial sutures without damaging the underlying perineurium improves the functional outcomes following terminolateral nerve repair without compromising the function of the donor nerve (Figure 2)[44].
To date, there have been no large clinical series describing either satisfactory or disappointing results after end-to-side neurorrhaphy. In 1993, Viterbo[11] first applied end-to-side neurorrhaphy in recent clinical practice with the use of cross-facial nerve graft transplantation for the treatment of facial palsy. Reinnervation was observed in selected patients. A few years later, end-to-side neurorrhaphy was used to bridge the nerve gap after ulnar nerve injury. In this case, the median nerve was the donor nerve[47]. The authors reported ulnar nerve motor and sensory restoration without deterioration of the donor nerve. Yüksel et al[48] described a case of severe upper extremity nerve injury treated with end-to-side neurorrhaphy of the median and radial nerves to the ulnar nerve. The patient had satisfactory sensory recovery.
Amr et al[49] reported satisfactory results in 11 cases of brachial palsy injury treated with end-to-side and side-to-side grafting neurorrhaphy. Deterioration in donor muscle motor power was observed in one case, which improved a year later. Santamaria et al[50] sutured the lateral antebrachial cutaneous nerve in an end-to-side fashion to the cervical plexus, posterior auricular, or hypoglossal nerve where it was not possible to preserve the proximal stump of the lingual nerve in twenty-eight patients with tongue cancer who underwent hemiglossectomy and primary reconstruction with innervated radial forearm flaps. Sensory tests were significantly diminished when end-to-side nerve repair was used.
In digital nerve reconstruction, of 5 patients who underwent end-to-side neurorrhaphy four had sensitivity near to completely normal and one patient had a poor result[51]. Satisfactory results in all patients were also obtained in a series of ten nerve defects at the palm or digit level treated by end-to-side neurorrhaphy. Donor nerve injury was recorded in one case[52].
Experimental and clinical studies suggest that end-to-side neurorrhaphy can provide satisfactory functional recovery in the recipient nerve, without any deterioration of donor nerve function. The source of the regenerating axons traveling in the epineurium of the donor nerve is thought to be the proximal Ranvier’s nodes at the site of end-to-side neurorrhaphy, however, histologic evidence is still lacking. Partial neurotomy of the donor nerve may enhance regeneration of motor neurons through end-to-side neurorrhaphy and reinnervation of motor targets. To date, a limited number of reported cases in clinical practice have revealed that the end-to-side technique may become a viable means of repairing peripheral nerves in certain clinical situations.
Peer reviewers: R Shane Tubbs, PhD, Professor of Anatomy, Pediatric Neurosurgery, Children’s Hospital, 1600 7th Avenue South ACC 400, Birmingham, AL 35233, United States; Shuichi Kaneyama, MD, PhD, Department of Opthopaedic Surgery, Kobe Rosai Hospital. 4-1-23, Kagoike-dori, Chuo-ku, Kobe 6510053, Japan; Juan A Pretell-Mazzini, MD, Division of Orthopaedic Surgery, The Children´s Hospital of Philadelphia, 34th Street and Civic Center Boulevard-Wood Building 2nd Floor, Philadelphia, PA 19104, United States
S- Editor Yang XC L- Editor Webster JR E- Editor Yang XC
| 1. | Saito I, Oka Y, Odaka M. Promoting nerve regeneration through long gaps using a small nerve tissue graft. Surg Neurol. 2003;59:148-54; discussion 154-5. |
| 2. | Mackinnon SE, Dellon AL. A study of nerve regeneration across synthetic (Maxon) and biologic (collagen) nerve conduits for nerve gaps up to 5 cm in the primate. J Reconstr Microsurg. 1990;6:117-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 131] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 3. | Kakinoki R, Nishijima N, Ueba Y, Oka M, Yamamuro T, Nakamura T. Nerve regeneration over a 25 mm gap in rat sciatic nerves using tubes containing blood vessels: the possibility of clinical application. Int Orthop. 1997;21:332-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Fansa H, Dodic T, Wolf G, Schneider W, Keilhoff G. Tissue engineering of peripheral nerves: Epineurial grafts with application of cultured Schwann cells. Microsurgery. 2003;23:72-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | Viterbo F, Trindade JC, Hoshino K, Mazzoni Neto A. Latero-terminal neurorrhaphy without removal of the epineural sheath. Experimental study in rats. Rev Paul Med. 1992;110:267-275. [PubMed] |
| 6. | Ballance CA, Ballance HA, Stewart P. REMARKS on the OPERATIVE TREATMENT of CHRONIC FACIAL PALSY of PERIPHERAL ORIGIN. Br Med J. 1903;1:1009-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 111] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 7. | Harris W, Low VW. On the importance of accurate muscular analysis in lesions of the brachial plexus, and the treatment of Erb’s palsy and infantile paralysis of the upper extremity by cross-union of the nerve roots. BMJ. 1903;24:1035-1038. |
| 8. | Sherren J. Some points in the surgery of the peripheral nerves. Edinb Med J. 1906;20:297-332. |
| 9. | Le tie vant E. Traite des Sections Nerveuses. J.B. Baillie`re et Fils. 1873;. |
| 10. | Krivolutskaia EG, Chumasov EI, Matina VN, Mel’tsova GM, Kirillov AL. [End-to-side type of plastic repair of the facial nerve branches]. Stomatologiia (Mosk). 1989;68:35-38. [PubMed] |
| 11. | Viterbo F. A new method for treatment of facial palsy: the Cross-Face Nerve transplantation with end-to-side neurorraphy. Rev Soc Bras Cir Plast Estet Reconstr. 1993;8:29-38. |
| 12. | Viterbo F, Trindade JC, Hoshino K, Mazzoni Neto A. End-to-side neurorrhaphy with removal of the epineurial sheath: an experimental study in rats. Plast Reconstr Surg. 1994;94:1038-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 172] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 13. | Viterbo F, Trindade JC, Hoshino K, Mazzoni A. Two end-to-side neurorrhaphies and nerve graft with removal of the epineural sheath: experimental study in rats. Br J Plast Surg. 1994;47:75-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 142] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 14. | Ross DA, Matsuda H, Zuker RM. End-to-side nerve coaptation for muscle reinnervation. Transactions of the 11th Congress of the International Confederation of Plastic, Reconstructive and Aesthetic Surgery. Yokohama: [publisher unknown]; 1995; p4. |
| 15. | Lundborg G, Zhao Q, Kanje M, Danielsen N, Kerns JM. Can sensory and motor collateral sprouting be induced from intact peripheral nerve by end-to-side anastomosis? J Hand Surg Br. 1994;19:277-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 154] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 16. | Cao X, Shidao H, Yu J. [Experimental study on the collateral sprouting after end-to-side anastomosis of nerve trunk]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 1997;11:321-324. [PubMed] |
| 17. | Zhao JZ, Chen ZW, Chen TY. Nerve regeneration after terminolateral neurorrhaphy: experimental study in rats. J Reconstr Microsurg. 1997;13:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 83] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Tham SK, Morrison WA. Motor collateral sprouting through an end-to-side nerve repair. J Hand Surg Am. 1998;23:844-851. [PubMed] |
| 19. | Zhang Z, Soucacos PN, Beris AE, Bo J, Ioachim E, Johnson EO. Long-term evaluation of rat peripheral nerve repair with end-to-side neurorrhaphy. J Reconstr Microsurg. 2000;16:303-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 50] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Zhang Z, Soucacos PN, Bo J, Beris AE, Malizos KN, Ioachim E, Agnantis NJ. Reinnervation after end-to-side nerve coaptation in a rat model. Am J Orthop (Belle Mead NJ). 2001;30:400-46; discussion 407. [PubMed] |
| 21. | Hayashi A, Yanai A, Komuro Y, Nishida M, Inoue M, Seki T. Collateral sprouting occurs following end-to-side neurorrhaphy. Plast Reconstr Surg. 2004;114:129-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Lutz BS, Chuang DCC, Hsu JC, Wei FC. End-to-side neurorraphy: fuctional and double-labeling study in rat upper limb. J Reconstr Microsurg. 1998;14:590. |
| 23. | Andreopoulos GK, Skoulis TG, Terzis JK. Double labeling technique to trace axonal sprouting after end-to-side neurorrhaphy. Abstract presented at the 43rd annual meeting of the Plastic Surgery Research Council. Loma Linda: [publisher unknown] 1998; . |
| 24. | Zhang Z, Soucacos PN, Bo J, Beris AE. Evaluation of collateral sprouting after end-to-side nerve coaptation using a fluorescent double-labeling technique. Microsurgery. 1999;19:281-286. [PubMed] |
| 25. | Sterne GD, Coulton GR, Brown RA, Green CJ, Terenghi G. Neurotrophin-3-enhanced nerve regeneration selectively improves recovery of muscle fibers expressing myosin heavy chains 2b. J Cell Biol. 1997;139:709-715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 69] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 26. | Slack JR, Hopkins WG, Williams MN. Nerve sheaths and motoneurone collateral sprouting. Nature. 1979;282:506-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 51] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Buehler MJ, Seaber AV, Urbaniak JR. The relationship of functional return to varying methods of nerve repair. J Reconstr Microsurg. 1990;6:61-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 28. | EDDS MV. Collateral nerve regeneration. Q Rev Biol. 1953;28:260-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 232] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 29. | Torigoe K, Tanaka HG, Takahashi A, Hashimoto K. Early growth of regenerating neurites in acrylamide neuropathic mice: application of a film model. Brain Res. 1997;746:269-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 30. | Tarasidis G, Watanabe O, Mackinnon SE, Strasberg SR, Haughey BH, Hunter DA. End-to-side neurorrhaphy resulting in limited sensory axonal regeneration in a rat model. Ann Otol Rhinol Laryngol. 1997;106:506-512. [PubMed] |
| 31. | Yamauchi T, Maeda M, Tamai S, Tamai M, Yajima H, Takakura Y, Haga S, Yamamoto H. Collateral sprouting mechanism after end-to-side nerve repair in the rat. Med Electron Microsc. 2000;33:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 32. | Diamond J, Holmes M, Coughlin M. Endogenous NGF and nerve impulses regulate the collateral sprouting of sensory axons in the skin of the adult rat. J Neurosci. 1992;12:1454-1466. [PubMed] |
| 33. | Doubleday B, Robinson PP. The role of nerve growth factor in collateral reinnervation by cutaneous C-fibres in the rat. Brain Res. 1992;593:179-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 34. | Owen DJ, Logan A, Robinson PP. A role for nerve growth factor in collateral reinnervation from sensory nerves in the guinea pig. Brain Res. 1989;476:248-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 35. | Sebert ME, Shooter EM. Expression of mRNA for neurotrophic factors and their receptors in the rat dorsal root ganglion and sciatic nerve following nerve injury. J Neurosci Res. 1993;36:357-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 145] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 36. | Shen H, Chung JM, Chung K. Expression of neurotrophin mRNAs in the dorsal root ganglion after spinal nerve injury. Brain Res Mol Brain Res. 1999;64:186-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 55] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 37. | Bajrović F, Kovacic U, Pavcnik M, Sketelj J. Interneuronal signalling is involved in induction of collateral sprouting of nociceptive axons. Neuroscience. 2002;111:587-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 38. | McCallister WV, Tang P, Smith J, Trumble TE. Axonal regeneration stimulated by the combination of nerve growth factor and ciliary neurotrophic factor in an end-to-side model. J Hand Surg Am. 2001;26:478-488. [PubMed] |
| 39. | Matsuda H, Hata Y, Matsuka K, Ito O, Battal NM. Experimental study of nerve regeneration in end-to-side nerve coaptation. J Jpn Plast Reconstr Surg. 1995;15:910-918. |
| 40. | Battal MN, Hata Y, Matsuka K, Ito O, Matsuda H. Cross-facial nerve grafting by end-to-side neurorrhaphy with or without removal of the epineurium in rats. J Jpn Plast Reconstr Surg. 1996;16:641-647. |
| 41. | Bontioti E, Dahlin LB, Kataoka K, Kanje M. End-to-side nerve repair induces nuclear translocation of activating transcription factor 3. Scand J Plast Reconstr Surg Hand Surg. 2006;40:321-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 42. | Brenner MJ, Dvali L, Hunter DA, Myckatyn TM, Mackinnon SE. Motor neuron regeneration through end-to-side repairs is a function of donor nerve axotomy. Plast Reconstr Surg. 2007;120:215-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 49] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 43. | Bontioti E, Kanje M, Lundborg G, Dahlin LB. End-to-side nerve repair in the upper extremity of rat. J Peripher Nerv Syst. 2005;10:58-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 68] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 44. | Lykissas MG, Korompilias AV, Batistatou AK, Mitsionis GI, Beris AE. Can end-to-side neurorrhaphy bridge large defects? An experimental study in rats. Muscle Nerve. 2007;36:664-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 45. | Viterbo F, Teixeira E, Hoshino K, Padovani CR. End-to-side neurorrhaphy with and without perineurium. Sao Paulo Med J. 1998;116:1808-1814. [PubMed] |
| 46. | Bertelli JA, dos Santos AR, Calixto JB. Is axonal sprouting able to traverse the conjunctival layers of the peripheral nerve? A behavioral, motor, and sensory study of end-to-side nerve anastomosis. J Reconstr Microsurg. 1996;12:559-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 62] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 47. | Luo Y, Wang T, Fang H. [Preliminary investigation of treatment of ulnar nerve defect by end-to-side neurorrhaphy]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 1997;11:338-339. [PubMed] |
| 48. | Yüksel F, Peker F, Celiköz B. Two applications of end-to-side nerve neurorrhaphy in severe upper-extremity nerve injuries. Microsurgery. 2004;24:363-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 49. | Amr SM, Moharram AN. Repair of brachial plexus lesions by end-to-side side-to-side grafting neurorrhaphy: experience based on 11 cases. Microsurgery. 2005;25:126-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 50. | Santamaria E, Wei FC, Chen IH, Chuang DC. Sensation recovery on innervated radial forearm flap for hemiglossectomy reconstruction by using different recipient nerves. Plast Reconstr Surg. 1999;103:450-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 63] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 51. | Mennen U. End-to-side nerve suture in clinical practice. Hand Surg. 2003;8:33-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 57] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 52. | Voche P, Ouattara D. End-to-side neurorrhaphy for defects of palmar sensory digital nerves. Br J Plast Surg. 2005;58:239-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |