Published online Aug 28, 2025. doi: 10.4329/wjr.v17.i8.111076
Revised: July 16, 2025
Accepted: August 8, 2025
Published online: August 28, 2025
Processing time: 67 Days and 1.2 Hours
Lung cancer, particularly non-small cell lung cancer (NSCLC), remains a leading cause of cancer-related death globally, and a significant number of patients deve
Core Tip: Stereotactic radiotherapy (SRT) offers a precise and effective treatment option for brain metastasis (BM) in patients with non-small cell lung cancer (NSCLC), providing better local tumor control, fewer side effects, and improved survival rates compared with the effects of traditional therapies. Integrating SRT with immunotherapy has displayed promise in enhancing intracranial progression-free survival, but further research is needed to optimize treatment protocols and refine multimodal therapy strategies for patients with NSCLC and BM.
- Citation: Khan A, Zhao HF, Meng H, Wu N, Liu LL. Stereotactic radiotherapy for brain metastases of non-small cell lung cancer: A comprehensive review. World J Radiol 2025; 17(8): 111076
- URL: https://www.wjgnet.com/1949-8470/full/v17/i8/111076.htm
- DOI: https://dx.doi.org/10.4329/wjr.v17.i8.111076
Lung cancer remains a significant health issue globally, causing approximately 2 million new cases and 1.8 million fatalities each year[1]. Non-small cell lung cancer (NSCLC) is the most common form of lung cancer, accounting for 85% of all diagnosed cases[2]. Advanced NSCLC is associated with poor outcomes, as evidenced by 5-year survival rates of only 15% and 5% for especially stage III and metastatic stage IV NSCLC, respectively[3]. Throughout disease progression, as many as 60% of patients with NSCLC can experience central nervous system (CNS) involvement[4]. Brain metastasis (BM) in patients with NSCLC is correlated with reduced overall survival, progression-free survival, and quality of life; however, early detection has been demonstrated to improve patient outcomes[5]. BM occurs in nearly 50% of individuals with advanced NSCLC, significantly affecting the overall morbidity associated with the disease[6].
BM is associated with several symptoms, such as headache, seizures, and changes in vision, speech, and/or per
Chemotherapy is an effective treatment for BM, with response rates in the brain similar to those reported in other regions of the body[11]. Targeted systemic therapies are highly effective, especially in patients with driver mutations such as EGFR and ALK–MET mutations, and they can effectively reach the CNS[11]. Although there are insufficient pro
The management of NSCLC with BM mostly consists of surgical intervention and RT[13]. Whole-brain RT (WBRT) and stereotactic RT (SRT) are two forms of RT. SRT includes various techniques, such as fractionated SRT (FSRT), stereotactic body RT (SBRT), and stereotactic radiosurgery (SRS)[14]. Currently, most patients typically receive SRT, whereas individuals with a significant disease load receive WBRT[15]. SRT facilitates superior local control with little toxicity[16,17].
This study examined the clinical effectiveness of SRT and the underlying mechanisms in patients with lung cancer and BM. This study compared recent improvements in SRT procedures with traditional treatments to assess the effects of SRT on local control, side effects, and survival rates. This research also explored the integration of SRT with systemic medications to develop a comprehensive and successful treatment plan for patients with NSCLC.
Currently, the standard treatment for patients with limited BMs involves a combination of surgery and postoperative RT[18,19]. For individuals with an asymptomatic BM lesion, those with multiple BMs, or those ineligible for surgery because of comorbidities or challenging anatomical lesion locations, standalone RT is also considered[20]. Nevertheless, several studies reported no substantial difference in patient outcomes between RT alone and postoperative RT in terms of local disease management and overall survival[21,22]. SRT is currently recommended by the World Health Organization for the treatment of both malignant and benign tumors, in addition to neurological and vascular diseases. This treatment is especially suitable for certain difficult-to-treat or inoperable tumors, such as malignant brain tumors (e.g., glioblastoma, metastatic brain tumors), spinal tumors, lung cancer, liver cancer, and pancreatic cancer[23]. For benign tumors, such as meningioma, pituitary adenoma, and acoustic neuroma, SBRT can also effectively control tumor growth and avoid surgical risks[24]. This treatment method is widely used for tumor control and symptom relief, as it targets the tumor with precise radiation beams and minimizes damage to surrounding healthy tissues. SRT has become part of regular practice in the radiation oncology field, and it is used outside of clinical trials and specialist academic institutions[25]. The three forms of SRT (SRS, FSRT and SBRT) are distinguished by their indications, fractionation, and quality standards[26]. WBRT has traditionally been the primary method used because of its ability to rapidly address both apparent and hidden lesions[27]. WBRT plays a significant role in managing numerous brain lesions, enhancing both endemic and remote CNS tumor management. However, WBRT is associated with several adverse effects, including cognitive issues such as drowsiness and memory loss, decreased physical ability, diminished appetite, and increased fatigue[28,29]. Additionally, irradiation can cause radiation necrosis in the healthy brain parenchyma[30]. Consequently, the implementation of RT has recently transitioned to the use of less hazardous methodologies, such as SRT, which have led to increased cognitive preservation[31].
As noted by Topkan et al[32], SRS is an effective technique that allows a substantial dose of radiation to be precisely directed to a well-defined target in a single session. This ablative and focused strategy works similarly to surgical resection while causing less damage. SRS is the best treatment option for small BMs, and it is a good option when surgery is not possible, such as in cases in which metastases are located in deep or delicate brain areas and they cannot be removed or a patient has other health problems that preclude surgery[33-35]. Linear accelerators, CyberKnife®, and GammaKnife are the most widely utilized technologies for the radiosurgical treatment of BMs[36,37]. Schmitt et al[38] and Vellayappan et al[39] used SRS to treat BMs next to the optic nerve, brainstem, or other delicate brain structures, and they demonstrated that this approach had a better toxicity profile than other RT methods.
FSRT/multifraction stereotactic RT (MFSRT) is a viable approach for the management of extensive postoperative cavities after the excision of a BM[20]. This method divides radiation doses to increase the biologically effective dose (BEDs) delivered to the target lesion while simultaneously minimizing the risk of radiation necrosis in adjacent healthy brain tissue[40]. Researchers have treated patients with numerous BMs with three to five FSRT treatments[41]. FSRT/MFSRT is often used to treat lesions adjacent to vital regions, such as the brainstem[15,20]. Establishing the precise radiation doses administered by MFSRT remains difficult owing to the absence of prospective studies. Studies comparing RT approaches indicated that MFRST causes much lower rates of radiation necrosis (0%–8%) than SRS (13%–30%)[20]. Research reported that the 1-year local control rate after MFSRT alone varies between 65% and 96%[15,20]. According to Perlow et al[42], FSRT/MFSRT can provide therapeutic efficacy and cause few side effects when used after surgery. Additionally, FSRT/MFSRT can be used as an initial treatment before surgery.
Moreover, this method has been substantially applied clinically because it induces a lower rate of side effects and a higher rate of local control than SRS. Putz et al[43] reported that FSRT/MFSRT had different biological effectiveness than SRS. Although hypoxic tumor cells can persist after SRS, reoxidation-based FSRT/MFSRT achieves superior tumor control rates. Patients who undergo FSRT or MFSRT might not benefit from the SIR model because of this[44]. The development of prognostic indicators for patients with NSCLC who are receiving FSRT/MFSRT to treat brain oligometastasis is extremely important. The size of the metastatic lesion and the radiation dose are the new foci of prognostic research for FSRT/MFSRT in patients with oligometastasis[45]. Although several studies suggested different BEDs for radiation, there is no universally accepted technique for evaluating the extent of the tumor, radiation dosage, or fractionation schemes[46]. Consequently, it is essential to develop and improve prediction indices to assess the efficacy of treatment for patients with NSCLC and brain oligometastasis who are undergoing FSRT/MFSRT.
SBRT integrates various technologies and methods, such as 3D conformal radiation, intensity-modulated RT, image guidance, motion management, and stereotactic precision. However, the hallmark of SBRT is the application of a high, ablative, or nearly ablative dose in a limited number of treatment sessions (i.e., five or fewer fractions)[47]. Although the technologies and procedures used in the implementation of SBRT are intriguing, the primary purpose of this strategy is to administer a compact dosage precisely to a designated target possessing steep slopes in all directions, a technique known as geometric avoidance[48]. In contrast to traditional RT, which leverages the difference in healing between neoplastic and healthy tissues after RT to achieve a therapeutic benefit, SBRT primarily aims to target the tumor while ideally completely sparing the surrounding normal tissue[49]. Thus, this method markedly differs from traditional RT, which often affects extensive areas of healthy tissue[50].
SRS was originally designed to treat a range of cranial disorders opposed to being specifically intended for the treatment of brain tumors. In the late 1960s, SRS was successfully used to treat arteriovenous malformations (AVMs), which are congenital vascular defects in which blood flows directly from arteries to veins without passing through capillaries. AVMs are characterized by poorly developed blood channels, which are highly radiosensitive. A single SRS dose of 15–25 Gy obliterates 80%–90% of small AVMs, indicating early success in the use of SRS for treating abnormal vascular structures[50-52]. This success facilitated the use of SRS in the treatment of brain tumors and metastases[53].
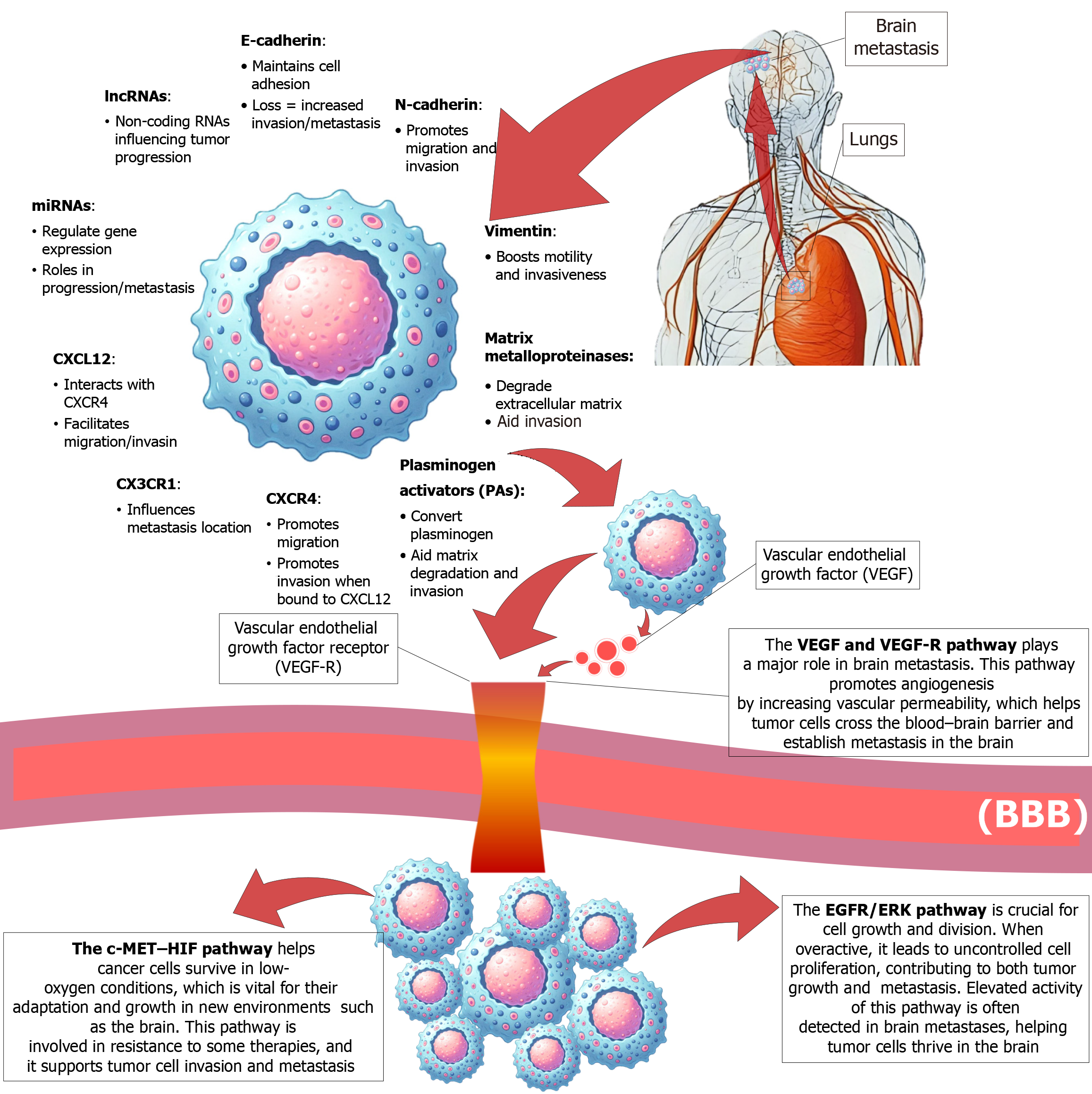
The tumor microenvironment (TME) is critical at every phase of metastasis, as it shapes tumor growth, progression, and spread to distant sites[54] (Figure 1). BM is common in lung cancer, as cancer cells can infiltrate the brain parenchyma by crossing the blood-brain barrier (BBB), disrupting its defense mechanism[4,55,56]. Reactive gliosis triggers changes in astrocytes that accelerate BM growth and reduce sensitivity to chemotherapy. Additionally, various TME cells are reprogrammed to facilitate cancer spread; for example, tumor-associated macrophages, especially the M2 subtype, promote angiogenesis and immune suppression[57-59].
Tumor spread is influenced by factors such as blood flow, proximity, and the TME. Key processes include tumor cells crossing the BBB and undergoing epithelial-mesenchymal transition, enabling tumor cell detachment and invasion. Markers, such as CXCL12/CXCR4, E-cadherin, and MMP-9, and pathways, such as the EGFR/ERK and VEGF pathways, play important roles in metastasis. Noncoding RNAs, including specific microRNAs (e.g., miRNA-200, miRNA-378) and lncRNAs (e.g., MALAT1), contribute to metastatic adaptation in the brain by regulating the expression of tumor suppressor genes and oncogenes[60].
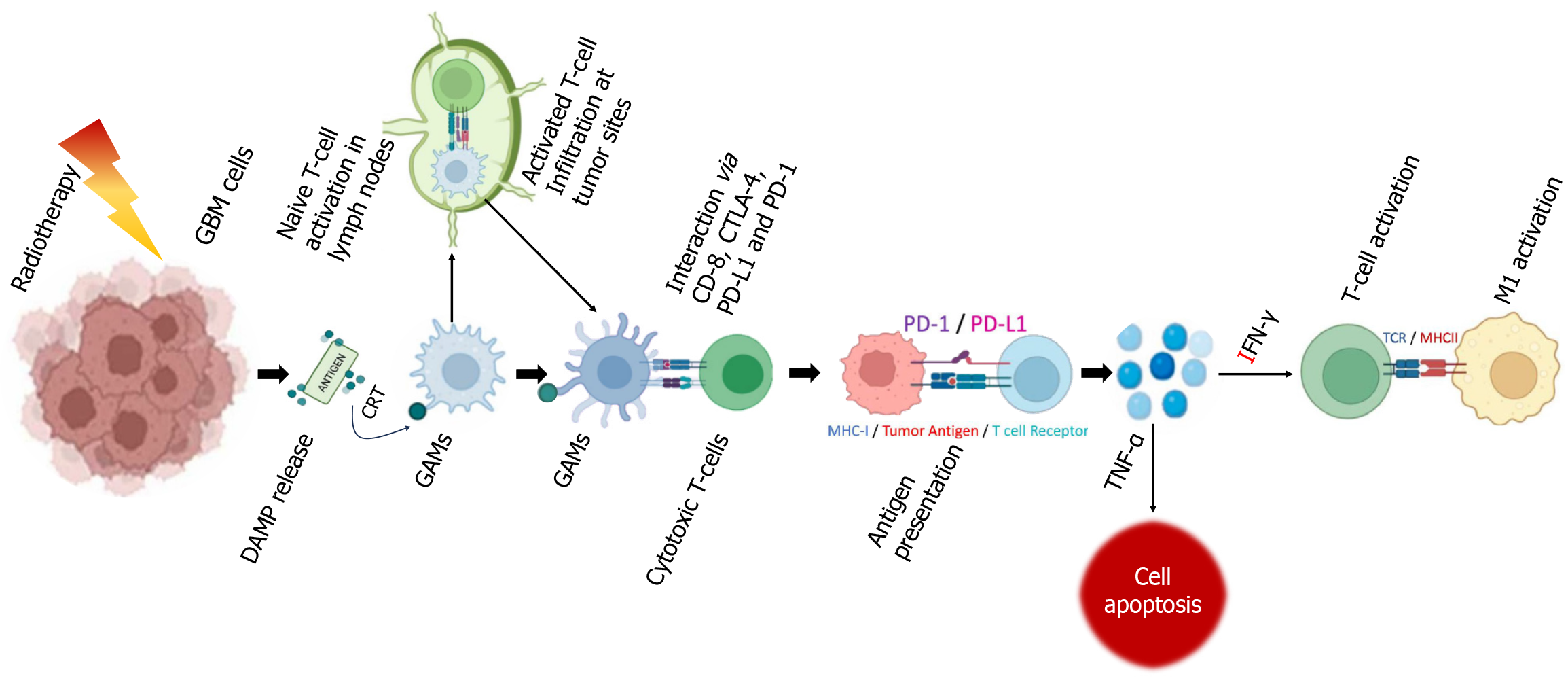
In NSCLC, dendritic cells (DCs) suppress T lymphocyte proliferation through regulatory T cells (Tregs), and tumor-associated neutrophils can shift to protumoral N2 phenotypes, contributing to extracellular matrix remodeling and immune evasion[61,62]. RT has complex effects on TME cells that potentially influence both treatment response and resistance. RT increases immune activation by increasing natural killer (NK) cell and CD8+ T lymphocyte cytotoxicity and promoting M1 macrophage differentiation while reducing Treg infiltration[63,64]. DCs also become more active in response to RT, increasing MHC molecule expression and triggering immune responses[65].
Nonetheless, RT stimulates the secretion of TGF-β, thus facilitating tumor invasion and immune evasion. In addition, inflammatory cytokines produced by CD8+ T cells and NK cells, such as tumor necrosis factor-alpha, interferon-gamma (IFN-γ), and interleukin-2, help establish a pro-apoptotic environment, potentially increasing the ability of the immune system to target cancer cells[34,66]. Tregs, which are resistant to radiation, impair CD8+ T-cell activity and support cancer-associated fibroblast differentiation and antiapoptotic signaling. High-dose hypofractionated stereotactic RT (HSRT) can counteract these processes by increasing CD8+ T-cell activity and reducing TGF-β secretion[67,68].
PD-L1 expression is another critical factor in the TME. Cancer cells often exploit PD-L1/PD-1 pathways to evade immune responses by inhibiting cytotoxic T lymphocytes (CTLs)[69]. In lung cancer with BM, the number of PD-L1–positive tumor-infiltrating cells is decreased, thereby reducing the efficacy of PD-L1 inhibitors compared with their efficacy in primary lung tumors[70]. RT increases PD-L1 expression in tumor cells, potentially facilitating immune evasion. However, this increase in PD-L1 expression could also increase the responsiveness of tumors to PD-L1–targeting treatments when used alongside RT[71]. High PD-L1 expression, especially in tumors with a high tumor mutational burden (TMB), generally increases the effectiveness of immunotherapies, such as nivolumab and pembrolizumab[72]. Conversely, NSCLC tumors with EGFR or ALK mutations have a lower TMB and PD-L1 expression, making them less responsive to these therapies[73].
Radiation also affects macrophage polarization, thus influencing tumor growth. Some studies revealed that low-dose radiation (10 Gy) increases the number of M1 macrophages while reducing the number of M2 macrophages and activating prosurvival pathways[74,75]. Modest radiation doses can increase IFN-γ expression, enhancing lymphocyte mobility and tumor-fighting abilities by upregulating MHC I and VCAM-1[76,77]. However, radioresistant tumors can block CTL infiltration, although RT-induced TLR upregulation and DAMP exposure increase CTL cytotoxicity[78]. During HSRT, high radiation doses increase immune-stimulating antigen production, further enhancing antitumor immunity[34].
The regulation of inflammation in the TME remains critical, as COX-2 overexpression promotes tumor growth and radioresistance. COX-2 inhibition can sensitize cancer cells to radiation, with some evidence suggesting improved survival in patients with breast or lung cancer[79,80]. However, hypoxia, which is driven by HIF-1 and VEGF, compli
SRS demands greater targeting precision, a sharply defined dose gradient, and rigorous dose verification, differing from other forms of RT[84]. Three main factors affect positioning accuracy in SRS[85].
Modern SRS systems have sub-millimeter mechanical accuracy, which is crucial for precisely targeting small areas[86].
CT offers high-resolution, low-distortion imaging. Conversely, magnetic resonance imaging often requires thicker slices for better signal quality, leading to potential distortions. Although 1.5T MR systems maintain distortion within approximately 0.5 mm, 3T MR systems require careful assessment to manage distortion for SRS[87-89].
During intracranial SRS, a fixed stereotactic frame is used to stabilize the head, whereas frameless SRS systems face challenges from involuntary movement. Modern SRS devices are equipped with monitoring systems to limit or adjust for movements within a sub-millimeter range[90,91].
The goal of SRS is to deliver steep dose gradients to exert maximum effects on targeted lesions[32,92]. High-dose-per-fraction strategies in fractionated therapy improve machine efficiency and patient throughput. Radiosurgery devices achieve precise dose distributions through different techniques. The GammaKnife converges approximately 200 beams at a focal point. Novalis uses a gantry that moves along multiple arcs, and CyberKnife delivers photons from various directions to create nonisocentric dose patterns[93,94].
Given the intense, localized doses that are administered in SRS, dose verification is essential to minimize the risk of tissue damage[95]. Elative dose distributions are accurately measured, and radiochromic films are often used for absolute dose quantification. In GammaKnife planning systems, the 1 mm/3% criterion typically yields gamma index values exceeding 97%, highlighting its precision. Accurate absorbed dose measurements, particularly with smaller fields, are critical for SRS applications[96-98].
The outcomes of SRT for BM treatment have been frequently reported across various populations. The key factors that influence prognosis include age, the number of metastases, the presence of extracranial metastases, and the Karnofsky Performance Status. According to the diagnosis-specific Graded Prognostic Assessment, patients with BM generally have a poor prognosis, with a median survival of 3–15 months[99,100]. Table 1 presents findings from various studies that assessed the effectiveness of SRT compared with that of WBRT.
| Ref. | Objective | Dosage | Key findings | Effectiveness | Toxicity | Concluding remarks |
| Luo et al[97] | Local and cerebral control in NSCLC patients treated with CyberKnife™ SRT | 27 Gy in three fractions (69%), 18–25 Gy in one fraction (18%), 30 Gy in five fractions (9%) | Local control rate = 78.7%, cerebral control | Effective for local control, minimal toxicity | Acute | Suitable for patients with few BMs, close follow-up is needed |
| Fessart et al[99] | Survival predictors in patients with lung cancer and BM | Single 20-Gy fraction for SRT, 7–10 Gy/fraction for HFSRT | Disease control rate = 84%, median overall | Effective with better control than WBRT | Grade III–IV toxicity = 4% | Well-tolerated, patient selection is key |
| Guo et al[100] | Palliative RT for BM and quality of life | WBRT = 30 Gy for 10 fractions, | Response rate = 50%–75%, neurological improvement rate = 75% | Improves quality of life, better than WBRT alone | Short term: Neurological symptoms; long term: Memory loss | Treatment should be individualized according to patients’ conditions |
| Gunnarsson et al[101] | Effectiveness of SRT in patients with lung cancer and BM | BED 10 SRT ≥ 50 Gy | Median overall | Effective with controlled extracranial disease | No marked toxicity difference | SRT is effective, particularly with stable extracranial disease |
| Hashmi et al[102] | Cognitive deterioration in SRS vs SRS + WBRT patients | SRS = 20–24 Gy, SRS + WBRT = 18–20 Gy (SRS) + 30 Gy (WBRT) | Cognitive deterioration: 63.5% (SRS) vs 91.7% (SRS + WBRT), median survival = 10.4 months (SRS), 7.4 months (SRS + WBRT) | SRS alone results in less cognitive decline | Cognitive deterioration | SRS preferred for minimizing cognitive decline without impacting survival |
For larger BMs (greater than 2 cm in diameter), SRS is being used more frequently alongside conservative approaches[18]. Patients typically exhibit prominent neurological symptoms and potentially experience vasogenic edema or mass effects, which frequently necessitate immediate surgical intervention. After achieving gross total resection, postoperative SRS at a median dose of 15 Gy has been associated with increased local control and overall survival in patients with tumors averaging 8.7–9.6 mL in volume[101-103]. However, SRS is associated with some risks, including potential neurological issues because of extensive resection and symptomatic radionecrosis, particularly with a large target volume margin
Radiation necrosis remains a notable risk of SRS, particularly in cases involving large treatment volumes[105]. This condition can appear 1 to 2 years post-treatment, and it is often indicated by radiographic changes or symptoms such as headaches, drowsiness, seizures, and death in severe cases[106]. Recent studies investigated the combination of SRS with immunotherapies and targeted therapies, focusing on the potential synergistic benefits of pairing SRS with immune checkpoint inhibitors[107-109].
SRT represents a significant advancement in treating BMs in patients with NSCLC. This technique increases precision and minimizes collateral damage to surrounding brain tissue, thereby improving local control and extending patient survival. To maximize the benefits of SRT, further clinical studies are needed to refine the timing, radiation dosage, and fractionation protocols. Future research efforts should focus on improving predictive metrics and identifying biomarkers to develop tailored, personalized treatment for patients with lung cancer BMs. Additionally, exploring emerging technologies, such as advanced imaging techniques or novel RT methods, holds great potential for improving the effectiveness of SRT. Areas that require further clinical validation include the combination of SRT with other treatment modalities or the optimization of fractionation schedules, as such advancements could significantly enhance patient outcomes. By outlining these potential research directions, this paper can serve as a guide for future work in the field, encouraging further exploration and development to optimize efficacy and expand treatment indications.
Sincere gratitude is extended to Professor Guan Wei for her invaluable assistance and insightful guidance throughout the process of writing this paper.
| 1. | Sharma R. Mapping of global, regional and national incidence, mortality and mortality-to-incidence ratio of lung cancer in 2020 and 2050. Int J Clin Oncol. 2022;27:665-675. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 110] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 2. | Rodak O, Peris-Díaz MD, Olbromski M, Podhorska-Okołów M, Dzięgiel P. Current Landscape of Non-Small Cell Lung Cancer: Epidemiology, Histological Classification, Targeted Therapies, and Immunotherapy. Cancers (Basel). 2021;13:4705. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 133] [Article Influence: 33.3] [Reference Citation Analysis (31)] |
| 3. | Chaft JE, Rimner A, Weder W, Azzoli CG, Kris MG, Cascone T. Evolution of systemic therapy for stages I-III non-metastatic non-small-cell lung cancer. Nat Rev Clin Oncol. 2021;18:547-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 238] [Article Influence: 59.5] [Reference Citation Analysis (0)] |
| 4. | Tsui DCC, Camidge DR, Rusthoven CG. Managing Central Nervous System Spread of Lung Cancer: The State of the Art. J Clin Oncol. 2022;40:642-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 5. | Buriolla S, Pelizzari G, Corvaja C, Alberti M, Targato G, Bortolot M, Torresan S, Cortiula F, Fasola G, Follador A. Immunotherapy in NSCLC Patients with Brain Metastases. Int J Mol Sci. 2022;23:7068. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 6. | Zhang Q, Abdo R, Iosef C, Kaneko T, Cecchini M, Han VK, Li SS. The spatial transcriptomic landscape of non-small cell lung cancer brain metastasis. Nat Commun. 2022;13:5983. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 108] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 7. | Mukherjee S, Ali S, Hashmi S, Jahan S. History, Origin and Types of Neurological Disorders. Applications of Stem Cells and derived Exosomes in Neurodegenerative. Springer, Singapore, 2023. [DOI] [Full Text] |
| 8. | Souza VGP, Forder A, Telkar N, Stewart GL, Carvalho RF, Mur LAJ, Lam WL, Reis PP. Identifying New Contributors to Brain Metastasis in Lung Adenocarcinoma: A Transcriptomic Meta-Analysis. Cancers (Basel). 2023;15:4526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 9. | Achrol AS, Rennert RC, Anders C, Soffietti R, Ahluwalia MS, Nayak L, Peters S, Arvold ND, Harsh GR, Steeg PS, Chang SD. Brain metastases. Nat Rev Dis Primers. 2019;5:5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 393] [Cited by in RCA: 686] [Article Influence: 114.3] [Reference Citation Analysis (0)] |
| 10. | Souza VGP, de Araújo RP, Santesso MR, Seneda AL, Minutentag IW, Felix TF, Hamamoto Filho PT, Pewarchuk ME, Brockley LJ, Marchi FA, Lam WL, Drigo SA, Reis PP. Advances in the Molecular Landscape of Lung Cancer Brain Metastasis. Cancers (Basel). 2023;15:722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 11. | Page S, Milner-Watts C, Perna M, Janzic U, Vidal N, Kaudeer N, Ahmed M, McDonald F, Locke I, Minchom A, Bhosle J, Welsh L, O'Brien M. Systemic treatment of brain metastases in non-small cell lung cancer. Eur J Cancer. 2020;132:187-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 12. | Nigen B, Goronflot T, Herbreteau G, Mathiot L, Sagan C, Raimbourg J, Bennouna J, Thillays F, Pons-Tostivint E. Impact of first-line immunotherapy on survival and intracranial outcomes in a cohort of non-small cell lung cancer patients with brain metastases at diagnosis. Lung Cancer. 2023;184:107321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 13. | Wrona A, Dziadziuszko R, Jassem J. Management of brain metastases in non-small cell lung cancer in the era of tyrosine kinase inhibitors. Cancer Treat Rev. 2018;71:59-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 14. | Su Z, Zhou L, Xue J, Lu Y. Integration of stereotactic radiosurgery or whole brain radiation therapy with immunotherapy for treatment of brain metastases. Chin J Cancer Res. 2020;32:448-466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Thomsen BJ, Soliman H. The modern management of untreated large (>2 cm) brain metastases: a narrative review. Chin Clin Oncol. 2022;11:16. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 16. | Guénolé M, Lucia F, Bourbonne V, Dissaux G, Reygagne E, Goasduff G, Pradier O, Schick U. Impact of concomitant systemic treatments on toxicity and intracerebral response after stereotactic radiotherapy for brain metastases. BMC Cancer. 2020;20:991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 17. | Loo M, Clavier JB, Attal Khalifa J, Moyal E, Khalifa J. Dose-Response Effect and Dose-Toxicity in Stereotactic Radiotherapy for Brain Metastases: A Review. Cancers (Basel). 2021;13:6086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 18. | Diehl CD, Giordano FA, Grosu AL, Ille S, Kahl KH, Onken J, Rieken S, Sarria GR, Shiban E, Wagner A, Beck J, Brehmer S, Ganslandt O, Hamed M, Meyer B, Münter M, Raabe A, Rohde V, Schaller K, Schilling D, Schneider M, Sperk E, Thomé C, Vajkoczy P, Vatter H, Combs SE. Opportunities and Alternatives of Modern Radiation Oncology and Surgery for the Management of Resectable Brain Metastases. Cancers (Basel). 2023;15:3670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 19. | Moravan MJ, Fecci PE, Anders CK, Clarke JM, Salama AKS, Adamson JD, Floyd SR, Torok JA, Salama JK, Sampson JH, Sperduto PW, Kirkpatrick JP. Current multidisciplinary management of brain metastases. Cancer. 2020;126:1390-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 20. | Zoghbi M, Moussa MJ, Dagher J, Haroun E, Qdaisat A, Singer ED, Karam YE, Yeung SJ, Chaftari P. Brain Metastasis in the Emergency Department: Epidemiology, Presentation, Investigations, and Management. Cancers (Basel). 2024;16:2583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 21. | Møller DS, Lutz CM, Khalil AA, Alber M, Holt MI, Kandi M, Schmidt HH, Tvilum M, Appelt A, Knap MM, Hoffmann L. Survival benefits for non-small cell lung cancer patients treated with adaptive radiotherapy. Radiother Oncol. 2022;168:234-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 22. | Savoca G, Calvaruso M, Minafra L, Bravatà V, Cammarata FP, Iacoviello G, Abbate B, Evangelista G, Spada M, Forte GI, Russo G. Local Disease-Free Survival Rate (LSR) Application to Personalize Radiation Therapy Treatments in Breast Cancer Models. J Pers Med. 2020;10:177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 23. | Xing Y, Yasinjan F, Cui J, Peng Y, He M, Liu W, Hong X. Advancements and current trends in tumor treating fields: a scientometric analysis. Int J Surg. 2024;110:2978-2991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 24. | Martin F, Magnier F, Berger L, Miroir J, Chautard E, Verrelle P, Lapeyre M, Biau J. Fractionated stereotactic radiotherapy of benign skull-base tumors: a dosimetric comparison of volumetric modulated arc therapy with Rapidarc® versus non-coplanar dynamic arcs. Radiat Oncol. 2016;11:58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 25. | Vlaskou Badra E, Baumgartl M, Fabiano S, Jongen A, Guckenberger M. Stereotactic radiotherapy for early stage non-small cell lung cancer: current standards and ongoing research. Transl Lung Cancer Res. 2021;10:1930-1949. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 26. | Mangoni M, Borghesi S, Aristei C, Becherini C. Radiobiology of stereotactic radiotherapy. Rep Pract Oncol Radiother. 2022;27:57-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 27. | Amin S, Baine M, Lin C. Immunotherapy plus stereotactic body radiation therapy or whole-brain radiation therapy in brain metastases. Immunotherapy. 2023;15:163-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 28. | Munshi A, Sarkar B, Pandey V, Sonkar DR. Radiotherapy Induced Central Nervous System Toxicity. Complications of Cancer Therapy: Best Practices in Prevention and Management. Springer, Singapore, 2024. [DOI] [Full Text] |
| 29. | Scampoli C, Cammelli S, Galietta E, Siepe G, Buwenge M, Macchia G, Deodato F, Cilla S, Strigari L, Chiesa S, Morganti AG. Memantine in the Prevention of Radiation-Induced Brain Damage: A Narrative Review. Cancers (Basel). 2022;14:2736. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 30. | Yang X, Ren H, Fu J. Treatment of Radiation-Induced Brain Necrosis. Oxid Med Cell Longev. 2021;2021:4793517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 31. | van Grinsven EE, Nagtegaal SHJ, Verhoeff JJC, van Zandvoort MJE. The Impact of Stereotactic or Whole Brain Radiotherapy on Neurocognitive Functioning in Adult Patients with Brain Metastases: A Systematic Review and Meta-Analysis. Oncol Res Treat. 2021;44:622-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 32. | Topkan E, Kucuk A, Senyurek S, Sezen D, Durankus NK, Akdemir EY, Saglam Y, Bolukbasi Y, Pehlivan B, Selek U. Radiosurgery Techniques for Brain Metastases. J Cancer Tumor Int. 2020;10:1-14. [DOI] [Full Text] |
| 33. | Halima A, Suh JH, Lo SS, Angelov L, Murphy ES, Chao ST. Repeat Stereotactic Radiosurgery for Brain Metastasis. Intracranial Stereotactic Radiosurgery Boca Raton: CRC Press, 2021. |
| 34. | Levis M, Gastino A, De Giorgi G, Mantovani C, Bironzo P, Mangherini L, Ricci AA, Ricardi U, Cassoni P, Bertero L. Modern Stereotactic Radiotherapy for Brain Metastases from Lung Cancer: Current Trends and Future Perspectives Based on Integrated Translational Approaches. Cancers (Basel). 2023;15:4622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 35. | McCutcheon IE. Stereotactic Radiosurgery to Prevent Local Recurrence of Brain Metastasis After Surgery: Neoadjuvant Versus Adjuvant. Acta Neurochir Suppl. 2021;128:85-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 36. | Chambrelant I, Jarnet D, Bou-Gharios J, Le Fèvre C, Kuntz L, Antoni D, Jenny C, Noël G. Stereotactic Radiation Therapy of Single Brain Metastases: A Literature Review of Dosimetric Studies. Cancers (Basel). 2023;15:3937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 37. | Soldá F, Tancu C, Kitchen N, Fersht N. Neurosurgical applications of radiotherapy. Surgery (Oxford). 2024;42:590-598. [DOI] [Full Text] |
| 38. | Schmitt D, Blanck O, Gauer T, Fix MK, Brunner TB, Fleckenstein J, Loutfi-Krauss B, Manser P, Werner R, Wilhelm ML, Baus WW, Moustakis C. Technological quality requirements for stereotactic radiotherapy: Expert review group consensus from the DGMP Working Group for Physics and Technology in Stereotactic Radiotherapy. Strahlenther Onkol. 2020;196:421-443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 91] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 39. | Vellayappan BA, Lim MC, Yong C, Teo K, Malone S, Lo S. Target Delineation for Radiosurgery (Including Postoperative Cavity Radiosurgery) in Brain Metastases. Radiotherapy in Managing Brain Metastases. Springer, Cham, 2020. [DOI] [Full Text] |
| 40. | McKelvey KJ, Hudson AL, Donaghy H, Stoner SP, Wheeler HR, Diakos CI, Howell VM. Differential effects of radiation fractionation regimens on glioblastoma. Radiat Oncol. 2022;17:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 41. | Piras A, Boldrini L, Menna S, Sanfratello A, D'Aviero A, Cusumano D, Di Cristina L, Messina M, Spada M, Angileri T, Daidone A. Five-Fraction Stereotactic Radiotherapy for Brain Metastases: A Single-Institution Experience on Different Dose Schedules. Oncol Res Treat. 2022;45:408-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 42. | Perlow HK, Ho C, Matsui JK, Prasad RN, Klamer BG, Wang J, Damante M, Upadhyay R, Thomas E, Blakaj DM, Beyer S, Lonser R, Hardesty D, Raval RR, Prabhu R, Elder JB, Palmer JD. Comparing pre-operative versus post-operative single and multi-fraction stereotactic radiotherapy for patients with resectable brain metastases. Clin Transl Radiat Oncol. 2023;38:117-122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 43. | Putz F, Weissmann T, Oft D, Schmidt MA, Roesch J, Siavooshhaghighi H, Filimonova I, Schmitter C, Mengling V, Bert C, Frey B, Lettmaier S, Distel LV, Semrau S, Fietkau R. FSRT vs. SRS in Brain Metastases-Differences in Local Control and Radiation Necrosis-A Volumetric Study. Front Oncol. 2020;10:559193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 44. | Chen T, Tang M, Zhou Y, Wang Z, Li S, Wang H, Lu Y, Wang J, Shen W. Pretreatment lymphocyte-to-monocyte ratio as a prognostic factor and influence on dose-effect in fractionated stereotactic radiotherapy for oligometastatic brain metastases in non-small cell lung cancer patients. Front Oncol. 2023;13:1216852. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 45. | Klement RJ, Sweeney RA. Metabolic factors associated with the prognosis of oligometastatic patients treated with stereotactic body radiotherapy. Cancer Metastasis Rev. 2023;42:927-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 46. | Moreno AC, Fellman B, Hobbs BP, Liao Z, Gomez DR, Chen A, Hahn SM, Chang JY, Lin SH. Biologically Effective Dose in Stereotactic Body Radiotherapy and Survival for Patients With Early-Stage NSCLC. J Thorac Oncol. 2020;15:101-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 47. | Cellini F, Arcelli A, Simoni N, Caravatta L, Buwenge M, Calabrese A, Brunetti O, Genovesi D, Mazzarotto R, Deodato F, Mattiucci GC, Silvestris N, Valentini V, Morganti AG. Basics and Frontiers on Pancreatic Cancer for Radiation Oncology: Target Delineation, SBRT, SIB technique, MRgRT, Particle Therapy, Immunotherapy and Clinical Guidelines. Cancers (Basel). 2020;12:1729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 48. | Conde-Moreno AJ, Zucca Aparicio D, Pérez-Calatayud MJ, López-Campos F, Celada Álvarez F, Rubio Rodríguez C, Fernández-Letón P, Gómez-Caamaño A, Contreras Martínez J. Recommended procedures and responsibilities for radiosurgery (SRS) and extracranial stereotactic body radiotherapy (SBRT): report of the SEOR in collaboration with the SEFM. Clin Transl Oncol. 2021;23:1281-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 49. | Schill NC, Smith HW. The Role of Radiation in the Treatment of Hepatocellular Carcinoma. In: Tsoulfas G, editor. Liver Cancer-Multidisciplinary Approach. United Kingdom: IntechOpen Limited, 2024. [DOI] [Full Text] |
| 50. | Song CW, Glatstein E, Marks LB, Emami B, Grimm J, Sperduto PW, Kim MS, Hui S, Dusenbery KE, Cho LC. Biological Principles of Stereotactic Body Radiation Therapy (SBRT) and Stereotactic Radiation Surgery (SRS): Indirect Cell Death. Int J Radiat Oncol Biol Phys. 2021;110:21-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 114] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 51. | Daou BJ, Palmateer G, Thompson BG, Maher CO, Hayman JA, Lam KL, Wahl DR, Kim M, Pandey AS. Stereotactic Radiosurgery for Brain Arteriovenous Malformations: Evaluation of Obliteration and Review of Associated Predictors. J Stroke Cerebrovasc Dis. 2020;29:104863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 52. | Shaaban A, Tos SM, Mantziaris G, Kotecha R, Fariselli L, Gorgulho A, Levivier M, Ma L, Paddick I, Pollock BE, Regis J, Suh JH, Yomo S, Sahgal A, Sheehan JP. Repeat Single-Session Stereotactic Radiosurgery for Cerebral Arteriovenous Malformations: A Systematic Review, Meta-Analysis, and International Stereotactic Radiosurgery Society Practice Guidelines. Neurosurgery. 2025;96:29-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 53. | Fatima N, Meola A, Ding VY, Pollom E, Soltys SG, Chuang CF, Shahsavari N, Hancock SL, Gibbs IC, Adler JR, Chang SD. The Stanford stereotactic radiosurgery experience on 7000 patients over 2 decades (1999-2018): looking far beyond the scalpel. J Neurosurg. 2021;135:1725-1741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 54. | Neophytou CM, Panagi M, Stylianopoulos T, Papageorgis P. The Role of Tumor Microenvironment in Cancer Metastasis: Molecular Mechanisms and Therapeutic Opportunities. Cancers (Basel). 2021;13:2053. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 265] [Article Influence: 66.3] [Reference Citation Analysis (0)] |
| 55. | McDonald B, Barth K, Schmidt MHH. The origin of brain malignancies at the blood-brain barrier. Cell Mol Life Sci. 2023;80:282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 56. | Mortezaee K. Organ tropism in solid tumor metastasis: an updated review. Future Oncol. 2021;17:1943-1961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 57. | Ge Z, Ding S. The Crosstalk Between Tumor-Associated Macrophages (TAMs) and Tumor Cells and the Corresponding Targeted Therapy. Front Oncol. 2020;10:590941. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 147] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 58. | Jablonska PA, Galán N, Barranco J, Leon S, Robledano R, Echeveste JI, Calvo A, Aristu J, Serrano D. Presence of Activated (Phosphorylated) STAT3 in Radiation Necrosis Following Stereotactic Radiosurgery for Brain Metastases. Int J Mol Sci. 2023;24:14219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 59. | Tan Y, Wang M, Zhang Y, Ge S, Zhong F, Xia G, Sun C. Tumor-Associated Macrophages: A Potential Target for Cancer Therapy. Front Oncol. 2021;11:693517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 79] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 60. | Sereno M, Hernandez de Córdoba I, Gutiérrez-Gutiérrez G, Casado E. Brain metastases and lung cancer: molecular biology, natural history, prediction of response and efficacy of immunotherapy. Front Immunol. 2023;14:1297988. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 61. | Wu L, Zhang XH. Tumor-Associated Neutrophils and Macrophages-Heterogenous but Not Chaotic. Front Immunol. 2020;11:553967. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 62. | Yan M, Zheng M, Niu R, Yang X, Tian S, Fan L, Li Y, Zhang S. Roles of tumor-associated neutrophils in tumor metastasis and its clinical applications. Front Cell Dev Biol. 2022;10:938289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 63. | Gómez V, Mustapha R, Ng K, Ng T. Radiation therapy and the innate immune response: Clinical implications for immunotherapy approaches. Br J Clin Pharmacol. 2020;86:1726-1735. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 64. | Wang Y. Advances in Hypofractionated Irradiation-Induced Immunosuppression of Tumor Microenvironment. Front Immunol. 2020;11:612072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 65. | Punnanitinont A, Kannisto ED, Matsuzaki J, Odunsi K, Yendamuri S, Singh AK, Patnaik SK. Sublethal Radiation Affects Antigen Processing and Presentation Genes to Enhance Immunogenicity of Cancer Cells. Int J Mol Sci. 2020;21:2573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 66. | Sprooten J, De Wijngaert P, Vanmeerbeerk I, Martin S, Vangheluwe P, Schlenner S, Krysko DV, Parys JB, Bultynck G, Vandenabeele P, Garg AD. Necroptosis in Immuno-Oncology and Cancer Immunotherapy. Cells. 2020;9:1823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 127] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 67. | Cinier J, Hubert M, Besson L, Di Roio A, Rodriguez C, Lombardi V, Caux C, Ménétrier-Caux C. Recruitment and Expansion of Tregs Cells in the Tumor Environment-How to Target Them? Cancers (Basel). 2021;13:1850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 68] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 68. | Patel RR, Verma V, Barsoumian HB, Ning MS, Chun SG, Tang C, Chang JY, Lee PP, Gandhi S, Balter P, Dunn JD, Chen D, Puebla-Osorio N, Cortez MA, Welsh JW. Use of Multi-Site Radiation Therapy for Systemic Disease Control. Int J Radiat Oncol Biol Phys. 2021;109:352-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 69. | Cui JW, Li Y, Yang Y, Yang HK, Dong JM, Xiao ZH, He X, Guo JH, Wang RQ, Dai B, Zhou ZL. Tumor immunotherapy resistance: Revealing the mechanism of PD-1 / PD-L1-mediated tumor immune escape. Biomed Pharmacother. 2024;171:116203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 57] [Reference Citation Analysis (0)] |
| 70. | Ma SC, Tang XR, Long LL, Bai X, Zhou JG, Duan ZJ, Wang J, Fu QJ, Zhu HB, Guo XJ, Zhang YP, Guo ZQ, Wu DH, Dong ZY. Integrative evaluation of primary and metastatic lesion spectrum to guide anti-PD-L1 therapy of non-small cell lung cancer: results from two randomized studies. Oncoimmunology. 2021;10:1909296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 71. | Wang Y, Zhang T, Huang Y, Li W, Zhao J, Yang Y, Li C, Wang L, Bi N. Real-World Safety and Efficacy of Consolidation Durvalumab After Chemoradiation Therapy for Stage III Non-small Cell Lung Cancer: A Systematic Review and Meta-analysis. Int J Radiat Oncol Biol Phys. 2022;112:1154-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 67] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 72. | Ricciuti B, Wang X, Alessi JV, Rizvi H, Mahadevan NR, Li YY, Polio A, Lindsay J, Umeton R, Sinha R, Vokes NI, Recondo G, Lamberti G, Lawrence M, Vaz VR, Leonardi GC, Plodkowski AJ, Gupta H, Cherniack AD, Tolstorukov MY, Sharma B, Felt KD, Gainor JF, Ravi A, Getz G, Schalper KA, Henick B, Forde P, Anagnostou V, Jänne PA, Van Allen EM, Nishino M, Sholl LM, Christiani DC, Lin X, Rodig SJ, Hellmann MD, Awad MM. Association of High Tumor Mutation Burden in Non-Small Cell Lung Cancers With Increased Immune Infiltration and Improved Clinical Outcomes of PD-L1 Blockade Across PD-L1 Expression Levels. JAMA Oncol. 2022;8:1160-1168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 224] [Article Influence: 74.7] [Reference Citation Analysis (0)] |
| 73. | Tafe LJ. Non-Small Cell Lung Cancer as a Precision Oncology Paradigm: Emerging Targets and Tumor Mutational Burden (TMB). Adv Anat Pathol. 2020;27:3-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 74. | Becherini C, Lancia A, Detti B, Lucidi S, Scartoni D, Ingrosso G, Carnevale MG, Roghi M, Bertini N, Orsatti C, Mangoni M, Francolini G, Marani S, Giacomelli I, Loi M, Pergolizzi S, Bonzano E, Aristei C, Livi L. Modulation of tumor-associated macrophage activity with radiation therapy: a systematic review. Strahlenther Onkol. 2023;199:1173-1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 75. | Zhang Y, Feng Z, Liu J, Li H, Su Q, Zhang J, Huang P, Wang W, Liu J. Polarization of tumor-associated macrophages by TLR7/8 conjugated radiosensitive peptide hydrogel for overcoming tumor radioresistance. Bioact Mater. 2022;16:359-371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 76. | Donlon NE, Power R, Hayes C, Reynolds JV, Lysaght J. Radiotherapy, immunotherapy, and the tumour microenvironment: Turning an immunosuppressive milieu into a therapeutic opportunity. Cancer Lett. 2021;502:84-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 118] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 77. | Jorgovanovic D, Song M, Wang L, Zhang Y. Roles of IFN-γ in tumor progression and regression: a review. Biomark Res. 2020;8:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 815] [Cited by in RCA: 766] [Article Influence: 153.2] [Reference Citation Analysis (0)] |
| 78. | Ashrafizadeh M, Farhood B, Eleojo Musa A, Taeb S, Najafi M. Damage-associated molecular patterns in tumor radiotherapy. Int Immunopharmacol. 2020;86:106761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 79. | Li S, Jiang M, Wang L, Yu S. Combined chemotherapy with cyclooxygenase-2 (COX-2) inhibitors in treating human cancers: Recent advancement. Biomed Pharmacother. 2020;129:110389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 91] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 80. | Wang D, Cabalag CS, Clemons NJ, DuBois RN. Cyclooxygenases and Prostaglandins in Tumor Immunology and Microenvironment of Gastrointestinal Cancer. Gastroenterology. 2021;161:1813-1829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 87] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 81. | Kabakov AE, Yakimova AO. Hypoxia-Induced Cancer Cell Responses Driving Radioresistance of Hypoxic Tumors: Approaches to Targeting and Radiosensitizing. Cancers (Basel). 2021;13:1102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 82. | Qi S, Deng S, Lian Z, Yu K. Novel Drugs with High Efficacy against Tumor Angiogenesis. Int J Mol Sci. 2022;23:6934. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 50] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 83. | Bilotta MT, Antignani A, Fitzgerald DJ. Managing the TME to improve the efficacy of cancer therapy. Front Immunol. 2022;13:954992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 147] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 84. | Chung H, Lee D. Stereotactic Radiosurgery. Prog Med Phys. 2020;31:63-70. [DOI] [Full Text] |
| 85. | Tanaka M. Basic Knowledge and Overview of Brain AVMs. J Neuroendovasc Ther. 2025;19:2024-0037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 86. | Freislederer P, Kügele M, Öllers M, Swinnen A, Sauer TO, Bert C, Giantsoudi D, Corradini S, Batista V. Recent advanced in Surface Guided Radiation Therapy. Radiat Oncol. 2020;15:187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 87. | Taghizadeh S, Labuda C, Yang CC, Morris B, Kanakamedala MR, Vijayakumar S, Rey-Dios R, Duggar WN, Florez E, Fatemi A. Optimizing MRI sequences and images for MRI-based stereotactic radiosurgery treatment planning. Rep Pract Oncol Radiother. 2019;24:12-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 88. | Grishchuk D, Dimitriadis A, Sahgal A, De Salles A, Fariselli L, Kotecha R, Levivier M, Ma L, Pollock BE, Regis J, Sheehan J, Suh J, Yomo S, Paddick I. ISRS Technical Guidelines for Stereotactic Radiosurgery: Treatment of Small Brain Metastases (≤1 cm in Diameter). Pract Radiat Oncol. 2023;13:183-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 89. | Nayak KS, Lim Y, Campbell-Washburn AE, Steeden J. Real-Time Magnetic Resonance Imaging. J Magn Reson Imaging. 2022;55:81-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 54] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 90. | Solberg TD, Balter JM, Benedict SH, Fraass BA, Kavanagh B, Miyamoto C, Pawlicki T, Potters L, Yamada Y. Quality and safety considerations in stereotactic radiosurgery and stereotactic body radiation therapy: Executive summary. Pract Radiat Oncol. 2012;2:2-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 137] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 91. | Taillez A, Bimbai AM, Lacornerie T, Le Deley MC, Lartigau EF, Pasquier D. Studies of Intra-Fraction Prostate Motion During Stereotactic Irradiation in First Irradiation and Re-Irradiation. Front Oncol. 2021;11:690422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 92. | Mendel JT, Jaster AW, Yu FF, Morris LC 3rd, Lynch PT, Shah BR, Agarwal A, Timmerman RD, Nedzi LA, Raj KM. Fundamentals of Radiation Oncology for Neurologic Imaging. Radiographics. 2020;40:827-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 93. | Griffin RJ, Ahmed MM, Amendola B, Belyakov O, Bentzen SM, Butterworth KT, Chang S, Coleman CN, Djonov V, Formenti SC, Glatstein E, Guha C, Kalnicki S, Le QT, Loo BW Jr, Mahadevan A, Massaccesi M, Maxim PG, Mohiuddin M, Mohiuddin M, Mayr NA, Obcemea C, Petersson K, Regine W, Roach M, Romanelli P, Simone CB 2nd, Snider JW, Spitz DR, Vikram B, Vozenin MC, Abdel-Wahab M, Welsh J, Wu X, Limoli CL. Understanding High-Dose, Ultra-High Dose Rate, and Spatially Fractionated Radiation Therapy. Int J Radiat Oncol Biol Phys. 2020;107:766-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 94. | Kilby W, Naylor M, Dooley JR, Maurer CR, Sayeh S. A Technical Overview of the CyberKnife System. Handbook of Robotic and Image-Guided Surgery. Elsevier. 2020;15-38. [DOI] [Full Text] |
| 95. | Padelli F, Aquino D, Fariselli L, De Martin E. IBA myQA SRS Detector for CyberKnife Robotic Radiosurgery Quality Assurance. Appl Sci. 2022;12:7791. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 96. | Casolaro P. Radiochromic Films for the Two-Dimensional Dose Distribution Assessment. Appl Sci. 2021;11:2132. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 97. | Luo D, Han EY, Wen Z, Mackin DS, Krafft SP, Li J, Wang X, Briere TM. Use of uniform shots for robust planning of mask-based treatment in Gamma Knife Icon. Phys Med. 2020;73:135-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 98. | Muñoz L, Kron T, Petasecca M, Bucci J, Jackson M, Metcalfe P, Rosenfeld AB, Biasi G. Consistency of small-field dosimetry, on and off axis, in beam-matched linacs used for stereotactic radiosurgery. J Appl Clin Med Phys. 2021;22:185-193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 99. | Fessart E, Mouttet Audouard R, Le Tinier F, Coche-Dequeant B, Lacornerie T, Tresch E, Scherpereel A, Lartigau E, Mirabel X, Pasquier D. Stereotactic irradiation of non-small cell lung cancer brain metastases: evaluation of local and cerebral control in a large series. Sci Rep. 2020;10:11201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 100. | Guo D, Liu J, Li Y, Chen Q, Zhao Y, Guo X, Zhu S, Ji S. A Novel Score Combining Magnetic Resonance Spectroscopy Parameters and Systemic Immune-Inflammation Index Improves Prognosis Prediction in Non-Small Cell Lung Cancer Patients With Brain Metastases After Stereotactic Radiotherapy. Front Oncol. 2022;12:762230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 101. | Gunnarsson K, Hallqvist A. Stereotactic radiotherapy for brain metastases in patients with lung cancer; outcome and toxicity in clinical practice. Rep Pract Oncol Radiother. 2022;27:410-418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 102. | Hashmi MF, Agarwal MS, Maani EV, Arora RD, Cascella M. Palliative Radiation Therapy for Brain Metastases. 2023 Jun 4. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing, 2025. [PubMed] |
| 103. | Nardone V, Romeo C, D'Ippolito E, Pastina P, D'Apolito M, Pirtoli L, Caraglia M, Mutti L, Bianco G, Falzea AC, Giannicola R, Giordano A, Tagliaferri P, Vinciguerra C, Desideri I, Loi M, Reginelli A, Cappabianca S, Tassone P, Correale P. The role of brain radiotherapy for EGFR- and ALK-positive non-small-cell lung cancer with brain metastases: a review. Radiol Med. 2023;128:316-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 104. | Shiue K, Sahgal A, Lo SS. Precision Radiation for Brain Metastases With a Focus on Hypofractionated Stereotactic Radiosurgery. Semin Radiat Oncol. 2023;33:114-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 105. | Minniti G, Niyazi M, Andratschke N, Guckenberger M, Palmer JD, Shih HA, Lo SS, Soltys S, Russo I, Brown PD, Belka C. Current status and recent advances in resection cavity irradiation of brain metastases. Radiat Oncol. 2021;16:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 106. | Wang K, Tepper JE. Radiation therapy-associated toxicity: Etiology, management, and prevention. CA Cancer J Clin. 2021;71:437-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 234] [Article Influence: 58.5] [Reference Citation Analysis (0)] |
| 107. | Desagneaux A, Charles J, Kastler A, Dols A-, Leccia M, Elhalawani H, Aizer A, Flandin I, Verry C. Safety and outcomes of combined immune checkpoint inhibitor and radiation therapy for patients with melanoma brain metastases. 2022 Preprint. [DOI] [Full Text] |
| 108. | Hendriks LEL, Henon C, Auclin E, Mezquita L, Ferrara R, Audigier-Valette C, Mazieres J, Lefebvre C, Rabeau A, Le Moulec S, Cousin S, Duchemann B, le Pechoux C, Botticella A, Ammari S, Gazzah A, Caramella C, Adam J, Lechapt E, Planchard D, De Ruysscher D, Dingemans AM, Besse B. Outcome of Patients with Non-Small Cell Lung Cancer and Brain Metastases Treated with Checkpoint Inhibitors. J Thorac Oncol. 2019;14:1244-1254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 186] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 109. | Kotecha R, Kim JM, Miller JA, Juloori A, Chao ST, Murphy ES, Peereboom DM, Mohammadi AM, Barnett GH, Vogelbaum MA, Angelov L, Suh JH, Ahluwalia MS. The impact of sequencing PD-1/PD-L1 inhibitors and stereotactic radiosurgery for patients with brain metastasis. Neuro Oncol. 2019;21:1060-1068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 88] [Article Influence: 22.0] [Reference Citation Analysis (0)] |