Published online Dec 28, 2016. doi: 10.4329/wjr.v8.i12.902
Peer-review started: June 17, 2016
First decision: August 4, 2016
Revised: August 29, 2016
Accepted: October 22, 2016
Article in press: October 24, 2016
Published online: December 28, 2016
Processing time: 188 Days and 14.9 Hours
Since its introduction in the 1970s, computed tomography (CT) has revolutionized diagnostic decision-making. One of the major concerns associated with the widespread use of CT is the associated increased radiation exposure incurred by patients. The link between ionizing radiation and the subsequent development of neoplasia has been largely based on extrapolating data from studies of survivors of the atomic bombs dropped in Japan in 1945 and on assessments of the increased relative risk of neoplasia in those occupationally exposed to radiation within the nuclear industry. However, the association between exposure to low-dose radiation from diagnostic imaging examinations and oncogenesis remains unclear. With improved technology, significant advances have already been achieved with regards to radiation dose reduction. There are several dose optimization strategies available that may be readily employed including omitting unnecessary images at the ends of acquired series, minimizing the number of phases acquired, and the use of automated exposure control as opposed to fixed tube current techniques. In addition, new image reconstruction techniques that reduce radiation dose have been developed in recent years with promising results. These techniques use iterative reconstruction algorithms to attain diagnostic quality images with reduced image noise at lower radiation doses.
Core tip: The rapid increase in computed tomography (CT) utilisation has brought with it significant public concern with regards to the doses of ionising radiation delivered during scanning due to the fact that some experimental and epidemiological evidence has linked exposure to low-dose radiation to the development of solid organ cancers and leukaemia. It now seems that a threshold-model of risk might be more appropriate with the risk increasing exponentially once cumulative doses of 100 mSv or more are reached. Nevertheless, there is an inherent responsibility on the medical community to keep radiation doses “as low as reasonably achievable”. Each imaging procedure needs to be justified and optimised and the minimum radiation dose possible used to obtain a diagnostic CT should remain the goal in each clinical scenario.
- Citation: Power SP, Moloney F, Twomey M, James K, O’Connor OJ, Maher MM. Computed tomography and patient risk: Facts, perceptions and uncertainties. World J Radiol 2016; 8(12): 902-915
- URL: https://www.wjgnet.com/1949-8470/full/v8/i12/902.htm
- DOI: https://dx.doi.org/10.4329/wjr.v8.i12.902
Since its introduction in the 1970s, computed tomography (CT) has revolutionized diagnostic decision making[1,2]. It has resulted in better surgery, better diagnosis and treatment of cancer, better treatment after injury and major trauma, better treatment of stroke and better treatment of cardiac conditions[3,4]. CT has many advantages over other imaging modalities in that it can be performed in minutes and is widely available which can allow physicians to rapidly confirm or exclude a diagnosis with improved conviction. It has had a major impact on the field of surgery where it has decreased the need for emergency surgery from 13% to 5% and has almost made many exploratory surgical procedures extinct. The widespread uptake of CT in clinical practice has been shown to decrease the proportion of patients requiring inpatient admission[5,6]. The progressive year on year technological advances in CT have also helped to make it an increasingly appealing imaging modality with higher spatial resolution and shorter scanning times leading to vastly increased number of clinical applications, e.g., CT colonography, CT angiography, CT urography, etc.
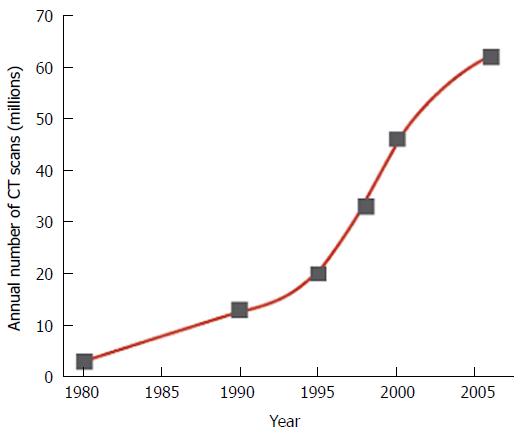
Given these advantages, it is no surprise that CT has seen an explosion in its utilization since its inception[7]. In 2007, it was estimated that around 62 million CT scans were being obtained each year in the United States, compared with around 3 million per year in 1980[8]. One of the major concerns associated with the widespread uptake of CT is the associated increased radiation exposure incurred by patients. A United States study in 2009 found that CT is now responsible for 75.4% of the effective radiation dose delivered from all imaging procedures, while it accounts for only 11% of X-ray based examinations[9]. This increased reliance on CT scanning has resulted in the cumulative per-capita effective radiation dose received from medical imaging in the United States to increase almost six-fold between the years 1980-2006[10] (from 0.5 mSv to 3.0 mSv) and medical imaging is now the largest source of radiation exposure to humans other than natural background radiation[11] (in 2009, it contributed to over 24% of the United States population’s radiation dose)[12]. Since the mid-1990’s there has been an annual increase of almost 10% in the utilization of CT scanning[7]. The rapid expansion in the utilization of fluoroscopic and interventional radiologic procedures has also helped to contribute to the increases in ionizing radiation delivered by the medical community[13,14]. Combine these guided procedures with the potential advent of CT-based screening programs (e.g., CT colonography[15], CT lung screening[16]) and there is an expectation that the reliance on CT scanning could continue to increase further in years ahead (Figure 1). This reliance on CT scanning is often further exacerbated by a lack of alternative imaging modalities, especially in smaller centres[17].
The rapid increase in CT utilisation has brought with it significant public concern[18] with regards to the doses of ionising radiation delivered during scanning given that some experimental and epidemiologic evidence has linked exposure to low-dose radiation to the development of solid organ cancers and leukaemia[19]. It is widely accepted that large doses of ionising radiation increase the likelihood that an individual will go on to develop cancer during their lifetime but the association between low-dose radiation (of the order used in standard diagnostic examinations) and oncogenesis is unclear. The link between radiation and the subsequent development of neoplasia has been largely based on extrapolating data from studies of survivors of the atomic bombs dropped in Japan in 1945[20] and on assessments of the increased relative risk of neoplasia in those occupationally exposed to radiation within the nuclear industry[21]. Using this method of extrapolation where small hypothetical risks are multiplied by huge patient numbers, Brenner et al[22] estimated that in the future 1%-2% of all cancers in the United States would occur secondary to the effects of ionising radiation delivered by medical imaging, while a similar study by Berrington de González et al[23] in 2009 predicted that 29000 additional cancers and 14500 additional deaths could be expected each year.
While there is little dispute that large exposures to ionizing radiation such as are seen in nuclear disasters place an individual at an exponentially increased risk of developing cancer (analysis of the fall-out from the Chernobyl disaster has also highlighted an increased risk in thyroid cancer in those children exposed in utero downwind of Chernobyl)[24] there is widespread disagreement as to level of cumulative radiation dose delivered by medical imaging which increases the risk of cancer. While many authors argue that a linear no-threshold (LNT) model applies to the association between radiation and oncogenesis[22,25,26] others argue that a practical threshold exists below which the risks of cancer are no greater than an individual’s background spontaneous risk[27,28]. A recent report has even suggested that exposure of individuals to low-dose radiation may elevate the immune response and thereby protect the individual from cancer, a concept known as hormesis[29,30]. The assertion that radiation induces cancer is a very broad statement. Particular organ systems are distinctly radiosensitive while others have more robust defences against the effects of ionising radiation. For example, organs such as the oesophagus, breast and bladder are particularly susceptible while organs such as the rectum, pancreas and prostate are much less sensitive[31].
The validity of the linear no-threshold model has come under even further scrutiny in more recent times[32]. An analysis of the Radiation Effects Research Foundation (REFR) data (which has followed the victims of the Hiroshima and Nagaskai attacks) compared cancer incidence in these cities with other Japanese cities which were not affected by the nuclear bombings. The group specifically looked at the incidence of colon cancer (commonly used as a cancer indicator in the Japanese population) and found that its incidence was not increased in those who received doses of radiation less than about 100 mSv[25]. It is suggested that ascribing cancer risks to radiation exposures of less than 100 mSv is confounded by other risk factors for malignancy within an individual population[28]. The REFR data was more consistent with the threshold-quadratic model of radiation-induced cancer than with a LNT model. Another issue in extrapolating experience of atomic bomb survivors in Japan to those exposed to ionising radiation in the medical setting is the inherent baseline differences in cancer risk amongst Japanese individuals vs populations of a different ethnic distribution (for example, stomach cancer is 10 times more prevalent in the Japanese community compared with United States subject, while breast cancer is three times more prevalent in the United States than in Japan)[25].
The linear-no-threshold model was initially adopted to assess radiation risk not because it has a solid biological and scientific foundation but because of its simplicity and its conservative nature (i.e., the model is more likely to over-predict rather than under-predict the neoplastic risk associated with imaging)[33]. As far back as 1946, when Muller accepted his Nobel Prize for his work investigating genetic mutations in Drosphilia generated by the effects of X-ray (proposing the LNT model as a basis for predicting oncogenesis), there has been disagreement with regards to this model[34]. International societies are beginning to doubt its validity. The Health Physics Society concluded that at doses below 50-100 mSv “risks of health effects are either too small to be observed or are non-existent”[35]. The American Association of Physicists in Medicine supported this view stating that at dosages less than 50 mSv for single procedures and less than 100 mSv for multiple procedures the “predictions of hypothetical cancer incidence and deaths in patient populations exposed to such low doses are highly speculative and should be discouraged”. Most tellingly, the United Nations Scientific Committee on the Effects of Atomic Radiation, one of the foremost international authorities on the effects of radiation in health, have also supported this position and have detailed that “statistically significant elevations in risk are observed at doses of 100 to 200 mGy and above” and that at dose ranges less than this no definitive risk can be ascribed to ionising radiation[31]. Doses of ionizing radiation delivered by common radiological procedures are outlined in Table 1[36].
| Examination | Average effective dose (mSv) | Values reported in literature |
| Posterioranterior study of chest | 0.02 | 0.007-0.05 |
| Head CT | 2 | 0.9-4.0 |
| Thorax CT | 7 | 4.0-18.0 |
| CT Pulmonary angiogram | 15 | 13.0-40.0 |
| Abdomen CT | 8 | 3.5-25 |
| Pelvic CT | 6 | 3.3-10 |
| Coronary angiography | 16 | 5.0-32 |
While previously it had been insisted that even low doses of radiation were associated with risk of oncogenesis with a linear increase in risk with increased exposure, it now seems that a threshold-model of risk might be more appropriate with the risk increasing exponentially once cumulative doses of 100 mSv or more are reached[37]. This, however does not negate the danger associated with radiation or allow complacency when deciding on the validity of an indication for a particular scan. In patients with long-term chronic medical conditions, for instance, the requirement for repeated imaging makes them more likely candidates for incurring radiation exposure in the range of > 100 mSv. In a study of Crohn’s patients (this patient subgroup have an increased risk of small bowel lymphoma at baseline)[38] over a 15-year period, it was shown that 16% of these patients had radiation exposure of > 75 mSv[39] and a similar study assessing maintenance haemodialysis patients found that 13% of this population experienced a cumulative dose of > 75 mSv over a median follow-up of 3.4 years[40]. In critically ill trauma patients the cumulative effective dose delivered to each patient averages 106 ± 59 mSv[41] (although in this patient group the risks of avoiding imaging usually far outweigh the potential risk of future malignancy). Given that most CT studies can average at two to three imaging phases per study the doses incurred by each individual exam can quickly accumulate, especially in the patient with chronic medical complaints requiring ongoing radiologic investigation.
A simple dismissal of the linear-no-threshold model has engendered controversy since the recent publication of prospective data involving a large cohort study of paediatric patients in the United Kingdom who had undergone at least once CT scan between 1985 and 2002, when they were younger than 22 years of age. This data, albeit within the paediatric population, has been the first to suggest that medical imaging and the associated radiation exposure does indeed predispose to the development of cancer[42] and that the link is not just a speculative one based on extrapolation from prior disasters or occupational exposure in the nuclear industry. Pearce et al[42] and his team highlighted a linear association between the radiation dose to the brain and brain tumour risk and a similar association between doses received by the bone marrow and the development of leukaemia[43,44]. The authors chose to follow the incidence of these tumours following radiation therapy as these have been the malignancies which have been observed in irradiated children. These data were validated by the work of Mathews et al[45] who found a 24% increase in cancer incidence in a paediatric population exposed to a CT scan at least one year before a cancer diagnosis and followed up for 9.5 years. While these reports have helped to clarify the situation in the paediatric population the effects of radiation exposure in the adult population is less clear and whether or not this data can be directly applied to adult patients is ambiguous given that: (1) for any given CT examination, the doses delivered to adults is smaller than their paediatric counterparts[46,47]. The effective dose delivered to a neonate when assessing a particular anatomic site can be double those which an adult will receive for the same investigation[48]; and (2) children have been shown to have an inherently higher sensitivity to the effects of ionising radiation[20,48,49].
What is particularly concerning about the findings of these investigators is that it is within the paediatric population the expansion in CT utilisation is increasing at the most significant rate[50,51]. For example, between the years of 1991-1994 there was an increase of 63% in the utilisation of CT examinations in children less than 15 years of age[52]. This has been driven by a decrease in the scanning time for CT which reduces the need for sedation in younger or uncooperative children[53]. Conversely, despite the risks of radiation exposure in this sensitive cohort, the use of CT has had dramatic benefits in the paediatric population. Between 1990-2007, the expansion in utilisation of CT decreased the negative appendectomy rate from 23% to 1.7% with an associated decrease in the number of operations performed[54].
While the dangers of radiation exposure in the extremely young have been highlighted by recent population studies the situation with regard to the foetus in pregnant patients remains uncertain. While physicians have been demonstrated to have a poor understanding regarding the risks of imaging in pregnancy[55,56], this is likely due to the fact that there is no solid scientific evidence regarding the exact dangers. Data in animal studies has demonstrated teratogenicity but the doses used in these studies were much higher than those used in diagnostic scanning[57]. Studies of individuals exposed in utero at Hiroshima and Nagasaki have demonstrated growth restriction, microcephaly, mental retardation and increased risk of seizures from high dose radiation exposure[58,59]. While protocols exist which direct the need for scanning in pregnant or potentially pregnant patients these are primarily based on the linear no-threshold model rather than a specifically defined carcinogenic risk[60-62]. These protocols advocate minimising the radiation dose to which the foetus is exposed and concentrating the dose on the anatomy of interest; for example in suspected appendicitis, the scan volume should be restricted to include only potentially diagnostic areas and dual pass studies should be avoided[63,64]. Clearly, the use of imaging in pregnancy and particularly the use of CT always evokes enormous anxiety and is usually met with reluctance among radiologists and radiographers/radiology technologists. However, as in all clinical situations balancing of risk vs benefit is required based on best available evidence and considering how diagnostic information which may be gained by imaging may change management and improve clinical outcome vs potential risk to fetus and mother as a result of radiation exposure. The use of ultra-low dose protocols in pregnancy is therefore vital, until higher level evidence is available to inform decisions regarding imaging in pregnancy.
While we know that ionising radiation confers certain risks to a patient, news media can sensationalise and exaggerate the potential adverse effects of radiation on carcinogenicity[65] which can induce anxiety in patients, particularly parents of children undergoing investigation[66,67]. In most instances the benefits of performing CT completely outweigh the potential risks. Medical doctors are increasingly encountering difficult situations when patients refuse CT scanning in clinical scenarios when CT scanning is clearly required; an example of this would be when a patient with newly diagnosed cancer who requires a CT for staging, declines the exam based on perceived risks associated with radiation exposure. Despite media coverage, patient understanding of the exact risks associated with CT scanning can, at times, be poor. Popular media have a tendency to focus on perceived and sometimes sensationalized dangers associated with radiation exposure associated with CT scanning while ignoring the enormous benefits in terms of expeditious and accurate diagnosis. Occasionally, excessive focus or lack of balance in the reporting of very rare incidences of error leading to extremely excessive radiation exposures from CT scanning as happened when it was discovered that one centre had been erroneously exposing patients to radiation doses eight times of normal during CT perfusion scanning[68].
On the other hand, patients have been shown to underestimate the relative amount of radiation delivered by CT scanning and, surprisingly given media coverage, underestimate the carcinogenic potential of exposure to ionizing radiation. In fact, one study has shown that patients will often have a higher degree of faith in their treating physician if they undergo computed tomography scanning as part of their work-up[69].
There is no question that there is a requirement for better patient education by imaging providers prior to the performance of CT scans. For example, it has been shown that 93% of adults referred for abdominal CT did not receive any information regarding the potential risks of this procedure and that only 3% of adult emergency patients are even aware of the potential association between CT and carcinogenesis[70]. Despite the inherent risks associated with paediatric CT[71] there is often reluctance on the part of physicians to convey the dangers associated with radiation for fear of confusing or upsetting parents with this information[72]. Striking a balance is difficult as the exact risk associated with radiation exposure in the range associated with CT scanning to patients, and particularly in children is unclear. However, informing parents about the slight risks associated with CT has not been shown to affect their willingness to allow their child to undergo scanning[73]. A balanced discussion of risks vs benefits with parents about the risks is paramount[74,75] as pressure is often exerted on physicians by parents encouraging the utilisation of CT in order to expedite diagnosis[76], without a thorough awareness of the dangers associated with this scanning. There is a need for multidisciplinary discussion involving experts in many disciplines (including radiology, radiation biology, medical physicist, public health physicians) so that a consensus can be agreed to guide physicians in providing advise to patients of varying ages with regard to risk associated with CT scanning. Proper counselling and education can help parents become more willing to accept a more conservative strategy[73]. Despite limited knowledge amongst some physicians regarding the carcinogenic potential of CT scanning there are concerted efforts amongst radiologists and physicists to reduce radiation exposure through imaging to patients. Using newer technologies, and strategies such as iterative reconstruction, radiation exposures associated with CT scanning are diminishing incrementally[77].
Difficulty arises when balancing the immediate need for diagnosis with the unlikely potential for harm associated with a CT scan. To this effect, there tends to be a reliance on the individual health care providers to be cognisant of potential dangers and to minimize patient exposure to “as low as reasonably achievable”. There can be a lack of recognition from health care workers, however, regarding potential dangers associated with CT. A United States study of health care providers found that less than 50% of radiologists and only 9% of emergency department personnel were aware that there was a potential association between CT and the development of malignancy[70]. Data have also shown that many physicians are also unaware of the doses of radiation associated with individual examinations[78,79]. A systematic review on physicians’ knowledge of radiation exposure and risk found that there was often a “low level of knowledge and radiation risk awareness”[80]. An assessment of American paediatric surgeons found that 53% of all respondents thought that the lifetime risk of cancer was increased from exposure to one abdomino-pelvic CT scan, although 75% underestimated the dose delivered by this scan compared with a chest X-ray. The report also found that the majority of paediatric surgeons did not discuss the potential risks associated with these scans with their patients[81].
Poor physician awareness has also been observed in the United Kingdom and other parts of the EU[79], where appreciation of the consequences of radiation exposures was similar to the United States with most underestimating the dose of radiation delivered by common radiological investigations[78,82]. Similarly, in an Australian cohort of doctors, it has been shown that the “knowledge of radiation exposure from medical imaging is poor[83]”. It has also been highlighted that not only is there deficient knowledge amongst doctors regarding radiation dose incurred through imaging but that radiation dose is often not considered to be an important consideration when referring for radiological investigation[84]. The reasons why there is such a poor understanding amongst clinicians regarding the dangers associated with radiation could be explained by a lack of training at undergraduate and postgraduate level[85]. It has clearly been shown that there is a lack of awareness at undergraduate level[86].
Research in the postgraduate population has found that there is often limited focus on radiation safety and radiation protection within training programmes and have highlighted the importance of increased education initiatives in this area, both within radiology and other specialities[87]. Teaching of radiology at an undergraduate level and delivery of dedicated radiation protection education improves student’s awareness.
While we may not be certain as to the exact oncogenic potential of ionising radiation there is an inherent responsibility on the medical community to keep radiation doses “as low as reasonably achievable (ALARA)”. Each imaging procedure needs to be justified and optimised and the minimum radiation dose possible used to obtain a diagnostic CT should remain the goal in each clinical scenario. With improved technology, significant advances have already been achieved with regards to radiation dose reduction. The dosage delivered from a combined CT study of the abdomen and pelvis has declined by a factor of between two and three since the 1980s due to a number of different technological innovations[88]. However despite these technological advances and emphasis on the ALARA principle, radiation exposure has been shown to vary over a tenfold range in clinical practice for the same investigation, depending on variable parameters[23]. This type of variation can exist both within and between different institutions with wide discrepancies in average dose reported[89]. While standards and limits exist for health care workers and those routinely exposed to radiation occupationally (e.g., nuclear workers) there is currently no legal requirement for routine monitoring of cumulative effective radiation dose which patients may be exposed to during the diagnostic process[90-92].
Integration of hospital PACS systems on a national and international level would help to allow cumulative radiation exposure for each patient to be tracked. This type of database is currently being developed by the scanning industry (GE healthcare’s Dosewatch® system being an example of this). These platforms also allow optimisation tools which can be utilised by both radiographers and radiologists to try to minimise radiation exposure while, at the same time, maximising the clinical information which will be attained by each scan and limiting the risk of duplicating scans which have already been carried out at other institutions. Defined exposure limits can be stipulated for each type of scan and the technology will inform the physician if these pre-defined limits are exceeded. This would also allow departments the opportunity to audit and streamline their practices. Also, this online collection of radiation dose data associated with imaging procedures, will alert individual departments to sporadic incidences of high radiation exposures and allow immediate action to prevent large cohorts of patients from suffering very high radiation exposures as a result of diagnostic imaging.
Scanning techniques can be optimally adjusted (Table 2) in order to try to achieve an acceptable image at lower exposure level. Dose reduction can be achieved via a wide variety of means[93] as below.
| Current dose reductionstrategies in CT scanning | Dose reduction strategies gaining interest[92] |
| Solid state scintillating detectors | Manual/automated adjustment of scanner output according to patient size via: Tube current modulation; Selection of the most dose-efficient tube potential |
| Electronic circuits with lower levels of background noise | Iterative reconstruction methods |
| Multi-detector row arrays | Increased spiral pitch or non-spiral methods in cardiac CT |
| More powerful X-ray tubes and generators | |
| Beam shaping filters which vary the X-ray intensity across the patient cross section |
Tube current modulation and automatic exposure control[94]
Different patients, depending on their size, will all require different radiation doses and the most basic feature which can be modulated in each patient is the tube current[76]. For example, the amperage utilised in paediatric scanning should be significantly lower than that utilised in their adult counterparts[95] (and needs to be higher in obese patients). The tube current should be modulated based on the overall attenuation of the anatomic area being assessed[96]. Other techniques, such as ECG based current modulation can be used to help reduce the dose during cardiac CT[94,97]. Automatic exposure control, is a relatively new technique, which modulates the tube current during an individual scan based on the different attenuations of different anatomic regions. This also has the added advantage of delivering the optimal dose to achieve the optimal diagnostic image[98]. Radiologists can define the quantity of noise, which is acceptable to individual clinical scenarios, prior to the scan thus aiding the difficult task of balancing of image quality and radiation exposure.
Strategies to design an ideal tube potential for individual patient sizes and different diagnostic tasks have been published and these have been demonstrated to reduce doses by 70% for the chest and by 40% for the abdomen[99]. In the case of cardiac CT, electrocardiographic based tube current modulation can allow doses to be reduced by levels of between 30%-90%[97]. Further advances using a dual source CT system allows dose reductions by a factor of up to 10-12[100].
Iterative reconstruction has been one of the most significant advances in dose reduction technology in CT scanning in recent times. This type of technology, when used in conjunction with or in place of filtered back projection, may improve noise and spatial qualities within the image[101]. Iterative reconstruction techniques allows images of improved quality to be acquired at significantly lower radiation doses[102]. As technology and software continues to improve it is likely that iterative reconstruction algorithms will progress concurrently[103].
This technique has the potential to optimise quality of acquired image by eliminating noise and have been demonstrated to substantially reduce radiation dose[104,105].
Low dose strategies for abdominal CT scanning in children and young adults have been shown to be non-inferior to standard dose CT with respect to negative appendectomy rates[108,109]. These low dose strategies can use up to four times less radiation than the standard dose protocol.
Using a large number of thin adjacent CT slices can result in significant increases in radiation dose to the patient. Multi-slice CT scanners also deliver considerably more radiation dose due to scan overlap, positioning of the CT scanner in closer proximity to the patient and increased scatter radiation[110]. There is therefore an important balance to be met when selecting a slice small enough to achieve the optimal diagnostic image and large enough to ensure that the radiation dose delivered is acceptable[111].
All too often during image acquisition in CT the area being scanned includes extra images which are outside the field of original interest. For example, one study found that when assessing the utilisation of abdomino-pelvic CTs that extra images above the diaphragm were obtained in 97% of cases and that images below the symphysis pubis were obtained in 94% of patients[112]. This equated to an additional 1280 images in 106 patients and while the images provided additional radiation exposure in each patient there was little additional diagnostic information in the majority of these cases. Maintaining the field to only the area of interest can allow smaller cumulative dosing and potentially improved images via focused imaging[113].
Automated prompts and advice as part of online radiology ordering systems can help to reduce the number of low utility examinations carried out[114] (one study demonstrated that this type of system can reduce the number of low utility examinations threefold)[115].
Typical CT urography protocols have required multiple image acquisitions to obtain the unenhanced, contrast-enhanced nephrographic, and contrast-enhanced excretory phase images. This method of multiple image acquisition requires a significant radiation burden (quoted between 15-35 mSv)[116,117]. However, the utilisation of split bolus protocols can significantly reduce this burden and exposes the patient to doses similar to that experienced in standard unenhanced and contrast enhanced abdomino-pelvic CT[118].
Rational scanning: The strategies outlined above can play a huge role in minimising the dose administered to the patient during various scanning procedures. However, the best dose reduction strategy is to avoid needless scanning. Unfortunately, it has been shown that large numbers of scans are undertaken each year which are lacking in a valid clinical indication[119]. In fact, it has been suggested that perhaps 20%-40% of all CT scans could be avoided if decisions to scan were based on available guidelines[120,121]. In the paediatric population it has been shown that one third of all CT scans could be replaced by alternative approaches or not performed at all[122] and questions have also been raised regarding the routine use of CT for diagnosing appendicitis within the same population, despite its impressive results in reducing negative appendectomy rates[123]. There is scope for replacing or reducing CT in favour of other diagnostic modalities. Magnetic resonance imaging and ultrasound have the benefit of not exposing the patient to any ionizing radiation but their utility is compromised by availability [in the case of magnetic resonance imaging (MRI)][124,125] and image quality (in the case of ultrasound[126]. Also in some clinical scenarios, MRI does not offer equivalent diagnostic information when compared to CT. Decision support software programmes which rate the appropriateness of a CT scan as it is ordered by a physician, are difficult to develop, but have shown impressive reductions in the expansion of CT scanning[114]. Given that between 20%-40% of CT scans are ordered inappropriately as per evidence based guidelines[127], the introduction of these types of initiatives to encourage physicians to re-assess the clinical necessity for each scan is encouraging. The American College of Radiologists have recognised the need for thorough guidelines to assist physicians in deciding when particular scans should be utilised[128]. However, a caveat to the introduction of these types of decision support is that the application of a no-denial policy on radiological imaging, surprisingly, did not result in increased utilisation of imaging modalities[129].
Of course when imaging is clinically indicated then the benefit-risk balance is almost always overwhelmingly in favour of imaging[128,130]. However, all too often the decision to image is based on time constraints, medico-legal concerns or patient preference. There is, as yet, no study which attempts to quantify and assess the risk-benefit ratio for radiological investigations and responsibility lies with the referring physician and radiologist[131]. The need to optimise clinical decision making with regards to imaging therefore needs to be guideline based as this alone has the potential to reduce the influence of convenience factors[132]. The risk/benefit ratio is individual to each patient. The following factors contribute to oncogenic risk from radiation.
Genetic considerations: Certain populations and individuals may be more radiosensitive and have more of a propensity to develop cancers post radiation exposure[133]. For example, some patient groups with a genetic abnormality which predisposes to cancer have been shown to be more sensitive to the effects of radiation[134,135].
Age at exposure: The BEIR VII report demonstrated the relationship between the life-time attributable risk of cancer incidence and age at exposure, showing that the risks of carcinogenesis was much higher the earlier that patient was exposed to high doses of radiation[21]. Older patients undergo the majority of medical imaging but limited life expectancy reduces risk of radiation induced cancers[21]. Criteria for imaging in these patients should not necessarily be the same as for those for younger patients with curable disease[136]. The longer post-radiation life expectancy in the paediatric population allows greater scope for the generation of malignancy and this fact has been borne out by recent population based studies from the United Kingdom[42] and Australia[45].
Sex: There appears to be a trend towards a higher incidence of cancer in females post exposure to radiation as opposed to men (even with similar exposures to radiation)[137].
Illness: Many patients who undergo repeated imaging while being treated for illness likely to reduce life expectancy. Oncogenic effects of this imaging radiation are unlikely to to materialise[136].
Fractionation and protraction of exposure: In general, it is believed that there is a greater risk from high doses of radiation delivered over a short time period in comparison with the same (or lower doses) delivered over a protracted course due to the influence of DNA damage repair[138]. However, the influence of the cumulative dose being delivered over a longer period has been suggested to be, surprisingly, small[139,140].
The reality is that rational scanning will rely on the appropriate knowledge base amongst physicians and trainees. Therefore, the role of education of medical staff, both at undergraduate, postgraduate and even more senior level cannot be underestimated given the shortcomings in knowledge of radiation exposure identified above. These types of educational initiatives have previously shown to be successful in reducing scanning numbers when implemented appropriately[141].
Clarity regarding the association between radiation exposure and oncogenesis is, as yet, not fully elucidated. However, despite this, the goal when imaging patients should always be to use a dose that is “as low as reasonably achievable”. Imaging, irrespective of the risk, should only be used when the potential clinical benefit outweighs the potential risk. The three fundamental principles of radiation which are laid out by the International Commission of Radiologic Protection include[142]: (1) justification; (2) dose optimization; and (3) dose limitation.
There is a responsibility to adhere to these fundamental principles. Given that it has been shown that low-dose protocols do not impact diagnostic yield, such protocols need to become the standard[108]. Recent data has shown that a single scan has low risk but given CT expansion cumulative doses can escalate. The extrapolation of small carcinogenic risks in the individual to cumulative cancer figures in the population is often sensationalized by the popular media resulting in significant distress and anxiety amongst the public, which can make patients and their families reluctant to undergo scans which may be in their best interests.
The future of radiation optimisation will include education of physicians and patients. Such initiatives include the Image Gently® and Image Wisely® campaigns. Image Gently® provides information regarding paediatric population radiation safety to parents and physicians and guides dose optimisation[143,144]. The Image Wisely® campaign promotes radiation safety in the adult population and has developed an honour roll for facilities and associations who have pledged to “image wisely” within their practice[145]. The Image Gently® initiative has been further developed to include specific guidance on paediatric interventional procedures under the title of Step Lightly®[146,147], In response to the Cedar-Sinai controversy in the United States, the Food and Drug Administration has also launched a national initiative to reduce unnecessary radiation exposure to patients[148]. It is apparent that physicians are not effectively discussing the potential risks of radiation exposure with their patients, however small[149]. When potential radiation dose exposure is substantial, for example, during interventional procedures, radiation risk needs to be a component of consent prior to the procedure. With increased prevalence of radiologic investigations, patient education regarding the risks of radiation exposure needs to be tackled by the medical community in order to accurately convey potential risk. The Interventional Radiology Patient Safety Program among others have issued guidelines resulting in practice modifications where excessive radiation doses were being delivered intra-procedurally[150,151]. Incorporating audit as standard into radiology departments can also help to decrease the dose delivered to each patient[152,153] and will also help when discussing these scans with our patients. The establishment of national reference levels for specific CT examinations will allow audit at a local, national and international level[154-157]. While controversy still exists regarding the exact oncogenic risk associated with CT scanning simply ignoring the issue is not acceptable but audit, education and reassessment are key to improved understanding and safer practices.
Manuscript source: Invited manuscript
Specialty type: Radiology, nuclear medicine and medical imaging
Country of origin: Ireland
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Tomizawa N, Li YZ S- Editor: Kong JX L- Editor: A E- Editor: Wu HL
| 1. | Esses D, Birnbaum A, Bijur P, Shah S, Gleyzer A, Gallagher EJ. Ability of CT to alter decision making in elderly patients with acute abdominal pain. Am J Emerg Med. 2004;22:270-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 48] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 2. | Mettler FA, Thomadsen BR, Bhargavan M, Gilley DB, Gray JE, Lipoti JA, McCrohan J, Yoshizumi TT, Mahesh M. Medical radiation exposure in the U.S. in 2006: preliminary results. Health Phys. 2008;95:502-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 370] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 3. | Hricak H, Brenner DJ, Adelstein SJ, Frush DP, Hall EJ, Howell RW, McCollough CH, Mettler FA, Pearce MS, Suleiman OH. Managing radiation use in medical imaging: a multifaceted challenge. Radiology. 2011;258:889-905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 213] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 4. | van Randen A, Bipat S, Zwinderman AH, Ubbink DT, Stoker J, Boermeester MA. Acute appendicitis: meta-analysis of diagnostic performance of CT and graded compression US related to prevalence of disease. Radiology. 2008;249:97-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 210] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 5. | Rosen MP, Sands DZ, Longmaid HE, Reynolds KF, Wagner M, Raptopoulos V. Impact of abdominal CT on the management of patients presenting to the emergency department with acute abdominal pain. AJR Am J Roentgenol. 2000;174:1391-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 151] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 6. | Rosen MP, Siewert B, Sands DZ, Bromberg R, Edlow J, Raptopoulos V. Value of abdominal CT in the emergency department for patients with abdominal pain. Eur Radiol. 2003;13:418-424. [PubMed] |
| 7. | Smith-Bindman R, Miglioretti DL, Johnson E, Lee C, Feigelson HS, Flynn M, Greenlee RT, Kruger RL, Hornbrook MC, Roblin D. Use of diagnostic imaging studies and associated radiation exposure for patients enrolled in large integrated health care systems, 1996-2010. JAMA. 2012;307:2400-2409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 568] [Cited by in RCA: 599] [Article Influence: 46.1] [Reference Citation Analysis (0)] |
| 8. | IMV 2006 CT Market Summary Report. Des Plains, IL: IMV Medical Information Division, 2006. |
| 9. | Fazel R, Krumholz HM, Wang Y, Ross JS, Chen J, Ting HH, Shah ND, Nasir K, Einstein AJ, Nallamothu BK. Exposure to low-dose ionizing radiation from medical imaging procedures. N Engl J Med. 2009;361:849-857. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1061] [Cited by in RCA: 953] [Article Influence: 59.6] [Reference Citation Analysis (0)] |
| 10. | Mettler FA, Bhargavan M, Faulkner K, Gilley DB, Gray JE, Ibbott GS, Lipoti JA, Mahesh M, McCrohan JL, Stabin MG. Radiologic and nuclear medicine studies in the United States and worldwide: frequency, radiation dose, and comparison with other radiation sources--1950-2007. Radiology. 2009;253:520-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 581] [Cited by in RCA: 589] [Article Influence: 36.8] [Reference Citation Analysis (0)] |
| 11. | Mettler FA, Wiest PW, Locken JA, Kelsey CA. CT scanning: patterns of use and dose. J Radiol Prot. 2000;20:353-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 481] [Cited by in RCA: 429] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 12. | Schauer DA, Linton OW. National Council on Radiation Protection and Measurements report shows substantial medical exposure increase. Radiology. 2009;253:293-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 114] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 13. | Tsapaki V, Ahmed NA, AlSuwaidi JS, Beganovic A, Benider A, BenOmrane L, Borisova R, Economides S, El-Nachef L, Faj D. Radiation exposure to patients during interventional procedures in 20 countries: initial IAEA project results. AJR Am J Roentgenol. 2009;193:559-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 90] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 14. | Mettler FA, Bhargavan M, Thomadsen BR, Gilley DB, Lipoti JA, Mahesh M, McCrohan J, Yoshizumi TT. Nuclear medicine exposure in the United States, 2005-2007: preliminary results. Semin Nucl Med. 2008;38:384-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 15. | Heiken JP, Peterson CM, Menias CO. Virtual colonoscopy for colorectal cancer screening: current status. Cancer Imaging. 2005;5 Spec No A:S133-S139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 16. | Henschke CI, Yankelevitz DF, Libby DM, Pasmantier MW, Smith JP, Miettinen OS. Survival of patients with stage I lung cancer detected on CT screening. N Engl J Med. 2006;355:1763-1771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1163] [Cited by in RCA: 1171] [Article Influence: 61.6] [Reference Citation Analysis (0)] |
| 17. | Dempster B. National rationing of MRI in Australia has impact on use of CT. BMJ. 2013;346:f3929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 18. | Freudenberg LS, Beyer T. Subjective perception of radiation risk. J Nucl Med. 2011;52 Suppl 2:29S-35S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Royal HD. Effects of low level radiation-what’s new? Semin Nucl Med. 2008;38:392-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 20. | Health Effects of Exposure to Low Levels of Ionizing Radiations: Time for Reassessment?. Washington (DC): National Academies Press (US), 1998. |
| 21. | Cardis E, Vrijheid M, Blettner M, Gilbert E, Hakama M, Hill C, Howe G, Kaldor J, Muirhead CR, Schubauer-Berigan M. The 15-Country Collaborative Study of Cancer Risk among Radiation Workers in the Nuclear Industry: estimates of radiation-related cancer risks. Radiat Res. 2007;167:396-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 462] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 22. | Brenner DJ, Hall EJ. Computed Tomography - An Increasing Source of Radiation Exposure. N Engl J Med. 2007;357:2277-2284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6212] [Cited by in RCA: 5801] [Article Influence: 322.3] [Reference Citation Analysis (3)] |
| 23. | Berrington de González A, Mahesh M, Kim KP, Bhargavan M, Lewis R, Mettler F, Land C. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169:2071-2077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1348] [Cited by in RCA: 1370] [Article Influence: 91.3] [Reference Citation Analysis (0)] |
| 24. | Hatch M, Ron E, Bouville A, Zablotska L, Howe G. The Chernobyl disaster: cancer following the accident at the Chernobyl nuclear power plant. Epidemiol Rev. 2005;27:56-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 60] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Preston DL, Ron E, Tokuoka S, Funamoto S, Nishi N, Soda M, Mabuchi K, Kodama K. Solid cancer incidence in atomic bomb survivors: 1958-1998. Radiat Res. 2007;168:1-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1224] [Cited by in RCA: 1218] [Article Influence: 67.7] [Reference Citation Analysis (0)] |
| 26. | Berrington de González A, Darby S. Risk of cancer from diagnostic X-rays: estimates for the UK and 14 other countries. Lancet. 2004;363:345-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1191] [Cited by in RCA: 1078] [Article Influence: 51.3] [Reference Citation Analysis (0)] |
| 27. | Tubiana M. Dose-effect relationship and estimation of the carcinogenic effects of low doses of ionizing radiation: the joint report of the Académie des Sciences (Paris) and of the Académie Nationale de Médecine. Int J Radiat Oncol Biol Phys. 2005;63:317-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 118] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 28. | Hendee WR, O’Connor MK. Radiation risks of medical imaging: separating fact from fantasy. Radiology. 2012;264:312-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 237] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 29. | Doss M. Shifting the paradigm in radiation safety. Dose Response. 2012;10:562-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Doss M. Linear No-Threshold Model VS. Radiation Hormesis. Dose Response. 2013;11:480-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 69] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 31. | Report of the United Nations Scientific Committee on the Effects of Atomic Radiation 2010. [accessed 2013 Mar 1]. Available from: http://www.unscear.org/docs/reports/2010/UNSCEAR_2010_Report_M.pdf. |
| 32. | Tubiana M, Feinendegen LE, Yang C, Kaminski JM. The linear no-threshold relationship is inconsistent with radiation biologic and experimental data. Radiology. 2009;251:13-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 343] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 33. | Scott BR. Low-dose radiation risk extrapolation fallacy associated with the linear-no-threshold model. Hum Exp Toxicol. 2008;27:163-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 34. | Calabrese EJ. Muller’s Nobel lecture on dose-response for ionizing radiation: ideology or science? Arch Toxicol. 2011;85:1495-1498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | Health Physics Society. Position statement of Health Physics Society. Radiation risk in perspective. Available from: http://hps.org/documents/radiationrisk.pdf. |
| 36. | Mettler FA, Huda W, Yoshizumi TT, Mahesh M. Effective doses in radiology and diagnostic nuclear medicine: a catalog. Radiology. 2008;248:254-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1427] [Cited by in RCA: 1364] [Article Influence: 80.2] [Reference Citation Analysis (0)] |
| 37. | Doss M. Linear no-threshold model may not be appropriate for estimating cancer risk from CT. Radiology. 2014;270:307-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 38. | Bernstein CN, Blanchard JF, Kliewer E, Wajda A. Cancer risk in patients with inflammatory bowel disease: a population-based study. Cancer. 2001;91:854-862. [PubMed] |
| 39. | Desmond AN, O’Regan K, Curran C, McWilliams S, Fitzgerald T, Maher MM, Shanahan F. Crohn’s disease: factors associated with exposure to high levels of diagnostic radiation. Gut. 2008;57:1524-1529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 265] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 40. | Kinsella SM, Coyle JP, Long EB, McWilliams SR, Maher MM, Clarkson MR, Eustace JA. Maintenance hemodialysis patients have high cumulative radiation exposure. Kidney Int. 2010;78:789-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 42] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 41. | Kim PK, Gracias VH, Maidment AD, O’Shea M, Reilly PM, Schwab CW. Cumulative radiation dose caused by radiologic studies in critically ill trauma patients. J Trauma. 2004;57:510-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 42. | Pearce MS, Salotti JA, Little MP, McHugh K, Lee C, Kim KP, Howe NL, Ronckers CM, Rajaraman P, Sir Craft AW. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet. 2012;380:499-505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2528] [Cited by in RCA: 2637] [Article Influence: 202.8] [Reference Citation Analysis (1)] |
| 43. | Noshchenko AG, Bondar OY, Drozdova VD. Radiation-induced leukemia among children aged 0-5 years at the time of the Chernobyl accident. Int J Cancer. 2010;127:412-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 44. | Ron E, Modan B, Boice JD, Alfandary E, Stovall M, Chetrit A, Katz L. Tumors of the brain and nervous system after radiotherapy in childhood. N Engl J Med. 1988;319:1033-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 685] [Cited by in RCA: 618] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 45. | Mathews JD, Forsythe AV, Brady Z, Butler MW, Goergen SK, Byrnes GB, Giles GG, Wallace AB, Anderson PR, Guiver TA. Cancer risk in 680,000 people exposed to computed tomography scans in childhood or adolescence: data linkage study of 11 million Australians. BMJ. 2013;346:f2360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1349] [Cited by in RCA: 1393] [Article Influence: 116.1] [Reference Citation Analysis (0)] |
| 46. | Huda W, Atherton JV, Ware DE, Cumming WA. An approach for the estimation of effective radiation dose at CT in pediatric patients. Radiology. 1997;203:417-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 163] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 47. | Ware DE, Huda W, Mergo PJ, Litwiller AL. Radiation effective doses to patients undergoing abdominal CT examinations. Radiology. 1999;210:645-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 104] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 48. | Brenner DJ, Doll R, Goodhead DT, Hall EJ, Land CE, Little JB, Lubin JH, Preston DL, Preston RJ, Puskin JS. Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Natl Acad Sci USA. 2003;100:13761-13766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1180] [Cited by in RCA: 1129] [Article Influence: 51.3] [Reference Citation Analysis (0)] |
| 49. | Huang WY, Muo CH, Lin CY, Jen YM, Yang MH, Lin JC, Sung FC, Kao CH. Paediatric head CT scan and subsequent risk of malignancy and benign brain tumour: a nation-wide population-based cohort study. Br J Cancer. 2014;110:2354-2360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 162] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 50. | Hall EJ. Invited editorial: CT scanning; risk versus benefit. J Radiol Prot. 2000;20:347-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 51. | Miglioretti DL, Johnson E, Williams A, Greenlee RT, Weinmann S, Solberg LI, Feigelson HS, Roblin D, Flynn MJ, Vanneman N. The use of computed tomography in pediatrics and the associated radiation exposure and estimated cancer risk. JAMA Pediatr. 2013;167:700-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1033] [Cited by in RCA: 1029] [Article Influence: 85.8] [Reference Citation Analysis (0)] |
| 52. | Coren ME, Ng V, Rubens M, Rosenthal M, Bush A. The value of ultrafast computed tomography in the investigation of pediatric chest disease. Pediatr Pulmonol. 1998;26:389-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 53. | White KS. Invited article: helical/spiral CT scanning: a pediatric radiology perspective. Pediatr Radiol. 1996;26:5-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 54. | Raja AS, Wright C, Sodickson AD, Zane RD, Schiff GD, Hanson R, Baeyens PF, Khorasani R. Negative appendectomy rate in the era of CT: an 18-year perspective. Radiology. 2010;256:460-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 189] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 55. | Johnson AM, Dahn CM, Cadiz LA. Deficiencies in physician knowledge of the risks of imaging in pregnancy. Obstet Gynecol. 2014;123 Suppl 1:58S. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 56. | Ratnapalan S, Bona N, Chandra K, Koren G. Physicians’ perceptions of teratogenic risk associated with radiography and CT during early pregnancy. AJR Am J Roentgenol. 2004;182:1107-1109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 109] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 57. | Brent RL. The effect of embryonic and fetal exposure to x-ray, microwaves, and ultrasound: counseling the pregnant and nonpregnant patient about these risks. Semin Oncol. 1989;16:347-368. [PubMed] |
| 58. | Yamazaki JN, Schull WJ. Perinatal loss and neurological abnormalities among children of the atomic bomb. Nagasaki and Hiroshima revisited, 1949 to 1989. JAMA. 1990;264:605-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 59. | Schull WJ, Otake M. Cognitive function and prenatal exposure to ionizing radiation. Teratology. 1999;59:222-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 60. | Chen MM, Coakley FV, Kaimal A, Laros RK. Guidelines for computed tomography and magnetic resonance imaging use during pregnancy and lactation. Obstet Gynecol. 2008;112:333-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 272] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 61. | Dauer LT, Thornton RH, Miller DL, Damilakis J, Dixon RG, Marx MV, Schueler BA, Vañó E, Venkatesan A, Bartal G. Radiation management for interventions using fluoroscopic or computed tomographic guidance during pregnancy: a joint guideline of the Society of Interventional Radiology and the Cardiovascular and Interventional Radiological Society of Europe with Endorsement by the Canadian Interventional Radiology Association. J Vasc Interv Radiol. 2012;23:19-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 62. | Leung AN, Bull TM, Jaeschke R, Lockwood CJ, Boiselle PM, Hurwitz LM, James AH, McCullough LB, Menda Y, Paidas MJ. American Thoracic Society documents: an official American Thoracic Society/Society of Thoracic Radiology Clinical Practice Guideline--Evaluation of Suspected Pulmonary Embolism in Pregnancy. Radiology. 2012;262:635-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 93] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 63. | Ames Castro M, Shipp TD, Castro EE, Ouzounian J, Rao P. The use of helical computed tomography in pregnancy for the diagnosis of acute appendicitis. Am J Obstet Gynecol. 2001;184:954-957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 58] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 64. | Wagner LK, Huda W. When a pregnant woman with suspected appendicitis is referred for a CT scan, what should a radiologist do to minimize potential radiation risks? Pediatr Radiol. 2004;34:589-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 65. | The Associated Press. Report Links Increased Cancer Risk to CT Scans. N. Y. Times. 2007. [accessed. 2014;May 13] Available from: http://www.nytimes.com/2007/11/29/us/29scan.html. |
| 66. | Sternberg S. CT scans in children linked to cancer later. USA Today. 2001;. |
| 67. | Schenkman L. Radiology. Second thoughts about CT imaging. Science. 2011;331:1002-1004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 68. | Zarembo A. Cedars-Sinai radiation overdoses went unseen at several points. Los Angel. Times. 2009. [accessed. 2014;May 26] Available from: http://articles.latimes.com/2009/oct/14/local/me-cedars-sinai14. |
| 69. | Baumann BM, Chen EH, Mills AM, Glaspey L, Thompson NM, Jones MK, Farner MC. Patient perceptions of computed tomographic imaging and their understanding of radiation risk and exposure. Ann Emerg Med. 2011;58:1-7.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 93] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 70. | Lee CI, Haims AH, Monico EP, Brink JA, Forman HP. Diagnostic CT scans: assessment of patient, physician, and radiologist awareness of radiation dose and possible risks. Radiology. 2004;231:393-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 471] [Cited by in RCA: 450] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 71. | Brenner D, Elliston C, Hall E, Berdon W. Estimated risks of radiation-induced fatal cancer from pediatric CT. AJR Am J Roentgenol. 2001;176:289-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2366] [Cited by in RCA: 2183] [Article Influence: 91.0] [Reference Citation Analysis (0)] |
| 72. | Frush DP. Responsible use of CT. Radiology. 2003;229:289-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 73. | Larson DB, Rader SB, Forman HP, Fenton LZ. Informing parents about CT radiation exposure in children: it’s OK to tell them. AJR Am J Roentgenol. 2007;189:271-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 93] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 74. | Gunderman RB, Applegate KE. Managing risk: threat or opportunity? AJR Am J Roentgenol. 2005;185:43-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 75. | Earnest F, Swensen SJ, Zink FE. Respecting patient autonomy: screening at CT and informed consent. Radiology. 2003;226:633-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 76. | Linton OW, Mettler FA. National conference on dose reduction in CT, with an emphasis on pediatric patients. AJR Am J Roentgenol. 2003;181:321-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 251] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 77. | Kalra MK, Maher MM, Toth TL, Hamberg LM, Blake MA, Shepard JA, Saini S. Strategies for CT radiation dose optimization. Radiology. 2004;230:619-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 746] [Cited by in RCA: 690] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 78. | Soye JA, Paterson A. A survey of awareness of radiation dose among health professionals in Northern Ireland. Br J Radiol. 2008;81:725-729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 79. | Shiralkar S, Rennie A, Snow M, Galland RB, Lewis MH, Gower-Thomas K. Doctors’ knowledge of radiation exposure: questionnaire study. BMJ. 2003;327:371-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 205] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 80. | Krille L, Hammer GP, Merzenich H, Zeeb H. Systematic review on physician’s knowledge about radiation doses and radiation risks of computed tomography. Eur J Radiol. 2010;76:36-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 72] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 81. | Rice HE, Frush DP, Harker MJ, Farmer D, Waldhausen JH. Peer assessment of pediatric surgeons for potential risks of radiation exposure from computed tomography scans. J Pediatr Surg. 2007;42:1157-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 82. | Arslanoğlu A, Bilgin S, Kubal Z, Ceyhan MN, Ilhan MN, Maral I. Doctors’ and intern doctors’ knowledge about patients’ ionizing radiation exposure doses during common radiological examinations. Diagn Interv Radiol. 2007;13:53-55. |
| 83. | Keijzers GB, Britton CJ. Doctors’ knowledge of patient radiation exposure from diagnostic imaging requested in the emergency department. Med J Aust. 2010;193:450-453. [PubMed] |
| 84. | Scatarige JC, Diette GB, Haponik EF, Merriman B, Fishman EK. Availability, requesting practices, and barriers to referral for high-resolution CT of the lungs: results of a survey of U.S. pulmonologists. Acad Radiol. 2002;9:1370-1377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 85. | Subramaniam R, Hall T, Chou T, Sheehan D. Radiology knowledge in new medical graduates in New Zealand. N Z Med J. 2005;118:U1699. [PubMed] |
| 86. | O’Sullivan J, O’Connor OJ, O’Regan K, Clarke B, Burgoyne LN, Ryan MF, Maher MM. An assessment of medical students’ awareness of radiation exposures associated with diagnostic imaging investigations. Insights Imaging. 2010;1:86-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 87. | Sadigh G, Khan R, Kassin MT, Applegate KE. Radiation safety knowledge and perceptions among residents: a potential improvement opportunity for graduate medical education in the United States. Acad Radiol. 2014;21:869-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 88. | McCollough CH. CT dose: how to measure, how to reduce. Health Phys. 2008;95:508-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 70] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 89. | Smith-Bindman R, Lipson J, Marcus R, Kim KP, Mahesh M, Gould R, Berrington de González A, Miglioretti DL. Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med. 2009;169:2078-2086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1844] [Cited by in RCA: 1716] [Article Influence: 107.3] [Reference Citation Analysis (0)] |
| 90. | Durán A, Hian SK, Miller DL, Le Heron J, Padovani R, Vano E. A summary of recommendations for occupational radiation protection in interventional cardiology. Catheter Cardiovasc Interv. 2013;81:562-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 91. | Miller DL, Vañó E, Bartal G, Balter S, Dixon R, Padovani R, Schueler B, Cardella JF, de Baère T. Occupational radiation protection in interventional radiology: a joint guideline of the Cardiovascular and Interventional Radiology Society of Europe and the Society of Interventional Radiology. Cardiovasc Intervent Radiol. 2010;33:230-239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 184] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 92. | McCollough CH, Primak AN, Braun N, Kofler J, Yu L, Christner J. Strategies for reducing radiation dose in CT. Radiol Clin North Am. 2009;47:27-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 656] [Cited by in RCA: 540] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 93. | Nickoloff EL, Alderson PO. Radiation exposures to patients from CT: reality, public perception, and policy. AJR Am J Roentgenol. 2001;177:285-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 147] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 94. | McCollough CH, Bruesewitz MR, Kofler JM. CT dose reduction and dose management tools: overview of available options. Radiographics. 2006;26:503-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 606] [Cited by in RCA: 537] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 95. | Donnelly LF, Emery KH, Brody AS, Laor T, Gylys-Morin VM, Anton CG, Thomas SR, Frush DP. Minimizing radiation dose for pediatric body applications of single-detector helical CT: strategies at a large Children’s Hospital. AJR Am J Roentgenol. 2001;176:303-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 373] [Cited by in RCA: 325] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 96. | Boone JM, Geraghty EM, Seibert JA, Wootton-Gorges SL. Dose reduction in pediatric CT: a rational approach. Radiology. 2003;228:352-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 176] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 97. | Jakobs TF, Becker CR, Ohnesorge B, Flohr T, Suess C, Schoepf UJ, Reiser MF. Multislice helical CT of the heart with retrospective ECG gating: reduction of radiation exposure by ECG-controlled tube current modulation. Eur Radiol. 2002;12:1081-1086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 490] [Cited by in RCA: 431] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 98. | Brisse HJ, Robilliard M, Savignoni A, Pierrat N, Gaboriaud G, De Rycke Y, Neuenschwander S, Aubert B, Rosenwald JC. Assessment of organ absorbed doses and estimation of effective doses from pediatric anthropomorphic phantom measurements for multi-detector row CT with and without automatic exposure control. Health Phys. 2009;97:303-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 99. | Yu L, Li H, Fletcher JG, McCollough CH. Automatic selection of tube potential for radiation dose reduction in CT: a general strategy. Med Phys. 2010;37:234-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 177] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 100. | Flohr TG, Leng S, Yu L, Aiimendinger T, Bruder H, Petersilka M, Eusemann CD, Stierstorfer K, Schmidt B, McCollough CH. Dual-source spiral CT with pitch up to 3.2 and 75 ms temporal resolution: image reconstruction and assessment of image quality. Med Phys. 2009;36:5641-5653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 128] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 101. | Silva AC, Lawder HJ, Hara A, Kujak J, Pavlicek W. Innovations in CT dose reduction strategy: application of the adaptive statistical iterative reconstruction algorithm. AJR Am J Roentgenol. 2010;194:191-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 424] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 102. | Prakash P, Kalra MK, Kambadakone AK, Pien H, Hsieh J, Blake MA, Sahani DV. Reducing abdominal CT radiation dose with adaptive statistical iterative reconstruction technique. Invest Radiol. 2010;45:202-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 310] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 103. | Thibault JB, Sauer KD, Bouman CA, Hsieh J. A three-dimensional statistical approach to improved image quality for multislice helical CT. Med Phys. 2007;34:4526-4544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 711] [Cited by in RCA: 559] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 104. | Mendrik AM, Vonken EJ, Rutten A, Viergever MA, van Ginneken B. Noise reduction in computed tomography scans using 3-d anisotropic hybrid diffusion with continuous switch. IEEE Trans Med Imaging. 2009;28:1585-1594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 54] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 105. | Paul NS, Blobel J, Prezelj E, Burey P, Ursani A, Menezes RJ, Kashani H, Siewerdsen JH. The reduction of image noise and streak artifact in the thoracic inlet during low dose and ultra-low dose thoracic CT. Phys Med Biol. 2010;55:1363-1380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 106. | O’Connor OJ, Vandeleur M, McGarrigle AM, Moore N, McWilliams SR, McSweeney SE, O’Neill M, Ni Chroinin M, Maher MM. Development of low-dose protocols for thin-section CT assessment of cystic fibrosis in pediatric patients. Radiology. 2010;257:820-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 68] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 107. | Huda W. Radiation doses and risks in chest computed tomography examinations. Proc Am Thorac Soc. 2007;4:316-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 86] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 108. | Kim K, Kim YH, Kim SY, Kim S, Lee YJ, Kim KP, Lee HS, Ahn S, Kim T, Hwang SS. Low-dose abdominal CT for evaluating suspected appendicitis. N Engl J Med. 2012;366:1596-1605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 224] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 109. | Kim SY, Lee KH, Kim K, Kim TY, Lee HS, Hwang SS, Song KJ, Kang HS, Kim YH, Rhee JE. Acute appendicitis in young adults: low- versus standard-radiation-dose contrast-enhanced abdominal CT for diagnosis. Radiology. 2011;260:437-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 110. | Hidajat N, Mäurer J, Schröder RJ, Wolf M, Vogl T, Felix R. Radiation exposure in spiral computed tomography. Dose distribution and dose reduction. Invest Radiol. 1999;34:51-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 111. | Goldman LW. Principles of CT: radiation dose and image quality. J Nucl Med Technol. 2007;35:213-225; quiz 226-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 209] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 112. | Kalra MK, Maher MM, Toth TL, Kamath RS, Halpern EF, Saini S. Radiation from “extra” images acquired with abdominal and/or pelvic CT: effect of automatic tube current modulation. Radiology. 2004;232:409-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 45] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 113. | Kolditz D, Kyriakou Y, Kalender WA. Volume-of-interest (VOI) imaging in C-arm flat-detector CT for high image quality at reduced dose. Med Phys. 2010;37:2719-2730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 114. | Sistrom CL, Dang PA, Weilburg JB, Dreyer KJ, Rosenthal DI, Thrall JH. Effect of computerized order entry with integrated decision support on the growth of outpatient procedure volumes: seven-year time series analysis. Radiology. 2009;251:147-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 211] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 115. | Rosenthal DI, Weilburg JB, Schultz T, Miller JC, Nixon V, Dreyer KJ, Thrall JH. Radiology order entry with decision support: initial clinical experience. J Am Coll Radiol. 2006;3:799-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 119] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 116. | Caoili EM. Imaging of the urinary tract using multidetector computed tomography urography. Semin Urol Oncol. 2002;20:174-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 117. | Nawfel RD, Judy PF, Schleipman AR, Silverman SG. Patient radiation dose at CT urography and conventional urography. Radiology. 2004;232:126-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 117] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 118. | Chow LC, Kwan SW, Olcott EW, Sommer G. Split-bolus MDCT urography with synchronous nephrographic and excretory phase enhancement. AJR Am J Roentgenol. 2007;189:314-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 127] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 119. | Hadley JL, Agola J, Wong P. Potential impact of the American College of Radiology appropriateness criteria on CT for trauma. AJR Am J Roentgenol. 2006;186:937-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 120. | Stein SC, Fabbri A, Servadei F, Glick HA. A critical comparison of clinical decision instruments for computed tomographic scanning in mild closed traumatic brain injury in adolescents and adults. Ann Emerg Med. 2009;53:180-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 90] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 121. | Kuppermann N, Holmes JF, Dayan PS, Hoyle JD, Atabaki SM, Holubkov R, Nadel FM, Monroe D, Stanley RM, Borgialli DA. Identification of children at very low risk of clinically-important brain injuries after head trauma: a prospective cohort study. Lancet. 2009;374:1160-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1100] [Cited by in RCA: 1097] [Article Influence: 68.6] [Reference Citation Analysis (0)] |
| 122. | Slovis T, Berdon W. Panel Discussion. Pediatr Radiol. 2002;32:242-244. [DOI] [Full Text] |
| 123. | Stephen AE, Segev DL, Ryan DP, Mullins ME, Kim SH, Schnitzer JJ, Doody DP. The diagnosis of acute appendicitis in a pediatric population: to CT or not to CT. J Pediatr Surg. 2003;38:367-371; discsussion 367-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 64] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 124. | Ginde AA, Foianini A, Renner DM, Valley M, Camargo CA. Availability and quality of computed tomography and magnetic resonance imaging equipment in U.S. emergency departments. Acad Emerg Med. 2008;15:780-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 147] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 125. | Broder JS. CT utilization: the emergency department perspective. Pediatr Radiol. 2008;38 Suppl 4:S664-S669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 126. | van Randen A, Laméris W, van Es HW, van Heesewijk HP, van Ramshorst B, Ten Hove W, Bouma WH, van Leeuwen MS, van Keulen EM, Bossuyt PM. A comparison of the accuracy of ultrasound and computed tomography in common diagnoses causing acute abdominal pain. Eur Radiol. 2011;21:1535-1545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 160] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 127. | Lehnert BE, Bree RL. Analysis of appropriateness of outpatient CT and MRI referred from primary care clinics at an academic medical center: how critical is the need for improved decision support? J Am Coll Radiol. 2010;7:192-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 213] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 128. | Larson PA, Bettman MA, Waldrip C. Ionizing radiation in abdominal CT and the ACR Appropriateness Criteria®. J Am Coll Radiol. 2012;9:298; author reply 298-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 129. | Robinson JD, Hippe DS, Hiatt MD. The effect of a no-denial policy on imaging utilization. J Am Coll Radiol. 2013;10:501-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 130. | Zondervan RL, Hahn PF, Sadow CA, Liu B, Lee SI. Body CT scanning in young adults: examination indications, patient outcomes, and risk of radiation-induced cancer. Radiology. 2013;267:460-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 131. | Wall BF, Kendall GM, Edwards AA, Bouffler S, Muirhead CR, Meara JR. What are the risks from medical X-rays and other low dose radiation? Br J Radiol. 2006;79:285-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 88] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 132. | ICRP Publication 105. Radiation protection in medicine. Ann ICRP. 2007;37:1-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 411] [Cited by in RCA: 368] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 133. | Ronckers CM, Doody MM, Lonstein JE, Stovall M, Land CE. Multiple diagnostic X-rays for spine deformities and risk of breast cancer. Cancer Epidemiol Biomarkers Prev. 2008;17:605-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 113] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 134. | Flint-Richter P, Sadetzki S. Genetic predisposition for the development of radiation-associated meningioma: an epidemiological study. Lancet Oncol. 2007;8:403-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 135. | Jansen-van der Weide MC, Greuter MJ, Jansen L, Oosterwijk JC, Pijnappel RM, de Bock GH. Exposure to low-dose radiation and the risk of breast cancer among women with a familial or genetic predisposition: a meta-analysis. Eur Radiol. 2010;20:2547-2556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 136. | Brenner DJ, Shuryak I, Einstein AJ. Impact of reduced patient life expectancy on potential cancer risks from radiologic imaging. Radiology. 2011;261:193-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 89] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 137. | Einstein AJ, Henzlova MJ, Rajagopalan S. Estimating risk of cancer associated with radiation exposure from 64-slice computed tomography coronary angiography. JAMA. 2007;298:317-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1081] [Cited by in RCA: 992] [Article Influence: 55.1] [Reference Citation Analysis (0)] |
| 138. | Muirhead CR, O’Hagan JA, Haylock RG, Phillipson MA, Willcock T, Berridge GL, Zhang W. Mortality and cancer incidence following occupational radiation exposure: third analysis of the National Registry for Radiation Workers. Br J Cancer. 2009;100:206-212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 313] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 139. | Little MP, Boice JD. Comparison of breast cancer incidence in the Massachusetts tuberculosis fluoroscopy cohort and in the Japanese atomic bomb survivors. Radiat Res. 1999;151:218-224. [PubMed] |
| 140. | Brenner DJ. Does fractionation decrease the risk of breast cancer induced by low-LET radiation? Radiat Res. 1999;151:225-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 141. | Townsend BA, Callahan MJ, Zurakowski D, Taylor GA. Has pediatric CT at children’s hospitals reached its peak? AJR Am J Roentgenol. 2010;194:1194-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 66] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 142. | The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Ann ICRP. 2007;37:1-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 1072] [Article Influence: 63.1] [Reference Citation Analysis (0)] |
| 143. | Strauss KJ, Goske MJ, Kaste SC, Bulas D, Frush DP, Butler P, Morrison G, Callahan MJ, Applegate KE. Image gently: Ten steps you can take to optimize image quality and lower CT dose for pediatric patients. AJR Am J Roentgenol. 2010;194:868-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 199] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 144. | Applegate KE, Cost NG. Image Gently: a campaign to reduce children’s and adolescents’ risk for cancer during adulthood. J Adolesc Health. 2013;52:S93-S97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 145. | Brink JA, Amis ES. Image Wisely: a campaign to increase awareness about adult radiation protection. Radiology. 2010;257:601-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 155] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 146. | Sidhu M, Goske MJ, Connolly B, Racadio J, Yoshizumi TT, Strauss KJ, Coley BD, Utley T. Image Gently, Step Lightly: promoting radiation safety in pediatric interventional radiology. AJR Am J Roentgenol. 2010;195:W299-W301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 147. | Sidhu M. Radiation safety in pediatric interventional radiology: Step Lightly. Pediatr Radiol. 2010;40:511-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 148. | Initiative to Reduce Unnecessary Radiation Exposure from Medical Imaging. [accessed 2014 May 26]. Available from: http://www.fda.gov/Radiation-EmittingProducts/RadiationSafety/RadiationDoseReduction/default.htm. |
| 149. | Caverly TJ, Prochazka AV, Cook-Shimanek M, Pawlak M, Stickrath C. Weighing the potential harms of computed tomography: patient survey. JAMA Intern Med. 2013;173:588-590; discussion 590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 150. | Steele JR, Jones AK, Ninan EP. Quality initiatives: Establishing an interventional radiology patient radiation safety program. Radiographics. 2012;32:277-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 151. | Miller DL, Balter S, Schueler BA, Wagner LK, Strauss KJ, Vañó E. Clinical radiation management for fluoroscopically guided interventional procedures. Radiology. 2010;257:321-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 139] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 152. | Miglioretti DL, Zhang Y, Johnson E, Lee C, Morin RL, Vanneman N, Smith-Bindman R. Personalized technologist dose audit feedback for reducing patient radiation exposure from CT. J Am Coll Radiol. 2014;11:300-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 153. | Rodrigues JC, Negus IS, Manghat NE, Hamilton MC. A completed audit cycle of the lateral scan projection radiograph in CT pulmonary angiography (CTPA); the impact on scan length and radiation dose. Clin Radiol. 2013;68:574-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 154. | McCollough C, Branham T, Herlihy V, Bhargavan M, Robbins L, Bush K, McNitt-Gray M, Payne JT, Ruckdeschel T, Pfeiffer D. Diagnostic reference levels from the ACR CT Accreditation Program. J Am Coll Radiol. 2011;8:795-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 72] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 155. | Kim KP, Berrington de González A, Pearce MS, Salotti JA, Parker L, McHugh K, Craft AW, Lee C. Development of a database of organ doses for paediatric and young adult CT scans in the United Kingdom. Radiat Prot Dosimetry. 2012;150:415-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 156. | Foley SJ, McEntee MF, Rainford LA. Establishment of CT diagnostic reference levels in Ireland. Br J Radiol. 2012;85:1390-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 117] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 157. | Vassileva J, Rehani MM, Applegate K, Ahmed NA, Al-Dhuhli H, Al-Naemi HM, Al Suwaidi JS, Arandjic D, Beganovic A, Benavente T. IAEA survey of paediatric computed tomography practice in 40 countries in Asia, Europe, Latin America and Africa: procedures and protocols. Eur Radiol. 2013;23:623-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |