Published online Jun 18, 2021. doi: 10.5312/wjo.v12.i6.376
Peer-review started: January 17, 2021
First decision: March 8, 2021
Revised: April 2, 2021
Accepted: May 22, 2021
Article in press: May 22, 2021
Published online: June 18, 2021
Processing time: 145 Days and 4.8 Hours
Three-dimensional (3D)-printed titanium cups used in primary total hip arthroplasty (THA) were developed to combine the benefits of a low elastic modulus with a highly porous surface. The aim was to improve local vascularization and bony ingrowth, and at the same time to reduce periprosthetic stress shielding. Additive manufacturing, starting with a titanium alloy powder, allows serial production of devices with large interconnected pores (trabecular titanium), overcoming the drawbacks of tantalum and conventional manufacturing techniques. To date, 3D-printed cups have achieved dependable clinical and radiological outcomes with results not inferior to conventional sockets and with good rates of osseointegration. No mechanical failures and no abnormal ion release and biocompatibility warnings have been reported. In this review, we focused on the manufacturing technique, cup features, clinical outcomes, open questions and future developments of off-the-shelf 3D-printed titanium shells in THA.
Core Tip: Three-dimensional printed titanium cups theoretically provide a porous, rough surface that improves local vascularization and osseointegration, while avoiding stress shielding because of the low elastic modulus. We herein discuss the manufacturing, main features, and clinical results obtained with 3D-printed titanium cups, with a focus on the open questions and possible future developments to improve this newborn device and technology.
- Citation: Castagnini F, Caternicchia F, Biondi F, Masetti C, Faldini C, Traina F. Off-the-shelf 3D printed titanium cups in primary total hip arthroplasty. World J Orthop 2021; 12(6): 376-385
- URL: https://www.wjgnet.com/2218-5836/full/v12/i6/376.htm
- DOI: https://dx.doi.org/10.5312/wjo.v12.i6.376
Acetabular component loosening is one of the most frequent reasons for revision in primary total hip arthroplasty (THA)[1]. Uncemented third-generation sockets were designed to improve local vascularization and the subsequent bony ingrowth promoted by the porosity of the cup surface and the biocompatibility of the metal alloy coating[2,3]. The long-term clinical and radiological performance and reliable safety profile of third-generation tantalum cups in primary THA is outstanding, even if some sporadic concerns have arisen after consulting registry data[4]. Highly porous titanium implants were developed based on the previous solid experience with third-generation, highly porous tantalum cups. The aim was to overcome drawbacks related to the cost of tantalum, which is a rare metal. 3D printed cups are a specific stand-alone type of highly porous titanium cups[2]. While they share the material and porosity with other highly porous titanium cups, because of additive manufacturing, 3D printed cups have larger pore sizes and higher porosity, reproducing trabecular bone-like elastic modulus in unique monoblock implants with no coatings[2]. 3D-printed cups may exhibit differences in the surface and beyond the surface with different biological advantages than traditionally manufactured highly porous titanium cups, including improved osseointegration, reduced stress shielding, and have unique failure modalities like cracking and ion release[2,5].
3D printed cups are produced using additive manufacturing, an industrial process that transforms 3D computer models into devices by layer-on-layer, titanium alloy powder-based fusion technology. This production method is very different from the manufacturing of traditional implants, usually by subtracting machinery and forming. Additive manufacturing is particularly suitable for custom made implants, complex geometries, and diverse surfaces, even with serial production. On the other side, conventional techniques are mainly limited to mass production and off-the shelf devices because of manufacturing constraints[5]. These features allow additive manufacturing devices to integrate surface porosity within a monoblock implant, which improves osseointegration and potentially reduces stress shielding by mimicking the stiffness of the periprosthetic bone (Figure 1). The additive manufacturing process is a powder bed fusion technology that adopts electron beam melting (EBM) and/or laser beam melting (SLM) energy sources for producing titanium alloy cups[6]. Both techniques start with a titanium alloy powder, whose chemical and physical properties are chosen as a function of the production technique and the powder bed fusion technology. The powder can be processed using a computer-aided design (CAD) model. The device is produced by a beam, with melting and bonding the powder layer after layer with precisely controlled cooling[ 5,6].
Some important differences exist between the two main additive manufacturing technologies. EBM technology is driven by a higher power source, requires a vacuum chamber at 600-1000°C, and is controlled by electromagnetic lenses that focus the beam, which is regulated by a deflection coil. A preheating scan is required, however, the support during manufacturing is scarce because of the high speed and the vacuum environment. On the other hand, SLM is characterized by a much lower power source, is performed in a purified inert gas atmosphere at 200°C, and is controlled by a CAD mechanical mirror system. Preheating is not required, and the support during manufacturing is more intense. A most striking difference between EBM and SLM is the powder that is used. EBM used larger particles (45-150 μm vs 20-100 μm) and thicker powder layers (50-200 μm vs 20-100 μm)[5-7]. Post processing steps include cleaning (removal of excess powder), surface finishing, and thermal treatment, particularly for SLM.
The manufacturing technology has a noticeable impact on the final qualities of the device. The surface is rougher and the CAD design is more accurately reproduced after EBM manufacturing. Moreover, the mechanical properties of EBM devices are good because there are residual tensile stresses. The superior mechanical properties mean that heat treatment is not required, which reduces the manufacturing costs and makes EBM faster and more cost-effective than SLM[8]. The biocompatibility of the devices produced with the two different technologies is dependable, and neither dermal irritation nor delayed hypersensitivity were demonstrated in animals[6,9]. A comparative study cell reported that adhesion, proliferation, and activity were more pronounced in the SLM cohort[10].
A complete list of off-the-shelf 3D printed titanium cups is shown in Table 1. Nearly all the 3D printed cups have similar features, which include a porous surface structure of interconnected pores larger than 200 μm with a porosity higher than 50% (Figure 2). The elastic modulus and coefficient of friction were rarely provided by the manufacturers. Many cups had multihole designs with even augments, making the devices adaptable for massive bone stock loss and cup revision settings.
| Cup | Manufacturer | Pore size (µm) | Porosity (%) | Design | Modularity | Notes | Available clinical studies |
| Fixa Ti-Por | Adler Ortho, Milan, Italy | 700 | 70 | Hemispherical | Yes | Yes | |
| Agilis Ti-Por | Adler Ortho, Milan, Italy | 700 | 70 | Hemispherical | No, Delta ceramic | Large ceramic-on-ceramic heads | No |
| Polymax Ti-Por | Adler Ortho, Milan, Italy | 700 | 70 | Hemispherical | No, XLPE | Yes | |
| Omnia Ti-Por | Adler Ortho, Milan, Italy | 700 | 70 | Hemispherical | Yes | Multihole, Ti augments | No |
| Delta TT | Lima Corporate, San Daniele, Italy | 640 | 65 | Hemispherical | Yes | Elastic modulus: 1.12 GPa | Yes |
| Delta One TT | Lima Corporate, San Daniele, Italy | 640 | 65 | Hemispherical | Yes | Multihole, Ti augments | Yes |
| Delta Revision TT | Lima Corporate, San Daniele, Italy | 640 | 65 | Hemispherical, triflanged | Yes | Commercially pure Ti, multihole, Ti augments | Yes |
| SQRUM TT | Kyocera, Kyoto, Japan | 640 | 60 | Hemispherical | Yes | Coefficient of friction: 1.09 | Yes |
| Mpact 3D Metal | Medacta, Castel San Pietro, Switzerland | 600-800 | 75 | Hemispherical | Yes | Multihole, Ti augments | No |
| Trinity Plus | Corin, Cirencester, United Kingdom | 300-900 | 50-90 | Hemispherical | Yes | Even multihole | No |
| Ecofit Epore | Implatcast, Buxtehude, Germany | 100-500 | 60 | Hemispherical | Yes | Multihole | No |
| Redapt | Smith and Nephew, Menphis, United States | 202-934 | 60-80 | Hemispherical | Yes | Multihole | No |
| Trident II Tritanium | Stryker, Mahwah, United States | 100-700 | 55-65 | Hemispherical | Yes | Laser rapid melting technology | Yes |
| G7 | Zimmer, Warsaw, United States | 475 | 70 | Hemispherical | Yes | Even multihole shells | Yes |
| 3D ACT | ITI Medical Equipment, Changzhou City, China | 600-800 | 80 | Yes | Coefficient of friction: 1.08 | Yes |
Few preclinical studies of 3D printed titanium cups are available. Jahnke et al[11] compared the micromotion of 3D printed cups (EPore) and conventional sockets in sawbone models. The 3D printed cups had acceptable micromotion, lower than of conventional cups with low wall thickness. The investigators reported that the wall thickness, together with the highly porous surface, contributed to 3D cup stability, allowing peripheral micromotion of the sockets, and at the same time, provided rigidity with the thicker walls[11]. Dall’Ava et al[12] evaluated the outer surfaces of three off-the -shelf 3D printed titanium cups (Delta TT, Trident II Tritanium, and Mpact 3D Metal). The three cups have differences as wall thickness, pore size, porosity, and metallic structure. Some molten beads were present, with differences of size and density related to the production technology[12]. Dall’Ava et al[13] compared 3D printed cups (Delta TT) with conventional cups (Pinnacle Porocoat, DePuy, United States). Pore size, porosity, and the mechanical features of the pores of the 3D printed caps and the conventional cups were significantly different. A visual assessment revealed higher percentages of bony attachment and tissue ingrowth for the 3D printed cups, but the differences comparison with the conventional sockets were not significant[13]. Hothi et al[14] compared three 3D printed cups with conventional sockets. Two of the 3D-printed acetabular shells were produced by EBM (Delta TT and Mpact 3D) and one by laser rapid melting (Trident II Tritanium). Structural cavities were evident in all the 3D-printed cups and in none of the conventional sockets, with a significantly higher density, but no difference in cavity size, in the laser rapid melting socket. The presence of cavities was attributed to suboptimal manufacturing parameters, gas entrapment, or gaps in the starting powder beads[14].
A systematic review of the outcomes and survivorship of 3D printed titanium cups in THA was conducted following retrieval of published studies indexed in the PubMed/MEDLINE database. {query box: ["highly" (All Fields) AND "porous" (All Fields)]} OR ["trabecular" (All Fields) OR "trabecularization" (All Fields) OR "trabecularized" (All Fields)] OR "pore" (All Fields) OR {"3D" (All Fields) AND ["printed" (All Fields) OR "printing" (MeSH Terms) OR "printing" (All Fields) OR "print" (All Fields) OR "printings" (All Fields) OR "prints" (All Fields)]} OR ["printed" (All Fields) OR "printing" (MeSH Terms) OR "printing" (All Fields) OR "print" (All Fields) OR "printings" (All Fields) OR "prints" (All Fields)] OR "3D printed" (All Fields) OR {["porosity"(MeSH Terms) OR "porosity" (All Fields) OR "porosities" (All Fields)] OR ["addit manuf" (Journal) OR ["additive" (All Fields) AND "manufacturing" (All Fields)] OR "additive manufacturing" (All Fields)]} AND "cup" (All Fields) AND ["hip" (MeSH Terms) OR "hip" (All Fields)] AND ["arthroplasty" (MeSH Terms) OR "arthroplasty" (All Fields) OR "arthroplasties" (All Fields)]; double check for 3D printed titanium cup brands, as listed in the paper by Dall’Ava: Ti-Por, Delta TT, Trinity, EcoFit, C-Fit, Mpact, Redapt, Trident, G7 {query box: Cup brand AND "cup" (All Fields) AND ["hip" (MeSH Terms) OR "hip" (All Fields)]}.
Ten papers were included. The two largest studies were registry reports, one from Registro dell'Implantologia Protesica Ortopedica (RIPO) and the other from the New Zealand arthroplasty registry. The RIPO registry study investigated the mid-term outcomes of 9864 Ti-Por cups, compared with other cementless cups (Figure 3). The non-stratified survival rates showed that the 3D printed cups had a significantly higher survival rate (98.7% vs 97.9%) at 7 years. The survival rates with cup aseptic loosening as the study end point showed that Ti-Por cups performed better than the other cementless cups (99.9% vs 99.5%). With similar articulation surfaces, Ti-Por cups achieved survival rates comparable to those of the control group[15].
The New Zealand registry compared 2192 3D-printed cups (1397 Delta TT cups, 640 Ti-Por, and 155 Polymax cups) and other cementless sockets (Pinnacle, Trident, and RM). The outcomes were similar in both cohorts with no differences in the revision rates and functional scores (P = 0.058, hazard ratio: 1.29) while additive manufactured sockets were more frequently implanted in younger patients; the rates of aseptic cup loosening and deep infections were similar in both cohorts. Polymax cups achieved the lowest rates among the 3D cups[16].
Perticarini et al[17] published the first clinical and radiographic description of a 3D printed cup (Delta TT) cohort with a follow-up of 72 months. The clinical and radiographic outcomes of 134 THAs were satisfying, with 99.3% of the sockets radiographically stable at final follow-up. Only one case of aseptic cup loosening was reported. It occurred in a high-grade dysplastic patient, and was caused by the high dislodging forces of the lengthened abductor muscles (no femoral shortening was performed). The cup was successfully revised with another Delta TT device. The survival rate at mid-term was 99.3%[17].
Berend et al[18] evaluated 400 hips with G7 cups at a minimum follow-up of 1 year. Clinical and radiographic outcomes were satisfactory, with no radiolucent lines; two cup failures occurred because of impaired bony ingrowth[18].
Berend et al[19] updated the results of the previous case series at a minimum follow-up of 3 years in 152 implants. The clinical outcomes significantly improved, and the radiographic findings showed no radiolucent lines. The overall survival rate at 3.4 years was 99.5%, one case required cup revision because of failed bony ingrowth[19].
Massari et al[20] reported dual-energy x-ray absorptiometry outcomes of 3D printed cups (Delta TT): They found a reduction of bone mass density at 6 months, with slight recovery and stabilizing by 24 months. The bone mass variation was dependent on the body mass index. They reported also a significant clinical improvement with respect to the pre-operative status and no cases of failed cups[20].
Imai et al[21] compared the hip replacement outcomes of 101 3D printed cups (SQRUM TT) and 35 hydroxyapatite-coated sockets (SQRUM HA) in low-grade dysplastic patients receiving the same stems and the same bearings. The short-term clinical outcomes significantly improved in both the cohorts, achieving similar results. The radiographic 2-year outcomes showed that the 3D printed cups had a higher rate of radiolucent lines and fibrous ingrowth than the conventional hydroxyapatite sockets[21].
Bistolfi et al[22] evaluated the metal ion levels in the blood and urine in a cohort of 19 patients with conventional cups (Delta PF) and 19 with 3D printed cups (Delta TT). At 24 months, differences in the blood and urine levels of titanium, aluminum, and vanadium ions in of the two cohorts were not significant. A trend of decreasing ion levels was evident in both cohorts[22].
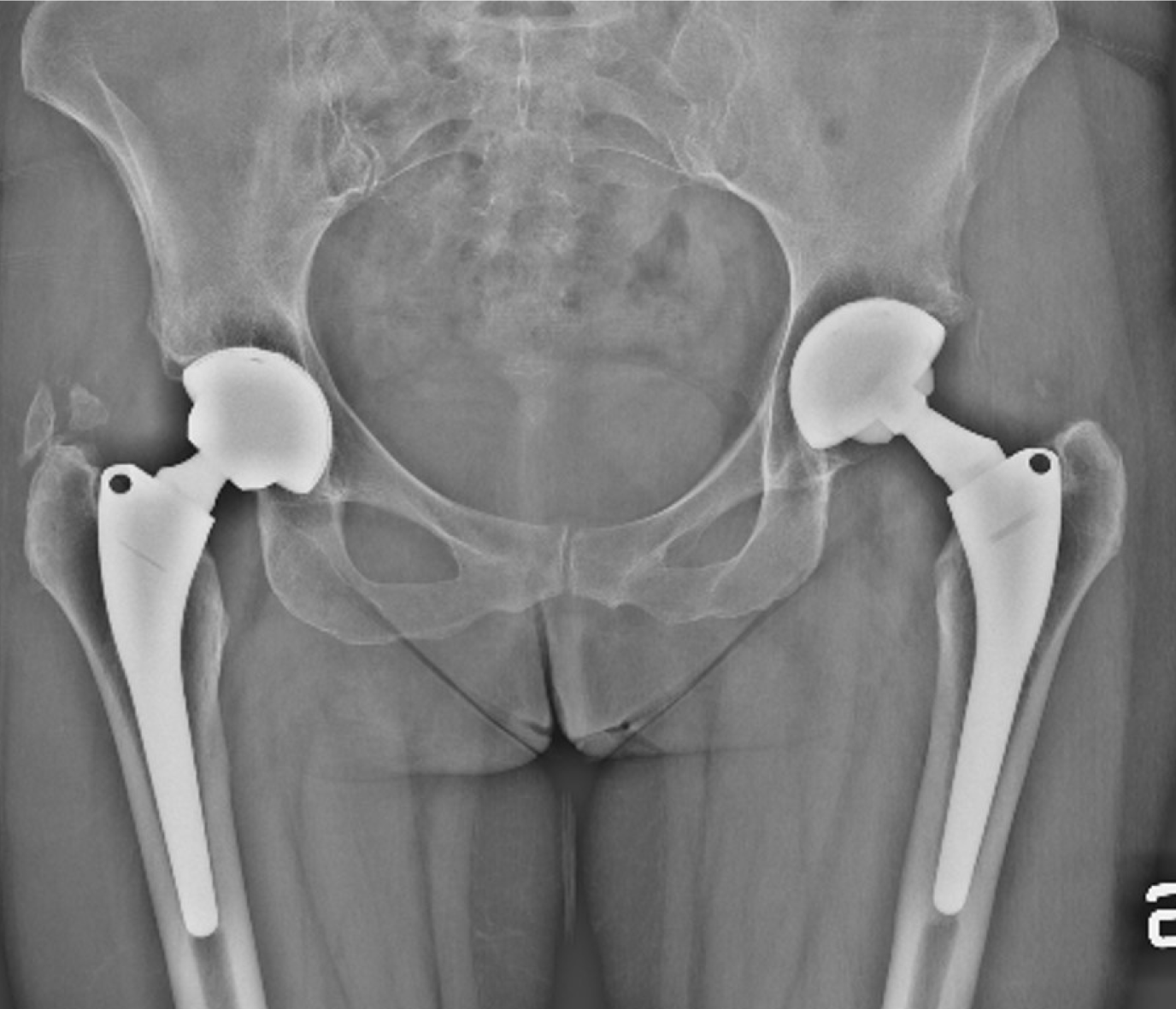
Castagnini et al[23] published a retrospective comparison of 24 metachronous bilateral THAs, a Ti-Por cup on one side and a hydroxyapatite-coated socket on the other side (Figure 4). The mid-term clinical outcomes showed comparable rates of cup loosening and radiographic osseointegration in both cohorts. The Ti-Por group had more evident medial stress shielding, but the difference was not significant (P = 0.067). Neither was the decrease in acetabular radiographic density in the DeLee and Charnley Zone II[23,24].
Alamanda et al[25] used multiacquisition variable-resonance image combination magnetic resonance imaging to assess the radiological osseointegration of 3D printed cups, evaluating 19 Trident II Tritanium shells and 20 Trident sockets as a control group. The 3D cup cohort achieved significantly better outcomes in terms of bony ingrowth (P = 0.009) with good osseointegration in 99.4% of the cases compared with 91.6% for the conventional cups. Only 0.6% of the bone-cup interface, measured in nine acetabular zones, were fibrotic compared with 8.3% of the conventional cups, mainly involving the central posterior zone[25].
The outcomes of 3D printed ACT sockets were retrospectively assessed in 108 hips by Geng et al[26] at an average follow-up of 48 months. A significant clinical improvement was observed, with 106 of 108 (91.3%) patients satisfied with the outcome. No acetabular cup failed: some transient radiolucent lines completely disappeared after 6 months. Radiographic signs of osseointegration were evident in all the cups. The overall survival rate was 99.1% at 72 months, with cup survival rate achieving 100%[26].
In summary, the clinical and radiological outcomes of 3D-printed titanium cups were dependable at mid-term, with large sized studies providing data not inferior to conventional cups. No mechanical failures were recorded and the metal ion release was reassuring. Some radiological findings of fibrotic ingrowth and radiolucent lines were also reported, making longer follow-up and more preclinical studies of 3D printed cups desirable.
The first open question about 3D-printed cups is about manufacturing. The layer-over-layer production and the highly porous structure may predispose to delamination and fatigue fracture, and the findings about cavities are not reassuring[14]. Another concern is related to the balling effect of metallic droplets on the molten surfaces, potentially causing cracking and undesired roughness on the inner surface that compromises the locking mechanism and the liner seating, wear, and hardness. While some concerns of fatigue fractures and liner failures may be raised, to date no mechanical failures of 3D-printed cups have been reported, only a case of hemisp
The stress shielding and the fibrous membrane interface issues in the medial zone may be reasons of concern in 3D printed cups. It is likely that these findings are mainly related to the different stress distribution around the periprosthetic acetabular bone and the improved circumferential grip of the highly porous sockets. Thus, it can be hypothesized that a device with nonuniform porosity may reduce medial stress shielding and improve long-term osseointegration. However, in mechanical testing, Le Cann et al[28] demonstrated that the opposite condition (micro-roughness around the peripheral rim and macro-roughness on the rest of the cup) promoted better primary stability by reducing the damage of the supportive circumferential bone[28]. To date, many doubts exist about the optimal pore size, porosity, and porous surface extension of titanium devices although fundamental investigations addressing this topic were carried out in the 1980s for tantalum metal blocks[29-32]. Some investigators have reported that the use of pores that were too large could be detrimental for osseointegration by promoting fibrous tissue growth, at least for a short time[33]. Some data on ultraporous titanium cups are available from laboratory and clinical cohort studies, but no definitive conclusions can be drawn, as long-term follow-up is lacking and the tested populations are small.
Another aspect to be developed is related to the cup stabilization provided by high friction coefficients in highly porous devices. Theoretically, the high friction coefficient and the better initial scratch fit would improve primary stability, possibly enhancing the secondary bony ingrowth. Some investigators have found improvements with some less porous cups (plasma-sprayed devices)[34]. Unfortunately, there have been no supporting studies with definitive results comparing ultraporous cups and more recent surface textures, as described by Goldman et al[35]. In that study, ultraporous DePuy Gription cups did not provide better resistance to bending loads in comparison to standard DePuy Porocoat titanium bead sockets following implantation in seven cadaver models.
3D-printed cups have a good biosafety profile, that needs to be confirmed in large clinical studies[6,22]. More in vivo and long-term studies are needed to evaluate and compare the biocompatibility of 3D-printed medical devices, considering the possibility of immunological rejection and inflammatory reactions that can account for the failure of metallic implants.
To date, the optimal 3D printing technique is still unknown. The 3D printed cups that have been evaluated in preclinical studies differ in pore size, porosity and structure, and some features that are dependent on the manufacturing method[12]. Moreover, while additive manufacturing is a highly efficient method with up to a 50% reduction of production costs and up to a 75% reduction of raw material use, the fine balance between efficiency and quality is yet to be determined[8,9,36]. More studies of the optimization of the energy requirements and scanning speed and their influence on the product quality and the subsequent need for post processing stages would be welcomed.
The possibility of achieving a good elastic modulus and a very highly porous surface makes 3D-printed cups particularly appealing for revision procedures when the bone stock is severely compromised. To date, short-term data on the use of 3D-printed cups are available in the literature; long-term data are lacking[37]. The preliminary short-to-midterm outcomes indicate that off-the-shelf 3D-printed titanium cups are dependable. Castagnini et al[38] reported no loosening of Ti-Por devices at a minimum follow-up of 5 years. Gallart et al[39] described two cases of Delta TT loosening out of 72 implants with augments, prevalently for revision hips, after more than 2 years. Steno et al[27] reported no re-revisions for loosening of Delta TT devices more than 3 years after implantation[27,38-40].
In summary, 3D printed cups produced using additive manufacturing demonstrated reliable clinical and radiographic mid-term outcomes, even in large patient series. These implants also had a reliable safety profile. The metal ion levels and rates of deep infection were comparable to control groups, and no cases of mechanical failure were reported. Nonetheless, to date no clinical studies have demonstrated significantly better outcomes when compared with conventional cups with the same bearing forces. Clinical trials and registry studies with longer follow-up are required to confirm the theoretical benefits of 3D-printed cups in terms of osseointegration and a decreased rate of aseptic loosening.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Personalized Arthroplasty Society.
Specialty type: Orthopedics
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Widmer KH S-Editor: Fan JR L-Editor: Filipodia P-Editor: Li JH
| 1. | Grave S. The Australian Orthopaedic Association National Joint Replacement Registry (AOANJRR) 2020. [cited 22 November 2020]. Aavailable from: https://aoanjrr.sahmri.com/. |
| 2. | Castagnini F, Traina F. Letter to the Editor on "Highly Porous Titanium Acetabular Components in Primary and Revision Total Hip Arthroplasty: A Systematic Review". J Arthroplasty. 2020;35:2302-2303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Malahias MA, Kostretzis L, Greenberg A, Nikolaou VS, Atrey A, Sculco PK. Highly Porous Titanium Acetabular Components in Primary and Revision Total Hip Arthroplasty: A Systematic Review. J Arthroplasty. 2020;35:1737-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 4. | Hemmilä M, Karvonen M, Laaksonen I, Matilainen M, Eskelinen A, Haapakoski J, Puhto AP, Kettunen J, Manninen M, Mäkelä KT. Survival of 11,390 Continuum cups in primary total hip arthroplasty based on data from the Finnish Arthroplasty Register. Acta Orthop. 2019;90:312-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Dall’Ava L, Hothi, H, Di Laura A, Henckel J, Hart A. 3D Printed Acetabular Cups for Total Hip Arthroplasty: A Review Article. Metals. 2019;9:729. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Wang H, Zhao B, Liu C, Wang C, Tan X, Hu M. A Comparison of Biocompatibility of a Titanium Alloy Fabricated by Electron Beam Melting and Selective Laser Melting. PLoS One. 2016;11:e0158513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 7. | Ni J, Ling H, Zhang S, Wang Z, Peng Z, Benyshek C, Zan R, Miri AK, Li Z, Zhang X, Lee J, Lee KJ, Kim HJ, Tebon P, Hoffman T, Dokmeci MR, Ashammakhi N, Li X, Khademhosseini A. Three-dimensional printing of metals for biomedical applications. Mater Today Bio. 2019;3:100024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 8. | Zhang Y, Wu L, Guo X, Kane S, Deng Y, Jung YG, Lee JH, Zhang J. Additive Manufacturing of Metallic Materials: A Review. J Materi Eng Perform. 2018;27:1-13. [DOI] [Full Text] |
| 9. | Ponader S, von Wilmowsky C, Widenmayer M, Lutz R, Heinl P, Körner C, Singer RF, Nkenke E, Neukam FW, Schlegel KA. In vivo performance of selective electron beam-melted Ti-6Al-4V structures. J Biomed Mater Res A. 2010;92:56-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 98] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 10. | Marin E, Fusi S, Pressacco M, Paussa L, Fedrizzi L. Characterization of cellular solids in Ti6Al4V for orthopaedic implant applications: Trabecular titanium. J Mech Behav Biomed Mater. 2010;3:373-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 83] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 11. | Jahnke A, Bott CC, Fonseca Ulloa CA, Jahnke GW, Rickert M, Ishaque BA, Ahmed GA. In vitro examination of the primary stability of three press-fit acetabular cups under consideration of two different bearing couples. Med Eng Phys. 2019;67:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Dall'Ava L, Hothi H, Henckel J, Di Laura A, Shearing P, Hart A. Comparative analysis of current 3D printed acetabular titanium implants. 3D Print Med. 2019;5:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 13. | Dall'Ava L, Hothi H, Henckel J, Di Laura A, Shearing P, Hart A. Characterization of dimensional, morphological and morphometric features of retrieved 3D printed acetabular cups for hip arthroplasty. J Orthop Surg Res. 2020;15:157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 14. | Hothi H, Dall'Ava L, Henckel J, Di Laura A, Iacoviello F, Shearing P, Hart A. Evidence of structural cavities in 3D printed acetabular cups for total hip arthroplasty. J Biomed Mater Res B Appl Biomater. 2020;108:1779-1789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | RIPO. Il Registro dell'Implantologia Protesica Ortopedica (RIPO). Database: RIPO. [cited 22 November 2020]. Available from: https://ripo.cineca.it/authzssl/Reports.html. |
| 16. | Wyatt MC, Kieser DC, Frampton CMA, Woodfield T, Hooper GJ. How do 3D printed primary uncemented acetabular components compare with established uncemented acetabular cups? Hip Int. 2020;27:1120700020918233. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Perticarini L, Zanon G, Rossi SM, Benazzo FM. Clinical and radiographic outcomes of a trabecular titanium™ acetabular component in hip arthroplasty: results at minimum 5 years follow-up. BMC Musculoskelet Disord. 2015;16:375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 18. | Berend KR, Adams JB, Morris MJ, Lombardi AV Jr. Three-Year Results with a Ringless Third-Generation Porous Plasma Sprayed Acetabular Component in Primary Total Hip Arthroplasty. Surg Technol Int. 2017;30:295-299. [PubMed] |
| 19. | Berend KR, Adams JB, Morris MJ, Lombardi AV Jr. Early Experience with a New Porous Hemispheric Acetabular Component. Surg Technol Int. 2015;27:263-267. [PubMed] |
| 20. | Massari L, Bistolfi A, Grillo PP, Borré A, Gigliofiorito G, Pari C, Francescotto A, Tosco P, Deledda D, Ravera L, Causero A. Periacetabular bone densitometry after total hip arthroplasty with highly porous titanium cups: a 2-year follow-up prospective study. Hip Int. 2017;27:551-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 21. | Imai H, Miyawaki J, Kamada T, Maruishi A, Takeba J, Miura H. Radiolucency around highly porous sockets and hydroxyapatite-coated porous sockets in total hip arthroplasty for hip dysplasia. Eur J Orthop Surg Traumatol. 2019;29:611-618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Bistolfi A, Cimino A, Lee GC, Ferracini R, Maina G, Berchialla P, Massazza G, Massè A. Does metal porosity affect metal ion release in blood and urine following total hip arthroplasty? Hip Int. 2018;28:522-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Castagnini F, Bordini B, Yorifuji M, Giardina F, Natali S, Pardo F, Traina F. Highly Porous Titanium Cups versus Hydroxyapatite-Coated Sockets: Midterm Results in Metachronous Bilateral Total Hip Arthroplasty. Med Princ Pract. 2019;28:559-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 24. | Moore MS, McAuley JP, Young AM, Engh CA Sr. Radiographic signs of osseointegration in porous-coated acetabular components. Clin Orthop Relat Res. 2006;444:176-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 167] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 25. | Alamanda VK, Demartino I, Potter HG, Koff MF, Lin B, Muskat A, Westrich GH. Multiacquisition Variable-Resonance Image Combination Magnetic Resonance Imaging Used to Study Detailed Bone Apposition and Fixation of an Additively Manufactured Cementless Acetabular Shell. Arthroplast Today. 2020;6:694-698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 26. | Geng X, Li Y, Li F, Wang X, Zhang K, Liu Z, Tian H. A new 3D printing porous trabecular titanium metal acetabular cup for primary total hip arthroplasty: a minimum 2-year follow-up of 92 consecutive patients. J Orthop Surg Res. 2020;15:383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 27. | Steno B, Kokavec M, Necas L. Acetabular revision arthroplasty using trabecular titanium implants. Int Orthop. 2015;39:389-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 28. | Le Cann S, Galland A, Rosa B, Le Corroller T, Pithioux M, Argenson JN, Chabrand P, Parratte S. Does surface roughness influence the primary stability of acetabular cups? Med Eng Phys. 2014;36:1185-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 29. | Pilliar RM, Lee JM, Maniatopoulos C. Observations on the effect of movement on bone ingrowth into porous-surfaced implants. Clin Orthop Relat Res. 1986;108-113. [PubMed] |
| 30. | Bobyn JD, Pilliar RM, Cameron HU, Weatherly GC. The optimum pore size for the fixation of porous-surfaced metal implants by the ingrowth of bone. Clin Orthop Relat Res. 1980;263-270. [PubMed] |
| 31. | Bobyn JD, Stackpool GJ, Hacking SA, Tanzer M, Krygier JJ. Characteristics of bone ingrowth and interface mechanics of a new porous tantalum biomaterial. J Bone Joint Surg Br. 1999;81:907-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 453] [Reference Citation Analysis (0)] |
| 32. | Tanzer M, Chuang PJ, Ngo CG, Song L, TenHuisen KS. Characterization of bone ingrowth and interface mechanics of a new porous 3D printed biomaterial: an animal study. Bone Joint J. 2019;101-B:62-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 33. | Taniguchi N, Fujibayashi S, Takemoto M, Sasaki K, Otsuki B, Nakamura T, Matsushita T, Kokubo T, Matsuda S. Effect of pore size on bone ingrowth into porous titanium implants fabricated by additive manufacturing: An in vivo experiment. Mater Sci Eng C Mater Biol Appl. 2016;59:690-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 432] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 34. | Small SR, Berend ME, Howard LA, Rogge RD, Buckley CA, Ritter MA. High initial stability in porous titanium acetabular cups: a biomechanical study. J Arthroplasty. 2013;28:510-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 35. | Goldman AH, Armstrong LC, Owen JR, Wayne JS, Jiranek WA. Does Increased Coefficient of Friction of Highly Porous Metal Increase Initial Stability at the Acetabular Interface? J Arthroplasty. 2016;31:721-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 36. | Wong KC, Scheinemann P. Additive manufactured metallic implants for orthopaedic applications. Sci China Mater. 2018;61:440-454. [DOI] [Full Text] |
| 37. | Migaud H, Common H, Girard J, Huten D, Putman S. Acetabular reconstruction using porous metallic material in complex revision total hip arthroplasty: A systematic review. Orthop Traumatol Surg Res. 2019;105:S53-S61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 38. | Castagnini F, Mariotti F, Tassinari E, Bordini B, Zuccheri F, Traina F. lsolated acetabular revisions of articular surface replacement (ASR) XL implants with highly porous titanium cups and Delta bearings. Hip Int. 2021;31:250-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 39. | Gallart X, Fernández-Valencia JA, Riba J, Bori G, García S, Tornero E, Combalía A. Trabecular TitaniumTM cups and augments in revision total hip arthroplasty: clinical results, radiology and survival outcomes. Hip Int. 2016;26:486-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 40. | Castagnini F, Bordini B, Stea S, Calderoni PP, Masetti C, Busanelli L. Highly porous titanium cup in cementless total hip arthroplasty: registry results at eight years. Int Orthop. 2019;43:1815-1821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |