Published online Sep 18, 2021. doi: 10.5312/wjo.v12.i9.720
Peer-review started: April 1, 2021
First decision: June 7, 2021
Revised: June 19, 2021
Accepted: August 20, 2021
Article in press: August 20, 2021
Published online: September 18, 2021
Processing time: 166 Days and 5.2 Hours
Osteochondromas are the most common benign bone tumor, accounting for 36% of benign bone tumors. Often found within the appendicular skeleton, osteochondromas of the spine are rare, comprising 4% to 7% of primary benign spinal tumors.
We report a case of a solitary lumbar osteochondroma in an 18-year-old male with a history of a suprasellar pineal germinoma treated with combined chemotherapy and radiation. He underwent mass excision and partial laminectomy with the ultrasonic bone scalpel (Misonix, Farmingdale, NY, United States) at the L5 Level without the use of adjuvants. The patient returned to work and full activities without back pain at 3 mo postoperatively.
Osteochondromas are common tumors of the appendicular skeleton but rarely occur within the spine. This case discussion supplements current osteochondroma literature by describing an unusual presentation of this tumor.
Core Tip: Osteochondromas of the spine are a rare but treatable condition. For symptomatic lesions, complete resection is largely curative without adjuvant therapy. The patient in this case report was pain free at his post-operative visits without signs or symptoms of recurrence or complication. He returned to work as a manual laborer at 3 mo. Further reports of patients diagnosed with osteochondromas and a history of childhood radiation will enable better understanding of radiation-induced osteochondromas and the rates and locations at which they occur.
- Citation: Suwak P, Barnett SA, Song BM, Heffernan MJ. Atypical osteochondroma of the lumbar spine associated with suprasellar pineal germinoma: A case report . World J Orthop 2021; 12(9): 720-726
- URL: https://www.wjgnet.com/2218-5836/full/v12/i9/720.htm
- DOI: https://dx.doi.org/10.5312/wjo.v12.i9.720
Osteochondromas are the most common benign bone tumor, accounting for 36% of benign bone tumors[1]. Often found within the appendicular skeleton, osteochondromas of the spine are rare, comprising 4% to 7% of primary benign spinal tumors[2,3]. Spine osteochondromas typically originate in the posterior elements as opposed to the vertebral bodies. The cervical spine is most affected (50%), followed by the thoracic and lumbar regions[4]. Characterized as outgrowths of bone covered with a cartila
Radiation-induced osteochondroma is a known phenomenon that occurs in patients with a childhood history of radiation therapy[8]. Compared to spontaneous solitary osteochondromas and hereditary multiple exostosis, radiation-induced lesions are radiologically and pathologically identical[8,9]. The presence of a radiation-induced osteochondroma in the lumbar spine is extremely unusual, as in our presented case. A review of literature reveals only two cases of radiation induced osteochondroma occurring in the lumbar spine (one occurring in the L1 body of a 29-year-old female and the L3 spinous process of a 15-year-old female[10-12]. The case of the 15-year-old female appeared in a larger childhood post-radiative tumor study and was not formally presented. Difficulty in diagnosis stems from inconsistent visualization on plain radiographs. Computed tomography (CT) and magnetic resonance imaging (MRI) are the gold standard in the evaluation of spinal osteochondromas, especially in the setting of spinal cord compressive symptoms[13,14].
We present an atypical, symptomatic lumbar spine osteochondroma in an 18-year-old male with a history of chemotherapy and radiation. This case report received institutional review board approval.
An 18-year-old-male presented to the pediatric orthopaedic clinic with the complaint of a posterior midline protuberance at the level of the lumbar spine.
Per the patient, the mass had been present for one year and was gradually enlarging. He noted pain with prolonged sitting and while lying supine.
There was no history of prior trauma or activity-related injury to his low back, however the patient worked as a manual laborer. The patient denied constitutional or neurologic symptoms. His history was significant for a suprasellar pineal germinoma successfully treated with chemotherapy and radiation at 9-years of age.
No family history of hereditary multiple exostosis was reported.
On physical examination, there was a palpable osseous protuberance approximately 5 cm in width along the posterior midline at the level of the lumbar spine. The mass was fixed and exquisitely tender to palpation. Further examination of the body and extremities revealed no other masses. Neurologic testing was benign with normal sensorimotor response in all extremities, symmetric reflexes, no upper motor neuron or radicular signs, and no balance or gait abnormalities.
No laboratory examinations were performed.
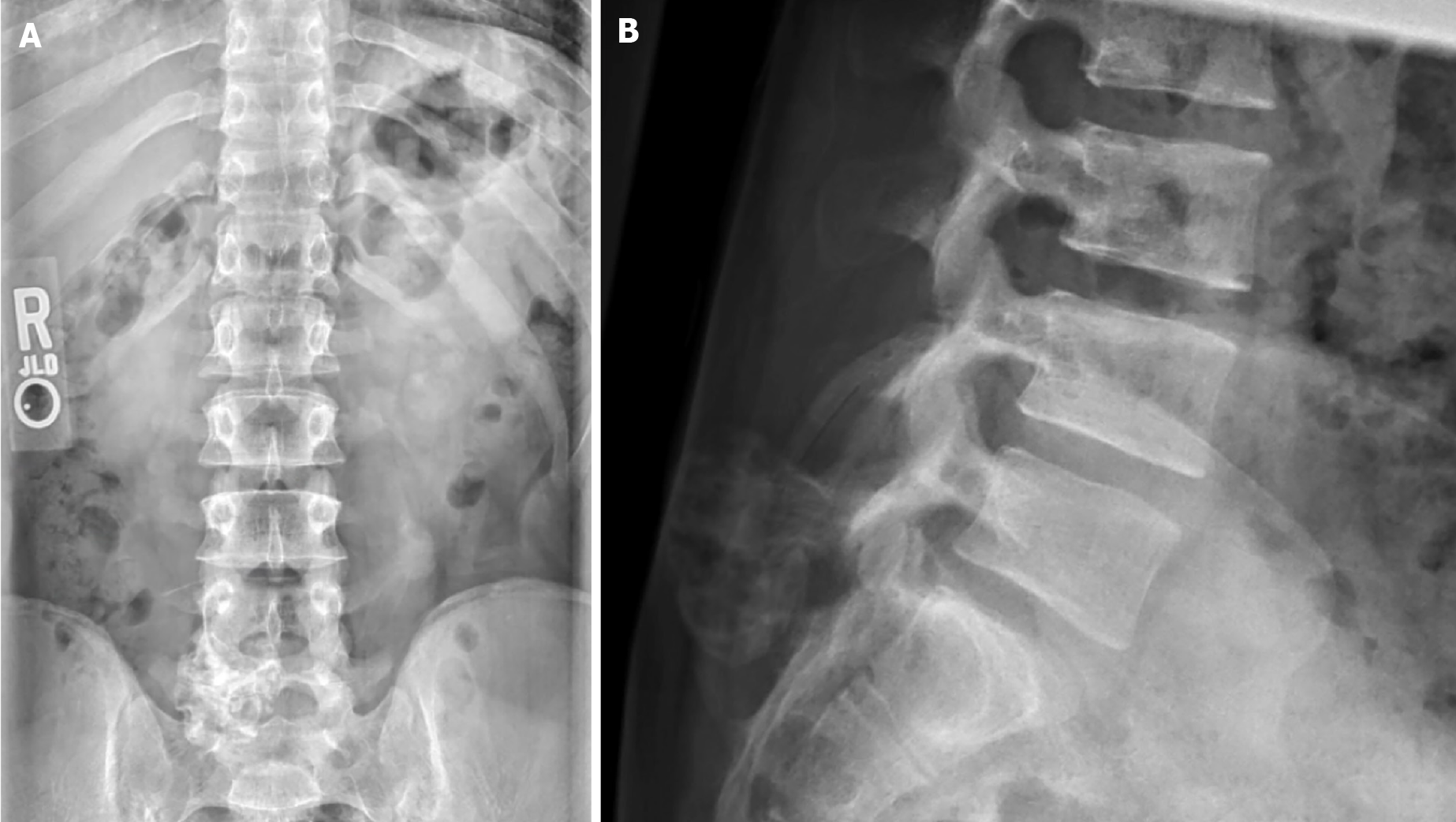
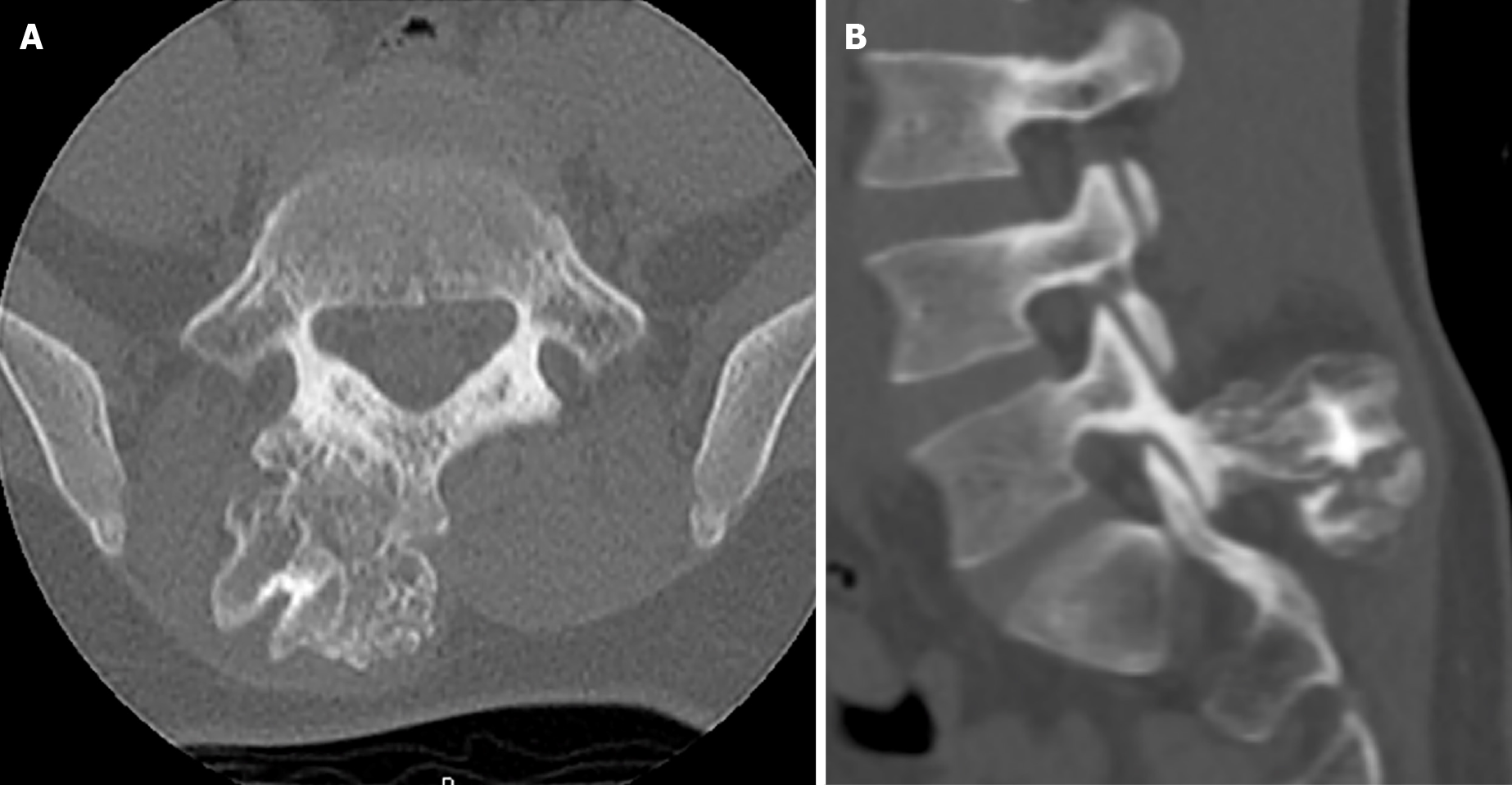
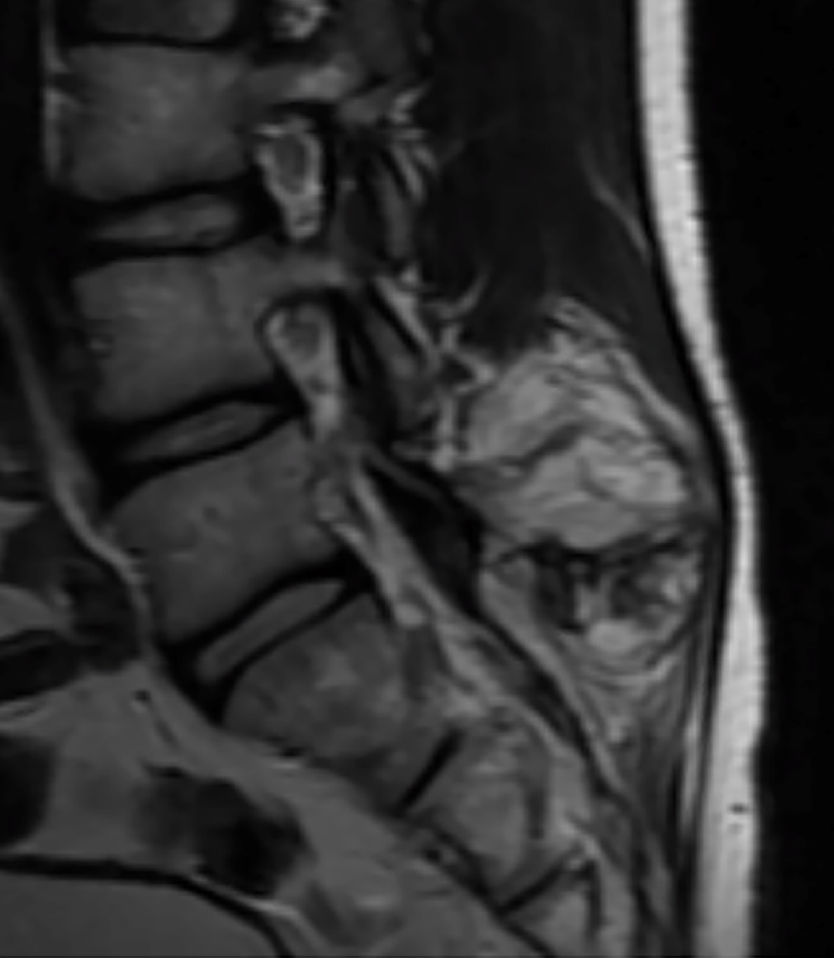
Initial radiographic evaluation demonstrated a large osteochondroma arising from the right spinous process/Lamina junction at the level of the L5 vertebral body (Figure 1). Subsequent CT and MRI imaging confirmed the radiographic findings without evidence of intraspinal abnormality or encroachment on the vertebral canal (Figures 2 and 3). Given the symptomatic nature and location of the mass, the patient was offered surgical treatment in the form of mass excision.
A diagnosis of an osteochondroma arising from the right spinous process/Lamina junction at the level of the L5 vertebral body was made, and this was later confirmed with surgical pathology.
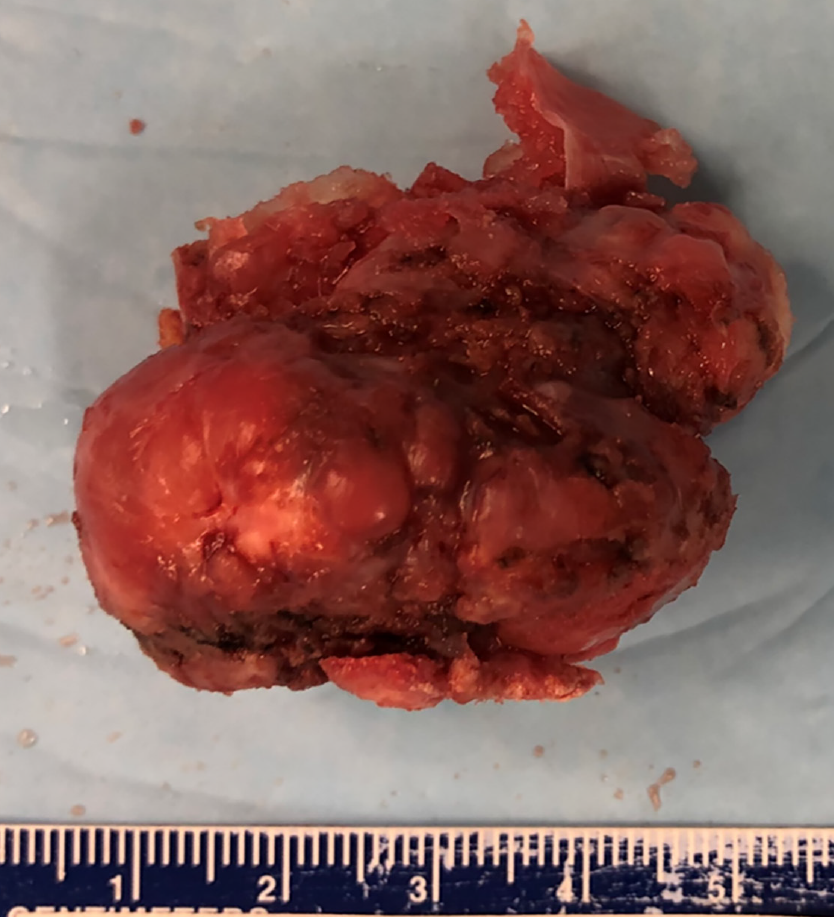
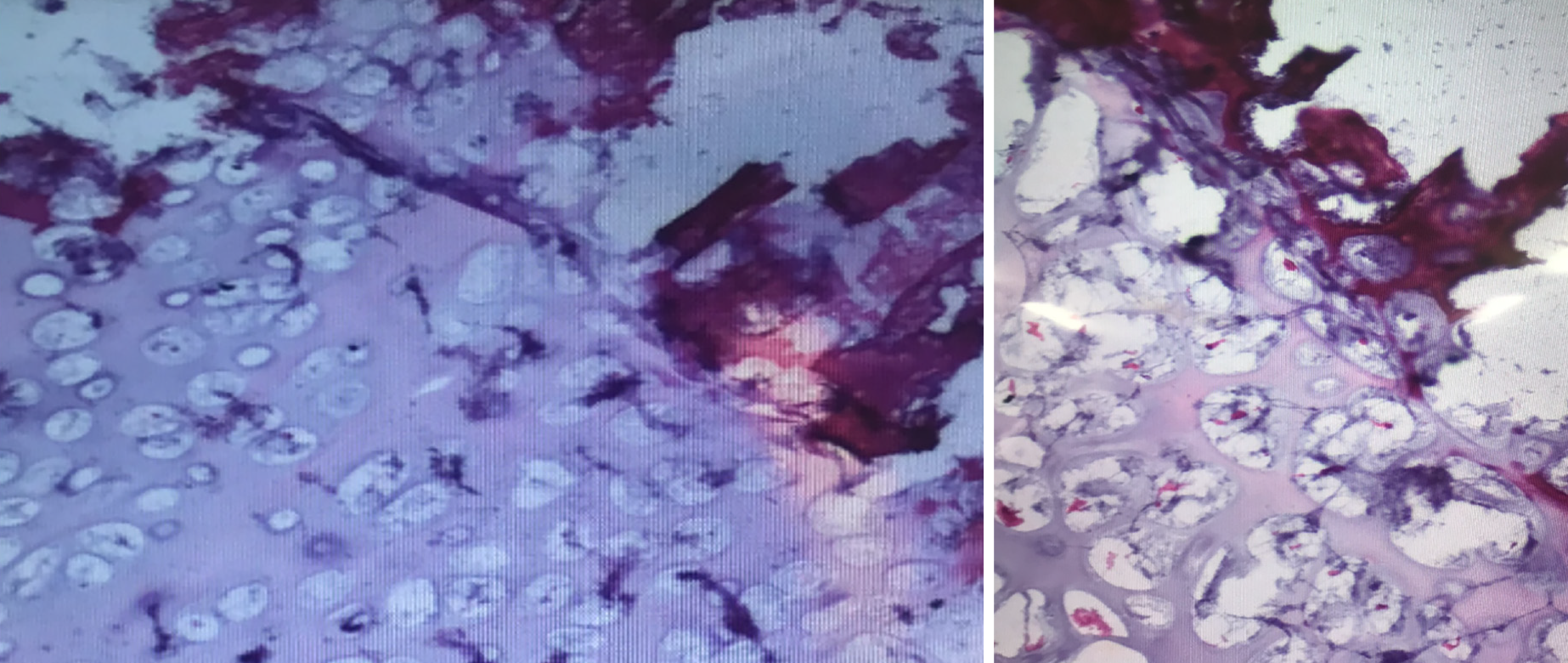
Intraoperatively, mass excision with partial laminectomy was performed through a posterior midline incision. Careful paramedian dissection was performed in order to free the soft tissues surrounding the mass without destabilization of the interspinous ligaments or the right L5-S1 facet joint. Once isolated, the mass was excised en bloc with use of a harmonic bone scalpel. A Woodson elevator was utilized under the lamina to gauge depth and ensure that the ventral surface of the lamina remained intact. After excision, a paraspinal muscular flap closure was performed to minimize the potential space created by the mass. Gross surgical pathology revealed a large bony mass measuring 3.8 cm × 3.5 cm × 2.5 cm that included a 1 cm thick sessile cartilaginous cap. Surgical margins were noted to be smooth (Figure 4). Microscopic examination demonstrated mature hyaline cartilage, fibrous perichondrium, and trabecular bone without cellular atypia or malignant features. Enchondral ossification was present within the sample without marrow elements identifiable within the trabecular bone. Pathology was consistent with benign osteochondroma (Figure 5).
Post-operatively, the patient was discharged on postoperative day 1 following removal of his drain. His post-operative course was uncomplicated. He returned to his work as a manual labor without pain or limitation at three months following surgery.
Osteochondromas are benign osteocartilaginous proliferations at a growth plate that typically stop increasing in size at skeletal maturity[5,15]. They infrequently occur in the spine (1% to 4% of osteochondromas) with the majority localized to the cervical spine[4]. Cases of lumbar osteochondromas, like the case presented, are scarce.
Patients typically present with axial pain which may be exacerbated with certain positions or activity like the patient in this study. Neurological manifestations from cord compression are rare but have previously been reported[16]. Initial radiographic evaluation may reveal a spinal lesion composed of a cortical protuberance in continuity with the medullary canal. Advanced imaging is recommended to aid in diagnosis. CT imaging provides detail of the osseous and cartilaginous margins whereas MRI can provide the thickness and architecture of the cartilaginous cap. A cartilage cap > 3 cm thickness can indicate malignant transformation[1,4].
Lumbar involvement of these exostoses is rare, perhaps due to increased stiffness of the lumbar spine when compared to the cervical spine and therefore less microtrauma to the epiphyseal cartilage. Osteochondromas occur within the posterior elements of the spine and may become symptomatic with growth into the paraspinal musculature[17]. A review of lumbosacral osteochondromas performed by Kuraishi et al[16] noted radicular symptoms in 4 out of 5 cases. In that series, involvement of the L4 inferior articular processes resulted in an L5 radiculopathy whereas involvement of the superior articular process of S1 corresponded with an L5 radiculopathy.
Given our patient’s history of a suprasellar pineal germinoma requiring chemo
Painful lesions or those causing neurologic deficit require surgical intervention. If resection entails destabilizing the spine, instrumentation and arthrodesis should be considered. The goal of surgery should be complete resection of the lesion[21]. Surgical nuance of our lumbar osteochondroma excision was the use of an ultrasonic bone scalpel. The ultrasonic bone scalpel amplifies electrical signals that oscillate a blunt blade, allowing for targeted resection of cortical bone without violation of soft tissues[22]. Given the location of our patients osteochondroma with abutment of the right L5 interspinous ligament and right L5-S1 facet capsule, excision of the mass without violating the soft tissues prevented posterior spinal destabilization requiring subsequent instrumentation and fusion.
Osteochondromas of the spine are a rare but treatable condition. For symptomatic lesions, complete resection is largely curative without adjuvant therapy. The patient in this case report was pain free at his post-operative visits without signs or symptoms of recurrence or complication. He returned to work as a manual laborer at 3 mo. Further reports of patients diagnosed with osteochondromas and a history of childhood radiation will enable better understanding of radiation-induced osteochondromas and the rates and locations at which they occur.
Manuscript source: Unsolicited manuscript
Specialty type: Orthopedics
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Vahedi P S-Editor: Wang JL L-Editor: AP-Editor: Ma YJ
| 1. | Motamedi K, Seeger LL. Benign bone tumors. Radiol Clin North Am. 2011;49:1115-1134, v. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 2. | O'Brien MF, Bridwell KH, Lenke LG, Schoenecker PL. Intracanalicular osteochondroma producing spinal cord compression in hereditary multiple exostoses. J Spinal Disord. 1994;7:236-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Pham MH, Cohen J, Tuchman A, Commins D, Acosta FL. Large solitary osteochondroma of the thoracic spine: Case report and review of the literature. Surg Neurol Int. 2016;7:S323-S327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Thakur NA, Daniels AH, Schiller J, Valdes MA, Czerwein JK, Schiller A, Esmende S, Terek RM. Benign tumors of the spine. J Am Acad Orthop Surg. 2012;20:715-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Murphey MD, Choi JJ, Kransdorf MJ, Flemming DJ, Gannon FH. Imaging of osteochondroma: variants and complications with radiologic-pathologic correlation. Radiographics. 2000;20:1407-1434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 528] [Cited by in RCA: 481] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 6. | Sinelnikov A, Kale H. Osteochondromas of the spine. Clin Radiol. 2014;69:e584-e590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Lotfinia I, Vahedi P, Tubbs RS, Ghavame M, Meshkini A. Neurological manifestations, imaging characteristics, and surgical outcome of intraspinal osteochondroma. J Neurosurg Spine. 2010;12:474-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 8. | Gorospe L, Madrid-Muñiz C, Royo A, García-Raya P, Alvarez-Ruiz F, López-Barea F. Radiation-induced osteochondroma of the T4 vertebra causing spinal cord compression. Eur Radiol. 2002;12:844-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Chew FS, Weissleder R. Radiation-induced osteochondroma. AJR Am J Roentgenol. 1991;157:792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 10. | Libshitz HI, Cohen MA. Radiation-induced osteochondromas. Radiology. 1982;142:643-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 46] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 11. | Heaston DK, Libshitz HI, Chan RC. Skeletal effects of megavoltage irradiation in survivors of Wilms' tumor. AJR Am J Roentgenol. 1979;133:389-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 48] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 12. | Cree AK, Hadlow AT, Taylor TK, Chapman GK. Radiation-induced osteochondroma in the lumbar spine. Spine (Phila Pa 1976). 1994;19:376-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 13. | Moriwaka F, Hozen H, Nakane K, Sasaki H, Tashiro K, Abe H. Myelopathy due to osteochondroma: MR and CT studies. J Comput Assist Tomogr. 1990;14:128-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 14. | Sultan M, Khursheed N, Makhdoomi R, Ramzan A. Compressive Myelopathy due to Osteochondroma of the Atlas and Review of the Literature. Pediatr Neurosurg. 2016;51:99-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 15. | Gunay C, Atalar H, Yildiz Y, Saglik Y. Spinal osteochondroma: a report on six patients and a review of the literature. Arch Orthop Trauma Surg. 2010;130:1459-1465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Kuraishi K, Hanakita J, Takahashi T, Watanabe M, Honda F. Symptomatic osteochondroma of lumbosacral spine: report of 5 cases. Neurol Med Chir (Tokyo). 2014;54:408-412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 17. | Nakaya Y, Ohue M, Baba I, Fujiwara K, Nakano A, Fukumoto S, Fujishiro T, Neo M. Paraarticular osteochondroma of a lumbar facet joint presenting with radiculopathy. J Orthop Sci. 2018;23:592-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 18. | King EA, Hanauer DA, Choi SW, Jong N, Hamstra DA, Li Y, Farley FA, Caird MS. Osteochondromas after radiation for pediatric malignancies: a role for expanded counseling for skeletal side effects. J Pediatr Orthop. 2014;34:331-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 19. | Taitz J, Cohn RJ, White L, Russell SJ, Vowels MR. Osteochondroma after total body irradiation: an age-related complication. Pediatr Blood Cancer. 2004;42:225-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 20. | Jaffe N, Ried HL, Cohen M, McNeese MD, Sullivan MP. Radiation induced osteochondroma in long-term survivors of childhood cancer. Int J Radiat Oncol Biol Phys. 1983;9:665-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 27] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Raswan US, Bhat AR, Tanki H, Samoon N, Kirmani AR. A solitary osteochondroma of the cervical spine: a case report and review of literature. Childs Nerv Syst. 2017;33:1019-1022. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 22. | Chen Y, Chang Z, Yu X, Song R, Huang W. Use of Ultrasonic Device in Cervical and Thoracic Laminectomy: a Retrospective Comparative Study and Technical Note. Sci Rep. 2018;8:4006. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |