Published online Oct 18, 2019. doi: 10.5312/wjo.v10.i10.348
Peer-review started: May 20, 2019
First decision: July 31, 2019
Revised: August 14, 2019
Accepted: September 4, 2019
Article in press: September 4, 2019
Published online: October 18, 2019
Processing time: 158 Days and 0.6 Hours
Periprosthetic joint infections (PJIs) are frequently caused by coagulase-negative Staphylococci (CoNS), which is known to be a hard-to-treat microorganism. Antibiotic resistance among causative pathogens of PJI is increasing. Two-stage revision is the favoured treatment for chronic CoNS infection of a hip or knee prosthesis. We hypothesised that the infection eradication rate of our treatment protocol for two-stage revision surgery for CoNS PJI of the hip and knee would be comparable to eradication rates described in the literature.
To evaluate the infection eradication rate of two-stage revision arthroplasty for PJI caused by CoNS.
All patients treated with two-stage revision of a hip or knee prosthesis were retrospectively included. Patients with CoNS infection were included in the study, including polymicrobial cases. Primary outcome was infection eradication at final follow-up.
Forty-four patients were included in the study. Twenty-nine patients were treated for PJI of the hip and fifteen for PJI of the knee. At final follow-up after a mean of 37 mo, recurrent or persistent infection was present in eleven patients.
PJI with CoNS can be a difficult to treat infection due to increasing antibiotic resistance. Infection eradication rate of 70%-80% may be achieved.
Core tip: Periprosthetic joint infections of the hip and knee caused by coagulase-negative Staphylococci can be difficult to treat. We retrospectively reviewed all patients treated with two-stage revision arthroplasty for such an infection in our hospital between 2003 and 2016. We treated 44 patients with coagulase-negative Staphylococci infection of the hip or knee with a two-stage revision using an antibiotic-loaded spacer. At final follow-up, infection was eradicated in 33 of these patients.
- Citation: Veltman ES, Moojen DJF, van Ogtrop ML, Poolman RW. Two-stage revision arthroplasty for coagulase-negative staphylococcal periprosthetic joint infection of the hip and knee. World J Orthop 2019; 10(10): 348-355
- URL: https://www.wjgnet.com/2218-5836/full/v10/i10/348.htm
- DOI: https://dx.doi.org/10.5312/wjo.v10.i10.348
Coagulase-negative staphylococci (CoNS) are a hard-to-treat group of microorganisms in relation to implanted foreign materials, due to a high rate of methicillin resistance and biofilm formation[1]. In recent years, the incidence of infections with CoNS has increased[2,3]. Periprosthetic joint infection (PJI) is a devastating complication after hip and knee arthroplasty that occurs in 1%-2% of patients[4]. When infection persists despite debridement procedures or when infection is diagnosed more than 3 mo postoperatively, it is considered a chronic infection[5]. In case of chronic PJI, removal of the prosthesis is usually indicated[6]. Two-stage revision arthroplasty with the use of an antibiotic-loaded spacer is the gold standard treatment in case of persistent or chronic infection[7,8].
The type of spacer used during two-stage revision does not influence the infection eradication rate[9,10]. In contrast, characteristics of the causative microorganism do influence the chance of infection eradication after two-stage revision[11]. Resistance to commonly prescribed antibiotics is an increasing problem as well[11]. Bacteria such as CoNS can form a biofilm on the prosthesis that prevents elimination by host defences and antimicrobial therapy[1,12]. In orthopaedic revision arthroplasty, the rate of resistance to antibiotics by CoNS is increasing[13]. The effects of infection exclusively by CoNS on the outcome after two-stage revision arthroplasty have not yet been described. The objective of this study was to evaluate infection eradication rate after two-stage revision arthroplasty of the hip and knee in patients with CoNS PJI.
We used the STROBE cohort checklist when writing our report[14]. This study was approved by the local medical ethics committee. After approval, we retrospectively reviewed the records of all patients who had two-stage revision arthroplasty of the hip or knee in our hospital between 2003 and 2016. We included all patients with CoNS PJI of the hip or knee in the study. Exclusion criteria were monomicrobial infection with bacteria other than CoNS and patients receiving one-stage revision. Patients with polymicrobial infection, in whom CoNS was one of the infecting organisms, were included in the study. In all patients, diagnosis of infection was affirmed according to the Musculoskeletal Infection Society criteria. Joint aspirations were routinely performed preoperatively and were positive in all patients.
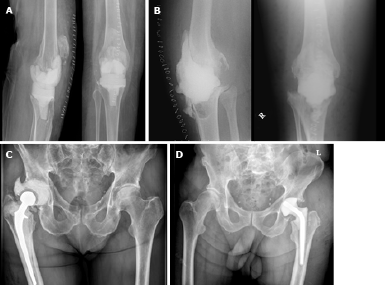
During first-stage surgery, we removed the infected prosthesis including all bone cement (when present). Multiple tissue samples were taken for culture, after which we administered cefuroxime antibiotic prophylaxis. We did not perform sonication of the removed prosthesis. After meticulous debridement, we implanted an antibiotic-loaded interval spacer with gentamicin and vancomycin. In patients with an infected total hip arthroplasty, we used either a functional articulating spacer or a prefabricated cement spacer (Figure 1A and B)[10]. Functional articulating spacers consist of (parts of) regularly used hip arthroplasty components combined with antibiotic-impregnated cement. Prefabricated cement spacers are commercially available in different head sizes and two different lengths. In patients with infected total knee arthroplasty we used either static spacers or dynamic spacers (Figure 1C and D)[15]. Static spacers are blocks of antibiotic-loaded cement that are moulded by hand intra-operatively. Patients were not allowed to bear weight on the static spacer and performing range of motion exercises was not possible. The dynamic spacers were either prefabricated cement blocks, or cement moulded by hand in the shape of a knee prosthesis.
We treated patients with antibiotics according to the recommendations as published by Zimmerli et al[4]. Patients received intravenous antibiotics for at least 2 wk based on the antibiogram of the cultured bacteria. Whenever possible, after 2 wk, we switched antibiotics to an oral substitute for an additional 4-wk minimum. The exact antibiotic treatment was determined in close consultation with a microbiologist and an infectious disease specialist. At 2 wk before the second-stage procedure, we discontinued antibiotics to achieve a 2-wk antibiotic free interval.
During second-stage surgery, we extracted the antibiotic-loaded spacer. Again, we took multiple tissue samples for culture, after which we administered antibiotic prophylaxis. We adjusted the postoperative antibiotic prophylaxis for the antibiogram of the bacteria cultured after the first-stage procedure. We performed another thorough debridement, after which we implanted a revision prosthesis. Postoperatively, patients received intravenous cefuroxime until culture results were available after 2 wk. When culture results were negative, we ceased antibiotics and patients were discharged. In case cultures were still positive, we continued antibiotics for a total of 12 wk.
We retrieved general patient characteristics, preoperative and postoperative laboratory results, complications during treatment, and final outcomes from patients’ records. Primary outcome was infection eradication after second-stage procedure, which was defined as the absence of clinical, radiological, or laboratory signs of infection at the latest follow-up, with a minimum of 1 year after second-stage surgery. Secondary outcomes were complications registered during the spacer period and at final follow-up.
Failure of treatment was defined as persistent or repeated infection after second-stage procedure, making it necessary to perform another revision, resection arthroplasty, arthrodesis, or amputation of the limb or use of suppressive antibiotics at final follow-up[16]. We used descriptive statistics, mean, and range to represent the demographics of the patients. Excel and SPSS software were used to perform calculations and statistical analyses. We analysed patients with two-stage revision of hip or knee as one group, and divided them into groups according to the joint treated and the interval spacer used.
Between 2003 and 2016 we treated 44 patients with CoNS PJI of a total hip or total knee prosthesis with two-stage revision arthroplasty using an antibiotic-loaded interval spacer. General patient characteristics can be found in Table 1. Polymicrobial infection was present in six patients. CoNS were sensitive to flucloxacillin or clindamycin in 19 patients. Due to antibiotic resistance to flucloxacillin and clindamycin, we treated 23 patients with vancomycin. We treated 2 patients with linezolid for 4 wk.
| Hip | Knee | Total | |
| Number of patients | 29 | 15 | 44 |
| Mean age | 66 | 64 | 66 |
| Gender, female | 15 | 11 | 26 |
| Mean BMI | 27 | 30 | 28 |
| BMI > 30, patients | 8 | 9 | 17 |
| Indication for primary prosthesis | |||
| Osteoarthritis | 19 | 15 | 34 |
| Posttraumatic | 10 | 0 | 10 |
| Comorbidity | |||
| Immune suppression | 3 | 2 | 5 |
| Previous PJI | 2 | 5 | 7 |
| Diabetes mellitus | 5 | 3 | 8 |
| Obesity, BMI > 30 | 8 | 9 | 17 |
| Active smoking | 7 | 4 | 11 |
| ASA 1/2/3 | 1/18/10 | 1/11/3 | 2/29/13 |
Laboratory results showed a mean C-reactive protein (CRP) level of 58 mg/L (range, 2-195) before first-stage surgery. During the spacer interval, the CRP gradually decreased to a mean 17 mg/L (range, 2-186) before second-stage surgery. At final follow-up, the CRP normalised at a mean 8 mg/L (range, 1-28). The leukocyte count remained within normal limits before first- and second-stage surgery and at final follow-up.
At the time of final follow-up, three patients had died due to reasons unrelated to treatment. The mean follow-up period was 37 mo (range, 12-119 mo; median 31 mo). Recurrent infection was present in 11 patients (7 hips and 4 knees), of whom 4 had persistent infection with CoNS; the others had re-infection with other bacteria. In addition to the patients with persistent infection, we considered 2 more patients failure of treatment.
We treated 29 patients with two-stage revision arthroplasty of an infected total hip prosthesis, for which we used 8 functional articulating spacers and 21 prefabricated spacers (Table 1). Polymicrobial infection was present in four patients. Additional causative microorganisms were Propionibacterum acnes in one patient, Pseudomonas aeruginosa in one patient, P. aeruginosa and Enterococcus faecalis in one patient, and haemolytic Streptococci group C in one patient. The other 25 patients had monomicrobial infection with CoNS.
The spacer interval was complicated by dislocation of the spacer in 4 of 21 patients with a prefabricated spacer, and in 1 of 8 patients with a functional articulating spacer. We performed spacer revision because of dislocation in two patients with a prefabricated spacer. Closed reduction was performed in the other two patients with a prefabricated spacer and the patient with a functional articulating spacer. Because of persistent wound effusion, we performed spacer revision within 2 wk after first-stage surgery in four patients with a prefabricated spacer. No spacer exchanges were performed after more than 2 wk.
Second-stage surgery was performed at a median of 8 wk (range, 2-15 wk) after the first-stage procedure. During revision surgery, an uncemented modular femoral revision stem was used in 17 patients and a dual mobility cup was used in 8 patients. All other components used were primary cemented or uncemented stems and cups (head diameter, 32 mm). Postoperatively, 12 patients received antibiotic treatment during the first 2 wk until culture results were negative. Four patients received antibiotic treatment for 6 wk, five patients received antibiotics for 12 wk, and one patient received antibiotics for 26 wk. Patients who had resection arthroplasty of the hip received antibiotics during 6 wk in four cases and during 12 wk in the other patient. Two patients received lifelong suppressive antibiotic therapy: The first due to persistent CoNS infection and the latter due to re-infection with another bacteria.
At final follow-up, we treated 22 patients successfully and considered 7 patients as failures after a mean follow-up of 42 mo (range, 12-119; median, 31 mo). Of the seven patients considered failure of treatment, six were treated with a prefabricated spacer. Due to persistent infection, we eventually accepted a Girdlestone situation in five patients. Two patients received lifelong suppressive antibiotics.
We treated 15 patients with two-stage revision arthroplasty of an infected knee prosthesis, using 4 static and 11 dynamic spacers (Table 1). Polymicrobial infection was present in two patients. The additional causative microorganisms were E. cloacae in one patient and E. faecalis in one patient. The other 13 patients had a monomicrobial infection with CoNS. Spacer interval was complicated by spacer exchange because of persistent wound effusion in two patients with a static spacer. In one patient with a dynamic spacer, a quadriceps tendon rupture occurred peroperatively.
Second-stage surgery was performed at a median of 8 wk (range, 4-27 wk) after the first-stage procedure. During second-stage surgery, a hinged type prosthesis was implanted in 11 patients, a constrained prosthesis in 2 patients, and a primary prosthesis in 2 patients. All knee prostheses were cemented. Postoperatively, eight patients received antibiotic treatment during the first 2 wk until culture results were negative. Two patients received antibiotic treatment for 6 wk, two received antibiotics for 12 wk, and one received antibiotics for 26 wk. Two patients received lifelong suppressive antibiotic therapy, both due to persistent CoNS infection.
At final follow-up, we treated nine patients successfully and considered six patients as failures after a mean follow-up of 28 mo (range, 12-59; median 31 mo). Due to persistent infection of the knee, two patients treated with a static spacer underwent further surgical procedures. We performed a second two-stage revision procedure, which eradicated the infection in one patient and an arthrodesis of the knee in the other patient. Two patients treated with a dynamic spacer received lifelong suppressive antibiotics. We performed an above the knee amputation because of persistent pain in one patient, who was treated with a static spacer and arthrodesis of the knee because of insufficiency of the extension mechanism in one patient who was treated with a dynamic spacer. The latter two patients had no demonstrable infection during second-stage revision, but were considered to have failed treatment.
This study retrospectively evaluated the infection eradication rate after two-stage revision arthroplasty with the use of an antibiotic-loaded interval spacer of PJI of the hip and knee caused by CoNS. At final follow-up, infection was eradicated in 33 of 44 cases; however we considered 2 more cases as failure of treatment. Poor rates of infection eradication have been reported in cases with polymicrobial infection of the hip or knee[17]. In our series of six patients with polymicrobial infection, one patient failed treatment. Existence of polymicrobial infection did not seem to influence chance of infection eradication negatively; however, the number of polymicrobial infections was too small to draw definite conclusions.
The infection eradication rate was comparable for two-stage revision of the hip (22 of 29) and the knee (11 of 15). The incidence of obesity (body mass index over 30) was higher in the knee group compared to the hip group (8 of 29 vs 9 of 15 patients). Obesity is a known risk factor for PJI[18-20]. In this series of patients, obesity was not related to a higher risk of persistent infection after two-stage revision arthroplasty of the hip or knee. Recurrence of infection after two-stage revision arthroplasty was also not related to gender, age, smoking status, ASA-classification, or time interval between the first- and second-stage procedure. Functional articulating spacers of the hip and dynamic spacers of the knee seem to lead to lower risk of failure; however the sample size of our study was too small to draw definite conclusions.
One-third of patients (10 of 29 patients) in the group of two-stage revisions of the hip received a primary hip prosthesis due to a proximal femoral fracture. In the other patients, the primary procedure was performed due to osteoarthritis of the hip. In the Netherlands, annually 4% of total hip arthroplasties are implanted because of a femoral neck fracture[21]. This suggests that the risk of infection is higher in patients receiving total hip arthroplasty after a femoral neck fracture. Physicians need to be aware of the increased risk of infection when providing information about hip arthroplasty to patients with hip fractures. Efforts should be made to optimally prepare the patient preoperatively. Treatment of comorbidities causing trauma, the timing and duration of surgery, perioperative antibiotic prophylaxis, and soft tissue management may all influence the chance of periprosthetic infection after total hip prosthesis for a proximal femur fracture. Infection eradication rate after two-stage revision hip arthroplasty was similar in trauma and elective patients (7 of 10 patients vs 14 of 19 patients, respectively, without infection at follow-up).
A limitation of this study is reflected by the retrospective design. The number of patients included in this study was relatively low, which was caused by the scarcity of PJI requiring two-stage revision and the fact that in this study we only focused on CoNS infections. Treatment of patients treated before 2007 was more heterogeneous compared to patients treated after 2007 due to the implementation of stricter perioperative protocols concerning treatment of infected prostheses.
Current literature lacks high-quality studies determining optimal treatment strategy in case of specific causative microorganisms such as CoNS in PJI of the hip and knee. As prospective studies of PJIs are hard to perform due to the scarcity of prosthetic infections, a retrospective multicentre study combining groups of patients to achieve a greater number of patients with CoNS PJI can provide more evidence on how to treat this specific infection. Orthopaedic surgeons should consider treating their patients with a functional articulating spacer of the hip or a dynamic spacer of the knee, as these may improve infection eradication rate. Whether or not functional outcome after two-stage revision with a functional articulating spacer of the hip or a dynamic spacer of the knee is improved compared to their more static counterparts has yet to be studied.
Due to biofilm formation CoNS can be a difficult to treat organism in PJI. The results of this study show that infection eradication rate comparable to that of other causative pathogens may be achieved following two-stage revision arthroplasty of the hip and knee[22-24].
The authors acknowledge Eduard L.A.R. Mutsaerts and S. John Ham for their efforts in treating the patients included in this study.
Coagulase-negative Staphylococci (CoNS) are difficult-to-treat microorganisms in periprosthetic joint infections (PJIs) of the hip and knee. The resistance of these bacteria to antibiotics is increasing.
To date, no infection eradication rates of treatment for this type of specific infection have been reported.
To evaluate the infection eradication rate of two-stage revision arthroplasty for CoNS PJI of the hip and knee.
Retrospective cohort study of all patients treated with two-stage revision for CoNS PJI of a hip or knee prosthesis.
In 33 of 44 patients, the infections were eradicated at a mean of 37 mo after two-stage revision surgery of the hip and knee.
Two-stage revision surgery of the hip and knee for PJI infections with CoNS leads to infection eradication rate comparable to other causative pathogens.
Two-stage revision yields an acceptable infection eradication rate for treatment of CoNS infection of the hip and knee. Future studies should consider combining cohorts of patients from multiple centres to achieve larger cohorts of patients.
Manuscript source: Invited manuscript
Specialty type: Orthopaedics
Country of origin: Netherlands
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: DeSousa K, Peng BG, Widmer KH S-Editor: Yan JP L-Editor: Filipodia E-Editor: Liu MY
| 1. | Becker K, Heilmann C, Peters G. Coagulase-negative staphylococci. Clin Microbiol Rev. 2014;27:870-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1011] [Cited by in RCA: 991] [Article Influence: 90.1] [Reference Citation Analysis (0)] |
| 2. | Murgier J, Laffosse JM, Cailliez J, Cavaignac E, Murgier P, Bayle-Iniguez X, Chiron P, Bonnevialle P. Is the prognosis the same for periprosthetic joint infections due to Staphylococcus aureus versus coagulase-negative staphylococci? A retrospective study of 101 patients with 2-year minimum follow-up. Arch Orthop Trauma Surg. 2016;136:1357-1361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Mühlhofer HML, Deiss L, Mayer-Kuckuk P, Pohlig F, Harrasser N, Lenze U, Gollwitzer H, Suren C, Prodinger P, VON Eisenhart-Rothe R, Schauwecker J. Increased Resistance of Skin Flora to Antimicrobial Prophylaxis in Patients Undergoing Hip Revision Arthroplasty. In Vivo. 2017;31:673-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Zimmerli W, Trampuz A, Ochsner PE. Prosthetic-joint infections. N Engl J Med. 2004;351:1645-1654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2193] [Cited by in RCA: 2174] [Article Influence: 103.5] [Reference Citation Analysis (0)] |
| 5. | Parvizi J, Gehrke T. International consensus on periprosthetic joint infection: Let cumulative wisdom be a guide. J Bone Joint Surg Am. 2014;96:441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 6. | Lichstein P, Gehrke T, Lombardi A, Romano C, Stockley I, Babis G, Bialecki J, Bucsi L, Cai X, Cao L, de Beaubien B, Erhardt J, Goodman S, Jiranek W, Keogh P, Lewallen D, Manner P, Marczynski W, Mason JB, Mulhall K, Paprosky W, Patel P, Piccaluga F, Polkowski G, Pulido L, Stockley I, Suarez J, Thorey F, Tikhilov R, Velazquez JD, Winkler H. One-stage versus two-stage exchange. J Orthop Res. 2014;32 Suppl 1:S141-S146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Gehrke T, Alijanipour P, Parvizi J. The management of an infected total knee arthroplasty. Bone Joint J. 2015;97-B:20-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 161] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 8. | Citak M, Argenson JN, Masri B, Kendoff D, Springer B, Alt V, Baldini A, Cui Q, Deirmengian GK, Del Sel H, Harrer MF, Israelite CL, Jahoda D, Jutte PC, Levicoff E, Meani E, Motta F, Pena OR, Ranawat AS, Safir O, Squire MW, Taunton MJ, Vogely CH, Wellman SS. Spacers. J Arthroplasty. 2014;29:93-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Romanò CL, Gala L, Logoluso N, Romanò D, Drago L. Two-stage revision of septic knee prosthesis with articulating knee spacers yields better infection eradication rate than one-stage or two-stage revision with static spacers. Knee Surg Sports Traumatol Arthrosc. 2012;20:2445-2453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 120] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 10. | Veltman ES, Moojen DJ, Glehr M, Poolman RW. Similar rate of infection eradication for functional articulating, prefabricated and custom-made spacers in 2-stage revision of the infected total hip: A literature review. Hip Int. 2016;26:319-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 11. | Carijo JH, Courtney PM, Goswami K, Groff H, Kendoff D, Matos J, Sandiford NA, Scheper H, Schmaltz CAS, Shubnyakov I, Tan TL, Wouthuyzen-Bakker M. Hip and Knee Section, Pathogen Factors: Proceedings of International Consensus on Orthopedic Infections. J Arthroplasty. 2019;34:S381-S386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 12. | Mirza YH, Tansey R, Sukeik M, Shaath M, Haddad FS. Biofilm and the Role of Antibiotics in the Treatment of Periprosthetic Hip and Knee Joint Infections. Open Orthop J. 2016;10:636-645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Malhas AM, Lawton R, Reidy M, Nathwani D, Clift BA. Causative organisms in revision total hip & knee arthroplasty for infection: Increasing multi-antibiotic resistance in coagulase-negative Staphylococcus and the implications for antibiotic prophylaxis. Surgeon. 2015;13:250-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 14. | von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. BMJ. 2007;335:806-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3438] [Cited by in RCA: 6179] [Article Influence: 343.3] [Reference Citation Analysis (0)] |
| 15. | Pivec R, Naziri Q, Issa K, Banerjee S, Mont MA. Systematic review comparing static and articulating spacers used for revision of infected total knee arthroplasty. J Arthroplasty. 2014;29:553-7.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 16. | Diaz-Ledezma C, Higuera CA, Parvizi J. Success after treatment of periprosthetic joint infection: A Delphi-based international multidisciplinary consensus. Clin Orthop Relat Res. 2013;471:2374-2382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 312] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 17. | Tan TL, Kheir MM, Tan DD, Parvizi J. Polymicrobial Periprosthetic Joint Infections: Outcome of Treatment and Identification of Risk Factors. J Bone Joint Surg Am. 2016;98:2082-2088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 89] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 18. | Werner BC, Higgins MD, Pehlivan HC, Carothers JT, Browne JA. Super Obesity Is an Independent Risk Factor for Complications After Primary Total Hip Arthroplasty. J Arthroplasty. 2017;32:402-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 19. | Wagner ER, Kamath AF, Fruth KM, Harmsen WS, Berry DJ. Effect of Body Mass Index on Complications and Reoperations After Total Hip Arthroplasty. J Bone Joint Surg Am. 2016;98:169-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 153] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 20. | Kunutsor SK, Whitehouse MR, Blom AW, Board T, Kay P, Wroblewski BM, Zeller V, Chen SY, Hsieh PH, Masri BA, Herman A, Jenny JY, Schwarzkopf R, Whittaker JP, Burston B, Huang R, Restrepo C, Parvizi J, Rudelli S, Honda E, Uip DE, Bori G, Muñoz-Mahamud E, Darley E, Ribera A, Cañas E, Cabo J, Cordero-Ampuero J, Redó MLS, Strange S, Lenguerrand E, Gooberman-Hill R, Webb J, MacGowan A, Dieppe P, Wilson M, Beswick AD; Global Infection Orthopaedic Management Collaboration. One- and two-stage surgical revision of peri-prosthetic joint infection of the hip: A pooled individual participant data analysis of 44 cohort studies. Eur J Epidemiol. 2018;33:933-946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 71] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 21. | Nederlandse Orthopaedische Vereniging and Stichting LROI. LROI jaarrapportage 2014-Orthopedische Implantaten in Beeld, 2014. Available from: https://www.lroi.nl/base/downloads/lroi-jaarrapportage-2014.pdf. |
| 22. | Johnson AJ, Zywiel MG, Jones LC, Delanois RE, Stroh DA, Mont MA. Reduced re-infection rates with postoperative oral antibiotics after two-stage revision hip arthroplasty. BMC Musculoskelet Disord. 2013;14:123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 23. | Lange J, Troelsen A, Thomsen RW, Søballe K. Chronic infections in hip arthroplasties: Comparing risk of reinfection following one-stage and two-stage revision: A systematic review and meta-analysis. Clin Epidemiol. 2012;4:57-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 24. | Leung F, Richards CJ, Garbuz DS, Masri BA, Duncan CP. Two-stage total hip arthroplasty: How often does it control methicillin-resistant infection? Clin Orthop Relat Res. 2011;469:1009-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 122] [Article Influence: 8.7] [Reference Citation Analysis (0)] |