Published online Aug 15, 2015. doi: 10.4291/wjgp.v6.i3.73
Peer-review started: March 12, 2015
First decision: April 13, 2015
Revised: April 23, 2015
Accepted: July 11, 2015
Article in press: July 14, 2015
Published online: August 15, 2015
Processing time: 159 Days and 13.5 Hours
Given the increasing commercial and clinical relevance of probiotics, improving their stress tolerance profile and ability to overcome the physiochemical defences of the host is an important biological goal. Herein, I review the current state of the art in the design of engineered probiotic cultures, with a specific focus on their utility as therapeutics for the developing world; from the treatment of chronic and acute enteric infections, and their associated diarrhoeal complexes, to targeting HIV and application as novel mucosal vaccine delivery vehicles.
Core tip: Genetically engineered probiotic bacteria, with improved in vivo stress survival and persistence, have the potential to enhance, and in some instances replace, conventional prophylactic and therapeutic measures. This is particularly relevant in the developing world, where chronic and acute infections, and their associated sequelae impose a significant clinical and economic burden.
- Citation: Sleator RD. Designer probiotics: Development and applications in gastrointestinal health. World J Gastrointest Pathophysiol 2015; 6(3): 73-78
- URL: https://www.wjgnet.com/2150-5330/full/v6/i3/73.htm
- DOI: https://dx.doi.org/10.4291/wjgp.v6.i3.73
Probiotics are commensal organisms that can be harnessed for therapeutic benefit[1]. In acute infections probiotics may enhance protection mediated by commensal flora through direct antagonism, i.e., completion for niches and nutrients, or via the production of antimicrobials, such as bacteriocins[2]. In chronic clinical conditions, such as immuno-suppression, microbe-host signalling is likely more relevant to effective probiotic function. This bacterial-host dialogue within the gut lumen, functions to maintain an effective mucosal barrier while also priming the host for further responses to injury[3]. These health promoting benefits of probiotics, coupled with fact that they are cheap to produce, transport and store, makes them an attractive alternative to traditional therapies, especially in underdeveloped and war-torn territories.
Herein, I review key milestones in the development of probiotic based therapies, focusing particularly on problems encountered in developing countries such as acute and chronic enteric infections, as well HIV (AIDS) which continues to devastate sub-Saharan Africa[4]. Little or no access to appropriate medical care, often coupled with compromised immunity means that the malnourished are significantly more predisposed to infections by enteric pathogens, leading to incapacitating and dehydrating diarrhoea which in turn leads to a dramatic worsening of an already severely compromised nutritional status. Superimposing the HIV (AIDS) pandemic on an already distressed situation has created a state of affairs which needs to be urgently addressed[5].
Probiotic therapy, specifically the use of engineered probiotic strains, is a viable alternative to traditional approaches to alleviate suffering, to fight existing diseases and to protect against future infections[6]. Herein, I review the current state of the art in the design and application of probiotic cultures as therapeutics for the developing world.
Almost a third of the world’s population are malnurished[7], a quarter of which are children in the developing world. Malnutrition significantly reduces cell-mediated immunity, immunoglobulin A (IgA) concentrations and cytokine production[7]. This in turn leads to an increased risk of infection, accompanied by bouts of acute and recurrent diarrhoea – which further exasperates an already depressed nutritional status. Indeed, almost half of all diarrhoea-related mortalities are linked to malnutrition[8], while morbidity is also disturbingly high (approximately 4 times more episodes per child per annum in developing countries than in the developed world). Diarrhoeal disorders exists as either acute diarrhoea; associated with sudden onset infections, characterized by recovery within two weeks, and chronic diarrhoea which lasts more than two weeks and usually arises as a symptom of malnutrition or immunodeficiency[9].
Probiotic bacteria have been shown to considerably limit the incidence and duration of diarrhoea associated with both acute infectious illness and chronic episodes linked to malnutrition[10]. Shornikova et al[11] showed that Lactobacillus reuteri can reduce the duration of acute diarrhoea in infants by one full day. Guandalini[12] observed similar effects with Lactobacillus rhamnosus, which also decreased the duration of hospital stays. Furthermore, in addition to alleviating the symptoms of malnutrition and diarrhoea, probiotics have also been used to specifically target bacterial and viral pathogens[13-16]. Lb. casei Shirota, for example was shown by Ogawa et al[17] to reduce Escherichia coli O157:H7 colonization, while Pascual et al[18] observed complete exclusion of Salmonella enteritidis by Lb. salivarius. Furthermore, even more impressive effects have been observed with mixed probiotic cocktails. Casey et al[19] reported significant amelioration of clinical symptoms of Salmonella Typhimurium infection in pigs using LIVE5; a cocktail of two Lactobacillus murinus strains with one strain each of Lb. salivarius subsp. salivarius, Lb. pentosus and Pediococcus pentosaceous. Pigs administered this mixture exhibited significantly lower levels of Salmonella infection, reduced frequency, severity and duration of diarrhoea, and enhanced weight gain relative to animals fed on a skim milk placebo. Nisbet et al[20] observed similar decreases in Salmonella gallinarum mediated mortality using a commercial probiotic mixture, while Johnson-Henry et al[21] showed that a Lactobacillus mixture reduced inflammation in Helicobacter pylori-infected animals. Furthermore, clinical trials in colonized humans revealed significantly lower levels of H. pylori, and decreased adverse side effects[22]. Probiotics are also effective against rotavirus, an enteric virus which accounts for approximately 60% of all diarrhoeal episodes in developing countries[23,24]. Specifically, Lactobacillus casei subsp. casei strain GG (LGG) has been shown to stimulate a rotavirus-specific IgA antibody response, which may confer immunity against future rotavirus infections[25].
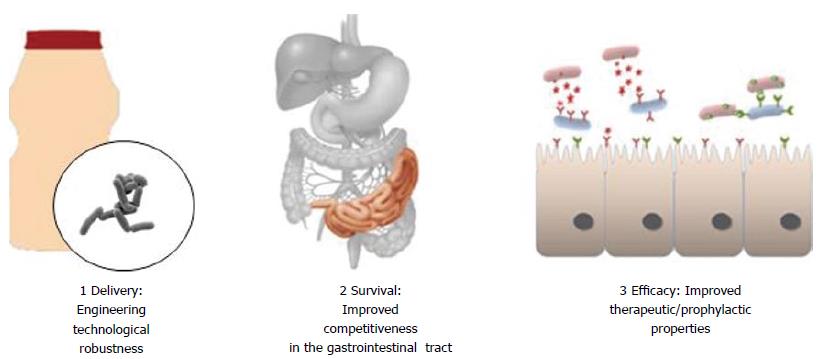
However, one of the most significant limitations in the clinical application of probiotics is that the most clinically relevant probiotics are often the most physiologically fragile. Improving probiotic stress tolerance is thus a biological imperative. Below we describe the application of the “Patho-biotechnology” concept for the development of improved probiotic cultures[26-29] (Figure 1).
Fluctuations in temperature and water availability (aw) are the most common stresses associated with food production[30,31]. The ability to overcome these stresses is thus an important criterion in the selection of commercially viable and clinically effective probiotics[32]. A common strategy to overcome temperature and osmotic stress is the accumulation of compatible solutes, such as the plant derived trimethyl ammonium compound glycine betaine[33], which serves to stabilise cellular function under stressful conditions[34].
Improving compatible solutes accumulation is thus an important first step in the development of more physiologically robust probiotic strains[35]. Several sophisticated mechanisms for compatible solute accumulation have evolved over time[34]. Indeed, the intracellular pathogen L. monocytogenes, which serves as a useful model for Gram positive osmotolerance[36], has three compatible solute uptake systems (BetL, Gbu and OpuC[37]), the first to be identified being the secondary betaine transporter BetL[38-40]. By cloning betL downstream of the nisin inducible promoter PnisA, we were able to assess the ability of BetL to contribute to probiotic survival under a variety of stresses[41,42]. As expected, the Lb. salivarius strain heterologously expressing BetL exhibited a significant increase in betaine uptake compared to the wild type, untransformed control. Indeed, the increased betaine uptake was sufficient to confer improved resistance to chill and cryotolerance, freeze-drying, spray-drying and barotolerance[41,43-46].
In addition to ex vivo stresses, probiotic bacteria must also overcome the in vivo defences of the host[30,31,47,48]. We demonstrated heterologous expression of BetL in Bifidobacterium breve UCC2003, significantly improved survival of the probiotic in gastric juice[49]. In support of this, Termont et al[50] also reported similar effects in a L. lactis strain expressing the E. coli trehalose synthesis genes, suggesting a novel protective role for compatible solutes in the gastric environment. Furthermore, we have reported roles for carnitine and proline in contributing to bacterial gastrointestinal survival[51-54]. Upon exiting the stomach, bacteria enter the upper small intestine where they are exposed to elevated osmolarity (equivalent to 0.3 mol/L NaCl). As was observed with L. salivarius[41], a significant osmoprotective effect was evident following BetL expression in B. breve, facilitating growth of the probiotic in conditions similar to those encountered in vivo. Furthermore, whilst stable colonisation of the murine intestine was achieved by oral administration of B. breve UCC2003, strains expressing BetL were recovered at significantly higher levels than the parent in the faeces, intestines and caecum of inoculated animals. Additionally, BetL significantly improved the clinical efficacy of the probiotic; resulting in significantly reduced levels of systemic infection following oral inoculation with L. monocytogenes, compared to the control.
In addition to improving physiological stress tolerance, “designer probiotics” have been designed to specifically inhibit enteric infections by blocking ligand-receptor interactions between the pathogen and/or secreted toxins and the host[55]. Blocking receptor binding prevents infection, while toxin neutralization dampens clinical symptoms. Engineered to express receptor-mimic structures on their surface[56], orally administered probiotics neutralize toxins and inhibit pathogen adherence to the intestinal epithelium. Examples of such constructs include an E. coli strain expressing a chimeric lipopolysaccharide terminating in a shiga toxin (Stx) receptor, which binds to and neutralises Stx1 and Stx2[56], as well those with receptor blocking potential against cholera toxin (Ctx) and Enterotoxigenic E. coli (ETEC) toxin LT[57,58].
As well as treating enteric infections, “designer probiotics” have also been enlisted to target HIV. Every day approximately 14000 people become infected with HIV, a majority of which are in developing countries. Rao et al[59] recently described the construction of an E. coli strain engineered to secrete HIV-gp41-haemolysin A hybrid peptides, which block HIV fusion and entry into target cells. This “live microbicide”, administered either orally or via rectal suppository, colonizes the gut mucosa creating a protective barricade against HIV infection for four weeks[60]. Other anti-HIV probiotics include a human vaginal isolate of Lactobacillus jensenii engineered to secrete two-domain CD4 which inhibits HIV entry into target cells and Streptococcus gordonii modified to produce cyanovirin-N, a HIV-inactivating protein originally isolated from cyanobacterium[61].
The most effective prophylaxis for infectious diseases is vaccination; resulting in the mobilisation of an immune response capable of specifically targeting invading pathogens[29]. In addition to the classical approach to vaccination, involving induction of acquired immunity to specific antigens, there is a growing awareness of the importance of innate immunity, associated primarily with our commensal microflora[2,62-64]. Indeed, optimal development and functioning of the mucosal immune response is dependent on microbial exposure early in post-natal life[65]. In the absence of such stimulation, development of the intestinal-associated lymphoid tissue is stunted and immune responses are suboptimal[66].
The development of efficient vaccine delivery vehicles remains challenging since traditional vaccines are typically based on either recombinant proteins or killed whole pathogens which, although safe, typically induce only weak immunity[67-69]. The alternative is to use viable or attenuated pathogens[15]. However, while this approach improves targeted delivery, it carries with it the possibility of reversion to virulence[70]. Using a patho-biotechnology based approach, probiotics are being engineered to function as novel vaccine delivery vehicles which, lack the possibility of reversion to virulence, and effectively stimulate both the innate and acquired arms of the immune response[68,69]. In line with this approach, Guimarães et al[71] engineered a Lactococcus lactis strain capable of delivering either DNA or protein into the epithelial cells of the small intestine. Heterologous expression of inlA, encoding a listerial eukaryotic cell adhesion factor, converted the otherwise non-invasive L. lactis strain into a safe and effective vaccine delivery platform. Furthermore, the addition of hlyA (encoding listeriolysin) to L. lactis inlA+ enables phagosomal escape within the macrophage allowing MHC I and II stimulation[72].
Mucosal vaccine delivery, achieved using probiotic delivery platforms, has significant medical and methodical advantages, particularly for use in the developing world, including: reduced pain and the possibility of cross contamination associated with intramuscular injection, no requirement for expensive medically trained personnel and no cold chain requirement - a significant advantage in the tropical climes most often associated with the developing world.
While conventional medical research continues to provide effective prophylactics and therapeutics, these often remain beyond the reach of the developing world. In this context, probiotics provide a viable and cost effective alternative to fighting infection, modulating the immune response and alleviating the symptoms of malnutrition and its associated sequelae, all of which will ultimately contribute to health and social gain, particularly in the developing world.
P- Reviewer: Jia L, Julian S, Rahimi R S- Editor: Ma YJ L- Editor: A E- Editor: Wu HL
| 1. | Fuller R. Probiotics in man and animals. J Appl Bacteriol. 1989;66:365-378. [PubMed] |
| 2. | Sleator RD, Hill C. New frontiers in probiotic research. Lett Appl Microbiol. 2008;46:143-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 43] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 3. | Madara J. Building an intestine--architectural contributions of commensal bacteria. N Engl J Med. 2004;351:1685-1686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 4. | Sleator RD. Not so neglected diseases. J Infect Dev Ctries. 2007;1:350-351. [PubMed] |
| 5. | Sleator RD. Probiotics -- a viable therapeutic alternative for enteric infections especially in the developing world. Discov Med. 2010;10:119-124. [PubMed] |
| 6. | Sleator RD. Probiotic therapy - recruiting old friends to fight new foes. Gut Pathog. 2010;2:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 7. | Solis B, Samartín S, Gómez S, Nova E, de la Rosa B, Marcos A. Probiotics as a help in children suffering from malnutrition and diarrhoea. Eur J Clin Nutr. 2002;56 Suppl 3:S57-S59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 8. | Mittal SK. Chronic diarrhea in tropics. Indian J Pediatr. 1999;66:S4-15. [PubMed] |
| 9. | Thapar N, Sanderson IR. Diarrhoea in children: an interface between developing and developed countries. Lancet. 2004;363:641-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 230] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 10. | Van Niel CW, Feudtner C, Garrison MM, Christakis DA. Lactobacillus therapy for acute infectious diarrhea in children: a meta-analysis. Pediatrics. 2002;109:678-684. [PubMed] |
| 11. | Shornikova AV, Casas IA, Mykkänen H, Salo E, Vesikari T. Bacteriotherapy with Lactobacillus reuteri in rotavirus gastroenteritis. Pediatr Infect Dis J. 1997;16:1103-1107. [PubMed] |
| 12. | Guandalini S. Treatment of acute diarrhea in the new millennium. J Pediatr Gastroenterol Nutr. 2000;30:486-489. [PubMed] |
| 13. | Sleator RD, Hill C. Engineered pharmabiotics with improved therapeutic potential. Hum Vaccin. 2008;4:271-274. [PubMed] |
| 14. | Sleator RD, Hill C. Designer probiotics: a potential therapeutic for Clostridium difficile. J Med Microbiol. 2008;57:793-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Sleator RD, Hill C. Rational design of improved pharmabiotics. J Biomed Biotechnol. 2009;2009:275287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Kechaou N, Chain F, Gratadoux JJ, Blugeon S, Bertho N, Chevalier C, Le Goffic R, Courau S, Molimard P, Chatel JM. Identification of one novel candidate probiotic Lactobacillus plantarum strain active against influenza virus infection in mice by a large-scale screening. Appl Environ Microbiol. 2013;79:1491-1499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 77] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 17. | Ogawa M, Shimizu K, Nomoto K, Takahashi M, Watanuki M, Tanaka R, Tanaka T, Hamabata T, Yamasaki S, Takeda Y. Protective effect of Lactobacillus casei strain Shirota on Shiga toxin-producing Escherichia coli O157: H7 infection in infant rabbits. Infect Immun. 2001;69:1101-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 92] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Pascual M, Hugas M, Badiola JI, Monfort JM, Garriga M. Lactobacillus salivarius CTC2197 prevents Salmonella enteritidis colonization in chickens. Appl Environ Microbiol. 1999;65:4981-4986. [PubMed] |
| 19. | Casey PG, Gardiner GE, Casey G, Bradshaw B, Lawlor PG, Lynch PB, Leonard FC, Stanton C, Ross RP, Fitzgerald GF. A five-strain probiotic combination reduces pathogen shedding and alleviates disease signs in pigs challenged with Salmonella enterica Serovar Typhimurium. Appl Environ Microbiol. 2007;73:1858-1863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 155] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 20. | Nisbet DJ, Tellez GI, Lowry VK, Anderson RC, Garcia G, Nava G, Kogut MH, Corrier DE, Stanker LH. Effect of a commercial competitive exclusion culture (Preempt) on mortality and horizontal transmission of Salmonella gallinarum in broiler chickens. Avian Dis. 1998;42:651-656. [PubMed] |
| 21. | Johnson-Henry KC, Mitchell DJ, Avitzur Y, Galindo-Mata E, Jones NL, Sherman PM. Probiotics reduce bacterial colonization and gastric inflammation in H. pylori-infected mice. Dig Dis Sci. 2004;49:1095-1102. [PubMed] |
| 22. | Gotteland M, Brunser O, Cruchet S. Systematic review: are probiotics useful in controlling gastric colonization by Helicobacter pylori. Aliment Pharmacol Ther. 2006;23:1077-1086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 160] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 23. | Cashman O, Lennon G, Sleator RD, Power E, Fanning S, O’Shea H. Changing profile of the bovine rotavirus G6 population in the south of Ireland from 2002 to 2009. Vet Microbiol. 2010;146:238-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 24. | Collins PJ, Martella V, Sleator RD, Fanning S, O’Shea H. Detection and characterisation of group A rotavirus in asymptomatic piglets in southern Ireland. Arch Virol. 2010;155:1247-1259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 25. | Heyman M. Effect of lactic acid bacteria on diarrheal diseases. J Am Coll Nutr. 2000;19:137S-146S. [PubMed] |
| 26. | Sleator RD, Hill C. Battle of the bugs. Science. 2008;321:1294-1295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Sleator RD, Hill C. Patho-biotechnology; using bad bugs to make good bugs better. Sci Prog. 2007;90:1-14. [PubMed] |
| 28. | Sleator RD, Hill C. ‘Bioengineered Bugs’ - a patho-biotechnology approach to probiotic research and applications. Med Hypotheses. 2008;70:167-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Sleator RD, Hill C. Patho-biotechnology: using bad bugs to do good things. Curr Opin Biotechnol. 2006;17:211-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 30. | Sleator RD, Hill C. Food reformulations for improved health: A potential risk for microbial food safety. Med Hypotheses. 2007;69:1323-1324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 31. | Sleator RD, Hill C. Molecular analysis of the microbial food safety implications of food reformulations for improved health. Foodborne Pathog Dis. 2008;5:499-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 32. | Watson D, Sleator RD, Hill C, Gahan CG. Enhancing bile tolerance improves survival and persistence of Bifidobacterium and Lactococcus in the murine gastrointestinal tract. BMC Microbiol. 2008;8:176. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 48] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 33. | Dreux N, Albagnac C, Sleator RD, Hill C, Carlin F, Morris CE, Nguyen-the C. Glycine betaine improves Listeria monocytogenes tolerance to desiccation on parsley leaves independent of the osmolyte transporters BetL, Gbu and OpuC. J Appl Microbiol. 2008;104:1221-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 34. | Sleator RD, Hill C. Bacterial osmoadaptation: the role of osmolytes in bacterial stress and virulence. FEMS Microbiol Rev. 2002;26:49-71. [PubMed] |
| 35. | Sleator RD, Francis GA, O’Beirne D, Gahan CG, Hill C. Betaine and carnitine uptake systems in Listeria monocytogenes affect growth and survival in foods and during infection. J Appl Microbiol. 2003;95:839-846. [PubMed] |
| 36. | Sleator RD, Gahan CG, Hill C. A postgenomic appraisal of osmotolerance in Listeria monocytogenes. Appl Environ Microbiol. 2003;69:1-9. [PubMed] |
| 37. | Wemekamp-Kamphuis HH, Wouters JA, Sleator RD, Gahan CG, Hill C, Abee T. Multiple deletions of the osmolyte transporters BetL, Gbu, and OpuC of Listeria monocytogenes affect virulence and growth at high osmolarity. Appl Environ Microbiol. 2002;68:4710-4716. [PubMed] |
| 38. | Sleator RD, Gahan CG, Abee T, Hill C. Identification and disruption of BetL, a secondary glycine betaine transport system linked to the salt tolerance of Listeria monocytogenes LO28. Appl Environ Microbiol. 1999;65:2078-2083. [PubMed] |
| 39. | Sleator RD, Gahan CGM B, Hill C. Analysis of the role of betL in contributing to the growth and survival of Listeria monocytogenes LO28. Int J Food Microbiol. 2000;60:261-268. [PubMed] |
| 40. | Hoffmann RF, McLernon S, Feeney A, Hill C, Sleator RD. A single point mutation in the listerial betL σ(A)-dependent promoter leads to improved osmo- and chill-tolerance and a morphological shift at elevated osmolarity. Bioengineered. 2013;4:401-407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 41. | Sheehan VM, Sleator RD, Fitzgerald GF, Hill C. Heterologous expression of BetL, a betaine uptake system, enhances the stress tolerance of Lactobacillus salivarius UCC118. Appl Environ Microbiol. 2006;72:2170-2177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 42. | Sleator RD, Wood JM, Hill C. Transcriptional regulation and posttranslational activity of the betaine transporter BetL in Listeria monocytogenes are controlled by environmental salinity. J Bacteriol. 2003;185:7140-7144. [PubMed] |
| 43. | Considine KM, Kelly AL, Fitzgerald GF, Hill C, Sleator RD. High-pressure processing--effects on microbial food safety and food quality. FEMS Microbiol Lett. 2008;281:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 212] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 44. | Considine KM, Sleator RD, Kelly AL, Fitzgerald GF, Hill C. Identification and characterization of an essential gene in Listeria monocytogenes using an inducible gene expression system. Bioeng Bugs. 2013;2:150-159. [PubMed] |
| 45. | Considine KM, Sleator RD, Kelly AL, Fitzgerald GF, Hill C. A role for proline synthesis and transport in Listeria monocytogenes barotolerance. J Appl Microbiol. 2011;110:1187-1194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 46. | Considine KM, Sleator RD, Kelly AL, Fitzgerald GF, Hill C. Novel listerial genetic loci conferring enhanced barotolerance in Escherichia coli. J Appl Microbiol. 2011;110:618-630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 47. | Sleator RD, Clifford T, Hill C. Gut osmolarity: a key environmental cue initiating the gastrointestinal phase of Listeria monocytogenes infection. Med Hypotheses. 2007;69:1090-1092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 48. | Culligan EP, Marchesi JR, Hill C, Sleator RD. Mining the human gut microbiome for novel stress resistance genes. Gut Microbes. 1990;3:394-397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 49. | Sheehan VM, Sleator RD, Hill C, Fitzgerald GF. Improving gastric transit, gastrointestinal persistence and therapeutic efficacy of the probiotic strain Bifidobacterium breve UCC2003. Microbiology. 2007;153:3563-3571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 77] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 50. | Termont S, Vandenbroucke K, Iserentant D, Neirynck S, Steidler L, Remaut E, Rottiers P. Intracellular accumulation of trehalose protects Lactococcus lactis from freeze-drying damage and bile toxicity and increases gastric acid resistance. Appl Environ Microbiol. 2006;72:7694-7700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 51. | Sleator RD, Wouters J, Gahan CG, Abee T, Hill C. Analysis of the role of OpuC, an osmolyte transport system, in salt tolerance and virulence potential of Listeria monocytogenes. Appl Environ Microbiol. 2001;67:2692-2698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 116] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 52. | Sleator RD, Gahan CG, Hill C. Identification and disruption of the proBA locus in Listeria monocytogenes: role of proline biosynthesis in salt tolerance and murine infection. Appl Environ Microbiol. 2001;67:2571-2577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 56] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 53. | Sleator RD, Banville N, Hill C. Carnitine enhances the growth of Listeria monocytogenes in infant formula at 7 degrees C. J Food Prot. 2009;72:1293-1295. [PubMed] |
| 54. | Feeney A, Sleator RD. Functional Screening of the Cronobacter sakazakii BAA-894 Genome reveals a role for ProP (ESA_02131) in carnitine uptake. Bioengineered. 2015;6:161-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 55. | Paton AW, Morona R, Paton JC. Designer probiotics for prevention of enteric infections. Nat Rev Microbiol. 2006;4:193-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 56. | Paton AW, Morona R, Paton JC. A new biological agent for treatment of Shiga toxigenic Escherichia coli infections and dysentery in humans. Nat Med. 2000;6:265-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 135] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 57. | Paton AW, Jennings MP, Morona R, Wang H, Focareta A, Roddam LF, Paton JC. Recombinant probiotics for treatment and prevention of enterotoxigenic Escherichia coli diarrhea. Gastroenterology. 2005;128:1219-1228. [PubMed] |
| 58. | Paton AW, Morona R, Paton JC. Neutralization of Shiga toxins Stx1, Stx2c, and Stx2e by recombinant bacteria expressing mimics of globotriose and globotetraose. Infect Immun. 2001;69:1967-1970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 51] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 59. | Rao S, Hu S, McHugh L, Lueders K, Henry K, Zhao Q, Fekete RA, Kar S, Adhya S, Hamer DH. Toward a live microbial microbicide for HIV: commensal bacteria secreting an HIV fusion inhibitor peptide. Proc Natl Acad Sci USA. 2005;102:11993-11998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 95] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 60. | Lagenaur LA, Berger EA. An anti-HIV microbicide comes alive. Proc Natl Acad Sci USA. 2005;102:12294-12295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 61. | Chang TL, Chang CH, Simpson DA, Xu Q, Martin PK, Lagenaur LA, Schoolnik GK, Ho DD, Hillier SL, Holodniy M. Inhibition of HIV infectivity by a natural human isolate of Lactobacillus jensenii engineered to express functional two-domain CD4. Proc Natl Acad Sci USA. 2003;100:11672-11677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 116] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 62. | Sleator RD, Shortall C, Hill C. Metagenomics. Lett Appl Microbiol. 2008;47:361-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 88] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 63. | Culligan EP, Hill C, Sleator RD. Probiotics and gastrointestinal disease: successes, problems and future prospects. Gut Pathog. 2009;1:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 64. | Sleator RD. The human superorganism - of microbes and men. Med Hypotheses. 2010;74:214-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 72] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 66. | Forchielli ML, Walker WA. The role of gut-associated lymphoid tissues and mucosal defence. Br J Nutr. 2005;93 Suppl 1:S41-S48. [PubMed] |
| 67. | Johnston CD, Bannantine JP, Govender R, Endersen L, Pletzer D, Weingart H, Coffey A, O’Mahony J, Sleator RD. Enhanced expression of codon optimized Mycobacterium avium subsp. paratuberculosis antigens in Lactobacillus salivarius. Front Cell Infect Microbiol. 2014;4:120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 68. | Johnston C, Douarre PE, Soulimane T, Pletzer D, Weingart H, MacSharry J, Coffey A, Sleator RD, O’Mahony J. Codon optimisation to improve expression of a Mycobacterium avium ssp. paratuberculosis-specific membrane-associated antigen by Lactobacillus salivarius. Pathog Dis. 2013;68:27-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 69. | Johnston C, Coffey A, O’ Mahony J, Sleator RD. Development of a novel oral vaccine against Mycobacterium avium paratuberculosis and Johne disease: a patho-biotechnological approach. Bioeng Bugs. 2010;1:155-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 70. | Roland KL, Tinge SA, Killeen KP, Kochi SK. Recent advances in the development of live, attenuated bacterial vectors. Curr Opin Mol Ther. 2005;7:62-72. [PubMed] |
| 71. | Guimarães VD, Gabriel JE, Lefèvre F, Cabanes D, Gruss A, Cossart P, Azevedo V, Langella P. Internalin-expressing Lactococcus lactis is able to invade small intestine of guinea pigs and deliver DNA into mammalian epithelial cells. Microbes Infect. 2005;7:836-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 72. | Bahey-El-Din M, Casey PG, Griffin BT, Gahan CG. Efficacy of a Lactococcus lactis ΔpyrG vaccine delivery platform expressing chromosomally integrated hly from Listeria monocytogenes. Bioeng Bugs. 2010;1:66-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |