Copyright
©2014 Baishideng Publishing Group Inc.
World J Orthop. Jul 18, 2014; 5(3): 247-254
Published online Jul 18, 2014. doi: 10.5312/wjo.v5.i3.247
Published online Jul 18, 2014. doi: 10.5312/wjo.v5.i3.247
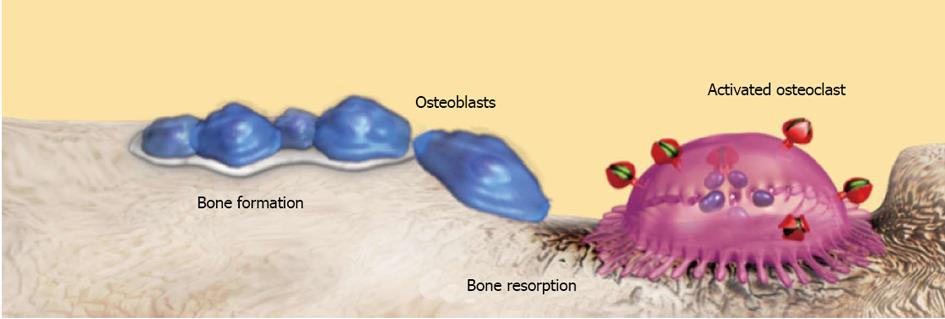
Figure 1 Osteoblasts and osteoclasts are close related in determining bone formation and bone resorption (bone turnover) that maintain the quantitative and qualitative characteristics of bone.
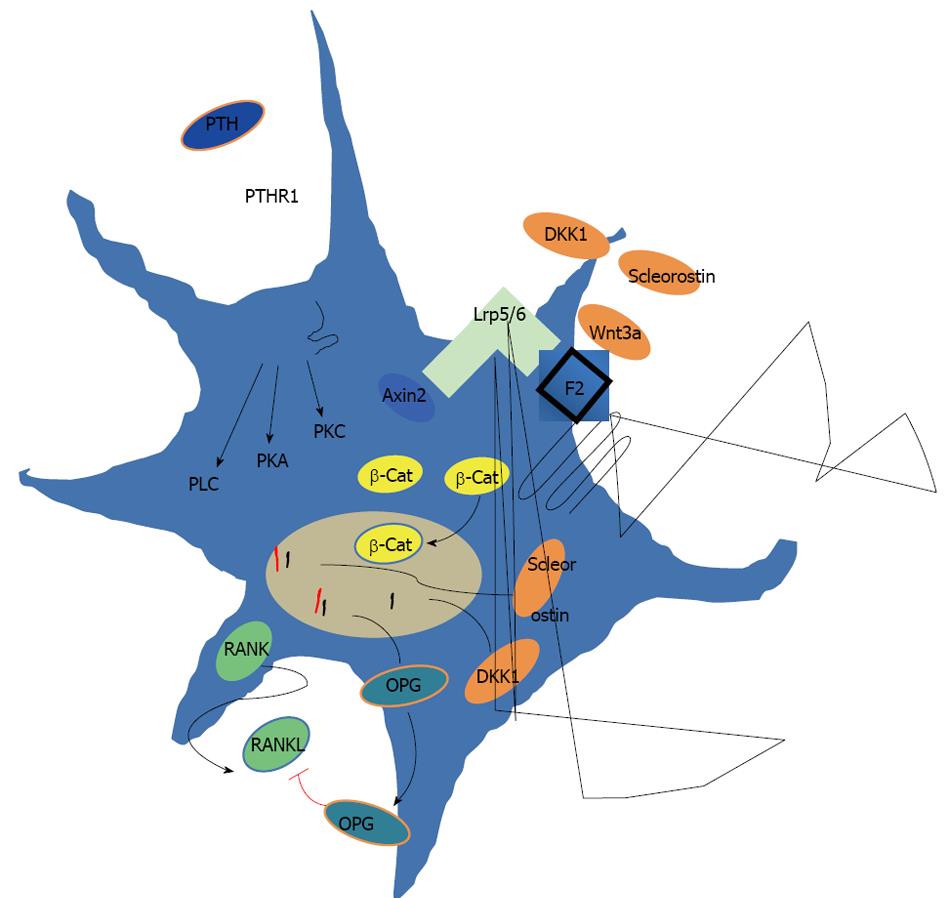
Figure 2 Signaling and cross talk of the parathyroid hormone and Wnt signaling pathways in the late osteoblast (osteocyte).
Parathyroid hormone (PTH) binds to its seven-transmembrane-spanning receptor and activates phosphatidyl inositol-specific phospholipase C (PLC), cAMP-dependent protein kinase A (PKA), and the protein kinase C (PKC) downstream signaling cascades, all contributing to the bone anabolic effect of PTH. In the late osteoblast activation of the canonical Wnt signaling pathway occurs upon simultaneous binding of the secreted glycoprotein Wnt3a to the seven-helix-receptor frizzled (Fz) family and the coreceptors Lrp 5/6. Binding of Wnt3a to Lrp5/6 changes the conformation of the cytoplasmic receptor domain, causing the recruitment of Axin2. β-Catenin accumulates in the cytosol and translocates into the nucleus, thereby stimulating the expression of the Lrp5/6 antagonists dickkopf-1 and sclerostin, and the RANKL inhibitor osteoprotegerin.
- Citation: Montagnani A. Bone anabolics in osteoporosis: Actuality and perspectives. World J Orthop 2014; 5(3): 247-254
- URL: https://www.wjgnet.com/2218-5836/full/v5/i3/247.htm
- DOI: https://dx.doi.org/10.5312/wjo.v5.i3.247