Published online May 28, 2013. doi: 10.4329/wjr.v5.i5.220
Revised: April 11, 2013
Accepted: May 16, 2013
Published online: May 28, 2013
Processing time: 86 Days and 24 Hours
A biloma is a rare disease characterized by an abnormal intra- or extrahepatic bile collection due to a traumatic or spontaneous rupture of the biliary system. Laboratory findings are nonspecific. The diagnosis is usually suspected on the basis of a typical history (right upper quadrant abdominal pain, chills, fever and recent abdominal trauma or surgery) and is confirmed by detection of typical radiologic features. We report the case of a patient with a history of previous cholecystectomy for lithiasis who presented with clinical symptoms and laboratory data suggestive of acute pancreatitis. Imaging studies also revealed the presence of a chronic and asymptomatic biloma, which could be mistaken for a pseudocyst. The atypical location and ultrasound findings suggested an alternative diagnosis. We therefore reviewed the known literature for bilomas, focusing on the role of ultrasonography, which can reveal some typical aspects, such as location and imaging features. We conclude that ultrasound plays a key role in the assessment of a suspected biloma in patients with appropriate history and clinical features and provides valuable diagnostic clues even in the absence of these.
Core tip: We report the case of a patient with a history of previous cholecystectomy for lithiasis who presented with a clinical picture suggestive of acute pancreatitis. Imaging studies revealed the presence of an asymptomatic biloma, which could be mistaken for a pseudocyst. We therefore reviewed the literature, focusing on the role of ultrasonography, which can reveal some typical aspects, such as location and imaging features. We conclude that ultrasound plays a key role in the assessment of a suspected biloma in patients with appropriate history and clinical features and provides valuable diagnostic clues even in the absence of these.
- Citation: Tana C, D’Alessandro P, Tartaro A, Tana M, Mezzetti A, Schiavone C. Sonographic assessment of a suspected biloma: A case report and review of the literature. World J Radiol 2013; 5(5): 220-225
- URL: https://www.wjgnet.com/1949-8470/full/v5/i5/220.htm
- DOI: https://dx.doi.org/10.4329/wjr.v5.i5.220
A biloma is a rare disease characterized by an abnormal intra- or extrahepatic bile collection secondary to a traumatic or spontaneous rupture of the biliary system. Post-traumatic cases were first reported by Whipple[1], who described the case of a patient kicked by a horse, but the term “biloma” was coined by Gould et al[2], who described a subject with extrahepatic bile leakage in the upper right quadrant of the abdomen after trauma from fighting. In the past, the common bile duct damage after open cholecystectomy was rated about 0.1%. Nowadays, with the laparoscopic technique, rates range from 0.3%-0.6%[3]. In a retrospective study that identified 18 patients with one or more documented intra-abdominal bilomas, the most frequent causes were iatrogenic (n = 16), in particular after cholecystectomy, partial hepatectomy and bile-duct catheter drainage; only two were post-traumatic[4]. After laparoscopic cholecystectomy, most of the lesions occur within 7 d[5]. The majority of minor bilomas resolve spontaneously without further complications[6]. Occasionally, spontaneous rupture of the biliary duct is reported, sometimes associated with choledocholithiasis. Other possible causes are cholangiocarcinomas, acute cholecystitis, tuberculosis, hepatic abscesses or infarctions. Rarely, an association with pancreatic cancer is described[7,8]. Biloma can complicate sickle cell disease[9,10]. The clinical features consist primarily of pain or abdominal distension, malaise, anorexia, nausea, chills and fever. If associated with choledocholithiasis, the bilomas may occur with jaundice, dark urine and acholic stools. Less frequently they are asymptomatic. Usually the lesions are diagnosed in an average time of 1-2 wk[11].
Laboratory exams may document the presence of neutrophilic leucocytosis and increased values of erythrocyte sedimentation rate and C-reactive protein (CRP), and these may suggest a concomitant cholangitis[7]. Occasionally, abnormal values of aspartate aminotransferase and alanine aminotransferase may be detected[12]. In the presence of jaundice, laboratory tests may show signs of cholestasis (elevation of serum alkaline phosphatase, total and direct bilirubin) as the result of biliary obstruction by gallstones or, less frequently, of extrahepatic biliary ductal compression caused by the biloma[13,14].
The diagnosis is suspected on the basis of the clinical history (e.g., recent cholecystectomy or abdominal trauma), location of the lesion, ultrasound and computed tomography (CT) appearance and can be confirmed by magnetic resonance (MR) cholangiopancreatography and by the features of the material obtained by ultrasonography-guided percutaneous aspiration. The bilomas must be differentiated from other similar findings, such as lymphocele, abscesses, hematomas, pseudocysts, liver cysts and seroma[4]. Gallbladder scintigraphy with technetium-99 may help to differentiate the biloma from hematomas or liver abscesses. Endoscopic retrograde cholangiography may provide not only further diagnostic confirmation but also a therapeutic option, allowing decompression of the bile duct and biliary drainage of the collection[15-19]. In the case of recurrence or persistence of the biloma, more invasive treatment strategies may be considered. In one study, surgical treatment was found to be associated with a higher complication rate[17]. If the collection is well confined or if there are small residual gallstones, surgical access by subcostal laparotomy is appropriate[20,21]. In case of bile leakage arising after hepatectomy, percutaneous transhepatic biliary drainage is the treatment of choice[21]. Kyoden et al[22] evaluated the use of prophylactic abdominal drainage performed in 1269 consecutive cases of elective liver resection in order to reduce the frequency of the development of subphrenic fluid collections and bile leakage. Placement of drains was effective in a significant number of patients undergoing hepatectomy.
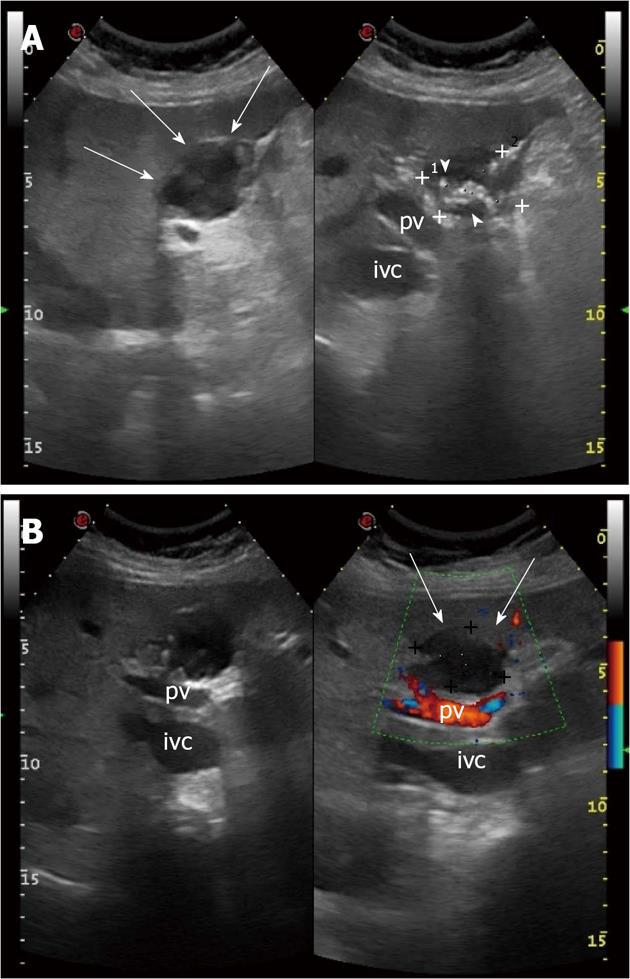
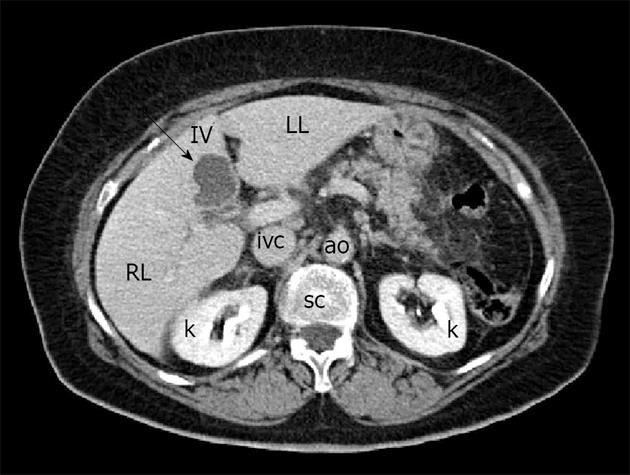
A 72-year-old woman was admitted to our Hospital for the persistence, from the day before, of acute and stabbing epigastric pain, not associated with nausea, vomiting, diarrhea or fever. The patient did not report unusual food intake or recent travel. She reported a history of ischemic heart disease, previous multiple pulmonary infarctions treated with oral anticoagulants, type 2 diabetes mellitus in good metabolic control, Hashimoto’s thyroiditis and a past history of cholecystectomy for lithiasis (12 years before). The physical examination revealed the presence of evoked pain by deep palpation in the epigastrium and torpid peristalsis. The remaining examination was normal. Laboratory tests documented fasting hyperglycemia (> 200 mg/dL), hyperamylasemia (3420 U/L), increased values of lipase (24667 U/L), CRP (8.3 mg/dL) and leukocytosis (14000/mL) with neutrophilia. Hemoglobin, serum electrolytes, indices of cholestasis, liver enzymes and cardiac markers were normal. An electrocardiogram, performed in the emergency room, showed signs of previous myocardial infarction. Abdominal X-ray showed poor distension of the bowel loops in the absence of other findings. An ultrasound examination of the upper abdomen revealed, in the IV hepatic segment and in proximity of the site of previous cholecystectomy, the presence of a heterogeneous hypo-anechoic rounded lesion, with a maximum size of 3.89 cm × 3.42 cm and hyperechoic, calcified walls; it was equipped with numerous hyperechoic debris generating acoustic shadow (Figure 1A). The lesion did not demonstrate an increase in color Doppler signal (Figure 1B). The pancreas showed a normal size with heterogeneous echotexture and blurred margins. The clinical, laboratory and pancreatic ultrasound findings were suggestive for acute pancreatitis. These data were confirmed by a contrast-enhanced abdominal CT, which showed inflammation of the pancreatic tissue and peritoneal effusion. The focal lesion localized in the IV hepatic segment had a hypodense appearance and did not enhance after intravenous administration of contrast agent (Figure 2). It was bounded by a wall thickening of a few mm and showed a thin septum in its lateral portion. The lesion’s appearance was not specific and was not supported by the clinical and laboratory findings, which strongly indicated a diagnosis of acute pancreatitis.
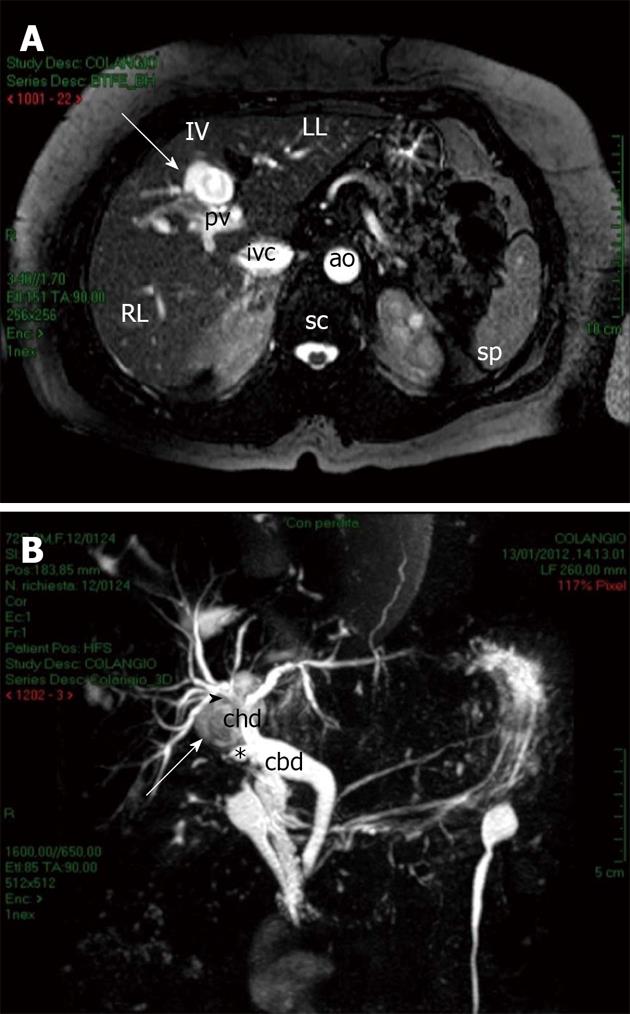
With the establishment of fasting and appropriate supportive care, the patient became asymptomatic again with optimal values of pancreatic function. However, the hepatic lesion persisted despite adequate treatment and was not due to simple pseudocyst, because of the atypical location and appearance. At magnetic resonance imaging (MRI) the lesion appeared intense on T1-weighted images and hyperintense on T2-weighted images (Figure 3A). In addition, because of its location in proximity of the site of the previous cholecystectomy, a contrast-enhanced MR cholangiopancreatography was performed, which revealed a close proximity between the lesion and the stump of the remnant cystic duct. The lesion seemed to arise posteriorly to the confluence of the right and left hepatic ducts into the common hepatic duct (Figure 3B).
Biloma was diagnosed. Considering the surgical risk of the patient and the fact that the lesion was found many years after cholecystectomy, the surgeon did not advise intervention. The patient is undergoing follow-up with ultrasonography every 6 mo to confirm the stability of the lesion over time.
Vazquez et al[4] identified 21 bilomas in eighteen patients, using ultrasound, CT or both. A solitary bile collection was found in fifteen patients, while two distinct bilomas were detected in three patients each. Width, depth, and axial length ranged from 2 cm × 2 cm × 3 cm to 10 cm × 19 cm × 25 cm, respectively. The maximal transverse diameter was ≤ 5 cm, between 6 and 10 cm and greater than 10 cm in four, eight and eight bilomas, respectively. Sixteen of these were located in the right upper quadrant of the abdomen, four of which were intrahepatic, six subhepatic, and six subphrenic. The last five were located in the left upper quadrant of the abdomen[4]. Therefore, the right quadrant is more frequently affected[7].
Ultrasound plays a key role in diagnosis of bilomas, representing the first instance investigation[3,15,23,24], with the advantage of being a non-invasive and rapidly executable exam. This factor is particularly important in post-traumatic cases, where a rapid diagnosis is essential for subsequent therapeutic intervention. Focused assessment with sonography for trauma (FAST) is indicated for screening hemodynamically stable patients with blunt abdominal trauma; in low-grade injuries it may disclose or exclude a potentially unknown pathology, such as bile leaks, free peritoneal fluid and hematomas with a positive cost-to-benefit ratio and high negative predictive value[25-27]. In high-grade injuries, ultrasound may be useful in association with CT for definitive interval assessment[26].
This method is able to show the presence of single or multiple well circumscribed anechoic lesions with prominent distal sonic enhancement[23]. These may contain a small amount of debris or have few septa but are usually devoid of capsules. They are sometimes surrounded by a thin rim which is thicker in the case of longer duration bilomas. The accuracy of ultrasonographic findings in the diagnosis of biloma is enhanced by the clinical pre-test probability, based on a thorough clinical-anamnestic assessment[24]. In the presence of a history of recent trauma or interventions such as cholecystectomy and hepatectomy, usually associated with clinical features (pain or abdominal distension, jaundice, chills, fever) and laboratory abnormalities, the ultrasound finding of well-delimited anechoic lesions in typical locations (sub- or intrahepatic or subphrenic) may suggest the presence of biloma. In this context, ultrasonography-guided percutaneous aspiration can attain a significant diagnostic value; a high aspirated fluid/serum bilirubin ratio is strongly suggestive of bile leakage and can confirm the diagnosis[27]. It may also be useful as a therapeutic option but is associated with discomfort and infection, whereas surgery, which is usually limited to refractory cases, has high morbidity and mortality rates. Therefore, Shami et al[28] recently suggested the use of endoscopic ultrasound (EUS) to drain bilomas, obtaining promising results. In this study, a total of five patients underwent EUS-guided transenteric drainage of symptomatic bilomas adjacent to the gastrointestinal lumen. The method included transenteric EUS-guided puncture, placement of a guidewire into the biloma and creation of an enteral-biloma fistula with positioning of a plastic endoprosthesis. This technique was successfully performed, resolving the biloma in all five patients, in the absence of significant morbidity. Recently, contrast-enhanced ultrasound (CEUS) has been applied to detect bile leakage by showing the passage of contrast agent into the perihepatic space[29]. The usefulness of this technique has recently been confirmed by Mao et al[30] in the diagnosis of biliary leakage following T-tube removal, but further studies with a larger number of patients are necessary to evaluate this new application of CEUS.
Finally, ultrasonography can be useful in the follow-up of patients undergoing drainage or surgery, to evaluate biloma resolution, and in those conservatively treated in order to document lesion stability without further complications[4].
CT can confirm the presence of bilomas, which appear as well-confined collections with low intraparenchymal or perihepatic attenuation values[31]. Bilomas are usually clearly delineated by liver margins, diaphragm, mesenteries and other adjacent structures; however, they have no identifiable capsule. Occasionally, they may have a thin rim of 1-2 mm which can be larger in the case of older biloma; it may be enhanced after administration of intravenous contrast agent[4]. CT cannot show bile duct injuries[17]. Sometimes the lesions are associated with the presence of ascitic fluid in the peritoneal cavity[4].
In doubtful cases and/or in the presence of CT contraindications (severe renal insufficiency or iodinated contrast sensitivity), MRI can be a valuable tool to diagnose and differentiate the biloma from other focal liver lesions, such as subacute hematoma: the biloma can appear heterogeneously intense on T1-weighted images and homogeneously hyperintense on T2-weighted images, while the hematoma usually appears hyperintense on both T1- and T2-weighted MR sequences[32]. Unlike CT, MR cholangiography enhanced with hepatocyte-specific contrast agents can accurately delineate the anatomy of the biliary system and its relationship with a suspected biloma. This method has proved of high diagnostic accuracy in differentiating biliary from nobiliary lesions[33]. Pecchi et al[34] have recently found that MR cholangiography can attain sensitivity, specificity, positive and negative predictive values and diagnostic accuracy of 93.5%, 94.4%, 96.7%, 89.5% and 93.9%, respectively, in the diagnosis of biliary complications (e.g., bilomas) after orthotopic liver transplantation. After the administration of gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid (Gd-EOB-DTPA), MR cholangiography can reveal an intrahepatic biloma as a liver fluid accumulation with delayed filling of the contrast agent. Furthermore, by demonstrating the passage of contrast material, such as mangafodipir trisodium or Gd-EOB-DTPA, MR cholangiography can clearly outline an extrahepatic biloma[33,35]. In our case, MR cholangiography confirmed the presence of bile leakage, suspected on the basis of ultrasound findings, in the absence of typical history and clinical features.
In an early stage, the gallbladder scintigraphy with technetium-99 can highlight one or more areas of reduced uptake of the radioactive substance, while in the late phase, 2 h after administration, it can document an uptake area. This examination helps to differentiate the biloma from hematomas or liver abscesses but is currently little used[36].
Endoscopic retrograde cholangiography accurately diagnoses the cause of postcholecystectomy bile leakage and biloma formation, at the same time allowing a definitive treatment determining decompression of the bile duct (through a sphincterotomy or nasobiliary endoprosthesis placement) and biliary drainage of the collection[15,17-19].
In conclusion, ultrasonography plays a key role in the assessment of suspected biloma: in patients with a history of recent trauma or hepatobiliary surgery, who present with right upper quadrant abdominal pain, chills, fever or other symptoms, the finding of single or multiple well circumscribed anechoic lesions with prominent distal sonic enhancement, debris or few septations, located in typical sites (more often subphrenic, subhepatic or intrahepatic) can orient toward the diagnosis, which can be confirmed by second level imaging and/or ultrasonography-guided percutaneous aspiration or endoscopic drainage. The latter can reveal a high aspirated fluid/serum bilirubin ratio, strongly suggestive of bile leakage, also allowing a rapid resolution of the lesion. In doubtful cases, like our patient, sonography may raise the suspicion of biloma, providing precious diagnostic clues, but confirmation with second level imaging, such as MR cholangiography, is needed. Finally, ultrasonography can be a valuable tool to follow-up untreated lesions in order to document their stability or any increase over time.
P- Reviewers Deter R, Liu YY S- Editor Gou SX L- Editor A E- Editor Ma S
| 1. | Whipple C. A case of traumatic rupture of the liver: formation of cystic swelling containing bile stained fluid. Lancet. 1898;1:719. [DOI] [Full Text] |
| 2. | Gould L, Patel A. Ultrasound detection of extrahepatic encapsulated bile: “biloma”. AJR Am J Roentgenol. 1979;132:1014-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 60] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 3. | Kannan U, Parshad R, Regmi SK. An unusual presentation of biloma five years following cholecystectomy: a case report. Cases J. 2009;2:8048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 4. | Vazquez JL, Thorsen MK, Dodds WJ, Quiroz FA, Martinez ML, Lawson TL, Stewart ET, Foley WD. Evaluation and treatment of intraabdominal bilomas. AJR Am J Roentgenol. 1985;144:933-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 42] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 5. | Kozarek R, Gannan R, Baerg R, Wagonfeld J, Ball T. Bile leak after laparoscopic cholecystectomy. Diagnostic and therapeutic application of endoscopic retrograde cholangiopancreatography. Arch Intern Med. 1992;152:1040-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 46] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 6. | Dev V, Shah D, Gaw F, Lefor AT. Gastric outlet obstruction secondary to post cholecystectomy biloma: case report and review of the literature. JSLS. 1998;2:185-188. [PubMed] |
| 7. | Bas G, Okan I, Sahin M, Eryılmaz R, Isık A. Spontaneous biloma managed with endoscopic retrograde cholangiopancreatography and percutaneous drainage: a case report. J Med Case Rep. 2011;5:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 8. | Trivedi PJ, Gupta P, Phillips-Hughes J, Ellis A. Biloma: an unusual complication in a patient with pancreatic cancer. World J Gastroenterol. 2009;15:5218-5220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 9] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Middleton JP, Wolper JC. Hepatic biloma complicating sickle cell disease. A case report and a review of the literature. Gastroenterology. 1984;86:743-744. [PubMed] |
| 10. | Lebensburger J, Esbenshade A, Blakely M, Hankins J, Wang W. Biloma and pneumobilia in sickle cell disease. Pediatr Blood Cancer. 2008;51:288-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Carroll BJ, Birth M, Phillips EH. Common bile duct injuries during laparoscopic cholecystectomy that result in litigation. Surg Endosc. 1998;12:310-313; discussion 314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 104] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 12. | Lee JH, Suh JI. A case of infected biloma due to spontaneous intrahepatic biliary rupture. Korean J Intern Med. 2007;22:220-224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 13. | Mansour AY, Stabile BE. Extrahepatic biliary obstruction due to post-laparoscopic cholecystectomy biloma. JSLS. 2000;4:167-171. [PubMed] |
| 14. | Cólović R, Perisić-Savić M. Retroperitoneal biloma secondary to operative common bile duct injury. HPB Surg. 1991;3:193-197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 15. | Mergener K, Strobel JC, Suhocki P, Jowell PS, Enns RA, Branch MS, Baillie J. The role of ERCP in diagnosis and management of accessory bile duct leaks after cholecystectomy. Gastrointest Endosc. 1999;50:527-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Bala M, Gazalla SA, Faroja M, Bloom AI, Zamir G, Rivkind AI, Almogy G. Complications of high grade liver injuries: management and outcomewith focus on bile leaks. Scand J Trauma Resusc Emerg Med. 2012;20:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 65] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Mizuno O, Kawamoto H, Fukatsu H, Harada R, Tsutsumi K, Fujii M, Kurihara N, Nakanishi T, Ogawa T, Ishida E. An iatrogenic hepatic subcapsular biloma successfully treated by percutaneous drainage and endoscopic biliary stenting. Endoscopy. 2008;40:E42-E43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 18. | Christoforidis E, Vasiliadis K, Goulimaris I, Tsalis K, Kanellos I, Papachilea T, Tsorlini E, Betsis D. A single center experience in minimally invasive treatment of postcholecystectomy bile leak, complicated with biloma formation. J Surg Res. 2007;141:171-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 19. | Miro AG, De Seta C, Arenga G, Russo M, Lombardi D. Treatment of major iatrogenic lesions of the bile ducts. Ann Ital Chir. 2002;73:35-39. [PubMed] |
| 20. | Nordin A, Halme L, Mäkisalo H, Isoniemi H, Höckerstedt K. Management and outcome of major bile duct injuries after laparoscopic cholecystectomy: from therapeutic endoscopy to liver transplantation. Liver Transpl. 2002;8:1036-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 48] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 21. | Hoekstra LT, van Gulik TM, Gouma DJ, Busch OR. Posthepatectomy bile leakage: how to manage. Dig Surg. 2012;29:48-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Kyoden Y, Imamura H, Sano K, Beck Y, Sugawara Y, Kokudo N, Makuuchi M. Value of prophylactic abdominal drainage in 1269 consecutive cases of elective liver resection. J Hepatobiliary Pancreat Sci. 2010;17:186-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 23. | Esensten M, Ralls PW, Colletti P, Halls J. Posttraumatic intrahepatic biloma: sonographic diagnosis. AJR Am J Roentgenol. 1983;140:303-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 22] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 24. | Sgourakis G, Lanitis S, Korontzi M, Kontovounisios C, Zacharioudakis C, Armoutidis V, Karaliotas C, Dedemadi G, Lepida N, Karaliotas C. Incidental findings in focused assessment with sonography for trauma in hemodynamically stable blunt trauma patients: speaking about cost to benefit. J Trauma. 2011;71:E123-E127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 25. | Kornezos I, Chatziioannou A, Kokkonouzis I, Nebotakis P, Moschouris H, Yiarmenitis S, Mourikis D, Matsaidonis D. Findings and limitations of focused ultrasound as a possible screening test in stable adult patients with blunt abdominal trauma: a Greek study. Eur Radiol. 2010;20:234-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 26. | Chiu WC, Wong-You-Cheong JJ, Rodriguez A, Shanmuganathan K, Mirvis SE, Scalea TM. Ultrasonography for interval assessment in the nonoperative management of hepatic trauma. Am Surg. 2005;71:841-846. [PubMed] |
| 27. | Ahn YJ, Kim TH, Moon SW, Choi SN, Kim HJ, Jung WT, Lee OJ, Ko GH. [A case of perforated xanthogranulomatous cholecystitis presenting as biloma]. Korean J Gastroenterol. 2011;58:153-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 28. | Shami VM, Talreja JP, Mahajan A, Phillips MS, Yeaton P, Kahaleh M. EUS-guided drainage of bilomas: a new alternative? Gastrointest Endosc. 2008;67:136-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 29. | Ignee A, Baum U, Schuessler G, Dietrich CF. Contrast-enhanced ultrasound-guided percutaneous cholangiography and cholangiodrainage (CEUS-PTCD). Endoscopy. 2009;41:725-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 30. | Mao R, Xu EJ, Li K, Zheng RQ. Usefulness of contrast-enhanced ultrasound in the diagnosis of biliary leakage following T-tube removal. J Clin Ultrasound. 2010;38:38-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 31. | Yoon W, Jeong YY, Kim JK, Seo JJ, Lim HS, Shin SS, Kim JC, Jeong SW, Park JG, Kang HK. CT in blunt liver trauma. Radiographics. 2005;25:87-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 122] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 32. | Shigemura T, Yamamoto F, Shilpakar SK, Kojima T, Yamamoto S, Pu Y. MRI differential diagnosis of intrahepatic biloma from subacute hematoma. Abdom Imaging. 1995;20:211-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 33. | Lee NK, Kim S, Lee JW, Lee SH, Kang DH, Kim GH, Seo HI. Biliary MR imaging with Gd-EOB-DTPA and its clinical applications. Radiographics. 2009;29:1707-1724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 161] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 34. | Pecchi A, De Santis M, Di Benedetto F, Gibertini M, Gerunda G, Torricelli P. Role of magnetic resonance cholangiography in biliary complications of orthotopic liver transplantation. Radiol Med. 2010;115:1065-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 35. | Park MS, Kim KW, Yu JS, Kim MJ, Kim KW, Lim JS, Cho ES, Yoon DS, Kim TK, Lee SI. Early biliary complications of laparoscopic cholecystectomy: evaluation on T2-weighted MR cholangiography in conjunction with mangafodipir trisodium-enhanced 3D T1-weighted MR cholangiography. AJR Am J Roentgenol. 2004;183:1559-1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |