Published online Sep 28, 2014. doi: 10.4329/wjr.v6.i9.726
Revised: June 25, 2014
Accepted: July 25, 2014
Published online: September 28, 2014
Processing time: 138 Days and 13.5 Hours
Modification of 4-dimensional computed tomography (4D-CT) technique with volume rendering reconstructions and significant dose reduction is a safe and accurate method of pre-operative localization for primary hyperparathyroidism. Modified low dose 4D-CT with volume rendering reconstructions provides precise preoperative localization and is associated with a significant reduction in radiation exposure compared to classic preoperative localizing techniques. It should be considered the preoperative localization study of choice for primary hyperparathyroidism.
Core tip: To our knowledge, this is the first paper detailing the technical aspects of a low dose 4-dimensional computed tomography with volume rendering reconstruction. It is our aim to share our institute’s technique and experience in the hope of improved utilization of this modality. With this technique, our results are comparable to those published in the literature for diagnostic accuracy regarding correlation to intraoperative pathology. The 3D Volume rendering reconstruction of the parathyroid pathology shown in relation to the clavicle, thyroid gland, and skin provide superior surgical guidance and an essentially “cut here” approach for directed parathyroidectomy.
- Citation: Platz TA, Kukar M, Elmarzouky R, Cance W, Abdelhalim A. Low dose four-dimensional computerized tomography with volume rendering reconstruction for primary hyperparathyroidism: How I do it? World J Radiol 2014; 6(9): 726-729
- URL: https://www.wjgnet.com/1949-8470/full/v6/i9/726.htm
- DOI: https://dx.doi.org/10.4329/wjr.v6.i9.726
Improvements in imaging techniques for primary hyperparathyroidism have been critical in the ability to transition from formal cervical four-gland exploration to minimally invasive/directed parathyroidectomy. A precise anatomic localization study is the key to the success of minimally invasive parathyroidectomy. Traditionally, sestamibi single photon emission computed tomography (SPECT) and ultrasound (US) have been used with varying success rates from 29%-79%[1-10]. With advent of 4-dimensional computed tomography (4D-CT) technology, there is improved sensitivity and a higher intraoperative correlation rate ranging from 70%-89% demonstrated by multiple institutions[1-5,11-15]. Despite this clear advantage, the use of 4D-CT has been limited. Numerous factors including the concern of higher cost, increased radiation exposure and a lack of expertise/knowledge have been proposed. At our institution we have modified our technique to address some of these concerns. In this manuscript, we detail our modified 4D-CT technique providing an in-depth review of technical aspects, image processing and adaptations to decrease the effective radiation exposure. The term 4D is used to describe the combination of cross-sectional imaging and basic functional analysis through perfusion information of parathyroid adenomas. The first three dimensions refer to multiplanar CT: axial acquisitions, sagittal and coronal reformatted images. The fourth dimension is the change in enhancement overtime from non-contrast images to arterial and delayed phase imaging.
At our institution all patients diagnosed with primary hyperparathyroidism undergo low dose 4D-CT with volume rendering reconstructions. Additional workup includes history/physical, laboratories (serum calcium, intact parathyroid hormone level, 24-h urine calcium, vitamin D), and review of existing imaging modalities if performed (United States, sestamibi SPECT). After confirmation with localization studies, parathyroidectomy is performed via standard minimally invasive/directed technique or formal four-gland exploration.
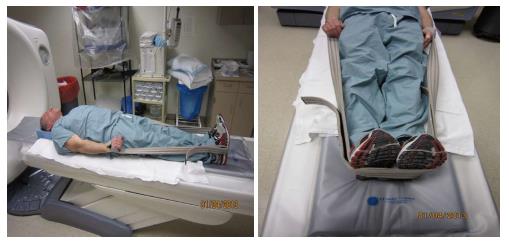
All imaging is performed on a 64 multi-slice CT scanner (VCT 64; GE Medical Systems, Milwaukee, Wis). The patient is positioned supine in the CT scanner. The patient enters head first with the upper extremities at their side. The manufacturer-supplied head holder is used for all scans. The patient’s arms are pulled caudally to minimize shoulder artifact using the manufacturer-supplied shoulder straps (Figure 1). This is very well tolerated by most patients. IV access is obtained in the right or left antecubital vein with an 18-gauge cannula and flushed with heparinized saline. The scanning protocol consists of three phases of CT imaging performed from the hard palate to the level of the carina in all phases using a 0.625 mm slice width. Scanning parameters include a voltage of 120 peak kilovolts (kVp), 200 milliamperes (mA) for the pre-contrast and delayed post-contrast phases (venous), 400 mA for the early post contrast phase (arterial), pitch of 1, and a rotation time of 0.7 s. A 64 mm × 0.625 mm detector configuration with a 10-mm beam width and a table speed of 39.37 cm per gantry rotation is utilized. Imaging is initiated with the non-contrast phase with the anticipation of the normal thyroid tissue being brighter than any parathyroid tissues due to the fact of its increased iodine concentration. The non-contrast phase is followed by intra-venous injection of 90 mL (4 mL/s) of non-ionic contrast medium (iohexol 350, 350 mg of iodine per milliliter; GE health care; Princeton, NJ). A 25 s delay is followed by repeat imaging which constitutes the arterial phase. A delay of 25 s is chosen to coincide with peak enhancement of the parathyroid adenoma as compared to enhancement of the thyroid gland and any regional lymph nodes. A higher mA (400 mA) is used in this phase to facilitate detection of small adenomas including possible multiple gland disease. Approximately 90 s after the injection, a delayed phase scan is performed. This final phase is utilized to confirm the presence of the parathyroid adenoma as it rapidly washes out the IV contrast material compared to the adjacent thyroid tissue. The duration of each phase is dependent on the distance between the hard palate and the carina as well as the table speed. Each phase takes approximately 15-17 s to complete based on the patient’s body habitus.
A major dose reduction is achieved by reducing the tube current to 200 mA in precontrast and delayed phases. This allows us to detect attenuation differences in lesions adjacent to thyroid gland without compromising imaging quality while achieving significant dose reduction. The effective radiation dose administered utilizing this modification was 11-13 millisieverts (mSv). This dose was calculated from the dose-length product provided by the scanner at the end of each exam[16]. Confirmatory CT dose index measurements using standard 16 cm head phantom and standard CT ionization chamber were also performed yielding the same numbers. Recently, our institution has implemented ASIR technology (Adaptive Statistical Iterative Reconstruction), which incorporates a new reconstructive CT algorithm with an average effective radiation dose of 9-10 mSv.
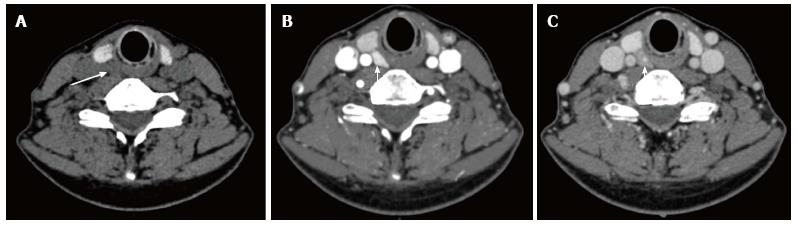
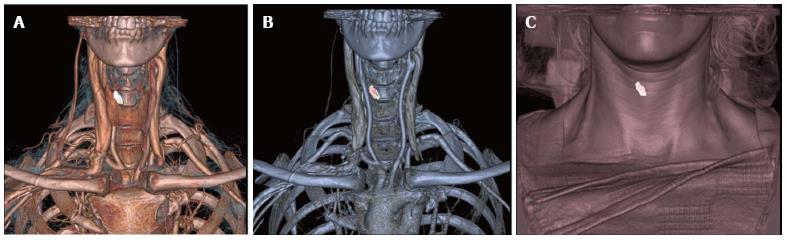
Standard post processing of imaging is performed by a fellowship-trained neuroradiologist on a separate workstation (Advantage Windows Workstation, version 4.5; GE Medical Systems). Two-dimensional sagittal, coronal and oblique multi-planar reformations are obtained from the arterial phase images. This is followed by 3-D reconstruction with volume rendering. The parathyroid adenoma is segmented from the axial images creating a 3-D volume of the adenoma that is merged with the original 3-D volume of the arterial phase of the study. The adenoma is assigned a different color and is shown against the original 3-D volume in three different thresholds and shown in relation to the thyroid gland, skin, and bony landmarks. Measurements are performed from the presumed parathyroid abnormality to the clavicular head and the overlying skin to aid the surgeon in operative guidance (Figures 2 and 3). Images processed on the workstation take an average of 8-10 min. The resulting images are interpreted by the attending neuroradiologist on a PACS workstation and a formal report is issued.
Multiple studies including our institute’s experience have shown excellent diagnostic accuracy of 4D-CT as a preoperative localization study for primary hyperparathyroidism ranging from 70%-89%[1-5,11-13]. Despite these promising results, many institutions continue to utilize sestamibi SPECT and US as the primary modalities for preoperative localization for directed parathyroidectomy. Despite superiority, 4D-CT has not gained widespread acceptance for reasons unclear to us. Plausible explanations include technical challenges, fear of increased radiation exposure and added costs.
We have utilized this technique in 150 consecutive patients undergoing parathyroidectomy for primary hyperparathyroidism. The true positive rate for modified 4D-CT with volume rendering for this cohort was 133 (89%) of 150 with a false negative (FN) of 17 (11%) of 150. In addition, utilizing our technique, the effective radiation exposure dose is 11-13 mSv. This essentially is an equivalent radiation exposure dose to that of sestamibi SPECT (9-11 mSv) but lower than that previously published for 4D-CT (27 mSv)[17]. To put this in perspective, radiation exposure from some commonly performed procedures such as CT scan of abdomen and pelvis is 14 mSv or CT angiogram is 15 mSv. This reduced effective radiation exposure of our technique is most likely due to the fact that we utilize a lower tube current for non-contrast and delayed images. The most recent implementation of ASIR technology has further reduced the radiation exposure from 4D-CT to essentially equivalent levels to that of sestamibi SPECT.
4D CT is especially useful to identify parathyroid glands in ectopic locations as the axial imaging extends from the hard palate to the level of the pulmonary artery. It is also very useful in recurrent/persistent hyperparathyroidism and improves the success rate of minimally invasive parathyroidectomy in the reoperative setting. Despite these advantages it has some limitations especially in patients with short obese necks, multinodular goiters and multiple exophytic nodules. Another limitation is the observer experience with post image processing and reconstructing 3D images.
To our knowledge, this is the first paper detailing the technical aspects of a low dose 4D-CT with volume rendering reconstruction. It is our aim to share our institute’s technique and experience in the hope of improved utilization of this modality. With this technique, our results are comparable to those published in the literature for diagnostic accuracy regarding correlation to intraoperative pathology. The 3D volume rendering reconstruction of the parathyroid pathology shown in relation to the clavicle, thyroid gland, and skin provide superior surgical guidance and an essentially “cut here” approach for directed parathyroidectomy.
In conclusion, low dose 4D-CT with volume rendering reconstruction provides superior quality images while minimizing radiation exposure. The technique is easily reproducible and in our opinion should be the diagnostic modality of choice in patients with primary hyperparathyroidism.
P- Reviewer: Fulop T S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Eichhorn-Wharry LI, Carlin AM, Talpos GB. Mild hypercalcemia: an indication to select 4-dimensional computed tomography scan for preoperative localization of parathyroid adenomas. Am J Surg. 2011;201:334-338; discussion 338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 2. | Mortenson MM, Evans DB, Lee JE, Hunter GJ, Shellingerhout D, Vu T, Edeiken BS, Feng L, Perrier ND. Parathyroid exploration in the reoperative neck: improved preoperative localization with 4D-computed tomography. J Am Coll Surg. 2008;206:888-895; discussion 895-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 130] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 3. | Cheung K, Wang TS, Farrokhyar F, Roman SA, Sosa JA. A meta-analysis of preoperative localization techniques for patients with primary hyperparathyroidism. Ann Surg Oncol. 2012;19:577-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 253] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 4. | Starker LF, Mahajan A, Björklund P, Sze G, Udelsman R, Carling T. 4D parathyroid CT as the initial localization study for patients with de novo primary hyperparathyroidism. Ann Surg Oncol. 2011;18:1723-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 106] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 5. | Rodgers SE, Hunter GJ, Hamberg LM, Schellingerhout D, Doherty DB, Ayers GD, Shapiro SE, Edeiken BS, Truong MT, Evans DB. Improved preoperative planning for directed parathyroidectomy with 4-dimensional computed tomography. Surgery. 2006;140:932-940; discussion 940-941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 276] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 6. | Glynn N, Lynn N, Donagh C, Crowley RK, Smith D, Thompson CJ, Hill ADK, Keeling F, Agha A. The utility of 99mTc-sestamibi scintigraphy in the localisation of parathyroid adenomas in primary hyperparathyroidism. Ir J Med Sci. 2011;180:191-194. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 7. | Witteveen JE, Kievit J, Stokkel MP, Morreau H, Romijn JA, Hamdy NA. Limitations of Tc99m-MIBI-SPECT imaging scans in persistent primary hyperparathyroidism. World J Surg. 2011;35:128-139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 8. | Gómez-Ramírez J, Sancho-Insenser JJ, Pereira JA, Jimeno J, Munné A, Sitges-Serra A. Impact of thyroid nodular disease on 99mTc-sestamibi scintigraphy in patients with primary hyperparathyroidism. Langenbecks Arch Surg. 2010;395:929-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Swanson TW, Chan SK, Jones SJ, Bugis S, Irvine R, Belzberg A, Levine D, Wiseman SM. Determinants of Tc-99m sestamibi SPECT scan sensitivity in primary hyperparathyroidism. Am J Surg. 2010;199:614-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 10. | Thomas DL, Bartel T, Menda Y, Howe J, Graham MM, Juweid ME. Single photon emission computed tomography (SPECT) should be routinely performed for the detection of parathyroid abnormalities utilizing technetium-99m sestamibi parathyroid scintigraphy. Clin Nucl Med. 2009;34:651-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 11. | Stark DD, Gooding GA, Moss AA, Clark OH, Ovenfors CO. Parathyroid imaging: comparison of high-resolution CT and high-resolution sonography. AJR Am J Roentgenol. 1983;141:633-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Beland MD, Mayo-Smith WW, Grand DJ, Machan JT, Monchik JM. Dynamic MDCT for localization of occult parathyroid adenomas in 26 patients with primary hyperparathyroidism. AJR Am J Roentgenol. 2011;196:61-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 13. | Randall GJ, Zald PB, Cohen JI, Hamilton BE. Contrast-enhanced MDCT characteristics of parathyroid adenomas. AJR Am J Roentgenol. 2009;193:W139-W143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Chazen JL, Gupta A, Dunning A, Phillips CD. Diagnostic accuracy of 4D-CT for parathyroid adenomas and hyperplasia. AJNR Am J Neuroradiol. 2012;33:429-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Gafton AR, Glastonbury CM, Eastwood JD, Hoang JK. Parathyroid lesions: characterization with dual-phase arterial and venous enhanced CT of the neck. AJNR Am J Neuroradiol. 2012;33:949-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 16. | Huda W, Ogden KM, Khorasani MR. Converting dose-length product to effective dose at CT. Radiology. 2008;248:995-1003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 17. | Hunter GJ, Schellingerhout D, Vu TH, Perrier ND, Hamberg LM. Accuracy of four-dimensional CT for the localization of abnormal parathyroid glands in patients with primary hyperparathyroidism. Radiology. 2012;264:789-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 105] [Article Influence: 8.1] [Reference Citation Analysis (0)] |